Abstract
There has been an increase in morbidity and mortality related to coronary heart disease (CHD) in China in recent years. Numerous clinical experiences and studies have shown that traditional Chinese medicine (TCM) plays an important role in the prevention, treatment, and prognosis of CHD. However, the mechanism of TCM in the treatment of CHD has not yet been elucidated. The circRNA-miRNA-mRNA network consists of miRNA that is competitively bound by circRNA, and miRNA regulates the transcription level of mRNA. Through literature review, we found that the circRNA-miRNA-mRNA network acts to contribute to certain effects to CHD such as myocardial hypertrophy, myocardial fibrosis, and heart failure. TCM contains constituents that act against CHD by antiatherosclerosis and apoptosis inhibition action, cardiac and cardiomyocyte protection, and these components also promote cell growth and protection of the vascular system by regulating miRNAs. Therefore, we consider that the circRNA-miRNA-mRNA network may be a new regulatory mechanism for the effective treatment of CHD by TCM.
1. Introduction
The morbidity and mortality caused by coronary heart disease (CHD) are steadily increasing in China. According to a 2018 report on cardiovascular disease in China, in 2013, 11,396,104 people died from CHD, with CHD mortality in 2016 of 113.46/100 thousand for urban residents and 118.78/100 thousand for rural residents. Therefore, it is urgent to find effective methods for the prevention and treatment of CHD events [1].
The etiology of CHD is cavity vascular stenosis, coronary insufficiency, acute or transient ischemia, and hypoxia of myocardial cells caused by coronary atherosclerosis (AS). CHD belongs to the XiongBi or heartache category in traditional Chinese medicine (TCM). With a history of thousands of years, TCM is unique in theory and rich in clinical practice. Syndrome differentiation and treatment and holistic concept are two basic characteristics of TCM. A syndrome reflects the pathological essence of a certain stage in the development of disease. Phlegm and blood syndrome (PBSS) is one of the common syndrome types in CHD, and its formation and development are closely related to blood stasis, obesity, hyperlipidemia, hyperglycemia, body deficiency, old age, and environment. For PBSS, a satisfactory clinical outcome is achieved by promoting blood circulation and dispersing phlegm. There are three types of Chinese medicine used for the prevention and treatment of CHD, namely, a single Chinese medicine (e.g., redroot salvia, Rhodiola, and pseudoginseng), effective ingredients (e.g., tanshinone IIA, salvianolic acid, and ligustrazine), and prescriptions (e.g., Xuefu-Zhuyu decoction, Chaihu-Shugan powder, Gualou-Xiebai-Banxia decoction, and Ditan decoction). However, the mechanisms of these Chinese herbal medicines (CHMs) have not been fully elucidated.
Circular RNAs (circRNA) and micro-RNAs (miRNA) are noncoding RNAs and have been confirmed to contribute to the pathogenesis and progression of CHD [2, 3]. CircRNA and miRNA interact with each other and regulate the mRNA expression of critical genes in CHD genesis and progression. Therefore, we speculated that there is a connection between the circRNA-miRNA-mRNA network and TCM and gathered supporting data through reviewing and summarizing the literature.
2. The Therapeutic Effect of TCM in CHD
TCM aims to treat CHD based on holistic regulation and syndrome differentiation and treatment. According to the model theory of gene constitution syndrome pattern, the occurrence and development of TCM syndromes are mainly based on the genomics of gene expression, which is a powerful approach to gain insight into the prevention and treatment of diseases [4].
In TCM, CHD is also described as XiongBi, which belongs to the category of chest pain, thoracic obstruction, heartache, or angina pectoris as a consequence of cold invasion, improper diet, mood disorder, fatigue, bodily internal injury, or body deficiency due to old age. CHD entails mainly obstruction of the heart channel with injury to other organs including the lungs, liver, spleen, and kidneys [5]. The syndromes of CHD in Chinese clinical medicine manifest as intermingled deficiency in origin (mostly Qi, blood, but also Qi-Yin and Yang) and excess in superficiality (mainly blood stasis and phlegm turbidity, but also cold coagulation and Qi stagnation) [6]. Qi in the TCM is a kind of substance with strong vitality and constant movement and extremely subtle, which is the unity of life substance and physiological function. Yin and Yang represent two opposite aspects of every object and their implicit conflict and interdependence. Generally, anything that is moving, ascending, bright, progressing, and hyperactive, including functional disease of the body, pertains to yang. The characteristics of stillness, descending, darkness, degeneration, and hypoactivity, including organic disease, pertain to Yin.
Traditional Chinese syndromes in XiongBi involve (a) heart-blood stasis syndrome (Xin Xue Yu Zu), (b) cold coagulation in heart-vessel syndrome (Han Ning Xin Mai), (c) Qi stagnation blood stasis syndrome (Qi Zhi Xin Xiong), (d) phlegm stasis in heart-vessel syndrome (Tan Zhuo Bi Zu), (e) deficiency of both Qi and Yin syndrome (Qi Yin Liang Xu), (f) Yin deficiency of heart and kidney syndrome (Xin Shen Yin Xu), and (g) Yang deficiency of heart and kidney syndrome (Xin Shen Yang Xu). These syndrome types are shown in Table 1 [6].
Table 1.
Chinese clinical medicine for XiongBi.
