Abstract
To establish the prevalence of mobile colistin resistance (mcr) genes amongst Salmonella enterica isolates obtained through public health surveillance in England (April 2014 to September 2017), 33 205 S . enterica genome sequences obtained from human, food, animal and environmental isolates were screened for the presence of mcr variants 1 to 8. The mcr-positive genomes were assembled, annotated and characterized according to plasmid type. Nanopore sequencing was performed on six selected isolates with putative novel plasmids, and phylogenetic analysis was used to provide an evolutionary context for the most commonly isolated clones. Fifty-two mcr-positive isolates were identified, of which 32 were positive for mcr-1, 19 for mcr-3 and 1 for mcr-5. The combination of Illumina and Nanopore sequencing identified three novel mcr-3 plasmids and one novel mcr-5 plasmid, as well as the presence of chromosomally integrated mcr-1 and mcr-3. Monophasic S. enterica serovar Typhimurium accounted for 27/52 (52 %) of the mcr-positive isolates, with the majority clustering in clades associated with travel to Southeast Asia. Isolates in these clades were associated with a specific plasmid range and an additional extended-spectrum beta-lactamase genotype. Routine whole-genome sequencing for public health surveillance provides an effective screen for novel and emerging antimicrobial determinants, including mcr. Complementary long-read technologies elucidated the genomic context of resistance determinants, offering insights into plasmid dissemination and linkage to other resistance genes.
Keywords: Salmonella, MCR, colistin, WGS
Data Summary
All fastq files and assemblies were submitted to the National Centre for Biotechnology Information (NCBI). All data can be found under BioProject: PRJNA248792, PRJNA248064 https://www.ncbi.nlm.nih.gov/bioproject/?term=PRJNA315192. Strain-specific details can be found in Table S1.
Impact Statement.
In this study we present the results of screening for mcr variants against Public Health England’s considerable set of Salmonella genomes collected via routine national surveillance of gastrointestinal disease over a 4-year period, thereby elucidating the presence of mcr-containing Salmonella entering the food chain. Employing combined Illumina and Nanopore sequencing, we identified three novel mcr-3 plasmids and one novel mcr-5 plasmid, as well as the presence of chromosomally integrated mcr-1 and mcr-3. This study highlights how routine whole-genome sequencing for public health surveillance provides an effective screen for novel and emerging antimicrobial determinants.
Introduction
Colistin has been utilized as an antimicrobial last resort against Gram-negative bacteria exhibiting pan-resistance to multiple antimicrobials [1]. Colistin resistance has historically been attributed to intrinsic resistance or chromosomal mutations that are not transmissible within the population; however, the acquisition of mobile colistin resistance (mcr) genes borne on plasmids has recently been described [2, 3]. Since 2015, eight variants of plasmid-mediated mcr genes have been detected in Gram-negative bacteria, including Salmonella enterica , Escherichia coli , Moraxella spp. and Klebsiella pneumoniae isolated from clinical human cases as well as pigs, calves, poultry, food and environmental sources [3–9]
The variety of mcr genes reported is mirrored in the variability of their genetic context. For mcr-1, at least 12 different plasmid replicons have been described, namely: IncX3, IncX4, an IncX3-X4 hybrid, IncH1, IncHI1, IncHI2, IncP, IncI2, IncF, IncFII, an IncI2–IncFIB hybrid and IncY [3, 10–12]. mcr-2 has been associated with an IncX4 plasmid [8], mcr-3 has been found on a IncHI2, IncP and an IncR plasmid type [9, 11, 13], and both mcr-4 and mcr-5 have been described on a ColE-like plasmid [5, 6]. mcr-6 has been isolated integrated into the chromosome and mcr-7 and mcr-8 have been reported on an IncI2 and IncFII plasmid, respectively [4, 7, 14]. Recently, mcr-9 was described in a Salmonella Typhimurium isolated in 2010 from a human patient in the USA [15].
Whole-genome sequencing (WGS) of bacterial isolates facilitates the detection of known resistance genes and resistance-associated mutations. The adoption of WGS for routine pathogen surveillance has consequently improved antimicrobial resistance (AMR) surveillance by enabling earlier detection of resistant strains [16] and by providing a framework for passive AMR surveillance. In 2016, a retrospective study to understand the prevalence of mcr-1 in England identified 15 mcr-1-positive isolates within a large collection of genomes obtained from the surveillance of Gram-negative pathogens by Public Health England [17].
The aim of this study was to further investigate the prevalence of mcr-1 to -8 in S. enterica isolates submitted to Public Health England (PHE) through routine surveillance, to characterize the genomic context of mcr and to provide an insight into the dissemination of mcr-mediated colistin resistance in England from 2012 to September 2017.
Methods
WGS isolates
Since April 2014, WGS analysis has been performed routinely for the identification and typing of presumptive S. enterica isolates at PHE. Data obtained from WGS have been deposited and archived in the National Center for Biotechnology Information (NCBI) Sequence Read Archive – BioProject: PRJNA248064. From April 2014 to September 2017, 31 292 Salmonella isolates were referred; 27 611 were isolated from human faeces, 1469 were isolated from human blood isolates, 335 were isolated from human urine, 171 were isolated from other human sites, 769 were isolated from non-human sources (environmental, animal and unspecified samples) and 937 were isolated from food. Additionally, the database also consisted of 1913 genome sequences obtained from clinical isolates from 2012 to 2013, making up 5 % of the collection. For clinical isolates, information concerning the age, sex and recent foreign travel of patients was obtained from the isolate referral form.
Sample preparation
Genomic DNA purification was performed in accordance with the manufacturer’s procedures using the Qiasymphony DSP DNA Midi kit (Qiagen, Hilden, Germany), while DNA extraction was performed using a Qiasymphony SP (Qiagen, Hilden, Germany) with DNA eluted with nuclease-free water. DNA concentration and quality was measured using a GloMax (Promega, WI, USA) and LabChip DX (Perkin Elmer, MA, USA), respectively. The sequencing DNA libraries of each sample were prepared using a Nextera XT DNA Library Prep kit (Illumina, CA, USA). Paired-end sequencing was achieved using a HiSeq 2500 instrument that produced 2×101 base pair (bp) reads. The reads were quality-trimmed using Trimmomatic v0.36 [18].
Multi-locus sequence typing (MLST) analysis was performed to identify genetically related isolates by sequence type (ST) using MOST [19]. Related STs were then designated into clonal complexes collectively termed eburst groups (eBGs) [20].
