Due to their dynamic characteristics, hydroxyapatite nanoparticles (HAP-NPs) have been employed numerous times in nanomedicine and in tissue engineering, particularly as diagnostic and therapeutic agents.
Due to their dynamic characteristics, hydroxyapatite nanoparticles (HAP-NPs) have been employed numerous times in nanomedicine and in tissue engineering, particularly as diagnostic and therapeutic agents.
Abstract
Due to their dynamic characteristics, hydroxyapatite nanoparticles (HAP-NPs) have been employed numerous times in nanomedicine and in tissue engineering, particularly as diagnostic and therapeutic agents. However, there are outstanding findings from various studies that question whether these NPs are safe when they are used in the human body. Therefore, a more in-depth toxicity assessment should be carried out to give a clear answer regarding the fate of these particles. Here we aim to investigate the possible cytotoxicity, genotoxicity and inflammation induced by HAP-NPs, as well as predict the synergistic antioxidative effect of chitosan nanoparticles (CsNPs) and curcumin nanoparticles (CurNPs) in mitigating this pronounced toxicity. The present study was conducted on eighty Wistar male rats, divided into eight equal groups. The results showed that, at the molecular level, HAP-NPs significantly induced gene expression of tumor suppressor protein p53, tumor necrosis factor-α (TNF-α), interleukin-6 (IL-6) and also Kidney Injury Molecule-1 (KIM-1) and Lipocalin-2 (LCN2). In addition, kidney biochemical parameters (total bilirubin, urea, uric acid and creatinine) increased, but albumin levels decreased in the group treated with HAP-NPs alone. Meanwhile, co-treatment with CsNPs and/or CurNPs with HAP-NPs showed an improvement in the activities of the kidney parameters and reduced inflammation. This study shows that the nephrotoxicity mechanism of HAP-NPs may involve various signaling pathways including alterations in biochemical parameters, gene expression of KIM-1 and LCN2 and disturbing the production of cytokines and p53. Furthermore, these insights showed that the combined effect of both CsNPs and CurNPs was more pronounced than the effect of each one on its own.
1. Introduction
The outstanding properties of nanoparticles (NPs) fundamentally rely on their physicochemical properties: the particles may exist in separated or aggregated forms and can be hexagonal, tube-like, irregular, or spherically shaped. Regardless of the increased publicity of nanotechnology in many fields of life, their applications are limited due to their long-term adverse effects and possible toxicity.1 The physicochemical characteristics of NPs, such as their large surface areas, small size and resilient chemical structures have favored their use in nano-medicine, and reinforced their toxicological side effects.2 In particular, the surface area and particle size are substantial factors that contribute directly to the toxicity of nanoparticles.3 Moreover, the surface of nanoparticles dictates biomolecule and ion adsorption, consequently influencing the cellular responses formulated, and thereby taking part in the induced toxicity of the nanoparticles.4
The influence of nanotechnology in many fields of life has led to various synthesis techniques for producing nano-sized materials. These techniques have been developed to produce hydroxyapatite nanoparticles (HAP-NPs) with further modifications, such as increased surface area, reduced size and controlled morphology.5 HAP is a calcium phosphate that is similar to bone and teeth minerals in composition and morphology.6 Apart from its biocompatibility and bioactivity, its porous structure and mechanical intensity are also important when applied within the human body as a bone substitute.7 Moreover, there are adequate osteo-inductive, osteo-integrative and osteo-conductive properties for this material.7 It is now possible to influence the chemical, physical and surface characteristics of synthetic HAP-NPs as they have shown great prospects in drug delivery as carriers for therapeutic agents including proteins, anticancer drugs, antibiotics, genes, radioisotopes and antigens for vaccines.8 These reports have proved, using the HAP-NPs in such injectable systems, that they clearly have the potential to be used as delivery vehicles due to the ease with which DNA, drugs and proteins can be attached or loaded.
There are several types of nano-antioxidant that possess fundamental properties that do not depend on their functionalization but on the surface properties of the material itself. Chitosan nanoparticles (CsNPs) represent one of the natural materials that have excellent antimicrobial, biological, and physicochemical properties, and this makes them outstanding environmentally friendly and bioactive materials that do not produce any harmful effects on humans.9 Chitosan is also a highly proven antioxidant agent that can chelate metal ions and scavenge free radicals by donating hydrogen or lone pairs of electrons.10 The hydroxyl and amino functional groups of chitosan interact with metal ions, eliciting many activities such as chelation, adsorption, and ion exchange.11 The strong hydrogen bonds and the semi-crystalline structure of chitosan guarantee that chitosan cannot be separated from the metal ions.12 Various studies have been reported regarding the capability of CsNPs to improve drug bioavailability through pharmacokinetic modifying and encapsulated drug protecting.13,14 Despite being used as oral delivery carriers, CsNPs can also be applied to other mucous membrane systems to deliver proteins and peptides.15
Alternatively, curcumin is considered to be a polyphenolic compound that has anti-inflammatory, antimicrobial, antioxidant, cholesterol-lowering and wound healing properties.16 Besides cancers, curcumin can also be utilized in cystic fibrosis, malaria, anti-inflammatory diseases and Alzheimer's treatments.17 Targeting RNA and DNA, due to the pleiotropic trait of curcumin, happens concurrently.18,19 Exclusively, curcumin pleiotropic characteristics are unlike other chemo-therapeutic agents and can regulate mitogen-activated protein kinase, nuclear factor-kappaB, nuclear b-catenin signaling, serine/threonine protein kinase (AKT) signaling pathways, transcription factor activator protein-1 and tumor suppressor protein 53 (p53).18,20,21 Curcumin can also suppress the expression of some growth factors such as the epidermal growth receptor and estrogen receptors, which are associated with cancers.20 In addition, it can down-regulate the expression of IL-6, IL-1, TNF α, IL-8, C-reactive protein and adhesive molecules.22,23
From a review of the literature, few studies have been done on the possible renal toxicity induced by HAP-NPs, and several recent research has been published using the bulk size of chitosan and curcumin but seldom using their nano-forms. Therefore, we aim in the present study to investigate the possible nephro-toxic effects of 45 days oral intake of HAP-NPs, by exploring changes in kidney genomic DNA, gene expression at the molecular level, interleukin 6, tumor suppressor P53, and tumor necrosis factor α. In addition, we determine the possible ameliorative effect of the nano-forms of chitosan and curcumin against this toxicity.
