Abstract
Mosquitoes are a serious threat to the society, acting as vector to several dreadful diseases. Mosquito management programes profoundly depend on the routine of chemical insecticides that subsequently lead to the expansion of resistance midst the vectors, along with other problems such as environmental pollution, bio magnification, and adversely affecting the quality of public and animal health, worldwide. The worldwide risk of insect vector transmitted diseases, with their associated illness and mortality, emphasizes the need for effective mosquitocides. Hence there is an immediate necessity to develop new eco-friendly pesticides. As a result, numerous investigators have worked on the development of eco-friendly effective mosquitocidal compounds of plant origin. These products have a cumulative advantage of being cost-effective, environmentally benign, biodegradable, and safe to non-target organisms. This review aims at describing the current state of research on behavioral, physiological, and biochemical effects of plant derived compounds with larvicidal effects on mosquitoes. The mode of physiological and biochemical action of known compounds derived from various plant families as well as the potential of plant secondary metabolites, plant extracts, and also the essential oils (EO), as mosquitocidal agents are discussed. This review clearly indicates that the application of vegetal-based compounds as mosquito control proxies can serve as alternative biocontrol methods in mosquito management programes.
Keywords: biopesticide, vector, secondary metabolites, phytochemical, physiology, enzyme, toxicity
Introduction
Vector borne diseases account for more than seven million deaths annually (World Health Organization [WHO], 2017), among which mosquito borne diseases are the most threatening due to their wide spread occurrence, consequently featuring a higher frequency of disease transmission (Lounibos, 2002; Tyagi et al., 2015). Among different mosquito families, Culicidae is a large family (3,300 Service species-41 genera) comprising Toxorhynchitinae, Anophelinae (anophelines), and also Culicinae (culicines) sub-families (Service, 1996; Senthil-Nathan et al., 2005b). Among the 31 genera, Anopheles, Culex, and Aedes are the most detrimental. Anopheles species, are carriers of major life-threatening diseases (malaria and filariasis-transmitting agents, such as Wuchereria bancrofti, Brugia malayi, and Brugia timori) and also of a few arboviruses (Kalaivani et al., 2012; Benelli et al., 2018; Thanigaivel et al., 2019; Vasantha-Srinivasan et al., 2019).
The discovery of DDT’s insecticidal properties in late 1930s/beginning of 1940s and the following progress of organochlorine invention and organophosphate insecticides concealed biological pesticide merchandise-research since the responses to mosquito regulation were supposed to have remained established (Shaalan et al., 2005; Senthil-Nathan et al., 2006a, b). The ranges of many of the mosquito species were not limited and keep expanding, thereby up surging the rates of disease incidence. Until recently, the use of several of the earlier synthetic-insecticides, such as permethrin and malathion, along with other organophosphates in vector control programes has been partial. This is due to absence of unique-insecticides, expense of synthetic-insecticides, apprehension for ecological sustainability, damaging influence on human health, besides further non-target populations, their persistent nature, greater amount of “biological magnification” through ecosystem and also the development of insecticide resistance (Ghosh et al., 2012). The emergence of DDT resistance in Aedes species (Ae. tritaeniorhynchus and Ae. sollicitans) lead to numerous drawbacks in mosquito control programs (Brown, 1986). Several categories of Mosquitocides are being implemented in malaria control programs (BHC, organophosphorus, carbamate, and pyrethroid). The ability of mosquitoes to evade the insecticidal action of these synthetic compounds are attributed to the increase in the rate of synthesis of detoxifying enzymes such as monoxygenases (MFOs), glutathione-S-transferases (GST) and carboxyl-cholinesterase (CCE). MFOs are often associated with metabolic resistance to pyrethroids, such as permethrin, while GSTs are usually associated with organochloride resistance such as DDT. Resistance to pyrethroids, organophosphates and carbamates, such as bendiocarb are incurred by the magnification of CCE activity (Hemingway and Ranson, 2000). Added insecticides, benzylphenyl urea and the larvicide, Bacillus thuringiensis israelensis (Bti), have partial use against mosquitoes. Unpredicted natural or anthropogenic associated ecological variations that modify the original habitats severely affect the vector biology thereby positively influencing their existence and disease incidence, thus constraining the frame-work of mosquito control strategies.
Biological Management of Mosquitoes
Several phytochemicals from several plant families are identified with larvicidal activities against different mosquito species (Table 1). Plant extracts with their augmented phytochemical elements have a recognized potential as a substitute to conventional mosquito control agents (Sukumar et al., 1991; Tripathi et al., 2009; Tehri and Singh, 2015). The main strategy for mosquito control deals with the restriction of the vector population. As a promising biocontrol agent, the compounds from the plants of the family Meliaceae such as neem Azadirachta indica A. Juss (Senthil-Nathan et al., 2005b; Senthil-Nathan, 2013), Indian white cedar, Dysoxylum malabaricum Bedd. (Senthil-Nathan et al., 2006a), D. beddomei and chinaberry tree, Melia azedarach L. (Senthil-Nathan et al., 2006b) were effective against An. stephensi (Senthil-Nathan et al., 2008). “Secondary metabolites” from Eucalyptus tereticornis Sm. (forest redgum, Myrtaceae) exhibited effective mosquitocidal activities against An. stephensi as reported by Senthil-Nathan (2007). Also, the crude metabolic extracts of Acanthospermum hispidum leaves were active against An. stephensi, Ae. Aegypti, as well as Cx. quinquefasciatus as reported by Vivekanandhan et al. (2018a, b). A study conducted on testing the mosquitocidal activity of Justicia adhatoda L. (Acanthaceae) leaf extracts revealed the potential of natural larvicidal agent against Ae. Aegypti (Thanigaivel et al., 2012, 2017a,b).
TABLE 1.
Phytochemicals identified from the specific plant families and their larvicidal activity on the mosquito species.
| Family and plant species | Major constituents | Mosquito species | References |
| Acanthaceae | |||
| Andrographis paniculata | Andrographolide | Aedes aegypti | Edwin et al., 2016 |
| Alangiaceae | |||
| Alangium salvifolium | Asarinin, sesamin and (+)-xanthoxylol-γ,γ-dimethylallylether, Hexadecanoicacid,1 hydroxymethyl-1,2-ethanediyl ester | Aedes aegypti | Thanigaivel et al., 2017a |
| Amaranthaceae | |||
| Chenopodium ambrosioides | α-Terpineol | Aedes aegypti | Leyva et al., 2009b |
| Amaryllidaceae | |||
| Alium macrostemon | Methyl propyl disulfide; mimethyl trisulfide | Aedes albopictus | Liu et al., 2014a |
| Alium monanthum | Dimethyl trisulfide; dimethyl tetrasulfide | Aedes aegypti | Moon, 2011 |
| Anacardiaceae | |||
| Pistacia terebinthus | α-Pinene; cyclopentane | Culex quinquefasciatus | Cetin et al., 2011 |
| Spondias purpurea | Caryophyllene oxide and α-cadinol | Aedes aegypti | Lima et al., 2011 |
| Annonaceae | |||
| Cananga odorata | Benzyl acetate, linalool, methyl benzoate | Aedes aegypti | Vera et al., 2014 |
| Guatteria blepharophylla | Caryophyllene oxide | Aedes aegypti | Aciole et al., 2011 |
| Guatteria friesiana | β-Eudesmol | Aedes aegypti | Aciole et al., 2011 |
| Guatteria hispida | β-Pinene and α-pinene | Aedes aegypti | Aciole et al., 2011 |
| Rollinia leptopetala | Spathulenol | Aedes aegypti | Feitosa et al., 2009 |
| Apiaceae | |||
| Angelica purpuraefolia | 4’-Chloro-4,4-dimethyl-3-(1-imidazolyl)-valerophenone, 1-Dodecanol, | Aedes aegypti | Nagella et al., 2012 |
| Anethum graveolens | Limonene, carvone | Aedes albopictus | Seo et al., 2015 |
| Apium graveolens | R-+-Limonene | Aedes aegypti | Pitasawat et al., 2007 |
| Limonene, carvone | Aedes albopictus | Seo et al., 2015 | |
| Bupleurum fruticosum | α-Pinene; β-pinene | Culex pipiens | Evergetis et al., 2009 |
| Carum carvi | Carvone | Aedes aegypti | Pitasawat et al., 2007 |
| Conopodium capillifolium | α-Pinene; sabinene | Aedes aegypti | Evergetis et al., 2009 |
| Coriandrum sativum | Linalool, 2,6-octadien-1-ol, 3,7- dimethyl-, acetate, E- | Aedes aegypti | Nagella et al., 2012 |
| Cuminum cyminum | ρ-cymene, β-pinene, cuminaldehyde | Aedes albopictus | Seo et al., 2015 |
| Daucus carota | Carotol | Aedes albopictus | Seo et al., 2015 |
| Elaeoselinum asclepium | α-Pinene; sabinene | Aedes aegypti | Evergetis et al., 2009 |
| Foeniculum vulgare | trans-Anethole, Limonene | Aedes aegypti | Rocha et al., 2015 |
| Heracleum pastinacifolium | Octyl acetate, Hexyl | Aedes aegypti | Tabanca et al., 2012a |
| Ligusticum chuanxiong | octadecenoic acids | Aedes aegypti, Culex quinquefasciatus | Evergetis et al., 2009 |
| Oenanthe pimpinelloides | γ-Terpinene; o-cymene | Aedes aegypti | Pavela, 2015 |
| Pimpinella anisum | Trans-anethole, α-Pinene; sabinene, β-phellandrene | Aedes aegypti | Pavela, 2015 |
| Petroselinum crispum | β-phellandrene,myristicin, α & β-pinene, myrcene | Anopheles culicifacies | Evergetis et al., 2012 |
| Pe. Sativum | Myristicin,1,8-cineole, 1,3,8-p-menthatriene | Aedes albopictus | Seo et al., 2015 |
| Trachyspermum ammi | Thymol | Anopheles stephensi | Pandey et al., 2009 |
| ρ-Cymene, γ-Terpinene | Aedes albopictus | Seo et al., 2015 | |
| Apocynaceae | |||
| Cionura erecta L. | Edren-9-one, alpha cadinol, eugenol and alpha muurolene | Anopheles stephensi | Mozaffari et al., 2014 |
| Araliaceae | |||
| Dendropanax morbifera | γ-Elemene | Aedes aegypti | Chung et al., 2009 |
| Aristolochiaceae | |||
| Aristolochia indica | Aristolochic acid I and II | Aedes aegypti | Pradeepa et al., 2015 |
| Asarum heterotropoides | Methyleugenol and safrole | Aedes aegypti | Perumalsamy et al., 2009 |
| Asteraceae | |||
| Achillea millefolium | Eucalyptol, β-pinene, borneol, sabinene, camphene | Aedes albopictus | Conti et al., 2010 |
| Artemisia absinthium | (Z)-β-ocimene, (E)-β-farnesene (Z)-en-yn-dicycloether | Aedes aegypti, Culex quinquefasciatus, Anopheles stephensi | Govindarajan and Benelli, 2016 |
| Ar. dracunculus | Hexanal, isovaleric acid, (Z)-3-hexenol, | Anopheles stephensi | Pour et al., 2016 |
| Hexadecanol | |||
| Artemisia vulgaris | Camphor, Linalool, terpenen-4-ol, a-and bthujone, b-pinene | Aedes aegypti | Bora and Sharma, 2011 |
| camphor, alpha-thujone, betacaryophyllene, gammamuurolene, camphene | |||
| Artemisia vulgaris | Myrcene, limonene, cineol | Aedes aegypti | Sujatha et al., 2013 |
| Ar. Nilagirica | Capillin | Aedes aegypti, Aedes albopictus | Bora and Sharma, 2011 |
| Blumea densiflora | Borneol, germacrene D, β-caryophyllene, γ-terpinene, sabinene, β-bisabolene | Anopheles anthropophagus | Zhu and Tian, 2011 |
| Blumea mollis | Linalool, γ-elemene, copaene, estragole, Allo-ocimene, γ-terpinene Alloaromadendrene | Culex quinquefasciatus | Senthilkumar et al., 2008 |
| Chamaemelum nobile | α-pinene | Aedes aegypti, Culex quinquefasciatus | Amer and Mehlhorn, 2006 |
| Chrysanthemum indicum | verbenol, 1,8-cineole, α-pinene, camphor, borneol, bornyl acetate | Aedes aegypti | Shunying et al., 2005 |
| Aedes aegypti | Wu et al., 2010 | ||
| Eupatorium betonicaeforme | β-Caryophyllene | Aedes aegypti | Albuquerque et al., 2004 |
| Matricaria recutita | α-bisabolol | Aedes aegypti | Heuskin et al., 2009 |
| Pectis oligocephala | p-Cymene and thymol | Aedes aegypti | Albuquerque et al., 2007 |
| Tagetes erecta | Piperitone | Aedes aegypti | Marques et al., 2011 |
| Tagetes filifolia | trans-Anethole | Aedes aegypti | Ruiz et al., 2011 |
| Tagetes lucida | Methyl chavicol | Aedes aegypti | Vera et al., 2014 |
| Tagetes minuta | Trans-ocimenone | Aedes aegypti | Ruiz et al., 2011 |
| Tagetes minuta | 5E-ocimenone | Aedes aegypti | Maradufu et al., 1978 |
| Tagetes patula | Limonene and terp | Aedes aegypti | Dharmagadda et al., 2005 |
| Bignoniaceae | |||
| Cybistax antisyphilitica | quinone | Aedes aegypti | Rodrigues et al., 2005 |
| Boraginaceae | |||
| Auxemma glazioviana | α-Bisabolol, α-cadinol, and T-muurolol | Aedes aegypti | Costa et al., 2004 |
| Cordia curassavica | Cordiaquinones J and K | Aedes aegypti | Ioset et al., 2000 |
| α-Pinene | Aedes aegypti | Santos et al., 2006 | |
| Cordia leucomalloides | δ-Cadinene and E- caryophyllene | Aedes aegypti | Santos et al., 2006 |
| Cucurbiataceae | |||
| Bryonopsis laciniosa | Goniothalamin | Culex pipiens | Kabir et al., 2003 |
| Cupressaceae | |||
| Callitris glaucophylla | Guaiol & citronellic acid | Aedes aegypti | Shaalan et al., 2006 |
| Chamaecyparis formosensis | Myrtenol | Aedes aegypti | Kuo et al., 2007 |
| Cryptomeria japonica | 16-Kaurene and elemol | Aedes aegypti, Aedes albopictus | Cheng et al., 2009c |
| Cunninghamia konishii | Cedrol, α-Pinene | Aedes aegypti | Cheng et al., 2013 |
| Cupressus arizonica var. glabra | α-Pinene & epi-zonarene | Aedes aegypti | Ali et al., 2013 |
| Cupressus arizonica | Limonene, umbellulone α-pinene | Anopheles stephensi | Sedaghat et al., 2011 |
| Cupressus benthamii | Limonene; umbellulone | Aedes albopictus | Giatropoulos et al., 2013 |
| Cupressus macrocarpa | Sabinene; α-Pinene; terpinen-4-ol | Aedes albopictus | Giatropoulos et al., 2013 |
| Cupressus sempervirens | α-Pinene; δ-3-carene | Aedes albopictus | Giatropoulos et al., 2013 |
| Cupressus torulosa | α-Pinene; δ-3-carene | Aedes albopictus | Giatropoulos et al., 2013 |
| Chamaecyparis | Myrtenol; myrtenal | Aedes aegypti, Aedes aegypti | Kuo et al., 2007 |
| formosensis | Limonene; oplopanonyl acetate; beyerene | Aedes albopictus | Giatropoulos et al., 2013 |
| Chamaecyparis lawsoniana | α-Pinene; sabinene; δ-3-carene | Culex pipiens | Vourlioti-Arapi et al., 2012 |
| Juniperus communis ssp. | |||
| Hemisphaerica | α-Pinene; limonene | Culex pipiens | Vourlioti-Arapi et al., 2012 |
| Juniperus drupacea | Sabinene; 4-methyl-1-1-methylethyl-3-cyclohexen-1-ol | Culex pipiens | Vourlioti-Arapi et al., 2012 |
| Juniperus foetidissima | Myrcene; germacrene-D; α-Pinene | Culex pipiens | Vourlioti-Arapi et al., 2012 |
| Juniperus oxycedrus L. ssp. | |||
| oxycedrus | α –pinene | Culex pipiens | Vourlioti-Arapi et al., 2012 |
| Juniperus oxycedrus L. | |||
| subsp. Macrocarpa | α-Pinene; δ-3-carene; β-phellandrene; α-terpinyl acetate | Aedes albopictus | Giatropoulos et al., 2013 |
| Juniperus phoenicea | |||
| Tetraclinis articulate | α-Pinene; bornyl acetate | Aedes albopictus | Giatropoulos et al., 2013 |
| Dioncophyllaceae | |||
| Triphyophyllum peltatum | dioncophylline A | Anopheles stephensi | François et al., 1996 |
| Euphorbiaceae | |||
| Croton nepetaefolius | Methyleugenol | Aedes aegypti | Morais et al., 2006 |
| Croton regelianus | Ascaridole & p-Cymene | Aedes aegypti | Torres et al., 2008 |
| Croton zehntneri | E-anethole, p-anisaldehyde | Aedes aegypti | Morais et al., 2006 |
| Fabaceae | |||
| Copaifera multijuga | β-caryophyllene | Anopheles darling, Aedes aegypti | Trindade et al., 2013 |
| Hymenaea courbaril | α-Copaene, spathulenol | Aedes aegypti | Aguiar et al., 2010 |
| Germacrene D and β-caryophyllene | |||
| Myroxylon pereirae | Benzyl benzoate | Aedes aegypti | Yenesew et al., 2003 |
| Millettia dura | Rotenoids, deguelin and tephrosin caryophyllene oxide; phenol,4-3,7-dimethyl-3-ethenylocta-1,6-dienyl; caryophyllene | Culex quinquefasciatus | Dua et al., 2013 |
| Psoralea corylifolia | Citronellol | Aedes aegypti | Benelli et al., 2017 |
| Geraniaceae | Culex quinquefasciatus | Cavalcanti et al., 2004 | |
| Pelargonium graveolens | Neral; geranial | Culex quinquefasciatus | |
| Gramineae | α-Pinene | Aedes aegypti | Cetin et al., 2011 |
| Cymbopogon citratus | Thymol | Culex pipiens | Govindarajan et al., 2013 |
| Hypericaceae | |||
| Hypericum scabrum | Δ-3-carene, 1,8-cineole, β-caryophyllene, bicyclogermacrene | Culex tritaeniorhynchus, Aedes albopictus, and Anopheles subpictus | Araújo et al., 2003 |
| Lamiaceae | |||
| Coleus aromaticus | β-caryophyllene, bergamotene, and terpinolene | Aedes aegypti | Jaenson et al., 2006 |
| Hyptis martiusii | |||
| Hyptis suaveolens | |||
| Lavandula gibsoni | α-Terpinolen and thymol | Aedes aegypti, Anopheles stephensi | Kulkarni et al., 2013 |
| Culex quinquefasciatus. | |||
| Lavandula stoechas | Fenchone, 1,8-Cineole | Culex pipiens | Traboulsi et al., 2002 |
