Abstract
STUDY QUESTION
Does embryo transfer medium containing hyaluronate (HA) promote the attachment phase of human embryo implantation?
SUMMARY ANSWER
HA-containing medium does not promote human blastocyst attachment to endometrial epithelial cells in vitro.
WHAT IS KNOWN ALREADY
Embryo transfer media containing high concentrations of HA are being used to increase implantation and live birth rates in IVF treatment, although the mechanism of action is unknown.
STUDY DESIGN, SIZE, DURATION
Expression of HA-interacting genes in frozen-thawed oocytes/embryos was assessed by microarray analysis (n = 21). Fresh and frozen human blastocysts (n = 98) were co-cultured with human endometrial epithelial Ishikawa cell layers. Blastocyst attachment and the effects of a widely used HA-containing medium were measured.
PARTICIPANTS/MATERIALS, SETTING, METHODS
Human embryos surplus to treatment requirements were donated with informed consent from several ART centres. Blastocyst-stage embryos were transferred at day 6 to confluent Ishikawa cell layers; some blastocysts were artificially hatched. Blastocyst attachment was monitored from 1 to 48 h, and the effects of blastocyst pre-treatment for 10 min with HA-containing medium were determined.
MAIN RESULTS AND THE ROLE OF CHANCE
Human embryos expressed the HA receptor genes CD44 and HMMR, hyaluronan synthase genes HAS1–3, and hyaluronidase genes HYAL1–3, at all stages of preimplantation development. Attachment of partially hatched blastocysts to Ishikawa cells at 24 and 48 h was related to trophectoderm grade (P = 0.0004 and 0.007, respectively, n = 34). Blastocysts of varying clinical grades that had been artificially hatched were all attached within 48 h (n = 21). Treatment of artificially hatched blastocysts with HA-containing medium did not significantly affect attachment at early (1–6 h) or late (24 and 48 h) time points, compared with control blastocysts (n = 43).
LIMITATIONS, REASONS FOR CAUTION
Using an adenocarcinoma-derived cell line to model embryo-endometrium attachment may not fully recapitulate in vivo interactions. The high levels of blastocyst attachment seen with this in vitro model may limit the sensitivity with which the effects of HA can be observed.
WIDER IMPLICATIONS OF THE FINDINGS
Morphological trophectoderm grade can be correlated with blastocyst attachment in vitro. HA-containing medium may increase pregnancy rates by mechanisms other than promoting blastocyst attachment to endometrium.
STUDY FUNDING/COMPETING INTEREST(S)
This work was funded by a grant from the Wellbeing of Women, the NIHR Local Comprehensive Research Network and NIHR Manchester Clinical Research Facility, the Department of Health Scientist Practitioner Training Scheme, and the Ministry of Higher Education, The State of Libya. None of the authors has any conflict of interest to declare.
Keywords: ART, hyaluronate-containing medium, add-ons, human embryo, embryology, embryo quality, implantation, trophectoderm
WHAT DOES THIS MEAN FOR PATIENTS?
Hyaluronate (HA) is a naturally occurring substance found throughout the body, which has been used as an additive to the liquid solution in which embryos are transferred into the womb as part of an IVF cycle. There is evidence from clinical studies showing that adding HA to this transfer medium improves live birth rates, and it has been proposed that it helps the embryo attach to the wall of the womb. The most commonly used HA-containing transfer medium is called EmbryoGlue®, in accordance with this proposed mode of action. Such embryo transfer media are widely offered to patients as so-called ‘add-ons’ to their IVF treatment. However, there have been no direct studies demonstrating that HA acts on the attachment of embryos to the endometrial epithelial cells which line the wall of the womb.
In this study, human embryos donated to research at different stages were shown to have the biological machinery required to respond to HA. The attachment of day 6 embryos to endometrial epithelial cells grown in a dish was carefully assessed, and adding EmbryoGlue® to the solution did not affect attachment when compared with embryos in control transfer medium without EmbryoGlue®. These experiments suggest that HA-containing embryo transfer medium might not directly promote embryo attachment to the womb lining but might improve IVF success rates in a different way, for example by promoting embryo growth. Further research is required in this area.
Introduction
Current evidence suggests that the type of commercially available embryo culture media used in ART is not associated with success rates (Mantikou et al. 2013, Youssef et al. 2015). However, when used at the point of embryo transfer, there is evidence that media containing high levels of hyaluronate (HA) increase live birth rate per transfer and implantation rate per embryo (Bontekoe et al. 2014, Farquhar and Marjoribanks 2018). HA is a glycosaminoglycan prevalent in follicular, oviductal and uterine fluid, and the oocyte cumulus extracellular matrix (Nagyova 2018). HA levels are increase in the endometrial stroma during the secretory phase of the menstrual cycle and fall to very low levels at menstruation (Carson et al. 1987, Salamonsen et al. 2001), suggestive of a role in implantation. Thus, HA is considered a physiologically relevant additive to embryo medium with a relatively low risk profile (Harper et al. 2012, Brison et al. 2013). However, despite initial data from a mouse model, two decades ago, that showed increased rates of implantation and foetal development in HA-containing embryo transfer medium (Gardner et al. 1999), and clinical evidence that has led to a widespread use of HA-containing medium in ART (Bontekoe et al. 2014), the mechanism of action of HA at implantation is not known.
HA has a range of biological functions, including stimulation of cell proliferation, migration and adhesion (Block et al. 2009), and control of swelling and shape in collagenous extracellular matrices and the pericellular environment (Wight 2017). HA is synthesised as various relative molecular masses (Mr) by synthase enzymes HAS1–3, while hyaluronidases HYAL1–3 cleave high Mr forms of HA into low Mr forms that have trophic signalling properties: receptors CD44, hyaluronan mediated mobility receptor (HMMR) and Toll-like receptor 4 (TLR4) bind HA at the cell surface (Tavianatou et al. 2019). In mouse, the addition of an HAS inhibitor negatively impacts blastocyst formation, and in bovine, the addition of Hyal2 to embryo culture medium improves development in a CD44-dependent manner (Fouladi-Nashta et al. 2017, Marei et al. 2017). However, little is known of the impacts of the HA system during human preimplantation development, other than the demonstration of HAS2, CD44 and HMMR expression in embryos (Campbell et al. 1995, Choudhary et al. 2007, Fouladi-Nashta et al. 2017).
