Key Points
Question
Is there a difference in clinical outcomes among patients with acute myocardial infarction (AMI) complicated by cardiogenic shock treated with intravascular microaxial left ventricular assist device (LVAD) vs intra-aortic balloon pump (IABP)?
Findings
In this registry-based retrospective cohort study that included 3360 propensity-matched patients undergoing percutaneous coronary intervention for AMI, treatment with intravascular microaxial LVAD vs IABP was associated with a significantly higher risk of in-hospital death (45.0% vs 34.1%) and in-hospital major bleeding (31.3% vs 16.0%).
Meaning
The use of intravascular microaxial LVAD compared with IABP may be associated with worse in-hospital clinical outcomes among patients with AMI and cardiogenic shock undergoing percutaneous coronary intervention, although study interpretation is limited by the observational design.
Abstract
Importance
Acute myocardial infarction (AMI) complicated by cardiogenic shock is associated with substantial morbidity and mortality. Although intravascular microaxial left ventricular assist devices (LVADs) provide greater hemodynamic support as compared with intra-aortic balloon pumps (IABPs), little is known about clinical outcomes associated with intravascular microaxial LVAD use in clinical practice.
Objective
To examine outcomes among patients undergoing percutaneous coronary intervention (PCI) for AMI complicated by cardiogenic shock treated with mechanical circulatory support (MCS) devices.
Design, Setting, and Participants
A propensity-matched registry-based retrospective cohort study of patients with AMI complicated by cardiogenic shock undergoing PCI between October 1, 2015, and December 31, 2017, who were included in data from hospitals participating in the CathPCI and the Chest Pain-MI registries, both part of the American College of Cardiology’s National Cardiovascular Data Registry. Patients receiving an intravascular microaxial LVAD were matched with those receiving IABP on demographics, clinical history, presentation, infarct location, coronary anatomy, and clinical laboratory data, with final follow-up through December 31, 2017.
Exposures
Hemodynamic support, categorized as intravascular microaxial LVAD use only, IABP only, other (such as use of a percutaneous extracorporeal ventricular assist system, extracorporeal membrane oxygenation, or a combination of MCS device use), or medical therapy only.
Main Outcomes and Measures
The primary outcomes were in-hospital mortality and in-hospital major bleeding.
Results
Among 28 304 patients undergoing PCI for AMI complicated by cardiogenic shock, the mean (SD) age was 65.0 (12.6) years, 67.0% were men, 81.3% had an ST-elevation myocardial infarction, and 43.3% had cardiac arrest. Over the study period among patients with AMI, an intravascular microaxial LVAD was used in 6.2% of patients, and IABP was used in 29.9%. Among 1680 propensity-matched pairs, there was a significantly higher risk of in-hospital death associated with use of an intravascular microaxial LVAD (45.0%) vs with an IABP (34.1% [absolute risk difference, 10.9 percentage points {95% CI, 7.6-14.2}; P < .001) and also higher risk of in-hospital major bleeding (intravascular microaxial LVAD [31.3%] vs IABP [16.0%]; absolute risk difference, 15.4 percentage points [95% CI, 12.5-18.2]; P < .001). These associations were consistent regardless of whether patients received a device before or after initiation of PCI.
Conclusions and Relevance
Among patients undergoing PCI for AMI complicated by cardiogenic shock from 2015 to 2017, use of an intravascular microaxial LVAD compared with IABP was associated with higher adjusted risk of in-hospital death and major bleeding complications, although study interpretation is limited by the observational design. Further research may be needed to understand optimal device choice for these patients.
This study uses registry data to compare risk of in-hospital mortality and major bleeding among patients with acute coronary syndrome and cardiogenic shock managed with an intravascular microaxial left ventricular assist device (LVAD) vs intra-aortic balloon pulsation (IABP).
Introduction
Based on data collected from 1995 to 2013, cardiogenic shock occurs in an estimated 4% to 12%1,2,3 of patients with acute myocardial infarction (AMI) and is associated with substantial morbidity and mortality. Percutaneous coronary intervention (PCI) is the cornerstone of management with a consideration of hemodynamic support with mechanical circulatory support (MCS) devices—most commonly intra-aortic balloon pumps (IABPs) and Impella devices (intravascular microaxial left ventricular assist devices [LVADs]).
Although intravascular microaxial LVADs improve hemodynamic parameters more than IABPs, it is not known whether this translates into improved outcomes among patients. The first intravascular microaxial LVAD received US Food and Drug Administration (FDA) clearance (in 2008 through the 510[k] regulatory pathway) for temporary support for up to 6 hours during cardiac procedures based on substantial equivalence to previously approved circulatory support devices but without a pivotal trial to demonstrate clinical efficacy compared with a control group.4 Intravascular microaxial LVADs were later reclassified as higher-risk class III medical devices in 2014, which now require premarket approval. In April 2016, FDA-approved indications for intravascular microaxial LVADs were expanded through premarket approval to include treatment of cardiogenic shock following AMI. This was based, in part, on a randomized clinical trial (RCT) that showed improved hemodynamics as compared with IABP,5 as well as data from a manufacturer-initiated registry demonstrating improved outcomes relative to historical data.6 Two RCTs that compared intravascular microaxial LVAD and IABP have demonstrated no statistically significant difference in 30-day mortality in AMI complicated by cardiogenic shock.5,7 A matched-pair analysis of 474 patients treated with intravascular microaxial LVAD in clinical practice compared with treatment using IABP (from patients in the IABP-SHOCK II trial) similarly showed no statistically significant mortality difference.8 Despite limited data demonstrating improvements in clinical outcomes relative to IABP, use of intravascular microaxial LVAD has steadily increased over time.9,10
Accordingly, this study sought to use the clinical data collected in 2 national registries to examine clinical outcomes associated with intravascular microaxial LVAD and IABP among patients with AMI complicated by cardiogenic shock undergoing PCI.
Methods
Data Source
For this study, we linked CathPCI and Chest Pain-MI, 2 registries under the American College of Cardiology’s National Cardiovascular Data Registry (described previously).11,12 The CathPCI Registry is a national voluntary registry of diagnostic cardiac catheterizations and PCIs. More than 1500 hospitals across the United States participate and are required to submit data on all PCI procedures. The Chest Pain-MI Registry includes patients with AMI and is used in more than 1000 US hospitals. Both registries capture standardized data elements, including patient demographics, medical history, laboratory data, procedural details, and in-hospital outcomes including mortality and major bleeding. Version 4.4 of the CathPCI Registry includes details of angiographic findings and can identify whether a patient received an IABP or any other MCS device. Version 2.4.2 of the Chest Pain-MI data collection form (released in the third quarter of 2015) includes the type of MCS device. All data submissions must meet prespecified quality standards. The registries include automatic system validation, education and training of staff, reporting of completeness, and random on-site auditing.13 The human investigation committee of the Yale University School of Medicine approved the use of a limited data set from the registry for research without requiring informed consent.
Study Population
All patients who underwent PCI for AMI complicated by cardiogenic shock between October 1, 2015, and December 31, 2017, were identified. Patients with cardiogenic shock were identified as those in the Chest Pain-MI Registry who had cardiogenic shock at first medical contact, as an in-hospital event, or those defined in the CathPCI Registry who had cardiogenic shock within 24 hours prior to and up to PCI, at the start of PCI, or as an intra- or postprocedure event. Cardiogenic shock is defined in both registries as 1, 2, or all 3 of the following: systolic blood pressure lower than 90 mm Hg, a cardiac index of less than 2.2 L/minute/m2 for at least 30 minutes that is secondary to ventricular dysfunction, or requirement for parenteral inotropic or vasopressor or MCS devices to support blood pressure and cardiac index.14 For patients who underwent multiple PCIs during the hospitalization, only data from the initial PCI were included.
Registry Linkage
A probabilistic linkage15 of patients across the 2 registries was performed to include detailed procedural data from the CathPCI Registry and the specific MCS device type from the Chest Pain-MI Registry. Multiple iterations of matching were then performed, with each subsequent match omitting variables that had been previously included. The matching variables were patient sex, date of admission, time of arrival to facility, age at hospital arrival, a unique hospital identifier, discharge date, and whether PCI was performed as documented in the Chest Pain-MI Registry. This match algorithm identified patients with entries in both registries at the same hospital. To identify patients with AMI complicated by cardiogenic shock who were transferred to another hospital for PCI or who had minor missing data elements that may have affected the match, up to 4 variables were allowed to be mismatched, but these variables always included sex and at least 1 date variable to ensure temporal factors limited matches for similar patients at different hospital encounters. The resulting linked CathPCI-Chest Pain-MI registry cohort formed our analytic cohort.
