Abstract
BACKGROUND AND PURPOSE:
MR angiography (MRA) is increasingly used as a noninvasive imaging technique for the follow-up of coiled intracranial aneurysms. However, the need for contrast enhancement has not yet been elucidated. We compared 3D time-of-flight MRA (TOF-MRA) and contrast-enhanced MRA (CE-MRA) at 3T with catheter angiography.
MATERIALS AND METHODS:
Sixty-seven patients with 72 aneurysms underwent TOF-MRA, CE-MRA, and catheter-angiography 6 months after coiling. Occlusion status on MRA was classified as adequate (complete and neck remnant) or incomplete by 2 independent observers. For TOF-MRA and CE-MRA, interobserver agreement, intermodality agreement, and correlation with angiography were assessed by κ statistics.
RESULTS:
Catheter-angiography revealed incomplete occlusion in 12 (17%) of the 69 aneurysms; 3 aneurysms were excluded due to MR imaging artifacts. Interobserver agreement was good for CE-MRA (κ = 0.77; 95% confidence interval [CI], 0.55–0.98) and very good for TOF-MRA (κ = 0.89; 95% CI, 0.75–1.00). Correlation of TOF-MRA and CE-MRA with angiography was good. The sensitivity of TOF-MRA and CE-MRA was 75% (95% CI, 43%–95%); the specificity of TOF-MRA was 98% (95% CI, 91%–100%) and of CE-MRA, 97% (95% CI, 88%–100%). All 5 incompletely occluded aneurysms, which were additionally treated, were correctly identified with both MRA techniques. Areas under the receiver operating characteristic curve for TOF-MRA and CE-MRA were 0.90 (95% CI, 0.79–1.00) and 0.91 (95% CI, 0.79–1.00). Intermodality agreement between TOF-MRA and CE-MRA was very good (κ = 0.83; 95% CI, 0.65–1.00), with full agreement in 66 (96%) of the 69 aneurysms.
CONCLUSIONS:
In this study, TOF-MRA and CE-MRA at 3T were equivalent in evaluating the occlusion status of intracranial aneurysms after coiling. Because TOF-MRA does not involve contrast administration, this method is preferred over CE-MRA.
Coiling of intracranial aneurysms is an established method for occluding them.1 A shortcoming of coiling is a risk for reopening of the aneurysm, which occurs in about 20% of coiled aneurysms.2–5 Because patients with reopened aneurysms are at risk for hemorrhage, additional treatment is advocated. Follow-up imaging is, therefore, highly recommended. The standard follow-up imaging technique after coiling is catheter angiography, but this diagnostic procedure is invasive, uses ionizing radiation, and exposes the patient to a small risk of serious complications.6,7
MR angiography (MRA) has been used as an alternative noninvasive imaging technique to assess the occlusion of coiled intracranial aneurysms with promising, but not yet conclusive, results.8–22 MRA can be performed without contrast enhancement with 3D time-of-flight (TOF-MRA) or with contrast enhancement (CE-MRA). Contrast administration has several disadvantages, such as patient discomfort, risk of renal damage,23 risk of allergic reaction, and higher cost. From published data, it is unclear which of the 2 MRA techniques, if any, provides the better diagnostic accuracy. To our knowledge, no studies are available that directly compare TOF-MRA and CE-MRA at 3T with angiography as a reference standard in the detection of reopening of coiled aneurysms.
The purpose of our study was to compare the diagnostic performance of TOF-MRA and CE-MRA at 3T with catheter angiography as a reference, at 6-month follow-up of coiled intracranial aneurysms.
Materials and Methods
Patients
The study was approved by the institutional review board. Written informed consent was obtained from all patients.
Between May 2005 and November 2007, all patients with coiled intracranial aneurysms scheduled for 6-month follow-up angiography were selected to participate in this study. Patients were requested to undergo TOF-MRA and CE-MRA at 3T on the same day as their standard follow-up angiography. Patients were not considered eligible when they were younger than 18 years of age, when additional aneurysms were treated with neurosurgical clips, when they had claustrophobia, or when a pacemaker was implanted. Complications of catheter angiography and CE-MRA were recorded.
Imaging Technique: Angiography
Follow-up angiography was performed on a single-plane angiographic unit (Integris Allura Neuro; Philips Medical Systems, Best, the Netherlands). Six to 8 mL of nonionic contrast material (iodixanol, Visipaque 320 mg I/mL; Amersham Health, Cork, Ireland) was injected into the internal carotid or vertebral artery with a power injector at 4–6 mL/s. Three views were acquired in each patient, including the working projection of the endovascular treatment. 3D rotational angiography was usually not performed at follow-up.
Imaging Techniques: MRA
MR imaging examinations were performed on a 3T system (Intera R10; Philips Medical Systems) by using the sensitivity encoding (SENSE) 8-channel phased-array head coil. The protocol included transversal T1-weighted spin-echo and T2-weighted fast spin-echo sequences, phase contrast survey MRA as a preparation for MRA, 3D TOF-MRA with multiple overlapping thin slab acquisition (MOTSA), and CE-MRA sequences.
