Abstract
Bone metabolism is a lifelong process that includes bone formation and resorption. Osteoblasts and osteoclasts are the predominant cell types associated with bone metabolism, which is facilitated by other cells such as bone marrow mesenchymal stem cells (BMMSCs), osteocytes and chondrocytes. As an important component in our daily diet, fatty acids are mainly categorized as long‐chain fatty acids including polyunsaturated fatty acids (LCPUFAs), monounsaturated fatty acids (LCMUFAs), saturated fatty acids (LCSFAs), medium‐/short‐chain fatty acids (MCFAs/SCFAs) as well as their metabolites. Fatty acids are closely associated with bone metabolism and associated bone disorders. In this review, we summarized the important roles and potential therapeutic implications of fatty acids in multiple bone disorders, reviewed the diverse range of critical effects displayed by fatty acids on bone metabolism, and elucidated their modulatory roles and mechanisms on specific bone cell types. The evidence supporting close implications of fatty acids in bone metabolism and disorders suggests fatty acids as potential therapeutic and nutritional agents for the treatment and prevention of metabolic bone diseases.
Keywords: bone diseases, bone homeostasis, bone metabolism, fatty acids
1. INTRODUCTION
Bone metabolism including osteoclasts‐mediated bone resorption and osteoblasts‐mediated bone formation is a lifelong process occurring within cancellous as well as cortical bones. Bone resorption starts with recruitment of osteoclasts to mineralized bone tissues and leads to acidification of extracellular microenvironment. Osteoclasts dissolve hydroxyapatite mineral crystals by producing hydrogen ions and digesting organic bone matrix via synthesis of hydrolytic enzymes, both resulting in calcium transfer from bone tissue into blood.1, 2 Bone formation is initiated by bone marrow mesenchymal stem cells (BMMSCs) migrating from vascular channels circulation to bone surface. Osteoblasts deposit organic bone matrix and regulate its mineralization and eventually differentiate into osteocytes that are embedded in the cavities of mineralized matrix.2, 3 In addition to osteoclasts, BMMSCs and osteoblasts, other bone cell types participating in bone metabolism include macrophages, surface bone‐lining cells, chondrocytes as well as osteocytes (Figure 1).4, 5, 6, 7, 8, 9, 10
Figure 1.
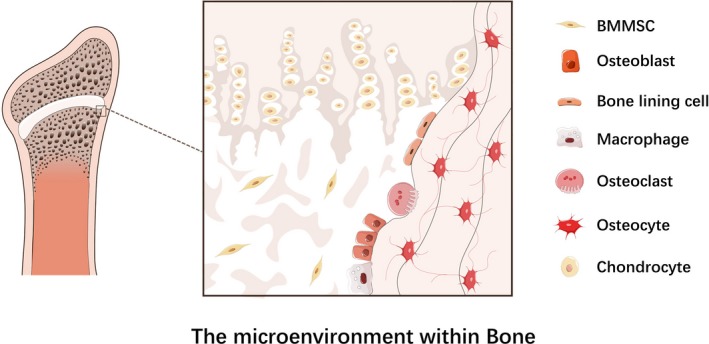
The microenvironment within bone. This figure displays the composition of bone microenvironment at cellular level. BMMSCs are multipotent cells capable of differentiating into multiple cell types such as osteoblasts. Osteoblasts are mononuclear cells responsible for bone formation. Bone lining cells are flat‐shaped cells located along the bony surfaces, maintaining their proliferative ability into other osteogenic cells. Macrophages are common precursors of osteoclasts and bone marrow‐resident macrophages in bone tissue. Osteoclasts are multinucleated giant cells with bone resorptive activity. Osteocytes are cells reside in bone lacunae and support bone structure, also with capacity to regulate the activities of both osteoclasts and osteoblasts. Chondrocytes are the main cartilage cell type existing in cartilaginous interstitium and cartilage lacuna. All these cell types existing in microenvironment within bone are implicated in bone homeostasis and thereby bone diseases
Accumulating evidence has established essential roles of fatty acids in bone metabolism 11 (Table 1). Categorization of fatty acids involved in bone metabolism has been reviewed by Natalia S. Harasymowicz et al 12 In general, ω‐3 long‐chain polyunsaturated fatty acids (LCPUFAs) are a group of well‐known fatty acids obtained from diet and supplemented via in vivo synthesis,13 and eicosapentaenoic acid (EPA), alpha‐linolenic acid (ALA) and docosahexaenoic acid (DHA) are the three major representatives of ω‐3 LCPUFAs. ω‐3 LCPUFAs could mediate bone metabolism via processes including lipid oxidation, calcium absorption and prostaglandin synthesis,14 and they can exert beneficial effects on bone remodelling by inhibiting osteoclast activity and enhancing osteoblast activity.15 Several studies have investigated the therapeutic properties of ω‐3 LCPUFAs. By promoting bone formation, ω‐3 LCPUFAs significantly affect peak bone mass,16 increase bone calcium levels as well as bone mineral content (BMC) and density.17, 18, 19, 20 Therefore, they represent a non‐pharmacological strategy for preventing bone loss and accelerating fracture healing 21 and thus to reduce risks of osteoporosis and rheumatoid arthritis.16, 22, 23 In addition, ingestion of ω‐3 LCPUFAs eliminates adriamycin‐ or cyclophosphamide‐induced toxicity in bone marrow and bone tissues, suggesting potential roles of ω‐3 LCPUFAs in combating side effects of specific bone‐targeted drugs.24
Table 1.
Overview of fatty acids involved in specific bone cell types and bone diseases
| Fatty acid | Effects on bone metabolism | Molecular formula | Structural formula | Class | Targeted cell | Receptor | Pathway | Relevant disease | References |
|---|---|---|---|---|---|---|---|---|---|
| Eicosapentaenoic acid (EPA) | Promote bone formation | C20H30O2 |
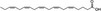
|
ω‐3 LCPUFAs |
Osteoblasts Osteoclasts BMMSCs Chondrocytes |
PPAR‐γ PTH1R GPR120 GPR40 |
MAPK mTOR |
Rheumatoid arthritis; Periodontitis; Osteocarcinoma | 181‐184, 205 |
| Docosahexaenoic acid (DHA) |
Promote bone formation; Inhibit bone resorption |
C22H32O2 |
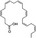
|
ω‐3 LCPUFAs |
Osteoblasts Osteoclasts BMMSCs Chondrocytes |
PPAR‐γ PTH1R TLR4 GPR120 GPR40 |
MAPK NF‐kB |
Rheumatoid arthritis; Periodontitis; Bone fracture; Osteocarcinoma |
181‐184, 205 |
| a‐Linolenic Acid (ALA) | Inhibit bone resorption | C18H30O2 |

|
ω‐6 LCPUFAs |
Osteoclasts Chondrocytes |
GPR40 |
MAPK NF‐kB |
— | 181 |
| Arachidonic Acid (AA) | Inhibit bone resorption | C20H32O2 |

|
ω‐6 LCPUFAs |
Osteoclasts Chondrocytes |
TLR4 |
MAPK NF‐kB |
Rheumatoid arthritis; Bone fracture |
181‐184, 205 |
| Myristic acid(MA) | Inhibit bone resorption | C14H26O2 |

|
ω‐5 LCMUFAs | Osteoclasts |
GPR40 |
RANKL |
Osteolysis; Osteoporosis |
25, 26 |
| Palmitoleic acid(PLA) | Inhibit bone resorption | C16H30O2 |

|
ω‐7 LCMUFAs |
Osteoclasts |
GPR40 |
NF‐kB MAPK |
Rheumatoid arthritis; Osteoporosis; Osteosclerosis |
166 |
| Oleic acid (OA) | Inhibit bone resorption | C18H34O2 |

|
ω‐9 LCMUFAs |
Osteoclasts BMMSCs |
GPR40 |
NF‐kB MAPK |
Bone healing; Osteoporosis; Periodontitis |
168, 202 |
| Palmitic acid (PA) |
Enhance bone resorption; Inhibit bone formation |
C16H32O2 |
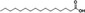
|
LCSFAs |
Osteoclasts, Osteoblasts BMMSCs Osteocytes Chondrocytes |
TLR4 PPAR‐γ |
MAPK, mTOR BMP NF‐kB |
Osteoarthritis; Bone loss; Periodontitis |
105, 180 |
| Stearic acid(SA) |
Enhance bone resorption; Inhibit bone formation |
C18H36O2 |
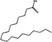
|
LCSFAs | Chondrocytes | — |
NF‐kB |
— | |
| Capric acid(CA) | Inhibit bone resorption | C10H20O2 |
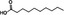
|
MCFAs | Osteoclasts |
GPR120 GPR40 GPR84 |
MAPK, NF‐kB |
Osteoporosis; Rheumatoid arthritis |
214 |
| Short chain FAs |
Osteoclasts Osteoblasts Chondrocytes |
GPR41 GPR43 GPR109 |
Inflammatory bone loss; Arthritis; Osteoporosis |
29, 64 | |||||
| ResolvinE1 (RvE1) |
Promote bone formation; Inhibit bone resorption |
C20H30O5 |
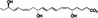
|
EPA metabolites |
Osteoblasts Osteoclasts |
ChemR23 | NF‐κB, MAPK | Periodontitis | 151 |
| Lipoxin A4 (LXA4) | Inhibit bone resorption | C20H32O5 |
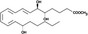
|
AA metabolites | Osteoclasts | FPR2/ALX |
NF‐κB, MAPK mTOR |
— | 215 |
| Prostaglandin E2 (PGE2) |
Promote bone formation; Enhance bone resorption |
C20H32O5 |
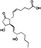
|
AA metabolites |
Osteoblasts Osteoclasts Osteocytes Chondrocytes |
EP2, EP4 | RANKL |
Periodontitis; Bone fracture |
276 |
Long‐chain monounsaturated fatty acids (LCMUFAs) such as ω‐5, ω‐7 and ω‐9 categories are commonly recognized as potential agents against osteoporosis and other osteolytic diseases. They promote bone formation and inhibit bone degeneration and thereby facilitate bone metabolism. By contrast, long‐chain saturated fatty acids (LCSFAs) might negatively affect bone metabolism. Intake of common dietary SFAs such as lauric acid (LA, C12:0), myristic acid (MA, C14:0), palmitic acid (PA, C16:0) or stearic acid (SA, C18:0) might initiate inflammatory osteoarthritis and obesity.25, 26, 27 Moreover, medium‐chain fatty acids (MCFAs) such as capric acid (CA) have been reported to suppress osteoclastogenesis and thereby alleviate bone resorption. Short‐chain fatty acids (SCFAs)28, 29, 30 including acetate, butyrate and propionate have been suggested to inhibit bone resorption and combat inflammation. As a result, SCFAs are promising in the prevention of inflammatory bone loss and arthritis. Furthermore, fatty acid derivatives such as lipoxin A4 (LXA4) and resolvin E1 (RvE1) have also been involved in bone resorption attenuation. Therefore, considering large quantities of fatty acids in our daily diets, it is worthwhile to understand influences of fatty acids on bone metabolism and the underlying mechanisms, for further exploring their beneficial therapeutic applications in a wide variety of metabolic bone disorders.
2. IMPLICATIONS OF FATTY ACIDS IN BONE DISEASES
2.1. Periodontitis
Periodontitis is a chronic bacterial infection disease characterized by primary gingival and extended alveolar bone inflammation, accompanied by periodontal tissue damage 31, 32 with connective tissue degradation and even tooth loss.33 Consistent links between fatty acids and periodontitis have been established by evidence derived from animal and human subjects. Investigations in animal models from different groups such as Bendyk et al 34 and Azuma et al 35, 36 come to conclusions that tissue levels of ω‐3 LCPUFAs is inversely associated with periodontic alveolar bone loss,34 and ω‐3 LCPUFAs EPA metabolite RvE1 is also established to enhance bone formation and reduce bone resorption in rabbit periodontitis models.37 As for the anti‐inflammatory effects, LCMUFAs oleic acid (OA) exhibits anti‐inflammatory potentials to decrease alveolar bone loss, while LCSFAs PA shows contrary effects with elevated tumour necrosis factor α (TNF‐α) levels in obesity mice models,38 suggesting that the anti‐inflammatory potentials of fatty acids in periodontitis might be varied based on specific fatty acids types. In human subjects, increased concentrations of specific SCFAs (lactic acid, propionic acid, butyric acid, isovaleric acid) have been found in the gingival fluid of periodontitis patients, demonstrating a possible association between SCFAs and inflammatory alveolar bone loss.39, 40 Moreover, a preliminary clinical study by El‐Sharkawy et al suggests that dietary supplementation of ω‐3 LCPUFAs might have therapeutic values against periodontitis.41, 42, 43, 44, 45 However, there are also clinical investigations report that benefits of dietary ω‐3 LCPUFAs might not be applied to periodontitis prevention and treatment.44, 46
In mechanism, fatty acids might exert effects on periodontitis pathogenesis and intervention via direct and indirect mechanisms. Fatty acids could directly affect periodontitis‐associated bone destruction. LCSFAs such as PA could trigger P gingivalis‐induced alveolar bone loss directly.31 In benefit, EPA metabolite RvE1 could target BLT1 receptors in osteoclasts to inhibit osteoclast fusion and maturation, and RvE1 can induce the release of osteoprotegerin (OPG) to antagonize the proresorptive role of osteoclast‐stimulating receptor activator of nuclear kappa‐β ligand (RANKL), and thus facilitates the prevention of alveolar bone loss and enhances periodontal bone regeneration in periodontitis patients.47 The indirect effects of fatty acids in periodontitis are mainly through inflammatory response. Studies have shown that LCSFAs (such as PA) at high levels in plasma may facilitate P gingivalis‐induced chemokine production in human gingival fibroblasts and further promote inflammatory response in periodontium.31 PA‐induced chemokine secretion in human gingival fibroblasts could be inhibited by LCPUFAs (such as DHA), and such effects presumably involving the suppression of toll‐like receptor (TLR) dimerization as well as nuclear factor‐kappa B (NF‐κB) activation.48 In addition to exert effects on chemokine, fatty acids such as RvE1 could also act on inflammatory cells by enhancing the migration of monocytes and neutrophils and promoting the clearance of apoptotic neutrophils to enhance pro‐inflammatory response.31 Last but not the least, ω‐3 fatty acids such as DHA and EPA exhibit extensive antibacterial effects against putative periodontal pathogens including F nucleatum and P gingivalis, and SCFA butyrate derived from anaerobic bacterial metabolism could inhibit the differentiation of gingival fibroblasts to promote chronic periodontitis.49 Given that refractory periodontitis significantly decreases the life quality of patients, studies investigating the interaction between fatty acids and periodontitis are required to develop novel intervention strategies.
2.2. Osteoporosis
Osteoporosis, marked by low bone mineral density (BMD) and deteriorated bone tissue microarchitecture, contributes to a high incidence of bone fracture on average up to 50% of women > 50 years.50 Osteoporosis is mainly caused by excessive bone resorption resulting from imbalance between overactive osteoclasts and inactive osteoblasts.51, 52 Hence, inhibiting bone resorption or promoting bone formation are promising strategies for osteoporosis prevention and treatment.53 It has been well acknowledged that osteoporosis is associated with levels of fatty acid in bone microenvironment.54 As reviewed earlier by Salari et al,55 investigations conducted in humans have shown inconsistent correlations between fatty acids and osteoporosis, while studies in animal models have confirmed that supplementation of ω‐3 LCPUFAs alleviates osteoporosis by suppressing bone breakdown, promoting calcium absorption from diet, reducing prostaglandin E2 (PGE2) production and increasing skeletal calcium.56 In mechanism, ω‐6 LCPUFAs intake results in a high ratio of ω‐6 versus ω‐3 LCPUFAs, and thus facilitating osteoporosis by promoting low‐grade chronic inflammation and regulating MSC lineage commitment.57 ω‐3 LCPUFAs inhibit osteoclastogenesis, decrease PGE2 content, and thus increasing BMD to benefit osteoporosis prevention and alleviation.51, 58 In addition, fatty acids such as palmitate could enhance energy generation for osteoblast differentiation, thus accelerating bone formation.52 Moreover, since LCPUFAs are highly prone to reactive oxygen species (ROS)‐induced oxidative damage, adoption of antioxidant CoQ as adjuvant could eliminate the disadvantages of LCPUFAs during osteoporosis therapeutics.59, 60
Estrogen deficiency‐induced postmenopausal osteoporosis is the most common type of osteoporosis. Along with decrease in estrogen levels, reduction in OPG delays osteoblast maturation and attenuates bone formation61; also, drop in OPG/RANKL ratio enhances osteoclast differentiation and promotes bone resorption and eventually results in bone loss.47 Moreover, endogenous fatty acids could serve as energy sources of skeletal and bone marrow cells to contribute to postmenopausal women bone health,62 while exogenous supply of fatty acids might favour or harm postmenopausal women bone condition. As illustrated by animal models, supplementation of fatty acids such as and SCFAs63 and ω‐3 LCPUFAs56 substantially reduces bone loss and restores bone mass and thus ameliorates postmenopausal bone loss in ovariectomized mice, and the protective roles of SCFAs on bone loss were mainly attributed to the suppression of osteoclast differentiation and function.64 According to data derived from human subjects, although earlier investigations indicate that ω‐3 LCPUFAs intake plays positive roles in enhancing bone mass and limiting postmenopausal osteoporosis risks,65 effects of PUFAs on bone are shown to be contradictory in general. For example, there is one study demonstrated that PUFA supplementation significantly enhanced lumbar spine and femoral neck BMD in a population of 65 postmenopausal women; however, another trial reported no significant therapeutic effects in 42 postmenopausal women receiving similar PUFA supplements.56 Taken together, understanding the functions and mechanisms of fatty acids in osteoporosis might help to develop novel preventive or therapeutic strategies to benefit bone health maintenance in osteoporotic patients.60, 66
2.3. Bone fracture
The high risk of bone fractures may result from osteoporosis with low BMD, or more specifically, deterioration of bone structure and loss of bone mass.67, 68, 69 Studies in mice models have suggested that endogenously produced ω‐3 LCPUFAs could facilitate fracture healing process, and supplementation of ω‐3 LCPUFAs exert positive effects on fracture healing.21 Consistently, investigations in human subjects by Sadeghi et al 68 and Harris et al 67 have indicated that increased intake of total PUFAs is positively correlated with higher BMD and reduced bone fracture risk in populations including elder men. However, epidemiological investigation by Virtanen et al demonstrates that low total PUFA, ω‐6 PUFA or LA intakes might promote the risk of hip fractures in women.70 Apart from heterogeneity in study design, sample inclusion and data process among different studies, diversity in fatty acid types might be an important factor contributing to the conflicting involvement of fatty acids in bone fractures. Correspondingly, specific mechanisms of fatty acid modulation on bone fractures vary a lot. For example, ω‐6 LCPUFAs such as arachidonic acid (AA) could stimulate PGE2 production to regulate bone metabolism and fracture healing, while ω‐3 PUFAs increase BMD by increasing calcium resorption and bone collagen synthesis, decreasing urinary calcium excretion, and thus inhibiting bone resorption.67, 68 Overall, fatty acids of different types might exert differential effects on bone fractures pathophysiology, and much more work needs to be done on exploiting them for bone fractures prevention and therapeutics.
