Abstract
Background
Left ventricular (LV) remodeling is the most common target organ damage in hypertension. Previously, our study found that plasma microRNA-29a (miR-29a) level was associated with the LV remodeling in hypertensive patients. However, the causal relationship between miR-29a and LV remodeling remains unknown. Thus, the aim of this study was to investigate the regulation mechanism of miR-29a in LV remodeling.
Methods & Results
Overexpression and knockdown miR-29a mice were generated by tail-intravenous injection of miR-29a-mimic and inhibitor lentivirus for one week respectively. Then the mice were subjected to angiotensin-II (AngII) induced LV remodeling by subcutaneous AngII capsule osmotic pumping into AngII for four weeks. AngII-induced LV remodeling mice as the model group (n = 9). Age-matched male SPF C57/BL6J mice (6–8 weeks old) were treated with the pumping of saline as a vehicle (n = 6). In vivo, overexpression miR-29a ameliorated AngII-induced LV remodeling, while knockdown miR-29a deteriorated LV remodeling. Simultaneously, we observed that overexpression miR-29a mice inhibited but knockdown miR-29a mice increased cardiac cross-sectional area, indicating that miR-29a has an antagonistic effect on cardiac hypertrophy. Further studies found that overexpression miR-29a inhibited the content of the LV collagen including collagen I and III. Moreover, the expression of transforming growth factor-β (TGF-β) and phosphorylated SMAD2/3 decreased with the down-regulation of collagen I and III in overexpression miR-29a mice.
Conclusions
Our finding indicates that overexpression miR-29a attenuates LV remodeling by inhibiting collagen deposition, TGF-β, and phosphorylated SMAD2/3 expression. Thus, intervention miR-29a may be a therapeutic target for attenuating LV remodeling.
Keywords: Left ventricular remodeling, MicroRNA-29a, SMAD2/3, Transforming growth factor-β
1. Introduction
Left ventricular (LV) remodeling is the most common target organ damage in hypertension, which is also a critical cause of cardiovascular disease such as coronary heart disease, heart failure, arrhythmia, atrial fibrillation.[1] A previous study found that the prevalence of LV hypertrophy (LVH) is 42.7% in hypertensive patients among Han Chinese.[2] Hypertension is the main cause of LV remodeling, effective control blood pressure cannot completely prevent the occurrence of LV remodeling, suggesting other possible causes. Recent studies have found that epigenetics involved in the occurrence and development of ventricular remodeling.[3],[4]
MicroRNAs (miRNAs) are endogenous non-coding small RNAs that regulate gene expression. Accumulating studies have found that miRNAs play important roles in regulating cardiovascular disease.[5]–[11] Several miRNAs are shown to be involved in cardiac remodeling, namely: miR-1, miR-133, miR-26, miR-9, and miR-98 inhibit cardiac remodeling, and miR-143, miR-199a, miR-208, miR-23, miR-499, miR-21 contribute to cardiac remodeling.[12]–[17] However, the molecular mechanisms of individual miRNAs remain unknown. A recent study found that microRNA-29a (miR-29a) prevented myocardial fibrosis after acute myocardial infarction by down-regulating the expression of extracellular matrix collagen.[18],[19] In addition, the up-regulation of miR-29a was associated with cardiac hypertrophy and fibrosis in patients with hypertrophic cardiomyopathy.[20] Our previous study also found circulating miR-29a is associated with the LV remodeling in hypertensive patients.[21] However, the regulatory mechanism of miR-29a in the remodeling process is still unclear.
In order to address this issue, we used overexpression and knockdown miR-29a mice model to investigate the regulation mechanism of miR-29a in LV remodeling.
2. Materials and methods
2.1. Animals
All the studies were performed in accordance with the relevant guidelines and regulations,[22] and approved by the Ethics Committee for Animal Study of Chinese PLA General Hospital. Healthy male SPF C57/BL6J mice (6–8 weeks old) were randomly assigned to four groups: (1) control group (pumping of saline, n = 6); (2) model group [pumping of angiotensin-II (AngII), n = 9]; (3) miR-29a-mimic group (n = 9) with injection of miR-29a-mimic lentivirus; and (4) miR-29a-knockdown (miR-29a-KD) group (n = 9) with injection of miR-29a-inhibitor lentivirus.
