Abstract
As part of a combined antiretroviral regimen, doravirine is safe and effective at suppressing viral replication in both treatment-naive and treatment-experienced adults living with human immunodeficiency virus (HIV)-1 who have no history of drug resistance against doravirine. In virologically suppressed individuals switching to a combination of doravirine, lamivudine, and tenofovir disoproxil fumarate, no resistance was found after 48 weeks. In treatment-naive individuals, rare cases (<2%) of emergent drug resistance have been reported, often involving the development of substitutions at position V106. From these few clinical cases, it is inferred that cross-resistance with other non-nucleoside reverse transcriptase inhibitors (NNRTIs) should be limited. In contrast, the use of doravirine as a second NNRTI should be evaluated on a case-by-case basis in the presence of pre-existing resistance. Importantly, doravirine remains active against K103N viruses in vitro, and limited clinical evidence suggests this to be the case in patients as well. Since K103N is by far the most prevalent (<70%) NNRTI substitution found in clinical practice, resistance against doravirine-based antiretroviral therapies is expected to be rare, even for treatment-experienced individuals. This review summarizes chemical, pharmacological, and clinical information about doravirine with an emphasis on drug resistance. The efficacy results from an early phase clinical trial evaluating doravirine in combination with islatravir are also provided.
Keywords: antiretroviral therapy, doravirine, drug resistance, highly active, HIV, reverse transcriptase inhibitors
Introduction
Currently, there are seven drug classes with over 30 different antiviral compounds active against human immunodeficiency virus (HIV)-1, and single-tablet regimens are widely utilized in clinical practice. However, with the increase in antiretroviral therapy (ART) uptake, HIV drug resistance is becoming a serious threat globally.1 Resistance has emerged against all antiretroviral drugs, and therefore choosing the most effective HIV regimen for a given individual remains a challenge, particularly for persons who are treatment-experienced. Treatment determination is dependent on several factors, including virological efficacy, expected patient adherence, and genotyping resistance background, as well as other pharmacological aspects, medication side effects, and drug–drug interactions. The long-term goal of treatment is generally to provide the maximal duration of viral suppression for patients using first-line therapy, delay the development of drug resistance, and prevent HIV transmission. This concept of choosing the most likely durable initial regimen may be challenged soon by the advent of test-and-treat strategies and efficacious drug-reduction regimens, including two-drug combinations. In this context, it is still important to discover new drugs and drug combinations with better pharmacology profiles, fewer side effects, fewer comorbidity concerns, and lower pill burden.
Doravirine (DOR, MK-1439) is a novel HIV-1 non-nucleoside reverse transcriptase inhibitor (NNRTI). Based on supporting data from the pivotal phase III clinical trials DRIVE-AHEAD and DRIVE-FORWARD, in August 2018, DOR was first approved by the US Food and Drug Administration (FDA) in two formulations – as a complete once-daily dose regimen in combination with two NRTIs, lamivudine and tenofovir disoproxil fumarate (DOR/3TC/TDF, Delstrigo [Merck & Co. Inc., Kenilworth, NJ]), and as a single tablet of 100 mg DOR (Pifeltro [Merck & Co. Inc.]) to be used in combination with other active antiretroviral drugs – both for the treatment of ART-naive adults living with HIV.2 Later, in November 2018, both formulations were approved in Europe for the treatment of adults living with HIV-1 without past or present evidence of NNRTI resistance.3,4
The pharmacodynamic and pharmacokinetic characteristics of DOR have been extensively discussed in previous reviews.5–7 Thus, this review summarizes important pharmacological aspects and clinical profiles of DOR while focusing on the HIV drug-resistance patterns against DOR identified from in vitro and clinical studies. The authors gathered information from MEDLINE/PubMed publications and the latest international conferences by searching for the following keywords: ‘MK-1439’, ‘pifeltro’, ‘delstrigo’, ‘doravirine’, and ‘resistance + [any of the previous]’. This review aims at compiling current information on resistance to the benefit of healthcare practitioners who may consider prescribing DOR despite the presence of NNRTI resistance mutations.
Chemistry of the compound
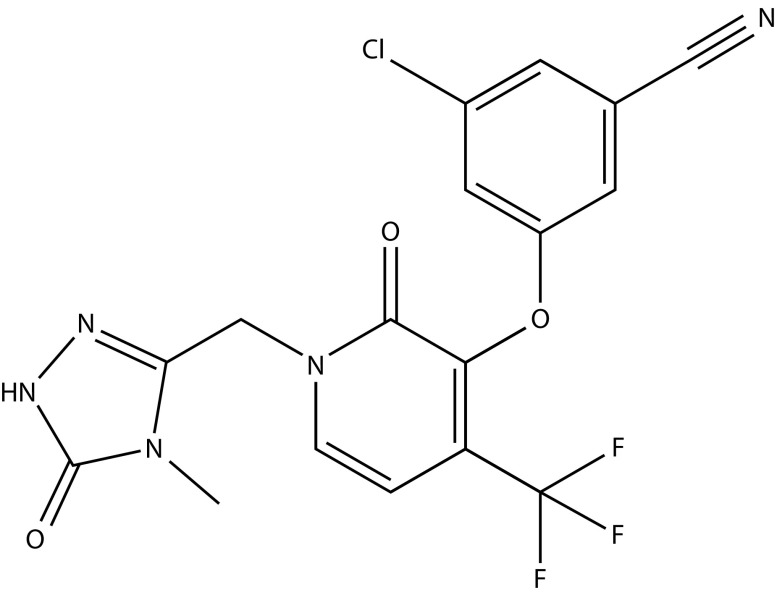
DOR is an HIV-1 pyridone non-NNRTI. The chemical name for DOR is 3-chloro-5-[[1-[(4,5-dihydro-4-methyl-5-oxo-1H-1,2,4-triazol-3-yl)methyl]-1,2-dihydro-2-oxo-4-(trifluoromethyl)-3-pyridinyl]oxy]benzonitrile (Figure 1). The empirical formula is C17H11ClF3N5O3, with a molecular weight of 425.75 g/mol. The water solubility of DOR is 2.73 mg/L (pH 7).2,3 Both DOR and DOR/3TC/TDF are for oral administration once daily with or without food. DOR is a 100-mg film-coated tablet. DOR/3TC/TDF is a fixed-dose combination, film-coated tablet containing three active antiretroviral drugs – DOR (100 mg), 3TC (300 mg), and TDF (300 mg).
