Abstract
Flavonoids are polyphenolic compounds subdivided into 6 groups: isoflavonoids, flavanones, flavanols, flavonols, flavones and anthocyanidins found in a variety of plants. Fruits, vegetables, plant-derived beverages such as green tea, wine and cocoa-based products are the main dietary sources of flavonoids. Flavonoids have been shown to possess a wide variety of anticancer effects: they modulate reactive oxygen species (ROS)-scavenging enzyme activities, participate in arresting the cell cycle, induce apoptosis, autophagy, and suppress cancer cell proliferation and invasiveness. Flavonoids have dual action regarding ROS homeostasis—they act as antioxidants under normal conditions and are potent pro-oxidants in cancer cells triggering the apoptotic pathways and downregulating pro-inflammatory signaling pathways. This article reviews the biochemical properties and bioavailability of flavonoids, their anticancer activity and its mechanisms of action.
Keywords: flavonoids, cancer, ROS, antioxidants, pro-oxidants, mitochondria
1. Introduction
Flavonoids are polyphenolic compounds synthesized in plants as bioactive secondary metabolites [1] responsible for their color, flavor and pharmacological activities [2]. The main flavonoid sources are fruits and vegetables [3], and they are also abundant in cocoa products (cocoa powder, chocolate) [4], black and green tea [3,5] and red wine [3,6]. Among the fruits, berries [7,8], plums, cherries [9,10] and apples [10,11] are the richest in flavonoids, whereas tropical fruits are poor in flavonoids [12]. Among the vegetables, the highest levels of flavonoids are found in broad beans [13], olives [14], onions [15], spinach [16] and shallot [17].
Flavonoids are potent antioxidants [11] protecting plants from unfavorable environmental conditions [1], therefore they have attracted attention and have been used in numerous epidemiological and experimental studies to assess their possible beneficial effects in multiple acute and chronic human disorders [18]. In vitro and in vivo studies have shown that flavonoids could exert anti-inflammatory, immunomodulatory [19] and strong anticancer activities [18,20,21].
2. Chemical Properties of Flavonoids
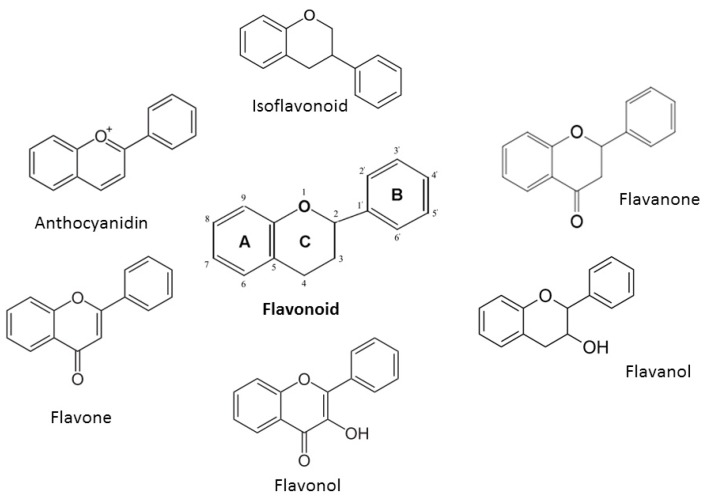
All flavonoids possess the basic flavan skeleton—a 15-carbon phenylpropanoid chain (C6-C3-C6 system), which forms two aromatic rings (A and B) linked by a heterocyclic pyran ring (C) (Figure 1). Based on their chemical structure, degree of oxidation, and linking chain unsaturation flavonoids could be further classified into 6 major groups: isoflavonoids, flavanones, flavanols, flavonols, flavones and anthocyanidins [20,22,23].
Figure 1.
Main chemical structures of flavonoids.
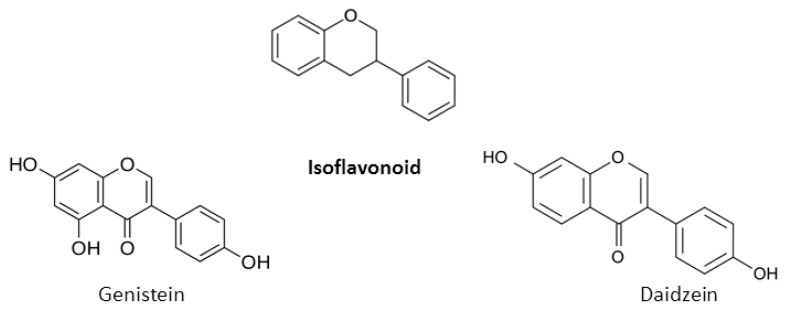
A chromane ring (A and C) is attached to a B ring (Figure 1) at C2 in flavonoids or C3 in isoflavonoids [22]. The main isoflavonoids are genistein and daidzein (Figure 2).
Figure 2.
Chemical structures of the main isoflavonoids.
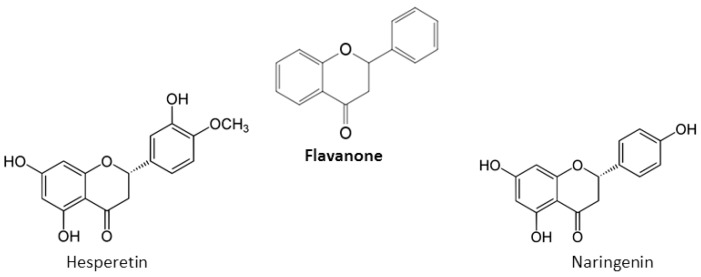
A saturated, oxidized C ring is present in flavanones, also described as di-hydroflavones [22]. Main flavanones are hesperetin and naringenin (Figure 3) [22].
Figure 3.
Chemical structures of main flavanones.
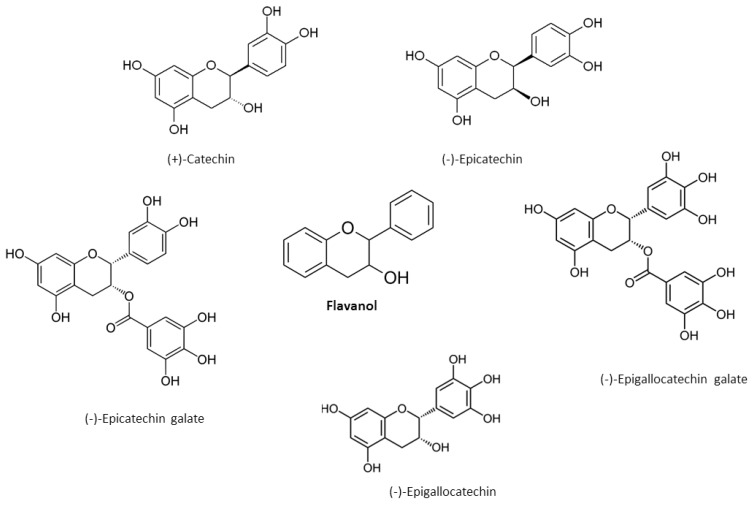
A saturated, unoxidized C ring with a hydroxyl group at C3 is common for flavanols, also known as green tea catechins. The most common catechin stereoisomers are cis ((-)-epicatechin) or trans ((+)-catechin according to C2 and C3 position in the molecule [5,24,25]. Flavanols can form gallic acid conjugates epicatechin gallate, epigallocatechin and epigallocatechin gallate during esterification with gallate groups (Figure 4) [5,24,25].
Figure 4.
Chemical structures of main flavanols.
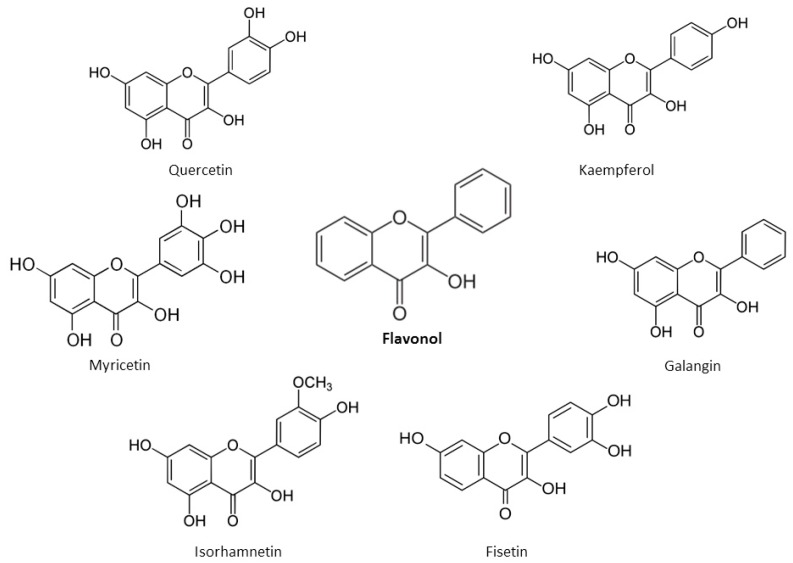
Flavonols possess an unsaturated C ring at the C2–C3 position, which is usually hydroxylated at C3 and oxidized at C4 [22]. The main flavonols are quercetin and kaempferol, followed by myricetin, isorhamnetin, fisetin and galangin found in lesser amounts (Figure 5). The –OH moieties in flavonols are responsible for their biological activities.
Figure 5.
Chemical structures of main flavonols.
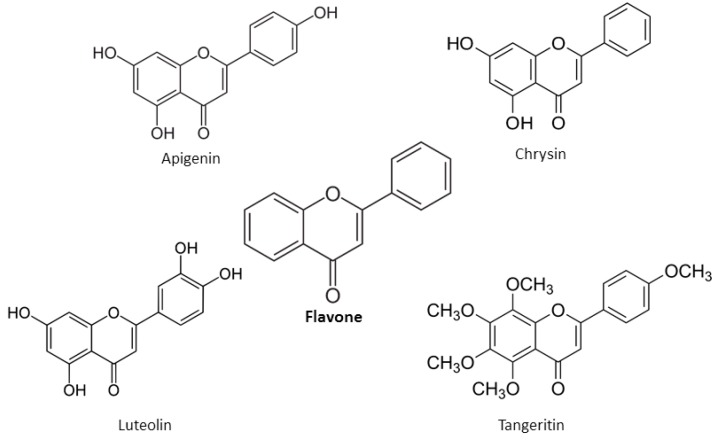
An unsaturated C ring at C2–C3, non-hydroxylated C3 and a ketonic group at C4 position are present in flavones [22]. The main flavones include apigenin, chrysin, luteolin, and tangeritin (Figure 6).
Figure 6.
Chemical structures of main flavones.
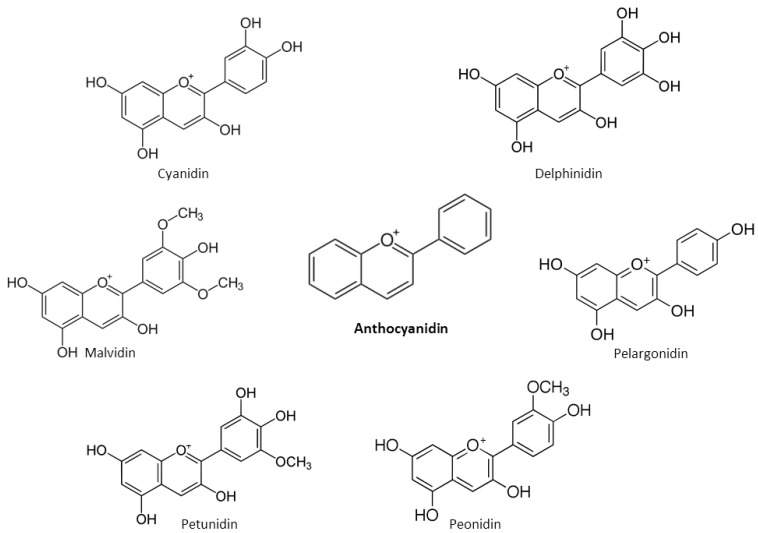
Anthocyanidins are water-soluble, unoxidized, unsaturated, flavonoids, mainly found as pH-dependent plant pigments. Anthocyanidins are based on the basic structure of the 2-phenyl-benzopyrylium chromophore–flavylium ion. They are hydroxylated at C3 position and at carbon atoms 3, 4 and 5 in the ring B of the molecule [20]. The main anthocyanidins include cyanidin, delphinidin, pelargonidin, peonidin, petunidin and malvidin (Figure 7) [20].
Figure 7.
Chemical structures of main anthocyanidins.
Flavonoids exist either as glycosides with linked sugars or as aglycones without linked sugars [18,20]. In the cytosol (pH 7.4), flavonoids form a mixture of phenolate anions and neutral phenols. Their proportion depends on the pKa of each phenolic group. Since flavonoids are weak hydrophobic acids, depending on their lipophilicity they have potential to cross cellular and mitochondrial membranes and act as protonophores [26,27,28].
3. Bioavailability of Flavonoids
Flavonoids can interact with other nutrients [29,30]: they can decrease glucose absorption due to suppression of carbohydrate-hydrolyzing enzymes (alpha-amylase and alpha-glucosidase) [31] and glucose transporter in the brush border [31]. Fat intake improves flavonoid bioavailability and increases their intestinal absorption via augmented secretion of bile salts which increase micellar incorporation of flavonoids [31]. However, protein intake can decrease flavonoid bioavailability [32,33], affecting both antioxidant efficacy and protein digestibility [32]. The gut microbiome is very important for the absorption and metabolism of flavonoids. After consumption, prior to absorption intestinal or colon microflora are able to hydrolyze glycosylated flavonoids such as flavones, isoflavones, flavonols and anthocyanins into their respective aglycones [33,34]. Aglycones are lipophilic, and therefore passive diffusion is responsible for their pathway to the intestinal epithelial cells while the uptake of glycosides into the intestinal epithelial cells is regulated by the epithelial transporters [34]. After absorption, flavonoids undergo metabolic transformations first in the small intestine, liver and kidney [34]. Methylation, sulfation, or glucuronidation of flavonoids before they reach the circulation and, afterwards, the tissues, could influence their biological activities. Unabsorbed flavonoids remaining in the proximal intestine are further digested in the colon by microbes able to split their heterocyclic oxygen containing ring and the hydroxylated phenyl carboxylic acids formed could be absorbed [34]. The highest concentration of plasma flavonoids in humans usually is reached 1 to 2 h after the intake of flavonoid-rich foods [35]. This depends on the type of flavonoid; for example, catechins and anthocyanins are characterized by a half-life elimination that is 5 to 10 times less compared to flavonols [33]. The concentration of plasma quercetin metabolites are found from 0.7 to 7.6 µM since quercetin is the most abundant dietary flavonoid [35]. Anthocyanins and pro-anthocyanidins have the lowest bioavailability, while quercetin glucosides, catechin, flavanones, isoflavones and gallic acid have the highest bioavailability [18].
4. Anticancer Effects of Flavonoids
The ability of flavonoids to scavenge free radicals, regulate cellular metabolism, and prevent oxidative stress–related diseases have been demonstrated in numerous studies [18,19,20,21,36,37]. There is accumulating evidence that many flavonoids exert anticancer activity, however, the molecular mechanisms responsible for this effect have not been fully elucidated yet.
