Abstract
Objective
Premenstrual syndrome (PMS) affects millions of women. While over-the-counter products have helped with the physical symptoms of PMS, emotional symptoms have been less well supported. The objective of this trial was to measure the effect of an oxaloacetate/vitamin C combination on the major emotional symptoms of PMS, including depression, anxiety, perceived stress, aggression, and suicidal ideation.
Methods
Forty-eight women experiencing PMS completed a baseline survey comprising the Beck's Depression Inventory, Cohen Perceived Stress Scale, Generalized Anxiety Disorder Test, and Buss-Perry Aggression Questionnaire. After baseline measurements, participants were randomly assigned to take either 2 capsules of 100 mg oxaloacetate/150 mg ascorbic acid, or 2 capsules of rice flour (placebo) for their entire menstrual cycle. At menstruation, the women completed the 4 surveys again. The women then switched capsules in a cross-over design and continued the study for an additional menstrual cycle. The final assessment was repeated at menstruation. Statistical analysis of the 4 surveys was performed to examine efficacy.
Results
Oxaloacetate/vitamin C supplementation during PMS significantly improved depression, perceived stress, anxiety, aggression, and suicidal ideation. The mean improvement in depression was 54.1%, 35.8% for perceived stress, 51.43% for generalized anxiety, and 17.8% for aggression. Suicidal ideation was reduced by 47.9%. All results were highly significant.
Conclusion
A combination of oxaloacetate and vitamin C supplementation helped to alleviate depression, anxiety, perceived stress, aggression, and suicidal ideation symptoms associated with PMS.
Trial Registration
ClinicalTrials.gov Identifier: NCT03509714
Keywords: Oxaloacetate, Premenstrual syndrome, Depression, Anxiety, Suicidal ideation
Introduction
Premenstrual syndrome (PMS) affects millions of women in a cyclical fashion, primarily 1 week before menstruation and can last from 1 to 10 days with each menstrual cycle. The effects can be segregated into physical effects, such as cramps, fatigue, acne and fluid retainage, emotional effects such as depression, anxiety, perceived stress, aggression, tension, lethargy, insomnia, and poor coordination in concentration, and mental disorders, such as the feeling of being reluctant toward oneself and being suppressed [1,2]. While there are many existing products for the physical effects of PMS, development of over-the-counter options for emotional symptoms has received less attention. Common complaints among those experiencing PMS include mood changes, depression, anxiety, perceived stress, and aggression.
The objective of this study was to examine emotional issues in PMS, and how they can be potentially improved with oral supplementation with a mix of the metabolite, “oxaloacetate” and vitamin C (ascorbic acid). Of particular interest was the treatment of the depression, anxiety, perceived stress, and aggression in PMS. These emotional states were selected due to the availability of validated measurement tools that could be used in a self-administered survey [3,4,5,6,7,8]. These validated measurement tools were modified for the subjects to only consider the 7 days before menstruation, to capture their effect on PMS in an on-line format that mirrors the Beck's Depression Inventory, Cohen's Perceived Stress, Generalized Anxiety Disorder, and Buss-Perry Aggression Scale surveys.
Oxaloacetate is an energy metabolite found in every cell of the human body. It holds a key place in the Krebs cycle within the mitochondria, is a critical early metabolite in gluconeogenesis, amino acid and fatty acid synthesis, and is found in the urea cycle. It is critical to human metabolism, proper cellular function, and is central to energy production and use in the body. In addition, oxaloacetate may affect emotional symptoms through multiple mechanisms. During PMS, there is a large increase in glucose utilization in the cerebellum of women who are affected by emotional mood swings [9]. The cerebellum plays key roles in motor control, attention, and regulating fear and pleasure responses [10]. Individuals with bipolar depression have been shown to exhibit an increased demand for glucose in the cerebellum, similar to patients with severe emotional PMS symptoms [11,12,13,14]. Shortages of glucose in the cerebellum during PMS, therefore, may be a causal factor in emotional issues. Women with emotional symptoms during PMS exhibit an increased desire for sugary/fatty foods such as chocolate [15]. While binge eating of sugar and fats brings some relief to the emotional symptoms of PMS [16,17], it can also cause worsening of these PMS-related symptoms after glucose levels in the body fall.
