Abstract
Background
Tumor lysis syndrome (TLS) is the most common life-threatening oncological emergency encountered by physicians treating children with lymphoproliferative malignancies. Healthcare providers should be aware of the condition in order to prevent occurrence and prompt timely management to avoid severe consequences.
Objective
To provide an update on the current understanding, evaluation, and management of tumor lysis syndrome in childhood malignancies.
Methods
A PubMed search was performed in Clinical Queries using the keywords ‘tumor lysis syndrome’ and ‘malignancies’ with Category limited to clinical trials and reviews for ages from birth to 18 years.
Results
There were 22 clinical trials and 37 reviews under the search criteria. TLS is characterized by acute electrolyte and metabolic disturbances resulting from massive and abrupt release of cellular contents into the circulation due to breakdown of tumor cells. If left untreated, it can lead to multiorgan compromise and eventually death. Apart from close monitoring and medical therapies, early recognition of risk factors for development of TLS is also necessary for successful management.
Conclusions
Prophylactic measures to patients at risk of TLS include aggressive fluid management and judicious use of diuretics and hypouricemic agents. Both allopurinol and urate oxidase are effective in reducing serum uric acid. Allopurinol should be used as prophylaxis in low-risk cases while urate oxidase should be used as treatment in intermediate to high-risk cases. There is no evidence on better drug of choice among different urate oxidases. The routine use of diuretics and urine alkalinization are not recommended. Correction of electrolytes and use of renal replacement therapy may also be required during treatment of TLS.
Keywords: hyperkalemia, hyperphosphatemia, hyperuricemia, hypocalcemia, lymphoproliferative malignancies
Introduction
Tumor lysis syndrome (TLS) is a potentially life-threatening complication of induction chemotherapy for treatment of childhood malignancies, in particular, lymphoproliferative malignancies.1–4 TLS is characterized by a group of acute electrolyte and metabolic disturbances caused by massive and abrupt release of intracellular contents such as nucleic acids, uric acid, phosphorus, and potassium into the circulation due to breakdown of tumor cells.3,5 It is most commonly seen in the first few days after the start of cytotoxic therapy especially in hematological malignancies such as acute lymphoblastic leukemia and high-grade lymphomas (particularly, Burkitt’s lymphoma).3,6 TLS in solid malignancies with a high tumor load, high proliferative rates, or high sensitivity to chemotherapy have also been published in several pediatric case reports.7–9 Spontaneous TLS, which develops in the absence of cytotoxic therapy, had also been described in children with hematological malignancies.10–12 As a result of breakdown of tumor cells, potentially fatal metabolic derangements such as hyperkalemia, hyperphosphatemia, hypocalcemia, and hyperuricemia may develop. First manifestations of TLS could be subtle and are commonly seen in oncologic patients without TLS, such as nausea, vomiting, diarrhea, muscle cramps, and paresthesia.9 Edema, arrhythmias, and seizures are often late presentations.9
Therefore, keys to prevention and treatment of TLS include a high index of suspicion, early recognition of metabolic and renal complications, and prompt of management of the condition, which includes both prophylaxis and treatment. An awareness of its physiologic consequences, predisposing risk factors, and knowledge on updated management measures are essential prerequisites. This review aims to provide an update on the current understanding, evaluation, and management of TLS in childhood malignancies.
Methods
A PubMed search was performed in Clinical Queries in April 2019 using the keywords ‘tumor lysis syndrome’ and ‘malignancies’ with Category limited to clinical trials and reviews for ages from birth to 18 years. There were 22 clinical trials and 37 reviews under the search criteria. A literature review of these articles was performed. This review is based on but not limited to these articles.
Definition
In 1993, Hande–Garrow proposed a system that identified TLS as laboratory or clinical TLS within 4 days of initial chemotherapy.6 However, this system did not account for patients who had pre-existing abnormal laboratory values before start of treatment and those who had developed abnormal laboratory values after the 4 days of initial therapy.6
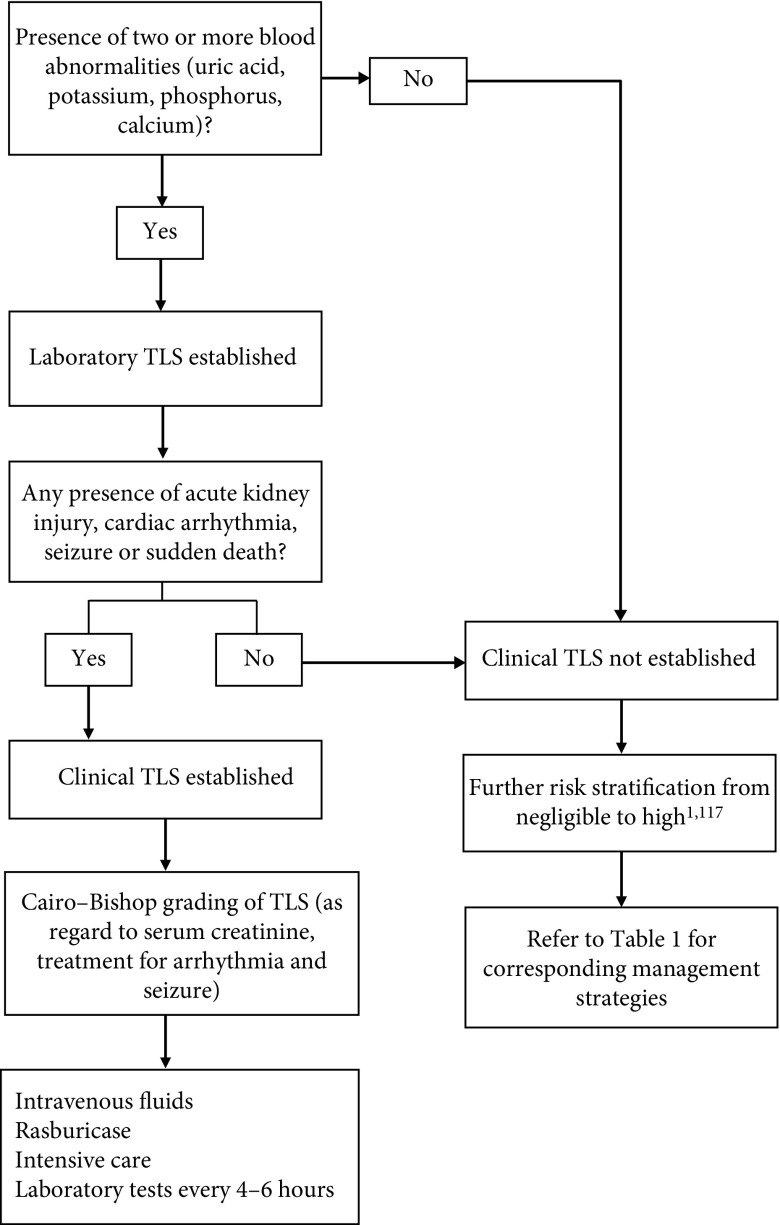
In 2004, Cairo–Bishop proposed a new classification of TLS (Figure 1).3,13 According to Cairo–Bishop’s definition of TLS, laboratory criteria for its diagnosis require two or more abnormal serum values at presentation, including either a 25% increase or decrease in levels of calcium, uric acid, potassium, or phosphorus within 3 days before or up to 7 days after the start of chemotherapy.3 The presumption for laboratory diagnosis is that the patient has or will receive adequate hydration (with or without alkalinization) and a hypouricemic agent(s).14 Criteria for the diagnosis of clinical TLS require the presence of laboratory TLS plus one or more clinical complications, which could be renal insufficiency (defined as serum creatinine more than or equal to 1.5 times the upper limit of normal), cardiac arrhythmias, seizures, or sudden death.3 These clinical complications should not be directly or probably attributable to a therapeutic agent.
Figure 1.
