Abstract
Polyphenols form a diverse group of compounds containing at least two hydroxyl groups in their chemical structure. Because of the common presence in plant kingdom, polyphenols are considered a significant component of food and an important group of compounds with antioxidant properties. The absorption of polyphenols present in food depends mostly on the activity of intestinal microflora. However, little is known about the processes and interactions responsible for such phenomenon in guts ecosystem. There are only few available publications that examine the effect on polyphenols on intestinal microbiota. Therefore, this work will focus on describing the relationship between polyphenol compounds present in food and bacteria colonizing the intestines, their mechanism, and impact on human’s health.
Keywords: polyphenols, gut microbiota, probiotics, antioxidants
1. Introduction
Intestinal Microflora
In human’s gastrointestinal tract, there are over one thousand species of bacteria. The representatives of eukaryotes and prokaryotes such as Firmicutes, Bacteroidetes, Actinobacteria, and Proteobacteria form a permanent or temporary component of microbiota [1]. Among them there are symbiotic and commensal microorganisms, but also those that can cause diseases. Intestinal microflora take part in creating short-chain fatty acids (SCFAs) and branched-chain fatty acids (BCFAs) that are important nutrients for intestinal cells [2]. SCFAs not only stimulate division and differentiation of enterocytes, but also regulate and maintain the mineral balance of organism and promote iron, calcium, and magnesium absorption [3]. The products that are created during fermentation of microorganisms regulate metabolism of lipids and glucose [4]. Microflora of gastrointestinal tract stimulates the synthesis of mucin, which protects organisms from toxic substances and pathogens [5]. The protective function of the intestine is also connected to the degradation of toxins and insulation of its walls. The microorganisms colonizing human’s gut are responsible for modulating the immune system [6] and participate in the production of vitamins such as B and K. Despite of all the above-mentioned benefits, the bacteria could also cause negative changes in health. Abnormal microflora can impact the growth of neoplasm in gastrointestinal tract, mainly in the large intestine due to production of mutagens and carcinogens (E. coli, E. faecalis, Basteroides) [7]. Low content of fiber, high content of protein and fats, but also unhealthy, highly processed food can cause disturbance of microbiota [8]. Moreover, taking medications such as antibiotics, chemotherapeutics, non-steroidal anti-inflammatory drugs, proton pump inhibitors, antineoplastic agents, stress, infections, and stimulants might affect microflora in a similar way to incorrect food content [9,10]. The possible effect of such a disturbance is the development of allergy [11] or diabetes mellitus type 1 [12]. Additionally, recent studies show the connection between intestinal microbiota and the central nervous system where the imbalance might be a possible cause of mental diseases e.g., depression [13,14]. Dysbiosis in gastrointestinal tract can also affect metabolic disorders and have an impact on obesity [12], heart diseases, and diabetes mellitus type 2 [15].
2. Phenol Compounds
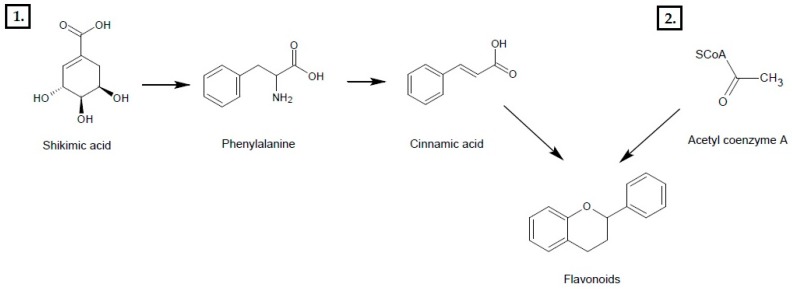
Phenols represent a very important group of antioxidant compounds, widespread among plants [16] that arise as products of plants’ metabolism in the wake of stress, damage, infection, or UV radiation. They are a cohort of compounds of diversified chemical structure emergent in two pathways. In shikimic acid’s transformation, hydroxycinnamic acids and coumarins come into existence. Simple structure polyphenol compounds are made of acetic acid. Structurally more complex flavonoids are formed as a result of these two routes (Figure 1). [17].
Figure 1.
Biosynthesis of Flavonoids. 1. The shikimic acid pathway; 2. The acetate pathway.
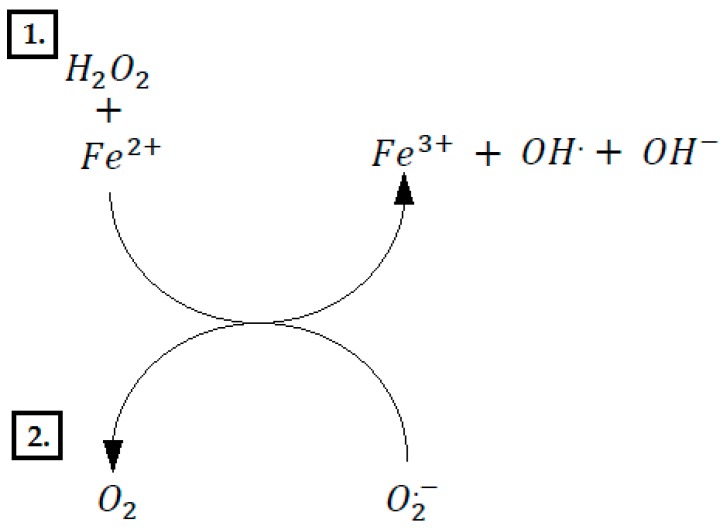
Due to the composition, polyphenol compounds might be differentiated into phenolic acids, flavonoids, stilbenes, and lignans (Table 1) [18]. Flavonoids are a large group of compounds composed of two benzene rings connected by a tri-carbon chain or heterocyclic ring [19]. Flavonoids can occur in two forms: Free - aglycons, or in the form of β-glycosides [combination of aglycone with the sugar part] [19]. Aglycones of most flavonoids are more hydrophobic than their glycosides [20]. They can penetrate the biological membranes of enterocytes as a result of passive transport [20]. The presence of a glycosidic substituent in the flavonoid molecule causes an increase in its mass and hydrophilicity, which limits its absorption as a result of diffusion [21]. Flavonoid compounds that have not been absorbed in the upper digestive tract reach the large intestine, where they are modified by bacterial enzymes [22]. These compounds can be metabolized by β-glucosidase, α-rhamnosidase, and β-galactosidase, synthesized by bacteria [23]. The resulting flavonoid aglycons can be absorbed in the large intestine or further metabolized [24]. As a result of metabolism, many low-molecular phenolic compounds, such as phenylacetic and phenylpropanoic acids, are formed [25]. Polyphenols commonly occur in plants; they are a significant ingredient of food. Fruits, especially berries, strawberries, raspberries, and chokeberry are abundant sources of them. Polyphenols occur in a fruit juice as well. Content in these products depends on the stage of manufacture. Juices made of freshly extruded fruits contain distinctly more polyphenols in comparison to manufactured concentrates [26]. Vegetables are a rich source of polyphenol compounds than fruits, mostly phenolic acids. Garlic, onion, broccoli, red cabbage, and red pepper pose the best source of them [27]. Green and black tea are rich in epicatechin, catechin, kaempferol, and quercetin [28]. Polyphenols’ antioxidant properties are connected to the presence of many hydroxyl groups, and thanks to this characteristic they protect plants from free radical actions. There are two mechanisms of polyphenol antioxidant action. Polyphenol reaction with radicals leads to the rise of a phenol radical, which due to a low reactivity does not cause any threat to the cells [29]. Antioxidant properties of polyphenol compounds result from the ability of metal chelation [30]. Transition metals (especially iron and copper) are elements present in many proteins and act out an important role in the functioning of the cells. Those metals are involved in Fenton’s reaction [31], which consists of two stages. The first one is the reaction between metal and hydrogen peroxide forming hydroxyl radical. Afterwards, the reduction of oxidized metal by superoxide anion takes place [32]. The arisen hydroxyl radical is a reactive form of oxygen that reacts with most of the compounds present in the cell e.g., protein, fatty acids, nucleic acids, or small metabolites [33]. Polyphenols inhibit the activity of oxidases, which catalyze the formation of water or hydrogen peroxide [34]. However, in specific situations, they can show prooxidase action via reduction of transition metals that allows catalysis of Fenton’s reaction (Figure 2) [35].
