Abstract
Oral administration of bovine collagen peptide (CP) combined with calcium citrate (CC) has been found to inhibit bone loss in ovariectomized rats. However, the protective effects of CP and CP–CC against bone loss have not been investigated in a tail-suspension simulated microgravity (SMG) rat model. Adult Sprague-Dawley rats (n = 40) were randomly divided into five groups (n = 8): a control group with normal gravity, a SMG control group, and three SMG groups that underwent once-daily gastric gavage with CP (750 mg/kg body weight), CC (75 mg/kg body weight) or CP–CC (750 and 75 mg/kg body weight, respectively) for 28 days. After sacrifice, the femurs were analyzed by dual-energy X-ray absorptiometry, three-point bending mechanical tests, microcomputed tomography, and serum bone metabolic markers. Neither CP nor CP–CC treatment significantly inhibited bone loss in SMG rats, as assessed by dual-energy X-ray absorptiometry and three-point bending mechanical tests. However, both CP and CP–CC treatment were associated with partial prevention of the hind limb unloading-induced deterioration of bone microarchitecture, as demonstrated by improvements in trabecular number and trabecular separation. CP–CC treatment increased serum osteocalcin levels. Dietary supplementation with CP or CP–CC may represent an adjunct strategy to reduce the risk of fracture in astronauts.
Keywords: collagen peptide, calcium citrate, bone loss, tail-suspended rats
1. Introduction
The development and maintenance of bone structure depends on mechanical stimulation. Numerous studies have demonstrated that mechanical loading promotes bone formation, whereas the absence of mechanical stimulation decreases bone mass [1]. During space flight, astronauts experience microgravity that leads to serious physiological changes, one of the most prominent being bone loss, which increases fracture risk [2]. Long-term exposure to microgravity is associated with increased bone resorption and decreased bone formation, with a reduction in bone mineral density (BMD) of approximately 2% after one month, equivalent to annual bone loss in postmenopausal women [3].
Drug intervention is not routinely used in space flight and exercise has been combined with nutrition improvement. However, because of the lack of mechanical load or duration of space flight, osteoblast stimulation is insufficient to maintain bone mass [4]. This has led to a focus on pharmaceutical interventions such as osteoporosis drugs, but their potential to prevent bone loss in space remains to be clarified. In particular, the effects of different drugs, alone or combined, and dose–response relationships for improvements in bone quality and regeneration have not been investigated. Currently available therapeutic agents that are conventionally used for the prevention of bone loss have several side effects [5]. Therefore, novel therapeutic approaches are being explored, which have fewer side effects, while effectively minimizing the loss of bone mass.
Collagen, which is a major component of all tissues can be produced from various sources. For example, they can be extracted from sea animals such as shark, marine sponges [6,7]. In addition, they can be extracted from terrestrial animals such as porcine, bovine animals. However, collagen is mostly produced from pork skin and bovine bones. Moreover, bovine collagen is currently widely used for many applications such as foods and cosmetics [6].
Collagen peptide (CP), known as collagen hydrolysates, is mainly composed of mixtures of peptides obtained by partial hydrolysis of gelatins [8]. Due to its higher digestive absorption and safety, oral supplementation with CP for the restoration of bone joints has gained increasing scientific attention [9]. Daily doses of 150 or 500 mg/kg of CP for up to three months significantly prevented bone loss in ovariectomized (OVX) rats compared with control rats [10]. CP also improved vertebral composition and biomechanical strength, and increased the quantity and volume ratio of lumbar trabecular bone, which demonstrated its effect on bone protection [8]. In postmenopausal women with osteopenia, administration of calcium–collagen chelate supplements was found to improve bone mineral density and increase the rate of bone formation and bone resorption [11].
A previous study in OVX rats demonstrated that daily doses of either CP alone (750 mg/kg) or CP–calcium citrate (CC; 750–75 mg/kg) had an osteoprotective effect by inhibiting the loss of bone mineral density. Moreover, CP–CC suppressed trabecular bone loss and improved the microarchitecture of the distal femur [12]. However, the effect of CP on prevention and restoration of bone loss induced by microgravity has not been reported. The main purpose of this study was to observe the effects of oral administration of CP, alone and in combination with CC, on bone structure and bone metabolism in rats under hind limb unloading simulated microgravity (SMG), and to provide a theoretical basis for the use of CP–CC to prevent and treat microgravity-related osteoporosis.
2. Results
2.1. Effect of Tail Suspension on Body Weight
Before tail suspension, there was no difference in the body weight of rats in the CN group and the four SMG groups (Figure 1). However, after 28 days of tail suspension, all SMG groups had lower body weight than the CN group (p = 0.000).
Figure 1.
