Abstract
STUDY QUESTION
Is it feasible to perform uterus transplantations (UTx) in a tertiary centre in the Netherlands?
SUMMARY ANSWER
Considering all ethical principles, surgical risks and financial aspects, we have concluded that at this time, it is not feasible to establish the UTx procedure at our hospital.
WHAT IS KNOWN ALREADY
UTx is a promising treatment for absolute uterine factor infertility. It is currently being investigated within several clinical trials worldwide and has resulted in the live birth of 19 children so far. Most UTx procedures are performed in women with the Mayer–Rokitansky–Küster–Hauser (MRKH) syndrome, a congenital disorder characterized by absence of the uterus. In the Netherlands, the only possible option for these women for having children is adoption or surrogacy.
STUDY DESIGN, SIZE, DURATION
We performed a feasibility study to search for ethical, medical and financial support for performing UTx at the Amsterdam UMC, location VUmc.
PARTICIPANTS/MATERIALS, SETTING, METHODS
For this feasibility study, we created a special interest group, including gynaecologists, transplant surgeons, researchers and a financial advisor. Also, in collaboration with the patients’ association for women with MRKH, a questionnaire study was performed to research the decision-making in possible recipients. In this paper, we present an overview of current practices and literature on UTx and discuss the results of our feasibility study.
MAIN RESULTS AND THE ROLE OF CHANCE
A high level of interest from the possible recipients became apparent from our questionnaire amongst women with MRKH. The majority (64.8%) positively considered UTx with a live donor, with 69.6% having a potential donor available. However, this ‘non-life-saving transplantation’ requires careful balancing of risks and benefits. The UTx procedure includes two complex surgeries and unknown consequences for the unborn child. The costs for one UTx are calculated to be around €100 000 and will not be compensated by medical insurance. The Clinical Ethics Committee places great emphasis on the principle of non-maleficence and the ‘fair distribution of health services’.
LIMITATIONS, REASONS FOR CAUTION
In the Netherlands, alternatives for having children are available and future collaboration with experienced foreign clinics that offer the procedure is a possibility not yet investigated.
WIDER IMPLICATIONS OF THE FINDINGS
The final assessment of this feasibility study is that that there are not enough grounds to support this procedure at our hospital at this point in time. We will closely follow the developments and will re-evaluate the feasibility in the future.
STUDY FUNDING/COMPETING INTEREST(S)
This feasibility study was funded by the VU Medical Center (Innovation grant 2017). No conflicts of interest have been reported relevant to the subject of all authors.
TRIAL REGISTRATION NUMBER
n.a
Keywords: absolute uterine factor infertility, medical ethics, feasibility study, MRKH syndrome, uterus transplantation
WHAT DOES THIS MEAN FOR PATIENTS?
In the Netherlands, women without a womb but with healthy ovaries can only have their own biological children through gestational surrogacy. This involves another woman who becomes pregnant using their eggs and gives birth to the child. A possible new option is a womb transplantation. A womb transplantation means that a womb is surgically removed, either from someone who is alive who chooses to do this—for example a mother or a friend—or from someone who has recently died. The womb is then placed into a woman with no womb. There are very strict medical checks that mean only a small number of women might be able to go through this procedure.
Sweden started with experimental womb transplantations in mice more than 20 years ago and developed this technique into a clinical procedure with successful womb transplantations. The first baby was born as a result of a womb transplant in 2014. Since then, this groundbreaking surgical procedure has been performed worldwide and has resulted in 19 children born from transplanted wombs. In the Netherlands, this surgery has not been carried out yet. We wanted to know how Dutch women feel about this procedure. We asked women who were born without a womb to fill out a questionnaire. Most of these women indicated that they would consider a womb transplantation if this technique becomes available in the Netherlands.
With a large team of professionals, we looked at whether it is possible to perform this procedure in our hospital. The surgery is complex and has risks for both the women who have the transplant and the donors. Furthermore, there are very high costs attached to this procedure, calculated to be around €100 000 that would not be covered by medical insurance. We have carefully balanced the risks and benefits, in the light of ethical considerations. We have decided that it is not possible to perform womb transplantations at our hospital, at the present time. We will closely follow the developments and improvements made in other countries and continue to look at the possibility of womb transplantations in our hospital in the future.
Introduction
With uterus transplantation (UTx), the uterine graft of a donor is removed and transplanted in a woman who has no uterus due to congenital or surgical reasons. The incentive for this procedure is a desire to have children in women with ovaries but no uterus. The first human UTx was performed in Saudi Arabia in 2000, but was rejected within 3 months (Fageeh et al., 2002). In 2014, the first child after a successful UTx was born in Sweden (Brannstrom et al., 2015) after years of “”animal research with successful transplantations in mice, rats, sheep and baboons (Racho El-Akouri et al., 2002; Dahm-Kähler et al., 2008; Wranning et al., 2008a; Wranning et al., 2008b; Enskog et al., 2010; Wranning et al., 2010). The UTx procedure in humans is currently being investigated by several clinical trials worldwide. At present, around 60 human UTx have been performed, resulting in the birth of 19 children (Brannstrom 2017; Flyckt et al., 2017; Ejzenberg et al., 2018; Testa et al., 2018; Andrew & Ahmed, 2019; Chmel et al., 2019; Jones et al., 2019; Kisu et al., 2019). In the Netherlands, no uterus transplantations have been performed so far.
