Abstract
Background
This study compared response rates and outcomes of combined radiotherapy and immunotherapy (iRT) based on the type of checkpoint inhibitor (anti-cytotoxic T-lymphocyte-associated protein 4 (CTLA4) vs antiprogrammed death-1 (PD1)) for metastatic non-small cell lung cancer (mNSCLC).
Methods
We retrospectively reviewed two prospective trials of radiation combined with anti-CTLA4 or anti-PD1 for patients with mNSCLC. Patients undergoing non-salvage stereotactic body radiation therapy (SBRT) to lung sites were selected from both trials and grouped by the immunotherapeutic compound received. Endpoints included in-field and out-of-field response rates, and overall response rate (complete or partial response) (all by response evaluation criteria in solid tumors). Progression-free survival (PFS) and overall survival (OS) were estimated with the Kaplan-Meier method.
Results
Median follow-up times for the 33 patients (n=17 SBRT+anti-CTLA4, n=16 SBRT+anti-PD1) were 19.6 and 19.9 months. Response rates for out-of-field lesions were similar between anti-PD1 (37%) and anti-CTLA4 (24%) (p=0.054). However, global response rates for all lesions were 24% anti-CTLA4 vs 56% anti-PD1 (p=0.194). The PFS was 76% for anti-CTLA4 vs 94% anti-PD1 at 3 months, 52% vs 87% at 6 months, 31% vs 80% at 12 months, and 23% vs 63% at 18 months (p=0.02). Respective OS values were 76% vs 87% at 6 months, 47% vs 80% at 12 months, and 39% vs 66% at 18 months (p=0.08).
Conclusions
Both anti-CTLA4 and anti-PD1 agents prompt a similar degree of in-field and out-of-field responses after iRT, although the global response rate and PFS were statistically higher in the anti-PD1 cohort. Further dedicated study and biological mechanistic assessment is required.
Trial registration numbers
NCT02239900 and NCT02444741.
Keywords: radiotherapy, immunotherapy
Introduction
Immune checkpoint inhibitors have emerged as a treatment option for several types of recurrent or metastatic cancers.1 The most widely used agents presently are anti-cytotoxic T-lymphocyte-associated protein 4 (CTLA4) and anti-programmed death-1 (PD1) antibodies. Anti-CTLA4, widely studied for melanoma, acts to block the inhibitory signal involving the CTLA4 molecule between antigen-presenting cells and T lymphocytes.2 Anti-PD1 compounds analogously block the inhibitory signal involving the PD1 receptor.3 In both cases, diminishing tumor-mediated immune-attenuating effects results in more robust T-cell activation and immune-mediated neoplastic destruction.4
These effects may be augmented by radiation therapy (RT), especially stereotactic body radiation therapy (SBRT), which can enhance antigen release for immune recognition and modulate the tumor stroma to facilitate immune cell infiltration.5 6 RT can also lead to responses in out-of-field (unirradiated) tumors, also known as the abscopal effect. Despite emerging evidence regarding favourable interactions between immunotherapy and RT (combined radiotherapy and immunotherapy (iRT)), whether the various immunotherapeutic compounds lead to different outcomes in combination with radiation is currently unknown. Optimizing the efficacy of immunotherapeutics used in iRT is critical not only because several compounds are available to treat metastatic cancers but also because enhancing treatment efficacy could affect the cost-effectiveness of these agents.7 8
To address these gaps in knowledge, we retrospectively analyzed two single-institution prospective clinical trials to evaluate whether combining SBRT with anti-CTLA4 versus with anti-PD1 leads to different response and survival outcomes for patients with metastatic non-small cell lung cancer (mNSCLC).