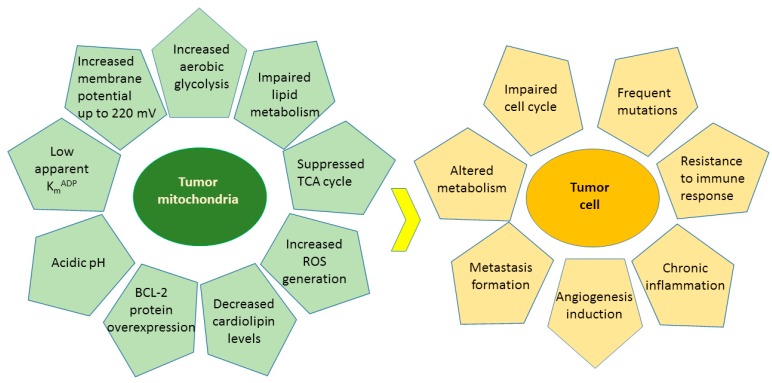
Cancer is a heterogeneous disease characterized by uncontrolled proliferation and impaired cell cycle leading to the growth of abnormal cells that invade and metastasize to other parts of the body [38,39]. Oxidative stress, hypoxia, genetic mutations and lack of apoptotic function are the main internal causes of cancer, whereas the external causes are related to increased exposure to stress, pollution, smoking, radiation and ultraviolet rays [40]. Altered metabolism, impaired cell cycles, frequent mutations, resistance to immune response, chronic inflammation, formation of metastasis, and induction of angiogenesis are the main characteristics of the cancer cells [38] (Figure 8). There is emerging evidence that cancer is a metabolic disease determined by various degrees of mitochondrial dysfunctions and metabolic alterations [38,39,41]. Mitochondria play essential roles in cellular energy supply, regulation of metabolism, cell death signaling and reactive oxygen species (ROS) generation. The main metabolic alterations of the tumor cells involve increased aerobic glycolysis [42], deregulated pH [43], impaired lipid metabolism [44], increased generation of ROS [45], and compromised enzyme activities [38,46] (Figure 8). As a direct consequence, the extracellular environment becomes acidic and more favorable to inflammation [47], glutamine-driven lipid biosynthesis increases and upregulates the pathways involved in tumorigenesis initiation and metastasis [48], cardiolipin levels decrease in membranes causing impaired enzyme activities [49,50,51], mitochondria are hyperpolarised [38], and this effect correlates with the malignancy and invasiveness of cancer cells [38].
Figure 8.
The main characteristics of tumor mitochondria and tumor cells.
Flavonoids exert a wide variety of anticancer effects: they modulate ROS-scavenging enzyme activities, participate in arresting the cell cycle, induce apoptosis, autophagy, and suppress cancer cell proliferation and invasiveness [18,19,20,21,36,37].
4.1. Flavonoids in Oxidative Stress
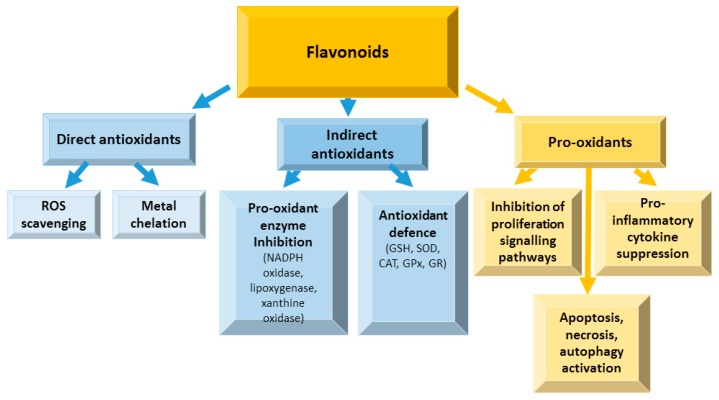
When the cellular homeostasis between the pro-oxidant activities and antioxidant defense is impaired, the production of ROS increases, and free radicals accumulate [18]. ROS are mainly generated in the electron transport chain in mitochondria as the byproducts of oxidative phosphorylation in the cell [52]. The amount of ROS produced causes oxidative stress which is involved in the development of inflammation processes leading to many degenerative diseases and cancer. Flavonoids have dual action regarding ROS homeostasis—they act as antioxidants under normal conditions and are potent pro-oxidants in cancer cells triggering the apoptotic pathways [53,54] (Figure 9).
Figure 9.
Antioxidant and pro-oxidant activities of flavonoids in oxidative stress. ROS—reactive oxygen species, NADPH-oxidase—nicotinamide adenine dinucleotide phosphate oxidase, GSH—glutathione, SOD—superoxide dismutase, CAT—catalase, GPx—glutathione peroxidase, GR—glutathione reductase.
Flavonoids can directly scavenge ROS, and chelate metal ions [55] due to their ability stabilize the free radicals due to the presence of phenolic hydroxyl groups [56]. Indirect flavonoid antioxidant effects are related to activation of antioxidant enzymes, suppression of pro-oxidant enzymes, and stimulating production of antioxidant enzymes and phase II detoxification enzymes [55]. Both antioxidant and pro-oxidant activities are involved in flavonoid anticancer effects [57,58].
Isoflavone genistein promoted breast cancer cell arrest at G2/M phase and subsequent ROS dependent apoptosis [59]. Daidzein promoted apoptosis in breast cancer MCF-7 cells due to the ROS generation [60]. Flavanone hesperetin induced apoptosis of gall bladder carcinoma [61], esophageal cancer [62], hepatocellular carcinoma [63] and human breast carcinoma MCF-7 cells [64] via activating the mitochondrial apoptotic pathway by increasing the ROS production. Flavanone naringenin exerted anti-cancer effects on choriocarcinoma JAR and JEG 3 cell lines by inducing the generation of ROS and activation of signaling pathways [65]. It also initiated an apoptotic cascade in human epidermoid carcinoma A431 cells [66]. In prostate cancer PC3 and LNCaP cell lines, naringenin suppressed proliferation and migration and induced apoptosis and ROS generation [67]. Furthermore, naringenin reduced ROS generation and enhanced the activity of superoxide dismutase, catalase, glutathione in chronic diseases and cancer [68]. Cocoa catechins and procyanidins have been shown to induce apoptotic morphological changes, DNA damage and apoptosis in epithelial ovarian cancer cells due to their prooxidant properties [69]. Cocoa polyphenolic extract activated the ERK1/2 pathway, thus increasing the activities of glutathione peroxidase and reductase in HepG2 cells [70]. Cocoa catechins and procyanidins also protected Caco2 cells against an induced oxidative stress and subsequent cellular death by reducing ROS production [71]. Due to antioxidant properties, cocoa flavanols exerted beneficial effects in the protection from colon cancer [72,73]. Flavonol quercetin exerted potent cancer chemopreventive properties [74,75]. Recent studies showed that quercetin reduced the proliferation of hepatocellular carcinoma HepG2 cells decreasing the intracellular ROS level [76]. It increased ROS production and the apoptotic cell number in human gastric cancer AGS [77] and human breast cancer MCF-7 cells [78]. Flavonol kaempferol inhibited the growth of cancerous bladder cells due to ROS level modulation-induced apoptosis and S phase arrest [79]. It activated caspases due to ROS generation and stimulated apoptosis in colorectal cancer HCT116, HCT15, and SW480 cell lines [80]. Furthermore, kaempferol exerted cytotoxic effects on rat hepatocellular carcinoma cells via ROS-mediated mitochondrial targeting [81]. The anticancer activities of flavones apigenin and luteolin in ovarian cancer cell lines (A2780, OVCAR-3 and SKOV-3) were also related to the changes in ROS signaling, as well as to the promotion of apoptosis [82,83]. Moreover, apigenin activated apoptosis also in human cervical cancer-derived cell lines including HeLa (human papillomavirus/HPV 18-positive), SiHa (HPV 16-positive), CaSki (HPV 16 and HPV 18-positive), and C33A (HPV-negative) cells due to increased ROS generation and launched mitochondrial apoptotic pathways [84]. Flavone chrysin was reported to augment ROS and lipid peroxidation levels, leading to the death of choriocarcinoma (JAR and JEG3) [85], bladder cancer [86] and ovarian cancer (ES2 and OV90) cells [87]. The antioxidant activity of flavonoids was also investigated in humans. It was found that serum total antioxidant capacity correlates with anthocyanin consumption in the diet [88]. Furthermore, cyanidin induced cell death via ROS modulation in the DU145 and LnCap human prostatic cancer cells [89]. Cyanidin and delphinidin accelerated cellular ROS accumulation, suppressed glutathione reductase, and depleted glutathione resulting in cytotoxicity in metastatic (LoVo and LoVo/ADR) colorectal cancer cells [90].
Thus, numerous studies show beneficial effects of flavonoids as potent antioxidants under normal and pro-oxidants under pathological conditions, capable of activating apoptosis and suppressing proliferation and inflammation.
4.2. Flavonoids in Apoptosis
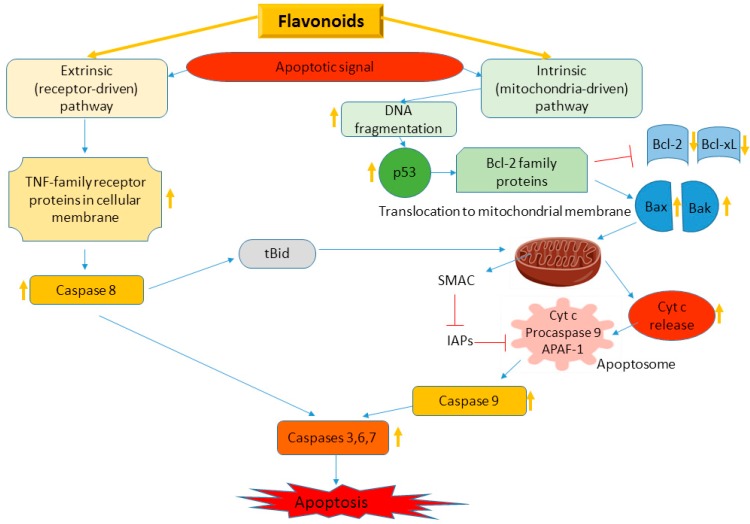
Cancer cells are resistant to apoptosis—a programmed cell death, usually induced by a series of signal transduction pathways and pro-apoptotic proteins—caspases and Bcl-2 family proteins [20,91]. There are two main signaling cascades of apoptosis—extrinsic, related to tumor necrosis factor (TNF) superfamily with main signaling protein—caspase 8; and intrinsic—mitochondrial pathway, where Bcl-2 family proteins launch the activation of caspases 9, 3 and 7 (Figure 10) [20,91]. There is an overexpression of oncogenic genes (e.g., c-Myc), leading to cellular proliferation and p53 suppression, and activated anti-apoptotic proteins of Bcl-2 family in cancer cells [92], whereas pro-apoptotic proteins and caspases could be downregulated [91,92]. Flavonoids could target apoptotic signaling cascade stimulating the cell death pathways [20,21] (Figure 10).
Figure 10.
Flavonoid targets in extrinsic and intrinsic apoptosis pathways. TNF—tumor necrosis factor, tBid—truncated Bid, Bcl-2—B-cell lymphoma protein 2, Bcl-xL—Bcl-2 homologue splice variants, Cyt c—cytochrome c, SMAC—second mitochondrial activator of caspases, IAPs—inhibitor of apoptosis proteins, APAF-1—apoptotic protease activating factor 1. Yellow arrows show the effect of flavonoids (activation or suppression).
Flavonoids acting as pro-oxidants could suppress proliferation of cancer cells by inhibition of epidermal growth factor receptor/mitogen activated protein kinase (EGFR/MAPK), phosphatidylinositide 3-kinases (PI3K), protein kinase B (Akt) as well as nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) [18,20,38].
Isoflavonoid genistein could regulate estrogen receptor-α expression and change Bax/Bcl-2 ratio downregulating proliferation, differentiation, and activating apoptosis in MCF-7 and 3T3-L1 cells [93]. Moreover, genistein suppressed Bcl-2, Bcl-xL, c-inhibitor of apoptosis protein 1 (c-IAP1), survivin, and NF-κB in C200 and A2780 cells [94], increased caspase-3 activity in HT-29 colon cancer cells [95] and activated intrinsic apoptotic signaling pathway in HCT-116 and LoVo cells [96]. Isoflavonoid daidzein also acted as phytoestrogen [97]. It promoted cytochrome c release from mitochondria, leading to caspase 7 and 9 activation and also altered Bax/Bcl-2 ratio in MCF-7 cells [60,98]. Daidzein induced apoptosis in the HCCSK-HEP-1 cell line via Bak upregulation and downregulation of anti-apoptotic proteins, resulting in cytochrome c release from mitochondria and activating subsequent apoptotic pathway involving caspases 3 and 9 [99]. Flavanone hesperetin induced cytochrome c release, activation of caspases-3 and -9, and reduced Bax to Bcl-2 ratio in gastric cancer cells [100], in the Eca109 cell line [62] as well as in the HT-29, MCF-7, and MDA-MB-231 cell lines [64,101]. In H522 cells, hesperetin induced extrinsic apoptotic pathway due to overexpression of TNF-protein superfamily members, caspase-9 activation, and decrease in p53 level [102]. Furthermore, hesperetin inhibited the NF-κB signaling pathway and reduced Bcl-2 transcription and translation in PC-3 cells [103]. Flavanone naringenin could induce apoptosis via increased p53 expression, Bax and caspase-3 cleaving, and downregulated Bcl-2 and survivin in SGC-7901 cell line [104,105]. Naringenin-induced extrinsic apoptotic pathway was related to overexpression of TNF-family proteins [20]. Flavanols catechins, especially epigallocatechin galate, induced apoptosis and cell-cycle arrest, inhibited NF-κB, leading to cyclooxygenase-2 (COX) overexpression [106]. Moreover, it increased Bax/Bcl-2 ratio, upregulated p53, p21, caspases-3, and -9, and down-regulated PI3K, Akt, and Bcl-2 in T47D and HFF cells [107]. Catechins could also alter the expression of anti- and pro-apoptotic genes [108,109,110]. Cocoa flavanols have been shown to moderate apoptosis pathways in HepG2 [111,112] and Caco-2 cells [72]. Flavonol quercetin, a widely abundant phytoestrogen [20], was able to induce intrinsic apoptotic pathway via Bax and caspase-3 upregulation and downregulation of Bcl-2 in MCF-7 cells [113,114,115]. Quercetin activated apoptosis in PC-3 and LNCaP cells regulating the p53 signaling pathway [116]. In HL-60 cells, quercetin activated intrinsic apoptotic cascade-modulating COX-2, activating caspase-3, modulating Bax, Bad, Bcl-2 expression and inducing cytochrome c release from mitochondria [117]. In a human hepatoma cell line, quercetin induced apoptosis via caspase activation, regulation of Bcl-2, and inhibition of PI-3-kinase/Akt and extracellular-signal-regulated kinase (ERK) pathways [118]. Quercetin was also able to suppress cancer cell proliferation due to inhibition of PI3K/Akt pathway [119]. Flavonol kaempferol, a phytoestrogen [120], induced intrinsic apoptosis in A2780/CP70, A2780wt and OVCAR-3 cell lines. Its main effects were related to the activation of caspases 3 and 7, the upregulation of p53, Bax and Bad and the downregulation of Bcl-xL protein [121]. In HeLa cells, kaempferol activated apoptosis elevating the Bax/Bcl-2 ratio [122]. Flavone apigenin also was reported to have estrogenic activity [123]. In PC-3 and DU145 cell lines apigenin induced Bax overexpression, the downregulation of Bcl-2 and Bcl-xL proteins, and stimulated cytochrome c release from mitochondria and subsequent activation of signaling cascades [124,125]. Apigenin upregulated p53 in ACHN, Caki-1 RCC cell lines [126]. In T24 cell line, apigenin inactivated PI3K/Akt signaling pathway, activated the intrinsic apoptotic pathway, promoted the cytochrome c release from mitochondria, inhibited Bcl-xL [127,128]. In HCT-116 cells, apigenin activated both extrinsic and intrinsic apoptotic pathways [129]. Flavone chrysin activated apoptosis in HeLa cells due to increased DNA fragmentation and stimulated p38 and NF-κB pathways [20]. Chrysin upregulated caspase 3 in the U937 cell line [130]. In SP6.5 and M17 melanoma cells, chrysin induced the intrinsic apoptotic pathway due to cytochrome c release-driven activation of caspases 3 and 9 [131]. Anthocyanidin pelargonidin stimulated the cytochrome c release from mitochondria, activated Bax, Bid, caspases 3 and 9, and inhibited the expression of Bcl-2 and Bcl-xL in HT-29 cells [132,133]. Furthermore, pelargonidin downregulated the PI3K/Akt signaling pathway thus suppressing proliferation of U2OS cell line [132]. Cyanidin could activate cytochrome c and upregulate Bax protein expression [20]. In U87 cells, cyanidin promoted Bax and p53 expression and downregulated Bcl-2 expression [134]. Delphinidin decreased proliferation of the SKOV3 cell line due to the suppression of PI3K/Akt and ERK1/2/MAPK signaling pathways [135]. Delphinidin was also able to activate caspases 3 and 9 in the NSCLC cell line [136].