Table 1.
| Class | Subclass | Examples of Compounds | Source |
|---|---|---|---|
| Phenolic acids | Hydroxycinnamic acids |
Curcumin Caffeic acid Ferulic acid |
Fruit and cereals |
| Hydroxybenzoic acids | Gallic acid Protocatechuic acid Vanillic acid |
Onion, raspberry, blackberry, strawberry | |
| Favonoids |
Flavonols |
Kaempferol, Quercetin, Myricetin |
Onions, tea, lettuce, broccoli, apples |
|
Flavanones |
Naringenin, Hesperetin |
Oranges, grapefruits | |
|
Flavanols |
Gallocatechin Catechins |
Tea, red wine, chocolate | |
|
Isoflavones |
Genistein, Glycitein, Daidzein |
Soybeans, legumes | |
|
Anthocyanins |
Pelargonidin, Delphinidin, Malvidin |
Blackcurrant, strawberries, red wine, chokeberry | |
|
Flavones |
Apigenin, Luteolin, |
Parsley, celery, red pepper, lemon, thyme | |
| Stilbenes | Resvertrol | Red wine | |
| Lignans | Pinoresinol, Lariciresinol, Secoisolariciresinol, Sesamin |
Flax seed, sesame seed, red wine |
Figure 2.
Fenton’s cycle. 1. is broken down into and in the presence of 2. Reduction of to by superoxide anion radical.
3. The Influence of Polyphenol Compounds Contained in Tea on Intestinal Microflora.
Tea, one of the most popular drinks in the world, is high in polyphenols such as catechin, epicatechin, and quercetin. Moreover, it is known for its antitumor properties, especially in the case of large intestine cancer. Several studies tried to examine the influence of polyphenol compounds present in tea on bacteria in gastrointestinal tract. Tzounis et al. [38] examined the influence of monomers of flavan-3-ols like (-) epicatechin and (+) catechin on fecal bacterial growth (Table 2). The experiment was carried out in conditions that correspond to those present in the human intestine with the usage of two doses, respectively 150 mg and 1000 mg. Presence of (+) catechin stimulated the growth of Clostridium coccoides–Eubacterium rectale, Bifidobacterium spp., and Escherichia coli, but at the same time inhibited the growth of C. histolyticum. In comparison, (-) epicatechin stimulated only the growth of population of C. coccoides–Eubacterium rectale. Due to prebiotic properties, the consumption of food enriched form polyphenols contributes to gut integrity and intestinal homeostasis [39]. Growth of the group Clostridium coccoides–Eubacterium rectale is beneficial for the host due to increased production of short-chain fatty acids (SCFAs) used by the cells as a source of energy [40]. SCFAs formed by bacteria also inhibit proliferation of tumor cells and accelerate conversion of cholesterol into bile acids [41]. Growth of Bifidobacterium spp. was minor, but statistically significant as those bacteria inhibit proliferation of pathogens by production of organic acids [42]. Moreover, decrease in a population of C. histolyticum could be observed. Those bacteria due to the proteolytic properties can redound to the progression of colon cancer and development of inflammatory bowel diseases such as Crohn’s disease or ulcerative colitis. Therefore, decline in the number of C. histolyticum brings benefits for the host. The mechanism of this process is connected to degradation of proteins. Undigested proteins undergo the fermentation in the large intestine; the process is carried out by proteolytic bacteria (mainly Bacteroides and Propionibacterium) during which ammonia, thiols, amines, phenol, and indole compounds are formed. Additionally, Amines in the nitrosation reaction can form carcinogens such nitrosamines. Phenol and indole compounds negatively affect intestinal epithelium as they stimulate development of cancer by increasing the strength of carcinogenic factors [43,44]. In contrast to glycosides in food that do not seem to cause any harm in the host organism, flavonoid aglycones present mutagenic and toxic effects. The contradiction in the results of the influence of flavonoid aglycones on human health is connected with the transformation to compounds either working profitably or unfavorably for the host [45].
Table 2.
The effects of the intake of polyphenols on intestinal bacteria.