Methods
Patients and study design
This retrospective review of two prospective iRT trials involving RT and anti-CTLA4 or anti-PD1 was approved by the institutional review board, as were the original trials, and written informed consent was obtained from all patients. Full information regarding inclusion/exclusion criteria, workup, follow-up, and RT planning (including dose constraints) is described in detail within the protocol for each study. Briefly, all patients in both trials were required to have ≥1 lesion amenable to RT and ≥1 additional non-contiguous lesion so that response to both in-field and out-of-field radiations could be monitored.
The first trial investigated SBRT with anti-CTLA4 (ipilimumab) for metastases from solid tumors to the liver, lung, or adrenal gland (n=143 enrolled). The trial protocol is available in online supplementary file 1. Patients received two cycles of anti-CTLA4 (3 mg/kg every 21 days), followed by SBRT and another two cycles of anti-CTLA4. SBRT was given either as 50 Gy in four daily fractions or as hypofractionated SBRT (60 Gy in 10 daily fractions) if a four-fraction regimen was deemed unfeasible or unsafe.
jitc-2019-000492supp001.pdf (9.1MB, pdf)
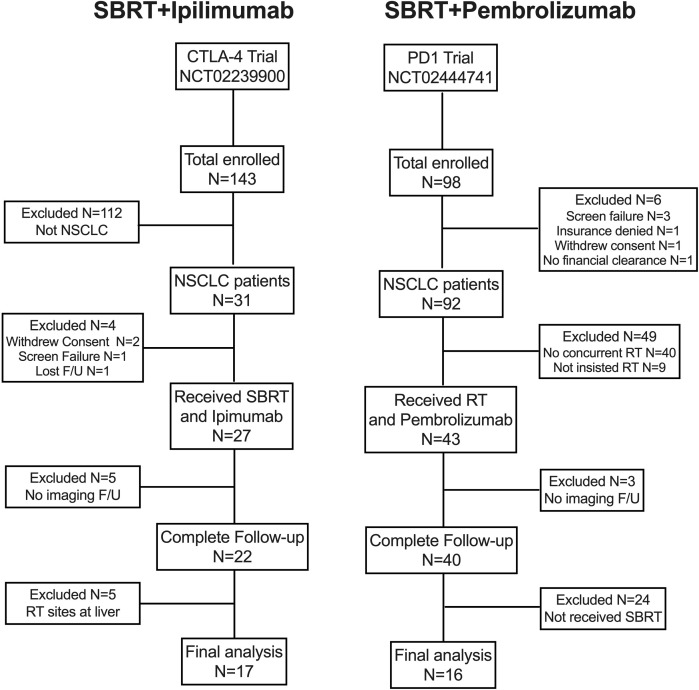
The other trial enrolled 98 patients with stage IV (metastatic) non-small cell lung cancer (NSCLC). The trial protocol is available in online supplementary file 2. RT and anti-PD1 (pembrolizumab 100 mg every 21 days, followed by 200 mg if tolerated) were given concurrently, followed by an additional anti-PD1. The phase I portion of this study examined the safety of the combined regimen; the phase II component randomized patients to anti-PD1 alone versus SBRT with anti-PD1 (patients with progressive disease (PD) on anti-PD1 only were allowed to undergo salvage RT). SBRT was given as 50 Gy in four daily fractions or, if that regimen was considered unfeasible or unsafe, as wide-field RT (non-SBRT) to a dose of 45 Gy in 15 daily fractions. The patient selection process for both of these trials is shown in figure 1.
Figure 1.
Flowchart of patient selection for this analysis. CTLA4, cytotoxic T-lymphocyte-associated protein 4; F/U, follow-up; NSCLC, non-small cell lung cancer; RT, radiation therapy; SBRT, stereotactic body radiation therapy.
jitc-2019-000492supp002.pdf (11.3MB, pdf)
Because the goal of this study was to evaluate and compare the effects of each immunotherapeutic agent given with SBRT, we compared the following two groups: (1) patients with mNSCLC from the first trial receiving classical or hypofractionated SBRT to lung sites and anti-CTLA4 (CTLA4 group) and (2) patients with mNSCLC from the other trial who received upfront (non-salvage) anti-PD1 and SBRT (PD1 group).