2.4. Rheumatoid arthritis
Rheumatoid arthritis, with manifestations of arthralgia, redness and swelling, and limited range of motion,71 is a chronic and autoimmune inflammatory disease affecting 0.5%−1% of the world population.72, 73, 74 If left untreated or ineffectively treated, rheumatoid arthritis typically leads to primary joints destruction caused by erosion of cartilage and bone, as well as subsequent systemic complications and even death.72, 73, 74, 75 Several studies have investigated the individual and combinational protective effects of LCPUFAs in rheumatoid arthritis. For example, ω‐3 LCPUFAs could lower the risk of cardiovascular disease in rheumatoid arthritis patients,23 and combinational utilization of ω‐3 LCPUFAs with low‐dose vitamin E could substantially reduce the side effects of disease‐modifying anti‐rheumatic drugs (DMARDs).75 The attenuation effects of ω‐3 LCPUFAs on rheumatoid arthritis‐induced bone and cartilage destruction are mainly mediated by reduced synthesis of cartilage‐degrading enzymes as well as the inflammatory response cytokines. ω‐3 LCPUFAs, especially EPA and DHA,23, 76 could alleviate morning stiffness and decrease number of swollen and tender joints in patients with rheumatoid arthritis and thus show anti‐inflammatory and restorative effects against rheumatoid arthritis. Importantly, since LCPUFAs AA could drive the synthesis of pro‐inflammatory cytokines, restriction of AA enhances ω‐3 LCPUFAs‐mediated anti‐inflammatory responses by decreasing the production of metalloproteinases and pro‐inflammatory cytokines as well as the migration of leucocytes in vivo, and thus strengthens the action of ω‐3 LCPUFAs in combating rheumatoid arthritis.75, 76 Another kind of LCPUFAs, ω‐6 LCPUFAs are eventually metabolized into AA and inflammatory eicosanoids and function as pro‐inflammatory agents,75, 77 ω‐3 LCPUFAs could reduce the synthesis of ω‐6 LCPUFAs by competing with the rate‐limiting delta‐6 desaturation enzyme and thus exert a therapeutic effect on rheumatoid arthritis.75, 78, 79 Moreover, SCFAs also play crucial roles in bone metabolism and immune responses in pathological bone loss and thus regulate systemic bone mass and protect from rheumatoid arthritis.64 Investigations are needed to further elucidate mechanisms underlying the pharmacological roles and therapeutic potentials of multiple types of fatty acids in arthritis such as temporomandibular joint arthritis.66
2.5. Tumour‐associated bone destruction
Multiple myeloma is a destructive cancer that mainly occurs in bone marrow.80 Studies have shown that fatty acids of different types play either pro‐death or pro‐survival roles in multiple myeloma. For example, PA could activate apoptosis in multiple myeloma cells and thereby serves as a potentially direct anti‐myeloma strategy.81 EPA and DHA could also initiate apoptosis and promote drug sensitivity in multiple myeloma cells, with a mechanism involving NF‐κB inhibition concomitant with activation of mitochondrial defects leading to caspase‐3 activation and apoptosis.82 In addition, EPA and DHA modulate p53/miR‐34a/Bcl‐2 axis to enhance dexamethasone (Dex)‐sensitivity in multiple myeloma cells where they trigger p53 expression and subsequent increase of miR‐34a levels in U266 cells, and finally activate Bcl‐2 to induce apoptosis of multiple myeloma cells.83, 84, 85 By contrast, SFAs and ω‐6 LCPUFAs represent energy sources for multiple myeloma cells, and ratio of ω‐3/ω‐6 fatty acid intake is critical for the maintenance of multiple myeloma cell survival.86, 87
Bone metastasis is a pernicious complication88 occurring in virtually 60% of patients with osteolytic breast or osteogenic prostate cancers and at a smaller rate in patients with other cancer types.89, 90 Patients with bone metastasis suffer from severe pain, bone fracture and osteolytic lesions, which symptoms are primarily attributed to aberrant bone resorption mediated by osteoclasts.91, 92 In osteolytic metastasis mice model originating from MDA‐MB‐231 human breast cancer, researchers found that supplementation with DHA and EPA‐enriched fish oil prevented breast cancer metastasis‐induced bone osteolysis,93 suggesting potential therapeutic effects of fatty acids for osteolytic bone metastasis. In mechanism, both DHA and EPA reduce the mRNA and protein levels of CD44 in breast cancer cells to inhibit cancer invasion; moreover, compared to EPA, DHA has profound anti‐inflammatory effects via inhibiting TNF‐α secretion and NF‐κB activation in macrophages and thus exhibits stronger suppression of osteoclast activity to attenuate the related osteolysis.94 However, in osteogenic metastasis derived from prostate cancer, fatty acids such as AA could facilitate metastatic cancer cell implantation and propagation via preparation of bone microenvironment “soil” for cancer cells by activating bone marrow adipocyte formation,95 demonstrating promotional roles of fatty acids in favour of osteogenic bone metastasis. This might be explained by the fact that fatty acids synthesized by bone marrow adipocytes could serve as energy source for certain types of tumour cells engaged in metastasis. During bone metastasis of prostate cancer, free fatty acid influx into cells induces the expression of lipid transport mediator fatty acid‐binding protein (FABP4), and expression of FABP4 between tumour cells and adipocytes could mediate adipocyte‐induced metabolic switch in prostate microenvironment and thus promotes osteogenic prostate cancer metastasis.96 Such roles of fatty acids in facilitating bone metastasis have also been confirmed in melanoma cancer,89 where bone marrow adipocytes play a pivotal role in bone metastasis by releasing free fatty acids to meet the energy demands of metastatic cancer cells for survival and growth. Therefore, fatty acids of different types behave significantly differently in cancer bone metastasis, and osteolytic or osteogenic or mixed bone lesion conditions derived from specific cancer types should be definitely taken into account when employing fatty acids for cancer bone metastasis therapeutics.
2.6. Other bone disorders
Fatty acids are also involved in non‐typical skeletal diseases such as osteomyelitis, a bone inflammatory process initiated by infection of pyogenic organisms 97 that predominantly occurs in long bones of children, and in hips, feet, jaws and spine of adults.98, 99, 100 This disease is characterized by severe damage to bone tissue and bone marrow, and probably accompanied by high morbidity and mortality.100 Accumulating evidence has shown that ω‐3 LCPUFAs could effectively combat microbial pathogenesis in osteomyelitis.101, 102, 103 Furthermore, combination of vancomycin and ω‐3 LCPUFAs has been suggested to be a reliable therapeutic strategy against S aureus‐induced osteomyelitis, with a mechanism involving inflammation alleviation by reducing TNF‐α and interleukin 6 (IL‐6) levels as well as antioxidant activity by decreasing SOD activity.97
Taken together, according to currently available pre‐clinical experiments (Table 2) and clinical studies (Table 3), various factors contribute to implications of different fatty acids types in multiple bone disorders. With most associations between fatty acids and bone disorders remain obscure (Figure 2), much more work needs to be done by collaboration of biological and clinical researchers to maximize the therapeutic potentials and minimize the side effects of fatty acids against bone diseases.
Table 2.
Animal experimental studies evaluating effects of fatty acids in bone disorders
| Class | Disease | Animal | Treatment | Study period | Bone‐related outcome | Conclusion | Year | Reference |
|---|---|---|---|---|---|---|---|---|
| ω‐3 LCPUFAs | Periodontitis | Male Wistar rats |
Control group (C) Group 1:ω‐3 PUFAs (C + O) Group 2: pulp exposure‐induced apical periodontitis (AP) Group 3: pulp exposure‐induced AP + ω‐3 PUFAs (AP + O) |
45d | Areas of bone resorption/inflammatory intensity :AP group > AP + O, C + O and C groups | ω‐3 LCPUFAs decrease inflammatory cell infiltration and AP bone resorption | 2018 | 35 |
| Osteoporosis | Male piglets |
Diet: suckling/standard formula/formula containing LCPUFAs Drug: placebo/ dexamethasone (DEX) |
15d |
DEX group: BMC of whole body, femur, and lumbar spine ↓ Suckled group: highest BMC of femur and whole body LCPUFA group: PGE2↑ |
ω‐3 LCPUFAs give rise to BMC of femur and whole body | 2002 | 277 | |
| Osteoporosis |
c57Bl/6 mice |
High‐fat diet/normal control | 24w |
Trabecula number and surface↑ Trabecular separation↓ |
HFD‐induced obesity promotes bone formation | 2010 | 278 | |
| Osteoporosis | Fat‐1 mice |
Group 1: Fat‐1 mice sham Group 2: Fat‐1 mice ovariectomized (OVX) Group 3: WT sham Group 4: OVX |
5mo |
Bone marrow adiposity↓ Bone parameters↑in the distal femoral metaphysis |
ω‐3 LCPUFAs improve osteoblastogenesis to treat osteoporosis |
2013 | 279 | |
| Osteoporosis | Fat‐1 mice |
Ovariectomized (Ovx) and sham operated AIN‐93M diet containing 10% corn oil |
24w |
Osteotropic factors↓ BMD↑ |
ω‐3 LCPUFAs effectively prevent post‐menopausal osteoporosis | 2009 | 280 | |
| Aging‐related bone loss | Gonad‐intact middle‐aged male rats |
Group 1: ω‐6 + ω‐3 diet (control) Group 2: ω‐6 diet (almost devoid of ω‐3 LCPUFA) Group 3: ω‐3 diet (rich in ω‐3 LCPUFA) |
20w |
Group 1:BMD↓ Group 2: bone PGE2 production↑ Group 3: bone‐specific alkaline phosphatase activity↑ + highest bone mineral and BMD |
ω‐3 LCPUFAs protect gonad‐intact middle‐aged male rats from bone loss |
2005 | 281 | |
| Aging‐related bone loss | Male Wistar rats | Diet: virgin olive oil/ sunflower oil/ (ω‐6 LCPUFAs)/ fish oil (ω‐3 LCPUFAs) | 24mo |
Bone loss:sunflower oil (+++) fish oil (++) virgin olive oil (+) |
Dietary ω‐3 LCPUFAs prevent aging‐associated bone loss ω‐6 LCPUFAs prevent aging‐related alveolar bone loss |
2013 | 282 | |
| Aging‐related bone loss |
Female Polycystic kidney disease (PKD) rats |
Group 1:casein + corn oil (Casein + CO) Group 2: casein + soybean oil (Casein + SO) Group 3: soy protein isolate + soybean oil (SPI + SO) Group 4: soy protein isolate + 1:1 soybean oil:salmon oil blend (SPI + SB) |
12w | Femur length: SPI + SO < Casein + CO | ω‐3 LCPUFAs influence bone longitudinal growth and mineral balance | 2015 | 283 | |
| Aging‐related bone loss | Male Wistar rats |
Group 1: fish oil Group 2: fish oil + coenzyme Q10 (CoQ10) |
24mo |
Aged rats bone mineral density: group 1<group 2 | CoQ10 avoids aging‐related bone loss | 2017 | 59 | |
| Osteoarthritis | Guinea pigs | High ω‐3 diet/typical western diet | 20w | ω‐3 diet group: OA average histological scores↓; cartilage parameters modified | ω‐3 LCPUFAs decrease OA in prone strain and increase no marker of pathology in either strain | 2011 | 284 | |
| Bone fracture | Fat‐1 ± C57BL/6 mice | Diets containing 10% corn oil | 12w | Fat‐1 + C57BL/6 mice exhibited acceleration in endochondral ossification, callus formationand remodeling process compared to fat‐1 ‐C57BL/6 mice group | ω‐3 PUFAs positively affect fracture healing | 2017 | 21 | |
| Bone growth | Female white rabbits |
Diet: soy bean oil (SBO control)/sesame oil (SO)/fish oil (FO)/algae oil |
100d |
Bone marrow fatty acids ↑ FO diet:highest ω‐3 LCPUFAs SBO diet:highest ω‐6 LCPUFAs |
ω‐6/ω‐3 LCPUFAs ratios are involved in bone resorption decrease and bone mass improvement during growth | 2014 | 285 | |
| Bone growth | Post‐partum female Wistar rats | Diet: flaxseed flour + semi‐purified diet |
51d |
Bone maximum force/breaking strength/ rigidity/ femoral head radiodensity ↑ | ω‐3 LCPUFAs enhance bone density and bone strength | 2017 | 286 | |
| Cancer bone metastasis | Immune‐compromised (nu/nu) mice | Lab chow diet/fish oil + intracardiac injection of the MDA‐MB‐231 cells | 6w | Fish oil diet group:osteolytic lesions ↓; migration of breast cancer cells↓; CD44 expression ↓ | ω‐3 LCPUFAs prevent breast cancer bone metastasis | 2011 | 93 | |
| Cancer bone metastasis | Female BALB/c mice | ω‐3/ω‐6 LCPUFAs diet + orthotopic implantation of 4T1 mammary tumor cells | 147d | Bone metastases frequencies: ω‐3 group<ω‐6 group | Dietary ω‐3 LCPUFAs reduce tumor metastasis to bone | 2018 | 287 | |
| ω‐6 LCPUFAs |
Periodontitis |
C57BL/6 mice |
Group 1: palmitic acid (PA)‐enriched high‐fat diet Group 2: oleic acid (OA)‐enriched high‐fat diet Group 3: normal caloric diet |
16w |
Weight↑(group 1 and group 2) alveolar bone loss and TNF‐α levels: group 1 (+++) bone remodeling markers: group 3 (+++) group 2 (++) group 1 (+) |
PA aggravates alveolar bone loss and osteoclast inflammation | 2016 | 38 |
| Aging‐related bone loss | Male Wistar rats | Diet: virgin olive oil/sunflower oil/(ω‐6 LCPUFAs)/fish oil (ω‐3 LCPUFAs) | 24mo |
Bone loss:sunflower oil (+++) fish oil (++) virgin olive oil (+) |
ω‐3 PUFAs prevent aging‐related bone loss ω‐6 LCPUFAs are associated with aging‐related alveolar bone loss |
2013 | 282 | |
| Aging‐related bone loss | Male Wistar rats | Diet: virgin olive oil (V group)/ sunflower oil (S group) | 24mo | Bone Mineral Density/Bone Mineral Content/ Bone Areal Size:V group>S group | MUFAs prevent aging‐related BMD decrease | 2017 | 288 | |
| Osteoarthritis | Female mice |
Group 1: C2/C3/C4 supplementation Group 2: fibre‐rich diet Group 3: bacterial transfer |
8w |
Group 1: bone volume per tissue volume↑, trabecular separation↓ Group 2: systemic bone mass↑, trabecular separation↓ Group 3: osteoclast numbers↓, systemic bone mass↓ |
SCFAs regulate bone metabolism to optimize arthritis severity | 2018 | 64 | |
| Osteoarthritis | 8‐week‐old female mice |
Group 1: C2/C3/C4 supplementation Group 2: fibre‐rich diet Group 3: Prevotella transfer into WT mice |
8w |
Group 1: bone volume per tissue volume↑, trabecular separation↓ Group 2: systemic bone mass↑, trabecular separation↓ Group 3: osteoclast numbers↓, systemic bone mass↓ |
SCFAs regulate bone metabolism and immune responses to alleviate arthritis | 2018 | 64 | |
| Partum‐related bone loss |
Wistar rats |
Control goup: placebo Test group: flaxseed flour |
51d |
Test group: arachidonic acid (ARA)↓ alpha‐linolenic acid (ALA) eicosapentaenoic (EPA)↑ femoral head radiodensity↑ |
ALA together with calcium increase bone density in post‐partum period |
2017 | 286 | |
| SFAs | Periodontitis | Male C57BL/6 mice | Diet:regular chow/ high‐fat diet | 16w |
LPS‐induce alveolar bone loss↑ LPS‐induce osteoclastogenesis ↑ |
SFAs are potentially involved in MetS‐related periodontitis | 2015 | 36 |
| Osteoarthritis | Male rats |
H group:20% beef tallow HLA group: 20% lauric acid HAS group: 20% stearic acid |
16w |
Articular cartilage degeneration Bone architecture changes Average osteocyte lacunae↓ |
SFAs prevent OA development | 2017 | 260 | |
| MUFAs | Periodontitis |
C57BL/6 mice |
Group 1: PA‐enriched high‐fat diet Group 2: OA‐enriched high‐fat diet Group 3: normal caloric diet |
16w |
Weight↑(group 1 and group 2) alveolar bone loss and TNF‐α levels: group 1 (+++) bone remodeling markers: group 3 (+++) group 2 (++) group 1 (+) |
OA can aggravate the alveolar bone loss and inflammation of osteoclasts | 2016 | 38 |
Table 3.
Epidemiological and clinical studies evaluating effects of fatty acids in bone disorders
| Class | Disease | Intervention | Study Period | Enrolment | Bone‐related outcome | Conclusion | Year | Reference |
|---|---|---|---|---|---|---|---|---|
| ω‐3 LCPUFAs | Periodontitis |
Control group: placebo Test group: DHA |
3mo | 55 |
IL‐1β↓ mean pocket depth↓ gingival index↓ |
DHA greatly contributes to moderate periodontitis and gingival inflammation | 2014 | 45 |
| Periodontitis |
Group 1: EPA 500 mg Group 2: borage oil 500 mg Group 3: EPA 500 mg and borage oil 5oo mg |
12w | 30 |
Periodontal probing depth and gingival inflammation group 1 (+) group 2 (+++) group 3 (++) |
Borage oil have better influences on periodontal inflammation than EPA | 2003 | 289 | |
| Periodontitis |
Control group: decalcified freeze dried bone allograft (DFDBA) + placebo Test group: DFDBA + omega‐3 polyunsaturated fatty acids combined with low‐dose aspirin o |
6mo | 40 |
Probing pocket depth↓(T) IL‐1b and IL‐10↓(T) |
ω‐3 LCPUFAs combined with low‐dose aspirin decrease gingival inflammation, pocket depth and attachment level gain |
2011 | 290 | |
| Rheumatoid arthritis |
Control group: placebo Test group: daily liquid nutrient supplementation |
4mo | 66 |
EPA, DHA and docosapentaenoic acid↑ arachidonic acid↓ |
EPA and GLA do not benefit RA patients at test doses |
2004 | 291 | |
| Rheumatoid arthritis |
Control group: diet group regarding the fatty acid intake Test group: Mediterranean diet |
52w | 13 |
Ratio of ω‐6 to ω‐3 fatty acids↓ intake of ω‐3 fatty acids↑ |
Revealed by dietary assessments and through fatty acids in s‐phospholipids, the fatty acid profile is different in the Cretan Mediterranean diet | 2005 | 292 | |
| Rheumatoid arthritis |
2 mL/kg fish oil emulsion intra‐ venously |
5mo | 34 |
Short‐term efficacy↑ rapid onset excellent tolerability |
ω‐3 PUFAs are safe and effective for RA | 2006 | 293 | |
| Rheumatoid arthritis |
Drug: cod liver oi Drug: placebo |
9mo | 97 | Daily NSAID requirement↓ |
ω‐3 LCPUFAs decrease NSAID‐ sparing agents |
2008 | 294 | |
| Rheumatoid arthritis |
Control group: placebo Test group: Step 1:0.2g of fish oil emulsion/kg intravenously Step 2:0.05 g of fish oil/kg orally |
14d | 23 |
Swollen joint count↓ Tender joint count↓ |
ω‐3 LCPUFAs improve symptoms of RA and extend the beneficial effects of infusion therapy |
2010 | 295 | |
| Rheumatoid arthritis |
Drug: ω‐3 LCPUFAs Drug: Placebo |
12w | 60 |
Clinical benefit concomitant analgesic medication↓ no weight change |
ω‐3 LCPUFAs decrease use of concomitant analgesic without weight changes | 2015 | 75 | |
| Rheumatoid arthritis | High‐dose/Low‐dose fish oil + disease‐modifying anti‐rheumatic drug(DMARD) | 12mo | 140 | Failure of DMARD therapy ↓ | ω‐3 LCPUFAs increase RA remission and decrease DMARD therapy failure | 2015 | 296 | |
| Rheumatoid arthritis | RA‐free participants at increased risk for RA | 10y | 136 | Percent of ω‐3 LCPUFAs in red blood cells↑→ rheumatoid factor (RF) positivity in shared epitope (SE)‐positive participants↓ | ω‐3 LCPUFAs exert pronounced effects on RA‐related autoimmunity | 2017 | 297 | |
| 2166 | ω‐3 LCPUFAs supplement use↑→RF positivity in SE‐positive participants↓ | |||||||
| ω‐6 LCPUFAs | Periodontitis |
Group 1: EPA 500 mg Group 2: borage oil 500 mg Group 3: EPA 500 mg and borage oil 5oo mg |
12w | 30 |
Periodontal probing depth and gingival inflammation group 1 (+) group 2 (+++) group 3 (++) |
Borage oil has better effects on periodontal inflammation than EPA | 2003 | 289 |
| SCFAs | Periodontitis | Periodontal treatment, | 6mo | 21 |
Levels of formic acid↑ Levels of lactic acid, propionic acid, butyric acid and isovaleric acid↓ |
Formic acid in gingival crevicular fluid is inversely associated with periodontitis severity Butyric and isovelaric acids can indicate development and progression of periodontitis |
2012 | 39 |
Figure 2.
