Summary
In amyotrophic lateral sclerosis (ALS) motor neurons (MNs) undergo dying-back, where the distal axon degenerates before the soma. The hexanucleotide repeat expansion (HRE) in C9ORF72 is the most common genetic cause of ALS, but the mechanism of pathogenesis is largely unknown with both gain- and loss-of-function mechanisms being proposed. To better understand C9ORF72-ALS pathogenesis, we generated isogenic induced pluripotent stem cells. MNs with HRE in C9ORF72 showed decreased axonal trafficking compared with gene corrected MNs. However, knocking out C9ORF72 did not recapitulate these changes in MNs from healthy controls, suggesting a gain-of-function mechanism. In contrast, knocking out C9ORF72 in MNs with HRE exacerbated axonal trafficking defects and increased apoptosis as well as decreased levels of HSP70 and HSP40, and inhibition of HSPs exacerbated ALS phenotypes in MNs with HRE. Therefore, we propose that the HRE in C9ORF72 induces ALS pathogenesis via a combination of gain- and loss-of-function mechanisms.
Keywords: C9ORF72, induced pluripotent stem cells, amyotrophic lateral sclerosis, gene editing, disease modeling, axonal trafficking, heat shock proteins, HSP70, HSP40
Highlights
-
•
Axonal trafficking is disrupted in MNs with hexanucleotide repeat expansion (HRE)
-
•
C9ORF72 knockout (KO) exacerbated phenotypes in MNs with HRE
-
•
C9ORF72 KO reduced heat shock proteins in MNs with HRE
-
•
Inhibition of heat shock proteins exacerbated ALS phenotypes in MNs with HRE
Sterneckert and colleagues generated isogenic induced pluripotent stem cell lines and demonstrated that MNs with hexanucleotide repeat expansion (HRE) in C9ORF72 show reduced axonal trafficking, which is not recapitulated by knocking out C9ORF72 in MNs from healthy individuals. In contrast, knocking out C9ORF72 exacerbated phenotypes in MNs with HRE, suggesting that both gain- and loss-of-function mechanisms contribute to C9ORF72-ALS.
Introduction
Amyotrophic lateral sclerosis (ALS) is a devastating neurodegenerative disease characterized by the progressive loss of motor neurons (MNs). Neurodegeneration proceeds in a dying-back manner, starting with the distal followed by the proximal axon, and finally MN soma death (Naumann et al., 2018). None of the currently available drugs meaningfully delay pathogenesis and new therapeutics are urgently needed.
The most common known genetic cause of ALS is a GGGGCC hexanucleotide repeat expansion (HRE) within the first intron of the gene C9ORF72. Approximately 40% of familial and 5% of sporadic ALS cases have an HRE in C9ORF72 (Majounie et al., 2012). Thus, understanding the pathogenesis of C9ORF72-ALS, particularly in the distal axon, could lead to more effective treatments for many ALS patients.
C9ORF72-ALS is inherited in an autosomal dominant manner, but the precise mechanism of pathogenesis is unclear. At least two studies have assessed C9ORF72 protein levels in patient brain tissue and demonstrated that C9ORF72 protein levels are reduced in patients with HRE (Frick et al., 2018, Waite et al., 2014), which is consistent with reports that RNA levels are also reduced (Gijselinck et al., 2012). Thus, one of the first hypotheses was that haploinsufficiency leading to a loss-of-function (LOF) mechanism may contribute to ALS and one report showed that reducing C9ORF72 levels in zebrafish resulted in motor defects (Ciura et al., 2013). However, multiple reports of genetic knockouts (KOs) in zebrafish and mice showed no evidence of MN degeneration and ALS pathology (Hruscha et al., 2013, Jiang et al., 2016). Subsequently, it was discovered that HRE is translated via a non-canonical form of translation called repeat-associated non-ATG (RAN) translation, resulting in neurotoxic dipeptide repeat proteins (DPRs). This has led to the currently predominant idea of a toxic gain-of-function (GOF) causing C9ORF72-ALS. However, the majority of mouse models overexpressing HRE or DPRs failed to develop ALS symptoms, in particular dying-back pathology, even though the mice showed significant levels of DPR proteins (Chew et al., 2015). Thus, both KO and overexpression mouse models could not recapitulate ALS phenotypes, arguing against simple LOF or GOF models and raising the need of a human disease model. A cooperation of LOF and GOF mechanisms driving C9ORF72-ALS pathogenesis was recently suggested (Shi et al., 2018). Yet, to date, no model is available that combines both LOF and GOF approaches endogenously.
Here, we used CRISPR/Cas9n gene editing to generate isogenic human induced pluripotent stem cells (iPSCs) and differentiated them into MNs for disease modeling, to better understand the mechanism by which the HRE in C9ORF72 causes ALS. We observed that MNs with the HRE showed decreased axonal trafficking, increased stress granule (SG) formation and significant transcriptome alterations compared with gene corrected MNs. To test the possibility of an LOF, we generated a C9ORF72 KO using a healthy control line. However, MNs lacking C9ORF72 protein showed no significant transcriptomic changes compared with isogenic controls, and axonal trafficking was similar in both wild-type (WT) MN lines (wtKO), suggesting that the HRE does not cause ALS via a simple LOF mechanism. However, rather than a simple GOF, it is possible that reduction of C9ORF72 protein levels exacerbates a toxic GOF associated with HRE. To test this, we generated iPSCs having both an HRE as well as C9ORF72 KO. We observed that those MNs showed increased apoptosis, disrupted axonal trafficking and aberrant transcriptome expression compared with MNs with HRE alone. Interestingly, these altered phenotypes were associated with decreased production of heat shock proteins (HSPs), particularly HSP70 as well as DNAJA4, a member of the HSP40 family, rather than increased DPR protein levels. Therefore, we propose that the HRE causes MN degeneration primarily via a toxic GOF that is exacerbated in HRE + C9ORF72 KO MNs, which is associated with reduced levels of HSPs.
Results
Generation of Isogenic iPSCs for Modeling C9ORF72-ALS
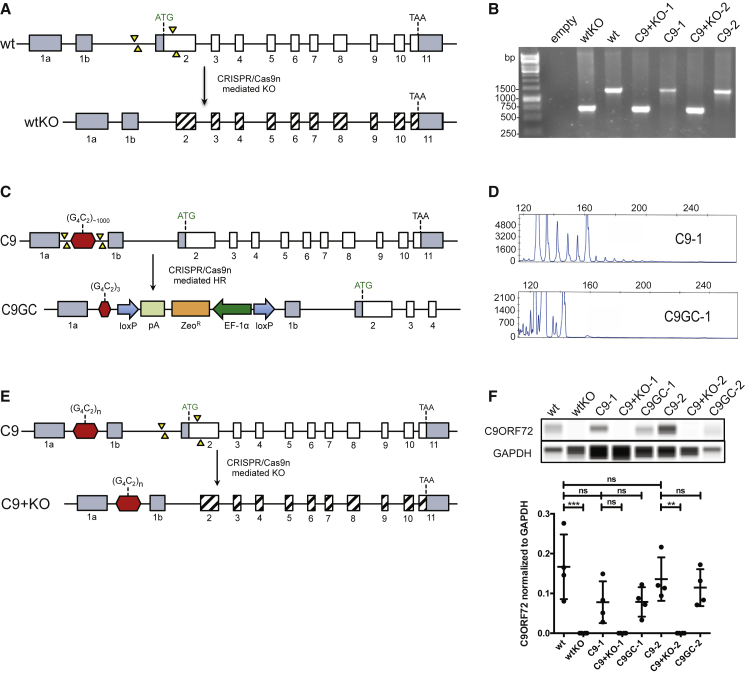
One of the critical outstanding questions is whether an HRE in C9ORF72 cause degeneration of human MNs via an LOF or a toxic GOF. To answer this question, we generated a series of isogenic iPSCs. To test if MN degeneration is caused by reduced levels of C9ORF72 protein, we generated a KO of C9ORF72 in WT cells using a quadruple Cas9-nickase (Cas9n) approach that introduced two different double-strand breaks surrounding the translational start codon in exon 2, which is used for all isoforms of C9ORF72 (Figure 1A). Cas9n was chosen because two different Cas9 proteins must cut in close proximity on opposing DNA strands to introduce a double-strand break. Guide RNAs were selected that would, together, only cut once in the genome, thus eliminating off-target effects. PCR-based genotyping was initially used to confirm deletion of the start codon of C9ORF72 (Figure 1B). The resulting iPSCs were designated wtKO.