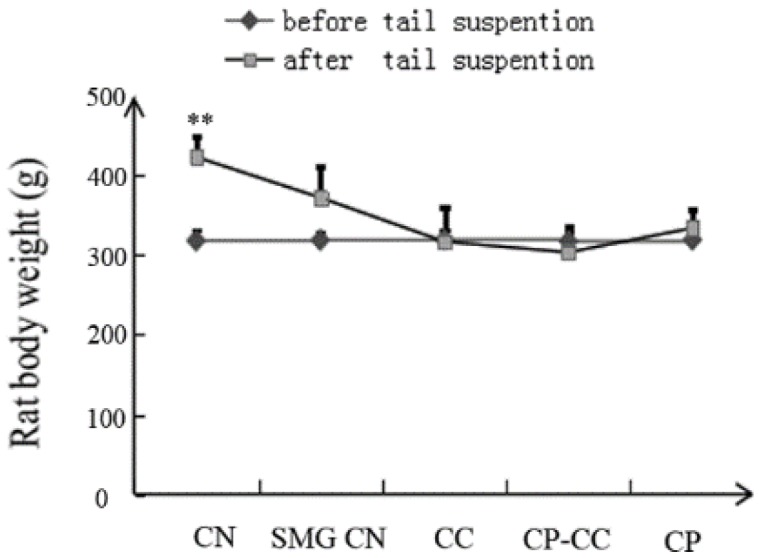
Body weight of rats before and after tail suspension (n = 8): CN, normal gravity control group; SMG CN, tail-suspended simulated microgravity control (SMG) groups; CC, SMG rats treated once-daily with 75 mg/kg calcium citrate (CC); CP–CC, SMG rats treated once-daily with 750 mg/kg bovine collagen peptide (CP) combined with 75 mg/kg CC; and CP, SMG rats treated once-daily with 750 mg/kg bovine CP. ** p < 0.01 vs. SMG CN.
2.2. Bone Mineral Density Assessment
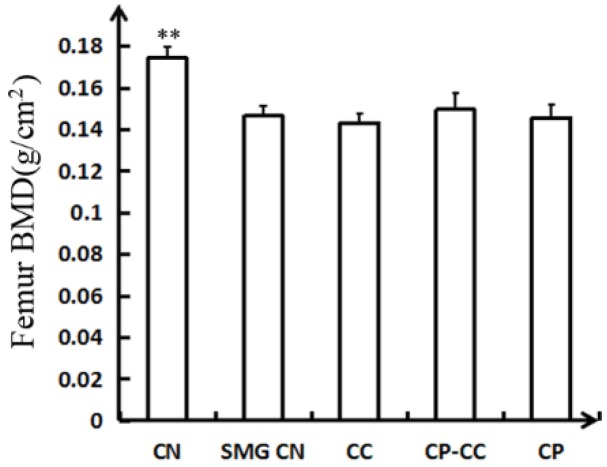
Changes in femoral bone mineral density, as determined by dual-energy X-ray absorptiometry analysis, are shown in Figure 2. The SMG group had a 17.82% reduction in femoral bone mineral density compared with the CN group (p = 0.000). No significant improvement in the femoral bone mineral density of tail-suspended rats was observed after oral administration of CC, CP, or CP–CC.
Figure 2.
Femoral BMD determined by dual-energy X-ray absorptiometry analysis (n = 6) CN, normal gravity control group; SMG CN, tail-suspended simulated microgravity control (SMG) groups; CC, SMG rats treated once-daily with 75 mg/kg calcium citrate (CC); CP–CC, SMG rats treated once-daily with 750 mg/kg bovine collagen peptide (CP) combined with 75 mg/kg CC; and CP, SMG rats treated once-daily with 750 mg/kg bovine CP. ** p <0.01 vs. SMG CN.
2.3. Biomechanical Performance
Table 1 summarizes the maximum force to failure, deformation at hardness, and hardness work cycle values for the CN, SMG CN, SMG CC, SMG CP, and SMG CP–CC groups. Maximum force applied to failure in the SMG groups was 95.25 ± 18.64 N compared with 139.80 ± 24.73 N (p = 0.000) in the CN group, with an average decline of 31.65% in the SMG groups over the 28-day period. Maximum force applied to failure in the CC and CP–CC groups was 104.235 ± 17.868 and 99.442 ± 18.639 N, respectively, with a slight increase after tail suspension for 28 days. Deformation at hardness and femoral energy absorption were not significantly different among the four SMG groups.
Table 1.
Femoral biomechanics analysis.
| CN | SMG CN | CC | CP–CC | CP | |
|---|---|---|---|---|---|
| FFS | 139.80 ± 24.73 ** | 95.25 ±1 8.64 | 104.24 ± 17.87 | 99.44 ± 16.19 | 97.10 ± 15.01 |
| FD | 7.39 ± 0.56 | 7.40 ± 0.68 | 7.48 ± 0.57 | 7.37 ± 0.58 | 7.4 ± 0.55 |
| FEA | 96.77 ± 42.94 | 67.478 ± 17.23 | 93.96 ± 24.37 | 76.94 ± 20.76 | 82.34 ± 10.85 |
FFS, Femoral fracture strength (N) FD, Femoral deformation (mm) FEA, Femoral energy absorption (mJ) CN, normal gravity control group; SMG CN, tail-suspended simulated microgravity (SMG) control groups; CC, SMG rats treated once-daily with 75 mg/kg calcium citrate (CC); CP–CC, SMG rats treated once-daily with 750 mg/kg bovine collagen peptide (CP) combined with 75 mg/kg CC; CP, SMG rats treated once-daily with 750 mg/kg bovine CP. Data are expressed as means ± SD (n = 6). ** p < 0.01 vs. SMG CN.
