Abstract
Objective:
Maternal depression increases risk for offspring cognitive vulnerabilities, which may be a mechanism underlying the intergenerational transmission of depression. Little is known about how cognitive vulnerabilities, particularly memory biases, develop in offspring of depressed mothers. Understanding the etiology of memory biases may lead to novel intervention targets. Therefore, the current study examined the prospective impact of maternal depression on the development of offspring overgeneral autobiographical memory (OGM; i.e., the tendency to recall less specific memories), a cognitive vulnerability implicated in the intergenerational transmission of depression.
Method:
Participants were offspring (age 8-14; 51% daughters, 81% Caucasian) of mothers with (n = 129) or without (n = 122) a history of major depressive disorder (MDD) during the offspring’s life. Mothers and offspring completed assessments every 6 months for 2 years.
Results:
Compared to offspring of never-depressed mothers, offspring of mothers with a history of MDD recalled less specific memories in response to negative, but not positive, cue words at the initial assessment, and this bias was maintained across the two-year follow-up. For offspring of depressed, but not never-depressed, higher levels of maternal depressive symptoms at a given assessment predicted prospective decreases in the children’s autobiographical memory specificity. Again, this finding was specific to negative, but not positive, cue words.
Conclusion:
These results suggest that maternal depression has both short- and long-term effects on the development of offspring OGM to negative cues, which may represent a malleable cognitive vulnerability for the intergenerational transmission of MDD that could be targeted for intervention.
Keywords: overgeneral autobiographical memory (OGM), intergenerational transmission of depression, major depressive disorder (MDD), cognitive vulnerability
About 10–15 million offspring under the age of 18 in the United States reside with a depressed parent (England & Sim, 2009). Research demonstrates that a maternal history of major depressive disorder (MDD) is strongly linked with a broad range of adverse outcomes among offspring, including a three-to-four fold increase in risk of developing MDD by early adulthood (for a review, see Goodman, 2007). Overall, maternal history of MDD constitutes one of the strongest risk factors for internalizing disorders in offspring (Goodman et al., 2011). Despite robust evidence of heightened risk for depression in offspring of depressed mothers (Goodman, 2007; Goodman et al., 2011), specific mechanisms of intergenerational risk remain poorly understood.
Theorists have proposed that biases in information processing and retrieval of emotional stimuli (via attention, interpretation, and memory), collectively referred to as cognitive vulnerabilities, represent promising putative mechanisms of risk in offspring of depressed mothers (Goodman & Gotlib, 1999; Gotlib, Joormann, & Foland-Ross, 2014). Indeed, evidence demonstrates that, compared to offspring of never-depressed mothers, offspring of depressed mothers exhibit higher levels of cognitive vulnerabilities (for a review, see Gotlib & Colich, 2014). However, this research has focused primarily on attention and interpretation biases, and much less research has examined the impact of maternal depression on memory biases. Future research examining memory biases in the intergenerational transmission of depression is warranted given that memory biases are implicated in the development and maintenance of depression (Hitchcock, Nixon, & Weber, 2014) and represent a promising treatment target for intervention and prevention efforts (Dalgleish & Werner-Seidler, 2014).
One key memory bias commonly observed among depressed, compared to non-depressed, individuals is overgeneral autobiographical memory (OGM; Williams et al., 2007), which represents a tendency to recall less specific memories in response to emotional cue words (i.e., recalling memories of extended periods or repeated events, as opposed to events that occurred at a specific time and place). Most of the research to date has examined OGM in the context of adult and adolescent major depressive disorder (MDD). Evidence in these samples suggests that OGM represents a trait-like marker of MDD (e.g., Champagne et al., 2016; Mackinger, Pachinger, Leibetseder, & Fartacek, 2000; Nandrino, Pezard, Poste, Reveillere, & Beaune, 2002; Spinhoven et al., 2006) and predicts increases in depressive symptoms over time (e.g., Sumner, Griffith, & Mineka, 2010; Van Daele, Griffith, Van den Bergh, & Hermans, 2014). Significantly fewer studies have examined OGM in children, despite the importance of early identification of such vulnerabilities for the prevention of future MDD. That said, similar to adult and adolescent samples, evidence suggests that OGM is associated with depression in children (for a review, see Hitchcock et al., 2014). However, unlike research in adult and adolescent samples, which suggests a unidimensional structure for overgeneral autobiographical memory (e.g., Griffith et al., 2012), there is some evidence that autobiographical memory specificity for positive versus negative cues may be distinct in children (Drummond, Dritschel, Astell, O’Carroll, & Dalgleish, 2006; Feurer et al., 2018; Hipwell, Sapotichne, Klostermann, Battista, & Keenan, 2011; Hitchcock et al., 2014; Williams et al., 2007; but see also Nuttall, Valentino, Comas, McNeill, & Stey, 2014; Valentino, Toth, & Cicchetti, 2009). Theorists and researchers have suggested that OGM may first develop in response to negative cues and then later generalize to positive cues, (Kuyken & Dalgleish, 2011; Rawal & Rice, 2012; Woody, Burkhouse, & Gibb, 2015), although there is evidence that OGM to positive cues may also play a unique role in the development of childhood depression (e.g., Hipwell et al., 2011). Together, these studies suggest that a two-dimensional approach examining the unique effects of OGM memory to negative versus positive cues is warranted in the study of risk for childhood depression.
Etiological models of OGM in children suggest that factors across cultural, contextual, familial, and individual levels influence the development of OGM, with factors at the most proximal levels (i.e., individual and familial) exerting the greatest influence (Valentino, 2011). In particular, there is strong evidence that mother-child interactions can shape the development of offspring OGM. For example, both longitudinal and experimental studies have shown that more elaborative maternal reminiscing about past events predicts more detailed memories in offspring (for a review, see Fivush, 2011). Further, several studies have suggested a causal pathway between maternal and offspring OGM through maternal reminiscing (Jobson, Burford, Burns, Baldry, & Wu, 2018; Valentino et al., 2014). Together, these studies provide evidence that mothers exert considerable influence on the development of OGM in their offspring. However, the impact of other factors, such as maternal depression, has been largely understudied in the etiology of OGM.