Most UTx procedures are performed in women with Mayer–Rokitansky–Küster–Hauser (MRKH) syndrome. These women are born without a uterus and upper part of the vagina. They have normal-functioning ovaries and normal secondary sexual characteristics. MRKH occurs in 1:5000 women (Herlin et al., 2016). Based on Dutch population statistics, around 1500 women in the Netherlands are affected, with 20 new cases of MRKH each year.
Due to absence of the uterus, these women have absolute uterine factor infertility. In the Netherlands, possible options for having children are adoption, and traditional and gestational surrogacy. Gestational surrogacy means an in vitro fertilization (IVF) procedure is performed and the resulting embryo is placed in a gestational carrier. Surrogacy, in general, is a controversial treatment and is not allowed by law in various countries in Europe, for example in Sweden. The only hospital in the Netherlands to perform this treatment is the Amsterdam UMC, location VU University Medical Center (VUmc). In the past 10 years, 34 live births were achieved by gestational surrogacy (Peters et al., 2018).
To investigate the possibility of performing uterus transplantations in the Amsterdam UMC, we performed a feasibility study to search for ethical, medical and financial support for this innovative, yet ethically and morally challenging, technique. Additionally, we performed a questionnaire study in women with the MRKH syndrome to study the support in patients. In this paper, we present an overview of current practices and literature on UTx and discuss the results of our feasibility study for performing UTx in the Netherlands.
Methods
For this feasibility study, we created a special interest group; this was initiated by the gynaecologists from Amsterdam UMC, location VUmc. The UTx team included gynaecologists, transplant surgeons, researchers and a financial advisor. We pointed out relevant stakeholders: the patients association of women with MRKH syndrome (in Dutch: ‘Stichting MRK-Vrouwen’) and the possible patients, the executive board of the hospital, the Netherlands Association for Obstetrics and Gynaecology (NVOG), the VUmc Clinical Ethics Committee and the potential future UTx team itself for discussing medical technical and ethical aspects.
Moreover, in collaboration with the patients association of women with MRKH syndrome, a questionnaire was designed consisting of questions about UTx, associated risks and financial aspects. A comprehensive information letter was attached to the questionnaire regarding the UTx procedure, financial aspects and possible risks. The questionnaire was sent by the patients association to all its members with diagnosis of MRKH syndrome, age between 18 and 36 years and Dutch nationality (n = 108). This study has been formally exempted from ethical approval, granted by the Institutional Review Board of the VUmc (reference 2019.151, date 20 March 2019).
UTx Procedure
Recipient and donor selection
Potential recipients for UTx are women with absolute uterine factor infertility caused by congenital absence of the uterus (MRKH syndrome) or when acquired after hysterectomy, due to obstetric complications or gynaecological disease. Also, women with a non-functioning uterus can be potential recipients, for example after therapy-resistant Asherman syndrome (severe intrauterine adhesions). Prior to the UTx procedure, an extensive multidisciplinary medical and psychological screening is essential (see Table I).
Table I.
Schematic summary of the program of uterus transplantation.
| Recipient | Living donor |
|---|---|
| Screening (12–18 months) Congenital or acquired absence of uterus, good mental and physical health, stable relationship, age 21–36 years. | Screening (12–18 months) Good mental and physical health, good relation to recipient, non-smoking, ≥1 uncomplicated term pregnancy, complete family, age < 60–65 years. |
| IVF treatment (3–6 months) Before start of UTx, an IVF treatment with successful cryopreservation of embryos should be performed. | |
| Transplantation Abdominal surgery, duration 6–7 hours. Followed by immunosuppressive therapy to reduce the risk of repulsion. | Transplantation Abdominal surgery, duration 8–9 h. Uterus will be removed, including vascular pedicle. Ovaries will stay in situ. |
| Embryo transfer (1 year after transplantation) Single embryo transfer. | |
| Pregnancy Intensive prenatal care and medical control for possible repulsion. Continuation immunosuppressive therapy. Delivery via caesarean section. | |
| Follow-up After 1 or 2 successful pregnancies the uterus will be removed. | |
| Duration for recipient: 3–4 years | Duration for donor: 1–2 years |
The donor uterus can originate from living (LD) or deceased donors (DD). Living donors are commonly a family member or friend. The recipients are themselves responsible for finding a donor. The donor will then be screened for suitability. They should at least have a fulfilled child wish with an uncomplicated obstetric history. The most important concerns in using LD-uterine grafts are the surgical and psychological risks for the donor. DD-uterine grafts can principally be retrieved from brain-dead women, who are formally registered as organ donor. The procedure of uterus retrieval from deceased donors is more simple than from living donors (Lavoue et al., 2017; Favre-Inhofer et al., 2018). However, the logistic organization is complex, with multiple surgical teams to be mobilized when a deceased donor becomes available. The increased transit time for the graft and the removal of vital organs prior to non-vital organs increases the ischemia time. This possibly reduces quality of the uterine graft (Kisu et al., 2013). Moreover, for a DD-graft, a less comprehensive assessment of the uterus prior to transplantation is possible (Lavoue et al., 2017).