2.4. CP–CC Treatment Inhibited Microgravity-Induced Deterioration of Bone Microarchitecture
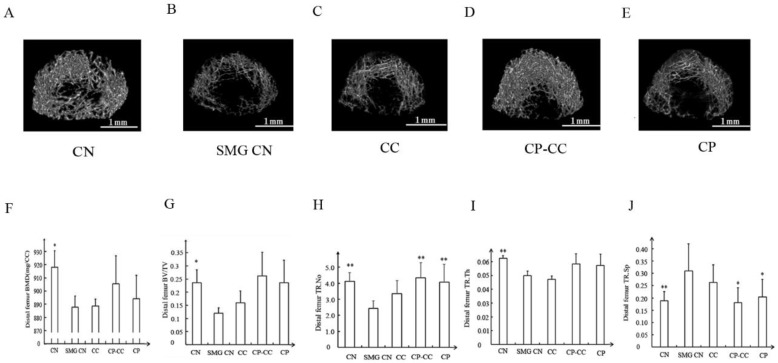
Three-dimensional microcomputed tomography images of the distal femurs in all groups are shown in Figure 3. The CN group showed dense, intact trabecular bone, whereas the area and density of trabecular bone was reduced in the SMG CN group. Oral administration of CP or CP–CC improved bone microarchitecture and density, with thicker trabecular bone, indicating that bone loss was ameliorated.
Figure 3.
Representative three-dimensional microcomputed images (A–E) and (F–J, BMD, BV/TV, TR.N, TR.Th, TR.Sp) analysis of the microstructural indices of distal femurs in each group (n = 5) CN, normal gravity control group; SMG CN, tail-suspended simulated microgravity (SMG) control groups; CC, SMG rats treated once-daily with 75 mg/kg calcium citrate (CC); CP–CC, SMG rats treated once-daily with 750 mg/kg bovine collagen peptide (CP) combined with 75 mg/kg CC; CP, SMG rats treated once-daily with 750 mg/kg bovine CP. BV/TV, bone volume/tissue volume; Tb.N (TR.No), trabecular number; Tb.Th (TR.Th), trabecular thickness; Tb.Sp (TR.Sp), trabecular separation. * p < 0.05 vs. SMG group. ** p < 0.01 vs. SMG group. Scale bars, 1 mm.
Analysis of microstructural indices demonstrated that, compared with CN rats, the SMG CN rats had reductions in bone mineral density (3.29%, p = 0.029), BV/TV (91.48%, p = 0.026), Tb.N (41.12%, p = 0.006), and Tb.Th (20.63%, p = 0.005). Treatment with CP or CP–CC did not affect the reductions in bone mineral density, BV/TV, and Tb.Th compared with SMG CN rats; in contrast, Tb.N was enhanced by CP (67.52%, p = 0.001) and CP–CC (79.17%, p = 0.004). However, there were no differences between SMG CN and SMG CC rats. In the SMG rats, CP and CP–CC treatment decreased Tb.Sp by 34.52% (p = 0.05) and 41.94% (p = 0.02), respectively, compared with SMG CN rats, which had greater Tb.Sp than CN rats (38.7%, p = 0.021).
2.5. Serum Levels of Bone Turnover Biomarkers
Bone turnover was evaluated by serum levels of biomarkers of resorption (CTX and TRAP-5b) and formation (ALP, osteocalcin, and PINP). After four weeks, serum levels of Ca, ALP, and osteocalcin in the SMG groups were lower than in the CN group (p < 0.05, Table 2), except for osteocalcin levels in the SMG CP–CC group. After oral administration of CP–CC, serum osteocalcin levels of SMG rats were significantly higher than those of the SMG CN and other SMG treatment groups. PINP levels in SMG CN and SMG CP rats were significantly higher than in CN rats, whereas treatment with CC inhibited the increase in PINP (p < 0.05).
Table 2.
