Abstract
Honeydew melon (Cucumis melo L.) is an oval-shaped delicious fruit of one cultivar group of the muskmelon with immense nutritional importance and is extensively consumed by many tropical countries. The effect of various organic solvents on the recovery of phytochemicals from honeydew melon plant fruits and seeds was assessed. Further, High-Performance Liquid Chromatography (HPLC) was used to examine and assess the contents of phenolic acid (gallic acid) and flavonoid (rutin) compounds. The use of gas chromatography–mass spectrometry (GC-MS) analysis explained the presence of volatile phytocompounds in the extracts. The use of organic solvents had a substantial impact on the total dry weight and extract yield. In general, the solvent-extracted constituents remained in the order of methanol>chloroform>distilled water for both honeydew melon seeds and whole fruit. 3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) was used to assess the cytotoxicity effect against PC3, HCT116, HeLa, and Jurkat cell lines. The chloroform extract exhibited a good cytotoxic activity against all cell lines as compared to other solvent extracts. HPLC analysis revealed the occurrence of gallic acid content of 0.102 ± 0.23 mg/10 mg of dry whole fruit extract, while 10 mg of dry seed extract contained only 0.022 ± 0.12 mg of gallic acid content. Likewise, rutin content was observed to be 0.224 ± 0.31 mg and 0.1916 ± 0.82 mg/10 mg of dry whole fruit and seed extract, respectively. Further, GC-MS analysis revealed the presence of a total of 37 compounds in chloroform extract of whole fruit, while only 14 compounds were found in seed extract. Nevertheless, more examinations are needed to identify and characterize other metabolites from honeydew melon and evaluate their pharmacological importance.
1. Introduction
Natural products play a significant role in the modern medicine. Ever since ancient period, naturally derived plant products are being widely used by mankind for treating several ailments. Nature has existed as a source of almost all drugs for many years, and herb-based products play an important role in primary human health care as the majority (80%) of the global population rely on traditional medical practices [1, 2]. Several drugs used in the current medicinal practices are mainly derived either from natural resources or from their chemical derivatives. Also, plant-derived phytocompounds are having huge demand in the pharmaceutical industry [2]. Currently, the prevailing health issues along with emergence of communicable infections and disorders are a major concern to the world population as they are seriously causing increased mortality rate day by day. About 50% of all mortality arising in nations is because of these diseases [3, 4]. Hence, a search for an effective curative agent is continuous. In this regard, natural products are the best choice of sources to isolate and identify new leads for discovering novel drug molecules. This is due to the fact that natural products are cost-effective, exhibit therapeutic potential with high efficacy, and possess no or negligible toxicity effect. The available traditional information related to the therapeutic importance of plants is the basis for their explorations by the experts [5].
Orange-fleshed honeydew melon, also known as (Cucumis melo L.), is an oval-shaped delicious fruit with immense nutritional importance and is extensively consumed by many tropical countries. It is one cultivar group of the muskmelon and belongs to the family, Cucurbitaceae. It is produced by crossing orange-fleshed cantaloupe with non-netted, green-fleshed honeydew [6–9]. The distinctive aroma of orange-fleshed honeydew melon fruit is due to the presence of several volatile compounds that are derived biosynthetically from fatty acids, amino acids, carotenoids, and terpenes. The major volatile constituents imparting include phenylethyl alcohol and (Z,Z)-3,6-nonadien-1-ol. The melon fruit is a good source of vital vitamins, i.e., thiamine, riboflavin, and folic acid; provitamin A; and ascorbic acid [7, 9]. It is stated that different parts of the orange-fleshed honeydew melon fruit possess varied levels of soluble solids, total sugars, sucrose, 5-methyltetrahydrofolic acid, and β-carotene [6]. Studies have revealed that secondary metabolites accumulated in plant species depend on different parameters including plant organs and ecological conditions [10, 11]. Also, the amount of the phytocompounds isolation via different extraction approaches may significantly fluctuate. Further, different solvents used for extracting plant-derived compounds can influence on the yield of phytocompounds [11]. Literature survey shows that meager information is available on phytochemicals and bioactivities of honeydew melon plant's different organs, and there is no comparative studies related to this aspect. Therefore, in this study, the effect of various organic solvents on the recovery of phytochemicals from honeydew melon plant fruits and seeds is assessed. Anticancer activities were assessed to compare between fruit and seed extracts. Further, High-Performance Liquid Chromatography (HPLC) was used to examine and assess the total phenolic content. The use of gas chromatography–mass spectrometry (GC-MS) analysis explained the presence of volatile phytocompounds in the extracts.
2. Materials and Methods
2.1. Collection of Honeydew Melon Fruits
Five completely ripened honeydew melon (Cucumis melo L.) fruits were collected directly from the agricultural field during the harvesting season (August, 2018). The fruits were then washed to remove the dust particles and then wiped neatly with sterile cotton cloth to remove any traces of water on the surface. Later, the fruits were cut open to obtain the seeds, and one whole fruit was cut in to pieces without removing any contents. The samples were then dried in a hot air oven till they were completely dried at 55°C–65°C and then ground to powder using a blender. All the powdered samples were then weighed and kept in air-tight containers for further use.