| Lippia origanoides | Carvacrol | Aedes aegypti | Mar et al., 2018 |
| Mentha longifolia | Piperitenone oxid | Aedes aegypti | Pavela et al., 2014 |
| M. microcorphylla | Piperitenone, Pulegone, Piperitenone oxide | Culex pipiens | Traboulsi et al., 2002 |
| M. spicata | Carvone | Aedes aegypti | Govindarajan et al., 2012 |
| Nepeta cataria | E,Z-Nepetalactone and Z, E-nepetalactone | Aedes aegypti | Zhu et al., 2006 |
| Ocimum americanum | E-Methyl-cinnamate | Aedes aegypti | Cavalcanti et al., 2004 |
| Ocimum basilicum | Linalool; methyl eugenol | Aedes aegypti | Govindarajan et al., 2013 |
| Ocimum gratissimum | Eugenol | Aedes aegypti | Cavalcanti et al., 2004 |
| Ocimum sanctum | Methyleugenol | Culex pipiens | Gbolade and Lockwood, 2008 |
| O. syriacum | Carvacrol, Thymol | Aedes aegypti | Traboulsi et al., 2002 |
| Perilla frutescens | oleic, S-limonene, perillaldehyde | Aedes aegypti | Pohlit et al., 2011 |
| Plectranthus amboinicus | Carvacrol | Aedes aegypti | Lima et al., 2011 |
| Plectranthus mollis | Piperitone oxide, fenchone | Aedes aegypti | Kulkarni et al., 2013 |
| Pogostemon cablin | Patchouli alcohol, Seyshellene, α-bulnesene, Norpatchoulenol | Aedes aegypti | Lima-Santos et al., 2019 |
| Pulegium vulgare | Pulegone; carvone | Aedes albopictus | Pavela, 2015 |
| Rosmarinus officinalis | 1,8-Cineole; camphor | Aedes aegypti | Giatropoulos et al., 2018 |
| Satureja hortensis | γ-Terpinene; carvacrol | Culex pipiens | Pavela, 2009 |
| Thymus capitatus (L.) | Thymol, alpha-Amyrin, Carvacrol + beta- | Mansour et al., 2000 | |
| Hoffm. & Link | Caryophyllene | Culex pipiens | |
| Thymus leucospermus | p-Cymene | Culex pipiens | Pitarokili et al., 2011 |
| Thymus satureoides | Thymol; borneol | Culex pipiens | Pavela, 2009 |
| Thymus teucrioides | p-Cymene; γ-terpinene; thymol | Aedes albopictus | Pitarokili et al., 2011 |
| Thymus vulgaris | a-terpinene,carvacrol, thymol | Giatropoulos et al., 2018 | |
| p-cymene, linalool, geraniol | Aedes aegypti | ||
| Vitex agnus castus | Trans-caryophyllene; 1,8 cineole | Culex quinquefasciatus | Niroumand et al., 2018 |
| Vitex trifolia | Methyl-p-hydroxybenzoate | Aedes aegypti | Kannathasan et al., 2011 |
| Lauraceae | |||
| Cinnamomum camphora | 1,8-Cineole | Anopheles sinensis | Zhang et al., 2018 |
| C. cassia | Cinnamaldehyde | Aedes aegypti | Zhu et al., 2006 |
| C. impressicostatum | Benzyl benzoate and α-phellandrene | Aedes aegypti | Jantan et al., 2005 |
| C. japonicum | Borneol | Anopheles sinensis | Zhang et al., 2018 |
| C. microphyllum | Benzyl benzoate | Aedes aegypti | Jantan et al., 2005 |
| C. mollissimum | Benzyl benzoate | Aedes aegypti | Jantan et al., 2005 |
| C. osmophloeum | trans-Cinnamaldehyde and cinnamyl acetate | Aedes aegypti | Cheng et al., 2004 |
| C. pubescens | Benzyl benzoate | Aedes aegypti | Jantan et al., 2005 |
| C. rhyncophyllum | Benzyl benzoate | Aedes aegypti | Jantan et al., 2005 |
| C. scortechinii | β-Phellandrene and linalool | Aedes aegypti | Jantan et al., 2005 |
| C. sintoc | Safrole | Aedes aegypti | Jantan et al., 2005 |
| C. subavenium | Eugenol | Anopheles sinensis | Zhang et al., 2018 |
| C. szechuanense | 1,8-Cineole | Anopheles sinensis | Zhang et al., 2018 |
| Laurus nobilis | 1,8-cineole, linalool | Culex pipiens | Patrakar et al., 2012 |
| Lindera obtusiloba | α-Copaene; β-caryophyllene | Aedes aegypti | Pavela, 2015 |
| Magnoliaceae | |||
| Magnolia salicifolia | Trans-anethole, Methyl eugenol, isomethyl eugenol, Costunolide, lactone and parthenolide | Aedes aegypti | Kelm et al., 1997 |
| Malvaceae | |||
| Abutilon indicum | β-sitosterol | Aedes aegypti, | Rahuman et al., 2008a |
| Azadirachtin, salannin, deacetylgedunin, gedunin, 17-hydroxyazadiradione and deaceytlnimbin | |||
| Meliaceae | |||
| Azadirachta indica | Saponins | Anopheles stephensi, | Senthil-Nathan et al., 2005a |
| 23-O-methylnimocinolide | |||
| 6α-O-acetyl-7-deacetylnimocinol | Culex quinquefasciatus | Ansari et al., 2005 | |
| Nimocinolide; 7-O-deacetyl-23-O-methyl- | Aedes aegypti | Siddiqui et al., 1999 | |
| 7α-O-senecioylnimocinolide | Banerji and Nigam, 1984 | ||
| desfurano-6α-hydroxyazadiradione | Naqvi, 1987 | ||
| 22,23-dihydronimocinol | Aedes aegypti | Siddiqui et al., 2002 | |
| 1α-acetyl-3α-propionylvilasinin | Aedes aegypti | Siddiqui et al., 2003 | |
| Meliatetraolenone | Aedes aegypti | Siddiqui et al., 2003 | |
| azadirachtin, salannin, deacetylgedunin, | Culex quinquefasciatus | Siddiqui et al., 2003 | |
| gedunin, 17- hydroxyazadiradione | |||
| deacetylnimbin | Anopheles stephensi | Senthil-Nathan et al., 2005a | |
| 3β,24,25-trihydroxycycloartane | Anopheles stephensi | ||
| Dysoxylum malabaricum | Beddomei lactone | Aedes aegypti | Senthil-Nathan et al., 2009 |
| D. beddomei | Caryophyllene epoxide | Aedes aegypti | Senthil-Nathan et al., 2009 |
| cis-Caryophyllene | |||
| Guarea humaitensis | 1α,7α,11β-triacetoxy-4α-carbomethoxy- | Aedes aegypti | Magalhães et al., 2010 |
| G. scabra | 12α-(2-methylpropanoyloxy)-14β,15β-epoxyhavanensin | Aedes aegypti | Magalhães et al., 2010 |
| Turraea floribunda | 1α,11β-diacetoxy-4α-carbomethoxy-7α- | Aedes aegypti | Ndung’u et al., 2004 |
| hydroxy-12α-(2-methylpropanoyloxy)-15- | Aedes aegypti | Ndung’u et al., 2004 | |
| oxohavanensin; 1α-acetyl-3α- | Culex pipiens | Ndung’u et al., 2004 | |
| propionylvilasinin | Culex pipiens | Ndung’u et al., 2003 | |
| Turraea wakefieldii | 11β,12α-diacetoxyneotecleanin | Culex pipiens | Ndung’u et al., 2003 |
| 11β,12α-diacetoxy-14β,15β- | Aedes aegypti | Ndung’u et al., 2003 | |
| epoxyneotecleanin | Aedes aegypti | Ndung’u et al., 2003 | |
| Myrtaceae | |||
| Eucalyptus benthamii | α-Pinene | Aedes aegypti | Lucia et al., 2012 |
| E. botryoides | p-Cymene, α-eudesmol, and 1,8-cineol | Aedes aegypti | Lucia et al., 2012 |
| E. camaldulensis | 1,8-Cineol, p-cymene and β-phellandrene | Aedes aegypti | Lucia et al., 2008 |
| E. citriodora | Citronellal; citronellol; | Aedes aegypti | Vera et al., 2014 |
| α-humulene isopulegol | |||
| E. dunnii | 1,8-Cineol and γ-terpinene | Aedes aegypti | Lucia et al., 2008 |
| E. fastigata | p-Cymene | Aedes aegypti | Lucia et al., 2012 |
| E. globulus | 1,8-Cineol | Aedes aegypti Anopheles arabiensis | Massebo et al., 2009 |
| E. grandis | α-Pinene | Aedes aegypti | Lucia et al., 2007 |
| E. gunnii | 1,8-Cineol and p-cymene | Aedes aegypti | Lucia et al., 2008 |
| E. nobilis | 1,8-Cineol | Aedes aegypti | Lucia et al., 2012 |
| E. radiata | 1,8-Cineol | Aedes aegypti | Lucia et al., 2012 |
| E. robusta | α-Pinene | Aedes aegypti | Lucia et al., 2012 |
| E. saligna | 1,8-Cineol and p-cymene | Aedes aegypti | Lucia et al., 2008 |
| E. tereticornis | β-Phellandrene and 1,8-cineol | Aedes aegypti | Lucia et al., 2008 |
| E. urophylla | 1,8-Cineol | Aedes aegypti | Cheng et al., 2009b |
| E. melanadenia | 1,8-Cineol | Aedes aegypti | Aguilera et al., 2003 |
| Myrtus communis | 1,8 Cineole, α-Pinene, Linalool | Culex quinquefasciatus | Traboulsi et al., 2002 |
| M. dissitiflora | Terpinen-4-ol | Aedes aegypti | Park et al., 2011 |
| M. leucadendron | 1,8-Cineol, α-pinene, and α-terpineol | Aedes aegypti | Leyva et al., 2008 |
| M. linariifolia | Terpinem-4-ol and γ-terpinene | Aedes aegypti | Park et al., 2011 |
| M. quinquenervia | 1,8-Cineol and E-nerolidol | Aedes aegypti | Park et al., 2011 |
| Pimenta dioica | Eugenol, linalool | Aedes aegypti | Pereira et al., 2014 |
| P. racemosa | Terpinem-4-ol and 1,8-cineol | Aedes aegypti | Aciole, 2009 |
| P. guajava | 1,8-Cineol and β-caryophyllene | Culex pipiens | Leyva et al., 2009a |
| 1,8-Cineol | Aedes aegypti | Lima et al., 2011 | |
| P. rotundatum | Eugenol | Aedes aegypti | Aguilera et al., 2003 |
| Syzygium aromaticum | Eugenol | Aedes aegypti | Costa et al., 2005 |
| Orchidaceae | |||
| Vanilla fragrans | 4-ethoxymethylphenol, 4-butoxymethylphenol, vanillin, 4-hydroxy-2-methoxycinnamaldehyde and 3,4-dihydroxyphenylacetic acid | Culex pipiens | Sun et al., 2001 |
| Pinaceae | |||
| Cupressus L., | limonene, α & β-pinene, | Aedes aegypti | Burfield, 2000 |
| Juniperus L. | 3-carene | Aedes aegypti | Burfield, 2000 |
| Pinus brutia | α-Pinene and β-pinene | Aedes albopictus | Koutsaviti et al., 2015 |
| P. halepensis | β-Caryophyllene | Aedes albopictus | Koutsaviti et al., 2015 |
| P. kesiya | α-Pinene, β-pinene, myrcene and germacrene D. | Aedes aegypti, Culex quinquefasciatus, | Govindarajan et al., 2016 |
| Anopheles stephensi | |||
| P. longifolia | k-terpineol | Culex quinquefasciatus, Anopheles | Ansari et al., 2005 |
| culicifacies | |||
| P. stankewiczii | Germacrene D α-Pinene and β-pinene | Aedes albopictus | Koutsaviti et al., 2015 |
| P. sylvestris | Eugenol 3, Cyclohexene-1-methanol, α-4- | Aedes aegypti, Culex quinquefasciatus | Fayemiwo et al., 2014 |
| trimethyl | |||
| Piperaceae | |||
|
Piper auritum P. betle P. capense P. decurrens |
Safrole Citronellal 2,3-Dihydro-2-(4′-hydroxyphenyl)-3-methyl-5(E)-propenylbenzofuran (conocarpan), 2-(4′-hydroxy-3′-methoxyphenyl)-3-methyl-5(E)-propenylbenzofuran (eupomatenoid-5), 2-(4′-hydroxyphenyl)-3-methyl-5(E)-propenylbenzofuran (eupomatenoid-6), 2,3-dihydro-5-formyl-2-(4′-hydroxyphenyl)-3-methylbenzofuran (decurrenal), and 3,7,11,15-tetramethyl-2(E)-hexadecen-1-ol (trans-phytol) |
Aedes aegypti Aedes aegypti Aedes atropalpus Aedes aegypti |
Leyva et al., 2009b Wahyuni, 2012 Chauret et al., 1996 de Morais et al., 2007 |
| P. gaudichaudianum | Caryophyllene oxide, β-selinene | Aedes aegypti | de Morais et al., 2007 |
| P. hostmanianum | Asaricin and myristicin | Aedes aegypti | de Morais et al., 2007 |
| P. humaytanum | β-selinene, caryophyllene oxide | Aedes aegypti | de Morais et al., 2007 |
| P. klotzschianum | 1-Butyl-3,4-methylenedioxybenzene, | Aedes aegypti | do Nascimento et al., 2013 |
| P. longum | limonene, and α-phellandrene | Culex pipiens | Lee, 2000 |
| Pipernonaline | Aedes aegypti | Yang et al., 2002 | |
| Aedes aegypti | Costa et al., 2004 | ||
| P. marginatum | Isoelemecin, apiole | Aedes aegypti | Autran et al., 2009 |
| (Z)-Asarone | Aedes aegypti | Autran et al., 2009 | |
| P. permucronatum | (E)-Asarone, patchouli alcohol | Aedes aegypti | de Morais et al., 2007 |
| Dillapiole and myristicin | |||
| Plumbaginaceae | |||
| Plumbago zeylanica | Plumbagin | Aedes aegypti | Pradeepa et al., 2016 |
| Poaceae | |||
| Cymbopogon citratus | Geranial | Aedes aegypti | Cavalcanti et al., 2004 |
| Cymbopogon flexuosus | citral a-pinene | Aedes aegypti | Syed and Leal, 2008 |
| Cymbopogon nardus | Geranial; neral | Aedes aegypti | Vera et al., 2014 |
| Girgensohnine | Aedes aegypti | Carreño-Otero et al., 2018 | |
| Vetiveria zizanioides | Citronellal | Aedes aegypti | Fradin and Day, 2002 |
| khusimol, isonootkatool, β-vetivenene, α & | Aedes aegypti | Vera et al., 2014 | |
| β-vetivones | |||
| Papilonaceae | |||
| Neorautanenia mitis | Neotenone, neorautanone, pterocarpans neoduline, nepseudin,4-methoxyneoduline | Culex quinquefasciatus, Anopheles gambiae | Joseph et al., 2004 |
| Aedes aegypti, Aedes albopictus | |||
| Elemol, Eudesmols | Culex quinquefasciatus | Zhu et al., 2006 | |
| Rutaceae | |||
| Chloroxylon swietenia | Heptacosanoic acid | Aedes aegypti, Culex quinquefasciatus | Balasubramani et al., 2015 |
| Citrus aurantifolia | Geijerene, Limonene, Germacrene D | Aedes aegypti, Anopheles stephensi | Kiran et al., 2006 |
| Citrus hystrix | α-terpineol | ||
| Citrus limon | β-Pinene; d-limonene; terpinene-4-ol | Culex pipiens | Sutthanont et al., 2010 |
| Limonene | Aedes aegypti | ||
| Citrus reticulata | D-Limonene; γ-terpinene | Culex quinquefasciatus | Michaelakis et al., 2009 |
| Citrus sinensis | Limonene | Aedes aegypti | Sutthanont et al., 2010 |
| Limonin, Nomilin, Obacunone | Culex quinquefasciatus | Jayaprakasha et al., 1997 | |
| Geijerene; limonene; germacrene D | Aedes aegypti | Vera et al., 2014 | |
| Chloroxylon swietenia | |||
| Kiran et al., 2006 | |||
| Clausena excavate | Safrole and terpinolene | Aedes aegypti, Aedes albopictus | Cheng et al., 2009a |
| Feronia limonia | Estragole and β-pinene | Aedes aegypti | Senthilkumar et al., 2013 |
| F. limonia | n-hexadecanoic acid | Culex quinquefasciatus | Rahuman et al., 2000 |
| Ruta graveolens | Undecan-2-one | Aedes aegypti | Tabanca et al., 2012b |
| Swinglea glutinosa | β-Pinene; piperitenone; | Aedes aegypti | Vera et al., 2014 |
| α-Pinene | |||
| Toddalia asiatica | Linalool | Aedes aegypti | Nyahanga et al., 2010 |
| Zanthoxylum armatum | Linalool | Aedes aegypti | Tiwary et al., 2007 |
| Z. articulatum | Viridiflorol | Aedes aegypti | Feitosa et al., 2007 |
| Z. avicennae | 1,8-Cineole | Aedes albopictus | Liu et al., 2014b |
| Limonene | Aedes aegypti | Pitasawat et al., 2007 | |
| Methyl heptyl ketone | Aedes aegypti | Borah et al., 2012 | |
| Z. piperitum | Asarinin, sesamin and (+)-xanthoxylol-γ,γ-dimethylallylether | Aedes aegypti, Culex pipiens | Kim and Ahn, 2017 |
| Z. monophyllum | Germacrene D-4-ol and a-Cadinol | Aedes albopictus, Culex quinquefasciatus, Anopheles stephensi | Pavela and Govindarajan, 2017 |
| Santalaceae | |||
| Santalum L. spp. | α-santalol | Aedes aegypti, Culex pipiens | Jones et al., 2007 |
| Santalum album | Guaiol, elemol, and eudesmol | Anopheles stephensi, | Amer and Mehlhorn, 2006 |
| Schisandraceae | Aedes aegypti | ||
| Illicium verum | Eugenol, α-Terpinyl acetate, Eucalypt, ol, (E)-anethole | Culex quinquefasciatus | Kimbaris et al., 2012 |
| Scrophulariaceae | |||
| Capraria biflora L. | α-Humulene | Aedes aegypti | Souza et al., 2012 |
| Stemodia maritima | β-Caryophyllene and caryophyllene oxide | Aedes aegypti | Arriaga et al., 2007 |
| Tiliaceae | |||
| Microcos paniculata | N-Methyl-6b-(deca-l’,3’,5’-trienyl)-3b-methoxy-2bmethylpiperidine | Aedes aegypti | Bandara et al., 2000 |
| Verbenaceae | |||
| Duranta repens | β-amyrin and 12-oleanene 3β, 21β-diol, | Culex quinquefasciatus | Nikkon et al., 2010 |
| Lantana camara | Bicyclogermacrene and E-caryophyllene | Aedes aegypti | Costa et al., 2010 |
| Eucalyptol, caryophyllene, | |||
| Lippia alba | Carvone; limonene | Aedes aegypti | Santiago et al., 2006 |
| L. gracilis | Carvacrol | Aedes aegypti | Santiago et al., 2006 |
| L. origanoides | Carvacrol; p-cymene | Aedes aegypti | Vera et al., 2014 |
| L. javanica | Allopurinol,camphor, Limonene, a –terpeneol, verbenone | Aedes aegypti | Mwangi et al., 1992 |
| L. microphylla | 1,8-cineole, thymol, α-pinene | Aedes aegypti | Santiago et al., 2006 |
| L. nodiflora | Camphor, p-cymene, γ−terpinene | Aedes aegypti | Santiago et al., 2006 |
| L. sidoides | Thymol | Aedes aegypti | Costa et al., 2005 |
| Zingiberaceae | |||
| Alpinia purpurata | β-Caryophyllene and β-pinene | Aedes aegypti | Santos et al., 2012 |
| Curcuma aromatic | 1H-3a,7-Methanoazulene and curcumene | Aedes aegypti | Choochote et al., 2005 |
| Turmerone, curcumene, and zingiberene | |||
| Curcuma longa | 1,8-Cineol and p-cymene | Aedes aegypti | Leyva et al., 2008 |
| Curcuma zedoaria | Dodecanal | Aedes aegypti | Pitasawat et al., 2007 |
| Hedychium coccineum | 1,8-Cineol and β-pinene | Aedes aegypti | Sakhanokho et al., 2013 |
| Hedychium sp. | 1,8-Cineol | Aedes aegypti | Sakhanokho et al., 2013 |
| Kaempferia galanga | Ethyl trans-p-methoxycinnamate | Aedes aegypti | Munda et al., 2018 |
| Kaempferia galanga | Ethyl cinnamate | Aedes aegypti | Munda et al., 2018 |
| Zingiber officinale | 4-Gingero | Aedes aegypti, Culex quinquefasciatus | Rahuman et al., 2008b |
| Zingiber officinale | 6-Dehydrogingerdione | Aedes aegypti, Culex quinquefasciatus | Rahuman et al., 2008b |
| Zingiber officinale | 6-Dihydrogingerdione | Aedes aegypti, Culex quinquefasciatus | Rahuman et al., 2008b |
| Zingiber zerumbet | α-Humulene; zerumbone | Aedes aegypti | Sutthanont et al., 2010 |
Besides secondary metabolites, essential oils (EOs) from plants were also recorded with effective mosquitocidal potentials. The EOs from the plants of Lamiaceae and Zingiberaceae were proved with bioactivity against Ae. aegypti (Kalaivani et al., 2012). The fern Actiniopteris radiata was testified with novel mosquitocidal activity against larvae of Ae. aegypti and An. Stephensi (Kamaraj et al., 2018). The seed oil extract of Acacia nilotica possessed robust larvicidal action against major mosquito vectors (Vivekanandhan et al., 2018a). A remarkable biological activity of EOs against Dengue vectors has been extensively reviewed by Chellappandian et al. (2017, 2018, 2019). Plant volatile oils were also conveyed with mosquitocidal potentials. As studied by Vasantha-Srinivasan et al. (2018), the crude volatile oil (CVO) from Piper beetle leaves possessed significant larvicidal, ovipositional, and repellency effects against Ae. Aegypti.
