Abstract
BACKGROUND AND PURPOSE: The appropriate choice of embolic materials with respect to the permanency of obliterated nidi after embolization and complications related to the procedure is essential for safe and effective embolization of cerebral arteriovenous malformations (AVMs). Our purpose was to ascertain the recanalization and complication rates after AVM treatment with polyvinyl alcohol (PVA) particles.
METHODS: Between 1988 and 1994, 36 AVMs were embolized with PVA particles at our institution. Follow-up angiographic findings and occurrence of complications during the embolization procedures were analyzed retrospectively.
RESULTS: Complete obliteration of the nidus immediately after embolization was achieved in five patients, and 80% to 99% obliteration was attained in 12 patients. Fifty-one follow-up angiographic examinations were performed 1 week to 60 months (mean, 7 months) after embolization in 31 patients. An increase in nidal size was seen on 15 follow-up angiograms (29%) and a decrease was seen in seven (14%). In 28 of the 51 angiograms obtained more than 1 month after follow up (mean, 13 months), 12 (43%) showed AVM enlargement. In four (80%) of five cases of complete obliteration, nidi reappeared on follow-up angiograms. Hemorrhagic complications occurred in three cases and ischemic ones in seven. One patient (3%) died and five (14%) suffered persistent neurologic deficits.
CONCLUSION: Embolization with PVA particles can produce significant volume reduction in AVM nidal size, but recanalization is a distinct possibility.
Recent technical advancements have increased the number of patients with cerebral arteriovenous malformations (AVMs) for whom embolization therapy may be appropriate, and gamma knife radiosurgery has proved to be an effective postembolization treatment (1, 2). Such progress led us to reconsider the role of embolization in the management of AVMs. The appropriate choice of embolization material is important in each case, because the permanency of obliterated nidi and the occurrence of complications during embolization are thought to be at least partially influenced by the characteristics of the material used (3–5). In our institution, the primary material used for AVM embolization from 1988 to 1994 was polyvinyl alcohol (PVA) particles. Preoperative embolization with PVA particles has been reported to control intraoperative bleeding (5, 6); however, obliteration with PVA particles has been considered to be less permanent than that achieved with liquid materials (5, 7, 8). Except for a couple of case reports describing the reappearance of embolized nidi (2, 9), few studies of angiographic changes in obliterated nidi embolized with PVA particles have been reported. To ascertain the rate of recanalization and of complications following PVA particle treatment of AVMs, we retrospectively analyzed angiographic changes and complications after embolization.
Methods
The data from 36 patients with intracerebral AVMs who underwent 62 embolization procedures with PVA particles at our institution from 1988 to 1994 were analyzed retrospectively. Their ages ranged from 16 to 72 years, with a mean age of 31 years; 20 patients were men and 16 were women. Initial symptoms were hemorrhage in 23 patients, convulsion in 10, headache in two, and ischemia in one. The locations of the AVMs were supratentorial in 29 cases and infratentorial in seven. The Spetzler-Martin grading system was used to classify all AVMs according to size, neurologic eloquence of adjacent brain, and venous drainage pattern (10). One AVM was grade 1, 11 were grade 2, 10 were grade 3, 11 were grade 4, and three were grade 5. Embolization with PVA particles was performed one to four times (average, 1.7 times) for each patient.
Transfemoral catheterization was performed under local anesthesia with systemic heparinization. A microcatheter (Tracker-18, Target Therapeutics, Fremont, CA) was coaxially introduced into feeding arteries through a guiding catheter, which was placed in the proximal carotid or vertebral artery. With the microcatheter in the proper position, a superselective provocative test with 2 to 3 mL of 1% lidocaine was performed just before embolization. The result of the provocative test was considered negative when no neurologic changes occurred. Three different sizes of PVA particles (49–149 μm, 150–249 μm, and 250–500 μm in diameter) suspended in a solution of ioxaglic acid (320 mg I/mL) diluted to half concentration with saline were used as embolic material in all patients. In some cases in which PVA particles passed through the nidi, additional embolic materials were used during the procedure to reduce blood flow: platinum coils were used 19 times in 15 cases; cyanoacrylate (isobutyl 2-cyanoacrylate [IBCA] and n-butyl cyanoacrylate [NBCA]), seven times in seven cases; silk sutures, six times in six cases; and 50% alcohol, six times in six cases. Embolization was discontinued when one of the following findings was observed during the procedure: the nidus disappeared, PVA particles refluxed into proximal normal vessels, or most PVA particles passed through a nidus. The duration of any one procedure was limited to 3 hours. Staged embolization with intervals of 1 week to 1 year was used in some patients with multiple feeders. The maximum diameter of the nidus before any treatment and immediately after the last embolization procedure was measured on the angiogram, and for an obliteration rate, the later was expressed as a percentage of the former. We confirmed the distribution of embolic material and the pathologic findings, including hemorrhage and infarction, on CT scans obtained 1 day after embolization.