2. Materials and methods
2.1. Tested compounds and doses
The reagents used for preparing the HAP-NPs were as follows: Na2CO3, which was purchased from Riedel-de-Haën, Germany and NaOH from El-Nasr pharmaceutical chemicals Co., Egypt. CsNPs (Mw 310–375 kDa) and CurNPs were purchased from Nanotech Egypt for Photo Electronics. The dose of HAP-NPs (300 mg per kg bw) was chosen according to Sabry,24 after being dissolved in distilled water. The dose of CsNPs (280 mg per kg bw) was chosen according to Tang and Abdel-Wahhab.25–27 In addition, the CurNPs were dissolved in distilled water and the dose (15 mg per kg bw) was chosen according to Yadav.28
2.2. Preparation of hydroxyapatite nanoparticles
2.2.1. Mechanical and alkali treatment
Salt tissue attached to the bone was removed mechanically by a sharp knife or Bard Parker blade no. 11. The bone was boiled for 2 hours in 30% sodium carbonate Na2CO3, followed by washing with hot water. Then it was boiled again in water for another 2 hours. This procedure was repeated several times to ensure maximum removal of the organic matter from the bone.29
2.2.2. Calcinations of the alkali treated bone
This procedure was performed in an electrically heated furnace with two holes for inlet and outlet purposes. A platinum disk of suitable size was used to introduce the bone regiments (cutoffs); the furnace temperature was gradually increased at a rate of 5 °C min–1 in a strong air stream to maintain an adequate oxidizing atmosphere, and also to get rid of the evolving carbon dioxide and water vapor. The temperature was increased until it reached 800 °C, with a soaking time of two hours at the final firing temperature.
2.2.3. Grinding of the treated cutoff bones
The prepared samples were dry-crushed in a mechanical porcelain mortar. The grinding was accomplished using grinding apparatus (Gilson company Inc. model USA, no Lc-91). The prepared hydroxyapatite particulates (fine powder) were dried before continuing the procedure to obtain the HAP-NPs. The HAP-NPs were obtained from the crushed bone using alkaline hydrolysis.30 The crushed bone (15 g) was heated with 2 M NaOH (El-Nasr Pharmaceutical chemicals Co., Egypt) for 1 h at 200 °C. This process was repeated until all traces of collagen were removed. HAP-NPs were collected into conical tubes, centrifuged (Combi-514 R, Hanil science industrial co., Ltd, Incheon, Korea) at 1000 rpm for 1 min, and then washed with H2O until they reached a neutral pH, and were then dried in an oven at 100 °C. The prepared HAP-NPs were packed into clean pre-sterilized glass containers and sterilized by gamma radiation at 2.5 M rad.
2.3. Characterization of the tested nanoparticles
All samples were characterized using Fourier-transform infrared spectroscopy (FTIR).
2.3.1. Fourier transform infrared spectroscopy analysis
Fourier-transform infrared (FTIR) spectra were measured using a Shimadzu FTIR-8400 Spectrometer (Shimadzu, Japan), and were recorded in the wavelength range 400–4000 cm–1. 100 mg of the dried samples was mixed with 100 mg of KBr spectral grade and pressed under hydraulic pressure into discs.31
2.4. Animals and experimental design
80 male Wistar rats were obtained from the faculty of Medicine in the University of Alexandria (Alexandria, Egypt), with an average weight of 170–175 g. They were kept on a tap water and basal diet, provided ad libitum, and were housed in standard environmental conditions. All procedures of the present work followed the guidelines of the Institutional Animal Care and Use Committee (IACUC) and were approved by the Animal Care and Use Committee of Alexandria University (Ethics Approval No. 1468–104, revised 2018). Taking this into consideration, their water and food consumption and body weights (gain or loss) were recorded weekly until the end of the experiments. After 2 weeks of acclimation, the animals were divided into 8 equal groups, housing a max. of 10 rats per cage. Group 1 was the control; group 2 was treated with CsNPs; group 3 received CurNPs; group 4 received CsNPs and CurNPs; group 5 received HAP-NPs; group 6 received CsNPs and HAP-NPs; group 7 received CurNPs and HAP-NPs; and group 8 received CsNPs, CurNPs and HAP-NPs. The animals were orally treated with the respective doses daily for 45 days.
2.5. Blood sample collection and tissue preparation
All animals were anesthetized, sacrificed and their blood samples were collected in heparin tubes. For separating the plasma, the blood was centrifuged at 860g for 20 minutes and the obtained plasma was preserved at –80 °C. The removed kidneys were washed with (0.9%) saline solution and the adhering fats and tissues were instantly removed. In addition, the kidneys were homogenized in a Potter–Elvehjem type homogenizer. The kidney homogenates were centrifuged at 10 000g at 4 °C for 20 minutes, and the supernatant was then stored for further analysis.
2.6. Measured parameters
2.6.1. Biochemical parameters
The plasma levels of albumin, urea, uric acid, creatinine, and total bilirubin were analyzed using (Biosystems S.A. Costa Brava 30, Barcelona, Spain) commercial kits.
2.6.2. Gene expression analysis of kidney injury molecule-1 and lipocalin-2 using RT-PCR
Quantitative analyses of KIM-1 and lipocalin-2 expressions in renal tissue were performed using qRT-PCR.32 The total RNA was isolated using the RNeasy Mini Kit (Qiagen, Germany) according to the manufacturer instructions, and then the isolated RNA was reverse transcribed using reverse transcriptase enzyme into complementary DNA (cDNA), which was then amplified and detected using specific primers by real-time PCR. Reverse transcription was done using the miScript II RT Kit (Qiagen, Germany), which is used to perform a one-step, single-tube reverse transcription reaction. The reverse-transcription master mix was prepared on ice according to Table 1, and the miScriptHiFlex Buffer promotes conversion of all the RNA species into cDNA.
Table 1. Reverse-transcription reaction components.
| Gene | Primer sequence |
|
| KIM-1 | F: | 5′-CGCAGAGAAACCCGACTAAG-3′ |
| R: | 5′-CAAAGCTCAGAGAGCCCATC-3′ | |
| Lipocalin-2 | F: | 5′-TCTGGGCCTCAAGGTAACAAC-3′ |
| R: | 5′-AGACAGGTGGGACCTGAACCA-3′ | |
| GAPDH | F: | 5′-GGGTGTGAACCACGAGAAATA-3′ |
| R: | 5′-AGTTGTCATGGATGACCTTGG-3′ | |
2.6.2.1. Relative quantification of KIM-1 and lipocalin-2 gene expression using PCR
Quantitative PCR was applied to determine the relative expression of KIM-1 and lipocalin-2 using the specific primer sets for each gene, shown in Table 2 and Fig. 1. The characteristic of real-time PCR is detecting the amplified DNA as the reaction progresses in real time and collecting the data during the PCR process and not at the end of the process, so the real-time cycler was programmed according to Table 3. A normalizer or reference gene, glyceraldehyde 3-phosphate dehydrogenase (GAPDH), was used as an internal control for experimental variability. This relative quantification method is called the ΔΔCt method or Livak method. It should be noted that the quantitative PCR assay was performed using the Rotor-Gene SYBR Green PCR Kit (Qiagen®, Germany).