| Syndromes | Therapeutic principle | Formulae | Chinese medicines of formulas |
|---|---|---|---|
| Xin Xue Yu Zu | Huo Xue Hua Yu, Tong Mai Zhi Tong | Xuefu-Zhuyu decoction | Taoren (Prunus persica (L.) Batsch), Honghua (Carthamus tinctorius L.), Danggui (Angelica sinensis (Oliv.) Diels), ShengDihuang (Rehmannia glutinosa Libosch.), Niuxi (Achyranthes bidentata Bl.), Chuanxiong (Ligusticum chuanxiong Hort.), Jiegeng (Platycodon、grandiflorum (Jacq.) A.DC.), Chishao (Paeonia lactiflora PalL), Zhike (Citrus aurantium L.), Gancao (Glycyrrhiza uralensis Fisch.), Chaihu (Bupleurum chinense) |
| Qi Zhi Xin Xiong | Shu Gan Li Qi, Huo XueTong Luo | Chaihu-Shu gan San | Chenpi (Citrus reticulata), Chaihu (Bupleurum chinense), Chuanxiong (Ligusticum chuanxiong Hort.), Xiangfu (Cyperus rotundus), Zhike (Citrus aurantium L.), Shaoyao (Paeonia lactiflora Pall), Gancao (Glycyrrhiza uralensis Fisch.) |
| Tan Zhuo Bi Zu | Tong Yang Xie Zuo, Huo Tan Xuan Bi | (1) Gualou-Xiebai-Banxia decoction; (2) Di-Tan decoction |
(1) Gualou (Trichosanthes kirilowii Maxim.), Xiebai (Allium macrostemon Bge.), Jiangbanxia (Rhizome Pinelliae Preparata), Huangju (rice wine); (2) Fuling (Poria cocos (Schw.) Wolf), Renshen (Panaxginseng C.A.Mey.), Gancao (Glycyrrhiza uralensis Fisch.),Chenpi (Citrus reticulata Blanco), Danxing (Arisaema Cum Bile), JiangBanxia (Rhizome Pinelliae Preparata), Zhuru (Caulis Bambusae in Taenia), Zhishi (Citrus aurantium L.), Shichangpu (Acorus tatarinowii Schott) |
| Han Ning Xin Mai | Xin Wen San Han, Xuan Tong Xin Yang | (1) Zhishi-Xiebai-Guizhi decoction (2) Si-Ning decoction |
(1) Zhishi (Citrus aurantium L.), Houpu (Magnolia officinalis Rehd. et Wils.), Xiebai (Allium macrostemon Bge.), Guizhi (Cinnamomum cassia Presl), Gualou (Trichosanthes kirilowii Maxim.); (2) Danggui (Angelica sinensis (Oliv.) Diels), Guizhi (Cinnamomum cassia Presl), Shaoyao(Paeonia lactiflora Pall), Xixin (Asarum heterotropoides), Tongcao (Tetrapanax papyrifer (Hook.) K. Koch), Gancao (Glycyrrhiza uralensis Fisch.), Dazao (Ziziphus jujuba Mill.) |
| Qi Yin Liang Xu | Yi Qi Yang Yin, Huo Xue Tong Mai | (1) Shengmai San, (2) Renshen -Yang-Rongdecoction |
(1) Renshen (Panax ginseng C.A.Mey.), Maimendong (Ophiopogon japonicus), Wuweizi (Schisandra chinensis (Turcz.) Baill.); (2) Renshen (Panax ginsengC.A.Mey.), Baizhu (Atractylodes macrocephala Koidz.Atractylodes macrocephala), Fuling (Koidz.Atractylodes macrocephala Koidz.), Gancao (Glycyrrhiza uralensis Fisch.), Chenpi (Citrus reticulata), Huangqi (Astragalus membranaceus (Fisch.) Bge.), Danggui (Angelica sinensis (Oliv.) Diels), Baishao (Paeonia lactiflora Pall.), ShuDihuang (Rehmannia glutinosa), Wuweizi (Schisandra chinensis (Turcz.) Baill.), Guixin (Cinnamomum cassia Presl), Zhiyuan (Polygala tenuifolia Willd.) |
| Xin Shen Yin Xu | Zi Yin Qing Huo, Yang Xin He Luo | 1. Tian-Wang-Bu-Xin Dan, 2. Zhi-Gancaodecoction |
(1) Renshen (Panax ginseng C.A.Mey.), Fuling (Poria cocos (Schw.) Wolf), Xuanshen (Scrophularia ningpoensis Hemsl.), Danshen (Salvia miltiorrhiza Bge.), Jiegeng (Platycodon grandiflorum (Jacq.) A.DC.), Yuanzhi (Polygala tenuifolia Willd.), Danggui (Angelica sinensis (Oliv.) Diels), Wuweizi (Schisandra chinensis (Turcz.) Baill.), Maimendong (Ophiopogon japonicus), Tianmendong (Asparagus cochinchinensis), Baiziren (Platycladus orientalis (L.) Franco), Suanzaoren (Semen Ziziphi Spinosae), ShengDihuang (Rehmannia glutinosa Libosch.); (2) Gancao (Glycyrrhiza uralensis Fisch.), Shengjiang (Zingiber officinale Rosc.), Guizhi (Cinnamomum cassia Presl), Renshen (Panax ginseng C.A.Mey.), ShengDihuang (Rehmannia glutinosa Libosch.), Ejiao (Asini Corii colla), Maimendong (Ophiopogon japonicus), Maren (Cannabis sativa L.), Dazao (Ziziphus jujuba Mill.) |
| Xin Shen Yang Xu | Wen Bu Yang Qi, Zhen Fen Xin Yang |
(1) Shen-Fu decoction, (2) You-Gui Yin |
(1) Renshen (Panax ginseng C.A.Mey.), Fuzi (Aconitum carmichaelii Debx.); (2) ShuDihuang (Rehmannia glutinosa), Shanyao (Dioscotea opposita Thunb), Shanzhuyu (Cornus officinalis Sieb.et Zucc.), Gouqizi (Lycium barbarum L.), Gancao (Glycyrrhiza uralensis Fisch.), Duzhong (Eucommia u1moides Oliv.), Rougui (Cinnamomum cassia Presl), ZhiFuzi (Aconitum carmichaelii Debx.) |
Based on the principle of holistic regulation and syndrome differentiation and treatment, CHMs have been widely used in disease prevention and treatment in China and have been proven to have great clinical effectiveness on cardiovascular system disease. A previous study that summarized and introduced the application of plant products in CHD, AS, dyslipidemia, and hypertension also sorted formulae and herbs according to specific cardiovascular diseases [7]. The plant products applied in AS and dyslipidemia were Allium fistulosum L. (Amaryllidaceae), Allium sativum L. (Amaryllidaceae), Astragalus propinquus (Fabaceae), Coptis chinensis Franch. (Ranunculaceae), Crataegus spp. (Rosaceae), Epimedium brevicornum Maxim (Berberidaceae), Fallopia multiflora Thunb. (Polygonaceae), Fermentum rubrum (Aspergillaceae), Olea europaea L. (Oleaceae), Panax ginseng C.A. Mey (Araliaceae), Pueraria lobata (Willd) ohwi (Leguminosae), Reynoutria japonica Houtt (Polygonaceae), Rheum palmatum Linn. (Polygonaceae), Salvia miltiorrhiza Bunge (Lamiaceae), Scutellaria baicalensis Georgi (Lamiaceae), Senna obtusifolia Linn. (Fabaceae), Cassia tora Linn. (Leguminosae), Ligusticum chuanxiong Hort. (Umbelliferae), Rhodiola crenulata H. Ohb (Crassulaceae), and Curcuma longa L. (Gingeraceae) [7–10].
Over the last decades, some TCM decoctions have been confirmed to be effective in the treatment of CHD. For instance, combination therapy with Xuefu Zhuyu decoction and traditional antianginal medications (TAMs) was more effective in treating angina pectoris than TAMs alone. Evidence has indicated that Xuefu Zhuyu decoction combined with TAMs was more effective in improving RAS (RR = 1.29; 95% CI = (1.20, 1.38)), ECG (RR = 1.37; 95% CI = (1.22, 1.54)), and blood HDL-C level (MD = 0.29 mmol/L; 95% CI = (0.23, 0.35)) compared with TAMs alone from a meta-analysis and systematic review of 14 randomized controlled trials [11]. There was benefit in the use of Tongxinluo (TXL) capsule for secondary prevention after acute myocardial infarction (AMI). A systematic review and meta-analysis of randomized controlled trials indicated that TXL improves cardiac function; regulates blood lipid total cholesterol (TC), triglycerides (TG), and low-density lipoprotein-C (LDL-C); and decreases the level of hs-C-reactive protein (hs-CRP) [12]. A meta-analysis indicated that Huoxue Huayu therapy (HXHY) is an effective and safe therapy for patients after percutaneous coronary intervention (PCI). HXHY had a greater beneficial effect on reducing the in-stent restenosis (ISR) rate and the degree of restenosis, improving Seattle Angina Questionnaire (SAQ) scores and increasing the revascularization rate compared with placebo. However, the rate of death and myocardial infarction (MI) of patients treated with HXHY was no different from that of those treated with placebo (P > 0.05) [13].
According to new studies, the pharmacological mechanisms of CHM are relevant to the effects on vascular smooth muscle cells (VSMCs), endothelial cells, cardiomyocytes, macrophages, and monocytes. Details are shown in Table 2 [7].