In regard to the intergenerational transmission of depression risk, research shows that OGM memory to negative cues may be a cognitive vulnerability that contributes to the development of depression in offspring of depressed parents (e.g., Rawal & Rice, 2012; Woody et al., 2015). For example, in a sample of 223 never-depressed offspring, offspring of mothers with a history of MDD, compared to offspring of never-depressed mothers, were more likely to exhibit OGM in response to negative, but not positive, cue words (Woody, Burkhouse, & Gibb, 2015). These findings were maintained even when statistically controlling for the influence of offspring’s current depressive symptoms, suggesting that OGM to negative cues may serve as a marker of depression risk among high-risk offspring before they develop depression themselves. Furthermore, Rawal & Rice (2012) found that, in a longitudinal study of 277 adolescents of depressed parents, offspring’s baseline levels of OGM to negative cues predicted depressive symptom increases and first-onset of depressive disorders across the 1-year follow-up. Critically, these findings were independent of the effects of offspring age, intelligence, and baseline depressive symptoms. Together, these studies suggest that OGM to negative cues may be a mechanism for the intergenerational transmission of depression. However, neither study measured OGM across multiple assessment points, and much less is known about how this bias changes over time in offspring of depressed parents and whether it exists as a malleable or stable risk factor. Thus, future research is needed to determine whether maternal depression predicts prospective changes in offspring OGM over time.
The current study was designed to address this important gap in the literature by prospectively examining the influence of maternal depression on the development of OGM in offspring of depressed mothers across a 2-year follow-up. First, we examined trajectories of OGM, assessed every 6 months over 2 years, in offspring of mothers with a history of MDD during the child’s life versus offspring of mothers with no lifetime history of depression. Our goal was to extend the cross-sectional findings previously reported in the same sample (i.e., Woody et al., 2015) showing that offspring of depressed, compared to never depressed, mothers exhibited OGM to negative, but not positive, cues. The key question addressed by the current study was whether these differences based on mothers’ MDD history were maintained across the 2-year follow-up. Second, we conducted a novel examination of the impact of mothers’ current depressive symptoms on prospective changes in offspring OGM between each assessment point. Given that day-to-day interactions with mothers influence offspring OGM (e.g., Jobson et al., 2018; Valentino et al., 2014), we predicted that higher levels of maternal depressive symptoms at a given assessment would predict prospective decreases in the number of specific memories to negative, but not positive, cues among offspring over the next 6 months. In addition, because historical risk factors such as maternal MDD (Rawal & Rice, 2012; Woody et al., 2015) uniquely influence OGM biases compared to contextual risk factors (Drummond et al., 2006; Hipwell et al., 2011), we conducted exploratory analyses to determine whether the effect of maternal depressive symptoms would be stronger among offspring of mothers with a history of MDD. Specifically, we hypothesized that increased maternal depressive symptoms (a contextual risk factor) would enhance OGM biases to negative cues, but only among offspring with a historical risk factor (i.e., history of maternal MDD). Finally, given the association between childhood depression and OGM biases (for a review, see Hitchcock et al., 2014), we conducted exploratory analyses to determine if the impact of maternal depression on offspring OGM would be at least partially independent from the impact of offspring depression and to determine whether offspring OGM would be impacted by offspring depressive symptoms at each time point.
Method
Participants
Participants in this study were 251 mothers and their offspring. Potential participants were recruited from the community through a variety of means (e.g., newspaper and bus ads, flyers). Mothers responding to the recruitment advertisements were initially screened over the phone to determine potential eligibility. To qualify for the study, mothers were required to either meet criteria for MDD during the offspring’s lifetime according to the Diagnostic and Statistical Manual of Mental Disorders – Fourth Edition (DSM-IV; American Psychiatric Association, 2000) (n = 129) or have no lifetime diagnosis of any DSM-IV mood disorder and no current Axis I diagnosis (n = 122). Exclusion criteria for both groups included symptoms of schizophrenia, organic mental disorder, alcohol or substance dependence within the last six months, or history of bipolar disorder. Offspring were required to be the biological child of the participating mother, be between 8 and 14 years old at the initial assessment, and not have a learning or developmental disability per mothers’ report. Only one child per family was included. If there was more than one eligible offspring, one was chosen at random for participation. The offspring age range of 8–14 years was selected to examine trajectories of OGM during a window of high risk for the development of cognitive vulnerabilities (Hankin et al., 2009). Demographic variables split by group (maternal depression: yes, no) are presented in Table 1. The only significant demographic differences between the groups were that offspring of never depressed mothers were more likely to be Caucasian and to have higher family incomes compared to offspring of depressed mothers (ps < .05).
Table 1.
Descriptives for Study Variables.
| Never Depressed Mothers (n = 122) |
Depressed Mothers (n = 129) |
|
|---|---|---|
| Offspring Sex (% girls) | 54% | 49% |
| Offspring Race (% Caucasian) | 91% | 72% |
| Offspring Ethnicity (% Hispanic) | 0% | 2% |
| Offspring Age (T1) | 10.92 (1.85) | 10.87 (2.01) |
| Family Income | $60,001-65,000 | $35,001-40,000 |
| Offspring AMT Specific Negative (T1) | 3.88 (1.15) | 3.47 (1.29) |
| Offspring AMT Specific Negative (T2) | 3.72 (1.17) | 3.48 (1.17) |
| Offspring AMT Specific Negative (T3) | 3.77 (0.98) | 3.46 (1.34) |
| Offspring AMT Specific Negative (T4) | 3.90 (1.05) | 3.63 (1.14) |
| Offspring AMT Specific Negative (T5) | 4.06 (0.95) | 3.83 (1.01) |
| Offspring AMT Specific Positive (T1) | 3.60 (1.25) | 3.54 (1.28) |
| Offspring AMT Specific Positive (T2) | 3.44 (1.04) | 3.33 (1.26) |
| Offspring AMT Specific Positive (T3) | 3.63 (1.12) | 3.47 (1.24) |
| Offspring AMT Specific Positive (T4) | 3.58 (1.06) | 3.45 (1.10) |
| Offspring AMT Specific Positive (T5) | 4.03 (0.92) | 3.75 (0.94) |
| Offspring CDI (T1) | 4.82 (5.22) | 7.48 (5.89) |
| Offspring CDI (T2) | 3.61 (4.12) | 6.81 (6.66) |
| Offspring CDI (T3) | 3.68 (4.15) | 5.93 (5.11) |
| Offspring CDI (T4) | 2.86 (3.61) | 5.22 (5.03) |
| Offspring CDI (T5) | 3.57 (4.76) | 5.92 (5.34) |
| Maternal BDI-II (T1) | 3.68 (4.97) | 14.59 (10.96) |
| Maternal BDI-II (T2) | 3.25 (3.62) | 12.32 (9.22) |
| Maternal BDI-II (T3) | 3.37 (3.94) | 11.61 (8.57) |
| Maternal BDI-II (T4) | 3.58 (4.78) | 10.04 (8.00) |
| Maternal BDI-II (T5) | 2.75 (3.75) | 11.33 (9.09) |
Note. AMT Specific Negative = number of specific autobiographical memories recalled in response to negative cue words (out of 5); AMT Specific Positive = number of specific autobiographical memories recalled in response to positive cue words (out of 5). CDI = Children’s Depression Inventory; BDI-II = Beck Depression Inventory II. To facilitate comparisons with other studies, untransformed T1-5 AMT, CDI, and BDI-II scores are presented.