Table 2. Primer sets of KIM-1, lipocalin-2 and GAPDH.
| Component | Volume/reaction |
| 5× miScript HiSpec buffer or 5× miScript Hiflex buffer | 4 μl |
| 10× nucleics mix | 2 μl |
| RNase-free water | Variable |
| miScript reverse transcriptase mix | 2 μl |
| Template RNA (added in step 3) | Variable (see Table 1 for recommendations) |
| Total volume | 20 μl |
Fig. 1. Right: The amplification plots of PCR runs for GAPDH (A1), KIM-1 (A2) and lipocalin-2 (A3) genes. The number of cycles is on the x axis and the fluorescence intensity of SYBR Green is on the y axis, whilst the horizontal lines show the threshold line level. Left: Melting curves of the PCR products of GAPDH (B1), KIM-1 (B2) and lipocalin-2 (B3).
Table 3. PCR Protocols for KIM-1, lipocalin-2 and GAPDH.
| KIM-1 | Lipocalin-2 | GAPDH | |
| Initial activation | 95 °C for 10 minutes | ||
| Number of cycles | 40 | 40 | 40 |
| Denaturation | 95 °C for 15 seconds | 95 °C for 15 seconds | 95 °C for 15 seconds |
| Annealing | 55 °C for 15 seconds | 55 °C for 15 seconds | 55 °C for 15 seconds |
| Extension | 60 °C for 15 seconds | 60 °C for 15 seconds | 60 °C for 15 seconds |
| Final extension | 60 °C for 10 minutes | ||
| Melting analysis | |||
2.6.3. ELISA measurements
Interleukin-6 (IL-6) was assayed using the ELISA kit for the in vitro quantitative measurements of rat IL-6 in the kidney tissue homogenates (Kamiya Biomedical C., 12779 Gateway Drive, Seattle, WA98168). Tumor necrosis factor-alpha (TNF-α) was determined using ELISA kits for the quantitative measurement of TNF-α in the rat supernatants and plasma samples (Abcam co., UK). Tumor suppressor gene p53 was assayed using ELISA kits (Active Motif co. 1914, Palomar Oaks Way, Suite 150, Carlsbad, CA 92008 USA).
2.7. Statistical analysis
Statistical analysis was performed using the general linear model (GLM) produced by the Statistical Analysis Systems Institute.33 Duncan's New Multiple Range Test was used to test the mean significant differences.34
3. Results
3.1. Characterization of the nanoparticles of hydroxyapatite, chitosan, and curcumin
3.1.1. Fourier-transform infrared spectroscopy analysis
Functional groups associated with hydroxyapatite nanoparticles (HAP-NPs) were identified by Fourier-Transform Infrared Spectroscopy (FTIR). Fig. 2 shows the FTIR spectrum recorded for the HAP-NPs, as it has revealed the presence of phosphate (PO43–), carbonate (CO32–) and hydroxyl (OH–) groups. The HAP-NP spectrum shows various peaks; the sharp narrow band at 3570.09 cm–1 and the wide band at 3448.69 cm–1 are associated with a hydroxyl group. The intensity bands at about 1461.79 and 1638.565 cm–1 are attributed to components of a trace amount of CO32– and the highest intensity in the phosphate group (PO43–) is detected at 471.464, 672.932, and 1048.94 cm–1 by the vibration bonding and P–O stretching vibration from the phosphate groups.
Fig. 2. FT-IR spectrum of the hydroxyapatite nanoparticles.
The identification of the functional groups of chitosan and the probable interaction between the constituents within the nanoparticle system were analyzed with a scanning range of 500–4000 cm–1. The FTIR spectrum of the CsNPs is presented in Fig. 3. In the chitosan spectra, different stretching vibration bands are observed in the range 3445–2891.89 cm–1, which are attributed to the peaks of N–H stretching from the type II amide (N–H) and the primary amine (NH2). The highly intense absorption band at 3445 cm–1 can be assigned to NH2, N–H and O–H, which are present in Cs in various amounts. The presence of a methyl group (CH3) in acetanilide (NHCOCH3), a methylene group (CH2) in hydroxymethyl (CH2OH) and a methine (CH) group in the pyranose ring was proven by the corresponding stretching vibrations of these groups in the range of 1431–1318 cm–1. The peak for the asymmetric stretch of C–O–C (glycosidic linkage) was found at around 1267 cm–1.
Fig. 3. FT-IR spectrum of the chitosan nanoparticles.
The FTIR spectra of the curcumin nanoparticles is shown in Fig. 4, and the peaks correspond to different functional groups. Among these, the characteristic stretching band at 3473.91 cm–1 is due to the presence of an O–H group, the absorption peak at 1658.84 cm–1 can be assigned to a systemic aromatic C C stretching band, and a peak at 1446.66 cm–1 can be assigned to the bending vibrations of a C O group. The absorption peak at 1080.17 cm–1 represents a benzoate trans C–H vibration, and this indicates that the major peaks of the CurNPs were retained.
Fig. 4. FT-IR spectrum of the curcumin nanoparticles.
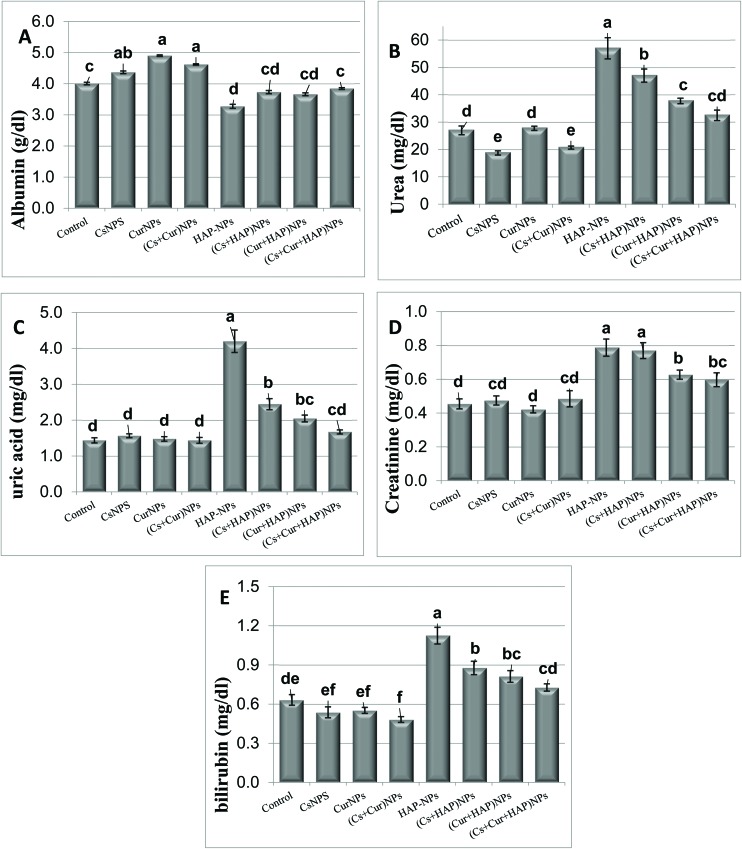
3.2. Kidney biochemical parameters
The statistical analysis of the plasma levels of albumin, total bilirubin, urea, uric acid, and creatinine are presented in Table 4 and Fig. 5. Rats treated with CsNPs or CurNPs alone or in combination showed no significant effect on the plasma levels of total bilirubin, uric acid, and creatinine, but the albumin levels were significantly increased. In contrast, the plasma urea level was significantly decreased in the group treated with CsNPs alone or in combination with CurNPs.