Screening for mcr variants
The reference fasta sequences of the mcr genes were obtained from GenBank with the accession numbers KP347127, LT598652, KY924928, MF543359, KY807921, MF176240, MG267386 and MG736312 for mcr-1, mcr-2, mcr-3, mcr-4, mcr-5, mcr-6, mcr-7 and mcr-8, respectively. The WGS paired-end reads of each sample were then scanned for the presence of mcr-1 to -8 variants using GeneFinder (https://github.com/phe-bioinformatics/gene_finder), a tool that incorporates Bowtie 2 v2.2.3 [21] and SAMtools v0.1.19 [22]. Positive hits were defined as those with nucleotide identity greater than or equal to 90 % over the length of the reference gene. Genomes that were positive for one or more mcr genes were further scanned against a reference database for AMR determinants available in the GeneFinder hitub repository using the same criteria as above (https://github.com/phe-bioinformatics/gene_finder/tree/master/refs).
Assembly and annotation
Genomes were assembled using SPades v3.8.0 [23] with the options ‘--only-assembler’ and ‘-k 21, 33, 55, 77’. Quast v4.5 [24] was used with default settings to check the contiguity of assembled reads before visualizing the assembly graph with Bandage v0.8.1 [25]. Prokka v1.12 [26] was used with the options ‘--kingdom Bacteria’ to annotate putative contigs of significance identified in Bandage, before being deposited into Artemis v16.0.0 [27] to be viewed interactively for any notable coding sequences flanking the mcr gene.
Screening for plasmids
PlasmidFinder v1.3 [28] was used to identify known replicons that could be present within the queried genome. blastn was used to align the mcr-containing contig to a representative plasmid of that replicon type. Once a plausible comparator plasmid was identified, Mauve v2.4.0 [29] was used to position the contigs of the query sequence to their aligned location on the reference plasmid. Genomes associated with the same putative plasmid type were then grouped and viewed using blast Ring Image Generator (brig) v0.95 [30] and the genetic context of the mcr gene was analysed using Easyfig [31].
Phylogenetic analysis of mcr-positive genomes
To explore the evolutionary relationships between isolates harbouring mcr genes, phylogenetic analysis was performed on the most common mcr-positive isolated Salmonella serotype, Salmonella Typhimurium. Illumina fastq reads from mcr-positive isolates belonging to eBG1 were mapped to the Salmonella Typhimurium LT2 reference genome (AE006468.1) using BWA MEM v0.7.13 [32]. Variant positions identified by GATK v2.6.5 UnifiedGenotyper [33] that passed the parameters >90 % consensus, minimum read depth of 10, mapping quality (MQ) >30, were imported into SnapperDB v0.2.5 [34]. Hierarchical single linkage clustering was performed on the pairwise SNP difference between all isolates at various distance thresholds (250, 100, 50, 25, 10, 5, 0). The result of the clustering is a SNP profile, or SNP address, that can be used to describe population structure based on clonal groups. Maximum-likelihood phylogenies were constructed based on the single-nucleotide polymorphism (SNP) using RaxML v8.2.8 [35].
Nanopore sequencing
Six isolates that failed to align to a previously described mcr-positive reference plasmid were selected for Oxford Nanopore Technologies MinION sequencing. DNA purification was performed using the Wizard Genomic DNA purification kit (Promega, Madison, WI, USA) for Gram-negative bacteria with minor modifications (double incubation and centrifugation times, substitution of vortex steps with manual inversions and elution in 100 µl of nuclease-free water). The DNA library was prepared using the Native Barcoding kit 1D, EXP-NBD103, and Ligation Sequencing kit 1D, SQK-LSK108, according to Oxford Nanopore Technologies’ protocol, and sequenced using a FLO MINI106 flow cell. Raw FAST5 reads were basecalled using Albacore v2.0.1, with the flags ‘-f FLO-MIN106’, ‘-k SQK-LSK108’ and ‘--barcoding’ added. The reads were then subsequently converted to fasta and fastq using Poretools v0.6.0 [36]. Unicycler v0.4.2 [37] was used to assemble the reads using the flags ‘--min_fasta_length 50’ and ‘--mode normal’, and the hybrid option ‘−1 path/to/paired_end1 −2 path/to/paired_end2 -l path/to/long_reads’ that combines Nanopore’s long reads and Illumina’s short reads. fastq files have been uploaded to NCBI BioProject – PRJNA248792.
Results
Prevalence
Of the 33 205 Salmonella isolates screened for mcr variants, 52 genomes were positive for an mcr determinant. mcr-1 was detected in 32 isolates, mcr-3 was detected in 19 isolates and mcr-5 was detected in a single isolate. Of the mcr-1-positive isolates, 19/32 were identified as S. enterica serovar Typhimurium, of which 13 were eBG1 (ST34 n=12, ST19 n=1) and 6 were eBG138 (all ST36). All S. Typhimurium-positive genomes were isolated from stool samples. The remaining mcr-1-positive isolates were identified as S. enterica serovars Stanley (n=3), Rissen (n=2) and Java (n=2), as well as single isolates of S. enterica serovars 4,[5],12:b:-, Thompson, Newport, Enteritidis, Agona and Virchow. One of the S. enterica serovar Java isolates was obtained from a poultry meat sample. The age distribution of mcr-1-positive cases was between 1 and 88 years old. Ten patients reported travel to Southeast Asia before the onset of symptoms, with Thailand (n=6) and Malaysia (n=2) being the countries visited most frequently, with single reports of travel to India, Borneo, Egypt, Spain and Poland. The highest number of mcr-1-positive isolates was received in 2017 (n=11), followed by 2016 (n=8), 2015 (n=8), 2014 (n=4) and 2012 (n=1).
Of the 19 mcr-3-positive isolates, 16 were S. Typhimurium, of which 13 were eBG1 (all ST34) and 3 were eBG138 (all ST36). Fifteen were from human faecal samples and one was from a food product. mcr-3 was identified in a Salmonella cholerae-suis ST139 isolate from human blood. Single isolates of Salmonella bovis-morbificans and Salmonella Stanley were also positive for mcr-3. The age distribution of patients with mcr-3-positive samples was 3 to 68 years old, with eight patients having travelled to Thailand and single reports of travel to Vietnam, Malaysia and Cambodia. The greatest number of mcr-3-positive isolates was received in 2015 (n=9), followed by 2014 (n=6), 2016 (n=4) and 2017 (n=1).
The mcr-5 gene was detected in an S. Typhimurium ST34 food isolate received in 2014. No isolates were positive for variants of mcr-2, mcr-4, mcr-6, mcr-7 or mcr-8.