Endpoints and statistics
The primary endpoint of our study was best treatment response, which was evaluated prospectively in both protocols by using V.1.1 of the response evaluation criteria in solid tumors (RECIST). The same imaging modality (CT of the chest/abdomen/pelvis with or without positron emission tomography) was used before and after treatment, with follow-up scans obtained every 3 months after SBRT. Responses of both in-field and out-of-field lesions (ie, best response of a lesion known but not irradiated) were evaluated together (global response) and separately (in-field vs out-of-field). The overall response rate (ORR) was defined in terms of the RECIST guidelines as partial response (PR)/complete response (CR), and the disease control rate was defined as any response other than PD.
The three secondary endpoints were progression-free survival (PFS), defined from the start of RT to the occurrence of a new lesion anywhere in the body or RECIST-defined progression of an existing lesion; overall survival (OS), defined from the start of RT to the date of death from any cause (or censored at last contact); and treatment-related toxicity, which was assessed prospectively per protocol according to the Common Terminology Criteria for Adverse Events V.4.0.
Statistical analyses were done with SPSS V.25. First, clinical characteristics of both groups were tabulated and compared by using χ2 or Mann-Whitney U-test. In-field, out-of-field, and global response rates were compared between groups with Fisher’s exact test. Kaplan-Meier analysis was used to plot PFS and OS, and intergroup comparisons were made with log-rank tests.
Results
From September 2014 through August 2016, 241 patients were enrolled in either trial; after exclusions, 33 patients were the subject of this analysis: 17 in the SBRT+CTLA4 group and 16 in the SBRT+PD1 group (figure 1). Median follow-up times were 19.6 months (CTLA4) and 19.9 months (PD1) (p=0.212). Baseline characteristics were generally well balanced between groups, although patients in the CTLA4 group seemed to have had more systemic therapies before receiving iRT (mean 2.12 vs 1.13, p=0.157) (table 1).
Table 1.
Patient characteristics
| Characteristic | Anti-CTLA4 (n=17) |
Anti-PD1 (n=16) |
P value |
| Age (years) | |||
| Median | 66 | 63 | 0.866 |
| Range | 38–80 | 37–91 | |
| Race | |||
| White | 15 | 14 | 0.998 |
| Black | 1 | 1 | |
| Asian | 1 | 1 | |
| Sex | |||
| Male | 10 | 12 | 0.325 |
| Female | 7 | 4 | |
| Tumor histology | |||
| Adenocarcinoma | 14 | 12 | 0.606 |
| Squamous cell carcinoma | 3 | 4 | |
| Smoking history | |||
| Yes | 12 | 13 | 0.475 |
| No | 5 | 3 | |
| KPS score at diagnosis | |||
| ≥80 | 9 | 14 | |
| <80 | 8 | 2 | 0.057 |
| Prior systemic therapy | |||
| Yes | 14 | 9 | 0.103 |
| No | 3 | 7 | |
| Number of systemic therapy regimens, mean (range) | 2.12 (0–8) | 1.13 (0–4) | 0.157 |
| Prior radiation therapy | |||
| Yes | 3 | 5 | 0.362 |
| No | 14 | 11 | |
| Prior immunotherapy | |||
| Yes | 2 | 2 | 0.9 |
| No | 15 | 14 | |
| History of autoimmune disease | |||
| Yes | 1 | 1 | 0.736 |
| No | 16 | 15 | |
| Metastatic sites mean (range) | 3.1 (2–9) | 2.4 (2–6) | 0.207 |
| ALC change mean (range) | −28% (1% to −70%) | −19% (−3% to −63%) | 0.148 |
ALC, absolute lymphocyte counts; KPS, Karnofsky performance status.
