Abstract
Background:
Potential drug–drug interactions (DDIs) are increasingly common in clinical practice, especially among individuals with chronic conditions, such as chronic kidney dysfunction. However, data relating to DDIs among chronically ill patients are limited in Nigeria. We, therefore, investigated the prevalence and pattern of DDIs among patients with kidney diseases on admission at a tertiary hospital in Lagos, Nigeria.
Materials and Methods:
This was a prospective observational study involving 61 adults with kidney diseases and on admission in medical wards of the study center, over a 3-month period. Data extractions were with a purposefully designed pro forma to extract relevant data on demographic, clinical, and dosing regimens of the prescribed drugs for individual patients. Potential DDIs were identified, and their severity was rated using the MICROMEDEX® software database (IBM® Watson–Truven Health Analytics), which is available online with limited access.
Results:
Of the 61 patients evaluated, majority were males (34; 55.7%), were elderly (26; 42.6%), and had chronic kidney disease Stage 3 (40; 65.5%). The most common cause of kidney disease was hypertension (20; 32.8%). Out of the 542 prescriptions received by the patients, potential DDI was observed in 508 (93.7%) prescriptions. Clinically significant drug interactions (CSDIs) were detected in 486 (85.7%) prescriptions. Pharmacodynamic DDIs (466; 91.7%) were the most common. Pill burden exceeding 25 pills/day was present in nine (14.8%) patients. The severities of the potential DDIs were major (135; 24.9%), moderate (333; 61.4%), and minor (38; 7.1%). Only two different potential DDIs were rated X (contraindicated).
Conclusion:
Exposure to drugs with potential DDIs was very common among patients with kidney diseases. Most of the CSDIs observed were of major severity. The use of DDI checker before prescribing drugs for individuals with kidney diseases could avert clinically significant interactions.
Keywords: Chronic kidney disease, co-administered drugs, contraindication, drug–drug interactions, pharmacodynamics, pharmacokinetics, polypharmacy, toxic effect
INTRODUCTION
Drug-drug interaction (DDI) is defined as a phenomenon that leads to alteration of the therapeutic efficacy or toxicity of a particular drug when co-administered with another drug.1 An interaction is said to be clinically significant if it requires a dosage adjustment of the object drug (s), requires therapy monitoring, or consists of a drug combination that is contraindicated due to its high potential for clinical adverse effects.2 In clinical practice, multiple drugs are often combined in the treatment of patients with chronic diseases. Drug combinations are commonly used in infectious diseases, such as tuberculosis, malaria, and HIV/AIDS, to avert drug resistance and also to utilize their synergistic potentials. However, this practice could lead to DDIs with expected beneficial effects, but, in some cases, undesirable outcomes may occur, such as ineffective treatment and severe adverse events.1,3 Among adult population without chronic kidney diseases (CKDs), the risks for potential DDIs are high and include polypharmacy, old age, impaired hepatic drug metabolism, and impaired renal elimination.1,2 For clinical purposes, most drug interaction databases have rated DDIs as A (no known interaction), B (minor severity and no action is needed), C (moderate severity but therapy monitoring is required), D (major severity and therapy modification is required), and X (contraindicated and such combination should be avoided). DDI is a cause of drug-related problems, which can result in morbidity, mortality, prolonged hospitalization, decreased quality of life, or increased cost of care of individual patients.4
It is well documented in the literature that CKD or renal dysfunction is a potential risk for DDI due to the practice of polypharmacy.4 The kidneys play a key role in the maintenance of homeostasis and in regulatory, excretory, and endocrine functions. Therefore, the gradual decrease in the glomerular filtration rate (GFR) and/or loss of kidney function associated with CKD can compromise the homeostasis of the entire body.4 Current evidence suggests that some of the adverse outcomes of CKD can be prevented or delayed by early detection and treatment.5 However, the clustered diseases that concomitantly exist with CKD, such as diabetes, hypertension, and hyperlipidemia, may require additional treatment with newer drugs, thus increasing the potential risk for DDIs. In a recent hospital-based study by Adibe et al. evaluating DDIs among patients with CKDs in South-Eastern Nigeria, pill burden and hypertension were reported as comorbidities that significantly predict DDIs.6 This was, however, a retrospective study and did not enumerate the pattern and types of DDI in the cohort. In a similar but a prospective study, Fasipe el al. reported an overall high prevalence of DDI (95.9%).7 Although the risks for DDIs in CKD have been fairly documented in the literature in developed countries, such data are, however, scarce in low- and middle-income countries. Hence, we determined the prevalence and pattern of DDIs among CKD patients at the Lagos University Teaching Hospital (LUTH), Idiaraba, Nigeria.
MATERIALS AND METHODS
Study design and location
This was a prospective observational study involving patients who consecutively presented with renal dysfunction and were admitted to the medical wards at LUTH between September 1 and December 31, 2016. LUTH is a tertiary health-care center situated at Surulere, South-West Nigeria. The hospital was established to provide tertiary care for over 20 million inhabitants of Lagos and its environs. It is a major referral center for the care of medical conditions including acute and CKDs. The first government renal dialysis center was established in the hospital in the 1980s, and the hospital currently has a functional renal transplant center. Patients with renal conditions are usually admitted via the emergency room or outpatient clinics by a renal team. The diagnosis of renal dysfunctions was based on the following criteria: GFR < 60 mL/min and/or overt proteinuria. The CKD stages were determined using the methods of Culleton et al.8
Study population
Over the study period, we prospectively studied 61 patients who were admitted to the medical wards with renal dysfunction and treated with multiple medicines with potential for interactions. Patients who were ≥18 years old, those diagnosed of kidney dysfunction, and those who were willing to give informed consents were enrolled in the study. Patients < 18 years old, critically ill, pregnant, or nonconsenting to the study were excluded from the study.
