Abstract
Relish, a transcription factor, is a critical downstream component of the immune deficiency (Imd) pathway and regulates host defense against bacterial infection by mediating antimicrobial peptide (AMP) synthesis. Understanding the immunological function of the mealworm beetle, Tenebrio molitor Relish (TmRelish) will be instructive in understanding insect immunity. In the present study, full-length ORF of TmRelish was retrieved from T. molitor-expressed sequence tags and RNA-seq database. The predicted TmRelish amino acid sequence contained an N-terminal Rel-homology domain; an Ig-like, plexin, and transcription factor domain; ankyrin repeat motifs; a nuclear localization signal; and a C-terminal death domain and shared the highly conserved structure of the Relish proteins of other insect species. TmRelish mRNA was detected in all developmental stages of the insect; however, the highest levels were detected in the larval gut tissue and adult hemocytes. TmRelish mRNA level was upregulated in the fat body, hemocyte, and gut tissue 9 h after infection of T. molitor larvae by the gram-negative bacteria, Escherichia coli. Furthermore, TmRelish knockdown led to significantly higher mortality of the E. coli-infected larvae, and significantly lower mortality of larvae infected with Staphylococcus aureus or Candida albicans. To elucidate the possible cause of mortality, we measured AMP transcription in the fat body, hemocytes, gut, and Malpighian tubules (MTs) of T. molitor larvae. TmRelish knockdown suppressed the expression of nine AMP genes in the larval fat body and gut tissue during E. coli infection, suggesting that TmRelish positively regulates AMP expression in both immune-related tissues, in response to E. coli challenge. Furthermore, negative regulation of some AMPs by TmRelish in the MTs, gut and hemocytes in response to C. albicans infection suggests a crosstalk between the Toll and Imd pathways.
Subject terms: Innate immunity, RNAi, Transcription
Introduction
The innate immune response is the first line of defense in vertebrates; however, it is the only form of defense in invertebrates. The lack of a specific adaptive immune response mechanism has conferred the innate immune components of invertebrate species with plasticity in their mechanism of action. Consequently, although similar immune signaling cascades are involved in clearing various microorganisms, invertebrate pathogen surveillance and immune activation systems lack high specificity and memory1. Most invertebrates possess a range of cellular and humoral defenses that allow them to overcome infectious agents entering through the gut or exoskeleton/cuticle. In insects, cellular defense mechanisms can be modulated by circulating hemocytes, sessile blood cells that can initiate wound repair/blood coagulation, phagocytosing pathogens in the hemocoel2, encapsulating multicellular parasites3, and formation of cell aggregates around microorganisms by a process termed nodulation4. Humoral insect defense components include antimicrobial peptides (AMPs), prophenoloxidase cascade intermediates, lectins, and complement-like factors. The fat body of insects synthesizes AMPs in response to non-self-microbes. These cationic peptides can disrupt microbial membranes, interfere with bacterial metabolism, and target cytoplasmic components. In Drosophila, levels of AMP induction and action have been associated with the type of infectious agent5; however, this is not universally true for other insects and invertebrate species. The prophenoloxidase enzymatic cascade causes the release of cytotoxins or opsonins against invading parasites and pathogens, but unlike AMPs, these do not affect host survival following infection6,7. Insects also possess numerous lectins that recognize a wide range of pathogens via their carbohydrate moieties and interact with the complement system to promote the lysis of microorganisms8.
The humoral immune system of Drosophila involves at least three independent signal transduction pathways that lead to the transcriptional induction of AMPs. The Toll signaling pathway is preferentially activated in response to the non-self recognition of fungal and gram-positive bacterial cell surface carbohydrates, such as β-1,3-glucan and Lys-type peptidoglycan, respectively. The Toll pathway requires the Toll ligand spätzle, dorsal, and dorsal group genes such as Tube, Pelle, and Cactus to activate the transcription of effector AMPs such as drosomycin and metchnikowin (antifungal peptides) and defensin (anti-gram- positive bacterial peptide)9,10. In the 18-wheeler-Dif pathway, which is related to the Toll pathway, the Toll-like gene, 18-wheeler, affects the transcription of antibacterial genes such as Attacin instead of diptericin or drosomycin. Both the Toll-dorsal and 18-wheeler-Dif pathways are required for regulation of the AMP, cecropin. The signaling cascade of the 18-wheeler-Dif pathway involves nuclear translocation of dorsal-like immunity factor (Dif) in the absence of Dorsal group genes and the presence of IκB kinase11. Conversely, the meso-diaminopimelic acid (DAP)-type peptidoglycans found on the cell-surface of gram-negative bacteria are recognized as non-self by the peptidoglycan-recognition proteins (PGRP)-LC and PGRP-LE. Signaling is then modulated downstream by the adapter protein IMD, as part of the IMD signaling pathway, and the NF-κB transcription factor Relish, to induce diptericin expression12. NF-κB/Rel proteins, which induces the transcription of robust pro-inflammatory responses with the help of AMPs, therefore appear to be crucial to our understanding of host humoral responses to microbial infection.
The three NF-κB transcription factors, Dorsal, Dif, and Relish share a highly conserved Rel homology domain (RHD; 300 amino acids) and can be grouped into two classes based on their C-terminal RHD sequences. Members of the first class, which includes Drosophila Relish (DmRelish), are composed of multiple Ankyrin repeats and activate AMP transcription by forming dimers with members of the second class, including Dorsal and Dif. The second class of NF-κB factors contain transactivation domains, which activate AMP gene transcription, although these tend to be less conserved13. In Drosophila, only Relish, not Dif or Dorsal, is involved in the coordination of the Toll, IMD, and 18-wheeler immunity pathways14. Relish activity relies greatly on the uncoupling of its C-terminal IκB-like domain; by the caspase DREDD and IκB-kinase (IKK) complex, which leads to IκB degradation15. DmRelish homologues have been identified in other insects where they participate in the regulation of AMP genes in response to pathogenic infections. Relish1 and Relish2 from Anopheles gambiae and Aedes aegypti, respectively, are key transcriptional activators of diptericin/drosomycin (antifungal defense) and defend against gram-positive S. aureus16. The DmRelish homolog of Culex quinquefasciatus is activated by the West Nile Virus (WNV), resulting in the triggering of an antiviral response17. Bombyx mori Relish encodes BmRelish1 and BmRelish2 (dominant negative factor of BmRelish1), which activate or inhibit cecropinB1, respectively18. The Manduca sexta Relish isoforms MsRel2A and MsRel2B have also been shown to induce the transcription of AMP genes16. Furthermore, Relish silencing in the adult honey bee, Apis mellifera, infected with Escherichia coli reduced the levels of abaecin and hymenoptaecin mRNA expression, but not defensin-119.
In Drosophila, the role of Relish in all three signaling pathways has been explained. Studying the regulatory role of Relish in the IMD signaling pathway has been straightforward; the induction of diptericin and other antibacterial defense genes has been studied by mutational analysis. In the IMD signaling pathway, the endoproteolytic cleavage of Relish by DREDD results in the release of the RHD domain from the C-terminal ankyrin repeat/IκB domain. Subsequently, RHD translocates to the nucleus and initiates the transcription of target genes20. Compelling evidence regarding the role of Relish as a master regulator of AMP gene expression following microbial challenge in Drosophila and in other insect models has provided important insights into insect humoral immunity. Using the mealworm beetle, Tenebrio molitor, we have elucidated intracellular events within the IMD signaling pathway leading to the activation of effector AMPs. We determined the role of T. molitor Relish (TmRelish) in regulating the expression of antibacterial and antifungal peptides and the survival of larvae following microbial challenge. We utilized the RNA interference (RNAi) approach to knockdown TmRelish in the immune tissues of T. molitor larvae and studied the regulation of AMP genes following bacterial and fungal infections.
Materials and Methods
Experimental insects and microorganisms
T. molitor was reared in an insectary in the dark at 27 ± 1 °C and 60 ± 5% relative humidity (RH). T. molitor larvae were fed an artificial diet consisting of 170 g wheat flour, 0.5 g chloramphenicol, 20 g roasted soy flour, 0.5 g sorbic acid, 0.5 mL propionic acid, 10 g soy protein, and 100 g wheat bran in 200 mL of distilled water, autoclaved at 121 °C for 20 min. Only 10th–12th instar larvae (approximately 2.4 cm, in length) were used in these experiments.
The gram-negative bacteria E. coli (strain K12), gram-positive bacteria Staphylococcus aureus (strain RN4220), and the fungus Candida albicans (strain AUMC 13529) were used for the immune challenge studies. E. coli and S. aureus were grown overnight at 37 °C in Luria-Bertani (LB) broth. C. albicans was cultured overnight at 37 °C in Sabouraud Dextrose broth. The overnight cultures were harvested by centrifugation at 5000 rpm for 10 min and subsequently washed twice with phosphate buffer saline (PBS, pH 7.0). The density of the cultures was measured at OD600, and the cells were resuspended in PBS at concentrations of 1 × 106 (E. coli and S. aureus) and 5 × 104 (C. albicans) cells/µL, and used to study host-pathogen interactions.
In silico identification and sequence characterization of TmRelish
To identify TmRelish, a local tblastn analysis was performed using the amino acid sequence of Tribolium castaneum Relish (TcRelish) (GenBank: EEZ97717.1) as a query against the locally curated T. molitor nucleotide database (derived from T. molitor RNA sequencing). The in silico results were sufficient to derive the full-length ORF of TmRelish. The deduced TmRelish amino acid sequence was used for domain analysis prediction using InterProScan 5.021 and blastx22. The nuclear localization signal (NLS) was predicted using cNLS Mapper23. To estimate the genetic relatedness of TmRelish, multiple sequence alignments were performed using ClustalX2.124, and phylogenetic analysis was performed using MEGA 725. The maximum-likelihood (ML) method was used to construct a phylogenetic tree using the JTT matrix-based model26 and the Relish protein of the mud crab, Scylla paramamosain (SpRelish: AZK36045.1), was used as the outgroup. To determine the confidence of the branches, we conducted a bootstrap analysis with 1,000 replicates. Multiple sequence analysis and phylogenetic analysis were carried out using amino acid sequences of Relish from orthologous insect groups (at least 15 sequences registered with the NCBI).
Expression analysis of TmRelish mRNA in different developmental stages and tissues
To evaluate developmental expression of TmRelish mRNA, samples (n = 20 for each stage) were collected from eggs (EG), young larvae (YL; 10th–12th instar), late instar larvae (LL; 19th–20th instar), pre-pupae (PP), 1–7-day-old pupae (P1–P7), and 1–5-day-old adults. To investigate tissue-specific expression, the fat body, MTs, gut, integument, hemocytes, ovary, and testis, were dissected from healthy larvae and adults and stored in RNA later solution at −20 °C until further use. For TmRelish mRNA expression analysis, 20 insects were used from which at least six insects were pooled together as one group (total of three groups). Tissue samples were collected from each group such that three samples for each tissue were obtained. Total RNA was extracted from the samples according to the modified LogSpin RNA isolation method with minor modifications27. Briefly, the samples were homogenized with a guanidine thiocyanate-based RNA lysis buffer [20 mM EDTA, 20 mM MES buffer, 3 M guanidine thiocyanate, 200 mM sodium chloride, 40 μM phenol red, 0.05% Tween-80, 0.5% acetic acid glacial (pH 5.5), and 1% isoamyl alcohol in 50 mL], incubated for 1 min at room temperature (approximately 25 °C), and centrifuged at 15,000 rpm for 30 s at 4 °C in a silica spin column (Bioneer, Korea, KA-0133-1). The aqueous phase was discarded, and the genomic DNA was digested by incubating the samples with DNase (Promega, USA, M6101) at 37 °C for 15 min. Thereafter, the silica spin column was washed with 450 mL of 3 M sodium acetate buffer, followed by centrifugation at 15,000 rpm at 4 °C for 30 s. Subsequently, 500 mL of 80% ethanol was added to the spin column and centrifuged at 15,000 rpm at 4 °C for 30 s. After drying the spin column for 1 min, total RNA was eluted in 30 µL of distilled water. For cDNA synthesis, 2 μg of total RNA was incubated with an oligo-(dT)12–18 primer at 72 °C for 5 min, 42 °C for 1 h, and 94 °C for 5 min in the AccuPower RT PreMix (Bioneer, Korea) solution.
