Abstract
Background
Antibiotics are not indicated for treating acute bronchitis cases, yet up to 70% of adult acute bronchitis medical visits in the USA result in an antibiotic prescription. Reducing unnecessary antibiotic prescribing for acute bronchitis is a key antibiotic stewardship goal set forth by the Centers for Disease Control and Prevention. Understanding what factors influence prescribing for bronchitis cases can inform antimicrobial stewardship initiatives. The goal of this study was to identify factors associated with antibiotic prescribing at a high-volume student health center at a large US university. The Pennsylvania State University Health Services offers on-campus medical care to a population of over 40,000 students and receives over 50,000 visits every year.
Methods
We conducted a retrospective chart review of acute bronchitis visits for the 2015–2016 academic year and used a multivariate logistic regression analysis to identify variables associated with antibiotic prescribing.
Results
Findings during lung exams increased the likelihood of an antibiotic prescription (rales OR 13.95, 95% CI 3.31–80.73; rhonchi OR 5.50, 95% CI 3.08–10.00; percussion abnormality OR 13.02, 95% CI 4.00–50.09). Individual clinicians had dramatically different rates of prescribing (OR range 0.03–12.3). Male patients were more likely than female patients to be prescribed antibiotics (OR 1.68, 95% CI 1.17–2.41). Patients who reported longer duration since the onset of symptoms were slightly more likely to receive prescriptions (OR 1.04 per day, 95% CI 1.03–1.06), as were patients who reported worsening symptoms (OR 1.78, 95% CI 1.03–3.10). Visits with diagnoses or symptoms associated with viral infections or allergies were less likely to result in prescriptions (upper respiratory tract infection (URI) diagnosis OR 0.33, 95% CI 0.18–0.58; sneezing OR 0.39, 95% CI 0.17–0.86; vomiting OR 0.31, 95% CI 0.10–0.83). An exam finding of anterior cervical lymphadenopathy was associated with antibiotic prescribing (tender OR 3.85, 95% CI 1.70–8.83; general OR 2.63, 95% CI 1.25–5.54).
Conclusions
Suspicious findings during lung examinations (rales, rhonchi, percussion abnormality) and individual healthcare providers were important factors influencing antibiotic prescribing rates for acute bronchitis visits. Patient gender, worsening symptoms, duration of illness, symptoms associated with viral infections or allergies, and anterior cervical lymphadenopathy also influenced prescribing rates.
Keywords: Antibiotic stewardship, Antibiotic prescribing, Acute bronchitis, Respiratory tract infections, Student health
Background
In the United States, 30% of outpatient antibiotic prescribing is estimated to be unnecessary, resulting in almost 47 million unnecessary antibiotic prescriptions each year [1, 2]. Excessive antibiotic prescribing drives the spread of antibiotic resistance, which contributes to increased morbidity, mortality, and economic costs associated with infections [3–5]. In response, the 2015 U.S. National Action Plan for Combatting Antibiotic-Resistant Bacteria set a goal of reducing inappropriate antibiotic prescribing in outpatient settings by 50% by 2020 [6].
A major source of unnecessary outpatient antibiotic prescriptions is acute bronchitis cases [7–10]. Acute bronchitis is a common self-limited respiratory illness, characterized predominantly by cough, typically lasting less than 3 weeks [7, 11]. In the US in 2011, cough was the most common illness-related reason for ambulatory care visits, accounting for 2.6 million outpatient visits [12]. A study in the UK estimated that 44/1000 adults are affected by acute bronchitis each year [13]. Antibiotics are not effective for treating acute bronchitis, which is usually of viral etiology [11], and long-standing professional guidelines recommend against antibiotics for uncomplicated cases [14, 15]. Nevertheless, US adults are prescribed antibiotics for acute bronchitis approximately 60–70% of the time [7–9, 16]. Further, relative to other upper respiratory tract infections for which antibiotic treatment is not indicated (e.g., nasopharyngitis, laryngitis), providers are especially likely to prescribe for acute bronchitis [8, 17–20]. Due to the prevalence of overprescribing, the U.S. Centers for Infectious Disease Control (CDC) has identified acute bronchitis cases as a major opportunity for reducing unnecessary outpatient antibiotic prescribing [21].
Although acute bronchitis presents an opportunity to improve antibiotic stewardship, there is little consensus regarding effective stewardship interventions for ambulatory care [22–24]. A diversity of interventions have been proposed, but evidence supporting their effectiveness remains sparse [23, 24]. Implementation of outpatient stewardship programs could be aided by identifying the factors driving overprescribing, which might point to interventions that target those drivers [22]. Factors driving antibiotic overprescribing may differ between hospital and outpatient settings and could include diagnostic uncertainty, real or perceived patient expectations for antibiotics, time pressures, or gaps in provider knowledge [25, 26].
Identifying drivers of prescribing for acute bronchitis could suggest potential interventions, but relatively few studies have focused on identifying these predictors. Prior studies of upper respiratory tract infection prescribing (including for acute bronchitis) in the USA have shown higher rates of antibiotic prescribing in rural (vs. urban) practices [8, 10], when patients have multiple diagnoses [27] or illness of longer duration [28], when providers are advanced practitioners rather than physicians [9], and when providers experience greater diagnostic uncertainty [27]. Since most studies have utilized data reported to insurance companies or national agencies [13, 16, 23], few previous studies have examined how physical exam findings influence prescribing for acute bronchitis. In the few studies that have included data from patient charts, purulent nasal discharge, purulent sputum, abnormal respiratory exam, tonsillar exudate, and sinus tenderness have been reported to be moderately associated with prescribing [20, 29]. In addition, US prescribing rates for uncomplicated acute bronchitis are higher for younger adults (18–39) than older adults (40+) [16], suggesting that factors influencing bronchitis prescribing for young adults are particularly good targets for evaluation and intervention.
University student health clinics provide an opportunity to study antibiotic prescribing in young adult patient populations. In the US, college students comprise a sizeable cohort of the population, with 20.1 million students enrolled in higher education, including 13.8 million students enrolled at 4-year degree-granting institutions [30]. At these 4-year institutions, there are 165.5 annual visits to student health centers for every 100 enrolled students, 37% of which are for respiratory tract infections [31]. Despite evidence that unnecessary antibiotic prescribing is high in young adult populations [16], antibiotic stewardship programs are almost nonexistent at most student health centers, and best stewardship practices are not yet defined. Understanding what drives unnecessary antibiotic prescribing in student health centers is a first step towards evidence-based stewardship policies in these settings, and findings can also inform stewardship efforts with providers treating young adults in similar contexts (e.g., urgent care clinics).
The goal of this study was to identify patient and visit factors associated with antibiotic prescribing for young adults diagnosed with acute bronchitis at a high-volume student health center at a large US university. We conducted a retrospective chart review of all visits with an acute bronchitis diagnosis for the 2015–2016 academic year at the Pennsylvania State University’s Student Health Center. This work is part of a multi-study interdisciplinary effort to improve antibiotic stewardship in emerging adult populations, with an initial focus on students at residential colleges.
Methods
Study site
The Pennsylvania State University Health Services (UHS) offers on-campus medical care to PSU students and their dependents, serving over 40,000 students in more than 50,000 visits yearly. At the time of the study, 28 clinicians saw patients at UHS. During the study period, 21 of these clinicians (9 doctors of medicine (MDs), 2 doctors of osteopathic medicine (DOs), 8 physician assistants (PAs), and 2 nurse practitioners (NPs)) diagnosed at least one patient with acute bronchitis. The remaining clinicians did not diagnose acute bronchitis in the period studied, and therefore they do not appear in the data set.