Sample size determination and sampling technique
The sample size was calculated using the following formulae: nf = n/(1+ [n/N]) and n = Z2pq/d2,9 where nf = final sample size, n = initial sample size, N = sample size based on the hospital frequency of CKD, Z = standard deviation set at 1.96 with confidence level of 95%, P = prevalence at 95% confidence level (set at 14.7% prevalence of CKD as obtained from a previous study10), q = 100 − p, and d = level of precision (set at 5% margin of error). The calculation yielded a sample size of 55. However, this was increased to 61 so as to make up for any withdrawal from the study. Overall, a total of sixty patients were enrolled in this study using a simple random selection technique.
Ethical approval
The Health Research Ethics Committee (HREC) of LUTH approved the study with a reference No. ADM/DCST/HREC/440. The study complied with all institutional guidelines and regulations as regards ethics, consent, and permission for the research. Consent was obtained from all individuals who participated in the study either directly or from their legal guardians.
Data collection
Data were obtained from eligible respondents using a well-structured, self-designed pro forma and case record of the individual patients, including nurses' records of drug administration. All the patients admitted during the study duration were followed up from the day of admission to the day of discharge, and all new prescriptions issued during hospital discharge were collected for a review. Data extracted included sociodemographic and clinical information such as age; gender; ethnic groups; the specific kidney disease suffered by each patient and comorbid diseases; indicated drug(s), for example, antihypertensive agents for those whose renal dysfunction was secondary to hypertension and antidiabetic drugs for those with diabetic nephropathies; and co-prescribed medications. The number of drugs taken per day constitutes the pill burden per patient. The potential interactions between the drugs prescribed for the patients were analyzed using IBM® Watson-Truven Health Analytics Micromedex® version 4.4, Ann Arbor, Michigan, United States drug interaction software mobile app available online with limited access.11 The Truven Health analytics Micromedex® database contains information on medications, potential DDIs of many co-prescribed medications, necessary interventions, etiology, epidemiology, diagnosis, and pharmacotherapy.11
Determination of severity rating of drug interactions between drugs indicated for kidney diseases and co-administered drugs
The interactions were rated as A (no known interaction), B (minor severity and no action needed), C (moderate severity but therapy monitoring required), D (major severity and therapy modification required), and X (contraindicated and drug combination should be avoided) as shown in Table 1.11,12
Table 1.
Severity rating of the interactions between drugs indicated for renal dysfunction and co-administered drugs
| Rating | Designation | Action | Explanation |
|---|---|---|---|
| A | Unknown | No known interaction | Unknown |
| B | Minor | No action needed | The interaction would have limited clinical effects. May include an increase in the frequency or severity of the side effects but generally would not require a major alteration in therapy |
| C | Moderate | Monitor therapy | The interaction may result to exacerbation of the patient’s condition and/or require an alteration in therapy |
| D | Major | Therapy modification | The interaction may be life threatening and/or require medical serious adverse events |
| X | Contraindicated | Avoid combination | The drugs are contraindicated for concurrent use |
A and B are considered nonclinically significant drug interactions, whereas C, D, and X are considered clinically significant drug interactions
Data analysis
The data obtained were recorded into Excel sheet and transported into Statistical Package for Social Sciences (SPSS®) Statistics for Windows, Version 20.0. Chicago, Illinois, United States for analysis. Descriptive statistics were used to analyze categorical variables, and results were presented as proportions or percentages, whereas continuous variables were expressed as means ± standard deviation. A univariate analysis was conducted for association between the sociodemographic or clinical variables of the patients and DDI severity, using Fisher's exact test, Pearson's Chi-square test, or Mann–Whitney test as appropriate. P < 0.05 was considered statistically significant, using a two-tailed test.
RESULTS
Sociodemographic characteristics of patients with potential drug-drug interaction
Of all the 61 respondents, 34 (55.7%) were males, and the mean age of the respondents was 53.8 ± 17.5 years. Table 2 shows the sociodemographic characteristics of the respondents. Majority were elderly (26; 42.6%), followed by those in the age group of 51–60 years (12; 19.5%). Yoruba (27; 44.3%) was the most common ethnicity, and hypertension (20; 32.8%) was the most common cause of nephropathy among our cohort of patients [Table 2]. Among the 542 prescriptions, the overall prevalence of potential DDI was seen in 508 (93.8%) prescriptions, and clinically significant DDI (CSDI) (486; 89.7%) was predominant. A few number of the patients (9, 14.8%) had significant pill burden, and the median daily pill taken per day per patient was 14 (4–30) [Table 2]. As for the types of DDI seen, pharmacodynamics type (466; 86%) was more common than pharmacokinetic DDI (76; 14%) in our cohort [Figure 1].
Table 2.
