Abstract
BACKGROUND AND PURPOSE:
Aqueductal CSF stroke volume (ACSV) measured by phase-contrast MR imaging is a tool for selection of surgical patients with idiopathic normal-pressure hydrocephalus (iNPH). The aim of the present study was to investigate whether there is a relationship between clinical outcome and changes in ACSV in patients with iNPH who have been shunted.
MATERIALS AND METHODS:
Sixty-five shunted patients with iNPH underwent clinical evaluation and ACSV measurements 7–30 days before and 1, 3, 6, and 12 months after surgery.
RESULTS:
Two patients were excluded from the study for the occurrence of a perioperative complication. In a group of 35 clinically improved patients, the mean preoperative ACSV (157.01 μL) decreased to 18% one month after ventriculoperitoneal shunt (VPS) and ≤49% at 12 months post-VPS. In a group of 15 unimproved patients, the lower mean preoperative ACSV (84.2 μL) decreased to 14.3% one month post-VPS and ≤34% at 12 months post-VPS. In the other 8 improved patients who developed a subdural fluid collection (SDFC), ACSV values decreased by 43%–75% in the 3 months post-VPS. A postoperative ACSV increase was noted in 6 patients with a shunt system malfunction. One patient experienced both SDCF and shunt malfunction.
CONCLUSIONS:
ACSV decreases in all patients in whom the VPS system works properly, with the rate of ACSV decrease being higher in the patients who show clinical improvement. Postoperative ACSV increase suggests shunt malfunction. A precipitous drop of ACSV values after VPS may be the consequence of increased drainage and herald the occurrence of SDFC.
Aqueductal CSF stroke volume (ACSV) measured by phase-contrast MR imaging has been proposed with controversy as a tool for the selection of patients with idiopathic normal-pressure hydrocephalus (iNPH) as candidates for shunt surgery.1–7 ACSV changes with time in untreated patients with iNPH have been previously described.8 Little is known as to how ACSV changes in patients with iNPH after shunt surgery. In this study, ACSV measurements were performed prospectively in patients with iNPH treated with ventriculoperitoneal shunt (VPS), and ACSV trends were correlated with the clinical courses of these patients, to evaluate the clinical utility of ACSV measurements in the management of shunted patients with iNPH.
Materials and Methods
Between March 2000 and December 2005, 65 consecutive patients (41 men and 24 women; mean age, 74.6 years) were treated with VPS. The patients presented with widening of ventricles (Evans ratio, >0.30) on MR imaging and at least 2 of the classic triad of neurologic disturbances associated with iNPH.(9) The patients underwent ACSV measurements 7–30 days before VPS surgery.
Clinical Evaluation
Patients were clinically diagnosed with iNPH on the basis of the cardinal symptoms of gait disturbance, cognitive impairment, and/or urinary bladder urgency or incontinence. Cognitive functions, including orientation, registration, attention, calculation, recall, and language, were evaluated by using the Mini-Mental State Examination (MMSE), in which the minimum score is zero and the maximum is 30. A score of ≤23 is indicative of cognitive impairment.(10) Urinary disorders were evaluated by a urinary incontinence scale (UIS) graded in 4 categories: normal urinary function (grade 0), urgent urination or sporadic incontinence (grade 1), frequent incontinence (grade 2), and complete incontinence (grade 3). Gait disorders were evaluated by a gait scale (GS) graded into 4 categories: normal gait (grade 0); discrete imbalance when turning with short steps, widened base, and occasional falling (grade 1); frequent falls and aid needed for ambulation (grade 2); and impossible gait (grade 3). The onset time of each of these disorders was recorded for each patient.
Phase-Contrast Cine MR Imaging Technique for Dynamic Flow Measurements at the Level of the Aqueduct
The technique for phase-contrast cine MR imaging for dynamic flow measurements at the level of the aqueduct has been previously described.(8) Briefly, the study was performed on a 0.5T superconductive MR imaging scanner (Philips, Best, the Netherlands) by using a phase-contrast cine MR imaging pulse sequence (TR, 54 ms; TE, 9–13 ms; NEX, 2; matrix, 190 × 256 pixels; section thickness, 5 mm; FOV, 12 cm). A cine acquisition with sensitivity to velocity in the section-select direction was obtained on an oblique axial plane of a section perpendicular to the aqueduct (at the inferior colliculus) just above the fourth ventricle. The acquired raw data were interpolated to produce 16 frames equally spaced in the cardiac cycle by means of finger plethysmography with retrospective cardiac gating. In all examinations, the velocity-encoding (VENC) value ranged between 10 and 25 cm/s. For each patient, we tried to use the same VENC value or adjusted it minimally to null the aliasing effect. In our study, ACSV was the average of systolic and diastolic volumes passing through the aqueduct. The area of measurement ranged from 0.062 to 0.146 cm2 and comprised at least 13 pixels. Errors in measurement due to partial volume averaging were minimized by orienting the section perpendicular to the aqueduct and because the pixel size was small relative to the aqueduct. A VENC of 10–25 cm/s allowed measurement without aliasing.