Most successful transplantations have been performed with living donors, as 17 out of 19 babies have been born after LD-UTx. In 2018, the first birth from a deceased donor was reported (Ejzenberg et al., 2018). In this report, we focus on UTx from living donors.
Surgical procedure
UTx is a complex procedure demanding major abdominal surgery in the donor and the recipient. The surgical isolation of the donor-uterus involves the delicate dissection of the uterine arteries and veins. Bilateral long vascular pedicles are required to allow for adequate anastomoses to the external iliac vein and artery in the recipient. Details have been extensively described by Brännström et al. (2014). After removal of the uterus, it is flushed and prepared at the back table and then transplanted in the recipient. The procedures in recipient and donor should be carried out synchronally, by teams consisting of a specialized gynaecologist and transplant surgeon.
Most donors reported in the literature had an uncomplicated post-operative course, with a hospital stay of around 6 days. Two major post-operative complications have been described: one ureterovaginal fistula requiring a nephrostomy catheter and a vaginal cuff dehiscence that was surgically repaired (Brännström et al., 2014; Kvarnstrom et al., 2017; Testa et al., 2017).
Follow-up
For the recipient, a combination of immunosuppressive drugs is administered to prevent rejection. This is initiated prior to surgery and is maintained until removal of the graft, i.e. also in the event of pregnancy. Post-operatively, the recipient will be closely monitored through clinical examination and the graft is monitored via imaging and cervical biopsies to detect histopathological signs of rejection. In the first clinical trial in Sweden, two out of nine grafts had to be removed in the first year after transplantation due to post-operative complications. The causes were severe uterine infection and acute thrombosis (Brännström et al., 2014; Johannesson et al., 2015).
IVF and possible pregnancy
Prior to performing the transplantation, an IVF procedure has to be completed with successful cryopreservation (freezing) of embryos. Six to twelve months post-transplantation, a first transfer of a cryo-embryo can be planned with no signs of rejection. In case of pregnancy, this will be closely monitored. In pregnancies following transplantations of solid organs such as kidney, the neonatal outcomes are well-studied and immunosuppressive therapy is deemed safe (Webster et al., 2017). In the Swedish UTx trial, the main obstetric complications were hypertensive disorders. The mean gestational age reached was 35 weeks, and all babies were delivered via elective caesarean section. No birth defects have been reported so far. After one or two pregnancies, hysterectomy is performed to remove the graft, in some at the same time as the caesarean section.
Alternatives
Alternatives for having children for these patients are adoption and surrogacy. Both options are established and allowed by law in the Netherlands. The adoption process is however a complex and lengthy procedure, and obviously the parents do not have a genetic relation with the child. In case of traditional surrogacy, the child is genetically related to the intended father, but not to the intended mother (as the gestational carrier is both genetic and birth mother). A procedure mentioned as an alternative for UTx to achieve a genetically related child is gestational surrogacy, in which an IVF treatment is performed with the intended mother and the resulting embryo is placed in a gestational carrier. With this procedure in the Netherlands, according to legal regulations, the patient herself is responsible for recruiting a woman as gestational carrier. For many women, finding a suitable gestational carrier is very difficult to impossible. Moreover, absence of specific regulation for surrogacy in civil rights is a source for legal uncertainty. This treatment also involves complex ethical considerations, since the gestational carrier takes the medical risks of pregnancy. The psychological implications for gestational carriers and parents have not been studied in detail. In children born after surrogacy, good psychological adjustment is reported although the psychological follow-up is currently very limited (Soderstrom-Anttila et al., 2016; Peters et al., 2018).
Support for UTx in the Netherlands
Professional support
The Netherlands Association for Obstetrics and Gynaecology (NVOG) had declared support for this feasibility study. The executive board of the hospital also supported the initiative. Ethical issues brought up by the local Clinical Ethics Committee will be discussed separately (see paragraph ‘Ethical considerations’).
Patient support
An important aspect in our feasibility study was the rate of support of the possible recipients. The questionnaire was anonymously completed online by 71 women with MRKH syndrome (response rate 66%), with a mean age of 26.7 years (range 18–36). The majority (64.8%) of the respondents would consider UTx with a live donor (see Fig. 1a, depicted per age group). In this group, 69.6% indicated having a potential donor available, with 75% of those being their mother.
Figure 1.
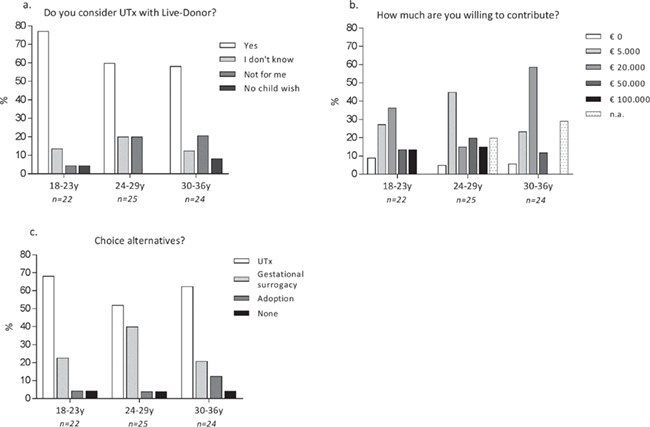
Results of questionnaire study amongst 71 women with MRKH syndrome. Questions were (a) ‘Do you consider UTx with Live-Donor?’; (b) ‘How much are you willing to contribute?’; and (c) ‘Which of the options, UTx, gestational surrogacy or adoption would you choose?’. Answers are depicted as percentage per age group.