Quantitative reverse-transcription PCR (qRT-PCR) was used to analyze the developmental and tissue distribution of TmRelish with gene-specific primers. The cDNA fragments obtained were diluted at a ratio of 1:20 and qRT-PCR was carried out under the following conditions: denaturation of 95 °C for 20 s followed by 45 cycles at 95 °C for 5 s and 60 °C for 20 s. Relative TmRelish mRNA expression was normalized to the T. molitor ribosomal protein (TmL27a), which was used as an internal control and was amplified under the same conditions. The gene-specific and internal control primers were designed using Primer3.0 plus (http://www.bioinformatics.nl/cgi-bin/primer3plus/primer3plus.cgi); sequence information has been provided in Table 1.
Table 1.
Sequences of the primers used in the study.
| Primer | Sequence (5′-3′) |
|---|---|
|
TmRelish_qPCR_Fw TmRelish_qPCR_Rv |
5′-AGCGTCAAGTTGGAGCAGAT-3′ 5′-GTCCGGACCTCATCAAGTGT-3′ |
|
TmRelish_Temp_Fw TmRelish_Temp_Rv |
5′-TGTGGGAAGATTACGGGAAA-3′ 5′-CAAATTGGCCACGATCTCTT-3′ |
|
dsTmRelish_Fw dsTmRelish_Rv |
5′-TAATACGACTCACTATAGGGT GACGTGCACCATCAATA-3′ 5′-TAATACGACTCACTATAGGGT GCGTGTTTGGCCTTGAT-3′ |
|
dsEGFP_Fw dsEGFP_Rv |
5′-TAATACGACTCACTATAGGG TACGTAAACGGCCACAAGTTC-3′ 5′-TAATACGACTCACTATAGGG TTGCTCAGGTAGTGTTGTCG-3′ |
|
TmTenecin-1_Fw TmTenecin-1_Rv |
5′-CAGCTGAAGAAATCGAACAAGG-3′ 5′-CAGACCCTCTTTCCGTTACAGT-3′ |
|
TmTenecin-2_Fw TmTenecin-2_Rv |
5′-CAGCAAAACGGAGGATGGTC-3′ 5′-CGTTGAAATCGTGATCTTGTCC-3′ |
|
TmTenecin-3_Fw TmTenecin-3_Rv |
5′-GATTTGCTTGATTCTGGTGGTC-3′ 5′-CTGATGGCCTCCTAAATGTCC-3′ |
|
TmTenecin-4_Fw TmTenecin-4_Rv |
5′-GGACATTGAAGATCCAGGAAAG-3′ 5′-CGGTGTTCCTTATGTAGAGCTG-3′ |
|
TmDefensin-1_Fw TmDefencin-1_Rv |
5′-AAATCGAACAAGGCCAACAC-3′ 5′-GCAAATGCAGACCCTCTTTC-3′ |
|
TmDefensin-2_Fw TmDefensin-2_Rv |
5′-GGGATGCCTCATGAAGATGTAG-3′ 5′-CCAATGCAAACACATTCGTC-3′ |
|
TmColeoptericin-1_Fw TmColeoptericin-1_Rv |
5′-GGACAGAATGGTGGATGGTC-3′ 5′-CTCCAACATTCCAGGTAGGC-3′ |
|
TmColeoptericin-2_Fw TmColeoptericin-2_Rv |
5′-GGACGGTTCTGATCTTCTTGAT-3′ 5′-CAGCTGTTTGTTTGTTCTCGTC-3′ |
|
TmAttacin-1a_Fw TmAttacin-1a_Rv |
5′-GAAACGAAATGGAAGGTGGA-3′ 5′-TGCTTCGGCAGACAATACAG-3′ |
|
TmAttacin-1b_Fw TmAttacin-1b_Rv |
5′-GAGCTGTGAATGCAGGACAA-3′ 5′-CCCTCTGATGAAACCTCCAA-3′ |
|
TmAttacin-2_Fw TmAttacin-2_Rv |
5′-AACTGGGATATTCGCACGTC-3′ 5′-CCCTCCGAAATGTCTGTTGT-3′ |
|
TmCecropin-2_Fw TmCecropin-2_Rv |
5′-TACTAGCAGCGCCAAAACCT-3′ 5′-CTGGAACATTAGGCGGAGAA-3′ |
|
TmThaumatin-like protein-1_Fw TmThaumatin-like protein-1_Rv |
5′-CTCAAAGGACACGCAGGACT-3′ 5′-ACTTTGAGCTTCTCGGGACA-3′ |
|
TmThaumatin-like protein-2_Fw TmThaumatin-like protein-2_Rv |
5′-CCGTCTGGCTAGGAGTTCTG-3′ 5′-ACTCCTCCAGCTCCGTTACA-3′ |
|
TmL27a_qPCR_Fw TmL27a_qPCR_Rv |
5′-TCATCCTGAAGGCAAAGCTCCAGT-3′ 5′-AGGTTGGTTAGGCAGGCACCTTTA-3′ |
※Underlined region indicates T7 promoter sequences.
TmRelish mRNA expression analysis after microbial challenge
qRT-PCR was conducted to examine the TmRelish mRNA induction profiles under microbial challenge. Healthy T. molitor larvae (10th–12th instar) were infected by injecting 1 µL each of E. coli (1 × 106 cells/µL), S. aureus (1 × 106 cells/µL), and/or C. albicans (5 × 104 cells/µL) into separate sets of larvae (n = 20). Tissue collection, total RNA extraction, cDNA synthesis, and qRT-PCR were carried out as described above. Tissue samples (fat body, hemocytes, gut, and Malpighian tubules) were collected from each set of infected larvae and the PBS-injected mock controls 3, 6, 9, 12, and 24 h post-infection. qRT-PCR was performed using on 20 μL reaction mixture with AccuPower® 2X GreenStar qPCR Master Mix (Bioneer, Korea) and specific primers. Relative TmRelish gene expression was calculated using the comparative CT method (2−ΔΔCT method)28.
TmRelish dsRNA production and RNAi efficiency
For synthesizing TmRelish double-stranded RNA (dsRNA), specific primers, containing T7 promoter sequences, were designed using the SnapDragon-long dsRNA design software (https://www.flyrnai.org/cgi-bin/RNAi_find_primers.pl). The primers were designed to amplify the 851 bp PCR product using AccuPower® Pfu PCR PreMix under the following conditions: denaturation at 95 °C for 2 min followed by 30 cycles of denaturation at 95 °C for 20 s, annealing at 56 °C for 30 s, and extension at 72 °C for 5 min. To synthesize dsTmRelish, PCR was carried out under the same conditions. The PCR product was purified using the AccuPrep® PCR Purification Kit (Bioneer, Korea), and the purified PCR product was used to synthesize dsTmRelish with the EZTM T7 High Yield in vitro Transcription Kit (Enzynomics, Korea), according to the manufacturer’s instructions. The final PCR product (1 μg, diluted 1:10) was mixed with 4 μL of 5X Transcription Buffer, 2 μL of 10X MgCl2, 2 μL of 100 mM DTT, 1 μL of RNase Inhibitor (40 U/μL), 1 μL each of 100 mM rATP, 100 mM rGTP, 100 mM rCTP, and 100 mM rUTP, and 1 μL of RNA polymerase. The mixture was incubated at 37 °C for 3 h and 25 °C for 1 h. The synthesized dsTmRelish was then gently mixed with one volume of 5 M ammonium acetate, kept on ice for 15 min, and centrifuged at 13,000 rpm at 4 °C for 10 min. The pellet, containing dsTmRelish was washed with 70%, 80%, and 99.9% ethanol and left to air dry. Thereafter, the pellet was resuspended in 30 μL of distilled water (Sigma, USA, W4502-1L). To synthesize double-stranded enhanced green fluorescent protein (dsEGFP) as a negative control, a 546 bp PCR product of EGFP, derived from the EGFP-C1 plasmid, was used as a template.
Subsequently, 1 μL of the synthesized dsTmRelish was injected into one set of larvae (n = 30), and 1 µL of dsEGFP was injected into another set of the same stage larvae (n = 30). To investigate the role of TmRelish in host-pathogen interactions, healthy T. molitor larvae were divided into three sets with two groups each (n = 30 in each group or n = 60 in each set). dsTmRelish was injected into one group of each set of larvae, whereas dsEGFP was injected into the other group, as a negative control. After TmRelish knockdown had been confirmed (24 h after dsRNA injection) in one group of T. molitor larvae, 1 × 106 cells/μL of E. coli and S. aureus and 5 × 104 cells/μL of C. albicans were injected into each larva, and larval survival was recorded every day for 10 days. The experiment was conducted at least three times independently to confirm the silencing of the transcripts.
Effect of TmRelish knockdown on AMP expression post-microbial challenge
In order to determine whether TmRelish knockdown affected AMP regulation, the expression profile of fourteen AMPs including members of the Tenecin family (TmTenecin-1 (TmTene1), TmTene2, TmTene3, and TmTene4)29–32; Attacin family (TmAttacin-1a (TmAtta1a), TmAtta1b, and TmAtta2)33; TmDefensin-1 (TmDef1); TmDef2; TmColeptericin-1 (TmCole1); TmCole2; TmCecropin-2 (TmCec2); and thaumatin-like protein (TmTLP1 and TmTLP2)34,35 was examined in TmRelish-silenced T. molitor larvae following microbial challenge. The immune tissues of the insect (fat body, hemocytes, gut, and MTs) were dissected 24 h post microbial challenge. Total RNA was extracted, and cDNA was synthesized as described above. qRT-PCR analysis was conducted using AMP-specific primers (Table 1). dsEGFP and PBS were used as the negative and mock controls, respectively.
Statistical analysis
Healthy 10th–12th instar larvae (approximately 2.4 cm in length) were randomly-selected for all experiments. All the developmental and tissue-specific profiling and microbial challenges tests were independently repeated three times (n = 20 per group). The survival experiments were repeated three times, with 30 larvae per group for each experiment. Values were reported as mean ± SE, and data were subjected to one-way analysis of variance (ANOVA) using SAS 9.4 (SAS Institute Inc., New South Wales, Australia). To evaluate the difference between groups (p < 0.05), Tukey’s multiple range tests were performed. The results for the survival assay were analyzed using a Kaplan-Meier plot (log-rank chi-square test) in Excel (http://www.real-statistics.com/survival-analysis/kaplan-meier-procedure/real-statistics-kaplan-meier/). Relative AMP gene expression was calculated using the comparative CT method (2−ΔΔCT method)28 and significant differences between dsTmRelish- and dsEGFP-injected groups were compared using Student’s t-test (p < 0.05).