Imaging parameters for the T1-weighted spin-echo sequence were TR/TE, 500/10 ms; 256 × 205 matrix (reconstructed to 512 × 410); FOV, 230 × 184 mm (ie, 80% rectangular FOV); and 4-mm section thickness with a 1-mm gap. Parameters for the T2-weighted fast spin-echo sequence were TR/TE, 3000/80 ms; 400 × 320 matrix (reconstructed to 512 × 410); FOV, 230 × 184 mm (ie, 80% rectangular FOV); 4-mm section thickness with a 1-mm gap and a turbo spin-echo factor of 15.
For MRA, gradient-echo techniques were used. TOF images were acquired in the transverse plane with the following parameters: TR/TE, 20/4 ms (shortest); flip angle, 20°; 512 × 328 matrix (reconstructed to 1024 × 870); FOV, 200 × 170 mm (ie, 85% rectangular FOV); 1.0-mm thick sections interpolated to 0.5 mm; 220 sections acquired in 10 chunks, resulting in a coverage area of 110 mm. The measured voxel size of the TOF-MRA image was 0.39 × 0.61 × 1 mm, and the reconstructed voxel size was 0.2 × 0.2 × 0.5 mm. The scanning time of MOTSA 3D TOF sequences was reduced by using SENSE. We used a SENSE reduction factor of 1.5, which resulted in an acquisition time of 7 minutes.
The timing for the 3D CE-MRA was calculated from the time-to- peak of a dynamic 2D (0.9-second cycle time) gradient-echo sequence with a midsagittal section of 50-mm thickness following injection of 1 mL of gadopentetate dimeglumine (Magnevist; Bayer Health Care Pharmaceuticals, Montville, NJ) intravenously. Subsequently, 15 mL of gadopentetate dimeglumine was injected at a rate of 2 mL/s for acquisition of 3D CE-MRA. Imaging parameters were as follows: TR/TE, 5.3/1.7 ms (shortest); flip angle, 30°; FOV, 250 × 200 mm (ie, 80% rectangular FOV); 368 × 263 matrix (reconstructed to 512 × 410); 1.0-mm-thick sections interpolated to 0.5 mm. The measured voxel size of the 3D CE-MRA image was 0.68 × 0.76 × 1.00 mm, and the reconstructed voxel size was 0.49 × 0.49 × 0.50 mm. SENSE was also used in 3D CE-MRA imaging with a SENSE reduction factor of 2 and a resulting scanning duration of 36 seconds. Also the randomly sampled central k-space technique was applied here.
Total scanning duration was 20 minutes. Adverse events during MRA were registered.
Image Evaluation
Aneurysm occlusion status on 6-month follow-up angiography was assessed by an experienced interventional neuroradiologist (W.J.v.R.), who was blinded to parallel MRA results. The occlusion status of the coiled aneurysms on TOF- and CE-MRA was classified both in a 3-tier scale as complete occlusion, neck remnant, and incomplete occlusion and in a 2-tier scale as adequate occlusion (complete occlusion and neck remnant) and incomplete occlusion. All TOF-MRA and CE-MRA images were evaluated independently and in random order by 2 experienced interventional neuroradiologists (R.v.d.B. and M.E.S.S.), who were unaware of the parallel angiography results. Source images, 3D maximum-intensity projections, and 3D volume-rendered reconstructions were available on a 3D Vitrea workstation (Vital Images, Minnetonka, Minn). Occlusion status of the coiled aneurysms was classified in the same way as for angiography. Discordant results in TOF-MRA images between the observers after completion of the evaluation were jointly reassessed in a second reading session to reach a consensus; a similar procedure was followed for the CE-MRA images.
Data Analysis
κ statistics were used to assess interobserver agreement for TOF-MRA and CE-MRA and intermodality agreement between TOF-MRA and CE-MRA and to correlate the consensus data of both MRA techniques with angiographic findings. The interpretation of κ was the following: <0.20, poor agreement; 0.21–0.40, fair agreement; 0.41–0.60, moderate agreement; 0.61–0.80, good agreement; and 0.81–1.00, very good agreement.24
Test characteristics of TOF-MRA and CE-MRA with corresponding 95% confidence intervals (CI) were calculated for the 2-tier occlusion scale. This was done because only the classification of incomplete occlusion is important in clinical decision making in terms of considering additional treatment. We compared the areas under the receiver operating characteristic curve (AUROC) for TOF-MRA and CE-MRA.
Results
Patients
Sixty-seven patients (46 women, 21 men; mean age, 49 ± 12 years) with 72 coiled aneurysms agreed to participate in the study. Of 72 coiled aneurysms, 60 were ruptured. Mean aneurysm size was 7 ± 5 mm. Twenty-nine aneurysms were located on the carotid artery; 28, on the anterior cerebral artery; 8, on the middle cerebral artery; and 7, in the posterior circulation. On CE-MRA, 3 aneurysms could not be assessed due to coil artifacts and venous overlap and were excluded, leaving 69 aneurysms evaluated with CE-MRA. On TOF-MRA, 1 aneurysm could not be assessed due to coil artifacts; this aneurysm showed similar coil artifacts on CE-MRA and had already been excluded, leaving 71 aneurysms evaluated with TOF-MRA.