Measures
The Structured Clinical Interview for DSM-IV Axis I Disorders (SCID-I; First, Spitzer, Gibbon, & Williams, 1994) was used to assess for mothers’ histories of psychiatric disorders. Within the sample, 129 mothers met for a history of MDD during their offspring’s life, 69 of whom had a history of recurrent MDD and 21 of whom met criteria for current MDD at the initial assessment. To assess inter-rater reliability, a subset of 20 SCID interviews from this project were coded by a second interviewer (all κ’s = 1.00).
Mother’s symptoms of depression were assessed using the Beck Depression Inventory II (BDI-II; Beck, Steer, & Brown, 1996). The BDI-II is a 21-item self-report questionnaire that has demonstrated satisfactory reliability and validity in previous research (Dozois, Dobson, & Ahnberg, 1998). The BDI-II was administered at each of the assessment points (α’s = .92-.94 across all time points).
Offspring’s histories of MDD and other Axis-I disorders were assessed at the initial time point using the Schedule for Affective Disorders and Schizophrenia for School Age Children–Present and Lifetime Version (K-SADS-PL; Kaufman et al., 1997). Eighteen offspring had a lifetime history of MDD at the initial assessment (16 of whom had mothers with a history of MDD). To assess inter-rater reliability, a subset of 20 K-SADS-PL interviews from this project were coded by a second interviewer (all κ’s = 1.00).
Offspring’s symptoms of depression were assessed using the Children’s Depression Inventory (CDI; Kovacs, 1981). The CDI is a 27-item self-report instrument that has demonstrated satisfactory reliability and validity in previous research (Smucker, Craighead, Craighead, & Green, 1986). The CDI was administered at each of the assessment points (α’s = .85-.89 across all time points).
Offspring’s autobiographical memory was assessed using the Autobiographical Memory Test (AMT; Williams & Broadbent, 1986). The AMT task consists of 10 emotional words, 5 positively valenced (happy, surprised, safe, successful, interested) and 5 negatively valenced (sad, lonely, hurt, careless, angry). Words were presented to participants in a pre-determined order on index cards, alternating between positive and negative words, and participants were asked to retrieve a specific memory for each cue word. The order of presented words remained the same across time points. To ensure that participants understood the task, all participants completed three practice trials involving neutral words with feedback. Participants were given 60 seconds to retrieve a memory and were instructed to not give the same memory for more than one cue word. All responses were audiotaped, transcribed, and then coded as specific or overgeneral (categoric or extended) following the coding scheme developed by Williams and colleagues (Williams et al., 2007; Williams & Broadbent, 1986). A specific memory is defined as a single event lasting less than one day whereas a categoric memory refers to a generic collection or class of events and extended memory is of a single event lasting more than one day. If participants provided a response that included semantic information but no personal memory, this was coded as a semantic associate. Finally, if participants were unable to retrieve a memory or if they provided the same memory for more than one cue, this was coded as no response. The AMT was administered at each of the assessment points and summary scores were calculated for each participant at each time point, reflecting the number of specific memories provided for each cue valence. All responses were coded by two independent raters (κ = .81). Any discrepancies across raters were discussed until consensus was achieved.
Of note, previous findings from the same sample have shown that a two-factor model provided a significantly better fit to our OGM data than a one-factor model (Feurer et al., 2018), which supports the analysis of OGM to negative versus positive cues separately. In addition, consistent with prior research from our group (Feurer et al., 2018; Woody et al., 2015), greater OGM biases were quantified by the number of specific memories recalled, such that fewer specific memories constituted greater overgeneral memory bias.
Procedure
Upon arrival at the laboratory for the baseline assessment, mothers were asked to provide informed consent and offspring were asked to provide assent to be in the study. Next, offspring were administered the K-SADS-PL and the AMT by a research assistant and then completed the CDI. Following this, the mother completed the BDI-II and was administered the SCID-I and the K-SADS-PL. Follow-up assessments occurred 6, 12, 18, and 24 months after the initial assessment, during which offspring completed the AMT and the CDI and mothers completed the BDI-II. Families were compensated a total of $275 for their participation. The project was approved by Binghamton University (SUNY) Institutional Review Board.
Results
Descriptive statistics for study variables are presented in Table 1. Of the mother-offspring pairs participating in the initial assessment, 210, 197, 170, and 174 participated in the 6, 12, 18, and 24 month follow-ups, respectively. A preliminary inspection of the data revealed significant skew for several variables (z> 3.29; cf. Tabachnick & Fidell, 2007). These variables were transformed prior to further analysis to satisfy assumptions of normality (square root: T1-T5 BDI-II, CDI; log10: T1-T5 AMT). Next, examining the pattern of missing data due to participant attrition, we found that Little’s missing completely at random (MCAR) test was nonsignificant, χ2(2583)= 2644.23, p = .20, suggesting data were missing at random. Given this, maximum likelihood estimates of missing data were created and used in all subsequent analyses (see Schafer & Graham, 2002).1
Impact of maternal MDD history on trajectories of offspring OGM across the 2-year follow-up
Hierarchical linear modeling (HLM; Raudenbush, Bryk, Cheong, & Congdon, 2004) was used to examine whether maternal MDD history (yes, no) was associated with offspring’s memories for positive and negative cues across the follow-up period. We conducted separate models for positive and negative specific memories. The Level 1 model for these HLM analyses was:
where OGMtij represents the number of specific autobiographical memories (positive or negative) generated by each offspring at time t for assessment i and participant j, π0j is the intercept (reflecting each offspring’s OGM score at the initial assessment point), π1j is the slope of the linear relation between Time (in weeks) and OGM scores across the follow up for participant j, and eij represents the error term.
The Level 2 model was:
where β01 is the cross-level interaction term representing the effect of maternal MDD history on the OGM intercept and β11 is the cross-level interaction term representing the effect of maternal MDD history on linear change in offspring’s OGM across the follow-up. Finally, β00 and β10 are the intercept terms for each of their respective equations, and r0j and r1j are the error terms.