An intriguing hypothesis posits that high Mr HA acts as an embryo attachment ‘glue’, facilitating the first interaction between blastocyst-stage embryo trophectoderm (TE) and endometrial epithelial cells by bridging between receptors on both surfaces (Gardner et al. 1999, Bontekoe et al. 2014, Aplin and Ruane 2017). Indeed, HA-containing embryo transfer media are sold under commercial trade names (e.g. EmbryoGlue®) implying this mode of action. Conversely, such media improve clinical ART success rate regardless of the developmental stage at which embryos are transferred to the uterus (Bontekoe et al. 2014). Embryos transferred at day 3 in the presence of HA are unlikely to benefit from an attachment ‘glue’ mode of action since attachment is not initiated until 2–3 days later. In support of an alternative hypothesis, bovine data reveal that HA may moderate implantation (Fouladi-Nashta et al. 2017, Marei et al. 2017). Similarly, we have recently shown that the enzymatic reduction of high Mr HA in endometrial epithelial cell cultures promotes mouse blastocyst attachment in an in vitro model of implantation (Berneau et al. 2019).
Studying human embryo implantation directly requires the use of in vitro models. Interactions between blastocysts and primary human epithelial cells in vitro have been previously reported (Lindenberg et al. 1985, Lindenberg et al. 1986, Bentin-Ley et al. 2000, Galan et al. 2000, Meseguer et al. 2001, Petersen et al. 2005a, Lalitkumar et al. 2007, Lalitkumar et al. 2013, Berger et al. 2015, Boggavarapu et al. 2016). However, the 2D culture phenotype of primary endometrial epithelial cells does not recapitulate the in vivo epithelium (Campbell et al. 2000). Moreover, the kinetics of blastocyst attachment at implantation have not been evaluated in such models. The human endometrial adenocarcinoma Ishikawa cell line (Nishida et al. 1985) offers a controlled in vitro epithelial system to study human embryo implantation, which is thought to begin on day 6 of embryo development (Kang et al. 2014, Aberkane et al. 2018). Ishikawa cells are moderately polarised (Chitcholtan et al. 2013, Buck et al. 2015), show a surface composition that resembles endometrial epithelium (Singh and Aplin 2015) and have proven receptivity to rodent blastocysts (Green et al. 2015, Ruane et al. 2017).
In light of the uncertainties surrounding HA activity on human embryos and implantation, we designed a study to investigate the effect of a commercial HA-containing medium (EmbryoGlue®) on human embryo attachment to Ishikawa cells. We find no evidence that HA transfer medium promotes embryo attachment.
Materials and Methods
Human embryos
Embryos unsuitable for treatment (based on morphological grade) from current ART cycles (fresh) or surplus to treatment requirements from previous ART cycles (frozen) were obtained with informed written consent from patients at Old St Marys Hospital, Manchester, or other IVF units in the north west of England, in accordance with ethics approval from the NRES committee south central (Berkshire) (REC reference: 12/SC/0649) and a research licence from the Human Fertilisation and Embryology Authority (HFEA; R0026), centre 0067 (Old St Mary’s Hospital; fresh embryo research) and the University of Manchester (0175; frozen-thawed embryo research). Embryos frozen at either the pronuclear (PN) or early cleavage stages were thawed using ThawKit Cleave (Vitrolife, Gothenburg, Sweden) according to the manufacturer’s instructions. None of the embryos used were from PGT cycles. Embryos were cultured in G1 and G2 sequential media or GTL continuous culture medium (Vitrolife) to day 6 post-fertilisation and graded using a standardised scheme (Cutting et al. 2008, ASiRMaESIGo 2011). Only embryos exhibiting blastocyst morphology on day 6 were used. Artificial hatching was performed by passing blastocysts through three drops of acid Tyrodes buffer, pH 2.5, (Sigma-Aldrich, Poole, UK). Removal of the zona pellucida (ZP) was assessed by microscopy before washing in G2 or GTL medium.
Microarray analysis of oocyte and embryo gene expression
The microarray analysis reported here (GeneChip® Human Genome U133 Plus 2.0 Array [Life Technolgies, Paisley, UK]) was performed previously (Shaw et al. 2013, Smith et al. 2019). cDNA samples were obtained from individual human oocyte, 4-cell, 8-cell single blastomeres, intact 8-cell, whole blastocyst, TE and inner cell mass (ICM) samples. The lower threshold of positive relative gene expression was set at 5.64, based on MAS5 normalisation.
EmbryoGlue® treatment of blastocysts
EmbryoGlue® (Vitrolife) was used as a high-HA medium, as it contains 0.5 mg/ml HA compared with 0.125 mg/ml HA in G2 embryo culture medium (Fancsovits et al. 2015). We were unable to obtain information from the manufacturer on the Mr profile of HA in EmbryoGlue® or a suitable control medium lacking HA. Day 6 blastocysts were treated with EmbryoGlue® or control medium (GTL) by 10 min incubation (as suggested in the clinical protocol) after hatching. Blastocysts were added directly to Ishikawa cells in a 3-μl treatment medium.
Blastocyst attachment assay
Ishikawa cells (ECACC 99040201) were grown to confluence on glass coverslips in 24-well plates coated with 2% Matrigel (Sigma) in growth medium (1:1 DMEM:Ham’s F12, 10% foetal bovine serum, 2-mM l glutamine, 100-ug/ml streptomycin, 100-IU/ml penicillin) (Sigma) at 37°C and in 95% air and 5% CO2. Cells were washed and replenished with serum-free growth medium immediately prior to the transfer of single day 6 blastocysts to individual wells and co-culture at 37°C, 95% air and 5% CO2. We have previously determined mouse blastocyst attachment stability to Ishikawa cells based on the levels of oscillation upon agitation (Ruane et al. 2017), however, the larger size of day 6–8 human blastocysts prevented such grading of attachment stability due to the oscillation of embryos despite visible, substantial areas of attachment (Fig. 1, arrows). Therefore, embryos were scored simply as ‘attached’ or ‘unattached’ during co-culture. All co-cultures were fixed at 48 h with 4% formaldehyde for 30 min, at which time attachment was re-classified as ‘stably attached’ (remaining attached at fixation), ‘weakly attached’ (detached at fixation) or ‘unattached’ (Singh et al. 2010).
Figure 1.