As a result, 69 similar aneurysms in 64 patients were used for the analyses of intermodality agreement and correlation between the imaging techniques (Fig 1). There were no complications, either from angiography or MRA.
Fig 1.
Flow chart of all coiled aneurysms at 6-month follow-up. *Three aneurysms were excluded due to coil artifacts and venous overprojection on CE-MRA. One of these aneurysms was also excluded due to coil artifacts on TOF-MRA.
Angiography
On 6-month follow-up angiography, 57 coiled aneurysms were adequately occluded and 12 aneurysms were incompletely occluded (Fig 1). Of the 12 incompletely occluded aneurysms, 5 were additionally treated.
Interobserver Agreement for MRA
Interobserver agreement for TOF-MRA for the 3-tier classification was good (κ = 0.74; 95% CI, 0.60–0.88), with full agreement in 58 (82%) of the 71 aneurysms that could be evaluated. For the 2-tier classification, interobserver agreement was very good (κ = 0.89; 95% CI, 0.75–1.00), with full agreement in 69 (97%) of 71 aneurysms.
Interobserver agreement for CE-MRA for the 3-tier classification was good (κ = 0.67; 95% CI, 0.51–0.82), with full agreement in 53 (77%) of 69 aneurysms. For the 2-tier classification, interobserver agreement was also good (κ = 0.77; 95% CI, 0.55–0.98), with full agreement in 65 (94%) of 69 aneurysms.
TOF-MRA Compared with Angiography
Correlation between TOF-MRA and angiography for the 3-tier classification was moderate (κ = 0.57; 95% CI, 0.40–0.74), with full agreement in 47 (68%) of 69 aneurysms. For the 2-tier classification, the correlation between TOF-MRA and angiography was good (κ = 0.78; 95% CI, 0.58–0.99), with full agreement in 65 (94%) of 69 aneurysms (Table 1).
Table 1:
Correlation between TOF-MRA and angiography*
| TOF-MRA | Angiography |
|||
|---|---|---|---|---|
| Incomplete Occlusion | Adequate Occlusion |
Total | ||
| Neck Remnant | Complete Occlusion | |||
| Incomplete occlusion | 9 | 0 | 1 | 10 |
| Adequate occlusion | ||||
| Neck remnant | 3 | 14 | 5 | 22 |
| Complete occlusion | 0 | 13 | 24 | 37 |
| Total | 12 | 27 | 30 | 69 |
Note:—TOF-MRA indicates time-of-flight MR angiography.
Correlation for the 3-tier classification was moderate (κ = 0.57; 95% confidence interval [CI]), 0.40–0.74) with a full agreement in 47 (68%) of 69 aneurysms. Correlation for the 2-tier classification, adequate and incomplete occlusion, was good (κ = 0.78; 95% CI, 0.58–0.99), with a full agreement in 65 (94%) of 69 aneurysms.
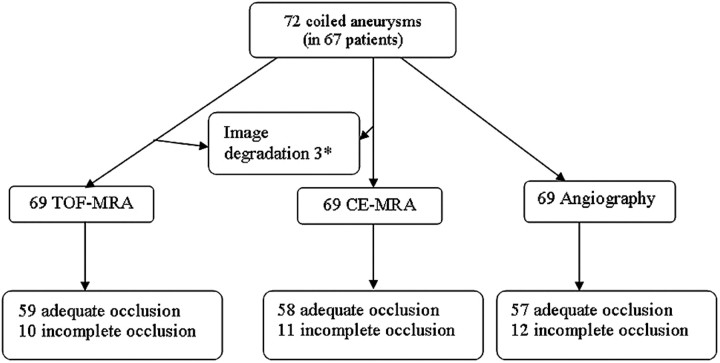
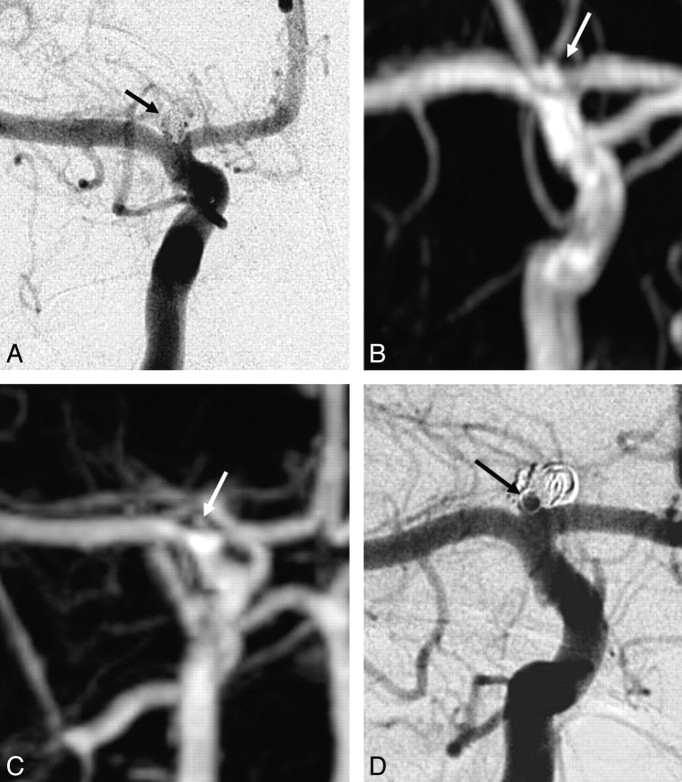
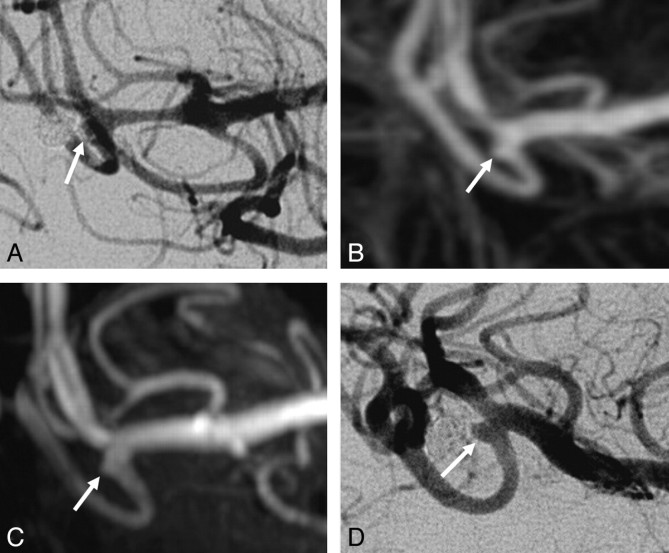
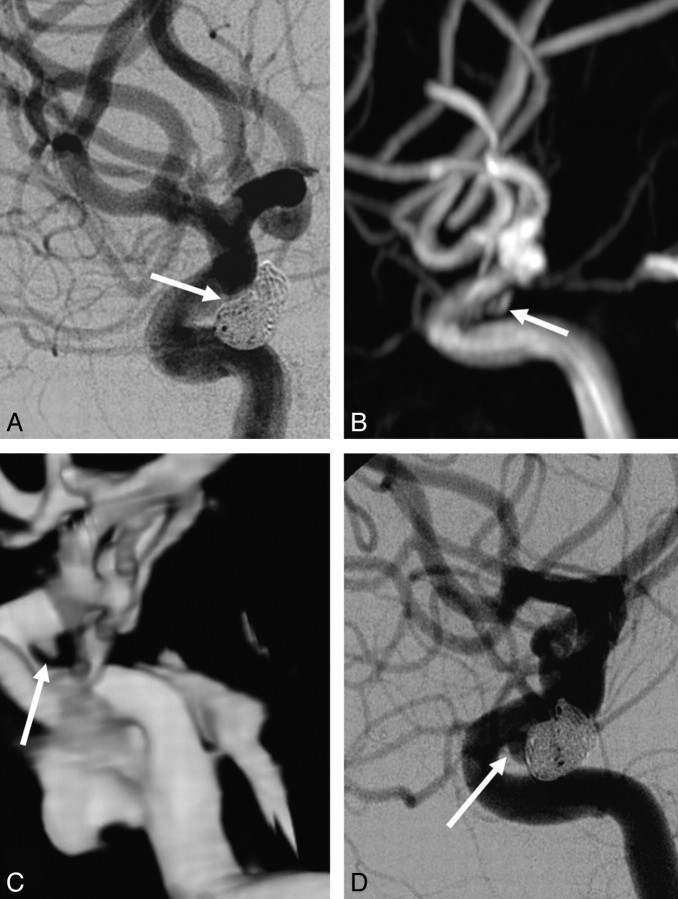
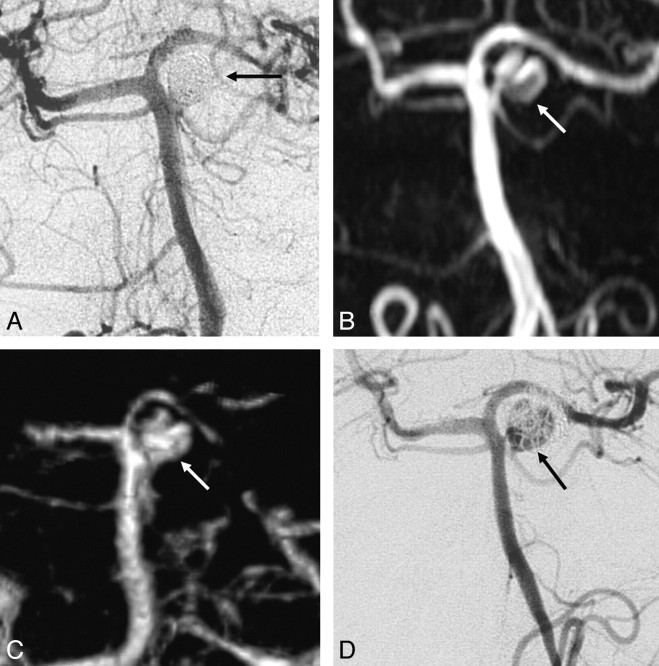
TOF-MRA wrongly classified 3 of the 12 incompletely occluded aneurysms as adequately occluded: a ruptured 4-mm carotid tip aneurysm (Fig 2), a ruptured 4-mm middle cerebral artery aneurysm (Fig 3), and a ruptured 7-mm carotid artery aneurysm (Fig 4). In none of these 3 aneurysms was additional treatment indicated. All 5 incompletely occluded aneurysms that were additionally treated were correctly identified with TOF-MRA as incompletely occluded (Fig 5). One ruptured 6-mm basilar tip aneurysm that was completely occluded on angiography was incorrectly classified by TOF-MRA as incompletely occluded. The sensitivity of TOF-MRA was 75% (95% CI, 43%–95%); the specificity was 98% (95% CI, 91%–100%). All test characteristics of TOF-MRA and AUROC are displayed in Table 2.