2.2. Generation of recombinant lentivirus
The short hairpin RNAs (shRNAs) containing mmu-mir-29a (miR-29a-mimic, sequence: 5′-ACCCCTTAGAGGATGACTGATTTCTTTTGGTGTTCAGAGTCAATAGAATTTTCTAGCACCATCTGAAATCGGTTATAATGATTGGGGA-3′) or a specific inhibitor of mmu-mir-29a (miR-29a-KD, sequence: 5′-TAGCACCAGACTAAATCGGTTAAAGTAGCACCACAGTAAATCGGTTAGGTTAGCACCACACAAAATCGGTTACAATAGCACCACAGAAAATCGGTTAGTCTAGCACCAGTCAAAATCGGTTAAGGTAGCACCACTGAAAATCGGTTATACTAGCACCAGTGTAAATCGGTTATGATAGCACCAGTGAAAATC GGTTA-3′) were cloned into the lentiviral vector pHS-AMR. shRNAs containing an unrelated sequence were used as negative controls, sequence: 5′-TAATTGTCAAATCAGAGTGCTTGTTTTGGCCACTGACTGACAAGCACTATTTGACAATTA-3′. Viral production was performed by using EpFect™ Transfection Reagent for the transfection of human embryonic kidney (HEK)-293 cells. Virus was concentrated by ultracentrifugation. Recipient cells were infected with 106 viral transducing units per mL plus polybrene and stably transduced cells were selected in puromycin for ten days or sorted by Fluorescence Activated Cell Sorter (FACS).
2.3. Construct model
Capsule osmotic pump was purchased from AlZET Corporation (MODEL 2004), and AngII was purchased from Sigma Corporation of the United States. The animals underwent tail-intravenously injection of recombinant lentivirus (109 titer/mL, 0.1 mL per mouse). Overexpression (n = 9) and knockdown (n = 9) miR-29a mice were generated by tail-intravenous injection of miR-29a-mimic and inhibitor lentivirus one week respectively. The control group and the model group were injected with normal saline intravenously. Besides the control group, the mice were subjected to AngII by subcutaneous AngII capsule osmotic pump (1400 ng/kg·min−1) for four weeks to induce LV remodeling.
2.4. Ultrasound assessment
At the end of the experiment, the mice were weighed and anesthetized intraperitoneally with 2.5% Avertin (0.018 mL/g). Transthoracic echocardiography was performed in all mice (Vevo 2100 imagine system, FuJIFILM Visualsonic Inc., Toronto, Canada) with a high transducer frequency probe (VisualSonics MS400, 18 MHz to 38 MHz). The LV dimensions, LV anterior wall (LVAW) thicknesses, and LV posterior wall (LVPW) thickness were averaged from more than three cardiac cycles at end-diastole and end-systole by the M-mode measurements.
2.5. Tissue preparation
The mice thoracic cavity were immediately opened. The hearts were removed and perfused with saline under physiological pressure until the liver turned white. The cardiac tissue slices (3 mm thick) were cut in the mid-ventricle, fixed in 4% paraformaldehyde for 24 hours, flushed by phosphate buffered saline (PBS), dehydrated in ascending series of ethanol and embedded with paraffin. The remaining LV slices were quick-frozen in liquid nitrogen and stored under –80 °C.
2.6. Histological analysis
2.6.1. Masson and Sirius red staining
Paraffin-embedded tissue sections were stained with Masson and Sirius red staining separately. Masson staining was performed as follows: after being dehydrated in gradient ethanol, the sections were immersed into the Alcian Blue staining solution, acid alcohol, ponceau, and phosphomolybdic acid solution in sequence to stain, and the change of cardiac structure, myocardial cells and cardiac fibroblasts were observed with a microscope (200 ×). Sirius red staining was performed by incubating sections in Sirius red solution, washing in acidified water and then dehydrating in ethanol to observe the expression of the LV myocardial collagen. The cardiac cross-sectional area (mm2) and the LV collagen volume were measured through IPP 6.0.
2.6.2. Immunofluorescent detection
Paraffin-embedded sections were dewaxed, enclosed for 12 min in 3% H2O2 and then flushed three times in PBS, repaired with microwave energy combined with Ethylenediaminetetraacetic Acid and flushed three times in PBS again. Subsequently, the sections were enclosed in fetal bovine serum protein solution for one hour at room temperature, after that, dropwise added first antibody of collagen I and III (1:50, Abcam and Proteintech), incubated for overnight in 4 °C. The next day, the sections were flushed three times in PBS firstly, and then dropwise added the goat secondary antibody labeled with fluorescence, incubated for one hour in 37 °C and finally stored in 4 °C without light. The expression of myocardial tissue collagen I and III was observed with a fluorescence microscope (400 ×) and the collagen area was measured by partial photography and IPP 6.0 software.