Data collection and Curation
UHS staff identified 1451 visits with acute bronchitis diagnoses during the 2015–2016 academic year (August–May). Honest brokers were then employed and trained to access the electronic medical records for these visits, extract deidentified data (data excluding information that could be used to identify individual patients), and enter it in the secure database manager REDCap for use by the researchers. Data extracted included patient characteristics, visit characteristics, symptoms recorded, exam findings, secondary diagnoses, tests ordered, and antibiotic prescriptions (see Table 1). A double-entry procedure was used to provide a reliability check on data extracted from a randomly selected sample (N = 69; ~ 5%) of the visits. This check indicated adequate data quality (agreement > 96% across all variables) for the intended analyses; identified discrepancies were corrected [32–34].
Table 1.
Descriptive statistics (n = 1031) and bivariate analysis
| Variable | Visit Count (%) | Odds Ratio (95% CI) | Bivariate p-value |
|---|---|---|---|
| Date and Time | |||
| visit date | 1028 (99.7%) | 0.99 (0.99-0.99) | p < 0.001 ** |
| week day | 1028 (99.7%) | range 0.64-1.28 | p = 0.63 |
| time of day | 1021 (99.0%) | 0.99 (0.99-0.99) | p = 0.24 |
| Patient Characteristics | |||
| gender | p = 0.03* | ||
| female (reference group) | 636 (61.7%) | - | |
| male | 390 (37.8%) | 1.34 (1.02-1.76) | |
| not recorded | 5 (0.5%) | - | |
| race | p = 0.43 | ||
| white (reference group) | 594 (57.6%) | - | |
| multiple | 92 (8.9%) | 0.90 (0.55-1.44) | |
| Asian | 50 (4.8%) | 0.97 (0.51-1.79) | |
| black | 21 (2.0%) | 1.04 (0.38-2.54) | |
| Hispanic | 6 (0.6%) | - | |
| international | 7 (0.7%) | - | |
| Pacific islander | 1 (0.1%) | - | |
| not recorded | 260 (25.2%) | - | |
| academic status | p = 0.27 | ||
| undergraduate student | 932 (90.4%) | - | |
| graduate student | 85 (8.2%) | 0.86 (0.52-1.39) | |
| spouse/dependent | 3 (0.3%) | - | |
| not recorded | 11 (1.1%) | - | |
| height (inches) | 1013 (98.2%) | 1.04 (1.00-1.08) | p = 0.02* |
| weight (pounds) | 1016 (98.5%) | 1.00 (0.99-1.01) | p = 0.09 |
| Visit Characteristics | |||
| provider | see Fig 3 | range 0.05–4.63 | p < 0.001** |
| days since onset (patient reported) †† | 1016 (98.5%) | 1.02 (1.01-1.03) | p < 0.001** |
| severity (patient reported) | p = 0.009* | ||
| mild (reference group) | 61 (5.9%) | - | |
| moderate | 342 (33.2%) | 0.48 (0.27-0.86) | |
| severe | 32 (3.1%) | 1.13 (0.46-2.70) | |
| not recorded | 596 (57.8%) | - | |
| progression (patient reported) | p < 0.001** | ||
| stable/no change (reference group) | 274 (26.6%) | - | |
| worsening | 317 (30.7%) | 2.27 (1.77-3.77) | |
| improving | 108 (10.5%) | 1.06 (0.60-1.84) | |
| not recorded | 332 (32.2%) | - | |
| antibiotics in past month | 40 (3.9%) | 0.46 (0.19-1.00) | p = 0.07 |
| Additional Diagnosis | |||
| upper respiratory infection | 197 (19.1%) | 0.26 (0.16-0.39) | p < 0.001** |
| suspicious cough | 77 (7.5%) | 1.02 (0.61-1.67) | p = 0.93 |
| allergic rhinitis | 32 (3.1%) | 0.51 (0.19-1.17) | p = 0.14 |
| fever | 17 (1.6%) | 2.58 (0.98-6.92) | p = 0.05 |
| viral syndrome | 13 (1.3%) | - | - |
| tonsillitis | 6 (0.6%) | - | - |
| influenza | 4 (0.4%) | - | - |
| mononucleosis | 3 (0.3%) | - | - |
| Common Symptoms Recorded† | |||
| throat symptoms | |||
| sore throat | 402 (39.0%) | 1.42 (1.08-1.85) | p = 0.01* |
| painful swallowing | 130 (12.6%) | 0.58 (0.37-0.89) | p = 0.02* |
| hoarseness | 167 (16.2%) | 0.80 (0.55-1.56) | p = 0.25 |
| swollen glands in neck | 112 (10.9%) | 1.08 (0.70-1.63) | p = 0.73 |
| systemic symptoms | |||
| headache | 244 (23.7%) | 0.79 (0.57-1.09) | p = 0.15 |
| documented fever | 68 (6.6%) | 0.70 (0.42-1.17) | p = 0.17 |
| fever symptoms (patient reported) | 255 (24.7%) | 0.94 (0.69-1.28) | p = 0.71 |
| chills | 147 (14.2%) | 0.94 (0.65-1.37) | p = 0.73 |
| sweats | 159 (15.4%) | 1.19 (0.82-1.75) | p = 0.36 |
| nasal symptoms | |||
| stuffy nose | 665 (64.5%) | 0.86 (0.66-1.14) | p = 0.29 |
| sinus congestion | 344 (33.4%) | 0.73 (0.55-0.98) | p = 0.03* |
| clear nasal discharge | 215 (20.9%) | 0.71 (0.50-0.99) | p = 0.04* |
| purulent nasal discharge | 184 (17.8%) | 0.83 (0.58-1.19) | p = 0.33 |
| post-nasal drip sensation | 390 (37.8%) | 0.93 (0.70-1.21) | p = 0.59 |
| sinus pain | 79 (7.7%) | 1.69 (1.05-2.69) | p = 0.03* |
| sneezing | 101 (9.8%) | 0.39 (0.22-0.66) | p < 0.001** |
| pulmonary symptoms | |||
| sleep disruption due to cough | 610 (59.2%) | 1.22 (0.93-1.59) | p = 0.15 |
| sputum production | 638 (61.9%) | 0.98 (0.75-1.29) | p = 0.91 |
| shortness of breath | 348 (33.8%) | 0.74 (0.56-0.97) | p = 0.03* |
| chest tightness | 277 (26.8%) | 0.94 (0.69-1.26) | p = 0.67 |
| wheezing | 275 (26.7%) | 0.64 (0.48-0.86) | p = 0.003* |
| chest pain | 232 (22.5%) | 0.82 (0.60-1.12) | p = 0.22 |
| paroxysms of cough | 315 (30.6%) | 0.98 (0.73-1.30) | p = 0.87 |
| ear symptoms | |||
| ear pain | 48 (4.7%) | 1.02 (0.53-1.88) | p = 0.94 |
| ear pressure sensation | 122 (11.8%) | 0.89 (0.58-1.35) | p = 0.60 |
