Abstract
Background:
Calcium phosphate materials have been employed clinically as bone void fillers for several decades. These materials are most often provided in the form of small, porous granules that can be packed to fill the wide variety of size and shape of bony defects encountered. ReBOSSIS-85 (RB-85) is a synthetic bioresorbable bone void filler for the repair of bone defects with handling characteristics of glass wool-like (or cotton ball-like). The objective of this study is to evaluate the in vivo performance of RB-85 (test material), compared to a commercially available bone void filler, Mastergraft Putty (predicate material), when combined with bone marrow aspirate and iliac crest autograft, in an established posterolateral spine fusion rabbit model.
Methods:
One hundred fifty skeletally mature rabbits had a single level posterolateral fusion performed. Rabbits were implanted with iliac crest bone graft (ICBG), Mastergraft Putty™ plus ICBG, or one of 4 masses of ReBOSSIS-85 (0.2, 0.3, 0.45, or 0.6 g) plus ICBG. Plain films were taken weekly until euthanasia. Following euthanasia at 4, 8, and 12 weeks, the lumbar spine were tested by manual palpation. Spinal columns in the 12 week group were also subjected to non-destructive flexibility testing. MicroCT and histology were performed on a subset of each implant group at each euthanasia period.
Results:
Radiographic scoring of the fusion sites indicated a normal healing response in all test groups. Bilateral radiographic fusion rates for all test groups were 0% at 4 weeks; ICBG 43%, Mastergraft Putty 50%, RB-85-0.2g 0%, RB-85-0.3g 13%, RB-85-0.45g 38%, and RB-85-0.6g 63% at 8 weeks; and ICBG 50%, Mastergraft Putty 50%, RB-85-0.2g 0%, RB-85-0.3g 25%, RB-85-0.45g 36%, and RB-85-0.6g 50% at 12 weeks.
Spine fusion was assessed by manual palpation of the treated motion segments. At 12 weeks, ICBG, MGP, and RB-85-0.6g were fused mechanically in at least 50% of the rabbits. All groups demonstrated significantly less range of motion in both flexion/extension, lateral bending, and axial rotation compared to normal unfused controls.
Histopathology analysis of the fusion masses, in all test groups, indicated an expected normal response of mild inflammation with macrophage and multinucleated giant cell response to the graft material at 4 weeks and resolving by 12 weeks. Regardless of test article, new bone formation and graft resorption increased from 4 to 12 weeks post-op.
Conclusions:
This animal study has demonstrated the biocompatibility and normal healing features associated with the ReBOSSIS-85 bone graft (test material) when combined with autograft as an extender. ReBOSSIS-85 was more effective when a larger mass of test article was used in this study.
Clinical Relevance:
ReBOSSIS-85 can be used as an extender negating the need for large amounts of local or iliac crest bone in posterolateral fusions.
Keywords: synthetic bone graft substitute, posterolateral fusion, animal model
Introduction
Iliac crest autograft (ICBG) has long been considered the gold standard in achieving long-term spinal arthrodesis,1-11 despite the morbidity rate associated with graft harvest.12, 13 In response, a number of commercially available synthetic bone graft extenders and autograft alternatives have been developed.14-20 Most common substitutes and extenders are allografts, demineralized bone matrices, collagen matrices with bone morphogenetic proteins (BMPs), and ceramics.1 Ceramic bone grafts are porous and resorbable making them optimal for bone ingrowth.21-29 Ceramic bone grafts are osteoconductive and can be combined with osteoinductive factors such as bone marrow aspirate to maximize bone growth.30
Calcium phosphate materials have been employed clinically as bone void fillers for several decades. These materials are most often provided in the form of small, porous granules that can be packed to fill the wide variety of size and shape of bony defects encountered. Recently, ReBOSSIS85 was developed and is a synthetic bioresorbable bone void filler for the repair of bone defects with handling characteristics of glass wool-like (or cotton ball-like) material. ReBOSSIS85 is a synthetic, resorbable bone void filler. It is composite material consisting of (by weight) 40% beta-tricalcium phosphate (β-TCP), 30% siloxane-containing vaterite (a form of calcium carbonate, CaCO3), and 30% poly(L-lactide-co-glycolide). Siloxane-containing vaterite has been synthesized by doping calcium carbonate with 2-3% of siloxane with an amino group.34 Fibrous composites were prepared by blending siloxane-containing vaterite and β-TCP with PLGA using kneading and subsequent electrospinning. The electrospinning process used in manufacturing ReBOSSIS-85 results in a glass wool-like physical form. The diameter of the fibers in the PLGA scaffold ranges from 50 μm to 100 μm. The interconnected structure allows for formation of new bone throughout the network of interconnecting pores.31-33 The silicon content of the final ReBOSSIS-85 material is 0.5% - 1% by weight. A trace amount of silicon species has been reported to stimulate osteogenic cells to mineralize and enhance bone formation.34-37 The product form allows for versatility in handling and ease of use and can be hydrated with bone marrow aspirate (BMA). ReBOSSIS-85 has demonstrated to be osteoconductive, providing a scaffold for cell attachment and supporting formation of osseous tissue across bony defects.
This purpose of this study was to evaluate and compare the spine fusion performance of the ReBOSSIS-85 to a FDA approved bone graft substitute Mastergraft Putty in a spine fusion animal model, using iliac crest autograft as a control. Both products are designed to be hydrated with bone marrow aspirate and can be used when combined with autologous bone.
Methods
One hundred fifty male skeletally mature New Zealand White rabbits weighing 4.5–5.5 kg were entered into the study (Table 1). Female rabbits were not used as they may undergo instantaneous ovulation and pseudopregnancy due to extraneous stimulation, and subsequent fluctuations in hormones may impact bone metabolism. To avoid potential impact on study outcomes, male animals were selected for this study. The number of animals per treatment was determined by a power analysis to show a 20% difference in flexion/ extension via biomechanical testing (alpha of 0.05 and a power of 90). All procedures were approved by the Institutional Animal Care Use Committee (#6031701) and conducted at The University of Iowa Department of Orthopaedics Animal Research Surgicenter, Bone Healing Research Lab, and the Iowa Spine Research Lab, USA. Throughout the study, animals were individually caged and monitored twice daily for signs of pain and discomfort.
Table 1.