Results
In silico analysis of TmRelish
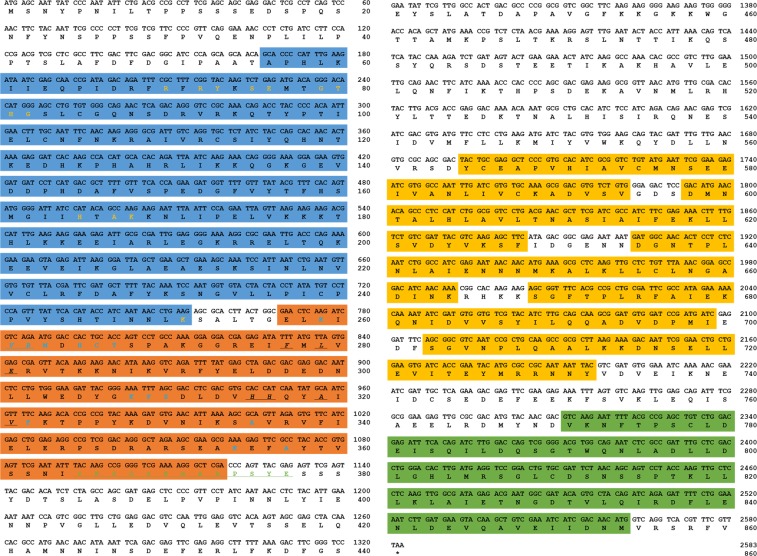
To identify Relish in the mealworm beetle T. molitor, we screened the RNA-seq data of T. molitor, using the TcRelish amino acid sequence as the query (GenBank: EEZ97717.1). Homology search against the RNA-seq database was performed using the local TBLASTN program. We identified a single Relish homolog (TmRelish) and studied the features of its sequence bioinformatically. The in silico-derived TmRelish and its deduced amino acid sequence were formatted using the Ultra-Edit program (https://www.ultraedit.com/) and are shown in Fig. 1. The full-length open reading frame (ORF) sequence of TmRelish, GenBank accession number MK863367, consists of 2,583 bp, encoding an 860 residue polypeptide. TmRelish contains an N-terminal Rel homology domain (RHD; A56–K251), an Ig-like, plexins, transcription factors domain (IPT domain; E257–R373), a death-like domain (DD; V771–M854), five ankyrin repeats (ANK; Y565–V594, D598–F628, D635–K664, S669–I699, and S703–Y732), and a nuclear localization signal (NLS, Y365KPGSKRARPSYE377). A putative DNA-binding motif (R68FRFRYKS75) was found at the N-terminus of the RHD domain. The Relish homolog of the Chinese mitten crab, Eriocheir sinensis (EsRelish), encodes a polypeptide of 1,214 amino acids and contains the typical RHD, an inhibitor kB (IkB)-like domain with six ankyrin repeats, and a DD36. However, EsRelish has been shown to contain two NLS sequences in contrast to the single NLS observed in TmRelish. In the decapod crustacean, Exopalaemon carinicauda, Relish consists of 2,141 bp and encodes a polypeptide of 660 amino acids, with an RHD domain and two NLS sequences, similar to EsRelish37. In the mosquito Aedes aegypti, AaRelish occurs as three alternatively spliced transcripts encoding separate proteins. The 3.9-kb transcript, which encodes an RHD and IkB-like domain, is the most predominant, showing similarities with DmRelish; the second most predominant transcript encodes an RHD and lacks the IkB-like domain; and the least predominant transcript lacks most of the RHD, but contains an intact IkB-like domain. A serine-rich region was identified immediately downstream of the NLS in TmRelish (S378–S380), similar to that found in DmRelish and unlike AaRelish, while both TmRelish and AaRelish contained the DD. TmRelish-associated conserved domains were compared at the amino acid level by multiple sequence alignment using ClustalX 2.1 (S1 Figure A, RHD; Figure B, IPT; Figure C, Ankyrin repeats; Figure D, DD). The DNA-binding motif (RFRFRYKS) showed higher identity compared to the other RHD regions (S1 Figure).
Figure 1.
Complete nucleotide and deduced amino acid sequence of Tenebrio molitor Relish (TmRelish) and its predicted amino acid sequence. Nucleotides and amino acids are numbered along the right margin. The InterProScan program (http://www.ebi.ac.uk/Tools/pfa/iprscan5/) was used to annotate the Rel homology domain (RHD), shown in blue (the DNA binding motif R68FRFRYKS75 has been underlined), the Ig-like, plexins, transcription factors domain (IPT) in orange, the death domain (DD) in green, and the nuclear localization signal (Y365KPGSKRARPSYE377) has been underlined in green. The five Ankyrin repeats in the TmRelish sequence have been shown in yellow.
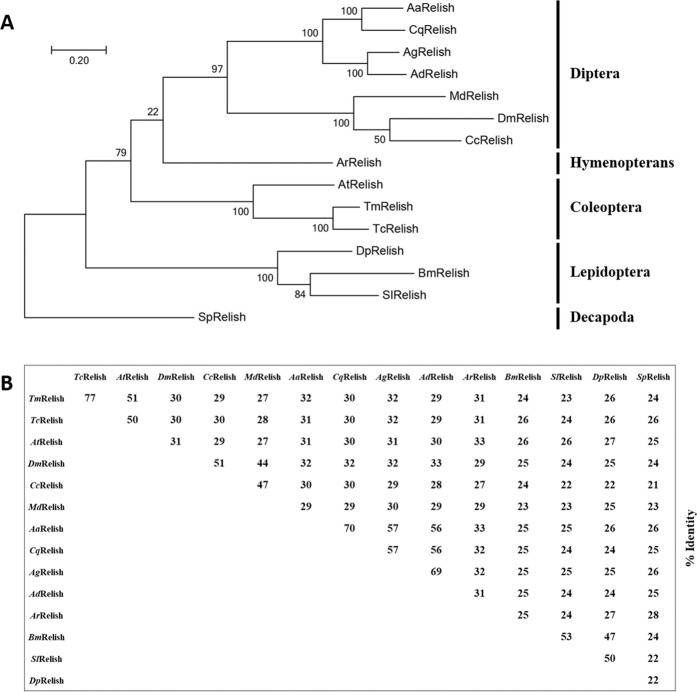
The deduced amino acid sequence of TmRelish was compared with Relish sequences from orthologous groups of insects to understand the evolutionary position of TmRelish and to predict functional divergence based on the features of its sequence. Phylogenetic analysis revealed that TmRelish showed close homology with TcRelish and clustered together under the order Coleoptera. Similarly, species belonging to the order Diptera (including the mosquito and Drosophila) were clustered together based on their Relish sequences but formed two distinct clades, one for mosquitoes [Aedes aegypti (AaRelish), Culex quinquefasciatus (CqRelish), Anopheles gambiae (AgRelish), and Anopheles darlingi (AdRelish)] and one for flies [Musca domestica MdRelish), Drosophila melanogaster (DmRelish), and Ceratitis capitata (CcRelish)]. The phylogenetic tree also revealed sequence divergence within insects and crustaceans, with the decapod Scylla paramamosain Relish (SpRelish) formed as an outgroup. Phylogenetic analysis of Relish sequences from T. molitor and other representative insects is shown in Fig. 2. Percent identity analysis showed that TmRelish had the highest identity with TcRelish, at 77%, followed by 51% identity with Aethina tumida Relish (AtRelish). Furthermore, TmRelish showed a maximum and minimum identity of 32% (with AgRelish and AaRelish) and 27% (with MdRelish), the hymenopteran and dipteran orthologous, respectively, and 23–26% identity with the lepidopteran orthologues.
Figure 2.
Phylogenetic tree (A) and percentage identity analysis (B) of the TmRelish and Relish protein sequences of representative insect species. Protein sequences were retrieved from NCBI, as described previously. The phylogenetic tree was constructed using the maximum likelihood method in MEGA7. The percentage of trees in which specific taxa clustered together is given by each branch. Bootstrap analysis values for 1,000 replicates are shown.
Developmental and tissue distribution of TmRelish
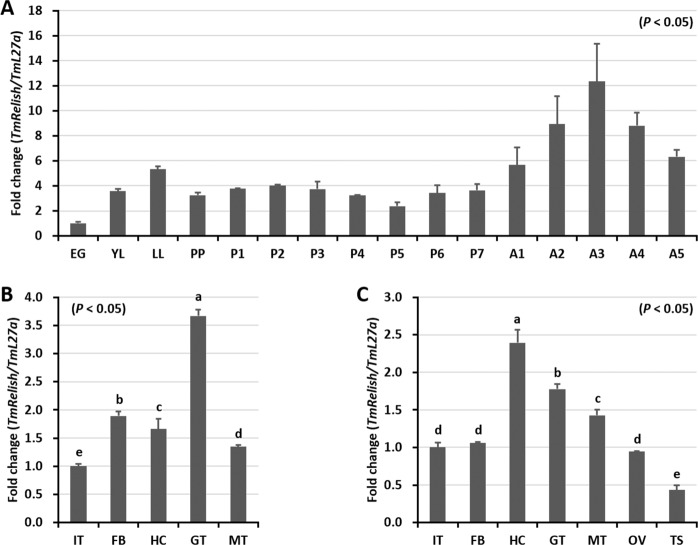
qRT-PCR was used to detect TmRelish mRNA expression in different developmental stages of T. molitor (Fig. 3A). TmRelish mRNA expression was detected in all developmental stages of the insect, with the highest level of expression observed in the adult stage. The expression of TmRelish mRNA in the larval and pupal stages did not change significantly. In the larval tissues examined, expression of TmRelish mRNA was highest in the gut, followed by the fat body, hemocytes, and MTs (Fig. 3B). In the 5-day-old adult tissues, TmRelish mRNA was expressed at the highest level in the hemocytes, followed by the gut and MTs. Furthermore, the TmRelish transcript was weakly detected in the integument, fat body, and ovary. The lowest transcription of TmRelish was observed in the testis (Fig. 3C). A higher level of TmRelish expression in the gut is expected as Relish acts as the NF-κB transcription factor in the Imd pathway, which is the master regulator of the gut response to microbiota. In the Drosophila gut, Relish affects host-microbiota interactions by altering the composition of 16S rRNA genes in gut associated microbes38.
Figure 3.
TmRelish mRNA expression in developmental stages (A) and tissues of Tenebrio molitor late-instar larvae (B) and 5-d-old adults (C) measured by qRT-PCR. (A) Relative expression levels of TmRelish mRNA in eggs (EG), young larvae (YL), late-instar larvae (LL), pre-pupae (PP), 1–7-day-old pupae (P1–P7), and 1–5-day-old adults (A1–A5) (B) Expression of TmRelish mRNA in the integument (IT), fat body (FB), hemocytes (HC), gut (GT), and Malpighian tubules (MT) of late-instar T. molitor larvae. (C) TmRelish mRNA expression in the integument (IT), fat body (FB), hemocytes (HC), gut (GT), Malpighian tubules (MT), ovary (OV), and testis (TS) of 5-day-old adults. T. molitor 60S ribosomal protein L27a (TmL27a) was used as an internal control to normalize RNA levels. Vertical bars represent the mean ± SE.