Oxaloacetate supplementation has been shown to support adequate glucose levels in the body [18,19]. In addition, oxaloacetate is a key metabolite in gluconeogenesis—the production of glucose when needed by the body [20]. An excess of oxaloacetate enables gluconeogenesis to occur on demand, thereby fueling the brain, and potentially meeting glucose needs in the cerebellum.
In addition to the ability of oxaloacetate to support proper glucose regulation, oxaloacetate affects 2 chemicals in the brain, gamma-amino butyric acid (GABA) and glutamate. Altering the GABA/glutamate ratio has been shown to be associated with other emotional conditions such as autism and schizophrenia spectra [21]. Clinical work has also tied GABA and glutamate concentrations to emotional processing [22]. The GABA/glutamate ratio may also play a major role in the emotional symptoms of PMS. Oxaloacetate supplementation can reduce glutamate levels in the brain via a process known as “glutamate scavenging” [23,24]. In addition, oxaloacetate supplementation has been shown to increase GABA levels in animal models [25]. By both lowering glutamate and increasing GABA levels, the GABA/glutamate ratio is altered, which may also help to alleviate emotional symptoms experienced by women during PMS.
Before the clinical trial, we noted multiple cases in which a mix of oxaloacetate/vitamin C supplementation resulted in a marked reduction in PMS-related aggression, depression, anxiety, and stress in women who attended our private practice. The combination of these individual clinical cases, and the functional ability of oxaloacetate to support proper glucose function and influence the GABA/glutamate ratio in the brain, which may exacerbate PMS, prompted us to conduct a formalized clinical trial to investigate the use of oxaloacetate supplementation to treat emotional symptoms during PMS. Due to the unstable nature of oxaloacetate, it was combined with ascorbic acid (vitamin C) to increase its stability at room temperature [26].
To our knowledge, this is the first report to describe the use of the metabolite oxaloacetate and vitamin C to significantly reduce emotional symptoms of depression, anxiety, perceived stress, and aggression in women experiencing PMS.
Materials and methods
Institutional review board approval was obtained for this study. All subjects were recruited and participated in the trial via the Internet. Subjects between 18 and 50 years of age, who believed they experienced emotional symptoms related to PMS (i.e., depression, perceived stress, general anxiety, or aggression), were selected for the trial. Women with diagnoses of premenstrual dysphoric disorder (PMDD) or general depression were specifically excluded because the United States Food & Drug Administration (US FDA) classifies these conditions as “diseases,” which are beyond the scope of nutritional supplement treatment. A total of 48 women were tested in 2 similar clinical trials.
Subjects completed the human consent form and subsequently participated in an on-line PMS Symptoms Questionnaire—a survey comprising the Beck's Depression Inventory, Cohen's Perceived Stress, Generalized Anxiety Disorder and Buss-Perry Aggression Scale—focused on the time that they were experiencing PMS symptoms, usually approximately one week before their period as a baseline measure. For part A of the study (26 women), subjects were randomly assigned in a double-blind manner: neither the subject nor the individual conducting the trial knew which product they received. Thirteen women received a one-month supply of the placebo first, and 13 received the active oxaloacetate/vitamin C combination first. They then switched treatments at their next menstruation, in a “cross-over” design. Participants were randomized as follows: they were assigned to either group, alternating to each group in the order in which they joined the study.
After the first 26 women completed the cross-over trial, the carry-over effect of the cross-over was evaluated. Statistical analysis suggested that the effect from the oxaloacetate/vitamin C mix, when administered first, carried over to the placebo period in the following month (P<0.05). Thus, a second trial was conducted (part B) using the same protocol as part A, with the only exception that in all subjects in whom the placebo was administered first to avoid a carryover effect. Twenty-two new women were tested in this fashion, bringing the total number of participants to 48.
