Abstract
α-Fetoprotein (AFP) is considered a good target for immunotherapy strategies against hepatocellular carcinoma (HCC); however, no immunodominant AFP-derived MHC class II-restricted helper T-lymphocyte (HTL) epitope has been reported. Therefore, we identified novel AFP-derived HTL epitopes possessing high immunogenicity. HTL epitopes were predicted using the online service, and peptides were subsequently synthesized. Four newly synthesized peptides showed positive reactivity in >20% patients on ELISPOT using peripheral blood mononuclear cells (PBMCs). Among these, the highest rate was shown by AFP1 (MKWVESIFLIFLLNFTESRT), which also showed the highest positive rate in cell proliferation assays. Binding assays demonstrated that AFP1 had strong binding properties toward MHC molecules. Further, blocking assays performed using an anti-HLA-DR antibody showed that immune response decreased, confirming the binding of AFP1 to HLA-DR molecules. Furthermore, the survival rates of patients with stages II–IV HCC indicated that T cell response against AFP1 led to significantly greater survival that of patients without T cell response. When evaluating immune response against AFP1 before and after HCC treatment, an increase in the frequency of peptide-specific T cells was observed after treatment in patients with HLA-DRB1*1502, *0405, and *0901 alleles. In conclusion, the identified epitopes may be useful for immunotherapy strategies against HCC.
Subject terms: Cancer, Immunology
Introduction
Among the different types of cancer, liver cancer is the second and sixth most common cause of death in males and females, respectively, worldwide1. Hepatocellular carcinoma (HCC) accounts for 70%–90% of primary liver cancers1. Chronic liver injury and liver cirrhosis are the major factors considered responsible for the development of liver cancer2. Current treatments for early-stage HCC include resection, transplantation, and locoregional therapy. However, the high rate of post-treatment relapse remains a challenge. Moreover, limited options are available for the treatment of advanced stage HCC3,4, with the exception of systemic therapies such as sorafenib, regorafenib, and lenvatinib5–7. The drug cabozantinib has been shown to prolong the survival period in HCC8; however, in general, these drugs prolong survival by only a few months.
In recent years, treatments using immune checkpoint inhibitors (ICIs) and chimeric antigen receptor-modified T cells have been clinically used in cancer immunotherapy and have shown positive therapeutic efficacies. Indeed, recently developed cancer immunotherapy strategies, which substantially differ from current cancer treatments, represent an innovative approach because they involve the host immune system as the target. Thus, novel cancer treatment regimens are being developed as a paradigm shift. Furthermore, the importance of recognizing the antigens expressed by cancer cells as targets and the removal of those cancer cells by T cells has been highlighted by the success of these immunotherapy treatments. Accordingly, research to identify highly immunogenic T cell epitopes that recognize such antigens, including neoantigens and T cell receptors, is underway.
Clinical trials are ongoing for several ICIs (anti-PD-1, PD-L1, and CTLA-4 antibodies) for HCC treatment; however, reports have indicated that the overall response rate is approximately 20%, with the efficacies of the treatment strategies using ICI alone considered insufficient. Therefore, the development of combination therapies, such as combinations of one ICI with another ICI, molecular target drug, or other immune therapy, has garnered increasing attention.
AFP is a cancer-related antigen frequently expressed in HCC and used as a marker for HCC diagnosis and therapeutic efficacy. As such, the expression of AFP in healthy individuals is believed to be rare; therefore, the antigen is considered a promising target for cancer immunotherapy9. Among the available immunotherapies for liver cancer using AFP as a target, therapies using dendritic cells (DCs) stimulated with AFP-derived epitopes and vaccine therapies by class I-restricted peptides have been developed. However, these therapies have not shown sufficient efficacy in clinical settings10,11.
We previously reported on AFP-derived major histocompatibility complex (MHC) class I-restricted epitopes12. In this previous study, we treated HCC patients with an AFP peptide vaccine against the peptide and successfully obtained specific TCRs from the patients who positively responded13. These results indicated the efficacy of immunotherapy strategies using epitopes with strong immunogenicity for HCC. On the other hand, cases with limited efficacy indicated the necessity of an improved strategy to improve overall treatment efficacy. In recent years, it has been demonstrated that the function of helper T lymphocytes (HTLs) is important for increasing the therapeutic efficacy of immunotherapy strategies, in addition to that of cytotoxic T lymphocytes (CTLs). HTLs are believed to contribute to cancer therapy by increasing the reactivity of CTLs14,15. In fact, clinical trials using wild-type 1 peptide-pulsed DCs have indicated that activation of both CTLs and HTLs results in a better clinical response compared with the CTL vaccine alone16.
To date, several studies have reported on AFP-derived MHC class II-restricted epitopes. However, the identification of immunodominant epitopes with strong immunogenicity has not been reported17–19. Therefore, in this study, we identified and characterized novel AFP-derived MHC class II-restricted epitopes.
Materials and Methods
Patient population
Samples were collected from 47 patients (39 males and 8 females) aged 40 to 81 years (median: 68 years). Of these, 36 patients had HCC and 11 patients had a history of liver cancer. These patients regularly visited the Department of Gastroenterology at Kanazawa University Hospital (Kanazawa, Japan) between 2007 and 2013. Blood samples were collected from 36 cancer-bearing patients before treatment. In addition, blood samples were collected after treatment (3 days to 42 days) in 5 patients to compare responses before and after treatment. Eleven non-cancerous patients had a period of 0.6–64 months after the previous treatment at the time of blood collection. After receiving consent, samples were collected from 14 healthy adult individuals (Table 1). The diagnosis of HCC was based on typical patterns, such as hyperattenuation areas in the early phase and hypoattenuation areas in the late phase, by dynamic CT or MRI. We performed CT scan (from chest to pelvis), upper gastrointestinal endoscopy, and colonoscopy to confirm that there were no other malignancies. We further confirmed that the healthy individuals had no history of malignant tumors and were negative for the hepatitis B virus surface antigen (HBsAg) and HCV antibody. All methods were carried out in accordance with relevant guidelines and regulations. According to the Helsinki Declaration, the study protocol was explained to the subjects and informed consent was obtained from all who wished to participate. This study was approved by the regional ethics committee (Medical Ethics Committee of Kanazawa University, Protocol #829).
Table 1.