Thus, flavonoids could activate cell death signaling pathways in cancer cells by a dual mechanism—activating anti-apoptotic proteins and suppressing pro-apoptotic proteins and caspases.
4.3. Immunomodulatory and Anti-Inflammatory Effects of Flavonoids
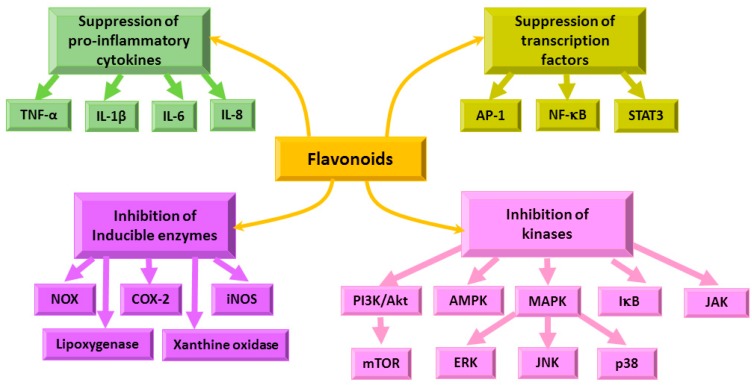
Chronic inflammation leads to tumor development, modulating cellular transformation, survival, proliferation, invasion, metastasis, and angiogenesis pathways [137]. Flavonoids were shown to exert anti-inflammatory action via immune cell regulation, suppression of chemokines, COX-2, cytokines and pro-inflammatory transcription factors, inhibition of PI3K/Akt, inhibitor of kappa kinase/c-Jun amino-terminal kinases (IKK/JNK) [19,21,137]. The NF-kB signaling pathway is crucial in the regulation of inflammation [19,21,137] and is related to the modulation of a wide variety of oncogenes (Figure 11) [137].
Figure 11.
Flavonoid targets during inflammation processes. TNF—tumor necrosis factor, IL—interleukin, AP-1—activator protein 1, NF-κB—nuclear factor kappa-light- chain-enhancer of activated B cells, STAT3—signal transducer and activator 3, NOX—NADPH oxidase, COX-2—cyclooxygenase-2, iNOS—inducible nitric oxide synthase, AMPK—AMP—activated protein kinase, PI3K—phosphatidylinositide 3-kinases, Akt—protein kinase B, mTOR—mammalian target of rapamycin, MAPK—mitogen activated protein kinase, ERK—extracellular-signal-regulated kinase, JNK—c-Jun N-terminal kinase, p38—p38 kinse, IκB—IκB kinase, JAK—Janus kinase.
The immune system is a key player in protecting an organism from infections and cancer. B and T lymphocytes and macrophages are the major cells responsible for the immunity. B cells secret antibodies which are able to attach to pathogens, marking them so they are recognized and destroyed by phagocytes [138,139]. T cytotoxic cells are able to kill tumor cells directly, and T helper cells secrete cytokines and mediators which regulate the activities of B lymphocytes and macrophages [138,139]. Flavonoids have been shown to modulate directly the differentiation and count of the cells belonging to the immune system [138,139]. Furthermore, flavonoids can inhibit the activity of the mammalian target of rapamycin (mTOR) and thus reduce T effector differentiation and induce T regulatory cells [140]. Programmed cell death protein 1 (PD-1) is present on the surface of B cells, T cells and macrophages [141]. When programmed death-ligand 1 (PD-L1)—a protein present on the surface of tumor cells—binds the PD-1, the signal is sent to suppress the immune system response; therefore, the inhibitors of PD-L1/PD-1 signaling pathway could be promising agents in cancer immunotherapy [141]. The studies on flavonoids as possible suppressors of PD-L1/PD-1 immune checkpoint have not been very intense yet, nevertheless the first encouraging results were obtained demonstrating the inhibition of PD-L1 expression by flavone apigenin in A375 melanoma cells [142] and PD-1/PD-L1 inhibition in vitro by flavonols quercetin [143] and fisetin [143] as well as isoflavonoid glyasperin C [144].
Isoflavone genistein has been shown to modulate the expression of several genes involved in cell cycle regulation, migration, inflammation, and the PI3K and MAPK pathways in HeLa cells [145]. Genistein exerted influence on the expression of inflammatory-related genes in breast cancer MCF-7 (high ERα/ERβ ratio), T47D (low ERα/ERβ ratio), and MDA-MB-231 (ERα-negative) cell lines [146]. Furthermore, genistein inhibited the increased M2 polarization of macrophages and stemness of ovarian cancer SKOV3 and OVCA-3R cell lines by the co-culture of macrophages with ovarian cancer stem-like cells through disrupting the interleukin (IL)-8/STAT3 signaling axis [147]. Isoflavone daidzein downregulated the pro-inflammatory NF-kB and JNK signaling pathways in adipocyte and macrophage co-cultures [148]. Flavanone hesperetin suppressed secretion of TNF-α, IL-6, and IL-1β; decreased inducible nitric oxide synthase (iNOS) and COX-2 gene expression; down-regulated NF-κB (p65) phosphorylation in lipopolysaccharide -induced RAW 264.7 cells [149]. Hesperetin inhibited cell proliferation markers, angiogenic growth factors, COX-2 mRNA expression in 1,2-dimethylhydrazine-induced colon cancer [150].
Quercetin and naringenin prevented the lowered mRNA expression of liver IL-4, p53 and Bcl-2 in a diethylnitrosamine/2-acetylaminofluorene-induced hepatocarcinogenesis model in rats [151]. Naringenin inhibited the migration of breast cancer MDA-MR-231 cell line via modulation of inflammatory and apoptotic signaling pathways [152]. It also suppressed the migration and invasion of glioblastoma cells due to inhibition of ERK and p38 activities [153]. Catechins, especially epigallocatechin galate, inhibited NF-κB pathway and suppressed COX-2 overexpression [106]. Epicatechin induced NF-κB, AP-1 and Nrf2 via PI3K/AKT and ERK signalling in HepG2 cells [112]. Cocoa polyphenols prevented inflammation in the colon of azoxymethane-treated rats and in TNF-α-stimulated Caco-2 cells [154]. Flavonol quercetin inhibited the expression of matrix metallopeptidases MMP9 and MMP2 in human glioblastoma U251 cell line [155]. In ascite cells of Dalton’s lymphoma-bearing mice, quercetin downregulated the phosphorylation of Akt and PDK1 resulting in suppressed phosphorylation of downstream survival factors such as Bad, glycogen synthase kinase-3 (GSK-3β), mTOR, and nuclear factor of kappa light polypeptide gene enhancer in B-cells inhibitor alpha (IkBα) [156]. Furthermore, quercetin attenuated the levels of angiogenic factor vascular endothelial growth factor A (VEGF-A) and inflammatory enzymes COX-2 and iNOS [156]. Quercetin inhibited the migration and invasion of the human colon cancer Caco-2 cell line via regulation of the toll-like receptor 4 (TLR4)/NF-kB pathway [157]. Quercetin has been shown to be a potent inhibitor of mTOR activity and the PI3K/Akt signaling pathway in cancer cells [158]. Flavonol kaempferol downregulated TNF-alpha induced IL-8 promoter activation and gene expression in HEK 293 cells [159]. Furthermore, kaempferol reduced the plasma levels of the cytokines IL-6, IL-1β and TNF-α and suppressed the MAPK and NF-κB signaling pathways [160,161,162]. Flavone apigenin downregulated TNF-α-related inflammatory signaling in the A375 human melanoma cell line [163]. Apigenin decreased myeloperoxidase (MPO), inflammatory cytokine and COX-2 levels and downregulated NF-κB and STAT3, thereby inhibiting inflammation and inflammation-induced carcinogenesis in an inflammatory bowel disease and colitis-associated cancer model [164]. Apigenin could suppress Akt, ERK, MAPK, COX-2, IL-6, TNF-α, IL-1, iNOS activities in vitro and in vivo [163,165]. Flavone chrysin inhibited iNOS and COX-2 expression, and decreased the levels of proinflammatory cytokines IL-6, TNF-α, and prostaglandin E(2) (PGE(2)) in a renal cancer model in rats [166]. Cyanidin has been shown to inhibit pro-inflammatory cytokine interleukin-17A (IL-17A) [167]. Pelargonidin suppressed the production of TNF-α or IL-6 and the activation of NF-κB or ERK½ in vitro [168]. Cocoplum anthocyanins inhibited the production of TNF-α, IL-6 and the activation of NF-κB or ERK ½ in HT-29 colorectal adenocarcinoma cells [169]. Delphinidin suppressed the activation of NF-κB through MAPK signaling pathways in MCF-7 human breast carcinoma cells [170].
Chronic inflammation often precedes tumor development, therefore anti-inflammatory effects of flavonoids could be very important in decreasing the inflammation and enhancing the antitumor activity of immune cells.
4.4. Effects of Flavonoids on Mitochondrial Functions
Tumor-cell metabolism is altered compared to normal cells due to highly abnormal mitochondrial functions (Figure 8) [171]. Therefore, recent interest in natural compounds reverting the mitochondria to normal mode has emerged, and flavonoids have also been tested among potential drug candidates [20,36,37].
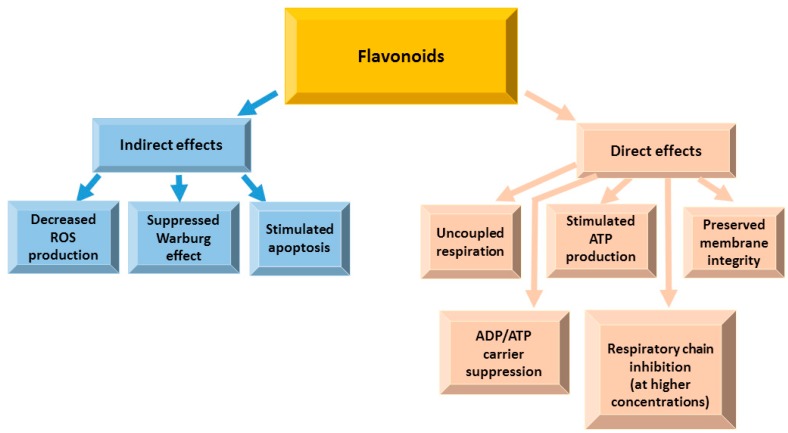
Hexokinase and voltage-dependent anion channel (VDAC) coupling in mitochondria prevents induction of apoptosis in tumors [14]. In human breast carcinoma (MDA-MB-231 and MCF-7) cells, an O-methylated flavone oroxylin A was reported to promote the detachment of hexokinase from mitochondria, resulting in inhibition of glycolysis [17]. Overexpression of antiapoptotic proteins of the BCL-2 family in mitochondria results in resistance to apoptotic pathways [37]. Flavanone hesperetin reduced antiapoptotic BCL-2 family protein transcription and translation in the human prostate cancer PC-3 cell line [103]. Naringenin and epigallocatechin-3-gallate decreased the BCL-2 expression accordingly in gastric cancer (SGC-7901) cells [104] and in cholangiocarcinoma (HuCC-T1) cells [172]. Mitochondrial adenine nucleotide translocase is a protein embedded in the mitochondrial inner membrane and responsible for ATP/ADP exchange [173]. It is one of the component of mitochondrial permeability transition pore complex, which is a key factor triggering apoptosis [174]. Quercetin (50 µM) was able to inhibit adenine nucleotide translocase by 46% in mitochondria isolated from rat kidney cortex [175], whereas apigenin (20 µM) inhibited it in human prostate cancer DU145 cells [176]. A procyanidin-rich French maritime pine (Pinus pinaster) bark extract inhibited the electron transport chain in isolated rat liver mitochondria and in submitochondrial particles, affecting complexes I, II and III [177]. An isoflavone genistein induced mitochondrial permeability transition in isolated rat liver mitochondria due to increased ROS generation at the complex III of the mitochondrial respiratory chain [178]. Epigallocatechin-3-gallate suppressed the growth of highly aggressive malignant pleural mesothelioma cells inhibiting complex I, II, and ATP synthase [179]. Moreover, epigallocatechin-3-gallate modulated mitochondrial bioenergetic functions and regulated apoptosis signaling cascade [180]. Anthocyanins were able to reduce cytosolic cytochrome c preventing apoptosis and support the electron transfer between NADH dehydrogenase and cytochrome c [181,182]. The inhibition of the tricarboxylic acid (TCA) cycle is one of the hallmarks of cancer [37]. Quercetin [183], kaempferol [184], hesperetin and naringenin [185] have been shown to stimulate the TCA cycle shifting anaerobic glycolysis to oxidative phosphorylation, normally suppressed in cancer cells. The effects of flavonoids on mitochondrial functions are summarized in Figure 12.
Figure 12.
Indirect and direct effects of flavonoids on mitochondrial functions.
In tumor cells, mitochondria are usually hyperpolarized, and their membrane potential reaches 220 mV [186,187] making them resistant to cell death signaling. Our group evaluated direct effects of selected flavonoids on the functions of cardiac mitochondria respiring on pyruvate and malate as substrates [188,189,190,191]. The results demonstrated that (-)-epicatechin [190,191], procyanidin B2 [190,191], hyperoside [189,190], quercetin [189,190], quercitrin [189,190] and rutin [189,190] uncoupled oxidation from phosphorylation. Furthermore, all flavonoids were reported to induce apoptosis (reviewed in [20]) and initially decrease mitochondrial membrane potential [20].
Most flavonoids have pKa values ranging between 6 and 9, i.e., close to the physiological pH of the cytosol and mitochondrial compartments, and favorable distribution coefficients [26,27,28], and therefore they have the ability to reach the mitochondrial matrix and release a proton in its relatively basic environment (pH 7.8). This effect might be crucial in the chemoprevention of cancer since the mild mitochondrial uncoupling effectively protects cells from oxidative stress.