Number of Animals per Treatment Group
| Treatment Group | #Animals/ Time Point | |||
|---|---|---|---|---|
| 4 weeks | 8 weeks | 12 weeks | Total | |
| Iliac (ICBG) Crest Bone Graft | 5 | 8 | 12 | 25 |
| Mastergraft ICBG Putty + | 5 | 8 | 12 | 25 |
| ReBOSSIS-85 (0.2g)/ BMA + ICBG (1:1) | 5 | 8 | 12 | 25 |
| ReBOSSIS-85 (0.3 g)/ BMA + ICBG | 5 | 8 | 12 | 25 |
| ReBOSSIS-85 (0.45 g)/ BMA + ICBG | 5 | 8 | 12 | 25 |
| ReBOSSIS-85 (0.6 g)/ BMA + ICBG | 5 | 8 | 12 | 25 |
| Total #Animals | 30 | 48 | 72 | 150 |
Surgical procedure
All operative procedures were performed in a dedicated surgical suite using inhalation anesthesia and aseptic techniques. Surgical anesthesia was maintained with 1–5% isoflurane delivered in oxygen. Cardiorespiratory monitoring was conducted throughout the procedure.
All rabbits were prepared for surgical procedures using standard operating procedures. After the pre-anesthetic had taken effect, rabbits were clipped free of fur over the surgical area and scrubbed with Chlorhexidine soap and wiped with isopropyl alcohol (alternated three times each). Chlorhexidine solution was applied just prior to surgical incision. Rabbits were placed prone on the operating table. The surgical approach to the spine was identical in all rabbits. A dorsal midline skin incision, approximately 15 centimeters long, was made from L1 to the sacrum, and then the fascia and muscle were incised over the L5-L6 transverse processes (TPs). The TPs were then decorticated with a high-speed burr to ensure bleeding host bone. Vertebral bodies and lamina were avoided and not decorticated.
All animals had approximately 2.5–3.0 cc of corticocancellous bone graft obtained bilaterally from the iliac crest (total of 5-6 cc). This volume of graft is the maximum amount which can be harvested from the rabbit iliac crest without significant animal morbidity.31,38-42
In the Mastergraft Putty™ animals, 1.5 cc of implant was hydrated with bone marrow aspirate (BMA, 1.5 cc) + ICBG (1.5cc) was placed in the paraspinal bed between the transverse processes. The ReBossis-85™ (RB-85), regardless of amount, was hydrated with 1.5 cc of BMA and 1.5 cc of ICBG (Figure 1) was added and then placed in the paraspinal bed. ICBG only rabbits received approximately 3.0 cc of ICBG per side. Fascia and skin were closed with 3-0 Vicryl and the skin stapled.
Figure 1.
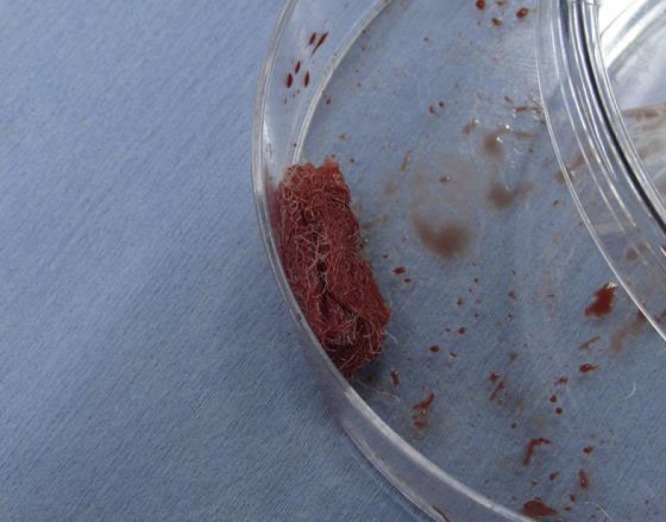
RB-85 hydrated with BMA and filled with ICBG.
Radiographic assessment
Ventral/dorsal radiographs were obtained with a Simon DR (Quantum) RAD-X High Frequency Radiographic Imaging System, (model: E7242X), and stored using Whitecap PACs system. Radiographs were examined to confirm graft placement and assess graft migration, osteolysis, fracture, and/or any other adverse events. In addition, radiographs were graded for bilateral fusion. Three reviewers blinded to treatment group and time point determined bilateral fusion rates for each study group at each time point. Trabecular bone patterns, change in implant appearance, and graft density of term radiographs were compared to immediate post-op radiographs to assess fusion. Final fusion results were determined by agreement of at least two of the three observers.
Five specimens (histology animals) were scanned using a SkyScan 1176 Micro-CT. Internal structures were reconstructed as a series of 2D cross sections which were then used to analyze morphological parameters of the specimen. The SkyScan 1176 was calibrated according to the manufacturers recommended schedule for contrast, positioning and density.
A rectangular ROI of 250 mm2 (10 x 25 mm) was placed across the fusion site inclusive of the transverse processes sagittal plane, bilateral, center of defect) and areas of bone, implant, and soft tissue were calculated.
Manual palpation analysis
The primary outcome used to determine fusion was manual palpation56 and flexibility analysis.24,57 After removing the spine, fusion was graded by three independent observers (blinded to treatment group and time point) as ‘fused’ if no detectable motion was present at the treated segment when tested in flexion and extension. The fusion was graded as ‘not fused’ if motion was present. Final results were determined by agreement of at least two of the three observers.
Mechanical testing
Biomechanical non-destructive stiffness testing was performed following manual palpation in the 12 week group to quantify stiffness and differences between groups. Testing consisted of flexion/extension, lateral bending, and torsion to a pre-determined, sub-failure load.
The vertebral bodies cranial and caudal to the fused motion segment were embedded in Bondo/Fiberglass material using 2 inch PVC piping. The specimens were mounted in a biaxial servo-hydraulic materials testing machine (858 Bionix II, MTS Corporation, Eden Prairie, MN, USA) retrofitted with two spine gimbals and a passive XZ table. Custom-made rigid body markers comprising infrared light emitting diodes affixed between two small aluminum plates were placed on each vertebral body and the two gimbals to track the segmental motions. Nondestructive flexibility tests were performed about each axis of rotation (i.e., flexion-extension, right-left lateral bending, and right-left axial rotation) by applying an isolated ±0.27 Nm (0 Nm, 0.09 Nm, 0.18 Nm, and 0.27 Nm) moment about each of the primary axes. Each test initiated and concluded in the neutral position with zero load. Three loading and unloading cycles were performed with motion data collected on the third cycle (the first two cycles served as preconditioning). The displacement of each vertebrae was measured using an optoelectronic motion capture system (Optotrak, Northern Digital, Waterloo, Ontario, Canada); the output of which was synchronized with that of the MTS. During testing the specimens were kept moist with saline solution spray. Stiffness was determined and compared to normal controls (10 normal rabbit lumbar columns, historic internal laboratory controls).