Derivatives of plants are enriched with active molecules with exceptional mosquitocidal properties and can be advanced as low cost environmentally friendly bio-pesticides. Many botanical extracts along with their chief constituents showed effective insect metabolism inhibition or stimulation of digestive enzymes (Senthil-Nathan et al., 2009; Napoleão et al., 2012; Senthil-Nathan, 2013). Unlike synthetic chemicals, previous literature on plant compounds doesn’t provide any indication for the emergence of resistance so far. This is most likely due to the blend of several bioactive compounds with different mechanisms of action and therefore it is difficult for mosquito vectors to develop resistance (Mulla and Su, 1999; Shaalan et al., 2005).
Impact of Phytochemicals on the Physiology of Mosquito Larvae
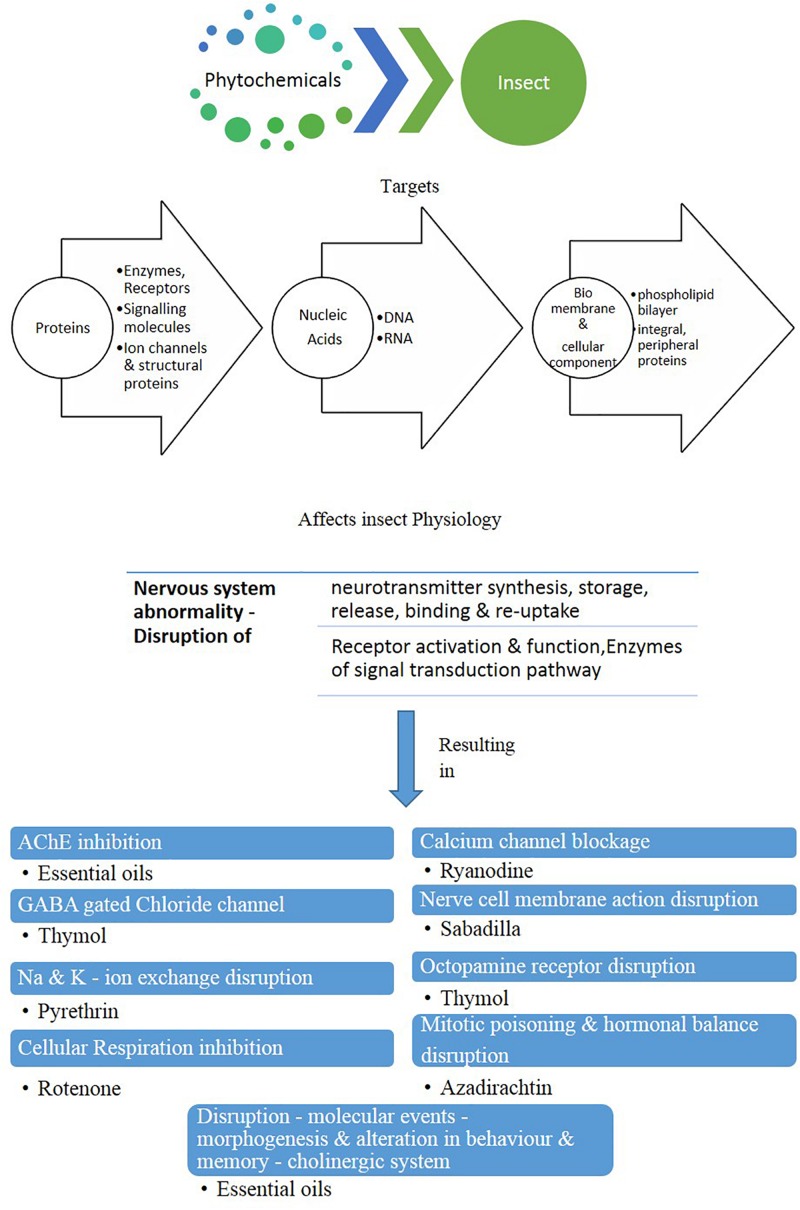
As in general, plant secondary metabolites are evolved as protection mechanism against herbivory. When these toxic substances are encountered by the mosquitoes, a relatively unambiguous response is triggered that has a non-specific influence on a wide range of molecular targets such as proteins, nucleic-acids, bio-membranes, besides added cellular components. Consequently, the physiology is disrupted at numerous receptor sites, eventually causing an abnormality in the nervous system. Plant metabolites affect several vital physiological functions that include inhibition of “AChE” as well as “GABA-gated” chloride channel, disruption of Na–K ion exchange besides constricting the cellular respiration. As a subsequent event, the alteration of these enzyme levels gives rise to several anomalies that include the obstruction of nerve cell membranes and octopamine receptors along with calcium channel blockage, resulting in hormonal imbalance, mitotic poisoning, and also modifications of the molecular basis of morphogenesis (Rattan, 2010).
Synthetic insecticides generally increase the level of detoxifying enzymes. Phytochemicals target the mentioned cellular mechanisms and potentially disturb their functions (Figure 1; Zibaee and Bandani, 2010; Zibaee, 2011; Kaur et al., 2014; Senthil-Nathan, 2015). Physiological effects of phytochemicals are discussed below.
FIGURE 1.
Mode of action of phytochemicals in insect body (modified after Ghosh et al., 2012).
Impact of Phytochemicals on Detoxifying Enzymes
The antioxidant and detoxification enzymes of mosquito vectors are vital in detoxification of reactive oxygen species (ROS) synthesized by the toxic chemicals (Rattan, 2010). Esterase and phosphatase of the mosquito vectors plays a key role in several physiological events (Koodalingam et al., 2014). Excessive usage of toxic chemicals on mosquito control caused insecticide resistance through sodium channel mutations, activation of detoxification enzymes, and upregulation of key genes and other regulatory components like MicroRNAs (miRNAs). The CYP450s, GSTs, SOD, and esterase gene families are recognized as the foremost four enzymes accountable for the metabolic-resistance of the insects (Hemingway et al., 2004). Generally, detoxifying enzymes are involved in digestion, reproduction, juvenile hormone metabolism, neuronal conduction, moulting, and more importantly detoxification of toxic chemicals (Koodalingam et al., 2014). Phosphatases are involved in tissue development, cellular differentiation, carbohydrate metabolisms, and synthesis of ATP (Koodalingam et al., 2014). Mainly these two major classes of detoxifying enzymes are considered for evaluating the impact of toxic chemicals on physiological or biochemical events of arthropod vectors.
Carboxyl-esterases (EC3.1.1.1) are non-specific omnipresent enzymes that are associated to the major “endogenous” functions in insects, which hydrolyze a different carboxylic-acid ester (Lija-Escaline et al., 2015). Generally, the metabolic pathway of these enzymes was targeted by the chemical pesticides, especially the fourth generation class of Pyrethroids, which acts on the voltage sensitive sodium channels and blocks the mosquito nervous system (Hong et al., 2014). Esterases can also target by sequestering the insecticide through rapid binding and slowly releasing the insecticide metabolites (Karunaratne et al., 1993). This latter type of resistance requires the presence of increased quantities of esterase due to the 1:1 stoichiometry of the reaction and decreases the metabolic breakdown time.
Plant extracts and their derivatives have been widely reported to decrease the levels of carboxylesterase (α- β-carboxylesterase) level in the Ae. aegypti larva (Koodalingam et al., 2014; Lija-Escaline et al., 2015). Besides exhibiting larvicidal activity Alangium salvifolium, also substantially reduced the levels of α, β-carboxylesterase as well as superoxide dismutase (SOD) in Ae. Aegypti (Thanigaivel et al., 2017a). Myrrh commiphora molmol (oil and oleo-resin extract) instigated biochemical changes in Cx. pipiens that affected the cell proteins, as well as loss of enzyme activity (Massoud et al., 2001).
Higher rates of enzyme activities, such as SOD (Agra-Neto et al., 2014; Lija-Escaline et al., 2015) and physiological enzymes like esterase (Wheelock et al., 2005; Lija-Escaline et al., 2015), phosphatases (Walter and Schütt, 1974; Urich, 1994) are recorded with increasing developmental stages and these are considered responsible for increased pyrethroid resistance. The Mosquito vectors that established resistance to Temephos have been found to possess genes that insensitized ACHE on exposure to pesticides. Insects were also characterized by the over expression of varied forms of detoxifying enzymes (GST, SOD, and esterases) (Larson et al., 2010).
Glutathione-S-transferases are a class of detoxification enzymes considered to play a vital role in the existence of insects exposed to toxic metabolites. Increased GST activities are connected with the expression of metabolic resistance toward insecticides (Clark, 1990). GSTs can break down a broad range of substances; amplified GST activity is possibly as a response to an environmental stress. Generally, Cytochrome P450s (CYP450) displayed upregulation when induced by plant secondary metabolites in diverse insect pests especially against the vectors of human diseases (Caballero et al., 2008) and have members which are considered as major elements conferring resistance against insecticides (i.e., CYP2, CYP4, and CYP6) (Sun et al., 2001). The upregulation of GST enzymes usually at the exposure of a prominent dosage of plant compounds suggests the activity of a major detoxification process (Edwin et al., 2016). Consequently, the levels of GST expression may be used as a biomarker to detect the development of resistance (Jukic et al., 2007).
CYP450 group of enzyme family are also designated as key indicators of metabolic resistance besides susceptibility to insecticides (David et al., 2013). Many previous research outcomes proved alteration or inhibition in the expression of major detoxifying enzymes exposed to plant chemicals. Thanigaivel et al. (2017a) showed increase in the rate of GST activity in IV instar larvae of dengue mosquito exposed to methanolic leaf extract of J. adhatoda with their major derivative 3-hydroxy-2,3-dihydropyrrolo[2,1-b]quinazolin-9(1h) one (26.37%). Likewise, carboxylesterase activities differed significantly in Ae. aegypti post treatment with the leaf extracts of P. nigrum with their major derivatives thymol (20.77%) (Lija-Escaline et al., 2015). Correspondingly, the activity of major enzymes (esterases, GST, and CYP450) of dengue mosquito severely affected post treated with dynamic plant compound andrographolide derived from Andrographis paniculata (Acanthaceae) at the maximum dosage of 12 ppm (Edwin et al., 2016). DDT resistance in the mosquito An. gambiae is correlated elevated glutathione transferase (GST) E2 activity (AgGSTE2) (Enayati et al., 2005). The DDT resistant An. gambiae evades the insecticidal activity by the dehydrochlorination of DDT to its non-insecticidal metabolite DDE. Muleya et al. (2008) reported that compounds -epiphyllocoumarin (Tral-1), knipholone anthrone, isofuranonaphthoquinones (Mr 13/2, Mr13/4), and the polyprenylated benzophenone (GG1) were potent inhibitors of AgGSTE2.
Besides the botanical extracts, EO derived from the plants also have strong inhibition of detoxifying enzymes of arthropod vectors (Pavela, 2015). EOs may provide substitute sources of vector control since they are enriched with diverse phyto-molecules with insecticidal properties (Cheng et al., 2013). Insecticide phytochemicals from EOs belong to terpenoids chiefly and Phenylpropanoids to a limited extent. In which, Terpenoids includes monoterpenes and sesquiterpenes as the major compositions of EOs (Chellappandian et al., 2018). Lee et al. (2003) specified that volatile and lipophilic monoterpenoids infiltrate insect body, where they afflict physiological processes, and hence their mode of action is hard to elucidate. Previous research of Vasantha-Srinivasan et al. (2017) showed that the CVO derived from Piper betle (L.) (Pb-CVO) showed upregulation in the level GST and CYP450 and down regulate the expression of Carboxylesterases activity against the field and laboratory strains of Ae. aegypti. Moreover, the above results also showed that the changes in the level of enzymes are steady in both field and laboratory strains compared to the chemical pesticides. Due to enriched chemical diversity and potential mosquitocidal activity, CVO have acquired greater interest from researchers looking for new besides natural replacements to chemical-pesticides in controlling medically challenging pests (Pavela, 2015). Correspondingly, EO constituent’s nootkatone and carvacrol from Alaskan yellow cedar tree inhibits 50% of acetylcholine esterase activity in Ae. aegypti compared to the carbaryl, a known acetylcholinesterase inhibitor (Anderson and Coats, 2012). The impact of major plant molecules against the mosquito larvicides was tabulated (Table 1). Hence, expression of these molecules on detoxifying and metabolic enzymes is considered an important biomarker to evaluate the mosquitocidal potential of bio-rational plant metabolites.
Pradeepa et al. (2014) have reported the antimalarial activities from the compound plumbagin, identified from the rhizome of Plumbago zeylanica against An. stephensi. Also, it was revealed that plumbagin constrains the vector AchE enzyme, An. Stephensi in a dose dependent manner and also can be considered for controlling resistant vectors whose insecticide resistance is associated to an increased SOD activity (Pradeepa et al., 2016). The detection of SOD activity in the anal gills of An. stephensi larvae could be associated with their resistance provided against damaging oxygen products (Nivsarkar et al., 1991). The sensitivity of an insect to an insecticide can hence be increased by identifying certain compounds that can deactivate these enzymes (Larson et al., 2010).