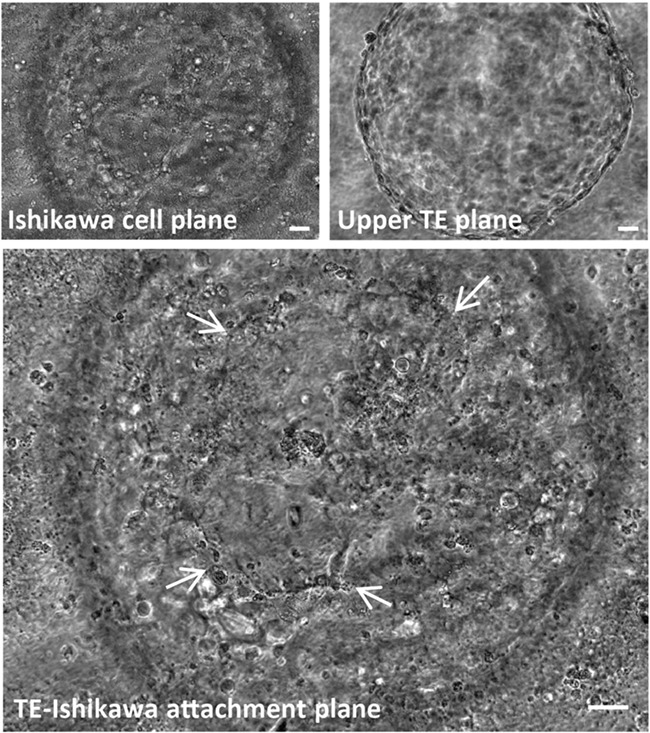
Phase contrast image of a live day 8, human blastocyst attached to Ishikawa cells after 48 h in co-culture. An area of trophectoderm (TE)-mediated attachment to Ishikawa cells (arrows) is visible. Scale bars 20 μm.
Statistical analysis
Attachment rates and embryo grades were analysed using the Chi-squared test. Repeated Chi-squared analysis with a Bonferroni correction for multiple comparisons was used to assess the effect of EmbryoGlue® treatment on embryo attachment at each experimental time point. Statistical significance was set as P < 0.05 and measured using SPSS (IBM, Portsmouth, UK).
Results
Expression of HA system components in pre-implantation human embryos
We screened our existing human pre-implantation embryo transcriptome data (Shaw et al. 2013, Smith et al. 2019) for the major components of the HA system. HA receptors CD44 and HMMR (Fig. 2A and B), but not TLR4 (data not shown), were expressed at all stages, including oocyte, 4-cell, 8-cell and blastocyst, and equally in TE compared with ICM. The HA synthase enzymes, HAS1–3, were expressed at the above stages, although HAS2 was weakest at 8-cell and blastocyst stages (Fig. 2C–E). Hyaluronidases HYAL1–3 were expressed in both ICM and TE, although HYAL1 was low or absent at earlier stages (Fig. 2F–H). Human blastocyst expression was confirmed by quantitative PCR analysis for two genes selected at random, HMMR and HYAL1 (unpublished data). These data suggest that HA receptors and systems for synthesising and degrading HA are present throughout pre-implantation development.
Figure 2.
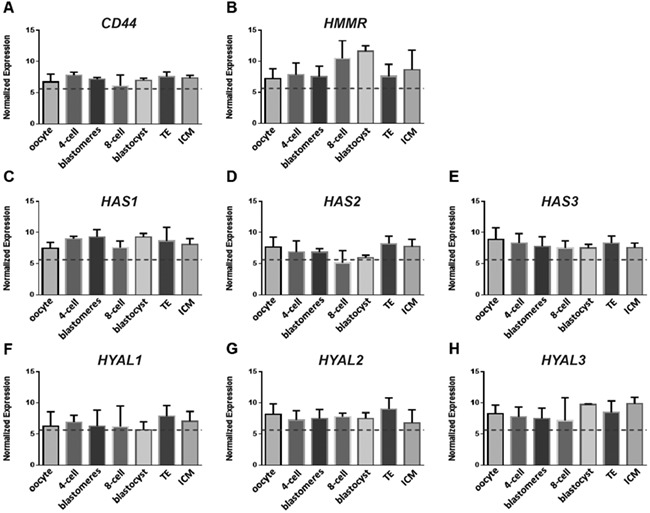
Gene expression of the hyaluronate (HA) system. Expression was studied by the microarray analysis of individual human oocytes (n = 4), 4-cell embryos (n = 4), 8-cell stage blastomeres (n = 8 blastomeres from one embryo), intact 8-cell embryos (n = 3), intact blastocysts (n = 4) and isolated ICM and TE samples (n = 6 each), using Mas5 normalisation. Data are mean ±SEM, with the lower threshold of positive relative gene expression set at 5.64 (dotted line), based on MAS5 normalisation. HA receptor genes CD44 and HMMR, hyaluronan synthase genes HAS1–3 and hyaluronidase genes HYAL1–3.
Hatching status and TE grade predict blastocyst attachment to Ishikawa cells in an in vitro model of implantation
In order to examine the effect of HA transfer medium, we first characterised the attachment of day 6 human blastocysts to Ishikawa cells. Co-cultures were monitored after 24 and 48 h; 7/17 (41.2%) blastocysts from current IVF cycles (fresh embryos) had attached to Ishikawa cells after 24-h co-culture. By 48 h, 6/14 (42.9%) were attached: 5/14 (35.7%) weakly and 1/14 (7.1%) stably (Fig. 3A). Frozen embryos were developed to day 6 after thawing at either PN (13/17) or at day 2 (4/17). We observed that 9/17 (52.9%) of these blastocysts were attached to the cells after 24-h co-culture, and at 48 h, 2/15 (13.3%) displayed weak attachment and 8/15 (53.3%) stable attachment (Fig. 3B). Stable attachment rates were higher for frozen compared with fresh embryos (P = 0.007).
Figure 3.
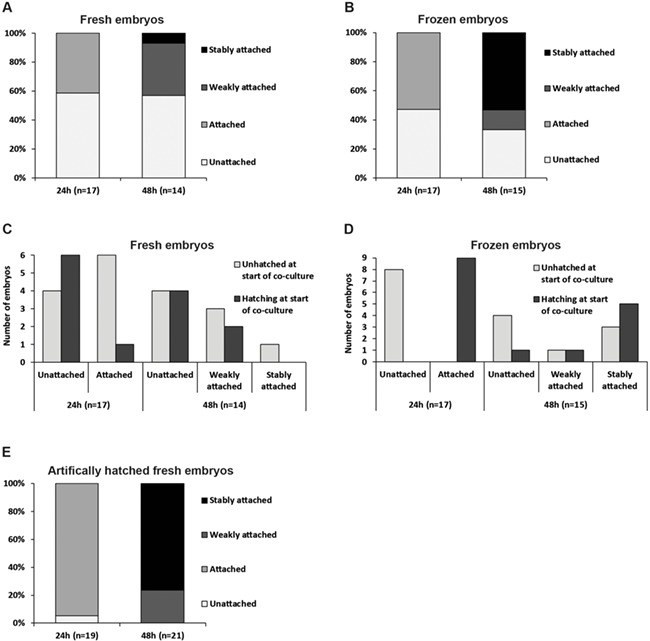
Attachment of day 6 human blastocysts to Ishikawa cells. (A) Day 6 human blastocysts from current IVF cycles (fresh) were co-cultured with Ishikawa cells for 48 h and attachment (attached or not attached) was assessed after 24 h. At 48 h, co-cultures were fixed with paraformaldehyde (PFA) and blastocysts that remained attached after fixation were scored as stably attached, whereas those, which became detached during fixation, were scored as weakly attached. Two co-cultures were fixed after 24 h to assess attachment stage (data not reported here). (B) Day 6 human blastocysts developed from frozen embryos were co-cultured with Ishikawa cells for 48 h. Attachment was assessed at 24 h (attached or not attached), and at 48 h, co-cultures were fixed with PFA. Blastocysts that remained attached after fixation were scored as stably attached, whereas those that became detached during fixation were scored as weakly attached. Two co-cultures were fixed after 24 h to assess attachment stage (data not reported here). (C) Representation of fresh blastocyst attachment data comparing embryos graded as unhatched or hatching at the start of co-culture. (D) Representation of frozen blastocyst attachment data comparing embryos graded as unhatched or hatching at the start of co-culture. (E) Day 6 human blastocysts from current IVF cycles (fresh) were artificially hatched from the zona pellucida using acid Tyrode’s solution prior to co-culture with Ishikawa for 48 h. Attachment was assessed after 24 h (attached or not attached). At 48 h, co-cultures were fixed with PFA and blastocysts that remained attached after fixation were scored as stably attached, whereas those that became detached during fixation were scored as weakly attached. Attachment of two co-cultures was not assessed at 24 h.