Figure 1.
Chemical structure of doravirine.
3-chloro-5-[[1-[(4,5-dihydro-4-methyl-5-oxo-1H-1,2,4-triazol-3- yl)methyl]-1,2-dihydro-2-oxo-4-(trifluoromethyl)-3-pyridinyl]oxy]benzonitrile
Pharmacodynamics, mechanism of action, and antiviral activity
As an allosteric inhibitor, similar to other NNRTIs, DOR binds to a hydrophobic pocket located in the p66 subunit of the p66/p51 heterodimer of HIV-1 reverse transcriptase (RT), about 10 Å away from the RT polymerase active site, causing conformational changes that inhibit HIV-1 deoxyribonucleic acid (DNA) synthesis.8,9 Similar levels of DOR susceptibility in vitro were also observed for 10 HIV-1 subtypes, including A, A1, AE, AG, B, BF, C, D, G, and H, with EC50 values from 0.6 nM to 10 nM.2–4,10 For all the subtypes tested, subtype H seems to be hypersensitive to DOR as well as other NNRTIs.2,10 Meanwhile, DOR activity is less effective against HIV-2 in peripheral blood mononuclear cells (PBMCs) with an EC50 of 1.25 μM (over 100-fold higher than the concentration that inhibits HIV-1).3,11
DOR cytotoxicity was investigated in different cell types, including resting or activated PBMCs, CD4+ T cells, monocytes, macrophages as well as MT4, SupT1, and HL60 cell lines. DOR exhibited no cytotoxicity effects at concentrations below 100 μM.10 DOR’s potential off-target activity was tested in biochemical assays against more than 110 cellular enzymes and receptors. DOR did not significantly inhibit human DNA polymerases α, β, and γ.10 In a ligand-binding test, DOR had a moderate affinity to 5-hydroxytryptamine receptor 2b with an IC50 of 2.5 μM, but no agonistic or antagonistic activity was observed in a cell-based assay, which suggested that binding of DOR to serotonin receptor 2b does not affect the normal functioning of those receptors.3,4,10
No antagonistic effect was observed in the CEM-SS cell line when DOR was combined with any of the 18 FDA-approved anti-HIV-1 drugs, including NRTIs such as lamivudine, abacavir, zidovudine, stavudine, zalcitabine, didanosine, emtricitabine, tenofovir disoproxil fumarate; NNRTIs such as delaviridine, efavirenz (EFV), etravirine (ETV), nevirapine (NVP), and rilpivirine (RPV); protease inhibitors (PIs) such as darunavir and indinavir; and entry/fusion inhibitors such as maraviroc and enfuvirtide. Only a slightly synergistic effect was observed with the integrase strand-transfer inhibitor raltegravir (RAL).2
DOR EC50 for hepatitis B virus (HBV) is >10 μM (the highest concentration tested) in HepG2 cell line, which means that DOR is not active against HBV at clinically relevant concentrations. As a result, it is unlikely to pose the risk of HBV resistance in HIV patients co-infected with HBV.3
Pharmacokinetics
Bioavailability, absorption, and distribution
The PK characteristics of DOR have been investigated in both healthy volunteers and people living with HIV-1. Food effects on DOR were studied in healthy volunteers in two trials: P029 and P037. In these studies, either the fixed-dose combination of DOR/3TC/TDF or DOR 100 mg was used in both fed and fasted states. A population PK study for DOR using pooled data from clinical trials phase I, IIb, and III, including 341 healthy and 959 adults living with HIV-1, demonstrated that PK values were comparable between fed and fasting groups. At a DOR dose of 100 mg daily, the steady-state AUC0-24h was 37.8 μM·h (27%), Cmax was 2,260 nM (18.4%), and C24h was 930 nM (41.6%).3,12 The absolute bioavailability of DOR was about 64%.2,3 There were no significant impacts in PK values, including AUC0-∝ and Cmax upon oral administration with or without food.2,3,13 Accordingly, in the pivotal phase III clinical trials termed P018 and P021, DOR was used in a fixed-dose combination (DOR/ABC/3TC) or as a single tablet regardless of food intake.2,4 DOR was also investigated in dose-escalation studies in healthy males receiving single doses of 6–1,200 mg or 30–750 mg for 10 days (P001, EudraCT 2010-024245-70, and P006; EudraCT 2011-004260-30).3,14 DOR orally given in the fasted state was rapidly absorbed with a median Tmax of 1–5 hours post-administration to achieve the maximum plasma concentration. Time to achieve steady-state was day 7 for once-daily administration. A single DOR 100 mg dose displayed a long terminal half-life T1/2 of 15 hours. All doses tested yielded C24h values > 19 nM.14 The volume distribution of a single-dose intravenous of DOR 100 μg is 60 L with a low clearance of 3.73 L/h.15 In vitro, DOR has a protein unbound fraction of approximately 25% and has good passive permeability.3,15 No data were reported for plasma protein binding in hepatic and renal impairment patients.3 No data are available on crushing DOR or DOR/ABC/3TC tablets, so this method of administration is not recommended.
Metabolism and elimination
The major mechanism of DOR elimination is metabolism. DOR is excreted primarily (90%) in feces, mainly as unchanged drug (84%), and in urine (10%: 2.2% as unchanged drug and 7.2% as metabolites). The primary component is an oxidative metabolite termed M9 that represents 6.7% and 2.7% of total doses of DOR detected in urine and feces, respectively.15 The metabolism of DOR by recombinant human CYPs in vitro demonstrated that DOR was mainly catalyzed by CYP3A4/5 enzymes with ~20-fold-higher catalytic efficiency for CYP3A4 versus CYP3A5. It is unlikely that other additional oxidation pathways contribute to the oxidative metabolism of DOR.15
Pharmacokinetic interaction
In agreement with its elimination profile, concentrations of DOR are significantly reduced by the concomitant use of rifampicin, a potent CYP3A inducer (Table 1). Multiple doses of rifampin (600 mg QD) decreased plasma DOR concentrations up to 88% for AUC, 97% for C24, and 57% for Cmax, which may pose a risk of development of resistance. Strong CYP3A inducers such as rifampicin are therefore contraindicated when given with DOR-containing regimens. With rifabutin (300 mg) QD, a moderate CYP3A inducer, the reduction in DOR concentrations is less pronounced, but dose adjustment to 100 mg BID is recommended. DOR exposure, when administered alone at 100 mg QD, is similar to that of 100 mg BID co-administered with rifabutin 300 mg QD. Similarly, the use of DOR BID is recommended when co-administration with other moderate CYP3A inducers such as dabrafenib, lesinurad, bosentan, thioridazine, nafcillin, modafinil, and telotristat ethyl.2,3 Another situation of interest concerns individuals previously treated with another NNRTI called EFV, also a CYP3A inducer, who wish to switch to a DOR-based regimen. EFV coadministration reduces DOR AUC0-24 by 62% and C24 by 85%. Thus, patients switching from EFV to DOR should be carefully monitored to ensure a safe transition. Reciprocally, no dose adjustment is recommended when DOR is co-administered with CYP3A inhibitors, including ketoconazole and ritonavir, which both increase DOR plasma concentrations. There were no clinically significant changes in DOR PK parameters with midazolam, dolutegravir, lamivudine, tenofovir DF, elbasvir, grazoprevir, ledipasvir, sofosbuvir, GS-331007, ethinyl estradiol, levonorgestrel, atorvastatin, metformin, or methadone (R or S forms).2,3
Table 1.