Impact of Phytochemicals on Midgut Tissues
The midgut of the mosquito larvae is the chief interface of exterior environment and chip in major process like digestion, ion transport, absorption, and osmoregulation process (Bernick et al., 2008; Elumalai et al., 2016). Generally, gut region is the target of numerous insecticidal complexes and its integrity is dynamic for digestion and conferring of resistance against toxins (Stenfors Arnesen et al., 2008). With the insect midgut being the important site for synthesis of digestive enzymes, plant derived molecules primarily targets thee gut epithelium layer (EL) (Senthil-Nathan et al., 2008). This might be the significant cause for condensed metabolic rate in addition to a reduced enzyme activity (Selin-Rani et al., 2016). The peritrophic membrane (pM) gaurds the EL from the surrounding the gut lumen (GL) (Lija-Escaline et al., 2015). Phyto-chemicals are proven to exert a serious impact on the digestive epithelial cells and further decrease the growth rate of arthropods (Yu et al., 2015). Neira-Oviedo et al. (2008) stated that plant compounds flow into the gastric caeca and the malpighian tubules thereby affecting the midgut epithelium. For instance, extracts of M. azedarach have been reported to cause extensive harm on the EL and pM of filarial vector Cx. quinquefasciatus (Al-Mehmadi and Al-Khalaf, 2010). The pM may influence the growth and development of parasites vectors by creating a mechanical barrier to invasion by ookinetes (Rudin and Hecker, 1989). Plant extracts and their metabolites are crucial for the impairment of pest mid-gut epithelium (Rey et al., 1999). The compound catechin isolated from Leucas aspera affects the mid-gut of the three mosquito larvae Ae. aegypti, An. stephensi, and Cx. quinquefasciatus (Elumalai et al., 2016). Previous photomicrographic study on the midgut tissues of the dengue mosquito (Field and laboratory strains of Ae. aegypti) treated with the CVO of P. betle displayed severe injuries to the GL and EL (Vasantha-Srinivasan et al., 2018). Correspondingly, leaf extracts of Aristolochia indica L. (Aristolochiaceae) and their derivatives aristolochic acid I and II showed severe damage on the midgut vacuolated gut epithelial columnar cells (epi), GL, and pM (Pradeepa et al., 2015). Likewise, methanolic leaf extracts of P. nigrum severely affected the midgut cellular organelles of Ae. aegypti at the minimal dosage of 10 ppm (Lija-Escaline et al., 2015). Similarly, Vasantha-Srinivasan et al. (2018) reported that P. betle CVO derived from P. betle at the sub-lethal dosage damage the pM, and major alteration in the alignment of EL and GL of dengue mosquito comparable to the control. Previous research on Andrographolide a major derivative of A. paniculata against dengue mosquito gut cells proved that there was an unembellished collapse in the mid-gut pM, in addition to a chief variation in the El and GL alignment (Edwin et al., 2016). Selin-Rani et al. (2016) reported that the active plant molecules may damage the gut epithelium is the vital reason for concentrated metabolic rate and decrease in the enzyme-activity. Midgut cell damage is directly linked to the digestive and detoxifying enzymes dysregulation (Senthil-Nathan et al., 2008). This was also confirmed by histological studies of the mosquitoes that displayed midgut cell damage, post treatment with various botanical compounds (Yu et al., 2015). Further, treatment with plant compounds were also associated with altered protein (Fallatah, 2014) and biochemical profiles in mosquitoes (Senthilkumar et al., 2013).
Biochemical studies on Cx. pipipens exposed to Allium satvium, Citrus limon, and Bti were observed by Saeed et al. (2010). Results revealed that the use of plant oil extracts and Bti have great effect on total protein content of treated mosquito larvae. Fallatah (2010) reported the effect of water extract of fenugreek have high larvicidal effect against Cx. quinquefasciatus, causing noticeable effects on numerous body tissues together with the midgut and nervous system as well as total protein content. Aristolochic acids isolated from A. indica Linn, mainly affected the midgut EL and secondly the larval muscles and cells (Pradeepa et al., 2015). Similar results were also observed in mosquitoes treated with plant extracts (Costa et al., 2012). The orientation of the cytoplasmic protrusions of the apical surfaces of columnar cells toward the lumen suggests the secretion of apocrine and/or apoptosis.
Al-Mekhlafi (2018) reported the effect of Arum copticum (Apiaceae) extract against Culex pipiens larvae. Apart from exhibiting larvicidal activity, the extract was able to display cytopathological alterations of the midgut epithelium. EO and enriched fraction of Peumus boldus displayed larvicidal activity against Cu. Quinquefasciatus. The treated larvae displayed morphological changes in the midgut cells (de Castro et al., 2016). Velu et al. (2015) tested the peel extract of A. hypogaea against Aedes aegypti and Anopheles stephensi. The histopathological studies exposed midgut tissue damage and cuticle injury. Costa et al. (2012) reported similar aberrations in Ae. aegypti larvae (III instar) treated with Annona coriacea extract. Ae. aegypti larvae exposed to squamocin from Annona mucosa Jacq. (Annonaceae) displayed larvicidal and cytotoxic action with changes in the midgut epithelium and digestive cells by increasing the expression of autophagy genes (Costa et al., 2014, 2017). da Silva Costa et al. (2018) also reported that squamocin affected the osmoregulation and ion-regulation of Ae. aegypti larvae which resulted in a lethal effect caused by the development of a great vacuolization in the anal papillae wall.
The histopathological study of Ae. aegypti treated with methanol extract derived from seaweeds Sargassum binderi showed that larvae treated with seaweed extracts had cytopathological alteration of the midgut epithelium. The morphological observation revealed that the anal papillae and terminal spiracles of larvae were the common sites of aberrations (Yu et al., 2015). Phytochemicals (oleic, linoleic, linolenic, palmitic, and stearic acids) and their respective methyl esters were tested against fourth instar Cu. quinquefasciatus larvae. The compounds were found to affect its metabolism and the morphology of midgut along with their fat body (de Melo et al., 2018).
Impact of Phytochemicals on the Insect Behavior
With the development of resistance by this time attained to almost all available chemicals, strategies integrating “plant derived” compounds to influence “semiochemical”-mediated behaviors by means of interruption of mosquito-olfactory sensory system have substantially developed (Muema et al., 2017). As a consequence, the physiological status related to the olfactory sensory system is disrupted. The phytochemicals will bind to these odorant chemoreceptors and subsequent flight orientations of the mosquitoes are hindered (Bohbot et al., 2010). Henceforth the physiological status for instance “circadian-regulated appetitive stimulus” or “gonotrophic status” that triggers olfaction in pursuit of nutritious sources, mates and oviposition sites are disturbed. Plant-based semiochemicals can be exploited to lure the mosquitoes to an insecticide trap, thereby forming an integral part of an integrated vector control programe (Kamala-Jayanthi et al., 2015). Rice volatiles on evaluation with BioGent (BG) sentinel traps elicited antennal responses that stimulated long range oviposition site seeking behavior. Also, p-cresol, from Bermuda grass hay infusion was reported with avoidance response to gravid An. Gambiae (Eneh et al., 2016).
Future Perspectives
Higher rates of anthropogenic activities that are expected to expand with the population increase will increase the incidence of vector borne diseases. Additionally, the development of resistance among the vector population against the synthetic chemical insecticides along with their persistence in the environment and toxicity for non-target organisms are reducing the efficiencies of vector management practices globally. Hence novel plant-based compounds that are safe and effective are being focused for the development of improved management of vectors.
The research has now moved on from the isolation of bioactive compounds with anti-vector potentials to formulate novel application methods. Apart from the direct application of plant metabolites in vector control, nanoparticles (NPs) synthesized from plants using green technology are emerging as a new trend. Nanotechnology is presently “revolutionizing” the manufacture of commercial pesticides. Production of green NPs and nanoencapsulation compounds upsurges the permanence of EOs through “slow-release” phenomenon deliberating sustained fortification against mosquito bites. As reported by Jinu et al. (2018), silver nanoparticles (AgNPs) from Cleistanthus collinus Karra and Strychnos nux-vomica Linn nux-vomica presented highest larvicidal activity against A. stephensi and A. aegypti. Murugan et al. (2018a, b) proved the efficacy of zinc oxide NPs fabricated using the brown macroalga Sargassum wightii Greville ex J. Agardh. against An. stephensi. In another study reported by Murugan et al. (2018b), Poly (Styrene Sulfonate)/Poly (allylamine hydrochloride) encapsulation of TiO2 NPs were found to enhance their toxicity against mosquito vectors of Zika virus.
Conclusion
Mosquito vector borne diseases are a major human health problem in all countries. There has been an alteration toward plant-based insecticides to overcome the problems related with the use of synthetic mixtures in mosquito control programe. Botanicals can be used as mosquitocides for killing both larvae and adult mosquitoes. However, only very few botanicals have moved from laboratory to the field use, which may be due to the light and heat variability of phytochemicals compared to synthetic insecticides. Further these botanicals have been widely explored, but only a comparatively small number of patents have been filed with the persistence of regulating the formulations for use against mosquito species in the field level.
Although the activity of phytochemicals are generally attributed to some specific compounds, but there is increasing evidence that the combination of botanicals and biopesticides will result in an increased bioactivity compared to single phytochemicals (Senthil-Nathan et al., 2005a; Senthil-Nathan and Kalaivani, 2005, 2006).
At present, botanical insecticides make <1% of the world’s pesticide market (Sola et al., 2014). Isolation of active principles and synthesis of secondary metabolites of botanicals against mosquito threat are very important for the management of vector borne diseases. The positive results of initial studies on larvicidal potential of botanicals encourage further interest to investigate the bioactive compounds. Identifying botanical insecticides that are effective as well as appropriate and adaptive to overcome ecological hazards, biodegradable, and have a broad spectrum of larvicidal properties will work as a new defense in the arsenal of insecticides and it may act as an appropriate alternative product to fight against vector-borne diseases.
Thus, the present review collects important information on plant extracts along with their active molecules as agents affecting the physiology and behavior of medically threatening mosquito vectors. Now collective efforts are needed to take advantage of the accumulated knowledge on phytochemical action on mosquitos in order to integrate their application in integrated pest management programs.
Author Contributions
SS-N collected all the information and wrote the review.
Conflict of Interest
The author declares that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Acknowledgments
I am very grateful to Dr. Sylvia Anton for her thorough and constructive review and suggestion on the first draft of the manuscript.
References
- Aciole S. D., Piccoli C. F., Costa E. V., Navarro-Silva M. A., Marques F. A., Sales Maia B. H., et al. (2011). Insecticidal activity of three species of Guatteria (Annonaceae) against Aedes aegypti (Diptera: Culicidae). Rev. Colomb. Entomol. 37 262–268. [Google Scholar]
- Aciole S. D. G. (2009). Avaliação da Actividade Insecticida Dos Óleos Essenciais Nas Plantas Amazônicas Annonaceae, Boraginaceae e de Mata Atlântica Myrtaceae como Alternativa De Controle às Larvas de Aedes aegypti (Linnaeus, 1762) (Diptera: Culicidae). Doctoral dissertation, Universidade de lisboa, Lisbon. [Google Scholar]
- Agra-Neto A. C., Napoleão T. H., Pontual E. V., Santos N. D. L., Luz L. A., Oliveira C. M. F., et al. (2014). Effect of moringa oleifera lectins on survival and enzyme activities of Aedes aegypti larvae susceptible and resistant to organophosphate. Parasitol. Res. 113 175–184. 10.1007/s00436-013-3640-8 [DOI] [PubMed] [Google Scholar]
- Aguiar J. C. D., Santiago G. M., Lavor P. L., Veras H. N., Ferreira Y. S., Lima M. A., et al. (2010). Chemical constituents and larvicidal activity of hymenaea courbaril fruit peel. Nat. Prod. Commu. 5 1977–1980. [PubMed] [Google Scholar]
- Aguilera L., Navarro A., Tacoronte J. E., Leyva M., Marquetti M. C. (2003). Efecto letal de myrtaceas cubanas sobre Aedes aegypti (Diptera: Culicidae). Rev. Cub. Med. Trop. 55 100–104. [PubMed] [Google Scholar]
- Albuquerque M. R. J. R., Costa S. M. O., Bandeira P. N., Santiago G. M. P., Andrade-Neto M., Silveira E. R., et al. (2007). Nematicidal and larvicidal activities of the essential oils from aerial parts of Pectis oligocephala and Pectis apodocephala Baker. An. Acad. Bras. Ciênc. 79 209–213. 10.1590/s0001-37652007000200003 [DOI] [PubMed] [Google Scholar]
- Albuquerque M. R. J. R., Silveira E. R., De A., Uchôa D. E., Lemos T. L. G., Souza E. B., et al. (2004). Chemical composition and larvicidal activity of the essential oils from Eupatorium betonicaeforme (DC) Baker (Asteraceae). J. Agric. Food Chem. 52 6708–6711. 10.1021/jf0352881 [DOI] [PubMed] [Google Scholar]
- Ali A., Tabanca N., Demirci B., Baser K. H. C., Ellis J., Gray S., et al. (2013). Composition, mosquito larvicidal, biting deterrent and antifungal activity of essential oils of different plant parts of Cupressus arizonica var. glabra (‘Carolina Sapphire’). Nat. Prod. Commun. 8 257–260. 10.1177/1934578X1300800232 [DOI] [PubMed] [Google Scholar]
- Al-Mehmadi R. M., Al-Khalaf A. A. (2010). Larvicidal and histological effects of melia azedarach extract on Culex quinquefasciatus say larvae (Diptera: Culicidae). J. King Saud. Univ. Sci. 22 77–85. 10.1016/j.jksus.2010.02.004 [DOI] [Google Scholar]
- Al-Mekhlafi F. A. (2018). Larvicidal, ovicidal activities and histopathological alterations induced by Carum copticum (Apiaceae) extract against Culex pipiens (Diptera: Culicidae). Saudi J. Biol. Sci. 25 52–56. 10.1016/j.sjbs.2017.02.010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amer A., Mehlhorn H. (2006). Repellency effect of forty-one essential oils against Aedes, Anopheles, and Culex mosquitoes. Parasitol. Res. 99:478. 10.1007/s00436-006-0184-1 [DOI] [PubMed] [Google Scholar]
- Anderson J. A., Coats J. R. (2012). Acetylcholinesterase inhibition by nootkatone and carvacrol in arthropods. Pestic. Biochem. Physiol. 102 124–128. 10.1016/j.pestbp.2011.12.002 [DOI] [Google Scholar]
- Ansari M., Mittal P., Razdan R., Sreehari U. (2005). Larvicidal and mosquito repellent activities of pine (pinus longifolia, family: pinaceae) oil. J. Vector Borne Dis. 42:95. [PubMed] [Google Scholar]
- Araújo E. C., Silveira E. R., Lima M. A. S., Neto M. A., de Andrade I. L., Lima M. A. A., et al. (2003). Insecticidal activity and chemical composition of volatile oils from hyptis martiusii Benth. J. Agri. Food Chem. 51 3760–3762. 10.1021/jf021074s [DOI] [PubMed] [Google Scholar]