Characteristics of the patients studied.
| Clinical diagnosis | Patient No. | Sex, M/F | Age, years | ALT, IU/L | Lymph cell, /dL | AFP, ng/mL | Child-Pugh, A/B/C | Tumor size, large/small | Tumor multiplicity, multiple/solitary | Sstage, I/II/III/IV |
|---|---|---|---|---|---|---|---|---|---|---|
| Tumor-bearing patients | 36 | 31/5 | 68 (40–81) | 37.5 (10–159) | 1005 (380–2100) | 27 (6–40550) | 31/3/1a | 11/25 | 24/12 | 8/16/5/7 |
| Previous treatment subjects | 11 | 8/3 | 66 (54–80) | 42 (10–89) | 1140 (450–2520) | 11 (4–139) | 9/2/0 | 4/7 | 2/9 | 6/4/1/0 |
| Normal Donors | 14 | 12/2 | 32 (28–38) | 14 (11–23) | ND | ND | ND | ND | ND | ND |
Data are shown as the median (range); Large, >2 cm; small, ≤2 cm; ND, not determined.
aOne patient on anticoagulant medication was excluded.
Laboratory and virological testing
Blood samples were tested for the HBsAg and HCV antibody using commercial assay kits (Fuji Rebio, Tokyo, Japan). For human leukocyte antigen (HLA) typing, peripheral blood samples collected from the patients and healthy individuals were outsourced for analysis (BMI Inc., Tokyo, Japan) and confirmed using PCR. Serum AFP levels were measured by EIA (AxSYM AFP, Abbott Japan, Tokyo, Japan).
Synthetic peptides
Amino acid sequences of AFP (GenBank accession number AAB58754)-derived MHC class II-binding HTL epitopes were predicted using ProPred, an epitope prediction algorithm (https://webs.iiitd.edu.in/raghava/propred/index.html). The predicted threshold was set to the default value of 3%. The prediction score was calculated using Propred and TEPITOPE (http://datamining-iip.fudan.edu.cn/service/TEPITOPEpan/index.html). Each peptide was synthesized to contain nine core amino acids, which were selected according to the AFP amino acid sequence. Peptide synthesis was performed using Mimotope (Melbourne, Australia) and purity ≥ 80% was confirmed using high-performance liquid chromatography.
Preparation of peripheral blood mononuclear cells (PBMCs)
PBMCs were prepared using a method described previously12. Briefly, whole blood samples were collected from patients and healthy individuals, and PBMCs were isolated by centrifugation using Lymphoprep (AXIS-SHIELD PoC AS, Oslo, Norway). The isolated PBMCs were resuspended in RPMI 1640 (Gibco, Grand Island, NY) containing 10% heat-inactivated fetal bovine serum (FBS), 100 U/mL penicillin, and 100 μg/mL streptomycin. Fresh PBMCs were used for proliferation assays and unused PBMCs were stored frozen at −80 °C until use.
Enzyme-linked immunospot (ELISPOT) analysis
For ELISPOT analysis, 96-well plates (MultiScreen; Merck Millipore, Cork, Ireland) were coated with the anti-human IFN-γ antibody (Mabtech, Nacka, Sweden). The antibody was fixed by overnight incubation at 4 °C. The following day, each well was washed four times with phosphate-buffered saline (PBS) and blocked with RPMI 1640 containing 5% FBS for 2 h at 25 °C. Using two wells for each peptide, 10 μg/mL of the peptide and 3 × 105 PBMCs were added to each well and the mixture was cultured in RPMI 1640 containing 5% FBS. After a 24 h incubation period for the PBMCs with peptides, the plates were washed eight times and 100 μL of the biotin-labeled anti-human IFN-γ antibody was added and the plates were further incubated overnight at 4 °C. After washing the plates four times, streptavidin AP (Mabtech) was added to each well and the plates were incubated for 2 h. After washing the plates four times with PBS, freshly prepared NBT/BCIP solution (Biorad, Hercules, CA) was added. The plates were washed with distilled water to stop the reaction and dried at room temperature. The number of specific spots was calculated by subtracting the number of spots in a well without peptides from the number of spots in a well with peptides. The reaction was considered positive when >10 specific spots were calculated and there were more than twice the number of spots in the wells with peptides than in those without peptides. The negative control wells contained only PBMCs in the medium, whereas the positive control wells contained 10 ng/mL phorbol 12-myristate 13-acetate (Sigma, St. Louis, MO, USA).
The reactivity of the PBMCs against the peptides after the depletion of CD8+ and CD4+ cells was evaluated by ELISPOT, as described above. The determination of the depletion of the CD8+ and CD4+ cells was performed by labeling the PBMCs with MicroBeads (Miltenyi Biotec, Auburn, CA) followed by the use of a magnetic separator.
Proliferation assays
For proliferation assays, 10 μg/mL of each peptide and 2 × 105 freshly prepared PBMCs were added to each well of a 96-well plate and cultured in RPMI 1640 containing 10% FBS. After a 5 day culture period, 1 μCi/mL 3H-thymidine was added to each well and the cells were cultured for a further 24 h. The contents of each well were then transferred onto a 96-well filter plate using a cell harvester, and the radioactive uptake by the cells in each well was measured using a liquid scintillation counter. With regard to the stimulation index, the results were considered positive when the mean value of a well containing a target peptide was more than twice the mean value of a negative control well. The negative control wells contained only PBMCs and medium, while the positive control wells contained 10 ng/mL phytohemagglutinin (PHA).
Peptide-binding assays
An immortalized EBV-infected human B-lymphocyte cell line (kindly provided by RIKEN BRC through the National Bio-Resource Project of MEXT, Japan), expressing HLA class II molecules, was used for the peptide-binding assays as HLA class II-expressing cells. The immortalized human cells were cultured in RPMI 1640 containing 20% FBS. The immortalized human cells and fluorescein isothiocyanate (FITC)-labeled peptides (1 mg/mL) were inoculated in each well of a 96-well plate and incubated for 2 h. Each well was washed twice with PBS, and the cells were analyzed using flow cytometry. The wells containing FITC-labeled preS1 (23–33)20 (a peptide derived from the hepatitis B virus, which is an HLA-DRB1*0405-restricted epitope) were used as control wells, while those containing the immortalized human cells without the addition of a peptide were used as the negative control. In addition, the leukemia cell line K562 was used as a negative control.