Response
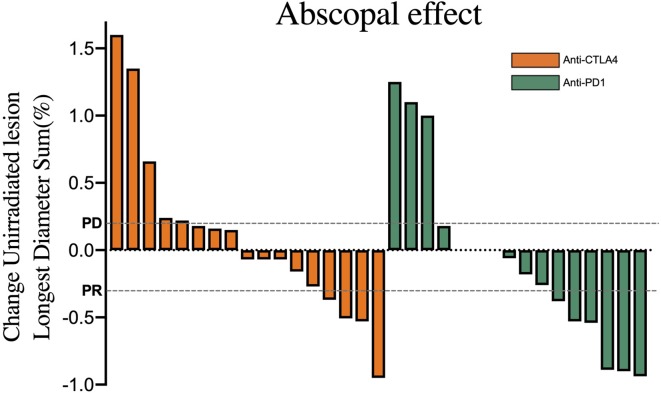
Images from at least one follow-up visit were required to evaluate treatment response. The out-of-field response rates were different, although not significantly so, for the two groups (figure 2): in the CTLA4 group, eight patients (24%) achieved PR, eight (47%) stable disease, and five (29%) PD; corresponding numbers in the PD1 group were six (37%) PR, seven (44%) stable disease, and three (19%) PD (p=0.454). The ORRs (PR/CR) were thus 24% (4 of 17) for the CTLA4 group and 37% (6 of 16) for the PD1 group (p=0.383), and the disease control rates (ie, any response other than PD) were 71% (12 of 17) for the CTLA4 group and 81% (13 of 16) for the PD1 group (p=0.674). Similarly, global response of all targeted lesions (ie, in-field and out-of-field) was no different between the CTLA4 and PD1 groups (p=0.194) (online supplementary figure S1A), although the ORR may have been higher in the PD1 group (p=0.054). Best responses of irradiated sites were also no different in the CTLA4 versus PD1 groups (p>0.05 for all) (online supplementary figure S1B).
Figure 2.
Waterfall and distribution plots of out-of-field responses. Values were derived from changes in the sum of the longest diameter of the out-of-field lesions, assessed according to response evaluation criteria in solid tumors guidelines: overall response rate (ie, PR/complete response) and disease control rate (ie, any response other than PD). CTLA4, cytotoxic T-lymphocyte-associated protein 4; PD, progressive disease; PD1, programmed death-1; PR, partial response; SD, stable disease.
jitc-2019-000492supp003.pdf (181.5KB, pdf)
Survival
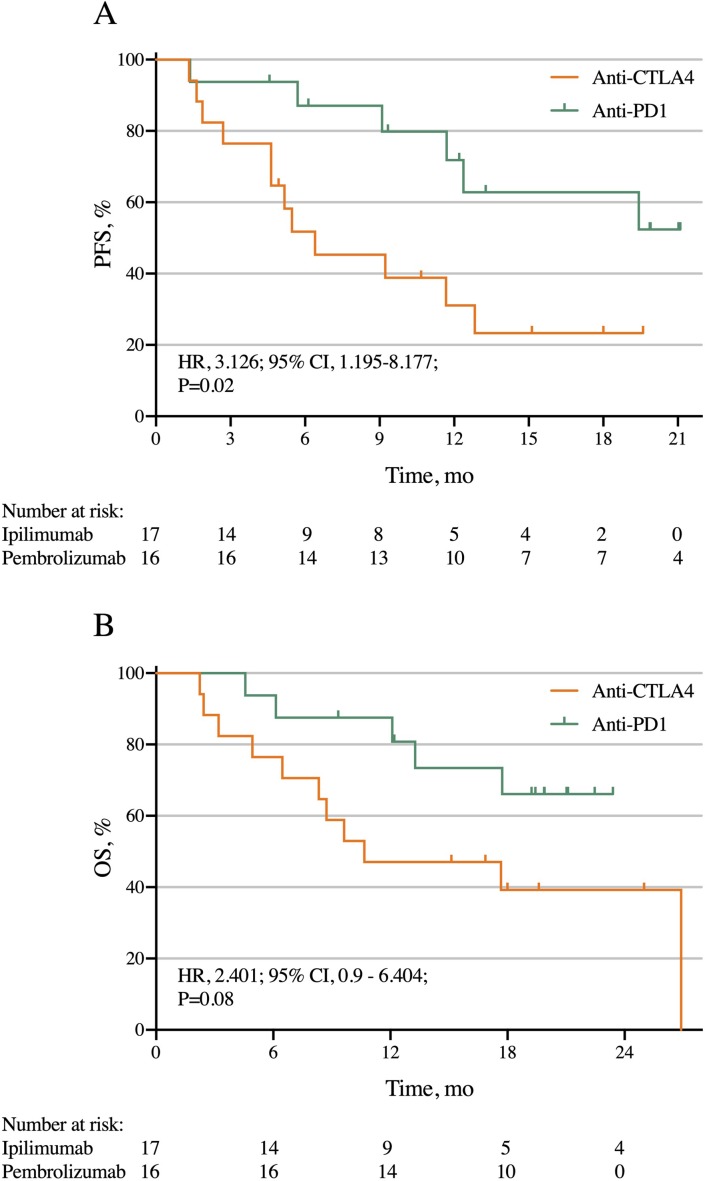
For all patients, the median PFS time for the CTLA4 group was 6.4 months and was not reached for the PD1 group (HR 3.126, 95% CI 1.195 to 8.177, p=0.02; figure 3A). The PFS rates were 76% CTLA4 vs 94% PD1 at 3 months, 52% vs 87% at 6 months, 31% vs 80% at 12 months, and 23% vs 63% at 18 months.
Figure 3.
PFS (A) and OS according to immunotherapy agent in two trials of stereotactic body radiation therapy given with either anti-CTLA4 or anti-PD1 for metastatic non-small cell lung cancer. CTLA4, cytotoxic T-lymphocyte-associated protein 4; OS, overall survival; PD1, programmed death-1; PFS, progression-free survival.
The median OS times were 10.7 months for the CTLA4 group versus not reached for the PD1 group (HR 2.401, 95% CI 0.9 to 6.404, p=0.08; figure 3B). Corresponding OS rates were 76% for the CTLA4 group vs 87% for the PD1 group at 6 months, 47% vs 80% at 12 months, and 39% vs 66% at 18 months.