Focusing first on offspring’s OGM for positive cues, we found that maternal MDD history was not significantly related to OGM at the baseline assessment, t(249) = −0.13, p = .71, reffect size = .01. Although there was an overall increase in the number of specific memories for positive cues across the follow-up (significant slope for Time), t(249) = 3.90, p < .001, reffect size = .24, maternal history of MDD did not moderate this trajectory, t(249) = −1.30, p = .20, reffect size = .08. These findings show that a history of maternal MDD was not associated with offspring’s OGM to positive cues at the initial assessment nor did it predict trajectories of OGM to positive cues across the follow-up assessments.
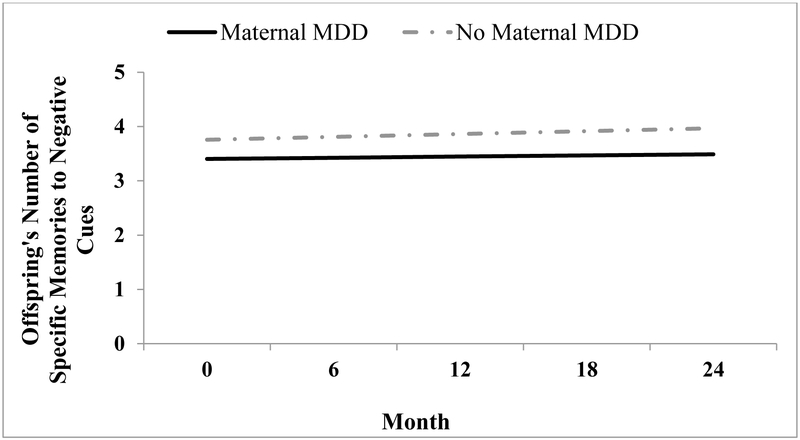
Turning next to OGM for negative cues, we found that maternal MDD history was significantly related to OGM at the baseline assessment, t(249) = −2.77, p = .01, reffect size = .17, indicating that offspring of mothers with a history of maternal MDD, compared to offspring of never-depressed mothers, reported fewer specific memories for negative cues at the initial assessment. Although there was a trending overall increase in the number of specific memories for negative cues across the follow-up, t(249) = 1.92, p = .06, reffect size = .12, maternal history of MDD did not moderate the time slope, t(249) = 0.82, p = .42, reffect size = .05, indicating that the initial difference in OGM to negative cues was maintained over the course of the follow-up. These findings are visually depicted in Figure 1.
Figure 1.
Trajectories of memory specificity to negative cues among offspring of mothers with and without a history of MDD over a two-year follow-up.
Tests of robustness showed that the influence of maternal MDD on OGM to negative cues at baseline remained significant even after accounting for offspring CDI scores, family income, and offspring race (Caucasian: yes, no) by including these factors as covariates (p = .03), suggesting that the influence of maternal MDD on offspring OGM is at least partially independent of offspring depressive symptoms, socioeconomic status, and race. Finally, exploratory analyses were conducted to examine whether any of the effects of maternal MDD on OGM to negative or positive cues were moderated by offspring’s age or sex. None of these analyses were significant (lowest p = .17).
Impact of maternal depressive symptoms on prospective changes in offspring OGM between assessments
A second series of HLM analyses was used to examine whether elevated levels of maternal depressive symptoms at each assessment point predicted prospective changes in the number of offspring’s specific negative and positive memories between each 6-month follow-up and whether the impact of maternal depressive symptoms was moderated by mothers’ history of MDD. The Level 1 model for these HLM analyses was:
where OGMtij represents the number of specific autobiographical memories (positive or negative) generated by each offspring at time t for assessment i and participant j, OGMt-1ij represents the number of specific memories (positive or negative) generated by each offspring at time t-1 for assessment i and participant j, and BDI-IIt-1ij represents the mother’s level of depressive symptoms at time t-1 for assessment i and participant j. In addition, π0j is the OGMtij intercept, π1j is the slope of the relation between the number of offspring specific memories between time t and time t-1 at each assessment i for participant j (i.e., the autocorrelation), π2j is the slope of the relation between maternal depressive symptoms and number of specific memories at time t, and eij represents the error term.
The Level 2 model was:
where β01 is the cross-level interaction term representing the effect of maternal MDD history (absent versus present in the offspring’s life) on the OGM intercept, β11 is the cross-level interaction term representing the effect of maternal MDD history on the slope of the relation between offspring’s lagged and current number of specific memories, and β21 is the cross-level interaction representing the effect of maternal MDD on the slope of the relation between maternal depressive symptoms and prospective changes in the number of specific memories from time t-1 to time t. Finally, β00, β10, and β20 are the intercept terms for each of their respective equations, and r0j, r1j, and r2j, are the error terms.
Focusing first on OGM for positive cues, we found that maternal depressive symptoms did not predict prospective changes in the number of offspring’s specific memories for positive cues during the follow-up, t(249) = 1.46, p = .15, reffect size = .09, nor was the mother MDD × maternal depressive symptoms interaction significant, t(249) = −1.60, p = .11, reffect size = .10, suggesting that mothers’ depressive symptoms during the follow-up did not predict prospective change in offspring OGM to positive cues, either for the full sample or for either MDD group.
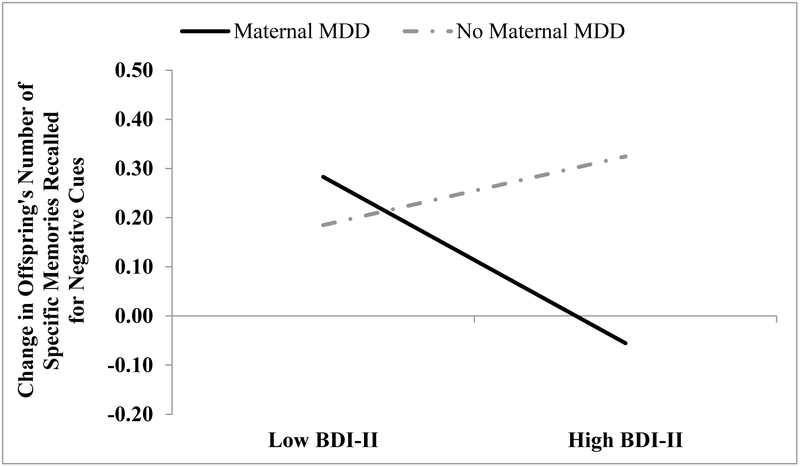
Turning next to OGM for negative cues, although we did not find a significant main effect of maternal depressive symptoms on prospective changes in offspring’s OGM to negative cues during the follow-up, t(249) = 0.92, p = .36, reffect size = .06, we did find a significant mother MDD history × maternal depressive symptoms interaction predicting change in OGM, t(249) = −2.44, p = .02, reffect size = .15. To explore the form of this interaction, we examined the impact of mothers’ depressive symptoms on changes in OGM separately for offspring of mothers with and without a history of MDD. Among offspring of mothers with a history of MDD, higher levels of maternal depressive symptoms at a given time point predicted prospective decreases in offspring’s number of specific memories to negative cues, t(128) = −2.84, p = .01, reffect size = .24. In contrast, maternal depressive symptoms did not predict prospective changes in number of offspring’s specific memories for negative cues among offspring of never-depressed mothers, t(121) = 0.99, p = .33, reffect size = .09. These findings are visually depicted in Figure 2.