| decreased hearing | 34 (3.3%) | 0.81 (0.35-1.69) | p = 0.58 |
| GI symptoms | |||
| loss of appetite | 129 (12.5%) | 0.82 (0.54-1.23) | p = 0.34 |
| abdominal pain | 25 (2.4%) | 0.30 (0.07-0.87) | p = 0.05 |
| post-tussive vomiting | 74 (7.2%) | 0.67 (0.41-1.09) | p = 0.10 |
| nausea | 63 (6.1%) | 0.46 (0.22-0.86) | p = 0.02* |
| vomiting | 48 (4.6%) | 0.44 (0.19-0.89) | p = 0.03* |
| diarrhea | 34 (3.3%) | 1.24 (0.59-2.49) | p = 0.56 |
| neuro-vascular symptoms | |||
| lightheadedness | 47 (4.6%) | 1.29 (0.69-2.35) | p = 0.41 |
| Commonly Ordered Labs | |||
| chest x-ray | 177 (17.2%) | 2.09 (1.50-2.90) | p < 0.001** |
| rapid strep screen | 31 (3.0%) | 0.65 (0.26-1.45) | p = 0.32 |
| complete blood count | 85 (8.2%) | 1.40 (0.88-2.21) | p = 0.15 |
| Monospot | 27 (2.6%) | 1.83 (0.83-3.95) | p = 0.12 |
| influenza A + B | 16 (1.6%) | 0.32 (0.05-1.14) | p = 0.13 |
| Common Exam Findings† | |||
| ear exam | |||
| tympanic membrane (TM) | 27 (2.6%) | 0.08 (0.004-0.40) | p = 0.01* |
| bulging | |||
| TM retraction | 42 (4.1%) | 0.79 (0.38-1.55) | p = 0.51 |
| visible fluid behind TM | 148 (14.3%) | 0.17 (0.09-0.29) | p < 0.001** |
| cerumen in canal | 29 (2.8%) | 0.46 (0.15-1.12) | p = 0.12 |
| nose exam | |||
| mucosal edema | 580 (56.2%) | 0.83 (0.63-1.08) | p = 0.16 |
| mucosal erythema | 510 (49.5%) | 0.78 (0.59-1.01) | p = 0.06 |
| nasal discharge | 324 (31.4%) | 1.59 (1.20-2.09) | p = 0.001** |
| maxillary sinus tenderness | 30 (2.9%) | 1.75 (0.83-3.64) | p = 0.13 |
| throat exam | |||
| erythema | 209 (20.3%) | 0.79 (0.56-1.10) | p = 0.17 |
| lymphoid hyperplasia | 104 (10.1%) | 1.47 (0.96-2.22) | p = 0.07 |
| post-nasal drip | 157 (15.2%) | 1.44 (1.00-2.04) | p = 0.04* |
| tonsil exam | |||
| surgically absent | 42 (4.1%) | 1.13 (0.57-2.14) | p = 0.71 |
| erythema | 84 (8.1%) | 0.43 (0.23-0.74) | p = 0.004** |
| enlarged | 47 (4.5%) | 1.17 (0.62-2.32) | p = 0.64 |
| lymphatics exam | |||
| anterior cervical lymphadenopathy, tender | 53 (5.1%) | 1.93 (1.10-3.38) | p = 0.02* |
| anterior cervical lymphadenopathy, non-tender | 87 (8.4%) | 0.70 (0.41-1.14) | p = 0.16 |
| posterior cervical lymphadenopathy, non-tender | 51 (4.9%) | 0.60 (0.29-1.16) | p = 0.15 |
| anterior cervical lymphadenopathy | 99 (9.6%) | 3.05 (2.01-4.66) | p < 0.001** |
| posterior cervical lymphadenopathy | 26 (2.5%) | 1.00 (0.41-2.25) | p = 0.10 |
| lung exam | |||
| wheezing | 215 (20.9%) | 1.92 (1.40-2.61) | p < 0.001** |
| rales | 21 (2.0%) | 10.05 (3.69-35.18) | p < 0.001** |
| rhonchi | 223 (21.6%) | 2.33 (1.71-3.16) | p < 0.001** |
| percussion abnormality | 25 (2.4%) | 9.55 (3.83-28.91) | p < 0.001** |
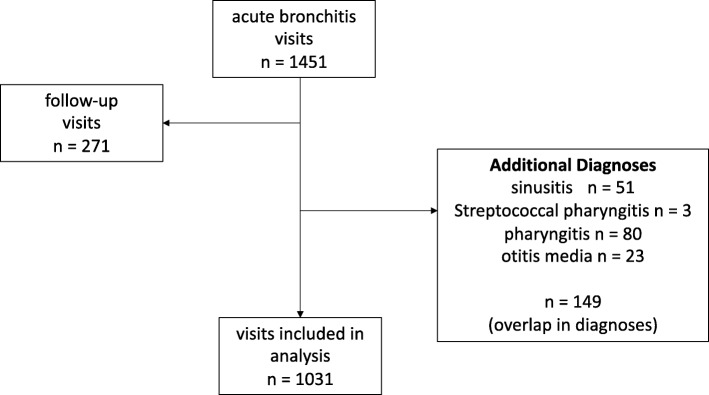
We subsequently excluded data on 271 follow-up visits within UHS for previously diagnosed conditions and 149 visits with additional diagnoses for which antibiotics might be appropriate (sinusitis, pharyngitis, streptococcal pharyngitis, otitis media). One thousand thirty-one visits were included in the final analysis (Fig. 1).
Fig. 1.
Flow of study inclusion and exclusion criteria for acute bronchitis visits (n = number of visits). Side arrows indicate exclusion criteria
Data from electronic patient charts included variables for all symptoms and exam findings listed in the record system. Many of these symptoms (e.g. eye discharge, mouth sores) were uncommon in acute bronchitis visits. To narrow the list to variables that might be important in acute bronchitis visits, as well as to eliminate variables with zero frequency cells in univariate contingency tables, we only considered symptoms and exam findings recorded for > 20 patients for subsequent analysis (Table 1).
Four visits had onset durations that were extreme outliers (> 100 days since onset), and we substituted missing values for these onset durations. Models excluded visits with missing values in predictor variables. This strategy resulted in 33 visits being excluded from analysis in the final multivariate model due to missing values in predictor variables. It is important to note that for two patient-reported variables, severity and progression, “not recorded” was coded as a factor level, and these entries were not considered missing values.
Statistical methods
In all analyses, the response variable was whether an antibiotic was prescribed at a visit. All variables listed in Table 1 were tested as possible predictive factors. Bivariate logistic regression analyses were used to identify a narrowed list of potential predictors of antibiotic prescribing (Table 1) [35]. Provider traits were not included in the logistic regression analysis due to the small number of providers in the data set (21 total). All variables identified as significant in the bivariate analyses were entered into multivariate logistic regression analyses to identify independent predictors of antibiotic prescribing for acute bronchitis. Backward stepwise removal of nonsignificant variables was used to generate the final multivariate model [35]. Factors were considered significant in the regression analyses when they had p-values < 0.05. Analyses were carried out using R (version 3.4.3).