Time course analysis of TmRelish following microbial challenge
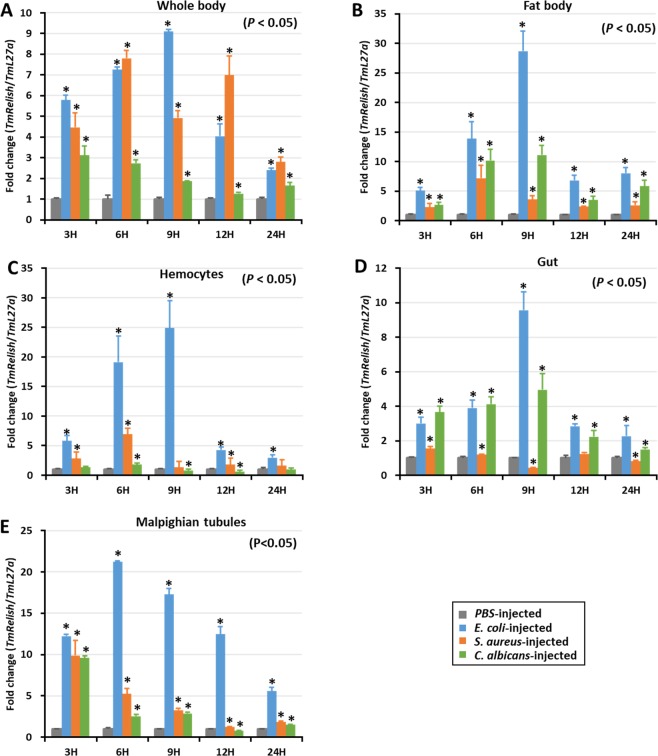
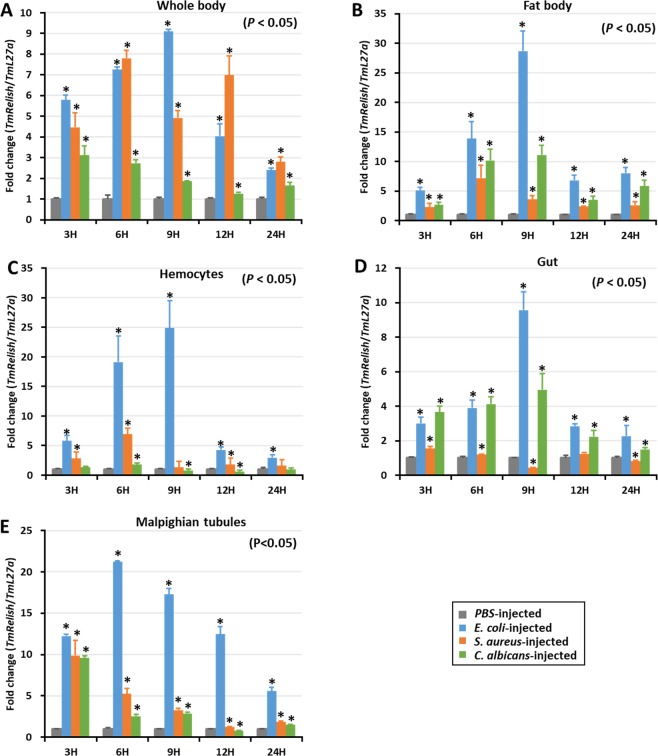
We observed the temporal expression of TmRelish mRNA in the fat body, hemocytes, gut, MTs, and the whole-body of T. molitor larvae challenged with E. coli, S. aureus, and C. albicans at various time points (3, 6, 9, 12, and 24 h) (Fig. 4). TmRelish expression levels were calculated relative to the expression (set to 1) of the mock control (PBS-injected). TmRelish mRNA expression was highly upregulated in the whole body of T. molitor larvae, challenged with E. coli, 9 h post-infection (Fig. 4A). The induction of TmRelish expression in the whole body of C. albicans-challenged T. molitor was also significant (p < 0.05), relative to the mock control; however, the expression level was lower than that of the E. coli and S. aureus-challenged groups. TmRelish expression was high 9 h post-infection with E. coli in the fat body (Fig. 4B), hemocytes (Fig. 4C), and gut tissue (Fig. 4D). TmRelish mRNA expression was non-significantly different in the hemocytes or gut tissue of S. aureus-challenged T. molitor larvae; a similar pattern of TmRelish expression was recorded in the MTs. Higher level of TmRelish mRNA expression was observed in the E. coli-challenged group, 6 h post-infection; declining at later time points (Fig. 4E). Hence, the time-course expression data suggests greater induction of TmRelish mRNA expression after E. coli challenge.
Figure 4.
TmRelish expression profiles of the whole body (A), fat body (B), hemocytes (C), gut (D), and Malpighian tubules (E) after Escherichia coli, Staphylococcus aureus, and Candida albicans infection, measured by qRT-PCR. T. molitor 60S ribosomal protein L27a (TmL27a) was used as an internal control. Expression levels of TmRelish in PBS-injected mock controls was set to 1. Vertical bars represent the mean ± SE (n = 20). ‘*’ indicates significant difference (p < 0.05).
TmRelish gene knockdown and T. molitor larval survival
Temporal induction of TmRelish prompted us to investigate its role in the immune response to microbial challenge. To determine whether TmRelish was involved in antibacterial or antifungal defense against E. coli and S. aureus or C. albicans, respectively, we depleted TmRelish through RNAi treatment. We found 83% knockdown of TmRelish expression upon RNAi treatment, compared to the EGFP injected controls on the third day post-dsRNA injection (Fig. 5A). Upon TmRelish knockdown, E. coli, S. aureus, and C. albicans were used to challenge T. molitor larvae and the survivability was monitored for 10 days. The percent survival of TmRelish-silenced larvae significantly dropped (90% mortality) in the E. coli challenged individuals (Fig. 5B). Survival of TmRelish knockdown larvae also reduced upon S. aureus and C. albicans challenge to 87% (Fig. 5C) and 80% (Fig. 5D), respectively, when compared to the EGFP dsRNA injected control. Taken together, the results suggested the requirement of TmRelish for survival of T. molitor larvae against E. coli infections. TmRelish could also be critical in conferring immunity against the gram-positive bacteria S. aureus and the fungus C. albicans.
Figure 5.
RNAi silencing efficiency of TmRelish mRNA in T. molitor larvae and survival in the 10 days following microbial challenge. qRT-PCR-based estimation of TmRelish knockdown relative to EGFP control, 3 days post-dsRNA injection (A). Effects of TmRelish mRNA knockdown on the survival of Tenebrio molitor larvae post Escherichia coli (B), Staphylococcus aureus (C), and Candida albicans (D) infection. dsEGFP-treated groups infected with the same microbes were used as negative controls. Survival was monitored for 10 d. The experiment was performed thrice with similar results. ‘*’ indicates significant differences between dsTmRelish and dsEGFP-treated groups (p < 0.05).
Effect of TmRelish knockdown on AMP gene expression
The reduced survival of TmRelish knockdown T. molitor larvae after being challenged with E. coli and other microorganisms suggested a promiscuous role of TmRelish in conferring immunity against the pathogens. We hypothesized that TmRelish depletion would affect immunocompetent tissues of the insect to produce antimicrobial factors in response to microbial insults. We therefore investigated the expression of fourteen T. molitor AMP genes in the TmRelish knockdown larval fat body, hemocytes, gut, and MTs post-E. coli, -S. aureus, and -C. albicans challenge.
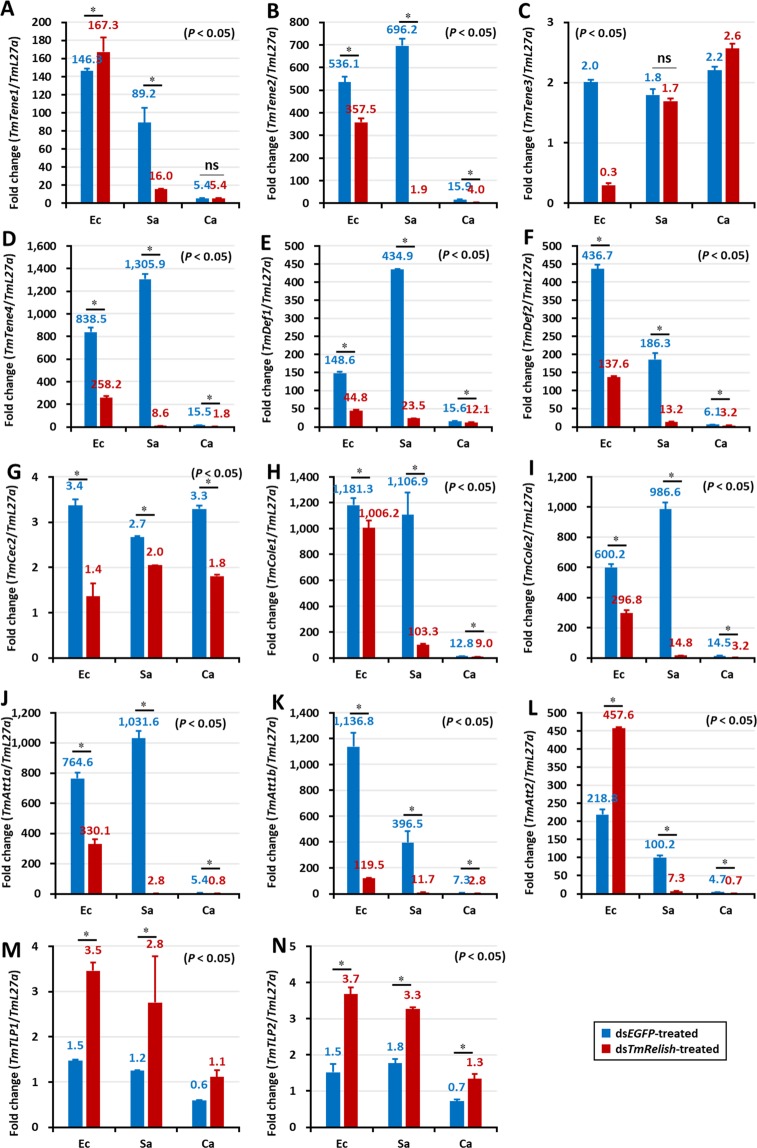
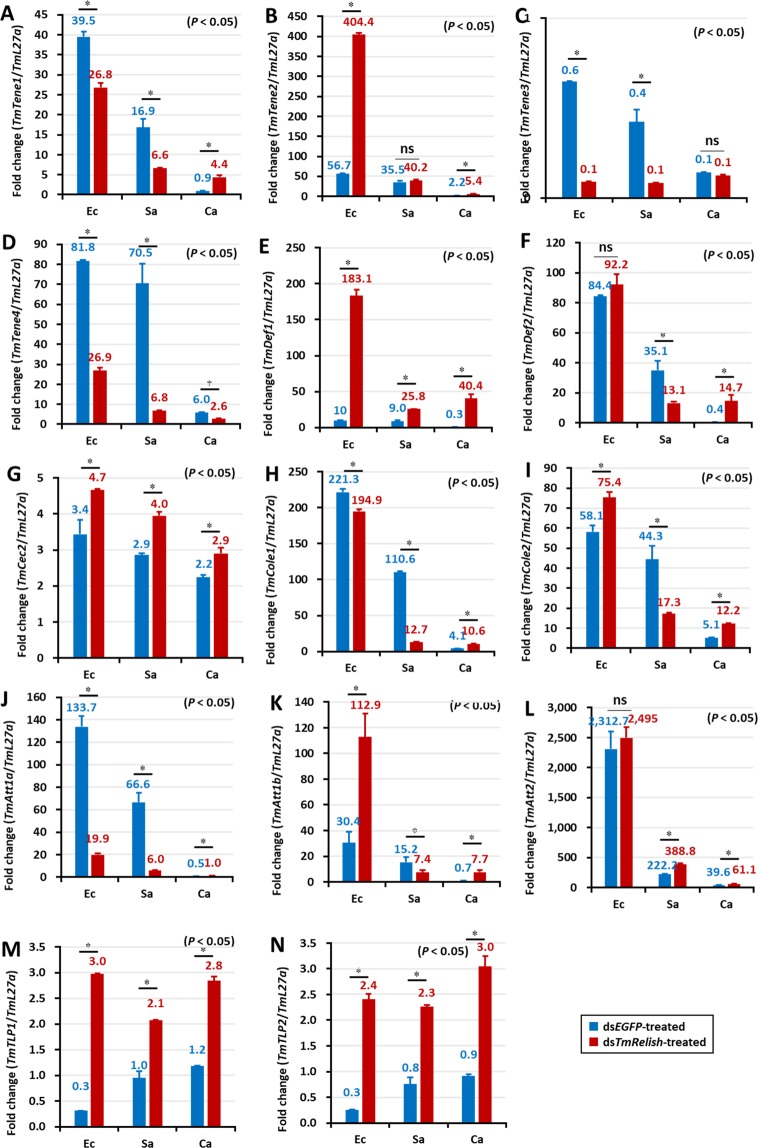
In the larval fat body of dsEGFP-treated cohorts, the mRNA expression levels of eleven AMP genes showed an increase after microbial challenge (Fig. 6). In TmRelish silenced larvae, the mRNA expression of TmTene2 and -4 (Fig. 6B,D); TmDef1 and -2 (Fig. 6E,F); TmCec2 (Fig. 6G); TmCole1 and -2 (Fig. 6H,I); and TmAtta1a and -1b (Fig. 6J,K) was downregulated. Conversely, the expression of TmTene1 (Fig. 6A) and TmAtta2 (Fig. 6L) was higher in the dsTmRelish-injected group compared to the dsEGFP-treated control group, after E. coli challenge. However, in S. aureus infected T. molitor larvae the expression of TmTene1 and TmAtta2 was found to be downregulated (Fig. 6A,L). The antimicrobial response of the larval fat body to S. aureus infection in dsEGFP-treated larvae was strikingly higher compared to the E. coli and C. albicans infected groups. Notably, AMPs such as TmTene2 and -4 (Fig. 6B,D), TmDef1 (Fig. 6E), TmCole2 (Fig. 6I), and TmAtta-1a (Fig. 6J) were found to show higher expression after S. aureus infection. Further, TmTene3 expression in the fat body of T. molitor larvae was not affected in TmRelish knockdown individuals, following S. aureus or C. albicans challenge (Fig. 6C). The antifungal AMPs namely TmTLP1 (Fig. 6M) and TmTLP2 (Fig. 6N) were found to be increased in TmRelish knockdown larvae, after microbial infection. The results are in agreement with a previous study on the toll pathway NF-κB factor, TmDorsal isoform 2 (TmDorX2)39. Thus, the expression of nine, eleven, and ten AMPs was downregulated in the dsTmRelish-injected group after systemic exposure to E. coli, S. aureus, and C. albicans, respectively, suggesting a requirement for TmRelish in regulating AMP gene expression in the fat body.