- Arriaga A. M., Rodrigues F. E., Lemos T. L., de Oliveira M. D. C., Lima J. Q., Santiago G. M., et al. (2007). Composition and larvicidal activity of essential oil from stemodia maritima L. Nat. Prod. Commun. 2 1237–1239. [Google Scholar]
- Autran E., Neves I., Da Silva C., Santos G., Da Câmara C., Navarro D. (2009). Chemical composition, oviposition deterrent and larvicidal activities against Aedes aegypti of essential oils from Piper marginatum Jacq (Piperaceae). Biores. Technol. 100 2284–2288. 10.1016/j.biortech.2008.10.055 [DOI] [PubMed] [Google Scholar]
- Balasubramani G., Ramkumar R., Krishnaveni N., Sowmiya R., Deepak P., Arul D., et al. (2015). GC–MS analysis of bioactive components and synthesis of gold nanoparticle using Chloroxylon swietenia DC leaf extract and its larvicidal activity. J. Photochem. Photobiol B: Biol. 148 1–8. 10.1016/j.jphotobiol.2015.03.016 [DOI] [PubMed] [Google Scholar]
- Bandara K. P., Kumar V., Jacobsson U., Molleyres L.-P. (2000). Insecticidal piperidine alkaloid from Microcos paniculata stem bark. Phytochemistry 54 29–32. 10.1016/S0031-9422(00)00025-X [DOI] [PubMed] [Google Scholar]
- Banerji B., Nigam S. (1984). Wood constituents of meliaceae: a review. Fitoter 55 3–36. [Google Scholar]
- Baraza L. D., Joseph C. C., Munissi J. J. E., Nkunya M. H. H., Arnold N., Porzel A., et al. (2008). Antifungal rosane diterpenes and other constituents of Hugonia castaneifolia. Phytochemistry 69 200–205. 10.1016/j.phytochem.2007.06.021 [DOI] [PubMed] [Google Scholar]
- Benelli G., Pavela R., Canale A., Cianfaglione K., Ciaschetti G., Conti F., et al. (2017). Acute larvicidal toxicity of five essential oils (Pinus nigra, Hyssopus officinalis, Satureja montana, Aloysia citrodora and Pelargonium graveolens) against the filariasis vector Culex quinquefasciatus: synergistic and antagonistic effects. Parasitol. Inter. 66 166–171. 10.1016/j.parint.2017.01.012 [DOI] [PubMed] [Google Scholar]
- Benelli G., Pavela R., Petrelli R., Cappellacci L., Canale A., Senthil-Nathan S., et al. (2018). Not just popular spices! essential oils from Cuminum cyminum and Pimpinella anisum are toxic to insect pests and vectors without affecting non-target invertebrates. Indus. Crop. Prod. 124 236–243. 10.1016/j.indcrop.2018.07.048 [DOI] [Google Scholar]
- Bernick E. P., Moffett S. B., Moffett D. F. (2008). Ultrastructure and morphology of midgut visceral muscle in early pupal Aedes aegypti mosquitoes. Tissue Cell 40 127–141. 10.1016/j.tice.2007.11.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bohbot J. D., Lu T., Zwiebel L. J. (2010). Molecular Regulation of Olfaction In Mosquitoes. Olfaction in Vector-Host Interactions. Wageningen: Wageningen Academic Publishers, 17–38. [Google Scholar]
- Bora K. S., Sharma A. (2011). The genus Artemisia: a comprehensive review. Pharm. Biol. 49 101–109. 10.3109/13880209.2010.497815 [DOI] [PubMed] [Google Scholar]
- Borah R., Saikia K., Talukdar A. K., Kalita M. C. (2012). Chemical composition and biological activity of the leaf essential oil of Zanthoxylum oxyphyllum. Planta Med. 78:100 10.1055/s-0032-1307608 [DOI] [Google Scholar]
- Brown A. W. (1986). Insecticide resistance in mosquitoes: a pragmatic review. J. Amer. Mosq. Control Assoc. 2 123–140. [PubMed] [Google Scholar]
- Burfield T. (2000). Safety of essential oils. Inter. J. Arom. 10 16–29. [Google Scholar]
- Caballero R., Hoshi T., Kashyap A. (2008). Zombie lending and depressed restructuring in Japan. Am. Econ. Rev. 98 1943–1977. 10.1257/aer.98.5.1943 [DOI] [Google Scholar]
- Carreño-Otero A. L., Palacio-Cortés A. M., Navarro-Silva M. A., Kouznetsov V. V., Duque L. J. E., Jonny E. (2018). Behavior of detoxifying enzymes of Aedes aegypti exposed to girgensohnine alkaloid analog and Cymbopogon flexuosus essential oil. Comp. Biochem. Physiol. Part C 204 14–25. 10.1016/j.cbpc.2017.11.002 [DOI] [PubMed] [Google Scholar]
- Cavalcanti E. S. B., Morais S. M. D., Lima M. A., Santana E. W. P. (2004). Larvicidal activity of essential oils from Brazilian plants against Aedes aegypti L. Mem. Instit. Oswaldo Cruz 99 541–544. 10.1590/S0074-02762004000500015 [DOI] [PubMed] [Google Scholar]
- Cetin H., Yanikoglu A., Cilek J. E. (2011). Larvicidal activity of selected plant hydrodistillate extracts against the house mosquito, Culex pipiens, a West Nile virus vector. Parasitol. Res. 108 943–948. 10.1007/s00436-010-2136-z [DOI] [PubMed] [Google Scholar]
- Chauret D. C., Bernard C. B., Arnason J. T., Durst T., Krishnamurty H., Sanchez-Vindas P., et al. (1996). Insecticidal neolignans from Piper decurrens. J. Nat. Prod. Res. 59 152–155. 10.1021/np960036y [DOI] [PubMed] [Google Scholar]
- Chellappandian M., Senthil-Nathan S., Vasantha-Srinivasan P., Karthi S., Thanigaivel A., Kalaivani K., et al. (2019). Target and non-target botanical pesticides effect of Trichodesma indicum (Linn) R. Br. and their chemical derivatives against the dengue vector, Aedes aegypti L. Environ. Sci. Poll. Res. 26 16303–16315. 10.1007/s11356-019-04870-3 [DOI] [PubMed] [Google Scholar]
- Chellappandian M., Thanigaivel A., Vasantha-Srinivasan P., Edwin E. S., Ponsankar A., Selin-Rani S., et al. (2017). Toxicological effects of Sphaeranthus indicus linn (asteraceae) leaf essential oil against human disease vectors, Culex quinquefasciatus say and Aedes aegypti linn., and impacts on a beneficial mosquito predator. Environ. Sci. Poll. Res. 25 10294–10306. 10.1007/s11356-017-8952-2 [DOI] [PubMed] [Google Scholar]
- Chellappandian M., Vasantha-Srinivasan P., Senthil-Nathan S., Karthi S., Thanigaivel A., Ponsankar A., et al. (2018). Botanical essential oils and uses as mosquitocides and repellents against dengue. Environ. Intern. 113 214–230. 10.1016/j.envint.2017.12.038 [DOI] [PubMed] [Google Scholar]
- Cheng S. S., Chang H. T., Lin C. Y., Chen P. S., Huang C. G., Chen W. J., et al. (2009a). Insecticidal activities of leaf and twig essential oils from Clausena excavata against Aedes aegypti and Aedes albopictus larvae. Pestic. Manag. Sci. 65 339–343. 10.1002/ps.1693 [DOI] [PubMed] [Google Scholar]
- Cheng S. S., Chua M. T., Chang E. H., Huang C. G., Chen W. J., Chang S. T. (2009b). Variations in insecticidal activity and chemical compositions of leaf essential oils from Cryptomeria japonica at different ages. Bioresour. Technol. 100 465–470. 10.1016/j.biortech.2007.11.060 [DOI] [PubMed] [Google Scholar]
- Cheng S. S., Huang C. G., Chen Y. J., Yu J. J., Chen W. J., Chang S. T. (2009c). Chemical compositions and larvicidal activities of leaf essential oils from two Eucalyptus species. Bioresour. Technol. 100 452–456. 10.1016/j.biortech.2008.02.038 [DOI] [PubMed] [Google Scholar]
- Cheng S. S., Lin C. Y., Chung M. J., Liu Y. H., Huang C. G., Chang S. T. (2013). Larvicidal activities of wood and leaf essential oils and ethanolic extracts from Cunninghamia konishii Hayata against the dengue mosquitoes. Ind. Crops Prod. 47 310–315. 10.1016/j.indcrop.2013.03.016 [DOI] [Google Scholar]
- Cheng S. S., Liu J. Y., Tsai K. H., Chen W. J., Chang S. T. (2004). Chemical composition and mosquito larvicidal activity of essential oils from leaves of different Cinnamomum osmophloeum provenances. J. Agri. Food Chem. 52 4395–4400. 10.1021/jf0497152 [DOI] [PubMed] [Google Scholar]
- Choochote W., Chaiyasit D., Kanjanapothi D., Rattanachanpichai E., Jitpakdi A., Tuetun B., et al. (2005). Chemical composition and anti-mosquito potential of rhizome extract and volatile oil derived from Curcuma aromatica against Aedes aegypti (Diptera: Culicidae). J. Vector Biol. 30 302–309. [PubMed] [Google Scholar]
- Chung I. M., Seo S. H., Kang E. Y., Park S. D., Park W. H., Moon H. I. (2009). Chemical composition and larvicidal effects of essential oil of Dendropanax morbifera against Aedes aegypti L. Biochem. Syst. Ecol. 37 470–473. 10.1016/j.bse.2009.06.004 [DOI] [Google Scholar]
- Clark A. G. (1990). “The glutathione S-transferases and resistance to insecticides,” in Glutathione S-transferases and Drug Resistance, eds Hayes J. D., Pickett C. B., Mantle T. J. (London: Taylor and Francis; ), 369–378. [Google Scholar]
- Conti B., Canale A., Bertoli A., Gozzini F., Pistelli L. (2010). Essential oil composition and larvicidal activity of six mediterranean aromatic plants against the mosquito Aedes albopictus (Diptera: Culicidae). Parasitol. Res. 107 1455–1461. 10.1007/s00436-010-2018-4 [DOI] [PubMed] [Google Scholar]
- Costa J. G., Pessoa O. D., Menezes E. A., Santiago G. M., Lemos T. L. (2004). Composition and larvicidal activity of essential oils from heartwood of Auxemma glazioviana taub (Boraginaceae). Flav. Frag. J. 19 529–531. 10.1002/ffj.1332 [DOI] [Google Scholar]
- Costa J. G. M., Rodrigues F. F. G., Angélico E. C., Silva M. R., Mota M. L., Santos N. K. A., et al. (2005). Estudo químico-biológico dos óleos essenciais de hyptis martiusii, lippia sidoides e Syzigium aromaticum frente às larvas do Aedes aegypti. Rev. Bras. Farmacogn. 15 304–309. [Google Scholar]
- Costa J. G. M., Rodrigues F. F. G., Sousa E. O., Junior D. M. S., Campos A. R., Coutinho H. D. M., et al. (2010). Composition and larvicidal activity of the essential oils of Lantana camara and Lantana montevidensis. Chem. Nat. Comp. 46 313–315. 10.1007/s10600-010-9601-x [DOI] [Google Scholar]
- Costa M., Cossolin J., Pereira M., Sant’Ana A., Lima M., Zanuncio J., et al. (2014). Larvicidal and cytotoxic potential of squamocin on the midgut of Aedes aegypti (Diptera: Culicidae). Toxins 6 1169–1176. 10.3390/toxins6041169 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Costa M. S., Pinheiro D. O., Serrão J. E., Pereira M. J. B. (2012). Morphological changes in the midgut of Aedes aegypti L. (Diptera: Culicidae) larvae following exposure to an Annona coriacea (Magnoliales: Annonaceae) extract. Neotrop. Entomol. 41 311–314. 10.1007/s13744-012-0050-z [DOI] [PubMed] [Google Scholar]
- Costa M. S., Santana A. E., Oliveira L. L., Zanuncio J. C., Serrão J. E. (2017). Toxicity of squamocin on Aedes aegypti larvae, its predators and human cells. Pestic. Manag. Sci. 73 636–640. 10.1002/ps.4350 [DOI] [PubMed] [Google Scholar]
- da Silva Costa M., de Paula S. O., Martins G. F., Zanuncio J. C., Santana A. E. G., Serrão J. E. (2018). Modes of action of squamocin in the anal papillae of Aedes aegypti larvae. Physiol. Mol. Plant Pathol. 101 172–177. 10.1016/j.pmpp.2017.04.001 [DOI] [Google Scholar]
- David J. P., Ismail H. M., Chandor-Proust A., Paine M. J. I. (2013). Role of cytochrome P450s in insecticide resistance: impact on the control of mosquito-borne diseases and use of insecticides on Earth. Philos Trans. R Soc. Lond B Biol. Sci. 3:68. 10.1098/rstb.2012.0429 [DOI] [PMC free article] [PubMed] [Google Scholar]
- de Castro D. S. B., da Silva D. B., Tibúrcio J. D., Sobral M. E. G., Ferraz V., Taranto A. G., et al. (2016). Larvicidal activity of essential oil of Peumus boldus Molina and its ascaridole-enriched fraction against Culex quinquefasciatus. Exp. Parasitol. 171 84–90. 10.1016/j.exppara.2016.10.008 [DOI] [PubMed] [Google Scholar]
- de Melo A. R., Garcia I. J. P., Serrão J. E., Santos H. L., dos Santos Lima L. A. R., Alves S. N. (2018). Toxicity of different fatty acids and methyl esters on Culex quinquefasciatus larvae. Ecotoxicol. Environ. Saf. 154 1–5. 10.1016/j.ecoenv.2018.02.009 [DOI] [PubMed] [Google Scholar]
- de Morais S. M., Facundo V. A., Bertini L. M., Cavalcanti E. S. B., Dos Anjos Júnior J. F., Ferreira S. A., et al. (2007). Chemical composition and larvicidal activity of essential oils from Piper species. Biochem. Syst. Ecol. 35 670–675. 10.1016/j.bse.2007.05.002 [DOI] [Google Scholar]
- Dharmagadda V. S. S., Naik S. N., Mittal P. K., Vasudevan P. (2005). Larvicidal activity of Tagetes patula essential oil against three mosquito species. Bioresour. Technol. 96 1235–1240. 10.1016/j.biortech.2004.10.020 [DOI] [PubMed] [Google Scholar]
- do Nascimento J. C., David J. M., Barbosa L. C., De Paula V. F., Demuner A. J., David J. P., et al. (2013). Larvicidal activities and chemical composition of essential oils from Piper klotzschianum (Kunth) C. DC. (Piperaceae). Pestic. Manag. Sci. 69 1267–1271. 10.1002/ps.3495 [DOI] [PubMed] [Google Scholar]
- Dua V. K., Kumar A., Pandey A. C., Kumar S. (2013). Insecticidal and genotoxic activity of Psoralea corylifolia Linn (Fabaceae) against Culex quinquefasciatus Say, 1823. Parasit. Vectors 6:30. 10.1186/1756-3305-6-30 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Edwin E.-S., Vasantha-Srinivasan P., Senthil-Nathan S., Thanigaivel A., Ponsankar A., Pradeepa V., et al. (2016). Anti-dengue efficacy of bioactive andrographolide from Andrographis paniculata (Lamiales: Acanthaceae) against the primary dengue vector Aedes aegypti (Diptera: Culicidae). Acta Trop. 163 167–178. 10.1016/j.actatropica.2016.07.009 [DOI] [PubMed] [Google Scholar]
- Elumalai D., Kaleena P. K., Ashok K., Suresh A., Hemavathi M. (2016). Green synthesis of silver nanoparticle using Achyranthes aspera and its larvicidal activity against three major mosquito vectors. Eng. Agric. Environ. Food 9 1–8. 10.1016/j.eaef.2015.08.002 [DOI] [Google Scholar]
- Enayati A. A., Ranson H., Hemingway J. (2005). Mini review: insectglutathione transferases and insecticide resistance. Insect. Mol. Biol. 14 3–8. 10.1111/j.1365-2583.2004.00529.x [DOI] [PubMed] [Google Scholar]
- Eneh L. K., Okal M. N., Borg-Karlson A. K., Fillinger U., Lindh J. M. (2016). Gravid Anopheles gambiae sensu stricto avoid ovipositing in Bermuda grass hay infusion and it’s volatiles in two choice egg-count bioassays. Malaria J. 15:276 10.1186/s12936-016-1330-6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evergetis E., Michaelakis A., Haroutounian S. A. (2012). Essential Oils of Umbelliferae (Apiaceae) Family Taxa as Emerging Potent Agents for Mosquito Control. London: InTechOpen. [Google Scholar]
- Evergetis E., Michaelakis A., Kioulos E., Koliopoulos G., Haroutounian S. (2009). Chemical composition and larvicidal activity of essential oils from six Apiaceae family taxa against the West Nile virus vector Culex pipiens. Parasitol. Res. 105 117–124. 10.1007/s00436-009-137 [DOI] [PubMed] [Google Scholar]
- Fallatah H. I. (2014). Noninvasive biomarkers of liver fibrosis: an overview. Adv. Hepatol. 2014:8 10.1155/2014/357287 [DOI] [Google Scholar]
- Fallatah S. A. (2010). Histopathological effects of fenugreek (Trigonella foenumgraceum) extracts on the larvae of the mosquito Culex quinquefasciatus. J. Arab Soc. Med. Res. 5 123–130. [Google Scholar]
- Fayemiwo K. A., Adeleke M. A., Okoro O. P., Awojide S. H., Awoniyi I. O. (2014). Larvicidal efficacies and chemical composition of essential oils of Pinus sylvestris and Syzygium aromaticum against mosquitoes. Asian Pac. J. Trop. Biomed. 4 30–34. 10.1016/S2221-1691(14)60204-5 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feitosa E., Arriaga A., Santiago G. M., De Lemos T. L., Oliveira M. C. F. (2009). Chemical composition and larvicidal activity of Rollinia leptopetala (Annonaceae). J. Braz. Chem. Soc. 20 375–378. 10.1590/S0103-50532009000200024 [DOI] [Google Scholar]
- Feitosa E. M. A., Arriaga A. M. C., Lemos T. L. G., Lima J. Q., Nunes Vasconcelos J., Oliveira M. C. F., et al. (2007). Zanthoxylum articulatum engler (Rutaceae) essential oil: chemical composition and larvicidal activity. J. Essen. Oil Res. 19 384–386. 10.1080/10412905.2007.9699311 [DOI] [Google Scholar]
- Fradin M. S., Day J. F. (2002). Comparative efficacy of insect repellents against mosquito bites. New Engl. J. Med. 347 13–18. 10.1056/NEJMoa011699 [DOI] [PubMed] [Google Scholar]
- François G., van Looveren M., Timperman G., Chimanuka B., Akéassi L., Holenz J., et al. (1996). Larvicidal activity of the naphthylisoquinoline alkaloid dioncophylline a against the malaria vector Anopheles stephensi. J. Ethnopharm. 54 125–130. 10.1016/S0378-8741(96)01459-6 [DOI] [PubMed] [Google Scholar]