Figure 1.
Generation of Isogenic iPSC Lines
(A) Strategy for targeting C9ORF72 to knockout C9ORF72 protein production in WT iPSCs (wtKO). Quadruple Cas9-nickase (Cas9n) introduced two double-strand breaks (yellow arrows).
(B) PCR confirmed the deletion in KO iPSC lines.
(C) Strategy for gene correction by reducing HRE to WT length of three repeats.
(D) Repeat-primed PCR confirmed absence of HRE in C9GC lines.
(E) Scheme of KO deletion in iPSCs with HRE in C9ORF72 (C9 + KO).
(F) Capillary electrophoresis confirmed loss of C9ORF72 protein in KO cells. Note that no significant differences between C9-1 and C9-1 in comparison with WT were not significant. N = 4 biological replicates. All values are presented as mean ± SEM. One-way ANOVA showed p < 0.05. Tukey's post-test for multiple comparisons was performed (**p < 0.01, ***p < 0.001).
See also Figures S1–S3.
It is also possible that the HRE in C9ORF72 cause ALS in a GOF manner. To test this, we used CRISPR-Cas9n-mediated gene editing to correct the HRE in iPSCs from two different ALS patients with heterozygous HRE in C9ORF72 using a donor vector that contained the WT length of three repeats (Figure 1C). Unmodified ALS patient-derived iPSCs were designated C9-1 and C9-2, and their gene corrected isogenic lines were designated C9GC-1 and C9GC-2 (Figure S1A). Repeat-primed PCR demonstrated the presence of the HRE in C9-1 and C9-2, but not in C9GC-1 and C9GC-2 iPSCs (Figures 1D and S1C). Southern blotting validated these results and determined repeat length (Figure S1B). Finally, amplicon length demonstrated C9GC-1 and C9GC-2 iPSCs were heterozygous, containing 5/3 and 2/3 repeats, respectively (Figure S1D).
Finally, we hypothesized that reducing C9ORF72 protein levels could exacerbate the pathological effects of a GOF. To test this, we used a quadruple-nickase approach to delete the translational start codon in an ALS patient iPSC line, resulting in retained repeat expansion in combination with a loss of C9ORF72 protein. These gene-edited iPSCs are hereafter called C9 + KO-1 and C9 + KO-2 (Figure 1E). PCR-based genotyping confirmed the presence of the deletion in C9ORF72 (Figure 1B).
qRT-PCR analysis was used to characterize the effects of gene editing on the expression levels of isoforms of C9ORF72as well as antisense RNA levels. Differentiated MNs from all iPSC lines expressed comparable levels of isoform 1, which contains the HRE (Figures S2A and S2B), as well as isoform 2, which encodes the long form of C9ORF72 protein (Figures S2A–S2C). Isoform 3 was not detected. Antisense transcripts were detected in all differentiated cultures of MNs, but no significant differences were observed (Figure S2D).
Capillary electrophoresis was used to quantify the levels of C9ORF72 protein in differentiated MNs (Figure 1F). We observed considerable variation in the levels of C9ORF72 protein. Relative to GAPDH, the levels in WT varied by more than 3-fold. Due to one particular data point, the average C9ORF72 protein levels was higher in WT compared with C9-1 and C9-2. However, due to the considerable heterogeneity, this difference was not significant, more likely due to stochastic differences between cultures than the HRE. Previous studies reported (Frick et al., 2018, Shi et al., 2018) that patient CNS tissue showed statistically significant reductions in C9ORF72 compared with controls. Thus, further reductions in C9ORF72 protein levels are needed in iPSC-derived neurons to recapitulate the conditions observed in patients. Therefore, the comparison between C9 and C9 + KO is of considerable interest, and, in agreement with our genotyping data, C9ORF72 protein was detected in MNs differentiated from C9-1 and C9-2, but not in wtKO, C9 + KO-1 and C9 + KO-2. Of note, multiple efforts to excise the selection cassette at the loxP sites were not successful due to insufficient recombinase activity. As a result of the selection cassette being present, the C9ORF72 protein levels were similar between the gene corrected lines and their respective isogenic parent (Figure 1F). The comparable levels of C9ORF72 protein means that phenotypic differences between C9 and C9GC lines cannot be caused by an LOF mechanism, making them ideal tools to assess possible GOF mechanisms.
Quality control was performed for all iPSC lines. Immunofluorescence confirmed that all lines homogeneously expressed the pluripotent stem cell markers OCT4 and SOX2 (Figure S3A). Karyotyping was performed for all iPSC lines (Figure S3B) showing that WT-, wtKO-, C9-1-, and C9-1-derived lines were euploid, but C9-2 contained three copies of chromosome 20, which was equally present in all iPSCs derived from C9-2. However, it will be demonstrated in the following sections that the majority of observed phenotypes were comparable in both C9-1- and C9-2-derived lines, indicating that the additional chromosome had only a minor effect on ALS phenotypes. These data confirm that the iPSC lines are isogenic and express markers of pluripotency.
Differentiation of Functional MNs from Isogenic iPSCs
In preparation for modeling ALS, each iPSC line was differentiated into MNs using small molecules (Figure 2A). Immunostaining showed that almost all of the cells in the differentiated cultures expressed the neuronal marker MAP2 as well as the MN marker SMI32 (Figure 2B). In addition, approximately 50%–70% of all neurons expressed the MN marker Islet1 (Figure 2B). There was no significant difference between cell lines independent of the genetic background (Figures 2C and S4A). In addition, MAP2 and MN markers SMI32 and CHAT were quantified using qRT-PCR and showed similar expression levels between the isogenic cells derived from C9-1 (Figure S4B). To better characterize the cultures, we performed Eigengene classification of transcriptome data, which identified the cells in these cultures with whole spinal cord with 80% probability (Figure S5A). Analysis of neuronal subtype markers showed that the cultures contained mostly MNs and also spinal interneurons (Figure S5B).
Figure 2.
Differentiation of iPSCs into Functional MNs
(A) Differentiation scheme.
(B) MNs express the neuronal marker MAP2 (green) and the MN markers Islet1 (red) and SMI32 (cyan). Scale bar, 100 μm.
(C) MN differentiation efficiency is comparable between all cell lines (n = 3 biological replicates). One-way ANOVA showed no statistical significance.
(D–F) Representative voltage-gated sodium inward and potassium outward currents of an MN recorded in whole-cell voltage-clamp mode (D). Peak sodium currents INa at maximal amplitude (E) and (F) at −40 mV holding potential.
(G) Recording of a C9GC-1 MN spiking repetitive spontaneous action potentials (APs).
(H and I) Number of spontaneously active MNs (H) and their AP frequency is significantly higher in C9GC-1 than C9-1 MNs (I).
(J) A C9GC-1 MN firing multiple APs upon depolarization in the current-clamp mode.
(K and L) The number of MNs with multiple elicited APs (K) and the maximal amplitudes of elicited APs were most pronounced in C9GC-1 cells (L). A minimum of n = 32 cells was measured per line. All values are presented as mean ± SEM. Statistical analysis was performed using one-way ANOVA followed by Tukey's post-test for multiple comparisons (∗p < 0.05, ∗∗p < 0.01).
See also Figures S4 and S5.
Next, we tested the electrophysiological functionality of the isogenic MNs. Whole-cell patch-clamp analysis demonstrated the development of essential functional properties during MN maturation. There was no significant difference between isogenic WT and wtKO cells (Figures S4C–S4H). Voltage-gated sodium inward and potassium outward currents were comparable between the C9-1-derived cell lines (C9-1, C9 + KO-1, and C9GC-1) although peak sodium current amplitudes were more pronounced in C9GC-1 MNs (Figures 2D–2F). The number of spontaneously active MNs and their action potential (AP) frequency were significantly decreased in C9-1 MNs compared with its isogenic control C9GC-1 (Figures 2G–2I). In addition, the number of MNs with multiple APs and the maximal amplitudes of elicited APs were most pronounced in C9GC-1 MNs (Figures 2J–2L), which is consistent with previously published reports of a hypoexcitability phenotype in HRE carrying MNs (Devlin et al., 2015, Sareen et al., 2013).
Interestingly, Devlin et al. (2015) reported a switch from hyperexcitability (Wainger et al., 2014) to hypoexcitability during differentiation of iPSC-derived MNs with mutations in the genes TARDBP and C9ORF72. Although in a recent study the excitability in mutant C9ORF72 iPSC-derived MNs, healthy and isogenic controls was not significantly different (Selvaraj et al., 2018), we found a hypoexcitability phenotype in mutant C9ORF72 MNs (C9-1) compared with their control (C9GC-1), which was also seen in other mutant ALS genotypes (Naujock et al., 2016, Naumann et al., 2018, Zhang et al., 2013).