After follow-up angiography, the residual nidi in noneloquent areas were removed surgically. For a variety of reasons (eg, the AVM was located in an eloquent area of the brain, the patient was in poor medical condition, or the patient refused surgery), follow-up angiographic studies were performed without other treatments in patients who did not undergo surgery. Since 1993, however, gamma knife radiosurgery has been performed at other institutions in patients with residual nidi less than 2 cm in diameter. AVMs that completely disappeared after embolization were followed up angiographically, and when the AVMs reappeared, embolization was attempted again. Once a patient received treatment other than embolization with PVA particles, he or she was excluded from the study.
Results
Obliteration Rate of Nidus Immediately after Last Embolization
The maximum diameter of the nidus on the angiogram obtained immediately after the last embolization was compared with that on the angiogram obtained before the first embolization. Total (100%) obliteration of the nidus was achieved in five cases (14%), 80% to 99% in 12 cases (34%), 60% to 79% in eight cases (23%), and less than 60% in 11 cases (30%). Table 1 summarizes the results in 36 patients with an AVM treated by embolization with PVA particles. On angiograms of the AVMs that were 80% obliterated, nidi were seen only in the late arterial to venous phases, and neither an intranidal aneurysm nor an arteriovenous fistula was observed.
TABLE 1:
Summary of 36 patients with AVMs embolized with PVA particles
Angiographic Changes after Embolization
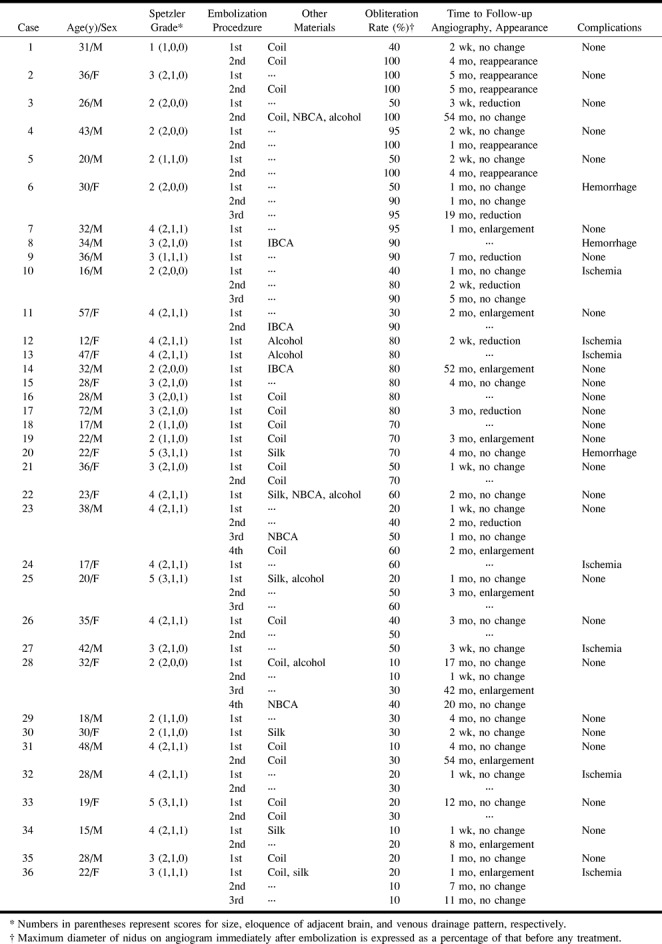
Among the 36 patients (62 embolization procedures), 31 (51 embolizations) had follow-up angiography 1 week to 60 months (mean, 7 months) after treatment. The size of the nidus on follow-up angiograms decreased after seven embolizations (14%) and increased after 15 (29%). For the remaining 29 embolizations (57%) that were followed up, no change was seen. Twenty-three (45%) of these 51 follow-up angiographic examinations were performed 1 month or less after the initial embolization procedure (Table 2). Only three (13%) of these 23 early follow-up angiographic studies showed AVM enlargement, whereas the other 12 cases (43%) of nidal enlargement were found on the 28 angiograms obtained 1 month or more (mean, 13 months) after initial treatment. In all, 14 (45%) of the 31 patients who underwent follow-up angiography showed an increase in the size of the nidus, six (19%) showed a decrease, and the remaining 11 (36%) showed no change on follow-up angiograms. Additional embolic materials (ie, platinum coils, acrylate, silk, and alcohol) were used in 31 embolization procedures in 25 patients. The angiographic changes seen after embolization with different materials are summarized in Table 3.