Results
Study population and antibiotic prescribing
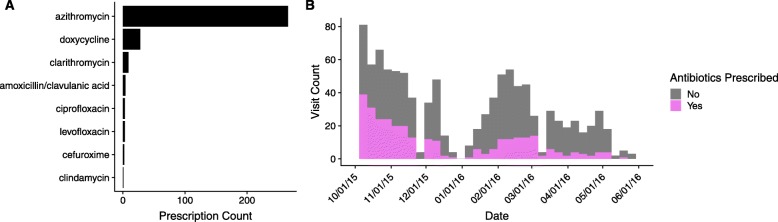
The data set included 1031 visits with an acute bronchitis diagnosis (Table 1). 61.7% of patients were female, and 90.1% of patients were undergraduate students. Antibiotics were prescribed at 30.8% of visits. Azithromycin was the most commonly prescribed antibiotic (83.9% of prescriptions) (Fig. 2a). Figure 2b shows the distribution of acute bronchitis visits and rates of antibiotic prescribing over the course of the 2015–2016 academic year. Table 1 shows the frequency of antibiotic prescribing by variable.
Fig. 2.
Antibiotic prescribing. a Antibiotic prescriptions by drug. b Visits and antibiotic prescribing over time
Factors associated with antibiotic use
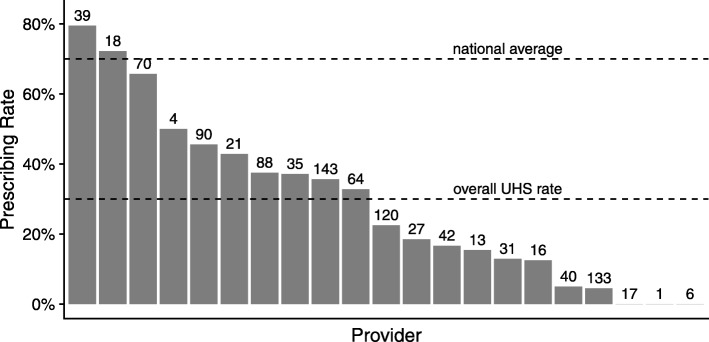
Factors independently associated with antibiotic prescribing in a multivariate regression model are summarized in Table 2. The factors with the greatest impacts on prescribing were individual providers and suspicious findings during lung examinations. The 21 providers in the data set had dramatically different rates of prescribing for acute bronchitis cases ranging from 0 to 80% (Fig. 3), and provider was an important predictor of prescribing (odds ratios (OR) ranged from 0.03 to 12.3 for individual providers). Suspicious findings during lung examinations were highly associated with antibiotic prescribing (rales OR 13.95, 95% CI 3.31–80.73; rhonchi OR 5.50, 95% CI 3.08–10.00; percussion abnormality OR 13.02, 95% CI 4.00–50.09).
Table 2.
Factors independently associated with prescribing in a multivariate model
| Variable | Odds Ratio (95% CI) | p-value |
|---|---|---|
| Visit and Patient Characteristics | ||
| visit date (days) | 0.99 (0.99-0.99) | p < 0.001** |
| gender | ||
| female (reference group) | ||
| male | 1.68 (1.17-2.41) | p = 0.005** |
| provider | 0.03-12.3 | p < 0.001** |
| onset duration (days) | 1.04 (1.03-1.06) | p < 0.001** |
| progression | ||
| stable/no change (reference group) | ||
| worsening | 1.78 (1.03-3.10) | p = 0.04* |
| improving | 0.74 (0.35-1.54) | p = 0.43 |
| not recorded | 1.69 (0.87-3.27) | p = 0.12 |
| Additional Diagnosis | ||
| URI diagnosis | 0.33 (0.18-0.58) | p < 0.001** |
| Symptoms | ||
| sneezing | 0.39 (0.17-0.86) | p = 0.02* |
| vomiting | 0.31 (0.10-0.83) | p = 0.03* |
| Exam Findings | ||
| lymphatics | ||
| anterior cervical lymphadenopathy, tender | 3.85 (1.70-8.83) | p = 0.001** |
| anterior cervical lymphadenopathy | 2.63 (1.25-5.54) | p = 0.01* |
| lungs | ||
| rales | 13.95 (3.31-80.73) | p = 0.001** |
| rhonchi | 5.50 (3.08-10.00) | p < 0.001** |
| percussion abnormality | 13.02 (4.00-50.09) | p < 0.001** |
Fig. 3.
Antibiotic prescribing rates by provider. Prescribing rates for acute bronchitis visits were highly variable among providers. The total number of acute bronchitis visits for each provider is shown above the bar, together with the national average [7–9, 16], and the overall rate at the PSU health facility
The model showed that prescribing rates decreased slightly over the course of the academic year (OR 0.99 per day, 95% CI 0.99–0.99). Male patients were more likely than female patients to be prescribed antibiotics (OR 1.68, 95% CI 1.17–2.41). Patients who reported longer duration since the onset of symptoms were slightly more likely to receive prescriptions (OR 1.04 per day, 95% CI 1.03–1.06), as were patients who reported their symptoms were worsening (OR 1.78, 95% CI 1.03–3.10). Visits with additional diagnoses or symptoms associated with viral infections or allergies were less likely to result in prescriptions (URI diagnosis OR 0.33, 95% CI 0.18–0.58; sneezing OR 0.39, 95% CI 0.17–0.86; vomiting OR 0.31, 95% CI 0.10–0.83). An exam finding of anterior cervical lymphadenopathy was associated with antibiotic prescribing (tender OR 3.85, 95% CI 1.70–8.83; general OR 2.63, 95% CI 1.25–5.54).
As a check, we repeated these analyses without excluding the data from follow-up visits (N = 149) for previously diagnosed conditions (Figure 4 in Appendix). The results of this analysis were qualitatively similar to the primary analysis, with the addition of antibiotic prescriptions in the past month as a predictor of prescribing (Tables 3 and 4 in Appendix). Patients who reported taking antibiotics in the past month were less likely to be prescribed antibiotics (OR 0.31, 95% CI 0.14–0.66). Provider and lung exam findings were the strongest predictors of prescribing in both analyses. Visit date, duration since onset, progression, URI diagnosis, sneezing, and anterior cervical lymphadenopathy were also significant predictors in both analyses.
Discussion
This study’s results indicated two key drivers of antibiotic prescribing: variation between individual providers and diagnostic uncertainty. We take each of these in turn. Individual providers had extraordinarily variable rates of antibiotic prescribing for acute bronchitis (ranging from 0 to 80%), despite treating the same patient population at the same clinic. These results suggest that a subset of providers can drive a disproportionate amount of unnecessary antibiotic prescribing for acute bronchitis. In the current study, provider traits (e.g. provider specialty, age) were not included in the logistic regression analysis due to the small number of providers in the data set (21 total). Previous studies have identified provider specialty, provider age, and perceived patient demand for antibiotics as factors influencing provider prescribing rates for upper respiratory tract infections [9, 20, 26, 36, 37].
A second important driver may be diagnostic uncertainty. In the present study, prescriptions were much more likely when findings of rales, rhonchi, or percussion abnormalities were recorded during lung examination, and somewhat more likely when external anterior cervical lymphadenopathy was reported. Rales and percussion abnormalities increased prescribing 13-fold, and rhonchi increased prescribing 5-fold. This increase in prescribing may reflect suspicion of pneumonia. Orders of chest x-rays, which also indicate suspicion of pneumonia, were a significant predictor of prescribing in a bivariate analysis, but were not significant in a multivariate model due to high correlation with other lung exam findings. Providers may prescribe antibiotics when there is suspicion of a condition that would respond to antibiotics or general diagnostic uncertainty [27], and this may not be reflected in the diagnosis code.