2.2. Solvent Extraction of Phytochemicals
The phytochemicals from powdered seeds and whole fruits were extracted using three different solvent systems such as distilled water, chloroform, and methanol to determine the ideal solvent extraction system. Each dried powdered sample weighing 20 g was added into 300 mL solvents such as distilled water, chloroform, and methanol in a conical flask and stirred for 24 h. Later, the solutions were double filtered using Whatman filter No.1 filter paper, and the filtrates were then evaporated completely in incubator at 40°C. After complete evaporation, the extracts were weighed, and the extract yield (%) and total dry weight were determined. Later, the dried extracts were preserved at 4°C in for further use.
2.3. Cytotoxic Assay through MTT Method
The cytotoxic activity of phytoextracts was determined by 3-(4,5-dimethylthiazol-2-yl)-2, 5-diphenyl tetrazolium bromide (MTT) reduction assay. The cytotoxic activity of different extracts was determined against human prostate cancer cell line (PC-3), human colon cancer cell line (HCT116), immortalized human T lymphocyte cells (Jurkat), and immortal cell line (HeLa). For MTT assay, cell lines were seeded in 96 well culture plates, at a cell density of 20,000 cells per well (200 μL cell suspension), without the test agent and allowed to grow for about 12 h. The IC50 values of different phytoextracts against these cell lines were determined using the concentrations 50, 150, 250, 350, and 450 μg/mL, and the assay was carried out in triplicates. The culture medium without cells was used as medium control, whereas, medium with cells, but without the experimental drug/compound, was used as negative control, and medium with cell lines containing curcumin (15 μM) was used as a positive control. All the plates were incubated for 24 h at 37°C in humidified CO2 (5%) incubator. After the incubation period, the plates were taken out from incubator, and spent media was removed followed by the addition of MTT reagent to a final concentration of 0.5 mg/mL. The plates were returned to the incubator and incubate for 3 h and then the MTT reagent was removed, and 100 μL of solubilization solution (DMSO) was added. The absorbance was in an ELISA reader at 570 nm with reference wavelength 630 nm. The IC50 values for each extract were determined by using linear regression equation, i.e., Y = mX + C. Here, Y = 50, m (angular coefficient) and C (linear coefficient) values were derived from the viability graph, by the relationship between extract concentration and absorbance (cell viability).
2.4. High-Performance Liquid Chromatography (HPLC) Analysis of Polyphenols
The analysis was made in isocratic mode with the mobile phase acetonitrile and water in the ratio 7 : 3 with the RP-HPLC C-18 column at a flow rate of 1 mL/min. The standards, gallic acid and rutin (Sigma Aldrich, St. Louis, MO, USA) (0.4 mg/mL), and samples (10 mg/mL) were dissolved in mobile phase, and 20 μL was injected, and the elution was monitored. The spectral data was obtained between 190 and 500 nm. The available standards, their UV spectra, and retention times were compared with the samples to identify and quantify phenolic compounds.
2.5. Gas Chromatography–Mass Spectrometry (GC-MS) Analysis
Each solvent extract was subjected to GC-MS analysis using the model instrument, GCMS-QP2010 Ultra (Shimadzu Co., Japan) attached with a capillary column DB-1 (0.25 μm film × 0.25 mm I.d.×30 m length). Analysis was performed by injecting 1 μL of the sample with a split ratio of 20 : 1, and Helium gas (99.9%) was used as the carrier gas at a flow rate of 1 mL/min. The analysis was performed in the electron impact (EI) mode with 70 eV of ionization energy. The injector temperature was maintained at 250°C (constant). The column oven temperature was set at 50°C (held for 3 min), raised at 10°C per min to 280°C (held for 3 min) and finally held at 300°C for 10 min. The compounds were identified after comparing the spectral configurations obtained with that of available mass spectral database (NIST and WILEY libraries).
2.6. Statistical Analysis
The results were statistically analyzed using GraphPad Prism (version 5.0) statistical software and expressed as the mean ± standard deviation (SD). Multiple comparisons were done through Dunnett's test by comparing the test data with control/untreated data to determine the statistically significant differences. Further, Student's t-test was used to compare the cytotoxicity effect between whole fruit and seed extracts. p value ≤0.05 was considered statistically significant.
3. Results
The use of organic solvents had a substantial impact on the total dry weight and extracts yield. Comparatively, the methanolic whole fruit extract showed with a higher extraction yield of 11.12%, and total dry weight was observed to be 546.01 ± 1.91 μg of dry weight. Likewise, the methanolic seed extract showed the highest extraction yield of 11.62% with 552.02 ± 2.23 μg of total dry weight. The water extract of both seeds and whole fruit yielded the lowest dry weight and yield. In general, the solvent-extracted constituents remained in the order of methanol>chloroform>distilled water for both honeydew melon seeds and whole fruit.