Figure 6.
AMP expression levels in the TmRelish-knockdown Tenebrio molitor larval fat body upon Escherichia coli (Ec), Staphylococcus aureus (Sa), and Candida albicans (Ca) infection. Healthy larvae (10th–12th instar) were injected with dsTmRelish and infected with a suspension of E. coli, S. aureus, or C. albicans on the third day, post-dsRNA injection. PBS-injected larvae were used as controls. After 24 h, the fat body tissue was dissected. Expression level of the AMP genes TmTenecin-1 (A), TmTenecin-2 (B), TmTenecin-3 (C), TmTenecin-4 (D), TmDefensin1 (E), TmDefensin2 (F), TmCecropin-2 (G), TmColeoptericin-1 (H), TmColeoptericin-2 (I), TmAttacin1a (J), TmAttacin-1b (K), TmAttacin-2 (L), TmThaumatin-like protein-1 (M), and TmThaumatin-like protein-2 (N) was measured using qRT-PCR and compared with the dsEGFP-treated groups. dsEGFP was used as a negative control and TmL27a as an internal control. The numbers above the bars indicate AMP expression levels. All experiments were repeated thrice, with similar results. Statistical analysis was performed using Student’s t-test (p < 0.05); ns: not significant.
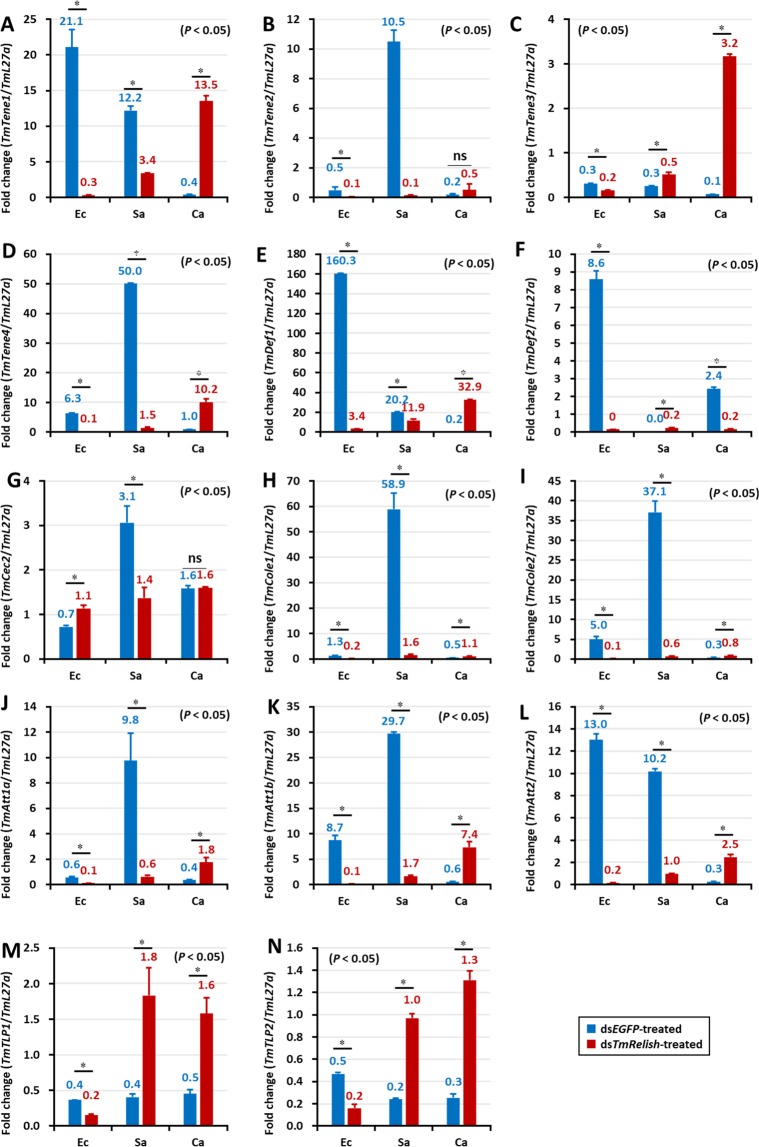
Interestingly, the dsEGFP group larvae showed lower expression of AMPs in the hemocytes compared to the fat body, gut, and MTs following E. coli, S. aureus, and C. albicans challenge. Moreover, after E. coli and S. aureus challenge, most of the AMP genes were upregulated in the control groups, while AMP genes were upregulated in response to C. albicans in the dsTmRelish larvae (Fig. 7). Furthermore, the expression of TmTene1, -2, and -4 (Fig. 7A,B,D); TmDef1 (Fig. 7E); TmCole1 and -2 (Fig. 7H,I); and TmAtta1a, -1b, and -2 (Fig. 7J–L) was downregulated in the dsTmRelish-treated groups in comparison with those in the dsEGFP-treated groups, following E. coli and S. aureus challenges. In contrast, mRNA expression of all AMPs, except for TmTene2 (Fig. 7B), TmDef2 (Fig. 7F), and TmCec2 (Fig. 7G), was upregulated in the TmRelish knockdown groups compared to those in the dsEGFP group, following C. albicans challenge (Fig. 7). These results suggested that TmRelish is not required for eliciting an antimicrobial immune response to C. albicans infection39.
Figure 7.
AMP expression levels in TmRelish-knockdown Tenebrio molitor larval hemocytes upon Escherichia coli (Ec), Staphylococcus aureus (Sa), and Candida albicans (Ca) infection on the third day post-TmRelish silencing. The AMP genes used for analysis include TmTenecin-1 (A), TmTenecin-2 (B), TmTenecin-3 (C), TmTenecin-4 (D), TmDefensin-1 (E), TmDefensin 2 (F), TmCecropin-2 (G), TmColeoptericin-1 (H), TmColeoptericin-2 (I), TmAttacin-1a (J), TmAttacin-1b (K), TmAttacin-2 (L), TmThaumatin-like protein1 (M), and TmThaumatin like protein2 (N). dsEGFP was used as a negative control and TmL27a was used as an internal control. Numbers above the bars indicate AMP mRNA expression levels. All experiments were repeated thrice with similar results. Statistical analysis was performed using Student’s t-tests (p < 0.05) and ns: not significant.
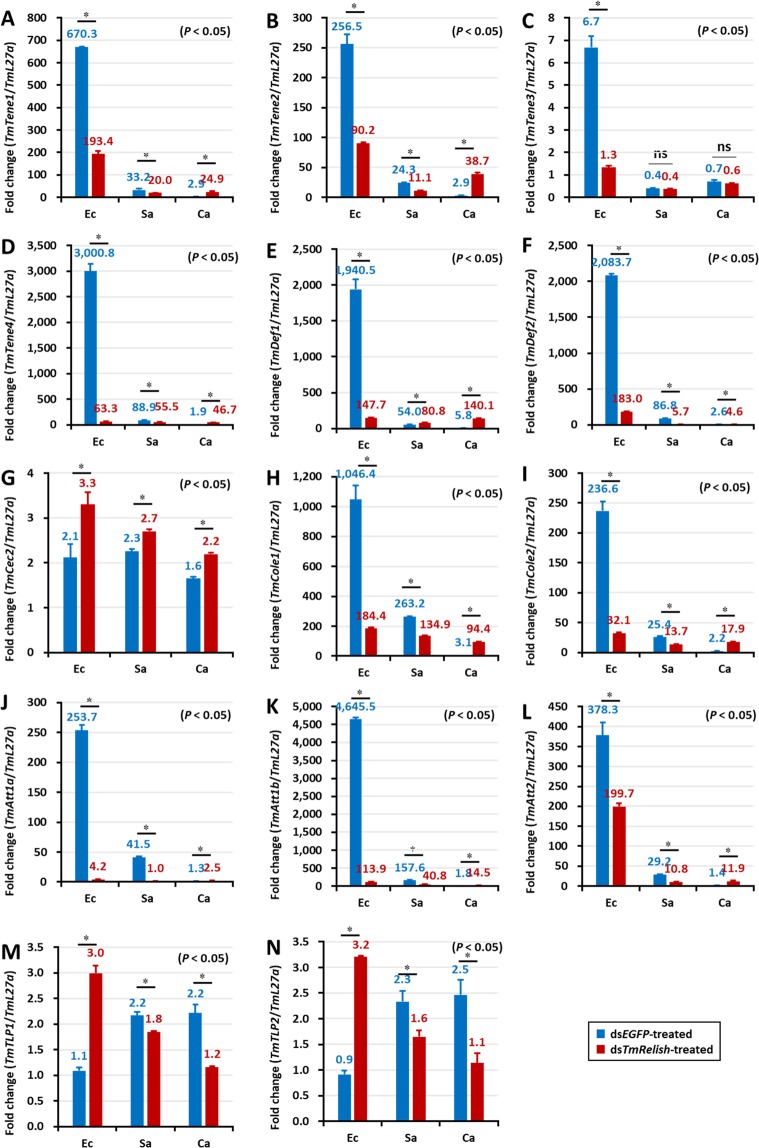
In the gut of dsEGFP-treated control larvae, TmRelish strongly enhanced E. coli-mediated induction of eleven AMP genes including TmTene1, -2, -3, and -4 (Fig. 8A–D); TmDef1 and -2 (Fig. 8E,F); TmCole1 and -2 (Fig. 8H,I); and TmAtta1a, -1b, and -2 (Fig. 8J–L). This effect was dramatically reduced in TmRelish silenced individuals (Fig. 8). In the case of S. aureus infection, TmRelish was required for the induction of TmTene1, -2, -3, and -4 (Fig. 8A–D); TmDef1 and -2 (Fig. 8E,F); TmCole1 and -2 (Fig. 8H,I); TmAtta1a, -1b, and -2 (Fig. 8J–L), and TmTLP1 and -2 (Fig. 8M,N) in the control groups. Further, the AMP genes upregulated after C. albicans infection were not affected in TmRelish silenced individuals. Notably, eleven AMP genes comprising TmTene1, -2, -3, and -4 (Fig. 8A–D); TmDef1 and -2 (Fig. 8E,F); TmCec2 (Fig. 8G); TmCole1 and -2 (Fig. 8H,I); and TmAtta1a, -1b, and -2 (Fig. 8J–L) were upregulated in dsTmRelish-injected cohorts. This is relevant, as in an earlier study we have found downregulation of all AMP genes in the dsTmDorX2-treated groups, following C. albicans exposure. This suggests the importance of TmDorX2 in the immune response to C. albicans in the gut39. In the MTs, following E. coli and S. aureus infection, five AMPs (TmTene1, -3, and -4; TmAtta1a; TmCole1) were slightly downregulated after dsTmRelish injection (Fig. 9A,C,D,J,H, respectively). In addition, TmCec2 induction was higher in the MTs of E. coli, S. aureus, and C. albicans-challenged TmRelish knockdown T. molitor larval groups (Fig. 9G). Additionally, knocking down TmRelish by RNAi led to reduced expression of eleven, ten, eleven, and seven AMP genes, after S. aureus infection, in the fat body, hemocytes, gut, and MTs, respectively. Collectively, our results demonstrate that TmRelish promotes TmTene2, TmTene4, TmDef2, TmCole1, TmCole2, TmAtta1a, and TmAtta1b expression in response to E. coli and S. aureus infections in the larval fat body and gut. We created a scheme summarizing our findings on the functional characterization of TmRelish (Fig. 10), swhich shows that E. coli infection caused highest mortality in T. molitor larvae after the knockdown of TmRelish, a component downstream of the Imd pathway, in the fat body and gut. Ten and eleven AMPs were highly upregulated in the fat body and gut tissues following E. coli infection, indicating a defense response in the host contributing towards the survival of the larvae. However, upon TmRelish knockdown, the expression of these AMPs significantly declined leading to increased host susceptibility to E. coli infection.