Table 2.
The pharmacological mechanisms of CHM.
| VSMCs | Endothelial cells | Cardiomyocytes |
|---|---|---|
| Inhibiting expression or activity of contractile and structural proteins | Activation of NO signaling pathway | Alleviation of cardiac hypertrophy |
| Regulating expression of extracellular matrix proteins | Inhibition of inflammation | Attenuation of oxidative stress |
| Regulating calcium levels in a PKA/PKG/PKC-dependent way | Attenuation of oxidative stress | Inhibition of apoptosis |
| Attenuating proliferation and migration of VSMCs | Inhibition of apoptosis, | Opening KATP channels |
| Anti-inflammation improving mitochondrial function | Inducing angiogenesis suppression of endothelial permeability | ANP secretion |
3. Regulation of miRNAs in CHD
With the improvement in modern living standards, phlegm and blood stasis commonly occur and then aggravate each other during CHD progression [14, 15]. Phlegm is a pathological substance caused by the disturbance of body fluid. Blood stasis is a form of pathology caused by the disturbance of blood circulation. Research on CHD with PBSS has mainly focused on lipid metabolism, inflammatory factors, hemorheology, coagulation function, endothelial cell injury, endoplasmic reticulum autophagy, insulin resistance, genomics, proteomics, and metabolomics [16].
MiRNAs are nuclear genome-encoded single-stranded small (18−23 bases) RNAs that are highly expressed in heart tissue, and they play central roles in the miRNA-miRNA functional synergistic network [17]. The specificity and timing of miRNA expression are very similar to the dynamic spatiotemporal characteristics of TCM syndromes. The expression of miR-146b-5p, miR-199a-3p, and miR-199a-5p is upregulated, CALR and TP53 are downregulated in blood stasis syndrome, and miR-363-5p and miR-668 are downregulated, and RIPK2 and STK4 are upregulated in phlegm-turbidity syndrome. In PBSS, the expression of miR-1207-5p, miR-1321, miR-320d, miR-765, and other miRNAs is upregulated, while the expression of miR-1181, miR-1225-3p, miR-1248, miR-668, miR-1223, miR-1281, miR-1538, miR-181d, miR-342-3p, miR-483-3p, miR-491-3p, miR-494, miR-532-3p, miR-668, and miR-98 is downregulated [18–20]. Peroxisome proliferator-activated receptor-α (PPAR-α) is an important factor affecting PBSS that can be regulated by miR-21 [21]. MiR-4433-3p and miR-363-5p are downregulated in patients with Qi deficiency, phlegm, and blood stasis syndrome. In patients with Qi deficiency and blood stasis syndrome, miR-17-5P, miR-320a, and miR-320c are downregulated [22].
The target genes of miRNA related to CHD with PBSS may regulate cell signaling pathways through the transforming growth factor-β (TGF-β) signaling pathway. MiRNAs such as miR-107, miR-146b-5p, miR-199a-5p, and miR-661 are upregulated, while miR-1321 is downregulated after HuoxueHuatan Anshen formula intervention in angina pectoris CHD with PBSS [23]. In addition, TGF-β1 is the initiating factor for synthesizing and depositing collagen fibers and other extracellular active components. After AMI, miR-21 induces myocardial fibrosis through the TGF-0205/Smad7 pathway [24], and circRNA-010567 promotes myocardial fibrosis by inhibiting miR-141 via targeting TGF-β1 [25]. In addition, abnormal proliferation or migration of VSMCs can lead to vessel lesions, resulting in AS and in stent-restenosis. The expression of miR-378a-5p was increased in the group with stent restenosis compared with healthy people, and miR-378a-5p overexpression promoted proliferation and migration in VSMCs by targeting CDK1 [26]. The changes in VSMC phenotype and vascular calcification are major characteristics of AS. VSMCs express miRNAs, and there is a causal link between miRNAs such miR-21, miR-125b, miR-133a, miR-136, miR-143/145, miR-1, and miR-206 and the pathogenesis of vascular disorders in cardiovascular disease with diabetes mellitus [27]. It is important that miRNAs such as miRNA-143/-145, miR-133, miR-125a-5p, miR-23b, miR-638, miR-663, miR-21, miR-100, miR-143, and miR-145 can regulate VMSC function, and miR-126-3p, miR-126-5p, miR-92a, miR-221, and miR-222 can also regulate endothelial cell function [28, 29].
The various aspects of treatment for CHD with PBSS are also closely related to miRNAs. Treatment of spleen phlegm-turbidity and blood stasis syndrome of CHD may be accomplished via multiple targets, including miRNA [30]. Studies have detected 12 differentially expressed miRNAs in dyslipidemia patients with spleen deficiency syndrome, and the results showed 10 downregulated (miR-124-3p, miR-9-5p, miR-133b, miR-1, miR-136-5p, miR-144-3p, miR-133a, miR-149-5p, miR-200a-3p, and miR-219-5p) and 2 upregulated (miR-193a-5p, miR-665) miRNAs that were involved in fatty acid metabolism, phagocytosis, and proliferation- and apoptosis-related signal pathways [31]. In addition, miRNAs can discriminate the severity of blood stasis syndrome, allowing prognostication for patients with acute coronary syndrome. For example, miR-208a-3p expression can be used to distinguish acute coronary syndrome patients with blood stasis syndrome from healthy volunteers. MiR-222-3p and miR-198 may help to grade the severity of blood stasis [32]. Additionally, it was reported that TCM remedies contain active components that can protect cardiomyocytes, protect against AS, inhibit apoptosis, promote cell growth, and protect the vascular system by regulating miRNAs. Details are shown in Tables 3 and 4.
Table 3.
The influence of active ingredients-miRNA in TCM on cardiovascular diseases.
| Active components | Cell types | MiRNAs | Targets | Effects (references) |
|---|---|---|---|---|
| Tanshinone IIA | Macrophages | miR-375 | KLF4 | Anti-AS [33] |
| NRCMs | miR-133 | MAPK ERK1/2 | Protecting cardiomyocytes [34] | |
| H9c2 | miR-152-3p | PTEN | Inhibiting apoptosis [35] | |
| Salvianolate | H9c2 | miR-122-5p | Bax, Bcl-2 | Inhibiting apoptosis [36] |
| Astragaloside IV | H9c2 | miR-23a/miR-92a. | PI3K/AKT | Protects rat cardiomyocytes [37] |
| Notoginsenoside R1 | H9c2 | miR-21 | PI3K/AKT | Cardioprotective actions [38] |
| Ginsenoside Rb1 | NRCMs | miR-208 | — | Protecting H/I impaired NRCMs [39] |
| Ginkgolide B | EPC | miR-126 | Akt | Promoted cell growth [40] |
| Crataegus special extract | HUVEC | miR-155 | eNOS | Protecting vascular [41] |
EPC: endothelial progenitor cells; HUVEC: human umbilical vein endothelial cells; NRCMs: neonatal rat cardiomyocytes.
Table 4.