Hemodynamic Support
Patients were categorized based on hemodynamic support: IABP only, intravascular microaxial LVAD only, and other (such as use of a percutaneous extracorporeal ventricular assist system, extracorporeal membrane oxygenation, LVAD, or patients receiving multiple devices during the hospitalization). Patients coded as not receiving any MCS device constituted the medical therapy group.
Outcomes
The primary outcomes were all-cause in-hospital death and in-hospital major bleeding. Death was captured in the Chest Pain-MI Registry. Major bleeding was defined using the Chest Pain-MI Registry as a decline in hemoglobin level of at least 3 g/dL; transfusion of whole blood or packed red blood cells; procedural intervention/surgery at bleeding site to treat the bleeding; or documented or suspected retroperitoneal bleed, gastrointestinal bleed, genitourinary bleed, or a bleed in a location not specified elsewhere.16
Covariates
Covariates were obtained from the CathPCI and Chest Pain-MI registries and included patient demographics, medical history, clinical presentation, laboratory values, administered medications, procedural characteristics, and coronary anatomic data. For continuous values with missing values, the mean was imputed. For binary (yes/no) variables, all missing variables were coded as no, and for categorical variables, all missing variables were coded as no or other (if there was not a no category).
Race and ethnicity were included in this study because our goal was to use all available patient information when risk-standardizing through propensity matching. This determination was made by the patient or family member and then entered into the CathPCI Registry. This determination was based on fixed categories, although multiple response options were possible. For race, the categories were white, black/African American, American Indian/Alaskan Native, Asian, and Native Hawaiian/Pacific Islander. For ethnicity, the categories were Hispanic/Latino or not.
Statistical Analysis
First, overall use of hemodynamic support among all patients was characterized. Characteristics of patients receiving intravascular microaxial LVAD vs those receiving IABP were compared using χ2 tests for categorical variables and 1-way analysis of variance or Kruskal-Wallis tests for continuous variables.
Clinical outcomes of mortality and major bleeding among patients undergoing PCI for AMI complicated by cardiogenic shock were characterized using propensity-matched analyses to compare patients who received either intravascular microaxial LVAD or IABP only. Seventy-five variables were preselected for matching using previously described methods.17 Among patients who received either an intravascular microaxial LVAD or IABP, a probabilistic model was developed that calculated the log-odds probability of receiving an intravascular microaxial LVAD. To develop the log-odds probability and to handle higher-dimensional, nonlinear relationships between covariates, a gradient descent–boosted decision tree algorithm was used to develop the propensity model (called extreme gradient boosting).18 The hyperparameters of learning rate were set to 0.1, as is common in slow-learning algorithms, and the number of trees and maximum depth of each tree was selected optimally in a 5-fold cross-validation analysis (depth range, 1-10; number of trees range, 50-1000 in increments of 10). The final model used a depth of 3 for each decision tree and 100 decision trees, which optimally maximized the C statistic for discriminating between intravascular microaxial LVAD and IABP (eTables 1 and 2 in the Supplement).
For each patient who received an intravascular microaxial LVAD, we found all IABP patients with a similar propensity for intravascular microaxial LVAD usage (within 0.6 standard deviations, a value that eliminates approximately 90% of the bias in observed confounders)19 and randomly selected 1 IABP patient for paired matching. This pair was then removed from the cohort, and the process was repeated until all patients were either matched or could not be matched due to probability differences.
The standardized mean difference of each covariate was calculated in the propensity-matched cohort. Next, outcomes in the cohort were examined and the absolute risk difference (ARD) and associated 95% CIs were calculated. To verify results, a second independent statistician blinded to the results of the initial analysis confirmed results from the gradient descent–boosted decision tree algorithm using standard logistic regression to propensity match patients using 75 variables.
For sensitivity, analyses were repeated stratified by timing of MCS device placement (either before or after initiation of PCI, when these data were available) in patients from hospitals that had placed at least 1 intravascular microaxial LVAD and IABP, therefore demonstrating capability to use both devices, and in patients who were not transferred to a facility.
As a secondary analysis, a comparison of patients receiving IABP vs medical therapy only was made (using the methods previously described) to determine whether outcomes using propensity matching were similar to those observed from the IABP-SHOCK II trial.20
As an additional step to address potential unmeasured confounding, an instrumental variable analysis was conducted using hospital-level propensity to use intravascular microaxial LVAD during our study period as the instrumental variable. A 2-stage ordinary least-squares regression analysis was conducted. In the first stage, the predicted probability of receiving intravascular microaxial LVAD at the facility-level was calculated after adjustment for covariates included in our propensity-score matching. The F statistic was calculated to determine the strength of the instrumental variable (a value >10 suggested proceeding to the second stage). In the second stage, the predicted probability of receiving intravascular microaxial LVAD (determined during the first stage) was used as the primary predictor, again adjusting for the same covariates, to examine differences in in-hospital clinical outcomes. The instrumental variable analysis was conducted in 2 populations: in the entire cohort of patients with AMI complicated by cardiogenic shock and in the patients who received intravascular microaxial LVAD only or IABP only. Analyses were conducted in R, with packages XGBoost for gradient descent boosting18 and pROC for C statistic calculations.21 The primary analyses examining outcomes of intravascular microaxial LVAD vs IABP were repeated using SAS version 9.4. All statistical analyses were 2-sided (α=.05 for statistical significance).
Results
Study Cohort
Of the 269 303 patients with AMI receiving PCI between October 1, 2015, and December 31, 2017, and matched across the Chest Pain-MI and CathPCI registries, 28 304 (10.5%) were classified as having cardiogenic shock. The mean (SD) age was 65.0 (12.6) years (Table). Approximately two-thirds of patients were men and 86% were white. Approximately 25% had been transferred from another acute care hospital, 81% presented with acute ST-segment elevation myocardial infarction, 38.9% had anterior infarct location, and 43.3% had cardiac arrest either at first medical contact or during hospitalization. Among those with cardiogenic shock at first medical contact, the mean (SD) systolic blood pressure was 94.9 (51.4) mm Hg.
Table. Characteristics of Patients Undergoing Percutaneous Coronary Intervention for Acute Myocardial Infarction Complicated by Cardiogenic Shock and of Propensity-Matched Patients Receiving Intravascular Microaxial Left Ventricular Assist Device vs Intra-aortic Balloon Pump From October 1, 2015, Through December 31, 2017.