Criteria for Surgical Selection
Between March 2000 and July 2003, selection for VPS surgery in patients with iNPH was based on continuous intracranial pressure (ICP) monitoring (ICPmo). According to our institutional protocol at that time, 52 patients presenting with a mean ICP >9 mm Hg and/or an ICP trend characterized by plateau waves(11) were selected for VPS. ICPmo consisted of monitoring patients for 48 hours. Instruments used for data acquisition were a strain gauge ICP microsensor (Codman MicroSensor; Johnson & Johnson Medical, Berkshire, UK) with a corresponding ventricle catheter, introduced at the level of the frontal horn of the lateral ventricle of the dominant hemisphere, connected to a pressure monitor with an incorporated printing system, BSM-4100J/K (Nihon Kohden, Tokyo, Japan).
Patients were in a supine position for the duration of the procedure, with trunk and head raised by 30°. In addition, between August 2003 to December 2005, 10 patients were selected for VPS on the basis of good response to CSF prolonged external lumbar drainage (PELD). With a Perifix R Kit (B. Braun Medical, Bethlehem, Pa), a 20-gauge closed-tip catheter was introduced through an 18-gauge Tuohy needle in the lumbar (L4–L5 or L5–S1) subarachnoid space. The catheter was connected to a 500-mL open CSF collection system, in which CSF was continuously drained for 24 hours at the velocity of 12–15 mL/h. Good response was defined as a decrease of at least 2 points by the union of UIS and GS scales or when the patient obtained, within 1 week after the external lumbar drainage, an increase of 1 point in UIS or GS and at least 3 points on the MMSE. Finally, VPS was also offered to 3 patients who presented with an unclear response to the prolonged external lumbar drainage, having persistent clinical worsening and a positive ICP.
VPS Surgery Technique
VPS surgery consisted of positioning a catheter in the frontal horn of the lateral ventricles of the nondominant hemisphere ventricle, connected through an interposition of a medium pressure Pudenz valve (Integra Lifesciences, Plainsboro, NJ) to a peritoneal antireflux draining system, whose distal end was positioned in the suprahepatic space. Intravenous antibiotic prophylaxis with ceftazidime 6 g daily and vancomycin 1500 g daily was started the day of surgery and continued for 3 days.
Statistical Analysis
Descriptive statistics are given as mean ± SD. The statistical significance of differences between groups was evaluated by using the nonparametric Wilcoxon matched pairs signed rank test, whereas the Mann-Whitney U test was used for unpaired comparisons. A 2-tailed P value <.05 was considered significant. Statistical Package for the Social Sciences software, Version 16.0, (SPSS, Chicago, Ill) was used for calculation.
Results
The onset time of symptoms ranged from 2 to 72 months (mean, 22.54 ± 16.45). Sixty-two patients (95%) presented with gait disturbances (15 patients, grade 1; 38 patients, grade 2; and 9 patients, grade 3 on the GS). Three patients without gait disturbances (grade 0) all showed marked tetra-hyperreflexia, which we considered as an incipient involvement of pyramidal tracts without clinically apparent gait disorders. Urinary disorders were present in 60 patients (92%) (20 patients, grade 1; 29 patients, grade 2; and 11 patients, grade 3 on the UIS). MMSE values ranged from 12 to 28 (mean, 21.7 ± 3.59). Because of the occurrence of postoperative intracerebral hemorrhage in 1 patient and the occurrence of allergic rejection of his silicon VPS in another patient, 2 patients were excluded from the study. The remaining 63 patients underwent clinical evaluation and ACSV measurements, following the same preoperative modalities at 1, 3, 6, and 12 months after VPS. Extra-schedule checks were performed in the patients who presented with clinical signs suggestive of over- or underdrainage.
Clinical improvement was considered present if a patient obtained the decrease of at least 2 points by the union of UIS and GS or when the patient obtained the increase of 1 point in UIS or GS and at least 3 points in the MMSE. Patients who did not meet these criteria were considered not improved.