TABLE 2:
Comparison of angiographic changes in nidi at 1 month or less with those at more than 1 month after embolization with PVA particles

TABLE 3:
Angiographic changes in nidi after embolization with PVA particles and additional materials
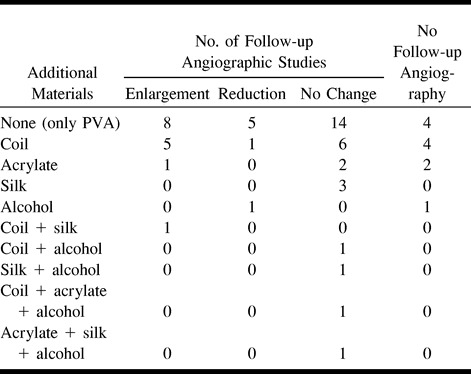
The presumptive main causes of nidal enlargement on angiograms were recanalization of feeders occluded during embolization (seven cases) (Fig 1), increase of blood supply from other feeders (six cases) (Fig 2), decrease in mass effect of hematoma (one case), and unclear origin (one case). In four (80%) of five cases of total obliteration, the nidi reappeared on follow-up angiograms; in the remaining case, complete obliteration was still apparent 54 months after embolization.
fig 1.
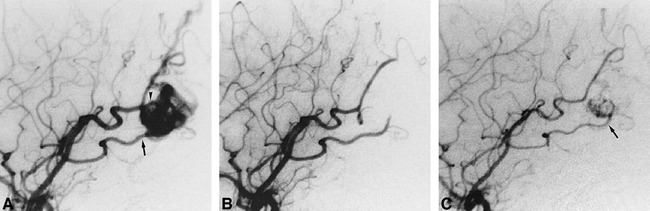
Case 5: Recanalization of feeders once obliterated during embolization. Only PVA particles were used in this case.
A, Before treatment, the left temporooccipital AVM is supplied by the angular (arrowhead) and temporooccipital (arrow) arteries on left internal carotid angiogram.
B, Both the angular and temporooccipital arteries are occluded during embolization, and the AVM disappears on angiogram obtained immediately after embolization.
C, The temporooccipital artery (arrow) recanalizes, and part of the AVM reappears on angiogram obtained 4 months after embolization.
fig 2.
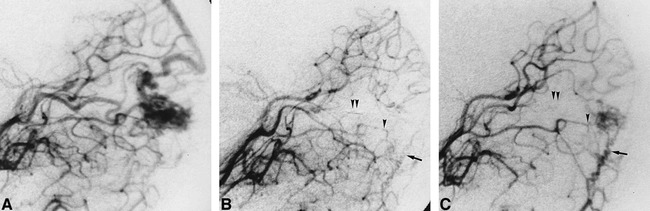
Case 19: Increase of blood supply from residual small feeders. Embolization was performed with PVA particles and platinum coils.
A, Before treatment, the right occipital AVM is supplied mainly by the parietooccipital artery on left vertebral angiogram.
B, The parietooccipital artery is occluded during embolization, and a small part of the AVM is fed by the calcarine and the posterior temporal (single arrowhead) arteries and by the meningeal branch of the vertebral artery (arrow) on angiogram obtained immediately after embolization. Double arrowheads indicate platinum coils in the parietooccipital artery.