Blocking assays
Blocking assays using the HLA antibodies were evaluated by ELISPOT. In brief, 10 μg/mL of the HLA-ABC (W6/32; BioLegend, Dan Diego, CA), HLA-DP (BRAFB6: Santa Cruz Biotechnology, Dallas, TX), HLA-DQ (SPV-L3: AbD Serotec, Oxford, UK), and HLA-DR (L243: BioLegend, SanDiego, CA) antibodies was added to the wells containing PBMCs and peptides for co-culture. The wells containing PBMCs alone or PBMCs with a peptide were used as controls.
Statistical analysis
Univariate analyses comparing the background factors of the T cell response positive and negative groups against the AFP-derived epitopes were performed using the Mann–Whitney U-test and chi-square test. Cause-specific survival (CSS) estimates were calculated using the Kaplan–Meier method. P-values <0.05 were considered statistically significant.
Results
Patient profiles
Clinical data from the HCC patients who participated in this study are summarized in Table 1. During blood collection, 36 patients had HCC and 11 patients had a history of HCC treatment, with median serum AFP levels of 27 and 11 ng/mL, respectively. Of the 36 HCC patients, 11 had a primary tumor >2 cm in diameter, while the remaining patients had a primary tumor of ≤2 cm in diameter. In addition, 24 patients had multiple tumors with ≥ 2 nodes, while the remaining patients had a solitary tumor. Based on the Union for International Cancer Control TNM classification, 8, 16, 5, and 7 patients had stage I, II, III (A/B), and IV (A/B) tumors, respectively.
Selection of potential MHC class II-restricted peptides derived from AFP
ProPred was used to predict the amino acid sequences. The amino acid sequence for the MHC class II allele was predicted from the entire set of HLA-DRB1 epitopes selected by the online service. In total, there were 14 types of peptides, of which 3 were for pre-existing peptides. Therefore, the remaining 11 types were selected from the peptides predicted to bind to multiple DRB1 alleles within the amino acid sequence, followed by the selection of additional peptides for HLA-DRB1*0405 (13.4% of the Japanese population21), *1502 (10.6% of the Japanese population), *1501 (7.7% of the Japanese population), *1302 (5.9% of the Japanese population), and *0101 (5.8% of the Japanese population), which occur at high frequencies in the Japanese population (Table 2). The prediction score of each peptide was calculated using ProPred and TEPITOPE.
Table 2.
Peptides.
| Peptide | Source | Start position | Amino acid sequence | Prediction score (ProPred) | Prediction score (TEPITOPE) | ||||
|---|---|---|---|---|---|---|---|---|---|
| DRB1 *0405 | DRB1 *0901 | DRB1 *1502 | DRB1 *0405 | DRB1 *0901 | DRB1 *1502 | ||||
| AFP1 | AFP | 1 | MKWVESIFLIFLLNFTESRT | 4.4500 | 5.6000 | 2.4264 | 2.814 | 5.5083 | |
| AFP22 | AFP | 22 | HRNEYGIASILDSYQCTAEI | 2.9000 | 3.6500 | 3.0698 | 2.0349 | 3.6029 | |
| AFP 62 | AFP | 62 | EVSKMVKDALTAIEKPT | 2.0000 | 1.8000 | 0.3903 | −0.3918 | 1.7524 | |
| AFP 134 | AFP | 134 | ASIPLFQVPEPVTSC | 1.0000 | 1.2000 | 0.9899 | 0.943 | 1.2312 | |
| AFP 155 | AFP | 155 | RETFMNKFIYEIARRHPFLY | 1.2000 | 2.8000 | 1.3064 | 2.6014 | 2.7413 | |
| AFP 169 | AFP | 169 | RHPFLYAPTILLWAARYDKII | 3.7800 | 3.7600 | 3.2598 | 2.7795 | 3.6407 | |
| AFP 210 | AFP | 210 | TKELRESSLLNQHACAVMKN | 0.1000 | 0.9800 | 0.1937 | 0.3807 | 0.998 | |
| AFP 224 | AFP | 224 | CAVMKNFGTRTFQAITVTKLSQ | 3.6000 | 2.2000 | 2.7082 | 2.0703 | 2.145 | |
| AFP 246 | AFP | 246 | KFTKVNFTEIQKLVLD | ND | 1.5000 | −0.4778 | 0.82 | 1.3595 | |
| AFP 346 | AFP | 346 | EKNIFLASFVHEYSRRHPQL | 2.6000 | 0.7000 | 2.6849 | 1.3533 | 0.7363 | |
| AFP 361 | AFP | 361 | RHPQLAVSVILRVAKG | ND | 1.4500 | −0.909 | −1.1422 | 1.3422 | |
| AFP 416 | AFP | 416 | CGLFQKLGEYYLQNAFLVAYT | 1.4500 | 2.8000 | 1.2723 | 3.2562 | 2.7063 | |
| AFP 428 | AFP | 428 | QNAFLVAYTKKAPQLTSSELM | 2.1000 | 1.9000 | 2.4694 | 1.5843 | 1.8786 | |
| AFP 549 | AFP | 549 | KQEFLINLVKQKPQITEEQ | 0.6000 | ND | 1.7001 | 1.0736 | 0.0559 | |
| preS1 | HBV preS | 23 | GFFPDHQLDPA | ND | 0.7000 | −1.3916 | −1.9603 | 0.6149 | |
AFP134, AFP246 and AFP361 contain the previously published epitopes. “preS1” is the previously published epitope.
ND, no data.
PBMC response to AFP-derived epitopes
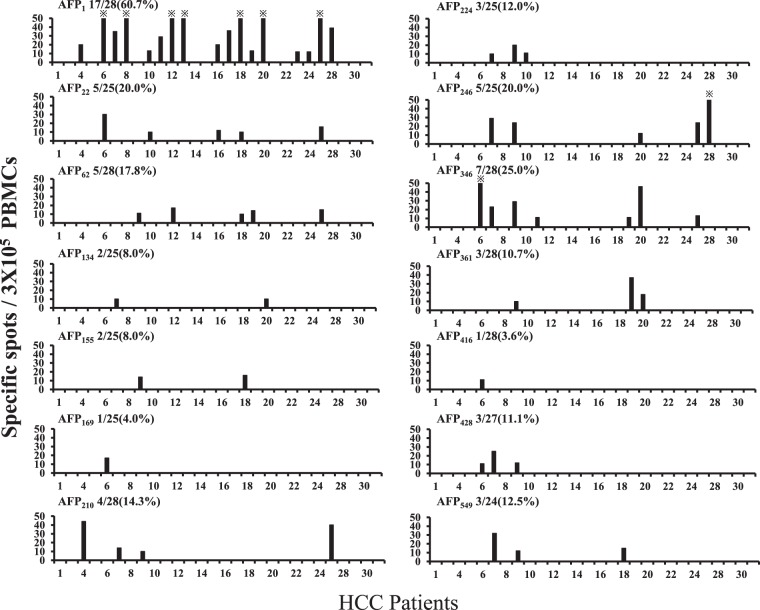
An IFN-γ ELISPOT assay was performed to evaluate whether PBMCs from the 28 HCC patients reacted to the synthesized peptides. The peptides that showed positive reactivity in >20% of the patients were AFP1, AFP22, AFP246, and AFP346, with reaction rates of 60.7%, 20.0%, 20.0%, and 25.0%, respectively (Fig. 1). In contrast, the peptides that showed positive reactivity in <20% of the patients were AFP62, AFP134, AFP155, AFP169, AFP210, AFP224, AFP361, AFP416, AFP428, and AFP549. Among all the peptides, AFP1 showed the highest positive reaction rate for PBMCs and had the highest number of wells with >50 spots.