| Examined Polyphenols |
Dose | Duration | Type of Experiment | Subjects | Diet | Diseases | Medications Taken | Stimulation of Bacterial Growth | Inhibition of Bacterial Growth | Reference |
|---|---|---|---|---|---|---|---|---|---|---|
| (+) – catechin (-) - epicatechin |
150 mg/1000 mg | 48 h | In vitro | Escherichia coli Bifidobacterium spp. Group Clostridium coccoides- Eubacterium rectale |
Clostridium histolyticum | [38] | ||||
| Tea catechins | 300 mg | 6 weeks | In vivo [Open label pilot] |
31 females and 4 males from 66 to 98 years of age |
38.7 g proteins, 21.1 g lipids, 191 g carbohydrates, minerals, and vitamins. |
Hypertension, cerebral stroke, senile dementia |
None antibiotics |
Bifidobacterium Lactobacillus |
[46] | |
| (+) – epicatechin (-) – catechin 3-O-methylgallic acid Gallic acid Caffeic acid Phloretic acid 3-Phenylpropionic acid 4-Hydroxyphenylacetic acid |
1 mg/mL |
24 h | In vitro | Clostridium spp., Bifidobacterium spp., Lactobacillus spp. |
Clostridium perfringens, Clostridium difficile, Escherichia coli, Salmonella, Staphylococcus spp.. |
[47] | ||||
| Extract from grapes’ seeds containing proanthocyanidins | 0,19 g 0,38 g |
2 weeks | In vivo [Open label pilot] |
9 healthy adults from 37 to 42 years of age 8 elderly inpatients from 67 to 98 years of age |
None red wine, green tea, products prepared using lactic bacteria 62,5±4,7 g/d proteins, 38,0 ± 4,7 g/d lipids, 212,0 ± 15,0 g/d carbohydrate 9,4 ± 1,3 g/d sodium, 803,9 ± 105,8 mg/d moisture, 568,1 ± 89,9 mg/d calcium |
Cerebral stroke, bone fracture, senile dementia, articular rheumatism |
None antibiotics None antibiotics |
Bifidobacterium spp. |
Enterobacteriaceae | [48] |
| Cocoa flavonoids | 23 mg 494 mg |
4 weeks | In vivo (Randomized, double-blind, placebo- controlled) |
22 healthy volunteers (12 male and 10 female - premenopausal ) from 18 to 50 years of age | The patients did not change their dietary habits |
None antibiotics |
Bifidobacterium spp. Lactobacillus spp. |
Clostridium spp. | [50] | |
| Polyphenols in wine | 272 mL/d | 4 weeks | In vivo (Randomized, crossover, controlled, intervention) |
10 healthy adult men from 45–50 years of age | The patients did not change their dietary habits None alcohol and red wine |
None antibiotics and supplements |
Enterococcus, Prevotella, Bacteroides, Bifidobacterium, Bacteroides uniformis, Eggerthella lenta, Blautia coccoides–Eubacterium rectale group |
[52] | ||
| Polyphenols in wine | 272 mL/d | 30 days | In vivo (Randomized, crossover, controlled intervention) |
20 adult men (10 obese and 10 healthy) from 45 to 50 years old: |
The American Heart Association dietary guidelines | None antibiotics, prebiotic, probiotics, symbiotics, vitamin supplements and any medical treatment influencing intestinal microbiota |
Bifidobacterium spp. Lactobacillus spp. |
Enterobacter cloacae Escherichia coli |
[56] | |
| Resveratrol Epigallocatechin-3-gallate |
80 mg/d 282 mg/d |
12 week | In vivo (Randomized, double-blind, placebo- controlled) |
18 males and 19 females from 20 to 50 years of age | < 600 mg caffeine, < 3 cups green tea, < 20 g alcohol | Overweight and obese | None antibiotics, medication, supplements | Bacteroidetes | [57] | |
| Anthocyanins Gallic acid |
24 h | In vitro | Bifidobacterium spp. Lactobacillus spp. |
Clostridium histolyticum | [64] | |||||
| Quercetin | 12.5, 25, 50, 75μg/mL |
24 h | In vitro | Bifidobacterium catenulatum, Enterococcus caccae | [73] | |||||
| Naringenin, Naringin, Hesperetin, Hesperidin, Rutin, Quercetin Catechin |
20, 100, 250 μg/mL* * [for quercetin 4, 20, 50 μg/mL] |
24 h | In vitro | Lactic acid bacteria | [74] | |||||
| Grapes’ polyphenols | 0.25, 0.5, 1 mg/mL | 24 h | In vitro | Bifidobacterium spp. Lactobacillus spp. |
[75] | |||||
| Extracts from: blackcurrant blueberry cranberry cloudberry lingonberry raspberry berry of sea-buckthorn strawberry Polyphenols: Apigenin, Caffeic acid, (+) - Catechin, Chlorogenic acid, Coumarin-3-carboxylic acid, Cyanidin chloride, Delphinidin chloride, Ferulic acid, Isoquercetin, Kaempferol, Cyanidin-3-O-glucoside, Luteolin, Myricetin, Pelargonidin chloride, Quercetin dihydrate, Rutin trihydrate, Trans-cinnamic acid |
0,5, 1, 5 mg/mL |
24 h | In vitro | Lactobacillus spp. | [76] |
In Goto et al.’s study [46], the polyphenols present in tea has a positive effect on the growth of Bifidobacterium spp.. Decline in the number of putrefactive bacteria like Enterobacteriaceae spp. and Clostridium spp. was noticeable as well. The consumption of tea evidently improved conditions in the intestines by lowering the levels of sulfides, ammonia, and pH.
In Lee et al.’s experiment [47], the main aim was to examine the effect of polyphenols and their aromatic metabolites on bacterial growth. A significant inhibition of the growth of Clostridium perfringens, Clostridium difficile, E. coli, Salmonella, and Staphylococcus spp was observed. The examined compounds were also inhibited, but in a smaller extent. The growth of probiotic bacteria e.g., Clostridium spp., Bifidobacterium spp., Lactobacillus spp was noticed.
4. The Influence of Polyphenols Present in Plant Extracts on Microbiota
Plant extracts form a large class of compounds with multi-directional effects on human health. Their properties are the sum of individual functions of complexes included in a specific extract. Commonly, together with polyphenols, other antioxidants like vitamin C, E, and carotene are present. Those substances have protective properties towards different tissues. The pharmaceutic industry offers abundant numbers of products containing distinct plant extracts used as a prevention or treatment in conditions like cardiovascular disease, liver dysfunction, and to mitigate symptoms of menopause.
Yamakoshi et al. [48] studied the effect of grapes’ seeds extract that contained, respectively, 89.3% and 38.5% proanthocyanidins on intestinal bacteria. The beverage was administered to two groups for two weeks. The number of Bifidobacterium increased when at the same time, the colony of Enterobacteriaceae decreased. Bifidobacterium as bacteria exhibit positive effects on human health through the formation of SCFAs, but also by lowering the pH and inhibiting the development of putrefactive bacteria.
Since intestinal microflora is very sensitive and easily detects changes in external conditions, it is of high importance to supplement with substances such as prebiotics (non-absorbable compounds) that stimulate growth of one or few groups of beneficial bacteria in colon [49].
Tzounis et al. [50] evaluated the influence of flavonols derived from cocoa with the potential of being used as prebiotics. Patients were divided into two groups where the first one obtained 494 mg of cocoa flavonols per day and the other group was given 23 mg of the same flavonols daily for the period of four weeks. Researchers determined a statistically significant growth of Bifidobacterium and Lactobacillus. Both types of bacteria inhibit development of pathogenic microorganisms. Moreover, studied extract affected the population of C. histolyticum by relevantly decreasing it. Cocoa-derived flavonols can also inhibit the growth of Clostridium perfringens that might contribute to progression of colon cancer and participate in arising of inflammatory bowel disease. The change in intestinal microflora was connected to a change in the level of triglycerides in patients’ blood. Therefore, the study confirms the possibility of applying the polyphenols as a potential prebiotic compound.
5. The Effect of Polyphenols Contained in Wine on Intestinal Microflora
Red wine is a source of multiple polyphenol compounds e.g., catechin, proanthocyanidins, stilbenes, anthocyanins, and flavonols that exhibit cardioprotective and antioxidant properties. Despite many studies on the mechanism of action of red wine, the contribution of individual components was not unequivocally determined. Compounds that potentially have the greatest meaning are resveratrol, phenolic acids, and proanthocyanidins. The positive effect of red wine on the gut microbiota is due to prebiotic activity and inhibition of pathogen growth [51].