- Gbolade A. A., Lockwood G. B. (2008). Toxicity of Ocimum sanctum L. essential oil to Aedes aegypti larvae and its chemical composition. J. Essen. Oil Bear. Plants 11 148–153. 10.1080/0972060X.2008.10643611 [DOI] [Google Scholar]
- Ghosh A., Chowdhury N., Chandra G. (2012). Plant extracts as potential mosquito larvicides. Ind. J. Med. Res. 135:581. [PMC free article] [PubMed] [Google Scholar]
- Giatropoulos A., Kimbaris A., Michaelakis A., Papachristos D. P., Polissiou M. G., Emmanouel N. (2018). Chemical composition and assessment of larvicidal and repellent capacity of 14 Lamiaceae essential oils against Aedes albopictus. Parasitol. Res. 117 1953–1964. 10.1007/s00436-018-5892-9 [DOI] [PubMed] [Google Scholar]
- Giatropoulos A., Pitarokili D., Papaioannou F., Papachristos D. P., Koliopoulos G., Emmanouel N., et al. (2013). Essential oil composition, adult repellency and larvicidal activity of eight Cupressaceae species from Greece against Aedes albopictus (Diptera: Culicidae). Parasitol. Res. 112 1113–1123. 10.1007/s00436-012-3239-5 [DOI] [PubMed] [Google Scholar]
- Govindarajan M., Benelli G. (2016). Artemisia absinthium-borne compounds as novel larvicides: effectiveness against six mosquito vectors and acute toxicity on non-target aquatic organisms. Parasitol. Res. 115 4649–4661. 10.1007/s00436-016-5257-1 [DOI] [PubMed] [Google Scholar]
- Govindarajan M., Rajeswary M., Benelli G. (2016). Chemical composition, toxicity and non-target effects of Pinus kesiya essential oil: an eco-friendly and novel larvicide against malaria, dengue and lymphatic filariasis mosquito vectors. Ecotoxicol. Environ. Saf. 129 85–90. 10.1016/j.ecoenv.2016.03.007 [DOI] [PubMed] [Google Scholar]
- Govindarajan M., Sivakumar R., Rajeswari M., Yogalakshmi K. (2012). Chemical composition and larvicidal activity of essential oil from Mentha spicata (Linn.) against three mosquito species. Parasitol. Res. 110 2023–2032. 10.1007/s00436-011-2731-7 [DOI] [PubMed] [Google Scholar]
- Govindarajan M., Sivakumar R., Rajeswary M., Veerakumar K. (2013). Mosquito larvicidal activity of thymol from essential oil of Coleus aromaticus benth. Against Culex tritaeniorhynchus, Aedes albopictus, and Anopheles subpictus (Diptera: Culicidae). Parasitol. Res. 112 3713–3721. 10.1007/s00436-013-3557-2 [DOI] [PubMed] [Google Scholar]
- Hemingway J., Hawkes N., Mccarroll L., Ranson H. (2004). The molecular basis of insecticide resistance in mosquitoes. Insect. Biochem. Mol. Biol. 34 653–655. 10.1016/j.ibmb.2004.03.018 [DOI] [PubMed] [Google Scholar]
- Hemingway J., Ranson H. (2000). Insecticide resistance in insect vectors of human disease. Annu. Rev. Entomol. 45 371–391. 10.1146/annurev.ento.45.1.371 [DOI] [PubMed] [Google Scholar]
- Heuskin S., Godin B., Leroy P., Capella Q., Wathelet J. P., Verheggen F., et al. (2009). Fast gas chromatography characterisation of purified semiochemicals from essential oils of Matricaria chamomilla L. (Asteraceae) and Nepeta cataria L.(Lamiaceae). J. Chromatograph. A 1216 2768–2775. 10.1016/j.chroma.2008.09.109 [DOI] [PubMed] [Google Scholar]
- Hong S., Guo Q., Wang W., Hu S., Fang F., Lv Y., et al. (2014). Identification of differentially expressed micro RNAs in Culex pipiens and their potential roles in pyrethroid resistance. Insect. Biochem. Mol. Biol. 55 39–50. 10.1016/j.ibmb.2014.10.007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ioset J. R., Marston A., Gupta M. P., Hostettmann K. (2000). Antifungal and larvicidal cordiaquinones from the roots of Cordia curassavica. Phytochem. 53 613–617. 10.1016/S0031-9422(99)00604-4 [DOI] [PubMed] [Google Scholar]
- Jaenson T. G., Pålsson K., Borg-Karlson A. K. (2006). Evaluation of extracts and oils of mosquito (Diptera: Culicidae) repellent plants from Sweden and Guinea-Bissau. J. Med. Entomol. 43 113–119. 10.1093/jmedent/43.1.113 [DOI] [PubMed] [Google Scholar]
- Jantan I. B., Yalvema M. F., Ahmad N. W., Jamal J. A. (2005). Insecticidal activities of the leaf oils of eight Cinnamomum. species against Aedes aegypti and Aedes albopictus. Pharm. Biol. 43 526–532. 10.1080/13880200500220771 [DOI] [Google Scholar]
- Jayaprakasha G., Singh R., Pereira J., Sakariah K. (1997). Limonoids from Citrus reticulata and their moult inhibiting activity in mosquito Culex quinquefasciatus larvae. Phytochemistry 44 843–846. 10.1016/S0031-9422(96)00589-4 [DOI] [PubMed] [Google Scholar]
- Jinu U., Rajakumaran S., Senthil-Nathan S., Geetha N., Venkatachalam P. (2018). Potential larvicidal activity of silver nanohybrids synthesized using leaf extracts of Cleistanthus collinus (Roxb.) Benth. ex Hook. f. and Strychnos nux-vomica L. nux-vomica against dengue, Chikungunya and Zika vectors. Physiol. Mol. Plant Pathol. 101 163–171. 10.1016/j.pmpp.2017.05.003 [DOI] [Google Scholar]
- Jones G., Smith J., Watson K. (2007). “Bioactive properties of native Australian medicinal plants,” in Advances in Medicinal Plant Research, eds Acharya S. N. Thomas J. E. (Thiruvananthapuram: Research Signpost; ), 257–286. [Google Scholar]
- Joseph C. C., Ndoile M. M., Malima R. C., Nkunya M. H. H. (2004). Larvicidal and mosquitocidal extracts, a coumarin, isoflavonoids and pterocarpans from neorautanenia mitis. Trans. R. Soc.Trop. Med. Hyg. 98 451–455. 10.1016/j.trstmh.2003.10.008 [DOI] [PubMed] [Google Scholar]
- Jukic M., Politeo O., Maksimovic M., Milos M., Milos M. (2007). In Vitro acetylcholinesterase inhibitory properties of thymol, carvacrol and their derivatives thymoquinone and thymohydroquinone. Phytother. Res. 21 259–261. 10.1002/ptr.2063 [DOI] [PubMed] [Google Scholar]
- Kabir K., Khan A., Mosaddik M. (2003). Goniothalamin–a potent mosquito larvicide from Bryonopsis laciniosa L. J Appl. Entomol 127 112–115. 10.1046/j.1439-0418.2003.00716.x [DOI] [Google Scholar]
- Kalaivani K., Senthil-Nathan S., Murugesan A. G. (2012). Biological activity of selected Lamiaceae and Zingiberaceae plant essential oils against the dengue vector Aedes aegypti L. (Diptera: Culicidae). Parasitol. Res. 110 1261–1268. 10.1007/s00436-011-2623-x [DOI] [PubMed] [Google Scholar]
- Kamala-Jayanthi P. D., Aurade R. M., Kempraj V., Chakravarthy A. K., Verghese A. (2015). “Glimpses of semiochemical research applications in Indian horticulture: present status and future perspectives,” in New Horizons in Insect Science: Towards Sustainable Pest Management, ed. Chakravarthy A. (New Delhi: Springer; ), 239–257. 10.1007/978-81-322-2089-3_22 [DOI] [Google Scholar]
- Kamaraj C., Deepak P., Balasubramani G., Karthi S., Arul D., Aiswarya D., et al. (2018). Target and non-target toxicity of fern extracts against mosquito vectors and beneficial aquatic organisms. Ecotoxicol. Environ. Saf. 161 221–230. 10.1016/j.ecoenv.2018.05.062 [DOI] [PubMed] [Google Scholar]
- Kannathasan K., Senthilkumar A., Venkatesalu V. (2011). Mosquito larvicidal activity of methyl-p-hydroxybenzoate isolated from the leaves of Vitex trifolia Linn. Acta Trop. 120 115–118. 10.1016/j.actatropica.2011.07.001 [DOI] [PubMed] [Google Scholar]
- Karunaratne S. H. P. P., Jayawardena K. G. I., Hemingway J., Ketterman A. J. (1993). Characterisation of a B-type esterase involved in insecticide resistance from the mosquito Culex quinquefasciatus. Biochem. J. 294 575–579. 10.1042/bj2940575 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaur H., Arora D. S., Sharma V. (2014). Isolation, purification and characterization of antimicrobial compound 6-[1, 2-dim.ethyl-6-(2-methyl-allyloxy)-hexyl]-3-(2-methoxy-phenyl)-chromen-4-one from Penicillium sp. HT 28. Appl. Biochem. Biotechnol. 173 1963–1976. 10.1007/s12010-014-0979-Y [DOI] [PubMed] [Google Scholar]
- Kelm M. A., Nair M. G., Schutzki R. A. (1997). Mosquitocidal compounds from Magnolia salicifolia. Inter. J. Pharm. 35 84–90. 10.1076/phbi.35.2.84.13279 [DOI] [Google Scholar]
- Kim S. I., Ahn Y.-J. (2017). Larvicidal activity of lignans and alkaloid identified in Zanthoxylum piperitum bark toward insecticide-susceptible and wild Culex pipiens pallens and Aedes aegypti. Parasit. Vectors 10:221. 10.1186/s13071-017-2154-0 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kimbaris A. C., Koliopoulos G., Michaelakis A., Konstantopoulou M. A. (2012). Bioactivity of Dianthus caryophyllus, Lepidium sativum, Pimpinella anisum, and Illicium verum essential oils and their major components against the West Nile vector Culex pipiens. Parasitol. Res. 111 2403–2410. 10.1007/s00436-012-3097-1 [DOI] [PubMed] [Google Scholar]
- Kiran S. R., Bhavani K., Devi P. S., Rao B. R., Reddy K. J. (2006). Composition and larvicidal activity of leaves and stem essential oils of Chloroxylon swietenia DC against Aedes aegypti and Anopheles stephensi. Bioresour. Technol. 97 2481–2484. 10.1016/j.biortech.2005.10.003 [DOI] [PubMed] [Google Scholar]
- Koodalingam A., Deepalakshmi R., Ammu M., Rajalakshmi A. (2014). Effects of neemazal on marker enzymes and hemocyte phagocytic activity of larvae and pupae of the vector mosquito Aedes aegypti. J. Asia Pac. Entomol. 17 175–181. 10.1016/j.aspen.2013.12.007 [DOI] [Google Scholar]
- Koutsaviti K., Giatropoulos A., Pitarokili D., Papachristos D., Michaelakis A., Tzakou O. (2015). Greek Pinus essential oils: larvicidal activity and repellency against Aedes albopictus (Diptera: Culicidae). Parasitol. Res. 114 583–592. 10.1007/s00436-014-4220-2 [DOI] [PubMed] [Google Scholar]
- Kulkarni R. R., Pawar P. V., Joseph M. P., Akulwad A. K., Sen A., Joshi S. P. (2013). Lavandula gibsoni and Plectranthus mollis essential oils: chemical analysis and insect control activities against Aedes aegypti, Anopheles sfttephensi and Culex quinquefasciatus. J. Pestic. Sci. 86 713–718. 10.1007/s10340-013-0502-1 [DOI] [Google Scholar]
- Kuo P. M., Chu F. H., Chang S. T., Hsiao W. F., Wang S. Y. (2007). Insecticidal activity of essential oil from Chamaecyparis formosensis matsum. Holzforschung 61 595–599. 10.1515/HF.2007.087 [DOI] [Google Scholar]
- Larson M. J., Baldwin S. A., Good D. A., Fair J. E. (2010). Temporal stability of the error-related negativity (ERN) and post-error positivity (Pe): the role of number of trials. Psychophysiology 47 1167–1171. 10.1111/j.1469-8986.2010.01022.x [DOI] [PubMed] [Google Scholar]
- Lee J., Iwai T., Yokota T., Yamashita M. (2003). Temporally and spatially selective loss of Rec8 protein from meiotic chromosomes during mammalian meiosis. J. Cell Sci. 116 2781–2790. 10.1242/jcs.00495 [DOI] [PubMed] [Google Scholar]
- Lee S. E. (2000). Mosquito larvicidal activity of pipernonaline, a piperidine alkaloid derived from long pepper Piper longum. J. Am.Mosq. Control Assoc. 16 245–247. [PubMed] [Google Scholar]
- Leyva M., Del Carmen Marquetti M., Tacoronte J. E., Scull R., Tiomno O., Mesa A., et al. (2009b). Larvicidal activity of plant essential oils Aedes aegypti (L.) (Diptera: Culicidae). Rev. Bioméd. 20 5–13. [Google Scholar]
- Leyva M., Marquetti M. C., Tacoronte J. E., Scull R., Tiomno O., Mesa A., et al. (2009a). Actividad larvicida de aceites esenciales de plantas contra Aedes aegypti (L.) (Diptera: Culicidae). Rev. Biomed. 20 5–13. 10.32776/revbiomed.v20i1.529 [DOI] [Google Scholar]
- Leyva M., Tacoronte J. E., Marquetti M. D. C., Scull R., Montada D., Rodríguez Y., et al. (2008). Actividad insecticida de aceites esenciales de plantas en larvas de Aedes aegypti (Diptera: Culicidae). Rev. Cubana Med. Trop. 60 78–82. [Google Scholar]
- Lija-Escaline J., Senthil-Nathan S., Thanigaivel A., Pradeepa V., Vasantha-Srinivasan P., Ponsankar A., et al. (2015). Physiological and biochemical effects of botanical extract from Piper nigrum Linn (Piperaceae) against the dengue vector Aedes aegypti Liston (Diptera: Culicidae). Parasitol. Res. 114 4239–4249. 10.1007/s00436-015-4662-4661 [DOI] [PubMed] [Google Scholar]
- Lima M. A., De Oliveira F. F. M., Gomes G. A., Lavor P. L., Santiago G. M., Nagao-Dias A. T., et al. (2011). Evaluation of larvicidal activity of the essential oils of plants species from Brazil against Aedes aegypti (Diptera: Culicidae). Afr. J. Biotechnol. 10:11716. [Google Scholar]
- Lima-Santos L., Barreto Brandão L., Lopes Martins R., de Menezes Rabelo E., Lobato Rodrigues A. B., da Conceição Vieira Araújo C. M., et al. (2019). Evaluation of the larvicidal potential of the essential oil Pogostemon cablin (Blanco) Benth in the control of Aedes aegypti. Pharmaceuticals 12:53. 10.3390/ph12020053 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu X. C., Liu Q., Zhou L., Liu Z. L. (2014a). Evaluation of larvicidal activity of the essential oil of Allium macrostemon Bunge and its selected major constituent compounds against Aedes albopictus (Diptera: Culicidae). Parasit. Vectors 7:1. 10.1186/1756-3305-7-184 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu X. C., Liu Q. Y., Zhou L., Liu Q. R., Liu Z. L. (2014b). Chemical composition of Zanthoxylum avicennae essential oil and its larvicidal activity on Aedes albopictus Skuse. Trop. J. Pharm. Res. 13 399–404. [Google Scholar]
- Lounibos L. P. (2002). Invasions by insect vectors of human disease. Ann. Rev. Entomol. 47 33–66. 10.1146/annurev.ento.47.091201.145206 [DOI] [PubMed] [Google Scholar]
- Lucia A., Audino P. G., Seccacini E., Licastro S., Zerba E., Masuh H. (2007). Larvicidal effect of Eucalyptus grandis essential oil and turpentine and their major components on Aedes aegypti larvae. J. Am. Mosq. Control Assoc. 23 299–304. 10.2987/8756-971X200723 [DOI] [PubMed] [Google Scholar]
- Lucia A., Juan L. W., Zerba E. N., Harrand L., Marcó M., Masuh H. M. (2012). Validation of models to estimate the fumigant and larvicidal activity of Eucalyptus essential oils against Aedes aegypti (Diptera: Culicidae). Parasitol. Res. 110 1675–1686. 10.1007/s00436-011-2685-9 [DOI] [PubMed] [Google Scholar]
- Lucia A., Licastro S., Zerba E., Masuh H. (2008). Yield, chemical composition, and bioactivity of essential oils from 12 species of Eucalyptus on Aedes aegypti larvae. Entomol. Exp. Appl. 129 107–114. 10.1111/j.1570-7458.2008.00757.x [DOI] [Google Scholar]
- Magalhães A. M. L., da Paz Lima M., Marques O. M. M., Facanali R., Pinto A. C. D. S., Pedro T. W. (2010). Chemical composition and larvicidal activity against Aedes aegypti larvae of essential oils from four Guarea species. Molecules 15 5734–5741. 10.3390/molecules15085734 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mansour S., Messeha S., El-Gengaihi S. (2000). Botanical biocides. 4. Mosquitocidal activity of certain Thymus capitatus constituents. J. Nat. Toxins 9 49–62. [PubMed] [Google Scholar]
- Mar J. M., Silva L. S., Azevedo S. G., França L. P., Goes A. F., dos Santos A. L., et al. (2018). Lippia origanoides essential oil: an efficient alternative to control Aedes aegypti, Tetranychus urticae and Cerataphis lataniae. Ind. Crops Prod. 111 292–297. 10.1016/j.indcrop.2017.10.033 [DOI] [Google Scholar]
- Maradufu A., Lubega R., Dorn F. (1978). Isolation of (5E)-Ocimerone, a mosquito larvicide from Tagetes minuta. Lloydia 41 181–183. [Google Scholar]
- Marques M. M., Morais S. M., Vieira I. G., Vieira M. G., Silva A. R. A., De Almeida R. R., et al. (2011). Larvicidal activity of tagetes erecta against Aedes aegypti. J. Am. Mosq. Control Assoc. 27 156–158. 10.2987/10-6056.1 [DOI] [PubMed] [Google Scholar]
- Massebo F., Tadesse M., Bekele T., Balkew M., Gebre-Michael T. (2009). Evaluation on larvicidal effects of essential oils of some local plants against anopheles arabiensis Patton and Aedes aegypti linnaeus (Diptera, Culicidae) in ethiopia. Afr. J. Biotechnol. 8 4183–4188. [Google Scholar]
- Massoud R., Askov K., Reinke J., Franco L. M., Bornstein T., Knebel E., et al. (2001). A Modern Paradigm for Improving Healthcare Quality. Quality Assurance Monograph. Available at: http:// www.qaproject.org (accessed March 19, 2019). [Google Scholar]