The placebo consisted of 250 mg of encapsulated rice flour, and the active ingredients were 100 mg of oxaloacetate and 150 mg vitamin C (ascorbic acid). The oxaloacetate/vitamin C combination is a commercial dietary supplement mix that has been available for >10 years (Terra Biological LLC, San Diego, CA, USA). Vitamin C is added to oxaloacetate to increase its stability, which can quickly decarboxylate at room temperature [26]. Subjects were instructed to take 2 capsules daily with breakfast (or lunch if they forgot to take it with breakfast). They began taking the provided supplement at the end of their first menstrual cycle of the study. Subjects continued taking the supplement until their second menstrual cycle of the study began. Women in the study repeated the on-line questionnaire reflecting their emotional PMS symptoms before the next menstrual cycle. When menstruation ended, subjects began taking the supplement they were not provided before, in a cross-over clinical design. At the start of their next menstrual cycle, they completed the final PMS survey. Subjects were given a bonus of 12 bottles of active product for completing the entire study.
All outcome measures are expressed as mean and standard deviation. Graphical representations illustrate means, standard error bars, and statistical significance in all participants. Changes between assessment time points were calculated as percentage changes. A linear mixed effects model with subject specific random effects was used to analyze changes within and between treatment groups for the cross-over study. The carry over effect was assessed by evaluating the interaction between group and sequence order. All P-values were 2-sided and differences with P<0.05 were considered to be statistically significant. Excel (Microsoft Corporation, Redmond, WA, USA) was used to calculate P-values.
Results
A total of 48 women completed parts A and B of the study. Women who experienced mood issues in PMS but had not received a formal diagnosis of clinical depression nor PMDD were recruited. On joining the clinical trial, the women in the study were administered a series of surveys to assess depression, anxiety, perceived stress, and aggression. The average baseline results are presented in Table 1. The mean of all participants indicated that, as a group, the women had moderate depression and anxiety, with above average perceived stress and aggression [27,28,29,30].
Table 1. Baseline averages for women in the study using self-assessment surveys.
| Characteristics | Becks depression inventory | Cohen perceived stress | Generalized anxiety disorder | Buss-Perry aggression |
|---|---|---|---|---|
| Raw mean score | 27.8 | 25.8 | 14.1 | 86.3 |
| Standard deviation | 11.3 | 5.2 | 4.9 | 23.6 |
| Classification of group average at initial baseline | Moderate depression (note: a score of 14.5 is the cutoff for depression) [28] | Above average stress (average for women is 13.7) [29] | Moderate anxiety (note: a score of 15+ is the cutoff for “severe anxiety”) [30] | Above average aggression (average for women is 68.2) [31] |
Number of women in the study: 48. Average age of the women in the study: 37.
Initially, a clinical examination was conducted as a randomized cross-over study of oxaloacetate/vitamin C mix versus placebo in 26 subjects, in which subjects were double-blinded, randomized to receive the oxaloacetate/vitamin C mix first, followed by placebo, or vice-versa (they were randomized by the order in which they were enrolled) (part A participants). A second clinical study was conducted when evaluation of the “carry-over” effect indicated that if the oxaloacetate/vitamin C mix was administered first, there was also a statistically significant improvement in depression when the placebo was subsequently administered (P<0.05). Due to this “carry-over” effect, an additional 22 subjects were treated in the same manner, except that the oxaloacetate/vitamin C mix was provided to them after the cross-over from placebo, so as to not bias the results with a false positive of the placebo (part B participants).
Table 2 summarizes that the percentage changes in depression, perceived stress, general anxiety, and aggression, which were highly and significantly improved over baseline with oxaloacetate/vitamin C treatment (P<0.0001, P<0.0001, P<0.0001 and P=0.0001, respectively). Compared with placebo (rather than baseline) depression, perceived stress and general anxiety were significantly improved with oxaloacetate/vitamin C treatment, with aggression trending toward significance (P<0.05, P<0.05, P<0.05 and P=0.060, respectively).