Figure 1.
Analysis of peripheral blood T cell responses to AFP-derived epitopes. The number of spots on IFN-γ ELISPOT is shown. The response was determined to be positive when >10 specific spots were calculated and there were more than twice the number of spots in wells with peptides than in those without peptides. The amino acid sequence of each peptide is shown in Table 2. *indicates >50 spots.
Similarly, the reactivity of PBMCs collected from the 14 healthy individuals to the synthesized peptides was evaluated using IFN-γ ELISPOT. The proportion of PBMCs showing positive reactivity tended to be lower than that of the PBMCs collected from the HCC patients. The peptides AFP1, AFP246, AFP346, and AFP361 showed positive reactivity in >20% of the healthy individuals; however, none of the wells with these peptides had a spot count >50 (Supplementary Fig. 1). Representative images of Fig. 1 and Supplementary Fig. 1 are shown in Supplementary Fig. 2.
IFN-γ ELISPOT for CD8+ and CD4+ cell-depleted PBMCs
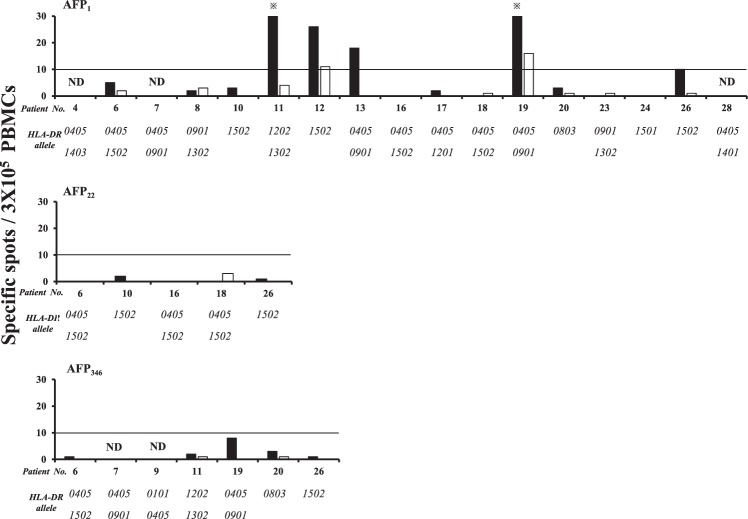
To confirm whether the lymphocytes that showed reactivity in the ELISPOT screening (Fig. 1) were CD4+ or CD8+ cells, MicroBeads were used to deplete CD8 and CD4 cells from the PBMCs, and IFN-γ ELISPOT was performed. The purity of the population was confirmed using flow cytometry (Supplementary Fig. 3). In all cases where CD8/4 was depleted, the fraction was validated by flow cytometry analysis. The peptides AFP1, AFP22, and AFP346, which showed a high rate of reactivity on ELISPOT, were used for this experiment (Fig. 2). The CD8+ cell-depleted PBMCs had a higher number of spots than the CD4+ cell-depleted PBMCs, indicating that the positive reactivity observed on ELISPOT (Fig. 1) was due to CD4+ cells. Representative images of Fig. 2 are shown in Supplementary Fig. 4.
Figure 2.
Responses of CD8+ and CD4+ cell-depleted PBMCs to AFP-derived epitopes. The numbers of spots on IFN-γ ELISPOT and the HLA-DR alleles of each patient are shown. Black and white bars denote the results for CD8+ and CD4+ cell-depleted PBMCs, respectively. The positive rate of CD8+ cell-depleted PBMCs against AFP1 was 35.7% (5/14), while that of CD4+ cell-depleted PBMCs was 14.3% (2/14). ND denotes “not determined”. *indicates >30 spots.
In addition, analysis of the HLA-DR alleles of the patients whose PBMCs reacted positively after being depleted of CD8+ cells showed the presence of many HLA-DRB1*0405, *0901, and *1502 alleles, indicating that these HLA-DR alleles may have contributed to the observed reactions. The number of spots observed for AFP1, which showed the highest reactivity during the earlier screening by ELISPOT (Fig. 1), was also demonstrated to be high in the second ELISPOT (Fig. 2).
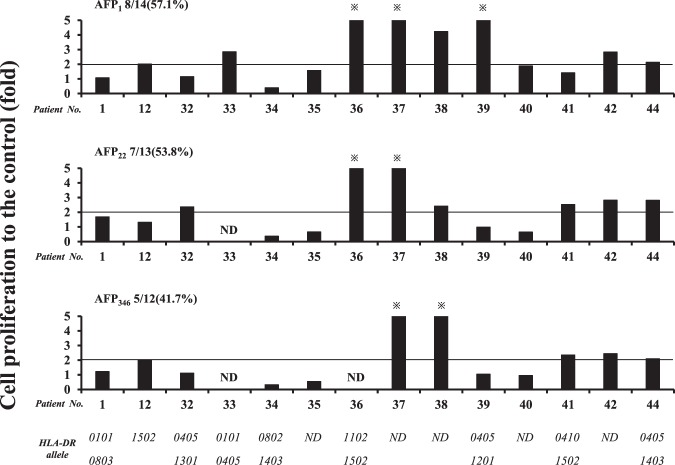
Proliferation assays
Proliferation assays were performed to evaluate the reactivity of PBMCs to peptides according to the degree of cellular proliferation (Fig. 3). As shown in Fig. 2, the peptides used for these assays included AFP1, AFP22, and AFP346, which were the peptides that showed high reactivity in the screening by ELISPOT (Fig. 1). In addition to the PBMCs from patients used during the screening by ELISPOT, fresh PBMCs were collected from the HCC patients and those with a history of HCC treatment. All three peptides showed a positive reactivity of >40%, with AFP1 showing the highest positive rate. When the measured value was compared with the negative control, it was significantly higher for AFP1 and AFP22 (Supplementary Fig. 5). In several patients, the IFN-γ ELISPOT of PBMCs co-cultured with the peptide for 5 days was performed in the same manner as in the proliferation assays and the response was stronger than at the time of the screening ELISPOT (Supplementary Fig. 6). Therefore, we confirmed antigen-specific proliferation. Thus, the proliferation assays confirmed that the PBMCs reacted to the peptides. Furthermore, HLA-DRB1*0405 and *1502 were found to be present in the patients whose HLA-DR alleles could be analyzed.