Histology
Five specimens from each group were randomly selected for histological evaluation. Fusion sites of each animal were processed for histology and sectioned in the sagittal plane to obtain a total of six sections per animal (3 per side of each fusion mass). For each side, sections were created adjacent to the vertebral body, through the center of the fusion mass, and through the lateral aspect of the fusion mass, spaced approximately 3 mm apart. Sections were stained with H&E.
All six sections from each fusion mass (3 per side) were subject to semiquantitative histopathology analysis. Specimens from each slide were evaluated by the PI and a board certified veterinary pathologist and assigned scoring based on a modified ISO 10993-6, Annex E (Table 2). An overall assessment was recorded for each fusion mass (one score per side).
Table 2.
Histopathology Analysis Scoring System
| Response | Score (*phf = per high powered field) | ||||
|---|---|---|---|---|---|
| 0 | 1 | 2 | 3 | 4 | |
| Polymorphonuclear cells | 0 | Rare, 1-5/phf* | 5-10/phf | Heavy infiltrate | Packed |
| Lymphocytes | 0 | Rare, 1-5/phf* | 5-10/phf | Heavy infiltrate | Packed |
| Plasma cells | 0 | Rare, 1-5/phf* | 5-10/phf | Heavy infiltrate | Packed |
| Macrophages | 0 | Rare, 1-5/phf* | 5-10/phf | Heavy infiltrate | Packed |
| Giant cells | 0 | Rare, 1-5/phf* | 3-5/phf | Heavy infiltrate | Packed |
| Necrosis | 0 | Slight | Moderate | Marked | Severe |
| Infection | 0 | Slight | Moderate | Marked | Severe |
| Fibrinous exudates | 0 | Slight | Moderate | Marked | Severe |
| Neovascularisation | 0 | Minimal capillary proliferation focal, 1-3 buds | Groups of 4-7 capillaries with supporting fibroblastic structures | Broad band of capillaries with supporting structures | Extensive band of capillaries with supporting fibroblastic structures |
| Fibrocytes/ tissue, A fibrosis fibro-connective | 0 | Narrow band | Moderately | Thick band | Extensive band |
| Material resorption degradation / Graft | 0 | Slight | Moderate | Marked | Severe (100% degraded) |
| Bone Formation | 0 | Slight | Moderate | Marked | Extensive |
Statistical analysis
Statistical analysis was performed on the microCT and histomorphometry data, as well as the flexion-extension, lateral bending and axial rotation data from the flexibility analysis. All data was analyzed to a 95% confidence level (p<0.05) using a 2-tailed t-test assuming unequal variance in Microsoft Excel. Unless otherwise noted, all data are reported as the mean and one standard deviation.
Results
All test articles were administered successfully. Rabbits were alert and eating within 3 hours of surgery. Daily observations of each rabbit were conducted and general overall health was determined. The rabbits, regardless of treatment, were in good health at time of euthanasia. There were no complications related to the test articles in-vivo. Three (3) rabbits were euthanized prior to end of study due to complications associated with ICBG harvest (1 ICBG 8w, 1 RB-85-0.45g 12w, and 1 RB-85-0.6g 4w). These complications have been noted in previous studies by the PI and others.12, 14-20, 34, 44-47
Necropsy of the animals was unremarkable regardless of test group. Macroscopic analysis of the implant sites demonstrated healthy tissue with no apparent adverse effects such as inflamed, necrotic, or devascularized tissue surrounding the defect sites.
Radiographic Assessment: Radiographs at 4, 8 and 12 weeks showed a normal healing response over time with no adverse reactions for all test groups (Figure 2). The loss of graft distinction at the host bone margins indicates a progression in host integration, implant remodeling and new bone formation over time. No fractures, osteolysis, or other adverse reactions were evident during radiographic examination for all test groups. Graft position migration from immediate post-op radiographs to radiographs greater than 1-week post-op is typically observed as the forces of overlaying muscle compresses the graft. These migration observations are typical of synthetic bone grafts applied in this animal model and are not a confounding variable. The radiographic fusion analysis is summarized in Table 3.
Figure 2.

Post-op and twelve week plain radiographs.
Table 3.
Bilateral Radiographic Fusion Rate
| 4 weeks | 8 weeks | 12 weeks | |
|---|---|---|---|
| ICBG | 0% | 43% | 50% |
| MGP | 0% | 50% | 50% |
| RB-85 (0.2) | 0% | 0% | 0% |
| RB-85 (0.3) | 0% | 13% | 25% |
| RB-85 (0.45) | 0% | 38% | 36% |
| RB-85 (0.6) | 0% | 63% | 50% |
The MicroCT scans supported the radiographic findings, showing a normal osteoconductive healing response over time with no adverse reactions for all test groups. The 4 week microCT scans showed host integration and new bone formation originating from the transverse process margins in all animals. The 8 and 12 week scans showed a progression of new bone formation and graft remodeling across the paraspinal bed from the 4 week time point (Figure 3).
Figure 3.

Sagittal slices of fusion masses at twelve weeks.
MicroCT morphometry analysis was performed using a rectangular region of interest (ROI) of 250 mm2 placed across the fusion site inclusive of the transverse processes (sagittal plane, bilateral, center of defect) and areas of bone, residual implant (Mastergraft Putty, MGP) and soft tissue were calculated. ReBOSSIS-85, regardless of group, was not distinguishable from background noise and/or soft tissue and therefore was not measured. Mean tissue areas are presented in Table 4.
Table 4.
MicroCT Tissue Ares
| 4 weeks | 8 weeks | 12 week | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Bone Area mm2 | Implant Area mm2 | Soft Tissue Area mm2 | Bone Area mm2 | Implant Area mm2 | Soft Tissue Area mm2 | Bone Area mm2 | Implant Area mm2 | Soft Tissue Area mm2 | |
| ICBG | 55.99 | N/A | 194.01 | 59.56 | N/A | 190.44 | 53.93 | N/A | 196.07 |
| Mastergraft Putty | 39.72 | 41.14 | 169.15 | 59.12 | 11.33 | 179.55 | 42.13 | 4.17 | 203.70 |
| ReBOSSIS-85 (0.2 g) | 45.70 | N/A | 204.30 | 37.52 | N/A | 212.48 | 47.75 | N/A | 202.25 |
| ReBOSSIS-85 (0.3 g) | 57.68 | N/A | 192.32 | 32.10 | N/A | 217.90 | 23.42 | N/A | 226.58 |
| ReBOSSIS-85 (0.45 g) | 61.48 | N/A | 188.52 | 33.71 | N/A | 216.29 | 28.10 | N/A | 221.90 |
| ReBOSSIS-85 (0.6 g) | 58.15 | N/A | 191.85 | 48.88 | N/A | 201.12 | 37.60 | N/A | 212.40 |
Flexibility Assessment of Stiffness: Spine fusion was assessed by manual palpation of the treated motion segments. No implanted motion segments were fused at 4 weeks, regardless of implant type. At 12 weeks, ICBG, MGP, and RB-85-0.6g were fused mechanically in at least 50% of the rabbits. No other test groups reached the 50% threshold for fusion in this model. Fusion results are listed in Table 5.