| Patient Characteristicsa | Medical Therapy Alone | Other Mechanical Circulatory Support | Intravascular Microaxial Left Ventricular Assist Device Only | Intra-aortic Balloon Pump Only | P Valueb | Propensity Matched | Standardized Mean Difference | |
|---|---|---|---|---|---|---|---|---|
| Intravascular Microaxial Left Ventricular Assist Device | Intra-aortic Balloon Pump | |||||||
| Total patients | 16 227 (57.3) | 1838 (6.5) | 1768 (6.2) | 8471 (29.9) | 1680 | 1680 | ||
| Age, mean (SD), y | 65.3 (12.8) | 63.0 (12.4) | 64.2 (12.0) | 65.2 (12.4) | <.001 | 64.3 (11.9) | 64.0 (11.9) | 0.03 |
| Body mass index, mean (SD)c | 28.9 (6.5) | 29.3 (6.6) | 29.7 (6.3) | 28.9 (6.1) | <.001 | 29.6 (6.3) | 30.0 (6.4) | 0.06 |
| Men | 10 517 (64.8) | 1326 (72.1) | 1260 (71.3) | 5865 (69.2) | <.001 | 1194 (71.1) | 1198 (71.3) | 0.005 |
| Women | 5710 (35.2) | 512 (27.9) | 508 (28.7) | 2606 (30.8) | 486 (28.9) | 482 (28.7) | 0.005 | |
| Raced | ||||||||
| White | 13 915 (85.8) | 1552 (84.4) | 1465 (82.9) | 7194 (84.9) | <.001 | 1390 (82.7) | 1414 (84.2) | 0.04 |
| Black | 1422 (8.8) | 167 (9.1) | 185 (10.5) | 739 (8.7) | 178 (10.6) | 170 (10.1) | 0.02 | |
| Asian | 491 (3.0) | 75 (4.1) | 43 (2.4) | 334 (3.9) | 41 (2.4) | 48 (2.9) | 0.03 | |
| American Indian | 178 (1.1) | 16 (0.9) | 42 (2.4) | 89 (1.1) | 37 (2.2) | 27 (1.6) | 0.04 | |
| Native Hawaiian/Pacific Islander | 42 (0.3) | 2 (0.1) | 5 (0.3) | 34 (0.4) | 5 (0.3) | 9 (0.5) | 0.04 | |
| Not recorded | 222 (1.4) | 34 (1.8) | 33 (1.9) | 123 (1.5) | NA | NA | NA | |
| Hispanic or Latino ethnicitye | 1057 (6.5) | 126 (6.9) | 116 (6.6) | 576 (6.8) | .82 | NA | NA | NA |
| Insurancef | ||||||||
| Private | 9172 (56.5) | 1058 (57.6) | 1003 (56.7) | 4828 (57.0) | .79 | 957 (57.0) | 959 (57.1) | 0.002 |
| Medicare | 8330 (51.3) | 797 (43.4) | 857 (48.5) | 4232 (50.0) | <.001 | 812 (48.3) | 792 (47.1) | 0.02 |
| Medicaid | 2072 (12.8) | 225 (12.2) | 205 (11.6) | 1060 (12.5) | .51 | 194 (11.5) | 206 (12.3) | 0.02 |
| None | 1540 (9.5) | 189 (10.3) | 178 (10.1) | 796 (9.4) | .57 | 173 (10.3) | 189 (11.3) | 0.002 |
| Otherg | 916 (5.6) | 105 (5.7) | 105 (5.9) | 454 (5.4) | .70 | 101 (6.0) | 95 (5.7) | 0.02 |
| Clinical history | ||||||||
| Cardiovascular risk factors | ||||||||
| Hypertension | 11 394 (70.2) | 1214 (66.1) | 1239 (70.1) | 5784 (68.3) | <.001 | 1176 (70.0) | 1160 (69.0) | 0.02 |
| Dyslipidemia | 9443 (58.2) | 979 (53.3) | 1023 (57.9) | 4760 (56.2) | <.001 | 971 (57.8) | 923 (54.9) | 0.06 |
| Current/recent smoker | 6169 (38.0) | 625 (34.0) | 548 (31.0) | 2800 (33.1) | <.001 | 516 (30.7) | 545 (32.4) | 0.04 |
| Diabetes mellitus | 5118 (31.5) | 637 (34.7) | 646 (36.5) | 2857 (33.7) | <.001 | 609 (36.3) | 621 (37.0) | 0.02 |
| Family history of premature coronary artery disease | 1914 (11.8) | 201 (10.9) | 191 (10.8) | 933 (11.0) | .19 | 184 (11.0) | 168 (10.0) | 0.03 |
| Currently on dialysis | 496 (3.1) | 37 (2.0) | 52 (2.9) | 211 (2.5) | .01 | 48 (2.9) | 61 (3.6) | 0.04 |
| Established coronary artery disease | ||||||||
| Prior PCI | 3772 (23.2) | 389 (21.2) | 423 (23.9) | 1861 (22.0) | .03 | 398 (23.7) | 369 (22.0) | 0.04 |
| Prior MI | 3570 (22.0) | 369 (20.1) | 394 (22.3) | 1755 (20.7) | .04 | 373 (22.2) | 376 (22.4) | 0.004 |
| Prior coronary artery bypass graft | 1368 (8.4) | 128 (7.0) | 127 (7.2) | 605 (7.1) | .001 | 125 (7.4) | 124 (7.4) | 0.002 |
| Established cardiovascular disease | ||||||||
| Prior heart failure | 1939 (11.9) | 223 (12.1) | 226 (12.8) | 963 (11.4) | .30 | 212 (12.6) | 231 (13.8) | 0.02 |
| Heart failure within 2 weeks | 2941 (18.1) | 550 (29.9) | 603 (34.1) | 2177 (25.7) | <.001 | 549 (32.7) | 563 (33.5) | 0.02 |
| New York Heart Association class I-III | 1364 (8.4) | 141 (7.7) | 182 (10.3) | 798 (9.4) | .002 | 175 (10.4) | 167 (9.9) | 0.02 |
| New York Heart Association class IV | 1556 (9.6) | 406 (22.1) | 419 (23.7) | 1372 (16.2) | <.001 | 373 (22.2) | 395 (23.5) | 0.03 |
| Cardiomyopathy or left ventricular dysfunction | 1886 (11.6) | 350 (19.0) | 366 (20.7) | 1350 (15.9) | <.001 | 333 (19.8) | 350 (20.8) | 0.02 |
| Atrial fibrillation/flutter | 1373 (8.5) | 109 (5.9) | 144 (8.1) | 626 (7.4) | <.001 | 138 (8.2) | 137 (8.2) | 0.002 |
| Established vascular disease | ||||||||
| Cerebrovascular disease | 1898 (11.7) | 176 (9.6) | 191 (10.8) | 877 (10.4) | .002 | 181 (10.8) | 176 (10.5) | 0.01 |
| Peripheral artery disease | 1618 (10.0) | 120 (6.5) | 172 (9.7) | 641 (7.6) | <.001 | 161 (9.6) | 147 (8.8) | 0.03 |
| Chronic lung disease | 2503 (15.4) | 216 (11.8) | 223 (12.6) | 1072 (12.7) | <.001 | 205 (12.2) | 215 (12.8) | 0.02 |
| Cancer | 1463 (9.0) | 103 (5.6) | 152 (8.6) | 721 (8.5) | <.001 | 146 (8.7) | 144 (8.6) | 0.004 |
| Heart rate, median (IQR), beats/min | 76 (57-97) | 83 (63-106) | 87 (68-106) | 83 (63-104) | <.001 | 83.7 (35.3) | 82.6 (36.1) | 0.03 |
| Systolic blood pressure, median (IQR), mm Hg | 120 (92-147) | 114 (87-140) | 116 (90-142) | 118 (92-143) | <.001 | 112.7 (46.3) | 111.4 (47.5) | 0.03 |
| Cardiac arrest within 24 h of PCI | 4441 (27.4) | 739 (40.2) | 624 (35.3) | 2666 (31.5) | <.001 | 578 (34.4) | 601 (35.8) | 0.03 |
| Cardiac arrest at first medical contact | 3791 (23.4) | 531 (28.9) | 449 (25.4) | 2060 (24.3) | <.001 | 421 (25.1) | 455 (27.1) | 0.05 |
| Heart failure at first medical contact | 2611 (16.1) | 476 (25.9) | 464 (26.2) | 1835 (21.7) | <.001 | 425 (25.3) | 474 (28.2) | 0.07 |
| Acute ST-segment elevation myocardial infarction before PCI | 12 938 (79.7) | 1552 (84.4) | 1383 (78.2) | 7148 (84.4) | <.001 | 1324 (78.8) | 1327 (79.0) | 0.004 |
| Thrombolytics given | 518 (3.2) | 60 (3.3) | 50 (2.8) | 257 (3.0) | .78 | 50 (3.0) | 62 (3.7) | 0.04 |