Figure 2.
Maternal history of major depressive disorder (MDD) moderates the impact of maternal depressive symptoms on prospective changes in the number of specific memories recalled for negative cues generated by offspring
Note. The y-axis depicts a change score between assessments in the number of specific memories recalled for negative cues (as estimated by the HLM equation). Thus, a positive score indicates an improvement in memory specificity whereas a negative score indicates a decline.
Tests of robustness showed that, among offspring of mothers with a history of MDD, the effect of maternal depressive symptoms on offspring’s number of specific memories to negative cues remained significant even after statistically controlling for the influence of offspring CDI scores, family income, and offspring race (p = .01), suggesting that the findings are at least partially independent of offspring depressive symptoms, socioeconomic status, and race. Finally, exploratory analyses were conducted to examine whether any of the significant main effects of maternal depressive symptoms or interactions between maternal MDD and depressive symptoms were moderated by offspring’s age or sex. None of these analyses were significant (lowest p = .82).
Impact of offspring depressive symptoms on prospective changes in offspring OGM between assessments
An exploratory series of HLM analyses were used to examine if elevated levels of offspring depressive symptoms at each assessment point predicted prospective changes in the number of offspring’s specific negative and positive memories between each 6-month follow-up above and beyond the impact of maternal depressive symptoms and whether the impact of offspring depressive symptoms was moderated by mothers’ history of MDD. The Level 1 model for these HLM analyses was:
where OGMtij represents the number of specific autobiographical memories (positive or negative) reported by the offspring at time t for assessment i and participant j, OGMt-1ij represents the number of specific memories (positive or negative) reported by the offspring at time t-1 for assessment i and participant j, BDI-IIt-1ij represents the mother’s level of depressive symptoms at time t-1 for assessment i and participant j, and CDIt-1ij represents the offspring’s level of depressive symptoms at time t-1 for assessment i and participant j. In addition, π0j is the OGMtij intercept, π1j is the slope of the relation between the number of offspring’s specific memories between time t and time t-1 at each assessment i for participant j (i.e., the autocorrelation), π2j is the slope of the relation between maternal depressive symptoms and number of specific memories at time t, π3j is the slope of the relation between offspring depressive symptoms and number of specific memories at time t, and eij represents the error term.
The Level 2 model was:
where β01 is the cross-level interaction term representing the effect of maternal MDD history (absent versus present in offspring’s life) on the OGM intercept, β11 is the cross-level interaction term representing the effect of maternal MDD history on the slope of the relation between offspring’s lagged and current number of specific memories, β21 is the cross-level interaction representing the effect of maternal MDD on the slope of the relation between maternal depressive symptoms and prospective changes in the number of specific memories from time t-1 to time t, and β31 is the cross-level interaction representing the effect of maternal MDD on the slope of the relation between offspring depressive symptoms and prospective changes in the number of specific memories from time t-1 to time t. Finally, β00, β10, β20, and β30 are the intercept terms for each of their respective equations, and r0j, r1j, r2j, and r3j, are the error terms.
Focusing first on OGM for positive cues, we found that there was a significant main effect of offspring depressive symptoms on prospective changes in offspring’s OGM to positive cues during the follow-up, t(249) = −4.12, p < .001, reffect size = .25, which was moderated by mother MDD history, t(249) = −2.74, p = .01, reffect size = .17. To explore the form of this interaction, we examined the impact of offspring depressive symptoms on changes in OGM separately for offspring of mothers with and without a history of MDD. Among offspring of mothers with a history of MDD, offspring depressive symptoms did not predict prospective changes in number of specific memories for positive cues, t(128) = 0.26, p = .79, reffect size = .02. In contrast, among offspring of never depressed mothers, higher levels of offspring depressive symptoms at a given time point predicted prospective decreases in number of specific memories for positive cues, t(121) = −4.08, p < .001, reffect size = .35. Tests of robustness showed that these findings remained significant even after accounting for family income and offspring race (p = .04).
For OGM for negative cues, there was a significant main effect of offspring depressive symptoms on prospective changes in offspring’s OGM to negative cues during the follow-up such that higher offspring depressive symptoms at a given time point predicted prospective decreases in offspring’s number of specific memories to negative cues, t(249) = −2.66, p = .01, reffect size = .17, an effect that was not moderated by mother MDD history, t(249) = 0.98, p = .33, reffect size = .06. However, this effect did not survive tests of robustness that included family income and offspring race as covariates (p = .09).
Discussion
The primary goal of the current study was to examine the impact of maternal depression on the development of offspring OGM. We found that offspring of mothers with a history of MDD reported less-specific memories to negative, but not positive, cue words than offspring of never-depressed mothers, a difference that was maintained across a two-year follow-up. Critically, this study provided an important extension of our previous research (Woody et al., 2015) by showing that the influence of maternal MDD on OGM biases is maintained over time, providing preliminary evidence for the hypothesis that overgeneral memory for negative cue words represents a trait-like vulnerability in offspring of mothers with a history of MDD. Focusing next on mothers’ depressive symptoms during the follow-up, we found that, among offspring of mothers with a history of MDD, elevated maternal depressive symptoms at a given time point predicted prospective decreases in memory specificity to negative, but not positive, cue words between each assessment period. Importantly, the current findings were at least partially independent of the effect of offspring depression, race, and socioeconomic status, suggesting that the influence of maternal depression on the development of offspring OGM is not merely a concomitant of these factors. Taken together, findings suggest that, in addition to the broad, group-level differences in OGM to negative cues, mothers’ ongoing symptoms of depression predict a continued decrease in offspring memory specificity for negative, but not positive, cues over time. In addition, our findings provide further support for a two-dimensional model of OGM among children (see also, Feurer et al., 2018; Hipwell et al., 2011; Kuyken & Dalgleish, 2011; Rawal & Rice, 2012; Woody et al., 2015) given that OGM biases among offspring of depressed mothers were specific to negative, but not positive, cues,.