Plasmid types and mcr context mcr-1
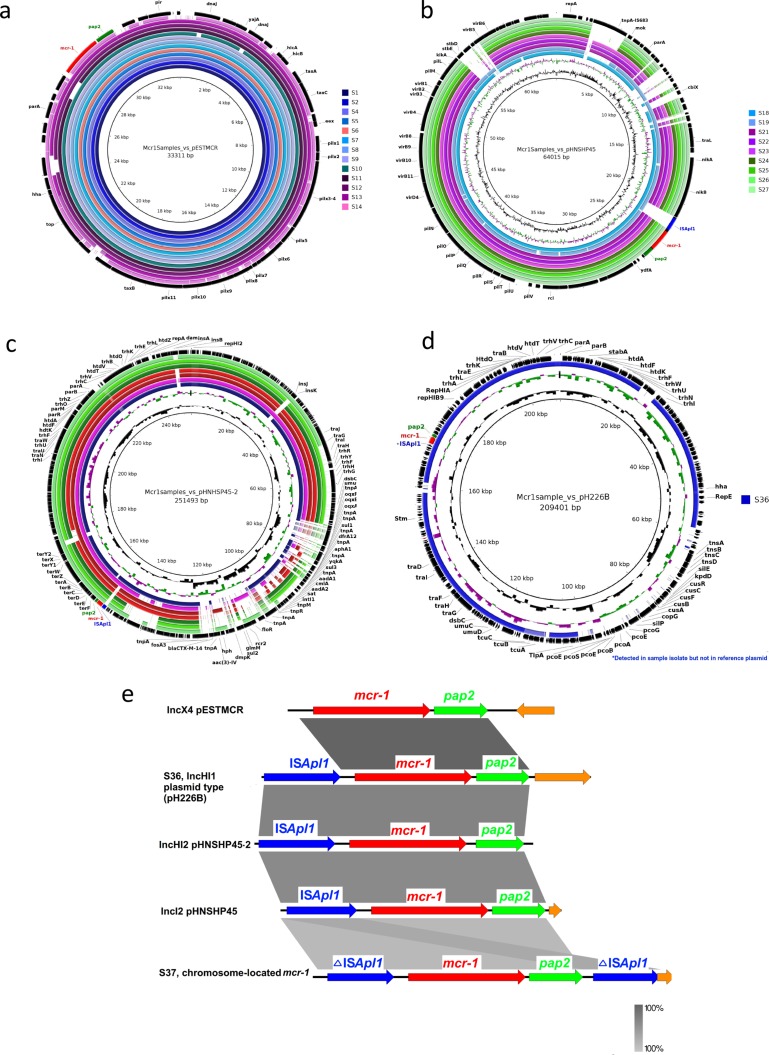
The mcr-1-positive isolates contained one of four plasmid types that have been previously described, namely: pESTMCR (KU743383), pHNSHP45 (KP347127), pHNSHP45-2 (KU341381) and pH226B (KX129784). Thirteen shared a similar backbone to pESTMCR, an IncX4 plasmid (88–100 % identity) (Fig. 1a); nine to pHNSHP45, an IncI2 plasmid (80–95 % identity) (Fig. 1b); seven to pHNSHP45-2, an IncHI2 plasmid (70–90 % identity) (Fig. 1c); while one isolate had a similar backbone to pH226B, an IncHI1 plasmid (80 % identity) (Fig. 1d). Neither S. Rissen isolate (S37 and S38;obtained from second-degree relatives) aligned to any reference plasmid. Nanopore sequencing detected two copies of the mcr-1 gene on the chromosome of isolate S37.
Fig. 1.
Brig plots showing mcr-1-positive plasmids aligned to (a) pESTMCR, (b) pHNSHP45, c) pHNHSP45-2 and (d) pH226B. (e) The genetic context upstream and downstream of mcr-1.
Among the 32 mcr-1-positive isolates, 3 distinct genetic environments were observed immediately upstream and downstream of the mcr-1 gene (Fig. 1e). The ISApl1 element, thought to be involved in mcr-1 mobilization, was found upstream of the mcr-1 gene in two isolates with the IncI2 plasmid type, two isolates with an IncHI2 plasmid type and one isolate with the IncHI1 plasmid type, but was not present in all isolates with the IncX4 backbone. Both copies of chromosomally located mcr-1 were located within a composite ISApl1 transposon designated as Tn6330 [38]. All 32 isolates, including both chromosomally located mcr-1 copies, had the pap2 gene located downstream of mcr-1, with no flanking transposons near the mcr-1 region when the ISApl1 element was absent. These three genetic environments have previously been observed in other studies [3, 3, 3, 36, 37, 37, 38, 38, 38, 39, 39, 40].
mcr-3
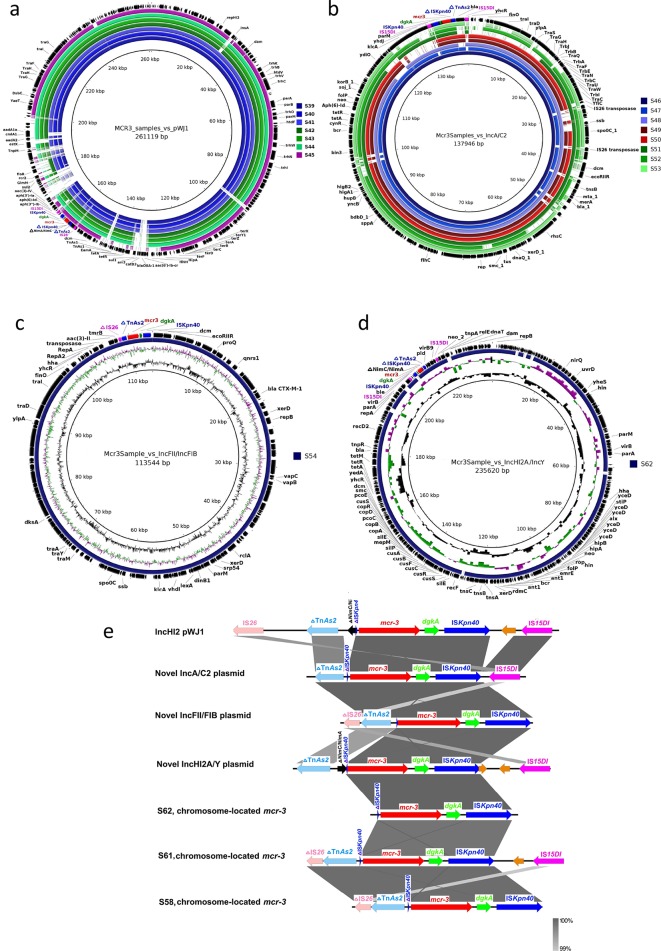
Seven isolates were found to harbour the mcr-3 gene on an IncHI2 plasmid with a similar backbone (95–95 % identity) to pWJ1 (KY924928) (Fig. 2a). The remaining 12 mcr-3-positive isolates did not align to a reference plasmid and a range of replicon types were detected by PlasmidFinder. To characterize the putative novel plasmids, a hybrid long-read/short-read sequencing strategy was deployed and three novel plasmids were described. Nanopore sequencing of S. Typhimurium isolates S46 and S48 revealed the presence of a novel 138 kb mcr-3 plasmid belonging to IncA/C2 that was shared amongst eight isolates (Fig. 2b). The S. Typhimurium isolate S54 was found to harbour a novel 113 kb plasmid containing the mcr-3 gene along with both IncFII and IncFIB replicons (Fig. 2c). Finally, isolate S62 (S. Stanley) was found to harbour the mcr-3 gene on a novel IncHI2A/IncY plasmid that was 236 kb in length (Fig. 2d) with a separate copy of the mcr-3 gene detected in the chromosome. mcr-3 genes were also detected in the chromosome of the S. Typhimurium ST34 isolate S58 and in S61, an S. bovis-morbificans (ST1499) isolate.