| Anterior infarction | 5207 (32.1) | 903 (49.1) | 894 (50.6) | 4019 (47.4) | <.001 | 846 (50.4) | 879 (52.3) | 0.01 |
| Left main and/or proximal left anterior descending artery disease | 5434 (33.5) | 1092 (59.4) | 1106 (62.6) | 4630 (54.7) | <.001 | 1037 (61.7) | 1046 (62.3) | 0.04 |
| Multivessel disease | 7886 (48.6) | 1127 (61.3) | 1171 (66.2) | 5376 (63.5) | <.001 | 1110 (66.1) | 1110 (66.1) | 0.0 |
| Chronic total occlusion | 502 (3.1) | 84 (4.6) | 55 (3.1) | 317 (3.7) | .001 | 51 (3.0) | 65 (3.9) | 0.05 |
| Kidney failure | 1271 (7.8) | 128 (7.0) | 171 (9.7) | 666 (7.9) | .02 | 163 (9.7) | 167 (9.9) | 0.008 |
| Chronic kidney disease | ||||||||
| GFR ≥60 mL/min | 8325 (51.3) | 862 (46.9) | 821 (46.4) | 4102 (48.4) | <.001 | 786 (46.8) | 752 (44.8) | 0.04 |
| GFR ≥45-<60 mL/min | 3276 (20.2) | 400 (21.8) | 377 (21.3) | 1842 (21.7) | .02 | 360 (21.4) | 364 (21.7) | 0.006 |
| GFR ≥30-<45 mL/min | 1798 (11.1) | 243 (13.2) | 229 (13.0) | 1018 (12.0) | .003 | 211 (12.6) | 225 (13.4) | 0.003 |
| GFR <30 mL/min or current dialysis | 1232 (7.6) | 124 (6.7) | 167 (9.4) | 636 (7.5) | .014 | 159 (9.5) | 163 (9.7) | 0.008 |
| GFR data missing | 1617 (10.0) | 210 (11.4) | 176 (10.0) | 885 (10.4) | .20 | 164 (9.8) | 176 (10.5) | 0.02 |
| GFR, median (IQR), mL/min | 64.5 (48.5-81.6) | 62.2 (47.2-78.0) | 61.5 (45.2-77.8) | 62.9 (47.4-78.7) | <.001 | 59.9 (20.6) | 59.4 (21.0) | 0.03 |
| Left ventricular ejection fraction, mean (SD), % | 42.6 (13.5) | 31.6 (14.7) | 29.1 (13.5) | 34.6 (14.0) | <.001 | 32.1 (8.6) | 32.0 (8.6) | 0.02 |
| Electrocardiogram findings | ||||||||
| ST elevation | 12 614 (77.7) | 1506 (81.9) | 1328 (75.1) | 6933 (81.8) | <.001 | 1277 (76.0) | 1288 (76.7) | 0.02 |
| Left bundle-branch block | 160 (1.0) | 37 (2.0) | 38 (2.1) | 97 (1.1) | <.001 | 32 (1.9) | 23 (1.4) | 0.04 |
| Isolated posterior MI | 146 (0.9) | 15 (0.8) | 14 (0.8) | 83 (1.0) | .82 | 15 (0.9) | 15 (0.9) | 0.0 |
| Initial laboratory results, median (IQR)h | ||||||||
| Brain-type natriuretic peptide, pg/mL | 245.0 (69.0-703.0) | 300.0 (103.2-873.2) | 409.5 (121.0-1003.0) | 326.0 (100.0-807.3) | <.001 | 745.2 (692.0)i | 757.8 (746.1)i | 0.02 |
| N-terminal pro–B-type natriuretic peptide, pg/mL | 1796.5 (330.8-6081.3) | 1920.0 (353.0-6210.8) | 2280.0 (585.0-6630.0) | 2030.0 (362.8-6940.3) | .14 | 5795.6 (3375.2)i | 5944.6 (3984.7)i | 0.04 |
| Hemoglobin, g/dL | 13.9 (12.4-15.2) | 14.1 (12.4-15.4) | 14.0 (12.3-15.4) | 14.0 (12.4-15.3) | .01 | 13.7 (2.3)i | 13.8 (2.2)i | 0.05 |
| Hemoglobin A1c, % | 5.9 (5.5-7.1) | 6.0 (5.5-7.5) | 6.0 (5.6-7.4) | 6.0 (5.5-7.4) | .005 | 6.8 (1.3)i | 6.8 (1.4)i | 0.01 |
| International normalized ratioe | 1.1 (1.0-1.2) | 1.1 (1.0-1.3) | 1.1 (1.0-1.3) | 1.1 (1.0-1.2) | <.001 | 1.3 (1.0) | 1.4 (1.2) | 0.03 |
| <2, No. (%) | 15 663 (96.5) | 1759 (95.7) | 1668 (94.3) | 8108 (95.7) | <.001 | NA | NA | NA |
| ≥2, No. (%) | 564 (3.5) | 79 (4.3) | 100 (5.7) | 363 (4.3) | NA | NA | NA | |
| Preprocedure laboratory results, median (IQR)j | ||||||||
| Creatine kinase–MB fraction, ng/mL | 6.8 (2.6-31.6) | 8.6 (3.0-40.9) | 8.8 (3.4-36.3) | 7.8 (3.0-37.8) | <.001 | 52.0 (59.0) | 53.0 (107.9) | 0.01 |
| Troponin I, ng/mL | 0.42 (0.06-4.54) | 0.61 (0.07-6.15) | 1.04 (0.10-8.53) | 0.59 (0.08-6.83) | <.001 | 12.6 (30.0) | 12.5 (30.5) | 0.002 |
| Troponin T, ng/mL | 0.07 (0.01-0.69) | 0.15 (0.01-0.99) | 0.20 (0.01-0.94) | 0.11 (0.01-0.99) | .004 | 5.2 (3.8) | 5.2 (1.6) | 0.02 |
| Creatinine, mg/dL | 1.1 (0.9-1.4) | 1.2 (1.0-1.5) | 1.2 (1.0-1.6) | 1.2 (0.9-1.5) | <.001 | 1.5 (1.1) | 1.5 (1.2) | 0.04 |
| Hemoglobin, g/dL | 13.8 (12.2-15.1) | 13.9 (12.3-15.3) | 13.8 (12.1-15.3) | 13.9 (12.3-15.2) | .002 | 13.6 (2.2) | 13.7 (2.1) | 0.05 |
| Medications administered within first 24 h | ||||||||
| Aspirin | 15 508 (95.6) | 1682 (91.5) | 1643 (92.9) | 7987 (94.3) | <.001 | 1561 (92.9) | 1564 (93.1) | 0.007 |
| Ticagrelor | 7146 (44.0) | 782 (42.5) | 793 (44.9) | 3554 (42.0) | .008 | 756 (45.0) | 754 (44.9) | 0.002 |
| Clopidogrel | 5964 (36.8) | 512 (27.9) | 462 (26.1) | 2857 (33.7) | <.001 | 448 (26.7) | 449 (26.7) | 0.001 |
| Prasugrel | 1655 (10.2) | 151 (8.2) | 148 (8.4) | 638 (7.5) | <.001 | 140 (8.3) | 132 (7.9) | 0.02 |
| β-Blocker | 7244 (44.6) | 447 (24.3) | 439 (24.8) | 2420 (28.6) | <.001 | 427 (25.4) | 411 (24.5) | 0.02 |
| Angiotensin-converting enzyme inhibitor | 2279 (14.0) | 91 (5.0) | 123 (7.0) | 627 (7.4) | <.001 | 117 (7.0) | 131 (7.8) | 0.03 |
| Angiotensin receptor blocker | 444 (2.7) | 23 (1.3) | 35 (2.0) | 132 (1.6) | <.001 | 35 (2.1) | 30 (1.8) | 0.08 |
| PCI status | ||||||||
| Emergency | 12 715 (78.4) | 1352 (73.6) | 1223 (69.2) | 6808 (80.4) | <.001 | 1200 (71.4) | 1168 (69.5) | 0.04 |
| Salvage | 860 (5.3) | 307 (16.7) | 322 (18.2) | 878 (10.4) | <.001 | 273 (16.3) | 297 (17.7) | 0.04 |
| Urgent | 2511 (15.5) | 168 (9.1) | 213 (12.0) | 747 (8.8) | <.001 | 197 (11.7) | 205 (12.2) | 0.02 |
| Elective | 135 (0.8) | 11 (0.6) | 10 (0.6) | 36 (0.4) | .003 | 10 (0.6) | 10 (0.6) | 0 |
| Timing of mechanical circulatory support placement | ||||||||
| Before initiation of PCI | NA | 925 (50.3) | 717 (40.6) | 2078 (24.5) | <.001 | 653 (38.9) | 747 (44.5) | 0.11 |
| After initiation of PCI | NA | 939 (51.1) | 724 (41.0) | 5633 (66.5) | <.001 | 720 (42.9) | 635 (37.8) | 0.10 |
| Transferred into CathPCI–reporting hospital | 3966 (24.4) | 426 (23.2) | 494 (27.9) | 2002 (23.6) | .001 | 461 (27.4) | 451 (26.8) | 0.01 |
| Transferred into Chest Pain-MI–reporting hospital | 4024 (24.8) | 428 (23.3) | 418 (27.3) | 2013 (23.8) | .176 | 450 (26.8) | 449 (26.7) | 0.001 |
Abbreviations: GFR, glomerular filtration rate; IQR, interquartile range; MI, myocardial infarction; PCI, percutaneous coronary intervention.