DOR drug–drug interactions and recommended dose adjustment.
| Drug | Effect on DOR | Mechanism | Recommendation |
|---|---|---|---|
| Rifampin | Decreased plasma concentrations | CYP3A inducer | Contraindication |
| Rifabutin | Dose adjustment | ||
| Dabrafenib | |||
| Lesinurad | |||
| Bosentan | |||
| Thioridazine | |||
| Nafcillin | |||
| Modafinil | |||
| Telotristat ethyl | |||
| Efavirenz | Monitor when switching from EFV to DOR | ||
| Ketoconazole | Increased plasma concentrations | CYP3A inhibitor | No adjustment required |
| Ritonavir | |||
| Midazolam | No significant effect | ||
| DTG | |||
| 3TC | |||
| TDF | |||
| Elbasvir | |||
| Grazoprevir | |||
| Ledipasvir | |||
| Sofosbuvir | |||
| GS-331007 | |||
| Ethinyl estradiol | |||
| Levonorgestrel | |||
| Atorvastatin | |||
| Metformin | |||
| Methadone |
This list is non-exhaustive. Dose adjustment is indicated for information only. Consult package insert and your healthcare specialist before changing the dosage of any drug.
3TC: lamivudine; DOR: doravirine; DTG: dolutegravir; EFV: efavirenz; TDF: tenofovir disproxil fumarate.
Special populations and lactation
Severe renal impairment was not associated with significant changes in DOR pharmacokinetics. Therefore, no dose adjustment of DOR is required in patients with mild, moderate, or severe renal impairment. However, for patients with estimated creatinine clearance <50 mL/min or patients with end-stage renal disease who need regular dialysis, the fixed-dose DOR/3TC/TDF is not recommended since dosing for both 3TC and TDF require adjustment. Although this approach is not part of current official recommendations, in some clinical settings, 3TC dose adjustments are made only for patients with creatinine clearance lower than 30 mL/min.16 At a single dose, DOR pharmacokinetics was not found to be different in patients with moderate hepatic impairment, and no dose adjustment is required for this population. DOR has not been studied in subjects with severe hepatic impairment. Other factors such as gender, race, body weight, and age (≥18 years of age) are not expected to have a clinically relevant effect on DOR pharmacokinetics.
The safety and efficacy of DOR for pediatric use have not been studied. A phase I/II clinical trial is ongoing to evaluate PK, safety, and tolerability of DOR and DOR/3TC/TDF in children and adolescents living with HIV-1 (NCT03332095). DOR has not been studied in pregnant women or elderly patients. In rats, no adverse effects on maternal health or embryo/fetal development were observed for DOR doses up to 450 mg/kg/day. Following oral administration to pregnant/lactating rats, DOR was secreted into the milk at day 14 postpartum. No effects on reproductive performance, fertility, or embryo/fetal viability were observed in rats on daily oral doses of DOR. No maternal toxicity at the maximum feasible dose of 450 mg/kg/day was observed in non-pregnant and pregnant rabbits.
Pivotal phase III studies
Table 2 summarizes the three phase III studies evaluating the safety and efficacy of DOR for the treatment of adults living with HIV.
Table 2.
Overview of pivotal phase III clinical trials conducted to evaluate the safety and efficacy of DOR.
| DRIVE-FORWARDP018-NCT02275780 | DRIVE-AHEADP021-NCT02403674 | DIRVE-SHIFTP024-NCT02397096 | ||
|---|---|---|---|---|
| Trial design | Phase | Phase III | Phase III | Phase III |
| Design | Randomized, double-blind, non-inferiority | Randomized, double-blind, non-inferiority | Randomized, non-inferiority, double-blind, switch study | |
| Participants | 766 treatment-naïve adults with no documented RAMs | 734 treatment-naïve adults with no documented RAMs | 673 adults with viral suppression >6 month with no documented RAMs | |
| Duration | 96 weeks | 96 weeks double-blind period + 96 weeks open-label period | 48 weeks | |
| Arms | DOR arm | DOR (100 mg) + TDF/FTC or ABC/3TC with matching placebo (n=383) | Single tablet regimen DOR/3TC/TDF (100/300/300 mg) with matching placebo (n=364) | Single tablet regimen DOR/3TC/TDF (100/300/300 mg): immediate switch (n=450), or 24-week delay switch (n=223) |
| Comparator | DRV + RTV (800/100 mg) + TDF/FTC or ABC/3TC with matching placebo (n=383) | Single tablet regimen EFV/TDF/FTC (600/200/300 mg) with matching placebo (n=364) | b/DRV, b/ATV, b/LPV, b/EVG, EFV, NVP, or RPV + 2 NRTIs | |
| Results: participants (%) with viral suppression | Week 48 | 84 versus 80% (MD: 2.1%, 95% CI: −2.725 to 6.924) | 84 versus 80.8% (MD: 3.5%, 95% CI: −2.0 to 9.0) | 90.8 versus 94.6% (MD: −3.8, 95% CI: −7.9 to 0.3) |
| Week 96 | 72 versus 66% (MD: 7.1%, 95% CI: 0.5 to 13.7) | 78 versus 74% (MD: 3.9%, 95% CI: −2.4 to 10) | N/A | |
| Results: Mean CD4+ cell count from baseline (cell/μL) | Week 48 | +193 versus +186 (MD 7.1, 95% CI: −20.8 to 35.0) | +198 cells versus +188 cells (MD 10.1, 95% CI: −16.1 to 36.3) | +14 in early switch group |
| Results: conclusion | Superiority | Non-inferiority | Non-inferiority |
3TC, lamivudine; 95% CI, 95% confidence interval; ABC, abacavir; ATV, atazanavir; b/, boosted; DOR, doravirine; DRV, darunavir; EFV, efavirenz; EVG, elvitegravir; FTC, emtricitabine; LPV, lopinavir; MD, mean difference; NA, non-available; NVP, nevirapine; RAMs, resistance-associated mutations; RPV, rilpivirine; TDF, tenofovir disoproxil fumarate.