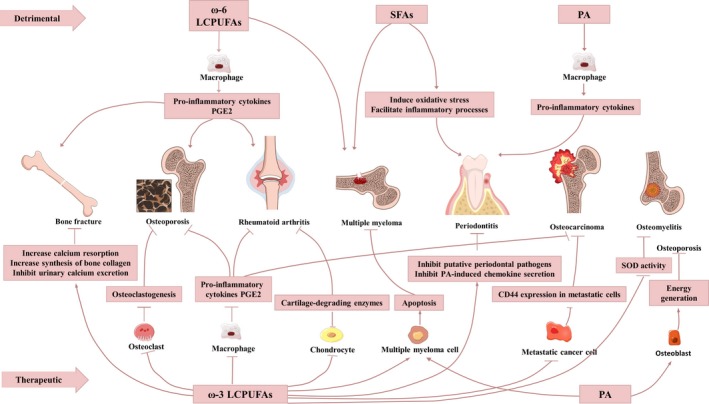
Implications of fatty acids in bone diseases. Fatty acids exert dual effects on bone either by alleviating or initiating bone diseases. ω‐6 LCPUFAs, SFAs and PA predominantly promote pathological bone remodelling by facilitating pro‐inflammatory processes and lead to osteoporosis, rheumatoid arthritis, periodontitis, etc Other fatty acids like ω‐3 LCPUFAs have therapeutic value in blocking bone disorders. Their targets include bone tissue components such as osteoblasts, osteoclasts, macrophages, chondrocytes and aberrant multiple myeloma cells, metastatic cancer cells, suppressing skeletal inflammation, carcinoma and bone fracture through complicated mechanisms. LCPUFAs, long‐chain polyunsaturated fatty acids; SFAs, saturated fatty acids; PA, palmitic acid
3. SIGNALLING PATHYWAYS INVOLVED IN FATTY ACIDS‐MODULATED BONE METABOLISM
Fatty acids and their metabolites could modulate bone metabolism via mechanisms such as inflammation,104 apoptosis,105 autophagy 106 and oxidative stress.104 Normally, fatty acids bind to specific cellular membrane‐bound or nucleus‐located targets, induce subsequent transduction of transmembrane/nucleus‐specific signals, further result in modulation of target gene transcription and protein synthesis and finally contribute to the regulation of cell growth, behaviour and function. Given that a multitude of factors are involved in these processes, understanding the underlying mechanisms will substantially facilitate the nutritional and therapeutic applications of fatty acids in bone homeostasis and disorders.
3.1. Receptors involved in fatty acids‐modulated bone metabolism
Cellular membrane‐bound and nuclear receptors, such as G protein‐coupled receptors (GPRs), peroxisome proliferator‐activated receptors (PPARs), TLRs and receptors for metabolites such as chemokine‐like receptor (ChemR), play essential roles in mediating the effects of fatty acids on bone metabolism.
GPRs are a superfamily of more than 1000 distinct membrane receptors; several GRPs among these have been reported to be modulated by fatty acids. GPR18, GPR41, GPR43 and GPR109A are receptors for SCFAs (C2‐C5) found in both osteoclasts and osteoblasts, where GPR41 could regulate leptin production, while GPR43 is the main receptor in mediating effects of SCFAs on osteoclasts.30, 107, 108 GPR40, which is expressed on osteoclasts and could be activated by medium/long‐chain fatty acids with a chain length of C8‐C22,109, 110, 111 positively affects bone metabolism by downregulating osteoclastogenesis, combating bone loss and protecting cartilage.112, 113, 114 GPR84, whose expression in macrophages and adipocytes could be enhanced under inflammatory conditions, is a receptor for MCFAs (C9‐C14).115, 116, 117 GPR120, which is expressed on osteoblasts and osteoclasts and could be stimulated by long‐chain saturated (C14‐C18) and long‐chain unsaturated fatty acids (C16‐C22),109, 110, 111 has been shown to mediate the anti‐inflammatory effects of DHA in macrophages.118 And GRP120 could enhance ω‐3 LCPUFAs‐induced osteoblastic bone formation by inducing β‐catenin activation and reduce osteoclastic bone resorption by suppressing NF‐κB signalling,14 and GPR120 could also modulate the bi‐potential differentiation of BMMSC in a dose‐dependent manner.119 In addition to the acknowledged roles of GPR40 and GPR120 in preventing bone disorders such as osteoporosis and osteoarthritis,120 GPR2 family member parathyroid hormone type 1 receptor (PTH1R) also plays a role in bone metabolism. PTH1R could mediate ω‐3 LCPUFAs‐induced activation of extracellular signal‐regulated kinases (ERK) to enhance osteoblasts proliferation and differentiation121, 122; moreover, EPA and DHA could act as agonists of PTH1R to attenuate osteoblast apoptosis and promote bone formation.123
PPARs, with known ligands including LCPUFAs and metabolites such as PGE2, are nuclear receptors that regulate lipid metabolism by acting as transcription factors in BMMSCs, osteoblasts and osteoclasts.124, 125, 126, 127, 128 When BMMSCs are exposed to a mixture of palmitic, oleic and linoleic acids, upregulation of PPARs and reduction of Runx2 facilitate differentiation of towards adipocyte‐like cells.129 Influences of PPARs on osteoblasts and osteoclasts depend on specific receptor isoform. Specifically, PPARα/β promotes bone resorption,130 whereas PPARγ is known inhibitors of osteoclastogenesis.131 Roles of PPARγ in osteoblasts are still disputed; it has been shown that conditional deletion of PPARγ in osteoblasts enhances bone mass and increased bone formation by activating mTOR signalling,132 while studies from other groups reported conflicting results.133, 134, 135 Moreover, recent findings have indicated that treatment of multiple myeloma cells with PPARs resulted in apoptotic effects,136 suggesting PPARs might serve as promising therapeutic targets for bone diseases.
TLRs mainly mediate the inflammatory action of fatty acids in bone cells. PA particularly activates TLR2 and induces IL‐1β expression and secretion to promote inflammatory response.137, 138, 139 Binding of SFAs to TLR4 on osteoclasts induces chronic inflammation 140, 141, 142 by enhancing the expression of macrophage inflammatory protein‐1a, which leads to hyperactivation of NF‐κB and subsequent enhancement of osteoclastic activities 143 as well as further decrease in bone size, BMC and BMD.144 Moreover, studies have shown that DHA treatment could block the pro‐inflammatory effects of lauric acid‐induced TLR2/4 activation in Raw264.7 cells,145 suggesting TLRs might be involved in the crosstalk among multiple downstream signalling pathways of different fatty acids types.
ChemR23 can act as chemerin receptor 146, 147 as well as RvE1 receptor in bone tissue cells such as monocytes.148 Binding of RvE1with ChemR23 could prevent inflammation by inhibiting NF‐κB activation,149 enhancing bone formation150 and reducing bone loss via RANKL/OPG ratio modulation,151, 152 while the detailed mechanisms involved in the downstream of Chem23 have yet to be fully elucidated.
3.2. RANK/RANKL/OPG signalling in fatty acids‐modulated bone metabolism
To our knowledge, various signalling pathways including RANKL,153 NF‐κB,154 mitogen‐activated protein kinase (MAPK),155 Wnt,156 Notch,157 Hedgehog,158 transforming growth factor‐β (TGF‐β),155 mTOR 159 and bone morphogenetic protein (BMP)155 are involved in bone metabolism. Among these, RANK/RANKL/OPG signalling is most frequently implicated in bone remodelling via modulation by a wide variety of fatty acids160, 161 (Figure 3). Upon activation, RANK/RANKL/OPG signalling substantially inhibits osteoclastogenesis but enhances osteogenesis via downstream signalling cascades such as MAPK, NF‐κB and phosphatidylinositol 3‐kinase (PI3K)/mTOR.160 Specifically, MAPK signalling162, 163, 164, 165 could be activated by ω‐7 LCMUFAs,166 PA and MCFAs167 and activation of MAPK signalling normally leads to enhanced proliferation of both osteoblasts and chondrocytes.155, 163, 165 By contrast, ALA,168 ω‐7 LCMUFAs166 and MCFAs167 could inhibit NF‐κB cascade, and repression of NF‐κB cascade attenuates osteoclastogenesis by enhancing both cell death and differentiation.154, 165, 169, 170 Moreover, PI3K/mTOR pathway could be downregulated by EPA or LXA4 but upregulated by PA159 and thus involved in BMMSC differentiation, osteoblast function and osteocyte formation during bone metabolism.171, 172, 173, 174, 175, 176, 177
Figure 3.
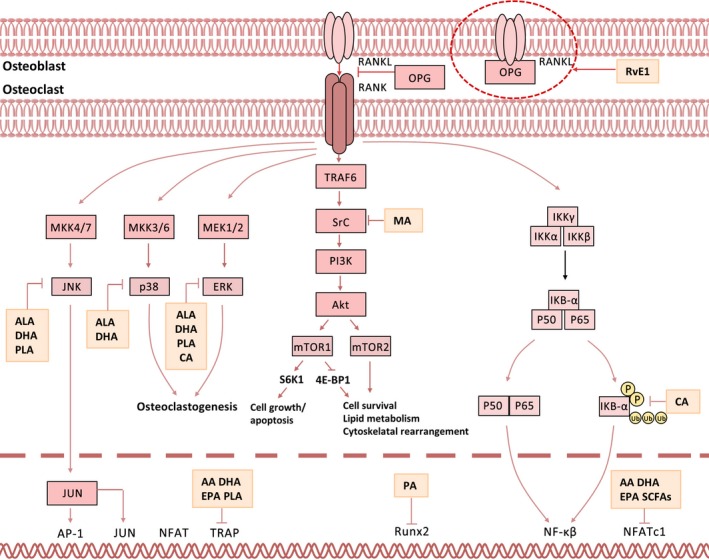
RANK/RANKL/OPG pathway in fatty acids‐modulated bone metabolism. The well‐documented RANKL signalling pathway exerts essential role in osteoclastogenesis. RANKL binds to RANK on the surface of osteoclast precursor cells and activates three distinct downstream signalling pathways. The MAPK pathways characterized by downstream factors ERK, p38 and JNK play pivotal role in cell death and survival. The NF‐κB signalling pathway is activated following IκBα phosphorylation and degradation. The p50 and p65 subunits of NF‐κB are released and translocated into the nucleus to activate the transcription of target genes. The PI3K/mTOR pathway is also activated upon binding of RANKL to RANK, which triggers the activation of PDK1s and Akt leading to the inhibition of the TSC complex and subsequent mTORC1 formation. The mTORC1 phosphorylates S6K1 as well as 4E‐BP1, which further regulate protein synthesis, cell proliferation, angiogenesis and autophagy. However, mTORC2 acts as an essential modulator of actin cytoskeleton, cell survival and lipid metabolism. RANKL, receptor activator of nuclear kappa‐β ligand; TGF‐β, transforming growth factor β; JNK, c‐jun NH2‐terminal kinase; Akt, protein kinase B; S6Ks, S6 kinases; 4E‐BP1, 4E‐binding protein 1
4. MODULATION OF FATTY ACIDS ON SPECIFIC BONE CELL TYPES
4.1. Fatty acids and osteoblasts
Osteoblasts are mononuclear cells predominantly involved in bone formation 4, 5. A growing body of evidence supports the promotional or inhibitory action of fatty acids on osteoblasts. In general, fatty acids such as PA suppress osteoblast function, whereas EPA, DHA and RvE1 predominantly promote osteoblastic function. Exploring the modulation effects of fatty acids on osteoblasts might provide new insights into therapeutic intervention targeting skeletal disorders associated with dysregulated bone formation.
4.1.1. Fatty acids as negative regulators of osteoblasts
Palmitate, a kind of LCSFAs, impedes osteoblast differentiation and induces cell death via lipotoxicity.105 Palmitate could induce autophagy in osteoblasts dependent on Beclin and PI3K,178 and autophagy serves as a protection mechanism in preserving osteoblasts from lipotoxicity.179 Palmitate also promotes apoptosis of osteoblasts through both extrinsic and intrinsic pathways, and PA‐induced high expression of cytosolic cytochrome C could be disrupted by inhibition of c‐Jun N‐terminal kinase (JNK).105 In foetal rat calvarial cell cultures, palmitate affects neither proliferation nor apoptosis of calvarial cells but represses BMP‐7‐induced osteoblastic differentiation by reducing the activity of transcription factor SMAD, and thus further abrogating expression of osteogenic markers Runx2, osteocalcin, alkaline phosphatase and bone sialoprotein.180 Interestingly, enhancing fatty acid oxidation could block all lipotoxic effects of palmitate suggested above, indicating that fatty acid oxidation might relieve the negative effects of palmitate on osteoblasts.105
4.1.2. Fatty acids as positive regulators of osteoblasts
LCPUFAs and SCFAs
ω‐3 LCPUFAs such as EPA and DHA could stimulate osteoblasts survival by activating pro‐survival Akt signal and suppressing glucocorticoid‐induced pro‐death pathway.123 They also promote osteoblastogenesis and prevent bone resorption by altering membrane function, regulating calcium balance and enhancing osteoblast activity.57 Involvement of EPA and DHA in preosteoblasts differentiation and maturation is largely associated with their anti‐inflammatory effects, which function by reducing the synthesis of inflammatory ARA‐derived PGE2,181 modulating PPAR‐γ signalling and thus lower levels of inflammatory cytokines such as IL‐1, IL‐6 and TNF‐α,182 and suppressing AA‐derived synthesis of eicosanoids183 as well as activity of cyclooxygenase and 5‐lipoxygenase.184 Therefore, as illustrated above, intake of EPA and DHA might have potent therapeutic implications in inflammatory bone disorders such as osteoporosis.24
ω‐6 LCPUFAs are activators of PPARγ, and lower dietary ratio of ω‐6/ω‐3 LCPUFAs blocks PPAR‐γ activation and thus enhancing osteoblastogenesis.56 Besides, SCFAs such as butyrate promote osteoblast formation and differentiation by enhancing production of bone sialoprotein and osteopontin; moreover, it stimulates osteoblasts to secret OPG and thus facilitating the blocking of osteoclast differentiation.185
Fatty acids derivatives
RvE1 is an EPA metabolite that is closely associated with inflammation‐induced bone disorders. In IL‐6‐stimulated osteoblasts, supplement of RvE1 leads to significant disruption of PI3K‐Akt pathway, which interacts with NF‐κB, MAPK and p53 signalling to modulate protein synthesis, cell differentiation and apoptosis. In inflammatory bone disorders, changes in production of pro‐inflammatory cytokines such as TNF‐α, IL‐6, IL‐1 and Gas6 186 modulate RANKL/OPG ratio and downstream events151, 152 and thus enhance osteoclasts‐mediated pathological inflammation‐induced bone resorption.
As a metabolite of AA, PGE2 exerts its effects on BMMSCs, osteoblasts and osteoclasts in dose‐dependent manner. In BMMSCs, PGE2 binds with EP4 receptor, which activates sphingosine kinase and inhibits caspases activities and thus prevents apoptosis of BMMSCs.187 In osteoblasts, intermittent administration,188 short‐term exposure to high doses or prolonged treatment with lower doses of PGE2189 could enhance proliferation and activity of osteoblasts and lead to remarkably enhanced bone formation; such influences of PGE2 on osteoblasts are mediated by EP2/EP4‐MAPK signalling pathways.190 In addition, effects of PGE2 on osteoblasts are accompanied by osteoclast stimulation, which might reverse the overall influence of PGE2 on bone system.191
4.2. Fatty acids and osteoclasts
Osteoclasts are multinucleated giant cells with bone resorptive activity. Two essential factors secreted by osteoblasts, macrophage colony‐stimulating factor (M‐CSF) and RANKL, are responsible for osteoclast precursors proliferation and osteoclastogenesis. Importantly, RANKL could prevent apoptosis of osteoclasts3, 4 and induce expression of osteoclast‐specific markers and transcription factors such as nuclear factor of activated T cells c1(NFATc1).4, 6 As bone‐resorbing cells,192, 193, 194, 195 osteoclasts highly express bone resorption‐associated proteins including osteoclast‐specific markers cathepsin K (CTSK), tartrate resistant acid phosphatase (TRAP) and matrix metalloproteinase 9 (MMP‐9).4, 196 Specifically, CTSK breaks down organic components in bone,197, 198 TRAP is implicated in cell adhesion upon activation by CTSK,199, 200 and high levels of MMP‐9 commonly occur in resorption lacunae.201 Multiple fatty acids have been found to promote or suppress osteoclast activity, in most cases via regulation of RANKL signalling. Effects of fatty acids on osteoclast functions demonstrate their potential applications as therapeutic reagents against resorption‐associated bone disorders such as osteoporosis and rheumatoid arthritis.