Serum levels of bone turnover biomarkers analysis.
| CN | SMG CN | CC | CP–CC | CP | |
|---|---|---|---|---|---|
| Ca | 2.60 ± 0.11011 | 2.38 ± 0.14 ## | 2.37 ± 0.08 ## | 2.38 ± 0.08 ## | 2.38 ± 0.05 ## |
| P | 2.64 ± 0.22 | 2.66 ± 0.19 | 3.01 ± 0.41 | 2.86 ± 0.22 | 2.65 ± 0.41 |
| ALP (U/L) | 202.33 ± 21.05 ** | 155.86 ± 25.53 ## | 174.57 ± 38.20 # | 148.33 ± 0.38 ## | 162.00 ± 24.36 ## |
| Osteocalcin (ng/mL) | 3.90 ± 0.82 ** | 2.66 ± 0.37 ## | 2.70 ± 0.51 ## | 3.96 ± 0.51 ** | 2.98 ± 0.70 ## |
| PINP (ng/mL) | 34.00 ± 2.19 ** | 77.00 ± 5.29 ## | 37.20 ± 2.17 ** | 51.20 ± 17.46 | 68.00 ± 8.81 ## |
| CTX (ng/mL) | 66.14 ± 8.80 | 60.43 ± 12.00 | 64.71 ± 8.54 | 60.25 ± 9.30 | 65.00 ± 6.00 |
| TRAP-5b | 2.07 ± 0.45 | 2.18 ± 0.22 | 3.71 ± 1.88 | 4.29 ± 1.50 | 3.34 ± 1.28 |
CN, normal gravity control groups; SMG CN, tail-suspension simulated microgravity control groups; CC, SMG rats were administrated 75 mg/kg calcium citrate daily; CP–CC, SMG rats were administrated 750 mg/kg bovine collagen peptide combined with 75 mg/kg calcium citrate daily; CP, SMG rats were administrated 750 mg/kg bovine collagen peptide daily, as indicated. ALP, bone-specific alkaline phosphatase; PINP, procollagen type I N-terminal propeptide; CTX, carboxyterminal telopeptide of collagen type 1. Data are expressed as means ± SD (n = 8). # p < 0.05 vs. CN. ## p < 0.01 vs. CN. ** p < 0.01 vs. SMG CN.
3. Discussion
Bone is a complex tissue. Hydroxyapatite salts (calcium and phosphorus) with collagen form a unique matrix that plays an important role in bone hardness. In addition, the Ca/P ratio in bones is vital for osteoporosis and may provide high reliability for diagnosis, prevention, and treatment of bone disorders. Collagen fibril diameter is related to bone site and Ca/P ratio. Ca/P ratio can serve as a reliable index of bone quality [13].
CP-based drugs play a role in the prevention and treatment of osteoporosis as orally administered, intestinally absorbed forms [14]. During space flight, weightlessness leads to calcium deficiency. High calcium intake from dietary supplementation does not affect bone metabolism, but prevents an elevation in serum calcium levels through increased calcitriol levels [15]. Therefore, intragastric administration was thought to be the most appropriate delivery route for bovine CP compounds combined with CC because of relative proximity to the pathologic process in the in vivo environment.
The results of this study show that rats in all SMG groups had significantly lower body weight than rats in the CN group, which is consistent with previous reports [16,17]. This may be related to loss of water electrolytes and loss of appetite caused by redistribution of body fluid under SMG.
Conventionally, the diagnosis and treatment of osteoporosis is assessed by bone mineral density, as measured by dual-energy X-ray absorptiometry [18]. Evaluation of bone biomechanical properties is indispensable to determine the quality of bone, and the intensity of bone fracture directly correlates to the relationship between the structure of the bone and the strength and hardness of the bone [19,20,21]. The present data show that SMG caused marked reductions in bone mineral density and femoral fracture strength of rats, which is consistent with previous reports [2,22]. These findings demonstrate that real or simulated weightlessness can cause bone changes, which are characterized by a decrease in cortical bone and cancellous bone formation [23].
Previous studies found that CP–CC led to substantial improvements in the matrix structure and quality of trabecular bone in the femurs of ovariectomized rats [12]. Dual-energy X-ray absorptiometry and biomechanical tests showed that CP–CC had a significant effect on femoral bone mineral density and fracture strength of ovariectomized rats [24]. Therefore, in the present study, the effects of CP–CC on bone remodeling were evaluated in a rat model of SMG. However, neither CP alone nor in combination with CC inhibited bone loss in SMG rats, based on dual-energy X-ray absorptiometry and three-point bending mechanical test analyses. Furthermore, assessment of the femoral microarchitecture using microcomputed tomography revealed that neither CP nor CP–CC had obvious effects on bone mineral density, BV/TV, or Tb.Th in tail-suspended SMG rats. A possible reason may be that CP and CP–CC act primarily via stimulation of bone formation to inhibit bone loss [8]. However, microgravity causes uncoupling of formation and resorption in bone remodeling, which may contribute to bone loss [25,26].
Nevertheless, CP and CP–CC treatment partially ameliorated microgravity-induced deterioration of bone microarchitecture, as indicated by suppressing both the reduction in Tb.N and the increase in Tb.sp induced by hind limb unloading simulated microgravity. This result is consistent with a previous report that oral administration of CP or CP–CC inhibited bone loss in ovariectomized rats [12]. This may represent an adjunctive dietary strategy to reduce the risk of fracture in astronauts.