Table 5.
Manual Palpation Rates
| 4 weeks | 8 weeks | 12 weeks | |
|---|---|---|---|
| ICBG | 0% | 43% | 50% |
| MGP | 0% | 50% | 50% |
| RB-85 (0.2) | 0% | 0% | 8% |
| RB-85 (0.3) | 0% | 13% | 25% |
| RB-85 (0.45) | 0% | 38% | 36% |
| RB-85 (0.6) | 0% | 63% | 50% |
Motion analysis of the test groups in this study were compared to historical internal laboratory data of normal unfused rabbit spines. Normal motion of the rabbit lumbar spine was determined by testing 10 normal (untreated/ unfused) rabbit lumbar spinal columns using the same testing methods. Motion in the flexion/ extension plane was used as the primary measurement to demonstrate fusion (Figure 4) as manual palpation testing is based off of motion detected in flexion/extension. General findings from the biomechanics indicate that the 6 test article groups had significantly less motion than the unfused normals. Mastergraft Putty, RB-85-0.45g, and RB-85-0.6g were not significantly different from ICBG positive control. No significant differences were seen among Mastergraft Putty and RB-85-0.3g, RB-85-0.45g, or RB-85-0.6g treated fusions. Both RB-85-0.45g and RB-85-0.6g had significantly less motion than RB-85-0.2g motion segments.
Figure 4.

Range of motion at twelve weeks.
Histologic Assessment: The majority of specimens, regardless of implant type or time of implantation, had mild inflammation. Most often there were very low numbers of macrophages and multinucleated giant cells with some scattered, often erythrocytes, and plasma cells in the early time points. In all sections, there is moderate neovascularization and fibrous tissue interspersed throughout the sections (Table 6). Graft resorption and new bone formation increased with time from 4 to 12 weeks (Figure 5).
Table 6.
Hisopathology Scoring
| Implant | Term | Polymorpho-nuclear cells | Lymphocytes | Plasma cells | Macrophages | Giant cells | Necrosis | Infection | Fibrinous exudates | Neovascular-isation | Fibrosis | Graft resorption | New Bone Formation | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| ICBG | 4 | Mean | 1.00 | 1.00 | 1.00 | 1.00 | 1.30 | 0.00 | 0.00 | 0.00 | 2.40 | 3.00 | 1.40 | 1.20 |
| St.Dev. | 0.00 | 0.00 | 0.00 | 0.00 | 0.48 | 0.00 | 0.00 | 0.00 | 0.52 | 0.00 | 0.52 | 0.42 | ||
| MGP | 4 | Mean | 1.00 | 2.00 | 2.00 | 1.70 | 1.80 | 0.00 | 0.00 | 0.00 | 2.00 | 3.00 | 1.30 | 1.00 |
| St.Dev. | 0.00 | 0.00 | 0.00 | 0.48 | 0.42 | 0.00 | 0.00 | 0.00 | 0.00 | 0.00 | 0.48 | 0.00 | ||
| RB-85- 0.2g | 4 | Mean | 0.80 | 0.70 | 0.30 | 1.80 | 1.78 | 0.00 | 0.00 | 0.00 | 1.10 | 3.00 | 0.00 | 1.00 |
| St.Dev. | 0.42 | 0.48 | 0.48 | 0.42 | 0.44 | 0.00 | 0.00 | 0.00 | 0.32 | 0.00 | 0.00 | 0.00 | ||
| RB-85- 0.3g | 4 | Mean | 0.90 | 0.80 | 0.20 | 2.10 | 1.70 | 0.00 | 0.00 | 0.00 | 1.50 | 3.00 | 0.00 | 1.00 |
| St.Dev. | 0.57 | 0.42 | 0.42 | 0.74 | 0.67 | 0.00 | 0.00 | 0.00 | 0.53 | 0.00 | 0.00 | 0.00 | ||
| RB-85- 0.45g | 4 | Mean | 0.80 | 0.70 | 0.10 | 2.10 | 1.80 | 0.00 | 0.00 | 0.00 | 1.60 | 3.00 | 0.00 | 1.00 |
| St.Dev. | 0.42 | 0.48 | 0.32 | 0.74 | 0.79 | 0.00 | 0.00 | 0.00 | 0.52 | 0.00 | 0.00 | 0.00 | ||
| RB-85- 0.6g | 4 | Mean | 0.88 | 1.00 | 0.13 | 2.00 | 1.88 | 0.00 | 0.00 | 0.00 | 1.63 | 3.00 | 0.00 | 1.00 |
| St.Dev. | 0.35 | 0.00 | 0.35 | 0.00 | 0.64 | 0.00 | 0.00 | 0.00 | 0.52 | 0.00 | 0.00 | 0.00 | ||
| ICBG | 8 | Mean | 0.50 | 1.00 | 1.00 | 1.20 | 1.90 | 0.00 | 0.00 | 0.00 | 2.40 | 3.00 | 1.80 | 2.20 |
| St.Dev. | 0.53 | 0.00 | 0.00 | 0.42 | 0.32 | 0.00 | 0.00 | 0.00 | 0.52 | 0.00 | 0.42 | 0.63 | ||
| MGP | 8 | Mean | 1.00 | 2.90 | 2.00 | 2.00 | 2.00 | 0.00 | 0.00 | 0.00 | 1.10 | 3.00 | 2.00 | 2.30 |
| St.Dev. | 0.00 | 0.32 | 0.00 | 0.00 | 0.47 | 0.00 | 0.00 | 0.00 | 0.32 | 0.00 | 0.47 | 0.48 | ||
| RB-85- 0.2g | 8 | Mean | 0.80 | 0.60 | 0.30 | 2.10 | 1.50 | 0.00 | 0.00 | 0.00 | 1.40 | 2.60 | 1.00 | 1.60 |
| St.Dev. | 0.63 | 0.52 | 0.48 | 1.10 | 0.85 | 0.00 | 0.00 | 0.00 | 0.52 | 0.52 | 0.00 | 0.84 | ||
| RB-85- 0.3g | 8 | Mean | 0.90 | 0.20 | 0.30 | 2.20 | 1.20 | 0.00 | 0.00 | 0.00 | 1.30 | 2.70 | 1.30 | 1.90 |
| St.Dev. | 0.57 | 0.42 | 0.48 | 0.79 | 0.42 | 0.00 | 0.00 | 0.00 | 0.48 | 0.48 | 0.48 | 0.74 | ||
| RB-85- 0.45g | 8 | Mean | 0.90 | 0.70 | 0.40 | 2.00 | 1.70 | 0.00 | 0.00 | 0.00 | 1.50 | 2.70 | 1.40 | 2.70 |
| St.Dev. | 0.32 | 0.48 | 0.52 | 0.82 | 0.95 | 0.00 | 0.00 | 0.00 | 0.71 | 0.67 | 0.52 | 0.95 | ||
| RB-85- 0.6g | 8 | Mean | 0.80 | 0.50 | 0.30 | 1.90 | 1.30 | 0.00 | 0.00 | 0.00 | 1.50 | 2.00 | 1.40 | 2.80 |
| St.Dev. | 0.42 | 0.53 | 0.48 | 0.57 | 0.67 | 0.00 | 0.00 | 0.00 | 0.53 | 1.05 | 0.52 | 0.92 | ||
| ICBG | 12 | Mean | 0.50 | 1.00 | 1.00 | 0.90 | 0.90 | 0.00 | 0.00 | 0.00 | 2.80 | 3.00 | 2.00 | 2.70 |