Toxicity
Toxic effects possibly, probably, or definitely related to protocol treatment of any grade were noted in 19 (71%) of the CTLA4 group and 33 (69%) of the PD1 group (p=0.908). Similarly, no difference in severe (grade ≥3) events was found between groups (29% CTLA4 vs 19% PD1, p=0.475). A complete toxicity profile for all patients is shown in online supplementary table S1.
Discussion
RT has been found to interact with immunotherapeutic compounds so as to augment the immune response; however, whether that interaction depends on the particular type of immunotherapeutic agent has not been well studied. In this novel comparison of two prospective trials of iRT for mNSCLC, both anti-CTLA4 and anti-PD1 compounds led to similar in-field and out-of-field response rates; however, the global response may have been higher for the anti-PD1 cohort along with PFS. Although these findings came from prospective trials, they should nevertheless be considered hypothesis-generating and require verification in other, larger prospective trials of patients with mNSCLC.
There are multiple reasons to explain the equivocal in-field (irradiated existing lesions) and out-of-field (unirradiated existing lesions) response rates in this study. First, it is unlikely that immunotherapy (regardless of agent) would appreciably increase in-field control relative to RT alone, given that SBRT has shown high rates of local control.9 Second, the abscopal effect remains clinically uncommon (regardless of the agent used),10 implying that another mechanism may underlie the PFS and OS findings in the current study. In addition to higher global ORR, the reduced development of new out-of-field lesions could have driven our PFS findings. This notion seems to be supported by the results of an aforementioned trial of a CTLA4 inhibitor versus a PD1 inhibitor, hinting that the distant control of micrometastatic disease may be enhanced by PD1 inhibitors.11 12 However, there are other possible causes of the PFS results, such as biological factors (activation of distinct immune-galvanizing pathways that produce different degrees of immune response, especially when optimally timed with RT). Moreover, there was a trend toward higher performance status in the anti-PD1 cohort and more prior courses of systemic therapy in the anti-CTLA4 cohort (which may imply therapy-resistant disease and/or being further into the disease course than the anti-PD1 group).