Some previous reports suggest that serum calcium levels are not altered by tail suspension [27,28]. On the contrary, calcium levels in the tail-suspended rats were significantly reduced in another previous study [22]. This implies that intestinal calcium absorption was reduced during tail suspension [29]. Serum concentrations of bone turnover markers are reflective of bone remodeling activity, and can potentially be used as surrogate markers of the rate of bone formation or bone resorption [30]. The present study also showed that ALP activity and osteocalcin levels were decreased under SMG conditions, which is consistent with a previous report [31,32], indicating that osteoblast activity was inhibited by microgravity [33]. The main function of osteocalcin is to maintain the normal mineralization rate of bone. Interesting, osteocalcin levels in CP–CC-treated SMG rats were similar to those of CN rats, demonstrating that CP–CC promotes osteocalcin levels of osteoblasts by hind limb unloading simulated microgravity. PINP is a well-established marker of bone formation, which is produced by formation of type I collagen, a major component of the bone matrix, by amino-terminal and carboxy-terminal splicing of type I procollagen in osteoblasts [34]. Conversely, β-CTX is a marker of bone resorption, reflecting the degradation of type I collagen by osteoclasts to produce amino-terminal and carboxy-terminal fragments [35]. After ovariectomy or orchidectomy, serum TRACP-5b levels, which reflect the number of osteoclasts rather than their activity [36], are expected to decline, and the histomorphometrically determined total number of osteoclasts in bone tissue is decreased owing to substantial bone loss [37]. Changes in serum PINP or β-CTX levels induced by microgravity have reported before [38]. High serum PINP levels in SMG or CP-treated rats in our study demonstrated that hind limb unloading promoted formation of type I collagen. However, treatment with CC significantly suppressed the hind limb unloading-induced increase in type I collagen formation. A possible reason for the lack of effects of CP–CC on serum PINP levels was that the actions of CP and CC counteracted one another. In this study, serum β-CTX levels were slightly reduced in tail-suspended rats, which is contrary to a previous report that CP–CC or CP supplementation inhibited the degradation of collagen in ovariectomized rats [22], and serum TRACP-5b levels of tail-suspended rats were not altered. It may be speculated that this is related to the time of blood sampling or the high extent of bone loss under hind limb unloading simulated microgravity.
This study has some limitations. CP–CC treatment did not display a dose–response effect, and the duration of tail suspension was relatively short. As a dietary supplement, the effective time of CP–CC treatment in ovariectomized rats is three months, while, in this study, tail suspension was only maintained for 28 days. Thus, differing results among studies may be due to the mechanisms underlying the two animal models, and, in the tail suspension model, rapid bone loss in the process of CP–CC is not caused by the changes of bone mineral density obviously. Second, continuous blood sampling was not performed for observation of bone turnover markers.
4. Materials and Methods
4.1. Animals and Treatment
Bovine bone CP (prepared by our laboratory) and CC (Dongtai Food Ingredients, Lianyungang, China) were used. Male, three-month-old Sprague-Dawley rats (n = 40, body weight: 300 ± 20 g) were obtained from the animal facility of the China Astronaut Research and Training Center (Beijing). Animals were maintained in cages under standard conditions (12-h light/dark cycle with free access to food and water). After feeding adaptation for seven days, the rats were divided into five groups (n = 8 each): a control group with normal gravity (CN), an untreated hind limb unloading simulated microgravity group (SMG CN), and three SMG groups that underwent once-daily treatment by gastric gavage with CC (75 mg/kg), CP (750 mg/kg), or CP–CC (750 and 75 mg/kg). Bovine CP and CC were dissolved in distilled water. The tail-suspended rats were fixed by the tail at a 30° head-down angle to mimic microgravity [39]. Briefly, the rats were individually caged, suspended by the tail using a strip of adhesive surgical tape attached to a chain hanging from a pulley, and subjected to hind limb unloading by tail suspension for 28 days. After sacrifice, serum was collected, and the bilateral femurs and tibiae were dissected and processed for dual-energy X-ray absorptiometry, three-point bending mechanical tests, microcomputed tomography, and evaluation of serum bone metabolic markers. All experimental procedures were approved by the Committees of Animal Ethics and Experimental Safety of the China Astronaut Research and Training Center.
4.2. Bone Mineral Density Assessment
The bone mineral density of femurs was measured by dual-energy X-ray absorptiometry equipped with appropriate software for small laboratory animals (GE Lunar PIXImus, GE Healthcare, Madison, WI, USA). All right femurs were placed in the same direction. Values were expressed as the observed mean (g/cm2) ± standard deviation (SD) of the whole group.
4.3. Biomechanical Testing
Biomechanical analysis was performed by three-point bending mechanical tests. Experiments were conducted using TexturePro CT V1.3 Build 14 (Brookfield Engineering Labs, Inc., Stoughton, MA, USA). Femora were placed horizontally on the frame on rounded edges at a distance of 20 mm. To minimize variability, the specimens were placed in a consistent position and orientation. Maximum force applied to failure, deformation at hardness and hardness work cycle values were recorded.