The influence of gormulas of TCM-miRNA on cardiovascular diseases.
| TCM formulas | miRNAs | Targets | Effects (references) |
|---|---|---|---|
| Compound Danshen dropping pills | miR-200b | VEGF/TGF-β1 | Diabetic cardiomyopathy [42] |
| Fuzheng Huayu Capsule | miR-29 | — | Myocardial fibrosis [43] |
| Xinfukang pill | miR-21 | Fas/FasL | Inhibiting apoptosis [44] |
| Xinfukang pills | miR-1 | Fibullin-2 | Cardiac hypertrophy [45] |
| Qi li qiangxin capsule | niR-199a-5p | — | Cardiac remodeling and hypertrophy [46] |
4. CircRNA-miRNA-mRNA in CHD
CircRNA is an endogenous noncoding RNA. It has no 5′-end cap or 3′-end poly (A) tail as a covalently closed loop from precursor mRNA back-splicing by the spliceosome machinery. Some circRNAs have been shown to act as miRNA sponges, to interact with RNA-binding proteins, to regulate transcription, or to be translated into proteins. CircRNAs have been widely explored in antiaging strategies, diabetes, cardiovascular diseases, and malignant tumors [44–50]. In various cardiovascular tissues and organs, circRNA participates in physiological and pathological processes such as myocardial repair regulation and fibrosis of vascular smooth muscle cells and myocardium, without being affected by the expression of maternal genes. Thus, circRNAs affect the occurrence of cardiovascular diseases such as heart failure and hypertension. CircRNAs and miRNAs interact with each other in CHD genesis and progression. The circRNA-miRNA network may regulate mRNA participating in the development of CHD through competitive endogenous RNA (ceRNA). A new regulation model of gene expression and a complex network system has been developed that involves ceRNA, in which coding RNA and noncoding RNA interact with each other. This regulatory network is constructed to affect the mutual regulation, restriction, and interaction among lncRNA, circRNA, miRNA, and mRNA, thus affecting the target genes participating in CHD [51].
The circRNAs that have been reported to be differentially expressed in Qi stagnation and blood stasis syndrome include circRNA09849, circRNA11523, circRNA18046, and circRNA24450, with circRNA 11523 being related to hsa-circ-0005860 in circBase [52]. Through the KEGG pathway enrichment analysis of differential circRNA in Qi stagnation and blood stasis syndrome, five enrichment pathways were found, namely, path:hsa05144 (malaria), path:hsa04914 (progesterone-mediated oocyte maturation), path:hsa04650 (natural killer cell-mediated cytotoxicity), path:hsa04110 (cell cycle), and path:hsa05203 (viral carcinogenesis). These circRNAs also participate in the regulation of natural killer cell-mediated cytotoxicity and cell cycle. Previous studies showed that a total of 1670 circRNAs and 13 miRNAs were differentially expressed in AMI, and 110 circRNAs and 11 miRNAs were differentially expressed in CHD. Gene ontology (GO) and pathway analyses for differentially expressed circRNAs showed that many pathways, diseases, and functions participate in the development of AMI [53]. One study also showed that there were nine circRNAs, namely, hsa_circ_0089378, hsa_circ_0083357, hsa_circ_0082824, hsa_circ_0068942, hsa_circ_0057576, hsa_circ_0054537, hsa_circ_0051172, hsa_circ_0032970, and hsa_circ_0006323, that could negatively regulate miR-130A-3p, resulting in the upregulation of transient receptor potential cation channel subfamily M member 3 (TRPM3) in patients with CHD, with nine circRNAs promoting TRPM3 expression by inhibiting hsa-miR-130a-3p in CHD patients [54]. The effects of the circRNA-microNA-mRNA network on CHD include myocardial hypertrophy, myocardial fibrosis, and heart failure, which are shown in Table 5.
Table 5.
The influence of PES1 (P) on cardiovascular diseases.
| circRNA | miRNA (P/N) | MRNA (P/N) | Effect |
|---|---|---|---|
| circANRIL | — | PES1 (P) | AS [55] |
| CDR1as | miR-7a (P) | PARP, SP1 (P) | Myocardial infarction [56] |
| circRNA-HRCR | miR-223 (N) | ARC (P) | Myocardial hypertrophy [57] |
| MFACR | miR-625-3p (N) | MTP18 (N) | Apoptosis [58] |
| circRNA_010567 | miR-141 (N) | TGF-β1 (P) | Myocardial fibrosis [25] |
| circNCX1 | miR-133a-3p (N) | CDIP1 (P) | Apoptosis [59] |
| Circ-SATB2 | miR-939 (N) | STIM1 (P) | Cell proliferation and differentiation [60] |
| circCHFR | miR-370 (N) | FOXO1/Cyclin D1 (N) | Migration of vascular smooth muscle [61] |
| circSlc8a1 | miR-133a (N) | SRF, CTGF, Adrb1, Adcy6 (P) | Myocardial hypertrophy [62] |
| circNfix | miR-214 (N) | Gsk3β (P) | Cardiac regeneration [63] |
| circHIPK3 | miR-29b-3p (N) | a-SMA, COL1A1, COL3A1 (N) | Cardiac fibroblasts [64] |
P/N: positive regulation or negative regulation.
Adrb1: adrenoceptor beta 1; Adcy6, adenylate cyclase 6; ARC: activity‐regulated cytoskeletal protein; CDR1as: Cerebellar degeneration-related protein 1 antisense ciRS-7; CDIP1: cell death-inducing protein 1; circANRIL: Circular antisense non-coding RNA in the INK4 locus; COL1A1: type I collagen; COL3A1: type III collagen; EGR1: early growth response protein 1; CTGF: connective tissue growth factor; FOXO1: Forkhead Box Protein O1; Gsk3β: glycogen synthase kinase 3β; IER2: immediate early response protein 2; MFACR: mitochondrial fission and apoptosis-related circRNA; MTP18: mitochondrial protein, 18 kDa; NCX1 sodium/calcium exchanger 1 (ncx1) gene; NR4A1: nuclear receptor subfamily 4 group A member 1; NRG-1: Neuregulin-1; PARP: Poly (ADP-ribose) polymerase; PES1: pescadillo homologue 1; α-SMA: α-smooth muscle actin; SP1: transcription factor Sp1; SRF: serum response factor; STIM1: stromal interaction molecule 1; TGF-β1: transforming growth factor-β1.
CircRNAs with a covalently closed continuous loop are an abundant class of endogenous RNAs that are formed during the maturation of precursor mRNA, and they have been widely researched with respect to cardiovascular diseases. When CircANRIL binds to pescadillo homologue 1 (PES1), it impairs exonuclease-mediated pre-rRNA processing and ribosome biogenesis in vascular smooth muscle cells and macrophages and induces nucleolar stress and p53 activation, resulting in the induction of apoptosis and inhibition of proliferation for conferring atheroprotection [55]. Cdr1as also functioned as a powerful miR-7a sponge in myocardial cells, involving the function of miR-7a targets PARP and SP1 in MI injury [56]. CircRNA HRCR can sequester and inhibit miR-223 activity and was then used in the treatment of cardiac hypertrophy and heart failure through increasing the ARC expression [57]. MFACR regulates mitochondrial dynamics and apoptosis through miR-652-3p and MTP18 [58]. CircRNA_010567 promotes myocardial fibrosis via suppressing miR-141 by targeting TGF-β1 [25]. CircNCX1 promotes cardiomyocyte apoptosis via the miR-133a-3p/CDIP1 [59]. Circ-SATB2 can regulate vascular smooth muscle cell phenotypic differentiation, proliferation, apoptosis, and migration by promoting the expression of STIM1 (a target gene of miR-939) [60]. CircCHFR facilitates the proliferation and migration of vascular smooth muscle via the miR-370/FOXO1/cyclin D1 pathway [61]. CircSlc8a1 can function as an endogenous sponge for miR-133a in cardiomyocytes, and it can regulate the serum response factor, connective tissue growth factor, adrenoceptor beta 1, and adenylate cyclase 6 through directly intervening the knockdown and overexpression in heart failure [62]. CircNfix can suppress Ybx1 ubiquitin-dependent degradation and increase miR-214 activity. It also can promote cardiac regenerative repair and functional recovery after MI [63]. Overexpression of circHIPK3 reverses miR-29b-3p-induced inhibition of cardiac fibrosis proliferation and migration, and then alters the expression levels of miR-29b-3p-targeting genes (a-SMA, COL1A1, COL3A1) in vitro [64].