2.6.3. Western blotting analysis
The frozen LV tissue of the mice was added the tissue Lysis Buffer, then ground and centrifuged to get supernatant. The isovolumetric 2 × gel-loading buffer was added to the supernatant after protein excretion. Subsequently, the supernatant boiled for 10 min. The proteins were loaded onto 8% Sodium Dodecyl Sulfate polyacrylamide gel for separation, and transferred to nitrocellulose membrane, then marked the target protein band after the ponceau staining. The first antibody was enclosed for one min in 5% skimmed milk and then incubated for one min at room temperature, incubated overnight at 4 °C. The antibodies were then stripped by washing the membrane 3 × 10 min with 0.2% TPBS (Triton X-100/PBS). The secondary antibody was incubated for one hour at room temperature. The membrane was then rinsed 4 × 10 min in 0.2% TPBS. The primary proteins used were the first antibody of transforming growth factor-β (TGF-β) (Cell Signaling Technology, 1:1000), p-SMAD2/3 (Cell Signaling Technology, 1:1000), Collagen-I (Abcam, 1:500), Collagen-III (Proteintech, 1:500), β-actin (Abcam, 1:4000), and the secondary antibody of rabbit anti-goat IgG (1:2000), goat anti-mouse IgG (1:5000), goat anti-rabbit IgG (1:2000).
2.7. Statistical analysis
The experimental data analysis was performed by using SPSS 20.0. All values were presented as mean ± S.E. Statistical differences among the groups were performed by One-Way ANOVA. A P-value < 0.05 was considered as statistically significant.
3. Results
3.1. Overexpression of miR-29a inhibits LVH in vivo
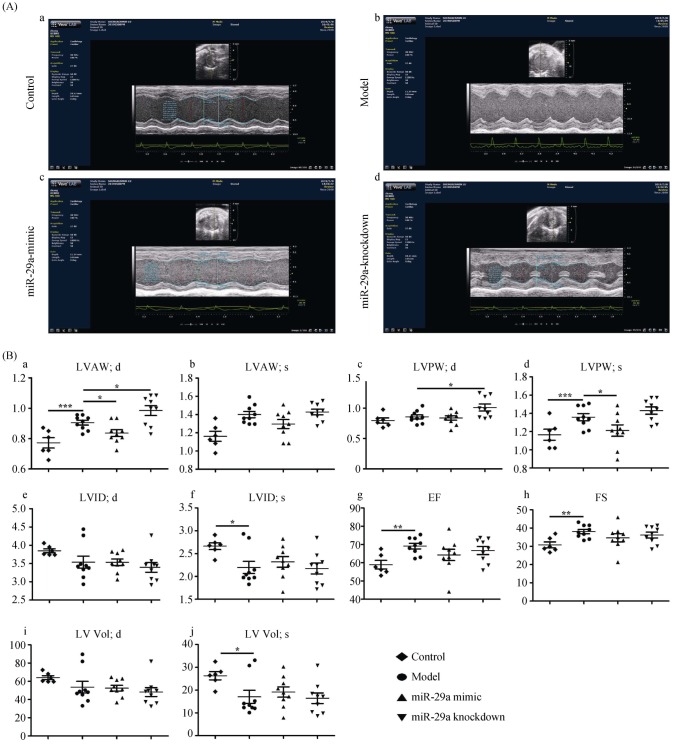
To investigate the effect of miR-29a on LVH, we constructed overexpression and knockdown miR-29a mice. We found that ultrasound parameters of LVH were higher in the model group than those in the control group, indicating that the construction of the model was successful. And, compared with the model group, miR-29a overexpression obviously reduced the thickness of the LVAW at the end-diastolic and the LVPW at the end-systolic (as shown in Figure 1Ab–c and Figure 1Ba–d). Conversely, the miR-29a-KD increased the thickness of LVAW and LVPW. These results indicated that overexpression of miR-29a ameliorated LV remodeling induced by AngII, while knockdown miR-29a deteriorated left ventricular remodeling in vivo.
Figure 1. Overexpression of miR-29a inhibits left ventricular remodeling by using ultrasound assessment.