Classification and management algorithm in children with risk of tumor lysis syndrome (TLS) 1,3,13,117
In 2011, Howard and colleagues suggested that two or more abnormal laboratory values should be present simultaneously to define laboratory TLS as abnormality that developed at a later time could be unrelated to TLS. These authors also suggested that a 25% change should not be considered as a criterion, as such a change could be insignificant unless the values are out of the normal range. Lastly, symptomatic hypocalcemia is also recommended to be included in clinical TLS.1,13,15
Nonetheless, the original Cairo–Bishop classification has been widely used to establish guidelines for the prevention and management of pediatric and adult TLS.14,16
A grading system for severity of TLS is also proposed by Cairo–Bishop.14 There are a total of five grades ranging from Grade 0 to Grade 5, which range from absence of TLS to death, respectively (Figure 1).13
Epidemiology
The incidence and prevalence of TLS varies among different malignancies and management strategies. Generally, bulky, aggressive, treatment-sensitive tumors, as well as non-implementation of prophylactic measures, are associated with higher frequencies of TLS.15 Studies have quoted incidence of TLS ranging from 4.4% to 53.6% in hematological malignancies of childhood.10,15–20
The majority of these patients have laboratory TLS alone. However, some may progress into clinical TLS.15,16 In a resource-limited setting, this proportion of patients can reach 15.9%.16 TLS-related mortality of up to 21.4% was reported in pediatric patients with hematological malignancies.21–23
Pathophysiology
TLS is a direct consequence of rapid lysis of malignant cells with the release of intracellular metabolites into the circulation.1,7,24,25 As the load of circulating intracellular products increases, the homeostatic control mechanisms of such substances are overwhelmed, resulting in the clinical sequelae of TLS. Among the cellular products, potassium, phosphorus, and nucleic acids play a major role in the pathophysiology of TLS. Although TLS can occur spontaneously, it is most often seen 48–72 hours after initiation of chemotherapy.26,27
Hyperkalemia
Hyperkalemia is the most rapidly fatal consequence of TLS. It is often the first observed effect of TLS and can occur within 6 hours after the start of chemotherapy.28,29 Lysis of tumor cells lead to the release of large amounts of potassium, a major intracellular ion, into the circulation.25,30 Moreover, serum potassium level is also raised by co-existing renal failure and acidosis.31 Severe hyperkalemia results when uptake capacity by muscle and liver is exceeded.32,33 Initial manifestations of hyperkalemia include neuromuscular effects, such as muscle weakness and paresthesia, as well as electrocardiogram abnormalities, such as peaked T waves, prolonged PR and QRS interval, and sine wave morphology. Without intervention, devastating complications, for example, cardiac dysrhythmias, ventricular tachycardia, ventricular fibrillation, or cardiac arrest, may ensue.34
Hyperphosphatemia
Tumor cells may contain four times more intracellular phosphate as compared to normal mature lymphoid cells.6 Rapid release of phosphate stores can overload the kidney’s excretory capacity, resulting in hyperphosphatemia.35–37 Symptoms of hyperphosphatemia include nausea, vomiting, diarrhea, lethargy, and seizures. In general, hyperphosphatemia is more commonly associated with TLS that occurs after chemotherapy initiation rather than spontaneous TLS. This is because in spontaneous TLS, tumor cells are able to reuse the released phosphorus for regeneration of new tumor cells, and thus they are less likely to exhibit extreme hyperphosphatemia.38
More importantly, phosphates in the blood can bind to calcium cations, leading to hypocalcemia. Severe hypocalcemia can cause arrhythmias, hypotension, tetany, and muscle cramps.34,39 In addition, calcium phosphate precipitation in renal tubules and cardiac conduction systems can lead to renal failure and cardiac failure, respectively.35–37 In acute renal failure, decreased calcitriol levels can also cause hypocalcemia. In fact, upon the use of highly effective hypouricemic drugs, calcium phosphate precipitation becomes the major cause of acute kidney injury in TLS.6,37,40
Hyperuricemia
Lysed tumor cells release purine nucleic acids, which are metabolized into xanthine. Xanthine is then metabolized into uric acid by hepatic xanthine oxidase, resulting in hyperuricemia. Uric acid is poorly soluble in water, and its solubility is even poorer in an acidic environment. As the concentration of uric acid increases along the kidney tubules, the likelihood of uric acid crystal formation and precipitation increases, causing tubular obstruction and urate nephropathy. With the development of hypouricemia drugs, hyperuricemia is no longer the major consequence of TLS.41 Xanthine, on the other hand, has lower water solubility than uric acid.42 High concentration of xanthine, especially in patients treated with allopurinol, predisposes these patients to xanthine nephropathy or urolithiasis.43–48
Acute renal failure
TLS is also commonly associated with acute renal failure. The most common cause of acute renal failure in TLS is due to the precipitation of uric acid crystals in renal tubules.3 Decreased tubular flow rate and renal medullary hemoconcentration also contributes to uric acid crystallization.49 Other mechanisms include calcium-phosphate crystal deposition, xanthine crystallization, tumor infiltration in the kidneys, tumor-associated obstructive uropathy, drug-associated nephrotoxicity, sepsis, and pre-existing volume depletion and/or renal dysfunction.3,14 Furthermore, lysis of tumor cells releases cytokines, which can exacerbate acute kidney injury, elicit a systemic inflammatory response syndrome, and eventually cause multiorgan failure.24,50
Risk factors
In recent decades, a number of risk-prediction models for TLS have been proposed for children with hematological malignancies.9,19,51–53 However, each of these models is applicable to certain oncological diseases only. They also lack standardized supportive care guidelines for respective diseases and have complex scoring systems.1,49 In 2010, an international expert TLS panel developed recommendations for risk stratification of TLS in children and adults with solid and hematological malignancies.11 This model classified TLS into low, intermediate, and high-risk types. The classification was based on three phases: first, to confirm the presence of laboratory TLS (defined by Hande–Garrow and Cairo–Bishop), then to define risk according to patient and disease characteristics, and lastly to perform risk adjustment by the degree of renal impairment. Associated TLS prophylaxis for each type is also proposed. This has provided a relatively simple clinical tool for health care providers in the management of both pediatric and adult oncology patients. In 2011, Howard and colleagues developed another approach to standardize the definition of TLS and the supportive care guidelines for each cancer type.1,49 In this approach, the risk types for TLS are classified into five categories with respective management recommendations (Table 1). In summary, risk for development of TLS depends on multiple factors, which could be categorized into tumor and patient characteristics and management-related issues (Table 2).1,14,49,54–56
Table 1.
| Risk | Serum potassium, phosphorus, calcium, creatinine, uric acid, urine output | Cancer mass and cell-lysis potential | Management |
|---|---|---|---|
| Negligible | ≤1 abnormal value | Small or resected localized tumor OR Medium size tumor with low cell-lysis potential |
No prophylaxis or monitoring required |
| Low | ≤1 abnormal value | Medium size tumor with medium/unknown cell-lysis potential* OR Large cancer mass with low cell-lysis potential |
Intravenous fluids Allopurinol Daily laboratory tests |
| Intermediate | ≤1 abnormal value | Large cancer mass with medium/unknown cell-lysis potential | Intravenous fluids Allopurinol or rasburicase Laboratory tests every 8–12 hours |
| High | Can be absent | Medium or large cancer mass with high cell-lysis potential OR Established laboratory TLS |
Intravenous fluids Rasburicase Cardiac monitoring Laboratory tests every 6–8 hours |
| Established | ≥2 abnormal values | Established clinical TLS | Intravenous fluids Rasburicase Intensive care Laboratory tests every 4–6 hours |
Patient should not have pre-existing nephropathy, dehydration, acidosis, hypotension, or nephrotoxin exposure. If present, risk should be upgraded to intermediate.
TLS: tumor lysis syndrome.
Table 2.
| Category | Risk factors |
|---|---|
| Tumor characteristics | Tumor type: TLS occurs most frequently in patients with non-Hodgkin’s lymphoma and other hematologic malignancies, particularly Burkitt’s lymphoma, acute lymphoblastic leukemia, and acute myeloid leukemia. Tumor burden: Larger tumor mass (>10 cm), higher number of cells that will lyse with treatment (≥25,000 circulating tumor cells/μL),15 tumor infiltration as evidenced by organomegaly (particularly renal infiltration or urine outflow tract obstruction), and bone marrow involvement are signs of high tumor load. Rate of proliferation: The higher the level of lactate dehydrogenase (>2 times the upper limit of normal range), which is a marker for tumor proliferation, the higher the risk for TLS. Sensitivity to anticancer therapy: Malignancies that are more sensitive to chemotherapy have a higher rate of cell lysis and thus pose a greater risk to TLS. |
| Patient characteristics | Nephropathy before diagnosis of cancer: Pre-existing nephropathy of any cause, such as hypertension and diabetes, predisposes the patient to higher risk of TLS. Pre-existing or co-existing conditions causing impaired kidney perfusion: These include conditions such as dehydration, volume depletion, and hypotension. In such conditions, urine flow in kidney tubules are reduced. Thus, solutes concentration in tubules increases and can cause crystallization, leading to nephropathy. Pre-existing hyperuricemia: Raised baseline serum uric acid (>450 μmol/L) increases the risk of TLS. High pretreatment lactate dehydrogenase: Baseline serum lactate dehydrogenase greater than twice the upper limit of normal increases risk of TLS. |
| Management related | Inadequate hydration: Hypovolemic state decreases rate of urine flow in kidney tubules and facilitates solutes crystallization and precipitation, causing acute kidney injury. Exogenous potassium: Exogenous sources of potassium such as intravenous fluids, medications, or food may enhance the risk of development of TLS. Exogenous phosphate: Dietary phosphate could be an additional load to high levels of serum phosphate from tumor lysis, causing an extra burden to kidneys’ excretion. Exposure to nephrotoxin: Nephrotoxic agents such as vancomycin, aminoglycosides, and contrast agents for imaging increase the risk of acute kidney injury from tumor lysis. Delayed uric acid removal: The longer the uric acid level remains high, the greater the risk of crystal formation and kidney injury. Intensity of initial anticancer therapy: The higher the intensity of initial chemotherapy, the greater the rate of tumor cells lysis and thus a higher risk of TLS. Choice of chemotherapy: Cases of TLS with use of certain chemotherapeutic agents, for example, alvocidib, dinaciclib, obinutuzumab, venetoclax, and rituximab, have been reported. |
TLS: tumor lysis syndrome.