Table 4. Plasma levels of albumin, urea, uric acid, creatinine, and total bilirubin.
| Experimental groups | Parameter |
||||
| Albumin (g dl–1) | Urea (m dl–1) | Uric acid (mg dl–1) | Creatinine (m dl–1) | Bilirubin (mg dl–1) | |
| Control | 3.94 ± 0.07c | 27.0 ± 1.59d | 1.44 ± 0.07d | 0.5 ± 0.03d | 0.63 ± 0.04de |
| CsNPs | 4.41 ± 0.26ab | 18.8 ± 0.76e | 1.57 ± 0.05d | 0.5 ± 0.03cd | 0.54 ± 0.04ef |
| CurNPs | 4.83 ± 0.05a | 27.8 ± 0.76d | 1.48 ± 0.06d | 0.4 ± 0.02d | 0.55 ± 0.02ef |
| (Cs + Cur) NPs | 4.52 ± 0.07a | 20.7 ± 0.56e | 1.44 ± 0.08d | 0.5 ± 0.05cd | 0.48 ± 0.02f |
| HAP-NPs | 3.18 ± 0.03d | 57.0 ± 3.86a | 4.20 ± 0.31a | 0.8 ± 0.05a | 1.13 ± 0.06a |
| (HAP + Cs) NPs | 3.66 ± 0.21cd | 47.0 ± 2.39b | 2.45 ± 0.15b | 0.8 ± 0.05a | 0.88 ± 0.05b |
| (HAP + Cur) NPs | 3.66 ± 0.07cd | 37.8 ± 0.99c | 2.05 ± 0.09bc | 0.6 ± 0.03b | 0.81 ± 0.04bc |
| (HAP + Cs + Cur) NPs | 3.87 ± 0.18c | 32.5 ± 1.88cd | 1.67 ± 0.06cd | 0.6 ± 0.04bc | 0.73 ± 0.03cd |
Fig. 5. The plasma levels of albumin (A), urea (B), uric acid (C), creatinine (D), and total bilirubin (E) in male rats treated orally with CsNPs, CurNPs, and HAP-NPs, alone or in combination.
On the other hand, the rats treated with HAP-NPs showed significantly higher plasma levels of urea, creatinine, uric acid, and total bilirubin but significantly lower levels of albumin compared to the control rats, or to the rats with other nanoparticles. However, the rats treated with CsNPs and/or CurNPs along with HAP-NPs showed significantly lower levels of plasma urea, creatinine, uric acid and bilirubin but significantly higher levels of albumin compared to the rats that received HAP-NPs alone. Furthermore, the rats treated with both CsNPs and CurNPs showed completely normal levels of the above parameters (Table 4 and Fig. 5).
3.3. Kidney cytokines and inflammation mediators
Table 5 and Fig. 6 show the levels of tumor suppressor p53, tumor necrosis factor-α (TNF-α) and interleukin-6 (IL-6). The results show that the treatment with HAP-NPs alone showed a significant increase in P53, TNF-α and IL-6 levels compared to that of the control group. However, the presence of CsNPs and CurNPs alone or in combination with HAP-NPs minimized the toxicity induced by the HAP-NPs in all the above parameters. It is clear from the obtained results that nanoparticles of chitosan and curcumin are able to scavenge the induced toxic effect, but they still do not reach the control values.
Table 5. Mean values ± SE of kidney tumor suppressor P53, tumor necrosis factor-α and interleukin-6 levels.
| Experimental groups | Parameter |
||
| P53 (ng ml–1 protein) | TNF-α (ng ml–1 tissue) | IL-6 (ng ml–1 tissue) | |
| Control | 6.1 ± 0.24d | 50 ± 0.82e | 110 ± 3.1e |
| CsNPs | 5.9 ± 0.26d | 53 ± 2.22e | 110 ± 5.4e |
| CurNPs | 6.6 ± 0.35d | 50 ± 3.71e | 115 ± 3.1e |
| (Cs + Cur) NPs | 5.8 ± 0.29d | 35 ± 0.48f | 103 ± 4.3e |
| HAP-NPs | 40.5 ± 1.88a | 208 ± 5.17a | 351 ± 7.2a |
| (HAP + Cs) NPs | 31.8 ± 1.73b | 140 ± 3.30b | 251 ± 7.2b |
| (HAP + Cur) NPs | 25.3 ± 1.48c | 127 ± 2.78c | 208 ± 4.6c |
| (HAP + Cs + Cur) NPs | 21.8 ± 1.23c | 87 ± 2.27d | 158 ± 4.4d |
Fig. 6. Mean values ± SE of kidney P53 (A), TNF-α (B) and IL-6 (C) in male rats treated orally with CsNPs, CurNPs, and HAP-NPs, alone or in combination.
3.4. Gene expression analysis of kidney injury molecule-1 and lipocalin-2
The results of the renal expressions of Kidney Injury Molecule-1 (KIM-1) and Lipocalin-2 (LCN2) are presented in Table 6 and Fig. 7. The treatment of normal rats daily for 45 days with CsNPs and/or CurNPs alone, or in combination, caused significant down-regulation of the renal gene expression of KIM-1 and LCN2 compared to that of the rats of the control group. However, the rats that received CurNPs showed no significant effect on the renal expression of KIM-1 or LCN2 (Table 6 and Fig. 7). On the other hand, treatment with HAP-NPs resulted in marked up-regulation of renal KIM-1 by about 5.6 fold, and of LCN2 by about 3.6 fold, compared to in the control rats. However, the presence of CsNPs and CurNPs along with HAP-NPs in the combination group significantly suppressed the induced expression of KIM-1 and LCN2. Since the effect of CsNPs is more potent than that of CurNPs, the combined effect therefore showed the best results.
Table 6. Mean values ± SE of renal tissue expression of KIM-1 and lipocalin-2.
| Experimental groups | KIM-1 (fold change) | Lipocalin-2 (fold change) |
| Control | 1.00 ± 0.12d | 1.00 ± 0.27cd |
| CsNPs | 0.67 ± 0.19d | 0.75 ± 0.10d |
| CurNPs | 0.95 ± 0.15d | 0.97 ± 0.13cd |
| (Cs + Cur) NPs | 0.97 ± 0.11d | 0.76 ± 0.17d |
| HAP-NPs | 5.58 ± 1.91a | 3.63 ± 1.33a |
| (Cs + HAP) NPs | 1.50 ± 0.20c | 1.83 ± 0.24b |
| (Cur + HAP) NPs | 1.93 ± 0.34b | 2.01 ± 0.90b |
| (Cs + Cur + HAP) NPs | 1.35 ± 0.09c | 1.30 ± 0.16c |
Fig. 7. Mean values ± SE of kidney injury molecule-1 (A) and lipocalin-2 (B) in male rats treated orally with CsNPs, CurNPs, and HAP-NPs, alone or in combination.