SI conversion: to convert creatinine to μmol/L, multiply by 88.4.
Values are reported as No. (%) unless otherwise indicated.
P values indicate differences across the 4 categories (medical therapy alone, other mechanical circulatory support, intravascular microaxial left ventricular assist device only, or intra-aortic balloon pump only).
Calculated as weight in kilograms divided by height in meters squared.
The total percent values exceed 100 because patients may report more than 1 race.
These variables were not used in the propensity matching.
The total percent values exceed 100% because some patients have more than 1 insurance type.
Category includes military health care, state-specific plans, Indian Health Service, and non-US insurers.
Indicates the first laboratory specimens drawn between presentation and discharge.
Values are reported as mean (SD).
Indicates the last laboratory specimens drawn between date of arrival and PCI.
Mechanical Circulatory Support Device Utilization
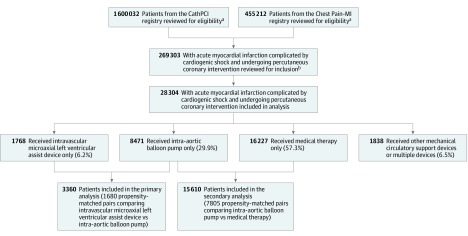
In this cohort of 28 304 patients with AMI complicated by cardiogenic shock undergoing PCI, 1768 (6.2%) received only an intravascular microaxial LVAD, 8471 (29.9%) received only an IABP, 1838 (6.5%) received other MCS devices or multiple devices, and 16 227 (57.3%) received medical therapy alone and were not treated with MCS (Figure 1). Patients receiving intravascular microaxial LVAD were significantly younger than patients receiving IABP (Table; eTable 3 in the Supplement). Patients with intravascular microaxial LVAD were significantly less likely to have acute ST-segment elevation myocardial infarction (78.2%) vs patients with IABP (84.4%; P < .001) but significantly more likely to be transferred into a Chest Pain-MI–reporting facility (patients with intravascular microaxial LVAD [27.3%] vs patients with IABP [23.8%]; P = .02). There was no significant difference in the percentage of patients who experienced cardiac arrest at first medical contact (intravascular microaxial LVAD [25.4%] vs IABP [24.3%]; P = .35).
Figure 1. Patient Population With Acute Myocardial Infarction Complicated by Cardiogenic Shock Undergoing Percutaneous Coronary Intervention.
aCathPCI and Chest Pain-MI are registries under the American College of Cardiology’s National Cardiovascular Data Registry. PCI indicates percutaneous coronary intervention; MI, myocardial infarction.
bPatient data were accessed from linked registries.
Outcomes of Intravascular Microaxial LVAD vs IABP
Unadjusted outcomes are provided in eTable 4 in the Supplement. The 1:1 propensity matching algorithm using data from all patients with AMI complicated by cardiogenic shock undergoing PCI yielded 1680 matched pairs, accounting for 95.0% (1680 of 1768) of patients who received an intravascular microaxial LVAD. Standardized mean differences for 74 of 75 (99%) characteristics of the propensity-matched cohorts were below 0.10 (Table).
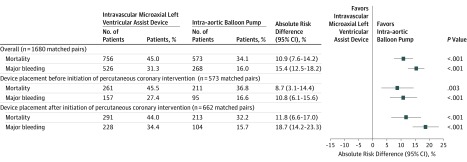
In the propensity-matched cohort, use of intravascular microaxial LVAD was associated with a significantly higher risk of in-hospital death (45.0%) when compared with use of IABP (34.1%; ARD, 10.9 percentage points [95% CI, 7.6-14.2]; P < .001; Figure 2). These statistically significant differences were consistent, regardless of the timing of device placement, among patients with intravascular microaxial LVAD placement before initiation of PCI (45.5%) compared with patients receiving IABP before initiation of PCI (36.8%; ARD, 8.7 percentage points [95% CI, 3.1-14.4]; P = .003), and among those with intravascular microaxial LVAD placement after initiation of PCI (44.0%) compared with IABP after initiation of PCI (32.2%; ARD, 11.8 percentage points [95% CI, 6.6-17.0]; P < .001).
Figure 2. In-Hospital Outcomes Among Propensity-Matched Patients With Acute Myocardial Infarction Complicated by Cardiogenic Shock Undergoing Percutaneous Coronary Intervention With Intravascular Microaxial Left Ventricular Assist Device vs Intra-aortic Balloon Pump.
Use of an intravascular microaxial LVAD was also associated with a significantly higher risk of in-hospital major bleeding (31.3%) compared with use of IABP (16.0%; ARD, 15.4 percentage points [95% CI, 12.5-18.2]; P < .001; Figure 2); both access site and nonaccess site bleeding were significantly higher with intravascular microaxial LVAD (eTable 5 in the Supplement).
A secondary 1:1 propensity-matching algorithm using data from the 390 hospitals that used both intravascular microaxial LVAD and IABP included 1570 matched pairs and found consistent results (eTables 6 and 7 and eFigure 1 in the Supplement). Results were also consistent in an additional 1:1 propensity-matching algorithm among 1201 matched pairs that excluded patients transferred into a treating facility (eTables 8 and 9 and eFigures 2 and 3 in the Supplement). Results were also consistent in an instrumental variable analysis among all patients with AMI complicated by cardiogenic shock and when limited to patients with AMI complicated by cardiogenic shock receiving intravascular microaxial LVAD or IABP only (eTables 10, 11, and 12 in the Supplement).
Outcomes of IABP vs Medical Therapy Alone
The 1:1 propensity-matching algorithm using data from all patients with AMI complicated by cardiogenic shock undergoing PCI yielded 7805 matched pairs. Standardized differences for the characteristics of both propensity-matched cohorts were all below 10% (eTable 13 in the Supplement).
In the propensity-matched cohort, IABP use was not associated with lower in-hospital mortality when compared with medical therapy alone; there was a small but statistically significantly higher risk (IABP, 28.6% vs 26.5% for medical therapy alone; ARD, 2.2 percentage points [95% CI, 0.8-3.6]; P = .002). In-hospital major bleeding was significantly higher among patients receiving IABP (14.5% vs 11.0%; ARD, 3.5 percentage points [95% CI, 2.5-4.5]; P < .001) (eTable 14 and eFigure 4 in the Supplement).
Discussion
Among patients with AMI complicated by cardiogenic shock, use of intravascular microaxial LVAD was associated with significantly higher risks of patients experiencing in-hospital mortality and major bleeding compared with use of IABP. These findings were consistent for patients regardless of the timing of device placement and transfer status.
The significantly higher risk of in-hospital mortality contrasts with prior RCTs, which failed to show a mortality benefit of intravascular microaxial LVAD but did not show overall harm. There are a number of potential explanations for the findings of this study relative to the previous clinical trials. First, by using national registry data, this study was larger than prior RCTs,5,7 which cumulatively enrolled only 74 total patients. Second, this study examined clinical experience, rather than device performance among highly selected patients treated by experienced physicians and hospitals in RCTs.5,7 The recent experience with Impella RP is instructive: the FDA’s May 2019 advisory warning22 suggests experience with devices as they are used in everyday clinical practice must be closely monitored. While a recent matched pair analysis of 237 patients from the IABP-SHOCK II trial and 237 patients receiving intravascular microaxial LVAD in a multinational registry found a point estimate of 2.1% higher mortality among patients receiving intravascular microaxial LVAD compared with IABP that was not statistically significant, that analysis did demonstrate higher risk of severe or life-threatening bleeding in patients receiving intravascular microaxial LVAD.8 The results showing higher risk of severe or life-threatening bleeding are consistent with the current analysis, which shows higher risk of severe in-hospital major bleeding among patients treated with intravascular microaxial LVAD. These results are also consistent with a large observational study of patients undergoing PCI with MCS, which found that use of intrasvascular microaxial LVAD was associated with higher risk of in-hospital adverse events, including death and major bleeding.23 Additionally, mortality risk when comparing patients receiving IABP vs medical therapy only were consistent with those observed in the IABP-SHOCK II trial,20 the largest RCT of IABP in cardiogenic shock, and support the robustness of this analytic approach. A potential explanation for the increase in mortality may be the increased bleeding with intravascular microaxial LVAD as compared with IABP, which is consistent with prior studies.7,24,25 Bleeding and transfusions are associated with adverse outcomes, including mortality, among patients with AMI26,27 and receiving PCI.28
Taken together, these results highlight the need for additional data to guide the optimal management of AMI complicated by cardiogenic shock in general and the role of MCS devices, in particular. Specifically, robust RCTs and complementary analyses of clinical populations are necessary. The former, including the ongoing DanGer trial of intravascular microaxial LVAD vs medical therapy in AMI complicated by cardiogenic shock,29 may provide definitive data on efficacy and safety, while the latter may provide important information about device performance in unselected settings and possible off-label indications.