Hatching from the ZP is required for implantation (McLaren 1970, Petersen et al. 2005b). All blastocysts were unhatched or were in the process of hatching from the ZP upon transfer to Ishikawa cells. Since the blastocyst could not be reoriented to reveal TE herniation, it was not possible to record accurately hatching status during co-culture. Hatching at the onset of co-culture was therefore correlated with attachment and was found to be a strong indicator of attachment at 24 h among frozen embryos, but not fresh: 9/9 hatching frozen blastocysts attached compared with 1/7 hatching fresh blastocysts (P = 0.0004) (Fig. 3C and D). However, 6/10 (60.0%) fresh blastocysts that were unhatched at the beginning of co-culture were attached after 24 h, and by 48 h, the only stably attached fresh blastocyst was one which was unhatched at the start of co-culture (Fig. 3C). We found that 3/8 (37.5%) frozen blastocysts unhatched at the onset of co-culture were stably attached after 48 h (Fig. 3D), but notably, all blastocysts (fresh and frozen) scored as stably attached after 48 h were at least partially hatched by this time; 6/9 (66.7%) had an associated ZP which was lost upon fixation, while the remainder had fully hatched before fixation.
These observations suggest that attachment at 24 h of blastocysts that were unhatched as co-culture began may reflect adherent interactions mediated by the ZP, however, scoring attachment as stable at 48 h is the reflective of robust TE-Ishikawa adherence. To eliminate the confounding factor of hatching status, a series of fresh blastocysts was chemically hatched prior to co-culture. Remarkably, 18/19 (94.7%) were attached after 24 h, and after 48 h, all had attached, with 5/21 (23.8%) achieving weak attachment and 16/21 (76.2%) reaching stable attachment (Fig. 3E).
Fresh, frozen, and artificially hatched fresh cohorts were compared to determine whether embryo quality underpinned the differences in attachment. The proportion of embryos that were of overall high grade on days 3, 5 and 6 did not differ between the cohorts (Table I). Individual grades for expansion or ICM on day 6 also did not differ; however, TE grades were significantly higher in frozen when compared with fresh or artificially hatched fresh cohorts (P = 0.00008 and 0.0009, respectively) (Table I). Therefore, TE grade was positively associated with the attachment of partially exposed TE in hatching blastocysts, whereas full TE exposure in artificially hatched embryos led to the attachment of blastocysts regardless of TE grade.
Table I.
Characteristics of human embryos from the experiments presented in Fig. 3 .
| Fresh embryos (n = 14) | Frozen embryos (n = 15) | Artificially hatched fresh embryos (n = 21) | |
|---|---|---|---|
| High overall grade day 3 | 6 | 8 | 8 |
| High overall grade day 5 | 1 | 1* | 0 |
| High overall grade day 6 | 1 | 4 | 2 |
| High expansion grade day 6 | 13 | 15 | 13 |
| High ICM grade day 6 | 3 | 5 | 3 |
| High TE grade day 6 | 1a | 12a,b | 5b |
Numbers of embryos from fresh, frozen [six embryos frozen at pronuclear stage (PN), 11 embryos frozen at day 2], and artificially hatched fresh cohorts with high grades. High grades are based on the eligibility/requirement for embryo freezing (Cutting et al. 2008, ASiRMaESIGo 2011). P < 0.05, repeated Chi-squared analysis with a Bonferroni correction.
*Eight out of the 15 frozen embryos were graded on day 5.
asignificant difference between fresh and frozen cohorts.
bsignificant difference between frozen and chemically hatched fresh cohorts.
Effects of HA-containing medium on human blastocyst attachment to Ishikawa cells
To test the effect of EmbryoGlue® on blastocyst attachment in our in vitro model, we artificially hatched blastocysts prior to treatment and monitored attachment at early timepoints, including 1, 2, 4 and 6 h, as well as 24 and 48 h. Control blastocysts (n = 20) were hatched and incubated in GTL, while treated blastocysts (n = 23) were hatched and incubated in EmbryoGlue®, before co-culture. There were no differences in blastocyst grade between the two groups (Table II). Rapid attachment of control blastocysts was observed, with 25, 35, 60 and 75% achieving attachment at 1, 2, 4 and 6 h, respectively (Fig. 4). EmbryoGlue®-treated blastocysts tended to exhibit slower kinetics of attachment, although this difference was not significant (43.5 and 47.8% at 4 and 6 h, respectively, Fig. 4). A total of 91.3 and 100% of EmbryoGlue®-treated blastocysts were attached after 24 and 48 h, respectively, compared with 75 and 85% of control blastocysts, respectively, but this difference was not significant (Fig. 4).
Table II.
Characteristics of human embryos from the experiment presented in Fig. 4 , graded on day 6.
| Control (n = 20) | EmbryoGlue® (n = 23) | |
|---|---|---|
| High overall grade day 6 | 5 (1 fresh, 4 frozen) | 4 (0 fresh, 4 frozen) |
| High expansion grade day 6 | 15 (9 fresh, 6 frozen) | 16 (8 fresh, 8 frozen) |
| High ICM grade day 6 | 5 (1 fresh, 4 frozen) | 5 (1 fresh, 4 frozen) |
| High TE grade day 6 | 7 (1 fresh, 6 frozen) | 13 (5 fresh, 8 frozen) |
Control embryos included 10 fresh embryos and 10 frozen: five frozen at PN; two frozen on day 2; and three frozen on day 3. EmbryoGlue®-treated embryos included 12 fresh embryos and 11 frozen: six frozen at PN; one frozen on day 2; and four frozen on day 3. High grades are based on eligibility/requirement for embryo freezing (Cutting et al. 2008, ASiRMaESIGo 2011).