4. Discussion
Nanoparticle induced toxicity is highly attributed to oxidative stress, starting with chromosomal fragmentation, strand breakages of DNA and gene mutation induction, followed by DNA damage and apoptosis.35,36 In 2017, Xue et al. reported that hydroxyapatite nanoparticles (HAP-NPs) could induce cell toxicity and inhibit cell growth in different tissues following intravenous (IV) injection, and this might be related to the residence time of nanoparticles in tissues and the metabolism characteristics of organs.37 In addition, in 2007, Chen et al. observed that HAP-NPs significantly reduced cell viability and induced apoptosis in human gastric cancer cell lines that were characterized by morphological changes, hypodiploid DNA and DNA fragmentation. It was therefore concluded that, regardless of whether these nanoparticles were inhaled or injected or produced from prosthesis, once they enter the body, they are able to be distributed everywhere with the blood circulation and can be deposited to interact with tissues or cells, leading to injury of some organs.38
Many pathophysiological mechanisms are immediately linked to nephrotoxicity induction because conventional markers such as serum creatinine and blood urea nitrogen (BUN) have low specificity and sensitivity, and so diagnosis and treatment timing is often delayed. Thus, new biomarker development is needed for nephrotoxicity specific diagnosis at early stages.39 Biomarkers dominate biomolecules that show the relationship between diseases and exogenous substances. Mainly, biomarkers provide premeditation into the onset mechanism of how these toxicants might adversely affect individuals or groups.40 Urine is considered to be an efficient and attractive sample because it is non-invasive and is easily obtained in significant amounts.41 Such biomarkers have been proposed to help predict adverse effects before damage can be indicated.
In several kidney injury models, an increase in the expressions of lipocalin-2/NGAL and kidney injury molecule-1 (KIM-1) has been confirmed to detect injury before the start of the main histopathological alterations.42,43 When the kidney becomes damaged by reperfusion or ischemia or is exposed to toxic substances such as cisplatin, KIM-1 can be used as a more sensitive biomarker than traditional biomarkers such as creatinine and BUN.44 The biomarker appears quickly and is easily detected in the urine upon nephrotoxicity, because the extracellular domain of KIM-1 is very stable.45 Furthermore, Neutrophil Gelatinase-Associated Lipocalin (NGAL) is synthesized mainly in the maturation process of granulocytes and is induced in epithelial cells by tumorigenesis or inflammation. NGAL is considered to be a well-known sensitive biomarker for early acute kidney injury diagnosis as its concentration in the blood increases upon inflammation or infection.46 Immediately following acute kidney injury, NGAL is massively upregulated in the distal part of the nephron. This leads to upregulated plasma and urinary NGAL levels, resulting from apical and basolateral secretion from nephron epithelia. Impaired proximal tubular reabsorption in the case of proximal tubular injury may further potentiate increased NGAL levels in urine.47–49 These characteristics may make NGAL superior or complementary to creatinine in the diagnosis of acute kidney injury.
Cytokines are low-molecular weight soluble proteins excreted by leukocytes and other cells, especially as a response to antigenic stimuli, and they behave as messengers of the immune system. Cytokines may receive specific denotation that refers to their mechanism of action and predominant synthesizing cells. Thus, cytokines mainly produced by lymphocytes are called lymphokines, while those dominantly synthesized by mononuclear phagocytes are called monokines. Cytokines that act on other leukocytes are called interleukins (IL). The IL is involved in antigen presentation and response, specifically by T-helper lymphocytes.50 Cytokines that control the inflammatory traffic and baseline of leukocytes through chemotaxis are called chemokines (i.e. chemotactic cytokines).50 Mobilization is a fundamental characteristic of the chemokines and immune cells are important facilitators of this process. In addition to chemotaxis, chemokines have a pro-angiogenic effect and promote the degranulation of leukocytes.51
Inflammation is an important host defense mechanism that is characterized by a number of interactions between inflammatory cells and inflammation mediators.52 Uncontrolled inflammation can lead to chronic diseases and tissue injury.53 Generally, inflammation treatment is aimed at either inhibition of the production of inflammatory mediators or inhibition of the activity of inflammatory cells.54 However, most inflammatory diseases are treated with steroidal and nonsteroidal anti-inflammatory drugs that suppress inducible nitric oxide synthase (iNOS), pro-inflammatory cytokine levels, prostaglandin E2 (PGE2), and cyclooxygenase-2.55 Despite this, prolonged use of conventional drugs may produce adverse side effects.56 The mechanisms in which inflammation leads to renal function deterioration have not yet been fully elucidated. Immune-inflammatory mediators are known to modulate adhesion, interstitial migration, and endothelial function of circulating immune cells (monocytes, neutrophils, or leukocytes), as well as being capable of activating resident fibroblasts.57,58
The sub-chronic oral exposure of HAP-NPs induced a pro-inflammatory status in the exposed rats, as indicated by marked elevated levels of inflammatory cytokines: interleukin-6 (IL-6), and tumour necrosis factor-α (TNF-α). In line with the present data, Velard indicated that neutrophils treated with HAP-NPs secreted chemokines and cytokines that contribute to leukocyte chemotaxis.59 In addition, Griffin found that HAP-NPs modulated the production of chemo-attractants in vivo in a particle size and shape dependent manner, leading to the recruitment of different cells.60 TNF-α elevated levels are involved in enhancing and nourishing the recruitment of neutrophils in inflammation sites. Consequently, exposure to HAP-NPs might produce an inflammation in the host tissues, especially in the kidneys. Both in vitro and in vivo studies showed that injury induced by nanoparticles leads to ROS-mediated activation of NF-κB, and to the production of pro-inflammatory mediators such as IL-8, TNF-α, IL-6 and IL-2.61
Furthermore, the oral exposure to HAP-NPs has significantly induced the levels of p53; the potent apoptotic inducer under oxidative stress conditions. The expression of p53 can be induced under a variety of cellular stresses including oxidative stress, DNA damage, and hypoxia.62 P53 may be involved in the induction of apoptosis following exposure to HAP-NPs, and the anti-apoptotic Bcl-2 family expression might be down-regulated by p53.63 It was assumed that p53 played a vital role in the cellular response to ROS-induced apoptosis and DNA damage.64,65
The data of the present study suggested that HAP-NPs activated a p53-mediated apoptosis response. This suggestion is in line with Mroz who documented that nanoparticles and ROS could result in mitochondrial mediated apoptosis pathways, activate p53 and induce DNA damage.66 P53 decreased the levels of the anti-apoptotic Bcl-2 family in mitochondria, and this caused the release of cytochrome c from the mitochondrial intermembrane space into the cytosol, and the activation of the caspase family.67 In agreement with this data, it was documented that HAP-NPs could induce apoptosis via oxidative stress and HAP-NP-induced ROS generation led to the direct damage of cellular components including lysosomal rupture, mitochondrial damage, and DNA damage.68 In response to this DNA damage, the cells either initiate the mechanisms of DNA repair or induce cell cycle arrest and apoptosis. The altered expression of the DNA damage response genes has been observed as a result of exposure to nanoparticles. If the DNA damage extent goes beyond the framework of repair by the body's mechanisms of DNA repair, the cells will initiate a programmed cell death.