We also asked a question about available alternatives to fulfil the desire to have children. If uterus transplantations, gestational surrogacy and adoption were all to be reimbursed by health insurance, still 60.6% would prefer a uterus transplantation, 28.2% preferred gestational surrogacy, 7.0% chose adoption and the remaining 4.2% chose ‘none of the above’ (Fig. 1b).
With regard to financial aspects, it was asked how much they would be willing to contribute, with the options allowing €0, €5000, €20 000, €50 000 and €100 000 (Fig. 1c). The most frequently given answers were €5000 (32.2%) and €20 000 (35.6%). The two younger groups of women were generally willing to contribute more :13.6% (18–23 years) and 15% (24–29 years) were willing to contribute €100 000, compared with 0% in the group of 30–36 years.
In addition, we asked the question of which complications they would be willing to accept for themselves and for their potential donor. The majority of the respondents indicated to accept the chance of a post-surgical haemorrhage for both herself (73.2%; Fig. 2a) and her potential donor (66.2%; Fig. 2b). The risk of rejection of a newly acquired uterus was accepted by 36.6%. The risk of dying during the procedure was unacceptable for all respondents. The women tended to accept more risks with their own surgery than the donor surgery.
Figure 2.

Results of questionnaire study amongst 71 women with MRKH syndrome. The questions were regarding potential surgical complications for their own surgery (a) and the surgery of their potential donor (b). Answers are depicted as percentage of the women accepting the proposed complication.
Legal and Financial Aspects
Legal aspects
Organ transplantations are filed under the Act on Special Medical Procedures (in Dutch: Wet bijzondere medische verrichtingen); this means that the organ transplant must be performed in a facility with a special license for this type of transplantation from the Dutch Ministry of Health, Welfare and Sport (in Dutch: Ministerie van Volksgezondheid, Welzijn en Sport). No licenses have been acquired for uterus transplantation in the Netherlands yet.
Financial aspects
The costs for one UTx are calculated in Table II, resulting in total costs of nearly €100 000. This table does not include the costs for a possible pregnancy and/or labour, because this is covered by medical insurance. The costs for UTx however, so far, are not reimbursed. Therefore, funding by research grants would initially be the only way to finance such a program.
Table II.
Calculated costs for UTx in the Netherlands (for the year 2018).
| Costs | Total | ||
|---|---|---|---|
| UTx | Screening donor and recipient | €8.750 | €93.850 |
| IVF treatment (3 cycles) | €13.650 | ||
| Surgery donor (incl. robot, 8 h surgery) and recipient (7 h surgery) | €54.750 | ||
| Follow-up and immunosuppressive therapy (2 years) | €14.200 | ||
| Removal donated uterus | € 2.500 |
Ethical Considerations
Along with medical and scientific progress on UTx, worldwide ethical questions have risen. It is clear that this ‘quality of life transplant’ requires balancing of risks and benefits (Bruno and Arora, 2018b; Shapiro and Ward, 2018; Williams, 2018). In the light of UTx, the ethical principles of beneficence, non-maleficence, autonomy and justice have been extensively discussed (Catsanos et al., 2011; Lefkowitz et al., 2012). To translate this into clinical practice, ‘the Montreal Criteria for ethical feasibility of uterine transplantation’ were constructed (Lefkowitz et al., 2013). These criteria define the acceptable conditions in which UTx could be performed in human.
The Clinical Ethics Committee of the Amsterdam UMC has considered the feasibility of UTx, in view of various ethical principles. They place great emphasis on the principle of non-maleficence, as UTx implicates potentially physical and psychological risks for both recipients and donors. Moreover, it involves the health of the (unborn) child. The ‘fair distribution of health services’ is also an important point in their discussion; by performing UTx in our hospital, it will potentially be at the expense of another treatment or patient population due to the high financial costs. Another essential issue raised is the availability of alternatives such as adoption or gestational surrogacy, in our country. They advised to follow and evaluate the international evolution of the UTx procedure and scientific reports concerning the development of the children born after UTx and reconsider this issue in the future.
Discussion
UTx is a promising experimental treatment for absolute uterine factor infertility. There have been 19 live births after UTx reported so far, and several clinical trials are currently being performed worldwide showing a high level of interest for this technique. In this feasibility study, we considered the various aspects of performing UTx in our hospital. A high level of interest from the possible recipients became apparent from our questionnaire amongst Dutch women with MRKH. However, strict screening regulations for both donor and recipient would apply, allowing only a small portion of women to be eligible (Taran et al., 2019). The UTx procedure puts two women at major health risk because of the extensive surgical procedures and, for the recipient, the immunosuppressive treatment. Moreover, emergency uterine graft removal is reported to be 28.6%, because of complications in the first 6 months after transplantation (Jones et al., 2019). Also, high costs apply and we do not know the long-term consequences for children born after these procedures. In the Netherlands, alternatives are available and future collaboration with experienced foreign clinics that offer the procedure is a possibility not yet investigated. The final assessment of this feasibility study is that that there are not enough grounds to support this procedure at our hospital at this point in time.
