Abstract
Renin angiotensin system (RAS) is an endocrine system widely known for its physiological roles in electrolyte homeostasis, body fluid volume regulation and cardiovascular control in peripheral circulation. However, brain RAS is an independent form of RAS expressed locally in the brain, which is known to be involved in brain functions and disorders. There is strong evidence for a major involvement of excessive brain angiotensin converting enzyme (ACE)/Angiotensin II (Ang II)/Angiotensin type-1 receptor (AT-1R) axis in increased activation of oxidative stress, apoptosis and neuroinflammation causing neurodegeneration in several brain disorders. Numerous studies have demonstrated strong neuroprotective effects by blocking AT1R in these brain disorders. Additionally, the angiotensin converting enzyme 2 (ACE2)/Angiotensin (1–7)/Mas receptor (MASR), is another axis of brain RAS which counteracts the damaging effects of ACE/Ang II/AT1R axis on neurons in the brain. Thus, angiotensin II receptor blockers (ARBs) and activation of ACE2/Angiotensin (1–7)/MASR axis may serve as an exciting and novel method for neuroprotection in several neurodegenerative diseases. Here in this review article, we discuss the expression of RAS in the brain and highlight how altered RAS level may cause neurodegeneration. Understanding the pathophysiology of RAS and their links to neurodegeneration has enormous potential to identify potentially effective pharmacological tools to treat neurodegenerative diseases in the brain.
Keywords: Brain, RAS, Angiotensin, Neurodegeneration, Neuroprotection
1. Introduction
Renin angiotensin system (RAS) is an endocrine system widely known for its physiological roles in electrolyte homeostasis, body fluid volume regulation and cardiovascular control in peripheral circulation. Renin, an enzyme produced from the kidney, acts on angiotensinogen (AGT), a liver-precursor, to release an inactive decapeptide, angiotensin I (Ang I). Another enzyme, angiotensin converting enzyme (ACE) cleaves Ang I to the active octapeptide Ang II, the effector peptide of RAS, which is important for various physiological functions. However, chronic activation of RAS and the increase in Ang II level may act on AT1R, leading to various pathophysiological processes, including inflammation, vasoconstriction, fibrosis, increased renal sodium absorption, aldosterone and arginine vasopressin (AVP) release (de Morais et al., 2018, Gao et al., 2014, Sparks et al., 2014, Hua et al., 2011). Apart from systemic RAS, local independent RAS has been reported in various tissues such as heart, kidney, lung, liver and retina (Ola et al., 2013, Ola et al., 2017). Although, relatively less information is available on both the expression and regulation of RAS in the brain (Stornetta et al., 1988, Lavoie et al., 2004), widespread distribution of angiotensin receptors (Angiotensin type-2 receptor [AT2R] and AT1R) has been found in the central nervous system. Interestingly, AGT, the precursor of Ang I, is mostly produced within astrocytes where it constitutively secretes various neuroactive peptides (Bodiga and Bodiga, 2013). In addition, renin, an enzyme, which cleaves AGT into Ang I, has been found to be expressed within neurons and astrocytes. ACE converts Ang I into Ang II, which binds to both the AT1R and AT2R that are expressed in neurons, astrocytes, oligodendrocytes and microglia of various sections of brain (Labandeira-Garcia et al., 2017).
A number of studies have reported that dysregulated brain RAS may be implicated in neurodegeneration due to neuroinflammation, oxidative stress and aging-related pathophysiological changes (Labandeira-Garcia et al., 2017). Altered RAS plays a key role in numerous degenerative diseases of the brain including parkinson disease (PD), alzheimer’s (AD), huntington disease, dementia, amyotrophic lateral sclerosis, Multiple sclerosis, Traumatic brain injury and Stroke (Takane et al., 2017, Mogi and Horiuchi, 2013, Wright and Harding, 2013). Several studies have established the fact that activation of local RAS in the brain influences pathological processes in damaging the neurons. For example, elevated Ang II decreases cognitive function since ACE inhibitors (ACEIs) improve cognition by crossing the blood brain barrier (BBB), independent of blood pressure [BP] (Rygiel, 2016). High levels of Ang II are found to increase oxidative stress and promote neuroinflammation while ARBs prevent many risk factors for AD and protect neurons (Saavedra, 2016, Liu et al., 2015). Activation of AT1R by Ang II due to increased expression of ACE is known to play important role in vasoconstriction in the brain and impairs cognition (Ahmed et al., 2018.), cell death (Zhang et al., 2012) and inflammation (Labandeira-Garcia et al., 2017).
2. The components of brain renin angiotensin system
The brain RAS is an independent system involved in different brain physiological functions and disorders (Ganten et al., 1971, Labandeira-Garcia et al., 2017, Wright and Harding, 2013). Different components of RAS in the brain have been identified (Grobe et al., 2010, Grobe et al., 2011, Saavedra, 1992). Cellular and molecular studies have reported that all RAS genes and their promoter regions are active in the brain, while other evidences also revealed the de novo production of RAS components in the brain (Hermann et al., 1987, Fuxe et al., 1980). The establishment of brain RAS is also supported by several pharmacological inhibition studies (Shinohara et al., 2016, Shinohara et al., 2017). Similarly, it has been found that all the components required for the synthesis of angiotensin peptides are synthesized locally within the brain (Harding et al., 1988). Surprisingly, peripheral angiotensins may also interact with the brain RAS at the circumventricular organs which lacks BBB (Fry and Ferguson, 2007). As a result, they might control certain physiological functions although only the brain angiotensins execute central activities (Ferguson et al., 1999, Ferguson et al., 2001). Thus, there is complex interplay between the tissue and peripheral systemic RAS that makes the whole system difficult to understand (de Morais et al., 2018).
2.1. Brain renin/pro-renin
Angiotensinogen (AGT) is cleaved by renin enzyme to form Ang I peptide. Renin is abundant in blood circulation while present at low levels in the brain (Bodiga and Bodiga, 2013, Grobe et al., 2008). Several reports suggested that both renin and AGT are co-expressed in many parts of the brain (Lavoie et al., 2004). Within neuronal cells, expression of renin was found both intracellularly and in secreted form in the form of pro-renin (Grobe et al., 2008). The pro-renin in the brain binds with higher affinity to pro-renin receptors (PRRs) compared to renin, leading to AGT cleavage. Thus, stimulation of renin/pro-renin signaling causes cognitive impairment by activating the angiotensin receptors (Wright and Harding, 2013). However, van Thiel et al suggested that intracellular renin could be the key to generation of angiotensins in the brain, and coordinate other non-RAS functions (van Thiel et al., 2017). In addition, the impaired activity or expression of the brain renin may cause neurogenic hypertension, altering drinking and metabolism (Shinohara et al., 2016, Shinohara et al., 2017). Few studies though still suggest that the expression of renin is controversial in the brain (Nakagawa and Sigmund, 2017).
2.2. Brain angiotensin converting enzymes (ACE/ACE2)
Angiotensin converting enzyme (ACE) is the major component of RAS in the brain. Predominantly, it is localized in the endothelia of cerebral vasculature, although its highest expression has been found in choroid plexus (CP), organum vasculosum of the lamina terminalis (OVLT), subfornical organ (SFO) and area postrema (AP) (Bodiga and Bodiga, 2013). ACE activity is found in the areas of brain that lacks BBB, such as the SFO and pineal gland [PG] (Saavedra, 1992, Chai et al., 1987). Even inside the BBB surprisingly, ACE activity has been found in rats and humans (Saavedra, 1992). Evidence from clinical studies revealed ACE involvement in cognitive dysfunction, since its inhibitors reduced the onset of dementia in AD (Jouquey et al., 1995). ACEIs might reduce cognitive dysfunction through anti-inflammatory actions, independently from BP lowering effects (Rygiel, 2016). Several studies found a correlation between hypertension and central ACE expression/activity, although the underlying mechanism remains elusive. In vivo transfection of human ACE into neuronal cells cleaved Ang I to Ang II in cerebrospinal fluid and increased sympathetic nervous activity, which were all reversed by ACEIs (Tani 1991).