Other predictors of prescribing in this study included symptoms of sneezing and vomiting, reported worsening of symptoms, diagnosis of an upper respiratory tract infection, duration of illness, and patient gender. Duration of illness has previously been associated with prescribing for upper respiratory tract infections [28]. Patient gender has not typically been associated with prescribing rates for acute bronchitis [16, 20, 28, 29], although some studies have reported that males are more likely to get antibiotic prescriptions for upper respiratory tract infections [8, 17].
The identification of provider variation and diagnostic uncertainty as drivers of prescribing suggests possible interventions for this clinic and similar settings. Provider variation points to a need for provider-targeted interventions such as audit and feedback, communication training, provider education, or clinical decision support tools [22, 23]. In an ‘audit and feedback’ intervention, individual clinicians receive personalized, ongoing feedback on their prescribing rates [22–24, 38, 39]. In one study, quarterly feedback resulted in a 50% relative reduction in broad-spectrum antibiotic use for respiratory tract infections [24]. Provider communication training has also been shown to decrease unnecessary antibiotic prescribing [23]. Communication training addresses provider concerns related to patient satisfaction and patient expectation for antibiotics [23]. In some cases, diagnostic uncertainty may be addressed through point of care diagnostic testing [22]. Point of care diagnostics are available for respiratory tract infections including Group A Streptococcus and influenza [22]. There is some evidence supporting point of care testing to reduce antibiotic prescribing for respiratory tract infections [23, 40].
While unnecessary prescribing for acute bronchitis was common in our data, the rate of prescribing was substantially lower than the nationwide average. In the 2015–2016 academic year, antibiotics were prescribed at less than a third of acute bronchitis visits, compared to national rates near 70% [7–9, 16]. There is still room for improvement, but overall, this suggests that lower rates of prescribing for acute bronchitis are achievable.
Our study is unique in its focus on antibiotic prescribing practices at a university health center. University health services are important centers for antibiotic prescribing serving millions of patients, yet they have largely been overlooked as sites for antibiotic stewardship. To our knowledge, the Pennsylvania State University is the first university with a student antibiotic stewardship program. This study is the first to identify drivers of antibiotic prescribing in a university health center, and one of the few to focus on young adults or consider exam findings and symptoms from patient charts as possible predictors of prescribing. We hope that these findings can be used to inform antibiotic stewardship initiatives at university health centers and similar clinical contexts. Our results suggest that unnecessary antibiotic prescribing is disproportionately driven by a subset of clinicians, and interventions targeting providers may be effective at reducing unnecessary prescribing.
Conclusions
Reducing unnecessary antibiotic prescribing for acute bronchitis cases is a national antibiotic stewardship goal, yet rates of unnecessary antibiotic prescribing remain stubbornly high nationwide. Here we identified factors that influence antibiotic prescribing for acute bronchitis cases at a large university health center. Suspicious findings during lung examinations (rales, rhonchi, percussion abnormality) and individual healthcare providers were the most influential factors affecting antibiotic prescribing rates for acute bronchitis visits. Patient gender, worsening symptoms, duration of illness, symptoms associated with viral infections or allergies, and anterior cervical lymphadenopathy also influenced prescribing rates.
Acknowledgments
We are grateful to the University Health Services Antibiotic Stewardship committee for their enthusiastic support. Karen Bascom, Karen Anderson, and Julia Lundy were instrumental to data entry. Ruth Anne Snyder, Bettyann Milliron, Carmel Kamens, and Cristie Happekotte helped coordinate access and training for the honest brokers. We also thank Sue Johnson for her help and support with computing. This study grew out of discussions initiated by Lewis Logan. We thank reviewers Katherine Ka Wai Lam, Yu Feng, and Sofiane Bakour and editor Simon Ching Lam for their helpful comments on this manuscript.
Abbreviations
- GI
Gastrointestinal
- PSU
Pennsylvania State University
- TM
Tympanic membrane
- UHS
University Health Services
- URI
Upper respiratory tract infection
Appendix
Analysis including follow-up visits
The logistic regression analyses described in the main text were repeated with a data set including follow-up visits for previously diagnosed conditions, which had been excluded from the original analysis. Antibiotics were prescribed at 30.0% of visits.
Fig. 4.
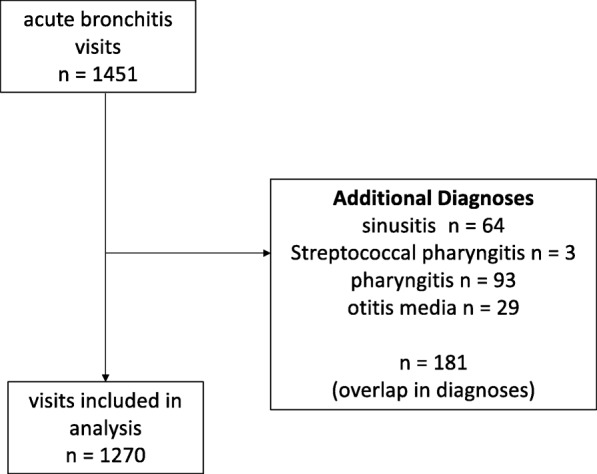
Flow of study inclusion and exclusion criteria for acute bronchitis visits (n = number of visits). Side arrows indicate exclusion criteria
Table 3.