Fig. 2.
Brig plots showing mcr-3-positive plasmids aligned to (a) pWJ1, (b) the novel IncA/C2 plasmid, (c) the novel IncFII/IncFIB plasmid and (d) the novel IncHI2A/IncY plasmid. (e) The genetic context upstream and downstream of mcr-3.
The genetic context of mcr-3 in these 19 isolates, including the chromosome-mediated mcr-3 positive sequences, had a diacylglycerol kinase (dgkA) gene downstream of mcr-3 flanked by a truncated (∆) ISKpn40 (41 bp) and an intact ISKpn40 (Fig. 2e). All except the chromosomal mcr-3 gene of S62 had a ∆TnAs2 element found upstream of the ∆ISKpn40-mcr-3-dgkA-ISKpn40 structure. Overall, four genetic environments were observed. Isolates with the IncHI2 pWJ1 backbone had an intact IS26 and IS15DI located upstream and downstream of the ∆TnAs2-∆ISKpn40-mcr-3-dgkA-ISKpn40 fragment, respectively. Isolates with the novel IncA/C2 and IncHI2A/IncY plasmid type had two complete IS15DI elements enclosing the fragment. Chromosome-mediated mcr-3-positive isolates and the single isolate with the novel IncFII/IncFIB plasmid type only had a ∆IS26 located upstream of the fragment, while the chromosomal mcr-3 sequence of S62 had a conserved ∆ISKpn40-mcr-3-dgkA-ISKpn40 structure with no flanking IS or transposons. ISKpn40, TnAs2, IS26 and IS15DI have been identified in previous papers as key elements in the transposition of the mcr-3 gene [13, 40, 41].
mcr-5
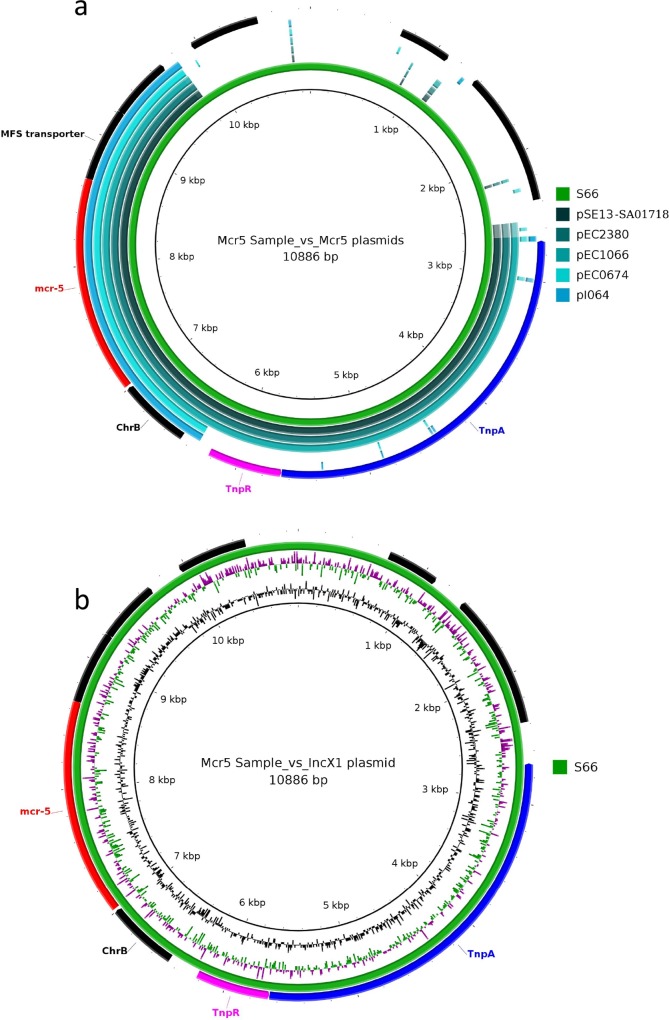
The mcr-5-positive isolate identified in this study did not align to the previous mcr-5 ColE plasmids pSE13-SA01718 (KY807921), pEC2380 (MG587004), pEC1066 (MG587003), pEC0674 (MF684783) and pI064-2 (MG800820), and instead was found on a small novel 10 kb plasmid with an uncategorized plasmid replication protein (Fig. 3). This plasmid harboured the same mcr-5 Tn3 family transposon, Tn6452, described in pSE13-SA01718, pEC2380 and pEC1066 [5, 42].
Fig. 3.
(a) Brig plot showing the mcr-5-positive plasmid aligned to previously described mcr-5 plasmids. (b) Brig plot showing novel mcr-5 plasmid aligned to a related IncX1 plasmid.
Co-resistance to other antimicrobials
Of the mcr-positive Salmonella isolates, all but four were defined as multidrug-resistant (MDR), with genotypically inferred resistance to at least three classes of antibiotics. Forty-seven isolates possessed β-lactamases, of which 10 encoded for extended-spectrum beta-lactamase (ESBL) production. Forty isolates were positive for bla TEM-1 and six isolates had an alternative TEM variant. Three isolates were positive for bla CMY-2, whilst none of the isolates possessed carbapenemase genes. The ESBL bla CTX-M-55 was found in nine isolates and one isolate contained bla CTX-M-14. The ESBL genotype was restricted to mcr-3 Salmonella samples. Forty-seven isolates harboured genes conferring resistance to a range of aminoglycosides. dfrA-mediated trimethoprim resistance determinants were detected in 24 isolates, with dfrA12 being the most common gene (n=16). Forty-seven isolates contained tetracycline resistance determinants [tet(A), tet(M) or tet(X)], while 34 isolates contained catA, floR or cmlA1 conferring resistance to chloramphenicol and/or florfenicol. Predicted reduced susceptibility to fluoroquinolones was largely mediated by qnr genes (n=24), with only five isolates displaying gyrA mutations in the quinolone resistance-determining region. Nine isolates displayed macrolide resistance determinants, with six isolates containing mph(A) in isolation or in conjunction with an erm gene. All genotypic profiles are described in Table S1 (available in the online version of this article).