Management
Early identification of patients at risk for TLS is essential for timely management. The best management of TLS is prevention. Prophylactic measures should be applied to patients at risk of TLS. Preventive measures include aggressive fluid management, use of diuretics, and hypouricemic agents. Treatment strategies, apart from the use of hypouricemic agents, include correction of electrolytes and use of renal replacement therapy. Close monitoring of renal and cardiac parameters is also important in the management of these patients.
Fluid management
Volume depletion is a major risk factor for TLS. Aggressive hydration and diuresis are fundamental to the prevention and management of TLS. Adequate hydration improves the intravascular volume, enhances renal perfusion and glomerular filtration, and further promotes the excretion of uric acid, potassium, and phosphate. This may also delay and prevent the need for renal replacement therapy.1,14,29,49,57 Aggressive hydration refers to hyperhydration using intravenous crystalloids of 2.5 (up to 3) L/m2/day to achieve a target urine output of at least 4 ml/kg/hour for infants and 100 ml/m2/hour for older patients.1,49,58 It is also prudent to limit the potassium content in intravenous fluids. Use of loop diuretics is also commonly recommended in clinical practice if urine output is suboptimal after achieving adequate hydration or if the patient is prone to volume overload.1,32,33,49 Furosemide may be considered for the normovolemic patient with hyperkalemia or for the patient with obvious fluid overload. However, routine use of diuretics is not recommended as use of diuretics may lead to volume depletion and may contribute to calcium phosphate and uric acid precipitation in renal tubules in patients with volume depletion. The use of diuretics is contraindicated in patients with obstructive uropathy.14,32,33 Current recommendations on diuretic use are based on expert opinions, and there have been no published evidenced-based studies assessing the role of diuretics in the management of TLS yet.
Urine alkalinization
Urine alkalinization with the use of intravenous sodium bicarbonate solution was widely advocated for the management of TLS. However, this practice has to be carried out with caution. Theoretically, urine alkalinization increases the solubility of uric acid and promotes its excretion. On the other hand, it decreases the solubility of calcium phosphate and may increase the risk of calcium phosphate precipitation. The resulting alkaline serum pH also promotes albumin-calcium binding and exacerbates hypocalcemia.7 Moreover, the solubility of xanthine and hypoxanthine is low in alkaline conditions and can cause xanthine crystal precipitation and obstructive nephropathies, especially in cases managed with allopurinol and rasburicase.3,14,41 The only indication for alkalinization of urine is in patients with metabolic acidosis.
Xanthine oxidase inhibitors
Allopurinol is a xanthine analog. It acts as a competitive inhibitor of xanthine oxidase, blocks the conversion of purine metabolites into uric acid, and has been shown to prevent the development of obstructive uropathy caused by precipitation of uric acid crystals in renal tubules.14,59,60 In view of its mechanism of action, allopurinol does not reduce pre-existing serum uric acid level. Therefore, the reduction of serum uric acid level may take several days.61 Thus, allopurinol is recommended to be used for prophylaxis in low to intermediate risk of TLS rather than treatment of established TLS.1,14,49,62 For patients with pre-existing hyperuricemia, urate oxidase is the preferred hypouricemia agent.
Allopurinol prophylaxis should be initiated at least 12–24 hours prior to start of chemotherapy and continued until uric acid levels are normalized, and tumor burden and white cells count have returned to low risk levels (≤50,000/μL for acute lymphoblastic lymphoma and ≤10,000/μL for acute myeloid leukemia).14 Allopurinol is usually given orally. For patients who are unable to tolerate the medication orally, allopurinol can be given intravenously. The oral dosage for children is 300–450 mg/m2/day in three divided doses up to 400 mg daily. In infants weighing <10 kg, the dose is 3.3 mg/kg every 8 hours.58,63 The dosage would need to be adjusted in the presence of acute kidney injury, leading to a reduction in creatinine clearance.
Urate oxidase
Urate oxidase is an enzyme that is present in most mammals that converts uric acid to allantoin. Allantoin is five- to ten-fold more soluble in urine than uric acid and is readily excreted.62 However, this enzyme is absent in humans due to a non-sense mutation in the coding region. Urate oxidase is therefore widely used in patients with cancer for prevention and treatment of hyperuricemia. There are two types of urate oxidase: non-recombinant type Uricozyme® (Sanofi-Aventis, United States) and recombinant type rasburicase. Non-recombinant urate oxidase Uricozyme is a natural uricase obtained from Aspergillus flavus cultures, while recombinant urate oxidase rasburicase is obtained by recombinant DNA technique from a genetically modified strain of Saccharomyces cerevisiae, which is cloned from a strain of A. flavis.33,64,65
A recent Cochrane systematic review of (randomized) controlled clinical trials on urate oxidase for prevention and treatment of TLS in children with cancer found that although urate oxidase might be effective in reducing serum uric acid in numerous uncontrolled studies, there is little evidence supporting its effectiveness in preventing or treating TLS.66 In the Cochrane systematic review, there were only two randomized controlled trials and five controlled clinical trials on the use of urate oxidase in the prophylaxis of TLS, and there were no trials on its use in the treatment of established TLS.66 Despite the paucity of evidence in significant clinical benefits, the authors of the Cochrane systematic review suggested that this could be a false negative result due to inadequate sample size of the existing clinical trials and that its probable effectiveness in reducing serum uric acid could be an important surrogate outcome.66 Trials of larger sample size are needed to evaluate the role of urate oxidase for the prevention and treatment of TLS in children.
Uricozyme has been used as a uricolytic agent for the past several decades. It is administered by intravenous infusion at 100 units/kg/day. However, due to product-related impurities, it can be complicated with severe acute hypersensitivity reactions such as anaphylaxis and bronchospams in 5% of patients.62,67–69
Rasburicase was then developed in 2001 to reduce the occurrence of these reactions. It is the drug of choice for intermediate- to high-risk TLS patients in view of its quicker onset of action than that of allopurinol.1,67,70–76 Rasburicase is administered by intravenous infusion at 0.2 mg/kg/dose once daily for up to 5 days.3,9,58
The optimal treatment duration has not been determined.70,71,77–83 Further research includes the optimal number of doses needed, optimal dose based on uric acid levels and tumor burden, dosing in obese patients, and maximum dose.84 Possible side effects from rasburicase, such as anaphylaxis and methemoglobinemia, are rare (<1%).19,70,82,83 It is also important to note that urate oxidases can cause hemolytic anemia in patients with glucose-6-phosphate dehydrogenase (G6PD) deficiency and is therefore contraindicated. It is recommended that clinicians screen for G6PD deficiency prior to urate oxidase use in patients at risk, specifically in males of African, Southeast Asian, and Mediterranean ethnicity.9,85,86 However, this is not always feasible, as TLS often progresses quickly if treatment is delayed. In such cases, strict monitoring for signs and symptoms of hemolytic anemia after administration of urate oxidase is crucial.79,85–87
Novel xanthine oxidase inhibitors and urate oxidase
Febuxostat is a novel xanthine oxidase inhibitor developed in early 2000s; it lacks the hypersensitivity profile of allopurinol. This is because febuxostat is not a purine analog and therefore had little effects on other enzymes involved in purine and pyrimidine metabolism.88,89 As it is metabolized in the liver, it does not require dosage adjustments in patients with renal impairment, making it a feasible alternative to allopurinol for patients with allopurinol hypersensitivity or renal impairment.7,76 A recent phase III study comparing febuxostat and allopurinol for the prevention of TLS in adults showed a significant lower serum uric acid in the group using febuxostat with comparable renal function preservation and safety profile.90
However, in 2019, based on the preliminary results from a safety clinical trial on febuxostat in adults, the US Food and Drug Administration recommended febuxostat to be reserved for use in patients who have failed or do not tolerate allopurinol, as febuxostat showed an increased risk of heart-related deaths and death from all causes.91 Moreover, there have been no clinical randomized trials on the use of febuxostat in childhood TLS.7 Large-scale studies are needed to define the optimal febuxostat dosage, explore the most appropriate population for its administration, and better define its safety profile.92
Y-700 is another newly synthesized xanthine oxidase inhibitor that showed a more potent and a longer lasting hypouricemic action than allopurinol in hyperuricemic rats.93 In rat models and in healthy adult male volunteers, Y-700 had high oral bioavailability.93,94 Unlike allopurinol, it is hardly excreted in kidneys but primarily eliminated by liver and excreted in feces, making it a safe alternative for patients with renal failure.93,94 However, to the best of our knowledge, there have been no clinical trials on use of Y-700 in TLS yet.