Descriptive statistics (n = 1270) and bivariate analysis
| Variable | Visits | Odds Ratio (95% CI) | Bivariatep-value |
|---|---|---|---|
| Date and Time | |||
| visit date | 1267 (99.8%) | 0.99 (0.99-0.99) | p < 0.001 ** |
| week day | 1267 (99.8%) | range 0.60-1.40 | p = 0.26 |
| time of day | 1258 (99.0%) | 0.99 (0.99-1.00) | p = 0.61 |
| Patient Characteristics | |||
| gender | p = 0.07 | ||
| female (reference group) | 788 (62.0%) | - | |
| male | 477 (46.3%) | 1.25 (0.98-1.60) | |
| not recorded | 5 (0.4%) | - | |
| race | p = 0.20 | ||
| white (reference group) | 737 (58.0%) | - | |
| multiple | 117 (9.2%) | 0.76 (0.48-1.17) | |
| Asian | 61 (4.8%) | 0.88 (0.49-1.54) | |
| black | 31 (2.4%) | 1.00 (0.45-2.11) | |
| Hispanic | 7 (0.6%) | - | |
| international | 8 (0.6%) | - | |
| Pacific islander | 2 (0.1%) | - | |
| not recorded | 307 (24.2%) | - | |
| academic status | p = 0.70 | ||
| undergraduate student (reference group) | 1153 (90.8%) | - | |
| graduate student | 101 (7.9%) | 0.83 (0.52-1.30) | |
| spouse/dependent | 4 (0.3%) | - | |
| not recorded | 12 (0.9%) | - | |
| height (inches) | 1242 (97.8%) | 1.03 (1.00-1.07) | p = 0.04* |
| weight (pounds) | 1236 (97.3%) | 1.00 (0.99-1.01) | p = 0.15 |
| Visit Characteristics | |||
| provider | 1254 (98.7%) | range 0.07 -3.27 | p < 0.001** |
| onset duration (patient reported) | 1250 (98.4%) | 0.99 (0.99-1.00) | p < 0.001** |
| severity (patient reported) | p = 0.02* | ||
| mild (reference group) | 69 (5.4%) | - | |
| moderate | 381 (30.0%) | 0.54 (0.32-0.95) | |
| severe | 37 (2.9%) | 1.20 (0.52-2.72) | |
| not recorded | 783 (61.6%) | - | |
| timing (patient reported) | p < 0.001** | ||
| stable/no change (reference group) | 322 (25.3%) | - | |
| worsening | 370 (29.1%) | 2.59 (1.84-3.65) | |
| improving | 176 (13.8%) | 0.67 (0.40-1.09) | |
| not recorded | 402 (31.6%) | - | |
| antibiotics in past month | 85 (6.7%) | 0.44 (0.23-0.77) | p = 0.006** |
| Additional Diagnosis | |||
| URI | 216 (17.0%) | 0.30 (0.19-0.44) | p < 0.001** |
| suspicious cough | 102 (8.0%) | 1.30 (0.84-1.98) | p = 0.22 |
| allergic rhinitis | 38 (3.0%) | 0.43 (0.16-0.96) | p = 0.06 |
| fever | 19 (1.5%) | 2.13 (0.84-5.31) | p = 0.10 |
| viral syndrome | 15 (1.2%) | - | - |
| tonsillitis | 6 (0.4%) | - | - |
| influenza | 5 (0.4%) | - | - |
| mononucleosis | 7 (0.5%) | - | - |
| Common Symptoms Recorded† | |||
| throat symptoms | |||
| sore throat | 459 (36.1%) | 1.45 (1.13-1.86) | p = 0.003** |
| painful swallowing | 146 (11.5%) | 0.60 (0.39-0.89) | p = 0.01* |
| hoarseness | 186 (14.6%) | 0.84 (0.59-1.18) | p = 0.32 |
| swollen glands in neck | 126 (9.9%) | 1.19 (0.80-1.75) | p = 0.39 |
| systemic symptoms | |||
| headache | 272 (21.4%) | 0.88 (0.65-1.18) | p = 0.40 |
| documented fever | 73 (5.7%) | 0.63 (0.39-1.04) | p = 0.06 |
| fever symptoms (patient reported) | 285 (22.4%) | 1.10 (0.83-1.46) | p = 0.51 |
| chills | 164 (12.9%) | 0.86 (0.60-1.22) | p = 0.38 |
| sweats | 182 (14.3%) | 1.02 (0.73-1.45) | p = 0.92 |
| nasal symptoms | |||
| stuffy nose | 782 (61.6%) | 1.04 (0.81-1.33) | p = 0.76 |
| sinus congestion | 402 (39.0%) | 0.83 (0.64-1.08) | p = 0.16 |
| clear nasal discharge | 251 (19.8%) | 0.86 (0.63-1.16) | p = 0.33 |
| purulent nasal discharge | 212 (16.7%) | 0.96 (0.69-1.32) | p = 0.79 |
| post-nasal drip sensation | 444 (35.0%) | 1.10 (0.86-1.41) | p = 0.46 |
| sinus pain | 85 (6.7%) | 1.79 (1.14-2.79) | p = 0.01* |
| sneezing | 108 (8.5%) | 0.47 (0.28-0.77) | p = 0.004** |
| pulmonary symptoms | |||
| sleep disruption due to cough | 710 (55.9%) | 1.08 (0.85-1.37) | p = 0.54 |
| sputum production | 743 (58.5%) | 0.94 (0.73-1.20) | p = 0.61 |
| shortness of breath | 412 (32.4%) | 0.69 (0.53-0.88) | p = 0.003** |
| chest tightness | 331 (26.1%) | 0.82 (0.62-1.07) | p = 0.14 |
| wheezing | 340 (26.8%) | 0.64 (0.49-0.84) | p = 0.001** |
| chest pain | 269 (21.2%) | 0.75 (0.56-0.99) | p = 0.047* |
| paroxysms of cough | 362 (28.5%) | 0.91 (0.70-1.18) | p = 0.46 |
| ear symptoms | |||
| ear pain | 59 (4.6%) | 1.03 (0.57-1.78) | p = 0.93 |
| ear pressure sensation | 143 (11.2%) | 0.93 (0.63-1.36) | p = 0.71 |
| decreased hearing | 41 (3.2%) | 0.96 (0.47-1.87) | p = 0.92 |
| GI symptoms | |||
| loss of appetite | 146 (11.5%) | 0.80 (0.53-1.17) | p = 0.26 |
| abdominal pain | 29 (2.3%) | 0.26 (0.06-0.75) | p = 0.03* |
| post-tussive vomiting | 85 (6.7%) | 0.59 (0.38-0.92) | p = 0.02* |
| nausea | 72 (5.7%) | 0.50 (0.26-0.89) | p = 0.02* |
| vomiting | 58 (4.6%) | 0.73 (0.38-1.32) | p = 0.32 |
| diarrhea | 37 (2.9%) | 1.27 (0.62-2.49) | p = 0.49 |
| neuro-vascular symptoms | |||
| lightheadedness | 59 (4.6%) | 1.30 (0.74-2.24) | p = 0.33 |
| Commonly Ordered Labs | |||
| chest x-ray | 223 (17.6%) | 2.17 (1.61-2.91) | p < 0.001** |
| rapid strep screen | 37 (2.9%) | 0.54 (0.21-1.16) | p = 0.14 |
| complete blood count | 115 (9.0%) | 1.38 (0.92-2.06) | p = 0.11 |
| monospot | 36 (2.8%) | 1.33 (0.65-2.61) | p = 0.41 |
| influenza A + B | 19 (1.5%) | 0.27 (0.04-0.95) | p = 0.08 |
| Common Exam Findings† | |||
| ear exam | |||
| tympanic membrane (TM) bulging | 32 (2.5%) | 0.07 (0.004-0.34) | p = 0.01* |
| TM retraction | 48 (3.8%) | 0.77 (0.38-1.46) | p = 0.44 |
| visible fluid behind TM | 173 (13.6%) | 0.18 (0.10-0.30) | p < 0.001** |
| cerumen in canal | 37 (2.9%) | 0.64 (0.27-1.34) | p = 0.26 |
| nose exam | |||
| mucosal edema | 707 (55.7%) | 0.83 (0.65-1.06) | p = 0.14 |
| mucosal erythema | 606 (47.7%) | 0.80 (0.63-1.02) | p = 0.07 |
| nasal discharge | 382 (30.1%) | 1.58 (1.22-2.04) | p < 0.001** |
| maxillary sinus tenderness | 35 (2.7%) | 2.01 (1.01-3.95) | p = 0.04* |
| throat exam | |||
| erythema | 240 (18.9%) | 0.76 (0.55-1.04) | p = 0.09 |
| lymphoid hyperplasia | 114 (9.0%) | 1.59 (1.07-2.36) | p = 0.02* |
| post-nasal drip | 181 (14.2%) | 1.41 (1.01-1.95) | p = 0.04* |
| tonsil exam | |||
| surgically absent | 56 (4.4%) | 0.93 (0.50-1.65) | p = 0.81 |
| erythema | 97 (7.6%) | 0.47 (0.27-0.79) | p = 0.006** |
| enlarged | 57 (4.5%) | 1.10 (0.62-2.04) | p = 0.74 |
| lymphatics exam | |||
| anterior cervical lymphadenopathy, tender | 64 (5.0%) | 1.88 (1.12-3.12) | p = 0.01* |
| anterior cervical lymphadenopathy, non-tender | 103 (8.1%) | 0.81 (0.51-1.27) | p = 0.38 |
| posterior cervical lymphadenopathy, non-tender | 62 (4.9%) | 0.67 (0.35-1.20) | p = 0.19 |
| anterior cervical lymphadenopathy | 132 (10.4%) | 2.80 (1.94-4.04) | p < 0.001** |
| posterior cervical lymphadenopathy | 38 (3.0%) | 1.08 (0.52-2.12) | p = 0.83 |
| lung exam | |||
| wheezing | 253 (19.9%) | 1.82 (1.37-2.43) | p < 0.001** |
| rales | 22 (1.7%) | 8.26 (3.24-25.27) | p < 0.001** |
| rhonchi | 273 (21.5%) | 2.30 (1.74-3.03) | p < 0.001** |
| percussion abnormality | 30 (2.4%) | 9.89 (4.27-26.89) | p < 0.001** |
† includes symptoms and findings recorded for > 20 visits
Table 4.