4.4. Microcomputed Tomography Analysis of the Distal Femur
The secondary spongiosa extracted from the left distal femurs of rats was scanned with a desktop, microcomputed tomography scanner (μCT40; Scanco Medical, Bruttisellen, Switzerland) using a voxel size of 10 μm, an X-ray tube voltage of 70 kVp, a current intensity of 114 μA, and an integration time of 600 ms. Briefly, slices were scanned at the region of the distal femur beginning at 0.1 mm from the most proximal aspect of the growth plate and extending proximally along the femur diaphysis. A volume of interest was manually drawn on each specimen. Microstructural measures included trabecular bone mineral density, bone volume per total volume (BV/TV), trabecular thickness (Tb.Th), trabecular number (Tb.N), and trabecular separation (Tb.Sp). Computation of these structural measures was performed using a previously described method [12].
4.5. Biochemical Serum Analysis
Blood was collected and serum were separated by centrifugation to determine alkaline phosphatase activity (ALP) using an autoanalyzer. Serum bone osteocalcin/bone GLA protein (BGP) content was measured with a carboxyglutamic acid radioimmunometric assay kit (BGP Radioimmunometric Assay, Beijing North Institute of Biological Technology, Beijing, China), according to the manufacturer’s protocol. Serum N-terminal propeptide of type I procollagen (PINP) was measured with a specific rat PINP enzyme immunoassay (Rat PINP EIA; IDS Ltd., UK). The Beta isomer of serum C-telopeptide of type I collagen (CTX) was measured by an ELISA specific for rat CTX (RatLaps ELISA; IDS. Serum tartrate-resistant acid phosphatase form 5b (TRACP-5b) was measured by an ELISA specific for rat TRACP-5b (RatTRAP Assay; IDS).
4.6. Statistical Analysis
All numerical data are expressed as means ± SD. Statistical analyses were performed using SPSS for Windows version 17.0 (IBM, Armonk, NY, USA). With sample size of 8 (n = 5 or 6), nonparametric statistical analysis were performed. After one-way ANOVA, least-significant differences or Dunnett’s post-hoc test was used to determine significant differences between groups. Values of p < 0.05 were considered to indicate statistical significance.
5. Conclusions
Bovine CP, alone or in combination with CC, did not inhibit bone loss in a tail-suspended rat model of microgravity. However, CP or CP–CC treatment partially ameliorated the microgravity-induced deterioration of bone microarchitecture. This may represent an adjunct dietary strategy to reduce the risk of fracture in astronauts and highly sedentary populations.
Author Contributions
Conceptualization, J.L. and Y.G.; investigation, J.W.; and writing—original draft preparation, J.L. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest. The founding sponsors had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, and in the decision to publish the results.
Footnotes
Sample Availability: Samples of the compounds bovine bone collagen peptides are available from the authors.
References
- 1.Lloyd S.A., Morony S.E., Ferguson V.L., Simske S.J., Stodieck L.S., Warmington K.S., Livingston E.W., Lacey D.L., Kostenuik P.J., Bateman T.A. Osteoprotegerin is an effective countermeasure for spaceflight-induced bone loss in mice. Bone. 2015;81:562–572. doi: 10.1016/j.bone.2015.08.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Licata A.A. Challenges of estimating fracture risk with DXA: Changing concepts about bone strength and bone Density. Aerosp. Med. Hum. Perform. 2015;86:628–632. doi: 10.3357/AMHP.4208.2015. [DOI] [PubMed] [Google Scholar]
- 3.Lang T., LeBlanc A., Evans H., Lu Y., Genant H., Yu A. Cortical and trabecular bone mineral loss from the spine and hip in long-duration spaceflight. J. Bone Miner Res. 2004;19:1006–1012. doi: 10.1359/JBMR.040307. [DOI] [PubMed] [Google Scholar]
- 4.Cavanagh P.R., Licata A.A., Rice A.J. Exercise and pharmacological countermeasures for bone loss during long-duration space flight. Gravit Space Biol. Bull. 2005;18:39–58. [PubMed] [Google Scholar]
- 5.Peng Y.L., Hu H.Y., Luo J.C., Hou M.C., Lin H.C., Lee F.Y. Alendronate, a bisphosphonate, increased upper and lower gastrointestinal bleeding: Risk factor analysis from a nationwide population-based study. Osteoporos. Int. 2014;25:1617–1623. doi: 10.1007/s00198-014-2647-z. [DOI] [PubMed] [Google Scholar]