(A): Representative M-mode echocardiography images; and (B): echocardiography parameters were detected in mice among four groups (a. LVAW; d. the thickness of the LVAW at the end-diastolic; b. LVAW; s. the thickness of the LVAW at the end-systolic; c. LVPW; d. the thickness of the LVPW at the end-diastolic; d. LVPW; s. the thickness of the LVPW at the end-systolic; e. LVID; d. the left ventricular interior diameter at the end-diastolic; f. LVID; s. the LVID at the end-systolic; g. EF%: LV ejection fraction; h. FS%: LV fractional shortening; i. LV Vol; d. LV volume at the end-diastolic; and j. LV Vol; s. LV volume at the end-systolic.). Data are indicated as the means ± S.E. *P < 0.05, **P < 0.01, ***P < 0.001. EF: ejection fraction; FS: fractional shortening; LV: left ventricular; LVAW: left ventricular anterior wall; LVID: left ventricular inner dimension; LVPW: left ventricular posterior wall.
3.2. miR-29a inhibits fibroblast proliferation
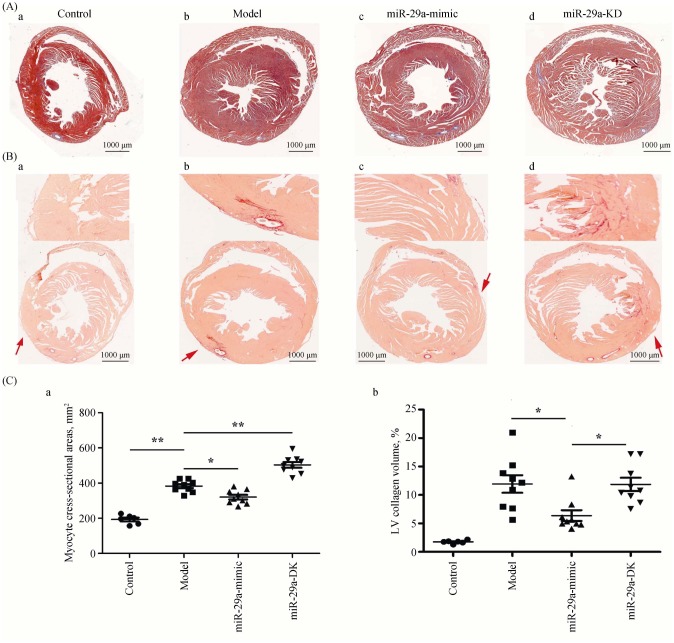
We observed the histological changes in the heart. Masson staining showed that the cardiac cross-sectional area was significantly larger in the model group than that in the control group (Figure 2A). The cardiac cross-sectional area was reduced in miR-29a-mimic group but increased in the miR-29a-KD group (Figure 2Ca). These results indicated that miR-29a attenuated cardiac hypertrophy. The ratio of the collagen area in the LV area by Sirius Red Staining (Collagen-specific Dyeing) showed that compared with the model group, miR-29a overexpression obviously inhibited the content of collagen, suggesting that it inhibited fibroblast proliferation. However, we did not find that miR-29a-KD group had a significant effect on the ratio of the collagen area in the LV area (Figure 2Cb). These results indicated miR-29a improves the myocardial remodeling by inhibiting fibroblast proliferation.
Figure 2. Overexpression of miR-29a inhibits fibroblast proliferation.
(A): Color images of the cardiac cross-sectional under the microscope (200 ×) after Masson staining to observe the ventricular wall thickness; (B): color images of the cardiac cross-sectional after Sirius red Staining, in which four arrows show the expression of the LV myocardial collagen, and the collagen and non-collagen components is red and orange stained respectively; and (C): scatter plots of the relevant parameters detected after staining (a: the area of cardiac myocyte cross-section; and b: the LV collagen volume.). Scale bar is 1000 µm in Figure A & B. Data are indicated as means ± S.E. *P < 0.05, **P < 0.01. LV: left ventricular.