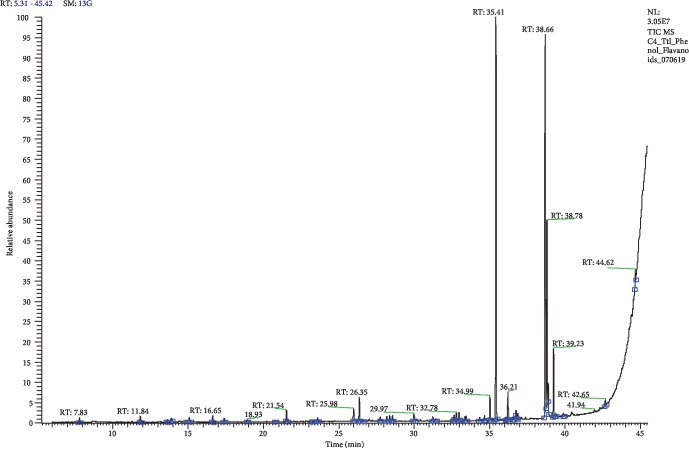
The HPLC analysis used to identify the presence of polyphenols in the chloroform extracts of seeds and whole fruit showed the maximum antiproliferative activity against all the cells. The gallic acid content was found to be 0.102 ± 0.23 mg/10 mg of dry whole fruit extract, while for seeds, it was observed to be 0.022 ± 0.12 mg/10 mg. Likewise, the flavonoid, rutin, was observed to be 0.224 ± 0.31 mg/10 mg of dry whole fruit extract. In the chloroform extract of seeds, it was found to be 0.1916 ± 0.82 mg/10 mg. Further, the GC-MS analysis revealed that the chloroform extract of whole fruit contains a total of 37 compounds, while seed extract contained only 14 compounds. The identified nonvolatile compounds of chloroform whole fruit extracts are given in Table 1 and Figure 1.
Table 1.
The GC-MS analysis report showing the presence of different metabolites.
| Sl. No. | Apex RT | Area | % area | Height | % height | Identification |
|---|---|---|---|---|---|---|
| 1 | 7.83 | 813202.579 | 0.31 | 330482.409 | 0.34 | Ribitol, 1,3:2,4-di-O-benzylidene |
| 2 | 13.63 | 415974.127 | 0.16 | 164857.783 | 0.17 | Beta-estradiol 3,17-disulfate |
| 3 | 13.9 | 898950.604 | 0.35 | 262301.786 | 0.27 | Malic acid |
| 4 | 14.95 | 356696.389 | 0.14 | 121270.499 | 0.12 | Undecane |
| 5 | 15.1 | 1003747.7 | 0.39 | 324390.461 | 0.33 | 2,3-Dihydroxy-2-methylpentanoic acid |
| 6 | 17.4 | 810167.007 | 0.31 | 280707.991 | 0.29 | Mephaneine |
| 7 | 18.93 | 421209.113 | 0.16 | 164030.915 | 0.17 | Oxazolam |
| 8 | 20.81 | 138570.826 | 0.05 | 58502.816 | 0.06 | 5-(Hydroxymethyl)-2-(dimethoxymethyl)furan |
| 9 | 23.23 | 132501.875 | 0.05 | 61695.305 | 0.06 | 9-Nonadecene |
| 10 | 23.43 | 326043.858 | 0.13 | 125731.585 | 0.13 | Tetradecane |
| 11 | 23.61 | 740844.025 | 0.28 | 291654.173 | 0.3 | Glafenin |
| 12 | 26.35 | 4846262.298 | 1.86 | 1783633.242 | 1.81 | 2,4-Di-tert-butylphenol |
| 13 | 26.6 | 326311.396 | 0.13 | 122525.028 | 0.12 | Lauric acid, methyl ester |
| 14 | 28.18 | 845949.533 | 0.33 | 331696.785 | 0.34 | 2-Dodecanol |
| 15 | 28.35 | 1219645.767 | 0.47 | 409647.806 | 0.42 | Hexadecane |
| 16 | 28.56 | 1134791.726 | 0.44 | 411097.895 | 0.42 | Methyl 3,5-dimethoxybenzoate |
| 17 | 31.22 | 895920.761 | 0.34 | 326795.903 | 0.33 | Myristic acid, methyl ester |
| 18 | 32.58 | 356138.721 | 0.14 | 154978.494 | 0.16 | Pentadecanoic acid, methyl ester |
| 19 | 32.64 | 754986.253 | 0.29 | 332653.706 | 0.34 | 1-Docosene |
| 20 | 32.78 | 2052151.175 | 0.79 | 575060.411 | 0.58 | Nonadecane |
| 21 | 32.96 | 1498170.836 | 0.58 | 564804.031 | 0.57 | N1-(tert-Butyldimethylsilyl)-6-nitrobenzene-1,3-diamine |
| 22 | 33.36 | 867260.244 | 0.33 | 307142.157 | 0.31 | Methyl 13-methyltetradecanoate |
| 23 | 34.32 | 764655.12 | 0.29 | 253979.509 | 0.26 | Phthalic acid, butyl isobutyl ester |
| 24 | 34.66 | 976392.889 | 0.38 | 359128.863 | 0.36 | Palmitic acid, methyl ester |
| 25 | 34.99 | 4397282.814 | 1.69 | 1756158.358 | 1.78 | (Z)-Methyl hexadec-11-enoate |
| 26 | 35.41 | 77711514.61 | 29.89 | 30214884.99 | 30.7 | Palmitic acid, methyl ester |
| 27 | 36.09 | 466517.711 | 0.18 | 183204.419 | 0.19 | Hexadecanoic acid |
| 28 | 36.21 | 5520414.14 | 2.12 | 2061336.773 | 2.09 | Dibutyl phthalate |
| 29 | 36.74 | 997875.035 | 0.38 | 447963.602 | 0.46 | Palmitic acid, ethyl ester |
| 30 | 36.81 | 669635.521 | 0.26 | 300464.6 | 0.31 | 2-Octadecoxyethanol |
| 31 | 38.66 | 71694171.07 | 27.58 | 28428235.56 | 28.88 | Linoleic acid, methyl ester |
| 32 | 38.78 | 41915737.09 | 16.12 | 13833942.29 | 14.05 | Linolenic acid, methyl ester |
| 33 | 38.87 | 3935994.727 | 1.51 | 1775814.73 | 1.8 | 13-Octadecenoic acid, methyl ester |
| 34 | 39.23 | 13455425.91 | 5.18 | 5116611.537 | 5.2 | Stearic acid, methyl ester |
| 35 | 39.88 | 649279.136 | 0.25 | 224145.171 | 0.23 | Linoleic acid ethyl ester |
| 36 | 42.65 | 1173609.722 | 0.45 | 447888.354 | 0.46 | Eicosanoic acid, methyl ester |
| 37 | 44.62 | 1960767.037 | 0.75 | 1100376.213 | 1.12 | Docosanoic acid, methyl ester |
Figure 1.