Figure 4.
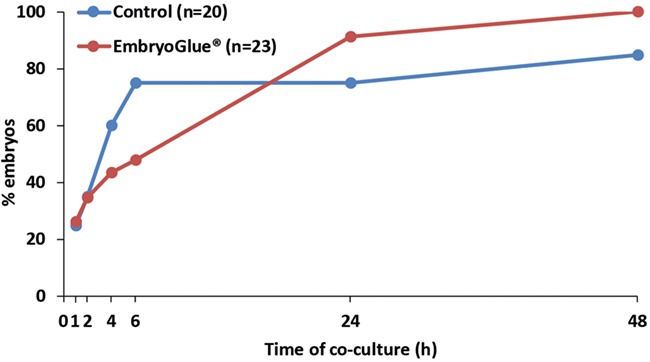
Effects of HA-containing medium on human blastocyst attachment to Ishikawa cells. Fresh blastocysts were treated with control GTL medium or EmbryoGlue® before co-culture with Ishikawa cells and monitoring of attachment over 48 h. Embryo attachment was assessed hourly from 1–6 h, at 24 and 48 h. Percentage of attached embryos is plotted over time of co-culture.
Discussion
Clinical evidence of efficacy has encouraged the use of HA-containing media to increase embryo implantation rates in ART. Blastocyst attachment experiments based on clinical application were performed here to shed much needed light on the activity of HA at implantation. No significant effect of HA on attachment to endometrial epithelial cells in vitro was found, although a trend to slower attainment of attachment was observed. These findings reflect recent in vivo and in vitro animal studies suggesting that instead of promoting blastocyst attachment, HA actually attenuates this aspect of implantation (Marei et al. 2017, Berneau et al. 2019), implying that HA must affect the implantation potential of human embryos by an alternative mechanism.
To test HA activity at implantation, we extended an existing in vitro model of implantation for use with human embryos. This model uses the Ishikawa cell line to provide a well-characterised and consistent in vitro endometrial epithelium that allows for the evaluation of blastocyst attachment kinetics (Ruane et al. 2017). Recent developments in primary endometrial epithelial organoid culture may yield implantation models that better recapitulate the in vivo process (Turco et al. 2017), although the evaluation of attachment processes in a 3D system presents some technical challenges. Here, we used fixation to differentiate between weakly and stably attached embryos and showed that complete hatching from the ZP is not required for stable attachment. Importantly, higher levels of attachment of the higher TE grade frozen embryo cohort suggest that attachment to Ishikawa cells reflects implantation rates in vivo, where TE grade appeared superior to ICM and expansion status in predicting implantation (Ahlstrom et al. 2011). Artificial hatching prior to co-culture, however, allowed stable attachment of almost all embryos independent of TE grade, underscoring the highly receptive nature of Ishikawa cells. A previous study demonstrated high levels of attachment of hatched day 6 human blastocysts to Ishikawa cells; however, only high grade blastocysts were used (Aberkane et al. 2018). In addition, our approach revealed that hatched blastocysts attach rapidly to Ishikawa cells, with 75% embryos adhering within 6 h. These findings indicate that herniating high grade TE is capable of mediating attachment, and provide in vitro evidence for assisted hatching of low TE grade embryos to enhance implantation potential in the clinical setting.
We characterised the expression of HA-related genes during human pre-implantation development, demonstrating that transcripts for the HA receptors—CD44 and HMMR—but not TLR4, and synthesis (HAS) and degradation (HYAL) enzymes are present in cleavage stage embryos and in both ICM and TE of the blastocyst. HA-containing medium could therefore stimulate CD44 and HMMR during cleavage stage transfer and lead to responses which may improve the implantation potential of the resulting blastocyst. Degradation of HA into active signalling forms by cleavage stage embryos is also possible through HYAL, and this may contribute to HA-containing medium activity during cleavage-stage transfer. As Ishikawa cells express CD44 at the apical domain (Berneau et al. 2019), HA could act as an adhesion factor by bridging CD44 receptors present in the apposing TE-Ishikawa membranes. Moreover, increased CD44 expression was noted in blastocysts that adhered more quickly to Ishikawa cells, (Aberkane et al. 2018). The microarray data also suggest that the human embryo is capable of both synthesising HA and degrading high Mr HA, consistent with murine, ovine and bovine studies (Fouladi-Nashta et al. 2017). HAS2, the isoform which synthesises high Mr HA, was not expressed in later stage human embryos, similar to the pattern seen in bovine (Marei et al. 2013). However, HYAL1 and HYAL2 were expressed throughout development and in both ICM and TE. These data are consistent with the model that later stage pre-implantation embryos develop in a predominantly low Mr HA environment and, moreover, that embryos are capable of degrading exogenous high Mr HA added to medium into low Mr forms which can be active as trophic signalling agents (Fouladi-Nashta et al. 2017).
EmbryoGlue® treatment of day 6 blastocysts had no significant effect on attachment to Ishikawa cells, although there was a trend towards impaired initial attachment that fits with our recent description of enhanced kinetics of mouse blastocyst attachment after hyaluronidase treatment (Berneau et al. 2019). EmbryoGlue® was added to the co-culture with blastocysts, mimicking clinical treatment, leading to a final concentration of 0.6%. As EmbryoGlue® is more viscous than co-culture medium (Reed and Said 2019), a bolus of HA may be retained for some time around the blastocyst. High HA concentrations around the blastocyst may stimulate CD44 and HMMR receptors in Ishikawa cells (Tamm-Rosenstein et al. 2013, Berneau et al. 2019), which could affect Ishikawa-blastocyst interactions. In vivo studies in sheep also highlighted inhibitory effects of HA on implantation (Marei et al. 2017). HA may therefore act like mucins, glycoproteins downregulated from the endometrial epithelial surface at implantation, to allow TE attachment (Aplin et al. 2001, Meseguer et al. 2001, Dharmaraj et al. 2014). We conclude that HA in EmbryoGlue® is not acting here as a CD44 bridging molecule in the initial stages of embryo attachment. This may be attributable to the use of poor quality human embryos, or to the fact that Ishikawa cells are highly receptive to human embryos, to a level that is not sensitive to exogenous HA.