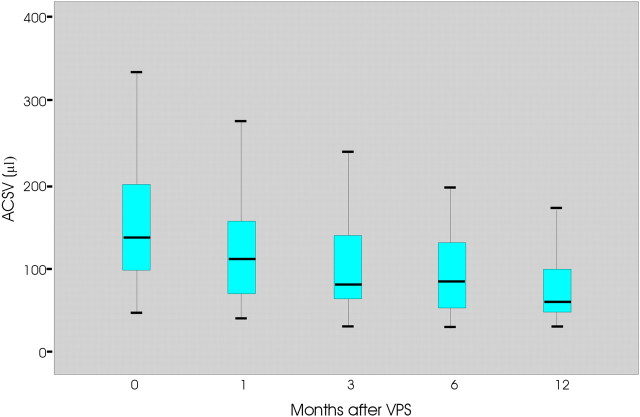
Thirty-five patients improved after VPS. In this group of patients, the mean time of symptom onset was 18 months. ACSV changes with time in the improved patients are reported in Fig 1. The mean preoperative ACSV value was 157.1 μL (ranging between 51 and 330.0 ± 82), which decreased by 18% (mean = 129 μL, ranging between 44 and 281 ± 69) 1 month after VPS and ≤49% at the last control at 12 months (mean = 81, ranging between 36 and 185 ± 41).
Fig 1.
The graph shows the ACSV changes at 0, 1, 3, 6, and 12 months after shunt placement in improved patients with iNPH.
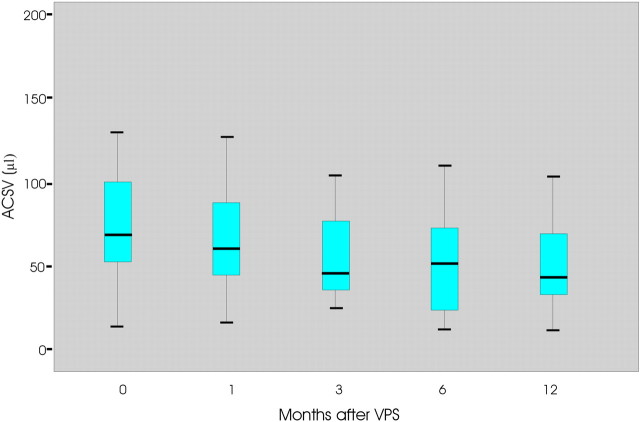
Fifteen patients did not improve after the shunt procedure. In this group, the mean time of symptom onset was 31 months. ACSV changes with time in the unimproved patients are reported in Fig 2. The mean preoperative ACSV value was 84.2 μL (ranging between 20 and 178 ± 39) and decreased to 14.3% (mean = 73 μL, ranging between 23 and 133 ± 34) 1 month after VPS and ≤34% (mean = 56 μL, ranging between 18 and 110 ± 17.5) at the last control at 12 months.
Fig 2.
The graph shows the ACSV changes at 0, 1, 3, 6, and 12 months after shunt placement in unimproved patients with iNPH.
Statistically, there was a significant difference between the preoperative ACSV of the improved and unimproved patients (P < .005) and between the onset time of symptoms between the 2 groups (P < .005). Also, there was a significant difference in P values between the changes in ACSV at 1, 3, 6, and 12 months in both the improved and unimproved patient groups (P < .05).
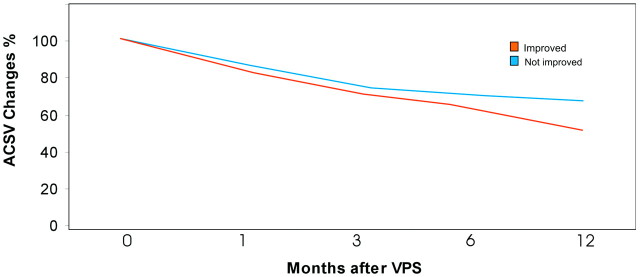
A comparison of the reduction of ACSV values in percentage in improved and not improved patients is reported in Fig 3.
Fig 3.
The graph shows the ACSV changes (percentage) at 0, 1, 3, 6, and 12 months after shunt placement in improved and unimproved patients with iNPH.
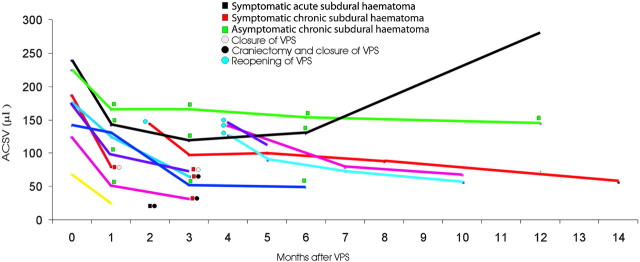
Eight patients who had improved after VPS developed a subdural fluid collection (SDFC) (7 chronic subdural hematomas, 1 acute subdural hematoma). SDFC occurred 1 month after VPS in 5 patients, 2 months after VPS in 1 patient, and 3 months after VPS in 2 patients. In 3 patients, the clinical deterioration coincided with the appearance of the SDFC, whereas in 2 patients, the clinical deterioration occurred 2 months after the imaging findings of the SDFC. In the remaining 3 patients, SDFC remained neurologically silent. Figure 4details the ACSV changes in this subgroup of patients. In the 5 patients who presented with SDFC 1 month after VPS, the mean preoperative ACSV value was 189.6 μL and decreased by 43% in the first month. In the patient who presented with SDFC 2 months after VPS, the preoperative ACSV was 68 μL and decreased by 75% in the first month. In the 2 patients who presented with SDFC 3 months after VPS, preoperative ACSV values of 141 μL and 173 μL decreased respectively by 7% and 28% at the first month and by 63% and 74% at the third month.