Co-location of resistance determinants within the mcr-harbouring plasmids was also explored. mcr-1-positive isolates with an IncHI2-like backbone had additional resistance genes located on the same plasmid conferring resistance to aminoglycosides, tetracyclines and phenicols (Fig. 1c). All mcr-3-positive isolates with the IncHI2 plasmid, pWJ1, also co-harboured resistance genes, including four that acquired floR, five that acquired tet(A) and aph [6]-Id, two with mph(A) and one with qnrS1 and bla CTX-M-55. Of the novel mcr-3 plasmids, the IncA/C2 plasmid co-harboured bla CTX-M-55 in 5/8 isolates and the erm gene in a single isolate, while all contained the resistance genes floR, tet(A), aph [6]-Id and sul2. The IncFII/IncFIB plasmid encoded bla CTX-M-55 and qnrS1 in conjunction with mcr-3, and the IncHI2A plasmid encoded bla TEM-1 and a tetracycline resistance operon. The small mcr-5 plasmid did not harbour any other resistance genes.
Phylogenetic analysis
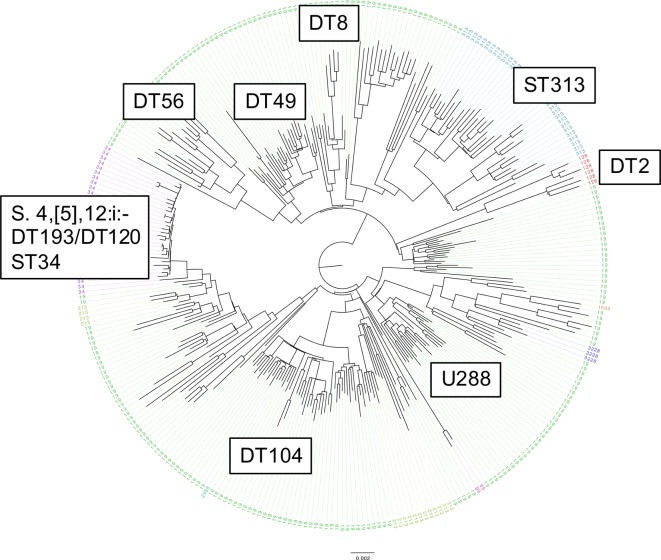
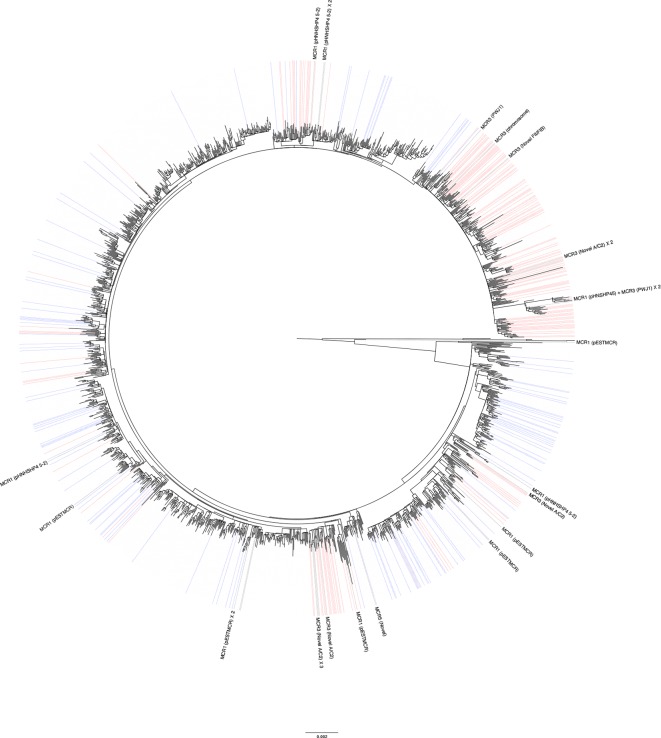
The majority (36/52) of the mcr-positive samples were S. Typhimurium, with 27 of these isolates belonging to clonal complex eBG1. Placing the mcr-positive isolates into a phylogeny of 288 diverse S. Typhimurium genomes (100 SNP representatives) revealed that 26/27 clustered within the monophasic variant 4,[5],12:i- ST34 clade (Fig. 4). One mcr-positive isolate (MLST 19) clustered adjacent to the ST34 monophasic clade, as defined by BAPS. Comparing the ST34 mcr-positive isolates against 1284 deduplicated (5 SNP representatives) ST34 genomes from routine surveillance of human clinical cases in England revealed that neither the mcr-1-positive isolates nor the mcr-3-positive isolates form a monophyletic group (Fig. 5). Overlaying patient travel information onto the phylogeny revealed several broad clusters where reported travel to Southeast Asia predominated. Of the mcr-positive ST34 EBG1 isolates, 16/26 cluster in clades where most background isolates are from patients reporting travel to Asia, including 7/16 cases that did not report any travel information. The remaining 10 mcr-positive EBG1 isolates are found in a diverse set of clusters that tend to be more associated with travel to Europe.
Fig. 4.
Maximum-likelihood SNP phylogeny of the 28 mcr-positive Salmonella Typhimurium eBG1 isolates against a background of 288 100-SNP representatives encompassing the diversity of the PHE Salmonella Typhimurium collection. Taxa are labelled and coloured by MLST and predominant phage types are labelled.
Fig. 5.
Maximum-likelihood SNP phylogeny of the 28 mcr-positive Salmonella Typhimurium eBG1 isolates against a background of 1284 5-SNP representatives encompassing the diversity of the PHE Salmonella Typhimurium ST34 collection. Taxa are labelled by mcr gene and plasmid type and coloured by reported travel to either Europe (Blue) or Asia (Red).
Several mcr plasmids were restricted to Asian-associated clades. The mcr-3 IncHI2 plasmid PJW1 was exclusively found in multiple Asian-associated clades of EBG1 but was also found in EBG36 associated with cases who had reported travel to Asia. The novel mcr-3 IncA/C2 plasmid was also restricted to the Asian-associated clades and furthermore associated with bla CTX-M-55. In contrast, the IncX4 mcr-1 plasmid pESTMCR was more commonly associated with EBG1 isolates that cluster with patients reporting travel within Europe, as was the IncHI2 plasmid pHNHSHP4 5–2.