Queipo-Ortuno et al. [52] analyzed the effect of consumption of red wine on intestinal microbiota. For four weeks, they studied the influence of wine (272 mL), non-alcoholic wine (272 mL), and alcohol (250 mL). There was a significant growth of Enterococcus, Prevotella, Bifidobacterium, Bacteroides uniformis, Eggerthella lenta, and group Blautia coccoides–Eubacterium rectale in patients receiving red wine. Simultaneously, during this study, blood pressure measurements and lipid concentration tests were carried out on patients. The obtained results showed some beneficial effects of polyphenols present in wine on circulatory system and lipid profile. Not only systolic and diastolic blood pressure, but also the concentration of triglycerides, total cholesterol, and HDL cholesterol significantly declined. Those changes could be associated with the growth of a species Bifidobacterium spp. Bifidobacteria can produce short-chain fatty acids capable of reducing cholesterol synthesis in the liver or can bind bile acids in the digestive tract [53]. The obtained results present some potential advantages of the usage of red wine polyphenols as a prebiotic source. The experiment did not detect any effect of wine or alcohol on the growth of Lactobacillus spp., while consumption of alcohol ensued from increase in proliferation of Bacteroides spp. and Clostridium spp. Additionally, the disappearance of Prevotellaceae spp. population in comparison with red wine and non-alcoholic wine occurred. The concentration of uric acid considerably decreased when patients received red wine possibly as a result of the growth of Proteobacteria that degrade the mentioned compound [54]. Lowering of CRP concentration after non-alcoholic and red wine period might be connected to the increase in the number of Bifidobacterium spp., and the change in the recalled parameter can indicate an advantageous effect of wine polyphenols on cardiovascular system [55].
A similar study was carried by Moreno-Indias et al. [56] where researchers examined prebiotic effect of red wine on intestinal microbiota and reduction of metabolic syndrome markers in obese patients. In this study, red wine (272 mL) and non-alcoholic wine (272 mL) were tested. After the period of red and non-alcoholic wine consumption, in patients with a metabolic syndrome, scientists observed a relevant increase in the number of Fusobacterium and Bacteroidetes while at the same time, a significant decline in the population of Firmicutes was noticed. Likewise, in the group of healthy subjects, a statistically important growth of Bacteroidetes in comparison with the initial value was noticeable. In the case of patients with dysmetabolic syndrome X, during red as well as non-alcoholic wine consumption, researchers registered decline in Clostridium and Clostridium histolyticum species that are within Firmicutes phylum. At the same time, a growth of bacteria in the group of Blautia coccoides–Eubacterium rectale, Faecalibacterium prausnitzii, Roseburia, and Lactobacillus was noticed. In healthy patients, scientists observed an increase in the number of Faecalibacterium prausnitzii and Roseburia.
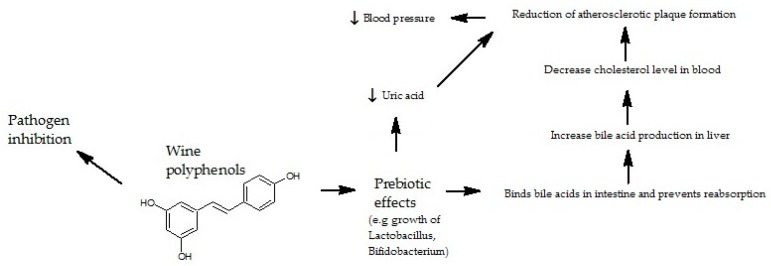
In the study conducted on obese women and men, a significant decrease in the number of Bacteroidetes and a tendency to reduce the number of Faecalibacterium prausnitzii in men was noted. These changes were not observed in women. The differences may be due to hormonal changes [57]. Faecalibacterium prausnitzii belongs to the group of important SCFA producers [58]. A decrease in the number of these bacteria is observed in patients with inflammatory bowel disease [IBD] [59]. The most common diseases from this group are Leśniowski-Crohn’ s disease and ulcerative colitis [59]. In IBD, the excessive activation of the immune system is increased by pathogenic bacteria [60]. Additionally, SCFA-inducing interleukin 10 [IL-10] reduces inflammatory reactions [60]. Akkermansia muciniphila is an important factor modulating F. prausnitzii count. A. muciniphila degrades mucus components to propionic acid, acetic acid, and oligosaccharides, which are used by F. prausnitzii [61]. Numerous clinical studies have investigated the benefits of polyphenol supplementation (anthocyanins, epigallocatechin gallate, resveratrol, and curcumin) in patients with IBD. The improvement of patients’ health resulted from multidirectional actions such as regulation of cytokine concentration, antioxidant enzymes, and modification of intestinal microflora [62,63]. Anthocyanins are natural pigments widespread in plant kingdom and are present in flowers, fruit peel, or in wine where they impart the color. Hidalgo et al. [64] determined the influence of mixture of anthocyanins, 3-glucoside of malvidin (oenin), and gallic acid on bacterial growth. The use of oenin on microorganisms caused the increase in the total number of bacteria. Scientists demonstrated the growth in population of C. coccoides–Eubacterium rectale that is responsible for production of SCFA. The usage of mixture of anthocyanins of low concentration on studied bacteria had a positive correlation with the growth of Bifidobacterium spp. and Lactobacillus spp. but did not affect C. coccoides-Eubacterium rectale and C. histolyticum populations. Gallic acid used in the experiment inhibited the growth of C. histolyticum without any negative consequences on the beneficial bacteria population in intestines. Moreover, it considerably increased the total number of bacteria and specific species called Atopobium spp., which is significant in gastrointestinal tract as Atopobium minutum induces the process of apoptosis of colon cancer cells. Therefore, the growth of this bacteria suggests a positive effect on human health [65]. Anthocyanins and their metabolites present in large intestine, increase the number of Lactobacillus spp. and Bifidobacterium spp. These bacteria have an ability to metabolize phenol complexes during augmentation, supplying energy to the cells and enriching the environment for other bacteria. Hence, anthocyanins, mainly the mixture of anthocyanins derived from grapes, might function as a stimulator for growth of Lactobacillus spp. and Bifidobacterium spp. that have a positive effect on large intestine. The increase in number of lactic acid bacteria in colon reduces the creation of procarcinogens in the large intestine, and lowers pH and cholesterol levels in patients (Figure 3).
Figure 3.
Interaction between wine polyphenols and gut microbiota.