3.3. miR-29a suppresses the secretion of collagen I and III
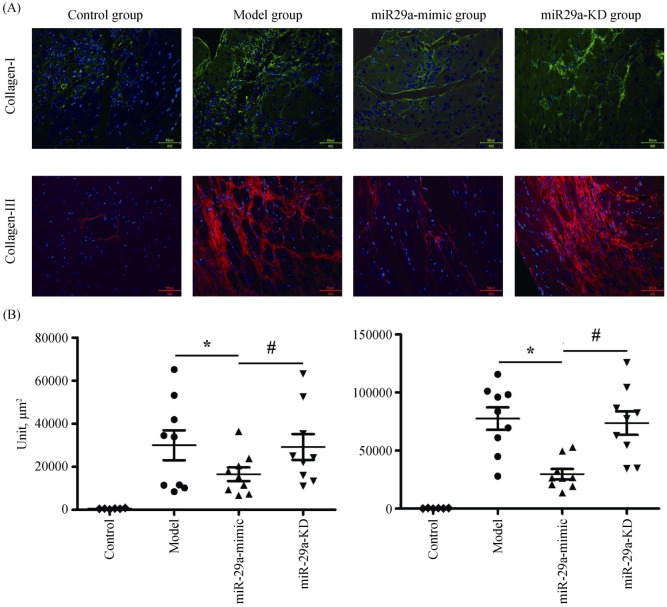
Immunofluorescent detection showed that compared with the model group, miR-29a overexpression significantly inhibited the levels of collagen I and III (Figure 3A & B); However, the levels of collagen I and III were no significant difference in the miR-29a-KD group. The result is consistent with the effect of miR-29a-mimic and miR-29a-KD on the ratio of the collagen area in the LV area (Figure 2), suggesting that miR-29a overexpression can dramatically inhibit secreting collagen I and III.
Figure 3. Inhibition of miR-29a on cardiac collagen.
(A): The image showing the expression of myocardial tissue collagen I and III under the fluorescence microscope (400 ×); and (B): the levels of collagen I and III. Data are indicated as the means ± S.E. *P < 0.05, miR-29a-mimic group vs. model group; #P < 0.05, miR-29a-KD group vs. miR-29a-mimic group. miR-29a-KD: miR-29a-knockdown.
3.4. miR-29a decreases the expression of TGF-β and SMAD2/3
The TGF-β and SMAD2/3 pathways are classic ways for collagen activation and secretion of myocardial fibroblasts.
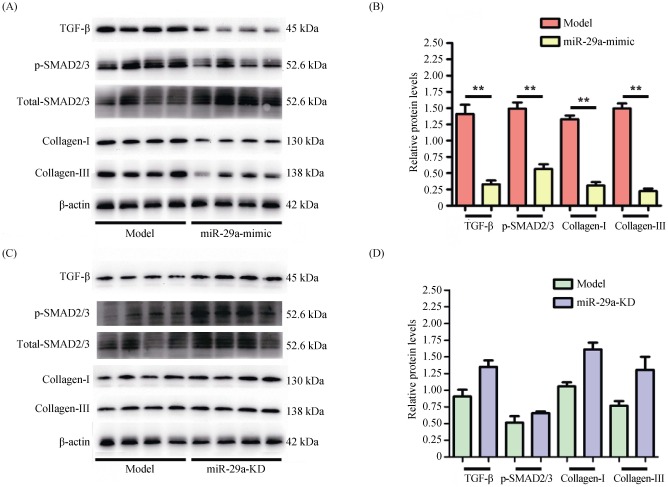
Our results suggested miR-29a overexpression protected from LV remodeling (Figures 1–3). Therefore, we further investigated the cardiac expression of TGF-β and SMAD2/3 in the model group and miR-29a-mimic group by using Western blot. Compared with the model group, miR-29a overexpression significantly inhibited the expression of TGF-β and phosphorylated SMAD2/3 (activated form) in the heart, accompanied by a significant decrease in the expressing quantity of collagen I and III (Figure 4A & B). However, there was no obvious difference in the cardiac expression of TGF-β and SMAD2/3 between miR-29a-KD group and model group (Figure 4C & D). These results indicated that overexpression miR-29a can reduce cardiomyocyte proliferation to ameliorated LV remodeling by inhibiting the content of LV collagen including collagen I and III, accompanied by decreasing TGF-β and phosphorylated SMAD2/3.
Figure 4. Inhibition of miR-29a on the expression of Collagen I, Collagen III, TGF-β and SMAD2/3.
The expression of Collagen I, Collagen III, TGF-β and p-SMAD2/3 were detected using Western blot and the mimic group was low as compared with the model group (A & B), while the knockdown group was no obvious difference (C & D). Data are indicated as means ± S.E. **P < 0.01. TGF-β: transforming growth factor-β.
4. Discussion
This is the first vivo study to investigate the mechanism of miR-29a in regulating LV remodeling. We found that cardiac overexpression of miR-29a attenuated cardiac remodeling, accompanied by collagen I, collagen III, TGF-β and phosphorylated SMAD2/3 expression decreasing.