4.5. Effects of Flavonoids on Gut Microbiota
The gastrointestinal tract, and especially the intestinal barrier, is very important in sustaining health [192,193]. Intestinal epithelium, besides nutrient absorption, provides a barrier controlling the entrance of microorganisms, their metabolic products and toxins as well as toxins present in ingested foods [194]. Due to anti-inflammatory action, flavonoids could protect the integrity of the intestinal barrier [192,195,196]. Flavanol epicatechin and flavonol quercetin suppressed systemic inflammation in rodent models of overfeeding (high fructose and high fat diets) [197,198,199]. Plant extracts rich in anthocyanins and pure anthocyanins could protect Caco-2 cell monolayers from permeabilization due to inflammation [200,201], whereas O-glucosides of delphinidin and cyanidin were more potent than the O-glucosides of petunidin, peonidin and malvidin [200]. In several metaanalysis, flavones, flavanols, flavonols, isoflavones, anthocyanidins and proanthocyanidins could reduce colorectal cancer risk [202,203,204,205]. Thus, some flavonoids were able to prevent and cure metabolic diseases directly at the gastrointestinal tract [206].
Flavonoids could suppress the activity of gut metabolizing enzymes—α-glucosidase, pancreatic lipoprotein lipase and amylase [192]. In vivo, proanthocyanidins inhibited triglyceride absorption in mice and in humans [207]. Oolong tea-derived epigallocatechin galate suppressed α-amylase [208]. In the gastrointestinal tract lumen, the decreased activities of α-glucosidase, pancreatic lipoprotein lipase and amylase would lead to a suppressed absorption of glucose from complex carbohydrates and fatty acids from triglycerides [192,209].
The microbiota present in the intestinal lumen is very important for the whole body. The link between pathological conditions, ingested food and the gut microbiota has not been established yet, although the primary investigations let hypothesize that it might be possible to prevent chronic diseases by modulating the intestinal microflora [210,211,212]. Most flavonoids (except flavanols) are naturally attached to sugars as β-glycosides, therefore they are not readily absorbed in the small intestine [213,214], and glycosylated flavonoids reach the colon [34] where the microbiota digest the flavonoids forming phenolic acids and other metabolites, which can later be absorbed [213,215]. Thus, flavonoids in the colon could influence the gut microbiome, whereas microbes could modulate flavonoid activity and bioavailability metabolizing them and these processes may be beneficial for health [192]. Flavonoids are known to exert antimicrobial activity, inhibiting specific microbes, such as pathogenic and commensal microorganisms [209]. Quercetin was shown to suppress the growth of Lactobacillus sp., Bacteroides galacturonicus and Ruminococcus gauvreauii [216]. The polyphenols present in cloudberry could decrease the growth of Candida albicans, Bacillus cereus, Helicobacter pylori, Campylobacter jejuni, Staphylococcus epidermidis, Staphylococcus aureus and Clostridium perfingens [217]. Furthermore, flavonoids could promote the growth of specific microbes in the gut [192]. Mice ingesting food rich in apple flavonoids demonstrated higher levels of bacteria belonging to a combined group of Bacteroides–Prevotella–Poryphyromonas and Bifidobacterium spp. but significantly decreased levels of Lactobacillus spp. [218]. Quercetin and rutin increased the growth of Bifidobacterium bifidum in vitro [219]. These studies show that flavonoids can affect microbial populations by changing endotoxin production, converting primary into secondary bile acids [220], sustaining immune homeostasis [221] and participating in bioactive and nutrient absorption and metabolism, thereby regulating short-chain fatty acid formation [222].
Thus, ingestion of flavonoids is related to the suppression of inflammatory markers via the downregulation of the transcription factor NF-κB signaling pathway in the gastrointestinal tract that could be a promising strategy in therapeutic approaches preventing chronic diseases and controlling inflammation due to the modulation of the microbiota. However, at high doses flavonoids could exert pro-oxidant properties, act as mutagens and inhibit enzymes involved in hormone metabolism [223,224,225]. Since adverse effects due to flavonoid overdose may outweigh the beneficial activities, the excessive intake of flavonoids in diets should be avoided [224,225].
5. Conclusions and Future Perspectives
Flavonoids are natural molecules, present in human foods and beverages since ancient times; therefore, they do not have dangerous side effects as synthetic anti-cancer drugs Numerous studies have shown their strong positive activities in reducing inflammation, modulating immune response, and supporting and restoring the normal functions of cells. Flavonoids exert a wide range of anticancer effects and, therefore, they could serve as potential compounds for further studies on the development of novel cancer chemopreventive agents and on understanding their detailed mechanisms of action. Furthermore, the daily intake of flavonoids as flavonoid-rich foods or flavonoid supplements could induce favorable changes in the gut microbiota, decreasing the risk of cancer and normalizing vital functions at cellular level.
Acknowledgments
The authors wish to thank Pharmaceutical and Health technology Open access center for support of this study.
Author Contributions
Conceptualization, D.M.K. and J.B.; Literature Review and Resources, D.M.K.; V.J.; A.S. and J.B.; Writing—Original Draft Preparation, D.M.K. and J.B.; Writing—Review and Editing, D.M.K.; V.J.; A.S. and J.B.; Visualization, D.M.K. and J.B.; Supervision, J.B. All authors reviewed the results and approved the final version of the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
References
- 1.Nabavi S.M., Samec D., Tomczyk M., Milella L., Russo D., Habtemariam S., Suntar I., Rastrelli L., Daglia M., Xiao J., et al. Flavonoid biosynthetic pathways in plants: Versatile targets for metabolic engineering. Biotechnol. Adv. 2018 doi: 10.1016/j.biotechadv.2018.11.005. [DOI] [PubMed] [Google Scholar]
- 2.Scarano A., Chieppa M., Santino A. Looking at Flavonoid Biodiversity in Horticultural Crops: A Colored Mine with Nutritional Benefits. Plants. 2018;7:98. doi: 10.3390/plants7040098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Liu J., Wang X., Yong H., Kan J., Jin C. Recent advances in flavonoid-grafted polysaccharides: Synthesis, structural characterization, bioactivities and potential applications. Int. J. Biol. Macromol. 2018;116:1011–1025. doi: 10.1016/j.ijbiomac.2018.05.149. [DOI] [PubMed] [Google Scholar]
- 4.Kofink M., Papagiannopoulos M., Galensa R. (-)-Catechin in cocoa and chocolate: Occurrence and analysis of an atypical flavan-3-ol enantiomer. Molecules. 2007;12:1274–1288. doi: 10.3390/12071274. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Braicu C., Ladomery M.R., Chedea V.S., Irimie A., Berindan-Neagoe I. The relationship between the structure and biological actions of green tea catechins. Food Chem. 2013;141:3282–3289. doi: 10.1016/j.foodchem.2013.05.122. [DOI] [PubMed] [Google Scholar]
- 6.Arts I.C., van De Putte B., Hollman P.C. Catechin contents of foods commonly consumed in The Netherlands. 2. Tea, wine, fruit juices, and chocolate milk. J. Agric. Food Chem. 2000;48:1752–1757. doi: 10.1021/jf000026+. [DOI] [PubMed] [Google Scholar]
- 7.Maatta-Riihinen K.R., Kamal-Eldin A., Torronen A.R. Identification and quantification of phenolic compounds in berries of Fragaria and Rubus species (family Rosaceae) J. Agric. Food Chem. 2004;52:6178–6187. doi: 10.1021/jf049450r. [DOI] [PubMed] [Google Scholar]
- 8.Wu X., Gu L., Prior R.L., McKay S. Characterization of anthocyanins and proanthocyanidins in some cultivars of Ribes, Aronia, and Sambucus and their antioxidant capacity. J. Agric. Food Chem. 2004;52:7846–7856. doi: 10.1021/jf0486850. [DOI] [PubMed] [Google Scholar]
- 9.De Pascual-Teresa S., Santos-Buelga C., Rivas-Gonzalo J.C. Quantitative analysis of flavan-3-ols in Spanish foodstuffs and beverages. J. Agric. Food Chem. 2000;48:5331–5337. doi: 10.1021/jf000549h. [DOI] [PubMed] [Google Scholar]
- 10.Arts I.C., van de Putte B., Hollman P.C. Catechin contents of foods commonly consumed in The Netherlands. 1. Fruits, vegetables, staple foods, and processed foods. J. Agric. Food Chem. 2000;48:1746–1751. doi: 10.1021/jf000025h. [DOI] [PubMed] [Google Scholar]
- 11.Vrhovsek U., Rigo A., Tonon D., Mattivi F. Quantitation of polyphenols in different apple varieties. J. Agric. Food Chem. 2004;52:6532–6538. doi: 10.1021/jf049317z. [DOI] [PubMed] [Google Scholar]
- 12.Landberg R., Naidoo N., van Dam R.M. Diet and endothelial function: From individual components to dietary patterns. Curr. Opin. Lipidol. 2012;23:147–155. doi: 10.1097/MOL.0b013e328351123a. [DOI] [PubMed] [Google Scholar]
- 13.Mejri F., Selmi S., Martins A., Benkhoud H., Baati T., Chaabane H., Njim L., Serralheiro M.L.M., Rauter A.P., Hosni K. Broad bean (Vicia faba L.) pods: A rich source of bioactive ingredients with antimicrobial, antioxidant, enzyme inhibitory, anti-diabetic and health-promoting properties. Food Funct. 2018;9:2051–2059. doi: 10.1039/C8FO00055G. [DOI] [PubMed] [Google Scholar]
- 14.Romani A., Mulinacci N., Pinelli P., Vincieri F.F., Cimato A. Polyphenolic content in five tuscany cultivars of Olea europaea L. J. Agric. Food Chem. 1999;47:964–967. doi: 10.1021/jf980264t. [DOI] [PubMed] [Google Scholar]
- 15.Slimestad R., Fossen T., Vagen I.M. Onions: A source of unique dietary flavonoids. J. Agric. Food Chem. 2007;55:10067–10080. doi: 10.1021/jf0712503. [DOI] [PubMed] [Google Scholar]
- 16.Pandjaitan N., Howard L.R., Morelock T., Gil M.I. Antioxidant capacity and phenolic content of spinach as affected by genetics and maturation. J. Agric. Food Chem. 2005;53:8618–8623. doi: 10.1021/jf052077i. [DOI] [PubMed] [Google Scholar]
- 17.Fattorusso E., Iorizzi M., Lanzotti V., Taglialatela-Scafati O. Chemical composition of shallot (Allium ascalonicum Hort.) J. Agric. Food Chem. 2002;50:5686–5690. doi: 10.1021/jf020396t. [DOI] [PubMed] [Google Scholar]
- 18.Rodriguez-Garcia C., Sanchez-Quesada C. Dietary Flavonoids as Cancer Chemopreventive Agents: An Updated Review of Human Studies. Antioxidants. 2019;8:137. doi: 10.3390/antiox8050137. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yahfoufi N., Alsadi N., Jambi M., Matar C. The Immunomodulatory and Anti-Inflammatory Role of Polyphenols. Nutrients. 2018;10:1618. doi: 10.3390/nu10111618. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Abotaleb M., Samuel S.M., Varghese E., Varghese S., Kubatka P., Liskova A., Busselberg D. Flavonoids in Cancer and Apoptosis. Cancers. 2018;11:28. doi: 10.3390/cancers11010028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Chirumbolo S., Bjorklund G., Lysiuk R., Vella A., Lenchyk L., Upyr T. Targeting Cancer with Phytochemicals via Their Fine Tuning of the Cell Survival Signaling Pathways. Int. J. Mol. Sci. 2018;19:3568. doi: 10.3390/ijms19113568. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Panche A.N., Diwan A.D., Chandra S.R. Flavonoids: An overview. J. Nutr. Sci. 2016;5:e47. doi: 10.1017/jns.2016.41. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Durazzo A., Lucarini M., Souto E.B., Cicala C., Caiazzo E., Izzo A.A., Novellino E., Santini A. Polyphenols: A concise overview on the chemistry, occurrence, and human health. Phytother. Res. 2019;33:2221–2243. doi: 10.1002/ptr.6419. [DOI] [PubMed] [Google Scholar]
- 24.Rosen T. Green tea catechins: Biologic properties, proposed mechanisms of action, and clinical implications. J. Drugs Derm. 2012;11:e55–e60. [PubMed] [Google Scholar]
- 25.Babu P.V., Liu D. Green tea catechins and cardiovascular health: An update. Curr. Med. Chem. 2008;15:1840–1850. doi: 10.2174/092986708785132979. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Ravanel P. Uncoupling activity of a series of flavones and flavonols on isolated plant mitochondria. Phytochemistry. 1986;25:1015–1020. doi: 10.1016/S0031-9422(00)81546-0. [DOI] [Google Scholar]
- 27.van Dijk C., Driessen A.J., Recourt K. The uncoupling efficiency and affinity of flavonoids for vesicles. Biochem. Pharmacol. 2000;60:1593–1600. doi: 10.1016/S0006-2952(00)00488-3. [DOI] [PubMed] [Google Scholar]
- 28.Dorta D.J., Pigoso A.A., Mingatto F.E., Rodrigues T., Prado I.M., Helena A.F., Uyemura S.A., Santos A.C., Curti C. The interaction of flavonoids with mitochondria: Effects on energetic processes. Chem. Biol. Interact. 2005;152:67–78. doi: 10.1016/j.cbi.2005.02.004. [DOI] [PubMed] [Google Scholar]
- 29.Scholz S., Williamson G. Interactions affecting the bioavailability of dietary polyphenols in vivo. Int. J. Vitam Nutr. Res. 2007;77:224–235. doi: 10.1024/0300-9831.77.3.224. [DOI] [PubMed] [Google Scholar]
- 30.Jakobek L. Interactions of polyphenols with carbohydrates, lipids and proteins. Food Chem. 2015;175:556–567. doi: 10.1016/j.foodchem.2014.12.013. [DOI] [PubMed] [Google Scholar]
- 31.Gonzales G.B., Smagghe G., Grootaert C., Zotti M., Raes K., Van Camp J. Flavonoid interactions during digestion, absorption, distribution and metabolism: A sequential structure-activity/property relationship-based approach in the study of bioavailability and bioactivity. Drug Metab. Rev. 2015;47:175–190. doi: 10.3109/03602532.2014.1003649. [DOI] [PubMed] [Google Scholar]