Gene Correction and Transcriptome Sequencing Suggests that HRE Induces MN Degeneration via a GOF
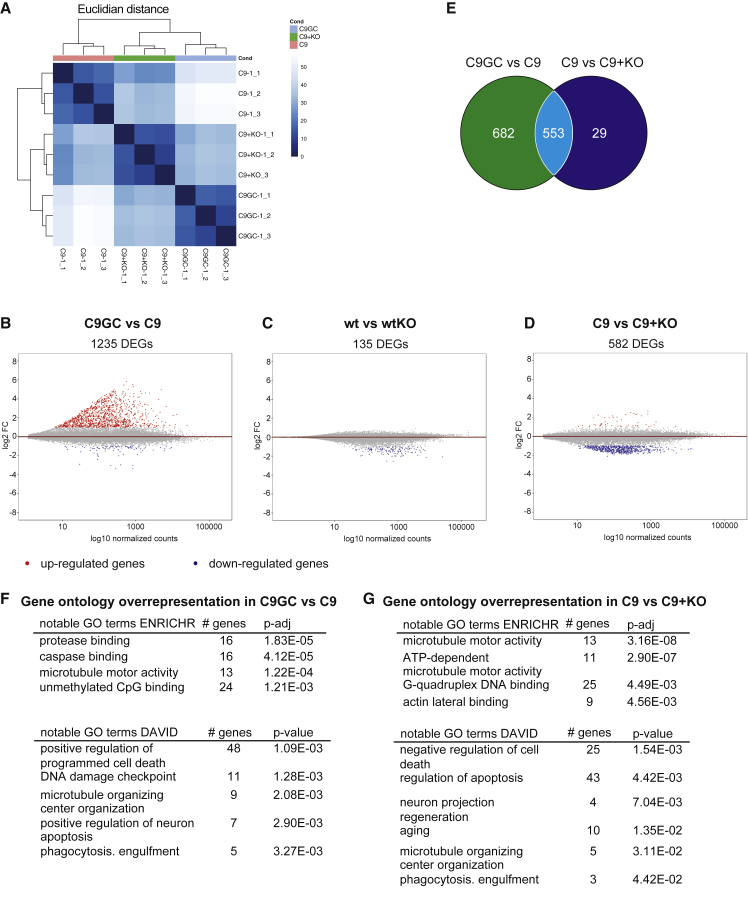
As an unbiased approach, we used transcriptome sequencing to determine differences in gene expression and pathways affected by the HRE (Figure 3A). We compared transcriptome profiles of C9-1 and C9-1GC MNs. A total of 1,235 genes were differentially expressed (Figure 3B) of which the majority were upregulated in C9-1 (Figure S6A). Gene ontology analysis showed enrichment of multiple terms related to ALS pathogenesis, including positive regulation of programmed cell death, DNA damage checkpoint, microtubule organization, and microtubule motor activity (Figures 3F, S6G, and S6I).
Figure 3.
Transcriptome Analysis of Isogenic MNs
(A) Heatmap and hierarchical clustering of transcriptome results from the indicated MNs.
(B–D) MA plots depict significantly (p < 0.05) upregulated (red) and downregulated genes (blue). (B) shows C9GC versus C9, (C) shows wt versus wtKO, and (D) shows C9 versus C9 + KO.
(E) Venn diagram shows numbers of differentially expressed genes (DEGs) between C9GC versus C9 MNs (green), between C9 versus C9 + KO MNs (blue), and the overlap of both comparisons (cyan).
(F and G) Gene ontology studies on DEGs between (F) C9GC versus C9 and (G) C9 versus C9 + KO using ENRICHR (top) and DAVID (bottom).
See also Figure S6.
Next, the transcriptome of WT and wtKO MNs was analyzed (NCBI GEO: GSE143744). The LOF model predicts that transcriptome changes in wtKO cells compared with WT would be comparable with HRE-associated changes. In contrast, the GOF model predicts HRE-associated changes would not be recapitulated in wtKO neurons compared with WT. We observed that only 135 genes were differentially expressed in wtKO cells compared with WT (Figure 3C). Although there was some overlap with the genes altered in C9 versus C9GC (Figure S6B), gene ontology analysis could not identify any significantly enriched terms. This suggests that C9ORF72 LOF alone does not cause neurodegeneration.
We hypothesized that reduced levels of C9ORF72 would exacerbate the pathogenicity of a toxic GOF. To test this, we sequenced the transcriptome of MNs differentiated from C9 + KO-1 iPSCs and compared it to isogenic C9-1 MNs (NCBI GEO: GSE143743). We identified 582 differentially expressed genes (Figure 3D), of which 553 were also differentially expressed between C9GC-1 versus C9-1 (Figure 3E), demonstrating a considerable overlap. Interestingly, most of these overlapping proteins were downregulated in C9 + KO compared with C9 (Figures S6C and S6E). There was some overlap with the genes differentially regulated between WT and wtKO (Figure S6D). Gene ontology analysis showed enrichment for terms related to MN degeneration, including negative regulation of cell death, regulation of apoptosis, microtubule motor activity and neuron projection regeneration (Figures 3G, S6H, and S6J). These results could suggest that C9ORF72 KO might exacerbate the pathogenicity of HRE. This is underpinned by the fact that rather all differentially expressed genes between C9 and C9 + KO overlap with genes dysregulated in C9GC versus C9, which is, however, not the case vice versa (Figure 3E).
Previously, Selvaraj et al. (2018) reported transcriptome analysis of MNs differentiated from isogenic C9ORF72 iPSCs. Unexpectedly, we found very little overlap with our results (Figure S6F). However, it is known that C9ORF72 pathology, phenotypes, and penetrance differ from patient to patient, suggesting that HRE-mediated transcriptome changes are strongly dependent on the genetic background. Interestingly, one of genes downregulated by HRE in both datasets was DNAJA4, which is an HSP40 family member, and could suggest that HSPs play an important role in the pathogenesis of C9ORF72-ALS.
HRE Induces Distal Axon Trafficking Defects that Are Exacerbated in Neurons with HRE + C9ORF72 KO
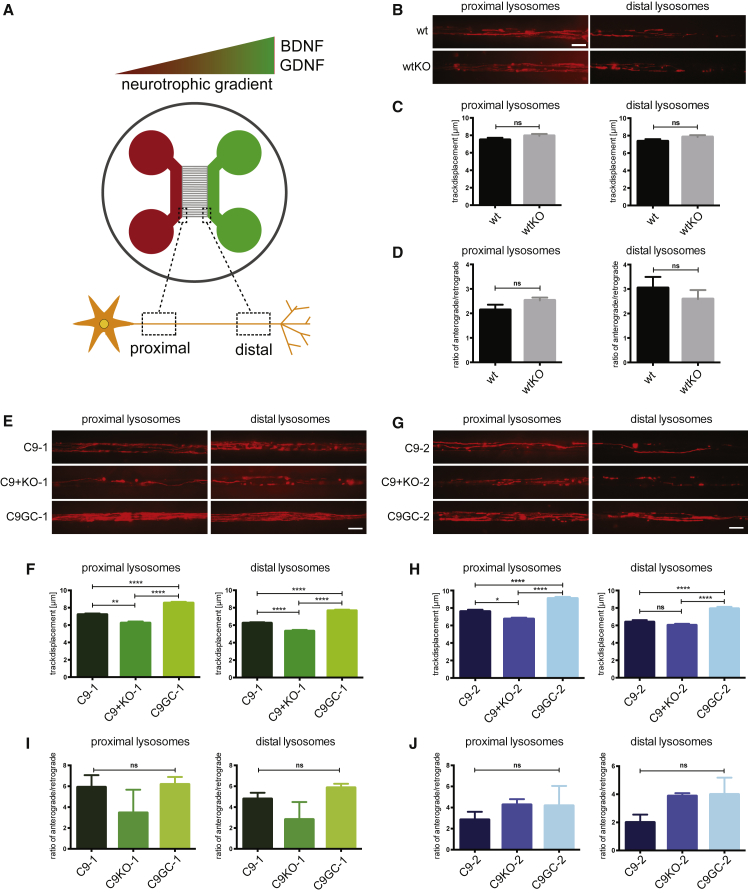
During ALS, MNs undergo dying-back in which distal axon degeneration precedes MN death (Naumann et al., 2018). A possible reason for the distal axon degenerating first is a disruption of axonal transport, which is used to deliver RNA, proteins and even organelles to the synapse to maintain its function. Mutations in motoric proteins, such as KIF5A and DCTN1 have been associated with ALS (Nicolas et al., 2018, Puls et al., 2003) and our transcriptome data suggested that axonal trafficking is affected by HRE as well as HRE + C9ORF72 KO, which could contribute to dying-back pathogenesis. As C9ORF72 protein function is linked to autophagy and lysosomes (Shi et al., 2018), we focused on lysosomal trafficking in MN axons using microfluidic devices. MNs were seeded into one side of the device, defined as “proximal,” and axons grew through channels to the distal side attracted by brain-derived neurotrophic factor (BDNF) and glial cell line-derived neurotrophic factor (GDNF) (Figure 4A). Lysosomes were labeled using LysoTracker, and live imaging of the axons was performed to assess the distance traveled, referred to as track displacement. Similar to our transcriptome data, WT and wtKO cells did not show any differences in the track displacement or directionality of lysosomal trafficking at either the proximal or the distal axon (Figures 4B–4D), suggesting that the HRE does not cause neurodegeneration via a simple LOF.