Figure 3.
Analysis of the proliferative activity of PBMCs against AFP-derived epitopes. Multiple analyses comparing the control and the HLA-DR alleles of each patient are shown. Positivity was assigned when the stimulation index was more than twice that of the control. Positive frequencies for each peptide are shown. ND denotes “not determined”. *indicates >5.
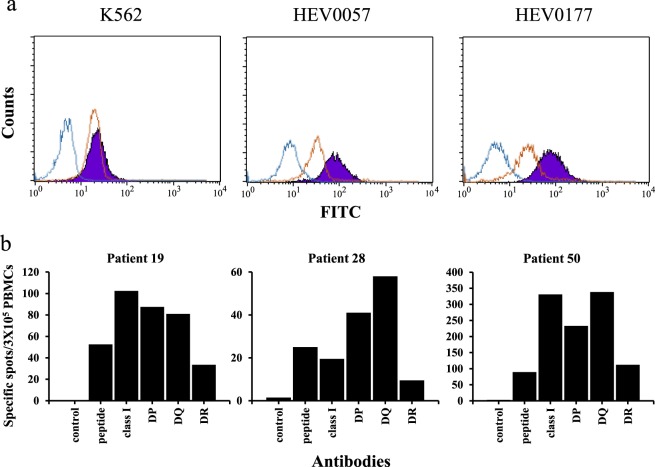
Binding assays
Binding assays were performed using AFP1, which was the peptide that showed the highest reactivity to PBMCs, in order to analyze its affinity towards HLA-DR molecules (Fig. 4a). Using FITC-labeled AFP1, the binding affinity of the peptide to cell lines expressing HLA-DR was analyzed by flow cytometry. Cell lines expressing homozygous HLA-DR molecules, including HEV0057 (HLA-DRB1*0901) (RRID:CVCL_T098) and HEV0177 (HLA-DRB1*0405) (RRID:CVCL_T166), were used. The K562 cell line, which does not express HLA molecules, was used as a negative control. FITC-labeled preS1 (23–33)20, an HLA-DRB1*0405-restricted epitope, was used as a control peptide. For the K562 cells, the fluorescence intensities of preS1 and AFP1 were at similar levels; however, for the HEV0577 and HEV0177 cells, the fluorescence intensity of AFP1 was stronger than that of preS1. These results confirmed the binding of AFP1 to the HLA molecules. Each experiment was performed three times on different days, with all three repetitions showing similar results. Representative data are shown.
Figure 4.
Analysis of the binding of labeled peptides to MHC molecules and the direct ex vivo analysis of PBMCs against AFP1 epitopes using an HLA antibody. (a) The affinity of each FITC-labeled AFP1 epitope and positive control peptides (preS1 23–32) to the human immortalized cells was measured using flow cytometry. HEV0057 and HEV0177 cells expressed homozygous HLA-DRB1*0901 and *0405, respectively. Control (without peptides), positive control peptide, and AFP1 are shown in blue, reddish brown, and purple, respectively. Each experiment was performed three times. (b) Direct ex vivo analyses (IFN-γ ELISPOT) of PBMCs against AFP1 were conducted using the HLA class I, HLA-DP, HLA-DQ, or HLA-DR antibodies. The numbers of spots are shown. Control well contains PBMCs only (without HLA antibody or peptides). Peptide wells contained peptide and PBMCs (without HLA antibody). Antibody wells contained PBMCs, peptides, and each antibody.
Blocking assays to confirm MHC restriction
To confirm the MHC restriction of AFP1, IFN-γ ELISPOT was conducted using MHC class I and class II antibodies (Fig. 4b). Using these antibodies, the binding of AFP1 to appropriate HLA molecules was shown to be inhibited and the type of HLA molecule contributing to the response was confirmed. The HLA-DR alleles of the HCC patients whose PBMCs were used were as follows: HLA-DRB1*0405/0901 for patient 19, HLA-DRB1*0405/1401 for patient 28, and HLA-DRB1*1501/1502 for patient 50. The production of IFN-γ was shown to be suppressed by the HLA-DR antibodies in patients 19 and 28. In patient 50, the production of IFN-γ was approximately the same with or without the antibodies. On the other hand, the class I antibodies HLA-DP and HLA-DQ did not suppress the production of IFN-γ in any patient. Thus, the binding of AFP1 to the HLA-DR molecules was confirmed.
Clinical profiles of the patients who showed a positive or negative T cell response against AFP1
To examine the characteristics of the HCC patients who showed an immune response against the AFP-derived MHC class II-restricted epitopes, the clinical profiles of these patients were analyzed. According to the IFN-γ ELISPOT (Fig. 1) and the proliferation assays (Fig. 3), the patients who showed a T cell response against the AFP1 peptide were classified into a positive group (n = 24), while those who did not show such a response were classified into a negative group (n = 16; Supplementary Table 1). Notably, the proportion of females tended to be significantly higher in the positive group than in the negative group (p = 0.07); however, no significant factors could be identified. In other words, the T cell response against AFP1 was induced regardless of tumor size, tumor multiplicity, the presence/absence of vascular invasion, or the etiology of liver disease. Next, the survival rates of the HCC patients between initial treatment and death due to HCC were analyzed to obtain cause-specific survival (CSS). When CSS was analyzed in the HCC patient group during blood collection, in which patients were determined to have clinical stage II, III, or IV HCC according to the TNM classification, the CSS of the positive group (n = 14) that showed a T cell response against the AFP1-derived epitope was significantly better than that of the negative group (n = 8), which did not show a response (p = 0.039; Fig. 5).