The major phytocompounds detected in the chloroform whole fruit extract of honeydew melon using GC-MS analysis.
To explore the cytotoxicity effect of methanol, chloroform and water extracts of seeds and whole fruit of honeydew melon were determined by MTT assay. The anticancer activity was carried out against different cancer cells, such as PC3, HCT116, HeLa, and Jurkat cell lines, and the results showed a decreased viability (%) with increased concentration of the phytoextracts. The results have shown that different solvent extracts of whole fruit and seeds inhibited different cancer cells in a dose-dependent manner. The methanol and chloroform extracts exhibited a good cytotoxic activity against HeLa cell lines as compared to distilled water extracts (Table 2).
Table 2.
Effect of phytoextracts on cancer cell line, HeLa.
| Drug/phytoextracts | Concentration | Percentage cell viability | ||
| Control/untreated cell lines | — | 100 ± 0.005 | ||
| Curcumin | 15 μM | 49.85±0.05∗∗∗∗ | ||
|
| ||||
| Methanol extract | Distilled water extract | Chloroform extract | ||
| Whole fruit | 50 μg/mL | 89.80±0.007∗∗∗∗ | 91.40±0.006∗∗∗∗ | 86.12±0.004∗∗∗∗ |
| Seeds | 86.02±0.004∗∗∗∗ | 84.36±0.004∗∗∗∗ | 93.68±0.006∗∗∗∗ | |
| Whole fruit | 150 μg/mL | 75.25±0.005∗∗∗∗ | 76.19±0.001∗∗∗∗ | 72.46±0.005∗∗∗∗ |
| Seeds | 77.95±0.002∗∗∗∗ | 77.95±0.002∗∗∗∗ | 76.08±0.004∗∗∗∗ | |
| Whole fruit | 250 μg/mL | 46.27±0.003∗∗∗∗ | 64.18±0.001∗∗∗∗ | 51.35±0.005∗∗∗∗ |
| Seeds | 63.04±0.003∗∗∗∗ | 61.07±0.005∗∗∗∗ | 56.02±0.02∗∗∗∗ | |
| Whole fruit | 350 μg/mL | 32.60±0.007∗∗∗∗ | 54.86±0.004∗∗∗∗ | 32.40±0.005∗∗∗∗ |
| Seeds | 54.76±0.002∗∗∗∗ | 50.51±0.005∗∗∗∗ | 43.78±0.003∗∗∗∗ | |
| Whole fruit | 450 μg/mL | 18.32±0.004∗∗∗∗ | 25.87±0.004∗∗∗∗ | 14.59±0.005∗∗∗∗ |
| Seeds | 32.09±0.003∗∗∗∗ | 36.12±0.002∗∗∗∗ | 38.01±0.004∗∗∗∗ | |
Comparative cytotoxic effect of different solvent extracts of honeydew melon on HeLa cell lines after 24 h of incubation. Data is expressed as the mean ± SD (each treatment, n = 3). ∗∗∗∗ indicates p < 0.0001 in comparison with control/untreated cells by one-way ANOVA followed by Dunnett's test for multiple comparisons.
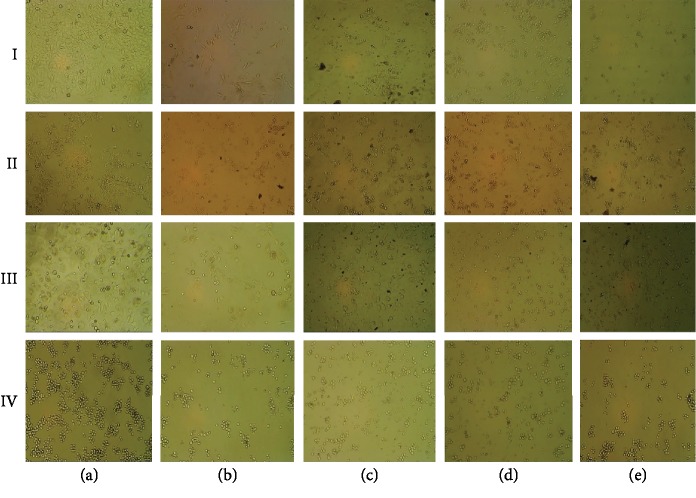
However, almost similar cytotoxic activity has been exhibited against HCT116 by all different solvent extracts tested (Table 3). The chloroform extract of seed has shown a better cytotoxicity against PC3 when compared to the whole fruit; however, distilled water and methanol extracts of whole fruit exhibited better cytotoxicity compared to seed extract (Table 4). The cytotoxicity against Jurkat cell lines was found to be almost similar among all different extracts (Table 5). Both whole fruit (Figure 2) and seed extract extracts (figure not shown) exhibited good cytotoxic activity against different cancer cell lines.