- Michaelakis A., Papachristos D., Kimbaris A., Koliopoulos G., Giatropoulos A., Polissiou M. G. (2009). Citrus essential oils and four enantiomeric pinenes against Culex pipiens (Diptera: Culicidae). Parasitol. Res. 105:769. 10.1007/s00436-009-1452-7 [DOI] [PubMed] [Google Scholar]
- Moon H.-I. (2011). Larvicidal activity of major essential oils from stems of Allium monanthum maxim. against Aedes aegypti L. J. Enzyme Inhib. Med. Chem. 26 827–830. 10.3109/14756366.2011.558842 [DOI] [PubMed] [Google Scholar]
- Morais S. M., Cavalcanti E. S., Bertini L. M., Oliveira C. L. L., Rodrigues J. R. B., Cardoso J. H. L. (2006). Larvicidal activity of essential oils from Brazilian Croton species against Aedes aegypti L. J. Am. Mosq. Control Assoc. 22 161–165. 10.2987/8756-971X200622 [DOI] [PubMed] [Google Scholar]
- Mozaffari E., Abai M. R., Khanavi M., Vatandoost H., Sedaghat M. M., Moridnia A., et al. (2014). Chemical composition, larvicidal and repellency properties of Cionura erecta (L.) griseb. Against malaria vector, Anopheles stephensi Liston (Diptera: Culicidae). J. Arthr. Dis. 8:147. [PMC free article] [PubMed] [Google Scholar]
- Muema J. M., Bargul J. L., Njeru S. N., Onyango J. O., Imbahale S. S. (2017). Prospects for malaria control through manipulation of mosquito larval habitats and olfactory-mediated behavioural responses using plant-derived compounds. Parasit. Vectors. 10:184. 10.1186/s13071-017-2122-8 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muleya V., Hayeshi R., Ranson H., Abegaz B., Bezabih M. T., Robert M., et al. (2008). Modulation of Anopheles gambiae epsilon glutathione transferase activity by plant natural products in vitro. J. Enzyme Inhib. Med. Chem. 23 391–399. 10.1080/14756360701546595 [DOI] [PubMed] [Google Scholar]
- Mulla M. S., Su T. (1999). Activity and biological effects of neem products against arthropods of medical and veterinary importance. J. Am. Mosq. Control Assoc. 15 133–152. [PubMed] [Google Scholar]
- Munda S., Saikia P., Lal M. (2018). Chemical composition and biological activity of essential oil of Kaempferia galanga: a review. J. Essen. Oil Res. 30 303–308. 10.1080/10412905.2018.1486240 [DOI] [Google Scholar]
- Murugan K., Jaganathan A., Rajaganesh R., Suresh U., Madhavan J., Senthil-Nathan S., et al. (2018a). Poly (styrene sulfonate)/poly (allylamine hydrochloride) encapsulation of TiO2 nanoparticles boosts their toxic and repellent activity against zika virus mosquito vectors. J. Cluster Sci. 29 27–39. 10.1007/s10876-017-1300-3 [DOI] [Google Scholar]
- Murugan K., Roni M., Panneerselvam C., Suresh U., Rajaganesh R., Aruliah R., et al. (2018b). Sargassum wightii-synthesized ZnO nanoparticles reduce the fitness and reproduction of the malaria vector Anopheles stephensi and cotton bollworm Helicoverpa armigera. Physiol. Mol. Plant Pathol. 101 202–213. 10.1016/j.pmpp.2017.02.004 [DOI] [Google Scholar]
- Mwangi J., Addae-Mensah I., Muriuki G., Munavu R., Lwande W. A., Hassanali A. (1992). Essential oils of lippia species in Kenya. IV: Maize weevil (Sitophilus zeamais) repellancy and larvicidal activity. Inter. J. Pharm. 30 9–16. 10.3109/13880209209054622 [DOI] [Google Scholar]
- Nagella P., Ahmad A., Kim S.-J., Chung I.-M. (2012). Chemical composition, antioxidant activity and larvicidal effects of essential oil from leaves of Apium graveolens. Immunopharmacol. Immunotoxicol. 34 205–209. 10.3109/08923973.2011.592534 [DOI] [PubMed] [Google Scholar]
- Napoleão T. H., Pontual E. V., Lima T. A., Santos N. D. L., Sá R. A., Coelho L. C. B. B., et al. (2012). Effect of Myracrodruon urundeuva leaf lectin on survival and digestive enzymes of Aedes aegypti larvae. Parasitol. Res. 110 609–616. 10.1007/s00436-011-2529-7 [DOI] [PubMed] [Google Scholar]
- Naqvi S. (1987). “Biological evaluation of fresh neem extracts and some neem components, with reference to abnormalities and esterase activity in insects,” in Proceedings of the First International Neem Conference, (Nairobi: ), 315–330. [Google Scholar]
- Ndung’u M., Hassanali A., Hooper A. M., Chhabra S., Miller T. A., Paul R. L., et al. (2003). Ring A-seco mosquito larvicidal limonoids from turraea wakefieldii. Phytochemistry 64 817–823. 10.1016/j.phytochem.2003.08.020 [DOI] [PubMed] [Google Scholar]
- Ndung’u M. W., Kaoneka B., Hassanali A., Lwande W., Hooper A. M., Tayman F., et al. (2004). New mosquito larvicidal tetranortriterpenoids from Turraea wakefieldii and Turraea floribunda. J. Agric. Food Chem. 52 5027–5031. 10.1021/jf049474y [DOI] [PubMed] [Google Scholar]
- Neira-Oviedo M., Vanekeris L., Corena-Mcleod M. D. P., Linser P. J. (2008). A microarray-based analysis of transcriptional compartmentalization in the alimentary canal of Anopheles gambiae (Diptera: Culicidae) larvae. Insect Mol. Biol. 17 61–72. 10.1111/j.1365-2583.2008.00779.x [DOI] [PubMed] [Google Scholar]
- Nikkon F., Salam K. A., Yeasmin T., Mosaddik A., Khondkar P., Haque M. E. (2010). Mosquitocidal triterpenes from the stem of Duranta repens. Pharm. Biol. 48 264–268. 10.3109/13880200903096570 [DOI] [PubMed] [Google Scholar]
- Niroumand M. C., Heydarpour F., Farzaei M. H. (2018). Pharmacological and therapeutic effects of Vitex agnus-castus L.: A review. Pharm. Rev. 12:103 10.4103/phrev.phrev_22_17 [DOI] [Google Scholar]
- Nivsarkar M., Kumar G.-P., Laloraya M., Laloraya M.-M. (1991). Superoxide dismutase in the anal gills of the mosquito larvae of Aedes aegypti: its inhibition by alpha-terthienyl. Arch. Insect. Biochem. Physiol. 4 249–255. 10.1002/arch.940160404 [DOI] [PubMed] [Google Scholar]
- Nyahanga T., Jondika J. I., Manguro L. O. A., Orwa J. A. (2010). Chemical composition, antiplasmodial, larvicidal and antimicrobial activities of essential oils of Toddalia asiatica leaves and fruits. Int. J. Essen. Oil Ther. 4 54–58. [Google Scholar]
- Pandey S., Upadhyay S., Tripathi A. (2009). Insecticidal and repellent activities of thymol from the essential oil of Trachyspermum ammi (Linn) sprague seeds against Anopheles stephensi. Parasitol. Res. 105 507–512. 10.1007/s00436-009-1429-6 [DOI] [PubMed] [Google Scholar]
- Park H.-M., Kim J., Chang K.-S., Kim B.-S., Yang Y.-J., Kim G.-H., et al. (2011). Larvicidal activity of Myrtaceae essential oils and their components against Aedes aegypti, acute toxicity on Daphnia magna, and aqueous residue. J. Med. Entomol. 48 405–410. 10.1603/ME10108 [DOI] [PubMed] [Google Scholar]
- Patrakar R., Mansuriya M., Patil P. (2012). Phytochemical and pharmacological review on Laurus nobilis. Inter. J. Pharm. Chem. Sci. 1 595–602. 30496684 [Google Scholar]
- Pavela R. (2009). Larvicidal property of essential oils against Culex quinquefasciatus Say (Diptera: Culicidae). Indust. Crop. Prod. 30 311–315. 10.1016/j.indcrop.2009.06.005 29748726 [DOI] [Google Scholar]
- Pavela R. (2015). Essential oils for the development of eco-friendly mosquito larvicides: a review. Ind. Crops Prod. 76 174–187. 10.1016/j.indcrop.2015.06.050 [DOI] [Google Scholar]
- Pavela R., Govindarajan M. (2017). The essential oil from Zanthoxylum monophyllum a potential mosquito larvicide with low toxicity to the non-target fish Gambusia affinis. J. Pest Sci. 90 369–378. 10.1007/s10340-016-0763-6 [DOI] [Google Scholar]
- Pavela R., Kaffkova K., Kumsta M. (2014). Chemical composition and larvicidal activity of essential oils from different Mentha L. and Pulegium species against Culex quinquefasciatus say (Diptera: Culicidae). Plant Prot. Sci. 50 36–41. 10.17221/48/2013-PPS [DOI] [Google Scholar]
- Pereira A. I. S., Pereira A. D. G. S., Sobrinho L., Palma O., Cantanhede E. D. K. P., Siqueira L. F. S. (2014). Antimicrobial activity in fighting mosquito larvae Aedes aegypti: homogenization of essential oils of linalool and eugenol. Educ. Quím. 25 446–449. [Google Scholar]
- Perumalsamy H., Kim N. J., Ahn Y. J. (2009). Larvicidal activity of compounds isolated from asarum heterotropoides against Culex pipiens pallens, Aedes aegypti, and Ochlerotatus togoi (Diptera: Culicidae). J. Med. Entomol. 46 1420–1423. 10.1603/033.046.0624 [DOI] [PubMed] [Google Scholar]
- Pitarokili D., Michaelakis A., Koliopoulos G., Giatropoulos A., Tzakou O. (2011). Chemical composition, larvicidal evaluation, and adult repellency of endemic greek thymus essential oils against the mosquito vector of West Nile virus. Parasitol. Res. 109 425–430. 10.1007/s00436-011-2271-1 [DOI] [PubMed] [Google Scholar]
- Pitasawat B., Champakaew D., Choochote W., Jitpakdi A., Chaithong U., Kanjanapothi D., et al. (2007). Aromatic plant-derived essential oil: an alternative larvicide for mosquito control. Fitoterapia 78 205–210. 10.1016/j.fitote.2007.01.003 [DOI] [PubMed] [Google Scholar]
- Pohlit A. M., Lopes N. P., Gama R. A., Tadei W. P., De Andrade Neto V. F. (2011). Patent literature on mosquito repellent inventions which contain plant essential oils - a review. Planta Med. 77 598–617. 10.1055/s-0030-1270723 [DOI] [PubMed] [Google Scholar]
- Pour H. T., Shayeghi M., Vat H., Abai M. R. (2016). Study on larvicidal effects of essential oils of three Iranian native plants against larvae Anopheles stephensi (Liston). Vector Biol. J. 1:2. [Google Scholar]
- Pradeepa V., Sathish-Narayanan S., Kirubakaran S. A., Senthil-Nathan S. (2014). Antimalarial efficacy of dynamic compound of plumbagin chemical constituent from Plumbago zeylanica Linn (Plumbaginaceae) against the malarial vector Anopheles stephensi Liston (Diptera: Culicidae). Parasitol. Res. 113 3105–3109. 10.1007/s00436-014-4015-5 [DOI] [PubMed] [Google Scholar]
- Pradeepa V., Sathish-Narayanan S., Kirubakaran S. A., Thanigaivel A., Senthil-Nathan S. (2015). Toxicity of aristolochic acids isolated from Aristolochia indica Linn (Aristolochiaceae) against the malarial vector Anopheles stephensi liston (Diptera: Culicidae). Exper. Parasitol. 153 8–16. 10.1016/j.exppara.2015.01.017 [DOI] [PubMed] [Google Scholar]
- Pradeepa V., Senthil-Nathan S., Sathish-Narayanan S., Selin-Rani S., Vasantha-Srinivasan P., Thanigaivel A., et al. (2016). Potential mode of action of a novel plumbagin as a mosquito repellent against the malarial vector Anopheles stephensi (Culicidae: Diptera). Pestic. Biochem.Physiol. 134 84–93. 10.1016/j.pestbp.2016.04.001 [DOI] [PubMed] [Google Scholar]
- Rahuman A. A., Gopalakrishnan G., Ghouse B. S., Arumugam S., Himalayan B. (2000). Effect of Feronia limonia on mosquito larvae. Fitoterapia 71 553–555. 10.1016/S0367-326X(00)00164-7 [DOI] [PubMed] [Google Scholar]
- Rahuman A. A., Gopalakrishnan G., Venkatesan P., Geetha K. (2008a). Isolation and identification of mosquito larvicidal compound from Abutilon indicum (Linn.) Sweet. Parasitol. Res. 102 981–988. 10.1007/s00436-007-0864-5 [DOI] [PubMed] [Google Scholar]
- Rahuman A. A., Gopalakrishnan G., Venkatesan P., Geetha K., Bagavan A. (2008b). Mosquito larvicidal activity of isolated compounds from the rhizome of Zingiber officinale. Phytoth. Res. 22 1035–1039. 10.1002/ptr.2423 [DOI] [PubMed] [Google Scholar]
- Rattan R. S. (2010). Mechanism of action of insecticidal secondary metabolites of plant origin. Crop Prot. 29 913–920. 10.1016/j.cropro.2010.05.008 [DOI] [Google Scholar]
- Rey D., Long A., Pautou M. P., Meyran J. C. (1999). Histopathological effects of tannic acid on the midgut epithelium of some aquatic diptera larvae. J. Invert. Pathol. 73 173–181. 10.1006/jipa.1998.4810 [DOI] [PubMed] [Google Scholar]
- Rocha D. K., Matos O., Novo M. T., Figueiredo A. C., Delgado M., Moiteiro C. (2015). Larvicidal activity against Aedes aegypti of Foeniculum vulgare essential oils from Portugal and Cape Verde. Nat. Prod. Commun. 4 677–682. 10.1177/1934578X1501000438 [DOI] [PubMed] [Google Scholar]
- Rodrigues A. M. S., De Paula J. E., Roblot F., Fournet A., Espíndola L. S. (2005). Larvicidal activity of Cybistax antisyphilitica against Aedes aegypti larvae. Fitoterapia 76 755–757. 10.1016/j.fitote.2005.08.015 [DOI] [PubMed] [Google Scholar]
- Rudin W., Hecker H. (1989). Lectin binding sites in the midgut of the mosquitoes Anopheles stephensi liston and Aedes aegypti L. (Diptera, Culicidae). Parasitol. Res. 75 268–279. 10.1007/bf00931811 [DOI] [PubMed] [Google Scholar]
- Ruiz C., Cachay M., Domínguez M., Velásquez C., Espinoza G., Ventosilla P., et al. (2011). Chemical composition, antioxidant and mosquito larvicidal activities of essential oils from Tagetes filifolia, Tagetes minuta and Tagetes elliptica from Perú. Planta Med. 77:E30 10.1055/s-0031-1282361 [DOI] [Google Scholar]
- Saeed M., Muhammad N., Khan H. (2010). Analysis of toxic heavy metals in branded pakistani herbal products. J. Chem. Soc. Pak. 32:471. 22186337 [Google Scholar]
- Sakhanokho H. F., Sampson B. J., Tabanca N., Wedge D. E., Demirci B., Baser K. H. C., et al. (2013). Chemical composition, antifungal and insecticidal activities of Hedychium essential oils. Molecules 18 4308–4327. 10.3390/molecules18044308 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Santiago G. M., Lemos T. L., Pessoa O. D., Arriaga A. M., Matos F. J., Lima M. A. S., et al. (2006). Larvicidal activity against Aedes aegypti L. (Diptera: Culicidae) of essential oils of Lippia species from Brazil. Nat. Prod. Commun. 1 573–576. 10.1177/1934578X0600100711 [DOI] [Google Scholar]
- Santos G. K., Dutra K. A., Barros R. A., da Câmara C. A., Lira D. D., Gusmão N. B., et al. (2012). Essential oils from Alpinia purpurata (Zingiberaceae): chemical composition, oviposition deterrence, larvicidal and antibacterial activity. Ind. Crops Prod. 40 254–260. 10.1016/j.indcrop.2012.03.020 [DOI] [Google Scholar]
- Santos R. P., Nunes E. P., Nascimento R. F., Santiago G. M. P., Menezes G. H. A., Silveira E. R., et al. (2006). Chemical composition and larvicidal activity of the essential oils of Cordia leucomalloides and Cordia curassavica from the Northeast of Brazil. J. Braz. Chem. Soci. 17 1027–1030. 10.1590/S0103-50532006000500030 [DOI] [Google Scholar]
- Sedaghat M. M., Dehkordi A. S., Khanavi M., Abai M. R., Mohtarami F., Vatandoost H. (2011). Chemical composition and larvicidal activity of essential oil of Cupressus arizonica EL Greene against malaria vector Anopheles stephensi liston (Diptera: Culicidae). Pharmacogn. Res. 3:135 10.4103/0974-8490.81962 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Selin-Rani S., Senthil-Nathan S., Thanigaivel A., Vasantha-Srinivasan P., Edwin E. S., Ponsankar A., et al. (2016). Toxicity and physiological effect of quercetin on generalist herbivore, Spodoptera litura Fab. and a non-target earthworm Eisenia fetida savigny. Chemosphere 165 257–267. 10.1016/j.chemosphere.2016.08.136 [DOI] [PubMed] [Google Scholar]
- Senthilkumar A., Jayaraman M., Venkatesalu V. (2013). Chemical constituents and larvicidal potential of Feronia limonia leaf essential oil against Anopheles stephensi, Aedes aegypti and Culex quinquefasciatus. Parasitol. Res. 112 1337–1342. 10.1007/s00436-012-3188-z [DOI] [PubMed] [Google Scholar]
- Senthilkumar A., Kannathasan K., Venkatesalu V. (2008). Chemical constituents and larvicidal property of the essential oil of Blumea mollis (D. Don) Merr. against Culex quinquefasciatus. Parasitol. Res. 103 959–962. 10.1007/s00436-008-1085-2 [DOI] [PubMed] [Google Scholar]
- Senthil-Nathan S. (2007). The use of eucalyptus leaf extract as a natural larvicidal agent against malarial vector Anopheles stephensi Liston (Diptera: Culicidae). Bioresour. Technol. 98 1856–1860. 10.1016/j.biortech.2006.07.044 [DOI] [PubMed] [Google Scholar]
- Senthil-Nathan S. (2013). Physiological and biochemical effect of neem and other Meliaceae plants secondary metabolites against Lepidopteran insects. Front. Physiol. 4:359. 10.3389/fphys.2013.00359 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Senthil-Nathan S. (2015). “A review of biopesticides and their mode of action against insect pests,” in Environmental Sustainability, ed. Sridevi G. (Berlin: Springer; ). [Google Scholar]