It is important to note that our examination of maternal MDD history versus current maternal depressive symptoms provide complementary but distinct information. Whereas mother MDD is a historical factor that may indicate a population at risk, mothers’ current depressive symptoms represent a contextual and modifiable risk factor that can be targeted to reduce offspring’s risk in the future. Thus, our findings regarding the impact of maternal MDD history on OGM trajectories suggested that OGM to negative cues had developed among offspring of mothers with a history of MDD in children between the ages of 8–14 years of age and that this memory bias was maintained across a two-year follow-up. Importantly, we also found that elevations in mothers’ current depressive symptoms predicted further impairments in children’s OGM for negative cues among offspring of mothers with a history of MDD. Given evidence that maternal reminiscence style impacts the development of offspring autobiographical memory (Fivush, 2011; Jobson et al., 2018; Valentino et al., 2014) and that ongoing depressive symptoms increase OGM biases in adults (e.g., Mackinger et al., 2000; Spinhoven et al., 2006), it may be that mothers with a history of MDD who are experiencing higher levels of depressive symptoms are more likely to model overgeneral reminiscence styles about negative events, which then excerbates existing OGM biases among their offspring. Future research is needed to test this possibility.
The current findings suggest that the development of OGM to negative cues is sensitive to ongoing fluctuations in mothers’ depressive symptoms among mothers with a history of MDD. Taken together with prior findings showing that OGM to negative cues at baseline predicts increases in depressive symptoms and disorders in adolescents and adults (Rawal & Rice, 2012; Sumner et al., 2010), these findings suggest that OGM could serve as an important therapeutic target for offspring of depressed mothers. For example, there is a promising line of research showing that memory therapeutics, which are designed to target autobiographical memory biases, can ameliorate symptoms of internalizing disorders in adults and adolescents (for a review, see Dalgleish & Werner-Seidler, 2014). One such memory therapeutic, MEmory Specificity Training (MEST; Raes et al., 2009), is administered by a therapist once a week for 4–6 weeks by teaching participants to recall more specific memories in a group therapy setting. Notably, MEST has proven successful in increasing the retrieval of specific memories and decreasing depressive symptoms in depressed adolescents (Neshat-Doost et al., 2013). In addition, broader cognitive behavioral therapies (CBT), such as Mindfulness-Based Cognitive Therapy (Williams, Teasdale, Segal, & Soulsby, 2000) and CBT for depression (McBride, Segal, Kennedy, & Gemar, 2007), have shown efficacy in reducing OGM biases in adults. However, no research of which we are aware has examined the effectiveness of CBT or memory therapeutics such as MEST in reducing OGM biases and preventing depression among offspring of depressed mothers.
In addition, given our findings that the development of OGM biases is sensitive to ongoing fluctuations in maternal depressive symptoms among offspring of depressed mothers, a more global approach toward reducing OGM biases in offspring of depressed mothers may be to provide treatment for the mother’s depression. Prior work has demonstrated that other vulnerability factors for depression in offspring significantly decrease following treatment for maternal depression (i.e., psychopharmacology, CBT, or a combination of these approaches) (e.g., Cuijpers, Weitz, Karyotaki, Garber, & Andersson, 2015; Pilowsky et al., 2008). Thus, future research might examine if OGM biases are also ameliorated following treatment of maternal depression. Further, it is possible that interventions that target maternal reminiscence styles (e.g., Reese & Newcombe, 2007; Taumoepeau & Reese, 2013), could also serve to reduce OGM biases in offspring given evidence that reminiscing styles are altered by depression and are a causal mechanism in the intergenerational transmission of OGM biases from mother to offspring (Jobson et al., 2018; Valentino et al., 2014). Clearly, future research is necessary to further understand how intervention efforts might address OGM biases in offspring of depressed mothers and what role these programs could have in preventing the intergenerational transmission of MDD.
Finally, it should be noted that there were unique effects of maternal and offspring depressive symptoms on changes in offspring OGM. In contrast to what was observed for the impact of maternal depression (across diagnoses and symptoms), we found that, among offspring of never depressed mothers, higher levels of offspring depressive symptoms at a given time point predicted prospective decreases in the number of specific memories recalled for positive cues. These findings are consistent with prior research that suggests that childhood dysphoria is related to OGM biases for positive cues in community samples of young children (Drummond et al., 2006) and adolescents (Hipwell et al., 2011), which may be indicative of difficulty recalling past positive events or successes due to depressed affect.
The current study exhibited several strengths, including the large sample size and the use of a multi-wave prospective design to examine the impact of maternal depression on the subsequent development of OGM. However, there were also limitations that highlight areas for future research. First, the current study examined the development of OGM beginning in late childhood to early adolescence (i.e., ages 8–14), and offspring of depressed mothers already displayed a bias in OGM to negative cues at the initial assessment. Thus, future research will benefit from the inclusion of younger offspring to examine the developmental antecedents of OGM to negative cues among offspring of mothers with a history of MDD. Second, the study did not assess for broader measures of cognitive functioning, such as verbal fluency, working memory, or other memory biases. Future research should include such measures to ensure OGM is not confounded by other measures of offspring cognitive functioning. However, the specificity of the current findings to OGM for negative, but not positive cues, suggests that differences between offspring of depressed vs. never-depressed mothers are not due to general memory impairments. Third, the current study did not assess for paternal history of MDD and therefore cannot draw any conclusions regarding the effects of paternal depression on the development of offspring OGM. Previous research has demonstrated that both maternal and paternal histories of MDD are associated with increased internalizing problems in offspring (Connell & Goodman, 2002), suggesting that paternal MDD plays an important role in the intergenerational transmission of MDD as well. Future research should examine the effects of both maternal and paternal MDD on offspring OGM as this approach could highlight important avenues for prevention and intervention efforts. Similarly, the current study did not assess for maternal or paternal treatment histories, which also could inform future research efforts. Finally, a large majority of offspring in the current study were non-Hispanic Caucasians, and future research is needed to replicate and extend these findings in a more ethnoracially diverse sample.