Figure 4.
Axonal Trafficking Is Altered by the HRE and Loss of C9ORF72
(A) Schematic representation of microfluidic chambers used for live-cell imaging.
(B) Maximum projection for WT and wtKO MNs of 400 frames acquired within a 2-min movie. Higher signal correlates with longer track displacement. Scale bar, 10 μm.
(C and D) Quantification shows (C) track displacement of lysosomes in μm and (D) direction of trafficking. N = 3 biological replicates; >1,000 lysosomes were analyzed per cell line and per side for all experiments.
(E) Maximum projection for C9-1 MNs. Scale bar, 10 μm.
(F) Quantification shows track displacement of C9-1 lysosomes in μm.
(G) Maximum projection for C9-2 MNs. Scale bar, 10 μm.
(H) Quantification shows track displacement of C9-2 lysosomes in μm.
(I and J) Quantification of direction of lysosomal trafficking. N = 3 biological replicates; >1,000 lysosomes were analyzed per cell line and per side for all experiments. Lysosome trafficking results are shown in (I) for C9-1, C9KO-1, and C9GC-1, and in (J) for C9-2, C9KO-2, and C9GC-2.
All values are presented as mean ± SEM. One-way ANOVA followed by Tukey's post-test for multiple comparisons was performed (∗∗p < 0.01, ∗∗∗p < 0.001, ∗∗∗∗p < 0.0001). Tracks were shorter at the distal axon end than on the proximal compartment, consistent with dying-back.
Next, we assessed axonal trafficking in MNs with the HRE in C9ORF72. We found that MNs from C9-1 and C9-2 showed significantly decreased lysosomal track displacement in distal as well as proximal axons compared with their gene corrected counterparts (Figures 4E–4H). Remarkably, lysosomes in the axons of C9 + KO-1 and C9 + KO-2 showed even shorter track displacement in proximal and distal axons, demonstrating that C9ORF72 KO exacerbated axonal trafficking defects of the HRE. No significant alterations were observed in the directionality of lysosomal trafficking (Figures 4I and 4J). These data demonstrate that the HRE induces degeneration of the distal MN axon before the proximal axon, which is consistent with dying-back pathogenesis.
C9ORF72 KO Exacerbates MN Degeneration in HRE MNs
In ALS patients, axonal degeneration is followed by the death of the MN soma. Because loss of C9ORF72 protein exacerbates defects in axonal transport in MNs containing an HRE, we predicted that MNs with HRE and deleted C9ORF72 protein would show the highest levels of apoptosis. Consistent with this idea, our transcriptome data showed that gene ontology terms related to neuronal death and apoptosis were enriched in MNs with HRE + C9ORF72 KO. Thus, we investigated apoptosis in differentiated MNs. To do this, iPSC-derived MNs were cultured for 14 days and immunostained for TUJ1 and the late apoptotic marker cleaved caspase-3 (CC3) (Figure 5A). Quantification showed that CC3 levels in C9 + KO-1 were more than 2-fold of CC3 levels in C9-1 and C9GC-1, which were comparable (Figure 5B). Similarly, C9 + KO-2 showed higher levels of CC3 compared with C9-2 and C9GC-2. These data demonstrate that deleting C9ORF72 protein increases apoptosis in MNs with HRE in C9ORF72.
Figure 5.
HRE + C9ORF72 KO Neurons Show Increased Apoptosis without Increasing DPR Protein Levels
(A) Immunostaining for the indicated markers. Images were segmented using CellProfiler to quantify cleaved caspase-3 (CC3), a marker of apoptosis. Scale bar, 50 μm.
(B) C9 + KO MNs show more apoptosis compared with gene corrected C9GC (n = 3 biological replicates).
(C) Quantification of endogenous poly-GP DPR levels using an ELISA assay (n = 7 biological replicates).
(D) Immunostaining for the indicated markers, including MAP2 and poly-GP DPR proteins. Poly-GP peptides could be detected in the cytoplasm of C9 and C9 + KO MNs (indicated by arrows) but not in gene corrected controls. Isogenic cell lines set 1 (left) and set 2 (right). Scale bars, 10 μm.
All values are presented as mean ± SEM. One-way ANOVA followed by Tukey's post-test for multiple comparisons was performed (∗p < 0.05, ∗∗p < 0.01, ∗∗∗p < 0.001, ∗∗∗∗p < 0.0001).
DPR Protein Levels Are Associated with HRE but Not Increased by C9ORF72 KO
Transcriptomics and axonal trafficking demonstrate the association of the HRE with a toxic GOF, and DPR proteins translated from HRE-containing RNA are currently the best and most discussed candidate for the toxic GOF. For this reason, we used an ELISA immunoassay to quantify the endogenous levels of poly-GP DPR proteins in differentiated MNs. As expected, no poly-GP protein was detected in WT, wtKO and the gene corrected lines C9GC-1 and C9GC-2 (Figure 5C). In contrast, C9-1 and C9-2 MNs expressed significantly higher levels of poly-GP compared with isogenic controls. However, there was no significant difference in the levels of poly-GP protein between C9-1 and C9-2, despite the different lengths of repeat expansion (C9-1 having ∼1,000 repeats; C9-2 having ∼750 repeats). Immunostaining was used to validate the presence of DPR proteins in iPSC-derived MNs. In contrast to gene corrected controls, we consistently observed punctate staining in the cytoplasm and neurites of MNs carrying an HRE (Figure 5D). However, the high level of unspecific staining made the immunostaining images difficult to quantify. Interestingly, ELISA results showed that C9 + KO-1 and C9 + KO-2 expressed about 40% less poly-GP protein compared with their respective parental lines (Figure 5C), which is particularly surprising as we previously found that MN degeneration was highest in these lines. This clearly indicates that C9ORF72 KO exacerbates MN degeneration without increasing DPR protein levels and suggests that other factors influence the pathology of C9ORF72-ALS. It is interesting to note that the decrease in DPR levels in C9 + KO versus C9 MNs correlates with an apparent reversion in the expression of genes differentially expressed in MNs with HRE (Figures S6A and S6C), suggesting that DPR levels may play a causal role in the expression of many differentially expressed genes in iPSC-derived MNs.
Since C9ORF72 protein functions in autophagy, we expected that DPR protein levels would be increased instead of decreased. To better understand the possible mechanism for this decrease, we further characterized the deletion present in C9ORF72 KO cells. The HRE residing in the first intron is normally spliced out of the mature C9ORF72 mRNA. However, in addition to removing the start codon, the splice acceptor site was also removed (Figure 1E). Thus, we predicted that mature mRNA transcript for C9ORF72 would contain the HRE. To test this hypothesis, transcriptome reads mapping to the intron were examined and C9 + KO-1 showed considerably more reads mapping to the intron compared with C9-1 (Figure S7A). A recent report showed retention of intron 1 decreases RAN translation due to the presence of an upstream open reading frame (uORF) (Tabet et al., 2018), but we did not observe any significant difference in the levels of the uORF transcript in C9 + KO MNs compared with C9 (Figure S2E). Nevertheless, it is possible that change in context of the HRE might contribute to the lower DPR levels observed in our C9 + KO-1 and C9 + KO-2 lines. Thus, we argue that a change in RNA splicing is likely responsible for the decrease in DPR protein levels.