Sociodemographic characteristics and clinical details of the patients with renal dysfunction experiencing potential drug-drug interaction
| Characteristics | n (%) |
|---|---|
| Age (years), mean±SD | 53.8±17.5 |
| Age categories | |
| ≤30 | 7 (11.5) |
| 31-40 | 9 (14.8) |
| 41-50 | 7 (11.5) |
| 51-60 | 12 (19.7) |
| ≥61 | 26 (42.6) |
| Gender | |
| Male | 34 (55.7) |
| Female | 27 (44.3) |
| Ethnicity | |
| Yoruba | 27 (44.3) |
| Igbo | 21 (34.4) |
| Hausa | 13 (21.3) |
| Kidney disease types | |
| Hypertensive nephropathy | 20 (32.8) |
| Diabetic nephropathy | 14 (22.9) |
| Chronic glomerulonephritis | 8 (13.1) |
| Obstructive uropathy | 11 (18) |
| Others | 8 (13.1) |
| Comorbid conditions | |
| Hypertension | 21 (34.4) |
| Diabetes | 16 (26.2) |
| Cardiovascular diseases | 14 (22.9) |
| Sepsis | 10 (16.4) |
| Presence of potential DDI | 508 (93.8) |
| Mean pill/day | 15.1±6.9 |
| Median pill/day | 14 (4-30) |
| Pill burden ˃25/day | 9 (14.8) |
Others refer to HIV nephropathy (3.2%), sickle cell nephropathy (4.1%), and toxic nephropathy (5.8%). DDI – Drug–drug interaction; SD – Standard deviation
Figure 1.
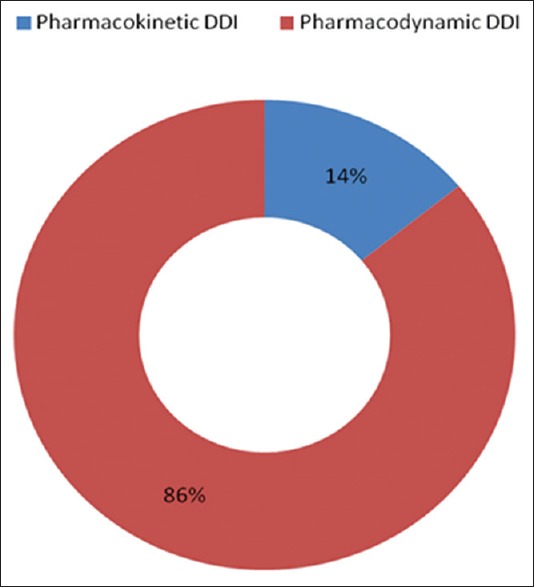
Distribution of drug–drug interaction according to pharmacological types among patients with renal dysfunction. The most common type of drug–drug interaction was pharmacodynamics drug–drug interaction (86%), whereas pharmacokinetic type (14%) was less common
Primary drug prescribed for patients with kidney disease
Antihypertensive drugs (259; 44.0%) were the most predominant primary drugs prescribed for patients with renal dysfunction [Figure 2]. Of the antihypertensive agents, angiotensin-converting enzyme inhibitors (ACEIs) (93; 36%) were the most commonly prescribed drugs involved in potential DDI when co-administered with other agents. Lisinopril (51; 19.7%) was the most common among the class of ACEIs prescribed. Other ACEIs were ramipril (32; 12.3%) and enalapril (10; 3.9%). Beta-blockers (12; 5.0%) were among the antihypertensive agents prescribed, for example, atenolol and metoprolol were the least prescribed. Other primary drugs prescribed included diuretics (104; 19.2%), for example, furosemide (40; 38%), spironolactone (35;33.7%), hydrochlorothiazide (16; 15.4%), and torsemide (13; 12.5%). Phosphate binders were among the primary drugs used for renal dysfunction (63; 11.6%). However, calcium carbonate was less commonly prescribed, but had potential for DDI when co-administered with other agents [Table 3].
Figure 2.
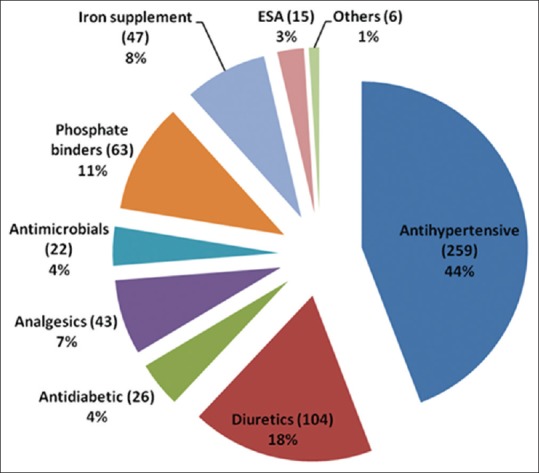
Category of drug used among patients with kidney disease. Antihypertensive agents (44%) were found to be the most prescribed drugs with potential drug–drug interaction followed by diuretics (18%), phosphate binders (11%), iron supplements (8%), analgesics (7%), antidiabetics (4%), antimicrobials (4%), and ESA- erythropoietin stimulating agents (3%)
Table 3.