Patients’ perspective
It is important to acknowledge the wish for a genetically related child and the possibility to take a role in the child’s health prior to birth by carrying a pregnancy (Richards et al., 2019). Our questionnaire study confirms earlier studies in Europe, that the majority of women with MRKH would consider UTx and have a preference for UTx over gestational surrogacy and adoption (Gauthier, et al., 2015; Saso et al., 2016; Chmel et al., 2018). After evaluation of the results, it appears that the younger women tend to be more positive towards UTx and are willing to contribute more financially. We hypothesize that younger women might have less insight in the risks and high financial burden. The participants in our study only received written comprehensive information on UTx, and we did not discuss this face-to-face. However, a previous study shows that after more extensive counselling, the majority of the women still see UTx as a good treatment option (Saso et al., 2016). By collaborating with the national patient organization, we have reached a broad range of MRKH patients, although our questionnaire study did not include possible recipients with other indications for UTx, such as a post-hysterectomy condition. Saso et al. (2016) reported no differences in perspective on UTx between MRKH and non-MRKH women. In addition, MRKH syndrome is by far the leading indication for requesting UTx (Huet et al., 2016).
Ethical concerns
Despite the fact that UTx is already conducted elsewhere in the world, the narrative on ethical aspects of uterus transplantations is currently still ongoing with contradictory opinions and views (Bruno and Arora, 2018; O'Donovan, 2018a). In our feasibility study, ethical considerations played a major role. Yet, medical ethics is not straightforward science; for example, the principle of non-maleficence is difficult to judge. Concerning the uterus donor, a healthy woman is subjecting herself to a high-risk surgical procedure. The risks of the hysterectomy that has to be performed in a living uterus donor are different from that of regular hysterectomy. The dissection of the connected blood vessels is more delicate, the duration of the surgery is longer and there is a higher risk of ureteral injury. Mortality rates in donors have been estimated to be similar to that for live kidney donation. In living kidney donors, there is no increased risk for mortality (O'Keeffe et al., 2018), although there is some discussion about a possible increased long-term risk for all-cause mortality (Mjoen et al., 2014). This is not likely to apply to uterus donors as the uterus is not an essential organ after reproductive age. Most problems are to be expected perioperative; there is no uterus donor mortality reported as far as we know.
Also, the recipient is subjecting herself and the unborn child to a high-risk pregnancy following transplantation. The most experience with post-transplant pregnancies comes from kidney transplant recipients. Although pregnancy following renal transplantation is considered to be safe, obstetric complications (affecting both mother and child) occur relatively often (Deshpande et al., 2011).
Mortality and morbidity risk for the donor would be eliminated when using deceased donors. We feel it is important to consider this option when DD-UTx is proved to be equally successful to LD-UTX. It has even been mentioned that ‘living uterus donation would no longer be ethically appropriate’ if DD-UTx appears to be effective (Bruno and Arora, 2018b).
Future considerations
Evidently, should uterus transplantation be carried out in our centre in the future, sufficient and extensive surgical training is required for both the procurement of the uterine graft and the recipient operation. It has been suggested by the Swedish team to perform first training on sheep, followed by training on bodies donated to science. Moreover, in many countries the experienced Swedish team was present during the first transplantation.
If UTx is going to be performed in the Netherlands, this should always be in the context of a clinical trial. Consequently, research grants are then needed for funding because of the high costs without insured reimbursement and to avoid UTx from becoming a privileged treatment for the wealthy. Further research should provide information on psychological and general health consequences for all parties involved, including the child. Also newer surgical techniques should be investigated. At present, a clinical trial in Sweden investigates the possibility for UTx using robot surgery in the donor, with the aim to minimize surgical risks (Brännström et al., 2018). Minimizing surgical risks also reduces the ethical principle of ‘non-maleficence’. The results of this study are expected in the near future.
In addition, we consider the possibility of an international collaboration with more experienced clinics in UTx to refer patients. The centralization of care for UTx would preserve the best medical program and a concentrated experience in specialized hospitals in a limited number of countries. Recently, a clinical trial for uterus transplantation from deceased donors was initiated in Ghent, Belgium, where they performed their first, and latest, UTx in October 2018. The continuity of performing the procedure is hampered by the limited availability of suitable donors. This was also shown by a screening program in Germany where more than 50% of the potential patients were excluded after screening as a result of self-withdrawal, unavailability of a donor and incompatibility between donor and recipient (Taran et al., 2019).