- Senthil-Nathan S., Choi M. Y., Paik C. H., Seo H. Y., Kalaivani K. (2009). Toxicity and physiological effects of neem pesticides applied to rice on the Nilaparvata lugens Stål, the brown planthopper. Ecotoxicol. Environ. Saf. 72 1707–1713. 10.1016/j.ecoenv.2009.04.024 [DOI] [PubMed] [Google Scholar]
- Senthil-Nathan S., Hisham A., Jayakumar G. (2008). Larvicidal and growth inhibition of the malaria vector Anopheles stephensi by triterpens from Dysoxylum spp. (Meliaceae). Fitoterapia 76 106–111. 10.1016/j.fitote.2007.07.013 [DOI] [PubMed] [Google Scholar]
- Senthil-Nathan S., Kalaivani K. (2005). Efficacy of nucleopolyhedrovirus and azadirachtin on Spodoptera litura fabricius (Lepidoptera: Noctuidae). Biol. Control 34 93–98. 10.1016/j.biocontrol.2005.03.001 [DOI] [Google Scholar]
- Senthil-Nathan S., Kalaivani K. (2006). Combined effects of azadirachtin and nucleopolyhedrovirus (SpltNPV) on Spodoptera litura fabricius (Lepidoptera: Noctuidae) larvae. Biol. Control 39 96–104. 10.1016/j.biocontrol.2006.06.013 [DOI] [Google Scholar]
- Senthil-Nathan S., Kalaivani K., Chung P. G. (2005a). The effects of azadirachtin and nucleopolyhedrovirus on midgut enzymatic profile of Spodoptera litura Fab.(Lepidoptera: Noctuidae). Pestic. Bichem. Physiol. 83 46–57. 10.1016/j.pestbp.2005.03.009 [DOI] [Google Scholar]
- Senthil-Nathan S., Kalaivani K., Murugan K. (2005b). Effects of neem limonoids on the malaria vector Anopheles stephensi liston (Diptera: Culicidae). Acta Trop. 96 47–55. 10.1016/j.actatropica.2005.07.002 [DOI] [PubMed] [Google Scholar]
- Senthil-Nathan S., Kalaivani S., Sehoon K. (2006a). Effects of Dysoxylum malabaricum Bedd (Meliaceae) extract on the malarial vector Anopheles stephensi Liston (Diptera: Culicidae). Bioresour. Technol. 97 2077–2083. 10.1016/j.biortech.2005.09.034 [DOI] [PubMed] [Google Scholar]
- Senthil-Nathan S., Savitha G., George D. K., Narmadha A., Suganya L., Chung P. G. (2006b). Efficacy of Melia azedarach L. extract on the malarial vector Anopheles stephensi Liston. Bioresour. Technol. 97 1214–1221. 10.1016/j.biortech.2005.05.019 [DOI] [PubMed] [Google Scholar]
- Seo S.-M., Jung C.-S., Kang J., Lee H.-R., Kim S.-W., Hyun J., et al. (2015). Larvicidal and acetylcholinesterase inhibitory activities of Apiaceae plant essential oils and their constituents against Aedes albopictus and formulation development. J. Agric. Food Chem. 63 9977–9986. 10.1021/acs.jafc.5b03586 [DOI] [PubMed] [Google Scholar]
- Service M. W. (1996). Medical Entomology for Students. London: Chapman and Hall. [Google Scholar]
- Shaalan E., Canyon D., Younes M. W. F., Abdel-Wahab H., Mansour A. (2005). A review of botanical phytochemicals with mosquitocidal potential. Environ. Int. 31 1149–1166. 10.1016/j.envint.2005.03.003 [DOI] [PubMed] [Google Scholar]
- Shaalan E. A. S., Canyon D. V., Bowden B., Younes M. W. F., Abdel-Wahab H., Mansour A. H. (2006). Efficacy of botanical extracts from Callitris glaucophylla against Aedes aegypti and Culex annulirostris mosquitoes. Trop. Biomed. 23 180–185. [PubMed] [Google Scholar]
- Shunying Z., Yang Y., Huaidong Y., Yue Y., Guolin Z. (2005). Chemical composition and antimicrobial activity of the essential oils of Chrysanthemum indicum. J. Ethnopharmacol. 96 151–158. 10.1016/j.jep.2004.08.031 [DOI] [PubMed] [Google Scholar]
- Siddiqui B. S., Afshan F., Afshan F., Ghiasuddin S. F., Naqvi S. N.-U.-H., Tariq R. M., et al. (1999). New insect-growth-regulator meliacin butenolides from the leaves of Azadirachta indica A. Juss. J. Chem. Soci. Perkin Trans. 1 2367–2370. 10.1039/A902326G [DOI] [Google Scholar]
- Siddiqui B. S., Afshan F., Faizi S., Naeem-Ul-Hassan Naqvi S., Tariq R. M. (2002). Two new triterpenoids from Azadirachta indica and their insecticidal activity. J. Nat. Prod. 65 1216–1218. 10.1021/np0105477 [DOI] [PubMed] [Google Scholar]
- Siddiqui B. S., Afshan F., Gulzar T., Sultana R., Naqvi S. N.-H., Tariq R. M. (2003). Tetracyclic triterpenoids from the leaves of Azadirachta indica and their insecticidal activities. Chem. Pharm. Bull. 51 415–417. 10.1248/cpb.51.415 [DOI] [PubMed] [Google Scholar]
- Sola P., Mvumi B. M., Ogendo J. O., Mponda O., Kamanula J. F., Nyirenda S., et al. (2014). Botanical pesticide production, trade and regulatory mechanisms in sub-Saharan Africa: making a case for plant-based pesticidal products. Food Secur. 6 369–384. 10.1007/s12571-014-0343-7 [DOI] [Google Scholar]
- Souza L. G. D. S., Almeida M. C. S., Monte F. J. Q., Santiago G. M. P., Braz-Filho R., Lemos T. L. G., et al. (2012). Chemical constituents of Capraria biflora (Scrophulariaceae) and larvicidal activity of essential oil. Quím. Nova 35 2258–2262. 10.1590/S0100-40422012001100032 [DOI] [Google Scholar]
- Stenfors Arnesen L. P., Fagerlund A., Granum P. E. (2008). From soil to gut: Bacillus cereus and its food poisoning toxins. FEMS Microbiol. Rev. 32 579–606. 10.1111/j.1574-6976.2008.00112.x [DOI] [PubMed] [Google Scholar]
- Sujatha G., Kumari R., Bollipo D. (2013). Composition and larvicidal activity of Artemisia vulgaris L. stem essential oil against Aedes aegypti. Jord. J. Biol. Sci. 147 1–5. [Google Scholar]
- Sukumar K., Perich M. J., Boobar L. R. (1991). Botanical derivatives in mosquito control: a review. J. Am. Mosq. Control Assoc. 7 210–237. [PubMed] [Google Scholar]
- Sun R., Sacalis J. N., Chin C.-K., Still C. C. (2001). Bioactive aromatic compounds from leaves and stems of Vanilla fragrans. J. Agric. Food Chem. 49 5161–5164. 10.1021/jf010425k [DOI] [PubMed] [Google Scholar]
- Sutthanont N., Choochote W., Tuetun B., Junkum A., Jitpakdi A., Chaithong U., et al. (2010). Chemical composition and larvicidal activity of edible plant-derived essential oils against the pyrethroid-susceptible and-resistant strains of Aedes aegypti (Diptera: Culicidae). J. Vector Ecol. 35 106–115. 10.1111/j.1948-7134.2010.00066.x [DOI] [PubMed] [Google Scholar]
- Syed Z., Leal W. S. (2008). Mosquitoes smell and avoid the insect repellent DEET. Proc. Nat. Acad. Sci. U.S.A. 105 13598–13603. 10.1073/pnas.0805312105 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tabanca N., Demirci B., Kiyan H. T., Ali A., Bernier U. R., Wedge D. E., et al. (2012a). Repellent and larvicidal activity of Ruta graveolens essential oil and its major individual constituents against Aedes aegypti. Planta Med. 78:90 10.1055/s-0032-1307598 [DOI] [PubMed] [Google Scholar]
- Tabanca N., Özek G., Ali A., Duran A., Hamzaoðlu E., Başer K. H. C., et al. (2012b). Chemical composition of Heracleum pastinacifolium subsp. transcaucasicum and subsp. incanum essential oils, and their biting deterrent and larvicidal activity against Aedes aegypti. Planta Med. 78:89 10.1055/s-0032-1307597 [DOI] [Google Scholar]
- Tehri K., Singh N. (2015). The role of botanicals as green pesticides in integrated mosquito management-A review. Inter. J. Mosq. Res. 2 18–23. [Google Scholar]
- Thanigaivel A., Chandrasekaran R., Revathi K., Nisha S., Sathish-Narayanan S., Kirubakaran S. A., et al. (2012). Larvicidal efficacy of Adhatoda vasica (L.) Nees against the bancroftian filariasis vector Culex quinquefasciatus Say and dengue vector Aedes aegypti L. in in vitro condition. Parasitol. Res. 110 1993–1999. 10.1007/s00436-011-2728-2 [DOI] [PubMed] [Google Scholar]
- Thanigaivel A., Chanthini K. M. P., Karthi S., Vasantha-Srinivasan P., Ponsankar A., Sivanesh H., et al. (2019). Toxic effect of essential oil and its compounds isolated from Sphaeranthus amaranthoides Burm. f. against dengue mosquito vector Aedes aegypti Linn. Pestic. Bichem. Physiol. 160 163–170. 10.1016/j.pestbp.2019.08.006 [DOI] [PubMed] [Google Scholar]
- Thanigaivel A., Senthil-Nathan S., Vasantha-Srinivasan P., Edwin E. S., Ponsankar A., Selin-Rani S., et al. (2017a). Chemicals isolated from Justicia adhatoda Linn reduce fitness of the mosquito, Aedes aegypti L. Arch. Insect Biochem. Physiol. 94:e21384. 10.1002/arch.21384 [DOI] [PubMed] [Google Scholar]
- Thanigaivel A., Vasantha-Srinivasan P., Edwin E.-S., Ponsankar A., Selin-Rani S., Chellappandian M., et al. (2017b). Development of an eco-friendly mosquitocidal agent from Alangium salvifolium against the dengue vector Aedes aegypti and its biosafety on the aquatic predator. Environ. Sci. Pollut. Res. 25 10340–10352. 10.1007/s11356-017-9102-6 [DOI] [PubMed] [Google Scholar]
- Tiwary M., Naik S. N., Tewary D. K., Mittal P. K., Yadav S. (2007). Chemical composition and larvicidal activities of the essential oil of Zanthoxylum armatum DC (Rutaceae) against three mosquito vectors. J. Vector Born. Dis. 44:198. [PubMed] [Google Scholar]
- Torres M. C. M., Assunção J. C., Santiago G. M. P., Andrade-Neto M., Silveira E. R., Costa-Lotufo L. V., et al. (2008). Larvicidal and nematicidal activities of the leaf essential oil of croton regelianus. Chem. Biodivers. 5 2724–2728. 10.1002/cbdv.200890227 [DOI] [PubMed] [Google Scholar]
- Traboulsi A. F., Taoubi K., El-Haj S., Bessiere J. M., Rammal S. (2002). Insecticidal properties of essential plant oils against the mosquito Culex pipiens molestus (Diptera: Culicidae). Pestic. Manag. Sci. 58 491–495. 10.1002/ps.486 [DOI] [PubMed] [Google Scholar]
- Trindade F. T. T., Stabeli R. G., Pereira A. A., Facundo V. A., de Almeida A. (2013). Copaifera multijuga ethanolic extracts, oilresin, and its derivatives display larvicidal activity against Anopheles darlingi and Aedes aegypti (Diptera: Culicidae). Rev. Bras. Farmacogn. 23 464–470. 10.1590/S0102-695X2013005000038 [DOI] [Google Scholar]
- Tripathi A. K., Upadhyay S., Bhuiyan M., Bhattacharya P. R. (2009). A review on prospects of essential oils as biopesticides in insect pest management. J. Pharmacogn. Phytother. 1 52–63. [Google Scholar]
- Tyagi B. K., Munirathinam A., Venkatesh A. (2015). A catalogue of indian mosquitoes. Inter. J. Mosq. Res. 2 50–97. 10.1186/s12864-018-4729-3 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Urich K. (1994). Comparative Animal Biochemistry. Berlin: Springer-Verlag. [Google Scholar]
- Vasantha-Srinivasan P., Karthi S., Chellappandian M., Ponsankar A., Thanigaivel A., Senthil-Nathan S., et al. (2019). Aspergillus flavus (Link) toxins reduces the fitness of dengue vector Aedes aegypti (Linn.) and their non-target toxicity against aquatic predator. Microb. Pathog. 128 281–287. 10.1016/j.micpath.2019.01.014 [DOI] [PubMed] [Google Scholar]
- Vasantha-Srinivasan P., Senthil-Nathan S., Ponsankar A., Thanigaivel A., Edwin E. S., Selin-Rani S., et al. (2017). Comparative analysis of mosquito (Diptera: Culicidae: Aedes aegypti Liston) responses to the insecticide Temephos and plant derived essential oil derived from Piper betle L. Ecotoxicol. Environ. Saf. 139 439–446. 10.1016/j.ecoenv.2017.01.026 [DOI] [PubMed] [Google Scholar]
- Vasantha-Srinivasan P., Thanigaivel A., Edwin E. S., Ponsankar A., Senthil-Nathan S., Selin-Rani S., et al. (2018). Toxicological effects of chemical constituents from Piper against the environmental burden Aedes aegypti Liston and their impact on non-target toxicity evaluation against biomonitoring aquatic insects. Environ. Sci. Pollut. Res. 25 10434–10446. 10.1007/s11356-017-9714-x [DOI] [PubMed] [Google Scholar]
- Velu K., Elumalai D., Hemalatha P., Janaki A., Babu M., Hemavathi M., et al. (2015). Evaluation of silver nanoparticles toxicity of Arachis hypogaea peel extracts and its larvicidal activity against malaria and dengue vectors. Environ. Sci. Poll. Res. 22 17769–17779. 10.1007/s11356-015-4919-3 [DOI] [PubMed] [Google Scholar]
- Vera S. S., Zambrano D. F., Méndez-Sanchez S. C., Rodríguez-Sanabria F., Stashenko E. E., Luna J. E. D. (2014). Essential oils with insecticidal activity against larvae of Aedes aegypti (Diptera: Culicidae). Parasitol. Res. 113 2647–2654. 10.1007/s00436-014-3917-6 [DOI] [PubMed] [Google Scholar]
- Vivekanandhan P., Senthil-Nathan S., Shivakumar M. S. (2018a). Larvicidal, pupicidal and adult smoke toxic effects of Acanthospermum hispidum (DC) leaf crude extracts against mosquito vectors. Physiol. Mol. Plant Pathol. 101 156–162. 10.1016/j.pmpp.2017.05.005 [DOI] [Google Scholar]
- Vivekanandhan P., Venkatesan R., Ramkumar G., Karthi S., Senthil-Nathan S., Shivakumar M. S. (2018b). Comparative analysis of major mosquito vectors response to seed-derived essential oil and seed pod-derived extract from Acacia nilotica. Intern. J. Environ. Res. Public Health. 15:388. 10.3390/ijerph15020388 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vourlioti-Arapi F., Michaelakis A., Evergetis E., Koliopoulos G., Haroutounian S. (2012). Essential oils of indigenous in Greece six Juniperus taxa. Parasitol. Res. 110 1829–1839. 10.1007/s00436-011-2706-8 [DOI] [PubMed] [Google Scholar]
- Wahyuni D. (2012). Larvicidal activity of essential oils of Piper betle from the Indonesian plants against Aedes aegypti L. J. Appl. Environ. Biol. Sci. 2 249–254. [Google Scholar]
- Walter K., Schütt C. (1974). “Acid and alkaline phosphatase in serum (Two–point method),” in Methods of Enzymatic Analysis, ed. Bergmeyer H. U. (Florida: Verlag Chemie GmbH; ), 856–860. 10.1016/b978-0-12-091302-2.50067-0 [DOI] [Google Scholar]
- Wheelock C. E., Shan G., Ottea J. A. (2005). Overview of carboxylesterases and their role in metabolism of insecticides. J. Pestic. Sci. 30 75–83. 10.1584/jpestics.30.75 [DOI] [Google Scholar]
- World Health Organization [WHO] (2017). Annex 1. Global Burden of Major Vector-Borne Diseases, as of March 2017. Geneva: WHO. [Google Scholar]
- Wu L.-Y., Gao H.-Z., Wang X.-L., Ye J.-H., Lu J.-L., Liang Y.-R. (2010). Analysis of chemical composition of Chrysanthemum indicum flowers by GC/MS and HPLC. J. Med. Plants Res. 4 421–426. [Google Scholar]
- Yang Y.-C., Lee S.-G., Lee H.-K., Kim M.-K., Lee S.-H., Lee H.-S. (2002). A piperidine amide extracted from Piper longum L. fruit shows activity against Aedes aegypti mosquito larvae. J. Agric. Food Chem. 50 3765–3767. 10.1021/jf011708f [DOI] [PubMed] [Google Scholar]
- Yenesew A., Derese S., Midiwo J. O., Heydenreich M., Peter M. G. (2003). Effect of rotenoids from the seeds of Millettia dura on larvae of Aedes aegypti. Pestic. Manag. Sci. 59 1159–1161. 10.1002/ps.740 [DOI] [PubMed] [Google Scholar]
- Yu C. H., Dang Y. K., Zhou Z. P., Wu C., Zhao F. Z., Sachs M. S., et al. (2015). Codon usage influences the local rate of translation elongation to regulate co-translational protein folding. Mol. Cell 59 744–754. 10.1016/j.molcel.2015.07.018 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang J., Huang T., Zhang J., Shi Z., He Z. (2018). Chemical composition of leaf essential oils of four Cinnamomum species and their larvicidal activity against Anopheleus sinensis (Diptera: Culicidae). J. Essen. Oil Bear. Plants 21 1284–1294. 10.1080/0972060X.2018.1552205 [DOI] [Google Scholar]
- Zhu J., Zeng X., Liu T., Qian K., Han Y., Xue S., et al. (2006). Adult repellency and larvicidal activity of five plant essential oils against mosquitoes. J. Am. Mosq. Control Assoc. 22 515–522. 10.2987/8756-971x(2006)22[515:aralao]2.0.co;2 [DOI] [PubMed] [Google Scholar]
- Zhu L., Tian Y. (2011). Chemical composition and larvicidal activity of Blumea densiflora essential oils against Anopheles anthropophagus: a malarial vector mosquito. Parasitol. Res. 109 1417–1422. 10.1007/s00436-011-2388-2 [DOI] [PubMed] [Google Scholar]
- Zibaee A. (2011). “Botanical insecticides and their effects on insect biochemistry and immunity,” in Pesticides in the Modern World - Pests Control and Pesticides Exposure and Toxicity Assessment, ed. Stoytcheva M. (London: IntechOpen; ), 10.5772/16550 [DOI] [Google Scholar]
- Zibaee A., Bandani A. R. (2010). Effects of Artemisia annua L. (Asteracea) on digestive enzymes profiles and cellular immune reactions of sunn pest, Eurygaster integriceps (Heteroptera: Scutellaridae), against Beauvaria bassiana. Bull. Entomol. Res. 100 185–196. 10.1017/S000748530999014 [DOI] [PubMed] [Google Scholar]