Cardiac remodeling is a chronic pathological process accompanied by cardiac hypertrophy and myocardial fibrosis. Previous studies suggested that miR-29 protected the heart from pathological hypertrophy and myocardial fibrosis.[19],[23] In patients with hypertrophic cardiomyopathy, circulating miR-29a was identified to be a biomarker for both hypertrophy and fibrosis.[20] The consistent results have also shown that miR-29a is positively correlated with LVH in hypertension patients.[24] In addition, experimental cardiac remodeling and reverse remodeling animal model confirmed that miR-29a involved in the cardiac reverse remodeling process.[25] Our study adds new evidence for the protective role of miR-29a in overexpression miR-29a mice and knockdown miR-29a mice subjected to AngII induced LV remodeling. We observed that overexpression miR-29a inhibited but knockdown miR-29a aggravated AngII-induced LV remodeling in vivo and pathological changes of cardiac remodeling (including cardiac cross-sectional area, and cardiac fibrosis), which also indicated the protective role of miR-29a in cardiac remodeling.
Accumulation of extracellular matrix molecules is one of the main pathological features of LV remodeling.[26],[27] Previous research showed that miR-29a was produced mostly by fibroblasts, and its family members regulated fibrosis by modulating the expression of collagen and other extracellular matrix genes, such as TGF-β, vascular endothelial growth factor-A, fibrillins, elastin, and myeloid cell leukemia 1 gene.[18],[28]–[30] In our study, miR-29a overexpression obviously decreased the content of LV collagen including collagen I and III, which indicated that it inhibited fibroblast proliferation. This finding is consistent with the results reported by Melo, et al.,[19] which shows swimming training-induced up-regulation of the expression of miR-29 is related to repression of collagen (collagen I and III) gene expression in myocardial-infarcted mice. In addition, by using bioinformatics websites (Target Scan Human), we also predicted that collagen I and III might be a target of miR-29a, which may participate in the regulation of cardiac hypertrophy. However, we did not find significant up-regulation of collagen I and III expression in knockdown miR-29a mice. Several reasons might explain the conflicting results. Firstly, exogenous delivery of the miR-29a inhibitor might not completely knock down cardiac-specific miR-29a expression. Secondly, miR-29a may not be the sole determinant of collagen expression, and other factors may also involve in it.
Another finding of our study was that the expression of TGF-β and phosphorylated SMAD2/3 was decreased accompanied by the down-regulation of collagen I and III in miR-29a-mimic group, whereas knockdown of miR-29a increased the expression. The results indicated that TGF-β and SMAD2/3 may involve in the regulation mechanism of miR-29a in LV remodeling. It has been well known that TGF-β is identified as a regulator of cardiac fibrosis,[31],[32] which can promote collagen protein synthesis. TGF-β and SMAD signaling pathway was considered to be one of the most classic ways of mediating fibroblasts activation and collagen secretion.[33] Moreover, other studies indicated that TGF-β repressed miR-29 expression.[18],[34] However, overexpression miR-29a can also inhibit TGF-β and SMAD2/3 expression. These data demonstrate that TGF-β and SMAD may be upstream signaling pathway molecules of miR-29a target gene collagen in regulating cardiac hypertrophy.
In the present study, we found that miR-29a attenuated cardiac remodeling accompanied by lowing collagen, TGF-β and phosphorylated SMAD2/3 expression. However, the signaling pathway of miR-29a targeted collagen in the regulation of LV remodeling could not be fully explained. Further studies are needed to reveal the complete mechanism of miR-29a expression and its association with the signaling pathway of target gene collagen.
In summary, our study showed that overexpression miR-29a contributed to attenuate AngII-induced cardiac remodeling by down-regulating collagen, TGF-β and phosphorylated SMAD2/3 expression. Thus, miR-29a may be a potential therapeutic target for inhibiting hypertensive LV remodeling.
Acknowledgments
This study was supported by the National Natural Science Foundation of China (No. 81570383). All authors had no conflicts of interest to disclose.