ACE2 is an enzyme expressed virtually throughout the brain and specifically in the areas controlling central BP (Tota et al., 2012, Nakamura et al., 1999). Even though, it exists as a transmembrane protein, there is evidence for its globular nature, although in a truncated form (Doobay et al., 2007). Proteolytically, ACE2 cleaves Ang II to Ang (1–7), a ligand for the Mas receptor (MASR). This signaling pathway has been shown to produce body functions that negate the effects of Ang II. Aside from Ang II substrate, ACE2 also acts on Ang I, releasing Ang (1–9). The action of ACE or other peptidases can then regenerate Ang (1–7) (Xia and Lazartigues, 2010, Tipnis et al., 2000, Xu et al., 2011). Several studies have revealed that brain ACE2 has been involved in the progression of neurogenic hypertension (Xu et al., 2017, Santos et al., 2003, Xia et al., 2009, Feng et al., 2010, Yamazato et al., 2007, Feng et al., 2008, Xia et al., 2015, Sriramula et al., 2011, Sriramula et al., 2015). As a result, ACE2 gene therapy has been proposed as a potential antihypertensive strategy, as it activates mechanisms of BP reduction. (Tipnis et al., 2000). The ACE2/Ang (1–7)/MASR axis, is highly distributed in almost all cells of the brain and exerts protection by counteracting the ACE/Ang II/AT1R axis (Doobay et al., 2007, Gallagher et al., 2006, Hamming et al., 2004, Jiang et al., 2013). In ischemic stroke, Ang-(1–7) injection in intracerebroventricular showed beneficial effects in neuroprotection (Mecca et al., 2011, Jiang et al., 2012). In addition, activation of ACE2/Ang(1–7)/MASR axis has also been shown to improve neurological deficits through its antioxidative and anti-inflammatory effects in ischemic insult. Thus, ACE2/Ang(1–7)/MASR axis is considered to play a protective role in the treatment of ischemic stroke, as well as other cerebrovascular diseases.
2.3. Brain angiotensinogen
Angiotensinogen (AGT) is a fatherly precursor peptide of RAS where other angiotensin peptides originate from. A wealth of evidence has shown that the 485 amino acid long AGT molecule exists in the brain even though the precise cellular location of its origin is still unknown (Grobe et al., 2008). Several studies reported that more than 90% of AGT peptides are produced in the astrocytes (Milsted et al., 1990, Grobe et al., 2008, Sherrod et al., 2005) and partly, their synthesis have also been reported in neurons (Lavoie et al., 2004, Agassandian et al., 2017, Stornetta et al., 1988, Yang et al., 1999) and glial cells (Lavoie et al., 2004, Morimoto et al., 2001). In addition, the mRNA (Uijl et al., 2018, Nguyen et al., 2002) and intracellular forms of the peptides have been detected in several brain regions (Bodiga and Bodiga, 2013, Sherrod et al., 2005). The function of AGT is obscure in the brain. However, few studies reported that hypertension and fluid balance are regulated by AGT modulation in rats and mice (Lavoie et al., 2004, Morimoto et al., 2001). This may be explained by the localization of AGT in the brain areas mediating cardiovascular functions (Uijl et al., 2018).
2.4. Angiotensin peptides and receptors
Angiotensin I (Ang I), a physiologically inactive peptide has been found in the brain, although at reportedly low levels (Nguyen et al., 2002). In addition, existence of Ang II and its receptors have also been confirmed in brain (Nakagawa and Sigmund, 2017, Schinke et al., 1999, Schelling et al., 1983). Furthermore, AT1R is found to be expressed in abundance while AT2R expression is restricted and lower in the brain (Guimond and Gallo-Payet, 2012). AT1R facilitates most of the damaging effects of Ang II while AT2R counters the action of the former. Infusion of Ang II in the brain increases AT1R expression thereby regulating the brain RAS activity (Wei et al., 2009). The cellular damaging effects of the activation of AT1R is mediated by signaling through mitogen-activated protein kinase (MAPK) and c-Jun N-terminal kinase (JNK) stimulation. (Wei et al., 2008). Ang II regulates the neurophysiology of several regions of the brain, including cardiovascular centres, probably by binding to neurons in the GABAergic and glutamergic pathways of the hypothalamus (Mascolo et al., 2017). Mogi and colleagues observed that Ang II signaling via AT1R led to neurotoxicity and BBB damage (Millan et al., 1991), while another study implicated the angiotensin peptide in the progression of AD (Iwai and Inagami, 1992). Conversely, Ang II acting along AT2R axis facilitated increase in brain mass and neuronal sprouting (Kakar et al., 1992). Ang II can be converted to another angiotensin peptide, angiotensin III (Ang III) by an enzyme, aminopeptidase (AMN) via the removal of aspartic acid residue at the amino terminal. It has also been demonstrated that Ang III binds to AT1R, AT2R and non AT-receptors (Mogi et al., 2006, Mogi and Horiuchi, 2013, Miners et al., 2010, Goel et al., 2017). Ang III has been shown to be an important peptide in BP regulation, even more than Ang II (Gao et al., 2014). Ang III can also form angiotensin IV (Ang IV) when acted upon by AMN, although ang IV can be formed from ang II directly via the action of dipeptidyl aminopeptidase (de Morais et al., 2018). Ang IV acts via angiotensin receptor type-4 to mediate protective mechanisms in neurodegenerative diseases (Llorens-Cortes and Mendelsohn, 2002).
3. Brain RAS mediated mechanisms of neurodegeneration and neuroprotection
3.1. Oxidative stress
Excessive levels of oxidative stress have been reported to be an underlining factor in several neurodegenerative diseases (Wright et al., 2012, Tota et al., 2012). Brain RAS could be a risk factor for oxidative stress that mediates neuronal dysfunction (de Morais et al., 2018). Accumulating evidences suggest that ang II plays a vital role in this process while the role of other components of RAS involved in neurodegeneration remains elusive (Comoglio et al., 2008). Through the AT1R, ang II activates the production of reactive oxygen species (ROS) mediated by nicotinamide adenine dinucleotide phosphate (NADPH) oxidase (NOX), an enzyme found in several regions of the brain (Fernando et al., 2005, Wright and Harding, 2011, Solleiro-Villavicencio and Rivas-Arancibia, 2018, Su et al., 2016).
Ang II induction of NOX complex stimulates superoxide generation by inflammed cells causing death of dopaminergic neurons (Chan et al., 2005). In addition, induction of AT1R also stimulates nuclear factor kappa -light-chain-enhancer of activated B cells (NF-kB) signal pathway that may contribute to neuronal death as reported in case of PD. (Joglar et al., 2009). Furthermore, ang II influences neuronal N-methyl-D aspartate (NMDA) currents, leading to increased NOX-2 dependent generation of ROS (Wang et al., 2013). Studies have shown that superoxide produced by NOX, activates NF-kB and the RhoA/Rho kinase pathway in microglial cells, which promotes NOX stimulation via p38 mitogen-activated protein kinase (Rodriguez-Perez et al., 2015). Angiotensin II may also influence nitric oxide (NO) production through mechanisms involving AT2R subtypes (Labandeira-Garcia et al., 2017). Nitric oxide is produced by brain-type NO-synthase (bNOS) present in many neurons of various brain regions (Iwase et al., 1998). In the brain, some studies indicate that NO might participate in some ang II-regulated processes including development of stroke in spontaneously hypertensive rats [SHRs] (Ahmad 1997). Furthermore, stimulation of brain RAS by increased production of ang II in hypertensive rats caused higher levels of superoxide and malondialdehydes, and lower levels of antioxidant enzymes such as superoxide dismutase and glutathione (Qing et al., 2017). Thus, these studies provide a strong evidence of the activation of RAS system in oxidative damage that may trigger apoptosis and generation of intracellular ROS in neurodegenerative diseases of brain.
3.2. Neuro-inflammation
Both circulatory and local RAS components have been reported to play key roles in neuroinflammation-neurodegeneration pathway, with ang II suggested as the main neuroinflammatory trigger (Chen et al., 2016). Peripheral RAS components do not have total access to the brain due to the blockage effect of the BBB. However, in a disease state, the integrity of this barrier may be affected, allowing hormonal RAS components entry into the cerebral regions, especially the cardiovascular regulatory areas (Palmer et al., 2013). Studies have shown that brain RAS triggers chronic inflammation leading to the release of ROS and inflammatory mediators through activating glial cells (Carvalho and Moreira, 2018). Uncontrolled effects of these mediators upregulate various inflammatory cascades resulting in cognitive dysfunction and neurodegeneration (Sun et al., 2018, Martire et al., 2015).