At the mRNA gene expression level, HAP-NPs remarkably induced the renal KIM-1 and lipocalin-2 gene expressions. From previous studies, it appeared that KIM-1 and lipocalin-2/NGAL may be the most auspicious biomarkers for renal damage, because it had been shown that their mRNA expression increases significantly following acute kidney injury.69 Moreover, it had been demonstrated that lipocalin-2/NGAL and KIM-1 were particularly induced at the toxicity target site in both animal models and in several human renal diseases, including the proximal tubule epithelium injury.45,69,70
With respect to the inflammatory cytokine levels, the co-administration of CsNPs and/or CurNPs along with HAP-NPs significantly decreased the elevated levels of IL-6 and TNF-α in kidney tissues. Also, the level of p53 changed, which indicates that there is a synergistic effect between the CsNPs or CurNPs when combined. It was also documented that the CurNPs showed anti-inflammatory activity by down-regulating the expression of IL-1β and TNF-α through impairing NF-κB activation.71 This is in agreement with the previous findings that have confirmed that CurNPs reduce the levels of many pro-inflammatory cytokines.72 Abdolahi and Trivedi found that the treatment with CurNPs showed significant reduction of pro-inflammatory cytokines, and improved the natural activity of killer cells and the extent of phagocytosis upon exposure to the CurNP product in cell-lines, because it has several pharmacological activities such as antioxidant-, anti-inflammatory-, antimicrobial-, and anticarcinogenic-activity.73,74 Ganugula reported that CurNPs prevented inflammation and apoptosis induced by streptozotocin in pancreatic beta cells in an animal model of Type 1 diabetes mellitus.75 On the other hand, Seferian and Martinez reported that chitosan has immune-stimulating activity.76 Furthermore, Abdel-Wahhab found that CsNPs modulated gene expression and prevented genotoxicity.27
At the mRNA level, CsNPs and/or CurNPs administration along with HAP-NPs-treated rats significantly ameliorated the renal expression of lipocalin-2 and KIM-1, with nearly complete normalization in the rats administered with CsNPs and/or CurNPs. These data are in line with the obtained data that has been documented, which states that curcumin inhibits atherogenesis by down-regulating LCN2 expression in apolipoprotein E-knockout mice.77 Also, in agreement with Wu, it was confirmed that curcumin has a reno-protective effect by down-regulating the expressions of KIM-1 and LCN2 in glycerol induced acute kidney injury in rats.78
5. Conclusion
In summary, the results suggested that oral treatment with HAP-NPs has induced nephrotoxicity in male rats at several levels, including kidney biochemical parameters, cytokine production, and gene expression of renal KIM-1 and lipocalin-2. Despite the distinguished use of HAP-NPs in the field of tissue engineering and medicine, the search for protection against their toxicity is of great importance. Hence, the present study aimed to explore the protective role of CsNPs and CurNPs, whether they were co-administered alone or in combination along with HAP-NPs, and the results clearly indicated that their combined effect is much stronger than the effect of each one alone.
Conflicts of interest
The authors gratefully acknowledge that there are no conflicts of interest.
References
- Lanone S., Boczkowski J. Curr. Mol. Med. 2006;6:651–663. doi: 10.2174/156652406778195026. [DOI] [PubMed] [Google Scholar]
- Donaldson K., Stone V., Tran C. L., Kreyling W., Borm P. J. J. Occup. Environ. Med. 2004;61:727–728. doi: 10.1136/oem.2004.013243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oberdörster G., Oberdörster E., Oberdörster J. Environ. Health Perspect. 2005;113:823–839. doi: 10.1289/ehp.7339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li X., Liu W., Sun L., Aifantis K. E., Yu B., Fan Y., Feng Q., Cui F., Watari F. J. Biomed. Mater. Res., Part A. 2015;103:2499–2507. doi: 10.1002/jbm.a.35384. [DOI] [PubMed] [Google Scholar]
- Furuzono T., Walsh D., Sato K., Sonoda K., Tanaka J. J. Mater. Sci. Lett. 2001;20:111–114. [Google Scholar]
- Wei G., Ma P. X. Biomaterials. 2004;25(19):4749. doi: 10.1016/j.biomaterials.2003.12.005. [DOI] [PubMed] [Google Scholar]
- Eliaz N., Metoki N. Materials. 2017;10(4):334. doi: 10.3390/ma10040334. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Canillas M., Pena P., Antonio H., Rodríguez M. A. Bol. Soc. Esp. Ceram. Vidrio. 2017;56(3):91–112. [Google Scholar]
- Malmiri H. J., Jahanian M. A. G., Berenjian A. Am. J. Biochem. Biotechnol. 2012;8:203–219. [Google Scholar]
- Rajalakshmi A., Krithiga N., Jayachitra A. Middle East J. Sci. Res. 2013;16(10):1446–1451. [Google Scholar]
- Laroche C., Delattre C., Mati-Baouche N., Salah R., Ursu A. V., Moulti-Mati F., Pierre G. Curr. Org. Chem. 2018;22:641–666. [Google Scholar]
- Xie W., Xu P., Liu Q. Bioorg. Med. Chem. Lett. 2001;11(13):1699–1701. doi: 10.1016/s0960-894x(01)00285-2. [DOI] [PubMed] [Google Scholar]
- Shi B., Shen Z., Zhang H., Bi J., Dai S. Biomacromolecules. 2011a;13(1):146–153. doi: 10.1021/bm201380e. [DOI] [PubMed] [Google Scholar]
- Shi L. E., Tang Z. K., Yi Y., Chen J. S., Xiong W. Y., Ying G. Q. Chem. Biochem. Eng. Q. 2011b;25(1):83–88. [Google Scholar]