- 6.Park S.H., Song T.J., Bae T.S., Khang G., Choi B.H., Park S.R., Min B.H. Comparative analysis of collagens extracted from different animal sources for application of cartilage tissue engineering. Int. J. Precis. Eng. Man. 2012;13:2059–2066. doi: 10.1007/s12541-012-0271-4. [DOI] [Google Scholar]
- 7.Tziveleka L.A., Loannou E., Tsiourvas D., Berillis P., Foufa E., Roussis V. Collagen from the marine sponges Axinella cannabina and Suberites carnosus: Isolation and morphological, biochemical, and biophysical characterization. Mar. Drugs. 2017;15:152. doi: 10.3390/md15060152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.De Almeida-Jackix E., Cúneo F., Amaya-Farfan J., de Assunção J.V., Quintaes K.D. A food supplement of hydrolyzed collagen improves compositional and biodynamic characteristics of vertebrae in ovariectomized rats. J. Med. Food. 2010;13:1385–1390. doi: 10.1089/jmf.2009.0256. [DOI] [PubMed] [Google Scholar]
- 9.Nomura Y., Oohashi K., Watanabe M., Kasugai S. Increase in bone mineral density through oral administration of shark gelatin to ovariectomized rats. Nutrition. 2005;21:1120–1126. doi: 10.1016/j.nut.2005.03.007. [DOI] [PubMed] [Google Scholar]
- 10.Kim H.K., Kim M.G., Leem K.H. Osteogenic activity of collagen peptide via ERK/MAPK pathway mediated boosting of collagen synthesis and its therapeutic efficacy in osteoporotic bone by back-scattered electron imaging and microarchitecture analysis. Molecules. 2013;18:15474–15489. doi: 10.3390/molecules181215474. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Hooshmand S., Elam M.L., Browne J. Evidence for bone reversal properties of a calcium-collagen chelate, a novel dietary supplement. J. Food Nutr. Disor. 2013;2:1–6. doi: 10.4172/2324-9323.1000102. [DOI] [Google Scholar]
- 12.Liu J.L., Wang Y.H., Song S.J., Wang X.J., Qin Y.Y., Si S., Guo Y.C. Combined oral administration of bovine collagen peptides with calcium citrate inhibits bone loss in ovariectomized rats. PLoS ONE. 2015;10:e0135019. doi: 10.1371/journal.pone.0135019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Berillis P., Emfietzoglou D., Tzaphlidou M. Collagen fibril diameter in relation to bone site and to calcium/phosphours ratio. Sci. World J. 2006;6:1109–1113. doi: 10.1100/tsw.2006.212. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Adam M., Spacek P., Hulejová H., Galiánová A., Blahos J. Postmenopausal osteoporosis. Treatment with calcitonin and a diet rich in collagen proteins. Cas. Lek. Cesk. 1996;135:74–78. [PubMed] [Google Scholar]
- 15.Iwamoto J., Takeda T., Sato Y. Interventions to prevent bone loss in astronauts during space flight. Keio J. Med. 2005;54:55–59. doi: 10.2302/kjm.54.55. [DOI] [PubMed] [Google Scholar]
- 16.Blaber E.A., Dvorochkin N., Lee C., Alwood J.S., Yousuf R., Pianetta P., Globus R.K., Burns B.P., Almeida E.A. Microgravity induces pelvic bone loss through osteoclastic activity, osteocytic osteolysis, and osteoblastic cell cycle inhibition by CDKN1a/p21. PLoS ONE. 2013;8:e61372. doi: 10.1371/journal.pone.0061372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Narici M.V., de Boer M.D. Disuse of the musculo-skeletal system in space and on earth. Eur. J. Appl. Physiol. 2011;111:403–420. doi: 10.1007/s00421-010-1556-x. [DOI] [PubMed] [Google Scholar]
- 18.Anastasopoulou C., Rude R.K. Bone mineral density screening: Assessment of influence on prevention and treatment of osteoporosis. Endocr. Pract. 2002;8:199–201. doi: 10.4158/EP.8.3.199. [DOI] [PubMed] [Google Scholar]
- 19.Miskey J.R. Osteoporosis and bone function adaption: Mechanobiological regulation of bone architecture in growing and adult bone, a review. J. Rehabil. Res. Dev. 2000;37:189–199. [PubMed] [Google Scholar]
- 20.Cui W., Liu C.L. Basic bone biomechanics (I) Chin. J. Osteoporos. 1997;3:82–85. [Google Scholar]
- 21.Chu Y.M., Jiang B.Q., Zhang D.Y., Fu Z.G., Xu F. Effect of bone mass, microarchitecture and impact direction on biomechani CCl properties of bone in ovariectomy-induced osterporotic rats. Chin. J. Osteoporos. 2007;13:651–654. [Google Scholar]
- 22.Berg-Johansen B., Liebenberg E.C., Li A., Macias B.R., Hargens A.R., Lotz J.C. Spaceflight-induced bone loss alters failure mode and reduces bending strength in murine spinal segments. J. Orthop. Res. 2016;34:48–57. doi: 10.1002/jor.23029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Vico L., Collet P., Guignandon A., Lafage-Proust M.H., Thomas T., Rehaillia M., Alexandre C. Effects of long-term microgravity exposure on cancellous and cortical weight-bearing bones of cosmonauts. Lancet. 2000;355:1607–1611. doi: 10.1016/S0140-6736(00)02217-0. [DOI] [PubMed] [Google Scholar]