A 2017 American Heart Association scientific statement noted little evidence to guide the timing or selection of patients with cardiogenic shock who are suitable for MCS devices.30 Furthermore, given that cardiogenic shock is a complex, heterogenous syndrome requiring complex team-based clinical care infrastructure and highly specialized clinicians, a recently released classification scheme31 may improve the phenotyping of patients with shock to better align therapeutic interventions with the cause and degree of hemodynamic derangement. In this context, this analysis of outcomes among patients receiving MCS devices may inform these efforts.
Limitations
This study has several limitations. First, the presence of cardiogenic shock was based on site documentation. More detailed hemodynamic and clinical data, including the use of vasopressor therapy at the time of MCS device placement, would have enabled a more granular patient profile but are not captured in either the Chest Pain-MI or CathPCI case report forms. However, the registry definition for shock is consistent with many clinical trials, and sites are subject to random audit.13 Moreover, the event rate among patients included in the propensity-matched analyses suggests these patients had cardiogenic shock.
Second, registry data provide clinical information, such as hemodynamics and laboratory values, at a single time point; cardiogenic shock is an evolving process, and the specific information at the time of decision to use a particular MCS device was not available. Therefore, the possibility cannot be excluded that comparable patient clinical status at presentation might have changed during subsequent hospital course prior to initiation of therapy and have affected the observed outcome differences between intravascular microaxial LVAD and IABP.
Third, there may be residual confounding whereby patients receiving intravascular microaxial LVADs had greater severity of illness than those receiving IABPs. While this study employed a propensity match using detailed demographics, clinical history and presentation, infarct location, coronary anatomy, and clinical laboratory data from a large, national registry, other clinical parameters that may affect or be associated with MCS device selection, such as right heart catheterization measurements, lactate levels, or success of reperfusion were not available. However, approximately 95% of all patients receiving intravascular microaxial LVADs were matched, suggesting that these results may represent the experience of the majority of patients receiving intravascular microaxial LVAD for AMI complicated by cardiogenic shock.
Fourth, different types of intravascular microaxial LVADs, specifically the Impella 2.5, CP, 5.0, and RP, could not be distinguished. While there are differences in the degree of hemodynamic support provided by these devices, the Impella 5.0 device requires specialized vascular access and is unlikely to be the initial support device used in a patient with AMI complicated by cardiogenic shock. Impella RP was approved in September 2017 and was only available for approximately 3 months of the study period. To further mitigate these concerns, this analysis was limited to patients who received only the intravascular microaxial LVAD or IABP such that patients who had an escalation in their support with device replacement were excluded.
Conclusions
Among patients undergoing PCI for AMI complicated by cardiogenic shock from 2015 to 2017, use of intravascular microaxial LVAD compared with IABP was associated with higher adjusted risk of in-hospital death and major bleeding complications, although study interpretation is limited by the observational design. Further research may be needed to understand optimal device choice for these patients.
eTable 1. C-statistic for Discrimination Between Intravascular Microaxial Left Ventricular Assist Device and Intra-aortic Balloon Pump Among All Hospitals
eTable 2. C-statistic for Discrimination Between Intravascular Microaxial Left Ventricular Assist Device and Intra-aortic Balloon Pump Among All Hospitals with At Least 1 Intra-aortic Balloon Pump and 1 Microaxial Left Ventricular Assist Device
eTable 3. Baseline Characteristics of Patients Undergoing Percutaneous Coronary Intervention for Acute Myocardial Infarction Complicated by Cardiogenic Shock from October 1, 2015 – December 31, 2017, Among All Hospitals with At Least 1 Intra-aortic Balloon Pump and 1 Intravascular Microaxial Left Ventricular Assist Device
eTable 4: Unadjusted Outcomes Among Patients Undergoing Percutaneous Coronary Intervention for Acute Myocardial Infarction Complicated by Cardiogenic Shock from October 1, 2015 – December 31, 2017
eTable 5. Characteristics of Patients in Matched Cohort Undergoing PCI and Receiving Intravascular Microaxial Left Ventricular Assist Device or Intra-aortic Balloon Pump for Acute Myocardial Infarction Complicated by Cardiogenic Shock, Among All Hospitals
eTable 6. Characteristics of Patients in Matched Cohort Undergoing PCI and Receiving Intravascular Microaxial Left Ventricular Assist Device or Intra-aortic Balloon Pump (IABP) for Acute Myocardial Infarction Complicated by Cardiogenic Shock, Among All Hospitals with At Least 1 Intra-aortic Balloon Pump and 1 Intravascular Microaxial Left Ventricular Assist Device
eTable 7. Characteristic of Bleeding Type in Matched Cohort Undergoing PCI and Receiving Intravascular Microaxial Left Ventricular Assist Device or Intra-aortic Balloon Pump for Acute Myocardial Infarction Complicated by Cardiogenic Shock, Among All Hospitals with At Least 1 Intra-aortic Balloon Pump and 1 Intravascular Microaxial Left Ventricular Assist Device
eTable 8. Characteristics of Patients Who Were Not Transferred to a Treating Facility in Matched Cohort Undergoing PCI and Reeiving Intravascular Microaxial Left Ventricular Assist Device or Intra-aortic Balloon Pump for Acute Myocardial Infarction Complicated by Cardiogenic Shock
eTable 9. Characteristics of Patients Who Were Not Transferred to a Treating Facility in Matched Cohort Undergoing PCI and Receiving Intravascular Microaxial Left Ventricular Assist Device or Intra-aortic Balloon Pump for Acute Myocardial Infarction Complicated by Cardiogenic Shock, Among All Hospitals with At Least 1 Intra-aortic Balloon Pump and 1 Intravascular Microaxial Left Ventricular Assist Device
eTable 10. Characteristics of Patients with Acute Myocardial Infarction Complicated by Cardiogenic Shock Undergoing PCI, Stratified by Median by Propensity to Use Intravascular Microaxial Left Ventricular Assist Device
eTable 11. Characteristics of Patients with Acute Myocardial Infarction Complicated by Cardiogenic Shock Undergoing PCI, Stratified by Median by Propensity to Use Intravascular Microaxial Left Ventricular Assist Device, Among Patients Receiving Intra-aortic Balloon Pump or Intravascular Microaxial Left Ventricular Assist Device Only
eTable 12. Instrumental Variable Analysis Results Among Patients Undergoing PCI and Receiving Intravascular Microaxial Left Ventricular Assist Device or Intra-aortic Balloon Pump for Acute Myocardial Infarction Complicated by Cardiogenic Shock
eTable 13. Characteristics of Patients in Matched Cohort Undergoing PCI and Receiving Intra-Aortic Balloon Pump or Medical Therapy Only for Acute Myocardial Infarction Complicated by Cardiogenic Shock, Among All Hospitals
eTable 14. Characteristics of Bleeding Type in Matched Cohort Undergoing PCI and Receiving Intra-Aortic Balloon Pump or Medical Therapy Only for Acute Myocardial Infarction Complicated by Cardiogenic Shock, Among All Hospitals