In view of the highly immunogenic potential of traditional urate oxidases, which predisposes patients to risk of anaphylaxis with repeated treatment courses, novel urate oxidases are developed.83 These urate oxidases are covalently conjugated with polyethylene glycol (PEG) to convert them into less antigenic variants with longer half-lives.95,96 Examples of such novel agents include pegloticase, which is derived from a mammalian source, and uricase PEG 20, which originates from Escherichia Coli. However, they are not approved for treatment of TLS currently. Adult clinical trials are underway, and further studies on their efficacy and safety profile in children with TLS are needed.95,96
Correction of electrolytes
Hyperkalemia is the most devastating component of TLS as it can cause sudden death due to cardiac dysrhythmia. Oral or intravenous sources of potassium should be limited or even eliminated in patients at risk of TLS.14 Standard treatment for asymptomatic hyperkalemia include oral or rectal administration of sodium polystyrene sulfonate.3
For symptomatic patients, more aggressive treatment should be used, such as administration of rapid acting insulin and dextrose infusion, which act by increasing intracellular movement of potassium ions from extracellular space.3 A serum potassium level ≥7 mmol/L represents medical emergency and probably warrants renal dialysis.14 In case of significant hyperkalemia leading to electrocardiogram changes, cardiac arrhythmias, cardiac dysfunction, and neurological dysfunction such as seizures, slow infusion of calcium gluconate with cardiac monitoring should be considered to block potassium effects on cardiac cell membranes.1,49 Upon use of intravenous calcium, especially in cases with a calcium–phosphorus product exceeding 70 (serum calcium [mg/dL] × serum phosphorus [mg/dL]), physicians have to watch out for the possibility of formation of calcium–phosphate products, which may lead to acute kidney injury and calcifications of soft tissue.1,49,97–99
Like the management of potassium, oral and intravenous intake of phosphorus should be restricted in patients at risk for TLS. Clinicians have to ensure adequate hydration and may use phosphate binders to reduce intestinal absorption of phosphorus present in meals.100 There are three types of phosphate binders: aluminum-containing (such as aluminum hydroxide), calcium-containing (such as calcium carbonate), and sevelamer.101–103 Calcium-based phosphate binders should not be used in patients with elevated calcium levels in view of risk of calcium phosphate crystallization and organ injury.14,34,104 In refractory cases, dialysis should be the management of choice.3 Intervention is not required in patients with asymptomatic hypocalcemia.1,14,49 For symptomatic hypocalcemia, for example, with cardiac arrhythmia, seizure, or tetany, patients should be treated with slow intravenous calcium gluconate infusion at lowest dose with cardiac monitoring.1,14,49 The aim is to relieve symptoms but not to solely treat the numbers as excessive serum calcium increases calcium phosphate product and crystallization.1,49
Renal replacement therapy
It was demonstrated that there is ongoing risk of acute kidney injury and associated need for renal replacement therapy in patients with TLS.105 Indications for renal replacement therapy in TLS are similar to other patients with other causes of acute kidney injury, such as significant fluid overload, uremia, and severe electrolyte, and metabolic disturbances, yet with lower thresholds.1,49,106 This is particularly true in TLS cases with oliguria because of potentially rapid release and accumulation of electrolytes and metabolites, which could lead to sudden death.1,14,34,49,104 Hyperphosphatemia-induced symptomatic hypocalcemia may also warrant dialysis, in which continuous renal replacement therapies may be the preferred modality as phosphate clearance with dialytic therapy is time dependent.1,107–109 For these reasons, continuous renal replacement therapies such as hemofiltration, hemodialysis, and hemodiafiltration are preferred rather than peritoneal dialysis, as these approaches result in better phosphate and uric acid clearance rates and faster clinical improvement.38,110 There are no major trials showing which approach of hemodialysis is more superior than the other. For most patients, intermittent hemodialysis may be enough. However, in patients with rebound of electrolytes with intermittent hemodialysis, continuous renal replacement therapies may be required.7,107,111 Dialysis should be carried out until there is adequate return of renal function and urine output. Prophylactic continuous renal replacement therapy in children at high risk of TLS was shown to be beneficial in a small study.112 Larger randomized studies will be necessary to provide more evidence for this strategy.
Monitoring
Physicians should stratify patients with malignancies for risk of TLS. As frequent monitoring and assessment is required for patients at high risk of TLS, patients might need to be transferred to centers with intensive care facilities.
Renal function monitoring in terms of urine output is of utmost importance during the management of TLS. Fluid balance should be monitored with urine output as TLS could lead to oliguria by obstructive uropathy with precipitation of calcium-phosphate, xanthine and uric acid products. Moreover, hyperhydration can lead to fluid overload in patients at risk.32,33 Measurement of electrolytes, creatinine, and uric acid should be done up to every 6–8 hours in patients at high risk for TLS after the start of cytotoxic therapy.1,49 During the period at risk for TLS, as well as with specific treatment such as intravenous calcium administration, cardiac monitoring is also necessary for detection of arrhythmias. Monitoring should continue over the entire period at risk.1,49
Prognosis
Many confounding factors may influence the clinical outcomes of patients with TLS, early identification of patients at risk, early initiation of prophylactic measures, and prompt treatment of complications such as electrolyte imbalances and acute renal failure.1,49 Severe hyperkalemia may predispose the patient to cardiac arrhythmia, and severe hypocalcemia may predispose the patient to seizures. While pediatric studies on factors predicting outcomes of TLS are lacking, adult studies have been carried out to look for predictors of mortality in TLS. Predictors for hospital mortality include the presence of acute kidney injury and dialysis requirements, cardiac arrhythmias, and sepsis.113–116 TLS is uncommon in relapsed malignancies as tumor cells are more chemoresistant.33
Summary
Clinicians should be aware of the risk factors for TLS in order to prompt timely management. Prophylactic measures to patients at risk of TLS include aggressive fluid management and judicious use of diuretics and hypouricemic agents. Both allopurinol and urate oxidase are effective in reducing serum uric acid. Allopurinol should be used as prophylaxis for low-risk cases while urate oxidase as treatment for intermediate- to high-risk cases. There is no evidence on better drug of choice among different types of urate oxidases. The routine use of diuretics and urine alkalinization are not recommended. Correction of electrolytes and use of renal replacement therapy may also be required during treatment of TLS.
Paucity of pediatric data on prognosis of TLS as well as lack of pediatric evidence on uricosuric agents precludes the development of specific guidelines on TLS of childhood. Further large-scale pediatric studies on predictors of outcome in TLS will be helpful in identifying children at risk of worse prognosis. Large-scale randomized controlled trials on urate oxidases and novel uricosuric agents in children are also particularly helpful to define effectiveness and safety profiles of various drugs and dosage regimes. Our extensive review confirms that there does not seem to be any randomized trials on novel xanthine oxidase inhibitors and urate oxidase or new information regarding TLS treatment for children.
Acknowledgements
None.
Footnotes
Contributions: All authors contributed equally to the preparation of this review. All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Disclosure and potential conflicts of interest: The authors declare that they have no conflicts of interest relevant to this manuscript. The International Committee of Medical Journal Editors (ICMJE) Potential Conflicts of Interests form for the authors is available for download at https://www.drugsincontext.com/wp-content/uploads/2020/01/dic.2019-8-2-COI.pdf
Funding declaration: There was no funding associated with the preparation of this article.
Correct attribution: Copyright © 2020 Cheung WL, Hon KL, Fung CM, Leung AKC. Published by Drugs in Context under Creative Commons License Deed CC BY NC ND 4.0.
Provenance: invited; externally peer reviewed.
Drugs in Context is published by BioExcel Publishing Ltd. Registered office: Plaza Building, Lee High Road, London, England, SE13 5PT.
BioExcel Publishing Limited is registered in England Number 10038393. VAT GB 252 7720 07.