Fig 2.
Disagreement between both TOF-MRA and CE-MRA with angiography on the occlusion of a carotid tip aneurysm. A, Angiogram obtained immediately after coiling shows adequate occlusion with a small neck remnant (arrow). B, Follow-up TOF-MRA at 6 months shows complete occlusion (arrow). C, Follow-up CE-MRA at 6 months shows a small neck remnant (arrow). D, Follow-up angiogram at 6 months shows incomplete occlusion (arrow). Because the geometry of the reopened aneurysm was unfavorable, this patient was not retreated but subjected to extended follow-up.
Fig 3.
Disagreement between both TOF-MRA and CE-MRA with angiography on the occlusion of a middle cerebral artery aneurysm. A, Angiogram obtained immediately after coiling shows a small neck remnant (arrow). B, Follow-up TOF-MRA at 6 months shows a small neck remnant (arrow). C, Follow-up CE-MRA at 6 months shows a small neck remnant (arrow). D, Follow-up angiogram shows incomplete occlusion (arrow). Because the geometry of the reopened aneurysm was unfavorable, this patient was not retreated but was subjected to extended follow-up.
Fig 4.
Disagreement between both TOF-MRA and CE-MRA with angiography on the occlusion of a carotid artery aneurysm. A, Angiogram obtained immediately after coiling shows a small neck remnant (arrow). B, Follow-up TOF-MRA at 6 months shows a small neck remnant (arrow). C, Follow-up CE-MRA at 6 months shows a small neck remnant (arrow). D, Follow-up angiogram at 6 months shows incomplete occlusion (arrow). Because the geometry of the reopened aneurysm was unfavorable, this patient was not retreated but was subjected to extended follow-up.
Fig 5.
Agreement between TOF-MRA, CE-MRA, and angiography on the occlusion of a partially thrombosed superior cerebellar artery aneurysm. A, Angiogram obtained immediately after coiling shows complete occlusion (arrow). B, Follow-up TOF-MRA at 6 months shows incomplete occlusion (arrow). C, Follow-up CE-MRA at 6 month shows incomplete occlusion (arrow). D, Follow-up angiogram at 6 month shows incomplete occlusion (arrow). The aneurysm was additionally coiled without complications, and complete occlusion was achieved.
Table 2:
Test characteristics of TOF-MRA and CE-MRA compared with angiography
| TOF-MRA (95% CI) | CE-MRA (95% CI) | |
|---|---|---|
| Sensitivity | 75% (43%–95%) | 75% (43%–95%) |
| Specificity | 98% (91%–100%) | 97% (88%–100%) |
| PPV | 90% (56%–100%) | 82% (48%–98%) |
| NPV | 95% (86%–99%) | 95% (86%–99%) |
| AUROC | 0.90 (0.79%–1.02%) | 0.91 (0.79%–1.02%) |
Note:—CE-MRA indicates contrast-enhanced MR angiography; PPV, positive predictive value; NPV, negative predictive value; AUROC, area under the receiver operating curve; CI, confidence interval.
CE-MRA Compared with Angiography
Correlation between CE-MRA and angiography for the 3-tier classification was moderate (κ = 0.52; 95% CI, 0.35–0.69), with full agreement in 44 (64%) of 69 aneurysms. For the 2-tier classification, correlation between CE-MRA and angiography was good (κ = 0.74; 95% CI, 0.52–0.96), with full agreement in 64 (93%) of 69 aneurysms (Table 3). CE-MRA wrongly classified 3 of the 12 incompletely occluded aneurysms as adequately occluded. These were the same 3 aneurysms that were not correctly classified as incompletely occluded with TOF-MRA (Figs 2–4).