In a recent review, O’Connor and colleagues reported that excessive ang II generated due to astrocyte dysfunction resulted in neuroinflammation and loss of neuronal activity (Amin et al., 2016). In Addition, inflammation and other abnormal neurovascular alterations resulting from the effect of RAS along the AT1R axis have been linked to AD, although detailed mechanisms are still elusive (Carvalho and Moreira, 2018). Ang II treatment of mesencephalic cultures, caused activation of the microglial RhoA/ROCK pathway and the upregulation of microglia tumor necrotic factor, TNF α, a proinflammatory cytokine that facilitate dopaminergic neurodegeneration (Labandeira-Garcia et al., 2017, Saavedra, 2012). Torika and colleagues reported abnormal amounts of ACE and ang II found in neuroinflammation predisposed alzheimer model (Torika et al., 2016). According to in vivo studies, induction of α-synuclein, a pro-microglial inflammatory molecule, also resulted in death of dopaminergic neurons in substantia nigra (Barkholt et al., 2012, Rodriguez-Perez et al., 2018). This trend of neuroinflammation in the brain has also been demonstrated in various pharmaco-inhibition studies. The use of perindopril, a potent ACEI, in combating neuro-cognition associated pathologies is reported and supported by Perindopril Protection Against Recurrent Stroke Study (Tzourio et al., 2003). Interestingly, oral administration of perindopril in SHRs improved memory and reduced neurodegeneration by lowering NF-kB pathway, possibly by lowering the level of ang II (Goel et al., 2015). A similar anti-inflammatory effect was observed in 5X familial AD mice after intranasal application of telmisartan [ARB] and perindopril [ACEI] (Torika et al., 2016). Other research groups have reported that candesartan and other potent AT1R blockers successfully attenuated death of dopaminergic neurons induced by the α-synclein. They reported that the blockade of the Ang II/AT1R axis and upregulation of peroxisome proliferator-activated receptor gamma might reduce inflammation by modulating neurotrophic factors (NFs) that activate neuroprotective pathways (Wang et al., 2014, Sathiya et al., 2013, Tong et al., 2016). In addition, telmisartan was found to attenuate lipopolysaccharide induced NO, TNF-α, interleukin 1 beta (IL1-β) as a possible novel neuroprotective mechanism, reinforcing the hypothesis that brain RAS may regulate memory via neuroinflammation (Montgomery and Bowers, 2012, Nakamura and Lipton, 2011, Goel et al., 2015).
3.3. Apoptosis
The concept that brain RAS stimulates apoptosis mediated neuronal cell death has been well reported and ang II is recognized as the major component acting via AT1R. Ang II induced oxidative stress mediated by NOX may trigger apoptosis (Zhao et al., 2015, Yamamoto et al., 2008). Ang II could also facilitate apoptosis via mitochondrial ROS mediated upregulation of both adenosine monophosphate activated protein kinase and peroxisome proliferator-activated receptor coactivator 1α cascades, thereby stimulating Bax, a pro-apoptotic protein that causes the death of neuronal cells (Kim et al., 2017). In a study when ang II was administered in PD rat model, caspase 3 levels increased abruptly leading to the death of dopaminergic neurons due to cleavage of neuronal membrane proteins (Gao et al., 2017). Additionally, AT1R regulated autophagy stimulation and mitochondrial mediated cell death cascades were also reported as separate mechanisms that may cause ang II induced dopaminergic cell death (Gao et al., 2016, Ou et al., 2016). Apoptosis induced phosphoinositol 3 kinase/protein kinase B (P13K/Akt) signaling cascade might also be one of the fundamental mechanisms responsible for neurodegeneration as reported in postnatal brain of rats (Auladell et al., 2017).
3.4. Neurotrophic factors
Neurotrophic factors (NFs) are proteins with a wide range of functions spanning across central and peripheral nervous system. Centrally, they play diverse functions including resuscitation of dying neurons, synaptic plasticity, axonal development and growth of dendrites. On the other hand, their distorted regulation represents a threat to the neuroprotective and cognitive roles they play thereby implicating them in a number of neurodegenerative diseases, including AD and PD (Ledda and Paratcha, 2016, Sampaio et al., 2017). Generally, NFs have been classified into 3 groups namely neurotrophins, glial cell line derived neurotrophic factors and neurocytokines (Ibáñez and Andressoo, 2017). Although few others with no particular group have also been reported, including mesencephalic astrocyte-derived neurotrophic factor and cerebral dopamine neurotrophic factor (Lindahl et al., 2017). Because a modulatory connection exists between NFs and brain RAS, several studies have suggested the use of brain angiotensin peptides as potential novel compounds to modulate brain functions such as in improving cognition (Jackson et al., 2018). The neurotrophins are made up of five classes, including nerve growth factor, brain-derived neurotrophic factor (BDNF) and neurotrophin-3, 4, 6 (NT-3, -4, -6) (Skaper 2018). The most widely studied neurotrophin 3 and BDNF cascades have been reported to be regulated by ang II (Schaich et al., 2016).
3.5. Neuroprotection
Oxidative stress has been reported to be an underlying factor that mediates neuronal dysfunction in the brain (de Morais et al., 2018, Comoglio et al., 2008). Ang II is considered as the main instigator that leads to the release of ROS and inflammatory mediators in neurodegenerative diseases (Chen et al., 2016). Ang II induced inflammatory cascades result in cognitive dysfunction and neurodegeneration in the brain (Sun et al., 2018, Martire et al., 2015). On the other hand, decreased amount of ACE2 and Ang (1–7) damages cells in cerebral arteries and enhances the level of oxidative stress (Peña Silva et al., 2012). Activation of ACE 2/Ang (1–7)/MASR axis is considered as a novel approach for neuroprotection, since it suppresses NF-kB signaling pathway and reduces oxidative stress. In cerebral ischemia, ang(1–7) showed anti-inflammatory effects to protect brain ischemia (Jiang et al., 2012). In addition, ang IV is reported to be the natural inhibitor of the insulin-regulated aminopeptidase receptor and used as a neuroprotective agent (Naveri et al., 1994). The ARBs could represent another class of drugs that safeguards the neurophysiological milieu from various cognitive defects (Villapol and Saavedra, 2015). ARBs have been shown to reduce brain disorders via several mechanisms, including reducing neuroinflammation and hypoxia (Saavedra et al., 2011). The two well-known ARBs, irbesartan and telmisartan have been reported to significantly reduce the production of ROS in the retina and retinal explants (Ola et al., 2013, White et al., 2015). Moreover, few studies reported neuroprotection by inhibition of NOX due to antioxidant effect of ARBs. It is also established that ARBs protected the cerebral vasculature and reversed other cerebrovascular pathologies (Farag et al., 2017). Thus, ARBs’ treatment of several neurodegenerative brain disorders in humans might be the new therapeutic intervention to protect physiological and pharmacological insults.
4. Conclusions
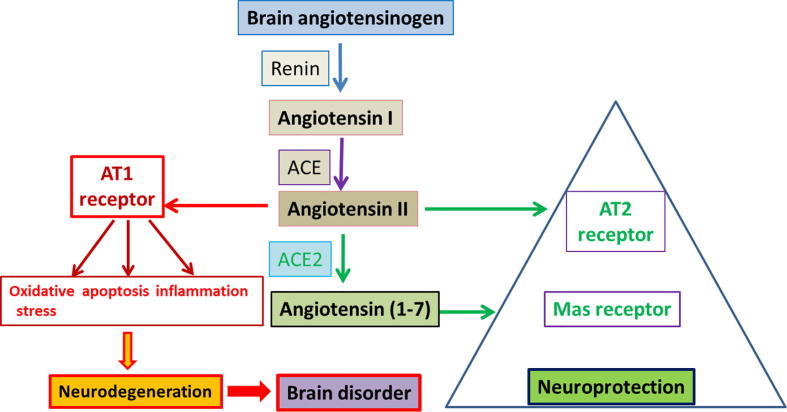
As evidenced from literature, enzymes and peptides related to RAS are expressed and interact with other RAS components (Renin, Ang I, ACE, Ang II, AT1R and AT2R) in the brain. There is strong evidence for the activation of AngII/AT1R axis in initiating a cascade of events leading to increase in oxidative stress, apoptosis, and neuroinflammation causing neurodegeneration in the several brain disorders as depicted in Fig. 1. A number of studies demonstrated strong neuroprotection by blocking the activation of ACE/AngII/AT1R axis using ARBs and also by activation of ACE2/Ang (1-7)/MASR axis. However, few scientists still argue the local expression of the components of RAS especially in the specific cell types within the brain mainly due to poorly characterized commercially available antibodies. The exact physiological and pathological roles of RAS are not evident in the brain and require more studies to elucidate their functions. The improved knowledge of RAS in the brain would open new approaches, novel methods and promising targets to protect several neurodegenerative disease of the brain.