HSP Levels Are Altered by HRE and Exacerbated by C9ORF72 KO
Next, we sought to identify a possible mechanism by which C9ORF72 KO exacerbated MN degeneration without increasing DPR levels. HSPs play a crucial role in the protein quality control system and recently it was shown that HSP70 and HSP40 were critical nodes in the transcriptome network in the frontal cortex of ALS patients with HRE in C9ORF72 (Prudencio et al., 2015). This led us to hypothesize that C9ORF72 KO could reduce the levels of specific HSPs, which might exacerbate the toxicity of DPR proteins for MNs. Measuring protein expression of HSP70 family members HSPA1A and HSPA1B using capillary electrophoresis revealed that C9-1 and C9-2 MNs expressed less HSPA1A and significantly less HSPA1B compared with the gene corrected controls C9GC-1 and C9GC-2 (Figures 6A and 6B). This reduction was even greater in C9 + KO-1 and C9 + KO-2. As indicated above, we found that DNAJA4, which is an HSP40 family member that forms complexes with HSP70, was downregulated in MNs with HRE compared with gene corrected controls in our data as well as in the results from Selvaraj et al. (2018). We observed a trend toward decreased DNAJA4 protein in HRE MNs compared with gene corrected, but there was a striking and significant decrease of DNAJA4 protein in C9ORF72 KO MNs, which showed only 10%–50% of the parental levels (Figure 6C). Therefore, changes in HSP levels correlate with the exacerbated ALS phenotypes, suggesting that they play a critical role in MN degeneration.
Figure 6.
HRE and Loss of C9ORF72 Reduce HSP40 and HSP70 Protein Levels and Alter SG Formation
(A and B) Capillary electrophoresis showing that (A) HSPA1A and (B) HSPA1B are reduced in C9 MNs compared with C9GC controls. Levels were even further reduced in C9 + KO MNs.
(C) Capillary electrophoresis showing that DNAJA4 is reduced in C9 MNs compared with C9GC, and strikingly downregulated in C9 + KO MNs (n = 8 biological replicates).
(D) Immunostaining of isogenic MNs with the indicated makers. Bottom panel shows segmentation of SGs, nuclei, and soma. Scale bar, 5 μm.
(E) SG mean intensity was unaltered in MNs within the isogenic sets.
(F) Quantification revealed significantly higher number of SGs per cell in C9 MNs compared with C9GC, which was increased in C9 + KO neurons (n = 3 biological replicates; >3,400 cells and >12,000 SGs).
All values are presented as mean ± SEM. One-way ANOVA followed by Tukey's post-test for multiple comparisons was performed (∗p < 0.05, ∗∗p < 0.01, ∗∗∗∗p < 0.0001, ∗∗∗∗p < 0.0001).
SG Formation Is Perturbed by HRE and Exacerbated by KO of C9ORF72
SGs have been suggested to be crucibles of ALS pathogenesis (Li et al., 2013). They are membraneless organelles formed by liquid-liquid protein phase separation. Although both DPR proteins as well as HSPs play critical roles in SG formation and maintenance, we predicted that SGs might be affected by the loss of C9ORF72 protein in MNs containing the HRE. Therefore, we assessed SG formation using our isogenic MNs. Differentiated MNs were treated for 1 h with 0.5 mM sodium arsenite to induce SG formation, which were assessed using immunostaining for the SG marker eukaryotic translation initiation factor 3 (eIF3) in combination with the neuronal marker MAP2. eIF3-positive SGs were observed in the cytoplasm and often in perinuclear regions (Figure 6D). The mean eIF3 intensity was comparable between the lines (Figure 6E). Quantification showed a significant increase of the number of SGs per cell with an HRE compared with gene corrected controls (Figure 6F). Importantly, the number of SGs per cell was even higher in cells with HRE + C9ORF72 KO, which is consistent with our previous results that C9ORF72 KO exacerbates ALS-associated phenotypes of HRE MNs. Therefore, we suggest that membraneless compartments, such as SGs, could be a point of pathological convergence of DPR proteins and reduced HSP levels leading to MN degeneration in C9ORF72-associated ALS.
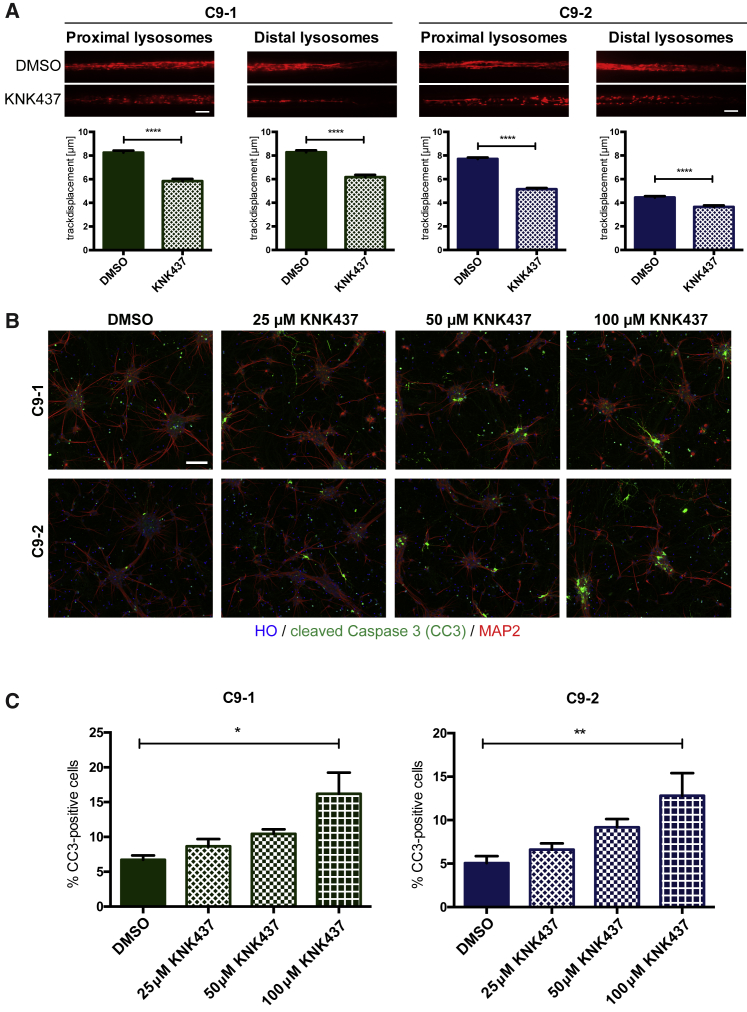
Inhibition of HSPs Exacerbates Axonal Trafficking Defects and Induces Apoptosis in MNs with HRE
C9 + KO MNs showed decreased HSP levels as well as exacerbated ALS phenotypes compared with C9 MNs, leading us to hypothesize that inhibiting HSPs would increase degeneration of C9 MNs. To test this, axonal trafficking was assessed using iPSC-derived MNs with the HRE treated with 200 μM KNK437, which inhibits the synthesis of HSPs, including HSP70 as well as HSP40 (Yokota et al., 2000). We observed that C9-1 as well as C9-2 MNs showed significantly reduced lysosomal track displacement in the proximal and distal axons (Figure 7A). In addition, the directionality of lysosomal trafficking was severely affected (Figure S7B). To confirm that HSPs are causally linked to the survival of MNs with HRE, we assessed apoptosis using immunostaining for CC3 (Figure 7B). We observed a significant and dose-dependent increase in CC3 staining of iPSC-derived C9-1 and C9-2 neurons treated with KNK437 (Figure 7C). These data demonstrate that HSPs play a critical role in MN survival and that knocking out C9ORF72 exacerbates ALS phenotypes in iPSC-derived MNs with HRE, which is associated with reduced levels of specific HSPs.
Figure 7.
HSP Inhibitor KNK437 Disrupts Axonal Trafficking and Induces Apoptosis in MNs with HRE
(A) Maximum projection for C9-1 and C9-2 MNs treated with either DMSO or 200 μM KNK437. Scale bars, 10 μm. Quantification for lysosomal track displacement in μm. Mann-Whitney test was performed for statistical significance (∗∗∗∗p < 0.0001). N = 6 biological replicates; >500 lysosomes were analyzed per cell line and per side for all experiments.
(B) Immunostaining for CC3 for C9-1 and C9-3 MNs treated with DMSO or the indicated concentration of KNK437. Scale bar, 50 μm.