Table 3:
Correlation between CE-MRA and angiography*
| CE-MRA | Angiography |
|||
|---|---|---|---|---|
| Incomplete Occlusion | Adequate Occlusion |
Total | ||
| Neck Remnant | Complete Occlusion | |||
| Incomplete occlusion | 9 | 2 | 0 | 11 |
| Adequate occlusion | ||||
| Neck remnant | 2 | 10 | 5 | 17 |
| Complete occlusion | 1 | 15 | 25 | 41 |
| Total | 12 | 27 | 30 | 69 |
Correlation for the 3-tier classification was moderate (κ = 0.52; 95% CI, 0.35–0.69) with full agreement in 44 (64%) of 69 aneurysms. Correlation for the 2-tier classification, adequate and incomplete occlusion, was good (κ = 0.74; 95% CI, 0.52–0.96), with a full agreement in 64 (93%) of 69 aneurysms.
All 5 incompletely occluded aneurysms that were additionally treated were correctly identified with CE-MRA as incompletely occluded (Fig 5).
CE-MRA incorrectly classified 2 adequately occluded aneurysms as incompletely occluded: a ruptured 3-mm anterior communicating artery aneurysm and a ruptured 6-mm pericallosal artery aneurysm. The sensitivity of CE-MRA was 75% (95% CI, 43%–95%); the specificity was 97% (95% CI, 88%–100%). All test characteristics of CE-MRA and AUROC are displayed in Table 2.
Intermodality Agreement
Intermodality agreement between TOF-MRA and CE-MRA for the 3-tier classification was good (κ = 0.71; 95% CI, 0.57–0.85), with full agreement in 54 (78%) of the 69 aneurysms (Table 4). Intermodality agreement between both MR imaging techniques for the 2-tier classification was very good (κ = 0.83; 95% CI, 0.65–1.00), with full agreement in 66 (96%) of the 69 aneurysms (Table 4).
Table 4:
Intermodality agreement between TOF-MRA and CE-MRA*
| TOF-MRA | CE-MRA |
|||
|---|---|---|---|---|
| Incomplete Occlusion | Adequate Occlusion |
Total | ||
| Neck Remnant | Complete Occlusion | |||
| Incomplete occlusion | 9 | 2 | 0 | 11 |
| Adequate occlusion | ||||
| Neck remnant | 1 | 12 | 4 | 17 |
| Complete occlusion | 0 | 8 | 33 | 41 |
| Total | 10 | 22 | 37 | 69 |
Intermodality agreement for the 3-tier classification was good (κ = 0.71; 95% CI, 0.57–0.85) with full agreement in 54 (78%) of the 69 aneurysms. Intermodality agreement for the 2-tier classification, adequate and incomplete occlusion, was very good (κ = 0.83; 95% CI, 0.65–1.00), with full agreement in 66 (96%) of the 69 aneurysms.
Discussion
In the evaluation of aneurysm-occlusion status 6 months after coiling, diagnostic performance of TOF-MRA equaled that of CE-MRA. Interobserver agreement of both MRA techniques was good. Also correlation with angiography as a reference standard was similar and good for both techniques. The negative predictive values for incomplete occlusion at 6-month follow-up of coiled aneurysms were exactly the same for TOF-MRA and CE-MRA. This implies that contrast enhancement does not have additional value in excluding incomplete occlusion if the TOF-MRA shows an adequate occlusion. The positive predictive value of CE-MRA was somewhat lower than that of TOF-MRA. Thus, in our study group, contrast enhancement had no additional value for ruling in incomplete aneurysms either.
Despite favorable test characteristics, 3 of 12 incompletely occluded aneurysms were wrongly classified as adequately occluded. Both TOF-MRA and CE-MRA failed to identify the same 3 incompletely occluded aneurysms. However, in none of these 3 aneurysms was additional treatment judged to be indicated, and the incorrect classification thus had no clinical consequences. The assessment of the occlusion status was to some extent limited by the use of a classification that allowed room for subjective differences. Apparently, it was sometimes difficult to classify a small residual lumen as borderline adequate or borderline incomplete occlusion.25 All 5 incompletely occluded aneurysms that were additionally treated were correctly identified with both MRA techniques.
Another 3 adequately occluded aneurysms were wrongly classified as incompletely occluded, 1 with TOF-MRA and 2 with CE-MRA. This discrepancy implies that classification of incomplete aneurysm occlusion with MRA should be verified with angiography.
Three Aneurysms were Excluded from Assessment Due to Artifacts on CE-MRA, Compared with 1 Aneurysm on TOF-MRA
We could not find other studies comparing TOF-MRA and CE-MRA at 3T assessed separately in the same patients with angiography as a reference. Previous studies have evaluated either TOF-MRA or CE-MRA for coiled aneurysms.15–18 Other studies have evaluated TOF-MRA and CE-MRA with catheter angiography as a reference,10,14,19,23 of which some reported better diagnostic performance of CE-MRA.19 However, all these studies were performed on 1.5T, and evaluation of the 2 MR imaging techniques independently was not done. One study compared independently TOF-MRA and CE-MRA at 1.5T, with catheter angiography as reference; no significant difference was found between TOF-MRA and CE-MRA.13 A meta-analysis of 16 studies on the diagnostic performance of MRA found no difference between TOF-MRA and CE-MRA.20 However, these findings should be interpreted with some caution because the included studies were of moderate methodologic quality and all pooled estimates were subject to heterogeneity. A recent study on 3T but without catheter angiography as a reference showed a similar classification of aneurysm occlusion on TOF-MRA and CE-MRA, though the visualization of residual flow was considered better on CE-MRA.10
Interobserver and intermodality agreements were substantially better in the 2-tier scales in concordance with a study on aneurysm assessment scales.24 Apparently, in the 2-tier scale, it was sometimes difficult to differentiate a completely occluded aneurysm and a small neck remnant. Although the 2-tier scales may not identify these subgroups, this outcome has little clinical impact because additional treatment is considered only in incompletely occluded aneurysms.