It is unknown which Mr species of HA are present in commercially available media as manufacturers are not obliged under current legislation to disclose the full composition of media to clinics and researchers, although there have been recent calls from professional bodies in ART for this to be reconsidered (Sunde et al. 2016). It is likely that EmbryoGlue® contains high Mr HA; however, human embryos also express hyaluronidase enzymes, such as HYAL2, and these may be released into culture medium and effect the breakdown of added high Mr HA into smaller species with trophic signalling capacity. HA may therefore enhance implantation through the signalling downstream of the receptors we found to be expressed in the cleavage and blastocyst stage. MMP9, a member of the matrix metalloproteinase family, has been identified as a downstream target of CD44 (Misra et al. 2015) and implicated in implantation in a number of species (Zhao et al. 2002, Beceriklisoy et al. 2007). MMP9 functions in murine and human trophoblast invasion (Canete-Soler et al. 1995, Rechtman et al. 1999, Zhang et al. 2004), and dysregulated expression of MMP9 has been implicated in infertility and early pregnancy loss (Skrzypczak et al. 2007). Thus, a mechanism whereby high levels of HA in EmbryoGlue® act to upregulate embryonic genes in preparation for implantation and trophoblast invasion could be envisaged.
In conclusion, we have characterised an in vitro model of human embryo attachment and shown that HA may not act as an implantation ‘glue’, at least with respect to the initial interaction with endometrial epithelium. Our study highlights the difficulties in uncovering basic mechanisms underlying clinical data in ART. Improvements in ART success rates are urgently required, with the development of an implantation-competent embryo and successful initiation of implantation being key bottlenecks. Although much clinical research has taken place into improving ART, the basic scientific understanding is lagging behind. This represents a critical gap in knowledge as the evidence base for treatments in ART and associated risks, and in particular, the use of ‘add-ons’ to IVF treatment (including HA-containing medium) is a highly controversial area (Spencer et al. 2016, Wilkinson et al. 2016, Macklon et al. 2019, Wilkinson et al. unpublished work, https://www.hfea.gov.uk/treatments/explore-all-treatments/treatment-add-ons/). We believe that ART treatments require as strong an evidence base as possible (Stocking et al. 2019). We further suggest that an increase in transparency in ART is required; in the case of culture medium, it is important that the full composition of media be disclosed in order to facilitate basic research and allow full understanding of clinical treatments (Biggers 2000, Sunde et al. 2016).
Acknowledgements
We would like to thank the IVF patients who kindly donated their embryos to this research programme, and the clinic staff at the Department of Reproductive Medicine, St Mary’s Hospital, Manchester; Manchester Fertility, Manchester; and the Hewitt Centre, Liverpool Women’s Hospital, Liverpool, who made this possible. We would especially like to thank our senior research nurse, Claudette Wright, for co-ordinating these activities.
Authors’ roles
S.J.K., M.W., J.D.A. and D.R.B. designed the study and obtained funding. P.T.R., C.B., P.A.B., W.A. and S.C.B. carried out experimental work. P.T.R. and D.R.B. wrote the manuscript, which was edited by S.J.K., M.W. and J.D.A.
Funding
Wellbeing of Women (grant RG1442); NIHR Local Comprehensive Research Network and NIHR Manchester Clinical Research Facility; Department of Health Scientist Practitioner Training Scheme; Ministry of Higher Education, The State of Libya.
Conflict of interest
The authors declare no conflicts of interest.
References
- Aberkane A, Essahib W, Spits C, De C, Sermon K, Adriaenssens T, Mackens S, Tournaye H, Brosens JJ, Van de H. Expression of adhesion and extracellular matrix genes in human blastocysts upon attachment in a 2D co-culture system. Mol Hum Reprod 2018;24:375–387. [DOI] [PubMed] [Google Scholar]
- Ahlstrom A, Westin C, Reismer E, Wikland M, Hardarson T. Trophectoderm morphology: an important parameter for predicting live birth after single blastocyst transfer. Hum Reprod 2011;26:3289–3296. [DOI] [PubMed] [Google Scholar]
- Aplin JD, Meseguer M, Simon C, Ortiz ME, Croxatto H, Jones CJ. MUC1, glycans and the cell-surface barrier to embryo implantation. Biochem Soc Trans 2001;29:153–156. [DOI] [PubMed] [Google Scholar]
- Aplin JD, Ruane PT. Embryo-epithelium interactions during implantation at a glance. J Cell Sci 2017;130:15–22. [DOI] [PubMed] [Google Scholar]
- ASiRMaESIGo The Istanbul consensus workshop on embryo assessment: proceedings of an expert meeting. Hum Reprod 2011;26:1270–1283. [DOI] [PubMed] [Google Scholar]
- Beceriklisoy HB, Walter I, Schafer-Somi S, Miller I, Kanca H, Izgur H, Aslan S. Matrix metalloproteinase (MMP)-2 and MMP-9 activity in the canine uterus before and during placentation. Reprod Domest Anim 2007;42:654–659. [DOI] [PubMed] [Google Scholar]
- Bentin-Ley U, Horn T, Sjogren A, Sorensen S, Falck Larsen J, Hamberger L. Ultrastructure of human blastocyst-endometrial interactions in vitro. J Reprod Fertil 2000;120:337–350. [DOI] [PubMed] [Google Scholar]
- Berger C, Boggavarapu NR, Menezes J, Lalitkumar PG, Gemzell-Danielsson K. Effects of ulipristal acetate on human embryo attachment and endometrial cell gene expression in an in vitro co-culture system. Hum Reprod 2015;30:800–811. [DOI] [PubMed] [Google Scholar]
- Berneau SC, Ruane PT, Brison DR, Kimber SJ, Westwood M, Aplin JD. Investigating the role of CD44 and hyaluronate in embryo-epithelial interaction using an in vitro model. Mol Hum Reprod 2019;25:265–273. [DOI] [PubMed] [Google Scholar]
- Biggers JD. Ethical issues and the commercialization of embryo culture media. Reprod Biomed Online 2000;1:74–76. [DOI] [PubMed] [Google Scholar]
- Block J, Bonilla L, Hansen PJ. Effect of addition of hyaluronan to embryo culture medium on survival of bovine embryos in vitro following vitrification and establishment of pregnancy after transfer to recipients. Theriogenology 2009;71:1063–1071. [DOI] [PubMed] [Google Scholar]