- 32.Swieca M., Gawlik-Dziki U., Dziki D., Baraniak B., Czyz J. The influence of protein-flavonoid interactions on protein digestibility in vitro and the antioxidant quality of breads enriched with onion skin. Food Chem. 2013;141:451–458. doi: 10.1016/j.foodchem.2013.03.048. [DOI] [PubMed] [Google Scholar]
- 33.Hollman P.C., Katan M.B. Absorption, metabolism and health effects of dietary flavonoids in man. Biomed. Pharmacother. 1997;51:305–310. doi: 10.1016/S0753-3322(97)88045-6. [DOI] [PubMed] [Google Scholar]
- 34.Cassidy A., Minihane A.M. The role of metabolism (and the microbiome) in defining the clinical efficacy of dietary flavonoids. Am. J. Clin. Nutr. 2017;105:10–22. doi: 10.3945/ajcn.116.136051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Lotito S.B., Frei B. Consumption of flavonoid-rich foods and increased plasma antioxidant capacity in humans: Cause, consequence, or epiphenomenon? Free Radic. Biol. Med. 2006;41:1727–1746. doi: 10.1016/j.freeradbiomed.2006.04.033. [DOI] [PubMed] [Google Scholar]
- 36.Perez-Vizcaino F., Fraga C.G. Research trends in flavonoids and health. Arch. Biochem. Biophys. 2018;646:107–112. doi: 10.1016/j.abb.2018.03.022. [DOI] [PubMed] [Google Scholar]
- 37.Gorlach S., Fichna J., Lewandowska U. Polyphenols as mitochondria-targeted anticancer drugs. Cancer Lett. 2015;366:141–149. doi: 10.1016/j.canlet.2015.07.004. [DOI] [PubMed] [Google Scholar]
- 38.Neagu M., Constantin C., Popescu I.D., Zipeto D., Tzanakakis G., Nikitovic D., Fenga C., Stratakis C.A., Spandidos D.A., Tsatsakis A.M. Inflammation and Metabolism in Cancer Cell-Mitochondria Key Player. Front. Oncol. 2019;9:348. doi: 10.3389/fonc.2019.00348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Kroemer G., Pouyssegur J. Tumor cell metabolism: cancer's Achilles' heel. Cancer Cell. 2008;13:472–482. doi: 10.1016/j.ccr.2008.05.005. [DOI] [PubMed] [Google Scholar]
- 40.Blackadar C.B. Historical review of the causes of cancer. World J. Clin. Oncol. 2016;7:54–86. doi: 10.5306/wjco.v7.i1.54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Bock F.J., Tait S.W.G. Mitochondria as multifaceted regulators of cell death. Nat. Rev. Mol. Cell Biol. 2019;21:85–100. doi: 10.1038/s41580-019-0173-8. [DOI] [PubMed] [Google Scholar]
- 42.Lebelo M.T., Joubert A.M., Visagie M.H. Warburg effect and its role in tumourigenesis. Arch. Pharmacal Res. 2019;42:833–847. doi: 10.1007/s12272-019-01185-2. [DOI] [PubMed] [Google Scholar]
- 43.Chiche J., Brahimi-Horn M.C., Pouyssegur J. Tumour hypoxia induces a metabolic shift causing acidosis: A common feature in cancer. J. Cell. Mol. Med. 2010;14:771–794. doi: 10.1111/j.1582-4934.2009.00994.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Zaidi N., Lupien L., Kuemmerle N.B., Kinlaw W.B., Swinnen J.V., Smans K. Lipogenesis and lipolysis: The pathways exploited by the cancer cells to acquire fatty acids. Prog. Lipid Res. 2013;52:585–589. doi: 10.1016/j.plipres.2013.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Weinberg F., Ramnath N., Nagrath D. Reactive Oxygen Species in the Tumor Microenvironment: An Overview. Cancers. 2019;11:1191. doi: 10.3390/cancers11081191. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Lu S., Wang Y. Nonmetabolic functions of metabolic enzymes in cancer development. Cancer Commun. 2018;38:63. doi: 10.1186/s40880-018-0336-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Lee N., Kim D. Cancer Metabolism: Fueling More than Just Growth. Mol. Cells. 2016;39:847–854. doi: 10.14348/molcells.2016.0310. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Vegliante R., Di Leo L., Ciccarone F., Ciriolo M.R. Hints on ATGL implications in cancer: Beyond bioenergetic clues. Cell Death Dis. 2018;9:316. doi: 10.1038/s41419-018-0345-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Seyfried T.N., Flores R.E., Poff A.M., D’Agostino D.P. Cancer as a metabolic disease: Implications for novel therapeutics. Carcinogenesis. 2014;35:515–527. doi: 10.1093/carcin/bgt480. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Zhong H., Xiao M., Zarkovic K., Zhu M., Sa R., Lu J., Tao Y., Chen Q., Xia L., Cheng S., et al. Mitochondrial control of apoptosis through modulation of cardiolipin oxidation in hepatocellular carcinoma: A novel link between oxidative stress and cancer. Free Radic. Biol. Med. 2017;102:67–76. doi: 10.1016/j.freeradbiomed.2016.10.494. [DOI] [PubMed] [Google Scholar]
- 51.Kiebish M.A., Han X., Cheng H., Chuang J.H., Seyfried T.N. Cardiolipin and electron transport chain abnormalities in mouse brain tumor mitochondria: Lipidomic evidence supporting the Warburg theory of cancer. J. Lipid Res. 2008;49:2545–2556. doi: 10.1194/jlr.M800319-JLR200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Murphy M.P. How mitochondria produce reactive oxygen species. Biochem. J. 2009;417:1–13. doi: 10.1042/BJ20081386. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Hadi S.M., Asad S.F., Singh S., Ahmad A. Putative mechanism for anticancer and apoptosis-inducing properties of plant-derived polyphenolic compounds. IUBMB Life. 2000;50:167–171. doi: 10.1080/152165400300001471. [DOI] [PubMed] [Google Scholar]
- 54.Link A., Balaguer F., Goel A. Cancer chemoprevention by dietary polyphenols: Promising role for epigenetics. Biochem. Pharmacol. 2010;80:1771–1792. doi: 10.1016/j.bcp.2010.06.036. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Youn H.S., Lee J.Y., Saitoh S.I., Miyake K., Kang K.W., Choi Y.J., Hwang D.H. Suppression of MyD88- and TRIF-dependent signaling pathways of Toll-like receptor by (-)-epigallocatechin-3-gallate, a polyphenol component of green tea. Biochem. Pharmacol. 2006;72:850–859. doi: 10.1016/j.bcp.2006.06.021. [DOI] [PubMed] [Google Scholar]
- 56.Fraga C.G., Galleano M., Verstraeten S.V., Oteiza P.I. Basic biochemical mechanisms behind the health benefits of polyphenols. Mol. Asp. Med. 2010;31:435–445. doi: 10.1016/j.mam.2010.09.006. [DOI] [PubMed] [Google Scholar]
- 57.Oliveira-Marques V., Marinho H.S., Cyrne L., Antunes F. Modulation of NF-kappaB-dependent gene expression by H2O2: A major role for a simple chemical process in a complex biological response. Antioxid Redox Signal. 2009;11:2043–2053. doi: 10.1089/ars.2008.2279. [DOI] [PubMed] [Google Scholar]
- 58.Valko M., Leibfritz D., Moncol J., Cronin M.T., Mazur M., Telser J. Free radicals and antioxidants in normal physiological functions and human disease. Int. J. Biochem. Cell Biol. 2007;39:44–84. doi: 10.1016/j.biocel.2006.07.001. [DOI] [PubMed] [Google Scholar]
- 59.Kaushik S., Shyam H., Agarwal S., Sharma R., Nag T.C., Dwivedi A.K., Balapure A.K. Genistein potentiates Centchroman induced antineoplasticity in breast cancer via PI3K/Akt deactivation and ROS dependent induction of apoptosis. Life Sci. 2019:117073. doi: 10.1016/j.lfs.2019.117073. [DOI] [PubMed] [Google Scholar]
- 60.Jin S., Zhang Q.Y., Kang X.M., Wang J.X., Zhao W.H. Daidzein induces MCF-7 breast cancer cell apoptosis via the mitochondrial pathway. Ann. Oncol. 2010;21:263–268. doi: 10.1093/annonc/mdp499. [DOI] [PubMed] [Google Scholar]
- 61.Pandey P., Sayyed U., Tiwari R.K., Siddiqui M.H., Pathak N., Bajpai P. Hesperidin Induces ROS-Mediated Apoptosis along with Cell Cycle Arrest at G2/M Phase in Human Gall Bladder Carcinoma. Nutr. Cancer. 2019;71:676–687. doi: 10.1080/01635581.2018.1508732. [DOI] [PubMed] [Google Scholar]
- 62.Wu D., Zhang J., Wang J., Li J., Liao F., Dong W. Hesperetin induces apoptosis of esophageal cancer cells via mitochondrial pathway mediated by the increased intracellular reactive oxygen species. Tumour Biol. 2016;37:3451–3459. doi: 10.1007/s13277-015-4176-6. [DOI] [PubMed] [Google Scholar]
- 63.Zhang J., Song J., Wu D., Wang J., Dong W. Hesperetin induces the apoptosis of hepatocellular carcinoma cells via mitochondrial pathway mediated by the increased intracellular reactive oxygen species, ATP and calcium. Med. Oncol. 2015;32:101. doi: 10.1007/s12032-015-0516-z. [DOI] [PubMed] [Google Scholar]
- 64.Palit S., Kar S., Sharma G., Das P.K. Hesperetin Induces Apoptosis in Breast Carcinoma by Triggering Accumulation of ROS and Activation of ASK1/JNK Pathway. J. Cell. Physiol. 2015;230:1729–1739. doi: 10.1002/jcp.24818. [DOI] [PubMed] [Google Scholar]
- 65.Park S., Lim W., Bazer F.W., Song G. Naringenin suppresses growth of human placental choriocarcinoma via reactive oxygen species-mediated P38 and JNK MAPK pathways. Phytomedicine. 2018;50:238–246. doi: 10.1016/j.phymed.2017.08.026. [DOI] [PubMed] [Google Scholar]
- 66.Ahamad M.S., Siddiqui S., Jafri A., Ahmad S., Afzal M., Arshad M. Induction of apoptosis and antiproliferative activity of naringenin in human epidermoid carcinoma cell through ROS generation and cell cycle arrest. PLoS ONE. 2014;9:e110003. doi: 10.1371/journal.pone.0110003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 67.Lim W., Park S., Bazer F.W., Song G. Naringenin-Induced Apoptotic Cell Death in Prostate Cancer Cells Is Mediated via the PI3K/AKT and MAPK Signaling Pathways. J. Cell. Biochem. 2017;118:1118–1131. doi: 10.1002/jcb.25729. [DOI] [PubMed] [Google Scholar]
- 68.Zaidun N.H., Thent Z.C., Latiff A.A. Combating oxidative stress disorders with citrus flavonoid: Naringenin. Life Sci. 2018;208:111–122. doi: 10.1016/j.lfs.2018.07.017. [DOI] [PubMed] [Google Scholar]
- 69.Taparia S.S., Khanna A. Procyanidin-rich extract of natural cocoa powder causes ROS-mediated caspase-3 dependent apoptosis and reduction of pro-MMP-2 in epithelial ovarian carcinoma cell lines. Biomed. Pharmacother. 2016;83:130–140. doi: 10.1016/j.biopha.2016.06.019. [DOI] [PubMed] [Google Scholar]
- 70.Martin M.A., Serrano A.B., Ramos S., Pulido M.I., Bravo L., Goya L. Cocoa flavonoids up-regulate antioxidant enzyme activity via the ERK1/2 pathway to protect against oxidative stress-induced apoptosis in HepG2 cells. J. Nutr. Biochem. 2010;21:196–205. doi: 10.1016/j.jnutbio.2008.10.009. [DOI] [PubMed] [Google Scholar]
- 71.Rodriguez-Ramiro I., Martin M.A., Ramos S., Bravo L., Goya L. Comparative effects of dietary flavanols on antioxidant defences and their response to oxidant-induced stress on Caco2 cells. Eur. J. Nutr. 2011;50:313–322. doi: 10.1007/s00394-010-0139-2. [DOI] [PubMed] [Google Scholar]
- 72.Martin M.A., Goya L., Ramos S. Preventive Effects of Cocoa and Cocoa Antioxidants in Colon Cancer. Diseases. 2016:4. doi: 10.3390/diseases4010006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Martin M.A., Goya L., Ramos S. Potential for preventive effects of cocoa and cocoa polyphenols in cancer. Food Chem. Toxicol. 2013;56:336–351. doi: 10.1016/j.fct.2013.02.020. [DOI] [PubMed] [Google Scholar]
- 74.Rather R.A., Bhagat M. Quercetin as an innovative therapeutic tool for cancer chemoprevention: Molecular mechanisms and implications in human health. Cancer Med. 2019 doi: 10.1002/cam4.1411. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Tang S.M., Deng X.T., Zhou J., Li Q.P., Ge X.X., Miao L. Pharmacological basis and new insights of quercetin action in respect to its anti-cancer effects. Biomed. Pharmacother. 2019;121:109604. doi: 10.1016/j.biopha.2019.109604. [DOI] [PubMed] [Google Scholar]
- 76.Jeon J.S., Kwon S., Ban K., Kwon Hong Y., Ahn C., Sung J.S., Choi I. Regulation of the Intracellular ROS Level Is Critical for the Antiproliferative Effect of Quercetin in the Hepatocellular Carcinoma Cell Line HepG2. Nutr. Cancer. 2019;71:861–869. doi: 10.1080/01635581.2018.1559929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 77.Shang H.S., Lu H.F., Lee C.H., Chiang H.S., Chu Y.L., Chen A., Lin Y.F., Chung J.G. Quercetin induced cell apoptosis and altered gene expression in AGS human gastric cancer cells. Environ. Toxicol. 2018;33:1168–1181. doi: 10.1002/tox.22623. [DOI] [PubMed] [Google Scholar]
- 78.Wu Q., Kroon P.A., Shao H., Needs P.W., Yang X. Differential Effects of Quercetin and Two of Its Derivatives, Isorhamnetin and Isorhamnetin-3-glucuronide, in Inhibiting the Proliferation of Human Breast-Cancer MCF-7 Cells. J. Agric. Food Chem. 2018;66:7181–7189. doi: 10.1021/acs.jafc.8b02420. [DOI] [PubMed] [Google Scholar]
- 79.Wu P., Meng X., Zheng H., Zeng Q., Chen T., Wang W., Zhang X., Su J. Kaempferol Attenuates ROS-Induced Hemolysis and the Molecular Mechanism of Its Induction of Apoptosis on Bladder Cancer. Molecules. 2018;23:2592. doi: 10.3390/molecules23102592. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Choi J.B., Kim J.H., Lee H., Pak J.N., Shim B.S., Kim S.H. Reactive Oxygen Species and p53 Mediated Activation of p38 and Caspases is Critically Involved in Kaempferol Induced Apoptosis in Colorectal Cancer Cells. J. Agric. Food Chem. 2018;66:9960–9967. doi: 10.1021/acs.jafc.8b02656. [DOI] [PubMed] [Google Scholar]
- 81.Seydi E., Salimi A., Rasekh H.R., Mohsenifar Z., Pourahmad J. Selective Cytotoxicity of Luteolin and Kaempferol on Cancerous Hepatocytes Obtained from Rat Model of Hepatocellular Carcinoma: Involvement of ROS-Mediated Mitochondrial Targeting. Nutr. Cancer. 2018;70:594–604. doi: 10.1080/01635581.2018.1460679. [DOI] [PubMed] [Google Scholar]