4.2.1. Fatty acids as positive regulators of osteoclasts
Accumulating evidence has shown that PA enhances RANKL‐mediated differentiation of osteoclasts by upregulating expression levels of RANK; importantly, PA has been reported to be sufficient for osteoclast differentiation in conditions even without RANKL.202
4.2.2. Fatty acids as negative regulators of osteoclasts
LCPUFAs
LCPUFAs such as DHA and AA could exert inhibitory effects on osteoclast proliferation, differentiation and maturation. In mechanism, DHA intervention could inhibit osteoclast precursors proliferation by inhibiting M‐CSF‐induced activation of AKT and expression of cyclin D1/D2, and DHA triggers apoptosis of mature osteoclasts by inducing Bim expression and thus leads to defective osteoclast formation.203 In addition, DHA and AA could regulate migration and adhesion of osteoclasts in bone by downregulating expression of RANK and VNR.204 As for osteoclastogenesis process, LCPUFAs including DHA and AA suppress the expression of osteoclast‐specific genes such as CTSK, TRAP, MMP‐9, NFATc1, c‐Fos and DC‐STAMP in differentiating osteoclasts, thus decreasing osteoclast numbers and bone resorption.110, 205, 206, 207 In detail, DHA and AA bind to TLR4 on cell membrane to suppress TLR4 signalling, MAPK pathways and NF‐κB signalling.208 This further leads to downregulation of c‐Fos205 and NFATc1,209 which is the master regulator for osteoclast proliferation and differentiation. Also, levels of key cell‐to‐cell fusion mediator DC‐STAMP179, 210 are decreased, followed by substantial reduction in osteoclast formation and osteoclast number. Of note, there are certain differences between DHA and AA in combating bone resorption. Specifically, AA displays a more profound effect than DHA in inhibiting osteoclast function at equal concentrations,206, 207 which probably results from a more significant inhibition of CA2 expression205 and further prevention of resorption lacunae acidification with facilitation of CTSK and MMP‐9 enzymatic activities.205 These findings provide molecular mechanisms underlying the benefits of DHA supplement, and intake of high doses of EPA and DHA supplements has been suggested to attenuate bone loss associated with breast cancer.211
And, ALA intervention leads to apoptosis reactivation and RANKL signalling repression in osteoclasts. In mechanism, ALA reduces RANKL‐stimulated phosphorylation of JNK, ERK and AKT together with NF‐κB and BCL‐2 proteins to exert pro‐apoptotic action,212 reduces inflammatory bone loss via downregulating NF‐kB‐iNOS‐COX‐2 signalling axis and further inhibits RANKL‐induced osteoclast differentiation. Moreover, ALA can be converted into downstream fatty acids and several eicosanoids such as DHA and EPA and further exerts more complicated effects on osteoclastogenesis.181
LCMUFAs
Studies have shown that MA could suppress N‐myristoyl‐transferase, a critical enzyme involved in Src myristicylation26 to endoplasmic membrane and further phosphorylation.25 MA‐induced Src inhibition then affects a large number of cytoskeletal changes in osteoclasts, reduces latter stages of osteoclast differentiation and prevents RANKL‐induced bone loss in vivo.25 Such inhibitory effects against osteoclast formation and function suggest MA might serve as a new therapeutic agent against osteolytic bone disorders. In addition, investigations by Heerden et al have suggested that PLA could inhibit RANKL‐induced osteoclast formation and promote apoptosis of mature osteoclasts.166 In mechanism, PLA downregulates the activity of NF‐κB, MAPKs, JNK and ERK, inhibits expression of genes involved in osteoclast activity such as DC‐STAMP and resorption markers CTSK, MMP9 and TRAP and reduces number of TRAP‐positive osteoclasts by repressing actin ring formation and blocking their osteolytic capability,166 suggesting PLA as a potential therapeutic option for bone disorders related to excessive osteoclast formation. Moreover, as stated previously, PA enhances RANKL‐mediated osteoclastogenesis by facilitating expression of TNF‐α and RANK; conversely, OA could increase expression of DGAT1 and intracellular accumulation of triglycerides in osteoclasts to attenuate PA‐induced osteoclastogenesis.202 In addition, OA might facilitate osteogenic differentiation of adipose tissue‐derived stromal cells168 and thus serve as potential bone induction agent.
SCFAs
SCFAs represent useful supplements to inhibit bone resorption and restore bone metabolism balance.64 Among SCFAs, although concentrations of these molecules in vivo are too low to affect bone metabolism, in vitro investigations suggest that butyrate and propionate alone or mixed could inhibit osteoclast differentiation.28 In mechanism, SCFAs exert effects on bone metabolism (mainly inhibit bone resorption) via direct30or indirect64 mechanisms. Directly, SCFAs bind to receptors (GPR18, GPR41, GPR43, GPR109A)30 present on osteoclast precursors; in specific, acetate and propionate show higher affinity for GPR41, while butyrate exerts effects mostly via GPR43 activation.30 Indirectly,64 SCFAs regulate bone mineral absorption by influencing signalling pathways and gene expression. Butyrate and propionate induce metabolic reprogramming of osteoclasts to enhance glycolysis and thus downregulating critical genes in osteoclasts such as TRAF6 and NFATc1.29 In addition, production of SCFAs increases serum IGF‐1 and peripheral serotonin levels, which affects bone metabolism and decreases PTH levels to inhibit bone resorption.28, 63, 213 And, SCFAs might play a role in immunoregulation by modulating inflammatory events to prevent inflammatory bone loss such as arthritis.
MCFAs
CA inhibits RANKL‐modulated osteoclastogenesis in bone marrow‐derived macrophages by preventing M‐CSF and RANKL‐induced cytoskeletal reorganization, suppresses RANKL‐stimulated IκBα phosphorylation and enhanced NF‐κB transcription and diminishes RANKL‐induced NFATc1 activation.214 Moreover, CA could promote apoptosis of mature osteoclasts by initiating Bim expression and inhibiting M‐CSF‐induced ERK activation,214 demonstrating CA treatment represents a potential strategy for amelioration of bone resorption‐associated diseases.214
Fatty acids derivatives
As a metabolite of AA generated by lipoxidase, LXA4 could dose‐dependently reduce levels of ROS, the expression of osteoclast‐specific genes and osteoclast‐related transcription factors and thus attenuate osteoclasts‐mediated bone loss.215 And exposure to EPA‐derived RvE1 could downregulate STAT1 and subsequently attenuate MAPK and NF‐κB signalling,152 which further restore favourable receptor inducer for RANKL/OPG ratio and rescue OPG production, thus regulating osteoclast differentiation.151 Moreover, PGE2 could induce activation of osteoclasts in a dose‐dependent manner. In vivo studies have revealed that continuous treatment of rats with PGE2 results in bone loss owing to increased osteoclasts stimulation, and higher rates of bone resorption compared with bone formation due to longer bone resorption period.188 In vitro, PGE2 has been shown to increase osteoclast size, enhance resorptive pit formation and reduce osteoclast apoptosis.216 In mechanism, PGE2 could trigger osteoclastogenesis in murine bone marrow cultures treated with RANKL and M‐CSF, possibly caused by EP2 and EP4 receptor‐modulated induction of adenylate cyclase,191, 217 and by suppression of OPG and osteoblast‐induced RANKL secretion and enhanced RANK expression in osteoclasts.218, 219
4.3. Fatty acids and BMMSCs
BMMSCs are multipotent cells characterized by surface markers of CD105, CD73, CD90, CD44, CD29 and CD1469 with differential potentials into osteoblasts, chondroblasts and bone marrow adipocytes.7 BMMSCs are critical in maintaining the dynamic homeostasis of bone tissue, and deficiencies of BMMSCs proliferation are correlated with reduced bone mass.220, 221 Various signalling pathways including Wnt, Notch, Hedgehog, TGF‐β and BMP are involved in BMMSCs osteogenesis. Notably, Runx2 plays the most pivotal role in this process by promoting expression of osteogenesis‐related genes, regulating cell cycle progression and improving bone microenvironments.8
4.3.1. Fatty acids as positive activators of BMMSCs
DHA, a special lipid component of osteoblast membrane, has been reported to fuel wide lipidomic remodelling of BMMSCs. DHA supplementation enhances Akt activation at plasma membrane and thereby potentiates osteogenic differentiation.222 Long‐term and high‐dose treatment of inflammatory diseases with Dex facilitates apoptosis of BMMSCs, leading to bone loss and associated metabolic bone diseases.223, 224 These effects can be eliminated by EPA via activating autophagy and suppressing apoptosis of BMMSCs. More specifically in the case of Dex‐induced apoptosis, activation of GPR120 by EPA triggers Ras‐Erk1/2 cascade, leading to suppression of Dex‐induced apoptosis, accompanied by activation of AMPK/mTOR to initiate autophagy.223, 224 Interestingly, EPA treatment in the absence of Dex has limited effects on autophagy induction,223, 224 demonstrating potential therapeutic role of EPA in managing long‐term side effects of Dex abuse.225
Oleate inhibits palmitate (palm)‐induced apoptosis and increases BMMSCs proliferation.27 Palm has been shown to induce lipotoxicity, whereas oleate fully neutralizes palm‐induced lipotoxicity and pro‐inflammatory response. Oleate exhibits cytoprotective effects by deactivating palm‐induced pathways and fostering esterification of Palm into triglycerides.226 More specifically, Ole inhibits palm‐induced activation of ERK and NF‐κB signalling, which results in pro‐apoptotic effects in BMMSCs.226, 227 Also, decline in IL‐6 and IL‐8 expression and secretion levels by Ole treatment was also observed.228 Furthermore, Ole maintains the oxidative levels of palmitate.27 Hence, OA represents a potential therapeutic agent in combating PA‐induced lipotoxicity in the bone.
4.3.2. Fatty acids as negative regulators of BMMSCs
As mentioned above, palmitate triggers BMMSCs apoptosis and reduces their proliferation.27 Gillet et al have reported that palmitate exerts cytotoxic effects by inducing endoplasmic reticulum stress and activating NF‐κB and ERK signalling pathways, thus further regulating secretion of cytokines and chemokines in BMMSCs and inducing binding of exogenous ligands to TLRs. Moreover, palmitate triggers pro‐inflammatory responses via upregulating TLR4 expression accompanied with enhanced expression and secretion of IL‐6 and IL‐8, whose overproduction facilitates differentiation of osteoclast precursor cells into mature osteoclasts and results in impaired bone formation and enhanced bone resorption.229, 230, 231, 232 And undifferentiated BMMSCs have been found to be less sensitive to lipotoxicity than BMMSC‐derived osteoblastic cells.226
4.4. Fatty acids and osteocytes
Osteocytes are osteoblast‐derived cells located in lacunae surrounded by mineralized bone matrix, with the ability to support bone structure and receive machine sensation. Importantly, osteocytes can serve as endocrine cells to synthesize and express important regulatory molecules including RANKL, Dickkopf‐1 (DKK1) and sclerostin (SOST)233, 234, 235 and thus participating in bone resorption and formation regulation by coupling osteoclast and osteoblast activities.6 Studies have shown that fatty acids such as PA and PGE2 have noteworthy influences on osteocyte metabolism, which might provide novel therapeutic strategies for bone diseases like osteoporosis.
4.4.1. Fatty acids in osteocytes‐mediated bone metabolism
PGE2 released by osteocytes are important regulators of bone formation. For example, PGE2 produced by low‐intensity pulsed ultrasound‐stimulated osteocytes could enhance osteoblasts differentiation but inhibit their proliferation in vitro.236 In addition, mechanical loading or fluid flow shear stress on osteocytes can release PGE2 to regulate osteoblast proliferation and differentiation.237 In mechanism, loading‐induced PGE2 can activate EP2/EP4 receptors to stimulate downstream PI3K/Akt pathway,238 which further facilitates gap junction communication by transcriptional regulation of Cx43 to promote osteocytes survival.239 And PGE2 can activate MAPK and subsequently induce phosphorylation of Cx43 at S279/282 and closure of Cx43 hemichannels, which thus modulating bone anabolism and protecting osteocytes from harmful effects caused by sustained hemichannels opening.239 Moreover, PGE2 could promote production of 8‐nitro‐cGMP in osteocytes to enhance osteoclasts differentiation.240
4.4.2. Fatty acids in osteocytes‐associated bone disorders
Investigations have suggested that PA can cause lipotoxicity in osteocytes. PA results in apoptosis and inhibits survival in osteocytes by induction of autophagy failure, which is indicated by conspicuous increase in LC3‐II and reduction of autophagosomes/lysosomes in cytoplasm.234 In addition, PA exerts effects on bone turnover by decreasing expression of DKK1, RANKL and sclerostin in osteocytes.234 Given osteocytes apoptosis and dysfunction are two common changes in osteoporotic bone, PA might play a part in the pathogenesis as well as potential therapeutic applications in osteoporosis. In addition, fatty acids oxidation can serve as energy source for osteocytes.241 In vivo evidence has shown that fatty acid oxidation could compensate dysfunction of energy metabolism and osteocytes formation caused by glucose transporter‐4 deficiency in osteoblasts and osteocytes of mice.242 Importantly, activation of β‐catenin regulated by Wnt‐Lrp5 signalling affects oxidative potential and fatty acids utilization in osteocytes and thus is responsible for expression of key enzymes during fatty acid oxidation.241 Therefore, fatty acid oxidation in osteocytes exerts regulatory effects on bone fat and body mass, which might have regulatory roles and therapeutic applications in metabolic disease‐associated bone disorders.
4.5. Fatty acids and chondrocytes
Chondrocytes is the main cartilage cell type existing in cartilaginous interstitium and cartilage lacuna, and they can produce cartilage extra cellular matrix that composed mainly of proteoglycans and collagen.243 Fatty acids are integrated into chondrocytes mainly in the form of phosphatidylcholine and triacylglycerols and then mediate downstream signalling pathways via receptors expressed on chondrocytes membrane such as GPR40, GPR120, CD36 and TLR4, as well as a few LRP and PPAR family members.12, 244 As an energy source for chondrocytes, fatty acids participate in chondrocytes energy metabolism245 and further alleviate or enhance chondrocytes damage and cartilage degeneration via multiple mechanisms. Understanding the regulation effects of fatty acids in chondrocytes might help to explore their potential therapeutic values for bone disorders associated with chondrocytes inflammation and cartilage degeneration.
4.5.1. Fatty acids as positive regulators of chondrocytes
ω‐3 PUFAs and metabolites
EPA plays anti‐inflammatory roles by competitively suppressing AA oxidation pathway,246 and EPA treatment could delay IL‐α‐induced chondrocyte death.247 In addition, EPA treatment could inhibit oxidative stress‐induced chondrocyte apoptosis via poly (ADP‐ribose) polymerase and caspase 3 cleavage, p38 MAPK, p53 phosphorylation and MMPs expression and thus ameliorating cartilage degeneration.248 p38 MAPK‐dependent mechanism is also involved in DHA‐involved alleviation of cartilage damage.249
EPA and DHA can be converted to SPM and novel bioactive lipid mediators such as resolvins in vivo.250 Articular chondrocytes could participate in SPM metabolism by expressing biosynthetic enzymes like15‐LO type 1,251 and SPM exhibits a more potent anti‐inflammatory effect than their precursors in protecting chondrocytes and cartilage.12, 250 As for resolvins, resolvin D1 demonstrated anti‐arthritic nature in a model of inflammatory arthritis indicated by significantly attenuated arthritic score and hind paw oedema and reduced leucocytes infiltration within paw.252
Resolvin D3 also shows similar effect on arthritis model.253 In mechanism, investigations by Benabdoune et al in an experimental osteoarthritis in human chondrocytes have found that RvD1 inhibits IL‐1β‐induced COX2, PGE2, inducible NO and MMP‐13 by stifling IL‐1β‐induced activation of p38/MAPK, JNK1/2 and NF‐κB/p65.254 Moreover, resolvin D1 could maintain cartilage integrity in inflammatory arthritis by stimulating the production of chondrocytes extracellular matrix and inhibiting IL‐1β‐induced cells degradation via direct ALX/FPR2 receptor ligation.252 And, resolvin D1 could reduce 4‐hydroxynonenal‐induced oxidative stress and chondrocytes apoptosis.254 These findings suggest that it is promising to develop novel therapeutic strategies based on the functional mechanisms of SPM for the therapeutics of chondrocyte‐related diseases such as osteoarthritis.251
AA derivatives
As epoxide metabolites of AA, epoxyeicosatrienoic acids (EETs) have been reported to reduce inflammatory cytokines such as TNF‐α and IL‐6 and decrease cytotoxicity in canine chondrocytes. However, since EETs could be rapidly metabolized into corresponding vicinal diols by soluble epoxide hydrolase (sEH), sEH inhibitors that are able to stabilize anti‐inflammatory EETs might have therapeutic potentials for chondrocytes survival and cartilage protection.255
SCFAs
Butyric acid and butyrate can reduce cartilage destruction mainly by inhibiting inflammation and MMPs expression. Studies have shown that in human chondrocytes, butyric acid could dose‐dependently suppress IL‐1β‐induced PGE2 synthesis as well as TNF‐α/IL‐17‐induced PGE2 production, with a mechanism involving COX‐2 expression inhibition.256 And butyric acid can reduce the release of IL‐1β‐induced proteoglycan from cartilage explants.256 Butyrate could inhibit the production of key MMPs in chondrocytes via pro‐inflammatory cytokines at both mRNA and protein levels, which further potently inhibit cartilage collagen breakdown.257 Moreover, sodium butyrate markedly inhibits IL‐1β‐induced expression of MMPs and ADAMTSs by suppressing phosphorylation of IκBα, NF‐κB p65 and IKK to abolish inflammatory NF‐κB activation.258 Importantly, GPR43 receptor is greatly relevant to efficacy of butyrate in inhibiting IL‐1β‐induced inflammation in chondrocytes and its chemoattractant effects.259
4.5.2. Fatty acids as negative regulators of chondrocytes
SFA and its metabolites
Several studies have shown that animals fed with high‐SFAs diet exhibit accelerated cartilage degeneration,260 and long‐chain SFAs are considered as important negative regulators of chondrocyte metabolism. Studies have shown that BMMSCs and adipose stem cells‐derived chondrocytes which generate long‐chain SFAs have decreased cartilaginous matrix production,261 and SFAs with different chain lengths might exert relative effects in chondrocytes. It has been found that diet rich in longer chain SFAs such as PA and SA promotes more expression of collagenase‐10 and MMP‐13 and increases much more chondrocyte apoptosis than diet rich in shorter chain SFAs.260
PA and SA have been reported to participate in inflammatory reactions by augmenting pro‐inflammatory markers such as IL‐6 in human chondrocytes.262 In primary mouse chondrocytes, SA could promote lactate dehydrogenase‐dependent production of lactate to stabilize HIF1α protein and facilitate pro‐inflammatory cytokines expression263 SA‐stimulated NF‐κβ p65 activation and pro‐inflammatory cytokines expression in chondrocytes could be attenuated by miRNA‐26a; conversely, NF‐κB p65 could also inhibit miRNA‐26a production by directly targeting the promoter region of miRNA‐26a.264 In addition, PA and SA treatment could enhance autophagy activation in chondrocytes, which is strongly associated with increased activation of NF‐κB signalling pathway,265 while opposite effects have been observed upon LA stimulation.
Palmitate has been reported to synergize with IL‐1β to induce caspase activation and chondrocyte apoptosis, as well as increase expression of cyclooxygenase 2 and IL‐6 in chondrocytes via TLR‐4 signalling, which are all involved in the pathological processes of cartilage destruction.266, 267 Lipotoxicity of palmitate could also be mediated by endoplasmic reticulum (ER) stress and further suppresses IGF‐1‐mediated signalling and succedent proteoglycans and collagen type II synthesis in chondrocytes.268 And utilization of either JNK inhibitor or small molecule chemical chaperone could weaken the effect of palmitate to facilitate cartilage matrix synthesis and chondrocytes survival.269, 270 Importantly, palmitate‐induced ER stress could activate unfolded protein response signalling and subsequently promote apoptosis of meniscus cells to affect the development of obesity‐related osteoarthritis.271 Moreover, in human chondrocytes, palmitate could induce expression of pro‐apoptotic molecules such as cleaved caspase‐3 (CC3) and negative cell survival regulators such as tribbles related protein 3 (TRB3) and nuclear protein 1 (Nupr1) and thus induces apoptosis of chondrocytes.272 Taken together, palmitate has potent therapeutic implications for inflammatory bone diseases such as osteoarthritis.
ω‐6 PUFAs and their metabolites
A growing body of evidence has shown that a higher ratio of ω‐6‐to‐ω‐3 PUFAs might exert negative influences on cartilage.273 As for specific mechanisms, ω‐6 PUFAs such as ALA and AA aggravate cartilage damage by serving as precursors for pro‐inflammatory prostanoids, while ω‐3 PUFAs such as EPA and DHA protect cartilage by being metabolized to anti‐inflammatory mediators such as protectins and resolvins.273, 274 Moreover, AA‐derived PGE2 could serve as important inflammatory mediator to regulate inflammatory reactions of chondrocytes. Studies have shown that PGE2 could suppress differentiation of chondrocytes by activating downstream receptors protein kinase A (PKA) and protein kinase C (PKC), which might be responsible for activation of transcription factors associated with collagen X production.275
Taken together, fatty acids exert multiple effects on specific bone cell types and thereby associated bone diseases (Table 4), which might be mediated via distinct mechanisms at cellular and molecular levels (Figure 4). Understanding the mechanistic implications of fatty acids in bone cells will greatly benefit their further utilization in related bone disorders.