Fig 4.
The graph shows the ACSV changes in iNPH patients who developed a SDFC after shunt placement.
Two patients with neurologic deterioration who had a chronic subdural hematoma with mass effect underwent a craniectomy for a surgical evacuation and concomitantly a closure of the distal end of the shunt (with a hemoclip application on the drainage system in the subclavicular region) for a period of 30–40 days. Following this procedure, the 2 patients regained the neurologic deficit associated with the SDFC and presented with re-emergence of iNPH preshunt symptoms. In these patients, the ACSV assessed with a closed valve 1 month after the procedures increased to reach average values 1% lower than the preshunt values.
With the opening of the shunt, clinical improvement continued as did further decrease in the ACSV values. The patient who developed an acute subdural hematoma with severe neurologic deterioration underwent a craniotomy for the evacuation of the hematoma and the closure of the drainage at the level of the neck. This patient later refused reopening of the drainage system and follow-up. Two patients with neurologic deterioration who had a chronic subdural hematoma without mass effect underwent the shunt system closure. In these patients, preshunt symptoms suggestive of iNPH reappeared 1 week after the procedure and the subdural collection decreased after 1 month to then dissolve 3 months after the shunt system closure. ACSV values in these patients, measured with the closed system, increased to a value of 1% lower than the preshunt values. Once the shunt was reopened, the symptoms suggestive of iNPH improved and the values of ACSV decreased.
Three patients with neurologically silent chronic subdural hematoma did not undergo any surgical procedure. In these patients, SDFC remained stable, nonclinical evolution was observed, and ACSV values remained stable with time.
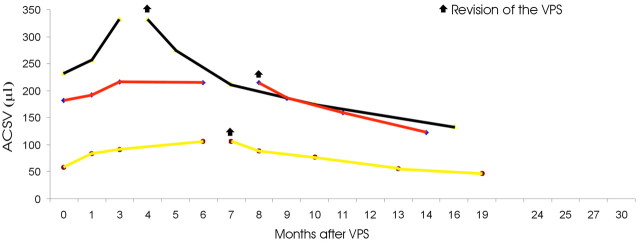
Six patients underwent surgical revision of the shunt system. In 3 of these patients, ACSV increased after VPS. Figure 5details the ACSV changes in this subgroup of patients. One patient of this group presented with a period of improvement for 1.5 months after VPS and an increase of ACSV values at 1, 3, and 6 months. The patient underwent PELD and ICP measuring 7 months after VPS. On the basis of a good response to ELD and mean ICP values of 5.6 mm Hg, the medium was changed with a low-pressure Pudenz valve. After the shunt revision, the patient improved and ACSV decreased at 1, 3, 6, and 12 months. The second patient (red line in Fig 4) in whom ACSV increased at 1, 3, and 6 months after the shunt placement did not improve clinically. The pumping maneuver did not indicate rigidity of the reservoir. The results of a PELD performed at the 6-month follow-up were positive, and the patient underwent shunt revision 8 months after VPS. At surgery, proximal detachment of the valve from the ventricular catheter was detected. After the shunt revision, the patient improved and ACSV decreased at 1, 3, and 6 months. The third of these patients (black line in Fig 4) who presented with increasing ACSV at 1, 3, and 6 months after VPS also did not improve clinically. With the pumping maneuver, the reservoir was not promptly expansible. MR imaging showed the ventricular catheter tangentially positioned at the frontal horn of the lateral ventricle. The patient underwent surgical revision of the system 4 months after VPS, with clinical improvement and ACSV decreasing at 1, 3, 6, and 12 months after the treatment.
Fig 5.
The graph shows the ACSV changes in patients with iNPH who underwent shunt system revision. In these patients, the shunt system did not work properly immediately after the shunt procedure.