Donor availability is an important aspect of the discussion on UTx. In this article, we have focused on related living donors (family members or friends). Non-directed living organ donation is allowed by law in the Netherlands for kidney and (part of the) liver. Evidently, this is voluntary, and financial compensation is prohibited (in Dutch: ‘Wet op orgaandonatie’). In the USA, the first four non-directed living UTx were performed in 2016. The women who volunteered to be a donor all expressed the desire for another woman to be able to carry her own child (Warren et al., 2018). However, it remains a controversial subject, especially in the case of non-life-saving donation. So-called ‘cross-over’ donation can be a solution for donor-recipient incompatibility. In the Netherlands, cross-over kidney transplantation has been introduced as an extra option in the living kidney donation program in 2004. With this program, patients who cannot receive their own partner’s kidney for immunological reasons receive a kidney from the partner of another patient in exchange for a kidney from their own partner (de Klerk et al., 2004). This might also be an option for future clinical trials with uterus transplantations. Female-to-male transgenders have also been suggested as potential donor candidates, because of the voluntary hysterectomy at sex-reassignment surgery (Api et al., 2017). As the Amsterdam UMC has a special focus for transgender care, we want to consider and investigate this unique patient group as possible donors in the future when the technique to harvest the uterus during laparoscopic robot surgery is well-developed.
In summary, considering all medical, ethical and financial aspects, we conclude that it is currently not feasible to establish the UTx procedure at the Amsterdam UMC. We will follow the impending developments concerning this treatment and will re-evaluate its feasibility in the future.
Acknowledgements
We thank the patients association (‘Stichting MRK-Vrouwen’) and all women with the MRKH syndrome who completed the questionnaire, as well as the Clinical Ethics Committee of the VU Medical Center for their thorough research in this topic.
Authors’ roles
The special interest group for UTx consisted of all the authors. Funding was raised by W.H. Design of feasibility study was performed by the research group as a whole. Organization and data collection of questionnaire study was performed by N.M., J.D. and L.J. Financial aspects were calculated by T.F. The manuscript draft was written by H. P and L.J., supervised by W.H; all authors have read and contributed to the manuscript before submission.
Funding
This feasibility study was funded by the VU Medical Center (Innovation grant 2017).
Conflict of interest
No conflicts of interest regarding this study have been reported relevant to the subject of all authors. Dr Lambalk reports grants from Ferring and grants from Merck outside the submitted work.
References
- Andrew S, Ahmed S. A woman delivered the first baby in the US born from the transplanted uterus of a dead donor [internet]. CNN 2019 [cited on 19 August 2019]. Available from: https://edition.cnn.com/2019/07/09/us/first-us-baby-transplanted-uterus-of-dead-donor-trnd/index.html2019. [Google Scholar]
- Api M, Boza A, Ceyhan M. Could the female-to-male transgender population be donor candidates for uterus transplantation? Turk J Obstet Gynecol 2017;14:233–237. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brannstrom M. Womb transplants with live births: an update and the future. Expert Opin Biol Ther 2017;17:1105–1112. [DOI] [PubMed] [Google Scholar]
- Brännström M, Dahm-Kähler P, Kvarnström N. Robotic-assisted surgery in live-donor uterus transplantation. Fertil Steril 2018;109:256–257. [DOI] [PubMed] [Google Scholar]
- Brannstrom M, Johannesson L, Bokstrom H, Kvarnstrom N, Molne J, Dahm-Kahler P, Enskog A, Milenkovic M, Ekberg J, Diaz-Garcia C et al. Livebirth after uterus transplantation. Lancet 2015;385:607–616. [DOI] [PubMed] [Google Scholar]
- Brännström M, Johannesson L, Dahm-Kähler P, Enskog A, Mölne J, Kvarnström N, Diaz-Garcia C, Hanafy A, Lundmark C, Marcickiewicz J et al. First clinical uterus transplantation trial: a six-month report. Fertil Steril 2014;101:1228–1236. [DOI] [PubMed] [Google Scholar]
- Bruno B, Arora KS. Uterus transplantation: response to open peer commentaries on the ethics of using deceased versus living donors. Am J Bioeth 2018a;18:W6–W8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bruno B, Arora KS. Uterus transplantation: the ethics of using deceased versus living donors. Am J Bioeth 2018b;18:6–15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Catsanos R, Rogers W, Lotz M. The ethics of uterus transplantation. Bioethics 2011;27:65–73. [DOI] [PubMed] [Google Scholar]