For all manuscript and submissions enquiries, contact the Editor-in-Chief gordon.mallarkey@bioexcelpublishing.com
For all permissions, rights and reprints, contact David Hughes david.hughes@bioexcelpublishing.com
Peer review comments to author: 30 October 2019
References
- 1.Howard SC, Jones DP, Pui CH. The tumor lysis syndrome. N Engl J Med. 2011;364:1844–54. doi: 10.1056/NEJMra0904569. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Calvo Villas JM. Síndrome de lisis tumoral. Med Clin (Barc) 2019;152:397–404. doi: 10.1016/j.medcli.2018.10.029. [DOI] [PubMed] [Google Scholar]
- 3.Cairo MS, Bishop M. Tumour lysis syndrome: new therapeutic strategies and classification. Br J Haematol. 2004;127:3–11. doi: 10.1111/j.1365-2141.2004.05094.x. [DOI] [PubMed] [Google Scholar]
- 4.Cheson BD, Heitner Enschede S, Cerri E, et al. Tumor lysis syndrome in chronic lymphocytic leukemia with novel targeted agents. Oncologist. 2017;22:1283–1291. doi: 10.1634/theoncologist.2017-0055. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Seegmiller JE, Laster L, Howell RR. Biochemistry of uric acid and its relation to gout. N Engl J Med. 1963;268:821–827. doi: 10.1056/NEJM196304112681507. [DOI] [PubMed] [Google Scholar]
- 6.Hande KR, Garrow GC. Acute tumor lysis syndrome in patients with high-grade non-Hodgkin’s lymphoma. Am J Med. 1993;94:133–139. doi: 10.1016/0002-9343(93)90174-N. [DOI] [PubMed] [Google Scholar]
- 7.Wilson FP, Berns JS. Tumor lysis syndrome: new challenges and recent advances. Adv Chronic Kidney Dis. 2014;21:18–26. doi: 10.1053/j.ackd.2013.07.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Rodríguez-Reimúndes E, Perazzo F, Vilches AR. [Tumor lysis syndrome in a patient with a renal carcinoma treated with sunitinib]. Medicina (B Aires) 2011;71:158–160. http://www.ncbi.nlm.nih.gov/pubmed/21550933. [PubMed] [Google Scholar]
- 9.Hsieh P-M, Hung K-C, Chen Y-S. Tumor lysis syndrome after transarterial chemoembolization of hepatocellular carcinoma: case reports and literature review. World J Gastroenterol. 2009;15(37):4726–4728. doi: 10.3748/wjg.15.4726. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Kobayashi D, Wofford MM, McLean TW, et al. Spontaneous tumor lysis syndrome in a child with T-cell acute lymphoblastic leukemia. Pediatr Blood Cancer. 2010;54:773–775. doi: 10.1002/pbc.22384. [DOI] [PubMed] [Google Scholar]
- 11.Kim SH, Yang EJ, Lim YT, et al. Spontaneous tumor lysis syndrome presenting acute kidney injury with extreme hyperuricemia and urinary stone: a rare case of spontaneous tumor lysis syndrome. Child Kidney Dis. 2017;21:31–34. doi: 10.3339/jkspn.2017.21.1.31. [DOI] [Google Scholar]
- 12.Shenoy MT, D’Souza B, Akshatha LN, et al. Spontaneous tumor lysis syndrome in an infant: a case report. Indian J Clin Biochem. 2015;30:360–362. doi: 10.1007/s12291-014-0472-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Adeyinka A, Bashir K. Tumor Lysis Syndrome. Treasure Island, FL: StatPearls; 2019. http://www.ncbi.nlm.nih.gov/pubmed/30085527. [PubMed] [Google Scholar]
- 14.Coiffier B, Altman A, Pui C-H, et al. Guidelines for the management of pediatric and adult tumor lysis syndrome: an evidence-based review. J Clin Oncol. 2008;26(16):2767–2778. doi: 10.1200/JCO.2007.15.0177. [DOI] [PubMed] [Google Scholar]
- 15.Williams SM, Killeen AA. Tumor lysis syndrome. Arch Pathol Lab Med. 2019;143:386–393. doi: 10.5858/arpa.2017-0278-RS. [DOI] [PubMed] [Google Scholar]
- 16.Cairo MS, Coiffier B, Reiter A, et al. TLS Expert Panel. Recommendations for the evaluation of risk and prophylaxis of tumour lysis syndrome (TLS) in adults and children with malignant diseases: an expert TLS panel consensus. Br J Haematol. 2010;149:578–586. doi: 10.1111/j.1365-2141.2010.08143.x. [DOI] [PubMed] [Google Scholar]
- 17.Bahoush G. Identification of children with acute lymphoblastic leukemia at low risk for tumor lysis syndrome. IJCA. 2015;1(2):29–33. http://ijca.iums.ac.ir/article-1-108-en.html. [Google Scholar]
- 18.Sevinir B, Demirkaya M, Baytan B, et al. Hyperuricemia and tumor lysis syndrome in children with non-Hodgkin’s lymphoma and acute lymphoblastic leukemia. Turk J Haematol. 2011;28(1):52–9. doi: 10.5152/tjh.2011.06. [DOI] [PubMed] [Google Scholar]
- 19.Wössmann W, Schrappe M, Meyer U, et al. Incidence of tumor lysis syndrome in children with advanced stage Burkitt’s lymphoma/leukemia before and after introduction of prophylactic use of urate oxidase. Ann Hematol. 2003;82:160–165. doi: 10.1007/s00277-003-0608-2. [DOI] [PubMed] [Google Scholar]
- 20.AlBagshi M, Hassan ES, Sadek AO, et al. Tumor lysis syndrome in children with acute leukemia: incidence and outcome. J Appl Hematol. 2013;4:100. doi: 10.4103/1658-5127.123304. [DOI] [Google Scholar]
- 21.Abdel-Baset HA, Eldin EN, Eltayeb AA, et al. Clinical and laboratory approach for the identification of the risk for tumour lysis syndrome in children with acute lymphoblastic leukemia. Life Sci J. 2012;9 doi: 10.7537/marslsj090112.27. [DOI] [Google Scholar]
- 22.Outcome of tumor lysis syndrome in pediatric patients with hematologic malignancies – a single-center experience from Pakistan. J Community Support Oncol. 2016 doi: 10.12788/jcso.0300. [DOI] [Google Scholar]
- 23.Alavi S, Arzanian MT, Abbasian MR, et al. Tumor lysis syndrome in children with non-Hodgkin lymphoma. Pediatr Hematol Oncol. 2006;23(1):65–70. doi: 10.1080/08880010500313561. [DOI] [PubMed] [Google Scholar]
- 24.Nakamura M, Oda S, Sadahiro T, et al. The role of hypercytokinemia in the pathophysiology of tumor lysis syndrome (TLS) and the treatment with continuous hemodiafiltration using a polymethylmethacrylate membrane hemofilter (PMMA-CHDF) Transfus Apher Sci. 2009;40:41–47. doi: 10.1016/j.transci.2008.11.004. [DOI] [PubMed] [Google Scholar]
- 25.Howard SC. Tumor lysis syndrome. Abeloff’s Clin Oncol. 2014:591–596. doi: 10.1016/B978-1-4557-2865-7.00038-2. [DOI] [Google Scholar]
- 26.Jasek AM, Day HJ. Acute spontaneous tumor lysis syndrome. Am J Hematol. 1994;47(2):129–131. doi: 10.1002/ajh.2830470212. [DOI] [PubMed] [Google Scholar]
- 27.Gangireddy M, Shrimanker I, Nookala VK, et al. Spontaneous tumor lysis syndrome in diffuse large B-cell lymphoma: early diagnosis and management. Cureus. 2019;11:e4679. doi: 10.7759/cureus.4679. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Flombaum CD. Metabolic emergencies in the cancer patient. Semin Oncol. 2000;27:322–334. http://www.ncbi.nlm.nih.gov/pubmed/10864220. [PubMed] [Google Scholar]
- 29.Locatelli F, Rossi F. Incidence and pathogenesis of tumor lysis syndrome. In: Ronco C, Rodeghiero, editors. Hyperuricemic Syndromes: Pathophysiology and Therapy. Basel: KARGER; 2004. pp. 61–68. [DOI] [Google Scholar]
- 30.Cunningham SG. Fluid and electrolyte disturbances associated with cancer and its treatment. Nurs Clin North Am. 1982;17:579–593. http://www.ncbi.nlm.nih.gov/pubmed/6757872. [PubMed] [Google Scholar]
- 31.Kelly KM, Lange B. Oncologic emergencies. Pediatr Clin North Am. 1997;44:809–830. doi: 10.1016/s0031-3955(05)70531-9. [DOI] [PubMed] [Google Scholar]
- 32.Cohen EP, Krzesinski J-M, Launay-Vacher V, et al. Onco-nephrology: core curriculum 2015. Am J Kidney Dis. 2015;66(5):869–883. doi: 10.1053/j.ajkd.2015.04.042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wilson FP, Berns JS. Onco-nephrology: tumor lysis syndrome. Clin J Am Soc Nephrol. 2012;7(10):1730–1739. doi: 10.2215/CJN.03150312. [DOI] [PubMed] [Google Scholar]