Table 4.
Modulatory roles and therapeutic potentials of fatty acids for bone diseases
| Disease | Pathologic mechanism | Fatty acid | Detrimental mechanism | Potential drug formula | Therapeutic mechanism | Reference |
|---|---|---|---|---|---|---|
| Periodontitis |
Specific bacterial colonization Increased function of osteoclasts Increased dysfunction of osteoblasts |
PA | Induces pro‐inflammatory response | ω‐3 LCPUFAs |
Inhibit putative periodontal pathogens Inhibit PA‐induced chemokine secretion |
31, 33, 49 |
| Butyrate | Inhibit differentiation of HGFs | Resolvin D1 |
Inhibit putative periodontal pathogens Inhibit PA‐induced chemokine secretion |
|||
| SFAs |
Induce oxidative stress Facilitate inflammatory processes |
RvE1 |
Reduce inflammation Inhibit osteoclast activities |
|||
| Osteoporosis |
Increased function of osteoclasts Increased dysfunction of osteoblasts |
ω‐6 LCPUFAs |
Induce chronic inflammation Induce MSC chronic deregulation |
ω‐3 LCPUFAs |
Inhibit osteoclastogenesis Reduce PGE2 |
51, 52, 58 |
| PA | Provide energy generation for differentiation of osteoblasts | |||||
| Osteoporosis |
Reduced OPG Facilitated osteoclast differentiation |
— | — | ω‐3 LCPUFAs |
Inhibit osteoclastogenesis Reduce PGE2 |
47, 61, 64, 65 |
| — | — | SCFAs |
Inhibit osteoclast differentiation Provide energy generation for differentiation of osteoblasts |
|||
| Bone fracture |
Deterioration of bone structure Loss of bone mineral |
ω‐6 LCPUFAs | Induce PGE2 production | ω‐3 LCPUFAs |
Increase calcium resorption Increase synthesis of bone collagen Inhibit urinary calcium excretion |
67, 68 |
| Rheumatoid arthritis | Autoimmune inflammatory disease of unknown aetiology | ω‐6 LCPUFAs | Induce production of pro‐inflammatory cytokines | ω‐3 LCPUFAs |
Reduce inflammation Reduce cartilage‐degrading enzymes |
75, 76 |
| Osteocarcinoma | Derives from primary bone sarcomas or prostate cancer, breast cancer etc | AA | Supports implantation and propagation of metastatic cells | DHA |
Reduce CD44 expression in metastatic cells Inhibit osteoclast formation |
88, 89, 90, 91, 92, 94, 95, 96 |
| EPA | Reduce CD44 expression in metastatic cells | |||||
| Osteomyelitis | Bone infection of pyogenic organisms | — | — | ω‐3 LCPUFAs |
Reduce levels of TNF‐α and IL‐6 Reduce SOD activity |
97, 101, 102, 103, 298 |
| Multiple myeloma | Cancer growing in bone marrow | SFAs | — | PA | Activate multiple myeloma cell apoptosis | 94, 95, 96, 97, 98, 99, 100, 101 |
| ω‐6 LCPUFAs | — | ω‐3 LCPUFAs |
Promote drug sensitivity of myeloma cell apoptosis Activate multiple myeloma cell apoptosis Inhibit function of ω‐6 LCPUFAs |
Figure 4.
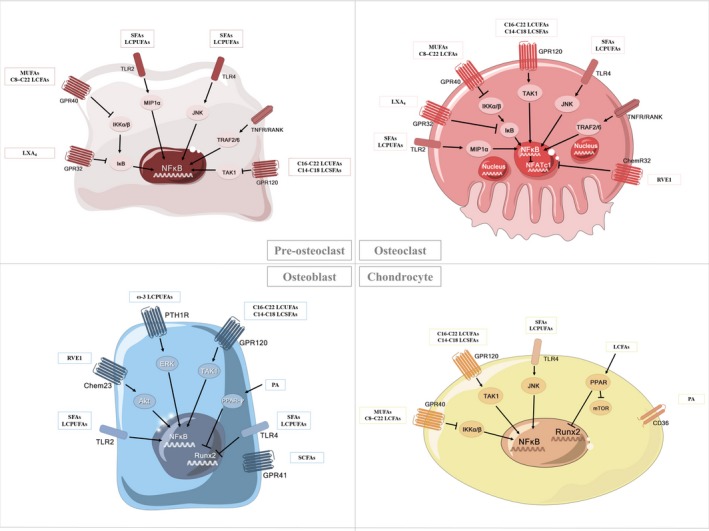
Modulation of fatty acids on specific bone cell types. Multiple receptors for fatty acids including GPRs, ChemR23, TLRs and PPARs are found in pre‐osteoclasts, mature osteoclasts, osteoblasts and chondrocytes. Several GPRs including GPR18, GPR41, GPR43 and GPR109A are receptors for SCFAs (C2‐C5) expressed in both osteoclasts and osteoblasts. GPR40, found on osteoclasts, could be activated by medium/long‐chain fatty acids with a chain length of C8‐C22. GPR84, whose expression in macrophages and adipocytes could be enhanced under inflammatory conditions, is a receptor for MCFAs (C9‐C14). GPR120 is expressed on osteoblasts and osteoclasts and could be stimulated by LCSFAs (C14‐C18) and LCUFAs (C16‐C22). PTH1R, belonging to GPR2 family, could be antagonized by ω‐3 LCPUFAs to promote osteoblast activity. PPARs, with known ligands including LCPUFAs and metabolites such as PGE2, are nuclear receptors that regulate lipid metabolism by acting as transcription factors in BMMSCs, osteoblasts, osteoclasts and chondrocytes. TLRs, including TLR2 and TLR4, are found in osteoblasts, pre‐osteoclasts, osteoclasts and chondrocytes. Their ligands are mainly SFAs and LCPUFAs and are involved in inflammatory action. ChemR23 can act as chemerin receptor as well as RvE1 receptor in bone tissue cells such as osteoclasts and osteoblasts. Interactions of fatty acids with specific receptors induce transduction of transmembrane specific signals and activation of various downstream signalling pathways including NF‐κB, NFATc1 or Runx2‐mediated transcriptional regulation, and further modulating bone microenvironment homeostasis and pathological bone remodelling. GPRs, G protein‐coupled receptors; chemR, chemokine‐like receptor; TLR, toll‐like receptor; SCFAs, short‐chain fatty acids; MCFAs, medium‐chain fatty acids; LCSFAs, long‐chain saturated fatty acids; LCUFAs, long‐chain unsaturated fatty acids; PTH1R, parathyroid hormone type 1 receptor; LCPUFAs, long‐chain polyunsaturated fatty acids; PPARs, peroxisome proliferator‐activated receptors; PGE2, prostaglandin E2; SFAs, saturated fatty acids; RvE1, resolvin E1; NF‐κB, nuclear factor‐kappa B; NFATc1, nuclear factor of activated T‐cell cytoplasmic 1; Runx2, runt‐related transcription factor 2
5. CONCLUDING REMARKS
In this review, we reviewed impacts of fatty acids on bone metabolism, summarized molecular mechanisms involved in actions of fatty acids in distinct bone cell types, and discussed their potential implications for metabolic bone disorders. Currently available findings imply that LCPUFAs mainly exert protective functions on bone by promoting functions of BMMSCs and osteoblasts while inhibiting activities of osteoclasts. MCFAs such as CA suppress osteoclastogenesis and thereby alleviate bone resorption. SCFAs and associated combinational treatment might inhibit bone resorption and inflammatory response for potential therapeutics against inflammatory bone loss including arthritis. Overall, these fatty acids might serve as potential therapeutic and nutritional agents in managing metabolic bone disorders such as osteoporosis, rheumatoid arthritis and oral‐maxillofacial diseases such as periodontitis. Moreover, as natural compounds occurring widely in human body, fatty acids are available in a variety of ways and might be potent to antagonize possible side effects of current drug therapies. Nevertheless, currently available investigations have only reported roles of fatty acids in a limited number of bone disorder conditions, and further bench and clinical investigations are needed to comprehensively elucidate the underlying mechanisms for their possible applications in additional skeletal disorders such as temporomandibular joint disorder and osteosarcoma. Taken together, we conclude that involvement of fatty acids in bone diseases pathogenesis might provide potential therapeutic targets for interventions of bone disorders, and promising fatty acids with therapeutic effects might be used directly or indirectly in nutritional or drug formulations for prevention and treatment of specific types of bone disorders.
CONFLICT OF INTERESTS
The authors declare no conflicts of interest.
AUTHOR CONTRIBUTIONS
Bao M and Zhang K gathered relevant literature and wrote the manuscript; Wei Y, Hua W and Gao Y interpreted data from pathological and experimental studies; and Li X and Ye L provided financial support, revised and reviewed the manuscript.
ACKNOWLEDGEMENTS
This work was supported by grants from the National Natural Science Foundation of China for Distinguished Young Scholars (81825005) to Prof. Ling Ye, and grants from the project of Science & Technology Department of Sichuan Province (2018JY0568) and the National Natural Science Foundation of China (81903033) to Dr Xin Li.
Bao M, Zhang K, Wei Y, et al. Therapeutic potentials and modulatory mechanisms of fatty acids in bone. Cell Prolif. 2020;53:e12735 10.1111/cpr.12735
Contributor Information
Xin Li, Email: lixin0914071@126.com.
Ling Ye, Email: yeling@scu.edu.cn.
DATA AVAILABILITY STATEMENT
The data that support the findings of this study are available from the corresponding author upon reasonable request.
REFERENCES
- 1. Teitelbaum SL. Bone resorption by osteoclasts. Science. 2000;1504‐1508. [DOI] [PubMed] [Google Scholar]
- 2. Doherty AH, Ghalambor CK, Donahue SW. Evolutionary physiology of bone: bone metabolism in changing environments. Physiology. 2015;17‐29. [DOI] [PubMed] [Google Scholar]
- 3. Locatelli V, Bianchi VE. Effect of GH/IGF‐1 on bone metabolism and osteoporsosis. Int J Endocrinol. 2014;53:235060. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4. Mediero A, Cronstein BN. Adenosine and bone metabolism. Trends Endocrin Met. 2013;290‐300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Thent ZC, Froemming GRA, Muid S. Bisphenol A exposure disturbs the bone metabolism: an evolving interest towards an old culprit. Life Sci. 2018;1‐7. [DOI] [PubMed] [Google Scholar]
- 6. Insua A, Monje A, Wang HL, Miron RJ. Basis of bone metabolism around dental implants during osseointegration and peri‐implant bone loss. J Biomed Mater Res A. 2017;2075‐2089. [DOI] [PubMed] [Google Scholar]
- 7. Kokabu S, Lowery JW, Jimi E. Cell fate and differentiation of bone marrow mesenchymal stem cells. Stem Cells Int. 2016;53:3753581. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Xu J, Li Z, Hou Y, Fang W. Potential mechanisms underlying the Runx2 induced osteogenesis of bone marrow mesenchymal stem cells. Am J Transl Res. 2015;2527‐2535. [PMC free article] [PubMed] [Google Scholar]
- 9. Galli D, Vitale M, Vaccarezza M. Bone marrow‐derived mesenchymal cell differentiation toward myogenic lineages: facts and perspectives. Biomed Res Int. 2014;53:762695. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10. Loi F, Cordova LA, Pajarinen J, et al. Inflammation, fracture and bone repair. Bone. 2016;119‐130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Beermann C, Jelinek J, Reinecker T, et al. Short term effects of dietary medium‐chain fatty acids and n‐3 long‐chain polyunsaturated fatty acids on the fat metabolism of healthy volunteers. Lipids Health Dis. 2003;10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Harasymowicz NS, Dicks A, Wu CL, et al. Physiologic and pathologic effects of dietary free fatty acids on cells of the joint. Ann N Y Acad Sci. 2019;36‐53. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13. Takahashi Y, Ide T. Dietary n‐3 fatty acids affect mRNA level of brown adipose tissue uncoupling protein 1, and white adipose tissue leptin and glucose transporter 4 in the rat. Br J Nutr. 2000;175‐184. [PubMed] [Google Scholar]
- 14. Ahn SH, Park SY, Baek JE, et al. Free fatty acid receptor 4 (GPR120) stimulates bone formation and suppresses bone resorption in the presence of elevated n‐3 fatty acid levels. Endocrinology. 2016;2621‐2635. [DOI] [PubMed] [Google Scholar]
- 15. Watkins BA, Li Y, Lippman HE, et al. Modulatory effect of omega‐3 polyunsaturated fatty acids on osteoblast function and bone metabolism. Prostag Leukotr Ess. 2003;387‐398. [DOI] [PubMed] [Google Scholar]
- 16. Griel AE, Kris‐Etherton PM, Hilpert KF, et al. An increase in dietary n‐3 fatty acids decreases a marker of bone resorption in humans. Nutr J. 2007;2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Mangano KM, Kerstetter JE, Kenny AM, et al. An investigation of the association between omega 3 FA and bone mineral density among older adults: results from the National Health and Nutrition Examination Survey years 2005–2008. Osteoporos Int. 2014;1033‐1041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Weiss LA, Elizabeth BC, Denise VM. Ratio of n‐6 to n‐3 fatty acids and bone mineral density in older adults: the Rancho Bernardo Study. Am J Clin Nutr. 2005;934‐938. [DOI] [PubMed] [Google Scholar]
- 19. Hogstrom M, Nordstrom P, Nordstrom A. n‐3 Fatty acids are positively associated with peak bone mineral density and bone accrual in healthy men: the NO2 Study. Am J Clin Nutr. 2007;803‐807. [DOI] [PubMed] [Google Scholar]
- 20. Dongxu S, Aparna K, Khaliquz Z, et al. Dietary n‐3 fatty acids decrease osteoclastogenesis and loss of bone mass in ovariectomized mice. J Bone Miner Res. 2010;1206‐1216. [DOI] [PubMed] [Google Scholar]
- 21. Bai X, Lin C, Jiang H, et al. Endogenous production of n‐3 polyunsaturated fatty acids promotes fracture healing in mice. J Healthc Eng. 2017;1‐6. [DOI] [PubMed] [Google Scholar]
- 22. Appleton KM, Fraser WD, Rogers PJ, et al. Supplementation with a low‐moderate dose of n‐3 long‐chain PUFA has no short‐term effect on bone resorption in human adults. Br J Nutr. 2011;1145‐1149. [DOI] [PubMed] [Google Scholar]
- 23. Navarini L, Afeltra A, Gallo Afflitto G, et al. Polyunsaturated fatty acids: any role in rheumatoid arthritis? Lipids Health Dis. 2017;197. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24. Lavado‐García J, Roncero‐Martin R, Moran JM, et al. Long‐chain omega‐3 polyunsaturated fatty acid dietary intake is positively associated with bone mineral density in normal and osteopenic Spanish women. PLoS ONE. 2018;53:e0190539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25. Kwon JO, Jin WJ, Kim B, et al. Myristoleic acid inhibits osteoclast formation and bone resorption by suppressing the RANKL activation of Src and Pyk2. Eur J Pharmacol. 2015;189‐198. [DOI] [PubMed] [Google Scholar]
- 26. Najumudeen AK, Kohnke M, Solman M, et al. Cellular FRET‐biosensors to detect membrane targeting Inhibitors of N‐Myristoylated proteins. PLoS ONE. 2013;53:e66425. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27. Fillmore N, Huqi A, Jaswal JS, et al. Effect of fatty acids on human bone marrow mesenchymal stem cell energy metabolism and survival. PLoS ONE. 2015;53:e0120257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28. Yan J, Takakura A, Zandi‐Nejad K, et al. Mechanisms of gut microbiota‐mediated bone remodeling. Gut Microbes. 2018;84‐92. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29. Weaver CM. Diet, gut microbiome, and bone health. Curr Osteoporos Rep. 2015;125‐130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30. Montalvany‐Antonucci CC, Duffles LF, de Arruda JAA, et al. Short‐chain fatty acids and FFAR2 as suppressors of bone resorption. Bone. 2019;112‐121. [DOI] [PubMed] [Google Scholar]
- 31. Shikama Y, Kudo Y, Ishimaru N, et al. Potential role of free fatty acids in the pathogenesis of periodontitis and primary Sjogren's syndrome. Int J Mol Sci. 2017;122‐134. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32. Peddis N, Musu D, Ideo F, et al. Interaction of biologic therapy with apical periodontitis and periodontitis: a systematic review. Aust Dent J. 2019;64:122‐134. [DOI] [PubMed] [Google Scholar]
- 33. Sima C, Van Dyke TE. Therapeutic targets for management of periodontitis and diabetes. Curr Pharm Des. 2016;2216‐2237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34. Bendyk A, Marino V, Zilm PS, et al. Effect of dietary omega‐3 polyunsaturated fatty acids on experimental periodontitis in the mouse. J Periodontal Res. 2009;211‐216. [DOI] [PubMed] [Google Scholar]
- 35. Azuma MM, Gomes‐Filho JE, Cardoso CBM, et al. Omega 3 fatty acids reduce the triglyceride levels in rats with apical periodontitis. Braz Dent J. 2018;173‐178. [DOI] [PubMed] [Google Scholar]
- 36. Li Y, Lu Z, Zhang X, et al. Metabolic syndrome exacerbates inflammation and bone loss in periodontitis. J Dent Res. 2015;362‐370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37. Balta MG, Loos BG, Nicu EA. Emerging concepts in the resolution of periodontal inflammation: a role for resolvin E1. Front Immunol. 2017;1682. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38. Muluke M, Gold T, Kiefhaber K, et al. Diet‐induced obesity and its differential impact on periodontal bone loss. J Dent Res. 2016;223‐229. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39. Qiqiang L, Huanxin M, Xuejun G. Longitudinal study of volatile fatty acids in the gingival crevicular fluid of patients with periodontitis before and after nonsurgical therapy. J Periodontal Res. 2012;740‐749. [DOI] [PubMed] [Google Scholar]
- 40. Lu R, Meng H, Gao X, et al. Effect of non‐surgical periodontal treatment on short chain fatty acid levels in gingival crevicular fluid of patients with generalized aggressive periodontitis. J Periodontal Res. 2014;574‐583. [DOI] [PubMed] [Google Scholar]
- 41. El‐Sharkawy H, Aboelsaad N, Eliwa M, et al. Adjunctive treatment of chronic periodontitis with daily dietary supplementation with omega‐3 Fatty acids and low‐dose aspirin. J Periodontol. 2010;1635‐1643. [DOI] [PubMed] [Google Scholar]
- 42. Deore GD, Gurav AN, Patil R, et al. Omega 3 fatty acids as a host modulator in chronic periodontitis patients: a randomised, double‐blind, palcebo‐controlled, clinical trial. J Periodontal Implant Sci. 2014;25‐32. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43. Wang SH, Hung HC, Tsai CC, et al. Plasma polyunsaturated fatty acids and periodontal recovery in Taiwanese with periodontitis: a significant relationship. Arch Oral Biol. 2014;800‐807. [DOI] [PubMed] [Google Scholar]
- 44. Keskiner I, Saygun I, Bal V, et al. Dietary supplementation with low‐dose omega‐3 fatty acids reduces salivary tumor necrosis factor‐alpha levels in patients with chronic periodontitis: a randomized controlled clinical study. J Periodontal Res. 2017;695‐703. [DOI] [PubMed] [Google Scholar]
- 45. Naqvi AZ, Hasturk H, Mu L, et al. Docosahexaenoic acid and periodontitis in adults: a randomized controlled trial. J Dent Res. 2014;767‐773. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46. Martinez GL, Koury JC, Brito F, et al. The impact of non‐surgical periodontal treatment on serum levels of long chain‐polyunsaturated fatty acids: a pilot randomized clinical trial. J Periodontal Res. 2014;268‐274. [DOI] [PubMed] [Google Scholar]