Primary drugs prescribed for patients with renal dysfunction
| Drug categories | Frequency |
|---|---|
| Antihypertensive | |
| ACEIs | 93 |
| Lisinopril | 51 |
| Ramipril | 32 |
| Enalapril | 10 |
| Angiotensin receptor blockers | 82 |
| Losartan | 63 |
| Valsartan | 14 |
| Telmisartan | 5 |
| Calcium channel blockers | 72 |
| Amlodipine | 45 |
| Nifedipine | 27 |
| Beta-blockers | 12 |
| Atenolol | 7 |
| Metoprolol | 5 |
| Diuretics | 104 |
| Frusemide | 40 |
| Spirololactone | 35 |
| Hydroclorothiazide | 16 |
| Torsemide | 13 |
| Phosphate binders | 63 |
| Calcium carbonate | 32 |
| Calcitriol | 31 |
| Erythropoietin-stimulating agents (erythropoetin) | 15 |
ACEIs – Angiotensin-converting enzyme inhibitors
Co-administered drugs prescribed in patients with kidney disease
Table 4 shows other prescribed drugs with the potential risk of DDI when co-administered with primary drugs used in patients with renal disease. Iron supplements were frequently co-administered (47; 8.7%), followed by analgesic (43; 7.9%), antidiabetic (26; 4.8%), and antimicrobial (22; 4.1%) agents. Of the iron supplements, iron sucrose (36; 76.6%) and ferrous gluconate (11; 23.4%) were recorded. However, ibuprofen (15; 34.9%) and aspirin (28; 65.1%) were among the analgesic and antiplatelet drugs prescribed, respectively. Antidiabetic agents recorded were metformin (18; 69.2%) and glimepiride (8; 30.8%). Antimicrobials, for example, metronidazole, levofloxacin, and ciprofloxacin were among the least prescribed drugs [Figure 2].
Table 4.
Co-administered drugs prescribed for patients with renal dysfunction
| Drug categories | Frequency |
|---|---|
| Iron supplement | 47 |
| Iron sucrose | 36 |
| Ferrous gluconate | 11 |
| Analgesics | 43 |
| Ibuprofen | 15 |
| Aspirin | 28 |
| Antidiabetic agents | 26 |
| Metformin | 18 |
| Glimeperide | 8 |
| Antimicrobials | 22 |
| Levofloxacin | 10 |
| Ciprofloxacin | 6 |
| Metronidazole | 6 |
| Others | 6 |
*Others refer to antiemetic (4) and antimalarial (2)
Drug combinations with potential drug–drug interaction, possible clinical management, and severity rating
There are 14 potential DDIs between the primary drugs for renal dysfunction and co-administered drugs in the study [Table 5]. The drug pairs, their potential DDI, and the likely outcomes of the interactions are presented in Table 4.
Table 5.
Drug pairs with potential drug-drug interaction, clinical management, and interaction rating of some medications co-prescribed among renal patients
| Drug pairs | Potential drug interaction | Clinical management | Possible outcome | Frequency | Rating |
|---|---|---|---|---|---|
| Lisinopril and losartan | May result in increased risk of hypotension, syncope, hyperkalemia, changes in renal function, acute renal failure | If co-administration is required, closely monitor renal function | Possible deterioration in renal function | 128 | D |
| Lisinopril and erythropoetin | None | None | None | 20 | A |
| Lisinopril and aspirin | Aspirin may decrease the effectiveness of lisinopril | Although studies have suggested an interaction between ACE inhibitors and aspirin, the benefit against the risk should be weighed before co-prescribing the two drugs | Inhibition of prostaglandin syntheses | 38 | C |
| Lisinopril and furosemide | May result in first dose postural hypotension | Discontinue furosemide 2-3 days prior to adding lisinopril, if hypertension is not controlled with lisinopril only, furosemide may be restarted or started with a very low dose of lisinopril in the evening and closely monitor blood pressure. Monitor for hypotension and fluid status and check body weight regularly | Vasodilation and relative intravascular volume depletion | 43 | C |
| Lisinopril and metformin | May result in increased risk of hypoglycemia | Monitor patient closely for hypoglycemia. Upon discontinuation of lisinopril, monitor patient closely for worsening glycemic control | Increased blood glucose-lowering effects | 36 | C |
| Furosemide and aspirin | May result in decreased diuretic and antihypertensive efficacy | Monitor patient for diuretic efficacy and for signs of renal failure | Decreased renal prostaglandin production | 40 | C |
| Calcium carbonate and atenolol | May result in reduced effectiveness of atenolol | Instruct patient to avoid taking atenolol and calcium-containing compounds concurrently | Decreased atenolol absorption | 10 | B |
| Calcium carbonate and iron sucrose | May result in decrease in iron effectiveness | Instruct patient to space drug use | Decreased iron absorption | 9 | B |
| Calcium carbonate/ferrous gluconate | May result in decrease in iron effectiveness | Instruct patient to space drug use | Decreased iron absorption | 10 | B |
| Calcium carbonate/aspirin | May result in salicylate ineffectiveness | Monitor aspirin for reduced effectiveness and for possible toxicity upon initiation and withdrawal of calcium, respectively. Use buffered aspirin to limit the degree to which the urine is alkalized. | Increased renal clearance and decreased absorption due to increased urinary pH | 39 | C |
| Calcium carbonate and levofloxacin | May result in ineffectiveness of levofloxacin | If concomitant therapy is necessary, administer levofloxacin at least 2 hours before or after calcium | Decreased levofloxacin absorption due to chelation | 41 | C |
| Ferrous gluconate and calcium carbonate | May result in iron ineffectiveness | Advise patient to space drug use | Decreased iron absorption | 10 | B |
| Ferrous gluconate and calcitriol | May result in altered absorption of Vitamin D analog | Concomitant use is not recommended | Altered phosphate concentrations in the gastrointestinal tract | 37 | C |
| Ferrous gluconate and levofloxacin | May result in decreased plasma level of levofloxacin | Levofloxacin should be administered 2 hours before or 2 hours after a dose oral iron | Decreased levofloxacin absorption due to chelation | 40 | C |
| Others (grouped - each having frequency<5) | Varied | Varied | Varied | >5 (41) |
ACE: Angiotensin-converting enzyme
Potential drug–drug interaction and pattern of severity between primary and co-prescribed drugs
Table 6 shows the pattern of severity of potential DDI between antihypertensive (primary drug) and co-prescribed drugs among our cohort. Among the 541 prescriptions, 2 were contraindicated and rated category X, whereas 135 (24.9%) were of major severity and were classified as D. The majority of co-administered drugs (333; 61.4%) had potential interaction of moderate severity (Category C), whereas minor severity (Category B) was identified in 38 (7.1%) prescriptions. The overall prevalence of clinically significant DDIs was detected in 93.8% of the prescriptions administered to the patients.