A limitation of our study is the small sample size of 69 aneurysms, of which 12 (17%) were incompletely occluded, which precludes definitive conclusions on whether MRA can replace catheter angiography in the follow-up of coiled aneurysms. However, all patients underwent both TOF-MRA and CE-MRA, resulting in 138 MRA datasets for comparison with angiography, which was sufficient to draw conclusions about the additional value of contrast enhancement in MRA.
Although in several previous studies CE-MRA was considered superior to TOF-MRA, this could not be confirmed in our study. Our TOF-MRA technique was optimized by using a short TE and the MOTSA technique instead of the single-volume 3D-TOF sequence that was used by others.10 In the MOTSA sequence, we used a section thickness of 1 mm, which might seem large for modern scanner technology. However, complete coverage within an acceptable timeframe with a good signal intensity–to-noise ratio limits minimum section thickness. To enhance the signal intensity–to-noise ratio, we used the overlapping slab technique. Halving the section thickness from 1 to 0.5 mm while maintaining the signal intensity–to-noise ratio would require a 4-fold increase in acquisition time. Because the parameters we used resulted in a relatively long acquisition time of 7 minutes, a substantially longer acquisition time was not considered an option.
MOTSA minimizes signal-intensity loss due to spin saturation and maintains small voxels and short TEs to minimize intravoxel phase dispersion. Our MOTSA TOF technique resulted in a reconstructed voxel size of 0.2 × 0.2 × 0.5 mm, whereas for the CE-MRA sequence, the reconstructed voxel size was 0.49 × 0.49 × 0.5 mm. The problem of image degrading by venous overprojection can be decreased by faster imaging with higher SENSE factors, at the expense of a decreased signal intensity–to-noise ratio.10 Large aneurysm remnants may be better visualized by CE-MRA than by TOF-MRA, due to saturation effects and signal-intensity loss with TOF.10 Because in our study no large aneurysm remnants were present, this possible advantage of CE-MRA could not be substantiated.22
In summary, in this study, TOF-MRA and CE-MRA at 3T were equivalent in evaluating the occlusion status of coiled intracranial aneurysms. Because TOF-MRA does not involve contrast administration, this method is preferred over CE-MRA in most patients if MRA is used instead of catheter angiography for the follow-up of coiled intracranial aneurysms.
References
- 1. Molyneux AJ, Kerr RS, Yu LM, et al. , for the International Subarachnoid Aneurysm Trial (ISAT) Collaborative Group. International Subarachnoid Aneurysm Trial (ISAT) of neurosurgical clipping versus endovascular coiling in 2143 patients with ruptured intracranial aneurysms: a randomised trial. Lancet 2002;360:1267 [DOI] [PubMed] [Google Scholar]
- 2. Sluzewski M, van Rooij WJ, Rinkel GJ, et al. Endovascular treatment of ruptured intracranial aneurysms with detachable coils: long-term clinical and serial angiography results. Radiology 2003;227:720–24 [DOI] [PubMed] [Google Scholar]
- 3. Cognard C, Weill A, Spelle L, et al. Long-term angiographic follow-up of 169 intracranial aneurysms occluded with detachable coils. Radiology 1999;212:348–56 [DOI] [PubMed] [Google Scholar]
- 4. Raymond J, Guilbert F, Weill A, et al. Long-term angiographic recurrences after selective endovascular treatment of aneurysms with detachable coils. Stroke 2003;34:1398–403 [DOI] [PubMed] [Google Scholar]
- 5. Piotin M, Spelle L, Mounayer C, et al. Intracranial aneurysms: treatment with bare platinum coils—aneurysm packing, complex coils, and angiographic recurrence. Radiology 2007;243:500–08 [DOI] [PubMed] [Google Scholar]
- 6. Cloft HJ, Joseph GJ, Dion JE. Risk of cerebral angiography in patients with subarachnoid hemorrhage, cerebral aneurysm, and arteriovenous malformation: a meta-analysis. Stroke 1999;30:317–20 [DOI] [PubMed] [Google Scholar]
- 7. Willinsky RA, Taylor SM, Terbrugge K, et al. Neurologic complications of cerebral angiography: prospective analysis of 2899 procedures and review of the literature. Radiology 2003;227:522–28. Epub 2003 Mar 13 [DOI] [PubMed] [Google Scholar]