- 47. Gyurko R, Van Dyke TE. The role of polyunsaturated omega‐3 fatty acid eicosapentaenoic acid‐derived resolvin E1 (RvE1) in bone preservation. Crit Rev Immunol. 2014;347‐357. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48. Varela‐Lopez A, Giampieri F, Bullon P, et al. Role of Lipids in the onset, progression and treatment of periodontal disease. A systematic review of studies in humans. Int J Mol Sci. 2016;15017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49. Szafranski SP, Deng ZL, Tomasch J, et al. Functional biomarkers for chronic periodontitis and insights into the roles of Prevotella nigrescens and Fusobacterium nucleatum; a metatranscriptome analysis. NPJ Biofilms Microbiomes. 2015;1:15017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50. Kanis JA, Reginster JY. European guidance for the diagnosis and management of osteoporosis in postmenopausal women–what is the current message for clinical practice? Pol Arch Med Wewn. 2008;538‐540. [PubMed] [Google Scholar]
- 51. Banu J. Causes, consequences, and treatment of osteoporosis in men. Drug Des Devel Ther. 2013;849‐860. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52. Lee WC, Guntur AR, Long F, et al. Energy metabolism of the osteoblast: implications for osteoporosis. Endocr Rev. 2017;255‐266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53. Chen JS, Sambrook PN. Antiresorptive therapies for osteoporosis: a clinical overview. Nat Rev Endocrinol. 2011;81‐91. [DOI] [PubMed] [Google Scholar]
- 54. Rosen CJ, Bouxsein ML. Mechanisms of disease: is osteoporosis the obesity of bone? Nat Clin Pract Rheumatol. 2006;35‐43. [DOI] [PubMed] [Google Scholar]
- 55. Salari P, Rezaie A, Larijani B, et al. A systematic review of the impact of n‐3 fatty acids in bone health and osteoporosis. Med Sci Monit. 2008;RA37‐44. [PubMed] [Google Scholar]
- 56. Vanek C, Connor WE. Do n‐3 fatty acids prevent osteoporosis? Am J Clin Nutr. 2007;647‐648. [DOI] [PubMed] [Google Scholar]
- 57. Kelly OJ, Gilman JC, Kim Y, et al. Long‐chain polyunsaturated fatty acids may mutually benefit both obesity and osteoporosis. Nutr Res. 2013;521‐533. [DOI] [PubMed] [Google Scholar]
- 58. Casado‐Diaz A, Santiago‐Mora R, Dorado G, et al. The omega‐6 arachidonic fatty acid, but not the omega‐3 fatty acids, inhibits osteoblastogenesis and induces adipogenesis of human mesenchymal stem cells: potential implication in osteoporosis. Osteoporos Int. 2013;1647‐1661. [DOI] [PubMed] [Google Scholar]
- 59. Varela‐Lopez A, Ochoa JJ, Llamas‐Elvira JM, et al. Age‐related loss in bone mineral density of rats fed lifelong on a fish oil‐based diet is avoided by coenzyme Q10 addition. Nutrients. 2017;14203‐14227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60. Lau BY, Cohen DJ, Ward WE, et al. Investigating the role of polyunsaturated fatty acids in bone development using animal models. Molecules. 2013;18:14203‐14227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61. Xu X, Jia X, Mo L, et al. Intestinal microbiota: a potential target for the treatment of postmenopausal osteoporosis. Bone Res. 2017;17046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62. Miranda M, Pino AM, Fuenzalida K, et al. Characterization of fatty acid composition in bone marrow fluid from postmenopausal women: modification after hip fracture. J Cell Biochem. 2016;2370‐2376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63. Yan J, Herzog JW, Tsang K, et al. Gut microbiota induce IGF‐1 and promote bone formation and growth. Proc Natl Acad Sci USA. 2016;E7554‐E7563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64. Lucas S, Omata Y, Hofmann J, et al. Short‐chain fatty acids regulate systemic bone mass and protect from pathological bone loss. Nat Commun. 2018;55. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65. Choi E, Park Y. The association between the consumption of fish/shellfish and the risk of osteoporosis in men and postmenopausal women aged 50 years or older. Nutrients. 2016;113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 66. Milam SB. Pathogenesis of degenerative temporomandibular joint arthritides. Odontology. 2005;7‐15. [DOI] [PubMed] [Google Scholar]
- 67. Longo AB, Ward WE. PUFAs, bone mineral density, and fragility fracture: findings from human studies. Adv Nutr. 2016;299‐312. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68. Sadeghi O, Djafarian K, Ghorabi S, et al. Dietary intake of fish, n‐3 polyunsaturated fatty acids and risk of hip fracture: A systematic review and meta‐analysis on observational studies. Crit Rev Food Sci Nutr. 2019;1320‐1333. [DOI] [PubMed] [Google Scholar]
- 69. Li J, He W, Liao B, et al. FFA‐ROS‐P53‐mediated mitochondrial apoptosis contributes to reduction of osteoblastogenesis and bone mass in type 2 diabetes mellitus. Sci Rep. 2015;12724. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70. Virtanen JK, Mozaffarian D, Willett WC, et al. Dietary intake of polyunsaturated fatty acids and risk of hip fracture in men and women. Osteoporos Int. 2012;2615‐2624. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 71. Guo Q, Wang Y, Xu D, et al. Rheumatoid arthritis: pathological mechanisms and modern pharmacologic therapies. Bone Res. 2018;15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72. Gerlag DM, Norris JM, Tak PP. Towards prevention of autoantibody‐positive rheumatoid arthritis: from lifestyle modification to preventive treatment. Rheumatology. 2016;607‐614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73. Kaneko Y, Takeuchi T. A paradigm shift in rheumatoid arthritis over the past decade. Intern Med. 2014;1895‐1903. [DOI] [PubMed] [Google Scholar]
- 74. Gioxari A, Kaliora AC, Marantidou F, et al. Intake of omega‐3 polyunsaturated fatty acids in patients with rheumatoid arthritis: a systematic review and meta‐analysis. Nutrition. 2018;114‐124.e4. [DOI] [PubMed] [Google Scholar]
- 75. Rajaei E, Mowla K, Ghorbani A, et al. The effect of omega‐3 fatty acids in patients with active rheumatoid arthritis receiving DMARDs therapy: double‐blind randomized controlled trial. Glob J Health Sci. 2015;18‐25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76. Miles EA, Calder PC. Influence of marine n‐3 polyunsaturated fatty acids on immune function and a systematic review of their effects on clinical outcomes in rheumatoid arthritis. Br J Nutr. 2012;57‐66. [DOI] [PubMed] [Google Scholar]
- 77. Abdulrazaq M, Innes JK, Calder PC. Effect of omega‐3 polyunsaturated fatty acids on arthritic pain: A systematic review. Nutrition. 2017;39–40:57‐66. [DOI] [PubMed] [Google Scholar]
- 78. Gan RW, Young KA, Zerbe GO, et al. Lower omega‐3 fatty acids are associated with the presence of anti‐cyclic citrullinated peptide autoantibodies in a population at risk for future rheumatoid arthritis: a nested case‐control study. Rheumatology. 2016;367‐376. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79. Woo SJ, Lim K, Park SY, et al. Endogenous conversion of n‐6 to n‐3 polyunsaturated fatty acids attenuates K/BxN serum‐transfer arthritis in fat‐1 mice. J Nutr Biochem. 2015;713‐720. [DOI] [PubMed] [Google Scholar]
- 80. Falank C, Fairfield H, Reagan MR. Signaling interplay between bone marrow adipose tissue and multiple myeloma cells. Front Endocrinol. 2016;67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81. Yasuyuki N, Itsuko I, Michihiko W, et al. Palmitic acid, verified by lipid profiling using secondary ion mass spectrometry, demonstrates anti‐multiple myeloma activity. Leuk Res. 2015;638‐645. [DOI] [PubMed] [Google Scholar]
- 82. Abdi J, Garssen J, Faber J, et al. Omega‐3 fatty acids, EPA and DHA induce apoptosis and enhance drug sensitivity in multiple myeloma cells but not in normal peripheral mononuclear cells. J Nutr Biochem. 2014;1254‐1262. [DOI] [PubMed] [Google Scholar]
- 83. Dai X, Li M, Geng F. Omega‐3 polyunsaturated fatty acids eicosapentaenoic acid and docosahexaenoic acid enhance dexamethasone sensitivity in multiple myeloma cells by the p53/miR‐34a/Bcl‐2 axis. Biochemistry. 2017;826‐833. [DOI] [PubMed] [Google Scholar]
- 84. Kaiyan H, Yang Y, Kejian S, et al. The p38 MAPK‐regulated PKD1/CREB/Bcl‐2 pathway contributes to selenite‐induced colorectal cancer cell apoptosis in vitro and in vivo. Cancer Lett. 2014;189‐199. [DOI] [PubMed] [Google Scholar]
- 85. Hai‐Yan S, Xiao‐Hui D, Guo‐Yan Y, et al. Expression of bcl‐2 and p53 in induction of esophageal cancer cell apoptosis by ECRG2 in combination with cisplatin. Asian Pac J Cancer Prev. 2014;1397‐1401. [DOI] [PubMed] [Google Scholar]
- 86. Fernandez E, Chatenoud L, La Vecchia C, et al. Fish consumption and cancer risk. Am J Clin Nutr. 1999;85‐90. [DOI] [PubMed] [Google Scholar]
- 87. Jurczyszyn A, Czepiel J, Gdula‐Argasińska J, et al. Plasma fatty acid profile in multiple myeloma patients. Leuk Res. 2015;400‐405. [DOI] [PubMed] [Google Scholar]
- 88. Song ZP, Xiong BR, Guan XH, et al. Minocycline attenuates bone cancer pain in rats by inhibiting NF‐kappaB in spinal astrocytes. Acta Pharmacol Sin. 2016;753‐762. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89. Wang J, Chen GL, Cao S, et al. Adipogenic niches for melanoma cell colonization and growth in bone marrow. Lab Invest. 2017;737‐745. [DOI] [PubMed] [Google Scholar]
- 90. Sowder ME, Johnson RW. Bone as a preferential site for metastasis. JBMR Plus. 2019;53:e10126. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91. Potepan P, Spagnoli I, Danesini GM, et al. The radiodiagnosis of bone metastases from melanoma. Radiol Med (Torino). 1994;741‐746. [PubMed] [Google Scholar]
- 92. Hesse E, Taipaleenmaki H. MicroRNAs in bone metastasis. Curr Osteoporos Rep. 2019;122‐128. [DOI] [PubMed] [Google Scholar]
- 93. Mandal CC, Ghosh‐Choudhury T, Yoneda T, et al. Fish oil prevents breast cancer cell metastasis to bone. Biochem Biophys Res Commun. 2010;602‐607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94. Rahman MM, Veigas JM, Williams PJ, et al. DHA is a more potent inhibitor of breast cancer metastasis to bone and related osteolysis than EPA. Breast Cancer Res Treat. 2013;341‐352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95. Brown MD, Hart C, Gazi E, et al. Influence of omega‐6 PUFA arachidonic acid and bone marrow adipocytes on metastatic spread from prostate cancer. Br J Cancer. 2010;403‐413. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96. Herroon MK, Rajagurubandara E, Hardaway AL, et al. Bone marrow adipocytes promote tumor growth in bone via FABP4‐dependent mechanisms. Oncotarget. 2013;2108‐2123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97. Zhou P, Wu J, Wang Y, et al. The synergistic therapeutic efficacy of vancomycin and omega‐3 fatty acids alleviates Staphylococcus aureus‐induced osteomyelitis in rats. Biomed Pharmacother. 2019;1228‐1233. [DOI] [PubMed] [Google Scholar]
- 98. Patel M, Rojavin Y, Jamali AA, et al. Animal models for the study of osteomyelitis. Semin Plast Surg. 2009;148‐154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 99. Calhoun JH, Manring MM, Shirtliff M. Osteomyelitis of the long bones. Semin Plast Surg. 2009;59‐72. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 100. Buch K, Thuesen ACB, Brons C, et al. Chronic non‐bacterial osteomyelitis: a review. Calcif Tissue Int. 2018;544‐553. [DOI] [PubMed] [Google Scholar]
- 101. Svahn SL, Ulleryd MA, Grahnemo L, et al. Dietary omega‐3 fatty acids increase survival and decrease bacterial load in mice subjected to Staphylococcus aureus‐induced sepsis. Infect Immun. 2016;1205‐1213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102. Desbois AP, Lawlor KC. Antibacterial activity of long‐chain polyunsaturated fatty acids against Propionibacterium acnes and Staphylococcus aureus . Mar Drugs. 2013;4544‐4557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103. Kim YG, Lee JH, Raorane CJ, et al. Herring oil and omega fatty acids inhibit Staphylococcus aureus biofilm formation and virulence. Front Microbiol. 2018;1241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104. Wauquier F, Leotoing L, Philippe C, et al. Pros and cons of fatty acids in bone biology. Prog Lipid Res. 2015;121‐145. [DOI] [PubMed] [Google Scholar]
- 105. Gunaratnam K, Vidal C, Boadle R, et al. Mechanisms of palmitate‐induced cell death in human osteoblasts. Biol Open. 2013;1382‐1389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106. Mizushima N. Autophagy: process and function. Genes Dev. 2007;2861‐2873. [DOI] [PubMed] [Google Scholar]
- 107. Ge H, Li X, Weiszmann J, et al. Activation of G protein‐coupled receptor 43 in adipocytes leads to inhibition of lipolysis and suppression of plasma free fatty acids. Endocrinology. 2008;4519‐4526. [DOI] [PubMed] [Google Scholar]
- 108. Xiong Y, Miyamoto N, Shibata K, et al. Short‐chain fatty acids stimulate leptin production in adipocytes through the G protein‐coupled receptor GPR41. Proc Natl Acad Sci USA. 2004;1045‐1050. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109. Briscoe CP, Tadayyon M, Andrews JL, et al. The orphan G protein‐coupled receptor GPR40 is activated by medium and long chain fatty acids. J Biol Chem. 2003;11303‐11311. [DOI] [PubMed] [Google Scholar]
- 110. Cornish J, MacGibbon A, Lin JM, et al. Modulation of osteoclastogenesis by fatty acids. Endocrinology. 2008;5688‐5695. [DOI] [PubMed] [Google Scholar]
- 111. Blad CC, Tang C, Offermanns S. G protein‐coupled receptors for energy metabolites as new therapeutic targets. Nat Rev Drug Discov. 2012;603‐619. [DOI] [PubMed] [Google Scholar]
- 112. Philippe C, Wauquier F, Leotoing L, et al. GW9508, a free fatty acid receptor agonist, specifically induces cell death in bone resorbing precursor cells through increased oxidative stress from mitochondrial origin. Exp Cell Res. 2013;3035‐3041. [DOI] [PubMed] [Google Scholar]
- 113. Wauquier F, Philippe C, Leotoing L, et al. The free fatty acid receptor G protein‐coupled receptor 40 (GPR40) protects from bone loss through inhibition of osteoclast differentiation. J Biol Chem. 2013;6542‐6551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114. Philippe C, Wauquier F, Landrier JF, et al. GPR40 mediates potential positive effects of a saturated fatty acid enriched diet on bone. Mol Nutr Food Res. 2017;34457‐34464. [DOI] [PubMed] [Google Scholar]
- 115. Wang J, Wu X, Simonavicius N, et al. Medium‐chain fatty acids as ligands for orphan G protein‐coupled receptor GPR84. J Biol Chem. 2006;281:34457‐34464. [DOI] [PubMed] [Google Scholar]
- 116. Bouchard C, Page J, Bedard A, et al. G protein‐coupled receptor 84, a microglia‐associated protein expressed in neuroinflammatory conditions. Glia. 2007;790‐800. [DOI] [PubMed] [Google Scholar]
- 117. Nagasaki H, Kondo T, Fuchigami M, et al. Inflammatory changes in adipose tissue enhance expression of GPR84, a medium‐chain fatty acid receptor: TNFalpha enhances GPR84 expression in adipocytes. FEBS Lett. 2012;368‐372. [DOI] [PubMed] [Google Scholar]
- 118. Oh DY, Talukdar S, Bae EJ, et al. GPR120 is an omega‐3 fatty acid receptor mediating potent anti‐inflammatory and insulin‐sensitizing effects. Cell. 2010;687‐698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119. Gao B, Huang Q, Jie Q, et al. GPR120: A bi‐potential mediator to modulate the osteogenic and adipogenic differentiation of BMMSCs. Sci Rep. 2015;14080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120. Chen Y, Zhang D, Ho KW, et al. GPR120 is an important inflammatory regulator in the development of osteoarthritis. Arthritis Res Ther. 2018;163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121. Xiao G, Jiang D, Thomas P, et al. MAPK pathways activate and phosphorylate the osteoblast‐specific transcription factor, Cbfa1. J Biol Chem. 2000;4453‐4459. [DOI] [PubMed] [Google Scholar]
- 122. Sowa H, Kaji H, Yamaguchi T, et al. Activations of ERK1/2 and JNK by transforming growth factor beta negatively regulate Smad3‐induced alkaline phosphatase activity and mineralization in mouse osteoblastic cells. J Biol Chem. 2002;36024‐36031. [DOI] [PubMed] [Google Scholar]
- 123. Candelario J, Tavakoli H, Chachisvilis M. PTH1 receptor is involved in mediating cellular response to long‐chain polyunsaturated fatty acids. PLoS ONE. 2012;53:e52583. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124. Berger J, Bailey P, Biswas C, et al. Thiazolidinediones produce a conformational change in peroxisomal proliferator‐activated receptor‐gamma: binding and activation correlate with antidiabetic actions in db/db mice. Endocrinology. 1996;4189‐4195. [DOI] [PubMed] [Google Scholar]
- 125. Siersbaek R, Nielsen R, Mandrup S. PPARgamma in adipocyte differentiation and metabolism–novel insights from genome‐wide studies. FEBS Lett. 2010;3242‐3249. [DOI] [PubMed] [Google Scholar]
- 126. Ambrogini E, Almeida M, Martin‐Millan M, et al. FoxO‐mediated defense against oxidative stress in osteoblasts is indispensable for skeletal homeostasis in mice. Cell Metab. 2010;136‐146. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127. Iyer S, Ambrogini E, Bartell SM, et al. FOXOs attenuate bone formation by suppressing Wnt signaling. J Clin Invest. 2013;3409‐3419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128. Rached MT, Kode A, Silva BC, et al. FoxO1 expression in osteoblasts regulates glucose homeostasis through regulation of osteocalcin in mice. J Clin Invest. 2010;357‐368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129. Diascro DD Jr, Vogel RL, et al. High fatty acid content in rabbit serum is responsible for the differentiation of osteoblasts into adipocyte‐like cells. J Bone Miner Res. 1998;96‐106. [DOI] [PubMed] [Google Scholar]
- 130. Mano H, Kimura C, Fujisawa Y, et al. Cloning and function of rabbit peroxisome proliferator‐activated receptor delta/beta in mature osteoclasts. J Biol Chem. 2000;8126‐8132. [DOI] [PubMed] [Google Scholar]
- 131. Chan BY, Gartland A, Wilson PJ, et al. PPAR agonists modulate human osteoclast formation and activity in vitro. Bone. 2007;149‐159. [DOI] [PubMed] [Google Scholar]
- 132. Sun H, Kim JK, Mortensen R, et al. Osteoblast‐targeted suppression of PPARgamma increases osteogenesis through activation of mTOR signaling. Stem Cells. 2013;2183‐2192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 133. Duque G, Li W, Vidal C, et al. Pharmacological inhibition of PPARgamma increases osteoblastogenesis and bone mass in male C57BL/6 mice. J Bone Miner Res. 2013;639‐648. [DOI] [PubMed] [Google Scholar]