Factors independently associated with prescribing in a multivariate model
| Variable | Odds Ratio (95% CI) | p-value |
|---|---|---|
| Visit and Patient Characteristics | ||
| visit date (days) | 0.99 (0.99-0.99) | p < 0.001** |
| height (inches) | 1.05 (1.00-1.09) | p = 0.03* |
| provider | range 0.04-8.42 | p < 0.001** |
| onset duration (days) | 1.04 (1.03-1.05) | p < 0.001** |
| progression | ||
| stable/no change (reference group) | ||
| worsening | 1.79 (1.11-2.90) | p = 0.02* |
| improving | 0.43 (0.23-0.81) | p = 0.01* |
| not recorded | 1.43 (0.83-2.48) | p = 0.20 |
| antibiotics in past month | 0.32 (0.14-0.65) | p = 0.003** |
| Diagnosis | ||
| URI diagnosis | 0.36 (0.21-0.62) | p < 0.001** |
| Symptoms | ||
| sore throat | 1.46 (1.04-2.05) | p = 0.03* |
| sneezing | 0.48 (0.22-0.97) | p = 0.048* |
| Exam Findings | ||
| lymphatics | ||
| anterior cervical lymphadenopathy, tender | 2.55 (1.27-5.13) | p = 0.008** |
| anterior cervical lymphadenopathy | 2.88 (1.55- 5.39) | p < 0.001** |
| lungs | ||
| rales | 10.21 (3.16-60.02) | p < 0.001** |
| rhonchi | 5.08 (3.10-8.49) | p < 0.001** |
| percussion abnormality | 9.69 (3.47-30.79) | p < 0.001** |
Authors’ contributions
VJM analyzed the data and wrote the manuscript. EPCF and RRV were major contributors to data collection and processing. YZ assisted with literature review and manuscript writing. MZ, AFR, and ELM conceived, designed, and managed the study, and contributed substantially to manuscript writing. All authors read and approved the final manuscript.
Funding
This study was funded by the Huck Institutes of the Life Sciences at Pennsylvania State University. The funding body did not have a role in designing the study, in collection, analysis, and interpretation of data, or in writing the manuscript.
Availability of data and materials
The datasets analyzed during the current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
The Pennsylvania State University Institutional Review Board (IRB) approved this research. Honest brokers were employed and trained to access electronic medical records for clinical visits, extract deidentified data (data excluding information that could be used to identify individual patients), and enter it in the secure database manager REDCap for use by the researchers. Informed consent was waived by the IRB given the retrospective nature of the study and the use of honest brokers for deidentification.
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Fleming-Dutra KE, Hersh AL, Shapiro DJ, et al. Prevalence of inappropriate antibiotic prescriptions among U.S. ambulatory care visits, 2010-2011. JAMA. 2016;315(17):1864–1873. doi: 10.1001/jama.2016.4151. [DOI] [PubMed] [Google Scholar]
- 2.Pew Research Center. Antibiotic use in outpatient settings. Washington D.C: The Pew Chartible Trusts; 2016.
- 3.Friedman ND, Temkin E, Carmeli Y. The negative impact of antibiotic resistance. Clin Microbiol Infect. 2016;22(5):416–422. doi: 10.1016/j.cmi.2015.12.002. [DOI] [PubMed] [Google Scholar]
- 4.Holmes AH, Moore LSP, Sundsfjord A, Steinbakk M, Regmi S, Karkey A, et al. Understanding the mechanisms and drivers of antimicrobial resistance. Lancet. 2016;387(10014):176–187. doi: 10.1016/S0140-6736(15)00473-0. [DOI] [PubMed] [Google Scholar]
- 5.zur Wiesch PA, Kouyos R, Engelstädter J, Regoes RR, Bonhoeffer S. Population biological principles of drug-resistance evolution in infectious diseases. Lancet Infect Dis. 2011;11(3):236–247. doi: 10.1016/S1473-3099(10)70264-4. [DOI] [PubMed] [Google Scholar]
- 6.The White House . National action plan for combating antibiotic-resistant bacteria. 2015. [Google Scholar]
- 7.Barnett ML, Linder JA. Antibiotic prescribing for adults with acute bronchitis in the United States, 1996-2010. JAMA. 2014;311(19):2020–2022. doi: 10.1001/jama.2013.286141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Brown DW, Taylor R, Rogers A, Weiser R, Kelley M. Antibiotic prescriptions associated with outpatient visits for acute upper respiratory tract infections among adult Medicaid recipients in North Carolina. N C Med J. 2003;64(4):148–156. [PubMed] [Google Scholar]
- 9.Schmidt ML, Spencer MD, Davidson LE. Patient, provider, and practice characteristics associated with inappropriate antimicrobial prescribing in ambulatory practices. Infect Control Hosp Epidemiol. 2018;39(3):307–315. doi: 10.1017/ice.2017.263. [DOI] [PubMed] [Google Scholar]
- 10.Gonzales R, Steiner JF, Sande MA. Antibiotic prescribing for adults with colds, upper respiratory tract infections, and bronchitis by ambulatory care physicians. JAMA J Am Med Assoc. 1997;278(11):901. doi: 10.1001/jama.1997.03550110039033. [DOI] [PubMed] [Google Scholar]
- 11.Smith SM, Fahey T, Smucny J, Becker LA. Antibiotics for acute bronchitis. Cochrane Database Syst Rev. 2017;6:CD000245. doi: 10.1002/14651858.CD000245.pub4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Talwalkar A, Hing E, Palso K. National Hospital Ambulatory Medical Care Survey: 2011 outpatient department summary tables. Hyattsville: U.S. Dept. of Health and Human Services, Public Health Service, Centers for Disease Control and Prevention, National Center for Health Statistics; 2011. [Google Scholar]
- 13.Macfarlane J, Holmes W, Gard P, Macfarlane R, Rose D, Weston V, et al. Prospective study of the incidence, aetiology and outcome of adult lower respiratory tract illness in the community. Thorax. 2001;56(2):109–114. doi: 10.1136/thorax.56.2.109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Gonzales R, Bartlett JG, Besser RE, Cooper RJ, Hickner JM, Hoffman JR, et al. Principles of appropriate antibiotic use for treatment of uncomplicated acute bronchitis: background. Ann Intern Med. 2001;134(6):521. doi: 10.7326/0003-4819-134-6-200103200-00021. [DOI] [PubMed] [Google Scholar]
- 15.Irwin RS, Baumann MH, Bolser DC, Boulet L-P, Braman SS, Brightling CE, et al. Diagnosis and management of cough executive summary. Chest. 2006;129(1):1S–23S. doi: 10.1378/chest.129.1_suppl.1S. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Grigoryan L, Zoorob R, Shah J, Wang H, Arya M, Trautner BW. Antibiotic prescribing for uncomplicated acute bronchitis is highest in younger adults. Antibiotics. 2017;6(4):E22. doi: 10.3390/antibiotics6040022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Xu KT, Roberts D, Sulapas I, Martinez O, Berk J, Baldwin J. Over-prescribing of antibiotics and imaging in the management of uncomplicated URIs in emergency departments. BMC Emerg Med. 2013;13(1):7. doi: 10.1186/1471-227X-13-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Aspinall SL, Good CB, Metlay JP, Mor MK, Fine MJ. Antibiotic prescribing for presumed nonbacterial acute respiratory tract infections. Am J Emerg Med. 2009;27(5):544–551. doi: 10.1016/j.ajem.2008.04.015. [DOI] [PubMed] [Google Scholar]