- Grenha A., Seijo B., Remunán-López C. Eur. J. Pharm. Sci. 2005;25:427–437. doi: 10.1016/j.ejps.2005.04.009. [DOI] [PubMed] [Google Scholar]
- Gupta S. C., Patchva S., Koh W., Aggarwal B. B. Clin. Exp. Pharmacol. Physiol. 2012;39(3):283–299. doi: 10.1111/j.1440-1681.2011.05648.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maheshwari R. K., Singh A. K., Gaddipati J., Srimal R. C. Life Sci. 2006;78:2081–2087. doi: 10.1016/j.lfs.2005.12.007. [DOI] [PubMed] [Google Scholar]
- Garodia P., Ichikawa H., Malani N., Sethi G., Aggarwal B. B. J. Soc. Integr. Oncol. 2007;5:25–37. doi: 10.2310/7200.2006.029. [DOI] [PubMed] [Google Scholar]
- Kakarala M., Brenner D. E., Korkaya H., Cheng C., Tazi K., Ginestier C., Wicha M. S. Breast Cancer Res. Treat. 2010;122:777–785. doi: 10.1007/s10549-009-0612-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hatcher H., Planalp R., Cho J., Torti F. M., Torti S. V. Cell. Mol. Life Sci. 2008;65:1631–1652. doi: 10.1007/s00018-008-7452-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Venkatesha S. H., Berman B. M., Moudgil K. D. Bioorg. Med. Chem. 2011;19:21–29. doi: 10.1016/j.bmc.2010.10.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shamsara J., Sharif S., Afsharnezhad S., Lotfi M., Raziee H. R., Ghaffarzadegan K., Behravan J. Cancer Invest. 2009;27:825–829. doi: 10.1080/07357900902783211. [DOI] [PubMed] [Google Scholar]
- Ganjali S., Sahebkar A., Mahdipour E., Jamialahmadi K., Torabi S., Akhlaghi S., Ghayour-Mobarhan M. Sci. World J. 2014;3:898361. doi: 10.1155/2014/898361. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sabry R., Preparation of hydroxyapatite nanoparticles by using emulsion liquid membrane, [PhD Thesis], Al-Azhar University, Egypt, 2012. [Google Scholar]
- Tang Z. X., Qian J. Q., Shi L. E. Appl. Biochem. Biotechnol. 2007a;136(1):77–96. doi: 10.1007/BF02685940. [DOI] [PubMed] [Google Scholar]
- Tang Z. X., Shi L. E., Qian J. Q. Biochem. Eng. J. 2007b;34(3):217–223. [Google Scholar]
- Abdel-Wahhab M. A., Aljawish A., El-Nekeety A. A., Abdel-Aiezm S. H., Abdel-Kader H. A., Rihn B. H., Joubert O. Toxicol. Rep. 2015;2:737–747. doi: 10.1016/j.toxrep.2015.05.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yadav A., Lomash V., Samim M., Flora S. J. Chem.-Biol. Interact. 2012;199(1):49–61. doi: 10.1016/j.cbi.2012.05.011. [DOI] [PubMed] [Google Scholar]
- Elkamary Y. M. H. and Elkholi M. B., A novel process for the preparation of medical natural hydroxyapatite, Patent cooperation treaty (PCT), 2012.
- Venkatesan J., Qian Z. J., Ryu B., Thomas N. V., Kim S. K. Biomed. Mater. 2011;6(3):035003. doi: 10.1088/1748-6041/6/3/035003. [DOI] [PubMed] [Google Scholar]
- Yousef N. M. Int. J. Dev. Res. 2014;4(9):1875–1879. [Google Scholar]
- Rached E., Hoffmann D., Blumbach K., Weber K., Dekant W., Mally A. Toxicol. Sci. 2008;103(2):371–381. doi: 10.1093/toxsci/kfn040. [DOI] [PubMed] [Google Scholar]
- SAS, Statistical Analysis System, SAS Procedure Guide. Release 6.03 Edition, SAS Institute Inc., Cary, Nc, U.S.A, 1998.
- Duncan D. B. Biometrics. 1955;11(1):1–42. [Google Scholar]
- Singh N., Manshian B., Jenkins G. J. S., Griffiths S. M., Williams P. M., Maffeis T. G. G., Wright C. J., Doak S. H. Biomaterials. 2009;30:3891–3914. doi: 10.1016/j.biomaterials.2009.04.009. [DOI] [PubMed] [Google Scholar]
- Al Gurabi M. A., Ali D., Alkahtani S., Alarifi S. OncoTargets Ther. 2015;8:295. doi: 10.2147/OTT.S77572. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xue Y., Chen Q., Sun J. J. Appl. Toxicol. 2017;37:1004–1016. doi: 10.1002/jat.3450. [DOI] [PubMed] [Google Scholar]
- Chen X., Deng C., Tang S., Zhang M. Biol. Pharm. Bull. 2007;30:128–132. doi: 10.1248/bpb.30.128. [DOI] [PubMed] [Google Scholar]
- Ferguson M. A., Vaidya V. S., Bonventre J. V. Toxicology. 2008;245:182–193. doi: 10.1016/j.tox.2007.12.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Finn W. F., Porter G. A. Clin. Nephrotoxins. 2003:621–655. [Google Scholar]
- Wu Y., Yang L., Su T., Wang C., Liu G., Li X. M. Clin. J. Am. Soc. Nephrol. 2010;5:1954–1959. doi: 10.2215/CJN.02370310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amin R. P., Vickers A. E., Sistare F., Thompson K. L., Roman R. J., Lawton M., Bennett L. Environ. Health Perspect. 2004;112:465–479. doi: 10.1289/ehp.6683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thukral S. K., Nordone P. J., Hu R., Sullivan L., Galambos E., Fitzpatrick V. D., Afshari C. A. Toxicol. Pathol. 2005;33(3):343–355. doi: 10.1080/01926230590927230. [DOI] [PubMed] [Google Scholar]
- Vaidya V. S., Ramirez V., Ichimura T., Bobadilla N. A., Bonventre J. V. Am. J. Physiol. Renal. Physiol. 2006;290:517–529. doi: 10.1152/ajprenal.00291.2005. [DOI] [PubMed] [Google Scholar]
- Ichimura T., Hung C. C., Yang S. A., Stevens J. L., Bonventre J. V. Am. J. Physiol. Renal. Physiol. 2004;286(3):552–563. doi: 10.1152/ajprenal.00285.2002. [DOI] [PubMed] [Google Scholar]