In summary, the current results support the role of OGM to negative cues as a trait-like vulnerability for the intergenerational transmission of MDD. Additionally, the findings suggest that the development of OGM to negative cues is sensitive to mother’s ongoing depressive symptoms among offspring of depressed mothers and thus may represent a malleable intervention target for prevention efforts. If these findings are replicated and extended, this work has important implications for better understanding the intergenerational transmission of MDD and developing break-through experimental therapeutics to target OGM biases in offspring of depressed mothers to prevent future depression in these high-risk youth.
Supplementary Material
Acknowledgments
This project was supported by NICHD grant HD057066 and NIMH grant MH098060 awarded to B.E.G. M.L.W. is supported by the NIMH grant MH119225, A.T. is supported by NIMH Grant MH114319, C.F. is supported by NSF GRF grant No. DGE1144464, and K.L.B. is supported by NIMH Grant MH113793. We would like to thank Ashley Johnson, Lindsey Stone, Andrea Hanley, Anastacia Kudinova, Michael Van Wie, Devra Alper, Eric Funk, and Effua Sosoo for their help in conducting assessments for this project.
Footnotes
Supplementary analyses regarding the influence of maternal history of anxiety disorders and PTSD and non-linear effects on offspring OGM development can be found in the Supplement.
References
- American Psychiatric Association. (2000). Diagnostic and statistical manual of mental disorders (4th ed.). Washington, D.C. [Google Scholar]
- Beck A, Steer RA, & Brown GK (1996). Beck Depression Inventory-II. San Antonio, TX, 78204–2498. [Google Scholar]
- Champagne K, Burkhouse KL, Woody ML, Feurer C, Sosoo EE, & Gibb BE (2016). Brief report: Overgeneral autobiographical memory in adolescent major depressive disorder. Journal of Adolescence, 52, 72–75. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Connell AM, & Goodman SH (2002). The association between psychopathology in fathers versus mothers and children’s internalizing and externalizing behavior problems: A meta-analysis. Psychological Bulletin, 128, 746–773. [DOI] [PubMed] [Google Scholar]
- Cuijpers P, Weitz E, Karyotaki E, Garber J, & Andersson G (2015). The effects of psychological treatment of maternal depression on children and parental functioning: A meta-analysis. European Child & Adolescent Psychiatry, 24, 237–245. [DOI] [PubMed] [Google Scholar]
- Dalgleish T, & Werner-Seidler A (2014). Disruptions in autobiographical memory processing in depression and the emergence of memory therapeutics. Trends in Cognitive Sciences, 18, 596–604. [DOI] [PubMed] [Google Scholar]
- Dozois DJ, Dobson KS, & Ahnberg JL (1998). A psychometric evaluation of the Beck Depression Inventory-II. Psychological Assessment, 10, 83–89. [Google Scholar]
- Drummond LE, Dritschel B, Astell A, O’Carroll RE, & Dalgleish T (2006). Effects of age, dysphoria, and emotion-focusing on autobiographical memory specificity in children. Cognition and Emotion, 20, 488–505. [DOI] [PubMed] [Google Scholar]
- England MJE, & Sim LJ (2009). Depression in parents, parenting, and children: Opportunities to improve identification, treatment, and prevention. National Academies Press. [PubMed] [Google Scholar]
- Feurer C, Woody ML, Tsypes A, Burkhouse KL, Champagne K, & Gibb BE (2018). Episodic Life Stress and the Development of Overgeneral Autobiographical Memory to Positive Cues in Youth. Journal of Abnormal Child Psychology. [DOI] [PMC free article] [PubMed] [Google Scholar]
- First MB, Spitzer RL, Gibbon M, & Williams JB (1994). Structured clinical interview for Axis I DSM-IV disorders. Patient Edition (SCID-I/P). [Google Scholar]
- Fivush J (2011). The development of autobiographical memory. Annual Review of Psychology, 62, 559–582. [DOI] [PubMed] [Google Scholar]
- Goodman SH (2007). Depression in mothers. Annual Review of Clinical Psychology, 3, 107–135. [DOI] [PubMed] [Google Scholar]
- Goodman SH, & Gotlib IH (1999). Risk for psychopathology in the children of depressed mothers: A developmental model for understanding mechanisms of transmission. Psychological Review, 106, 458–490. [DOI] [PubMed] [Google Scholar]
- Goodman SH, Rouse MH, Connell AM, Broth MR, Hall CM, & Heyward D (2011). Maternal depression and child psychopathology: A meta-analytic review. Clinical Child and Family Psychology Review, 14, 1–27. [DOI] [PubMed] [Google Scholar]
- Gotlib IH, & Colich NL (2014). Children of Parents with Depression. In Gotlib IH & Hammen C (Eds.), Handbook of Depression (3rd ed., pp. 240–258). New York, NY: Guilford Press. [Google Scholar]
- Gotlib IH, Joormann J, & Foland-Ross LC (2014). Understanding familial risk for depression: A 25-Year perspective. Perspectives on Psychological Science, 9, 94–108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griffith JW, Sumner J. a., Raes F, Barnhofer T, Debeer E, & Hermans D (2012). Current psychometric and methodological issues in the measurement of overgeneral autobiographical memory. Journal of Behavior Therapy and Experimental Psychiatry, 43, 21–31. [DOI] [PubMed] [Google Scholar]
- Hankin BL, Oppenheimer C, Jenness J, Barrocas A, Shapero BG, & Goldband J (2009). Developmental origins of cognitive vulnerabilities to depression: Review of processes contributing to stability and change across time. Journal of Clinical Psychology, 65, 1327–1338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hipwell AE, Sapotichne B, Klostermann S, Battista D, & Keenan K (2011). Autobiographical memory as a predictor of depression vulnerability in girls. Journal of Clinical Child & Adolescent Psychology, 40, 254–265. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hitchcock C, Nixon RDV, & Weber N (2014). A review of overgeneral memory in child psychopathology. British Journal of Clinical Psychology, 53, 170–193. [DOI] [PubMed] [Google Scholar]
- Jobson L, Burford K, Burns B, Baldry A, & Wu Y (2018). Investigating whether maternal memory specificity is indirectly associated with child memory specificity through maternal reminiscing. Memory, 26, 1335–1343. [DOI] [PubMed] [Google Scholar]