Table 6.
Potential drug-drug interaction and pattern of severity rating between primary and co-prescribed drugs
| Rating | Designation | Number of prescriptions (%) |
|---|---|---|
| A | Unknown | 34 (6.22) |
| B | Minor | 38 (7.1) |
| C | Moderate | 333 (61.4) |
| D | Major | 135 (24.9) |
| X | Contraindicated | 2 (0.41) |
Glomerular filtration rate distribution and chronic kidney disease stages among patients with renal dysfunction
Figure 3 shows the GFR distribution and CKD stages among patients with renal dysfunction. The majority of the patients were within CKD stage 3 (65.5%).
Figure 3.
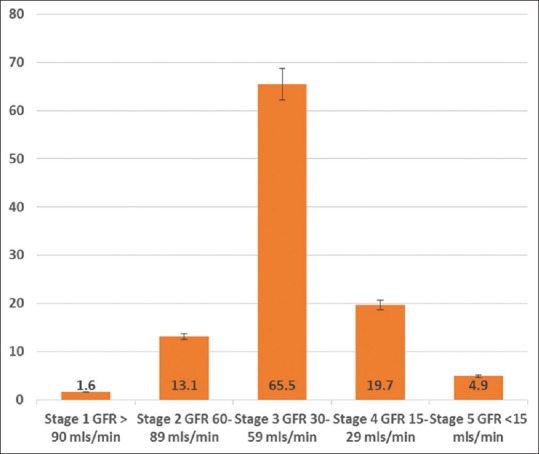
Glomerular filtration rate (GFR) distribution and chronic kidney disease (CKD) stages among the patients with renal dysfunction
Association between sociodemographic factors and the potential drug–drug interaction severity rating
Some sociodemographic factors, such as increasing age (P = 0.100), being a female (P = 0.697), being Yoruba ethnic group (P = 0.765), and higher level of education (P = 0.553), did not show any significant association with potential DDI severity among the patients admitted with renal dysfunction [Table 7].
Table 7.
Comparison of age, gender, ethnicity, and level of education with severity rating of potential drug-drug interaction in patients admitted for renal dysfunction
| Characteristics | Severity and prevalence of potential DDI (%) |
|||||
|---|---|---|---|---|---|---|
| Contraindication | Major | Moderate | Minor | χ2 | P | |
| Age group | ||||||
| <30 | 0 (0.0) | 13.3 (18) | 66.7 (27.0) | 20 (3.0) | 1.562 | 0.1 |
| 31-40 | 0 (0.0) | 18 (37.50) | 27 (56.30) | 3 (6.30) | ||
| 41-50 | 0 (0.0) | 8 (57.10) | 6 (42.90) | 0 (0.0) | ||
| 51-60 | 0 (0.0) | 7 (15.60) | 34 (75.6) | 4 (8.90) | ||
| >60 | 1 (1.0) | 25 (24.0) | 71 (68.30) | 7 (6.70) | ||
| Gender | ||||||
| Female | 0 (0.0) | 33 (27.3) | 78 (64.5) | 10 (8.3) | 1.436 | 0.697 |
| Male | 1 (1.0) | 27 (25.7) | 70 (66.7) | 7 (6.7) | ||
| Ethnicity | ||||||
| Igbo | 0 (0.0) | 20 (27.8) | 45 (62.5) | 7 (9.7) | 3.339 | 0.765 |
| Yoruba | 0 (0.0) | 23 (26.7) | 57 (66.3) | 5 (5.8) | ||
| Others | 1 (1.4) | 17 (24.6) | 46 (66.7) | 5 (7.2) | ||
| Education | ||||||
| Primary | 0 (0.0) | 1 (7.7) | 11 (84.6) | 1 (7.7) | 4.929 | 0.553 |
| Secondary | 0 (0.0) | 40 (28.4) | 91 (64.50) | 10 (7.10) | ||
| Tertiary | 1 (1.40) | 19 (26.40) | 46 (63.90) | 6 (8.30) | ||
Age, gender, ethnicity, and level of education were not significantly associated with the severity of DDI in patients admitted with renal dysfunction. DDI – Drug–drug interaction; EER – Exposure/exposed ratio
DISCUSSION
In this study, we reported the prevalence of DDI among patients admitted with renal dysfunction at a tertiary health facility in Lagos. Globally, clinicians are extremely concerned about potential DDIs in the management of CKD patients due to multiple drug therapies and pill burden that characterize the disease. Potential DDIs account for about 3%–5% of inpatient medication errors in the United States.13 Patients with kidney diseases are particularly prone to multiple drug therapies, and the risk of potential DDIs is high. Report on the prevalence of DDIs varies from region to region, and the rate varied widely from 32% to 96%.4,7,14 This may be due to the differences in the classification of DDI severity and the methods applied in each study. We documented 93.8% and 89.7% overall prevalence of DDI and clinically significant drug interaction (CSDI), respectively. The overall prevalence observed in our study was higher than those documented in other studies.15,16 This variation might have resulted from the different ways of determining the potential DDI, types of patients studied, and the types of co-prescribed drugs. It is well known that patients with renal disease are particularly administered multiple medications and are therefore susceptible to adverse drug reaction and adverse drug interactions.17,18 However, this high prevalence was similar to that reported by Fasipe et al.7 and Dubova et al.19 In another Brazilian study, the potential DDIs in CKD patients were investigated; Dijk et al. documented that potential DDIs were detected in 418 patients receiving 74.9% of the prescriptions.15 Similarly, Dubova et al. reported that 80% of the prescriptions for 624 ambulatory patients attending a family clinic in Mexico city had one or more potential DDIs compared to 93.8% overall prevalence of DDIs in our study.19 It is noted that majority of the data showed a high prevalence of potential DDIs even in other chronic conditions, apart from renal dysfunction. This may be attributed to the observation that multiple drug treatment and polypharmacy are common denominators in patients with chronic diseases and multiple co-morbidities.7,19