C, Blood supply of the posterior temporal artery (single arrowhead) and meningeal branch of the vertebral artery (arrow) increases, and the residual AVM enlarges 3 months after embolization. Double arrowheads indicate platinum coils in the parietooccipital artery.
Complications during Embolization
Symptomatic complications were observed in 10 patients. Three patients had hemorrhagic complications (arterial damage during catheter manipulation in two and rupture from the AVM due to hemodynamic changes immediately after embolization in one) and seven had ischemic complications (perforator territory ischemia associated with basilar arterial spasm induced by mechanical stress with a microcatheter in three, progression of retrograde thrombosis into proximal arteries in one, embolization of functioning arteries passing through a nidus in one, brain edema induced by additional injection of alcohol in one, and migration of PVA particles into normal vessels in one). In regard to the occurrence of complications and the use of embolic materials other than PVA particles, either acrylate or silk was used in the two patients with hemorrhagic complications associated with catheter manipulation; alcohol was used in the patient with brain edema and in the patient in whom functioning arteries were embolized; and coils and silk were used in the patient in whom PVA particles migrated into normal vessels. In the remaining five patients, only PVA particles were used.
Of the 10 patients who suffered from complications during embolization, one died of massive cerebral hemorrhage caused by vessel perforation (mortality, 3%) and one with hemorrhage and four with ischemia sustained persisting deficits (morbidity, 14%). The remaining four patients recovered from transient neurologic impairments.
Discussion
Treatment of AVMs by embolization carries the major disadvantage of being less permanent than treatment by surgical removal. In the present study, nidi that initially showed a reduction in size were found to have increased in 15 (29%) of 51 follow-up angiographic studies. Twenty-eight follow-up angiograms were obtained 1 month or more (mean, 13 months) after embolization, and, of these, 12 (43%) showed AVM enlargement. On the other hand, only three (13%) of the 23 early follow-up angiograms showed enlargement of nidi. Standard et al (9) described an AVM that had been embolized with PVA particles that showed recanalization 2 years later, although no evidence of recanalization had been seen on angiograms at the 9-month follow-up; the possibility of recanalization of nidi was thought to increase after embolization with PVA particles as the follow-up period became longer. Mathis et al (2) evaluated angiographic changes in AVMs embolized with PVA particles immediately before radiosurgery and observed an increase in nidal size in three (13%) of 24 patients on average 3 months after embolization, which was a shorter follow-up period than that in our study. Reappearance rates of 0% to 12% have been reported with cyanoacrylates (1, 7, 11, 12), which are regarded as a more permanent embolic agent than PVA particles (8).
Characteristics of embolic materials are closely related to mechanisms of angiographic reappearance, including recanalization of embolic material, migration of embolic material into vessel wall and adjacent tissue, stagnation of contrast agent after proximal artery ligation, development of collateral vessels or nidi, resolution of thrombus, and vascularization through organized thrombi (13). Germano et al (13) histologically observed recanalized vessels in 18% of PVA embolizations. PVA particles cannot pass through vessels with a small diameter; therefore, proximal arterial ligation is encountered more frequently with PVA particles than with liquid materials. PVA particles are thought to be easily washed away by blood flow as compared with liquid materials, which plug a nidus immediately after injection. Coils are useful for preventing recanalization of feeding arteries (3, 4); however, they do not always reduce the vascular bed of the nidus. In this series, platinum coils, acrylate, silk, and 50% alcohol were employed as additional embolic materials. However, because of the small number of patients in whom these materials were used, we are unable to make a definite statement regarding the use of PVA particles in conjunction with other embolic materials.