- Boggavarapu NR, Berger C, von C, Menezes J, Gemzell-Danielsson K, Lalitkumar PG. Effects of low doses of mifepristone on human embryo implantation process in a three-dimensional human endometrial in vitro co-culture system. Contraception 2016;94:143–151. [DOI] [PubMed] [Google Scholar]
- Bontekoe S, Heineman MJ, Johnson N, Blake D. Adherence compounds in embryo transfer media for assisted reproductive technologies. Cochrane Database Syst Rev 2014;2: CD007421. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brison DR, Roberts SA, Kimber SJ. How should we assess the safety of IVF technologies? Reprod Biomed Online 2013;27:710–721. [DOI] [PubMed] [Google Scholar]
- Buck VU, Gellersen B, Leube RE, Classen-Linke I. Interaction of human trophoblast cells with gland-like endometrial spheroids: a model system for trophoblast invasion. Hum Reprod 2015;30:906–916. [DOI] [PubMed] [Google Scholar]
- Campbell S, Larsen J, Seif MW, Allen TD, Knox F, Jones CJ, Aplin JD. Mosaic characteristics of human endometrial epithelium in vitro: analysis of secretory markers and cell surface ultrastructure. Mol Hum Reprod 2000;6:41–49. [DOI] [PubMed] [Google Scholar]
- Campbell S, Swann HR, Aplin JD, Seif MW, Kimber SJ, Elstein M. CD44 is expressed throughout pre-implantation human embryo development. Hum Reprod 1995;10:425–430. [DOI] [PubMed] [Google Scholar]
- Canete-Soler R, Gui YH, Linask KK, Muschel RJ. Developmental expression of MMP-9 (gelatinase B) mRNA in mouse embryos. Dev Dyn 1995;204:30–40. [DOI] [PubMed] [Google Scholar]
- Carson DD, Dutt A, Tang JP. Glycoconjugate synthesis during early pregnancy: hyaluronate synthesis and function. Dev Biol 1987;120:228–235. [DOI] [PubMed] [Google Scholar]
- Chitcholtan K, Asselin E, Parent S, Sykes PH, Evans JJ. Differences in growth properties of endometrial cancer in three dimensional (3D) culture and 2D cell monolayer. Exp Cell Res 2013;319:75–87. [DOI] [PubMed] [Google Scholar]
- Choudhary M, Zhang X, Stojkovic P, Hyslop L, Anyfantis G, Herbert M, Murdoch AP, Stojkovic M, Lako M. Putative role of hyaluronan and its related genes, HAS2 and RHAMM, in human early preimplantation embryogenesis and embryonic stem cell characterization. Stem Cells 2007;25:3045–3057. [DOI] [PubMed] [Google Scholar]
- Cutting R, Morroll D, Roberts SA, Pickering S, Rutherford A, BFS and ACE . Elective single embryo transfer: guidelines for practice British fertility society and association of clinical embryologists. Hum Fertil (Camb) 2008;11:131–146. [DOI] [PubMed] [Google Scholar]
- Dharmaraj N, Chapela PJ, Morgado M, Hawkins SM, Lessey BA, Young SL, Carson DD. Expression of the transmembrane mucins, MUC1, MUC4 and MUC16, in normal endometrium and in endometriosis. Hum Reprod 2014;29:1730–1738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fancsovits P, Lehner A, Murber A, Kaszas Z, Rigo J, Urbancsek J. Effect of hyaluronan-enriched embryo transfer medium on IVF outcome: a prospective randomized clinical trial. Arch Gynecol Obstet 2015;291:1173–1179. [DOI] [PubMed] [Google Scholar]
- Farquhar C, Marjoribanks J. Assisted reproductive technology: an overview of Cochrane reviews. Cochrane Database Syst Rev 2018;8: CD010537. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fouladi-Nashta AA, Raheem KA, Marei WF, Ghafari F, Hartshorne GM. Regulation and roles of the hyaluronan system in mammalian reproduction. Reproduction 2017;153:R43–R58. [DOI] [PubMed] [Google Scholar]
- Galan A, Herrer R, Remohi J, Pellicer A, Simon C. Embryonic regulation of endometrial epithelial apoptosis during human implantation. Hum Reprod 2000;15:74–80. [PubMed] [Google Scholar]
- Gardner DK, Rodriegez-Martinez H, Lane M. Fetal development after transfer is increased by replacing protein with the glycosaminoglycan hyaluronan for mouse embryo culture and transfer. Hum Reprod 1999;14:2575–2580. [DOI] [PubMed] [Google Scholar]
- Green CJ, Fraser ST, Day ML. Insulin-like growth factor 1 increases apical fibronectin in blastocysts to increase blastocyst attachment to endometrial epithelial cells in vitro. Hum Reprod 2015;30:284–298. [DOI] [PubMed] [Google Scholar]
- Harper J, Magli MC, Lundin K, Barratt CL, Brison D. When and how should new technology be introduced into the IVF laboratory? Hum Reprod 2012;27:303–313. [DOI] [PubMed] [Google Scholar]
- Kang YJ, Forbes K, Carver J, Aplin JD. The role of the osteopontin-integrin alphavbeta3 interaction at implantation: functional analysis using three different in vitro models. Hum Reprod 2014;29:739–749. [DOI] [PubMed] [Google Scholar]
- Lalitkumar PG, Lalitkumar S, Meng CX, Stavreus-Evers A, Hambiliki F, Bentin-Ley U, Gemzell-Danielsson K. Mifepristone, but not levonorgestrel, inhibits human blastocyst attachment to an in vitro endometrial three-dimensional cell culture model. Hum Reprod 2007;22:3031–3037. [DOI] [PubMed] [Google Scholar]
- Lalitkumar S, Boggavarapu NR, Menezes J, Dimitriadis E, Zhang JG, Nicola NA, Gemzell-Danielsson K, Lalitkumar LP. Polyethylene glycated leukemia inhibitory factor antagonist inhibits human blastocyst implantation and triggers apoptosis by down-regulating embryonic AKT. Fertil Steril 2013;100:1160–1169. [DOI] [PubMed] [Google Scholar]
- Lindenberg S, Hyttel P, Lenz S, Holmes PV. Ultrastructure of the early human implantation in vitro. Hum Reprod 1986;1:533–538. [DOI] [PubMed] [Google Scholar]
- Lindenberg S, Nielsen MH, Lenz S. In vitro studies of human blastocyst implantation. Ann N Y Acad Sci 1985;442:368–374. [DOI] [PubMed] [Google Scholar]
- Macklon NS, Ahuja KK, Fauser B. Building an evidence base for IVF 'add-ons'. Reprod Biomed Online 2019;38:853–856. [DOI] [PubMed] [Google Scholar]
- Mantikou E, Youssef MA, van M, van der F, Al-Inany HG, Repping S, Mastenbroek S. Embryo culture media and IVF/ICSI success rates: a systematic review. Hum Reprod Update 2013;19:210–220. [DOI] [PubMed] [Google Scholar]
- Marei WFA, Wathes DC, Raheem KA, Mohey-Elsaeed O, Ghafari F, Fouladi-Nashta AA. Influence of hyaluronan on endometrial receptivity and embryo attachment in sheep. Reprod Fertil Dev 2017;29:1763–1773. [DOI] [PubMed] [Google Scholar]