Trial DRIVE-SHIFT (P024-NCT02397096)
The DRIVE-SHIFT is a phase III, multicenter, open-label, randomized, non-inferiority clinical trial designed to study safety and efficacy of switching to DOR plus lamivudine and tenofovir disoproxil fumarate in HIV-1-infected adults. The study involved >600 participants from 122 hospitals and clinics in Europe, North America, Latin America, and Asia. Inclusion criteria were no history of virological failure, virological suppression for >6 months on a stable regimen consisting of either ritonavir- or cobicistat-boosted PI (atazanavir, darunavir or lopinavir), cobicistat-boosted EVG, or an NNRTI (EFV, NVP or RPV), each in combination with 2 NRTIs, and creatinine clearance >50 mL/min. Individuals with resistance to DOR, 3TC, or TDF were not enrolled. Participants were randomly assigned (2:1) to switch to a fixed-dose DOR/3TC/TDF, immediately (n=447) on day one or to remain on current therapy and then switch at week 24 (n=223). At week 24, 93.7% (419/447) in the ISG and 94.6% (211/223) in the DSG achieved HIV-1 ribonucleic acid (RNA) <50 copies/mL (treatment difference: −0.9, 95% CI: −4.7, 3.0). At week 48, a switch to DOR/3TC/TDF (90.8% viral suppression) was statistically non-inferior to maintaining current ART for 24 weeks (treatment difference: −3.8, 95% CI: −7.9, 0.3).17 Of note, from 670 participants who entered the trial, 114 (17%) had received an EFV-based regimen.18 In regard to safety, switching to a fixed-dose DOR/3TC/TDF once daily regimen was generally well tolerated, although, in this open-label setting, more participants in the ISG reported adverse events than those in the DSG (68.9 versus 52.5%, respectively). Switching to DOR/3TC/TDF from ritonavir-boosted PIs had positive effects on fasting low-density lipoprotein cholesterol (LDL-C) and non-high-density lipoprotein cholesterol (HDL-C) levels. No resistance mutation emerged against DOR/3TC/TDF.
Trial DRIVE-FORWARD (P018-NCT02275780)
DRIVE-FORWARD is a phase III, double-blind, randomized, and controlled non-inferiority study to compare the safety and efficacy of DOR versus ritonavir-boosted darunavir (r/DRV) both in combination with either TDF/FTC or ABC/3TC. Participants (n=766) were ART-naive adults with plasma HIV-1 RNA >1000 copies/mL, with no documented or known resistance to any of the study drugs, from 15 countries. They were randomized 1:1 (383 in each treatment group) to received DOR or r/DRV plus matching placebo for up to 96 weeks. Population characteristics including race, sex, ethnic origin, viral load (VL), non-B subtype, median CD4 cell count, and HBV/HCV co-infection were similar in both arms. At week 48, 84% (321/383) versus 80% (306/383) participants achieved plasma VL < 50 copies/mL (treatment difference: 2.1, 95% CI: −2.725, 6.924), establishing non-inferiority. At week 96, the point estimate favored the DOR treatment arm (72 versus 64% suppression with DOR versus r/DRV, respectively; treatment difference 7.6%, 95% CI: 1.0, 14.2).19 Diarrhea (14%) and headache (11%) were the most common adverse events reported for individuals using DOR. However, they were more frequent in the r/DRV arm (22.5% diarrhea and 14.1% headache). Both treatment groups reported comparable low rates of serious or drug-related adverse events. Patients in the DOR arm had a slight decrease in total cholesterol and triglyceride concentrations compared to the r/DRV group. Two cases of genotypic resistance were captured in the DOR group compared with a single case of phenotypic resistance in the r/DRV group (genotyping was unsuccessful). Resistance patterns are reported in Table 3.
Table 3.
NNRTI resistance mutations found in individuals from the DRIVE-FORWARD and DRIVE-AHEAD clinical trials.
| RT mutations | Trial |
|---|---|
| V106I, H221Y, F227C | DRIVE-FORWARD |
| V106A, P225Y/H | DRIVE-FORWARD |
| Y188L | DRIVE-AHEAD |
| Y318Y/F | DRIVE-AHEAD |
| V106I, F227C | DRIVE-AHEAD |
| V106V/I, H221H/Y, F227C | DRIVE-AHEAD |
| F227C | DRIVE-AHEAD |
| V106A, P225H, Y318Y/F | DRIVE-AHEAD |
| V106M/T, F227C/R | DRIVE-AHEAD |
The most frequently substituted position (V106) is indicated in bold.
NNRTI, non-nucleoside reverse transcriptase inhibitor.
Trial DRIVE-AHEAD (P021-NCT02403674)
DRIVE-AHEAD is a phase III, randomized, double-blind, non-inferiority trial to compare safety and efficacy of once-daily, fixed-dose DOR/3TC/TDF (100/300/300 mg) with EFV/FTC/TDF (600/200/300 mg) for 96 weeks, with a subsequent optional 96-week open-label stage for a total 192 weeks of follow-up.20 The trial is conducted at 126 clinical centers worldwide with 734 participants, of which 364 are given matching placebos. Eligible participants are ART-naive adults (≥18 years), with creatinine clearance >50 mL/min, plasma HIV-1 RNA ≥1000 copies/mL, and no documented resistance to any of the study drugs. DOR/3TC/TDF or placebo is taken QD orally without regard to food. Demographics and baseline factors were similar between the two groups, but more participants in the DOR arm are infected with a non-B HIV subtype (36 versus 30% in the EFV arm).20 For the primary endpoint, at week 48, DOR/3TC/TDF was non-inferior to EFV/FTC/TDF with 84.3% (307/364) in the DOR group achieving viral suppression <40 copies/mL versus 80.8% (294/364) in the EFV group (treatment difference: 3.5%; 95% CI: −2.0, 9.0). Results at week 96 were similar, with 78% suppression with DOR compared to 74% with EFV (3.9% of treatment difference, 95% CI: −2.4, 10).21
In regard to safety, compared to the EFV/FTC/TDF group, a smaller proportion of participants in the DOR/3TC/TDF arm reported adverse effects (82.7 versus 90.7%, treatment difference: 8%, 95% CI: −13.0, −3.1). Drug-related adverse events were also less frequent in the DOR/3TC/TDF group: 31.0 versus 62.9% in EFV/FTC/TDF group (treatment difference: −31.9%, 95% CI: −38.6, −24.8). Discontinuation due to adverse events was also lower with DOR. DOR also had a lowering effect on fasting lipid values versus EFV. There were 7 and 12 cases of emergent resistance mutations in the DOR and EFV arms, respectively, which are described in Table 3.