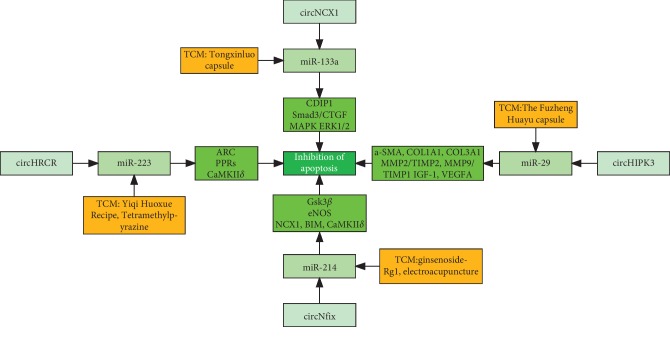
5. Potential circRNA-miRNA-mRNA Network in CHD Treated with TCM
TCM may play a positive role in the treatment of cardiovascular diseases through the circRNA/miRNA/mRNA regulatory network (Figure 1). In myocardial ischemia-reperfusion injury, increasing amounts of circNCX1 competitively bind to the miRNA-133a family, decreasing miRNA-133a-3p levels and increasing the activity of CDIP1, leading to cardiomyocyte deterioration [59]. Tongxinluo can increase the expression of the miRNA-133a family, weaken myocardial fibrosis, and reduce the damage caused by myocardial ischemia [65]. Therefore, Tongxinluo may play a regulatory role through the network of circNCX1/miRNA-133a/mRNA. Modulation of circHRCR/miR-223/ARC levels provides an attractive therapeutic target for the treatment of cardiac hypertrophy and heart failure [57]. TCM that tonifies Qi and activates blood (Shenmai injection, Shenqi Fuzheng injection, Danhong injection, and Qishen Yiqi pills) can significantly reduce the expression of miR-223-3p in the plasma of patients with AMI. Studies have also found that activating blood and dissolving phlegm can significantly reduce the expression level of miR-223 in the serum of hyperlipidemia rats in the myocardial ischemia-reperfusion model and play an anti-inflammatory role through the miR-223/PPRs pathway and inhibit apoptosis [66]. Ligustrazine, which is tetramethylpyrazine (TMP), can inhibit the proliferation, migration, apoptosis, and vascular remodeling of smooth muscle cells by downregulating the gene expression of miR-223 and then inhibiting the expression of CaMKIIδ [67]. Ginsenoside Rg1 upregulates the expression of eNOS by targeting miR-214, which is beneficial for promoting angiogenesis [68]. Electroacupuncture preconditioning can upregulate the expression of miR-214 and inhibit the increase in Ca2+, sodium/calcium exchanger 1 (NCX1), BCL2-like 11 (BIM), calmodulin-dependent protein kinase IIδ (CaMKIIδ), cyclophilin D (CypD), and other related proteins that have accumulated in hypoxia injury in H9c2 myocardial cells, and play a protective role in ischemia/reperfusion (I/R) [69]. The Fuzheng Huayu capsule has the effect of alleviating myocardial fibrosis after MI in rats. This mechanism may be related to upregulation of the expression levels in the miRNA-29 family and direct or indirect regulation of the ratio of matrix metalloproteinase 2/tissue inhibitor of metalloproteinases 2 (MMP2/TIMP2) and MMP9/TIMP1, improving the metabolic balance in the extracellular matrix [70].
Figure 1.
The potential role of the circRNA-miRNA-mRNA regulatory network in the cardiovascular diseases treated with TCM CDIP1: cell death-inducing protein 1; Smad3: recombinant human mothers against decapentaplegic homolog 3; CTGF: connective tissue growth factor; MAPK: mitogen-activated protein kinase; ERK1/2: extracellular signal regulated kinases 1 and 2; ARC: recombinant activity regulated cytoskeleton associated protein; PPRs: pentatricopeptide repeat proteins; CaMKIIδ: calmodulin-dependent protein kinase IIδ; GSK3β: glycogen synthase kinase-3β; eNOS: endothelial nitric oxide synthase; NCX1: sodium/calcium exchanger 1; BIM: BCL2-like 11; a-SMA: anti-α smooth muscle actin; COL1A1: recombinant collagen type I alpha 1; COL3A1: recombinant collagen type III alpha 1; MMP: mitochondrial membrane potential; TIMP: tissue inhibitor of metalloproteinase; IGF-1: insulin-like growth factor-1; and VEGFA: vascular endothelial cells growth factor A.
6. Conclusion
Currently, the main method used to treat CHD in China is to diagnose and treat the disease using modern medicine to determine syndrome differentiation and combine that with the usage of TCM [71]. Numerous studies have confirmed the role of circRNAs and miRNAs in the regulation of the formation and development of AS. CircRNA and miRNA play important roles not only in the regulation of the vascular structure and function but also in the process of CHD and AS. As previously mentioned, circRNA targets are circANRIL (PES1), CDR1as (PARP, SP1), circRNA-HRCR (ARC), MFACR (MTP18), circRNA_010567 (TGF-β1), circNCX1 (CDIP1), circ-SATB2 (STIM1), circCHFR (FOXO1/CyclinD1), circNfix (Gsk3β), and circHIPK3 (a-SMA, COL1A1, COL3A1). Substantial research has been performed showing that miRNAs coordinate their mutual interaction between chronic endothelial repair and endothelial senescence and mechanistically link the regulation of macrophage cholesterol homeostasis with defective efferocytosis. MiRNA and its target gene (s) are let-7a-5p and let-7b-5p (LOX-1), let-7g-5p (LOX-1, TGFBR1 and SMAD2), miR-26a-5p (TRPC6), miR-126-3p (RGS16), miR-126-5p (DLK1), miR-217-5p (SIRT1), miR-216a-5p (BECN1), miR-34a-5p (SIRT1), miR-181b-5p (IPOA3), miR-92a-3p (KLF2, KLF4, SOCS5, and SIRT1), and miR-712-5p (Timp3) in the development of AS.
Single herb medicine, effective components, formulae, and Chinese patent medicines are widely used in the prevention and treatment of CHD. The functions of TCM in clinical treatment mainly include tonifying and regulating Qi, activating blood circulation, reducing phlegm, and relieving pain and consumptive disease. The treatment principles of PBSS in CHD are activating blood circulation and reducing phlegm, which are potential regulators of lipid metabolism and antiplatelet aggregation. However, due to the complex chemical components in CHM, its efficacy is actually a combination of various factors. Therefore, the biggest challenge for the modernization of TCM is to reduce the ambiguity of effective substances and mechanisms.