Fig. 1.
Potential links between brain renin angiotensin system, neurodegeneration and neuroprotection.
Declaration of Competing Interest
The authors declared that there is no conflict of interest.
Acknowledgements
Authors want to thank Department of Biochemistry, College of Science, King Saud University for the help and support in preparation of the manuscript and research studies.
Footnotes
Peer review under responsibility of King Saud University.
References
- Agassandian K., Grobe J.L., Liu X., Agassandian M., Thompson A.P., Sigmund C.D. Evidence for intraventricular secretion of angiotensinogen and angiotensin by the subfornical organ using transgenic mice. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2017;312(6):R973–R981. doi: 10.1152/ajpregu.00511.2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ahmad S. Angiotensin receptor antagonists delay nitric oxide-deficient stroke in stroke-prone rats. Eur. J. Pharmacol. 1997;333(1):39–45. doi: 10.1016/s0014-2999(97)01089-3. [DOI] [PubMed] [Google Scholar]
- Ahmed H.A., Ishrat T., Pillai B., Bunting K.M., Patel A., Vazdarjanova A., Waller J.L., Arbab A.S., Ergul A., Fagan S.C. Role of angiotensin system modulation on progression of cognitive impairment and brain MRI changes in aged hypertensive animals – A randomized double- blind pre-clinical study. Behav. Brain Res. 2018;346:29–40. doi: 10.1016/j.bbr.2017.12.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amin F.U., Shah S.A., Kim M.O. Glycine inhibits ethanol-induced oxidative stress, neuroinflammation and apoptotic neurodegeneration in postnatal rat brain. Neurochem. Int. 2016;96:1–12. doi: 10.1016/j.neuint.2016.04.001. [DOI] [PubMed] [Google Scholar]
- Auladell C., de Lemos L., Verdaguer E., Ettcheto M., Busquets O., Lazarowski A., Beas-Zarate C., Olloquequi J., Folch J., Camins A. Role of JNK isoforms in the kainic acid experimental model of epilepsy and neurodegeneration. Front Biosci. 2017;22(5):795–814. doi: 10.2741/4517. [DOI] [PubMed] [Google Scholar]
- Barkholt P., Sanchez-Guajardo V., Kirik D., Romero-Ramos M. Long-term polarization of microglia upon alpha-synuclein overexpression in nonhuman primates. Neuroscience. 2012;208:85–96. doi: 10.1016/j.neuroscience.2012.02.004. (Epub 2012 Feb 9) [DOI] [PubMed] [Google Scholar]
- Bodiga V.L., Bodiga S. Renin angiotensin system in cognitive function and dementia. Asian J. Neurosci. 2013 Article ID 102602. [Google Scholar]
- Carvalho C., Moreira P.I. Oxidative stress: a major player in cerebrovascular alterations associated to neurodegenerative events. Front. Physiol. 2018;9:806. doi: 10.3389/fphys.2018.00806. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chai S.Y., McKinley M.J., Mendelsohn F.A. Distribution of angiotensin converting enzyme in sheep hypothalamus and medulla oblongata visualized by in vitro autoradiography. Clin. Exp. Hypertens. 1987;9:449–460. doi: 10.3109/10641968709164212. [DOI] [PubMed] [Google Scholar]
- Chan S.H.H., Hsu K.-S., Huang C.-C., Wang L.-L., Ou C.-C., Chan J.Y.H. NADPH oxidase–derived superoxide anion mediates angiotensin II–Induced pressor effect via activation of p38 mitogen–activated protein kinase in the rostral ventrolateral medulla. Circ. Res. 2005;97(8):772–780. doi: 10.1161/01.RES.0000185804.79157.C0. [DOI] [PubMed] [Google Scholar]
- Chen M., Lai L., Li X., Zhang X., He X., Liu W., Li R., Ke X., Fu C., Huang Z., Duan C. Baicalein attenuates neurological deficits and preserves blood–brain barrier integrity in a rat model of intracerebral hemorrhage. Neurochem. Res. 2016;41(11):3095–3102. doi: 10.1007/s11064-016-2032-8. [DOI] [PubMed] [Google Scholar]
- Comoglio P.M., Giordano S., Trusolino L. Drug development of MET inhibitors: targeting oncogene addiction and expedience. Nat Rev Drug Discov. 2008;7(6):504–516. doi: 10.1038/nrd2530. [DOI] [PubMed] [Google Scholar]
- de Morais S.D.B., Shanks J., Zucker I.H. Integrative Physiological Aspects of Brain RAS in Hypertension. Curr. Hypertens. Rep. 2018;20(2):10. doi: 10.1007/s11906-018-0810-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doobay M.F., Talman L.S., Obr T.D., Tian X., Davisson R.L., Lazartigues E. Differential expression of neuronal ACE2 in transgenic mice with overexpression of the brain renin-angiotensin system. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2007;292:R373–R381. doi: 10.1152/ajpregu.00292.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Farag E., Sessler D.I., Ebrahim Z., Kurz A., Morgan J., Ahuja S., Maheshwari K., John Doyle D. The renin angiotensin system and the brain: New developments. J. Clin. Neurosci. 2017;46:1–8. doi: 10.1016/j.jocn.2017.08.055. [DOI] [PubMed] [Google Scholar]
- Feng Y., Xia H., Cai Y., Halabi C.M., Becker L.K., Santos R.A. Brain-selective overexpression of human angiotensin-converting enzyme type 2 attenuates neurogenic hypertension. Circ. Res. 2010;106(2):373–382. doi: 10.1161/CIRCRESAHA.109.208645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feng Y., Yue X., Xia H., Bindom S.M., Hickman P.J., Filipeanu C.M., Wu G., Lazartigues E. Angiotensin-converting enzyme 2 Overexpression in the subfornical organ prevents the angiotensin II–mediated pressor and drinking responses and is associated with angiotensin II type 1 receptor downregulation. Circ. Res. 2008;102(6):729–736. doi: 10.1161/CIRCRESAHA.107.169110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ferguson A.V., Washburn D.L.S., Latchford K.J. Hormonal and neurotransmitter roles for angiotensin in the regulation of central autonomic function. Exp. Biol. Med. (Maywood) 2001;226(2):85–96. doi: 10.1177/153537020122600205. [DOI] [PubMed] [Google Scholar]
- Ferguson A.V., Washburn D.L.S., Bains J.S. Regulation of autonomic pathways by angiotensin. Curr. Opin. Endocrinol. Diabet. Obes. 1999;6(1):19. [Google Scholar]
- Fernando R.N., Larm J., Albiston A.L., Chai S.Y. Distribution and cellular localization of insulin-regulated aminopeptidase in the rat central nervous system. J. Comp. Neurol. 2005;487(4):372–390. doi: 10.1002/cne.20585. [DOI] [PubMed] [Google Scholar]
- Fry M., Ferguson A.V. The sensory circumventricular organs: brain targets for circulating signals controlling ingestive behavior. Physiol. Behav. 2007;91(4):413–423. doi: 10.1016/j.physbeh.2007.04.003. [DOI] [PubMed] [Google Scholar]
- Fuxe K., Ganten D., Hökfelt T., Locatelli V., Poulsen K., Stock G., Rix E., Taugner R. Renin-like immunocytochemical activity in the rat and mouse brain. Neurosci. Lett. 1980;18:245–250. doi: 10.1016/0304-3940(80)90292-x. [DOI] [PubMed] [Google Scholar]
- Gallagher P.E., Chappell M.C., Ferrario C.M., Tallant E.A. Distinct roles for ANG II and ANG-(1–7) in the regulation of angiotensin-converting enzyme 2 in rat astrocytes. Am. J. Physiol. Cell Physiol. 2006;290:C420–C426. doi: 10.1152/ajpcell.00409.2004. [DOI] [PubMed] [Google Scholar]
- Ganten D., Marquez-Julio A., Granger P., Hayduk K., Karsunky K.P., Boucher R. Renin in dog brain. Am. J. Physiol. 1971;221(6):1733–1737. doi: 10.1152/ajplegacy.1971.221.6.1733. [DOI] [PubMed] [Google Scholar]