HIV drug resistance against DOR
In vitro resistance and cross resistance
DOR efficacy was initially tested against mutants that are known to bear resistance against previous NNRTIs. Resistance to previous NNRTIs is heterogeneous, with resistance against first-generation NNRTIs (NVP and EFV) being often associated with the highly fit K103N substitution, whereas resistance against second-generation NNRTIs (ETR and RPV) is often linked to substitutions at position E138 together with the M184I NRTI-resistance substitution. Both second-generation inhibitors were selected for their retained antiretroviral activity against a single K103N substitution. Other substitutions such as Y181C (NVP) or G190A (NVP, EFV) display various degrees of cross resistance against different NNRTIs, regardless of their novelty. Finally, substitutions that disrupt the NNRTI-binding pocket, such as Y188L and M230L, confer pan-resistance against all NNRTIs. DOR was tested against various first- or second-generation NNRTI-resistant mutant viruses (Table 4). Most importantly, DOR was active against the most prevalent NNRTI-resistance mutations K103N, Y181C, and G190A.8,10,22 DOR exhibited <3-fold change in EC50 against K103N, Y181C, or double mutant K103N/Y181C strains.10,12 These cell-based observations are in agreement with biochemical assays that showed that DOR exhibits potent inhibitory activity against wild-type, K103N, and Y181C recombinant reverse transcriptase enzymes with half-inhibitory concentrations (IC50s) of 12.2, 9.7, and 9.7 nM, respectively.2,10
Table 4.
Effects on drug susceptibility of selected substitutions in vitro and in vivo against DOR, EFV, and RPV.
| Mutations selected with DOR | DOR | EFV | RPV | ||||||
|---|---|---|---|---|---|---|---|---|---|
| In vitro | In vivo | FC | In vitro | In vivo | FC | In vitro | In vivo | FC | |
| K101E | − | X | 4.5 | X | X | 11 | X | X | 10 |
| V106A | X | X | >10 | − | X | <10 | X* | − | <6 |
| V106I | X | X* | 1.4 | X | X | 1.1 | X | X | 1.2 |
| V106M | X | X | 3.3 | X | − | 106 | − | − | SUS |
| V106A/F227L | X | X | >100 | − | − | 22 | − | − | <10 |
| V106A/F227I | X | − | >100 | − | − | − | − | − | − |
| V106A/F227C | X | − | − | − | − | − | − | − | − |
| V106A/F227V | X | − | − | − | − | − | − | − | − |
| V106A/L234I | X | X | >100 | − | − | SUS | |||
| V106A/L234I/F227L | X | − | >100 | − | − | − | − | − | SUS |
| V106A/L234I/V108I | X | − | >100 | − | − | − | − | − | − |
| V106I/L234I/V108I | X | − | − | − | − | − | − | − | − |
| V90G/V106I/F227C | X | − | − | − | − | − | − | − | |
| V106M/F227L | X | X | − | − | − | − | − | − | − |
| V106M/F227C | X | − | − | − | − | − | − | − | − |
| V106M/F227V | X | − | − | − | − | − | − | − | − |
| V106M/L234I | − | X | − | − | − | − | − | − | − |
| V106M/V108I/F227C(R) | − | X | − | − | − | − | − | − | − |
| V108I | X | X | 4 | X | X | 1.6 | X* | − | 1.2 |
| V108I/L234I | X | − | − | − | − | − | − | − | |
| E138G | − | X | 1 | X | − | 2 | X | − | <10 |
| Y188C | − | X | SUS | X | − | 2.8 | − | − | SUS |
| Y188H | − | X | 2.8 | − | − | 3.9 | − | − | SUS |
| Y188L | X | X | >100 | X | X | >50 | − | − | <10 |
| G190E | X | >20 | − | X | >50 | X | − | >10 | |
| H221Y | X | X | <10 | − | − | 5 | − | X | − |
| F227C | X | X* | >10 | X* | X* | 5 | X* | X* | 4 |
| M230L | − | X | >20 | − | X | <10 | X | X | <10 |
| L234I | X | X | <10 | − | X | − | − | − | NA |
| P236L | − | X | >2 | − | − | SUS | − | − | SUS |
| Y318F | X | X | − | − | X | SUS | − | − | SUS |
| K103N/P225H | − | X | >10 | − | − | >50 | − | − | SUS |
| K103N/Y181C | − | X | >5 | − | − | >30 | − | − | 5.7 |
| P225S/A335T | − | X | − | − | − | − | − | − | − |
| A98G/P225L/F227C | − | X | − | − | − | − | − | − | − |
| A98G/V106A/P225L/Y318F | − | X | − | − | − | − | − | − | − |
| G190S/F227C(L/V)/M230I(L) | − | X | − | − | − | − | − | − | − |
| V90I/V106I/H221Y/F227C | − | X | − | − | − | − | − | − | − |
| A98G/V106I/H221Y/P225L/F227C | − | X | − | − | − | − | − | − | − |
Often occurs in mixture with other mutations; (−), no data available;
DOR: doravirine; EFV: efavirenz; FC: fold change; RPV: rilpivirine; SUS: susceptible/
DOR susceptibility was also evaluated against 102 clinical HIV-1 isolates bearing various mutations.3,4 Across those clinical isolates (no subtype information was provided), DOR displayed a good antiviral activity with fold changes in EC50<9 against most single mutant viruses, including A98G, E138A/G/K/Q, G190A, K101E/P, K103N/S, L100I, P236L, V106M, V108I, V197D, V90I, Y181C/V, and Y188H/C. Other single substitutions including G190E/S, V106A, Y188L, and M230L reduced DOR susceptibility >10-fold. The G190S, Y188L, and M230L substitutions confer >95-fold resistance.23–26 Among double and triple mutant viruses, 13 of 102 conferred >10-FC in EC50 against DOR, whereas this was true for 46/102, 11/102, and 15/102 for EFV, ETR and RPV, respectively.3,4 The highest levels of reduction in DOR susceptibility were associated with V106A or Y188L or each of these two mutations in combination with at least one secondary mutation, such as V106A/G190A/F227L, Y188L/K103N, Y188L/V106I, and E138K/Y181C/M230L. Other substitutions such as V106M, V108I, V179D, Y188H, or P236L conferred less than 10-FC against DOR.2,10,22,27
In vitro selection of resistance-associated mutations