- 24.Liu J.L., Song S.J., Si S.Y., GAO J., Wang Y.H., Qin Y.Y., Chen X.N., Guo Y.C. Effect of collagen peptide and calcium citrate on bone loss in ovariectomized rats. Chin. J. Osteoporos. Bone Miner Res. 2015;8:334–339. [Google Scholar]
- 25.Caillot-Augusseau A., Lafage-Proust M.H., Soler C., Pernod J., Dubois F., Alexandre C. Bone formation and resorption biological markers in cosmonauts during and after a 180-day space flight (Euromir 95) Clin. Chem. 1998;44:578–585. [PubMed] [Google Scholar]
- 26.Nichols H.L., Zhang N., Wen X. Proteomics and genomics of microgravity. Physiol. Genom. 2006;26:163–171. doi: 10.1152/physiolgenomics.00323.2005. [DOI] [PubMed] [Google Scholar]
- 27.Hashemian S.J., Rismanchi M., Esfahani E.N., Khoshvaghti A., Razi F. Effect of calcitriol supplementation and tail suspension on serum biomarkers of bone formation in rats. J. Diabetes Metab. Disor. 2015;14:14. doi: 10.1186/s40200-015-0142-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Yang R., Huang Z., Cai D.H., Zhang H., Lai A.N., Zhang Z., Sun P., Chen H. Preventive and therapeutic effect of strontium ranelate on osteoporosis in rats subjected to simulated weightlessness. J. South. Med. Univ. 2010;30:834–837. [PubMed] [Google Scholar]
- 29.Smith S.M., Wastney M.E., O’Brien K.O., Morukov B.V., Larina I.M., Abrams S.A., Davis-Street J.E., Oganov V., Shackelford L.C. Bone markers, calcium metabolism, and calcium kinetics during extended-duration space flight on the mir space station. J. Bone. Miner Res. 2005;20:208–218. doi: 10.1359/JBMR.041105. [DOI] [PubMed] [Google Scholar]
- 30.Vasikaran S.D. Utility of biochemical markers of bone turnover and bone mineral density in management of osteoporosis. Crit. Rev. Clin. Lab. Sci. 2008;45:221–258. doi: 10.1080/10408360801949442. [DOI] [PubMed] [Google Scholar]
- 31.Yotsumoto N., Takeoka M., Yokoyama M. Tail-suspended mice lacking calponin H1 experience decreased bone loss. Tohoku. J. Exp. Med. 2010;221:221–227. doi: 10.1620/tjem.221.221. [DOI] [PubMed] [Google Scholar]
- 32.Momken I., Stevens L., Bergouignan A., Desplanches D., Rudwill F., Chery I., Zahariev A., Zahn S., Stein T.P., Sebedio J.L., et al. Resveratrol prevents the wasting disorders of mechanical unloading by acting as a physical exercise mimetic in the rat. FASEB J. 2011;25:3646–3660. doi: 10.1096/fj.10-177295. [DOI] [PubMed] [Google Scholar]
- 33.Zerath E., Holy X., Roberts S.G., Andre C., Renault S., Hott M., Marie P.J. Spaceflight inhibits bone formation independent of corticosteroid status in growing rats. J. Bone Miner Res. 2000;15:1310–1320. doi: 10.1359/jbmr.2000.15.7.1310. [DOI] [PubMed] [Google Scholar]
- 34.Lee J., Vasikaran S. Current recommendations for laboratory testing and use of bone turnover markers in management of osteoporosis. Ann. Lab. Med. 2012;32:105–112. doi: 10.3343/alm.2012.32.2.105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Seibel M.J. Biochemical markers of bone turnover: Part I: Biochemistry and variability. Clin. Biochem. Rev. 2005;26:97–122. [PMC free article] [PubMed] [Google Scholar]
- 36.Alatalo S.L., Halleen J.M., Hentunen T.A., Mönkkönen J., Väänänen H.K. Rapid screening method for osteoclast differentiation in vitro that measures tartrate-resistant acid phosphatase 5b activity secreted into the culture medium. Clin. Chem. 2000;46:1751–1754. doi: 10.1093/clinchem/46.11.1751. [DOI] [PubMed] [Google Scholar]
- 37.Alatalo S.L., Peng Z., Janckila A.J., Kaija H., Vihko P., Vaananen H.K., Halleen J.M. A novel immunoassay for the determination of tartrate-resistant acid phosphatase 5b from rat serum. J. Bone Miner Res. 2003;18:134–139. doi: 10.1359/jbmr.2003.18.1.134. [DOI] [PubMed] [Google Scholar]
- 38.Jing D., Cai J., Wu Y., Shen G., Li F., Xu Q., Xie K., Tang C., Liu J., Guo W., et al. Pulsed electromagnetic fields partially preserve bone mass, microarchitecture, and strength by promoting bone formation in hindlimb-suspended rats. J. Bone Miner Res. 2014;29:2250–2261. doi: 10.1002/jbmr.2260. [DOI] [PubMed] [Google Scholar]
- 39.Lin Y., Liu Z.L., Qian A., Shang P. Construction of a modified Mouse Tail-suspension model for space bone loss. Spa. Med. Med. Eng. 2012;25:239–242. [Google Scholar]