- Chmel R, Novackova M, Janousek L, Matecha J, Pastor Z, Maluskova J, Cekal M, Kristek J, Olausson M, Fronek J. Revaluation and lessons learned from the first 9 cases of a Czech uterus transplantation trial: four deceased donor and 5 living donor uterus transplantations. Am J Transplant 2019;19:855–864. [DOI] [PubMed] [Google Scholar]
- Chmel R, Novackova M, Pastor Z, Fronek J. The interest of women with Mayer-Rokitansky-Kuster-Hauser syndrome and laparoscopic Vecchietti neovagina in uterus transplantation. J Pediatr Adolesc Gynecol 2018;31:480–484. [DOI] [PubMed] [Google Scholar]
- Dahm-Kähler P, Wranning C, Lundmark C, Enskog A, Mölne J, Marcickiewicz J, El-Akouri Randa R, McCracken J, Brännström M. Transplantation of the uterus in sheep: methodology and early reperfusion events. J Obstet Gynaecol Res 2008;34:784–793. [DOI] [PubMed] [Google Scholar]
- de Klerk M, Ijzermans JN, Kranenburg LW, Hilhorst MT, van Busschbach JJ, Weimar W. Cross-over transplantation; a new national program for living kidney donations. Ned Tijdschr Geneeskd 2004;148:420–423. [PubMed] [Google Scholar]
- Deshpande NA, James NT, Kucirka LM, Boyarsky BJ, Garonzik-Wang JM, Montgomery RA, Segev DL. Pregnancy outcomes in kidney transplant recipients: a systematic review and meta-analysis. Am J Transplant 2011;11:2388–2404. [DOI] [PubMed] [Google Scholar]
- Ejzenberg D, Andraus W, Baratelli Carelli Mendes LR, Ducatti L, Song A, Tanigawa R, Rocha-Santos V, Macedo Arantes R, Soares JM Jr, Serafini PC et al. Livebirth after uterus transplantation from a deceased donor in a recipient with uterine infertility. Lancet 2018. [DOI] [PubMed] [Google Scholar]
- Enskog A, Johannesson L, Chai DC, Dahm-Kähler P, Marcickiewicz J, Nyachieo A, Mwenda JM, Brännström M. Uterus transplantation in the baboon: methodology and long-term function after auto-transplantation. Hum Reprod 2010;25:1980–1987. [DOI] [PubMed] [Google Scholar]
- Fageeh W, Raffa H, Jabbad H, Marzouki A. Transplantation of the human uterus. Int J Gynaecol Obstet 2002;76:245–251. [DOI] [PubMed] [Google Scholar]
- Favre-Inhofer A, Rafii A, Carbonnel M, Revaux A, Ayoubi JM. Uterine transplantation: review in human research. J Gynecol Obstet Hum Reprod 2018;47:213–221. [DOI] [PubMed] [Google Scholar]
- Flyckt R, Kotlyar A, Arian S, Eghtesad B, Falcone T, Tzakis A. Deceased donor uterine transplantation. Fertil Steril 2017;107:e13. [DOI] [PubMed] [Google Scholar]
- Gauthier T, Garnault D, Therme JF, Piver P, Essig M, Pichon N, Marquet P, Aubard Y. Uterine transplantation: is there a real demand? Gynecol Obstet Fertil 2015;43:133–138. [DOI] [PubMed] [Google Scholar]
- Herlin M, Bjorn AM, Rasmussen M, Trolle B, Petersen MB. Prevalence and patient characteristics of Mayer-Rokitansky-Kuster-Hauser syndrome: a nationwide registry-based study. Hum Reprod 2016;31:2384–2390. [DOI] [PubMed] [Google Scholar]
- Huet S, Tardieu A, Filloux M, Essig M, Pichon N, Therme JF, Piver P, Aubard Y, Ayoubi JM, Garbin O et al. Uterus transplantation in France: for which patients? Eur J Obstet Gynecol Reprod Biol 2016;205:7–10. [DOI] [PubMed] [Google Scholar]
- Johannesson L, Kvarnstrom N, Molne J, Dahm-Kahler P, Enskog A, Diaz-Garcia C, Olausson M, Brannstrom M. Uterus transplantation trial: 1-year outcome. Fertil Steril 2015;103:199–204. [DOI] [PubMed] [Google Scholar]
- Jones BP, Saso S, Bracewell-Milnes T, Thum MY, Nicopoullos J, Diaz-Garcia C, Friend P, Ghaem-Maghami S, Testa G, Johannesson L et al. Human uterine transplantation: a review of outcomes from the first 45 cases. BJOG 2019. [DOI] [PubMed] [Google Scholar]
- Kisu I, Banno K, Mihara M, Suganuma N, Aoki D. Current status of uterus transplantation in primates and issues for clinical application. Fertil Steril 2013;100:280–294. [DOI] [PubMed] [Google Scholar]
- Kisu I, Liu Y, Chen G, Song MJ, Chang CY, Koon TH, Banno K, Aoki D. Current progress in uterus transplantation research in Asia. J Clin Med 2019;8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kvarnstrom N, Jarvholm S, Johannesson L, Dahm-Kahler P, Olausson M, Brannstrom M. Live donors of the initial observational study of uterus transplantation-psychological and medical follow-up until 1 year after surgery in the 9 cases. Transplantation 2017;101:664–670. [DOI] [PubMed] [Google Scholar]
- Lavoue V, Vigneau C, Duros S, Boudjema K, Leveque J, Piver P, Aubard Y, Gauthier T. Which donor for uterus transplants: brain-dead donor or living donor? A systematic review. Transplantation 2017;101:267–273. [DOI] [PubMed] [Google Scholar]
- Lefkowitz A, Edwards M, Balayla J. The Montreal criteria for the ethical feasibility of uterine transplantation. Transpl Int 2012;25:439–447. [DOI] [PubMed] [Google Scholar]