References
- 1.Chaturvedi S. The seventh report of the joint national committee on prevention, detection, evaluation, and treatment of high blood pressure (JNC 7): is it really practical? Natl Med J India. 2004;4:227. [PubMed] [Google Scholar]
- 2.Wang SX, Xue H, Zou YB, et al. Prevalence and risk factors for left ventricular hypertrophy and left ventricular geometric abnormality in the patients with hypertension among Han Chinese. Chin Med J (Engl) 2012;125:21–26. [PubMed] [Google Scholar]
- 3.Deschepper CF, Boutin-Ganache I, Zahabi A, et al. In search of cardiovascular candidate genes: interactions between phenotypes and genotypes. Hypertension. 2002;39:332–336. doi: 10.1161/hy0202.102787. [DOI] [PubMed] [Google Scholar]
- 4.Xue H, Wang S, Wang H, et al. Atrial natriuretic peptide gene promoter polymorphism is associated with left ventricular hypertrophy in hypertension. Clin Sci (Lond) 2008;114:131–137. doi: 10.1042/CS20070109. [DOI] [PubMed] [Google Scholar]
- 5.Cheng Y, Ji R, Yue J, et al. MicroRNAs are aberrantly expressed in hypertrophic heart: do they play a role in cardiac hypertrophy? Am J Pathol. 2007;170:1831–1840. doi: 10.2353/ajpath.2007.061170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Sayed D, Hong C, Chen IY, et al. MicroRNAs play an essential role in the development of cardiac hypertrophy. Circ Res. 2007;100:416–424. doi: 10.1161/01.RES.0000257913.42552.23. [DOI] [PubMed] [Google Scholar]
- 7.Tatsuguchi M, Seok HY, Callis TE, et al. Expression of microRNAs is dynamically regulated during cardiomyocyte hypertrophy. J Mol Cell Cardiol. 2007;42:1137–1141. doi: 10.1016/j.yjmcc.2007.04.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Thum T, Galuppo P, Wolf C, et al. MicroRNAs in the human heart: a clue to fetal gene reprogramming in heart failure. Circulation. 2007;116:258–267. doi: 10.1161/CIRCULATIONAHA.107.687947. [DOI] [PubMed] [Google Scholar]
- 9.van Rooij E, Olson EN. MicroRNAs: powerful new regulators of heart disease and provocative therapeutic targets. J Clin Invest. 2007;117:2369–2376. doi: 10.1172/JCI33099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.van Rooij E, Sutherland LB, Liu N, et al. A signature pattern of stress-responsive microRNAs that can evoke cardiac hypertrophy and heart failure. Proc Natl Acad Sci U S A. 2006;103:18255–18260. doi: 10.1073/pnas.0608791103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Orenes-Piñero E, Montoro-García S, Patel JV, et al. Role of microRNAs in cardiac remodelling: new insights and future perspectives. Int J Cardiol. 2013;167:1651–1659. doi: 10.1016/j.ijcard.2012.09.120. [DOI] [PubMed] [Google Scholar]
- 12.Da Costa Martins PA, De Windt LJ. MicroRNAs in control of cardiac hypertrophy. Cardiovasc Res. 2012;93:563–572. doi: 10.1093/cvr/cvs013. [DOI] [PubMed] [Google Scholar]
- 13.Carè A, Catalucci D, Felicetti F, et al. MicroRNA-133 controls cardiac hypertrophy. Nat Med. 2007;13:613–618. doi: 10.1038/nm1582. [DOI] [PubMed] [Google Scholar]
- 14.Li Q, Song XW, Zou J, et al. Attenuation of microRNA-1 derepresses the cytoskeleton regulatory protein twinfilin-1 to provoke cardiac hypertrophy. J Cell Sci. 2010;123:2444–2452. doi: 10.1242/jcs.067165. [DOI] [PubMed] [Google Scholar]
- 15.Ooi JY, Bernardo BC, Mcmullen JR. The therapeutic potential of miRNAs regulated in settings of physiological cardiac hypertrophy. Future Med Chem. 2014;6:205–222. doi: 10.4155/fmc.13.196. [DOI] [PubMed] [Google Scholar]
- 16.Sayed D, Hong C, Chen IY, et al. MicroRNAs play an essential role in the development of cardiac hypertrophy. Circ Res. 2007;100:416–424. doi: 10.1161/01.RES.0000257913.42552.23. [DOI] [PubMed] [Google Scholar]
- 17.Song XW, Li Q, Lin L, et al. MicroRNAs are dynamically regulated in hypertrophic hearts, and miR-199a is essential for the maintenance of cell size in cardiomyocytes. J Cell Physiol. 2010;225:437–443. doi: 10.1002/jcp.22217. [DOI] [PubMed] [Google Scholar]