- 19.Donnelly JP, Baddley JW, Wang HE. Antibiotic utilization for acute respiratory tract infections in U.S. emergency departments. Antimicrob Agents Chemother. 2014;58(3):1451–1457. doi: 10.1128/AAC.02039-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.McKay R, Mah A, Law MR, McGrail K, Patrick DM. Systematic review of factors associated with antibiotic prescribing for respiratory tract infections. Antimicrob Agents Chemother. 2016;60(7):4106–4118. doi: 10.1128/AAC.00209-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Antibiotic use in the United States . 2018 Update: Progress and opportunities. Atlantia: US Department of Health and Human Services, CDC; 2019. [Google Scholar]
- 22.Dobson EL, Klepser ME, Pogue JM, Labreche MJ, Adams AJ, Gauthier TP, et al. Outpatient antibiotic stewardship: interventions and opportunities. J Am Pharm Assoc. 2017;57(4):464–473. doi: 10.1016/j.japh.2017.03.014. [DOI] [PubMed] [Google Scholar]
- 23.Drekonja DM, Filice GA, Greer N, Olson A, MacDonald R, Rutks I, et al. Antimicrobial stewardship in outpatient settings: a systematic review. Infect Control Hosp Epidemiol. 2015;36(2):142–152. doi: 10.1017/ice.2014.41. [DOI] [PubMed] [Google Scholar]
- 24.Gerber JS, Prasad PA, Fiks AG, Localio AR, Grundmeier RW, Bell LM, et al. Effect of an outpatient antimicrobial stewardship intervention on broad-spectrum antibiotic prescribing by primary care pediatricians a randomized trial. JAMA. 2013;309(22):2345–2352. doi: 10.1001/jama.2013.6287. [DOI] [PubMed] [Google Scholar]
- 25.Avorn J, Solomon DH. Cultural and economic factors that (mis) shape antibiotic use: the nonpharmacologic basis of therapeutics. Ann Intern Med. 2000;133(2):128–135. doi: 10.7326/0003-4819-133-2-200007180-00012. [DOI] [PubMed] [Google Scholar]
- 26.O’Connor R, O’Doherty J, O’Regan A, Dunne C. Antibiotic use for acute respiratory tract infections (ARTI) in primary care; what factors affect prescribing and why is it important? A narrative review. Ir J Med Sci. 2018;187(4):969–986. doi: 10.1007/s11845-018-1774-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Whaley LE, Businger AC, Dempsey PP, Linder JA. Visit complexity, diagnostic uncertainty, and antibiotic prescribing for acute cough in primary care: a retrospective study. BMC Fam Pract. 2013;14(1):120. doi: 10.1186/1471-2296-14-120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Gonzales R, Camargo CA, MacKenzie T, Kersey AS, Maselli J, Levin SK, et al. Antibiotic treatment of acute respiratory infections in acute care settings. Acad Emerg Med. 2006;13(3):288–294. doi: 10.1197/j.aem.2005.10.016. [DOI] [PubMed] [Google Scholar]
- 29.Gonzales R, Barrett PH, Crane LA, Steiner JF, Steiner JF. Factors associated with antibiotic use for acute bronchitis. J Gen Intern Med. 1998;13(8):541–548. doi: 10.1046/j.1525-1497.1998.00165.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Snyder TD, de Brey C, Dillow S. Digest of education statistics: 2017. Washington, DC: National Center for Educational Statistics, US Department of Education, Institute of Education Sciences; 2019. [Google Scholar]
- 31.Turner JC, Keller A. College health surveillance network: epidemiology and health care utilization of college students at us 4-year universities. J Am Coll Heal. 2015;63(8):530–538. doi: 10.1080/07448481.2015.1055567. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Atkinson I. Accuracy of data transfer: double data entry and estimating levels of error. J Clin Nurs. 2012;21(19pt20):2730–2735. doi: 10.1111/j.1365-2702.2012.04353.x. [DOI] [PubMed] [Google Scholar]
- 33.Day S, Fayers P, Harvey D. Double data entry: what value, what price? Control Clin Trials. 1998;19(1):15–24. doi: 10.1016/S0197-2456(97)00096-2. [DOI] [PubMed] [Google Scholar]
- 34.Barchard KA, Pace LA. Preventing human error: the impact of data entry methods on data accuracy and statistical results. Comput Human Behav. 2011;27(5):1834–1839. doi: 10.1016/j.chb.2011.04.004. [DOI] [Google Scholar]
- 35.Hosmer David W., Lemeshow Stanley, Sturdivant Rodney X. Applied Logistic Regression. Hoboken, NJ, USA: John Wiley & Sons, Inc.; 2013. [Google Scholar]
- 36.Zuckerman IH, Perencevich EN, Harris AD. Concurrent acute illness and comorbid conditions poorly predict antibiotic use in upper respiratory tract infections: a cross-sectional analysis. BMC Infect Dis. 2007;7:47. doi: 10.1186/1471-2334-7-47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Dempsey PP, Businger AC, Whaley LE, Gagne JJ, Linder JA. Primary care clinicians’ perceptions about antibiotic prescribing for acute bronchitis: a qualitative study. BMC Fam Pract. 2014;15(1):194. doi: 10.1186/s12875-014-0194-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Meeker D, Linder JA, Fox CR, Friedberg MW, Persell SD, Goldstein NJ, et al. Effect of behavioral interventions on inappropriate antibiotic prescribing among primary care practices a randomized clinical trial. J Am Med Assoc. 2016;315(6):562–570. doi: 10.1001/jama.2016.0275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Hemkens LG, Saccilotto R, Reyes SL, Glinz D, Zumbrunn T, Grolimund O, et al. Personalized prescription feedback using routinely collected data to reduce antibiotic use in primary care a randomized clinical trial. JAMA Intern Med. 2017;177(2):176–183. doi: 10.1001/jamainternmed.2016.8040. [DOI] [PubMed] [Google Scholar]
- 40.Klepser Donald G., Klepser Michael E., Dering-Anderson Allison M., Morse Jacqueline A., Smith Jaclyn K., Klepser Stephanie A. Community pharmacist–physician collaborative streptococcal pharyngitis management program. Journal of the American Pharmacists Association. 2016;56(3):323-329.e1. doi: 10.1016/j.japh.2015.11.013. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets analyzed during the current study are available from the corresponding author on reasonable request.