- Lefkowitz A, Edwards M, Balayla J. Ethical considerations in the era of the uterine transplant: an update of the Montreal Criteria for the Ethical Feasibility of Uterine Transplantation. Fertil Steril 2013;100:924–926. [DOI] [PubMed] [Google Scholar]
- Mjoen G, Hallan S, Hartmann A, Foss A, Midtvedt K, Oyen O, Reisaeter A, Pfeffer P, Jenssen T, Leivestad T et al. Long-term risks for kidney donors. Kidney Int 2014;86:162–167. [DOI] [PubMed] [Google Scholar]
- O'Donovan L. Pushing the boundaries: uterine transplantation and the limits of reproductive autonomy. Bioethics 2018;32:489–498. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Keeffe LM, Ramond A, Oliver-Williams C, Willeit P, Paige E, Trotter P, Evans J, Wadstrom J, Nicholson M, Collett D et al. Mid- and long-term health risks in living kidney donors: a systematic review and meta-analysis. Ann Intern Med 2018;168:276–284. [DOI] [PubMed] [Google Scholar]
- Peters HE, Schats R, Verhoeven MO, Mijatovic V, de CJM, Sandberg JL, Peeters IP, Lambalk CB. Gestational surrogacy: results of 10 years of experience in the Netherlands. Reprod Biomed Online 2018. [DOI] [PubMed] [Google Scholar]
- Racho El-Akouri R, Kurlberg G, Dindelegan G, Molne J, Wallin A, Brannstrom M. Heterotopic uterine transplantation by vascular anastomosis in the mouse. J Endocrinol 2002;174:157–166. [DOI] [PubMed] [Google Scholar]
- Richards EG, Agatisa PK, Davis AC, Flyckt R, Mabel H, Falcone T, Tzakis A, Farrell RM. Framing the diagnosis and treatment of absolute uterine factor infertility: insights from in-depth interviews with uterus transplant trial participants. AJOB Empir Bioeth 2019;1–13. [DOI] [PubMed] [Google Scholar]
- Saso S, Clarke A, Bracewell-Milnes T, Saso A, Al-Memar M, Thum MY, Yazbek J, Del G, Hardiman P, Ghaem-Maghami S et al. Psychological issues associated with absolute uterine factor infertility and attitudes of patients toward uterine transplantation. Prog Transplant 2016;26:28–39. [DOI] [PubMed] [Google Scholar]
- Shapiro ME, Ward FR. Uterus transplantation: a step too far. Am J Bioeth 2018;18:36–37. [DOI] [PubMed] [Google Scholar]
- Soderstrom-Anttila V, Wennerholm UB, Loft A, Pinborg A, Aittomaki K, Romundstad LB, Bergh C. Surrogacy: outcomes for surrogate mothers, children and the resulting families-a systematic review. Hum Reprod Update 2016;22:260–276. [DOI] [PubMed] [Google Scholar]
- Taran FA, Scholler D, Rall K, Nadalin S, Konigsrainer A, Henes M, Bosmuller H, Fend F, Nikolaou K, Notohamiprodjo M et al. Screening and evaluation of potential recipients and donors for living donor uterus transplantation: results from a single-center observational study. Fertil Steril 2019;111:186–193. [DOI] [PubMed] [Google Scholar]
- Testa G, Koon EC, Johannesson L, McKenna GJ, Anthony T, Klintmalm GB, Gunby RT, Warren AM, Putman JM, dePrisco G et al. Living donor uterus transplantation: a single center's observations and lessons learned from early setbacks to technical success. Am J Transplant 2017;17:2901–2910. [DOI] [PubMed] [Google Scholar]
- Testa G, McKenna GJ, Gunby RT Jr, Anthony T, Koon EC, Warren AM, Putman JM, Zhang L, dePrisco G, Mitchell JM et al. First live birth after uterus transplantation in the United States. Am J Transplant 2018;18:1270–1274. [DOI] [PubMed] [Google Scholar]
- Warren AM, Testa G, Anthony T, McKenna GJ, Klintmalm GB, Wallis K, Koon EC, Gunby RT Jr, Johannesson L. Live nondirected uterus donors: psychological characteristics and motivation for donation. Am J Transplant 2018;18:1122–1128. [DOI] [PubMed] [Google Scholar]
- Webster P, Lightstone L, McKay DB, Josephson MA. Pregnancy in chronic kidney disease and kidney transplantation. Kidney Int 2017;91:1047–1056. [DOI] [PubMed] [Google Scholar]
- Williams NJ. Deceased donation in uterus transplantation trials: novelty, consent, and surrogate decision making. Am J Bioeth 2018;18:18–20. [DOI] [PubMed] [Google Scholar]
- Wranning CA, Akhi SN, Kurlberg G, Brannstrom M. Uterus transplantation in the rat: model development, surgical learning and morphological evaluation of healing. Acta Obstet Gynecol Scand 2008a;87:1239–1247. [DOI] [PubMed] [Google Scholar]
- Wranning CA, Dahm-Kähler P, Mölne J, Nilsson UA, Enskog A, Brännström M. Transplantation the uterus in the sheep: oxidative stress and reperfusion injury after short-time cold storage. Fertil Steril 2008b;90:817–826. [DOI] [PubMed] [Google Scholar]
- Wranning CA, Marcickiewicz J, Enskog A, Dahm-Kähler P, Hanafy A, Brännström M. Fertility after autologous ovine uterine-tubal-ovarian transplantation by vascular anastomosis to the external iliac vessels. Hum Reprod 2010;25:1973–1979. [DOI] [PubMed] [Google Scholar]