6. Polyphenols’ Negative Impact on Microflora of Intestines
Polyphenols act positively on a composition of intestines’ microflora and have a beneficial impact on human health. The impact of polyphenols on bacterial growth depends on the dose, structure, as well as phylum of bacteria [66]. Gram-negative are more resistant to polyphenols than gram-positive due to the differences in the cell wall structure [67]. The antibacterial effect is connected with various mechanisms, for instance cell wall damaging [68], H2O2 production [69], or changes in permeability of cell membrane [70]. Polyphenol compounds might derange quorum sensing [71]. Another hypothesis envisages the creation of metal-polyphenol compounds. The lack of iron suppresses the growth of sensitive bacteria, mostly aerobic microbes [72]. In Firmann et al.’s study [73] the influence of quercetin on intestinal commensal bacteria such as Ruminococcus gauvreauii, Bifidobacterium catenulatum and Enterococcus caccae, was evaluated. Outcome of studies show that quercetin does not influence the growth of Ruminococcus gauvreauii but mildly hinder the growth of Bifidobacterium catenulatum and Enterococcus caccae. The study held by Duda-Chodak [74] ascertained negative effect of aglycons (naringenin, hesperetin, quercetin, and catechin) and glycosides (naringin, hesperidin, and rutin) on intestinal microbiota represented by Bacteroides galacturonicus, Lactobacillus spp., Enterococcus caccae, Bifidobacterium catenulatum, Ruminococcus gauvreauii, and Escherichia coli. In this experiment, polyphenols in concentrations of 20, 100, and 250 μg/mL diluted in growth medium were used(for quercetin the concentrations were 4, 20, and 50 μg/mL). The examined aglycones presented inhibiting properties towards investigated bacteria (dependent on the concentration of the substance in microbiological medium) while glycosides did not exhibit any effect on intestinal microflora. Catechins did not affect the growth of microorganisms. Only the growth of B. catenulatum was slightly slowed down while the growth of E. caccae was stimulated by higher concentrations of catechins.
The study carried by Tabasco et al. [75] evaluated the influence of grapes’ polyphenols on the growth of lactic acid bacteria and Bifidobacterium with the usage of three extracts of concentrations 0.25, 0.5, and 1 mg/mL, respectively. The experiment showed a dose-dependent inhibition on growth of above-mentioned bacteria.
Puupponen-Pimia et al. [76] determined the effect of pure polyphenols and polyphenols contained in the extract from blueberries on the probiotic bacteria, Escherichia coli and Salmonella. Lactic acid bacteria were more resistant to pure polyphenol compounds than other examined microorganisms, except myricetin, which inhibited the growth of all lactic acid bacteria with no effect on the growth of Salmonella. Phenolic acids in high concentration mainly inhibited the growth of Gram-negative bacteria. The extracts showed the growth-inhibiting effect, mostly towards Gram-negative bacteria These results indicate that the inhibitory effect of the extracts is due to the synergistic action of polyphenols.
7. Conclusions
Reviewed studies present complex interactions between polyphenol compounds and intestinal microorganism with both positive and negative consequences. The presented studies examined the influence of polyphenols on gut’s microbiota in vivo and in vitro. Clinical studies, due to their design, do not give unequivocal answers. The first major limitation of the research is the number of individuals. A study group that is too small may not be enough to notice subtle changes. Another limitation is the short duration of the test. Making a comparison of the results between studies is often difficult due to differences in the accuracy of microorganism identification; some studies indicate changes at the phylum level, and others at the level of the species. In addition, the lack of clear information about the diet and lifestyle of patients means that caution should be exercised when extrapolating the results obtained. The in vitro studies were performed only on monobacterial cultures. As previously mentioned, intestinal microbiome is one of the most complex ecosystems on Earth. Biotransformation of polyphenol complexes by bacteria is still a subject of discussion and requires further studies. In respective stages, different microorganisms are responsible for transformation of polyphenols. Therefore, a lack of knowledge on this subject limits the analysis of potential benefits for human. Due to the limitations of the above-mentioned studies, further experiments are essential to examine the contribution of particular bacteria on different stages of biotransformation of polyphenol compounds and the consequences for human’s health.
Author Contributions
M.W. Research concept and design; J.G. Writing the article; E.M. Data analysis and interpretation; M.P. Collection and assembly of data; M.Ś. Critical revision of the article; P.S. Critical revision of the article; E.S. Writing the article; B.M. Data analysis and interpretation. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Conflicts of Interest
The authors declare no conflict of interest.
References
- 1.Bibbo S., Ianiro G., Giorgio V., Scaldaferri F., Masucci L., Gasbarrini A., Cammarota G. The role of diet on gut microbiota composition. Eur. Rev. Med. Pharmacol. Sci. 2016;20:4742–4749. [PubMed] [Google Scholar]
- 2.Nicholson J., Holmes E., Kinross J., Burcelin R., Gibson G., Jia W., Pettersson S. Host-Gut Microbiota Metabolic Interactions. Science. 2012;336:1262–1267. doi: 10.1126/science.1223813. [DOI] [PubMed] [Google Scholar]
- 3.Zeng H., Umar S., Rust B., Lazarova D., Bordonaro M. Secondary Bile Acids and Short Chain Fatty Acids in the Colon: A Focus on Colonic Microbiome, Cell Proliferation, Inflammation, and Cancer. Int. J. Mol. Sci. 2019;20:1214. doi: 10.3390/ijms20051214. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Raman M., Ambalam P., Doble M. Probiotics and Bioactive Carbohydrates in Colon Cancer Management. Springer; New Delhi, India: 2016. Probiotics and Bioactive Carbohydrates in Colon Cancer Management. [Google Scholar]