- 82.Tavsan Z., Kayali H.A. Flavonoids showed anticancer effects on the ovarian cancer cells: Involvement of reactive oxygen species, apoptosis, cell cycle and invasion. Biomed. Pharmacother. 2019;116:109004. doi: 10.1016/j.biopha.2019.109004. [DOI] [PubMed] [Google Scholar]
- 83.Salmani J.M.M., Zhang X.P., Jacob J.A., Chen B.A. Apigenin’s anticancer properties and molecular mechanisms of action: Recent advances and future prospectives. Chin. J. Nat. Med. 2017;15:321–329. doi: 10.1016/S1875-5364(17)30052-3. [DOI] [PubMed] [Google Scholar]
- 84.Souza R.P., Bonfim-Mendonca P.S., Gimenes F., Ratti B.A., Kaplum V., Bruschi M.L., Nakamura C.V., Silva S.O., Maria-Engler S.S., Consolaro M.E. Oxidative Stress Triggered by Apigenin Induces Apoptosis in a Comprehensive Panel of Human Cervical Cancer-Derived Cell Lines. Oxidative Med. Cell. Longev. 2017;2017:1512745. doi: 10.1155/2017/1512745. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Park W., Park S., Lim W., Song G. Chrysin disrupts intracellular homeostasis through mitochondria-mediated cell death in human choriocarcinoma cells. Biochem. Biophys. Res. Commun. 2018;503:3155–3161. doi: 10.1016/j.bbrc.2018.08.109. [DOI] [PubMed] [Google Scholar]
- 86.Xu Y., Tong Y., Ying J., Lei Z., Wan L., Zhu X., Ye F., Mao P., Wu X., Pan R., et al. Chrysin induces cell growth arrest, apoptosis, and ER stress and inhibits the activation of STAT3 through the generation of ROS in bladder cancer cells. Oncol. Lett. 2018;15:9117–9125. doi: 10.3892/ol.2018.8522. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 87.Lim W., Ryu S., Bazer F.W., Kim S.M., Song G. Chrysin attenuates progression of ovarian cancer cells by regulating signaling cascades and mitochondrial dysfunction. J. Cell. Physiol. 2018;233:3129–3140. doi: 10.1002/jcp.26150. [DOI] [PubMed] [Google Scholar]
- 88.Alipour B., Rashidkhani B., Edalati S. Dietary flavonoid intake, total antioxidant capacity and lipid oxidative damage: A cross-sectional study of Iranian women. Nutrition. 2016;32:566–572. doi: 10.1016/j.nut.2015.11.011. [DOI] [PubMed] [Google Scholar]
- 89.Sorrenti V., Vanella L., Acquaviva R., Cardile V., Giofre S., Di Giacomo C. Cyanidin induces apoptosis and differentiation in prostate cancer cells. Int. J. Oncol. 2015;47:1303–1310. doi: 10.3892/ijo.2015.3130. [DOI] [PubMed] [Google Scholar]
- 90.Cvorovic J., Tramer F., Granzotto M., Candussio L., Decorti G., Passamonti S. Oxidative stress-based cytotoxicity of delphinidin and cyanidin in colon cancer cells. Arch. Biochem. Biophys. 2010;501:151–157. doi: 10.1016/j.abb.2010.05.019. [DOI] [PubMed] [Google Scholar]
- 91.McArthur K., Kile B.T. Apoptotic Caspases: Multiple or Mistaken Identities? Trends Cell Biol. 2018;28:475–493. doi: 10.1016/j.tcb.2018.02.003. [DOI] [PubMed] [Google Scholar]
- 92.Jan R., Chaudhry G.E. Understanding Apoptosis and Apoptotic Pathways Targeted Cancer Therapeutics. Adv. Pharm. Bull. 2019;9:205–218. doi: 10.15171/apb.2019.024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Choi E.J., Jung J.Y., Kim G.H. Genistein inhibits the proliferation and differentiation of MCF-7 and 3T3-L1 cells via the regulation of ERalpha expression and induction of apoptosis. Exp. Ther. Med. 2014;8:454–458. doi: 10.3892/etm.2014.1771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Solomon L.A., Ali S., Banerjee S., Munkarah A.R., Morris R.T., Sarkar F.H. Sensitization of ovarian cancer cells to cisplatin by genistein: The role of NF-kappaB. J. Ovarian Res. 2008;1:9. doi: 10.1186/1757-2215-1-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Shafiee G., Saidijam M., Tavilani H., Ghasemkhani N., Khodadadi I. Genistein Induces Apoptosis and Inhibits Proliferation of HT29 Colon Cancer Cells. Int. J. Mol. Cell. Med. 2016;5:178–191. [PMC free article] [PubMed] [Google Scholar]
- 96.Qin J., Teng J., Zhu Z., Chen J., Huang W.J. Genistein induces activation of the mitochondrial apoptosis pathway by inhibiting phosphorylation of Akt in colorectal cancer cells. Pharm. Biol. 2016;54:74–79. doi: 10.3109/13880209.2015.1014921. [DOI] [PubMed] [Google Scholar]
- 97.Danciu C., Avram S., Pavel I.Z., Ghiulai R., Dehelean C.A., Ersilia A., Minda D., Petrescu C., Moaca E.A., Soica C. Main Isoflavones Found in Dietary Sources as Natural Anti-inflammatory Agents. Curr. Drug Targets. 2018;19:841–853. doi: 10.2174/1389450118666171109150731. [DOI] [PubMed] [Google Scholar]
- 98.Liu X., Suzuki N., Santosh Laxmi Y.R., Okamoto Y., Shibutani S. Anti-breast cancer potential of daidzein in rodents. Life Sci. 2012;91:415–419. doi: 10.1016/j.lfs.2012.08.022. [DOI] [PubMed] [Google Scholar]
- 99.Park H.J., Jeon Y.K., You D.H., Nam M.J. Daidzein causes cytochrome c-mediated apoptosis via the Bcl-2 family in human hepatic cancer cells. Food Chem. Toxicol. 2013;60:542–549. doi: 10.1016/j.fct.2013.08.022. [DOI] [PubMed] [Google Scholar]
- 100.Zhang J., Wu D., Vikash, Song J., Wang J., Yi J., Dong W. Hesperetin Induces the Apoptosis of Gastric Cancer Cells via Activating Mitochondrial Pathway by Increasing Reactive Oxygen Species. Dig. Dis. Sci. 2015;60:2985–2995. doi: 10.1007/s10620-015-3696-7. [DOI] [PubMed] [Google Scholar]
- 101.Sivagami G., Vinothkumar R., Bernini R., Preethy C.P., Riyasdeen A., Akbarsha M.A., Menon V.P., Nalini N. Role of hesperetin (a natural flavonoid) and its analogue on apoptosis in HT-29 human colon adenocarcinoma cell line--a comparative study. Food Chem. Toxicol. 2012;50:660–671. doi: 10.1016/j.fct.2011.11.038. [DOI] [PubMed] [Google Scholar]
- 102.Elango R., Athinarayanan J., Subbarayan V.P., Lei D.K.Y., Alshatwi A.A. Hesperetin induces an apoptosis-triggered extrinsic pathway and a p53- independent pathway in human lung cancer H522 cells. J. Asian Nat. Prod. Res. 2018;20:559–569. doi: 10.1080/10286020.2017.1327949. [DOI] [PubMed] [Google Scholar]
- 103.Sambantham S., Radha M., Paramasivam A., Anandan B., Malathi R., Chandra S.R., Jayaraman G. Molecular mechanism underlying hesperetin-induced apoptosis by in silico analysis and in prostate cancer PC-3 cells. Asian Pac. J. Cancer Prev. 2013;14:4347–4352. doi: 10.7314/APJCP.2013.14.7.4347. [DOI] [PubMed] [Google Scholar]
- 104.Bao L., Liu F., Guo H.B., Li Y., Tan B.B., Zhang W.X., Peng Y.H. Naringenin inhibits proliferation, migration, and invasion as well as induces apoptosis of gastric cancer SGC7901 cell line by downregulation of AKT pathway. Tumour Biol. 2016;37:11365–11374. doi: 10.1007/s13277-016-5013-2. [DOI] [PubMed] [Google Scholar]
- 105.Zhang H., Zhong X., Zhang X., Shang D., Zhou Y.I., Zhang C. Enhanced anticancer effect of ABT-737 in combination with naringenin on gastric cancer cells. Exp. Ther. Med. 2016;11:669–673. doi: 10.3892/etm.2015.2912. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Shirakami Y., Sakai H., Kochi T., Seishima M., Shimizu M. Catechins and Its Role in Chronic Diseases. Adv. Exp. Med. Biol. 2016;929:67–90. doi: 10.1007/978-3-319-41342-6_4. [DOI] [PubMed] [Google Scholar]
- 107.Moradzadeh M., Hosseini A., Erfanian S., Rezaei H. Epigallocatechin-3-gallate promotes apoptosis in human breast cancer T47D cells through down-regulation of PI3K/AKT and Telomerase. Pharmacol. Rep. 2017;69:924–928. doi: 10.1016/j.pharep.2017.04.008. [DOI] [PubMed] [Google Scholar]
- 108.Chen L., Zhang H.Y. Cancer preventive mechanisms of the green tea polyphenol (-)-epigallocatechin-3-gallate. Molecules. 2007;12:946–957. doi: 10.3390/12050946. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Liu J., Xing J., Fei Y. Green tea (Camellia sinensis) and cancer prevention: A systematic review of randomized trials and epidemiological studies. Chin. Med. 2008;3:12. doi: 10.1186/1749-8546-3-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Sutherland B.A., Rahman R.M., Appleton I. Mechanisms of action of green tea catechins, with a focus on ischemia-induced neurodegeneration. J. Nutr. Biochem. 2006;17:291–306. doi: 10.1016/j.jnutbio.2005.10.005. [DOI] [PubMed] [Google Scholar]
- 111.Granado-Serrano A.B., Martin M.A., Izquierdo-Pulido M., Goya L., Bravo L., Ramos S. Molecular mechanisms of (-)-epicatechin and chlorogenic acid on the regulation of the apoptotic and survival/proliferation pathways in a human hepatoma cell line. J. Agric. Food Chem. 2007;55:2020–2027. doi: 10.1021/jf062556x. [DOI] [PubMed] [Google Scholar]
- 112.Granado-Serrano A.B., Martin M.A., Haegeman G., Goya L., Bravo L., Ramos S. Epicatechin induces NF-kappaB, activator protein-1 (AP-1) and nuclear transcription factor erythroid 2p45-related factor-2 (Nrf2) via phosphatidylinositol-3-kinase/protein kinase B (PI3K/AKT) and extracellular regulated kinase (ERK) signalling in HepG2 cells. Br. J. Nutr. 2010;103:168–179. doi: 10.1017/S0007114509991747. [DOI] [PubMed] [Google Scholar]
- 113.Ranganathan S., Halagowder D., Sivasithambaram N.D. Quercetin Suppresses Twist to Induce Apoptosis in MCF-7 Breast Cancer Cells. PLoS ONE. 2015;10:e0141370. doi: 10.1371/journal.pone.0141370. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Duo J., Ying G.G., Wang G.W., Zhang L. Quercetin inhibits human breast cancer cell proliferation and induces apoptosis via Bcl-2 and Bax regulation. Mol. Med. Rep. 2012;5:1453–1456. doi: 10.3892/mmr.2012.845. [DOI] [PubMed] [Google Scholar]
- 115.Chou C.C., Yang J.S., Lu H.F., Ip S.W., Lo C., Wu C.C., Lin J.P., Tang N.Y., Chung J.G., Chou M.J., et al. Quercetin-mediated cell cycle arrest and apoptosis involving activation of a caspase cascade through the mitochondrial pathway in human breast cancer MCF-7 cells. Arch. Pharmacal Res. 2010;33:1181–1191. doi: 10.1007/s12272-010-0808-y. [DOI] [PubMed] [Google Scholar]
- 116.Wang P., Heber D., Henning S.M. Quercetin increased the antiproliferative activity of green tea polyphenol (-)-epigallocatechin gallate in prostate cancer cells. Nutr. Cancer. 2012;64:580–587. doi: 10.1080/01635581.2012.661514. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Niu G., Yin S., Xie S., Li Y., Nie D., Ma L., Wang X., Wu Y. Quercetin induces apoptosis by activating caspase-3 and regulating Bcl-2 and cyclooxygenase-2 pathways in human HL-60 cells. Acta Biochim. Biophys. Sin. 2011;43:30–37. doi: 10.1093/abbs/gmq107. [DOI] [PubMed] [Google Scholar]
- 118.Granado-Serrano A.B., Martin M.A., Bravo L., Goya L., Ramos S. Quercetin induces apoptosis via caspase activation, regulation of Bcl-2, and inhibition of PI-3-kinase/Akt and ERK pathways in a human hepatoma cell line (HepG2) J. Nutr. 2006;136:2715–2721. doi: 10.1093/jn/136.11.2715. [DOI] [PubMed] [Google Scholar]
- 119.Sun S., Gong F., Liu P., Miao Q. Metformin combined with quercetin synergistically repressed prostate cancer cells via inhibition of VEGF/PI3K/Akt signaling pathway. Gene. 2018;664:50–57. doi: 10.1016/j.gene.2018.04.045. [DOI] [PubMed] [Google Scholar]
- 120.Imran M., Rauf A., Shah Z.A., Saeed F., Imran A., Arshad M.U., Ahmad B., Bawazeer S., Atif M., Peters D.G., et al. Chemo-preventive and therapeutic effect of the dietary flavonoid kaempferol: A comprehensive review. Phytother. Res. 2019;33:263–275. doi: 10.1002/ptr.6227. [DOI] [PubMed] [Google Scholar]
- 121.Luo H., Rankin G.O., Li Z., Depriest L., Chen Y.C. Kaempferol induces apoptosis in ovarian cancer cells through activating p53 in the intrinsic pathway. Food Chem. 2011;128:513–519. doi: 10.1016/j.foodchem.2011.03.073. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.Kashafi E., Moradzadeh M., Mohamadkhani A., Erfanian S. Kaempferol increases apoptosis in human cervical cancer HeLa cells via PI3K/AKT and telomerase pathways. Biomed. Pharmacother. 2017;89:573–577. doi: 10.1016/j.biopha.2017.02.061. [DOI] [PubMed] [Google Scholar]
- 123.Dean M., Murphy B.T., Burdette J.E. Phytosteroids beyond estrogens: Regulators of reproductive and endocrine function in natural products. Mol. Cell. Endocrinol. 2017;442:98–105. doi: 10.1016/j.mce.2016.12.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Pandey M., Kaur P., Shukla S., Abbas A., Fu P., Gupta S. Plant flavone apigenin inhibits HDAC and remodels chromatin to induce growth arrest and apoptosis in human prostate cancer cells: In vitro and in vivo study. Mol. Carcinog. 2012;51:952–962. doi: 10.1002/mc.20866. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 125.Shukla S., Fu P., Gupta S. Apigenin induces apoptosis by targeting inhibitor of apoptosis proteins and Ku70-Bax interaction in prostate cancer. Apoptosis. 2014;19:883–894. doi: 10.1007/s10495-014-0971-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Meng S., Zhu Y., Li J.F., Wang X., Liang Z., Li S.Q., Xu X., Chen H., Liu B., Zheng X.Y., et al. Apigenin inhibits renal cell carcinoma cell proliferation. Oncotarget. 2017;8:19834–19842. doi: 10.18632/oncotarget.15771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Shi M.D., Shiao C.K., Lee Y.C., Shih Y.W. Apigenin, a dietary flavonoid, inhibits proliferation of human bladder cancer T-24 cells via blocking cell cycle progression and inducing apoptosis. Cancer Cell Int. 2015;15:33. doi: 10.1186/s12935-015-0186-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 128.Zhu Y., Mao Y., Chen H., Lin Y., Hu Z., Wu J., Xu X., Xu X., Qin J., Xie L. Apigenin promotes apoptosis, inhibits invasion and induces cell cycle arrest of T24 human bladder cancer cells. Cancer Cell Int. 2013;13:54. doi: 10.1186/1475-2867-13-54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 129.Liu R., Ji P., Liu B., Qiao H., Wang X., Zhou L., Deng T., Ba Y. Apigenin enhances the cisplatin cytotoxic effect through p53-modulated apoptosis. Oncol. Lett. 2017;13:1024–1030. doi: 10.3892/ol.2016.5495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Khoo B.Y., Chua S.L., Balaram P. Apoptotic effects of chrysin in human cancer cell lines. Int. J. Mol. Sci. 2010;11:2188–2199. doi: 10.3390/ijms11052188. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 131.Xue C., Chen Y., Hu D.N., Iacob C., Lu C., Huang Z. Chrysin induces cell apoptosis in human uveal melanoma cells via intrinsic apoptosis. Oncol. Lett. 2016;12:4813–4820. doi: 10.3892/ol.2016.5251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Chen Y., Wang S., Geng B., Yi Z. Pelargonidin induces antitumor effects in human osteosarcoma cells via autophagy induction, loss of mitochondrial membrane potential, G2/M cell cycle arrest and downregulation of PI3K/AKT signalling pathway. J. BUON. 2018;23:735–740. [PubMed] [Google Scholar]