Figure 5.
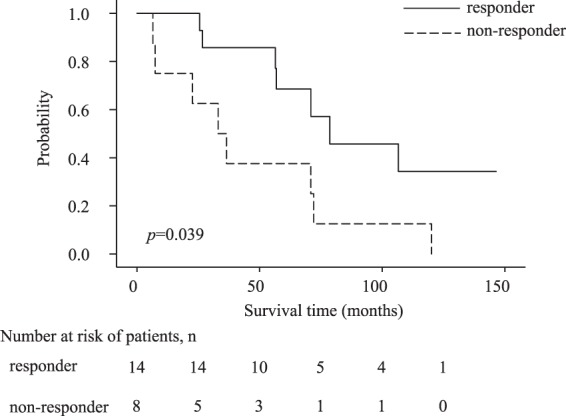
The Kaplan–Meier curve of cause-specific survival for death from liver cancer. Analysis of patients with stages II–IV HCC during blood collection. The relationship between duration (month) from the first day of HCC treatment and the cause-specific survival rate of death from liver cancer is shown using the Kaplan–Meier curve. A responder group that showed a T cell response against AFP1 and a non-responder group that did not show a response on ELISPOT using PBMCs or proliferation assays were compared.
T-cell response against AFP-derived epitopes before and after HCC treatment
To examine changes in the T cell response against AFP-derived MHC class II-restricted epitopes by HCC treatment, peripheral blood samples were collected before and after HCC treatment from the patients who underwent radiofrequency ablation (RFA) (n = 4) and transcatheter arterial chemoembolization (TACE, which was a vascular embolization using anticancer drugs for HCC) (n = 1). PBMCs were collected within 3 to 42 days after treatment (Supplementary Table 2). Following the isolation of PBMCs, IFN-γ ELISPOT was performed on five patients whose peripheral blood was collected before and after the treatment, whereas the PBMCs used were prepared within the post-treatment 3 month period. The peptides used were AFP1, AFP22, and AFP346 (Fig. 6), which were the peptides that showed a high response rate from the screening by ELISPOT (Fig. 1). Representative images of Fig. 6 are shown in Supplementary Fig. 7. For AFP1, four of five patients showed an increased frequency of epitope-specific T cells after treatment. Further examination, according to the presence of specific HLA-DR alleles (Supplementary Table 2), revealed that all patients with the HLA-DRB1*1502 allele, the one patient with the HLA-DRB1*0405 allele, and one of the two patients with the HLA-DRB1*0901 allele had an increased frequency of epitope-specific T cells.
Figure 6.
T cell response to MHC class II-restricted AFP-derived epitopes before and after HCC treatment. The numbers of spots obtained on IFN-γ ELISPOT, each peptide, and the patient number are shown. The control well contained only PBMCs (without peptides).
Discussion
AFP is a cancer-related antigen unique to HCC and has been reported to be a possible therapeutic target for immunotherapy. While clinical trials of a cancer vaccine have been conducted using an AFP-derived class I-restricted epitope as the antigen, the clinical efficacy of AFP remains uncertain10,11. However, from these clinical trials, the activation of CD4+ T cells has been suggested to be required for sufficient levels of clinical efficacy.
We previously succeeded in identifying human telomerase reverse transcriptase-specific HTL epitopes using an epitope prediction algorithm22. Similarly, this website was used to predict AFP-derived MHC class II-restricted HTL epitopes in this study. Peptides were synthesized based on the amino acid sequences of the epitopes predicted to bind to various DRB1 alleles, and IFN-γ ELISPOT was conducted using PBMCs obtained from the HCC patients. The peptides AFP1, AFP22, AFP246, and AFP346 showed a response in >20% of the patients based on IFN-γ ELISPOT. Furthermore, IFN-γ ELISPOT using CD8+ cell-depleted PBMCs showed a high spot count; therefore, it was indicated that the response was due to CD4+ cells. Among the patients who showed a response on ELISPOT (Fig. 1), many did not continue to show a response on ELISPOT after the depletion (Fig. 2). This is likely due to differences in the timing of blood collection and the process used for depletion, which may have impacted the immune response. When the HLA-DR alleles of the patients who showed a positive response on IFN-γ ELISPOT were analyzed, the frequencies of the HLA-DRB1*0405, *0901, and *1502 alleles were high; therefore, it was indicated that these alleles contributed to the response. Among the synthesized peptides, AFP1 showed the highest response and a high number of spots on the IFN-γ ELISPOT performed after depletion of the CD8+ cells. According to previous reports on AFP-derived MHC class II-restricted HTL epitopes, the ex vivo detection of AFP-specific T cells in peripheral blood is difficult23. Thus, T cells induced by peptide stimulation are generally used for experiments. However, PBMCs were stimulated only overnight in this study, indicating that AFP1 is an epitope with the ability to respond directly ex vivo. These results indicate that the immunogenicity of the AFP1 epitope identified in this study was strong. These epitopes with high immunogenicity were able to be isolated in this study because the epitopes were preliminarily designed using a computer software program and the PBMCs were obtained from Japanese patients, in whom HLA is relatively homogeneous.
AFP1 contains several MHC class I-restricted subdominant epitopes, i.e., AFP1 contains subdominant restricted epitopes of HLA-A2412 and HLA-A224. In fact, although low in frequency, 2 of 14 patients showed a positive response on IFN-γ ELISPOT using CD4+ cell-depleted PBMCs.
AFP-specific CD4+ cell reactions are observed at low frequency among healthy individuals25. However, AFP1 showed a positive response at a high frequency compared with other peptides (Supplementary Fig. 1). Therefore, AFP1 is an epitope that may have the potential to induce specific T cells among healthy individuals.
Furthermore, proliferation assays were performed to evaluate the response against peptides not only by IFN-γ production but also by the level of cellular proliferation. All three peptides analyzed showed a response rate >40%; thus, the T cell response against the peptides was also demonstrated by an increase in cellular proliferation. By examining the HLA-DR alleles of the HCC patients, we found that the patients possessed the HLA-DRB1*0405, *0901, or *1502 alleles. AFP1 showed a response at a high frequency even in proliferation assays. Therefore, we considered that AFP1 is an immunodominant epitope able to not only produce IFN-γ but also enhance cellular proliferation.