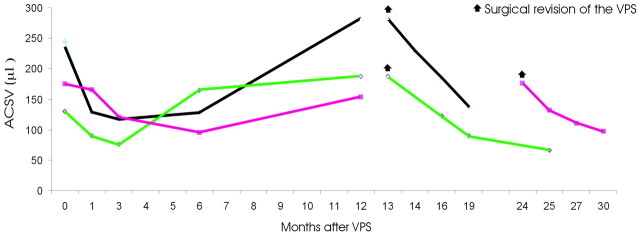
Three patients who underwent surgical revision of the system presented with an increase of ACSV after an initial decrease of ACSV after the shunt placement. Figure 6details the ACSV changes in this subgroup of patients. In 2 patients in this subgroup (1 of them, black line in Figs 4 and 6, has already been considered in the SDCF group), ACSV increased again after valve malfunctioning was detected, and clinical deterioration appeared 12 months after the shunt placement. In these patients, the shunt reservoir was rigid during the pumping maneuver, causing distal obstruction of the shunt. Surgical revision was performed in 1 patient (black line in Fig 6) 1 month after the clinical deterioration.
Fig 6.
The graph shows the ACSV changes in patients with iNPH who underwent shunt system revision after an initial period of improvement and subsequent shunt malfunctioning.
In the second patient (magenta line in Fig 6), surgical revision was performed 12 months after the appearance of the clinical signs due to a decision to postpone the procedure by the patient's guardian. ACSV performed at this time showed a further slight increase in value. After surgical revision, ACSV decreased in both of these patients at 1, 3, and 6 months, whereas clinical improvement was observed only in the patient undergoing the surgical revision at 1 month. In a third patient (green line in Fig 6), a re-increase of ACSV was observed 6 months after the shunt placement and 6 months before appearance of the clinical deterioration and detection of shunt malfunction. In this patient, a distal disconnection of the system was appreciated with x-ray findings. After revision of the system (13 months after the first surgery), the patient again improved and ACSV again continued to decrease at 3, 6, and 12 months.
Discussion
ACSV is defined as the mean volume of CSF passing through the Sylvian aqueduct in a craniocaudal direction during cardiac systole and a caudocranial direction during cardiac diastole, during a single heart cycle.(1) Differences in its value can be appreciated by using different systems and techniques.
During systole, blood normally flows into the brain under arterial pressure, causing inward and outward expansion of the brain parenchyma. Outward brain expansion produces the venous outflow, whereas the inward expansion produces the ventricular squeezing. In this phase, the aqueductal CSF flow induced by the ventricular squeezing is directed craniocaudally. During diastole, with the continuous venous outflow and the reduction of brain blood volume, the CSF in the aqueduct flows caudocranially (Fig 7A). In iNPH, the brain tissue is compressed against the inner table of the calvaria. According to Bradley's hypothesis,(12) with arterial inflow, the brain expansion is mainly directed inward, compressing the enlarged ventricles, leading to a much greater outflow of CSF through the aqueduct, which causes an increased ACSV (Fig 7B). In a previous study,(8) we demonstrated that in unshunted patients with iNPH, ACSV increases with progressive ventricular enlargement, while the tangential and radial shearing of the periventricular brain parenchyma produces a progressive collapsing of the periventricular arterioles, resulting in a reduction of the “pump” of the brain hydraulic system and a corresponding decrease of ACSV (Fig 7C).
Fig 7.
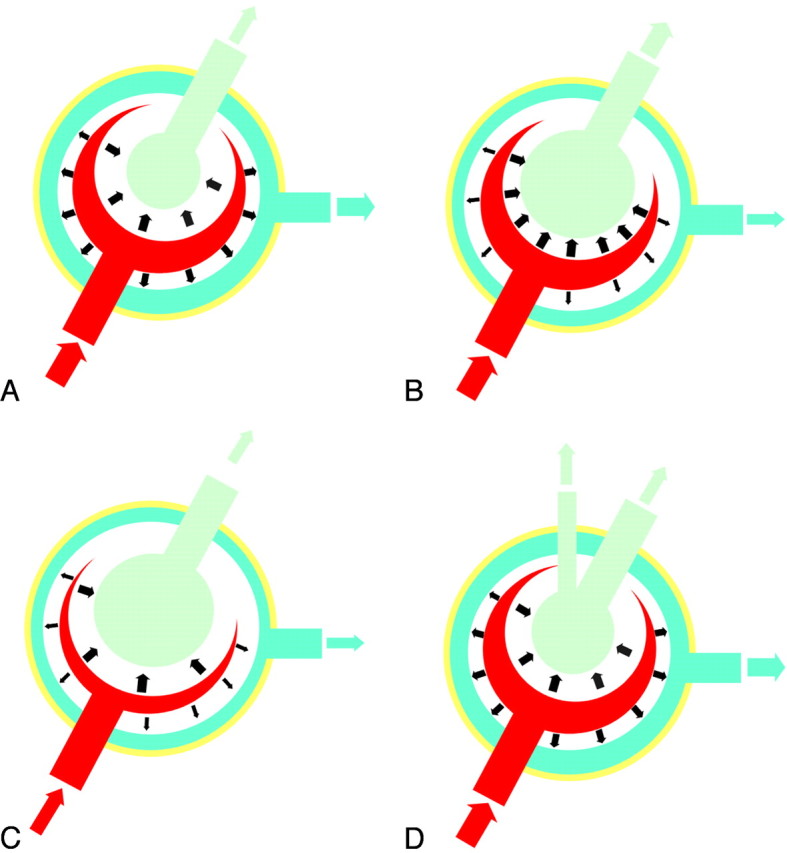
Proposed model demonstrating ACSV changes in iNPH. A, In healthy subjects, expansion of the cerebral hemispheres occurs both outwardly and inwardly. The outward expansion produces venous blood outflow as a result of compression on the cortical veins. Inward expansion produces flow of CSF into the aqueduct as a result of compression of the lateral and third ventricles. B, In communicating hydrocephalus, the brain has already expanded outward during diastole, compressing the cortical veins. However, during systole, with arterial blood entering, the systolic expansion is directed inwards, resulting in a much greater ACSV in the aqueduct. C, In unshunted patients with iNPH, progressive ischemia and a reduction of arterial inflow results in a decreased “ventricular CSF pump” and then in a subsequent decrease of ACSV. D, In shunted patients, the shunt creates an alternative to ventricular CSF outflow, and in the patients with reversible iNPH, the shunt recreates both outward and inward brain expansion and the renormalization of ACSV.