Although phenotypic testing using short-term infectivity assays is efficacious for rapid screening for high-level resistance, prolonged drug exposure to select for resistance is a more stringent way of testing for susceptibility. In this regard, results of in vitro selection for resistance against DOR have been reported for subtypes A, B, and C of HIV-1.28 In almost all cases, DOR selected first for V106A/M substitutions. Of note, the subtype-specificity of the substituted amino acid at position V106 relies on codon usage (often GTA in subtype B viruses versus GTG for subtype C). V106A is a non-polymorphic substitution selected by NVP, while V106M is commonly selected by both NVP and EFV from subtype C viruses. Mutations that emerged secondary to V106A/M included F227L/C/V or L234I. The double mutant viruses V106A/L234I (subtype B) and V108I/L234I (subtype A) eventually acquired a third mutation to give triple mutant viruses V106A/L234I/F227L (subtype B) and V108I/L234I/V106A(I) that both conferred over 150-fold decreases in DOR susceptibility.28 Other in vitro studies confirmed that viruses bearing both V106A and F227L substitutions reduced DOR susceptibility >500-fold.2,27,28 Of note, the viral breakthroughs described earlier were observed when selections were made with 3x EC95 of DOR; however, at 10x EC95, only F227C was selected.28 In theory, the pyridone core of DOR is in close proximity of but has limited contact with the F227 residue. It is thus possible that the F227L substitution per se may confer only low-level resistance against DOR.27,28
Supporting phenotypic testing, when DOR selections were initiated with K103N, Y181C, or K103N/Y181C viruses, they did not lead to the development of further RT mutations, an observation that may justify the use of DOR against these substitutions.
Clinical resistance against DOR
No resistance was found following a treatment switch to DOR-based regimens in the DRIVE-SHIFT trial. Two ART-naive individuals (n=2/382, <1%) from the DRIVE-FORWARD clinical trial experienced treatment failure with emergent NNRTI resistance mutations. In DRIVE-AHEAD, seven cases of genotypic resistance (n=7/364, <2%) were diagnosed in the DOR arm. Substitutions in these two trials are listed in Table 3. The most frequent substitutions were found at position V106. Important secondary substitutions were found at positions H221, P225, and/or F227. Notably, resistance against DOR was most commonly diagnosed with several concomitant substitutions, whereas unique K103N substitutions were found in half of the participants who had mutations in the EFV arm of the DRIVE-AHEAD trial, which may suggest that the dynamics of DOR resistance mutations in vivo seem to be more diverse and complicated than expected.29–31 F227C or Y318F alone may be linked to clinical resistance against DOR. Another substitution, K101E, was detected in one patient who had a virological failure in a phase II DOR dose-ranging study, although the virus bearing this mutation did not confer significant resistance against DOR in vitro.10,29,32 Importantly, preliminary clinical data obtained from a small number of participants (n=9) indicated that DOR could be used efficaciously to suppress viral replication for 96 weeks in individuals infected with K103N or G190A viruses.33
Doravirine plus islatravir
In addition to the clinical trials that led to its approval for clinical use, DOR is currently being evaluated in a two-drug combination with islatravir (ISL). ISL is a first-in-class nucleoside reverse transcriptase translocation inhibitor (NRTTI) that has been reviewed elsewhere.34 The DOR/ISL combination was evaluated in a phase IIB clinical trial that enrolled 121 treatment-naive adults living with HIV-1.35 Since DOR and ISL belong to two different drug classes and given that F227C increases the in vitro susceptibility of HIV-1 to ISL, this combination is expected to provide a combined high genetic barrier against the development of resistance.36 For the first 24 weeks, participants were treated once daily with 0.25 mg (n=29), 0.75 mg (n=30), or 2.25 mg (n=31) ISL plus DOR and 3TC (100 and 300 mg, respectively).35 The comparator was DOR/3TC/TDF (n=31). At week 24, 92% of participants using DOR/ISL/3TC achieved viral suppression (FDA snapshot analysis) compared to 87.1% with DOR/3TC/TDF. Between weeks 24 and 48, patients in the ISL arm stopped using 3TC while continuing their initial ISL dosing plus DOR. At week 48, 85.5% of participants using DOR/ISL were virologically suppressed versus 83.9% with DOR/3TC/TDF. No case of emergent resistance was reported in this study.
Conclusions
DOR is a safe and well-tolerated new NNRTI that has so far been used in treatment switch without the development of resistance. DOR yields an advantageous safety profile, particularly on lipids. In treatment-experienced individuals, DOR may be most beneficial to patients who wish to reduce pill burden or toxicity of other regimens, such as treatment anchored with a protease inhibitor. Its use in patients who previously had witnessed the development of mutations associated with NNRTI-resistance has not been fully validated, although initial clinical reports are encouraging. Common NNRTI-resistance through K103N seems innocuous to the efficacy of DOR-based regimens, whereas, in treatment-naive individuals, resistance to DOR can occur most commonly through the development of substitutions at position V106. Other rare substitutions, for example, at positions F227 and Y318, need to be monitored in individuals who use DOR. In treatment-naive individuals, DOR/ABC/3TC has suffered from indirect comparisons with recent integrase inhibitors.37 Early clinical results support the development of a DOR/ISL two-drug combination for both switch and treatment initiation.
Acknowledgements
None.
Footnotes
Contributions: All authors contributed equally to the preparation of this review. All named authors meet the International Committee of Medical Journal Editors (ICMJE) criteria for authorship for this article, take responsibility for the integrity of the work as a whole, and have given their approval for this version to be published.