- Gao J., Marc Y., Iturrioz X., Leroux V., Balavoine F., Llorens-Cortes C. A new strategy for treating hypertension by blocking the activity of the brain renin-angiotensin system with aminopeptidase A inhibitor. Clin. Sci. (Lond.) 2014;127(3):135–148. doi: 10.1042/CS20130396. [DOI] [PubMed] [Google Scholar]
- Gao Q., Jiang T., Zhao H.-R., Wu L., Tian Y.-Y., Ou Z., Zhang L., Pan Y., Lu J., Zhang Y.-D. Activation of autophagy contributes to the angiotensin II-triggered apoptosis in a dopaminergic neuronal cell line. Mol. Neurobiol. 2016;53(5):2911–2919. doi: 10.1007/s12035-015-9177-3. [DOI] [PubMed] [Google Scholar]
- Gao Q., Ou Z., Jiang T., Tian Y.Y., Zhou J.S., Wu L., Zhang Y.D. Azilsartan ameliorates apoptosis of dopaminergic neurons and rescues characteristic parkinsonian behaviors in a rat model of Parkinson’s disease. Oncotarget. 2017;8(15) doi: 10.18632/oncotarget.15732. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goel R., Bhat S.A., Hanif K., Nath C., Shukla R. Angiotensin II Receptor blockers attenuate lipopolysaccharide-induced memory impairment by modulation of NF-kappa B-mediated BDNF/CREB expression and apoptosis in spontaneously hypertensive rats. Mol. Neurobiol. 2017 doi: 10.1007/s12035-017-0450-5. [DOI] [PubMed] [Google Scholar]
- Goel R., Bhat S.A., Rajasekar N., Hanif K., Nath C., Shukla R. Hypertension exacerbates predisposition to neurodegeneration and memory impairment in the presence of a neuroinflammatory stimulus: Protection by angiotensin converting enzyme inhibition. Pharmacol. Biochem. Behav. 2015;133:132–145. doi: 10.1016/j.pbb.2015.04.002. [DOI] [PubMed] [Google Scholar]
- Grobe J.L., Buehrer B.A., Hilzendeger A.M., Liu X., Davis D.R., Xu D., Sigmund C.D. Angiotensinergic signaling in the brain mediates metabolic effects of deoxycorticosterone (DOCA)-salt in C57 mice. Hypertension. 2011;57:600–607. doi: 10.1161/HYPERTENSIONAHA.110.165829. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grobe J.L., Grobe C.L., Beltz T.G., Westphal S.G., Morgan D.A., Xu D., de Lange W.J., Li H., Sakai K., Thedens D.R., Cassis L.A., Rahmouni K., Mark A.L., Johnson A.K., Sigmund C.D. The brain renin-angiotensin system controls divergent efferent mechanisms to regulate fluid and energy balance. Cell Metab. 2010;12:431–442. doi: 10.1016/j.cmet.2010.09.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grobe J.L., Xu D., Sigmund C.D. An intracellular renin-angiotensin system in neurons: fact, hypothesis, or fantasy. Physiology. 2008;23(4):187–193. doi: 10.1152/physiol.00002.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Guimond M.O., Gallo-Payet N. The Angiotensin II type 2 receptor in brain functions. Int. J. Hypertens. 2012 doi: 10.1155/2012/351758. (Epub 2012 Dec 25) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamming I., Timens W., Bulthuis M.L.C, Lely A.T, Navis G.J., van Goor H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J. Pathol. 2004;203(2):631–637. doi: 10.1002/path.1570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Harding J.W., Sullivan M.J., Hanesworth J.M., Cushing L.L., Wright J.W. Inability of [125I] Sar 1, Ile8-angiotensin II to move between the blood and cerebrospinal fluid compartments. J. Neurochem. 1988;50(2):554–557. doi: 10.1111/j.1471-4159.1988.tb02946.x. [DOI] [PubMed] [Google Scholar]
- Hermann K., Raizada M.K., Sumners C., Phillips M.I. Presence of renin in primary neuronal and glial cells from rat brain. Brain Res. 1987;437:205–213. doi: 10.1016/0006-8993(87)91637-4. [DOI] [PubMed] [Google Scholar]
- Hua Z.Y., Jun G.J., Hua L.Z., Jing T. Research progress of ACE inhibitory peptide. Cereals Oils. 2011;25:44–46. [Google Scholar]
- Ibáñez C.F., Andressoo J.O. Biology of GDNF and its receptors - Relevance for disorders of the central nervous system. Neurobiol. Dis. 2017;97(Pt B):80–89. doi: 10.1016/j.nbd.2016.01.021. [DOI] [PubMed] [Google Scholar]
- Iwai N., Inagami T. Identification of two subtypes in the rat type I angiotensin II receptor. FEBS Lett. 1992;298(2–3):257–260. doi: 10.1016/0014-5793(92)80071-n. [DOI] [PubMed] [Google Scholar]
- Iwase K., Iyama K.-ichi., Akagi K., Yano S., Fukunaga K., Miyamoto E., Mori M., Takiguchi M. Precise distribution of neuronal nitric oxide synthase mRNA in the rat brain revealed by non-radioisotopic in situ hybridization. Mol. Brain Res. 1998;53(1-2):1–12. doi: 10.1016/s0169-328x(97)00139-3. [DOI] [PubMed] [Google Scholar]
- Jackson L., Eldahshan W., Fagan S.C., Ergul A. Within the brain: the renin angiotensin system. Int. J. Mol. Sci. 2018;19(3) doi: 10.3390/ijms19030876. pii: E876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang T., Gao L., Guo J., Lu J., Wang Y., Zhang Y. Suppressing inflammation by inhibiting the NF-κB pathway contributes to the neuroprotective effect of angiotensin-(1–7) in rats with permanent cerebral ischaemia. Br. J. Pharmacol. 2012;167:1520–1532. doi: 10.1111/j.1476-5381.2012.02105.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang T., Gao L., Lu J., Zhang Y.D. ACE2-Ang-(1–7)-mas axis in brain: a potential target for prevention and treatment of ischemic stroke. Curr. Neuropharmacol. 2013;11(2):209–217. doi: 10.2174/1570159X11311020007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Joglar B., Rodriguez-Pallares J., Rodriguez-Perez A.I., Rey P., Guerra M.J., Labandeira-Garcia J.L. The inflammatory response in the MPTP model of Parkinson's disease is mediated by brain angiotensin: relevance to progression of the disease J. J. Neurochem. 2009;109(2):656. doi: 10.1111/j.1471-4159.2009.05999.x. [DOI] [PubMed] [Google Scholar]
- Jouquey S., Mathieu M.N., Hamon G., Chevillard C. Effect of chronic treatment with trandolapril or enalapril on brain ACE activity in spontaneously hypertensive rats. Neuropharmacology. 1995;34:1689–1692. doi: 10.1016/0028-3908(95)00146-8. [DOI] [PubMed] [Google Scholar]
- Kakar S.S., Riel K.K., Neill J.D. Differential expression of angiotensin II receptor subtype mRNAs (AT-1A and AT-1B) in the brain. Biochem. Biophys. Res. Commun. 1992;185(2):688–692. doi: 10.1016/0006-291x(92)91680-o. [DOI] [PubMed] [Google Scholar]
- Kim M.-S., Lee G.-H., Kim Y.-M., Lee B.-W., Nam H.Y., Sim U.-C., Choo S.-J., Yu S.-W., Kim J.-J., Kim Kwon Y., Who Kim S. Angiotensin II causes apoptosis of adult hippocampal neural stem cells and memory impairment through the action on AMPK-PGC1α signaling in heart failure: role of Ang II on adult neurogenesis in HF. Stem Cells Transl. Med. 2017;6(6):1491–1503. doi: 10.1002/sctm.16-0382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Labandeira-Garcia J.L., Rodríguez-Perez A.I., Garrido-Gil P., Rodriguez-Pallares J., Lanciego J.L., Guerra M.J. Brain renin-angiotensin system and microglial polarization: implications for aging and neurodegeneration. Front. Aging Neurosci. 2017;9:129. doi: 10.3389/fnagi.2017.00129. (eCollection 2017) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lavoie J.L., Cassell M.D., Gross K.W., Sigmund C.D. Adjacent expression of renin and angiotensinogen in the rostral ventrolateral medulla using a dual-reporter transgenic model. Hypertension. 2004;43(53):1116–1119. doi: 10.1161/01.HYP.0000125143.73301.94. [DOI] [PubMed] [Google Scholar]