- 133.Lopez de Las Hazas M.C., Mosele J.I., Macia A., Ludwig I.A., Motilva M.J. Exploring the Colonic Metabolism of Grape and Strawberry Anthocyanins and Their in Vitro Apoptotic Effects in HT-29 Colon Cancer Cells. J. Agric. Food Chem. 2017;65:6477–6487. doi: 10.1021/acs.jafc.6b04096. [DOI] [PubMed] [Google Scholar]
- 134.Hosseini M.M., Karimi A., Behroozaghdam M., Javidi M.A., Ghiasvand S., Bereimipour A., Aryan H., Nassiri F., Jangholi E. Cytotoxic and Apoptogenic Effects of Cyanidin-3-Glucoside on the Glioblastoma Cell Line. World Neurosurg. 2017;108:94–100. doi: 10.1016/j.wneu.2017.08.133. [DOI] [PubMed] [Google Scholar]
- 135.Lim W., Song G. Inhibitory effects of delphinidin on the proliferation of ovarian cancer cells via PI3K/AKT and ERK 1/2 MAPK signal transduction. Oncol. Lett. 2017;14:810–818. doi: 10.3892/ol.2017.6232. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 136.Pal H.C., Sharma S., Strickland L.R., Agarwal J., Athar M., Elmets C.A., Afaq F. Delphinidin reduces cell proliferation and induces apoptosis of non-small-cell lung cancer cells by targeting EGFR/VEGFR2 signaling pathways. PLoS ONE. 2013;8:e77270. doi: 10.1371/journal.pone.0077270. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 137.Gupta S.C., Kunnumakkara A.B., Aggarwal S., Aggarwal B.B. Inflammation, a Double-Edge Sword for Cancer and Other Age-Related Diseases. Front. Immunol. 2018;9:2160. doi: 10.3389/fimmu.2018.02160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 138.Perez-Cano F.J., Castell M. Flavonoids, Inflammation and Immune System. Nutrients. 2016;8:659. doi: 10.3390/nu8100659. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 139.Ding S., Jiang H., Fang J. Regulation of Immune Function by Polyphenols. J. Immunol. Res. 2018;2018:1264074. doi: 10.1155/2018/1264074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 140.Hosseinzade A., Sadeghi O., Naghdipour Biregani A., Soukhtehzari S., Brandt G.S., Esmaillzadeh A. Immunomodulatory Effects of Flavonoids: Possible Induction of T CD4+ Regulatory Cells Through Suppression of mTOR Pathway Signaling Activity. Front. Immunol. 2019;10:51. doi: 10.3389/fimmu.2019.00051. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 141.Mooradian M.J., Sullivan R.J. Immunomodulatory effects of current cancer treatment and the consequences for follow-up immunotherapeutics. Future Oncol. 2017;13:1649–1663. doi: 10.2217/fon-2017-0117. [DOI] [PubMed] [Google Scholar]
- 142.Xu L., Zhang Y., Tian K., Chen X., Zhang R., Mu X., Wu Y., Wang D., Wang S., Liu F., et al. Apigenin suppresses PD-L1 expression in melanoma and host dendritic cells to elicit synergistic therapeutic effects. J. Exp. Clin. Cancer Res. 2018;37:261. doi: 10.1186/s13046-018-0929-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 143.Li W., Kim T.I., Kim J.H., Chung H.S. Immune Checkpoint PD-1/PD-L1 CTLA-4/CD80 are Blocked by Rhus verniciflua Stokes and its Active Compounds. Molecules. 2019;24:4062. doi: 10.3390/molecules24224062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 144.Bao F., Bai H.Y., Wu Z.R., Yang Z.G. Phenolic compounds from cultivated Glycyrrhiza uralensis and their PD-1/PD-L1 inhibitory activities. Nat. Prod. Res. 2019:1–8. doi: 10.1080/14786419.2019.1586698. [DOI] [PubMed] [Google Scholar]
- 145.Sundaram M.K., Unni S., Somvanshi P., Bhardwaj T., Mandal R.K., Hussain A., Haque S. Genistein Modulates Signaling Pathways and Targets Several Epigenetic Markers in HeLa Cells. Genes. 2019;10:955. doi: 10.3390/genes10120955. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 146.Pons D.G., Vilanova-Llompart J., Gaya-Bover A., Alorda-Clara M., Oliver J., Roca P., Sastre-Serra J. The phytoestrogen genistein affects inflammatory-related genes expression depending on the ERalpha/ERbeta ratio in breast cancer cells. Int. J. Food Sci. Nutr. 2019;70:941–949. doi: 10.1080/09637486.2019.1597025. [DOI] [PubMed] [Google Scholar]
- 147.Ning Y., Feng W., Cao X., Ren K., Quan M., Chen A., Xu C., Qiu Y., Cao J., Li X., et al. Genistein inhibits stemness of SKOV3 cells induced by macrophages co-cultured with ovarian cancer stem-like cells through IL-8/STAT3 axis. J. Exp. Clin. Cancer Res. 2019;38:19. doi: 10.1186/s13046-018-1010-1. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 148.Sakamoto Y., Kanatsu J., Toh M., Naka A., Kondo K., Iida K. The Dietary Isoflavone Daidzein Reduces Expression of Pro-Inflammatory Genes through PPARalpha/gamma and JNK Pathways in Adipocyte and Macrophage Co-Cultures. PLoS ONE. 2016;11:e0149676. doi: 10.1371/journal.pone.0149676. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 149.Ren H., Hao J., Liu T., Zhang D., Lv H., Song E., Zhu C. Hesperetin Suppresses Inflammatory Responses in Lipopolysaccharide-Induced RAW 264.7 Cells via the Inhibition of NF-kappaB and Activation of Nrf2/HO-1 Pathways. Inflammation. 2016;39:964–973. doi: 10.1007/s10753-016-0311-9. [DOI] [PubMed] [Google Scholar]
- 150.Nalini N., Aranganathan S., Kabalimurthy J. Chemopreventive efficacy of hesperetin (citrus flavonone) against 1,2-dimethylhydrazine-induced rat colon carcinogenesis. Toxicol. Mech. Methods. 2012;22:397–408. doi: 10.3109/15376516.2012.673092. [DOI] [PubMed] [Google Scholar]
- 151.Ahmed O.M., Ahmed A.A., Fahim H.I., Zaky M.Y. Quercetin and naringenin abate diethylnitrosamine/acetylaminofluorene-induced hepatocarcinogenesis in Wistar rats: The roles of oxidative stress, inflammation and cell apoptosis. Drug Chem. Toxicol. 2019 doi: 10.1080/01480545.2019.1683187. [DOI] [PubMed] [Google Scholar]
- 152.Zhao Z., Jin G., Ge Y., Guo Z. Naringenin inhibits migration of breast cancer cells via inflammatory and apoptosis cell signaling pathways. Inflammopharmacology. 2019;27:1021–1036. doi: 10.1007/s10787-018-00556-3. [DOI] [PubMed] [Google Scholar]
- 153.Chen Y.Y., Chang Y.M., Wang K.Y., Chen P.N., Hseu Y.C., Chen K.M., Yeh K.T., Chen C.J., Hsu L.S. Naringenin inhibited migration and invasion of glioblastoma cells through multiple mechanisms. Environ. Toxicol. 2019;34:233–239. doi: 10.1002/tox.22677. [DOI] [PubMed] [Google Scholar]
- 154.Rodriguez-Ramiro I., Ramos S., Lopez-Oliva E., Agis-Torres A., Bravo L., Goya L., Martin M.A. Cocoa polyphenols prevent inflammation in the colon of azoxymethane-treated rats and in TNF-alpha-stimulated Caco-2 cells. Br. J. Nutr. 2013;110:206–215. doi: 10.1017/S0007114512004862. [DOI] [PubMed] [Google Scholar]
- 155.Liu Y., Tang Z.G., Lin Y., Qu X.G., Lv W., Wang G.B., Li C.L. Effects of quercetin on proliferation and migration of human glioblastoma U251 cells. Biomed. Pharmacother. 2017;92:33–38. doi: 10.1016/j.biopha.2017.05.044. [DOI] [PubMed] [Google Scholar]
- 156.Maurya A.K., Vinayak M. Quercetin Attenuates Cell Survival, Inflammation, and Angiogenesis via Modulation of AKT Signaling in Murine T-Cell Lymphoma. Nutr. Cancer. 2017;69:470–480. doi: 10.1080/01635581.2017.1267775. [DOI] [PubMed] [Google Scholar]
- 157.Han M., Song Y., Zhang X. Quercetin Suppresses the Migration and Invasion in Human Colon Cancer Caco-2 Cells Through Regulating Toll-like Receptor 4/Nuclear Factor-kappa B Pathway. Pharmacogn. Mag. 2016;12:S237–S244. doi: 10.4103/0973-1296.182154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 158.Bruning A. Inhibition of mTOR signaling by quercetin in cancer treatment and prevention. Anti Cancer Agents Med. Chem. 2013;13:1025–1031. doi: 10.2174/18715206113139990114. [DOI] [PubMed] [Google Scholar]
- 159.Lee S., Kim Y.J., Kwon S., Lee Y., Choi S.Y., Park J., Kwon H.J. Inhibitory effects of flavonoids on TNF-alpha-induced IL-8 gene expression in HEK 293 cells. BMB Rep. 2009;42:265–270. doi: 10.5483/BMBRep.2009.42.5.265. [DOI] [PubMed] [Google Scholar]
- 160.Ren J., Lu Y., Qian Y., Chen B., Wu T., Ji G. Recent progress regarding kaempferol for the treatment of various diseases. Exp. Ther. Med. 2019;18:2759–2776. doi: 10.3892/etm.2019.7886. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 161.Devi K.P., Malar D.S., Nabavi S.F., Sureda A., Xiao J., Nabavi S.M., Daglia M. Kaempferol and inflammation: From chemistry to medicine. Pharmacol. Res. 2015;99:1–10. doi: 10.1016/j.phrs.2015.05.002. [DOI] [PubMed] [Google Scholar]
- 162.Kadioglu O., Nass J., Saeed M.E., Schuler B., Efferth T. Kaempferol Is an Anti-Inflammatory Compound with Activity towards NF-kappaB Pathway Proteins. Anticancer Res. 2015;35:2645–2650. [PubMed] [Google Scholar]
- 163.Ghitu A., Schwiebs A., Radeke H.H., Avram S., Zupko I., Bor A., Pavel I.Z., Dehelean C.A., Oprean C., Bojin F., et al. A Comprehensive Assessment of Apigenin as an Antiproliferative, Proapoptotic, Antiangiogenic and Immunomodulatory Phytocompound. Nutrients. 2019;11:858. doi: 10.3390/nu11040858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 164.Ai X.Y., Qin Y., Liu H.J., Cui Z.H., Li M., Yang J.H., Zhong W.L., Liu Y.R., Chen S., Sun T., et al. Apigenin inhibits colonic inflammation and tumorigenesis by suppressing STAT3-NF-kappaB signaling. Oncotarget. 2017;8:100216–100226. doi: 10.18632/oncotarget.22145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 165.Ginwala R., Bhavsar R., Chigbu D.I., Jain P., Khan Z.K. Potential Role of Flavonoids in Treating Chronic Inflammatory Diseases with a Special Focus on the Anti-Inflammatory Activity of Apigenin. Antioxidants. 2019;8:35. doi: 10.3390/antiox8020035. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Rehman M.U., Tahir M., Khan A.Q., Khan R., Lateef A., Oday O.H., Qamar W., Ali F., Sultana S. Chrysin suppresses renal carcinogenesis via amelioration of hyperproliferation, oxidative stress and inflammation: Plausible role of NF-kappaB. Toxicol. Lett. 2013;216:146–158. doi: 10.1016/j.toxlet.2012.11.013. [DOI] [PubMed] [Google Scholar]
- 167.Liu C., Zhu L., Fukuda K., Ouyang S., Chen X., Wang C., Zhang C.J., Martin B., Gu C., Qin L., et al. The flavonoid cyanidin blocks binding of the cytokine interleukin-17A to the IL-17RA subunit to alleviate inflammation in vivo. Sci. Signal. 2017;10:eaaf8823. doi: 10.1126/scisignal.aaf8823. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 168.Lee B.S., Lee C., Yang S., Park E.K., Ku S.K., Bae J.S. Suppressive effects of pelargonidin on lipopolysaccharide-induced inflammatory responses. Chem. Biol. Interact. 2019;302:67–73. doi: 10.1016/j.cbi.2019.02.007. [DOI] [PubMed] [Google Scholar]
- 169.Venancio V.P., Cipriano P.A., Kim H., Antunes L.M., Talcott S.T., Mertens-Talcott S.U. Cocoplum (Chrysobalanus icaco L.) anthocyanins exert anti-inflammatory activity in human colon cancer and non-malignant colon cells. Food Funct. 2017;8:307–314. doi: 10.1039/C6FO01498D. [DOI] [PubMed] [Google Scholar]
- 170.Im N.K., Jang W.J., Jeong C.H., Jeong G.S. Delphinidin suppresses PMA-induced MMP-9 expression by blocking the NF-kappaB activation through MAPK signaling pathways in MCF-7 human breast carcinoma cells. J. Med. Food. 2014;17:855–861. doi: 10.1089/jmf.2013.3077. [DOI] [PubMed] [Google Scholar]
- 171.Porporato P.E., Filigheddu N., Pedro J.M.B., Kroemer G., Galluzzi L. Mitochondrial metabolism and cancer. Cell Res. 2018;28:265–280. doi: 10.1038/cr.2017.155. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 172.Kwak T.W., Park S.B., Kim H.J., Jeong Y.I., Kang D.H. Anticancer activities of epigallocatechin-3-gallate against cholangiocarcinoma cells. Oncotargets Ther. 2017;10:137–144. doi: 10.2147/OTT.S112364. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 173.Klingenberg M. The ADP and ATP transport in mitochondria and its carrier. Biochim. Biophys. Acta. 2008;1778:1978–2021. doi: 10.1016/j.bbamem.2008.04.011. [DOI] [PubMed] [Google Scholar]
- 174.Halestrap A.P. Mitochondrial permeability: Dual role for the ADP/ATP translocator? Nature. 2004;430:984. doi: 10.1038/nature02816. [DOI] [PubMed] [Google Scholar]
- 175.Ortega R., Garcia N. The flavonoid quercetin induces changes in mitochondrial permeability by inhibiting adenine nucleotide translocase. J. Bioenerg. Biomembr. 2009;41:41–47. doi: 10.1007/s10863-009-9198-6. [DOI] [PubMed] [Google Scholar]
- 176.Oishi M., Iizumi Y., Taniguchi T., Goi W., Miki T., Sakai T. Apigenin sensitizes prostate cancer cells to Apo2L/TRAIL by targeting adenine nucleotide translocase-2. PLoS ONE. 2013;8:e55922. doi: 10.1371/journal.pone.0055922. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 177.Moini H., Arroyo A., Vaya J., Packer L. Bioflavonoid effects on the mitochondrial respiratory electron transport chain and cytochrome c redox state. Redox Rep. 1999;4:35–41. doi: 10.1179/135100099101534729. [DOI] [PubMed] [Google Scholar]