Notably, the ORRs (especially in-field) in this study were high, roughly two to three times the ORRs in another study of patients given anti-PD1 alone and five times to anti-CTLA4 alone.13 This could suggest that the immune priming provided by radiation may be an integral component to augment the system responses to checkpoint therapy. The response rate to anti-PD1 alone in NSCLC is about 19%, whereas the response rate to anti-CTLA4 in NSCLC is about 4.8%.14 According to these results, the addition of RT can enhance the response rate in NSCLC by about 98% for PD1 agents and by about 389% for anti-CTLA4 compounds. These notions are corroborated by preliminary results of the PEMBRO-RT study, which randomized patients with previously treated NSCLC (although, like the present study, patients were not stratified by PD-L1 status) to receive a PD1 inhibitor with or without preceding ablative RT (24 Gy in three fractions).15 Whereas PD1 without preceding RT led to an ORR of 19%, the addition of RT led to an ORR of 41% as well as longer PFS times (1.8 months vs 6.4 months, p=0.04) with no increase in rates of toxicity (22% vs 17%). Although these results show promise for combined-modality therapy, they should also be viewed cautiously because of the small numbers of patients (n=64), short follow-up (reported ORRs were at 12 weeks), and lack of PD-L1 stratification (given that higher PD-L1 cutoffs are associated with higher ORR). As to the high response rate in anti-CTLA4 and SBRT combination, it could be interpreted not only by the immune priming provided by radiation but also by the effect from anti-CTLA4 to block radiation-induced high Tregs.16 Our data could be confirmed by another CTLA4-RT study, and the objective response rate in their NSCLC cohort was 18%.17
Even though this study was based on prospectively collected data, several limitations must be addressed. First, this was an unspecified secondary analysis of prospective trials, which does not constitute the same level of evidence as a prespecified secondary analysis. The sample sizes were also relatively small, which could be why a doubling of the grade ≥3 toxicity rate with anti-CTLA4 seen here was not statistically significant. Notably, however, our study had very low lung toxicity rates that were numerically comparable to RT alone.18 Second, no intertrial comparison can adequately balance all baseline factors. In this study, the group given anti-CTLA4 had a numerically (but not statistically) higher incidence of previous systemic therapy (since they came from our phase I group), which could result in patients with more resistant tumors, greater number of metastatic sites, and reduced lymphocyte counts. Third, in any study of immunotherapy, quantification of response remains an inexact science; we chose to use RECIST to facilitate comparisons with other work,15 19 although the immune-related response criteria are now in common use as well. Also, radiographical response may not necessarily equate to continued cellular viability or further metastatic potential, especially at early time points. Fourth, this study (like others)19 did not stratify patients by PD-L1 status, tumor mutational burden, or other biological variables that could influence response and outcomes. Fifth, the high ORRs in this study may have stemmed from exclusion of patients who may have rapidly deteriorated and not been able to obtain their first imaging evaluation; this could have been a selection bias, although good responders with immature follow-up may have been excluded for this reason as well. Sixth, in the current study, PFS and OS were measured from the date of applied radiotherapy. Five patients in the ipilimumab cohort received two cycles of ipilimumab before SBRT, which seemed to produce a survival disadvantage compared with the patients with concurrent treatment. However, our previous study showed that there was no obvious difference between SBRT followed by the ipilimumab cohort and concurrent SBRT+Ipi cohort.20 Finally, this study cannot rule out effects of concurrent versus sequential iRT because all patients given anti-CTLA4 received sequential iRT, and all subjects given anti-PD1 underwent concurrent iRT. These shortcomings underscore the need to consider this study to be hypothesis-generating and not a substitute for randomized data.