- 5.Tremaroli V., Bäckhed F. Functional interactions between the gut microbiota and host metabolism. Nature. 2012;489:242–249. doi: 10.1038/nature11552. [DOI] [PubMed] [Google Scholar]
- 6.Hooper L.V., Littman D.R., MacPherson A.J. Interactions Between the Microbiota and the Immune System. Science. 2012;336:1268–1273. doi: 10.1126/science.1223490. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Mandal P. Molecular mechanistic pathway of colorectal carcinogenesis associated with intestinal microbiota. Anaerobe. 2018;49:63–70. doi: 10.1016/j.anaerobe.2017.12.008. [DOI] [PubMed] [Google Scholar]
- 8.Turnbaugh P.J., Ridaura V.K., Faith J.J., Rey F.E., Knight R., Gordon J.I. The Effect of Diet on the Human Gut Microbiome: A Metagenomic Analysis in Humanized Gnotobiotic Mice. Sci. Transl. Med. 2009;1:6ra14. doi: 10.1126/scitranslmed.3000322. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Modi S.R., Collins J.J., Relman D.A. Antibiotics and the gut microbiota. J. Clin. Investig. 2014;124:4212–4218. doi: 10.1172/JCI72333. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Reynolds M.A., Prudencio A., Aichelmann-Reidy M.E., Woodward K., Uhrich K. Non-Steroidal Anti-inflammatory Drug (NSAID)-Derived Poly(anhydrideesters) in Bone and Periodontal Regeneration. Curr. Drug Deliv. 2007;4:233–239. doi: 10.2174/156720107781023866. [DOI] [PubMed] [Google Scholar]
- 11.Hirata S., Kunisawa J. Gut microbiome, metabolome, and allergic diseases. Allergol Int. 2017;66:523–528. doi: 10.1016/j.alit.2017.06.008. [DOI] [PubMed] [Google Scholar]
- 12.Cenit M.C., Matzaraki V., Tigchelaar E., Zhernakova A. Rapidly expanding knowledge on the role of the gut microbiome in health and disease. Biochim. et Biophys. Acta (BBA) Mol. Basis Dis. 2014;1842:1981–1992. doi: 10.1016/j.bbadis.2014.05.023. [DOI] [PubMed] [Google Scholar]
- 13.Oriach C.S., Robertson R.C., Stanton C., Cryan J.F., Dinan T.G. Food for thought: The role of nutrition in the microbiota-gut–brain axis. Clin. Nutr. Exp. 2016;6:25–38. doi: 10.1016/j.yclnex.2016.01.003. [DOI] [Google Scholar]
- 14.Sharon G., Sampson T.R., Geschwind D.H., Mazmanian S.K. The Central Nervous System and the Gut Microbiome. Cell. 2016;167:915–932. doi: 10.1016/j.cell.2016.10.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Martinez J.A., Ramos-Lopez O., Riezu-Boj J.-I., Milagro F.I., Martinez J.A. Diet, Gut Microbiota, and Obesity: Links with Host Genetics and Epigenetics and Potential Applications. Adv. Nutr. 2019;10:S17–S30. doi: 10.1093/advances/nmy078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ozdal T., Sela D., Ulrih N.P., Boyacioglu D., Chen F., Capanoglu E. The Reciprocal Interactions between Polyphenols and Gut Microbiota and Effects on Bioaccessibility. Nutrients. 2016;8:78. doi: 10.3390/nu8020078. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Tsao R. Chemistry and Biochemistry of Dietary Polyphenols. Nutrients. 2010;2:1231–1246. doi: 10.3390/nu2121231. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Colomer R., Sarrats A., Lupu R., Puig T. Natural Polyphenols and their Synthetic Analogs as Emerging Anticancer Agents. Curr. Drug Targets. 2017;18:147–159. doi: 10.2174/1389450117666160112113930. [DOI] [PubMed] [Google Scholar]
- 19.Wang T.-Y., Li Q., Bi K. Bioactive flavonoids in medicinal plants: Structure, activity and biological fate. Asian J. Pharm. Sci. 2018;13:12–23. doi: 10.1016/j.ajps.2017.08.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Farhadi F., Khameneh B., Iranshahi M., Iranshahy M. Antibacterial activity of flavonoids and their structure-activity relationship: An update review. Phytother. Res. 2018;33:13–40. doi: 10.1002/ptr.6208. [DOI] [PubMed] [Google Scholar]
- 21.Fang Y., Liang F., Liu K., Qaiser S., Pan S., Xu X. Structure characteristics for intestinal uptake of flavonoids in Caco-2 cells. Food Res. Int. 2017;105:353–360. doi: 10.1016/j.foodres.2017.11.045. [DOI] [PubMed] [Google Scholar]
- 22.Murota K., Nakamura Y., Uehara M. Flavonoid metabolism: The interaction of metabolites and gut microbiota. Biosci. Biotechnol. Biochem. 2018;82:600–610. doi: 10.1080/09168451.2018.1444467. [DOI] [PubMed] [Google Scholar]
- 23.Oteiza P.I., Fraga C., Mills D.A., Taft D. Flavonoids and the gastrointestinal tract: Local and systemic effects. Mol. Asp. Med. 2018;61:41–49. doi: 10.1016/j.mam.2018.01.001. [DOI] [PubMed] [Google Scholar]
- 24.Rowland I., Gibson G., Heinken A., Scott K., Swann J., Thiele I., Tuohy K. Gut microbiota functions: Metabolism of nutrients and other food components. Eur. J. Nutr. 2018;57:1–24. doi: 10.1007/s00394-017-1445-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Tomás-Barberán F.A., Selma M.V., Espín J.C. Interactions of gut microbiota with dietary polyphenols and consequences to human health. Curr. Opin. Clin. Nutr. Metab. Care. 2016;19:471–476. doi: 10.1097/MCO.0000000000000314. [DOI] [PubMed] [Google Scholar]
- 26.Joseph S.V., Edirisinghe I., Burton-Freeman B.M. Fruit Polyphenols: A Review of Anti-inflammatory Effects in Humans. Crit. Rev. Food Sci. Nutr. 2015;56:419–444. doi: 10.1080/10408398.2013.767221. [DOI] [PubMed] [Google Scholar]
- 27.Pérez-Jiménez J., Neveu V., Voß F., Scalbert A. Identification of the 100 richest dietary sources of polyphenols: An application of the Phenol-Explorer database. Eur. J. Clin. Nutr. 2010;64:S112–S120. doi: 10.1038/ejcn.2010.221. [DOI] [PubMed] [Google Scholar]
- 28.Khan N., Mukhtar H. Tea polyphenols for health promotion. Life Sci. 2007;81:519–533. doi: 10.1016/j.lfs.2007.06.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Leopoldini M., Nino R., Toscano M. The molecular basis of working mechanism of natural polyphenolic antioxidants. Food Chem. 2011;125:288–306. doi: 10.1016/j.foodchem.2010.08.012. [DOI] [Google Scholar]
- 30.Wang T., Jonsdottir R., Olafsdottir G. Total phenolic compounds, radical scavenging and metal chelation of extracts from Icelandic seaweeds. Food Chem. 2009;116:240–248. doi: 10.1016/j.foodchem.2009.02.041. [DOI] [Google Scholar]