(C) Quantification shows percent CC3-positive cells. N = 4 biological replicates. One-way ANOVA was used to calculate statistical significance (∗p < 0.05, ∗∗p < 0.01).
See also Figure S7.
Discussion
To prevent ALS onset and progression, it is essential to prevent the degeneration of motor axons in addition to soma. This was illustrated using mice with mutant SOD1 where a KO of the gene Ppif, which encodes cyclophilin D, had no impact on disease onset, progression or survival although SOD1 protein aggregation was reduced and increased numbers of MN soma were protected in the spinal cord (Parone et al., 2013). Here, we demonstrate that axonal degeneration is a feature of the degeneration of MNs with HRE in C9ORF72, which is the most common mutation known to cause ALS.
Currently, it is not fully understood whether HRE in C9ORF72 cause ALS via LOF or GOF. Thus, isogenic control iPSC lines are powerful tools enabling the generation of MNs with specific genotypes in a defined genetic background to identify pathological mechanisms (Selvaraj et al., 2018). We produced a complete set of isogenic human iPSC-derived MNs to assess the contributions of LOF and GOF to ALS phenotypes. Because C9ORF72 transcript levels are reduced by the HRE, which can also retain the repeat expansion in ALS patients (Niblock et al., 2016), it is possible that both mechanisms contribute to pathogenesis. To test the possibility that reducing C9ORF72 protein levels could exacerbate ALS pathogenesis in MNs with HRE, we created isogenic MNs with a C9ORF72 KO as well as HRE. Importantly, this model produces DPR protein at endogenous levels and without any overexpression, which rely on promoters, such as the CMV-promoter, which usually results in RNA and protein levels that are higher than physiological levels. Moreover, we created an LOF model (wtKO) as well as a model for toxic GOF in which isogenic MNs with and without the HRE express similar levels of C9ORF72 protein. Our results show that axonal degeneration associated with HRE involves both GOF and LOF mechanisms by demonstrating that C9ORF72 KO exacerbated the effects of GOF caused by the HRE but the KO itself does not affect WT MNs. This exacerbation could have important implications for ALS therapeutics. Currently, antisense oligonucleotides (ASOs) are being developed to reduce the HRE-containing intronic RNA and downstream DPR protein levels (Jiang et al., 2016). Our results demonstrate the importance that these ASOs avoid unintentional reduction of C9ORF72 protein levels to maintain physiological levels of C9ORF72.
To better understand the mechanism of pathogenesis, we performed transcriptome sequencing, which identified over 1,000 differentially expressed genes between C9 versus C9GC MNs, most of which were upregulated. Interestingly, we found that many of these genes were downregulated in C9 + KO versus C9 MNs. One possible explanation is that DPR proteins induced this differential gene expression, and DPR levels were lower in C9 + KO MNs. However, another explanation could be that these differentially expressed genes are part of a compensatory response used by MNs to maintain viability in the presence of the HRE and that knocking out C9ORF72 protein may ablate this compensatory response, leading to more severe phenotypes. Both scenarios would explain why no phenotype was observed in wtKO MNs.
To our knowledge, our results are the first to report that decreased HSP activity could contribute to C9ORF72-ALS pathogenesis. Comparing our transcriptome data with those published by Selvaraj et al. (2018) showed that the HSP40 family member DNAJA4 was downregulated in both datasets, suggesting that HSPs play an important role in C9ORF72-ALS. We confirmed that DNAJA4 protein was misregulated, and showed that HSPA1A and HSPA1B were also misregulated. Consistent with this, transcriptome sequencing from the brain of C9ORF72-ALS patients show misregulation of HSPs (Prudencio et al., 2015). We showed that inhibiting HSPs significantly exacerbated C9ORF72-ALS phenotypes, including disrupting axonal transport and increasing apoptosis of MNs. Thus, it is possible that the most effective therapeutic strategy might be to combine ASOs, which aim at reducing DPR protein levels, together with the restoration of HSP levels, for example, using a gene therapy-based approach or a compound, such as arimoclomol, which is a co-inducer of HSPs currently being investigated in clinical trials for ALS that has shown promising results in mouse models (Benatar et al., 2018). However, the possibility has been raised that at least some HSPs might exacerbate ALS pathogenesis. For example, overexpression of HSF1, activating a heat shock response found in C9ORF72-ALS/FTLD patients, exacerbated eye degeneration in a Drosophila model (Mordes et al., 2018). Thus, further experiments are needed to test the role of specific HSPs in ALS phenotypes in human MNs.
One of the important functions of HSPs in human MNs is the regulation of SGs (Ganassi et al., 2016), which are crucial players in ALS pathogenesis. In addition, DPR proteins have already been shown to disrupt SG regulation (Lee et al., 2016), and, here, we demonstrate that HRE + C9ORF72 KO results in reduced levels of specific HSPs, exacerbating the SG phenotype. We found that the number of SGs per cell was increased in MNs with an HRE and even higher in MNs with HRE + C9ORF72 KO. Consistent with our data, a recent report demonstrated that removal of C9ORF72 led to increased accumulation of SGs in the cytoplasm and disrupted SG elimination by autophagy (Chitiprolu et al., 2018). This is of particular interest as it has been shown that disruption of microtubule transport proteins affects SG dynamics (Loschi et al., 2009). Because these same proteins also regulate axonal transport, it suggests that there is a general defect in microtubule-based transport in MNs with HRE in C9ORF72. Since SGs tend to accumulate misfolded proteins, this could indicate that MNs with HRE in C9ORF72 have defects in protein homeostasis and that C9ORF72 KO exacerbates these effects, which is consistent with our observation that HSPs were downregulated.
One interesting point is the existence of variability in how human MNs respond to reduced C9ORF72 levels. At least one group found that reducing C9ORF72 levels in human MNs led to degeneration (Shi et al., 2018). Although this is a different assay than our assessment of axonal trafficking, the differences in the severity of the phenotypes are noticeable. We suggest that variations in the genetic background could explain the different observations. Unlike mutations in rarer ALS loci, such as FUS or SOD1, HRE in C9ORF72 show age-dependent penetrance and do not achieve full penetrance until a very advanced age (Majounie et al., 2012). Even more striking, members of a single family inheriting the same mutation can exhibit different diseases, namely ALS or frontotemporal dementia or, occasionally, a mixture (Simon-Sanchez et al., 2012). Thus, the genetic background and, perhaps, environmental agents, can significantly impact the phenotype of patients. For example, ATXN2, TMEM106B, and SPT4 have already been shown to be critical modulators of C9ORF72 pathogenesis (van Blitterswijk et al., 2014). This implies that iPSCs could be combined with human genetics and gene editing to tease apart the influences of other loci on the phenotypes of human MNs with HRE in C9ORF72.
Experimental Procedures
A complete description of the methods can be found in the Supplemental Information.
iPSC Culture and MN Differentiation
iPSC generation and MN differentiation is described in Supplemental Information.
Immunocytochemical Staining
MNs were fixed for 20 min with 4% paraformaldehyde (EM Science) in PBS (Life Technologies). Permeabilization and blocking were achieved with 0.1% Triton X-(Sigma), 10% fetal calf serum (GE Healthcare) and 1% BSA (Roth) in PBS for 45 min and primary antibodies incubated overnight at 4°C. The cells were washed twice with 1% BSA in PBS and secondary antibodies were applied for 1 h at room temperature. The cells were washed twice with 1% Tween 20 (AppliChem) in PBS, then incubated with Hoechst for 5 min and washed with PBS. Imaging was performed using an inverted Apotome Zeiss Axio/Observer Z1 microscope (Zeiss) and analyzed using FIJI and Adobe Photoshop. The following primary antibodies were used: chicken anti MAP2 1:10,000 (Abcam, ab92434), goat anti-eIF3 1:300 (Santa Cruz, sc-16377), goat anti-SOX2 1:300 (Santa Cruz, sc-17320), mouse anti-OCT4 1:300 (Santa Cruz, sc-5279), mouse anti-SMI32 1:1,000 (Covance, SMI-32P), mouse anti-SSEA4 1:300 (DSHB, MC-813-70), mouse anti-TUJ1 1:1,000 (Covance, MMS-435P-250), rabbit anti-CC3 1:500 (Cell Signaling, 9661S), rabbit anti-Islet1 1:1,500 (Abcam, ab20670), rat anti-poly-GP 18 1:1,000 (kindly provided by Dieter Edbauer). The following secondary antibodies were diluted 1:1,000 (all Invitrogen): fluorescein isothiocyanate donkey anti-chicken (SA1-72000), Alexa Fluor 647 donkey anti-goat (A-21447), Alexa Fluor 488 donkey anti-mouse (A-21202), Alexa Fluor 568 donkey anti-mouse (A10037), Alexa Fluor 647 donkey anti-mouse (A-31571), Alexa Fluor 488 donkey anti-rabbit (A-21206), Alexa Fluor 568 donkey anti-rabbit (A10042), Alexa Fluor 647 donkey anti-rabbit (A-31573), Alexa Fluor 568 goat anti-rat (A-11077), Hoechst 33342 (H1399).