In this study, we showed that in all patients with iNPH who underwent a VPS procedure with a properly functioning shunt system, the ACSV gradually decreases with time. The shunt surgery provides an immediate alternative path to the aqueductal CSF outflow, while allowing continuous ventricular CSF subtraction and a consequent increase in brain compliance. All of this re-enables the outward brain expansion during the arterial inflow, with a reduction of the ventricular squeezing and a consequent reduction of the ACSV (Fig 7D).
The reduction of ACSV values with time after the shunt surgery is greater in the clinically improved patient group compared with the patients who did not improve clinically. We hypothesize that this is due to the different preoperative mean ACSV values between the 2 groups and the difference in the time of symptom onset. Both of these suggest a different preshunt status of the brain. As previously hypothesized,(8) a short disease course associated with high ACSV values indicates a minor cerebral insult as shown by the absence of neurodegenerative phenomena such as atrophy and ischemia, which render the iNPH irreversible; and with the opening of an alternative pathway for flow, the patient shows clinical improvement.
A drop of ACSV values after shunt placement was observed in patients who developed an SDFC. SDFC, the most frequent complication of shunt surgery with a negligible influence on outcome,(13,14) is directly linked to system overdrainage.(15) In patients with overdrainage, the CSF preferentially transits through the path of the shunt versus the aqueduct. This results in a reduction of aqueductal CSF flow and a more rapid and pronounced reduction of ACSV compared with shunted patients with no overdrainage. There were no significant differences in ACSV reduction between the symptomatic patient who developed acute subdural hematoma and the patients who developed chronic subdural hematoma. Furthermore there were no differences in ACSV decline between the symptomatic and asymptomatic patients with SDFC.
The lack of progression of SDFC observed in these cases may demonstrate a balance established between the shunt system, the abdominal compartment, and the intracranial compartments, including the SDFC. In 2 patients who developed a hematoma who subsequently underwent surgical evacuation and shunt system closure, we observed recurrence of iNPH signs and increase in ACSV values to higher levels than the preshunt values 1 month after treatment. In this case, the closure of the valve was in a patient whose shunt dependency had further compromised the ability of CSF reabsorption at the arachnoid villi level, inducing a CSF ventricular accumulation greater than that before the shunt surgery. After we reopened the system, ACSV progressively decreased as is normally expected in shunted patients. The reason we did not encounter further overdrainage is a matter of speculation. One can theorize that the shunt system underwent internal fibrosis induced by CSF stagnation during the closure time period, and the consequent increase in resistance of the system caused this phenomenon.
An increasing ACSV after an initial decrease in shunted patients should raise a suspicion of a possible shunt system malfunction. Determining the functional state of a CSF shunt system may be challenging. Palpation of the shunt reservoir is a maneuver that can present uncertainties, is very subjective, and is dependent on the physician's experience. In cases of suspected system malfunction, puncture of the reservoir, injection of a radioisotope, or injection of contrast material into the shunt system represent invasive techniques to assess malfunction, with associated risks and complications. On the other hand, noninvasive tools such as plain x-ray studies, CT, and standard MR imaging can frequently present uncertainty as to whether a shunt is functioning or not.