The circRNA-miRNA-mRNA network may be a new regulatory mechanism. CircRNAs and miRNAs interact with each other to regulate the mRNA expression of some critical genes in CHD genesis and progression. Previous studies showed that TCM contains active components that protect cardiomyocytes, act against atherosclerosis, inhibit apoptosis, promote cell growth, and protect the cardiovascular system by regulating miRNAs. Because there is a very tight connection among circRNAs, miRNAs, and mRNAs, it is likely that TCM may play a positive role in the treatment of CHD through the circRNA-miRNA-mRNA network. In conclusion, the circRNA-miRNA-mRNA network may be an important mechanism that can be affected by Chinese medicine, resulting in the amelioration of CHD. TCM can also be used to affect the circRNA-miRNA-mRNA network to assist with the diagnosis of CHD and the evaluation of the clinical effect of Chinese medicine on CHD.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Authors' Contributions
Fei Lin and Heng-Wen Chen contributed equally to this work.
References
- 1.Hu S. S., Gao R. L., Liu L. S., et al. Summary of 2018 report on cardiovascular diseases in China. Chinese Circulation Journal. 2019;34(3):6–17. in Chinese. [Google Scholar]
- 2.Nelson C. P., Hamby S. E., Saleheen D., et al. Genetically determined height and coronary artery disease. New England Journal of Medicine. 2015;372(17):1608–1618. doi: 10.1056/nejmoa1404881. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Manning K. S., Cooper T. A. The roles of RNA processing in translating genotype to phenotype. Nature Reviews Molecular Cell Biology. 2017;18(2):102–114. doi: 10.1038/nrm.2016.139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wang Z.-D., Jia L.-Q., Wu J., et al. Based on gene-constitution-syndrome research model to discuss syndromes of coronary heart disease. Chinese Archives of Traditional Chinese Medicine. 2016;34(5):1114–1117. [Google Scholar]
- 5.Zhou Z.-Y. Internal Medicine of Traditional Chinese Medicine. Beijing, China: Chinese Medicine Press; 2007. [Google Scholar]
- 6.Zhou Z.-Y. Chinese Internal Medicine. Beijing, China: Beijing China Press of Traditional Chinese Medicine; 2009. [Google Scholar]
- 7.Liu C., Huang Y. Chinese herbal medicine on cardiovascular diseases and the mechanisms of action. Frontiers in Pharmacology. 2016;7:469. doi: 10.3389/fphar.2016.00469. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wang G., Dai G., Song J., et al. Lactone component from Ligusticum chuanxiong alleviates myocardial ischemia injury through inhibiting autophagy. Frontiers in Pharmacology. 2018;9(301) doi: 10.3389/fphar.2018.00301. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Einbond L. S., Manservisi F., Wu H.-A., et al. A transcriptomic analysis of turmeric: curcumin represses the expression of cholesterol biosynthetic genes and synergizes with simvastatin. Pharmacological Research. 2018;132:176–187. doi: 10.1016/j.phrs.2018.01.023. [DOI] [PubMed] [Google Scholar]
- 10.Tao H., Wu X., Cao J., et al. Rhodiola species: a comprehensive review of traditional use, phytochemistry, pharmacology, toxicity, and clinical study. Medicinal Research Reviews. 2019;39(5):1779–1850. doi: 10.1002/med.21564. [DOI] [PubMed] [Google Scholar]
- 11.Yi G.-Z., Qiu Y.-Q., Xiao Y., Yuan L.-x. The usefulness of xuefu zhuyu tang for patients with angina pectoris: a meta-analysis and systematic review. Evidence-Based Complementary and Alternative Medicine. 2014;2014:11. doi: 10.1155/2014/521602.521602 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Li M., Li C., Chen S., et al. Potential effectiveness of Chinese patent medicine Tongxinluo capsule for secondary prevention after acute myocardial infarction: a systematic review and meta-analysis of randomized controlled trials. Frontiers in Pharmacology. 2018;9:p. 830. doi: 10.3389/fphar.2018.00830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Chen R., Xiao Y., Chen M., et al. A traditional Chinese medicine therapy for coronary heart disease after percutaneous coronary intervention: a meta-analysis of randomized, double-blind, placebo-controlled trials. Bioscience Reports. 2018;38(5) doi: 10.1042/bsr20180973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Han X.-J., Shen S.-G. Study the nature of angina pectoris of coronary heart disease marked by phlegm and stagnated blood. Chinese Journal of Basic Medicine in Traditional Chinese Medicine. 2002;8(10):53–54. [Google Scholar]
- 15.Bi Y.-F., Wang X.-L., Zhao Z.-Q., et al. Clinical epidemiological investigation on current syndrome characteristics of coronary heart disease in Chinese medicine. Journal of Traditional Chinese Medicine. 2017;58(23):2013–2019. [Google Scholar]
- 16.Liu Y.-J., Hu J.-Q., Jiang L.-J. Research progress on the molecular mechanism of the syndrome of phlegm and blood stasis in coronary heart disease. Modernization of Traditional Chinese Medicine and Materia Medica-World Science and Technology. 2016;18(5):791–799. [Google Scholar]
- 17.Li Y., Huo C., Pan T., et al. Systematic review regulatory principles of non-coding RNAs in cardiovascular diseases. Briefings in Bioinformatics. 2019;20(1):66–76. doi: 10.1093/bib/bbx095. [DOI] [PubMed] [Google Scholar]
- 18.Yu G. The Research of microRNA Biomarkers of Unstable Angina Patients with Blood Stasis Syndrome. Beijing, China: China Academy of Chinese Medical Sciences; 2013. [Google Scholar]
- 19.Lin F., Chen H.-W., Shi Z.-L., Zhao G.-A., Sun Y.-X. Explore molecular mechanism of Chinese herbs with promoting blood circulation and resolving phlegm effects on myocardial ischemia reperfusion injury based on correlation between microRNA and mtDNA. Zhongguo Zhong yao za zhi. 2017;42(5):1005–1010. doi: 10.19540/j.cnki.cjcmm.20170121.022. [DOI] [PubMed] [Google Scholar]
- 20.Wang J., Yao K.-W., Liu Y.-M., et al. Transcriptomic studies on coronary heart disease with blood stasis syndrome-research approaches and methods in terms of biomarker and disease symptoms. Chinese Journal of Experimental Traditional Medical Formulae. 2017;23(19):1–5. [Google Scholar]
- 21.Chuppa S., Liang M., Liu P., et al. MicroRNA-21 regulates peroxisome proliferator-activated receptor alpha, a molecular mechanism of cardiac pathology in cardiorenal syndrome type 4. Kidney International. 2017;93(2):375–389. doi: 10.1016/j.kint.2017.05.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zhu H.-Z. MicroRNA Different Expression on Coronary Heart Disease with Qi- Deficiency Blood Stasis Syndrome and Qi- Deficiency Phlegm Stasis Syndrome and Treatment of Tongguan Capsule. Guangzhou, China: Guangzhou University of Chinese Medicine; 2017. [Google Scholar]