Live-Cell Imaging and High Content Imaging
The technique has already been described (Pal et al., 2018). Microfluidic chambers (Xona Microfluidics) were used with glass bottom dishes coated with laminin. MNs (150,000) were seeded, and N2B27 with BDNF and GDNF was applied to the distal side. At day 14, lysosomes were labeled using LysoTracker (Thermo Fischer Scientific) and imaged using a Leica DMI6000 microscope in an incubator chamber.
Capillary Gel Electrophoresis
MNs were lysed in a 4% SDS buffer supplemented with 50 mM Tris base (Roth), protease- and phosphatase inhibitor (Roche), and protein concentration determined using a bicinchoninic acid assay (Thermo Fisher Scientific). Protein expression levels were measured with capillary electrophoresis using a 12-230 separation module from ProteinSimple Wes (ProteinSimple, Bio-Techne). Utilized primary antibodies included: rabbit anti GAPDH 1:1,000 (Cell Signaling, 2118S), rabbit anti-DNAJA4 1:40 (Atlas Antibodies, HPA041790), rabbit anti-HSPA1A 1:1,000 (BIOZOL, GTX111088), rabbit anti-HSPA1B 1:100 (BIOZOL, GTX106148), rabbit anti-C9ORF72 1:30 (Sigma, HPA023873).
Author Contributions
M.A.-R., J.L.S., and F.W. designed the research. M.A.-R. performed gene editing, disease modeling, and phenotypic assays. F.W., S.P., S.Staege, and N.K. performed electrophysiology experiments and qRT-PCR. A.P. and A.H. helped with axonal trafficking analysis. C.L. and D.E. performed ELISA essays and provided poly-GP antibody. W.J. and T.R. performed Southern blot, repeat-primed PCR and amplicon length PCR. M.B. and A.J. conducted high content imaging and analysis. S.Stefanov helped with transcriptome analysis. M.A.-R. and J.L.S. wrote the paper, which was critically revised by all authors.
Acknowledgments
This work was financed by DFG Research Center (DFG FZT 111) and Cluster of Excellence (DFG EXC 168). The Sterneckert lab is supported by the European Union's Horizon 2020 research and innovation program (643417) and the Bundesministerium für Bildung und Forschung (01ED1601B). This is an EU Joint Program – Neurodegenerative Disease Research (JPND) project. The project is supported by the following funding organizations under the aegis of JPND—www.jpnd.eu: Germany, Bundesministerium für Bildung und Forschung; Israel, Ministry of Health, Italy, Ministero dell’Istruzione, dell’Università e della Ricerca; Sweden, Swedish Research Council; and Switzerland, Swiss National Science Foundation. This project was additionally funded by the Robert Packard Center for ALS Research. F.W., S.P., and N.K. were supported by a grant of the Petermax-Müller-Stiftung (Hannover, Germany). This work was supported in part by the NOMIS Foundation (to D.E. and A.H.), the Helmholtz Virtual Institute “RNA dysmetabolism in ALS and FTD (VH-VI-510)” (to A.H.), BIOCREA GMBH (to A.H.), an unrestricted grant by the family of a deceased ALS patient via the Stiftung zur Förderung der Hochschulmedizin in Dresden (to A.H.) and by the Hermann und Lilly Schilling-Stiftung für medizinische Forschung im Stifterverband (to A.H.). We thank Prof. J. Rothstein and A.H. for providing patient-specific iPSCs. We thank G. Jurado Jiménez and S. Gholamhosseinian for their support as student assistants. We thank T. Levin for editing the manuscript. This work was additionally supported by the CRTD Light Microscopy facility as well as the next-generation sequencing facility, in particular, M. Lesche and A. Petzold.
Published: February 20, 2020
Footnotes
Supplemental Information can be found online at https://doi.org/10.1016/j.stemcr.2020.01.010.
Contributor Information
Florian Wegner, Email: wegner.florian@mh-hannover.de.
Jared L. Sterneckert, Email: jared.sterneckert@tu-dresden.de.
Supplemental Information
References
- Benatar M., Wuu J., Andersen P.M., Atassi N., David W., Cudkowicz M., Schoenfeld D. Randomized, double-blind, placebo-controlled trial of arimoclomol in rapidly progressive SOD1 ALS. Neurology. 2018;90:e565–e574. doi: 10.1212/WNL.0000000000004960. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chew J., Gendron T.F., Prudencio M., Sasaguri H., Zhang Y.J., Castanedes-Casey M., Lee C.W., Jansen-West K., Kurti A., Murray M.E. C9ORF72 repeat expansions in mice cause TDP-43 pathology, neuronal loss, and behavioral deficits. Science. 2015;348:1151–1154. doi: 10.1126/science.aaa9344. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chitiprolu M., Jagow C., Tremblay V., Bondy-Chorney E., Paris G., Savard A., Palidwor G., Barry F.A., Zinman L., Keith J. A complex of C9ORF72 and p62 uses arginine methylation to eliminate stress granules by autophagy. Nat. Commun. 2018;9:2794. doi: 10.1038/s41467-018-05273-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ciura S., Lattante S., Le Ber I., Latouche M., Tostivint H., Brice A., Kabashi E. Loss of function of C9orf72 causes motor deficits in a zebrafish model of amyotrophic lateral sclerosis. Ann. Neurol. 2013;74:180–187. doi: 10.1002/ana.23946. [DOI] [PubMed] [Google Scholar]
- Devlin A.C., Burr K., Borooah S., Foster J.D., Cleary E.M., Geti I., Vallier L., Shaw C.E., Chandran S., Miles G.B. Human iPSC-derived motoneurons harbouring TARDBP or C9ORF72 ALS mutations are dysfunctional despite maintaining viability. Nat. Commun. 2015;6:5999. doi: 10.1038/ncomms6999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Frick P., Sellier C., Mackenzie I.R.A., Cheng C.-Y., Tahraoui-Bories J., Martinat C., Pasterkamp R.J., Prudlo J., Edbauer D., Oulad-Abdelghani M. Novel antibodies reveal presynaptic localization of C9orf72 protein and reduced protein levels in C9orf72 mutation carriers. Acta Neuropathol. Commun. 2018;6:72. doi: 10.1186/s40478-018-0579-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ganassi M., Mateju D., Bigi I., Mediani L., Poser I., Lee H.O., Seguin S.J., Morelli F.F., Vinet J., Leo G. A surveillance function of the HSPB8-BAG3-HSP70 chaperone complex ensures stress granule integrity and dynamism. Mol. Cell. 2016;63:796–810. doi: 10.1016/j.molcel.2016.07.021. [DOI] [PubMed] [Google Scholar]
- Gijselinck I., Van Langenhove T., van der Zee J., Sleegers K., Philtjens S., Kleinberger G., Janssens J., Bettens K., Van Cauwenberghe C., Pereson S. A C9orf72 promoter repeat expansion in a Flanders-Belgian cohort with disorders of the frontotemporal lobar degeneration-amyotrophic lateral sclerosis spectrum: a gene identification study. Lancet Neurol. 2012;11:54–65. doi: 10.1016/S1474-4422(11)70261-7. [DOI] [PubMed] [Google Scholar]
- Hruscha A., Krawitz P., Rechenberg A., Heinrich V., Hecht J., Haass C., Schmid B. Efficient CRISPR/Cas9 genome editing with low off-target effects in zebrafish. Development. 2013;140:4982–4987. doi: 10.1242/dev.099085. [DOI] [PubMed] [Google Scholar]