Discussion
All mcr-positive S. enterica serovars isolated in this study are established zoonotic pathogens with serovars Typhimurium, Stanley, Rissen and cholerae-suis commonly isolated in pigs [43, 44]. This is consistent with the premise that the rise in colistin resistance in S. enterica has been driven by the selective pressure from colistin usage in agricultural animals rather than therapeutic use in humans [45]. Twenty-one patients that harboured mcr-positive strains reported travel to Southeast Asian countries, generally Thailand, where traditionally there are no veterinary antibiotic restrictions and antibiotics are readily available [46]. Furthermore, co-location of tet(A), tet(B), tet(M), catA, floR or cmlA1 on mcr-harbouring plasmids as a result of tetracycline and/or florfenicol use in food-producing animals could have also contributed to the selection of colistin resistance in Salmonella strains. Since the emergence of mcr genes, the European Medicines Agency (EMA) and the US Food and Drug Administration (FDA) have implemented guidelines for the restricted use of colistin in veterinary medicine in Europe and Thailand. However, plasmids harbouring mcr genes appear to be well established in bacterial species belonging to Enterobacteriaceae and it is likely that the number of mcr-positive isolates and the identification of other mcr variants and mcr-harbouring plasmids will continue to increase.
The predominant plasmid types harbouring mcr-1 in this routine surveillance study were IncX4 (n=13), IncI2 (n=9) and IncHI2 (n=7), which were the common plasmid types identified in other studies involving human and food S. enterica and E. coli isolates from Asia, Europe, the Americas and Africa [12, 38, 47, 48]. For mcr-3-positive samples, 8/19 had a novel IncA/C2 replicon type with the second most common (7/19) being an IncHI2 plasmid backbone previously described in Danish travellers returning from Asia [49, 50]. Two other novel mcr-3-containing plasmids were described in our data set, with one isolate harbouring an IncFII/FIB plasmid and another an IncHI2A/Y plasmid.
The mcr-1-pap2 and mcr-3-dgkA gene cassette was shared by all mcr-1 and mcr-3-positive isolates, respectively. Snesrud et al. demonstrated that mobilization of the mcr-1 cassette involves a composite transposon of ISApl1 [51]. The loss of a single copy of ISApl1, as seen in isolates with pHNSHP45-2, pHNSHP45 and pH226B backbones, or both copies of ISApl1, as seen in isolates with an pESTMCR backbone, is a result of its ‘copy out–paste in’ mechanism, which prevents the transposition of the mcr-1 cassette and transfixes it in the plasmid [51]. However, mcr-1 cassettes with a single-ended ISapl1 could still transpose if they maintained the inverted right repeat sequence of ISapl1 immediately after the pap2 gene [51]. Meanwhile, conjugation experiments have determined two circular forms that could have facilitated the dissemination of the mcr-3 gene. One circular form, mcr-3-dgkA-ISKpn40, was a 3535 bp derivative of the ISKpn40-mcr-3-dgkA-ISKpn40 fragment [41]. The second circular intermediate was ∆IS26-TnAs2-∆ISKpn40-mcr-3-dgkA-ISKpn40, 5990 bp in size, mediating mcr-3 transposition via homologous recombination between IS26 and IS15DI components [13].
These genetic combinations were also observed in the chromosomal integration of the mcr-1 and the mcr-3 gene cassettes, which adds a new perspective to their transmissible nature. mcr genes can therefore co-occur with other chromosome- and plasmid-located resistance genes and transpose themselves from plasmid-to-plasmid, from plasmid-to-chromosome, and vice versa. This transmissibility is of great concern in the clinical setting with respect to the treatment of invasive pathogens such as S. cholerae-suis. In silico analysis determined that mcr-3-positive isolates had more resistance genes co-located on the same mcr plasmid compared to mcr-1-positive isolates, and that the IncHI2 plasmid type harboured more resistance genes compared to other plasmid types. A study by Ruichao Li et al. hypothesized that IncHI2 plasmid types could be the most efficient vessels for the dissemination of co-located mcr genes due to their diverse MDR region, which incorporates several transposons, IS and resistance genes, supplying multiple insertion sites for mcr-1 [38].
Phylogenetic analysis of the most commonly detected mcr-positive serovar Typhimurium confirms previous reports of mcr-positive S. enterica circulating in Southeast Asia. The tree topology suggests multiple acquisitions of a restricted range of both mcr-1- and mcr-3-encoding plasmids within Asia and that those plasmids are more likely to encode additional antimicrobial resistance determinants, including ESBLs. These plasmids were also associated with other serovars of Salmonella associated with patients who reported travel to Asia. These data suggest that within Europe there appear to be alternative mcr-harbouring plasmids circulating that in general encode fewer additional antimicrobial resistance determinants.
This study demonstrates that routine WGS for public health surveillance can provide an effective screen for antimicrobial resistance determinants, including mcr genes. In addition, complementary long-read technologies can elucidate the genomic context of these determinants, offering insights into plasmid dissemination and linkage to other resistance genes. Finally, the phylogenetic context offers a perspective on the likely geographical origin of infection and plasmid transmission dynamics.
Data bibliography
NCBI BioProject: PRJNA248792, PRJNA248064.
Supplementary Data
Funding information
The work was funded by the National Institute for Health Research Health Protection Research Unit (NIHR HPRU) in Gastrointestinal Infections at University of Liverpool in partnership with Public Health England (PHE), in collaboration with University of East Anglia, University of Oxford and the Quadram Institute (109524).
Author contributions
T. J. D., N. W., M. D., G. G. and C. J. conceptualized the project. D. G. and M. D. performed the DNA extraction. D. G. and C. M. performed the ONT. sequencing. C. M., D. G., H. H. and A. P. performed the bioinformatics analysis. T. D. J. and C. M. wrote the manuscript. M. A. C., K. L. H. and D. M. critically reviewed the manuscript.
Conflicts of interest
The authors declare that there are no conflicts of interest.
Footnotes
Abbreviations: BLAST, Bsaic Local Alignment Search Tool; BRIG, BLAST Ring Image Generator; BURST, Based Upon Related Sequence Types; SAM, Sequence Alignment Map.
All supporting data, code and protocols have been provided within the article or through supplementary data files. One supplementary table is available with the online version of this article.