Conclusions
This novel study of two prospective trials of mNSCLC shows that both anti-CTLA4 and anti-PD1 agents prompt a similar degree of in-field and out-of-field responses after iRT, although the global response rate and PFS after iRT were statistically higher in the PD1 cohort. Although our findings were derived from prospective trials, these data should be considered hypothesis-generating and require verification by dedicated randomized studies, as well as biological mechanistic assessment.
Footnotes
DC and HM contributed equally.
Contributors: DC and JW designed the study, interpreted the data, analyzed the results and wrote the manuscript. All authors helped with manuscript writing, review and revision. VV, HB, HM, AY, DC, and CG assisted with manuscript review and revision.
Funding: This study was supported in part by Cancer Center Support (Core, grant number CA016672) from the National Cancer Institute, National Institutes of Health to The University of Texas MD Anderson Cancer Center.
Competing interests: JW has received grants from Bristol-Myers Squibb, Merck, Varian, and OncoResponse; he also is a cofounder of Healios, MolecularMatch, and OncoResponse (with ownership interest); he is on the scientific advisor board of Mavu, Reflexion Medical and Checkmate Pharmaceuticals, and receives laboratory research support from Varian, Incyte, Calithera, and Checkmate Pharmaceuticals.
Patient consent for publication: Not required.
Ethics approval: This post hoc analysis of single-institution clinical trials was reviewed and approved by the MD Anderson Cancer Center institutional review board.
Provenance and peer review: Not commissioned; externally peer reviewed.
Data availability statement: All data relevant to the study are included in the article or uploaded as supplementary information. All data has been presented in this article.
References
- 1. Sharma P, Allison JP. Immune checkpoint targeting in cancer therapy: toward combination strategies with curative potential. Cell 2015;161:205–14. 10.1016/j.cell.2015.03.030 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2. Marconcini R, Spagnolo F, Stucci LS, et al. Current status and perspectives in immunotherapy for metastatic melanoma. Oncotarget 2018;9:12452–70. 10.18632/oncotarget.23746 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3. Beavis PA, Henderson MA, Giuffrida L, et al. Dual PD-1 and CTLA-4 Checkpoint Blockade Promotes Antitumor Immune Responses through CD4+Foxp3- Cell-Mediated Modulation of CD103+ Dendritic Cells. Cancer Immunol Res 2018;6:1069–81. 10.1158/2326-6066.CIR-18-0291 [DOI] [PubMed] [Google Scholar]
- 4. Topalian SL, Drake CG, Pardoll DM. Immune checkpoint blockade: a common denominator approach to cancer therapy. Cancer Cell 2015;27:450–61. 10.1016/j.ccell.2015.03.001 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5. Demaria S, Coleman CN, Formenti SC. Radiotherapy: changing the game in immunotherapy. Trends Cancer 2016;2:286–94. 10.1016/j.trecan.2016.05.002 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6. Seyedin SN, Schoenhals JE, Lee DA, et al. Strategies for combining immunotherapy with radiation for anticancer therapy. Immunotherapy 2015;7:967–80. 10.2217/imt.15.65 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7. Verma V, Sprave T, Haque W, et al. A systematic review of the cost and cost-effectiveness studies of immune checkpoint inhibitors. J Immunother Cancer 2018;6:128. 10.1186/s40425-018-0442-7 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8. Joseph RW, Shillington AC, Macahilig C, et al. Factors associated with immunotherapy selection in patients with advanced melanoma. Immunotherapy 2018;10:1361–9. 10.2217/imt-2018-0150 [DOI] [PubMed] [Google Scholar]