- 34.Cheson BD, Frame JN, Vena D, et al. Tumor lysis syndrome: an uncommon complication of fludarabine therapy of chronic lymphocytic leukemia. J Clin Oncol. 1998;16:2313–2320. doi: 10.1200/JCO.1998.16.7.2313. [DOI] [PubMed] [Google Scholar]
- 35.Kabisch H, Niggemann B, Winkler K. Extreme Hyperphosphatämie mit Hypokalzämie im Rahmen eines Zellyse-Syndromes bei einem Kind mit T-ALL. Oncol Res Treat. 1989;12:64–68. doi: 10.1159/000216604. [DOI] [PubMed] [Google Scholar]
- 36.Aubert JD, Claeys M, Zwahlen A, et al. [Hyperphosphatemia and transient renal insufficiency following chemotherapy of acute lymphoblastic leukemia]. Schweiz Med Wochenschr. 1988;118:1953–1956. http://www.ncbi.nlm.nih.gov/pubmed/3212426. [PubMed] [Google Scholar]
- 37.Boles JM, Dutel JL, Briere J, et al. Acute renal failure caused by extreme hyperphosphatemia after chemotherapy of an acute lymphoblastic leukemia. Cancer. 1984;53:2425–2429. doi: 10.1002/1097-0142(19840601)53:11<2425::aid-cncr2820531111>3.0.co;2-r. [DOI] [PubMed] [Google Scholar]
- 38.Kjellstrand CM, Cambell DC, vonHartitzsch B, et al. Hyperuricemic acute renal failure. Arch Intern Med. 1974;133:349–359. doi: 10.1001/archinte.1974.00320150023002. [DOI] [PubMed] [Google Scholar]
- 39.Roberts KE. Pediatric fluid and electrolyte balance: critical care case studies. Crit Care Nurs Clin North Am. 2005;17:361–373. doi: 10.1016/j.ccell.2005.07.006. [DOI] [PubMed] [Google Scholar]
- 40.Kanfer A, Richet G, Roland J, Chatelet F. Extreme hyperphosphataemia causing acute anuric nephrocalcinosis in lymphosarcoma. BMJ. 1979;1(6174):1320–1321. doi: 10.1136/bmj.1.6174.1320-a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.van denBerg H, Reintsema AM. Renal tubular damage in rasburicase: risks of alkalinisation. Ann Oncol. 2004;15(1):175–176. doi: 10.1093/annonc/mdh029. [DOI] [PubMed] [Google Scholar]
- 42.Seegmiller JE. Xanthine stone formation. Am J Med. 1968;45(5):780–783. doi: 10.1016/0002-9343(68)90210-6. [DOI] [PubMed] [Google Scholar]
- 43.LaRosa C, McMullen L, Bakdash S, et al. Acute renal failure from xanthine nephropathy during management of acute leukemia. Pediatr Nephrol. 2007;22:132–135. doi: 10.1007/s00467-006-0287-z. [DOI] [PubMed] [Google Scholar]
- 44.Pais VM, Lowe G, Lallas CD, et al. Xanthine urolithiasis. Urology. 2006;67(5):1084.e9–1084.e11. doi: 10.1016/j.urology.2005.10.057. [DOI] [PubMed] [Google Scholar]
- 45.Veenstra J, Krediet RT, Somers R, et al. Tumour lysis syndrome and acute renal failure in Burkitt’s lymphoma. Description of 2 cases and a review of the literature on prevention and management. Neth J Med. 1994;45:211–216. http://www.ncbi.nlm.nih.gov/pubmed/7830847. [PubMed] [Google Scholar]
- 46.Band PR, Silverberg DS, Henderson JF, et al. Xanthine nephropathy in a patient with lymphosarcoma treated with allopurinol. N Engl J Med. 1970;283(7):354–357. doi: 10.1056/NEJM197008132830708. [DOI] [PubMed] [Google Scholar]
- 47.DeConti RC, Calabresi P. Use of allopurinol for prevention and control of hyperuricemia in patients with neoplastic disease. N Engl J Med. 1966;274(9):481–486. doi: 10.1056/NEJM196603032740902. [DOI] [PubMed] [Google Scholar]
- 48.Hande KR, Hixson CV, Chabner BA. Postchemotherapy purine excretion in lymphoma patients receiving allopurinol. Cancer Res. 1981;41:2273–2279. http://www.ncbi.nlm.nih.gov/pubmed/7237428. [PubMed] [Google Scholar]
- 49.Abu-Alfa AK, Younes A. Tumor lysis syndrome and acute kidney injury: evaluation, prevention, and management. Am J Kidney Dis. 2010;55(5):S1–S13. doi: 10.1053/j.ajkd.2009.10.056. [DOI] [PubMed] [Google Scholar]
- 50.Hijiya N, Metzger ML, Pounds S, et al. Severe cardiopulmonary complications consistent with systemic inflammatory response syndrome caused by leukemia cell lysis in childhood acute myelomonocytic or monocytic leukemia. Pediatr Blood Cancer. 2005;44(1):63–69. doi: 10.1002/pbc.20192. [DOI] [PubMed] [Google Scholar]
- 51.Truong TH, Beyene J, Hitzler J, et al. Features at presentation predict children with acute lymphoblastic leukemia at low risk for tumor lysis syndrome. Cancer. 2007;110:1832–1839. doi: 10.1002/cncr.22990. [DOI] [PubMed] [Google Scholar]
- 52.Kopecna L, Dolezel Z, Osvaldova Z, et al. The analysis of the risks for the development of tumour lysis syndrome in children. Bratisl Lek Listy. 2002;103:206–209. http://www.ncbi.nlm.nih.gov/pubmed/12448566. [PubMed] [Google Scholar]
- 53.Seidemann K, Meyer U, Jansen P, et al. Impaired renal function and tumor lysis syndrome in pediatric patients with non-Hodgkin’s lymphoma and B-ALL - Observations from the BFM-trials. Klin Padiatr. 1998;210:279–284. doi: 10.1055/s-2008-1043892. [DOI] [PubMed] [Google Scholar]
- 54.Weeks AC, Kimple ME. Spontaneous tumor lysis syndrome: a case report and critical evaluation of current diagnostic criteria and optimal treatment regimens. J Investig Med High Impact Case Reports. 2015;3(3):2324709615603199. doi: 10.1177/2324709615603199. https://doi.org/10.1177%2F2324709615603199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Coiffier B, Riouffol C. Management of tumor lysis syndrome in adults. Expert Rev Anticancer Ther. 2007;7:233–239. doi: 10.1586/14737140.7.2.233. [DOI] [PubMed] [Google Scholar]
- 56.D’Alessandro V, Greco A, Clemente C, et al. Severe spontaneous acute tumor lysis syndrome and hypoglycemia in patient with germ cell tumor. Tumori. 2010;96:1040–1043. doi: 10.1177/548.6530. [DOI] [PubMed] [Google Scholar]
- 57.Jones DP, Mahmoud H, Chesney RW. Tumor lysis syndrome: pathogenesis and management. Pediatr Nephrol. 1995;9:206–212. doi: 10.1007/bf00860751. [DOI] [PubMed] [Google Scholar]
- 58.Jones GL, Will A, Jackson GH, et al. British Committee for Standards in Haematology. Guidelines for the management of tumour lysis syndrome in adults and children with haematological malignancies on behalf of the British Committee for Standards in Haematology. Br J Haematol. 2015;169:661–671. doi: 10.1111/bjh.13403. [DOI] [PubMed] [Google Scholar]
- 59.Krakoffih M. Prevention of hyperuricemia in leukemia and lymphoma: use of allopurinol, a xanthine oxidase inhibitor. JAMA. 1965;193:1–6. doi: 10.1001/jama.1965.03090010007001. [DOI] [PubMed] [Google Scholar]
- 60.Mahmoud HH, Leverger G, Patte C, et al. Advances in the management of malignancy-associated hyperuricaemia. Br J Cancer. 1998;(S4):18–20. doi: 10.1038/bjc.1998.432. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Smalley RV, Guaspari A, Haase-Statz S, et al. Allopurinol: intravenous use for prevention and treatment of hyperuricemia. J Clin Oncol. 2000;18:1758–1763. doi: 10.1200/JCO.2000.18.8.1758. [DOI] [PubMed] [Google Scholar]
- 62.Brogard JM, Coumaros D, Franckhauser J, et al. Enzymatic uricolysis: a study of the effect of a fungal urate-oxydase. Rev Eur Etud Clin Biol. 1972;17:890–895. http://www.ncbi.nlm.nih.gov/pubmed/4631795. [PubMed] [Google Scholar]
- 63.Thaci D, Simpson EL, Beck LA, et al. Efficacy and safety of dupilumab in adults with moderate-to-severe atopic dermatitis inadequately controlled by topical treatments: a randomised, placebo-controlled, dose-ranging phase 2b trial. Lancet. 2016;387(10013):40–52. doi: 10.1016/S0140-6736(15)00388-8. [DOI] [PubMed] [Google Scholar]
- 64.Navolanic PM, Pui C-H, Larson RA, et al. Elitek-rasburicase: an effective means to prevent and treat hyperuricemia associated with tumor lysis syndrome, a Meeting Report, Dallas, Texas, January 2002. Leukemia. 2003;17(3):499–514. doi: 10.1038/sj.leu.2402847. [DOI] [PubMed] [Google Scholar]