- 18.van Rooij E, Sutherland LB, Thatcher JE, et al. Dysregulation of microRNAs after myocardial infarction reveals a role of miR-29 in cardiac fibrosis. Proc Natl Acad Sci U S A. 2008;105:13027–13032. doi: 10.1073/pnas.0805038105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Melo SF, Fernandes T, Baraúna VG, et al. Expression of microrna-29 and collagen in cardiac muscle after swimming training in myocardial-infarcted rats. Cell Physiol Biochem. 2014;33:657–669. doi: 10.1159/000358642. [DOI] [PubMed] [Google Scholar]
- 20.Roncarati R, Viviani Anselmi C, Losi MA, et al. Circulating miR-29a, among other up-regulated micrornas, is the only biomarker for both hypertrophy and fibrosis in patients with hypertrophic cardiomyopathy. J Am Coll Cardiol. 2014;63:920–927. doi: 10.1016/j.jacc.2013.09.041. [DOI] [PubMed] [Google Scholar]
- 21.Yao SY, Liu J, Xue H, et al. [Association between plasma microRNA-29a and left ventricular hypertrophy in patients with hypertension] Zhonghua Xin Xue Guan Bing Za Zhi. 2019;47:215–220. doi: 10.3760/cma.j.issn.0253-3758.2019.03.007. [In Chinese] [DOI] [PubMed] [Google Scholar]
- 22.Su M, Wang J, Wang C, et al. MicroRNA-221 inhibits autophagy and promotes heart failure by modulating the p27/CDK2/mTOR axis. Cell Death Differ. 2015;22:986–999. doi: 10.1038/cdd.2014.187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Soci UP, Fernandes T, Hashimoto NY, et al. MicroRNAs 29 are involved in the improvement of ventricular compliance promoted by aerobic exercise training in rats. Physiol Genomics. 2011;43:665–673. doi: 10.1152/physiolgenomics.00145.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Huang Y, Tang S, Huang C, et al. Circulating miRNA29 family expression levels in patients with essential hypertension as potential markers for left ventricular hypertrophy. Clin Exp Hypertens. 2017;39:119–125. doi: 10.1080/10641963.2016.1226889. [DOI] [PubMed] [Google Scholar]
- 25.Wang J, Xu R, Lin F, et al. MicroRNA: novel regulators involved in the remodeling and reverse remodeling of the heart. Cardiology. 2009;113:81–88. doi: 10.1159/000172616. [DOI] [PubMed] [Google Scholar]
- 26.Fomovsky GM, Thomopoulos S, Holmes JW, et al. Contribution of extracellular matrix to the mechanical properties of the heart. J Mol Cell Cardiol. 2010;48:490–496. doi: 10.1016/j.yjmcc.2009.08.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Li L, Zhao Q, Kong W. Extracellular matrix remodeling and cardiac fibrosis. Matrix Biol. 2018;68–69:490–506. doi: 10.1016/j.matbio.2018.01.013. [DOI] [PubMed] [Google Scholar]
- 28.Boon RA, Seeger T, Heydt S, et al. MicroRNA-29 in aortic dilation: implications for aneurysm formation. Circ Res. 2011;109:1115–1119. doi: 10.1161/CIRCRESAHA.111.255737. [DOI] [PubMed] [Google Scholar]
- 29.Zhang P, Huang A, Ferruzzi J, et al. Inhibition of microRNA-29 enhances elastin levels in cells haploinsufficient for elastin and in bioengineered vessels--brief report. Arterioscler Thromb Vasc Biol. 2012;32:756–759. doi: 10.1161/ATVBAHA.111.238113. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Kriegel AJ, Liu Y, Fang Y, et al. The miR-29 family: genomics, cell biology, and relevance to renal and cardiovascular injury. Physiol Genomics. 2012;44:237–244. doi: 10.1152/physiolgenomics.00141.2011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Wong CKS, Falkenham A, Myers T, et al. Connective tissue growth factor expression after angiotensin II exposure is dependent on transforming growth factor-β signaling via the canonical Smad-dependent pathway in hypertensive induced myocardial fibrosis. J Renin Angiotensin Aldosterone Syst. 2018;19:1470320318759358. doi: 10.1177/1470320318759358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Border WA, Noble NA. Transforming growth factor beta in tissue fibrosis. N Engl J Med. 1994;331:1286–1292. doi: 10.1056/NEJM199411103311907. [DOI] [PubMed] [Google Scholar]
- 33.Schmierer B, Hill CS. TGFbeta-SMAD signal transduction: molecular specificity and functional flexibility. Nat Rev Mol Cell Biol. 2007;8:970–982. doi: 10.1038/nrm2297. [DOI] [PubMed] [Google Scholar]
- 34.Wang G, Kwan BC, Lai F, et al. Urinary miR-21, miR-29, and miR-93: novel biomarkers of fibrosis. Am J Nephrol. 2012;36:412–418. doi: 10.1159/000343452. [DOI] [PubMed] [Google Scholar]