| St.Dev. | 0.53 | 0.00 | 0.00 | 0.32 | 0.74 | 0.00 | 0.00 | 0.00 | 0.42 | 0.00 | 0.00 | 0.48 | ||
| MGP | 12 | Mean | 0.40 | 1.10 | 0.70 | 0.80 | 0.90 | 0.00 | 0.00 | 0.00 | 1.00 | 0.80 | 3.20 | 3.20 |
| St.Dev. | 0.52 | 1.45 | 0.95 | 1.03 | 1.20 | 0.00 | 0.00 | 0.00 | 0.00 | 1.03 | 1.03 | 1.03 | ||
| RB-85- 0.2g | 12 | Mean | 0.80 | 0.20 | 0.30 | 1.60 | 1.20 | 0.00 | 0.00 | 0.00 | 1.20 | 2.00 | 2.00 | 2.00 |
| St.Dev. | 0.42 | 0.42 | 0.48 | 0.52 | 0.42 | 0.00 | 0.00 | 0.00 | 0.79 | 0.00 | 0.00 | 0.67 | ||
| RB-85- 0.3g | 12 | Mean | 0.80 | 0.50 | 0.30 | 1.40 | 1.20 | 0.00 | 0.00 | 0.00 | 1.40 | 1.60 | 2.20 | 2.40 |
| St.Dev. | 0.42 | 0.53 | 0.48 | 0.97 | 0.63 | 0.00 | 0.00 | 0.00 | 0.70 | 0.70 | 0.63 | 1.26 | ||
| RB-85- 0.45g | 12 | Mean | 0.60 | 0.20 | 0.00 | 0.90 | 0.80 | 0.00 | 0.00 | 0.00 | 1.50 | 1.50 | 2.30 | 2.10 |
| St.Dev. | 0.52 | 0.42 | 0.00 | 0.99 | 0.42 | 0.00 | 0.00 | 0.00 | 0.71 | 0.53 | 0.48 | 0.88 | ||
| RB-85- 0.6g | 12 | Mean | 0.30 | 0.70 | 0.00 | 0.60 | 0.80 | 0.00 | 0.00 | 0.00 | 0.90 | 1.10 | 2.70 | 3.30 |
| St.Dev. | 0.48 | 0.48 | 0.00 | 0.52 | 0.42 | 0.00 | 0.00 | 0.00 | 0.57 | 0.57 | 0.48 | 0.82 |
Figure 5.

Histology slides at twelve weeks.
Discussion
In this study we evaluated the in vivo performance of an electrospun PLGA fabric combined with calcium carbonate (vaterite) and β-TCP, (ReBOSSIS-85) and compared it to Mastergraft Putty. Both materials were combined with bone marrow aspirate and iliac crest autograft, in an established posterolateral spine fusion rabbit model. The study also included a control study group using only iliac crest autograft (ICBG). The test groups were evaluated for spine fusion rate, new bone formation, graft resorption and inflammatory response using radiographic, microCT, biomechanical and histological endpoints at 4, 8 and 12 weeks following implantation.
Radiographic scoring of the fusion sites indicated a normal healing response in all test groups, with no adverse reactions and similar progressions of new bone formation over time. Fusion rates for Mastergraft Putty and ReBOSSIS-85 (0.6g) were equivalent to ICBG at 8 and 12 weeks. The remaining ReBOSSIS-85 groups (0.2, 0.3, 0.45 g) were fused 0%, 25%, and 36% respectively at 12 weeks. MicroCT scans supported the radiographic observations, showing a normal osteoconductive healing response in all groups with remodelling of the fusion site over time.
Manual palpation results, at 4 weeks, demonstrated no fusions. At 12 weeks, ICBG, MGP, and RB-85-0.6g were fused mechanically in at least 50% of the rabbits. No other test groups reached the 50% threshold for fusion in this model. All groups demonstrated significantly less range of motion in both flexion/extension, lateral bending, and axial rotation compared to normal unfused controls.
Biodegradable polymer scaffolds are commonly used in bone reconstruction, tissue engineering, drug delivery systems, and three-dimensional biomaterials. When combined with ceramic composite bone fillers like calcium carbonate, these hybrids demonstrate excellent bone-forming and mechanical properties.50-54 Electrospinning of biodegradable polymer scaffolds can create mechanically strong polymer fiber meshes with even higher porosity and flexibility, allowing for easy handling, while control over pore sizing in turn can control soft tissue intrusion at the regeneration site.55-58 These electrospun PLA and PLGA fabrics combined with calcium carbonate (vaterite) show excellent hydroxyapatite (HA)-forming ability, cellular compatibility, and biocompatibility.59, 60 The bone forming ability of these hybrid materials can be increased by doping the calcium carbonate particles with siloxane.61, 62 The siloxane hybrids (SiPVHs) allow a controlled release of soluble silica species and calcium ions.48 Trace amounts of soluble silica and calcium have been suggested to enhance the mineralization and bone forming abilities of osteogenic cells and the soluble silica species and calcium ions released by the SiPVHs reportedly stimulate the proliferation and differentiation of murine osteoblast-like cells.63-65 This indicates a possibility of siloxane being used to create new bone-repairing and regenerating materials.
Histopathology analysis of the fusion masses, from each test article and time point, indicated an expected normal response for resorbable calcium phosphates and collagen graft material when present. Mild inflammation with macrophage and multinucleated giant cell response to the graft material was evident in all test groups. Regardless of test article, new bone formation and graft resorption increased from 4 to 12 weeks post-op.