eFigure 1. In-Hospital Outcomes among Propensity-Matched Patients with Acute Myocardial Infarction Complicated by Cardiogenic Shock Undergoing PCI with Intravascular Microaxial Left Ventricular Assist Device vs Intra-Aortic Balloon Pump, Among All Hospitals with At Least 1 Intra-aortic Balloon Pump and 1 Intravascular Microaxial Left Ventricular Assist Device
eFigure 2. In-Hospital Outcomes among Propensity-Matched Patients Who Were Not Transferred to a Treating Facility with Acute Myocardial Infarction Complicated by Cardiogenic Shock Undergoing PCI with Intravascular Microaxial Left Ventricular Assist Device vs Intra-Aortic Balloon Pump (IABP), Among All Hospitals
eFigure 3. In-Hospital Outcomes among Propensity-Matched Patients with Acute Myocardial Infarction Complicated by Cardiogenic Shock Undergoing PCI with Intravascular Microaxial Left Ventricular Assist Device vs Intra-Aortic Balloon Pump (IABP), Among All Hospitals with At Least 1 Intra-aortic Balloon Pump and 1 Intravascular Microaxial Left Ventricular Assist Device
eFigure 4. In-Hospital Outcomes among Propensity-Matched Patients with Acute Myocardial Infarction Complicated by Cardiogenic Shock Undergoing PCI with Intra-Aortic Balloon Pump vs Medical Therapy Alone
References
- 1.Redfors B, Angerås O, Råmunddal T, et al. 17-Year trends in incidence and prognosis of cardiogenic shock in patients with acute myocardial infarction in western Sweden. Int J Cardiol. 2015;185:256-262. doi: 10.1016/j.ijcard.2015.03.106 [DOI] [PubMed] [Google Scholar]
- 2.Jeger RV, Radovanovic D, Hunziker PR, et al. ; AMIS Plus Registry Investigators . Ten-year trends in the incidence and treatment of cardiogenic shock. Ann Intern Med. 2008;149(9):618-626. doi: 10.7326/0003-4819-149-9-200811040-00005 [DOI] [PubMed] [Google Scholar]
- 3.Kolte D, Khera S, Aronow WS, et al. Trends in incidence, management, and outcomes of cardiogenic shock complicating ST-elevation myocardial infarction in the United States. J Am Heart Assoc. 2014;3(1):e000590. doi: 10.1161/JAHA.113.000590 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Rathi VK, Kesselheim AS, Ross JS. The US Food and Drug Administration 515 Program Initiative: addressing the evidence gap for widely used, high-risk cardiovascular devices? JAMA Cardiol. 2016;1(2):117-118. doi: 10.1001/jamacardio.2016.0002 [DOI] [PubMed] [Google Scholar]
- 5.Seyfarth M, Sibbing D, Bauer I, et al. A randomized clinical trial to evaluate the safety and efficacy of a percutaneous left ventricular assist device versus intra-aortic balloon pumping for treatment of cardiogenic shock caused by myocardial infarction. J Am Coll Cardiol. 2008;52(19):1584-1588. doi: 10.1016/j.jacc.2008.05.065 [DOI] [PubMed] [Google Scholar]
- 6.US Food and Drug Administration . Summary of Safety and Effectiveness Data (SSED): Impella Ventricular Support Systems, 2016. https://www.accessdata.fda.gov/cdrh_docs/pdf14/P140003S004B.pdf. Accessed December 15, 2019.
- 7.Ouweneel DM, Eriksen E, Sjauw KD, et al. Percutaneous mechanical circulatory support versus intra-aortic balloon pump in cardiogenic shock after acute myocardial infarction. J Am Coll Cardiol. 2017;69(3):278-287. doi: 10.1016/j.jacc.2016.10.022 [DOI] [PubMed] [Google Scholar]
- 8.Schrage B, Ibrahim K, Loehn T, et al. Impella support for acute myocardial infarction complicated by cardiogenic shock. Circulation. 2019;139(10):1249-1258. doi: 10.1161/CIRCULATIONAHA.118.036614 [DOI] [PubMed] [Google Scholar]
- 9.Khera R, Cram P, Lu X, et al. Trends in the use of percutaneous ventricular assist devices: analysis of national inpatient sample data, 2007 through 2012. JAMA Intern Med. 2015;175(6):941-950. doi: 10.1001/jamainternmed.2014.7856 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sandhu A, McCoy LA, Negi SI, et al. Use of mechanical circulatory support in patients undergoing percutaneous coronary intervention: insights from the National Cardiovascular Data Registry. Circulation. 2015;132(13):1243-1251. doi: 10.1161/CIRCULATIONAHA.114.014451 [DOI] [PubMed] [Google Scholar]
- 11.Brindis RG, Fitzgerald S, Anderson HV, Shaw RE, Weintraub WS, Williams JF. The American College of Cardiology-National Cardiovascular Data Registry (ACC-NCDR): building a national clinical data repository. J Am Coll Cardiol. 2001;37(8):2240-2245. doi: 10.1016/S0735-1097(01)01372-9 [DOI] [PubMed] [Google Scholar]
- 12.Peterson ED, Roe MT, Rumsfeld JS, et al. A call to ACTION (Acute Coronary Treatment and Intervention Outcomes Network): a national effort to promote timely clinical feedback and support continuous quality improvement for acute myocardial infarction. Circ Cardiovasc Qual Outcomes. 2009;2(5):491-499. doi: 10.1161/CIRCOUTCOMES.108.847145 [DOI] [PubMed] [Google Scholar]
- 13.Messenger JC, Ho KK, Young CH, et al. ; NCDR Science and Quality Oversight Committee Data Quality Workgroup . The National Cardiovascular Data Registry (NCDR) data quality brief: the NCDR data quality program in 2012. J Am Coll Cardiol. 2012;60(16):1484-1488. doi: 10.1016/j.jacc.2012.07.020 [DOI] [PubMed] [Google Scholar]
- 14.Cannon CP, Battler A, Brindis RG, et al. American College of Cardiology key data elements and definitions for measuring the clinical management and outcomes of patients with acute coronary syndromes: a report of the American College of Cardiology Task Force on Clinical Data Standards (Acute Coronary Syndromes Writing Committee). J Am Coll Cardiol. 2001;38(7):2114-2130. doi: 10.1016/s0735-1097(01)01702-8 [DOI] [PubMed] [Google Scholar]
- 15.Brennan JM, Peterson ED, Messenger JC, et al. ; Duke Clinical Research Institute DEcIDE Team . Linking the National Cardiovascular Data Registry CathPCI Registry with Medicare claims data: validation of a longitudinal cohort of elderly patients undergoing cardiac catheterization. Circ Cardiovasc Qual Outcomes. 2012;5(1):134-140. doi: 10.1161/CIRCOUTCOMES.111.963280 [DOI] [PubMed] [Google Scholar]
- 16.American College of Cardiology National Cardiovascular Data Registry . What each registry collects. https://cvquality.acc.org/NCDR-Home/Data-Collection/What-Each-Registry-Collects. Accessed December 15, 2019.
- 17.Mauri L, Silbaugh TS, Garg P, et al. Drug-eluting or bare-metal stents for acute myocardial infarction. N Engl J Med. 2008;359(13):1330-1342. doi: 10.1056/NEJMoa0801485 [DOI] [PubMed] [Google Scholar]
- 18.Chen T, Guestrin C. Xgboost: a scalable tree boosting system. Paper presented at: Proceedings of the 22nd ACM SIGKDD International Conference on Knowledge Discovery and Data Mining; August 13-17, 2016; San Francisco, CA. [Google Scholar]
- 19.Gu XS, Rosenbaum PR. Comparison of multivariate matching methods: structures, distances, and algorithms. J Comput Graph Stat. 1993;2(4):405-420. doi: 10.1080/10618600.1993.10474623 [DOI] [Google Scholar]
- 20.Thiele H, Zeymer U, Neumann FJ, et al. ; IABP-SHOCK II Trial Investigators . Intraaortic balloon support for myocardial infarction with cardiogenic shock. N Engl J Med. 2012;367(14):1287-1296. doi: 10.1056/NEJMoa1208410 [DOI] [PubMed] [Google Scholar]
- 21.Robin X, Turck N, Hainard A, et al. pROC: An open-source package for R and S+ to analyze and compare ROC curves. BMC Bioinformatics. 2011;12(1):77. doi: 10.1186/1471-2105-12-77 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.US Food and Drug Administration . Update: increased rate of mortality in patients receiving Abiomed Impella RP System—letter to health care providers. https://www.fda.gov/medical-devices/letters-health-care-providers/update-increased-rate-mortality-patients-receiving-abiomed-impella-rp-system-letter-health-care. December 2, 2019. January 25, 2020.