The current study also assessed the pattern of DDI severity, and we reported that 0.4% of the co-administered drugs are very harmful and contraindicated. Furthermore, majority (61.4%) of the prescriptions are categorized as Class C, indicating moderate DDI in which the interaction may result in exacerbation of the patient's condition, which may require an alteration in drug therapy. Major interaction (Category D), where the interaction may be life threatening and requiring medical intervention to minimize or prevent serious adverse events, was observed in 24.6% of the prescriptions. The clinically significant DDI was found to be quite high (89.7%), suggesting major risk to renal disease patients. This is similar to the report of a previous study by Marquito et al.,4 where a grand total of 1364 drug interactions were identified, with severe DDI accounting for 16.8% of the cases and absolute contraindications responsible for 0.4%. Risk factors for the occurrence of DDIs in the comparative study were obesity, diabetes mellitus, hypertension, and advanced-stage CKD.4 In another similar study where drug interactions among renal failure patients at a tertiary care hospital in Erode were investigated,20 a total of 210 patients with 318 drug interactions were found comprising major (44; 13.8%), moderate (198; 62.2%), and minor (76; 23.8%) interactions.20 Findings from our study, i.e. major (135; 24.9%), moderate (333; 61.4%), and minor (38; 7.1%) interactions, were, however, contrasting to those of the comparative study.
Pill burden and polypharmacy are common in patients being managed for renal diseases.21,22 These two factors are known contributors to the risk of potential DDI. According to the literature, patients taking five drugs are 50% more likely to suffer from drug interactions, whereas patients on seven or more medications are 100% more likely to experience the effects of drug interactions.23 Pill burden exceeding 25 pills/day was present in more than one-sixth of the patients (14.8%) in this study. A previous study reported a similar finding, in which the median number of medications/patient administered in the study population was 11, indicating polypharmacy which is a major risk factor for potential DDI. A study done by Glintborg et al.24 among 200 patients found that the median number of drugs used were eight with a range of 1–24 drugs, and polypharmacy was a predictable risk factor for medication errors in the study.24
Pharmacokinetic and pharmacodynamic types of drug interaction occur often in patients with multiple drug therapies.25,26 We reported that pharmacokinetic type of DDIs (85.9%) was more common in this study, which is comparable to other studies.16,20,27 This is, however, contrasting to other studies where pharmacodynamic interaction was predominant.26 In pharmacokinetic interaction, reciprocal influence of absorption and distribution in the various compartments, metabolism, and elimination can affect the effective concentrations at their sites of action, resulting in the formation of complexes, competition for uptake transporters, or induction of metabolizing enzymes and efflux transporters.26 The systems are becoming increasingly better understood so that some of the interactions of various drugs can be well predicted, partly with the help of computer programs for at least some certain drug groups.28
Drug-related problems are common to all stages of CKD, similar to the report in our study in which CKD Stage 3 was most common.29 However, most studies enrolled patients with advanced-stage CKD on hemodialysis,30 which is at variance to this study, focusing mainly on predialysis patients. The most commonly prescribed primary drugs in renal dysfunction in this study were antihypertensives (e.g., lisinopril), diuretics (e.g., furosemide), and phosphate binders (e.g., calcium carbonate). Lisinopril and losartan, lisinopril and furosemide, lisinopril and metformin, furosemide and aspirin, calcium carbonate and atenolol, calcium carbonate and ferrous gluconate, ferrous gluconate and calcitriol, and ferrous gluconate and levofloxacin were the common drug combinations with potential DDI observed in our study. As the present study was a prospective one, we set out to observe the actual interaction with the drug combinations during the follow-up period. However, the interaction manifestations observed were mild and sometimes mimicking the underlying symptoms of renal dysfunction that patients were originally admitted and being treated for. This made it difficult to differentiate the actual outcome of the drug interactions from that of the underlying disease entity. For instance, lisinopril and losartan interaction gave outcome relating to possible deterioration in renal function. While fluctuations in renal functions were observed in these patients on those combinations, it was difficult to attribute the symptoms solely to drug interaction as the underlying kidney disease could present in the same manner. Similar findings had been reported in other studies where the medicines commonly involved in potential DDI were those used daily to treat patients with chronic disorders. Beta-blockers, calcium channel blockers, diuretics, digoxin, calcium carbonate, and furosemide were among the most frequently prescribed medications in those studies.4,16,28,30,31
It is important to note that age, gender, ethnicity, and level of education were not significantly associated with the severity of drug interaction in this study. This implies that those sociodemographic characteristics did not contribute to the risk of potential DDI. This is at variance to the findings from other studies where obesity, hypertension, diabetes mellitus, as well as advanced stage of CKD were the risk factors strongly associated with DDI occurrence.4,30 This shows that comorbidities rather than age, gender, or level of education are factors to be considered while managing patients at risk for DDI.