- 65.McDonnell AM, Lenz KL, Frei-Lahr DA, et al. Single-dose rasburicase 6 mg in the management of tumor lysis syndrome in adults. Pharmacotherapy. 2006;26(6):806–812. doi: 10.1592/phco.26.6.806. [DOI] [PubMed] [Google Scholar]
- 66.Cheuk DK, Chiang AK, Chan GC, et al. Urate oxidase for the prevention and treatment of tumour lysis syndrome in children with cancer. Cochrane Database Syst Rev. 2017;3:CD006945. doi: 10.1002/14651858.CD006945.pub4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Pui CH. Rasburicase: a potent uricolytic agent. Expert Opin Pharmacother. 2002;3:433–442. doi: 10.1517/14656566.3.4.433. [DOI] [PubMed] [Google Scholar]
- 68.Whelton A. Current and future therapeutic options for the management of gout. Am J Ther. 2010;17:402–417. doi: 10.1097/MJT.0b013e3181df8ad2. [DOI] [PubMed] [Google Scholar]
- 69.Laboureur P, Langlois C. [Urate oxidase of Aspergillus flavus. I. Isolation, purification, properties]. Bull Soc Chim Biol (Paris) 1968;50:811–825. http://www.ncbi.nlm.nih.gov/pubmed/5671592. [PubMed] [Google Scholar]
- 70.Shaikh SA, Marini BL, Hough SM, et al. Rational use of rasburicase for the treatment and management of tumor lysis syndrome. J Oncol Pharm Pract. 2018;24:176–184. doi: 10.1177/1078155216687152. [DOI] [PubMed] [Google Scholar]
- 71.Bosly A, Sonet A, Pinkerton CR. Rasburicase (recombinant urate oxidase) for the management of hyperuricemia in patients with cancer: report of an international compassionate use study. Cancer. 2003;98(5):1048–1054. doi: 10.1002/cncr.11612. [DOI] [PubMed] [Google Scholar]
- 72.Yim BT, Sims-McCallum RP, Chong PH. Rasburicase for the treatment and prevention of hyperuricemia. Ann Pharmacother. 2003;37(7–8):1047–1054. doi: 10.1345/aph.1C336. https://doi.org/10.1345%2Faph.1C336. [DOI] [PubMed] [Google Scholar]
- 73.Coiffier B, Mounier N, Bologna S, et al. Efficacy and safety of rasburicase (recombinant urate oxidase) for the prevention and treatment of hyperuricemia during induction chemotherapy of aggressive non-Hodgkin’s lymphoma: results of the GRAAL1 (Groupe d’Etude des Lymphomes de l’Adulte Trial on Rasburicase Activity in Adult Lymphoma) study. J Clin Oncol. 2003;21:4402–4406. doi: 10.1200/JCO.2003.04.115. [DOI] [PubMed] [Google Scholar]
- 74.Goldman SC. Rasburicase: potential role in managing tumor lysis in patients with hematological malignancies. Expert Rev Anticancer Ther. 2003;3(4):429–433. doi: 10.1586/14737140.3.4.429. [DOI] [PubMed] [Google Scholar]
- 75.Jeha S, Pui C-H. Recombinant urate oxidase (rasburicase) in the prophylaxis and treatment of tumor lysis syndrome. Contrib Nephrol. 2005;147:69–79. doi: 10.1159/000082545. [DOI] [PubMed] [Google Scholar]
- 76.Pession A, Barbieri E. Treatment and prevention of tumor lysis syndrome in children. Experience of Associazione Italiana Ematologia Oncologia Pediatrica. Contrib Nephrol. 2005;147:80–92. doi: 10.1159/000082546. [DOI] [PubMed] [Google Scholar]
- 77.Jeha S, Kantarjian H, Irwin D, et al. Efficacy and safety of rasburicase, a recombinant urate oxidase (Elitek), in the management of malignancy-associated hyperuricemia in pediatric and adult patients: final results of a multicenter compassionate use trial. Leukemia. 2005;19:34–38. doi: 10.1038/sj.leu.2403566. [DOI] [PubMed] [Google Scholar]
- 78.Pui CH, Jeha S, Irwin D, et al. Recombinant urate oxidase (rasburicase) in the prevention and treatment of malignancy-associated hyperuricemia in pediatric and adult patients: results of a compassionate-use trial. Leukemia. 2001;15:1505–1509. doi: 10.1038/sj.leu.2402235. [DOI] [PubMed] [Google Scholar]
- 79.Goldman SC, Holcenberg JS, Finklestein JZ, et al. A randomized comparison between rasburicase and allopurinol in children with lymphoma or leukemia at high risk for tumor lysis. Blood. 2001;97(10):2998–3003. doi: 10.1182/blood.v97.10.2998. [DOI] [PubMed] [Google Scholar]
- 80.Pui C-H, Mahmoud HH, Wiley JM, et al. Recombinant urate oxidase for the prophylaxis or treatment of hyperuricemia in patients with leukemia or lymphoma. J Clin Oncol. 2001;19(3):697–704. doi: 10.1200/JCO.2001.19.3.697. [DOI] [PubMed] [Google Scholar]
- 81.Savva DA, Herrera N, Rohatgi R. Comparison of fixed versus traditional weight-based dosing of rasburicase in a pediatric population. Pediatr Blood Cancer. 2018;65:e27236. doi: 10.1002/pbc.27236. [DOI] [PubMed] [Google Scholar]
- 82.Ibrahim U, Saqib A, Mohammad F, et al. Rasburicase-induced methemoglobinemia: the eyes do not see what the mind does not know. J Oncol Pharm Pract. 2018;24:309–313. doi: 10.1177/1078155217701295. [DOI] [PubMed] [Google Scholar]
- 83.Allen KC, Champlain AH, Cotliar JA, et al. Risk of anaphylaxis with repeated courses of rasburicase: a Research on Adverse Drug Events and Reports (RADAR) project. Drug Saf. 2015;38(2):183–187. doi: 10.1007/s40264-014-0255-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Bessmertny O, Robitaille LM, Cairo MS. Rasburicase: a new approach for preventing and/or treating tumor lysis syndrome. Curr Pharm Des. 2005;11:4177–4185. doi: 10.2174/138161205774913291. [DOI] [PubMed] [Google Scholar]
- 85.Alhalabi O, Yadav S, Jaiyesimi IA. Prescribing patterns of rasburicase and glucose-6-phosphate-dehydrogenase (G6PD) testing in a large community health system. J Clin Oncol. 2016;34(15_suppl):e18206–e18206. doi: 10.1200/JCO.2016.34.15_suppl.e18206. [DOI] [Google Scholar]
- 86.Nguyen AP, Ness GL. Hemolytic anemia following rasburicase administration: a review of published reports. J Pediatr Pharmacol Ther. 19(4):310–316. doi: 10.5863/1551-6776-19.4.310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Akande M, Audino AN, Tobias JD. Rasburicase-induced hemolytic anemia in an adolescent with unknown glucose-6-phosphate dehydrogenase deficiency. J Pediatr Pharmacol Ther. 2017;22:471–475. doi: 10.5863/1551-6776-22.6.471. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Okamoto K, Eger BT, Nishino T, et al. An extremely potent inhibitor of xanthine oxidoreductase. Crystal structure of the enzyme-inhibitor complex and mechanism of inhibition. J Biol Chem. 2003;278:1848–1855. doi: 10.1074/jbc.M208307200. [DOI] [PubMed] [Google Scholar]
- 89.Takano Y, Hase-Aoki K, Horiuchi H, et al. Selectivity of febuxostat, a novel non-purine inhibitor of xanthine oxidase/xanthine dehydrogenase. Life Sci. 2005;76:1835–1847. doi: 10.1016/j.lfs.2004.10.031. [DOI] [PubMed] [Google Scholar]
- 90.Spina M, Nagy Z, Ribera JM, et al. FLORENCE: a randomized, double-blind, phase III pivotal study of febuxostat versus allopurinol for the prevention of tumor lysis syndrome (TLS) in patients with hematologic malignancies at intermediate to high TLS risk. Ann Oncol. 2015;26:2155–2161. doi: 10.1093/annonc/mdv317. [DOI] [PubMed] [Google Scholar]
- 91.White WB, Saag KG, Becker MA, et al. Cardiovascular safety of febuxostat or allopurinol in patients with gout. N Engl J Med. 2018;378:1200–1210. doi: 10.1056/NEJMoa1710895. [DOI] [PubMed] [Google Scholar]
- 92.Bellos I, Kontzoglou K, Psyrri A, et al. Febuxostat administration for the prevention of tumour lysis syndrome: a meta-analysis. J Clin Pharm Ther. 2019;44:525–533. doi: 10.1111/jcpt.12839. [DOI] [PubMed] [Google Scholar]
- 93.Yamada I, Fukunari A, Osajima T, et al. Pharmacokinetics/pharmacodynamics of Y-700, a novel xanthine oxidase inhibitor, in rats and man. Nucleosides Nucleotides Nucleic Acids. 2004;23(8–9):1123–1125. doi: 10.1081/NCN-200027384. [DOI] [PubMed] [Google Scholar]