Figure 8.
AMP expression level in TmRelish-silenced Tenebrio molitor gut tissue upon Escherichia coli, Staphylococcus aureus, and Candida albicans infection. qRT-PCR expression profiles of TmTenecin-1 (A), TmTenecin-2 (B), TmTenecin-3 (C), TmTenecin-4 (D), TmDefensin1 (E), TmDefensin2 (F), TmCecropin2 (G), TmColeoptericin-1 (H), TmColeoptericin-2 (I), TmAttacin-1a (J), TmAttacin-1b (K), TmAttacin-2 (L), TmThaumatin like protein-1 (M), and TmThaumatin like protein-2 (N). dsEGFP was used as the negative control and TmL27a was used as an internal control. All experiments were performed at least thrice, and statistical analysis was performed using Student’s t-test (p < 0.05); ns: not significant.
Figure 9.
Expression levels of AMPs in TmRelish-silenced Tenebrio molitor Malpighian tubules upon Escherichia coli, Staphylococcus aureus, and Candida albicans infection. qRT-PCR expression profiles of TmTenecin-1 (A), TmTenecin-2 (B), TmTenecin-3 (C), TmTenecin-4 (D), TmDefensin-1 (E), TmDefensin-2 (F), TmCecropin-2 (G), TmColeoptericin-1 (H), TmColeoptericin-2 (I), TmAttacin-1a (J), TmAttacin-1b (K), TmAttacin-2 (L), TmThaumatin like protein-1 (M), and TmThaumatin like protein-2 (N). dsEGFP was used as the negative control and TmL27a was used as an internal control. All experiments were performed at least thrice, and statistical analysis was performed using Student’s t-tests (p < 0.05); ns: not significant.
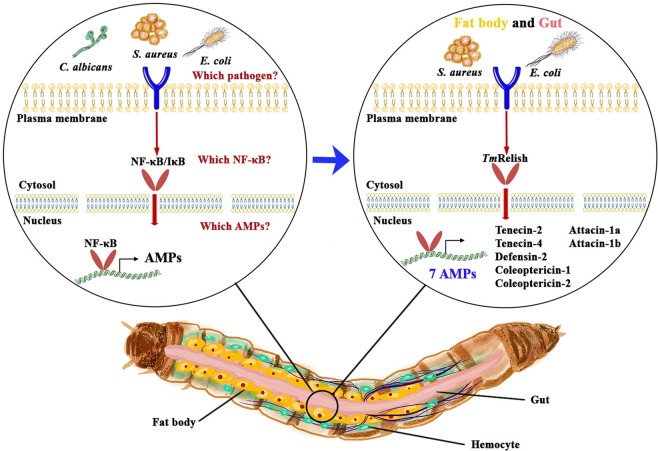
Figure 10.
A schematic representation of the humoral immunity pathway positively regulated by Relish in the T. molitor fat body and gut, upon Escherichia coli and Staphylococcus aureus infections, but not Candida albicans infection. Seven AMPs were downregulated in the TmRelish knockdown group, indicating that Relish is required for survival of the host.
Discussion
Beetles constitute 40% of all recognized arthropod species, and their success has been linked to their adaptive plasticity, which enables them to inhabit a variety of environmental niches. This success is unconventionally linked to their robust innate immune system40,41. Although genetic studies have elucidated the intricacies of innate immunity in D. melanogaster, little is known about the biochemical mechanisms of the innate immune response. The genomes and transcriptomes of beetles such as T. castaneum, T. molitor, and Holotrichia diomphalia have been studied to understand the host defense molecules that regulate immune reactions against a plethora of pathogens.
A genome-wide study of the T. castaneum innate immune system revealed information regarding pathogenic and non-pathogenic stress adaptation, and suggested the presence of crosstalk between the immune and stress responses42. The study also provided a data resource, which could be used for the discovery and functional characterization of genes involved in innate immunity in other beetle species. T. molitor has been shown to attack invading pathogens via transcriptional regulation of AMPs via the toll and Imd signaling cascades7,32. In beetles, the Toll pathway is activated by a serine protease cascade, which is activated upon recognition of gram-positive bacteria and fungi, whereas the Imd pathway directly senses pathogens via the peptidoglycan recognition proteins (PRRs) and is controlled by Imd adapter proteins30,43. In invertebrates such as beetles, the innate immune signaling components of the Toll pathway are comparatively better understood than those of the Imd pathway. There is, therefore, ample scope for expanding our understanding of the Imd pathway in relation to its modulation of host-pathogen interactions and immune surveillance in the host via effector AMPs.
The present study was conducted to determine the role of the Rel-homology domain (RHD) protein, Relish (downstream of the Imd protein), in T. molitor innate immunity and AMP gene regulation. Relish is an important member of the NF-κB transcription factor family and translocates to the nucleus upon the detection of gram-negative bacteria to elicit effector AMP functions. The involvement of Relish in AMP expression has been noted in other invertebrates, including Drosophila36,44. TmRelish contains RHD, IPT, ANK, and DD domains, and an NLS was identified in the C-terminal portion of the IPT domain. RHD is a characteristic of the eukaryotic Rel protein family and comprises two structural domains: an N-terminal DNA binding domain and a dimerization domain with a C-terminal immunoglobulin-like fold. In addition, TmRelish has an arginine (R)/lysine (K)-rich NLS that mediates the translocation of the protein to the nucleus45. Furthermore, TmRelish is a longer form of Relish, as the shorter form contains only the RHD and IPT domains. The short and long forms of Relish are more common in crustaceans such as shrimps; the giant tiger prawn, Penaeus monodon, and the giant freshwater prawn, Macrobrachium rosenbergii, encode long forms of Relish36,46, whereas shorter isoforms have been identified in the Chinese shrimp, Fenneropenaeus chinensis, and the white leg shrimp, Litopenaeus vannamei47. Among insects, both the long and short forms of Relish have been identified in Anopheles gambiae and are believed to be formed by the alternative splicing of Relish248. In the mosquito, Aedes aegypti, both isoforms of Relish1 were found to be the long forms that activate the Toll-antifungal pathway and induce the expression of AMPs such as diptericin and drosomycin. Furthermore, phylogenetic analysis revealed that TmRelish clustered with the beetle orthologs, supporting their evolutionary position. Interestingly, dipteran Relish separated into two clades: one for mosquitoes and the other for flies. As Relish is essential for mounting an appropriate humoral response against pathogens recognized as non-self via the Imd pathway, it is unlikely that positive selection pressures would have caused the adaptive evolution of the Relish complex, as has been observed in the termite, Nasutitermes49.
We also examined the expression pattern of TmRelish mRNA during development, and observed significantly higher mRNA expression in the adults than in the larvae or pupae. Cross-talk is known to exist between nuclear hormone receptors and innate immunity pathways, suggesting that the juvenile hormone (JH) and 20-hydroxy-ecdysone (20E; steroid hormone) modulate immune responses50. Furthermore, JH can act as an immune suppressor whereas 20E can induce AMP expression51. The increased TmRelish expression observed in adult T. molitor can be attributed to low JH levels. Furthermore, we speculate that the increased expression of the extracellular matrix protein, tenebrin, in the late-instar larvae is due to enhanced TmRelish mRNA expression. Tenebrin mRNA expression was shown to be positively regulated by 20E52, suggesting that increased 20E secretion could be related to high TmRelish mRNA expression levels in late-instar larvae.
Here, we have reported the biological functions of TmRelish in the absence of infection in different tissues. Higher TmRelish mRNA expression was observed in immune tissues such as the gut, hemocytes, fat body, and MTs of both T. molitor late-instar larvae and 5-day-old adults. In fact, Relish has been shown to be indispensable for eliciting humoral responses via AMP induction in Drosophila cuticles and epithelia, including the respiratory and digestive tracts, the MTs, and reproductive organs53,54. However, TmRelish mRNA expression was not enhanced in the ovary or testis of T. molitor adults, in our study. The mRNA of Relish in the freshwater prawn, M. rosenbergii (MrRelish), was also found to be highly expressed in the hemocytes and intestinal tissue36. Additionally, a recent study of the tobacco cutworm Spodoptera litura Relish (SlRelish) has revealed a strong expression in the fat body and hemolymph55. Hemocytes and gut compartments are sites of systemic and local inflammatory reactions; thus, the enhanced expression of TmRelish in these tissues suggests a role in inflammatory reactions56. Upon infection, the Imd pathway in the Drosophila gut regulates the shedding of enterocytes into the lumen via Relish, leading to the expression of AMPs to combat the invading pathogenic microorganisms57,58. Furthermore, Drosophila gut morphology is known to be largely influenced by Imd pathway genes, including the downstream component Relish38. In the present study, TmRelish mRNA was upregulated in the fat body, hemocytes, gut, and MTs of T. molitor late-instar larvae after E. coli challenge. The highest level of TmRelish mRNA expression was observed 9 h post-infection in the fat body, hemocytes, and gut tissue, and 6 h post-infection in the MTs. Relish is a downstream effector of the Imd signaling cascade, which induces AMP expression in response to microbe-associated molecular patterns (MAMPs), such as meso-diaminopimelic acid (DAP)-type peptidoglycan (PGN) which is found in most gram-negative and a few gram-positive (Bacillus and Listeria) bacteria. Our data demonstrated that TmRelish expression was induced in response to E. coli and S. aureus infection in a manner similar to that of Anopheles gambiae Relish2, which responds to both gram-negative and gram-positive bacteria48. TmRelish mRNA expression levels were lower in the fat body, hemocytes, and gut of the S. aureus-infected groups compared with the E. coli-infected groups, suggesting that E. coli elicit stronger TmRelish expression than S. aureus. Our results agree with those from an earlier study examining the induction of Relish homologs in the silkworm, Bombyx mori, in response to E. coli infection18. Consistently, SlRelish expression was exclusively induced by E. coli55. The Relish transcripts of crustaceans such as the Chinese shrimp, Fenneropenaeus chinensis, and the pearl oyster, Pinctada fucata, have also been shown to be upregulated in response to infection with the gram-negative bacteria Vibrio anguillarum and Vibrio alginolyticus, respectively59. In conclusion, TmRelish is involved in antibacterial immune defense in T. molitor.