This is a single-center study with a small sample size and was conducted over a 3-month period. A multicentric study spread over a longer period of time would be required in future to give a wider coverage of participation and ability to generalize our findings. Notwithstanding the limitations, this study provided significant clinical findings that are useful for clinicians to reduce the risk of drug interaction in at-risk patients such as those with renal dysfunction.
CONCLUSION
The rate of DDIs in patients admitted with renal dysfunction was very high, and majority were of clinically significant interactions. Polypharmacy and use of multiple medications may have contributed to this. It is, therefore, imperative for prescribers to be mindful of this phenomenon and use alternatives that can reduce the number of drugs administered to patients with kidney disease. Furthermore, healthcare providers should minimize DDIs by using software-based digital technologies for DDI check and other available methods.
Financial support and sponsorship
Nil.
Conflicts of interest
There are no conflicts of interest.
REFERENCES
- 1.Pirmohamed M. Drug-drug interactions and adverse drug reactions: Separating the wheat from the chaff. Wien Klin Wochenschr. 2010;122:62–4. doi: 10.1007/s00508-010-1309-1. [DOI] [PubMed] [Google Scholar]
- 2.Miller CD, El-Kholi R, Faragon JJ, Lodise TP. Prevalence and risk factors for clinically significant drug interactions with antiretroviral therapy. Pharmacotherapy. 2007;27:1379–86. doi: 10.1592/phco.27.10.1379. [DOI] [PubMed] [Google Scholar]
- 3.Leone R, Magro L, Moretti U, Cutroneo P, Moschini M, Motola D, et al. Identifying adverse drug reactions associated with drug-drug interactions: Data mining of a spontaneous reporting database in Italy. Drug Saf. 2010;33:667–75. doi: 10.2165/11534400-000000000-00000. [DOI] [PubMed] [Google Scholar]
- 4.Marquito AB, Fernandes NM, Colugnati FA, de Paula RB. Identifying potential drug interactions in chronic kidney disease patients. J Bras Nefrol. 2014;36:26–34. doi: 10.5935/0101-2800.20140006. [DOI] [PubMed] [Google Scholar]
- 5.Wouters OJ, O'Donoghue DJ, Ritchie J, Kanavos PG, Narva AS. Early chronic kidney disease: Diagnosis, management and models of care. Nat Rev Nephrol. 2015;11:491–502. doi: 10.1038/nrneph.2015.85. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Adibe MO, Ewelum PC, Amorha KC. Evaluation of drug-drug interactions among patients with chronic kidney disease in a South-Eastern Nigeria tertiary hospital: A retrospective study. Pan Afr Med J. 2017;28:199. doi: 10.11604/pamj.2017.28.199.13622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Fasipe OJ, Olayemi SO, Akinyede AA, Osho PO, Ibiyemi-Fasipe OB, Osho ES. How do we prevent the burden of extremely harmful and clinically nonbeneficial drug drug interactions among chronic kidney disease patients? Toxicol Res Appl. 2018;2:1–14. [Google Scholar]
- 8.Culleton BF, Larson MG, Evans JC, Wilson PW, Barrett BJ, Parfrey PS, et al. Prevalence and correlates of elevated serum creatinine levels: The Framingham heart study. Arch Intern Med. 1999;159:1785–90. doi: 10.1001/archinte.159.15.1785. [DOI] [PubMed] [Google Scholar]
- 9.Visser CM, Pathmanathan I, Brownlee A. Designing Conducting Health Systems Research Projects International Development Centre. Vol. 2. Geneva: Oltawa and World Health Organization; 2005. p. 216. [Google Scholar]
- 10.Ulasi II, Ijoma CK, Onodugo OD, Arodiwe EB, Ifebunandu NA, Okoye JU. Towards prevention of chronic kidney disease in Nigeria: A community-based study in Southeast Nigeria. Kidney Int Suppl. 2013;3:195–201. [Google Scholar]
- 11.Truven Health Analytic. New York, USA: IBM® Watson, Inc; 2017. [Last accessed on 2017 Feb 12]. Micromedex® Database on the Internet. Available from: https://www.micromedexsolutions.com/ibm/watsonhealth/learn/truvenhealthanalytics/micromede×2/4.77.0/WebHelp/Tools/MOBILE/Android_APPS . [Google Scholar]
- 12.Armahizer MJ, Kane-Gill SL, Smithburger PL, Anthes AM, Seybert AL. Comparing drug drug interaction severity for clinician opinion to propriety databases. ISRN Crit Care. 2013;2013:347346. [Google Scholar]