- Ledda F., Paratcha G. Assembly of neuronal connectivity by neurotrophic factors and leucine-rich repeat proteins. Front Cell Neurosci. 2016;10:199. doi: 10.3389/fncel.2016.00199. (eCollection 2016) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lindahl M., Saarma M., Lindholm P. Unconventional neurotrophic factors CDNF and MANF: structure, physiological functions and therapeutic potential. Neurobiol. Dis. 2017;97:90–102. doi: 10.1016/j.nbd.2016.07.009. [DOI] [PubMed] [Google Scholar]
- Liu J., Liu S., Matsumoto Y., Murakami S., Sugakawa Y., Kami A., Tanabe C., Maeda T., Michikawa M., Komano H., Zou K. Angiotensin type 1a receptor deficiency decreases amyloid β-protein generation and ameliorates brain amyloid pathology. Sci. Rep. 2015;5:12059. doi: 10.1038/srep12059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Llorens-Cortes C., Mendelsohn F.A. Organisation and functional role of the brain angiotensin system. J. Renin-Angiotensin-Aldosterone Syst. 2002;3(Suppl 1):S39–S48. doi: 10.3317/jraas.2002.029. [DOI] [PubMed] [Google Scholar]
- Martire S., Mosca L., d’Erme M. PARP-1 involvement in neurodegeneration: A focus on Alzheimer’s and Parkinson’s diseases. Mech. Ageing Dev. 2015;146–148:53–64. doi: 10.1016/j.mad.2015.04.001. [DOI] [PubMed] [Google Scholar]
- Mascolo A., Sessa M., Scavone C., De Angelis A., Vitale C., Berrino L., Rossi F., Rosano G., Capuano A. New and old roles of the peripheral and brain renin–angiotensin–aldosterone system (RAAS): Focus on cardiovascular and neurological diseases. Int. J. Cardiol. 2017;227:734–742. doi: 10.1016/j.ijcard.2016.10.069. [DOI] [PubMed] [Google Scholar]
- Mecca A.P., Regenhardt R.W., O’Connor T.E., Joseph J.P., Raizada M.K., Katovich M.J. Cerebroprotection by angiotensin-(1–7) in endothelin-1-induced ischaemic stroke. Exp. Physiol. 2011;96:1084–1096. doi: 10.1113/expphysiol.2011.058578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Millan M.A., Jacobowitz D.M., Aguilera G., Catt K.J. Differential distribution of AT1 and AT2 angiotensin II receptor subtypes in the rat brain during development. PNAS. 1991;88:11440–11444. doi: 10.1073/pnas.88.24.11440. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Milsted A., Barna B.P., Ransohoff R.M., Brosnihan K.B., Ferrario C.M. Astrocyte cultures derived from human brain tissue express angiotensinogen mRNA. Proc. Natl. Acad. Sci. U S A. 1990;87(15):5720–5723. doi: 10.1073/pnas.87.15.5720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miners J.S., van Helmond Z., Raiker M., Love S., Kehoe P.G. ACE variants and association with brain A-beta levels in Alzheimer’s disease. Am. J. Transl. Res. 2010;3(1):73–80. [PMC free article] [PubMed] [Google Scholar]
- Sun M.-S., Jin H., Sun X., Huang S., Zhang F.-L., Guo Z.-N., Yang Y. Free radical damage in ischemia-reperfusion injury: an obstacle in acute ischemic stroke after revascularization therapy. Oxid. Med. Cell. Longevity. 2018;2018:1–17. doi: 10.1155/2018/3804979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mogi M., Li J.M., Iwanami J., Min L.J., Tsukuda K., Iwai M. Angiotensin II type-2 receptor stimulation prevents neural damage by transcriptional activation of methyl methane sulfonate sensitive 2. Hypertension. 2006;48(1):141–148. doi: 10.1161/01.HYP.0000229648.67883.f9. [DOI] [PubMed] [Google Scholar]
- Mogi M., Horiuchi M. Effect of angiotensin II type 2 receptor on stroke, cognitive impairment and neurodegenerative diseases. Geriatr. Gerontol. Int. 2013;13(1):13–18. doi: 10.1111/j.1447-0594.2012.00900.x. [DOI] [PubMed] [Google Scholar]
- Montgomery S.L., Bowers W.J. Tumor necrosis factor-alpha and the roles it plays in homeostatic and degenerative processes within the central nervous system. J. Neuroimmune Pharmacol. 2012;7:42–59. doi: 10.1007/s11481-011-9287-2. PMID: 21728035. [DOI] [PubMed] [Google Scholar]
- Morimoto S., Cassell M.D., Beltz T.G., Johnson A.K., Davisson R.L., Sigmund C.D. Elevated blood pressure in transgenic mice with brain-specific expression of human angiotensinogen driven by the glial fibrillary acidic protein promoter. Circ. Res. 2001;89:365–372. doi: 10.1161/hh1601.094988. [DOI] [PubMed] [Google Scholar]
- Nakagawa P., Sigmund C.D. How is the brain renin-angiotensin system regulated? Hypertension (Dallas, Tex.: 1979) 2017;70(1):10–18. doi: 10.1161/HYPERTENSIONAHA.117.08550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nakamura S., Moriguchi A., Morishita R., Yamada K., Nishii T., Tomita N., Ohishi M., Kaneda Y., Higaki J., Ogihara T. Activation of the brain angiotensin system by in vivo human angiotensing converting enzyme gene transfer in rats. Hypertension. 1999;34:302–308. doi: 10.1161/01.hyp.34.2.302. [DOI] [PubMed] [Google Scholar]
- Nakamura T., Lipton S.A. Redox modulation by S-nitrosylation contributes to protein misfolding, mitochondrial dynamics, and neuronal synaptic damage in neurodegenerative diseases. Cell Death Differ. 2011;18:1478–1486. doi: 10.1038/cdd.2011.65. PMID: 21597461. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Naveri L., Stromberg C., Saavedra J.M. Angiotensin IV reverses the acute cerebral blood flow reduction after experimental subarachnoid hemorrhage in the rat. J. Cereb. Blood Flow Metab. 1994;14(6):1096–1099. doi: 10.1038/jcbfm.1994.143. [DOI] [PubMed] [Google Scholar]
- Nguyen G., Delarue F., Burckle C., Bouzhir L., Giller T., Sraer J.D. Pivotal role of the renin/prorenin receptor in angiotensin II production and cellular responses to renin. J. Clin. Invest. 2002;109(11):1417–1427. doi: 10.1172/JCI14276. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ola M.S., Ahmed M.M., Abuohashish H.M., Al-Rejaie S.S., Alhomida A.S. Telmisartan ameliorates neurotrophic support and oxidative stress in the retina of streptozotocin-induced diabetic rats. Neurochem. Res. 2013;38(8):1572–1579. doi: 10.1007/s11064-013-1058-4. [DOI] [PubMed] [Google Scholar]
- Ola M.S., Alhomida A.S., Ferrario C.M., Ahmad S. Curr. Med. Chem. 2017;24(28):3104–3114. doi: 10.2174/0929867324666170407141955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ou Z., Jiang T., Gao Q., Tian Y.Y., Zhou J.S., Wu L., Zhang Y.D. Mitochondrial-dependent mechanisms are involved in angiotensin II-induced apoptosis in dopaminergic neurons. J. Renin-Angiotensin-Aldosterone Syst.: JRAAS. 2016;17(4) doi: 10.1177/1470320316672349. 1470320316672349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palmer J.C., Tayler H.M., Love S. Endothelin-converting enzyme-1 activity, endothelin-1 production, and free radical-dependent vasoconstriction in Alzheimer’s disease. J. Alzheimers Dis. 2013;36:577–587. doi: 10.3233/JAD-130383. [DOI] [PubMed] [Google Scholar]
- Peña Silva R.A., Chu Y., Miller J.D., Mitchell I.J., Penninger J.M., Faraci F.M., Heistad D.D. Impact of ACE2 deficiency and oxidative stress on cerebrovascular function with aging. Stroke. 2012;43(12):3358–3363. doi: 10.1161/STROKEAHA.112.667063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Qing S., Chan-Juan H., Hong-Bao L. Renin-angiotensin system acting on reactive oxygen species in paraventricular nucleus induces sympathetic activation via AT1R/ PKCγ/Rac1 pathway in salt-induced hypertension. Sci. Rep. 2017;7:43107. doi: 10.1038/srep43107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rodriguez-Perez A.I., Borrajo A., Rodriguez-Pallares J., Guerra M.J., Labandeira-Garcia J.L. Interaction between NADPH-oxidase and Rho-kinase in angiotensin II-induced microglial activation. Glia. 2015;63:466–482. doi: 10.1002/glia.22765. [DOI] [PubMed] [Google Scholar]