Another problem with embolization is the occurrence of ischemic or hemorrhagic complications during and after treatment. The complication rate is influenced primarily by the technical aspects of the procedure, including vascular damage, which is related to operator skill and catheter features (5, 14), and by the characteristics of the embolization material, including hemodynamic changes after embolization (15–17) and inflammatory reaction of vessels (18). Among various materials used to embolize cerebral AVMs, PVA particles and cyanoacrylates (NBCA and IBCA) are representative materials that are readily available and have been well reported (6, 13, 19–22). Complication rates vary among studies. Shumacher and Horton (21) reported permanent deficits in 9% and no mortality with PVA particles. Purdy et al (19) found a 21% morbidity rate and an 8% mortality rate with PVA particles. Fox et al (23) reported 11% morbidity; and Guo et al (11) reported 19% morbidity with cyanoacrylates. In our series, permanent deficits were seen in five cases (14%) and death in one (3%). Two of the complications were caused by catheter operation, and one by additional injection of alcohol. The remaining three AVMs, which were located in eloquent areas, were embolized despite the high risk of symptomatic ischemia, because two were associated with hemorrhagic episodes and one with progressive ischemic symptoms. Therefore, the symptomatic complications in our series were not directly related to the decision to use PVA particles rather than another embolic agent. Because many conditions, especially the degree of technological advancement, differed among the studies cited, it is almost impossible to compare the complication rates on the basis of the embolic material used. Wallace et al (22), in an investigation of the safety and effectiveness of embolization using cyanoacrylates and PVA particles at a single institution, reported that comprehensive complication rates were lower with cyanoacrylates than with PVA particles; however, these rates were heavily influenced by the decreased number of surgical complications in the patients in whom acrylics were used. The authors stated the need to conduct a randomized, prospective, clinical trial to compare the relative safety and effectiveness of the two methods of embolization.
Our study has some limitations. The patient population was biased in that all patients were referred from other institutions, and some patients with small AVMs and/or AVMs in noneloquent areas were excluded because they were treated directly by surgery without embolization. We tried to embolize hemorrhagic AVMs located in eloquent and/or critical areas to eliminate intranidal aneurysms or arteriovenous fistulas that were thought to be the cause of the bleeding. Therefore, some AVMs in this series were regarded as unsuitable for surgery. Despite these limitations we believe that the results of this study are useful for gauging the permanency of embolization with PVA particles, because this is the first reported study that evaluates long-term angiographic changes after embolization with PVA particles.
Conclusion
Embolization with PVA particles can produce significant volume reduction in AVM nidal size, but reappearance of once-obliterated nidi and complications are distinct possibilities. Follow-up angiography is necessary to confirm obliterated nidal changes, especially after embolization with PVA particles.
Footnotes
Address reprint requests to Takatoshi Sorimachi, MD.
References
- 1.Gobin YP, Laurent A, Merienne L, et al. Treatment of brain arteriovenous malformations by embolization and radiosurgery. J Neurosurg 1996;85:19-28 [DOI] [PubMed] [Google Scholar]
- 2.Mathis JA, Barr JD, Horton JA, et al. The efficacy of particulate embolization combined with stereotactic radiosurgery for treatment of large arteriovenous malformations of the brain. AJNR Am J Neuroradiol 1995;16:299-306 [PMC free article] [PubMed] [Google Scholar]
- 3.Nakstad PH, Bakke SJ, Hald JK. Embolization of intracranial arteriovenous malformations and fistulas with polyvinyl alcohol particles and platinum fiber coils. Neuroradiology 1992;34:348-351 [DOI] [PubMed] [Google Scholar]
- 4.Purdy PD, Batjer HH, Risser RC, Samson D. Arteriovenous malformations of the brain: choosing embolic materials to enhance safety and ease of excision. J Neurosurg 1992;77:217-222 [DOI] [PubMed] [Google Scholar]