Disclosure and potential conflicts of interest: The authors have no other relevant affiliation or financial involvement with any organization or entity with financial interests or financial conflicts with the subject discussed in the manuscript. The International Committee of Medical Journal Editors (ICMJE) Potential Conflicts of Interests form for the authors is available for download at: https://www.drugsincontext.com/wp-content/uploads/2020/02/dic.2019-11-4-COI.pdf
Funding declaration: There was no funding associated with the preparation of this article.
Correct attribution: Copyright © 2020 Pham HT, Xiao MA, Principe MAV, Wong A, Mesplède T. Published by Drugs in Context under Creative Commons License Deed CC BY NC ND 4.0.
Provenance: invited; externally peer reviewed.
Drugs in Context is published by BioExcel Publishing Ltd. Registered office: Plaza Building, Lee High Road, London, England, SE13 5PT.
BioExcel Publishing Limited is registered in England Number 10038393. VAT GB 252 7720 07.
For all manuscript and submissions enquiries, contact the Editor-in-Chief gordon.mallarkey@bioexcelpublishing.com
For all permissions, rights and reprints, contact David Hughes david.hughes@bioexcelpublishing.com
Peer review comments to author: 18 December 2019
References
- 1.WHO. HIV drug resistance report 2019. 2019. [Accessed January 16, 2020]. https://www.who.int/hiv/pub/drugresistance/hivdr-report-2019/en/
- 2.FDA. Integrated review for pifeltro and delstrigo approval. 2018. [Accessed January 16, 2020]. https://www.fda.gov/media/128270/download .
- 3.EMA. Assessment report for Pifeltro 20 September 2018. 2018. [Accessed January 16, 2020]. EMA/821709/2018. https://www.ema.europa.eu/en/documents/assessment-report/pifeltro-epar-public-assessment-report_en.pdf.
- 4.EMA. Assessment report for Delstrigo. 2018. [Accessed January 16, 2020]. EMA/874672/2018. https://www.ema.europa.eu/en/documents/assessment-report/delstrigo-epar-public-assessment-report_en.pdf.
- 5.Boyle A, Moss CE, Marzolini C, Khoo S. Clinical pharmacodynamics, pharmacokinetics, and drug interaction profile of doravirine. Clin Pharmacokinet. 2019;58(12):1553–1565. doi: 10.1007/s40262-019-00806-9. [DOI] [PubMed] [Google Scholar]
- 6.Colombier MA, Molina JM. Doravirine: a review. Curr Opin HIV AIDS. 2018;13(4):308–314. doi: 10.1097/COH.0000000000000471. [DOI] [PubMed] [Google Scholar]
- 7.Deeks ED. Doravirine: first global approval. Drugs. 2018;78(15):1643–1650. doi: 10.1007/s40265-018-0993-4. [DOI] [PubMed] [Google Scholar]
- 8.Cote B, Burch JD, Asante-Appiah E, et al. Discovery of MK-1439, an orally bioavailable non-nucleoside reverse transcriptase inhibitor potent against a wide range of resistant mutant HIV viruses. Bioorg Med Chem Lett. 2014;24(3):917–922. doi: 10.1016/j.bmcl.2013.12.070. [DOI] [PubMed] [Google Scholar]
- 9.Sluis-Cremer N, Temiz NA, Bahar I. Conformational changes in HIV-1 reverse transcriptase induced by nonnucleoside reverse transcriptase inhibitor binding. Curr HIV Res. 2004;2(4):323–332. doi: 10.2174/1570162043351093. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Lai MT, Feng M, Falgueyret JP, et al. In vitro characterization of MK-1439, a novel HIV-1 nonnucleoside reverse transcriptase inhibitor. Antimicrob Agents Chemother. 2014;58(3):1652–1663. doi: 10.1128/AAC.02403-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ren J, Bird LE, Chamberlain PP, Stewart-Jones GB, Stuart DI, Stammers DK. Structure of HIV-2 reverse transcriptase at 2.35-A resolution and the mechanism of resistance to non-nucleoside inhibitors. Proc Natl Acad Sci U S A. 2002;99(22):14410–14415. doi: 10.1073/pnas.222366699. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yee KL, Ouerdani A, Claussen A, de Greef R, Wenning L. Population pharmacokinetics of doravirine and exposure-response analysis in individuals with HIV-1. Antimicrob Agents Chemother. 2019;63(4) doi: 10.1128/AAC.02502-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Behm MO, Yee KL, Liu R, Levine V, Panebianco D, Fackler P. The effect of food on doravirine bioavailability: results from two pharmacokinetic studies in healthy subjects. Clin Drug Investig. 2017;37(6):571–579. doi: 10.1007/s40261-017-0512-5. [DOI] [PubMed] [Google Scholar]
- 14.Anderson MS, Gilmartin J, Cilissen C, et al. Safety, tolerability and pharmacokinetics of doravirine, a novel HIV non-nucleoside reverse transcriptase inhibitor, after single and multiple doses in healthy subjects. Antivir Ther. 2015;20(4):397–405. doi: 10.3851/IMP2920. [DOI] [PubMed] [Google Scholar]
- 15.Sanchez RI, Fillgrove KL, Yee KL, et al. Characterisation of the absorption, distribution, metabolism, excretion and mass balance of doravirine, a non-nucleoside reverse transcriptase inhibitor in humans. Xenobiotica. 2019;49(4):422–432. doi: 10.1080/00498254.2018.1451667. [DOI] [PubMed] [Google Scholar]
- 16.Fischetti B, Shah K, Taft DR, Berkowitz L, Bakshi A, Cha A. Real-world experience with higher-than-recommended doses of lamivudine in patients with varying degrees of renal impairment. Open Forum Infect Dis. 2018;5(10) doi: 10.1093/ofid/ofy225. ofy225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Johnson M, Kumar P, Molina JM, et al. Switching to doravirine/lamivudine/tenofovir disoproxil fumarate (DOR/3TC/TDF) maintains HIV-1 virologic suppression through 48 weeks: results of the DRIVE-SHIFT trial. J Acquir Immune Defic Syndr. 2019;81(4):463–472. doi: 10.1097/QAI.0000000000002056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Greaves W, Wan H, Yee KL, Kandala B, Vaddady P, Hwang C. Doravirine exposure and HIV-1 suppression after switching from an efavirenz-based regimen to doravirine/lamivudine/tenofovir disoproxil fumarate. Antimicrob Agents Chemother. 2019. [DOI] [PMC free article] [PubMed]