In 1991, Drake et al(16) proposed MR phase imaging as a tool to determinate CSF shunt obstruction, by imaging the shunt over the skull, just distal to any reservoir. This method provides direct information on the presence or absence of flow in the lumen of the drainage system downstream from the reservoir with the patient in a supine position in the magnet. The method we use allows evaluation of how the system operates indirectly. A re-increase of ACSV values, associated with the rigidity of the reservoir during pumping, is indicative of obstruction at the distal end, whereas a re-increase of ACSV values in the absence of rigidity of the reservoir is indicative of malfunction of the system upstream of the valve. Investigation of a suspected shunt system malfunction, as proposed by Drake, which examines flow presence or absence in the drainage system within a given time, can produce false-positives in cases in which the measurement was taken in a given moment when the lack of flow was due to the efficiency of the system and not a shunt malfunction. An ACSV reduction after the shunt placement reflects the restoration of the outward brain expansion during the arterial inflow obtained over time. The persistence of decreased values after shunt surgery demonstrates the prolonged effectiveness of the CSF outflow alternative route and serves as an indication that the implanted system is operational.
A follow-up with a quantitative evaluation of the aqueductal CSF can detect a shunt system malfunction, even before the clinical symptoms reappear and the neurodegeneration process induced by iNPH resumes. It is not possible, on the basis of our data, to determine how long the brain of a patient who had improved after surgery can again tolerate a shunt malfunction before re-appearance of iNPH symptoms and progression of the neurodegeneration. However, as discussed above, 1 patient in our study who opted to not undergo shunt revision for 12 months did not improve clinically after the shunt was repaired.
The continuous increase of ACSV after shunt surgery may also indicate that the implanted system, even if working correctly, may not be an adequate representation of the ICP status of the brain. This was observed in 1 patient who underwent shunt surgery on the basis of a positive response to ELD, whose ACSV did not decrease after surgery. The patient improvement and ACSV decrease after the substitution of the medium with a low pressure valve indicate that the iNPH was still reversible, but the level of closure pressure of the system was not adequate for the CSF intracranial pressure. The introduction of programmable valves into clinical practice stems from the need to individualize the optimal closure pressure of the implanted system in each patient with iNPH. Optimization on the valve closure pressure value has already been demonstrated to increase the chance of success in shunted patients with iNPH.(17,18) In this regard, quantitative information offered by ACSV measurements might play an important role.
Although the purpose of this work was to analyze the usefulness of ACSV management in postshunt patients, this study contains elements that, when retrospectively analyzed, can affirm the utility of ACSV measurement in selected patients with iNPH who should undergo shunt surgery. Our results showed that iNPH shunt responders presented higher preshunt ACSV values and shorter onset times of symptoms with respect to nonresponders. As previously demonstrated, due to measuring ACSV many times during the progression of the disease in unshunted patients with iNPH, the ACSV has to be considered a “dynamic” parameter.(8) Indeed, during the disease progression, ACSV seems to increase between the onset of the symptoms and the following 18–20 months and then seems to plateau, followed in the next 18–20 months by a slight decline and finally a more precipitous drop in the next 12 months. As previously discussed, the increase in ACSV is linked to the progressive reduction of outward brain expansion, whereas a decrease in ACSV is associated with clinical deterioration, related to the neurodegenerative processes that reduce brain pulsatility and render the iNPH irreversible. Therefore, the ACSV-versus-time curve is characterized by an ascending phase in which the disease can still be considered reversible and a plateau phase and a descendant phase in which the disease becomes irreversible. Therefore, each preoperative ACSV value can be in either the reversible or irreversible phase of iNPH.(7) For this reason, the authors maintain that the ACSV, though not having a predictive value per se, could acquire it when coupled with the clinical onset parameters of each individual patient.
Conclusions
This study shows that ACSV may be a useful noninvasive tool for the preoperative selection of patients with iNPH and may be an effective postshunt management measurement. By analyzing the ACSV changes induced by a fixed shunt system, one can obtain clinically useful data for guiding the physician to regulate programmable valves better, which are now increasingly used in clinical practice, improving benefits of the shunt and preventing complications.