Table 3.
Effect of phytoextracts on cancer cell line, HCT116.
| Drug/phytoextracts | Concentration | Percentage cell viability | ||
| Control/untreated cell lines | — | 100 ± 0.006 | ||
| Curcumin | 15 μM | 50.63±0.003∗∗∗∗ | ||
|
| ||||
| Methanol extract | Distilled water extract | Chloroform extract | ||
| Whole fruit | 50 μg/mL | 80.15±0.007∗∗∗∗ | 84.27±0.001∗∗∗∗ | 89.25±0.01∗∗∗∗ |
| Seeds | 80.35±0.004∗∗∗∗ | 83.94±0.006∗∗∗∗ | 80.40±0.007∗∗∗∗ | |
| Whole fruit | 150 μg/mL | 70.53±0.003∗∗∗∗ | 66.75±0.004∗∗∗∗ | 73.98±0.003∗∗∗∗ |
| Seeds | 68.81±0.007∗∗∗∗ | 69.87±0.006∗∗∗∗ | 70.40±0.01∗∗∗∗ | |
| Whole fruit | 250 μg/mL | 49.30±0.007∗∗∗∗ | 56.93±0.004∗∗∗∗ | 60.45±0.007∗∗∗∗ |
| Seeds | 48.50±0.007∗∗∗∗ | 58.46±0.002∗∗∗∗ | 47.37±0.002∗∗∗∗ | |
| Whole fruit | 350 μg/mL | 31.25±0.007∗∗∗∗ | 46.25±0.01∗∗∗∗ | 42.01±0.007∗∗∗∗ |
| Seeds | 37.35±0.003∗∗∗∗ | 47.40±0.01∗∗∗∗ | 30.12±0.01∗∗∗∗ | |
| Whole fruit | 450 μg/mL | 28.46±0.003∗∗∗∗ | 16.72±0.002∗∗∗∗ | 27.07±0.004∗∗∗∗ |
| Seeds | 22.49±0.01∗∗∗∗ | 27.14±0.007∗∗∗∗ | 18.97±0.004∗∗∗∗ | |
Comparative cytotoxic effect of different solvent extracts of honeydew melon on HCT116 cell lines after 24 h of incubation. Data is expressed as the mean ± SD (each treatment, n = 3). ∗∗∗∗ indicates p < 0.0001 in comparison with control/untreated cells by one-way ANOVA followed by Dunnett's test for multiple comparisons.
Table 4.
Effect of honeydew melon phytoextracts on cancer cell line, PC3.
| Drug/phytoextracts | Concentration | Percentage cell viability | ||
| Control/untreated cell lines | — | 100 ± 0.008 | ||
| Curcumin | 15 μM | 49.08±0.002∗∗∗∗ | ||
|
| ||||
| Methanol extract | Distilled water extract | Chloroform extract | ||
| Whole fruit | 50 μg/mL | 96.77±0.01∗∗∗∗ | 86.33±0.004∗∗∗∗ | 89.00±0.007∗∗∗∗ |
| Seeds | 96.89±0.005∗∗∗∗ | 98.32±0.002∗∗∗∗ | 89.56±0.002∗∗∗∗ | |
| Whole fruit | 150 μg/mL | 80.47±0.002∗∗∗∗ | 75.77±0.002∗∗∗∗ | 77.45±0.01∗∗∗∗ |
| Seeds | 91.63±0.005∗∗∗∗ | 90.43±0.009∗∗∗∗ | 75.85±0.01∗∗∗∗ | |
| Whole fruit | 250 μg/mL | 70.35±0.004∗∗∗∗ | 62.70±0.007∗∗∗∗ | 65.49±0.001∗∗∗∗ |
| Seeds | 75.53±0.01∗∗∗∗ | 69.08±0.002∗∗∗∗ | 56.77±0.01∗∗∗∗ | |
| Whole fruit | 350 μg/mL | 58.80±0.002∗∗∗∗ | 50.91±0.003∗∗∗∗ | 52.66±0.004∗∗∗∗ |
| Seeds | 64.38±0.004∗∗∗∗ | 56.65±0.003∗∗∗∗ | 41.75±0.001∗∗∗∗ | |
| Whole fruit | 450 μg/mL | 34.66±0.003∗∗∗∗ | 46.62±0.009∗∗∗∗ | 36.81±0.005∗∗∗∗ |
| Seeds | 44.22±0.006∗∗∗∗ | 39.92±0.01∗∗∗∗ | 31.39±0.007∗∗∗∗ | |
Comparative cytotoxic effect of different solvent extracts of honeydew melon on PC3 cell lines after 24 h of incubation. Data is expressed as the mean ± SD (each treatment, n = 3). ∗∗∗∗ indicates p < 0.0001 in comparison with control/untreated cells by one-way ANOVA followed by Dunnett's test for multiple comparisons.
Table 5.