Loss of certain Relish-dependent target genes, such as AMP genes, has important consequences on humoral immunity. Knocking down the transcriptional activity of TmRelish mRNA suppresses expression of AMP genes, considerably weakening the host defense. In this study, survival results showed that when TmRelish was silenced, an early, highly significant mortality rate was observed in the E. coli-challenged larvae compared to the S. aureus and C. albicans-infected cohorts. This is consistent with the fact that Relish is essential for humoral defense against gram-negative bacteria and that short-term starvation prior to immune challenge increases survival. Furthermore, the knockdown of A. aegypti Relish2 was shown to dramatically increase the mortality of mosquitoes following infection with gram-positive and -negative bacteria60,61. We showed that E. coli was capable of killing almost 90% of TmRelish knockdown larvae. Thus, TmRelish appeared to be essential against E. coli infection. Further, SlRelish-depleted insects were highly susceptible to E. coli insult55. Although larval mortality was significant in the S. aureus- and C. albicans-treated groups, the rate was lower than that in the E. coli group.
The production of AMPs is an evolutionarily conserved mechanism, triggered when the cleaved RHD of Relish translocates to the nucleus. AMP expression was shown to be induced in the fat body, hemocytes, and gut tissues of Drosophila54,62. Analyzing the expression of T. molitor AMP genes in larvae with or without TmRelish knockdown revealed that expression levels of AMPs decreased upon silencing TmRelish during infections; TmRelish can therefore be proposed to be a positive regulator of AMPs, and TmRelish-silenced larvae are more susceptible to microbial infections compared to controls. Although we observed decreased levels of several AMPs in dsTmRelish-treated larvae following S. aureus, TmRelish depletion led to mild but significant mortality. In a previous study, the mRNA levels of TmTene1, TmTene2, TmTene4, TmDef2, TmCole1, TmCole2, TmAtta1a, TmAtta1b, and TmAtta2 were found to be significantly reduced in T. molitor following silencing of immune deficiency (TmIMD), an adapter molecule upstream of the Imd pathway, upon exposure to E. coli63. Similarly, upon E. coli challenge, expression of TmTene3, TmDef1, and the AMPs listed above, was downregulated in the gut of TmRelish-depleted larvae. Global expression analysis of the gut epithelium of Drosophila following oral infection with gram-negative bacteria revealed that the Imd pathway, and not the Toll pathway, is involved in eliciting a robust immune response64. Several AMP genes were found to be expressed at high levels in the fat body of the TmRelish non-knockdown groups; however, TmRelish knockdown downregulated the expression of TmTene2, TmTene3, TmTene4, TmDef1, TmDef2, TmCec2, TmCole1, TmCole2, TmAtta1a, and TmAtta1b. Eliminating TmRelish had profound consequences on Imd pathway activation and AMP expression in both the fat body and the gut, in response to E. coli, suggesting that these immunocompetent tissues are a vital part of the immune response in terms of AMP production following E. coli infection. When evaluating mortality rate, upon S. aureus infection, the role of TmRelish in different tissues is unexpected. However, new evidence supporting the role of the Imd pathway against S. aureus in the Drosophila gut suggests involvement of the Imd pathway in gram-positive clearance. In the present study, S. aureus infection induced TmTene1, TmTene4, TmCole1, TmCole2, TmAtta1a, and TmAtta1b in the fat body, hemocyte, gut, and MT of dsEGFP controls, while all six AMP genes were moderately downregulated in TmRelish knockdown larvae. Interestingly, eleven of the fourteen AMP genes were negatively regulated in the gut and hemocytes, but not the fat body, by TmRelish knockdown upon C. albicans challenge, suggesting the crosstalk between TmDorX2-Toll pathway in regulating AMP expression39. In addition to the gut, the fat body, hemocytes, and other epithelial tissues such as MTs (nephridia or kidney analogues) also play important roles in immune defense by producing AMPs65,66. A recent transcriptional analysis of Zophobas morio (Coleoptera: Tenebrionidae) showed that the fat body and MTs are versatile tissues and share important functions, such as immunity, detoxification, nitrogen metabolism, and eye pigmentation. Preliminary studies using Drosophila MTs suggested that Imd component genes lead to the induction of AMPs in response to microbial insults. Furthermore, immune response and AMP expression in the MTs of D. melanogaster have been associated with developmental regulation67. In the present study, only five AMP genes were downregulated in the TmRelish knockdown larvae infected with E. coli and S. aureus.
Conclusions
We identified a Relish homologue, which was expressed in all immune tissues, with the highest expression level being observed in the gut, in the coleopteran beetle, T. molitor. TmRelish expression increased during the early hours of E. coli infection in the hemocytes, gut, fat body, and MTs, with the highest level of expression seen in the gut tissue of the larvae. Loss of function studies, using RNAi directed against TmRelish, showed significant reduction in survival of the E. coli-, S. aureus-, and C. albicans-challenged larvae. Higher mortality was observed in the TmRelish knockdown and E. coli- infected groups than in the S. aureus- and C. albicans-infected groups, suggesting that the target gene is involved in defense against gram-negative bacteria by inducing the expression of nine AMP genes in both the fat body and the gut. Additionally, ten AMP genes were found to be downregulated in the larval fat body of T. molitor, in the TmRelish RNAi-treated groups, in response to S. aureus, indicating that TmRelish plays an essential role in antibacterial immune response of by larval fat body and gut, in response to E. coli and S. aureus infections.
Supplementary information
Author contributions
Han Y.S. and Jo Y.H. conceived and designed the experiments; Keshavarz M., Park K.B., Ko H.J., Kim C.E., and Edosa T.T. performed the experiments; Jo Y.H., Patnaik B.B., and Keshavarz M. analyzed the data; Han Y.S. and Lee Y.S. contributed reagents/materials/analysis tools; Han Y.S., Keshavarz M., Patnaik B.B., and Jo Y.H. wrote the manuscript; Lee Y.S. revised the manuscript.
Competing interests
The authors declare no competing interests.
Footnotes
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
These authors contributed equally: Maryam Keshavarz and Yong Hun Jo.
Change history
4/21/2020
An amendment to this paper has been published and can be accessed via a link at the top of the paper.
Supplementary information
is available for this paper at 10.1038/s41598-020-61157-1.
References
- 1.Rowley AF, Powell A. Invertebrate immune systems–specific, quasi-specific, or nonspecific? J. Immunology. 2007;179:7209–7214. doi: 10.4049/jimmunol.179.11.7209. [DOI] [PubMed] [Google Scholar]
- 2.Nazario-Toole, A. E. & Wu, L. P. In Advances in Insect Physiology Vol. 52 35–82 (Elsevier, 2017).
- 3.Dubovskiy I, Kryukova N, Glupov V, Ratcliffe N. Encapsulation and nodulation in insects. Invertebrate Survival J. 2016;13:229–246. [Google Scholar]
- 4.Satyavathi VV, Minz A, Nagaraju J. Nodulation: an unexplored cellular defense mechanism in insects. Cell. Signal. 2014;26:1753–1763. doi: 10.1016/j.cellsig.2014.02.024. [DOI] [PubMed] [Google Scholar]
- 5.Lemaitre B, Reichhart J-M, Hoffmann JA. Drosophila host defense: differential induction of antimicrobial peptide genes after infection by various classes of microorganisms. Proc. Natl Acad. Sci. 1997;94:14614–14619. doi: 10.1073/pnas.94.26.14614. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Cerenius L, Söderhäll K. The prophenoloxidase‐activating system in invertebrates. Immunological Rev. 2004;198:116–126. doi: 10.1111/j.0105-2896.2004.00116.x. [DOI] [PubMed] [Google Scholar]
- 7.Jo YH, et al. TmCactin plays an important role in Gram-negative and-positive bacterial infection by regulating expression of 7 AMP genes in Tenebrio molitor. Sci. Rep. 2017;7:46459. doi: 10.1038/srep46459. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Shokal U, Eleftherianos I. Evolution and function of thioester-containing proteins and the complement system in the innate immune response. Front. immunology. 2017;8:759. doi: 10.3389/fimmu.2017.00759. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ferrandon D, Imler JL, Hetru C, Hoffmann JA. The Drosophila systemic immune response: sensing and signalling during bacterial and fungal infections. Nat. Rev. Immunol. 2007;7:862–874. doi: 10.1038/nri2194. [DOI] [PubMed] [Google Scholar]
- 10.Imler, J.-L. & Bulet, P. In Mechanisms of epithelial defense Vol. 86 1–21 (Karger Publishers, 2005).