- 23.Amin AP, Spertus JA, Curtis JP, et al. The evolving landscape of Impella® use in the United States among patients undergoing percutaneous coronary intervention with mechanical circulatory support [published online November 17, 2019]. Circulation. doi: 10.1161/CIRCULATIONAHA.119.044007 [DOI] [PubMed] [Google Scholar]
- 24.Wernly B, Seelmaier C, Leistner D, et al. Mechanical circulatory support with Impella versus intra-aortic balloon pump or medical treatment in cardiogenic shock-a critical appraisal of current data. Clin Res Cardiol. 2019;108(11):1249-1257. doi: 10.1007/s00392-019-01458-2 [DOI] [PubMed] [Google Scholar]
- 25.Alushi B, Douedari A, Froehlig G, et al. Impella versus IABP in acute myocardial infarction complicated by cardiogenic shock. Open Heart. 2019;6(1):e000987. doi: 10.1136/openhrt-2018-000987 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Eikelboom JW, Mehta SR, Anand SS, Xie C, Fox KA, Yusuf S. Adverse impact of bleeding on prognosis in patients with acute coronary syndromes. Circulation. 2006;114(8):774-782. doi: 10.1161/CIRCULATIONAHA.106.612812 [DOI] [PubMed] [Google Scholar]
- 27.Chatterjee S, Wetterslev J, Sharma A, Lichstein E, Mukherjee D. Association of blood transfusion with increased mortality in myocardial infarction: a meta-analysis and diversity-adjusted study sequential analysis. JAMA Intern Med. 2013;173(2):132-139. doi: 10.1001/2013.jamainternmed.1001 [DOI] [PubMed] [Google Scholar]
- 28.Kinnaird TD, Stabile E, Mintz GS, et al. Incidence, predictors, and prognostic implications of bleeding and blood transfusion following percutaneous coronary interventions. Am J Cardiol. 2003;92(8):930-935. doi: 10.1016/S0002-9149(03)00972-X [DOI] [PubMed] [Google Scholar]
- 29.Udesen NJ, Møller JE, Lindholm MG, et al. ; DanGer Shock investigators . Rationale and design of DanGer shock: Danish-German cardiogenic shock trial. Am Heart J. 2019;214:60-68. doi: 10.1016/j.ahj.2019.04.019 [DOI] [PubMed] [Google Scholar]
- 30.van Diepen S, Katz JN, Albert NM, et al. ; American Heart Association Council on Clinical Cardiology; Council on Cardiovascular and Stroke Nursing; Council on Quality of Care and Outcomes Research; Mission: Lifeline . Contemporary management of cardiogenic shock: a scientific statement from the American Heart Association. Circulation. 2017;136(16):e232-e268. doi: 10.1161/CIR.0000000000000525 [DOI] [PubMed] [Google Scholar]
- 31.Baran DA, Grines CL, Bailey S, et al. SCAI clinical expert consensus statement on the classification of cardiogenic shock: this document was endorsed by the American College of Cardiology (ACC), the American Heart Association (AHA), the Society of Critical Care Medicine (SCCM), and the Society of Thoracic Surgeons (STS) in April 2019. Catheter Cardiovasc Interv. 2019;94(1):29-37. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
eTable 1. C-statistic for Discrimination Between Intravascular Microaxial Left Ventricular Assist Device and Intra-aortic Balloon Pump Among All Hospitals
eTable 2. C-statistic for Discrimination Between Intravascular Microaxial Left Ventricular Assist Device and Intra-aortic Balloon Pump Among All Hospitals with At Least 1 Intra-aortic Balloon Pump and 1 Microaxial Left Ventricular Assist Device
eTable 3. Baseline Characteristics of Patients Undergoing Percutaneous Coronary Intervention for Acute Myocardial Infarction Complicated by Cardiogenic Shock from October 1, 2015 – December 31, 2017, Among All Hospitals with At Least 1 Intra-aortic Balloon Pump and 1 Intravascular Microaxial Left Ventricular Assist Device
eTable 4: Unadjusted Outcomes Among Patients Undergoing Percutaneous Coronary Intervention for Acute Myocardial Infarction Complicated by Cardiogenic Shock from October 1, 2015 – December 31, 2017
eTable 5. Characteristics of Patients in Matched Cohort Undergoing PCI and Receiving Intravascular Microaxial Left Ventricular Assist Device or Intra-aortic Balloon Pump for Acute Myocardial Infarction Complicated by Cardiogenic Shock, Among All Hospitals
eTable 6. Characteristics of Patients in Matched Cohort Undergoing PCI and Receiving Intravascular Microaxial Left Ventricular Assist Device or Intra-aortic Balloon Pump (IABP) for Acute Myocardial Infarction Complicated by Cardiogenic Shock, Among All Hospitals with At Least 1 Intra-aortic Balloon Pump and 1 Intravascular Microaxial Left Ventricular Assist Device
eTable 7. Characteristic of Bleeding Type in Matched Cohort Undergoing PCI and Receiving Intravascular Microaxial Left Ventricular Assist Device or Intra-aortic Balloon Pump for Acute Myocardial Infarction Complicated by Cardiogenic Shock, Among All Hospitals with At Least 1 Intra-aortic Balloon Pump and 1 Intravascular Microaxial Left Ventricular Assist Device
eTable 8. Characteristics of Patients Who Were Not Transferred to a Treating Facility in Matched Cohort Undergoing PCI and Reeiving Intravascular Microaxial Left Ventricular Assist Device or Intra-aortic Balloon Pump for Acute Myocardial Infarction Complicated by Cardiogenic Shock
eTable 9. Characteristics of Patients Who Were Not Transferred to a Treating Facility in Matched Cohort Undergoing PCI and Receiving Intravascular Microaxial Left Ventricular Assist Device or Intra-aortic Balloon Pump for Acute Myocardial Infarction Complicated by Cardiogenic Shock, Among All Hospitals with At Least 1 Intra-aortic Balloon Pump and 1 Intravascular Microaxial Left Ventricular Assist Device
eTable 10. Characteristics of Patients with Acute Myocardial Infarction Complicated by Cardiogenic Shock Undergoing PCI, Stratified by Median by Propensity to Use Intravascular Microaxial Left Ventricular Assist Device
eTable 11. Characteristics of Patients with Acute Myocardial Infarction Complicated by Cardiogenic Shock Undergoing PCI, Stratified by Median by Propensity to Use Intravascular Microaxial Left Ventricular Assist Device, Among Patients Receiving Intra-aortic Balloon Pump or Intravascular Microaxial Left Ventricular Assist Device Only
eTable 12. Instrumental Variable Analysis Results Among Patients Undergoing PCI and Receiving Intravascular Microaxial Left Ventricular Assist Device or Intra-aortic Balloon Pump for Acute Myocardial Infarction Complicated by Cardiogenic Shock
eTable 13. Characteristics of Patients in Matched Cohort Undergoing PCI and Receiving Intra-Aortic Balloon Pump or Medical Therapy Only for Acute Myocardial Infarction Complicated by Cardiogenic Shock, Among All Hospitals
eTable 14. Characteristics of Bleeding Type in Matched Cohort Undergoing PCI and Receiving Intra-Aortic Balloon Pump or Medical Therapy Only for Acute Myocardial Infarction Complicated by Cardiogenic Shock, Among All Hospitals
eFigure 1. In-Hospital Outcomes among Propensity-Matched Patients with Acute Myocardial Infarction Complicated by Cardiogenic Shock Undergoing PCI with Intravascular Microaxial Left Ventricular Assist Device vs Intra-Aortic Balloon Pump, Among All Hospitals with At Least 1 Intra-aortic Balloon Pump and 1 Intravascular Microaxial Left Ventricular Assist Device
eFigure 2. In-Hospital Outcomes among Propensity-Matched Patients Who Were Not Transferred to a Treating Facility with Acute Myocardial Infarction Complicated by Cardiogenic Shock Undergoing PCI with Intravascular Microaxial Left Ventricular Assist Device vs Intra-Aortic Balloon Pump (IABP), Among All Hospitals
eFigure 3. In-Hospital Outcomes among Propensity-Matched Patients with Acute Myocardial Infarction Complicated by Cardiogenic Shock Undergoing PCI with Intravascular Microaxial Left Ventricular Assist Device vs Intra-Aortic Balloon Pump (IABP), Among All Hospitals with At Least 1 Intra-aortic Balloon Pump and 1 Intravascular Microaxial Left Ventricular Assist Device
eFigure 4. In-Hospital Outcomes among Propensity-Matched Patients with Acute Myocardial Infarction Complicated by Cardiogenic Shock Undergoing PCI with Intra-Aortic Balloon Pump vs Medical Therapy Alone