Effect of honeydew melon phytoextracts on cancer cell line, Jurkat.
| Drug/phytoextracts | Concentration | Percentage cell viability | ||
| Control/untreated cell lines | — | 100 ± 0.009 | ||
| Curcumin | 15 μM | 43.10±0.007∗∗∗∗ | ||
|
| ||||
| Methanol extract | Distilled water extract | Chloroform extract | ||
| Whole fruit | 50 μg/mL | 79.48±0.006∗∗∗∗ | 90.77±0.007∗∗∗∗ | 87.81±0.001∗∗∗∗ |
| Seeds | 87.45±0.009∗∗∗∗ | 73.63±0.002∗∗∗∗ | 88.62±0.004∗∗∗∗ | |
| Whole fruit | 150 μg/mL | 68.27±0.002∗∗∗∗ | 85.03±0.004∗∗∗∗ | 67.74±0.004∗∗∗∗ |
| Seeds | 71.59±0.002∗∗∗∗ | 66.21±0.002∗∗∗∗ | 74.55±0.001∗∗∗∗ | |
| Whole fruit | 250 μg/mL | 46.32±0.004∗∗∗∗ | 69.62±0.006∗∗∗∗ | 52.68±0.004∗∗∗∗ |
| Seeds | 58.06±0.004∗∗∗∗ | 56.36±0.003∗∗∗∗ | 47.31±0.004∗∗∗∗ | |
| Whole fruit | 350 μg/mL | 34.1±0.003∗∗∗∗ | 52.86±0.005∗∗∗∗ | 29.65±0.003∗∗∗∗ |
| Seeds | 43.99±0.003∗∗∗∗ | 47.04±0.002∗∗∗∗ | 32.61±0.009∗∗∗∗ | |
| Whole fruit | 450 μg/mL | 16.68±0.002∗∗∗∗ | 38.08±0.006∗∗∗∗ | 11.55±0.003∗∗∗∗ |
| Seeds | 27.41±0.008∗∗∗∗ | 28.13±0.005∗∗∗∗ | 16.12±0.008∗∗∗∗ | |
Comparative cytotoxic effect of different solvent extracts of honeydew melon on Jurkat cell lines after 24 h of incubation. Data is expressed as the mean ± SD (each treatment, n = 3). ∗∗∗∗ indicates p < 0.0001 in comparison with control/untreated cells by one-way ANOVA followed by Dunnett's test for multiple comparisons.
Figure 2.
Anticancer activity of honeydew melon whole fruit extract against different cancer cell lines, (a) untreated/control HeLa cell line, (b) treated with curcumin (15 μM), and (c–e) treated with methanol, water, and chloroform seed extracts, respectively, at IC50 values. I, II, III, and IV: HeLa, HCT116, PC3, and Jurkat cell lines, respectively.
The cytotoxicity was not observed in untreated cell lines; however, all the cell lines treated with curcumin (15 μM) showed the cytotoxicity. The statistical analysis by one-way ANOVA followed by Dunnett's test for multiple comparisons revealed high significance at p < 0.0001 in comparison with control/untreated cells against all the different tested cell lines. The seed extracts from all the three solvent systems exhibited anticancer activity against HeLa cell lines; the IC50 values were found to be 347.55, 349.43, and 331.33 μg/mL, respectively, for methanol, water, and chloroform extracts. Further, the cytotoxic activity of whole fruit extracts against HeLa cell lines revealed IC50 values of 264.27, 332.89, and 257.7 μg/mL, respectively, for methanol, water, and chloroform extracts. The IC50 values of all different extracts against different cell lines are shown in Table 6. Further, the cytotoxicity effects between whole fruit and seed extracts were assessed using t-test (p < 0.05). The results showed that there was no statistically significant difference between the extracts. Overall, the seed extract was more cytotoxic to HCT116 cell line, and whole fruit extract was more cytotoxic to HeLa, PC3, and Jurkat cell lines.
Table 6.
Cytotoxic activity of different phytoextracts against different cancer cell lines and IC50 values.
| Phytoextracts | IC50 values | ||||
|---|---|---|---|---|---|
| Fruit part | Solvent | HeLa | HCT116 | PC3 | Jurkat |
| Seed | Methanol | 347.55 μg/mL | 259.25 μg/mL | 436.36 μg/mL | 303.54 μg/mL |
| Water | 349.43 μg/mL | 304.42 μg/mL | 390.0 μg/mL | 293.13 μg/mL | |
| Chloroform | 331.33 μg/mL | 246.93 μg/mL | 319.8 μg/mL | 261.13 μg/mL | |
|
| |||||
| Whole fruit | Methanol | 264.27 μg/mL | 264.86 μg/mL | 376.92 μg/mL | 243.47 μg/mL |
| Water | 332.89 μg/mL | 277.93 μg/mL | 373.09 μg/mL | 376.64 μg/mL | |
| Chloroform | 257.70 μg/mL | 290.32 μg/mL | 341.01 μg/mL | 250.21 μg/mL | |
4. Discussion
Naturally derived products are the vital source for developing novel drugs. Studies have shown that phytochemicals possess incredible health benefits and play a significant role in human disease prevention. Phytochemicals like plant secondary metabolites and antioxidants exhibit important therapeutic properties [12–14]. In general, phytocompounds are extracted from different plant sources using many ways including decoction, maceration, Soxhlet extraction, supercritical fluid extraction, microwave-aided extraction, and ultrasound-aided extraction approaches [15]. The maceration technique is widely used at the preliminary investigation level due to its simplicity and easy to use [11, 15]. Hence, maceration was used in the present study to preliminarily examine the bioactive principles and bioactivities.