- 11.Williams MJ, Rodriguez A, Kimbrell DA, Eldon ED. The 18-wheeler mutation reveals complex antibacterial gene regulation in Drosophila host defense. EMBO J. 1997;16:6120–6130. doi: 10.1093/emboj/16.20.6120. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Paquette N, et al. Caspase-mediated cleavage, IAP binding, and ubiquitination: linking three mechanisms crucial for Drosophila NF-kappaB signaling. Mol. Cell. 2010;37:172–182. doi: 10.1016/j.molcel.2009.12.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Oeckinghaus, A. & Ghosh, S. The NF-κB family of transcription factors and its regulation. Cold Spring Harbor perspectives in biology, a000034 (2009). [DOI] [PMC free article] [PubMed]
- 14.Hedengren M, et al. Relish, a central factor in the control of humoral but not cellular immunity in Drosophila. Mol. Cell. 1999;4:827–837. doi: 10.1016/S1097-2765(00)80392-5. [DOI] [PubMed] [Google Scholar]
- 15.Erturk-Hasdemir D, et al. Two roles for the Drosophila IKK complex in the activation of Relish and the induction of antimicrobial peptide genes. Proc. Natl Acad. Sci. USA. 2009;106:9779–9784. doi: 10.1073/pnas.0812022106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zhong J, et al. Repeated ozone exposure exacerbates insulin resistance and activates innate immune response in genetically susceptible mice. Inhal. Toxicol. 2016;28:383–392. doi: 10.1080/08958378.2016.1179373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Paradkar PN, Duchemin JB, Voysey R, Walker PJ. Dicer-2-dependent activation of Culex Vago occurs via the TRAF-Rel2 signaling pathway. PLoS Negl. Trop. Dis. 2014;8:e2823. doi: 10.1371/journal.pntd.0002823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tanaka H, et al. Identification and functional analysis of Relish homologs in the silkworm, Bombyx mori. Biochim. Biophys. Acta. 2007;1769:559–568. doi: 10.1016/j.bbaexp.2007.07.001. [DOI] [PubMed] [Google Scholar]
- 19.Schluns H, Crozier RH. Relish regulates expression of antimicrobial peptide genes in the honeybee, Apis mellifera, shown by RNA interference. Insect Mol. Biol. 2007;16:753–759. doi: 10.1111/j.1365-2583.2007.00768.x. [DOI] [PubMed] [Google Scholar]
- 20.Stoven S, et al. Caspase-mediated processing of the Drosophila NF-kappaB factor Relish. Proc. Natl Acad. Sci. USA. 2003;100:5991–5996. doi: 10.1073/pnas.1035902100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Jones P, et al. InterProScan 5: genome-scale protein function classification. Bioinforma. 2014;30:1236–1240. doi: 10.1093/bioinformatics/btu031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Mount, D. W. Using the basic local alignment search tool (BLAST). Cold Spring Harbor Protocols2007, pdb. top17 (2007). [DOI] [PubMed]
- 23.Kosugi S, Hasebe M, Tomita M, Yanagawa H. Systematic identification of cell cycle-dependent yeast nucleocytoplasmic shuttling proteins by prediction of composite motifs. Proc. Natl Acad. Sci. USA. 2009;106:10171–10176. doi: 10.1073/pnas.0900604106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Larkin MA, et al. Clustal W and Clustal X version 2.0. Bioinforma. 2007;23:2947–2948. doi: 10.1093/bioinformatics/btm404. [DOI] [PubMed] [Google Scholar]
- 25.Kumar S, Stecher G, Tamura K. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Mol. Biol. Evol. 2016;33:1870–1874. doi: 10.1093/molbev/msw054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Jones DT, Taylor WR, Thornton JM. The rapid generation of mutation data matrices from protein sequences. Comput. Appl. Biosci. 1992;8:275–282. doi: 10.1093/bioinformatics/8.3.275. [DOI] [PubMed] [Google Scholar]
- 27.Yaffe H, et al. LogSpin: a simple, economical and fast method for RNA isolation from infected or healthy plants and other eukaryotic tissues. BMC Res. Notes. 2012;5:45. doi: 10.1186/1756-0500-5-45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Schmittgen TD, Livak KJ. Analyzing real-time PCR data by the comparative C(T) method. Nat. Protoc. 2008;3:1101–1108. doi: 10.1038/nprot.2008.73. [DOI] [PubMed] [Google Scholar]
- 29.Lee YJ, et al. Structure and Expression of the Tenecin 3 Gene inTenebrio molitor. Biochemical biophysical Res. Commun. 1996;218:6–11. doi: 10.1006/bbrc.1996.0002. [DOI] [PubMed] [Google Scholar]
- 30.Roh K-B, et al. Proteolytic cascade for the activation of the insect toll pathway induced by the fungal cell wall component. J. Biol. Chem. 2009;284:19474–19481. doi: 10.1074/jbc.M109.007419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Lee KH, et al. Identification and characterization of the antimicrobial peptide corresponding to C-terminal β-sheet domain of tenecin 1, an antibacterial protein of larvae of Tenebrio molitor. Biochemical J. 1998;334:99–105. doi: 10.1042/bj3340099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Chae J-H, et al. Purification and characterization of tenecin 4, a new anti-Gram-negative bacterial peptide, from the beetle Tenebrio molitor. Developmental Comp. Immunology. 2012;36:540–546. doi: 10.1016/j.dci.2011.09.010. [DOI] [PubMed] [Google Scholar]
- 33.Jo YH, et al. In silico identification, characterization and expression analysis of attacin gene family in response to bacterial and fungal pathogens in Tenebrio molitor. Entomological Res. 2018;48:45–54. doi: 10.1111/1748-5967.12287. [DOI] [Google Scholar]
- 34.Kim DH, Noh MY, Park KB, Jo YH. Expression profiles of two thaumatin‐like protein (TmTLP) genes in responses to various micro‐organisms from Tenebrio molitor. Entomological Res. 2017;47:35–40. doi: 10.1111/1748-5967.12197. [DOI] [Google Scholar]
- 35.Noh MY, Jo YH. Identification and sequence analysis of two thaumatin‐like protein (TmTLP) genes from Tenebrio molitor. Entomological Res. 2016;46:354–359. doi: 10.1111/1748-5967.12198. [DOI] [Google Scholar]
- 36.Shi YR, et al. Involvement of Relish gene from Macrobrachium rosenbergii in the expression of anti-microbial peptides. Dev. Comp. Immunol. 2015;52:236–244. doi: 10.1016/j.dci.2015.05.008. [DOI] [PubMed] [Google Scholar]
- 37.Ge QQ, et al. Molecular cloning and expression analysis of Relish gene from the ridgetail white prawn Exopalaemon carinicauda. Fish. Sci. 2015;81:699–711. doi: 10.1007/s12562-015-0898-z. [DOI] [Google Scholar]
- 38.Broderick NA, Buchon N, Lemaitre B. Microbiota-induced changes in drosophila melanogaster host gene expression and gut morphology. MBio. 2014;5:e01117–01114. doi: 10.1128/mBio.01117-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Keshavarz M, et al. Tm DorX2 positively regulates antimicrobial peptides in Tenebrio molitor gut, fat body, and hemocytes in response to bacterial and fungal infection. Sci. Rep. 2019;9:1–19. doi: 10.1038/s41598-019-53497-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Stork NE, McBroom J, Gely C, Hamilton AJ. New approaches narrow global species estimates for beetles, insects, and terrestrial arthropods. Proc. Natl Acad. Sci. USA. 2015;112:7519–7523. doi: 10.1073/pnas.1502408112. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Urbanski A, Adamski Z, Rosinski G. Developmental changes in haemocyte morphology in response to Staphylococcus aureus and latex beads in the beetle Tenebrio molitor L. Micron. 2018;104:8–20. doi: 10.1016/j.micron.2017.10.005. [DOI] [PubMed] [Google Scholar]
- 42.Altincicek B, Stotzel S, Wygrecka M, Preissner KT, Vilcinskas A. Host-derived extracellular nucleic acids enhance innate immune responses, induce coagulation, and prolong survival upon infection in insects. J. Immunol. 2008;181:2705–2712. doi: 10.4049/jimmunol.181.4.2705. [DOI] [PubMed] [Google Scholar]
- 43.Yokoi K, et al. Involvement of NF-κB transcription factors in antimicrobial peptide gene induction in the red flour beetle, Tribolium castaneum. Developmental Comp. Immunology. 2012;38:342–351. doi: 10.1016/j.dci.2012.06.008. [DOI] [PubMed] [Google Scholar]
- 44.Salcedo-Porras, N. & Lowenberger, C. Title: The innate immune system of kissing bugs, vectors of chagas disease. Dev Comp Immunol, 10.1016/j.dci.2019.04.007 (2019). [DOI] [PubMed]
- 45.Lange A, et al. Classical nuclear localization signals: definition, function, and interaction with importin α. J. Biol. Chem. 2007;282:5101–5105. doi: 10.1074/jbc.R600026200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Visetnan S, Supungul P, Hirono I, Tassanakajon A, Rimphanitchayakit V. Activation of PmRelish from Penaeus monodon by yellow head virus. Fish. Shellfish. Immunol. 2015;42:335–344. doi: 10.1016/j.fsi.2014.11.015. [DOI] [PubMed] [Google Scholar]
- 47.Huang L, et al. A Genome-Wide Survey for Host Response of Silkworm, Bombyx mori during Pathogen Bacillus bombyseptieus Infection. PloS One. 2009;4:e8098. doi: 10.1371/journal.pone.0008098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Meister S, et al. Immune signaling pathways regulating bacterial and malaria parasite infection of the mosquito Anopheles gambiae. Proc. Natl Acad. Sci. USA. 2005;102:11420–11425. doi: 10.1073/pnas.0504950102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Bulmer MS, Crozier RH. Variation in positive selection in termite GNBPs and Relish. Mol. Biol. Evol. 2006;23:317–326. doi: 10.1093/molbev/msj037. [DOI] [PubMed] [Google Scholar]
- 50.Chow EK, Razani B, Cheng G. Innate immune system regulation of nuclear hormone receptors in metabolic diseases. J. Leukoc. Biol. 2007;82:187–195. doi: 10.1189/jlb.1206741. [DOI] [PubMed] [Google Scholar]
- 51.Flatt T, et al. Hormonal regulation of the humoral innate immune response in Drosophila melanogaster. J. Exp. Biol. 2008;211:2712–2724. doi: 10.1242/jeb.014878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Royer V, Hourdry A, Fraichard S, Bouhin H. Characterization of a putative extracellular matrix protein from the beetle Tenebrio molitor: hormonal regulation during metamorphosis. Dev. Genes. Evol. 2004;214:115–121. doi: 10.1007/s00427-004-0389-1. [DOI] [PubMed] [Google Scholar]
- 53.Tzou P, et al. Tissue-specific inducible expression of antimicrobial peptide genes in Drosophila surface epithelia. Immun. 2000;13:737–748. doi: 10.1016/S1074-7613(00)00072-8. [DOI] [PubMed] [Google Scholar]
- 54.Tsakas S, Marmaras VJ. Insect immunity and its signalling: an overview. Isj-Invertebrate Survival. J. 2010;7:228–238. [Google Scholar]
- 55.Cai Y, Guo Z, Ma C, Su J. Knockdown of dorsal/relish increases larvae susceptibility to microbes in Spodoptera litura. J. Asia-Pacific Entomology. 2019;22:87–95. doi: 10.1016/j.aspen.2018.12.011. [DOI] [Google Scholar]
- 56.Ayyaz, A. & Jasper, H. Intestinal inflammation and stem cell homeostasis in aging Drosophila melanogaster. Frontiers in Cellular and Infection Microbiology3, 10.3389/fcimb.2013.00098 (2013). [DOI] [PMC free article] [PubMed]
- 57.Buchon N, Broderick NA, Lemaitre B. Gut homeostasis in a microbial world: insights from Drosophila melanogaster. Nat. Rev. Microbiol. 2013;11:615–626. doi: 10.1038/nrmicro3074. [DOI] [PubMed] [Google Scholar]
- 58.Zhai Z, Boquete J-P, Lemaitre B. Cell-Specific Imd-NF-κB Responses Enable Simultaneous Antibacterial Immunity and Intestinal Epithelial Cell Shedding upon Bacterial Infection. Immun. 2018;48:897–910. e897. doi: 10.1016/j.immuni.2018.04.010. [DOI] [PubMed] [Google Scholar]
- 59.Huang X-D, et al. Molecular cloning and characterization of class I NF-κB transcription factor from pearl oyster (Pinctada fucata) Fish. Shellfish. immunology. 2012;33:659–666. doi: 10.1016/j.fsi.2012.06.029. [DOI] [PubMed] [Google Scholar]
- 60.Antonova Y, Alvarez KS, Kim YJ, Kokoza V, Raikhel AS. The role of NF-κB factor REL2 in the Aedes aegypti immune response. Insect Biochem. Mol. Biol. 2009;39:303–314. doi: 10.1016/j.ibmb.2009.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Magalhaes T, Leandro D, Ayres C. Knock-down of REL2, but not defensin A, augments Aedes aegypti susceptibility to Bacillus subtilis and Escherichia coli. Acta tropica. 2010;113:167–173. doi: 10.1016/j.actatropica.2009.10.013. [DOI] [PubMed] [Google Scholar]
- 62.Stenbak CR, et al. Peptidoglycan Molecular Requirements Allowing Detection by the Drosophila Immune Deficiency Pathway. J. Immunology. 2004;173:7339–7348. doi: 10.4049/jimmunol.173.12.7339. [DOI] [PubMed] [Google Scholar]
- 63.Jo YH, et al. Regulation of the expression of nine antimicrobial peptide genes by TmIMD confers resistance against Gram-negative bacteria. Sci. Rep. 2019;9:10138. doi: 10.1038/s41598-019-46222-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Buchon N, Broderick NA, Poidevin M, Pradervand S, Lemaitre B. Drosophila intestinal response to bacterial infection: activation of host defense and stem cell proliferation. Cell Host Microbe. 2009;5:200–211. doi: 10.1016/j.chom.2009.01.003. [DOI] [PubMed] [Google Scholar]
- 65.McGettigan J, et al. Insect renal tubules constitute a cell-autonomous immune system that protects the organism against bacterial infection. Insect Biochem. Mol. Biol. 2005;35:741–754. doi: 10.1016/j.ibmb.2005.02.017. [DOI] [PubMed] [Google Scholar]
- 66.Zheng W, et al. Dehydration triggers ecdysone-mediated recognition-protein priming and elevated anti-bacterial immune responses in Drosophila Malpighian tubule renal cells. BMC Biol. 2018;16:60. doi: 10.1186/s12915-018-0532-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Verma P, Tapadia MG. Immune response and anti-microbial peptides expression in Malpighian tubules of Drosophila melanogaster is under developmental regulation. PLoS One. 2012;7:e40714. doi: 10.1371/journal.pone.0040714. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.