This animal study has demonstrated the biocompatibility and normal healing features associated with the ReBOSSIS-85 bone graft (test material) when combined with bone marrow aspirate and autograft as an extender. ReBOSSIS-85 was more effective when a larger mass of test article was used in this study.
References
- 1.Pugely AJ, Petersen EB, Devries-Watson N, Fredericks DC. Influence of 45S5 Bioactive Glass in A Standard Calcium Phosphate Collagen Bone Graft Substitute on the Posterolateral Fusion of Rabbit Spine. Iowa Orthop J. 2017;37:193–198. [PMC free article] [PubMed] [Google Scholar]
- 2.Arrington ED, Smith WJ, Chambers HG, et al. Complications of iliac crest bone graft harvesting. Clin.Orthop.Relat Res. 1996. pp. 300–9. [DOI] [PubMed]
- 3.Xu R, Ebraheim NA, Yeasting RA, Jackson WT. Anatomic considerations for posterior iliac bone harvesting. Spine. 1996;21(9):1017–20. doi: 10.1097/00007632-199605010-00004. [DOI] [PubMed] [Google Scholar]
- 4.Heppenstall B. Surgery of the Musculoskeletal System. New York: Churchill Livingston; 1983. Bone grafting; pp. 89–106. In: McCollster Evarts C, ed. [Google Scholar]
- 5.Chase SW, Herndon CH: The fate of autogenous and homogenous bone grafts: A historical review. J Bone Joint Surg. 1955;37A:809–841. [PubMed] [Google Scholar]
- 6.Friedlaender GE: Current concepts review: Bone grafts. J Bone Joint Surg. 1987;69A:786–790. [PubMed] [Google Scholar]
- 7.Goldberg VM, Stevenson S: Natural history of autografts and allografts. Clin Orthop. 1987;225:7–16. [PubMed] [Google Scholar]
- 8.Hall MB, Vallerand WP, Thompson D, Hartley G: Comparative anatomic study of anterior and posterior iliac crests as donor sites. J Oral Maxillofac Surg. 1991;49:560–563. doi: 10.1016/0278-2391(91)90335-j. [DOI] [PubMed] [Google Scholar]
- 9.Hsu K, Zucherman JF, White AH: Lumbar Spine Surgery. St Louis, CV Mosby; 1987. Bone Grafts and Implants in Spine Surgery; pp. 434–458. In White AH, Rothman RH, Ray CD (eds) [Google Scholar]
- 10.Kurz LT, Garfin SR, Booth RE: Harvesting autogenous iliac bone grafts: A review of complications and techniques. Spine. 1989;14:1324–1331. doi: 10.1097/00007632-198912000-00009. [DOI] [PubMed] [Google Scholar]
- 11.Fredericks DC, Smucker J, Petersen EB, et al. Effects of direct current electrical stimulation on gene expression of osteopromotive factors in a posterolateral spinal fusion model. Spine. 2007;32(2):174–81. doi: 10.1097/01.brs.0000251363.77027.49. [DOI] [PubMed] [Google Scholar]
- 12.Silber JS, Anderson DG, Daffner SD, et al. Donor site morbidity after anterior iliac crest bone harvest for single-level anterior cervical discectomy and fusion. Spine. 2003;28(2):134–9. doi: 10.1097/00007632-200301150-00008. [DOI] [PubMed] [Google Scholar]
- 13.Schnee CL, Freese A, Weil RJ, Marcotte PJ. Analysis of harvest morbidity and radiographic outcome using autograft for anterior cervical fusion. Spine. 1997;22(19):2222–2227. doi: 10.1097/00007632-199710010-00005. [DOI] [PubMed] [Google Scholar]
- 14.Smucker JD, Petersen EB, Al-hili A, Nepola JV, Fredericks DC. Assessment of SiCaP-30 in a Rabbit Posterolateral Fusion Model with Concurrent Chemotherapy. Iowa Orthop J. 2015;35:140–6. [PMC free article] [PubMed] [Google Scholar]
- 15.Smucker JD, Petersen EB, Nepola JV, Fredericks DC. Assessment of Mastergraft(®) strip with bone marrow aspirate as a graft extender in a rabbit posterolateral fusion model. Iowa Orthop J. 2012;32:61–8. [PMC free article] [PubMed] [Google Scholar]
- 16.Smucker JD, Fredericks DC. Assessment of Progenix(®) DBM putty bone substitute in a rabbit posterolateral fusion model. Iowa Orthop J. 2012;32:54–60. [PMC free article] [PubMed] [Google Scholar]
- 17.Smucker JD, Petersen EB, Fredericks DC. Assessment of MASTERGRAFT PUTTY as a graft extender in a rabbit posterolateral fusion model. Spine. 2012;37(12):1017–21. doi: 10.1097/BRS.0b013e31824444c4. [DOI] [PubMed] [Google Scholar]
- 18.Smucker JD, Bobst JA, Petersen EB, Nepola JV, Fredericks DC. B2A peptide on ceramic granules enhance posterolateral spinal fusion in rabbits compared with autograft. Spine. 2008;33(12):1324–9. doi: 10.1097/BRS.0b013e3181732a74. [DOI] [PubMed] [Google Scholar]
- 19.Fredericks D, Petersen EB, Watson N, Grosland N, Gibson-Corley K, Smucker J. Comparison of Two Synthetic Bone Graft Products in a Rabbit Posterolateral Fusion Model. Iowa Orthop J. 2016;36:167–73. [PMC free article] [PubMed] [Google Scholar]
- 20.Fredericks DC, Petersen EB, Sahai N, et al. Evaluation of a novel silicate substituted hydroxyapatite bone graft substitute in a rabbit posterolateral fusion model. Iowa Orthop J. 2013;33:25–32. [PMC free article] [PubMed] [Google Scholar]
- 21.Schliephake H, Neukam FW, Klosa D. Influence of pore dimensions on bone ingrowth into porous hydroxylapatite blocks used as bone graft substitutes. A histometric study. Int J Oral Maxillofac Surg. 1991;20:53–8. doi: 10.1016/s0901-5027(05)80698-8. doi: 10.1016/S0901-5027(05)80698-8. [DOI] [PubMed] [Google Scholar]
- 22.Holmes RE, Wardrop RW, Wolford LM. Hydroxylapatite as a bone graft substitute in orthognathic surgery. J Oral Maxillofac Surg. 1988;46:661–71. doi: 10.1016/0278-2391(88)90109-7. [DOI] [PubMed] [Google Scholar]
- 23.Holmes RE. Bone regeneration within a coralline hydroxyapatite implant. Plast Reconstr Surg. 1979;63:626–33. doi: 10.1097/00006534-197905000-00004. [DOI] [PubMed] [Google Scholar]