- 13.Duke JD, Han X, Wang Z, Subhadarshini A, Karnik SD, Li X, et al. Literature based drug interaction prediction with clinical assessment using electronic medical records: Novel myopathy associated drug interactions. PLoS Comput Biol. 2012;8:e1002614. doi: 10.1371/journal.pcbi.1002614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Oshikoya KA, Oreagba IA, Lawal S, Awodele O, Ogunleye OO, Senbanjo IO, et al. Potential drug-drug interactions in HIV-infected children on antiretroviral therapy in Lagos, Nigeria. HIV AIDS (Auckl) 2014;6:49–59. doi: 10.2147/HIV.S52266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Dijk KV, De-Vires CS, Van-Den-Berg PB, Brouwers JR, Van-Den-Berg LT. Occurrence of potential drug-drug interactions in nursing home residents. Int J Pharm Pract. 2001;9:45–54. [Google Scholar]
- 16.Rama M, Viswanathan G, Acharya LD, Attur RP, Reddy PN, Raghavan SV. Assessment of drug-drug interactions among renal failure patients of nephrology ward in a South Indian tertiary care hospital. Indian J Pharm Sci. 2012;74:63–8. doi: 10.4103/0250-474X.102545. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Dumbreck S, Flynn A, Nairn M, Wilson M, Treweek S, Mercer SW, et al. Drug-disease and drug-drug interactions: Systematic examination of recommendations in 12 UK national clinical guidelines. BMJ. 2015;350:h949. doi: 10.1136/bmj.h949. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Sharif-Askari FS, Sulaiman SA, Sharif-Askari N, Al Sayed Hussain A. Development of an adverse drug reaction risk assessment score among hospitalized patients with chronic kidney disease. PLoS One. 2014;9:e95991. doi: 10.1371/journal.pone.0095991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Dubova SV, Reyes-Morales H, Torres-Arreola Ldel P, Suárez-Ortega M. Potential drug-drug and drug-disease interactions in prescriptions for ambulatory patients over 50 years of age in family medicine clinics in Mexico city. BMC Health Serv Res. 2007;7:147. doi: 10.1186/1472-6963-7-147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Jose R, Varghese AA, Kannan C. A study on drug interactions among renal failure patients in tertiary care hospital, Erode. World J Pharm Pharm Sci. 2017;6:12. [Google Scholar]
- 21.Sutaria A, Liu L, Ahmed Z. Multiple medication (polypharmacy) and chronic kidney disease in patients aged 60 and older: A pharmacoepidemiologic perspective. Ther Adv Cardiovasc Dis. 2016;10:242–50. doi: 10.1177/1753944716634579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mason NA. Polypharmacy and medication-related complications in the chronic kidney disease patient. Curr Opin Nephrol Hypertens. 2011;20:492–7. doi: 10.1097/MNH.0b013e328349c261. [DOI] [PubMed] [Google Scholar]
- 23.Delafuente JC. Understanding and preventing drug interactions in elderly patients. Crit Rev Oncol Hematol. 2003;48:133–43. doi: 10.1016/j.critrevonc.2003.04.004. [DOI] [PubMed] [Google Scholar]
- 24.Glintborg B, Andersen SE, Dalhoff K. Drug-drug interactions among recently hospitalised patients frequent but mostly clinically insignificant. Eur J Clin Pharmacol. 2005;61:675–81. doi: 10.1007/s00228-005-0978-6. [DOI] [PubMed] [Google Scholar]
- 25.Cascorbi I. Drug interactions principles, examples and clinical consequences. Dtsch Arztebl Int. 2012;109:546–55. doi: 10.3238/arztebl.2012.0546. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Palleria C, Di Paolo A, Giofrè C, Caglioti C, Leuzzi G, Siniscalchi A, et al. Pharmacokinetic drug-drug interaction and their implication in clinical management. J Res Med Sci. 2013;18:601–10. [PMC free article] [PubMed] [Google Scholar]
- 27.Nazari MA, Moqhadam NK. Evaluation of Pharmacokinetic drug interactions in Prescriptions of Intensive Care Unit in a teaching hospital. Iran J Pharm Res. 2006;5:215–8. [Google Scholar]
- 28.Li L, Yu M, Chin R, Lucksiri A, Flockhart DA, Hall SD. Drug-drug interaction prediction: A Bayesian meta-analysis approach. Stat Med. 2007;26:3700–21. doi: 10.1002/sim.2837. [DOI] [PubMed] [Google Scholar]
- 29.Cardone KE, Bacchus S, Assimon MM, Pai AB, Manley HJ. Medication-related problems in CKD. Adv Chronic Kidney Dis. 2010;17:404–12. doi: 10.1053/j.ackd.2010.06.004. [DOI] [PubMed] [Google Scholar]
- 30.Al-Ramahi R, Raddad AR, Rashed AO, Bsharat A, Abu-Ghazaleh D, Yasin E, et al. Evaluation of potential drug- drug interactions among Palestinian hemodialysis patients. BMC Nephrol. 2016;17:96. doi: 10.1186/s12882-016-0317-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Hedge S, Udaykumar P, Manjuprasad MS. Potential drug interactions in chronic kidney disease patients. A cross-sectional study. Int J Recent Trends Sci Technol. 2015;16:56–60. [Google Scholar]