- 94.Fukunari A, Okamoto K, Nishino T, et al. Y-700 [1-[3-Cyano-4-(2,2-dimethylpropoxy)phenyl]-1H-pyrazole-4-carboxylic acid]: a potent xanthine oxidoreductase inhibitor with hepatic excretion. J Pharmacol Exp Ther. 2004;311:519–528. doi: 10.1124/jpet.104.070433. [DOI] [PubMed] [Google Scholar]
- 95.Zhang Z, Fiorino A. Pharmacokinetics of intravenous uricase-PEG 20, a drug for managing hyperuricemia in tumor lysis syndrome. Blood. 2009;114(22):3086. doi: 10.1182/blood.V114.22.3086.3086. [DOI] [Google Scholar]
- 96.Tiu RV, Mountantonakis SE, Dunbar AJ, et al. Tumor lysis syndrome. Semin Thromb Hemost. 2007;33:397–407. doi: 10.1055/s-2007-976175. [DOI] [PubMed] [Google Scholar]
- 97.O’Neill WC. The fallacy of the calcium-phosphorus product. Kidney Int. 2007;72(7):792–796. doi: 10.1038/sj.ki.5002412. [DOI] [PubMed] [Google Scholar]
- 98.Nomdedéu J, Martino R, Sureda A, et al. Acute tumor lysis syndrome complicating conditioning therapy for bone marrow transplantation in a patient with chronic lymphocytic leukemia. Bone Marrow Transplant. 1994;13:659–660. http://www.ncbi.nlm.nih.gov/pubmed/8054919. [PubMed] [Google Scholar]
- 99.Boccia RV, Longo DL, Lieber ML, et al. Multiple recurrences of acute tumor lysis syndrome in an indolent non-hodgkin’s lymphoma. Cancer. 1985;56(9):2295–2297. doi: 10.1002/1097-0142(19851101)56:9<2295::AID-CNCR2820560926>3.0.CO;2-X. [DOI] [PubMed] [Google Scholar]
- 100.Malberti F. Hyperphosphataemia: treatment Options. Drugs. 2013;73:673–688. doi: 10.1007/s40265-013-0054-y. [DOI] [PubMed] [Google Scholar]
- 101.St Peter WL, Wazny LD, Weinhandl E, et al. A review of phosphate binders in chronic kidney disease: incremental progress or just higher costs? Drugs. 2017;77:1155–1186. doi: 10.1007/s40265-017-0758-5. [DOI] [PubMed] [Google Scholar]
- 102.Floege J. Phosphate binders in chronic kidney disease: a systematic review of recent data. J Nephrol. 2016;29(3):329–340. doi: 10.1007/s40620-016-0266-9. [DOI] [PubMed] [Google Scholar]
- 103.Marcuccilli M, Chonchol M, Jovanovich A. Phosphate binders and targets over decades: do we have it right now? Semin Dial. 2017;30(2):134–141. doi: 10.1111/sdi.12568. [DOI] [PubMed] [Google Scholar]
- 104.Mirrakhimov AE, Voore P, Khan M, et al. Tumor lysis syndrome: a clinical review. World J Crit Care Med. 2015;4:130. doi: 10.5492/wjccm.v4.i2.130. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Brochard L, Abroug F, Brenner M, et al. An official ATS/ERS/ESICM/SCCM/SRLF statement: prevention and management of acute renal failure in the ICU patient: an international consensus conference in intensive care medicine. Am J Respir Crit Care Med. 2010;181:1128–1155. doi: 10.1164/rccm.200711-1664ST. [DOI] [PubMed] [Google Scholar]
- 106.Fleming GM, Walters S, Goldstein SL, et al. Nonrenal indications for continuous renal replacement therapy: a report from the Prospective Pediatric Continuous Renal Replacement Therapy Registry Group. Pediatr Crit Care Med. 2012;13:e299–e304. doi: 10.1097/PCC.0b013e31824fbd76. [DOI] [PubMed] [Google Scholar]
- 107.Sakarcan A, Quigley R. Hyperphosphatemia in tumor lysis syndrome: the role of hemodialysis and continuous veno-venous hemofiltration. Pediatr Nephrol. 1994;8:351–353. doi: 10.1007/bf00866363. [DOI] [PubMed] [Google Scholar]
- 108.Pichette V, Leblanc M, Bonnardeaux A, et al. High dialysate flow rate continuous arteriovenous hemodialysis: a new approach for the treatment of acute renal failure and tumor lysis syndrome. Am J Kidney Dis. 1994;23(4):591–596. doi: 10.1016/S0272-6386(12)80384-2. [DOI] [PubMed] [Google Scholar]
- 109.Heney D, Essex-Cater A, Brocklebank JT, et al. Continuous arteriovenous haemofiltration in the treatment of tumour lysis syndrome. Pediatr Nephrol. 1990;4:245–247. doi: 10.1007/bf00857665. [DOI] [PubMed] [Google Scholar]
- 110.Deger GE, Wagoner RD. Peritoneal dialysis in acute uric acid nephropathy. Mayo Clin Proc. 1972;47:189–192. http://www.ncbi.nlm.nih.gov/pubmed/4501569. [PubMed] [Google Scholar]
- 111.Agha-Razii M, Amyot SL, Pichette V, et al. Continuous veno-venous hemodiafiltration for the treatment of spontaneous tumor lysis syndrome complicated by acute renal failure and severe hyperuricemia. Clin Nephrol. 2000;54:59–63. http://www.ncbi.nlm.nih.gov/pubmed/10939758. [PubMed] [Google Scholar]
- 112.Saccente SL, Kohaut EC, Berkow RL. Prevention of tumor lysis syndrome using continuous veno-venous hemofiltration. Pediatr Nephrol. 1995;9:569–573. doi: 10.1007/bf00860936. [DOI] [PubMed] [Google Scholar]
- 113.Darmon M, Guichard I, Vincent F, et al. Prognostic significance of acute renal injury in acute tumor lysis syndrome. 2010:51. doi: 10.3109/10428190903456959. [DOI] [PubMed] [Google Scholar]
- 114.Durani U, Shah ND, Go RS. In-hospital outcomes of tumor lysis syndrome: a population-based study using the National Inpatient Sample. Oncologist. 2017;22:1506–1509. doi: 10.1634/theoncologist.2017-0147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.Pathak R, Giri S, Aryal MR, et al. Predictors of in-hospital mortality in tumor lysis syndrome. Blood. 2014;124 doi: 10.1182/blood.V124.21.4862.4862. [DOI] [Google Scholar]
- 116.Darmon M, Vincent F, Camous L, et al. Tumour lysis syndrome and acute kidney injury in high-risk haematology patients in the rasburicase era. A prospective multicentre study from the Groupe de Recherche en Réanimation Respiratoire et Onco-Hématologique. Br J Haematol. 2013;162:489–497. doi: 10.1111/bjh.12415. [DOI] [PubMed] [Google Scholar]
- 117.Howard SC, Trifilio S, Gregory TK, et al. Tumor lysis syndrome in the era of novel and targeted agents in patients with hematologic malignancies: a systematic review. Ann Hematol. 2016;95:563–573. doi: 10.1007/s00277-015-2585-7. [DOI] [PubMed] [Google Scholar]
- 118.Montesinos P, Lorenzo I, Martin G, et al. Tumor lysis syndrome in patients with acute myeloid leukemia: identification of risk factors and development of a predictive model. Haematologica. 2008;93:67–74. doi: 10.3324/haematol.11575. [DOI] [PubMed] [Google Scholar]
- 119.Baeksgaard L, Sørensen JB. Acute tumor lysis syndrome in solid tumors--a case report and review of the literature. Cancer Chemother Pharmacol. 2003;51:187–192. doi: 10.1007/s00280-002-0556-x. [DOI] [PubMed] [Google Scholar]
- 120.Yang H, Rosove MH, Figlin RA. Tumor lysis syndrome occurring after the administration of rituximab in lymphoproliferative disorders: high-grade non-Hodgkin’s lymphoma and chronic lymphocytic leukemia. Am J Hematol. 1999;62:247–250. doi: 10.1002/(SICI)1096-8652(199912)62:4<247::AID-AJH9>3.0.CO;2-T. [DOI] [PubMed] [Google Scholar]
- 121.Jensen M, Winkler U, Manzke O, et al. Rapid tumor lysis in a patient with B-cell chronic lymphocytic leukemia and lymphocytosis treated with an anti-CD20 monoclonal antibody (IDEC-C2B8, rituximab) Ann Hematol. 2019;77(1–2):89–91. doi: 10.1007/s002770050419. [DOI] [PubMed] [Google Scholar]
- 122.Jabr FI. Acute tumor lysis syndrome induced by rituximab in diffuse large B-cell lymphoma. Int J Hematol. 2005;82:312–314. doi: 10.1532/IJH97.NA0504. [DOI] [PubMed] [Google Scholar]
- 123.Hsu H-H, Huang C-C. Acute spontaneous tumor lysis in anaplastic large T-cell lymphoma presenting with hyperuricemic acute renal failure. Int J Hematol. 2004;79:48–51. doi: 10.1007/bf02983533. [DOI] [PubMed] [Google Scholar]