- 31.Brillas E., Sirés I., Oturan M.A. Electro-Fenton Process and Related Electrochemical Technologies Based on Fenton’s Reaction Chemistry. Chem. Rev. 2009;109:6570–6631. doi: 10.1021/cr900136g. [DOI] [PubMed] [Google Scholar]
- 32.Trujillo D., Font X., Sanchez A. Use of Fenton reaction for the treatment of leachate from composting of different wastes. J. Hazard. Mater. 2006;138:201–204. doi: 10.1016/j.jhazmat.2006.05.053. [DOI] [PubMed] [Google Scholar]
- 33.Ajiboye T.O., Aliyu M., Isiaka I., Haliru F., Ibitoye O., Uwazie J., Muritala H., Bello S., Yusuf I., Mohammed A. Contribution of reactive oxygen species to (+)-catechin-mediated bacterial lethality. Chem. Interactions. 2016;258:276–287. doi: 10.1016/j.cbi.2016.09.010. [DOI] [PubMed] [Google Scholar]
- 34.Zhang H., Tsao R. Dietary polyphenols, oxidative stress and antioxidant and anti-inflammatory effects. Curr. Opin. Food Sci. 2016;8:33–42. doi: 10.1016/j.cofs.2016.02.002. [DOI] [Google Scholar]
- 35.Zhou Y., Zheng J., Li Y., Xu N.-P., Li S., Chen Y.-M., Li H.-B. Natural Polyphenols for Prevention and Treatment of Cancer. Nutrients. 2016;8:515. doi: 10.3390/nu8080515. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Rothwell J.A., Knaze V., Zamora-Ros R. Polyphenols: Dietary assessment and role in the prevention of cancers. Curr. Opin. Clin. Nutr. Metab. Care. 2017;20:512–521. doi: 10.1097/MCO.0000000000000424. [DOI] [PubMed] [Google Scholar]
- 37.Lecci R.M., Logrieco A., Leone A. Pro-oxidative action of polyphenols as action mechanism for their pro-apoptotic activity. Anti-Cancer Agents Med. Chem. 2014;14:1363–1375. doi: 10.2174/1871520614666140922121014. [DOI] [PubMed] [Google Scholar]
- 38.Tzounis X., Vulevic J., Kuhnle G., George T., Leonczak J., Gibson G.R., Kwik-Uribe C., Spencer J.P. Flavanol monomer-induced changes to the human faecal microflora. Br. J. Nutr. 2007;99:782–792. doi: 10.1017/S0007114507853384. [DOI] [PubMed] [Google Scholar]
- 39.Shortt C., Hasselwander O., Meynier A., Nauta A., Fernández E.N., Putz P., Rowland I., Swann J., Türk J., Vermeiren J., et al. Systematic review of the effects of the intestinal microbiota on selected nutrients and non-nutrients. Eur. J. Nutr. 2017;57:25–49. doi: 10.1007/s00394-017-1546-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Koh A., De Vadder F., Kovatcheva-Datchary P., Bäckhed F. From Dietary Fiber to Host Physiology: Short-Chain Fatty Acids as Key Bacterial Metabolites. Cell. 2016;165:1332–1345. doi: 10.1016/j.cell.2016.05.041. [DOI] [PubMed] [Google Scholar]
- 41.Wilson M. Microbial Inhabitants of Humans. Cambridge University Press; Cambridge, UK: 2005. Short-chain fatty acids; pp. 388–390. [Google Scholar]
- 42.Redondo N., Gheorghe A., Díaz L.E., Villavisencio B., Marcos A., Nova E. Associations of Probiotic Fermented Milk (PFM) and Yogurt Consumption with Bifidobacterium and Lactobacillus Components of the Gut Microbiota in Healthy Adults. Nutrients. 2019;11:651. doi: 10.3390/nu11030651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Ma N., Tian Y., Wu Y., Ma X. Contributions of the Interaction between Dietary Protein and Gut Microbiota to Intestinal Health. Curr. Protein Pept. Sci. 2017;18:1. doi: 10.2174/1389203718666170216153505. [DOI] [PubMed] [Google Scholar]
- 44.Zhao J., Zhang X., Liu H., Brown M.A., Qiao S. Dietary Protein and Gut Microbiota Composition and Function. Curr. Protein Pept. Sci. 2019;20:145–154. doi: 10.2174/1389203719666180514145437. [DOI] [PubMed] [Google Scholar]
- 45.Nowak A., Lubudisz Z. Karcynogenna aktywność mikroorganizmów jelitowych. Żywność Nauka Technologia Jakość. 2008;6:25–39. [Google Scholar]
- 46.Goto K., Kanaya S., Nishikawa T., Hara H., Terada A., Ishigami T., Hara Y. The effects of tea catechins on fecal flora of elderly residents in long-term care facilities. J. Nutr. Sci. Vitaminol. (Tokyo). 1999;45:135–141. doi: 10.3177/jnsv.45.135. [DOI] [PubMed] [Google Scholar]
- 47.Lee H.C., Jenner A.M., Low C.S., Lee Y.-K. Effect of tea phenolics and their aromatic fecal bacterial metabolites on intestinal microbiota. Res. Microbiol. 2006;157:876–884. doi: 10.1016/j.resmic.2006.07.004. [DOI] [PubMed] [Google Scholar]
- 48.Yamakoshi J., Tokutake S., Kikuchi M. Effect of Proanthocyanidin-Rich Extract from Grape Seeds on Human Fecal Flora and Fecal Odor. Microb Ecol Health Dis. 2001;13:25–31. [Google Scholar]
- 49.Lee E.-S., Song E.-J., Nam Y.-D., Lee S.-Y. Probiotics in human health and disease: From nutribiotics to pharmabiotics. J. Microbiol. 2018;56:773–782. doi: 10.1007/s12275-018-8293-y. [DOI] [PubMed] [Google Scholar]
- 50.Tzounis X., Rodriguez-Mateos A., Vulevic J., Gibson G.R., Kwik-Uribe C., Spencer J.P. Prebiotic evaluation of cocoa-derived flavanols in healthy humans by using a randomized, controlled, double-blind, crossover intervention study. Am. J. Clin. Nutr. 2010;93:62–72. doi: 10.3945/ajcn.110.000075. [DOI] [PubMed] [Google Scholar]
- 51.Requena T., Monagas M., Pozo-Bayon M.A., Martin-Alvarez P., Bartolomé B., Del Campo R., Ávila M., Martínez-Cuesta M.C., Peláez C., Moreno-Arribas M. Perspectives of the potential implications of wine polyphenols on human oral and gut microbiota. Trends Food Sci. Technol. 2010;21:332–344. doi: 10.1016/j.tifs.2010.04.004. [DOI] [Google Scholar]
- 52.Ortuño M.I.Q., Boto-Ordóñez M., Murri M., Gómez-Zumaquero J.M., Clemente-Postigo M., Estruch R., Diaz F.C., Andres-Lacueva C., Tinahones F.J. Influence of red wine polyphenols and ethanol on the gut microbiota ecology and biochemical biomarkers. Am. J. Clin. Nutr. 2012;95:1323–1334. doi: 10.3945/ajcn.111.027847. [DOI] [PubMed] [Google Scholar]
- 53.Melanie H., Susilowati A., Maryati Y. Fermented inulin hydrolysate by Bifidobacterium breve as cholesterol binder in functional food application. Int. Symp. Appl. Chem. 2016;1803:259–266. [Google Scholar]