References
- 1.Falagas ME, Kasiakou SK, Saravolatz LD. Colistin: the revival of polymyxins for the management of multidrug-resistant gram-negative bacterial infections. Clin Infect Dis. 2005;40:1333–1341. doi: 10.1086/429323. [DOI] [PubMed] [Google Scholar]
- 2.Bialvaei AZ, Samadi Kafil H, Colistin SKH. Colistin, mechanisms and prevalence of resistance. Curr Med Res Opin. 2015;31:707–721. doi: 10.1185/03007995.2015.1018989. [DOI] [PubMed] [Google Scholar]
- 3.Li A, Yang Y, Miao M, Chavda KD, Mediavilla JR, et al. Complete sequences of mcr-1-Harboring plasmids from Extended-Spectrum-β-Lactamase- and carbapenemase-producing Enterobacteriaceae. Antimicrob Agents Chemother. 2016;60:4351–4354. doi: 10.1128/AAC.00550-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.AbuOun M, Stubberfield EJ, Duggett NA, Kirchner M, Dormer L, et al. mcr-1 and mcr-2 variant genes identified in Moraxella species isolated from pigs in Great Britain from 2014 to 2015. J Antimicrob Chemother. 2017;72:2745–2749. doi: 10.1093/jac/dkx286. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Borowiak M, Fischer J, Hammerl JA, Hendriksen RS, Szabo I, et al. Identification of a novel transposon-associated phosphoethanolamine transferase gene, mcr-5, conferring colistin resistance in d-tartrate fermenting Salmonella enterica subsp. enterica serovar paratyphi B. J Antimicrob Chemother. 2017;72:3317–3324. doi: 10.1093/jac/dkx327. [DOI] [PubMed] [Google Scholar]
- 6.Carattoli A, Villa L, Feudi C, Curcio L, Orsini S, et al. Novel plasmid-mediated colistin resistance mcr-4 gene in Salmonella and Escherichia coli, Italy 2013, Spain and Belgium, 2015 to 2016. Euro Surveill. 2017;22 doi: 10.2807/1560-7917.ES.2017.22.31.30589. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Wang X, Wang Y, Zhou Y, Li J, Yin W, et al. Emergence of a novel mobile colistin resistance gene, mcr-8, in NDM-producing Klebsiella pneumoniae . Emerg Microbes Infect. 2018;7:1–9. doi: 10.1038/s41426-018-0124-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Xavier BB, Lammens C, Ruhal R, Kumar-Singh S, Butaye P, et al. Identification of a novel plasmid-mediated colistin-resistance gene, mcr-2, in Escherichia coli, Belgium, June 2016. Euro Surveill Bull Eur Sur Mal Transm Eur Commun Dis Bull. 2016;21 doi: 10.2807/1560-7917.ES.2016.21.27.30280. [DOI] [PubMed] [Google Scholar]
- 9.Yin W, Li H, Shen Y, Liu Z, Wang S, et al. Novel Plasmid-Mediated Colistin Resistance Gene mcr-3 in Escherichia coli . mBio. 2017;8 doi: 10.1128/mBio.00543-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wang Q, Sun J, Li J, Ding Y, Li X-P, et al. Expanding landscapes of the diversified mcr-1-bearing plasmid reservoirs. Microbiome. 2017;5:70. doi: 10.1186/s40168-017-0288-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Zhang C, Feng Y, Liu F, Jiang H, Qu Z, et al. A Phage-Like IncY Plasmid Carrying the mcr-1 Gene in Escherichia coli from a Pig Farm in China. Antimicrob Agents Chemother. 2017;61 doi: 10.1128/AAC.02035-16. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Wang R, van Dorp L, Shaw LP, Bradley P, Wang Q, et al. The global distribution and spread of the mobilized colistin resistance gene mcr-1. Nat Commun. 2018;9:1179. doi: 10.1038/s41467-018-03205-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Wang Z, Fu Y, Du X-D, Jiang H, Wang Y. Potential transferability of mcr-3 via IS 26 -mediated homologous recombination in Escherichia coli . Emerg Microbes Infect. 2018;7:1–4. doi: 10.1038/s41426-018-0057-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Yang Y-Q, Li Y-X, Lei C-W, Zhang A-Y, Wang H-N. Novel plasmid-mediated colistin resistance gene mcr-7.1 in Klebsiella pneumoniae. J Antimicrob Chemother. 2018;73:1791–1795. doi: 10.1093/jac/dky111. [DOI] [PubMed] [Google Scholar]
- 15.Carroll LM, Gaballa A, Guldimann C, Sullivan G, Henderson LO, et al. Identification of novel mobilized colistin resistance gene mcr-9 in a multidrug-resistant, colistin-susceptible Salmonella enterica serotype typhimurium isolate. MBio. 2019;10 doi: 10.1128/mBio.00853-19. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Köser CU, Ellington MJ, Peacock SJ. Whole-Genome sequencing to control antimicrobial resistance. Trends Genet. 2014;30:401–407. doi: 10.1016/j.tig.2014.07.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Doumith M, Godbole G, Ashton P, Larkin L, Dallman T, et al. Detection of the plasmid-mediated mcr-1 gene conferring colistin resistance in human and food isolates of Salmonella enterica and Escherichia coli in England and Wales. J Antimicrob Chemother. 2016;71:2300–2305. doi: 10.1093/jac/dkw093. [DOI] [PubMed] [Google Scholar]
- 18.Bolger AM, Lohse M, Usadel B. Trimmomatic: a flexible trimmer for illumina sequence data. Bioinformatics. 2014;30:2114–2120. doi: 10.1093/bioinformatics/btu170. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tewolde R, Dallman T, Schaefer U, Sheppard CL, Ashton P, et al. Most: a modified MLST typing tool based on short read sequencing. PeerJ. 2016;4:e2308. doi: 10.7717/peerj.2308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Achtman M, Wain J, Weill F-X, Nair S, Zhou Z, et al. Multilocus sequence typing as a replacement for serotyping in Salmonella enterica. PLoS Pathog. 2012;8:e1002776. doi: 10.1371/journal.ppat.1002776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Langmead B, Salzberg SL. Fast gapped-read alignment with Bowtie 2. Nat Methods. 2012;9:357–359. doi: 10.1038/nmeth.1923. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Li H, Handsaker B, Wysoker A, Fennell T, Ruan J, et al. The sequence Alignment/Map format and SAMtools. Bioinformatics. 2009;25:2078–2079. doi: 10.1093/bioinformatics/btp352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Bankevich A, Nurk S, Antipov D, Gurevich AA, Dvorkin M, et al. SPAdes: a new genome assembly algorithm and its applications to single-cell sequencing. J Comput Biol. 2012;19:455–477. doi: 10.1089/cmb.2012.0021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Gurevich A, Saveliev V, Vyahhi N, Tesler G. QUAST: quality assessment tool for genome assemblies. Bioinformatics. 2013;29:1072–1075. doi: 10.1093/bioinformatics/btt086. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Wick RR, Schultz MB, Zobel J, Holt KE. Bandage: interactive visualization of de novo genome assemblies. Bioinformatics. 2015;31:3350–3352. doi: 10.1093/bioinformatics/btv383. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Seemann T. Prokka: rapid prokaryotic genome annotation. Bioinformatics. 2014;30:2068–2069. doi: 10.1093/bioinformatics/btu153. [DOI] [PubMed] [Google Scholar]