Next, the binding affinity of AFP1, which showed the highest response rate to T cells of the HCC patients, to HLA-DR molecules was examined using flow cytometry. As shown in Fig. 4a, AFP1 was confirmed to bind to human immortalized cells that were homozygous for the expression of HLA-DRB1*0405 or HLA-DRB1*0901. Furthermore, to examine MHC class II restriction, IFN-γ ELISPOT was performed in which the binding of AFP1 to each HLA molecule was inhibited by the respective HLA antibody. This effect of causing a decrease in IFN-γ production was higher with the addition of the HLA-DR antibody compared with that of other antibodies. These results indicated that AFP1 was able to bind to the HLA-DR molecules. The HLA-DR alleles of the patients used were *0405, *0901, and *1502 and it was indicated that AFP1 is a restricted epitope of these multiple alleles. Therefore, we considered that the strong immunogenicity of AFP1 was due to its ability to bind to multiple HLA-DR molecules.
The relationship between the clinical data of the HCC patients and the T cell response against the AFP-derived epitopes was analyzed. However, no significant differences in terms of serum AFP levels between the group that responded to the epitopes and the group that did not were observed. To initiate an immune response against cancer, antigen-presenting cells (APCs) must take up antigens from the tumors and an immune response must be triggered by the activation of T cells in response to the APCs26. In this study, AFP was the tumor antigen and was required for AFP-derived epitope-specific T cell activation. However, it has been reported that AFP reduces the ability of dendritic cells to stimulate T cell proliferation and inhibits the proliferation of CD8+ and CD4+ cells27. Thus, it is considered that T cell proliferation is suppressed when the serum AFP level is high. According to previous reports, the CD4+ T cell response against AFP is significantly high among HCC patients with low serum AFP levels and patients with early-stage HCC17. However, the CD4+ T cell response against AFP1 did not show a correlation with serum AFP levels or the clinical stage of HCC in this study. On the other hand, in patients with stage II-IV HCC, the CSS of the patient group that showed an immune response against AFP1 was better than that of the group that did not show such a response. These findings suggest that patients who exhibit a T cell response against AFP1 have a better prognosis even at an advanced clinical stage. In this context, our results indicated that AFP1 is an immunodominant epitope that may elicit an immune response in patients with high serum AFP levels or advanced HCC. Furthermore, having an immune response against AFP1, even at an advanced clinical stage, appeared to be beneficial for survival.
Next, changes in the immune response of patients before and after HCC treatment were examined by IFN-γ ELISPOT using the PBMCs obtained from the patients. Indeed, CD4 responses are known to increase after HCC treatment18,22. Our results showed that the number of responses against the AFP-derived epitopes increased in several patients, as reported previously. Therefore, an increase in the T cell response against AFP1 was indicated after HCC treatment. In contrast, there were patients in whom the T cell response decreased. As previously reported, immune responses are known to reach a peak 2–4 weeks after RFA treatment28. The sample from patient 52 was collected within 1 week after RFA treatment; therefore, the timing of sample collection may have influenced our results. Moreover, the involvement of dendritic cell activation associated with tumor cell death and heat shock proteins released after treatment is indicative of changes in the frequency of T cells specific to antigen epitopes after HCC treatment29. With regard to the decreased immune response after HCC treatment, a detailed mechanism has not been elucidated, thus further research on the underlying mechanism is warranted.
To the best of our knowledge, no reports of immunodominant HTL epitopes in AFP1 amino acid sequences have been published. In this study, AFP1 was indicated to be an immunodominant epitope capable of binding to multiple HLA-DR molecules. Therefore, immunotherapy using this epitope may be beneficial to HCC patients as part of a combination therapy with other immune therapies such as ICIs.
Supplementary information
Acknowledgements
The authors thank Maki Kawamura for technical assistance. This study was supported by research grants from the Ministry of Education, Culture, Sports, Science, and Technology of Japan. The authors would like to thank Enago (www.enago.jp) for the English language review.
Author contributions
Toshikatsu Tamai, Eishiro Mizukoshi, Masashi Kumagai, and Kazumi Fushimi performed and analyzed the experiments. Toshikatsu Tamai, Eishiro Mizukoshi, Masashi Kumagai, Takeshi Terashima, Noriho Iida, Masaaki Kitahara, Tetsuro Shimakami, Kazuya Kitamura, Kuniaki Arai, Taro Yamashita, Yoshio Sakai, Tatsuya Yamashita, Masao Honda, and Shuichi Kaneko designed the experiments. Toshikatsu Tamai and Eishiro Mizukoshi wrote the manuscript. Toshikatsu Tamai, Eishiro Mizukoshi, and Shuichi Kaneko edited and prepared the final manuscript.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
is available for this paper at 10.1038/s41598-020-60843-4.