- 178.Salvi M., Brunati A.M., Clari G., Toninello A. Interaction of genistein with the mitochondrial electron transport chain results in opening of the membrane transition pore. Biochim. Biophys. Acta. 2002;1556:187–196. doi: 10.1016/S0005-2728(02)00361-4. [DOI] [PubMed] [Google Scholar]
- 179.Valenti D., de Bari L., Manente G.A., Rossi L., Mutti L., Moro L., Vacca R.A. Negative modulation of mitochondrial oxidative phosphorylation by epigallocatechin-3 gallate leads to growth arrest and apoptosis in human malignant pleural mesothelioma cells. Biochim. Biophys. Acta. 2013;1832:2085–2096. doi: 10.1016/j.bbadis.2013.07.014. [DOI] [PubMed] [Google Scholar]
- 180.Oliveira M.R., Nabavi S.F., Daglia M., Rastrelli L., Nabavi S.M. Epigallocatechin gallate and mitochondria-A story of life and death. Pharmacol. Res. 2016;104:70–85. doi: 10.1016/j.phrs.2015.12.027. [DOI] [PubMed] [Google Scholar]
- 181.Bendokas V., Skemiene K., Trumbeckaite S., Stanys V., Passamonti S., Borutaite V., Liobikas J. Anthocyanins: From plant pigments to health benefits at mitochondrial level. Crit. Rev. Food Sci. Nutr. 2019:1–14. doi: 10.1080/10408398.2019.1687421. [DOI] [PubMed] [Google Scholar]
- 182.Liobikas J., Skemiene K., Trumbeckaite S., Borutaite V. Anthocyanins in cardioprotection: A path through mitochondria. Pharmacol. Res. 2016;113:808–815. doi: 10.1016/j.phrs.2016.03.036. [DOI] [PubMed] [Google Scholar]
- 183.Buss G.D., Constantin J., de Lima L.C., Teodoro G.R., Comar J.F., Ishii-Iwamoto E.L., Bracht A. The action of quercetin on the mitochondrial NADH to NAD(+) ratio in the isolated perfused rat liver. Planta Med. 2005;71:1118–1122. doi: 10.1055/s-2005-873174. [DOI] [PubMed] [Google Scholar]
- 184.Wang J., Fang F., Huang Z., Wang Y., Wong C. Kaempferol is an estrogen-related receptor alpha and gamma inverse agonist. FEBS Lett. 2009;583:643–647. doi: 10.1016/j.febslet.2009.01.030. [DOI] [PubMed] [Google Scholar]
- 185.Constantin R.P., do Nascimento G.S., Constantin R.P., Salgueiro C.L., Bracht A., Ishii-Iwamoto E.L., Yamamoto N.S., Constantin J. Citrus flavanones affect hepatic fatty acid oxidation in rats by acting as prooxidant agents. Biomed. Res. Int. 2013;2013:342973. doi: 10.1155/2013/342973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 186.Chen L.B. Mitochondrial membrane potential in living cells. Annu. Rev. Cell Biol. 1988;4:155–181. doi: 10.1146/annurev.cb.04.110188.001103. [DOI] [PubMed] [Google Scholar]
- 187.Fantin V.R., St-Pierre J., Leder P. Attenuation of LDH-A expression uncovers a link between glycolysis, mitochondrial physiology, and tumor maintenance. Cancer Cell. 2006;9:425–434. doi: 10.1016/j.ccr.2006.04.023. [DOI] [PubMed] [Google Scholar]
- 188.Bernatoniene J., Kopustinskiene D.M., Jakstas V., Majiene D., Baniene R., Kursvietiene L., Masteikova R., Savickas A., Toleikis A., Trumbeckaite S. The effect of Leonurus cardiaca herb extract and some of its flavonoids on mitochondrial oxidative phosphorylation in the heart. Planta Med. 2014;80:525–532. doi: 10.1055/s-0034-1368426. [DOI] [PubMed] [Google Scholar]
- 189.Bernatoniene J., Trumbeckaite S., Majiene D., Baniene R., Baliutyte G., Savickas A., Toleikis A. The effect of crataegus fruit extract and some of its flavonoids on mitochondrial oxidative phosphorylation in the heart. Phytother. Res. 2009;23:1701–1707. doi: 10.1002/ptr.2815. [DOI] [PubMed] [Google Scholar]
- 190.Trumbeckaite S., Bernatoniene J., Majiene D., Jakstas V., Savickas A., Toleikis A. The effect of flavonoids on rat heart mitochondrial function. Biomed. Pharmacother. 2006;60:245–248. doi: 10.1016/j.biopha.2006.04.003. [DOI] [PubMed] [Google Scholar]
- 191.Kopustinskiene D.M., Savickas A., Vetchy D., Masteikova R., Kasauskas A., Bernatoniene J. Direct effects of (-)-epicatechin and procyanidin B2 on the respiration of rat heart mitochondria. Biomed. Res. Int. 2015;2015:232836. doi: 10.1155/2015/232836. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 192.Oteiza P.I., Fraga C.G., Mills D.A., Taft D.H. Flavonoids and the gastrointestinal tract: Local and systemic effects. Mol. Asp. Med. 2018;61:41–49. doi: 10.1016/j.mam.2018.01.001. [DOI] [PubMed] [Google Scholar]
- 193.Wells J.M., Brummer R.J., Derrien M., MacDonald T.T., Troost F., Cani P.D., Theodorou V., Dekker J., Meheust A., de Vos W.M., et al. Homeostasis of the gut barrier and potential biomarkers. Am. J. Physiology. Gastrointest. Liver Physiol. 2017;312:G171–G193. doi: 10.1152/ajpgi.00048.2015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 194.Rowland I., Gibson G., Heinken A., Scott K., Swann J., Thiele I., Tuohy K. Gut microbiota functions: Metabolism of nutrients and other food components. Eur. J. Nutr. 2018;57:1–24. doi: 10.1007/s00394-017-1445-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 195.Qu G., Chen J., Guo X. The beneficial and deleterious role of dietary polyphenols on chronic degenerative diseases by regulating gene expression. Biosci. Trends. 2019;12:526–536. doi: 10.5582/bst.2018.01172. [DOI] [PubMed] [Google Scholar]
- 196.Espin J.C., Gonzalez-Sarrias A., Tomas-Barberan F.A. The gut microbiota: A key factor in the therapeutic effects of (poly)phenols. Biochem. Pharmacol. 2017;139:82–93. doi: 10.1016/j.bcp.2017.04.033. [DOI] [PubMed] [Google Scholar]
- 197.Bettaieb A., Vazquez Prieto M.A., Rodriguez Lanzi C., Miatello R.M., Haj F.G., Fraga C.G., Oteiza P.I. (-)-Epicatechin mitigates high-fructose-associated insulin resistance by modulating redox signaling and endoplasmic reticulum stress. Free Radic. Biol. Med. 2014;72:247–256. doi: 10.1016/j.freeradbiomed.2014.04.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 198.Gutierrez-Salmean G., Ortiz-Vilchis P., Vacaseydel C.M., Garduno-Siciliano L., Chamorro-Cevallos G., Meaney E., Villafana S., Villarreal F., Ceballos G., Ramirez-Sanchez I. Effects of (-)-epicatechin on a diet-induced rat model of cardiometabolic risk factors. Eur. J. Pharmacol. 2014;728:24–30. doi: 10.1016/j.ejphar.2014.01.053. [DOI] [PubMed] [Google Scholar]
- 199.Vazquez Prieto M.A., Bettaieb A., Rodriguez Lanzi C., Soto V.C., Perdicaro D.J., Galmarini C.R., Haj F.G., Miatello R.M., Oteiza P.I. Catechin and quercetin attenuate adipose inflammation in fructose-fed rats and 3T3-L1 adipocytes. Mol. Nutr. Food Res. 2015;59:622–633. doi: 10.1002/mnfr.201400631. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 200.Cremonini E., Mastaloudis A., Hester S.N., Verstraeten S.V., Anderson M., Wood S.M., Waterhouse A.L., Fraga C.G., Oteiza P.I. Anthocyanins inhibit tumor necrosis alpha-induced loss of Caco-2 cell barrier integrity. Food Funct. 2017;8:2915–2923. doi: 10.1039/C7FO00625J. [DOI] [PubMed] [Google Scholar]
- 201.Olejnik A., Kowalska K., Kidon M., Czapski J., Rychlik J., Olkowicz M., Dembczynski R. Purple carrot anthocyanins suppress lipopolysaccharide-induced inflammation in the co-culture of intestinal Caco-2 and macrophage RAW264.7 cells. Food Funct. 2016;7:557–564. doi: 10.1039/C5FO00890E. [DOI] [PubMed] [Google Scholar]
- 202.He X., Sun L.M. Dietary intake of flavonoid subclasses and risk of colorectal cancer: Evidence from population studies. Oncotarget. 2016;7:26617–26627. doi: 10.18632/oncotarget.8562. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 203.Grosso G., Godos J., Lamuela-Raventos R., Ray S., Micek A., Pajak A., Sciacca S., D'Orazio N., Del Rio D., Galvano F. A comprehensive meta-analysis on dietary flavonoid and lignan intake and cancer risk: Level of evidence and limitations. Mol. Nutr. Food Res. 2017;61 doi: 10.1002/mnfr.201600930. [DOI] [PubMed] [Google Scholar]
- 204.Rossi M., Bosetti C., Negri E., Lagiou P., La Vecchia C. Flavonoids, proanthocyanidins, and cancer risk: A network of case-control studies from Italy. Nutr. Cancer. 2010;62:871–877. doi: 10.1080/01635581.2010.509534. [DOI] [PubMed] [Google Scholar]
- 205.Li Y., Zhang T., Chen G.Y. Flavonoids and Colorectal Cancer Prevention. Antioxidants. 2018;7:187. doi: 10.3390/antiox7120187. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 206.Masumoto S., Terao A., Yamamoto Y., Mukai T., Miura T., Shoji T. Non-absorbable apple procyanidins prevent obesity associated with gut microbial and metabolomic changes. Sci. Rep. 2016;6:31208. doi: 10.1038/srep31208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 207.Sugiyama H., Akazome Y., Shoji T., Yamaguchi A., Yasue M., Kanda T., Ohtake Y. Oligomeric procyanidins in apple polyphenol are main active components for inhibition of pancreatic lipase and triglyceride absorption. J. Agric. Food Chem. 2007;55:4604–4609. doi: 10.1021/jf070569k. [DOI] [PubMed] [Google Scholar]
- 208.Fei Q., Gao Y., Zhang X., Sun Y., Hu B., Zhou L., Jabbar S., Zeng X. Effects of Oolong tea polyphenols, EGCG, and EGCG3″Me on pancreatic alpha-amylase activity in vitro. J. Agric. Food Chem. 2014;62:9507–9514. doi: 10.1021/jf5032907. [DOI] [PubMed] [Google Scholar]
- 209.Cardona F., Andres-Lacueva C., Tulipani S., Tinahones F.J., Queipo-Ortuno M.I. Benefits of polyphenols on gut microbiota and implications in human health. J. Nutr. Biochem. 2013;24:1415–1422. doi: 10.1016/j.jnutbio.2013.05.001. [DOI] [PubMed] [Google Scholar]
- 210.David L.A., Maurice C.F., Carmody R.N., Gootenberg D.B., Button J.E., Wolfe B.E., Ling A.V., Devlin A.S., Varma Y., Fischbach M.A., et al. Diet rapidly and reproducibly alters the human gut microbiome. Nature. 2014;505:559–563. doi: 10.1038/nature12820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 211.De Leoz M.L., Kalanetra K.M., Bokulich N.A., Strum J.S., Underwood M.A., German J.B., Mills D.A., Lebrilla C.B. Human milk glycomics and gut microbial genomics in infant feces show a correlation between human milk oligosaccharides and gut microbiota: A proof-of-concept study. J. Proteome Res. 2015;14:491–502. doi: 10.1021/pr500759e. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 212.Marchesi J.R., Adams D.H., Fava F., Hermes G.D., Hirschfield G.M., Hold G., Quraishi M.N., Kinross J., Smidt H., Tuohy K.M., et al. The gut microbiota and host health: A new clinical frontier. Gut. 2016;65:330–339. doi: 10.1136/gutjnl-2015-309990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 213.Walle T. Absorption and metabolism of flavonoids. Free Radic. Biol. Med. 2004;36:829–837. doi: 10.1016/j.freeradbiomed.2004.01.002. [DOI] [PubMed] [Google Scholar]
- 214.Kumar S., Pandey A.K. Chemistry and biological activities of flavonoids: An overview. Sci. World J. 2013;2013:162750. doi: 10.1155/2013/162750. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 215.Williamson G., Clifford M.N. Role of the small intestine, colon and microbiota in determining the metabolic fate of polyphenols. Biochem. Pharmacol. 2017;139:24–39. doi: 10.1016/j.bcp.2017.03.012. [DOI] [PubMed] [Google Scholar]
- 216.Duda-Chodak A. The inhibitory effect of polyphenols on human gut microbiota. J. Physiol. Pharmacol. 2012;63:497–503. [PubMed] [Google Scholar]
- 217.Nohynek L.J., Alakomi H.L., Kahkonen M.P., Heinonen M., Helander I.M., Oksman-Caldentey K.M., Puupponen-Pimia R.H. Berry phenolics: Antimicrobial properties and mechanisms of action against severe human pathogens. Nutr. Cancer. 2006;54:18–32. doi: 10.1207/s15327914nc5401_4. [DOI] [PubMed] [Google Scholar]
- 218.Espley R.V., Butts C.A., Laing W.A., Martell S., Smith H., McGhie T.K., Zhang J., Paturi G., Hedderley D., Bovy A., et al. Dietary flavonoids from modified apple reduce inflammation markers and modulate gut microbiota in mice. J. Nutr. 2014;144:146–154. doi: 10.3945/jn.113.182659. [DOI] [PubMed] [Google Scholar]
- 219.Gwiazdowska D., Jus K., Jasnowska-Malecka J., Kluczynska K. The impact of polyphenols on Bifidobacterium growth. Acta Biochim. Pol. 2015;62:895–901. doi: 10.18388/abp.2015_1154. [DOI] [PubMed] [Google Scholar]
- 220.Ridlon J.M., Kang D.J., Hylemon P.B., Bajaj J.S. Bile acids and the gut microbiome. Curr. Opin. Gastroenterol. 2014;30:332–338. doi: 10.1097/MOG.0000000000000057. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 221.Maynard C.L., Elson C.O., Hatton R.D., Weaver C.T. Reciprocal interactions of the intestinal microbiota and immune system. Nature. 2012;489:231–241. doi: 10.1038/nature11551. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 222.Shortt C., Hasselwander O., Meynier A., Nauta A., Fernandez E.N., Putz P., Rowland I., Swann J., Turk J., Vermeiren J., et al. Systematic review of the effects of the intestinal microbiota on selected nutrients and non-nutrients. Eur. J. Nutr. 2018;57:25–49. doi: 10.1007/s00394-017-1546-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 223.Kuo S.M. Dietary flavonoid and cancer prevention: Evidence and potential mechanism. Crit. Rev. Oncog. 1997;8:47–69. doi: 10.1615/CritRevOncog.v8.i1.30. [DOI] [PubMed] [Google Scholar]
- 224.Galati G., O'Brien P.J. Potential toxicity of flavonoids and other dietary phenolics: Significance for their chemopreventive and anticancer properties. Free Radic. Biol. Med. 2004;37:287–303. doi: 10.1016/j.freeradbiomed.2004.04.034. [DOI] [PubMed] [Google Scholar]
- 225.Skibola C.F., Smith M.T. Potential health impacts of excessive flavonoid intake. Free Radic. Biol. Med. 2000;29:375–383. doi: 10.1016/S0891-5849(00)00304-X. [DOI] [PubMed] [Google Scholar]