- 24.Holmes RE, Buchholz RW. Mooney Porous hydroxyapatite as a bone-graft substitute in metaphyseal defects. J Bone Joint Surg. 1986;68A:904–11. [PubMed] [Google Scholar]
- 25.Klawitter JJ, Hulbert SF. Application of porous ceramics for the attachment of load bearing internal orthopedic applications. J Biomed Mater Res Symposium. 1971;2:161–229. [Google Scholar]
- 26.Yuan H, Yang Z, Li Y, Zhang X, De Bruijn JD, De Groot K. Osteoinduction by calcium phosphate biomaterials. J Mater Sci Mater Med. 1998;9:723–6. doi: 10.1023/a:1008950902047. doi: 10.1023/A:1008950902047. [DOI] [PubMed] [Google Scholar]
- 27.Baas J, Svaneby D, Jensen TB, Elmengaard B, Bechtold J, Soballe K. Coralline hydroxyapatite granules inferior to morselized allograft around uncemented porous Ti implants: unchanged fixation by addition of concentrated autologous bone marrow aspirate. J Biomed Mater Res A. 2011;99:9–15. doi: 10.1002/jbm.a.33156. doi: 10.1002/jbm.a.33156. [DOI] [PubMed] [Google Scholar]
- 28.van Haaren EH, Smit TH, Phipps K, Wuisman PI, Blunn G, Heyligers IC. Tricalcium-phosphate and hydroxyapatite bone-graft extender for use in impaction grafting revision surgery. An in vitro study on human femora. J Bone Joint Surg Br. 2005;87:267–271. doi: 10.1302/0301-620x.87b2.14749. [DOI] [PubMed] [Google Scholar]
- 29.Johnson KD, Frierson KE, Keller TS, et al. Porous ceramics as bone graft substitutes in long bone defects: a biomechanical, histological, and radiographic analysis. J Orthop Res. 1996;14:351–69. doi: 10.1002/jor.1100140304. [DOI] [PubMed] [Google Scholar]
- 30.Odri G-A, Revert R, Delecrin J, et al. Effect of adding bone marrow to ceramic graft materials with different interconnectivities in lumbar arthrodesis. Journal of Orthopaedic Science. 2013;18(2):321–330. doi: 10.1007/s00776-012-0336-3. doi:10.1007/s00776-012-0336-3. [DOI] [PubMed] [Google Scholar]
- 31.Boden SD, Martin GJ, Morone M, et al. The use of coralline hydroxyapatite with bone marrow, autogenous bone graft, or osteoinductive bone protein extract for posterolateral lumbar spine fusion. Spine. 1999;24:320–7. doi: 10.1097/00007632-199902150-00003. [DOI] [PubMed] [Google Scholar]
- 32.Hutmacher D.W. Scaffolds in tissue engineering bone and cartilage. Biomaterials. 2000;21:>2529. doi: 10.1016/s0142-9612(00)00121-6. [DOI] [PubMed] [Google Scholar]
- 33.Reece GP, Patrick CW. Frontiers in tissue engineering. New York, USA: Elsevier Science; 1998. Tissue engineered construct design principles; pp. 166–96. In: Patrick Jr CW, Mikos AG, McIntire LV, editors. [Google Scholar]
- 34.Obata A, Hotta T, Wakita T, Ota Y, Kasuga T. Electrospun microfiber meshes of silicon-doped vaterite/poly(lactic acid) hybrid for guided bone regeneration. Acta Biomater. 2010;6(4):1248–57. doi: 10.1016/j.actbio.2009.11.013. [DOI] [PubMed] [Google Scholar]
- 35.Xynos ID, Edgar AJ, Buttery LD, Hench LL, Polak JM. Ionic products of bioactive glass dissolution increase proliferation of human osteoblasts and induce insulin-like growth factor II mRNA expression and protein synthesis. Biochem Biophys Res Commun. 2000;276(2):461–5. doi: 10.1006/bbrc.2000.3503. [DOI] [PubMed] [Google Scholar]
- 36.Reffitt D, Ogston N., Jugdaohsingh R., Cheung H., Evans B.A.J., Thompson R., Powell J., Hampson G. Orthosilicic acid stimulates collagen type 1 synthesis and osteoblastic differentiation in human osteoblast-like cells in vitro. Bone. 2003;32:127–135. doi: 10.1016/s8756-3282(02)00950-x. [DOI] [PubMed] [Google Scholar]
- 37.Thian ES, Huang J, Best SM, et al. The response of osteoblasts to nanocrystalline silicon-substituted hydroxyapatite thin films. Biomaterials. 2006;27(13):2692–8. doi: 10.1016/j.biomaterials.2005.12.019. [DOI] [PubMed] [Google Scholar]
- 38.Boden SD, Schimandle JH, Hutton WC. An experimental lumbar intertransverse process spinal fusion model. Radiographic, histologic, and biomechanical healing characteristics. Spine. 1995. pp. 412–20. [DOI] [PubMed]
- 39.Grauer JN, Erulkar JS, Patel TC et al. Biomechanical evaluation of the New Zealand white rabbit lumbar spine: a physiologic characterization. EurSpineJ. 2000;9:250–5. doi: 10.1007/s005860000141. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Grauer JN, Vaccaro AR, Kato M, et al. Development of a New Zealand white rabbit model of spinal pseudarthrosis repair and evaluation of the potential role of OP-1 to overcome pseudarthrosis. Spine. 2004;29:1405–12. doi: 10.1097/01.brs.0000129028.25671.96. [DOI] [PubMed] [Google Scholar]
- 41.Boden SD, Schimandle JH, Hutton WC. Lumbar intertransverse-process spinal arthrodesis with use of a bovine bone-derived osteoinductive protein. A preliminary report. Journal of Bone&Joint Surgery. 1995;77:1404–17. doi: 10.2106/00004623-199509000-00017. [DOI] [PubMed] [Google Scholar]
- 42.Boden SD, Schimandle JH, Hutton WC et al. In vivo evaluation of a resorbable osteoinductive composite as a graft substitute for lumbar spinal fusion. Journal of Spinal Disorders. 1997;10:1–11. [PubMed] [Google Scholar]
- 43.Valdes M, Palumbo M, Appel AJ, Mcallister S, Ehrlich M. Posterolateral intertransverse lumbar arthrodesis in the New Zealand White rabbit model: II. Operative technique. Spine J. 2004;4(3):293–9. doi: 10.1016/j.spinee.2003.08.022. [DOI] [PubMed] [Google Scholar]