- Rodriguez-Perez A.I., Sucunza D., Pedrosa M.A., Garrido-Gil P., Kulisevsky J., Lanciego J.L., Labandeira-Garcia J.L. Angiotensin Type 1 receptor antagonists protect against alpha-synuclein-induced neuroinflammation and dopaminergic neuron death. Neurotherapeutics. 2018;15(4):1063–1081. doi: 10.1007/s13311-018-0646-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rygiel K. Can angiotensin-converting enzyme inhibitors impact cognitive decline in early stages of Alzheimer's disease? An overview of research evidence in the elderly patient population. J. Postgrad. Med. 2016;62(4):242–248. doi: 10.4103/0022-3859.188553. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saavedra J.M. Brain and pituitary angiotensin. Endocr. Rev. 1992;13:329–380. doi: 10.1210/edrv-13-2-329. [DOI] [PubMed] [Google Scholar]
- Saavedra J.M., Sánchez-Lemus E., Benicky J. Blockade of brain angiotensin II AT1 receptors ameliorates stress, anxiety, brain inflammation and ischemia: therapeutic implications. Psychoneuroendocrinology. 2011;36(1):1–18. doi: 10.1016/j.psyneuen.2010.10.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saavedra J.M. Angiotensin II AT1 receptor blockers as treatments for inflammatory brain disorders. Clin. Sci. (Lond.) 2012;123:567–590. doi: 10.1042/CS20120078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saavedra J.M. Evidence to consider angiotensin II receptor blockers for the treatment of early Alzheimer’s disease. Cell. Mol. Neurobiol. 2016;36(2):259–279. doi: 10.1007/s10571-015-0327-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sampaio T.B., Savall A.S., Gutierrez M.E.Z., Pinton S. Neurotrophic factors in Alzheimer’s and Parkinson’s diseases: Implications for pathogenesis and therapy. Neural Regen. Res. 2017;12(4):549–557. doi: 10.4103/1673-5374.205084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santos R.A., Simoes e Silva A.C., Maric C. Angiotensin-(1–7) is an endogenous ligand for the G protein-coupled receptor Mas. Proc. Natl. Acad. Sci. U S A. 2003;100:8258–8263. doi: 10.1073/pnas.1432869100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sathiya S., Ranju V., Kalaivani P. Telmisartan attenuates MPTP induced dopaminergic degeneration and motor dysfunction through regulation of alpha-synuclein and neurotrophic factors (BDNF and GDNF) expression in C57BL/6J mice. Neuropharmacology. 2013;73:98–110. doi: 10.1016/j.neuropharm.2013.05.025. [DOI] [PubMed] [Google Scholar]
- Schaich C.L. BDNF acting in the hypothalamus induces acute pressor responses under permissive control of angiotensin II. Auton. Neurosci. 2016 doi: 10.1016/j.autneu.2016.02.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schelling P., Clauser E., Felix D. Regulation of angiotensinogen in the central nervous system. Clin. Exp. Hypertens. A. 1983;5:1047–1061. doi: 10.3109/10641968309048841. [DOI] [PubMed] [Google Scholar]
- Schinke M., Baltatu O., Bohm M., Peters J., Rascher W., Bricca G., Lippoldt A., Ganten D., Bader M. Blood pressure reduction and diabetes insipidus in transgenic rats deficient in brain angiotensinogen. Proc. Natl. Acad. Sci. 1999;96(7):3975–3980. doi: 10.1073/pnas.96.7.3975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sherrod M., Liu X., Zhang X., Sigmund C.D. Nuclear localization of angiotensinogen in astrocytes. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2005;288:R539–R546. doi: 10.1152/ajpregu.00594.2004. [DOI] [PubMed] [Google Scholar]
- Shinohara K., Liu X., Morgan D.A., Davis D.R., Sequeira-Lopez M.L., Cassell M.D., Grobe J.L., Rahmouni K., Sigmund C.D. Selective deletion of the brain specific isoform of renin causes neurogenic hypertension. Hypertension. 2016;68:1385–1392. doi: 10.1161/HYPERTENSIONAHA.116.08242. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shinohara K., Nakagawa P., Gomez J., Morgan D.A., Littlejohn N.K., Folchert M.D., Weidemann B.J., Liu X., Walsh S.A., Ponto L.L., Rahmouni K., Grobe J.L., Sigmund C.D. Selective deletion of renin-b in the brain alters drinking and metabolism. Hypertension. 2017;70(5):990–997. doi: 10.1161/HYPERTENSIONAHA.117.09923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Skaper S.D. Neurotrophic factors: an overview. Methods Mol. Biol. 2018;1727:1–17. doi: 10.1007/978-1-4939-7571-6_1. [DOI] [PubMed] [Google Scholar]
- Solleiro-Villavicencio H., Rivas-Arancibia S. Effect of chronic oxidative stress on neuroinflammatory response mediated by CD4+T cells in neurodegenerative diseases. Front. Cell. Neurosci. 2018;12:114. doi: 10.3389/fncel.2018.00114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sparks M.A., Crowley S.D., Gurley S.B., Mirotsou M., Coffman T.M. Classical renin-angiotensin system in kidney physiology. Compr. Physiol. 2014;4(3):1201–1228. doi: 10.1002/cphy.c130040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sriramula S., Cardinale J.P., Lazartigues E., Francis J. ACE2 overexpression in the paraventricular nucleus attenuates angiotensin II-induced hypertension. Cardiovasc Res. 2011;92(3):401–408. doi: 10.1093/cvr/cvr242. Epub 2011 Sep 27. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sriramula S., Xia H., Xu P., Lazartigues E. Brain-targeted angiotensin-converting enzyme 2 overexpression attenuates neurogenic hypertension by inhibiting cyclooxygenase-mediated inflammation. Hypertension. 2015;65(3):577–586. doi: 10.1161/HYPERTENSIONAHA.114.04691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stornetta R., Hawelu-Johnson C., Guyenet P., Lynch K. Astrocytes synthesize angiotensinogen in brain. Science. 1988;242(4884):1444–1446. doi: 10.1126/science.3201232. [DOI] [PubMed] [Google Scholar]
- Su Q., Liu J.-J., Cui W., Shi X.-L., Guo J., Li H.-B., Huo C.-J., Miao Y.-W., Zhang M., Yang Q., Kang Y.-M. Alpha lipoic acid supplementation attenuates reactive oxygen species in hypothalamic paraventricular nucleus and sympathoexcitation in high salt-induced hypertension. Toxicol. Lett. 2016;241:152–158. doi: 10.1016/j.toxlet.2015.10.019. [DOI] [PubMed] [Google Scholar]
- Takane K., Hasegawa Y., Lin B., Koibuchi N., Cao C., Yokoo T., Kim-Mitsuyama S. Detrimental effects of centrally administered angiotensin II are enhanced in a mouse model of alzheimer disease independently of blood pressure. J. Am. Heart Assoc. 2017;6(4) doi: 10.1161/JAHA.116.004897. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tani M. Angiotensin-converting enzyme in human brain: discrete localization and biochemical properties. Jpn. Heart J. 1991;32:189–201. doi: 10.1536/ihj.32.189. [DOI] [PubMed] [Google Scholar]
- Tipnis S.R., Hooper N.M., Hyde R., Karran E., Christie G., Turner A.J. A human homolog of angiotensin-converting enzyme: cloning and functional expression as a captopril-insensitive carboxypeptidase. J. Biol. Chem. 2000;275:33238–33243. doi: 10.1074/jbc.M002615200. [DOI] [PubMed] [Google Scholar]