- Kaufman J, Birmaher B, Brent D, Rao UMA, Flynn C, Moreci P, … Ryan ND (1997). Schedule for affective disorders and schizophrenia for school-age children - present and lifetime version (K-SADS-PL): Initial reliability and validity data. Journal of the American Academy of Child & Adolescent Psychiatry, 36, 980–988. [DOI] [PubMed] [Google Scholar]
- Kovacs M (1981). Rating scales to assess depression in school-aged children. Acta Paedopsychiatrica, 46, 305–315. [PubMed] [Google Scholar]
- Kuyken W, & Dalgleish T (2011). Overgeneral autobiographical memory in adolescents at risk for depression. Memory, 19, 241–250. [DOI] [PubMed] [Google Scholar]
- Mackinger HF, Pachinger MM, Leibetseder MM, & Fartacek RR (2000). Autobiographical memories in women remitted from major depression. Journal of Abnormal Psychology, 109, 331. [PubMed] [Google Scholar]
- McBride C, Segal ZV, Kennedy S, & Gemar M (2007). Changes in autobiographical memory specificity following cognitive behavior therapy and pharmacotherapy for major depression. Psychopathology, 40, 147–152. [DOI] [PubMed] [Google Scholar]
- Nandrino J-L, Pezard L, Poste A, Reveillere C, & Beaune D (2002). Autobiographical memory in major depression: A comparison between first-episode and recurrent patients. Psychopathology, 35, 335–340. [DOI] [PubMed] [Google Scholar]
- Neshat-Doost HT, Dalgleish T, Yule W, Kalantari M, Ahmadi SJ, Dyregrov A, & Jobson L (2013). Enhancing autobiographical memory specificity through cognitive training: An intervention for depression translated from basic science. Clinical Psychological Science, 1, 84–92. [Google Scholar]
- Nuttall AK, Valentino K, Comas M, McNeill AT, & Stey PC (2014). Autobiographical memory specificity among preschool-aged children. Developmental Psychology, 50, 1963–1972. [DOI] [PubMed] [Google Scholar]
- Pilowsky DJ, Wickramaratne P, Talati A, Tang M, Hughes CW, Garber J, … Weissmann M (2008). Children of depressed mothers 1 year after the initiation of maternal treatment: Findings from the STAR*D-Child study. The American Journal of Psychiatry, 165, 1136–1147. [DOI] [PubMed] [Google Scholar]
- Raes F, Hermans D, Williams JMG, & Eelen P (2005). Autobiographical memory specificity and emotional abuse. British Journal of Clinical Psychology, 44(1), 133–138. [DOI] [PubMed] [Google Scholar]
- Raes F, Williams JMG, & Hermans D (2009). Reducing cognitive vulnerability to depression: A preliminary investigation of MEmory Specificity Training (MEST) in inpatients with depressive symptomatology. Journal of Behavior Therapy and Experimental Psychiatry, 40, 24–38. [DOI] [PubMed] [Google Scholar]
- Raudenbush SW, & Bryk AS (2002). Hierarchical linear models: Applications and data analysis methods (2nd ed.). Thousand Oaks, CA: Sage. [Google Scholar]
- Raudenbush SW, Bryk AS, Cheong YF, & Congdon RT (2004). HLM 6: Hierarchical linear and nonlinear modeling. Incolnwood, IL: Scientific Software International. [Google Scholar]
- Rawal A, & Rice F (2012). Examining overgeneral autobiographical memory as a risk factor for adolescent depression. Journal of the American Academy of Child and Adolescent Psychiatry, 51, 518–527. [DOI] [PubMed] [Google Scholar]
- Reese E, & Newcombe R (2007). Training mothers in elaborative reminiscing enhances children’s autobiographical memory and narrative. Child Development, 78, 1153–1170. [DOI] [PubMed] [Google Scholar]
- Schafer JL, & Graham JW (2002). Missing data: Our view of the state of the art. Psychological Methods, 7, 147–177. [PubMed] [Google Scholar]
- Smucker MR, Craighead WE, Craighead LW, & Green BJ (1986). Normative and reliability data for the children’s depression inventory. Journal of Abnormal Child Psychology, 14, 25–39. [DOI] [PubMed] [Google Scholar]
- Spinhoven P, Bockting CLH, Schene AH, Koerter MWJ, Wekking EM, & Williams JMG (2006). Autobiographical memory in the euthymic phase of recurrent depression. Journal of Abnormal Psychology, 115, 590–600. [DOI] [PubMed] [Google Scholar]
- Sumner J. a., Griffith JW, & Mineka S (2010). Overgeneral autobiographical memory as a predictor of the course of depression: A meta-analysis. Behaviour Research and Therapy, 48, 614–625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tabachnick BG, & Fidell LS (2007). Using multivariate statistics (5th ed.). Needham Height, MA: Allyn & Bacon. [Google Scholar]
- Taumoepeau M, & Reese E (2013). Maternal reminiscing, elaborative talk, and children’s theory of mind: An intevention study. First Language, 33, 388–410. [Google Scholar]
- Valentino Kirsten. (2011). A developmental psychopathology model of overgeneral autobiographical memory. Developmental Review, 31, 32–54. [Google Scholar]
- Valentino Kristin, Nuttall AK, Comas M, McDonnell CG, Piper B, Thomas TE, & Fanuele S (2014). Mother-child reminiscing and autobiographical memory specificity among preschool-age children. Developmental Psychology, 50, 1197–1207. [DOI] [PubMed] [Google Scholar]
- Valentino Kristin, Toth SL, & Cicchetti D (2009). Autobiographical memory functioning among abused, neglected, and nonmaltreated children: the overgeneral memory effect. Journal of Child Psychology and Psychiatry, and Allied Disciplines, 50, 1029–1038. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Daele T, Griffith JW, Van den Bergh O, & Hermans D (2014). Overgeneral autobiographical memory predicts changes in depression in a community sample. Cognition & Emotion, 28, 1303–1312. [DOI] [PubMed] [Google Scholar]
- Williams J Mark G, Barnhofer T, Crane C, Hermans D, Raes F, Watkins ER, & Dalgleish T (2007). Autobiographical memory specificity and emotional disorder. Psychological Bulletin, 133, 122–148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams J Mark G, Teasdale JD, Segal ZV, & Soulsby J (2000). Mindfulness-based cognitive therapy reduces overgeneral autobiographical memory in formely depressed patients. Journal of Abnormal Psychology, 109, 150–155. [DOI] [PubMed] [Google Scholar]
- Williams J M G, & Broadbent K (1986). Autobiographical memory in suicide attempters. Journal of Abnormal Psychology, 95, 144–149. [DOI] [PubMed] [Google Scholar]
- Woody ML, Burkhouse KL, & Gibb BE (2015). Overgeneral autobiographical memory in children of depressed mothers. Cognition and Emotion, 29(1), 130–137. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.