- Ohlsson S., Wieslander J., Segelmark M. Clin. Exp. Immunol. 2003;131:528–535. doi: 10.1046/j.1365-2249.2003.02083.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mori K., Lee H. T., Rapoport D., Drexler L. R., Foster K., Yang J., Schmidt-Ott K. M., Chen X., Li J. Y., Weiss S. J. Clin. Invest. 2005;115:610–621. doi: 10.1172/JCI23056. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmidt-Ott K. M., Mori K., Li J. Y., Kalandadze A., Cohen D. J., Devarajan P., Barasch J. J. Am. Soc. Nephrol. 2007;18:407–413. doi: 10.1681/ASN.2006080882. [DOI] [PubMed] [Google Scholar]
- Schmidt-Ott K. M. Nephrol., Dial., Transplant. 2011;26:762–764. doi: 10.1093/ndt/gfr006. [DOI] [PubMed] [Google Scholar]
- Abbas A. K. and Lichtman A. H., Cellular and Molecular Immunology, 5th edn, Saunders, E. Sci., Philadelphia, 2003, pp. 243–274. [Google Scholar]
- Mackay C. R. Nat. Immunol. 2001;2:95–101. doi: 10.1038/84298. [DOI] [PubMed] [Google Scholar]
- Villarreal G., Zagorski J., Wahl S. M. Inflammation: Acut. 2001 [Google Scholar]
- Mantovani A., Allavena P., Sica A., Balkwill F. Nature. 2008;454:436–444. doi: 10.1038/nature07205. [DOI] [PubMed] [Google Scholar]
- Benjamini E., Sunshine G. and Leskowitz S., Immunology: A Short Course, John Wiley and Sons, New York, 3rd edn, 1996. [Google Scholar]
- Pohanka M., Snopkova S., Havlickova K., Bostik P., Sinkorova Z., Fusek J. Curr. Med. Chem. 2011;18:539–551. doi: 10.2174/092986711794480140. [DOI] [PubMed] [Google Scholar]
- Lin L., Hu K. Am. J. Clin. Exp. Immunol. 2014;3:30–36. [PMC free article] [PubMed] [Google Scholar]
- Ruiz-Ortega M., Lorenzo O., Suzuki Y., Rupérez M., Egido J. Curr. Opin. Nephrol. Hypertens. 2001;10:321–329. doi: 10.1097/00041552-200105000-00005. [DOI] [PubMed] [Google Scholar]
- Eddy A. A. Adv. Chronic Kidney Dis. 2005;12:353–365. doi: 10.1053/j.ackd.2005.07.011. [DOI] [PubMed] [Google Scholar]
- Velard F., Laurent-Maquin D., Guillaume C., Bouthors S., Jallot E., Nedelec J. M., Belaaouaj A., Laquerriere P. Acta Biomater. 2009;5(5):1708–1715. doi: 10.1016/j.actbio.2009.01.008. [DOI] [PubMed] [Google Scholar]
- Griffin G. K., Newton G., Tarrio M. L., Bu D. X., Maganto-Garcia E., Azcutia V., Alcaide P., Grabie N., Luscinskas F. W., Croce K. J., Lichtman A. H. J. Immunol. 2012;188:6287–6299. doi: 10.4049/jimmunol.1200385. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wallace J. L. Br. J. Pharmacol. 2007;152(4):421–428. doi: 10.1038/sj.bjp.0707396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simbula G., Columbano A., Ledda-Columbano G. M., Sanna L., Deidda M., Diana A., Pibiri M. Apoptosis. 2007;12(1):113–123. doi: 10.1007/s10495-006-0487-9. [DOI] [PubMed] [Google Scholar]
- Ok Y. J., Myoung H., Kim Y. K., Lee J. H., Kim M. J., Yun P. Y. Oral Oncol. 2009;45(3):266–272. doi: 10.1016/j.oraloncology.2008.05.001. [DOI] [PubMed] [Google Scholar]
- Cardaci S., Filomeni G., Rotilio G., Ciriolo M. R. Mol. Pharmacol. 2008;74:1234–1245. doi: 10.1124/mol.108.048975. [DOI] [PubMed] [Google Scholar]
- Chowdhury R., Chowdhury S., Roychoudhury P., Mandal C., Chaudhuri K. Apoptosis. 2009;14:108–123. doi: 10.1007/s10495-008-0284-8. [DOI] [PubMed] [Google Scholar]
- Mroz R. M., Schins R. P. F., Li H., Jimenez L. A., Drost E. M., Holownia A., Donaldson K. Eur. Respir. J. 2008;31(2):241–251. doi: 10.1183/09031936.00006707. [DOI] [PubMed] [Google Scholar]
- Hung H. S., Wu C. C., Chien S., Hsu S. H. Biomaterials. 2009;30:1502–1511. doi: 10.1016/j.biomaterials.2008.12.003. [DOI] [PubMed] [Google Scholar]
- Jin Y., Liu X., Liu H., Chen S., Gao C., Ge K., Zhang C., Zhang J. RSC Adv. 2017;7:13010–13018. [Google Scholar]
- Mishra J., Ma Q., Prada A., Mitsnefes M., Zahedi K., Yang J., Devarajan P. J. Am. Soc. Nephrol. 2003;14(10):2534–2543. doi: 10.1097/01.asn.0000088027.54400.c6. [DOI] [PubMed] [Google Scholar]
- Mishra J., Mori K., Ma Q., Kelly C., Barasch J., Devarajan P. Am. J. Nephrol. 2004;2:307–315. doi: 10.1159/000078452. [DOI] [PubMed] [Google Scholar]
- Hsieh C. H., Frink M., Hsieh Y. C., Kan W. H., Hsu J. T., Schwacha M. G., Choudhry M. A., Chaudry I. H. J. Immunol. 2008;181:2806–2812. doi: 10.4049/jimmunol.181.4.2806. [DOI] [PubMed] [Google Scholar]
- Anand P., Nair H. B., Sung B., Kunnumakkara A. B., Yadav V. R., Tekmal R. R., Aggarwal B. B. Biochem. Pharmacol. 2010;79:330–338. doi: 10.1016/j.bcp.2009.09.003. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- Abdolahi M., Tafakhori A., Togha M., Okhovat A. A., Siassi F., Eshraghian M. R., Sedighiyan M., Djalali M., Honarvar N. M., Djalali M. Immunogenetics. 2017;69:371–378. doi: 10.1007/s00251-017-0992-8. [DOI] [PubMed] [Google Scholar]
- Trivedi M. K., Mondal S. C., Gangwar M., Jana S. Inflammopharmacology. 2017;25(6):609–619. doi: 10.1007/s10787-017-0395-3. [DOI] [PubMed] [Google Scholar]
- Ganugula R., Arora M., Jaisamut P., Wiwattanapatapee R., Jørgensen H. G., Venkatpurwar V. P., Zhou B., Rodrigues Hoffmann A., Basu R., Guo S., Majeti N. V. R. K. Br. J. Pharmacol. 2017;174(13):2074–2084. doi: 10.1111/bph.13816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seferian P. G., Martinez M. L. Vaccine. 2000;19(6):661–668. doi: 10.1016/s0264-410x(00)00248-6. [DOI] [PubMed] [Google Scholar]
- Meng N., Gong Y., Zhang J., Mu X., Song Z., Feng R., Zhang H. J. Biomater. Appl. 2018;33(7):946–954. doi: 10.1177/0885328218815328. [DOI] [PubMed] [Google Scholar]
- Wu J., Pan X., Fu H., Zheng Y., Dai Y., Yin Y., Hou D. Sci. Rep. 2017;7(1):10114. doi: 10.1038/s41598-017-10693-4. [DOI] [PMC free article] [PubMed] [Google Scholar]