References
- 1. Bradley WG, Jr, Scalzo D, Queralt J, et al. Normal pressure hydrocephalus: evaluation with cerebrospinal fluid flow measurements at MR imaging. Radiology 1996; 198: 523– 29 [DOI] [PubMed] [Google Scholar]
- 2. Kim DS, Choi JU, Huh R, et al. Quantitative assessment of cerebrospinal fluid hydrodynamics using a phase-contrast cine MR image in hydrocephalus. Childs Nerv Syst 1999; 15: 461– 67 [DOI] [PubMed] [Google Scholar]
- 3. Mase M, Yamada K, Banno T, et al. Quantitative analysis of CSF flow dynamics using MRI in normal pressure hydrocephalus. Acta Neurochir Suppl 1998; 71: 350– 53 [DOI] [PubMed] [Google Scholar]
- 4. Dixon GR, Friedman JA, Luetmer PH, et al. Use of cerebrospinal fluid flow rates measured by phase-contrast MR to predict outcome of ventriculoperitoneal shunting for idiopathic normal-pressure hydrocephalus. Mayo Clin Proc 2002; 77: 509– 14 [DOI] [PubMed] [Google Scholar]
- 5. Bateman GA, Levi CR, Schofield P, et al. The pathophysiology of the aqueduct stroke volume in normal pressure hydrocephalus: can comorbidity with other forms of dementia be excluded? Neuroradiology 2005; 47: 741– 48 [DOI] [PubMed] [Google Scholar]
- 6. Kahlon B, Annertz M, Ståhlberg F, et al. Is aqueductal stroke volume, measured with cine phase-contrast magnetic resonance imaging scans, useful in predicting outcome of shunt surgery in suspected normal pressure hydrocephalus? Neurosurgery 2007; 60: 124– 30 [DOI] [PubMed] [Google Scholar]
- 7. Scollato A, Gallina P, Di Lorenzo N, et al. Comment on: Is aqueductal stroke volume, measured with cine phase-contrast magnetic resonance imaging scans useful in predicting outcome of shunt surgery in suspected normal pressure hydrocephalus? Neurosurgery 2008; 63: E1209, author reply E1209 [DOI] [PubMed] [Google Scholar]
- 8. Scollato A, Tenenbaum R, Bahl G, et al. Changes in aqueductal CSF stroke volume and progression of symptoms in patients with unshunted idiopathic normal pressure hydrocephalus. AJNR Am J Neuroradiol 2008; 29: 193– 98 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9. Adams RD, Fischer CM, Hakim S, et al. Symptomatic occult hydrocephalus with “normal” cerebrospinal fluid pressure: a treatable syndrome. N Engl J Med 1965; 273: 117– 26 [DOI] [PubMed] [Google Scholar]
- 10. Folstein MF, Folstein SE, McHugh PR. “Mini-mental state”: a practical method for grading the cognitive state of patients for the clinician. J Psychiatry Res 1975; 12: 189– 98 [DOI] [PubMed] [Google Scholar]
- 11. Raftopoulos C, Chaskis C, Delecluse F, et al. Morphological quantitative analysis of intracranial pressure waves in normal pressure hydrocephalus. Neurol Res 1992; 14: 389– 96 [DOI] [PubMed] [Google Scholar]
- 12. Bradley WG. MR prediction of shunt response in NPH: CSF morphology versus physiology. AJNR Am J Neuroradiol 1998; 19: 1285– 86 [PMC free article] [PubMed] [Google Scholar]
- 13. Boon AJ, Tans JT, Delwel EJ, et al. Dutch Normal-Pressure Hydrocephalus Study: randomized comparison of low- and medium-pressure shunts. J Neurosurg 1998; 88: 490– 95 [DOI] [PubMed] [Google Scholar]
- 14. Vanneste J, Augustijn P, Dirven C, et al. Shunting normal-pressure hydrocephalus: do the benefits outweigh the risks? A multicenter study and literature review. Neurology 1992; 42: 54– 59 [DOI] [PubMed] [Google Scholar]
- 15. Bergsneider M, Black PM, Klinge P, et al. Surgical management of idiopathic normal-pressure hydrocephalus. Neurosurgery 2005; 57 ( 3 suppl): S29– 39 [DOI] [PubMed] [Google Scholar]
- 16. Drake JM, Martin AJ, Henkleman RM. Determination of cerebrospinal fluid shunt obstruction with magnetic resonance phase imaging. J Neurosurg 1991; 75: 535– 40 [DOI] [PubMed] [Google Scholar]
- 17. Zemack G, Romner B. Adjustable valves in normal-pressure hydrocephalus: a retrospective study of 218 patients. Neurosurgery 2002; 51: 1392– 400 [PubMed] [Google Scholar]
- 18. Takeuchi T, Kasahara E, Iwasaky M, et al. Indication for shunting in patients with idiopathic normal pressure hydrocephalus presenting with dementia and brain atrophy (atypical idiopathic normal pressure hydrocephalus). Neurol Med Chir (Tokyo) 2000; 40: 38– 47 [DOI] [PubMed] [Google Scholar]