- 5.Wikholm G, Lundqvist C, Svendsen P. Transarterial embolization of cerebral arteriovenous malformations: improvement of results with experience. AJNR Am J Neuroradiol 1995;16:1811-1817 [PMC free article] [PubMed] [Google Scholar]
- 6.Spetzler RF, Martin NA, Carter LP, Flom RA, Randzeus PA, Wilkinson E. Surgical management of large AVMs by staged embolization and operative excision. J Neurosurg 1987;67:17-28 [DOI] [PubMed] [Google Scholar]
- 7.Fournier D, Terbrugge K, Rodesch G, Lasjaunias P. Revascularization of brain arteriovenous malformations after embolization with bucrylate. Neuroradiology 1990;32:497-501 [DOI] [PubMed] [Google Scholar]
- 8.Gruber A, Mazal PR, Bavinzski G, Killer M, Budka H, Richling B. Repermeation of partially embolized cerebral arteriovenous malformations: a clinical, radiologic, and histologic study. AJNR Am J Neuroradiol 1996;17:1323-1331 [PMC free article] [PubMed] [Google Scholar]
- 9.Standard SC, Guterman LR, Chavis TD, Hopkins LN. Delayed recanalization of a cerebral arteriovenous malformation following angiographic obliteration with polyvinyl alcohol embolization. Surg Neurol 1995;44:109-112 [DOI] [PubMed] [Google Scholar]
- 10.Spetzler RF, Martin NA. A proposed grading system for arteriovenous malformations. J Neurosurg 1986;65:476-483 [DOI] [PubMed] [Google Scholar]
- 11.Guo WY, Wikholm G, Karlsson B, Lindquist C, Snendsen P, Ericson K. Combined embolization and gamma knife radiosurgery for cerebral arteriovenous malformations. Acta Radiol 1993;34:600-606 [PubMed] [Google Scholar]
- 12.Vinuela F, Fox AJ, Pelz D, Debrun G. Angiographic follow-up of large cerebral AVMs incompletely embolized with isobutyl 2-cyanoacrylate. AJNR Am J Neuroradiol 1986;7:919-925 [PMC free article] [PubMed] [Google Scholar]
- 13.Germano IM, Davis RL, Wilson CB, Hieshima GB. Histopathological follow-up study of 66 cerebral arteriovenous malformations after therapeutic embolization with polyvinyl alcohol. J Neurosurg 1992;76:607-614 [DOI] [PubMed] [Google Scholar]
- 14.Frizzel RT, Fisher WS. Cure, morbidity, and mortality associated with embolization of brain arteriovenous malformations: a review of 1246 patients in 32 series over a 35-year period. Neurosurgery 1995;37:1031-1040 [DOI] [PubMed] [Google Scholar]
- 15.Takeuchi S, Kikuchi H, Karasawa J, et al. Cerebral hemodynamics in arteriovenous malformations: evaluation by single-photon emission CT. AJNR Am J Neuroradiol 1987;8:193-197 [PMC free article] [PubMed] [Google Scholar]
- 16.Takeuchi S, Abe H, Nishimaki K, et al. Cerebral haemodynamic changes after endovascular treatment of arteriovenous malformations: evaluation by single-photon emission CT. Acta Neurochir (Wien) 1994;127:142-150 [DOI] [PubMed] [Google Scholar]
- 17.Sorimachi T, Takeuchi S, Koike T, Minakawa T, Abe H, Tanaka R. Blood pressure monitoring in feeding arteries of cerebral arteriovenous malformations during embolization: a preventive role in hemodynamic complications. Neurosurgery 1995;37:1041-1048 [DOI] [PubMed] [Google Scholar]
- 18.Deveikis JP, Manz HJ, Luessenhop AJ, et al. A clinical and neuropathologic study of silk suture as an embolic agent for brain arteriovenous malformations. AJNR Am J Neuroradiol 1994;15:263-271 [PMC free article] [PubMed] [Google Scholar]
- 19.Purdy PD, Samson D, Batjer HH, Risser RC. Preoperative embolization of cerebral arteriovenous malformations with polyvinyl alcohol particles: experience in 51 adults. AJNR Am J Neuroradiol 1990;11:501-510 [PMC free article] [PubMed] [Google Scholar]
- 20.Vinuela F, Dion JE, Duckwiler G, et al. Combined endovascular embolization and surgery in the management of cerebral arteriovenous malformations: experience with 101 cases. J Neurosurg 1991;75:856-864 [DOI] [PubMed] [Google Scholar]
- 21.Schumacher M, Horton JA. Treatment of cerebral arteriovenous malformations with PVA: results and analysis of complications. Neuroradiology 1991;33:101-105 [DOI] [PubMed] [Google Scholar]
- 22.Wallace RC, Flom RA, Khayata MH, et al. The safety and effectiveness of brain arteriovenous malformation embolization using acrylic and particles: the experiences of a single institution. Neurosurgery 1995;37:606-618 [DOI] [PubMed] [Google Scholar]
- 23.Fox AJ, Girvin JP, Vinuela F, Drake CG. Rolandic arteriovenous malformations: improvement in limb function by IBC embolization. AJNR Am J Neuroradiol 1985;6:575-582 [PMC free article] [PubMed] [Google Scholar]