- 23.Li M. The microRNA Study of Huoxuehuatan’anshen Prescription for Patients with Angina Pectoris in CHD Phlegm and Blood Stasis Syndrome. Beijing, China: China Academy of Chinese Medical Sciences; 2016. [Google Scholar]
- 24.Yuan J., Chen H., Ge D., et al. Mir-21 promotes cardiac fibrosis after myocardial infarction via targeting smad7. Cellular Physiology and Biochemistry. 2017;42(6):2207–2219. doi: 10.1159/000479995. [DOI] [PubMed] [Google Scholar]
- 25.Zhou B., Yu J.-W. A novel identified circular RNA, circRNA_010567, promotes myocardial fibrosis via suppressing miR-141 by targeting TGF-β1. Biochemical and Biophysical Research Communications. 2017;487(4):769–775. doi: 10.1016/j.bbrc.2017.04.044. [DOI] [PubMed] [Google Scholar]
- 26.Liu S., Yang Y., Jiang S., et al. MiR-378a-5p regulates proliferation and migration in vascular smooth muscle cell by targeting CDK1. Frontiers in Genetics. 2019;10:22. doi: 10.3389/fgene.2019.00022. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Song Y., Hou M., Li Z., et al. TLR4/NF-κB/Ceramide signaling contributes to Ox-LDL-induced calcification of human vascular smooth muscle cells. European Journal of Pharmacology. 2017;794:45–51. doi: 10.1016/j.ejphar.2016.11.029. [DOI] [PubMed] [Google Scholar]
- 28.Tang N., Jiang S., Yang Y., et al. Noncoding RNAs as therapeutic targets in atherosclerosis with diabetes mellitus. Cardiovascular Therapeutics. 2018;36(4) doi: 10.1111/1755-5922.12436.e12436 [DOI] [PubMed] [Google Scholar]
- 29.Liu S., Yang Y., Jiang S., et al. Understanding the role of non-coding RNA (ncRNA) in stent restenosis. Atherosclerosis. 2018;272(272):153–161. doi: 10.1016/j.atherosclerosis.2018.03.036. [DOI] [PubMed] [Google Scholar]
- 30.Xv L., Jia L.-Q., Zhang Z., et al. Exploration on scientific connotation of coronary heart disease (CHD) phlegm-turbidity and blood stasis syndrome and treating CHD from spleen based on microRNAs. Chinese Archives of Traditional Chinese Medicine. 2017;35(9):2249–2252. [Google Scholar]
- 31.Yang Z.-M., Chen Y.-Y., Yang X.-R., et al. Bioinformatics analysis of differential expression profiles from serum microRNA in dyslipidemia patients with pi-deficiency syndrome. Chinese Journal of Integrated Traditional Chinese and Western Medicine. 2018;38(3):310–315. [Google Scholar]
- 32.Jia M., Miao Y., Zhao F.-H., et al. Significance of circulating microRNAs in the severity discrimination and prognosis evaluation of acute coronary syndrome patients with blood stasis syndrome. Chinese Journal of Integrated Traditional and Western Medicine. 2018;38(3):300–305. [Google Scholar]
- 33.Chen W., Li X., Guo S., et al. Tanshinone IIA harmonizes the crosstalk of autophagy and polarization in macrophages via miR-375/KLF4 pathway to attenuate atherosclerosis. International Immunopharmacology. 2019;70:486–497. doi: 10.1016/j.intimp.2019.02.054. [DOI] [PubMed] [Google Scholar]
- 34.Zhang L., Wu Y., Li Y., et al. Tanshinone IIA improves miR-133 expression through MAPK ERK1/2 pathway in hypoxic cardiac myocytes. Cellular Physiology and Biochemistry. 2012;30(4):843–852. doi: 10.1159/000341462. [DOI] [PubMed] [Google Scholar]
- 35.Zhang Z., Li Y., Sheng C., Yang C., Chen L., Sun J. Tanshinone IIA inhibits apoptosis in the myocardium by inducing microRNA-152-3p expression and thereby downregulating PTEN. American Journal of Translational Research. 2016;8(7):3124–3132. [PMC free article] [PubMed] [Google Scholar]
- 36.Lin J., Zheng X. Salvianolate blocks apoptosis during myocardial infarction by down regulating miR-122-5p. Current Neurovascular Research. 2018;14(4):323–329. doi: 10.2174/1567202614666171026114630. [DOI] [PubMed] [Google Scholar]
- 37.Gong L., Chang H., Zhang J., Guo G., Shi J., Xu H. Astragaloside IV protects rat cardiomyocytes from hypoxia-induced injury by down-regulation of miR-23a and miR-92a. Cellular Physiology and Biochemistry. 2018;49(6):2240–2253. doi: 10.1159/000493827. [DOI] [PubMed] [Google Scholar]
- 38.Liu Z., Wang H., Hou G., Cao H., Zhao Y., Yang B. Notoginsenoside R1 protects oxygen and glucose deprivation-induced injury by upregulation of miR-21 in cardiomyocytes. Journal of Cellular Biochemistry. 2019;120(6):9181–9192. doi: 10.1002/jcb.28194. [DOI] [PubMed] [Google Scholar]
- 39.Yan X., Liu J., Wu H., et al. Impact of miR-208 and its target gene nemo-Like kinase on the protective effect of ginsenoside Rb1 in hypoxia/ischemia injuried cardiomyocytes. Cellular Physiology and Biochemistry. 2016;39(3):1187–1195. doi: 10.1159/000447825. [DOI] [PubMed] [Google Scholar]
- 40.Chang J., Xue X., Song C., Liu B., Gao L. Ginkgolide B promotes cell growth in endothelial progenitor cells through miR-126 and the Akt signaling pathway. Molecular Medicine Reports. 2017;16(4):5627–5632. doi: 10.3892/mmr.2017.7254. [DOI] [PubMed] [Google Scholar]
- 41.Wang X., Liang Y., Shi J., Zhu H. J., Bleske B. E. Crataegus special extract WS 1442 effects on eNOS and microRNA 155. Planta Medica. 2018;84(15):1094–1100. doi: 10.1055/a-0601-7083. [DOI] [PubMed] [Google Scholar]
- 42.Fan Y., Liang L., Zhang T., et al. Effect of Compound Danshen Dropping Pills on heart of diabetic cardiomyopathy rats by regulating VEGF/TGF-β1 by miRNA-200b. Chinese Journal of Experimental Traditional Medical Formulae. 2019;25(17):34–42. [Google Scholar]
- 43.Qi Y., Zhu K., Ren X., et al. Institute of Basic Research in Clinical Medicine. Effects of Fuzheng Huayu Capsule on the expression of mir-29 family in rats with myocardial fibrosis after myocardial infarction. Global Traditional Chinese Medicine. 2019;12(6):839–844. [Google Scholar]
- 44.Guo P., Liang Y., Chen Z., Li Y., Sun L., Gao K. Effect of Xinfukang Pill on myocardial miRNA-21 of pressure overload rats. Chinese Journal of Experimental Traditional Medical Formulae. 2016;22(21):137–141. [Google Scholar]
- 45.Liang Y., Guo P., Chen Z., et al. Experimental study of Xinfukang pills on microRNA-1 in cardiac tissue of pressure overloaded rats. Tianjin Journal of Traditional Chinese Medicine. 2017;34(1):48–53. [Google Scholar]
- 46.Zhang H., Li S., Zhou Q., et al. Qiliqiangxin attenuates phenylephrine-induced cardiac hypertrophy through downregulation of MiR-199a-5p. Cellular Physiology and Biochemistry. 2016;38(5):1743–1745. doi: 10.1159/000443113. [DOI] [PubMed] [Google Scholar]