References
- 1.Torre LA, et al. Global cancer statistics, 2012. CA Cancer J. Clin. 2015;65:87–108. doi: 10.3322/caac.21262. [DOI] [PubMed] [Google Scholar]
- 2.Bosch FX, Ribes J, Díaz M, Cléries R. Primary liver cancer: Worldwide incidence and trends. Gastroenterology. 2004;127:S5–S16. doi: 10.1053/j.gastro.2004.09.011. [DOI] [PubMed] [Google Scholar]
- 3.Bruix J, Sherman M. American Association for the Study of Liver D. Management of hepatocellular carcinoma: an update. Hepatology. 2011;53:1020–1022. doi: 10.1002/hep.24199. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Heimbach JK, et al. AASLD guidelines for the treatment of hepatocellular carcinoma. Hepatology. 2018;67:358–380. doi: 10.1002/hep.29086. [DOI] [PubMed] [Google Scholar]
- 5.Llovet JM, et al. SHARP Investigators Study Group. Sorafenib in advanced hepatocellular carcinoma. N. Engl. J. Med. 2008;359:378–390. doi: 10.1056/NEJMoa0708857. [DOI] [PubMed] [Google Scholar]
- 6.Bruix J, et al. Regorafenib for patients with hepatocellular carcinoma who progressed on sorafenib treatment (RESORCE): a randomised, double-blind, placebo-controlled, phase 3 trial. Lancet. 2017;389:56–66. doi: 10.1016/S0140-6736(16)32453-9. [DOI] [PubMed] [Google Scholar]
- 7.Kudo M, et al. Lenvatinib versus sorafenib in first-line treatment of patients with unresectable hepatocellular carcinoma: a randomised phase 3 non-inferiority trial. Lancet. 2018;391:1163–1173. doi: 10.1016/S0140-6736(18)30207-1. [DOI] [PubMed] [Google Scholar]
- 8.Abou-Alfa GK, et al. Cabozantinib in patients with advanced and progressing hepatocellular carcinoma. N. Engl. J. Med. 2018;379:54–63. doi: 10.1056/NEJMoa1717002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Johnson PJ. Role of alpha-fetoprotein in the diagnosis and management of hepatocellular carcinoma. J. Gastroenterol. Hepatol. 1999;14(Suppl):S32–S36. doi: 10.1046/j.1440-1746.1999.01873.x. [DOI] [PubMed] [Google Scholar]
- 10.Butterfield LH, et al. A phase I/II trial testing immunization of hepatocellular carcinoma patients with dendritic cells pulsed with four alpha-fetoprotein peptides. Clin. Cancer Res. 2006;12:2817–2825. doi: 10.1158/1078-0432.CCR-05-2856. [DOI] [PubMed] [Google Scholar]
- 11.Sun Z, et al. Status of and prospects for cancer vaccines against hepatocellular carcinoma in clinical trials. Biosci. Trends. 2016;10:85–91. doi: 10.5582/bst.2015.01128. [DOI] [PubMed] [Google Scholar]
- 12.Mizukoshi E, Nakamoto Y, Tsuji H, Yamashita T, Kaneko S. Identification of alpha-fetoprotein-derived peptides recognized by cytotoxic T lymphocytes in HLA-A24+ patients with hepatocellular carcinoma. Int. J. Cancer. 2006;118:1194–1204. doi: 10.1002/ijc.21468. [DOI] [PubMed] [Google Scholar]
- 13.Nakagawa H, et al. Association between high-avidity T-cell receptors, induced by alpha-fetoprotein-derived peptides, and anti-tumor effects in patients with hepatocellular carcinoma. Gastroenterology. 2017;152:1395–1406 e1310. doi: 10.1053/j.gastro.2017.02.001. [DOI] [PubMed] [Google Scholar]
- 14.Bevan MJ. Helping the CD8(+) T-cell response. Nat. Rev. Immunol. 2004;4:595–602. doi: 10.1038/nri1413. [DOI] [PubMed] [Google Scholar]
- 15.Borst J, Ahrends T, Babala N, Melief CJM, Kastenmuller W. CD4(+) T cell help in cancer immunology and immunotherapy. Nat. Rev. Immunol. 2018;18:635–647. doi: 10.1038/s41577-018-0044-0. [DOI] [PubMed] [Google Scholar]
- 16.Koido S, et al. Treatment with chemotherapy and dendritic cells pulsed with multiple Wilms’ tumor 1 (WT1)-specific MHC class I/II-restricted epitopes for pancreatic cancer. Clin. Cancer Res. 2014;20:4228–4239. doi: 10.1158/1078-0432.CCR-14-0314. [DOI] [PubMed] [Google Scholar]
- 17.Alisa A, et al. Analysis of CD4+ T-Cell responses to a novel alpha-fetoprotein-derived epitope in hepatocellular carcinoma patients. Clin. Cancer Res. 2005;11:6686–6694. doi: 10.1158/1078-0432.CCR-05-0382. [DOI] [PubMed] [Google Scholar]
- 18.Ayaru L, et al. Unmasking of α-fetoprotein-specific CD4+ T cell responses in hepatocellular carcinoma patients undergoing embolization. J. Immunol. 2007;178:1914–1922. doi: 10.4049/jimmunol.178.3.1914. [DOI] [PubMed] [Google Scholar]
- 19.Alisa A, et al. Human CD4+ T cells recognize an epitope within-fetoprotein sequence and develop into TGF–producing CD4+ T cells. J. Immunol. 2008;180:5109–5117. doi: 10.4049/jimmunol.180.7.5109. [DOI] [PubMed] [Google Scholar]
- 20.Kim J, et al. In vitro binding analysis of hepatitis B virus preS-derived putative helper T-cell epitopes to MHC class II molecules using stable HLA-DRB1*0405/DRA*0101 transfected cells. IUBMB Life. 2000;50:379–384. doi: 10.1080/713803746. [DOI] [PubMed] [Google Scholar]
- 21.Ikeda N, et al. Determination of HLA-A, -C, -B, -DRB1 allele and haplotype frequency in Japanese population based on family study. Tissue Antigens. 2015;85:252–259. doi: 10.1111/tan.12536. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kumagai M, et al. Immune response to human telomerase reverse transcriptase-derived helper T cell epitopes in hepatocellular carcinoma patients. Liver Int. 2018;38:1635–1645. doi: 10.1111/liv.13713. [DOI] [PubMed] [Google Scholar]
- 23.Witkowski M, et al. Lack of ex vivo peripheral and intrahepatic alpha-fetoprotein-specific CD4+ responses in hepatocellular carcinoma. Int. J. Cancer. 2011;129:2171–2182. doi: 10.1002/ijc.25866. [DOI] [PubMed] [Google Scholar]
- 24.Butterfield LH, et al. T cell responses to HLA-A*0201-restricted peptides derived from human fetoprotein. J. Immunol. 2001;166:5300–5308. doi: 10.4049/jimmunol.166.8.5300. [DOI] [PubMed] [Google Scholar]
- 25.Evdokimova VN, Liu Y, Potter DM, Butterfield LH. AFP-specific CD4+ helper T-cell responses in healthy donors and HCC patients. J. Immunother. 2007;30:425–437. doi: 10.1097/CJI.0b013e31802fd8e2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Mellman I, Coukos G, Dranoff G. Cancer immunotherapy comes of age. Nature. 2011;480:480–489. doi: 10.1038/nature10673. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Pardee AD, Shi J, Butterfield LH. Tumor-derived alpha-fetoprotein impairs the differentiation and T cell stimulatory activity of human dendritic cells. J. Immunol. 2014;193:5723–5732. doi: 10.4049/jimmunol.1400725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Mizukoshi E, et al. Enhancement of tumor-associated antigen-specific T cell responses by radiofrequency ablation of hepatocellular carcinoma. Hepatology. 2013;57:1448–1457. doi: 10.1002/hep.26153. [DOI] [PubMed] [Google Scholar]
- 29.Zerbini A, et al. Radiofrequency thermal ablation of hepatocellular carcinoma liver nodules can activate and enhance tumor-specific T-cell responses. Cancer Res. 2006;66:1139–1146. doi: 10.1158/0008-5472.CAN-05-2244. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.