The organic solvents will have a substantial impact on the total dry weight and extract yield. Many studies have evidently suggested that methanol as the best solvent to be used for recovering higher extractable phytocompounds from honeydew melon, irrespective of the plant parts [11, 16, 17]. Likewise, in the present study also, methanol solvent recovered maximum extractable elements from the muskmelon whole fruit and seed extracts. Thus, the present study results are in agreement with earlier recorded observations. The presence of substantial variations in the obtained total yield of the extract in different solvents could be correlated to polar nature of the organic solvents evaluated [11, 18].
Previously, researchers have studied cytotoxicity effect of muskmelon fruits and seeds [19, 20]. However, the research efforts were limited to only a single plant parts (seeds, peel, or whole fruit) and to single cancer cells. Moreover, the use of different solvent extracts against different cancer cells is limited. This is a first comparative study to examine the cytotoxicity effect of different solvent extracts of whole fruit and seeds against several cancer cells, which was carried out for the first time. The highest cytotoxicity observed in the extract of whole fruit could be because of higher levels of bioactive compounds present in it. Similarly, it has been documented that biological activities are directly correlated to the amount of active metabolites that occur in plant extracts [5, 8, 11].
Polyphenols are a group of valuable plant secondary metabolites known for their free radical scavenging activity in cells and protects body from many diseases, such as neuronal disease, cardiovascular diseases, cataract, and cancers [5]. Mainly, secondary metabolites of plants include polyphenolics and flavonoids, and they possess superior pharmacological activities including antimicrobial, antioxidant, anti-inflammatory, and anticancer activities [21]. The quantitative analyses of honeydew melon extracts revealed that whole fruit extract possesses the highest gallic acid and rutin contents. This could be attributed to the fact that whole fruit is being exposed to external environmental stresses as compared to seeds; it contains higher levels of polyphenols. Similarly, a previous study by Rolim and coworkers have shown that peel extract having higher levels of polyphenols compared to seeds [20].
The biological properties of honeydew melon seed and whole fruit extracts could be due to the occurrence of different phytoconstituents. The literature survey discloses the presence of several nonvolatile compounds from aqueous, methanol, and ethanol extracts of muskmelon seeds and fruit peel around the globe. To the best of our awareness, there is no single report of GC-MS analysis to characterize honeydew melon metabolites. Therefore, this study also included GC-MS analysis to identify the possible bioactive metabolites of honeydew melon extracts. The whole fruit extract was identified with numerous metabolites, including previously known compounds possessing numerous pharmacological properties. In this investigation, the superior anticancer activities exhibited by chloroform extract of whole fruit could be related to the existence of more number of biologically active compounds, such as hexadecanoic acid, undecane, and 2-dodecanol [2, 18, 22]. Further, the presence of other newly determined compounds in the extract clearly opens a new avenue for the exploration of muskmelon plant in drug discovery and other medicinal applications. Yet, more examinations are needed to identify and characterize other metabolites from muskmelon and evaluate their pharmacological importance.
5. Conclusion
The present study revealed the occurrence of various soluble biologically active phytocompounds in honeydew melon whole fruit and seed extracts using different solvent systems. Overall, the study evidenced the role of phytocompounds occurring in honeydew melon extracts in inhibiting the growth of several types of cancer cells. Also, polyphenol content and yield of the extract varied depending on the solvent types used for the extraction. In the chloroform extract, more soluble metabolites were evidenced, and hence, it exhibited higher anticancer activity as compared to other solvent extracts. These observations clearly support the use of whole fruit for further investigations for treating various diseases, including cancers.
Data Availability
Not applicable.
Conflicts of Interest
The authors declare that there are no conflicts of interest.