- 19.Molina JM, Squires K, Sax PE, et al. Doravirine (DOR) versus ritonavir-boosted darunavir (DRV+r): 96-week results of the randomized, double-blind, phase 3 DRIVE-FORWARD Noninferiority Trial 22nd International AIDS Conference; 2018; Amsterdam, the Netherlands. [Google Scholar]
- 20.Orkin C, Squires KE, Molina JM, et al. Doravirine/lamivudine/tenofovir disoproxil fumarate is non-inferior to efavirenz/emtricitabine/tenofovir disoproxil fumarate in treatment-naive adults with human immunodeficiency virus-1 infection: week 48 results of the DRIVE-AHEAD trial. Clin Infect Dis. 2019;68(4):535–544. doi: 10.1093/cid/ciy540. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Orkin C, Squires K, Molina JM, et al. ID Week. San Francisco, CA, USA: 2018. Doravirine/lamivudine/tenofovir DF continues to be non-inferior to efavirenz/emtricitabine/tenofovir DF in treatment-naïve adults with HIV-1 infection: week 96 results of the DRIVE-AHEAD trial. [Google Scholar]
- 22.Feng M, Sachs NA, Xu M, et al. Doravirine suppresses common nonnucleoside reverse transcriptase inhibitor-associated mutants at clinically relevant concentrations. Antimicrob Agents Chemother. 2016;60(4):2241–2247. doi: 10.1128/AAC.02650-15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bacheler LT, Anton ED, Kudish P, et al. Human immunodeficiency virus type 1 mutations selected in patients failing efavirenz combination therapy. Antimicrob Agents Chemother. 2000;44(9):2475–2484. doi: 10.1128/aac.44.9.2475-2484.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Reuman EC, Rhee SY, Holmes SP, Shafer RW. Constrained patterns of covariation and clustering of HIV-1 non-nucleoside reverse transcriptase inhibitor resistance mutations. J Antimicrob Chemother. 2010;65(7):1477–1485. doi: 10.1093/jac/dkq140. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Bacheler L, Jeffrey S, Hanna G, et al. Genotypic correlates of phenotypic resistance to efavirenz in virus isolates from patients failing nonnucleoside reverse transcriptase inhibitor therapy. J Virol. 2001;75(11):4999–5008. doi: 10.1128/JVI.75.11.4999-5008.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Zhang Z, Xu W, Koh YH, et al. A novel nonnucleoside analogue that inhibits human immunodeficiency virus type 1 isolates resistant to current nonnucleoside reverse transcriptase inhibitors. Antimicrob Agents Chemother. 2007;51(2):429–437. doi: 10.1128/AAC.01032-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Smith SJ, Pauly GT, Akram A, et al. Rilpivirine and doravirine have complementary efficacies against NNRTI-resistant HIV-1 mutants. J Acquir Immune Defic Syndr. 2016;72(5):485–491. doi: 10.1097/QAI.0000000000001031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Feng M, Wang D, Grobler JA, Hazuda DJ, Miller MD, Lai MT. In vitro resistance selection with doravirine (MK-1439), a novel nonnucleoside reverse transcriptase inhibitor with distinct mutation development pathways. Antimicrob Agents Chemother. 2015;59(1):590–598. doi: 10.1128/AAC.04201-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Schurmann D, Sobotha C, Gilmartin J, et al. A randomized, double-blind, placebo-controlled, short-term monotherapy study of doravirine in treatment-naive HIV-infected individuals. AIDS. 2016;30(1):57–63. doi: 10.1097/QAD.0000000000000876. [DOI] [PubMed] [Google Scholar]
- 30.Sterrantino G, Borghi V, Callegaro AP, et al. Prevalence of predicted resistance to doravirine in HIV-1-positive patients after exposure to non-nucleoside reverse transcriptase inhibitors. Int J Antimicrob Agents. 2019;53(4):515–519. doi: 10.1016/j.ijantimicag.2019.02.007. [DOI] [PubMed] [Google Scholar]
- 31.Molina JM, Squires K, Sax PE, et al. Doravirine versus ritonavir-boosted darunavir in antiretroviral-naive adults with HIV-1 (DRIVE-FORWARD): 48-week results of a randomised, double-blind, phase 3, non-inferiority trial. Lancet HIV. 2018;5(5):e211–e220. doi: 10.1016/S2352-3018(18)30021-3. [DOI] [PubMed] [Google Scholar]
- 32.Gatell JM, Morales-Ramirez JO, Hagins DP, et al. Forty-eight-week efficacy and safety and early CNS tolerability of doravirine (MK-1439), a novel NNRTI, with TDF/FTC in ART-naive HIV-positive patients. J Int AIDS Soc. 2014;17(4 Suppl 3):19532. doi: 10.7448/IAS.17.4.19532. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wong A, Goldstein D, Mallolas J, et al. Efficacy and safety of doravirine/lamivudine/tenofovir disoproxil fumarate (DOR/3TC/TDF) in treatment-naive adults with HIV-1 and transmitted nonnucleoside reverse transcriptase inhibitor resistance mutations. J Acquir Immune Defic Syndr. 2019;82(4):e47–e49. doi: 10.1097/QAI.0000000000002153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Markowitz M, Grobler JA. Islatravir for the treatment and prevention of infection with the human immunodeficiency virus type 1. Curr Opin HIV AIDS. 2020;15(1):27–32. doi: 10.1097/COH.0000000000000599. [DOI] [PubMed] [Google Scholar]
- 35.Molina JM, Yazdanpanah Y, Afani Saud A, et al. Islatravir (ISL, MK-8591) at doses of 0.25 to 2.25 mg QD in combination with doravirine maintains viral suppression through 48 weeks in adults with HIV-1 infection. IAS 2019: Conference on HIV Pathogenesis Treatment and Prevention; 2019; Mexico City, Mexico. [Google Scholar]
- 36.Hazuda D, Xu M, Ngo W, et al. Understanding the resistance profile of the HIV-1 NNRTI doravirine in combination with the novel NRTTI MK-8591. 22nd International AIDS Conference; 2018; Amsterdam, The Netherlands. [Google Scholar]
- 37.Spinner CD, Barber TJ. Are 96-week data enough for doravirine to DRIVE FORWARD? Lancet HIV. 2020;7(1):e2–e3. doi: 10.1016/S2352-3018(19)30370-4. [DOI] [PubMed] [Google Scholar]