- 54.Guo Z., Zhang J., Wang Z., Ang K.Y., Huang S., Hou Q., Su X., Qiao J., Zheng Y., Wang L., et al. Intestinal Microbiota Distinguish Gout Patients from Healthy Humans. Sci. Rep. 2016;6:20602. doi: 10.1038/srep20602. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Melander O., Newton-Cheh C., Almgren P., Hedblad B., Berglund G., Engström G., Persson M., Smith J.G., Magnusson M., Christensson A., et al. Novel and Conventional Biomarkers for Prediction of Incident Cardiovascular Events in the Community. JAMA. 2009;302:49–57. doi: 10.1001/jama.2009.943. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Moreno-Indias I., Sanchez-Alcoholado L., Pérez-Martínez P., Andres-Lacueva C., Cardona F., Tinahones F.J., Ortuño M.I.Q. Red wine polyphenols modulate fecal microbiota and reduce markers of the metabolic syndrome in obese patients. Food Funct. 2016;7:1775–1787. doi: 10.1039/C5FO00886G. [DOI] [PubMed] [Google Scholar]
- 57.Most J., Penders J., Lucchesi M., Goossens G.H., E Blaak E. Gut microbiota composition in relation to the metabolic response to 12-week combined polyphenol supplementation in overweight men and women. Eur. J. Clin. Nutr. 2017;71:1040–1045. doi: 10.1038/ejcn.2017.89. [DOI] [PubMed] [Google Scholar]
- 58.Ferreira-Halder C.V., Faria A.V.S., Andrade S.S. Action and function of Faecalibacterium prausnitzii in health and disease. Best Pract. Res. Clin. Gastroenterol. 2017;31:643–648. doi: 10.1016/j.bpg.2017.09.011. [DOI] [PubMed] [Google Scholar]
- 59.Zhang M., Zhou L., Wang Y., Dorfman R.G., Tang D., Xu L., Pan Y., Zhou Q., Li Y., Yin Y., et al. Faecalibacterium prausnitzii produces butyrate to decrease c-Myc-related metabolism and Th17 differentiation by inhibiting histone deacetylase 3. Int. Immunol. 2019;31:499–514. doi: 10.1093/intimm/dxz022. [DOI] [PubMed] [Google Scholar]
- 60.Lopez-Siles M., Duncan S.H., Garcia-Gil J., Martinez-Medina M. Faecalibacterium prausnitzii: From microbiology to diagnostics and prognostics. ISME J. 2017;11:841–852. doi: 10.1038/ismej.2016.176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Lopez-Siles M., Enrich-Capó N., Aldeguer X., Sabat-Mir M., Duncan S.H., Garcia-Gil L.J., Martinez-Medina M. Alterations in the Abundance and Co-occurrence of Akkermansia muciniphila and Faecalibacterium prausnitzii in the Colonic Mucosa of Inflammatory Bowel Disease Subjects. Front. Microbiol. 2018;8:281. doi: 10.3389/fcimb.2018.00281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Biedermann L., Mwinyi J., Scharl M., Frei P., Zeitz J., Kullak- Ublick G.A., Vavricka S.R., Fried M., Weber A., Humpf H.U., et al. Bilberry ingestion improves disease activity in mild to moderate ulcerative colitis – An open pilot study. J. Crohns Colitis. 2013;7:271–279. doi: 10.1016/j.crohns.2012.07.010. [DOI] [PubMed] [Google Scholar]
- 63.Dryden G., Lam A., Beatty K., Qazzaz H.H., McClain C.J. A Pilot Study to Evaluate the Safety and Efficacy of an Oral Dose of (−)-Epigallocatechin-3-Gallate–Rich Polyphenon E in Patients With Mild to Moderate Ulcerative Colitis. Inflamm. Bowel Dis. 2013;19:1904–1912. doi: 10.1097/MIB.0b013e31828f5198. [DOI] [PubMed] [Google Scholar]
- 64.Hidalgo M., Concha M.J.O., Kolida S., Walton G.E., Kallithraka S., Spencer J.P.E., Gibson G.R., De Pascual-Teresa S. Metabolism of Anthocyanins by Human Gut Microflora and Their Influence on Gut Bacterial Growth. J. Agric. Food Chem. 2012;60:3882–3890. doi: 10.1021/jf3002153. [DOI] [PubMed] [Google Scholar]
- 65.Eslami M., Yousefi B., Kokhaei P., Hemati M., Nejad Z.R., Arabkari V., Namdar A. Importance of probiotics in the prevention and treatment of colorectal cancer. J. Cell. Physiol. 2019;234:17127–17143. doi: 10.1002/jcp.28473. [DOI] [PubMed] [Google Scholar]
- 66.Hervert-Hernández D., Goñi I. Dietary Polyphenols and Human Gut Microbiota: A Review. Food Rev. Int. 2011;27:154–169. doi: 10.1080/87559129.2010.535233. [DOI] [Google Scholar]
- 67.Puupponen-Pimiä R., Nohynek L., Hartmann-Schmidlin S., Kähkönen M., Heinonen M., Maatta-Riihinen K., Oksman-Caldentey K.-M. Berry phenolics selectively inhibit the growth of intestinal pathogens. J. Appl. Microbiol. 2005;98:991–1000. doi: 10.1111/j.1365-2672.2005.02547.x. [DOI] [PubMed] [Google Scholar]
- 68.Kemperman R., Bolca S., Roger L.C., Vaughan E.E. Novel approaches for analysing gut microbes and dietary polyphenols: Challenges and opportunities. Microbiol. 2010;156:3224–3231. doi: 10.1099/mic.0.042127-0. [DOI] [PubMed] [Google Scholar]
- 69.Anna V.S., Biondo E., Kolchinski E.M., Da Silva L.F.S., Correa A.P.F., Bach E., Brandelli A. Total Polyphenols, Antioxidant, Antimicrobial and Allelopathic Activities of Spend Coffee Ground Aqueous Extract. Waste Biomass Valorization. 2016;8:439–442. [Google Scholar]
- 70.Coppo E., Marchese A. Antibacterial activity of polyphenols. Curr. Pharm. Biotechnol. 2014;15:380–390. doi: 10.2174/138920101504140825121142. [DOI] [PubMed] [Google Scholar]
- 71.Nazzaro F., Fratianni F., Coppola R. Quorum Sensing and Phytochemicals. Int. J. Mol. Sci. 2013;14:12607–12619. doi: 10.3390/ijms140612607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Cardona F., Andres-Lacueva C., Tulipani S., Tinahones F.J., Ortuño M.I.Q. Benefits of polyphenols on gut microbiota and implications in human health. J. Nutr. Biochem. 2013;24:1415–1422. doi: 10.1016/j.jnutbio.2013.05.001. [DOI] [PubMed] [Google Scholar]
- 73.Firrman J., Liu L., Zhang L., Argoty G.A., Wang M., Tomasula P., Kobori M., Pontious S., Xiao W. The effect of quercetin on genetic expression of the commensal gut microbes Bifidobacterium catenulatum, Enterococcus caccae and Ruminococcus gauvreauii. Anaerobe. 2016;42:130–141. doi: 10.1016/j.anaerobe.2016.10.004. [DOI] [PubMed] [Google Scholar]
- 74.Duda-Chodak A. The inhibitory effect of polyphenols on human gut microbiota. J. Physiol. Pharmacol. Off. J. Pol. Physiol. Soc. 2012;63:497–503. [PubMed] [Google Scholar]
- 75.Tabasco R., Sánchez-Patán F., Monagas M., Bartolomé B., Moreno-Arribas M.V., Peláez C., Requena T. Effect of grape polyphenols on lactic acid bacteria and bifidobacteria growth: Resistance and metabolism. Food Microbiol. 2011;28:1345–1352. doi: 10.1016/j.fm.2011.06.005. [DOI] [PubMed] [Google Scholar]
- 76.Puupponen-Pimiä R., Nohynek L., Meier C., Kahkonen M., Heinonen M., Hopia A., Oksman-Caldentey K.-M. Antimicrobial properties of phenolic compounds from berries. J. Appl. Microbiol. 2001;90:494–507. doi: 10.1046/j.1365-2672.2001.01271.x. [DOI] [PubMed] [Google Scholar]