References
- 1.Gezahegn Z., Akhtar M. S., Woyessa D., Tariku Y. Antibacterial potential of Thevetia peruviana leaf extracts against food associated bacterial pathogens. Journal of Coastal Life Medicine. 2015;3(2):150–157. doi: 10.12980/jclm.3.201514j86. [DOI] [Google Scholar]
- 2.Swamy M., Sinniah U. A comprehensive review on the phytochemical constituents and pharmacological activities of Pogostemon cablin Benth.: an aromatic medicinal plant of industrial importance. Molecules. 2015;20(5):8521–8547. doi: 10.3390/molecules20058521. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Nii-Trebi N. I. Emerging and neglected infectious diseases: insights, advances, and challenges. BioMed Research International. 2017;2017:15. doi: 10.1155/2017/5245021.5245021 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Lobo D. A., Velayudhan R., Chatterjee P., Kohli H., Hotez P. J. The neglected tropical diseases of India and South Asia: review of their prevalence, distribution, and control or elimination. PLoS Neglected Tropical Diseases. 2011;5(10, article e1222) doi: 10.1371/journal.pntd.0001222. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Mohanty S. K., Mallappa K. S., Godavarthi A., Subbanarasiman B., Maniyam A. Evaluation of antioxidant, in vitro cytotoxicity of micropropagated and naturally grown plants of Leptadenia reticulata (Retz.) Wight & Arn.-an endangered medicinal plant. Asian Pacific Journal of Tropical Medicine. 2014;7:S267–S271. doi: 10.1016/S1995-7645(14)60244-3. [DOI] [PubMed] [Google Scholar]
- 6.Lester G. E. Antioxidant, sugar, mineral, and phytonutrient concentrations across edible fruit tissues of orange-fleshed honeydew melon (Cucumis melo L.) Journal of Agricultural and Food Chemistry. 2008;56(10):3694–3698. doi: 10.1021/jf8001735. [DOI] [PubMed] [Google Scholar]
- 7.Laur L. M., Tian L. Provitamin A and vitamin C contents in selected California-grown cantaloupe and honeydew melons and imported melons. Journal of Food Composition and Analysis. 2011;24(2):194–201. doi: 10.1016/j.jfca.2010.07.009. [DOI] [Google Scholar]
- 8.Perry P. L., Wang Y., Lin J. Analysis of honeydew melon (Cucumis melo var. inodorus) flavour and GC-MS/MS identification of (E,Z)-2,6-nonadienyl acetate. Flavour and Fragrance Journal. 2009;24(6):341–347. doi: 10.1002/ffj.1947. [DOI] [Google Scholar]
- 9.Zeb A. Phenolic profile and antioxidant activity of melon (Cucumis melo L.) seeds from Pakistan. Foods. 2016;5(4):p. 67. doi: 10.3390/foods5040067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sellem I., Kaaniche F., Chakchouk A. M., Mellouli L. Anti-oxidant, antimicrobial and anti-acetylcholinesterase activities of organic extracts from aerial parts of three Tunisian plants and correlation with polyphenols and flavonoids contents. Bangladesh Journal of Pharmacology. 2016;11(2):531–544. doi: 10.3329/bjp.v11i2.26001. [DOI] [Google Scholar]
- 11.Swamy M. K., Arumugam G., Kaur R., Ghasemzadeh A., Yusoff M. M., Sinniah U. R. GC-MS based metabolite profiling, antioxidant and antimicrobial properties of different solvent extracts of Malaysian Plectranthus amboinicus leaves. Evidence-Based Complementary and Alternative Medicine. 2017;2017:10. doi: 10.1155/2017/1517683.1517683 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ravindran R., Swamy M. K., Jaganathan R. Natural Bio-active Compounds. Springer; 2019. Therapeutic potential of plant polyphenolics and their mechanistic action against various diseases; pp. 313–351. [DOI] [Google Scholar]
- 13.Swamy M. K., Akhtar M. S. Natural Bio-Active Compounds: Volume 2: Chemistry, Pharmacology and Health Care Practices. Springer; 2019. [Google Scholar]
- 14.Swamy M. K., Patra J. K., Rudramurthy G. R. Medicinal Plants: Chemistry, Pharmacology, and Therapeutic Applications. CRC Press; 2019. [Google Scholar]
- 15.Zhang Q. W., Lin L. G., Ye W. C. Techniques for extraction and isolation of natural products: a comprehensive review. Chinese Medicine. 2018;13(1, article 177) doi: 10.1186/s13020-018-0177-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Sultana B., Anwar F., Ashraf M. Effect of extraction solvent/technique on the antioxidant activity of selected medicinal plant extracts. Molecules. 2009;14(6):2167–2180. doi: 10.3390/molecules14062167. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Anwar F., Shaheen N., Shabir G., Ashraf M., Alkharf K. M., Gilani A. H. Variation in antioxidant activity and phenolic and flavonoid contents in the flowers and leaves of Ghaneri (Lantana camara L.) as affected by different extraction solven. International Journal of Pharmacology. 2013;9(7):442–453. doi: 10.3923/ijp.2013.442.453. [DOI] [Google Scholar]
- 18.Swamy M. K., Sinniah U. R., Akhtar M. S. In vitro pharmacological activities and GC-MS analysis of different solvent extracts of Lantana camara leaves collected from tropical region of Malaysia. Evidence-Based Complementary and Alternative Medicine. 2015;2015:9. doi: 10.1155/2015/506413.506413 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Ittiyavirah S. P., George A., Santhosh A. M., Kurian S. T., Pappachan P., Jacob G. Studies of cytotoxic potential of aqueous fruit extract of Cucumis melo. Linn in prostate cancer cell lines PC-3 using MTT and neutral red assay. Iranian Journal of Pharmacology & Therapeutics. 2015;13(1):19–25. [Google Scholar]
- 20.Rolim P. M., Fidelis G. P., Padilha C. E. A., Santos E. S., Rocha H. A. O., Macedo G. R. Phenolic profile and antioxidant activity from peels and seeds of melon (Cucumis melo L. var. reticulatus) and their antiproliferative effect in cancer cells. Brazilian Journal of Medical and Biological Research. 2018;51(4):p. e6069. doi: 10.1590/1414-431x20176069. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Ghasemzadeh A., Ghasemzadeh N. Flavonoids and phenolic acids: role and biochemical activity in plants and human. Journal of Medicinal Plants Research. 2011;5(31):6697–6703. doi: 10.5897/jmpr11.1404. [DOI] [Google Scholar]
- 22.Amarowicz R. Squalene: a natural antioxidant? European Journal of Lipid Science and Technology. 2009;111(5):411–412. doi: 10.1002/ejlt.200900102. [DOI] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
Not applicable.