- 134. Viccica G, Francucci CM, Marcocci C. The role of PPARgamma for the osteoblastic differentiation. J Endocrinol Invest. 2010;9‐12. [PubMed] [Google Scholar]
- 135. Akune T, Ohba S, Kamekura S, et al. PPARgamma insufficiency enhances osteogenesis through osteoblast formation from bone marrow progenitors. J Clin Invest. 2004;846‐855. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136. Artur J, Jacek C, Joanna GA, et al. Erythrocyte membrane fatty acids in multiple myeloma patients. Leuk Res. 2014;1260‐1265. [DOI] [PubMed] [Google Scholar]
- 137. Snodgrass RG, Huang S, Choi IW, et al. Inflammasome‐mediated secretion of IL‐1beta in human monocytes through TLR2 activation; modulation by dietary fatty acids. J Immunol. 2013;4337‐4347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138. Nguyen MT, Favelyukis S, Nguyen AK, et al. A subpopulation of macrophages infiltrates hypertrophic adipose tissue and is activated by free fatty acids via Toll‐like receptors 2 and 4 and JNK‐dependent pathways. J Biol Chem. 2007;35279‐35292. [DOI] [PubMed] [Google Scholar]
- 139. Huang S, Rutkowsky JM, Snodgrass RG, et al. Saturated fatty acids activate TLR‐mediated proinflammatory signaling pathways. J Lipid Res. 2012;2002‐2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140. Hwang D. Modulation of the expression of cyclooxygenase‐2 by fatty acids mediated through toll‐like receptor 4‐derived signaling pathways. FASEB J. 2001;2556‐2564. [DOI] [PubMed] [Google Scholar]
- 141. Shi H, Kokoeva MV, Inouye K, et al. TLR4 links innate immunity and fatty acid‐induced insulin resistance. J Clin Invest. 2006;3015‐3025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 142. Dasu MR, Ramirez S, Isseroff RR. Toll‐like receptors and diabetes: a therapeutic perspective. Clin Sci (Lond). 2012;203‐214. [DOI] [PubMed] [Google Scholar]
- 143. Oh SR, Sul OJ, Kim YY, et al. Saturated fatty acids enhance osteoclast survival. J Lipid Res. 2010;892‐899. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144. Johnson GB, Riggs BL, Platt JL. A genetic basis for the "Adonis" phenotype of low adiposity and strong bones. FASEB J. 2004;1282‐1284. [DOI] [PubMed] [Google Scholar]
- 145. Lee JY, Zhao L, Youn HS, et al. Saturated fatty acid activates but polyunsaturated fatty acid inhibits Toll‐like receptor 2 dimerized with Toll‐like receptor 6 or 1. J Biol Chem. 2004;16971‐16979. [DOI] [PubMed] [Google Scholar]
- 146. Samson M, Edinger AL, Stordeur P, et al. ChemR23, a putative chemoattractant receptor, is expressed in monocyte‐derived dendritic cells and macrophages and is a coreceptor for SIV and some primary HIV‐1 strains. Eur J Immunol. 1998;1689‐1700. [DOI] [PubMed] [Google Scholar]
- 147. Arita M, Ohira T, Sun YP, et al. Resolvin E1 selectively interacts with leukotriene B4 receptor BLT1 and ChemR23 to regulate inflammation. J Immunol. 2007;3912‐3917. [DOI] [PubMed] [Google Scholar]
- 148. Arita M, Oh SF, Chonan T, et al. Metabolic inactivation of resolvin E1 and stabilization of its anti‐inflammatory actions. J Biol Chem. 2006;22847‐22854. [DOI] [PubMed] [Google Scholar]
- 149. Arita M, Bianchini F, Aliberti J, et al. Stereochemical assignment, antiinflammatory properties, and receptor for the omega‐3 lipid mediator resolvin E1. J Exp Med. 2005;713‐722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 150. Methner A, Hermey G, Schinke B, et al. A novel G protein‐coupled receptor with homology to neuropeptide and chemoattractant receptors expressed during bone development. Biochem Biophys Res Commun. 1997;336‐342. [DOI] [PubMed] [Google Scholar]
- 151. Gao L, Faibish D, Fredman G, et al. Resolvin E1 and chemokine‐like receptor 1 mediate bone preservation. J Immunol. 2013;689‐694. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 152. El Kholy K, Freire M, Chen T, et al. Resolvin E1 promotes bone preservation under inflammatory conditions. Front Immunol. 2018;1300. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 153. Infante M, Fabi A, Cognetti F, et al. RANKL/RANK/OPG system beyond bone remodeling: involvement in breast cancer and clinical perspectives. J Exp Clin Cancer Res. 2019;12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 154. Hutami IR, Tanaka E, Izawa T. Crosstalk between Fas and S1P1 signaling via NF‐kB in osteoclasts controls bone destruction in the TMJ due to rheumatoid arthritis. Jpn Dent Sci Rev. 2019;12‐19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 155. Wu M, Chen G, Li YP. TGF‐beta and BMP signaling in osteoblast, skeletal development, and bone formation, homeostasis and disease. Bone Res. 2016;16009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 156. Houschyar KS, Tapking C, Borrelli MR, et al. Wnt Pathway in Bone Repair and Regeneration ‐ What Do We Know So Far. Front Cell Dev Biol. 2018;170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 157. Canalis E. Notch in skeletal physiology and disease. Osteoporos Int. 2018;2611‐2621. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158. Cong Q, Xu R, Yang Y. Galphas signaling in skeletal development, homeostasis and diseases. Curr Top Dev Biol. 2019;281‐307. [DOI] [PubMed] [Google Scholar]
- 159. Bertoldo F, Silvestris F, Ibrahim T, et al. Targeting bone metastatic cancer: role of the mTOR pathway. Biochim Biophys Acta. 2014;248‐254. [DOI] [PubMed] [Google Scholar]
- 160. Sigl V, Jones LP, Penninger JM. RANKL/RANK: from bone loss to the prevention of breast cancer. Open Biol. 2016;576‐588. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161. Richards JB, Zheng HF, Spector TD. Genetics of osteoporosis from genome‐wide association studies: advances and challenges. Nat Rev Genet. 2012;13:576‐588. [DOI] [PubMed] [Google Scholar]
- 162. Choi HK, Kim TH, Jhon GJ, et al. Reactive oxygen species regulate M‐CSF‐induced monocyte/macrophage proliferation through SHP1 oxidation. Cell Signal. 2011;1633‐1639. [DOI] [PubMed] [Google Scholar]
- 163. Haagenson KK, Wu GS. The role of MAP kinases and MAP kinase phosphatase‐1 in resistance to breast cancer treatment. Cancer Metastasis Rev. 2010;143‐149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164. Raingeaud J, Gupta S, Rogers JS, et al. Pro‐inflammatory cytokines and environmental stress cause p38 mitogen‐activated protein kinase activation by dual phosphorylation on tyrosine and threonine. J Biol Chem. 1995;7420‐7426. [DOI] [PubMed] [Google Scholar]
- 165. Zhou JY, Liu Y, Wu GS. The role of mitogen‐activated protein kinase phosphatase‐1 in oxidative damage‐induced cell death. Cancer Res. 2006;4888‐4894. [DOI] [PubMed] [Google Scholar]
- 166. van Heerden B, Kasonga A, Kruger MC, et al. Palmitoleic acid inhibits RANKL‐induced osteoclastogenesis and bone resorption by suppressing NF‐kappaB and MAPK signalling pathways. Nutrients. 2017;53:e27739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 167. Park EJ, Kim SA, Choi YM, et al. Capric acid inhibits NO production and STAT3 activation during LPS‐induced osteoclastogenesis. PLoS ONE. 2011;6:e27739. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168. Cardoso GB, Chacon E, Chacon PG, et al. Fatty acid is a potential agent for bone tissue induction: In vitro and in vivo approach. Exp Biol Med. 2017;1765‐1771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 169. Son Y, Cheong YK, Kim NH, et al. Mitogen‐activated protein kinases and reactive oxygen species: how can ROS activate MAPK pathways? J Signal Transduct. 2011;53:792639. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 170. Dai L, Aye Thu C, Liu XY, et al. TAK1, more than just innate immunity. IUBMB Life. 2012;825‐834. [DOI] [PubMed] [Google Scholar]
- 171. Shen G, Ren H, Qiu T, et al. Mammalian target of rapamycin as a therapeutic target in osteoporosis. J Cell Physiol. 2018;3929‐3944. [DOI] [PubMed] [Google Scholar]
- 172. Ashworth RE, Wu J. Mammalian target of rapamycin inhibition in hepatocellular carcinoma. World J Hepatol. 2014;776‐782. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173. Sengupta S, Peterson TR, Sabatini DM. Regulation of the mTOR complex 1 pathway by nutrients, growth factors, and stress. Mol Cell. 2010;310‐322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 174. Zoncu R, Efeyan A, Sabatini DM. mTOR: from growth signal integration to cancer, diabetes and ageing. Nat Rev Mol Cell Biol. 2011;21‐35. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175. Bayascas JR, Alessi DR. Regulation of Akt/PKB Ser473 phosphorylation. Mol Cell. 2005;143‐145. [DOI] [PubMed] [Google Scholar]
- 176. Kim DH, Sarbassov DD, Ali SM, et al. mTOR interacts with raptor to form a nutrient‐sensitive complex that signals to the cell growth machinery. Cell. 2002;163‐175. [DOI] [PubMed] [Google Scholar]
- 177. Pyo JO, Nah J, Jung YK. Molecules and their functions in autophagy. Exp Mol Med. 2012;73‐80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 178. He C, Levine B. The Beclin 1 interactome. Curr Opin Cell Biol. 2010;140‐149. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 179. Caro LH, Plomp PJ, Wolvetang EJ, et al. 3‐Methyladenine, an inhibitor of autophagy, has multiple effects on metabolism. Eur J Biochem. 1988;325‐329. [DOI] [PubMed] [Google Scholar]
- 180. Yeh LC, Ford JJ, Lee JC, et al. Palmitate attenuates osteoblast differentiation of fetal rat calvarial cells. Biochem Biophys Res Commun. 2014;777‐781. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 181. Kruger MC, Coetzee M, Haag M, et al. Long‐chain polyunsaturated fatty acids: selected mechanisms of action on bone. Prog Lipid Res. 2010;438‐449. [DOI] [PubMed] [Google Scholar]
- 182. Maggio M, Artoni A, Lauretani F, et al. The impact of omega‐3 fatty acids on osteoporosis. Curr Pharm Des. 2009;4157‐4164. [DOI] [PubMed] [Google Scholar]
- 183. Calder PC. n‐3 polyunsaturated fatty acids, inflammation, and inflammatory diseases. Am J Clin Nutr. 2006;1505S‐1519S. [DOI] [PubMed] [Google Scholar]
- 184. Calder PC. n‐3 polyunsaturated fatty acids as pharmacologic agents: a fishy tale? Nutrition. 1997;1002‐1004. [DOI] [PubMed] [Google Scholar]
- 185. Katono T, Kawato T, Tanabe N, et al. Sodium butyrate stimulates mineralized nodule formation and osteoprotegerin expression by human osteoblasts. Arch Oral Biol. 2008;903‐909. [DOI] [PubMed] [Google Scholar]
- 186. Alciato F, Sainaghi PP, Sola D, et al. TNF‐alpha, IL‐6, and IL‐1 expression is inhibited by GAS6 in monocytes/macrophages. J Leukoc Biol. 2010;869‐875. [DOI] [PubMed] [Google Scholar]
- 187. Weinreb M, Shamir D, Machwate M, et al. Prostaglandin E2 (PGE2) increases the number of rat bone marrow osteogenic stromal cells (BMSC) via binding the EP4 receptor, activating sphingosine kinase and inhibiting caspase activity. Prostaglandins Leukot Essent Fatty Acids. 2006;81‐90. [DOI] [PubMed] [Google Scholar]
- 188. Tian XY, Zhang Q, Zhao R, et al. Continuous PGE2 leads to net bone loss while intermittent PGE2 leads to net bone gain in lumbar vertebral bodies of adult female rats. Bone. 2008;914‐920. [DOI] [PubMed] [Google Scholar]
- 189. Gao Q, Xu M, Alander CB, et al. Effects of prostaglandin E2 on bone in mice in vivo. Prostaglandins Other Lipid Mediat. 2009;20‐25. [DOI] [PubMed] [Google Scholar]
- 190. Minamizaki T, Yoshiko Y, Kozai K, et al. EP2 and EP4 receptors differentially mediate MAPK pathways underlying anabolic actions of prostaglandin E2 on bone formation in rat calvaria cell cultures. Bone. 2009;1177‐1185. [DOI] [PubMed] [Google Scholar]
- 191. Tsujisawa T, Inoue H, Nishihara T. SC‐19220, antagonist of prostaglandin E2 receptor EP1, inhibits osteoclastogenesis by RANKL. J Bone Miner Res. 2005;15‐22. [DOI] [PubMed] [Google Scholar]
- 192. Vaananen HK, Laitala‐Leinonen T. Osteoclast lineage and function. Arch Biochem Biophys. 2008;132‐138. [DOI] [PubMed] [Google Scholar]
- 193. Agrawal A, Buckley KA, Bowers K, et al. The effects of P2X7 receptor antagonists on the formation and function of human osteoclasts in vitro. Purinerg Signal. 2010;307‐315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194. Poulsen RC, Moughan PJ, Kruger MC. Long‐chain polyunsaturated fatty acids and the regulation of bone metabolism. Exp Biol Med. 2007;1275‐1288. [DOI] [PubMed] [Google Scholar]
- 195. Ferlin A, Pepe A, Facciolli A, et al. Relaxin stimulates osteoclast differentiation and activation. Bone. 2010;504‐513. [DOI] [PubMed] [Google Scholar]
- 196. Sorensen MG, Henriksen K, Schaller S, et al. Characterization of osteoclasts derived from CD14+ monocytes isolated from peripheral blood. J Bone Miner Metab. 2007;36‐45. [DOI] [PubMed] [Google Scholar]
- 197. Goto T, Yamaza T, Tanaka T. Cathepsins in the osteoclast. J Electron Microsc. 2003;551‐558. [DOI] [PubMed] [Google Scholar]
- 198. Johnson MR, Polymeropoulos MH, Vos HL, et al. A nonsense mutation in the cathepsin K gene observed in a family with pycnodysostosis. Genome Res. 1996;1050‐1055. [DOI] [PubMed] [Google Scholar]
- 199. Halleen JM, Tiitinen SL, Ylipahkala H, et al. Tartrate‐resistant acid phosphatase 5b (TRACP 5b) as a marker of bone resorption. Clin Lab. 2006;499‐509. [PubMed] [Google Scholar]
- 200. Oddie GW, Schenk G, Angel NZ, et al. Structure, function, and regulation of tartrate‐resistant acid phosphatase. Bone. 2000;575‐584. [DOI] [PubMed] [Google Scholar]
- 201. Sundaram K, Nishimura R, Senn J, et al. RANK ligand signaling modulates the matrix metalloproteinase‐9 gene expression during osteoclast differentiation. Exp Cell Res. 2007;168‐178. [DOI] [PubMed] [Google Scholar]
- 202. Drosatos‐Tampakaki Z, Drosatos K, Siegelin Y, et al. Palmitic acid and DGAT1 deficiency enhance osteoclastogenesis, while oleic acid‐induced triglyceride formation prevents it. J Bone Miner Res. 2014;1183‐1195. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203. Kim HJ, Ohk B, Yoon HJ, et al. Docosahexaenoic acid signaling attenuates the proliferation and differentiation of bone marrow‐derived osteoclast precursors and promotes apoptosis in mature osteoclasts. Cell Signal. 2017;226‐232. [DOI] [PubMed] [Google Scholar]
- 204. Nakamura I, Duong LT, Rodan SB, et al. Involvement of alpha(v)beta3 integrins in osteoclast function. J Bone Miner Metab. 2007;337‐344. [DOI] [PubMed] [Google Scholar]
- 205. Kasonga AE, Deepak V, Kruger MC, et al. Arachidonic acid and docosahexaenoic acid suppress osteoclast formation and activity in human CD14+ monocytes, in vitro. PLoS ONE. 2015;53:e0125145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206. Boeyens JC, Deepak V, Chua WH, et al. Effects of omega3‐ and omega6‐polyunsaturated fatty acids on RANKL‐induced osteoclast differentiation of RAW264.7 cells: a comparative in vitro study. Nutrients. 2014;2584‐2601. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207. Rahman MM, Bhattacharya A, Fernandes G. Docosahexaenoic acid is more potent inhibitor of osteoclast differentiation in RAW 264.7 cells than eicosapentaenoic acid. J Cell Physiol. 2008;201‐209. [DOI] [PubMed] [Google Scholar]
- 208. Mozaffarian D, Wu JH. Omega‐3 fatty acids and cardiovascular disease: effects on risk factors, molecular pathways, and clinical events. J Am Coll Cardiol. 2011;2047‐2067. [DOI] [PubMed] [Google Scholar]
- 209. Xing L, Chen D, Boyce BF. Mice Deficient in NF‐kappaB p50 and p52 or RANK Have Defective Growth Plate Formation and Post‐natal Dwarfism. Bone Res. 2013;336‐345. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 210. Kim K, Lee SH, Ha Kim J, et al. NFATc1 induces osteoclast fusion via up‐regulation of Atp6v0d2 and the dendritic cell‐specific transmembrane protein (DC‐STAMP). Mol Endocrinol. 2008;176‐185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211. Hutchins‐Wiese HL, Picho K, Watkins BA, et al. High‐dose eicosapentaenoic acid and docosahexaenoic acid supplementation reduces bone resorption in postmenopausal breast cancer survivors on aromatase inhibitors: a pilot study. Nutr Cancer. 2014;68‐76. [DOI] [PubMed] [Google Scholar]
- 212. Song J, Jing Z, Hu W, et al. alpha‐linolenic acid inhibits receptor activator of NF‐kappaB ligand induced (RANKL‐Induced) osteoclastogenesis and prevents inflammatory bone loss via downregulation of nuclear factor‐KappaB‐inducible nitric oxide synthases (NF‐kappaB‐iNOS) signaling pathways. Med Sci Monit. 2017;5056‐5069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213. Parvaneh K, Jamaluddin R, Karimi G, et al. Effect of probiotics supplementation on bone mineral content and bone mass density. Sci World J. 2014;53:595962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214. Kim HJ, Yoon HJ, Kim SY, et al. A medium‐chain fatty acid, capric acid, inhibits RANKL‐induced osteoclast differentiation via the suppression of NF‐kappaB signaling and blocks cytoskeletal organization and survival in mature osteoclasts. Mol Cells. 2014;598‐604. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215. Liu C, Guan H, Cai C, et al. Lipoxin A4 suppresses osteoclastogenesis in RAW264.7 cells and prevents ovariectomy‐induced bone loss. Exp Cell Res. 2017;293‐303. [DOI] [PubMed] [Google Scholar]
- 216. Ono K, Kaneko H, Choudhary S, et al. Biphasic effect of prostaglandin E2 on osteoclast formation in spleen cell cultures: role of the EP2 receptor. J Bone Miner Res. 2005;23‐29. [DOI] [PubMed] [Google Scholar]
- 217. Li X, Okada Y, Pilbeam CC, et al. Knockout of the murine prostaglandin EP2 receptor impairs osteoclastogenesis in vitro. Endocrinology. 2000;2054‐2061. [DOI] [PubMed] [Google Scholar]
- 218. Liu XH, Kirschenbaum A, Yao S, et al. Interactive effect of interleukin‐6 and prostaglandin E2 on osteoclastogenesis via the OPG/RANKL/RANK system. Ann N Y Acad Sci. 2006;225‐233. [DOI] [PubMed] [Google Scholar]
- 219. Suda K, Udagawa N, Sato N, et al. Suppression of osteoprotegerin expression by prostaglandin E2 is crucially involved in lipopolysaccharide‐induced osteoclast formation. J Immunol. 2004;2504‐2510. [DOI] [PubMed] [Google Scholar]