- 27.Carver T, Harris SR, Berriman M, Parkhill J, McQuillan JA. Artemis: an integrated platform for visualization and analysis of high-throughput sequence-based experimental data. Bioinformatics. 2012;28:464–469. doi: 10.1093/bioinformatics/btr703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Carattoli A, Zankari E, García-Fernández A, Voldby Larsen M, Lund O, et al. In silico detection and typing of plasmids using PlasmidFinder and plasmid multilocus sequence typing. Antimicrob Agents Chemother. 2014;58:3895–3903. doi: 10.1128/AAC.02412-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Darling ACE, Mau B, Blattner FR, Perna NT. Mauve: multiple alignment of conserved genomic sequence with rearrangements. Genome Res. 2004;14:1394–1403. doi: 10.1101/gr.2289704. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Alikhan N-F, Petty NK, Ben Zakour NL, Beatson SA. Blast ring image generator (BRIG): simple prokaryote genome comparisons. BMC Genomics. 2011;12:402. doi: 10.1186/1471-2164-12-402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Sullivan MJ, Petty NK, Beatson SA. Easyfig: a genome comparison visualizer. Bioinformatics. 2011;27:1009–1010. doi: 10.1093/bioinformatics/btr039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Li H, Durbin R. Fast and accurate long-read alignment with Burrows-Wheeler transform. Bioinformatics. 2010;26:589–595. doi: 10.1093/bioinformatics/btp698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.McKenna A, Hanna M, Banks E, Sivachenko A, Cibulskis K, et al. The genome analysis toolkit: a MapReduce framework for analyzing next-generation DNA sequencing data. Genome Res. 2010;20:1297–1303. doi: 10.1101/gr.107524.110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Dallman T, Ashton P, Schafer U, Jironkin A, Painset A, et al. SnapperDB: a database solution for routine sequencing analysis of bacterial isolates. Bioinformatics. 2018;34:3028–3029. doi: 10.1093/bioinformatics/bty212. [DOI] [PubMed] [Google Scholar]
- 35.Stamatakis A. RAxML version 8: a tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics. 2014;30:1312–1313. doi: 10.1093/bioinformatics/btu033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Loman NJ, Quinlan AR. Poretools: a toolkit for analyzing nanopore sequence data. Bioinformatics. 2014;30:3399–3401. doi: 10.1093/bioinformatics/btu555. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Wick RR, Judd LM, Gorrie CL, Holt KE. Unicycler: resolving bacterial genome assemblies from short and long sequencing reads. PLoS Comput Biol. 2017;13:e1005595. doi: 10.1371/journal.pcbi.1005595. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Li R, Xie M, Zhang J, Yang Z, Liu L, et al. Genetic characterization of mcr-1-bearing plasmids to depict molecular mechanisms underlying dissemination of the colistin resistance determinant. J Antimicrob Chemother. 2017;72:393–401. doi: 10.1093/jac/dkw411. [DOI] [PubMed] [Google Scholar]
- 39.Poirel L, Kieffer N, Nordmann P. In Vitro Study of IS Apl1 -Mediated Mobilization of the Colistin Resistance Gene mcr-1 . Antimicrob Agents Chemother. 2017;61 doi: 10.1128/AAC.00127-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Yu CY, Ang GY, Chong T-M, Chin PS, Ngeow YF, et al. Complete genome sequencing revealed novel genetic contexts of the mcr-1 gene in Escherichia coli strains. J Antimicrob Chemother. 2017;72:1253–1255. doi: 10.1093/jac/dkw541. [DOI] [PubMed] [Google Scholar]
- 41.Xiang R, Liu B-H, Zhang A-Y, Lei C-W, Ye X-L, et al. Colocation of the Polymyxin Resistance Gene mcr-1 and a Variant of mcr-3 on a Plasmid in an Escherichia coli Isolate from a Chicken Farm. Antimicrob Agents Chemother. 2018;62 doi: 10.1128/AAC.00501-18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Hammerl JA, Borowiak M, Schmoger S, Shamoun D, Grobbel M, et al. mcr-5 and a novel mcr-5.2 variant in Escherichia coli isolates from food and food-producing animals, Germany, 2010 to 2017. J Antimicrob Chemother. 2018;73:1433–1435. doi: 10.1093/jac/dky020. [DOI] [PubMed] [Google Scholar]
- 43.Hendriksen RS, Le Hello S, Bortolaia V, Pulsrikarn C, Nielsen EM, et al. Characterization of isolates of Salmonella enterica serovar Stanley, a serovar endemic to Asia and associated with travel. J Clin Microbiol. 2012;50:709–720. doi: 10.1128/JCM.05943-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Pornsukarom S, Patchanee P, Erdman M, Cray PF, Wittum T, et al. Comparative Phenotypic and Genotypic Analyses of Salmonella Rissen that Originated from Food Animals in Thailand and United States. Zoonoses Public Health. 2015;62:151–158. doi: 10.1111/zph.12144. [DOI] [PubMed] [Google Scholar]
- 45.Poirel L, Nordmann P. Emerging plasmid-encoded colistin resistance: the animal world as the culprit? J. Antimicrob. Chemother. 2016;71:2326–2327. doi: 10.1093/jac/dkw074. [DOI] [PubMed] [Google Scholar]
- 46.Holloway KA, Batmanabane G, Puri M, Tisocki K. Antibiotic use in South East Asia and policies to promote appropriate use: reports from country situational analyses. BMJ. 2017;358:j2291. doi: 10.1136/bmj.j2291. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Zurfluh K, Nüesch-Inderbinen M, Klumpp J, Poirel L, Nordmann P, et al. Key features of mcr-1-bearing plasmids from Escherichia coli isolated from humans and food. Antimicrob Resist Infect Control. 2017;6 doi: 10.1186/s13756-017-0250-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Matamoros S, van Hattem JM, Arcilla MS, Willemse N, Melles DC, et al. Global phylogenetic analysis of Escherichia coli and plasmids carrying the mcr-1 gene indicates bacterial diversity but plasmid restriction. Sci Rep. 2017;7:1–10. doi: 10.1038/s41598-017-15539-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Roer L, Hansen F, Stegger M, Sönksen UW, Hasman H, et al. Novel mcr-3 variant, encoding mobile colistin resistance, in an ST131 Escherichia coli isolate from bloodstream infection, Denmark, 2014. Eurosurveillance. 2017;22 doi: 10.2807/1560-7917.ES.2017.22.31.30584. [DOI] [PubMed] [Google Scholar]
- 50.Litrup E, Kiil K, Hammerum AM, Roer L, Nielsen EM, et al. Plasmid-Borne colistin resistance gene mcr-3 in Salmonella isolates from human infections, Denmark, 2009-17. Euro Surveill. 2017;22 doi: 10.2807/1560-7917.ES.2017.22.31.30587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Snesrud E, Ong AC, Corey B, Kwak YI, Clifford R, et al. Analysis of Serial Isolates of mcr-1 -Positive Escherichia coli Reveals a Highly Active IS Apl1 Transposon. Antimicrob Agents Chemother. 2017;61 doi: 10.1128/AAC.00056-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.