- McLaren A. The fate of the zona pellucida in mice. J Embryol Exp Morphol 1970;23:1–19. [PubMed] [Google Scholar]
- Meseguer M, Aplin JD, Caballero-Campo P, O'Connor JE, Martin JC, Remohi J, Pellicer A, Simon C. Human endometrial mucin MUC1 is up-regulated by progesterone and down-regulated in vitro by the human blastocyst. Biol Reprod 2001;64:590–601. [DOI] [PubMed] [Google Scholar]
- Misra S, Hascall VC, Markwald RR, Ghatak S. Interactions between Hyaluronan and its receptors (CD44, RHAMM) regulate the activities of inflammation and cancer. Front Immunol 2015;6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nagyova E. The biological role of Hyaluronan-rich oocyte-cumulus extracellular matrix in female reproduction. Int J Mol Sci 2018;19:E283. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nishida M, Kasahara K, Kaneko M, Iwasaki H, Hayashi K. Establishment of a new human endometrial adenocarcinoma cell line, Ishikawa cells containing estrogen and progesterone receptors. Nihon Sanka Fujinka Gakkai Zasshi 1985;37:1103–1111. [PubMed] [Google Scholar]
- Petersen A, Bentin-Ley U, Ravn V, Qvortrup K, Sorensen S, Islin H, Sjogren A, Mosselmann S, Hamberger L. The antiprogesterone org 31710 inhibits human blastocyst-endometrial interactions in vitro. Fertil Steril 2005a;83:1255–1263. [DOI] [PubMed] [Google Scholar]
- Petersen CG, Mauri AL, Baruffi RL, Oliveira JB, Massaro FC, Elder K, van Franco JGJ. Implantation failures: success of assisted hatching with quarter-laser zona thinning. Reprod Biomed Online 2005b;10:224–229. [DOI] [PubMed] [Google Scholar]
- Rechtman MP, Zhang J, Salamonsen LA. Effect of inhibition of matrix metalloproteinases on endometrial decidualization and implantation in mated rats. J Reprod Fertil 1999;117:169–177. [DOI] [PubMed] [Google Scholar]
- Reed ML, Said AH. Embryo transfer media – estimating viscosities, and consideration of viscosity as a factor impacting media and uterine fluid interactions. Reprod Biomed Online 2019; in press. [DOI] [PubMed] [Google Scholar]
- Ruane PT, Berneau SC, Koeck R, Watts J, Kimber SJ, Brison DR, Westwood M, Aplin JD. Apposition to endometrial epithelial cells activates mouse blastocysts for implantation. Mol Hum Reprod 2017;23:617–627. [DOI] [PubMed] [Google Scholar]
- Salamonsen LA, Shuster S, Stern R. Distribution of hyaluronan in human endometrium across the menstrual cycle. Implications for implantation and menstruation. Cell Tissue Res 2001;306:335–340. [DOI] [PubMed] [Google Scholar]
- Shaw L, Sneddon SF, Zeef L, Kimber SJ, Brison DR. Global gene expression profiling of individual human oocytes and embryos demonstrates heterogeneity in early development. PLoS One 2013;8:e64192. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh H, Aplin JD. Endometrial apical glycoproteomic analysis reveals roles for cadherin 6, desmoglein-2 and plexin b2 in epithelial integrity. Mol Hum Reprod 2015;21:81–94. [DOI] [PubMed] [Google Scholar]
- Singh H, Nardo L, Kimber SJ, Aplin JD. Early stages of implantation as revealed by an in vitro model. Reproduction 2010;139:905–914. [DOI] [PubMed] [Google Scholar]
- Skrzypczak J, Wirstlein P, Mikolajczyk M, Ludwikowski G. Zak T. TGF superfamily and MMP2, MMP9, TIMP1 genes expression in the endometrium of women with impaired reproduction. Folia Histochem Cytobiol 2007;45:S143–S148. [PubMed] [Google Scholar]
- Smith HL, Stevens A, Minogue B, Sneddon S, Shaw L, Wood L, Adeniyi T, Xiao H, Lio P, Kimber SJ et al. . Systems based analysis of human embryos and gene networks involved in cell lineage allocation. BMC Genomics 2019;20. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spencer EA, Mahtani KR, Goldacre B, Heneghan C. Claims for fertility interventions: a systematic assessment of statements on UK fertility Centre websites. BMJ Open 2016;6:e013940. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sunde A, Brison D, Dumoulin J, Harper J, Lundin K, Magli MC, Van den E, Veiga A. Time to take human embryo culture seriously. Hum Reprod 2016;31:2174–2182. [DOI] [PubMed] [Google Scholar]
- Tamm-Rosenstein K, Simm J, Suhorutshenko M, Salumets A, Metsis M. Changes in the transcriptome of the human endometrial Ishikawa cancer cell line induced by estrogen, progesterone, tamoxifen, and mifepristone (RU486) as detected by RNA-sequencing. PLoS One 2013;8:e68907. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tavianatou AG, Caon I, Franchi M, Piperigkou Z, Galesso D, Karamanos NK. Hyaluronan: molecular size-dependent signaling and biological functions in inflammation and cancer. FEBS J 2019; in press. [DOI] [PubMed] [Google Scholar]
- Turco MY, Gardner L, Hughes J, Cindrova-Davies T, Gomez MJ, Farrell L, Hollinshead M, Marsh SGE, Brosens JJ, Critchley HO et al. . Long-term, hormone-responsive organoid cultures of human endometrium in a chemically defined medium. Nat Cell Biol 2017;19:568–577. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wight TN. Provisional matrix: a role for versican and hyaluronan. Matrix Biol 2017;60–61:38–56. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wilkinson J, Roberts SA, Showell M, Brison DR, Vail A. No common denominator: a review of outcome measures in IVF RCTs. Hum Reprod 2016;31:2714–2722. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Youssef MM, Mantikou E, van M, Van der F, Al-Inany HG, Repping S, Mastenbroek S. Culture media for human pre-implantation embryos in assisted reproductive technology cycles. Cochrane Database Syst Rev 2015; CD007876. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang X, Wang HM, Lin HY, Liu GY, Li QL, Zhu C. Regulation of matrix metalloproteinases (MMPS) and their inhibitors (TIMPS) during mouse peri-implantation: role of nitric oxide. Placenta 2004;25:243–252. [DOI] [PubMed] [Google Scholar]
- Zhao YG, Xiao AZ, Cao XM, Zhu C. Expression of matrix metalloproteinase −2, −9 and tissue inhibitors of metalloproteinase −1, −2, −3 mRNAs in rat uterus during early pregnancy. Mol Reprod Dev 2002;62:149–158. [DOI] [PubMed] [Google Scholar]