- 47.Conn S. J., Pillman K. A., Toubia J., et al. The RNA binding protein quaking regulates formation of circRNAs. Cell. 2015;160(6):1125–1134. doi: 10.1016/j.cell.2015.02.014. [DOI] [PubMed] [Google Scholar]
- 48.Fan X. M., Weng X. Y., Zhao Y. F., Chen W., Gan T., Xu D. Circular RNAs in cardiovascular disease: an overview. Biomed Research International. 2017;2017(6):9. doi: 10.1155/2017/5135781.5135781 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Legnini I., Di Timoteo G., Rossi F., et al. Circ-ZNF609 is a circular RNA that can be translated and functions in myogenesis. Molecular Cell. 2017;66(1):22–37. doi: 10.1016/j.molcel.2017.02.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Wang Y. Y., Li G. P. The emerging landscape of circular RNAs in cardiovascular diseases. Tianjin Medical Journal. 2017;45(3):333–336. [Google Scholar]
- 51.Zhao G.-A. Significance of non-coding circular RNAs and microRNAs in the pathogenesis of cardiovascular diseases. Journal of Medical Genetics. 2018;55(11):713–720. doi: 10.1136/jmedgenet-2018-105387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Gao J.-L. Study on Diagnostic Scale and Differential Expression of circRNA in Qi Stagnation and Blood Stasis Syndrome. Beijing, China: China Academy of Chinese Medical Sciences; 2017. [Google Scholar]
- 53.Lin F., Zhao G.-A., Chen Z.-G., et al. Network correlation of circRNA-miRNA and the possible regulatory mechanism in acute myocardial infarction. Zhonghua Yi Xue Za Zhi. 2018;98(11):851–854. doi: 10.3760/cma.j.issn.0376-2491.2018.11.012. [DOI] [PubMed] [Google Scholar]
- 54.Pan R.-Y., Liu P., Zhou H.-T., et al. Circular RNAs promote TRPM3 expression by inhibiting hsa-miR-130a-3p in coronary artery disease patients. Oncotarget. 2017;8(36):60280–60290. doi: 10.18632/oncotarget.19941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Holdt L. M., Stahringer A., Sass K., et al. Circular non-coding RNA ANRIL modulates ribosomal RNA maturation and atherosclerosis in humans. Nature Communications. 2016;7:p. 12429. doi: 10.1038/ncomms12429. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Geng H. H., Rui L., Su Y. M., et al. The Circular RNA cdr1as promotes myocardial infarction by mediating the regulation of miR-7a on its target genes expression. PLoS One. 2016;11(3) doi: 10.1371/journal.pone.0151753.e0151753 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Wang K., Long B., Liu F., et al. A circular RNA protects the heart from pathological hypertrophy and heart failure by targeting miR-223. European Heart Journal. 2016;37(33):2602–2611. doi: 10.1093/eurheartj/ehv713. [DOI] [PubMed] [Google Scholar]
- 58.Wang K., Gan T. Y., Li N., et al. Circular RNA mediates cardiomyocyte death via miRNA-dependent upregulation of MTP18 expression. Cell Death & Differentiation. 2017;24(6):1111–1120. doi: 10.1038/cdd.2017.61. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Li M., Ding W., Tariq M. A., et al. A circular transcript of ncx1 gene mediates ischemic myocardial injury by targeting miR-133a-3p. Theranostics. 2018;8(21):5855–5869. doi: 10.7150/thno.27285. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.Mao Y. Y., Wang J. Q., Guo X. X., Bi Y., Wang C. X. Circ-SATB2 upregulates STIM1 expression and regulates vascular smooth muscle cell proliferation and differentiation through miR-939. Biochemical and Biophysical Research Communications. 2018;505(1):119–125. doi: 10.1016/j.bbrc.2018.09.069. [DOI] [PubMed] [Google Scholar]
- 61.Yang L., Yang F., Zhao H., Wang M., Zhang Y. Circular RNA circCHFR facilitates the proliferation and migration of vascular smooth muscle via miR-370/FOXO1/cyclin D1 pathway. Molecular Therapy-Nucleic Acids. 2019;16:434–441. doi: 10.1016/j.omtn.2019.02.028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Lim T. B., Aliwarga E., Luu T. D. A., et al. Targeting the highly abundant circular rna circslc8a1 in cardiomyocytes attenuates pressure overload induced hypertrophy. Cardiovascular Research. 2019;115(14):1998–2007. doi: 10.1093/cvr/cvz130. [DOI] [PubMed] [Google Scholar]
- 63.Huang S., Li X., Zheng H., et al. Loss of super-enhancer-regulated circRNA Nfix induces cardiac regeneration after myocardial infarction in adult mice. Circulation. 2019;139(25):2857–2876. doi: 10.1161/circulationaha.118.038361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Ni H., Li W., Zhuge Y., et al. Inhibition of circHIPK3 prevents angiotensin II-induced cardiac fibrosis by sponging miR-29b-3p. International Journal of Cardiology. 2019;292:188–196. doi: 10.1016/j.ijcard.2019.04.006. [DOI] [PubMed] [Google Scholar]
- 65.Luo W.-M. The Role and Mechanism of Unblocking and Collaterals Therapy on Target Organ Damages in Essential Hypertension. Jinan, China: Shandong University; 2016. [Google Scholar]
- 66.Zhu J. W., Dai G. H., Yao J., et al. Effect of Yiqi Huoxue Recipe on the expression of plasma MiR-223-3p and miR-132-5p in patients with acute myocardial infarction. Chinese Journal of Integrative Medicine on Cardio-/Cerebrovascuiar Disease. 2016;14(09):939–943. [Google Scholar]
- 67.Yang M. Effects of Tetramethylpyrazine on Calmodulin-dependent Protein Kinase II Delta Isoform Promote Rats Vascular Remodeling. Shanxi, China: Shanxi Provincial Institute of TCM; 2017. [Google Scholar]
- 68.Chan L. S., Yue P. Y., Mak N. K., Wong N. S. Role of microRNA-214 in ginsenoside-Rg1-induced angiogenesis. European Journal of Pharmaceutical Sciences. 2009;38(4):370–377. doi: 10.1016/j.ejps.2009.08.008. [DOI] [PubMed] [Google Scholar]
- 69.Liu P. Y., Tian Y., Xu S. Y. Mediated protective effect of electroacupuncture pretreatment by miRNA-214 on myocardial ischemia/reperfusion injury. Journal of Geriatric Cardiology. 2014;11(4):303–310. doi: 10.11909/j.issn.1671-5411.2014.04.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Liu W. W. Exosomal circHIPK3 Released from Cardiomyocytes Pretreated with Hypoxia Regulates Apoptosis and Proliferation of Cardiac Microvascular Endothelial Cells under Oxidative Stress. Zunyi, China: Zunyi Medical University; 2019. [Google Scholar]
- 71.Jie W., Fei L., Guo L. L., Xiong X. J., Xun F. Cardiovascular disease, mitochondria, and traditional chinese medicine. Evidence-Based Complementary and Alternative Medicine. 2015;2015:7. doi: 10.1155/2015/143145.143145 [DOI] [PMC free article] [PubMed] [Google Scholar]