- 9. Schonewolf CA, Heskel M, Doucette A, et al. Five-year long-term outcomes of stereotactic body radiation therapy for operable versus medically inoperable stage I non-small-cell lung cancer: analysis by operability, fractionation regimen, tumor size, and tumor location. Clin Lung Cancer 2019;20:e63–71. 10.1016/j.cllc.2018.09.004 [DOI] [PubMed] [Google Scholar]
- 10. Ngwa W, Irabor OC, Schoenfeld JD, et al. Using immunotherapy to boost the abscopal effect. Nat Rev Cancer 2018;18:313–22. 10.1038/nrc.2018.6 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11. Weber J, Mandala M, Del Vecchio M, et al. Adjuvant nivolumab versus ipilimumab in resected stage III or IV melanoma. N Engl J Med 2017;377:1824–35. 10.1056/NEJMoa1709030 [DOI] [PubMed] [Google Scholar]
- 12. Schachter J, Ribas A, Long GV, et al. Pembrolizumab versus ipilimumab for advanced melanoma: final overall survival results of a multicentre, randomised, open-label phase 3 study (KEYNOTE-006). Lancet 2017;390:1853–62. 10.1016/S0140-6736(17)31601-X [DOI] [PubMed] [Google Scholar]
- 13. Derangère V, Fumet JD, Boidot R, et al. Does bevacizumab impact anti-EGFR therapy efficacy in metastatic colorectal cancer? Oncotarget 2016;7 10.18632/oncotarget.7008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14. Zatloukal P, Heo DS, Park K, et al. Randomized phase II clinical trial comparing tremelimumab (CP-675, 206) with best supportive care (BSC) following first-line platinum-based therapy in patients (PTS) with advanced non-small cell lung cancer (NSCLC). J Clin Oncol 2009;27:8071. [Google Scholar]
- 15. Herbst RS, Baas P, Kim D-W, et al. Pembrolizumab versus docetaxel for previously treated, PD-L1-positive, advanced non-small-cell lung cancer (KEYNOTE-010): a randomised controlled trial. Lancet 2016;387:1540–50. 10.1016/S0140-6736(15)01281-7 [DOI] [PubMed] [Google Scholar]
- 16. Muroyama Y, Nirschl TR, Kochel CM, et al. Stereotactic radiotherapy increases functionally suppressive regulatory T cells in the tumor microenvironment. Cancer Immunol Res 2017;5:992–1004. 10.1158/2326-6066.CIR-17-0040 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17. Formenti SC, Rudqvist N-P, Golden E, et al. Radiotherapy induces responses of lung cancer to CTLA-4 blockade. Nat Med 2018;24:1845–51. 10.1038/s41591-018-0232-2 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18. Verma V, Cushman TR, Selek U, et al. Safety of combined immunotherapy and thoracic radiation therapy: analysis of 3 single-institutional phase I/II trials. Int J Radiat Oncol Biol Phys 2018;101:1141–8. 10.1016/j.ijrobp.2018.04.054 [DOI] [PubMed] [Google Scholar]
- 19. Reck M, Rodríguez-Abreu D, Robinson AG, et al. Pembrolizumab versus chemotherapy for PD-L1-positive non-small-cell lung cancer. N Engl J Med 2016;375:1823–33. 10.1056/NEJMoa1606774 [DOI] [PubMed] [Google Scholar]
- 20. Welsh JW, Tang C, de Groot P, et al. Phase II trial of ipilimumab with stereotactic radiation therapy for metastatic disease: outcomes, toxicities, and low-dose radiation-related Abscopal responses. Cancer Immunol Res 2019;7:1903–9. 10.1158/2326-6066.CIR-18-0793 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
jitc-2019-000492supp001.pdf (9.1MB, pdf)
jitc-2019-000492supp002.pdf (11.3MB, pdf)
jitc-2019-000492supp003.pdf (181.5KB, pdf)