- Jiang J., Zhu Q., Gendron T.F., Saberi S., McAlonis-Downes M., Seelman A., Stauffer J.E., Jafar-nejad P., Drenner K., Schulte D. Gain of toxicity from ALS/FTD-Linked repeat expansions in C9ORF72 is alleviated by antisense oligonucleotides targeting GGGGCC-containing RNAs. Neuron. 2016;90:535–550. doi: 10.1016/j.neuron.2016.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee K.-H., Zhang P., Kim H.J., Mitrea D.M., Sarkar M., Freibaum B.D., Cika J., Coughlin M., Messing J., Molliex A. C9orf72 dipeptide repeats impair the assembly, dynamics, and function of membrane-less organelles. Cell. 2016;167:774–788.e17. doi: 10.1016/j.cell.2016.10.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li Y.R., King O.D., Shorter J., Gitler A.D. Stress granules as crucibles of ALS pathogenesis. J. Cell Biol. 2013;201:361–372. doi: 10.1083/jcb.201302044. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Loschi M., Leishman C.C., Berardone N., Boccaccio G.L. Dynein and kinesin regulate stress-granule and P-body dynamics. J. Cell Sci. 2009;122:3973–3982. doi: 10.1242/jcs.051383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Majounie E., Renton A.E., Mok K., Dopper E.G.P., Waite A., Rollinson S., Chiò A., Restagno G., Nicolaou N., Simon-Sanchez J. Frequency of the C9orf72 hexanucleotide repeat expansion in patients with amyotrophic lateral sclerosis and frontotemporal dementia: a cross-sectional study. Lancet Neurol. 2012;11:323–330. doi: 10.1016/S1474-4422(12)70043-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mordes D.A., Prudencio M., Goodman L.D., Klim J.R., Moccia R., Limone F., Pietilainen O., Chowdhary K., Dickson D.W., Rademakers R. Dipeptide repeat proteins activate a heat shock response found in C9ORF72-ALS/FTLD patients. Acta Neuropathol. Commun. 2018;6:55. doi: 10.1186/s40478-018-0555-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Naujock M., Stanslowsky N., Bufler S., Naumann M., Reinhardt P., Sterneckert J., Kefalakes E., Kassebaum C., Bursch F., Lojewski X. 4-Aminopyridine induced activity rescues hypoexcitable motor neurons from amyotrophic lateral sclerosis patient-derived induced pluripotent stem cells. Stem Cells. 2016;34:1563–1575. doi: 10.1002/stem.2354. [DOI] [PubMed] [Google Scholar]
- Naumann M., Pal A., Goswami A., Lojewski X., Japtok J., Vehlow A., Naujock M., Günther R., Jin M., Stanslowsky N. Impaired DNA damage response signaling by FUS-NLS mutations leads to neurodegeneration and FUS aggregate formation. Nat. Commun. 2018;9:335. doi: 10.1038/s41467-017-02299-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Niblock M., Smith B.N., Lee Y.B., Sardone V., Topp S., Troakes C., Al-Sarraj S., Leblond C.S., Dion P.A., Rouleau G.A. Retention of hexanucleotide repeat-containing intron in C9orf72 mRNA: implications for the pathogenesis of ALS/FTD. Acta Neuropathol. Commun. 2016;4:18. doi: 10.1186/s40478-016-0289-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nicolas A., Kenna K.P., Renton A.E., Ticozzi N., Faghri F., Chia R., Dominov J.A., Kenna B.J., Nalls M.A., Keagle P. Genome-wide analyses identify KIF5A as a novel ALS gene. Neuron. 2018;97:1268–1283.e6. doi: 10.1016/j.neuron.2018.02.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pal A., Glass H., Naumann M., Kreiter N., Japtok J., Sczech R., Hermann A. High content organelle trafficking enables disease state profiling as powerful tool for disease modelling. Sci. Data. 2018;5:180241. doi: 10.1038/sdata.2018.241. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Parone P.A., Da Cruz S., Han J.S., McAlonis-Downes M., Vetto A.P., Lee S.K., Tseng E., Cleveland D.W. Enhancing mitochondrial calcium buffering capacity reduces aggregation of misfolded SOD1 and motor neuron cell death without extending survival in mouse models of inherited amyotrophic lateral sclerosis. J. Neurosci. 2013;33:4657–4671. doi: 10.1523/JNEUROSCI.1119-12.2013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Prudencio M., Belzil V.V., Batra R., Ross C.A., Gendron T.F., Pregent L.J., Murray M.E., Overstreet K.K., Piazza-Johnston A.E., Desaro P. Distinct brain transcriptome profiles in C9orf72-associated and sporadic ALS. Nat. Neurosci. 2015;18:1175–1182. doi: 10.1038/nn.4065. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Puls I., Jonnakuty C., LaMonte B.H., Holzbaur E.L.F., Tokito M., Mann E., Floeter M.K., Bidus K., Drayna D., Oh S.J. Mutant dynactin in motor neuron disease. Nat. Genet. 2003;33:455–456. doi: 10.1038/ng1123. [DOI] [PubMed] [Google Scholar]
- Sareen D., O'Rourke J.G., Meera P., Muhammad A.K.M.G., Grant S., Simpkinson M., Bell S., Carmona S., Ornelas L., Sahabian A. Targeting RNA foci in iPSC-derived motor neurons from ALS patients with a C9ORF72 repeat expansion. Sci. Translational Med. 2013;5:208ra149. doi: 10.1126/scitranslmed.3007529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Selvaraj B.T., Livesey M.R., Zhao C., Gregory J.M., James O.T., Cleary E.M., Chouhan A.K., Gane A.B., Perkins E.M., Dando O. C9ORF72 repeat expansion causes vulnerability of motor neurons to Ca(2+)-permeable AMPA receptor-mediated excitotoxicity. Nat. Commun. 2018;9:347. doi: 10.1038/s41467-017-02729-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi Y., Lin S., Staats K.A., Li Y., Chang W.-H., Hung S.-T., Hendricks E., Linares G.R., Wang Y., Son E.Y. Haploinsufficiency leads to neurodegeneration in C9ORF72 ALS/FTD human induced motor neurons. Nat. Med. 2018;24:313–325. doi: 10.1038/nm.4490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simon-Sanchez J., Dopper E.G., Cohn-Hokke P.E., Hukema R.K., Nicolaou N., Seelaar H., de Graaf J.R., de Koning I., van Schoor N.M., Deeg D.J. The clinical and pathological phenotype of C9ORF72 hexanucleotide repeat expansions. Brain. 2012;135:723–735. doi: 10.1093/brain/awr353. [DOI] [PubMed] [Google Scholar]
- Tabet R., Schaeffer L., Freyermuth F., Jambeau M., Workman M., Lee C.-Z., Lin C.-C., Jiang J., Jansen-West K., Abou-Hamdan H. CUG initiation and frameshifting enable production of dipeptide repeat proteins from ALS/FTD C9ORF72 transcripts. Nat. Commun. 2018;9:152. doi: 10.1038/s41467-017-02643-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- van Blitterswijk M., Mullen B., Heckman M.G., Baker M.C., DeJesus-Hernandez M., Brown P.H., Murray M.E., Hsiung G.Y., Stewart H., Karydas A.M. Ataxin-2 as potential disease modifier in C9ORF72 expansion carriers. Neurobiol. Aging. 2014;35:2421.e13–2421.e17. doi: 10.1016/j.neurobiolaging.2014.04.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wainger B.J., Kiskinis E., Mellin C., Wiskow O., Han S.S., Sandoe J., Perez N.P., Williams L.A., Lee S., Boulting G. Intrinsic membrane hyperexcitability of amyotrophic lateral sclerosis patient-derived motor neurons. Cell Rep. 2014;7:1–11. doi: 10.1016/j.celrep.2014.03.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Waite A.J., Baumer D., East S., Neal J., Morris H.R., Ansorge O., Blake D.J. Reduced C9orf72 protein levels in frontal cortex of amyotrophic lateral sclerosis and frontotemporal degeneration brain with the C9ORF72 hexanucleotide repeat expansion. Neurobiol. Aging. 2014;35:1779.e5–1779.e13. doi: 10.1016/j.neurobiolaging.2014.01.016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yokota S., Kitahara M., Nagata K. Benzylidene lactam compound, KNK437, a novel inhibitor of acquisition of thermotolerance and heat shock protein induction in human colon carcinoma cells. Cancer Res. 2000;60:2942–2948. [PubMed] [Google Scholar]
- Zhang Z., Almeida S., Lu Y., Nishimura A.L., Peng L., Sun D., Wu B., Karydas A.M., Tartaglia M.C., Fong J.C. Downregulation of microRNA-9 in iPSC-derived neurons of FTD/ALS patients with TDP-43 mutations. PLoS One. 2013;8:e76055. doi: 10.1371/journal.pone.0076055. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.