- 220. Long MW. Osteogenesis and bone‐marrow‐derived cells. Blood Cells Mol Dis. 2001;677‐690. [DOI] [PubMed] [Google Scholar]
- 221. Miura M, Chen XD, Allen MR, et al. A crucial role of caspase‐3 in osteogenic differentiation of bone marrow stromal stem cells. J Clin Invest. 2004;1704‐1713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222. Levental KR, Surma MA, Skinkle AD, et al. omega‐3 polyunsaturated fatty acids direct differentiation of the membrane phenotype in mesenchymal stem cells to potentiate osteogenesis. Sci Adv. 2017;53:eaao1193. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223. Canalis E, Mazziotti G, Giustina A, et al. Glucocorticoid‐induced osteoporosis: pathophysiology and therapy. Osteoporos Int. 2007;1319‐1328. [DOI] [PubMed] [Google Scholar]
- 224. Gao B, Huang Q, Jie Q, et al. Ginsenoside‐Rb2 inhibits dexamethasone‐induced apoptosis through promotion of GPR120 induction in bone marrow‐derived mesenchymal stem cells. Stem Cells Dev. 2015;781‐790. [DOI] [PubMed] [Google Scholar]
- 225. Gao B, Han YH, Wang L, et al. Eicosapentaenoic acid attenuates dexamethasome‐induced apoptosis by inducing adaptive autophagy via GPR120 in murine bone marrow‐derived mesenchymal stem cells. Cell Death Dis. 2016;53:e2235. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 226. Gillet C, Spruyt D, Rigutto S, et al. Oleate abrogates palmitate‐induced lipotoxicity and proinflammatory response in human bone marrow‐derived mesenchymal stem cells and osteoblastic cells. Endocrinology. 2015;4081‐4093. [DOI] [PubMed] [Google Scholar]
- 227. Lu J, Wang Q, Huang L, et al. Palmitate causes endoplasmic reticulum stress and apoptosis in human mesenchymal stem cells: prevention by AMPK activator. Endocrinology. 2012;5275‐5284. [DOI] [PubMed] [Google Scholar]
- 228. Mundy GR. Osteoporosis and inflammation. Nutr Rev. 2007;S147‐151. [DOI] [PubMed] [Google Scholar]
- 229. Tamura T, Udagawa N, Takahashi N, et al. Soluble interleukin‐6 receptor triggers osteoclast formation by interleukin 6. Proc Natl Acad Sci USA. 1993;11924‐11928. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 230. Bendre MS, Montague DC, Peery T, et al. Interleukin‐8 stimulation of osteoclastogenesis and bone resorption is a mechanism for the increased osteolysis of metastatic bone disease. Bone. 2003;28‐37. [DOI] [PubMed] [Google Scholar]
- 231. Houssiau FA, Devogelaer JP, Van Damme J, et al. Interleukin‐6 in synovial fluid and serum of patients with rheumatoid arthritis and other inflammatory arthritides. Arthritis Rheum. 1988;784‐788. [DOI] [PubMed] [Google Scholar]
- 232. Lisignoli G, Toneguzzi S, Pozzi C, et al. Proinflammatory cytokines and chemokine production and expression by human osteoblasts isolated from patients with rheumatoid arthritis and osteoarthritis. J Rheumatol. 1999;791‐799. [PubMed] [Google Scholar]
- 233. Zofkova I. Bone tissue as a systemic endocrine regulator. Physiol Res. 2015;439‐445. [DOI] [PubMed] [Google Scholar]
- 234. Al Saedi A, Bermeo S, Plotkin L, et al. Mechanisms of palmitate‐induced lipotoxicity in osteocytes. Bone. 2019;353‐359. [DOI] [PubMed] [Google Scholar]
- 235. Wang N, Xue P, Wu X, et al. Role of sclerostin and dkk1 in bone remodeling in type 2 diabetic patients. Endocr Res. 2018;29‐38. [DOI] [PubMed] [Google Scholar]
- 236. Li L, Yang Z, Zhang H, et al. Low‐intensity pulsed ultrasound regulates proliferation and differentiation of osteoblasts through osteocytes. Biochem Bioph Res Commun. 2012;296‐300. [DOI] [PubMed] [Google Scholar]
- 237. Vazquez M, Evans BA, Riccardi D, et al. A new method to investigate how mechanical loading of osteocytes controls osteoblasts. Front Endocrinol. 2014;208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 238. Armstrong VJ, Muzylak M, Sunters A, et al. Wnt/beta‐catenin signaling is a component of osteoblastic bone cell early responses to load‐bearing and requires estrogen receptor alpha. J Biol Chem. 2007;20715‐20727. [DOI] [PubMed] [Google Scholar]
- 239. Riquelme MA, Burra S, Kar R, et al. Mitogen‐activated protein kinase (MAPK) activated by prostaglandin E2 phosphorylates CONNEXIN 43 and closes osteocytic hemichannels in response to continuous flow shear stress. J Biol Chem. 2015;28321‐28328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 240. Nagayama K, Miyamoto Y, Kaneko K, et al. Production of 8‐nitro‐cGMP in osteocytic cells and its upregulation by parathyroid hormone and prostaglandin E2. Vitro Cell Dev Biol Anim. 2019;45‐51. [DOI] [PubMed] [Google Scholar]
- 241. Frey JL, Li Z, Ellis JM, et al. Wnt‐Lrp5 signaling regulates fatty acid metabolism in the osteoblast. Mol Cell Biol. 2015;1979‐1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 242. Li Z, Frey JL, Wong GW, et al. Glucose transporter‐4 facilitates insulin‐stimulated glucose uptake in osteoblasts. Endocrinology. 2016;4094‐4103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 243. Lee TJ, Jang J, Kang S, et al. Enhancement of osteogenic and chondrogenic differentiation of human embryonic stem cells by mesodermal lineage induction with BMP‐4 and FGF2 treatment. Biochem Bioph Res Co. 2013;793‐797. [DOI] [PubMed] [Google Scholar]
- 244. Damek‐Poprawa M, Golub E, Otis L, et al. Chondrocytes utilize a cholesterol‐dependent lipid translocator to externalize phosphatidylserine. Biochemistry. 2006;3325‐3336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 245. Villalvilla A, Gomez R, Largo R, et al. Lipid transport and metabolism in healthy and osteoarthritic cartilage. Int J Mol Sci. 2013;20793‐20808. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246. Wann AK, Mistry J, Blain EJ, et al. Eicosapentaenoic acid and docosahexaenoic acid reduce interleukin‐1beta‐mediated cartilage degradation. Arthritis Res Ther. 2010;R207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 247. Loef M, Schoones JW, Kloppenburg M, et al. Fatty acids and osteoarthritis: different types, different effects. Joint Bone Spine. 2019;451‐458. [DOI] [PubMed] [Google Scholar]
- 248. Sakata S, Hayashi S, Fujishiro T, et al. Oxidative stress‐induced apoptosis and matrix loss of chondrocytes is inhibited by eicosapentaenoic acid. J Orthop Res. 2015;359‐365. [DOI] [PubMed] [Google Scholar]
- 249. Wang Z, Guo A, Ma L, et al. Docosahexenoic acid treatment ameliorates cartilage degeneration via a p38 MAPK‐dependent mechanism. Int J Mol Med. 2016;1542‐1550. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 250. Serhan CN. Pro‐resolving lipid mediators are leads for resolution physiology. Nature. 2014;92‐101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 251. Perretti M, Norling LV. Actions of SPM in regulating host responses in arthritis. Mol Aspects Med. 2017;57‐64. [DOI] [PubMed] [Google Scholar]
- 252. Norling LV, Headland SE, Dalli J, et al. Proresolving and cartilage‐protective actions of resolvin D1 in inflammatory arthritis. JCI Insight. 2016;53:e85922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 253. Arnardottir HH, Dalli J, Norling LV, et al. Resolvin D3 is dysregulated in arthritis and reduces arthritic Inflammation. J Immunol. 2016;2362‐2368. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 254. Benabdoune H, Rondon EP, Shi Q, et al. The role of resolvin D1 in the regulation of inflammatory and catabolic mediators in osteoarthritis. Inflamm Res. 2016;635‐645. [DOI] [PubMed] [Google Scholar]
- 255. McReynolds CB, Hwang SH, Yang J, et al. Pharmaceutical effects of inhibiting the soluble epoxide hydrolase in canine osteoarthritis. Front Pharmacol. 2019;533. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 256. Chabane N, Zayed N, Afif H, et al. Histone deacetylase inhibitors suppress interleukin‐1beta‐induced nitric oxide and prostaglandin E2 production in human chondrocytes. Osteoarthritis Cartilage. 2008;1267‐1274. [DOI] [PubMed] [Google Scholar]
- 257. Young DA, Lakey RL, Pennington CJ, et al. Histone deacetylase inhibitors modulate metalloproteinase gene expression in chondrocytes and block cartilage resorption. Arthritis Res Ther. 2005;R503‐512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 258. Bo W, Zhou J, Wang K. Sodium butyrate abolishes the degradation of type II collagen in human chondrocytes. Biomed Pharmacother. 2018;1099‐1104. [DOI] [PubMed] [Google Scholar]
- 259. Pirozzi C, Francisco V, Guida FD, et al. Butyrate modulates inflammation in chondrocytes via GPR43 receptor. Cell Physiol Biochem. 2018;228‐243. [DOI] [PubMed] [Google Scholar]
- 260. Sekar S, Shafie SR, Prasadam I, et al. Saturated fatty acids induce development of both metabolic syndrome and osteoarthritis in rats. Sci Rep. 2017;46457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 261. Wu CL, Diekman BO, Jain D, et al. Diet‐induced obesity alters the differentiation potential of stem cells isolated from bone marrow, adipose tissue and infrapatellar fat pad: the effects of free fatty acids. Int J Obes. 2013;1079‐1087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 262. Frommer KW, Schaffler A, Rehart S, et al. Free fatty acids: potential proinflammatory mediators in rheumatic diseases. Ann Rheum Dis. 2015;303‐310. [DOI] [PubMed] [Google Scholar]
- 263. Miao H, Chen L, Hao L, et al. Stearic acid induces proinflammatory cytokine production partly through activation of lactate‐HIF1alpha pathway in chondrocytes. Sci Rep. 2015;13092. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 264. Xie Q, Wei M, Kang X, et al. Reciprocal inhibition between miR‐26a and NF‐kappaB regulates obesity‐related chronic inflammation in chondrocytes. Biosci Rep. 2015;144‐152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 265. Sekar S, Wu X, Friis T, et al. Saturated fatty acids promote chondrocyte matrix remodeling through reprogramming of autophagy pathways. Nutrition. 2018;54:144‐152. [DOI] [PubMed] [Google Scholar]
- 266. Alvarez‐Garcia O, Rogers NH, Smith RG, et al. Palmitate has proapoptotic and proinflammatory effects on articular cartilage and synergizes with interleukin‐1. Arthritis Rheumatol. 2014;1779‐1788. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 267. Herrero‐Beaumont G, Perez‐Baos S, Sanchez‐Pernaute O, et al. Targeting chronic innate inflammatory pathways, the main road to prevention of osteoarthritis progression. Biochem Pharmacol. 2019;24‐32. [DOI] [PubMed] [Google Scholar]
- 268. Walter P, Ron D. The unfolded protein response: from stress pathway to homeostatic regulation. Science. 2011;1081‐1086. [DOI] [PubMed] [Google Scholar]
- 269. Nazli SA, Loeser RF, Chubinskaya S, et al. High fat‐diet and saturated fatty acid palmitate inhibits IGF‐1 function in chondrocytes. Osteoarthritis Cartilage. 2017;1516‐1521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 270. Loeser RF. Growth factor regulation of chondrocyte integrins. Differential effects of insulin‐like growth factor 1 and transforming growth factor beta on alpha 1 beta 1 integrin expression and chondrocyte adhesion to type VI collagen. Arthritis Rheum. 1997;270‐276. [DOI] [PubMed] [Google Scholar]
- 271. Haywood J, Yammani RR. Free fatty acid palmitate activates unfolded protein response pathway and promotes apoptosis in meniscus cells. Osteoarthritis Cartilage. 2016;942‐945. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 272. Tan L, Yammani RR. Nupr1 regulates palmitate‐induced apoptosis in human articular chondrocytes. Biosci Rep. 2019;2076‐2083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 273. Wu CL, Jain D, McNeill JN, et al. Dietary fatty acid content regulates wound repair and the pathogenesis of osteoarthritis following joint injury. Ann Rheum Dis. 2015;74:2076‐2083. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 274. Patterson E, Wall R, Fitzgerald GF, et al. Health implications of high dietary omega‐6 polyunsaturated Fatty acids. J Nutr Metab. 2012;53:539426. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 275. Li TF, Zuscik MJ, Ionescu AM, et al. PGE2 inhibits chondrocyte differentiation through PKA and PKC signaling. Exp Cell Res. 2004;159‐169. [DOI] [PubMed] [Google Scholar]
- 276. Jung ID, Jeong YI, Lee CM, et al. COX‐2 and PGE2 signaling is essential for the regulation of IDO expression by curcumin in murine bone marrow‐derived dendritic cells. Int Immunopharmacol. 2010;760‐768. [DOI] [PubMed] [Google Scholar]
- 277. Weiler HA, Fitzpatrick‐Wong S. Dietary long‐chain polyunsaturated fatty acids minimize dexamethasone‐induced reductions in arachidonic acid status but not bone mineral content in piglets. Pediatr Res. 2002;282‐289. [DOI] [PubMed] [Google Scholar]
- 278. Lv S, Wu L, Cheng P, et al. Correlation of obesity and osteoporosis: effect of free fatty acids on bone marrow‐derived mesenchymal stem cell differentiation. Exp Ther Med. 2010;603‐610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 279. Chen TY, Zhang ZM, Zheng XC, et al. Endogenous n‐3 polyunsaturated fatty acids (PUFAs) mitigate ovariectomy‐induced bone loss by attenuating bone marrow adipogenesis in FAT1 transgenic mice. Drug Des Devel Ther. 2013;545‐552. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 280. Rahman MM, Bhattacharya A, Banu J, et al. Endogenous n‐3 fatty acids protect ovariectomy induced bone loss by attenuating osteoclastogenesis. J Cell Mol Med. 2009;1833‐1844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 281. Shen CL, Yeh JK, Rasty J, et al. Protective effect of dietary long‐chain n‐3 polyunsaturated fatty acids on bone loss in gonad‐intact middle‐aged male rats. Br J Nutr. 2006;462‐468. [DOI] [PubMed] [Google Scholar]
- 282. Bullon P, Battino M, Varela‐Lopez A, et al. Diets based on virgin olive oil or fish oil but not on sunflower oil prevent age‐related alveolar bone resorption by mitochondrial‐related mechanisms. PLoS ONE. 2013;53:e74234. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 283. Maditz KH, Smith BJ, Miller M, et al. Feeding soy protein isolate and oils rich in omega‐3 polyunsaturated fatty acids affected mineral balance, but not bone in a rat model of autosomal recessive polycystic kidney disease. BMC Nephrol. 2015;13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 284. Knott L, Avery NC, Hollander AP, et al. Regulation of osteoarthritis by omega‐3 (n‐3) polyunsaturated fatty acids in a naturally occurring model of disease. Osteoarthritis Cartilage. 2011;1150‐1157. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 285. Alnouri DM, El‐Din MF, Al‐Khalifa AS. The effect of long‐term supplementation with different dietary omega‐6/omega‐3 ratios on mineral content and ex vivo prostaglandin E2 release in bone of growing rabbits. Nutr Res Pract. 2014;360‐367. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 286. Ribeiro DC, Pereira AD, de Santana FC, et al. Incorporation of flaxseed flour as a dietary source for ALA increases bone density and strength in post‐partum female rats. Lipids. 2017;327‐333. [DOI] [PubMed] [Google Scholar]
- 287. Khadge S, Thiele GM, Sharp JG, et al. Long‐chain omega‐3 polyunsaturated fatty acids decrease mammary tumor growth, multiorgan metastasis and enhance survival. Clin Exp Metastasis. 2018;797‐818. [DOI] [PubMed] [Google Scholar]
- 288. Varela‐Lopez A, Ochoa JJ, Llamas‐Elvira JM, et al. Loss of bone mineral density associated with age in male rats fed on sunflower oil is avoided by virgin olive oil intake or coenzyme Q supplementation. Int J Mol Sci. 2017;213‐218. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 289. Rosenstein ED, Kushner LJ, Kramer N, et al. Pilot study of dietary fatty acid supplementation in the treatment of adult periodontitis. Prostaglandins Leukot Essent Fatty Acids. 2003;68:213‐218. [DOI] [PubMed] [Google Scholar]
- 290. Elkhouli AM. The efficacy of host response modulation therapy (omega‐3 plus low‐dose aspirin) as an adjunctive treatment of chronic periodontitis (clinical and biochemical study). J Periodontal Res. 2011;261‐268. [DOI] [PubMed] [Google Scholar]
- 291. Remans PH, Sont JK, Wagenaar LW, et al. Nutrient supplementation with polyunsaturated fatty acids and micronutrients in rheumatoid arthritis: clinical and biochemical effects. Eur J Clin Nutr. 2004;839‐845. [DOI] [PubMed] [Google Scholar]
- 292. Hagfors L, Nilsson I, Skoldstam L, et al. Fat intake and composition of fatty acids in serum phospholipids in a randomized, controlled, Mediterranean dietary intervention study on patients with rheumatoid arthritis. Nutr Metab. 2005;26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 293. Leeb BF, Sautner J, Andel I, et al. Intravenous application of omega‐3 fatty acids in patients with active rheumatoid arthritis. The ORA‐1 trial. An open pilot study. Lipids. 2006;29‐34. [DOI] [PubMed] [Google Scholar]
- 294. Galarraga B, Ho M, Youssef HM, et al. Cod liver oil (n‐3 fatty acids) as an non‐steroidal anti‐inflammatory drug sparing agent in rheumatoid arthritis. Rheumatology. 2008;665‐669. [DOI] [PubMed] [Google Scholar]
- 295. Bahadori B, Uitz E, Thonhofer R, et al. omega‐3 Fatty acids infusions as adjuvant therapy in rheumatoid arthritis. J Parenter Enteral Nutr. 2010;151‐155. [DOI] [PubMed] [Google Scholar]
- 296. Proudman SM, Cleland LG, Metcalf RG, et al. Plasma n‐3 fatty acids and clinical outcomes in recent‐onset rheumatoid arthritis. Br J Nutr. 2015;885‐890. [DOI] [PubMed] [Google Scholar]
- 297. Gan RW, Demoruelle MK, Deane KD, et al. Omega‐3 fatty acids are associated with a lower prevalence of autoantibodies in shared epitope‐positive subjects at risk for rheumatoid arthritis. Ann Rheum Dis. 2017;147‐152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 298. Perevozchikova TV, Avdeeva EY, Fait EA, et al. Influence of saussurea controversa and fillipendula ulmaria extracts on immunological reactivity of rats with experimental osteomyelitis. Eksp Klin Farmakol. 2016;16‐20. [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The data that support the findings of this study are available from the corresponding author upon reasonable request.