- Tong Q., Wu L., Jiang T., Ou Z., Zhang Y., Zhu D. Inhibition of endoplasmic reticulum stress-activated IRE1alpha-TRAF2- caspase-12 apoptotic pathway is involved in the neuroprotective effects of telmisartan in the rotenone rat model of Parkinson's disease. Eur. J. Pharmacol. 2016;776:106–115. doi: 10.1016/j.ejphar.2016.02.042. [DOI] [PubMed] [Google Scholar]
- Torika N., Asraf K., Roasso E., Danon A., Fleisher-Berkovich S. Angiotensin converting enzyme inhibitors ameliorate brain inflammation associated with microglial activation: possible implications for Alzheimer’s disease. J. Neuroimmune Pharmacol. 2016;11:774–785. doi: 10.1007/s11481-016-9703-8. [DOI] [PubMed] [Google Scholar]
- Tota S., Nath C., Najmi A.K., Shukla R., Hanif K. Inhibition of central angiotensin converting enzyme ameliorates scopolamine induced memory impairment in mice: Role of cholinergic neurotransmission, cerebral blood flow and brain energy metabolism. Behav. Brain Res. 2012;232:66–76. doi: 10.1016/j.bbr.2012.03.015. [DOI] [PubMed] [Google Scholar]
- Tzourio C., Anderson C., Chapman N., Woodward M., Neal B., MacMahon S. Effects of blood pressure lowering with perindopril and indapamide therapy on dementia and cognitive decline in patients with cerebrovascular disease. Arch. Intern. Med. 2003;163:1069–1075. doi: 10.1001/archinte.163.9.1069. [DOI] [PubMed] [Google Scholar]
- Uijl E., Ren L., Danser A.H.J. Angiotensin generation in the brain: a re-evaluation. Clin. Sci. (Lond.) 2018;132(8):839–850. doi: 10.1042/CS20180236. [DOI] [PubMed] [Google Scholar]
- van Thiel B.S., Góes Martini A., Te Riet L., Severs D., Uijl E., Garrelds I.M. Brain renin-angiotensin system, does it exist? Hypertension. 2017;69:1136–1144. doi: 10.1161/HYPERTENSIONAHA.116.08922. [DOI] [PubMed] [Google Scholar]
- Villapol S., Saavedra J.M. Neuroprotective effects of angiotensin receptor blockers. Am. J. Hypertens. 2015;28(3):289–299. doi: 10.1093/ajh/hpu197. Epub 2014 Oct 31. [DOI] [PubMed] [Google Scholar]
- Wang G., Coleman C.G., Chan J., Faraco G., Marques-Lopes J., Milner T.A., Pickel V.M. Angiotensin II slow-pressor hypertension enhances NMDA currents and NOX2-dependent superoxide production in hypothalamic paraventricular neurons. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2013;304(12):R1096–R1106. doi: 10.1152/ajpregu.00367.2012. Epub 2013 Apr 10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang J., Pang T., Hafko R., Benicky J., Sanchez-Lemus E., Saavedra J.M. Telmisartan ameliorates glutamate-induced neurotoxicity: roles of AT1 receptor blockade and PPAR gamma activation. Neuropharmacology. 2014;79:249–261. doi: 10.1016/j.neuropharm.2013.11.022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei S.G., Yu Y., Zhang Z.H., Felder R.B. Angiotensin II upregulates hypothalamic AT1 receptor expression in rats via the mitogen-activated protein kinase pathway. Am. J. Physiol. Heart Circ. Physiol. 2009;296(5):H1425–H1433. doi: 10.1152/ajpheart.00942.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wei S.G., Yu Y., Zhang Z.H., Weiss R.M., Felder R.B. Mitogen-activated protein kinases mediate upregulation of hypothalamic angiotensin II type 1 receptors in heart failure rats. Hypertension. 2008;52(4):679–686. doi: 10.1161/HYPERTENSIONAHA.108.113639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- White A.J., Heller J.P., Leung J., Tassoni A., Martin K.R. Retinal ganglion cell neuroprotection by an angiotensin II blocker in an ex vivo retinal explant model. J. Renin. Angiotensin. Aldosterone Syst. 2015;16(4):1193–1201. doi: 10.1177/1470320314566018. [DOI] [PubMed] [Google Scholar]
- Wright J.W., Mizutani S., Harding J.W. Focus on Brain Angiotensin III and Aminopeptidase A in the Control of Hypertension. Int J Hypertens. 2012;124758 doi: 10.1155/2012/124758. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wright J.W., Harding J.W. Brain renin-angiotensin—A new look at an old system. Prog. Neurobiol. 2011;95(1):49–67. doi: 10.1016/j.pneurobio.2011.07.001. Epub 2011 Jul 13. [DOI] [PubMed] [Google Scholar]
- Wright J.W., Harding J.W. The brain renin-angiotensin system: a diversity of functions and implications for CNS diseases. Pflugers Arch. 2013;465:133–151. doi: 10.1007/s00424-012-1102-2. [DOI] [PubMed] [Google Scholar]
- Xia H., de Queiroz T.M., Sriramula S., Feng Y., Johnson T., Mungrue I.N. Brain ACE2 overexpression reduces DOCA-salt hypertension independently of endoplasmic reticulum stress. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2015;308(5):R370–R378. doi: 10.1152/ajpregu.00366.2014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xia H., Feng Y., Obr T.D., Hickman P.J. Angiotensin LE II. type 1 receptor-mediated reduction of angiotensin-converting enzyme 2 activity in the brain impairs baroreflex function in hypertensive mice. Hypertension (Dallas, Tex: 1979) 2009;53(2):210–216. doi: 10.1161/HYPERTENSIONAHA.108.123844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xia H., Lazartigues E. Angiotensin-converting enzyme 2: Central regulator for cardiovascular function. Curr. Hypertens. Rep. 2010;12:170–175. doi: 10.1007/s11906-010-0105-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu J., Sriramula S., Xia H., Moreno-Walton L., Culicchia F., Domenig O. Clinical relevance and role of neuronal AT1 receptors in ADAM17-mediated ACE2 shedding in neurogenic hypertension. Circ. Res. 2017;121(1):43–55. doi: 10.1161/CIRCRESAHA.116.310509. Epub 2017 May 16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu P., Sriramula S., Lazartigues E. ACE2/ANG-(1–7)/Mas pathway in the brain: the axis of good. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2011;300:R804–R817. doi: 10.1152/ajpregu.00222.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yamamoto E., Tamamaki N., Nakamura T., Kataoka K., Tokutomi Y., Dong Y.F., Fukuda M., Matsuba S., Ogawa H., Kim-Mitsuyama S. Excess salt causes cerebral neuronal apoptosis and inflammation in stroke-prone hypertensive rats through angiotensin II-induced NADPH oxidase activation. Stroke J. Cereb. Circ. 2008;39(11):3049–3056. doi: 10.1161/STROKEAHA.108.517284. [DOI] [PubMed] [Google Scholar]
- Yamazato M., Yamazato Y., Sun C., Diez-Freire C., Raizada M.K. Overexpression of angiotensin-converting enzyme 2 in the rostral ventrolateral medulla causes long-term decrease in blood pressure in the spontaneously hypertensive rats. Hypertension (Dallas, Tex: 1979) 2007;49(4):926–931. doi: 10.1161/01.HYP.0000259942.38108.20. [DOI] [PubMed] [Google Scholar]
- Yang G., Gray T.S., Sigmund C.D., Cassell M.D. The angiotensinogen gene is expressed in both astrocytes and neurons in murine central nervous system. Brain Res. 1999;817:123–131. doi: 10.1016/s0006-8993(98)01236-0. [DOI] [PubMed] [Google Scholar]
- Zhang T.L., Fu J.L., Geng Z., Yang J.J., Sun X.J. The neuroprotective effect of losartan through inhibiting AT1/ASK1/MKK4/JNK3 pathway following cerebral I/R in rat hippocampal CA1 region. CNS Neurosci. Therapeut. 2012;18(12):981–987. doi: 10.1111/cns.12015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhao H.R., Jiang T., Tian Y.Y., Gao Q., Li Z., Pan Y., Zhang Y.D. Angiotensin II triggers apoptosis via enhancement of NADPH oxidase-dependent oxidative stress in a dopaminergic neuronal cell line. Neurochem. Res. 2015;40(4):854–863. doi: 10.1007/s11064-015-1536-y. Epub 2015 Feb 11. [DOI] [PubMed] [Google Scholar]