- 44.Gupta AR, Shah NR, Patel TC, Grauer JN. Peri-operative and long-term complications of iliac crest bone graft harvesting for spinal surgery: a quantitative review of the literature. Int Med J. 2001;8:163–6. [Google Scholar]
- 45.Magit DP, Maak T, Trioano N, et al. Healos/recombinant human growth and differentiation factor-5 induces posterolateral lumbar fusion in a New Zealand white rabbit model. Spine. 2006;31(19):2180–8. doi: 10.1097/01.brs.0000232823.82106.0a. [DOI] [PubMed] [Google Scholar]
- 46.Cockin J. Autologous bone grafting-complications at the donor site. J Bone Joint Surg. 1971;53B:153. [Google Scholar]
- 47.Shiels SM, Raut VP, Patterson PB, Barnes BR, Wenke JC. Antibiotic-loaded bone graft for reduction of surgical site infection in spinal fusion. Spine J. 2017;17(12):1917–1925. doi: 10.1016/j.spinee.2017.06.039. [DOI] [PubMed] [Google Scholar]
- 48.Bramwell V.W., Eyles J.E. Oya Alpar Particulate delivery systems for biodefence subunit vaccines. Adv Drug Deliv Rev. 2005;57:1247–1265. doi: 10.1016/j.addr.2005.01.010. [DOI] [PubMed] [Google Scholar]
- 49.Santerre J.P., Woodhouse K., Laroche G., Labow R.S. Understanding the biodegradation of polyurethanes: from classical implants to tissue engineering materials. Biomaterials. 2005;26:7457–7470. doi: 10.1016/j.biomaterials.2005.05.079. [DOI] [PubMed] [Google Scholar]
- 50.Kim S.S., Park M.S., Jeon O., Choi C.Y., Kim B.S. Poly(lactide-co-glycolide)/hydroxyapatite composted scaffolds for bone tissue engineering. Biomaterials. 2006;27:1399–1409. doi: 10.1016/j.biomaterials.2005.08.016. [DOI] [PubMed] [Google Scholar]
- 51.Zang K., Wang Y., Hillmyer M.A. Francis Processing and properties of porous poly(l-lactide)/ bioactive glass composites. Biomaterials. 2004;25:2489–2500. doi: 10.1016/j.biomaterials.2003.09.033. [DOI] [PubMed] [Google Scholar]
- 52.Kasuga T., Maeda H., Kato K., Nogami M., Hata K., Ueda M. Preparation of poly(lactic acid) composites containing calcium carbonate (vaterite) Biomaterials. 2003;24:3247–3253. doi: 10.1016/s0142-9612(03)00190-x. [DOI] [PubMed] [Google Scholar]
- 53.Liao H., Mutvei H., Sjöström M., Hammarström L., Li J. Tissue responses to natural aragonite (Margaritifera shell) implants in vivo. Biomaterials. 2000;21:457–468. doi: 10.1016/s0142-9612(99)00184-2. [DOI] [PubMed] [Google Scholar]
- 54.Vago R., Plotquin D., Bunin A., Sinelnikov I., Atar D., Itzhak D. Hard tissue remodeling using biofabricated coralline biomaterials. J Biochem Biophys Methods. 2002;50:253–259. doi: 10.1016/s0165-022x(01)00235-4. [DOI] [PubMed] [Google Scholar]
- 55.Still T.J., von Recum H.A. Electrospinning: applications in drug delivery and tissue engineering. Biomaterials. 2008;29:1989–2006. doi: 10.1016/j.biomaterials.2008.01.011. [DOI] [PubMed] [Google Scholar]
- 56.Li W.J., Laurencin C.T., Caterson E.J., Tuan R.S., Ko F.K. Electrospun nanofibrous structure: a novel scaffold for tissue engineering. J Biomed Mater Res. 2002;60:613–621. doi: 10.1002/jbm.10167. [DOI] [PubMed] [Google Scholar]
- 57.Xin X., Hussain M., Mao J.J. Continuing differentiation of human mesenchymal stem cells and induced chondrogenic and osteogenic lineages in electrospun PLGA nanofiber scaffold. Biomaterials. 2007;28:316–325. doi: 10.1016/j.biomaterials.2006.08.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Wakita T., Obata A., Poologasundarampillai G., Jones J.R., Kasuga T. Preparation of electrospun siloxane-poly(lactic acid)-vaterite hybrid fibrous membranes for guided bone regeneration. Compos. Sci. Technol. 2010;70(13):1889–1893. [Google Scholar]
- 59.Maeda H., Kasuga T., Hench L.L. Preparation of poly(l-lactic acid)–polysiloxane–calcium carbonate hybrid membranes for guided bone regeneration. Biomaterials. 2006;27:1216–1222. doi: 10.1016/j.biomaterials.2005.08.010. [DOI] [PubMed] [Google Scholar]
- 60.Maeda H., Kasuga T., Nogami M. Bonelike apatite coating on skeleton of poly(lactic acid) composite sponge. Mater Trans. 2004;45:989–993. [Google Scholar]
- 61.Obata A., Tokuda S., Kasuga T. Enhanced in vitro cell activity on silicon-doped vaterite/poly(lactic acid) composites. Acta Biomater. 2009;5:57–62. doi: 10.1016/j.actbio.2008.08.004. [DOI] [PubMed] [Google Scholar]
- 62.Obata A, Hotta T, Wakita T, Ota Y, Kasuga T. Electrospun microfiber meshes of silicon-doped vaterite/poly(lactic acid) hybrid for guided bone regeneration. Acta Biomater. 2010;6(4):1248–57. doi: 10.1016/j.actbio.2009.11.013. [DOI] [PubMed] [Google Scholar]
- 63.Xynos I.D., Edgar A.J., Buttery L.D.K., Hench L.L., Polak J.M. Ionic products of bioactive glass dissolution increase proliferation of human osteoblasts and induce insulin-like growth factor II mRNA expression and protein synthesis. Biochem Biophys Res Commun. 2000;276:461–465. doi: 10.1006/bbrc.2000.3503. [DOI] [PubMed] [Google Scholar]
- 64.Gough J.E., Jones J.R., Hench L.L. Nodule formation and mineralisation of human primary osteoblasts cultured on a porous bioactive glass scaffold. Biomaterials. 2004;25:2039–2046. doi: 10.1016/j.biomaterials.2003.07.001. [DOI] [PubMed] [Google Scholar]
- 65.Jones J.R., Tsigkou O., Coates E.E., Stevens M.M., Polak J.M., Hench L.L. Extracellular matrix formation and mineralization on a phosphate-free porous bioactive glass scaffold using primary human osteoblast (HOB) cells. Biomaterials. 2007;28:1653–1663. doi: 10.1016/j.biomaterials.2006.11.022. [DOI] [PubMed] [Google Scholar]