- 8. Majoie CB, Sprengers ME, van Rooij WJ, et al. MR angiography at 3T versus digital subtraction angiography in the follow-up of intracranial aneurysms treated with detachable coils. AJNR Am J Neuroradiol 2005;26:1349–56 [PMC free article] [PubMed] [Google Scholar]
- 9. Gibbs GF, Huston J, Bernstein MA, et al. Improved image quality of intracranial aneurysms: 3.0-T versus 1.5-T time-of-flight MR angiography. AJNR Am J Neuroradiol 2004;25:84–87 [PMC free article] [PubMed] [Google Scholar]
- 10. Anzalone N, Scomazzoni F, Cirillo M, et al. Follow-up of coiled cerebral aneurysms at 3T: comparison of 3D time-of-flight MR angiography and contrast-enhanced MR angiography. AJNR Am J Neuroradiol 2008;29:1530–36 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Agid R, Willinsky RA, Lee SK, et al. Characterization of aneurysm remnants after endovascular treatment: contrast-enhanced MR angiography versus catheter digital subtraction angiography. AJNR AM J Neuroradiol 2008;29:1570–74 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12. Gibbs GF, Huston J, Bernstein MA, et al. 3.0-Tesla MR angiography of intracranial aneurysms: comparison of time-of-flight and contrast-enhanced techniques. J Magn Reson Imaging 2005;21:97–102 [DOI] [PubMed] [Google Scholar]
- 13. Pierot L, Delcourt C, Bourquigny F, et al. Follow-up of intracranial aneurysms selectively treated with coils: prospective evaluation of contrast-enhanced MR angiography. AJNR Am J Neuroradiol 2006;27:744–49 [PMC free article] [PubMed] [Google Scholar]
- 14. Wikstrom J, Ronne-Enstrom E, Gal G, et al. Three dimensional time-of-flight (3DTOF) magnetic resonance angiography (MRA) and contrast-enhanced MRA of intracranial aneurysms treated with platinum coils. Acta Radiol 2008;49:190–96 [DOI] [PubMed] [Google Scholar]
- 15. Costalat V, Lebars E, Sarry L, et al. In vitro evaluation of 2D-digital subtraction angiography versus 3D-time-of-flight in assessment of intracranial cerebral aneurysm filling after endovascular therapy. AJNR Am J Neuroradiol 2006;27:177–84 [PMC free article] [PubMed] [Google Scholar]
- 16. Lubicz B, Neugroschl C, Collignon L, et al. Is digital subtraction angiography still needed for the follow-up of intracranial aneurysms treated by embolisation with detachable coils? Neuroradiology 2008;50:841–48. Epub 2008 Sep 16 [DOI] [PubMed] [Google Scholar]
- 17. Urbach H, Dorenbeck U, von Falkenhausen M, et al. Three dimensional time-of-flight MR angiography at 3 T compared to digital subtraction angiography in the follow-up of ruptured and coiled intracranial aneurysms: a prospective study. Neuroradiology 2008;50:383–89 [DOI] [PubMed] [Google Scholar]
- 18. Gauvrit JY, Leclerc X, Caron S, et al. Intracranial aneurysms treated with Guglielmi detachable coils: imaging follow-up with contrast-enhanced MR angiography. Stroke 2006;37:1033–37. Epub 2006 Mar 9 [DOI] [PubMed] [Google Scholar]
- 19. Farb RI, Nag S, Scott JN, et al. Surveillance of intracranial aneurysms treated with detachable coils: a comparison of MRA techniques. Neuroradiology 2005;47:507–15 [DOI] [PubMed] [Google Scholar]
- 20. Kwee TC, Kwee RM. MR angiography in the follow-up of intracranial aneurysms treated with Guglielmi detachable coils: systematic review and meta-analysis. Neuroradiology 2007;49:703–13 [DOI] [PubMed] [Google Scholar]
- 21. Deutschmann HA, Augustin M, Simbrunner J, et al. Diagnostic accuracy of 3D time-of-flight MR angiography compared with digital subtraction angiography for follow-up of coiled intracranial aneurysms: influence of aneurysm size. AJNR Am J Neuroradiol 2008;28:628–34 [PMC free article] [PubMed] [Google Scholar]
- 22. Nael K, Villablanca JP, Saleh R, et al. Contrast enhanced MR angiography at 3T in the evaluation of intracranial aneurysms: a comparison with time-of-flight MR angiography. AJNR Am J Neuroradiol 2006;27:2118–21 [PMC free article] [PubMed] [Google Scholar]
- 23. Kuo PH, Kanal E, Abu-Alfa AK, et al. Gadolinium-based MR contrast agents and nephrogenic systemic fibrosis. Radiology 2007;242:647–49 [DOI] [PubMed] [Google Scholar]
- 24. Altman DG. Practical Statistics for Medical Research. London, UK: Chapman and Hall; 1991 [Google Scholar]
- 25. Cloft HJ, Kaufmann T, Kallmes DF. Observer agreement in the assessment of endovascular aneurysm therapy and aneurysm recurrence. AJNR Am J Neuroradiol 2007;28:497–500 [PMC free article] [PubMed] [Google Scholar]