Table 2. Comparison of changes from baseline within and between oxaloacetate/vitamin C and placebo.
| Characteristics | Oxaloacetate | Placebo | P-valueb) | |||||
|---|---|---|---|---|---|---|---|---|
| Mean improvement (%) | Standard error (%) | P-valuea) | Mean improvement (%) | Standard error (%) | P-valuea) | |||
| Part A | ||||||||
| Becks depression | 52.2 | ±8.8 | <0.0001 | 25.9 | ±13.7 | <0.05 | <0.05 | |
| Cohen perceived stress | 36.2 | ±6.6 | <0.0001 | 21.4 | ±10.2 | <0.05 | <0.05 | |
| Generalized anxiety disorder | 51.3 | ±8.8 | <0.0001 | 24.0 | ±14.0 | <0.05 | <0.05 | |
| Buss-Perry aggression | 15.4 | ±4.8 | 0.0001 | 6.5 | ±5.9 | 0.128 | 0.060 | |
| Part B | ||||||||
| Becks depression | 56.4 | ±5.8 | <0.0001 | 28.1 | ±6.3 | <0.001 | <0.01 | |
| Cohen perceived stress | 37.4 | ±4.7 | <0.0001 | 15.0 | ±4.1 | <0.05 | 0.001 | |
| Generalized anxiety disorder | 52.6 | ±6.4 | <0.0001 | 15.1 | ±12.2 | 0.231 | <0.01 | |
| Buss-Perry aggression | 18.8 | ±4.0 | 0.0001 | 6.2 | ±3.9 | 0.132 | 0.01 | |
a)P-value for evaluating changes from baseline within treatment group; b)P-value for evaluating changes from baseline between oxaloacetate and placebo group (primary analysis).
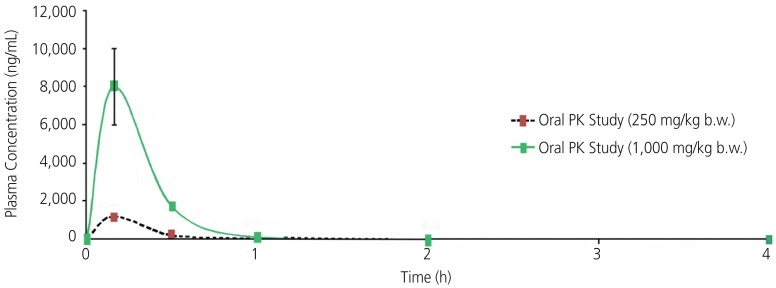
Analysis of the data for a cross-over effect indicated that there was a significant effect in depression when oxaloacetate/vitamin C was administered first (P<0.05). For perceived stress, generalized anxiety and aggression, there was a trend toward a carry-over effect when oxaloacetate/vitamin C was administered first. Previous work, shown in Fig. 1, examining the pharmacokinetics value of oxaloacetate in mouse models was reviewed to better understand the cross-over effect evident in this study. A single dose of oxaloacetate of 250 mg/kg body weight and 1,000 mg/kg body weight demonstrated a half-life of 0.28 and 0.14 hours, respectively. No residual increases in oxaloacetate were observed in the animals after 1 hour.
Fig. 1. Single dose oral pharmacokinetics study of oxaloacetate in male BALB/c mice.
PK, pharmacokinetics.
To overcome the carry-over effect in the cross-over design, the study was repeated with placebo being administered first in part B. This was a non-randomized cross-over study of oxaloacetate/vitamin C versus placebo in 22 subjects. All subjects received placebo first, followed by oxaloacetate/vitamin C; the results of part B of this study are summarized in Table 2. As shown, oxaloacetate/vitamin C supplementation resulted in highly significant improvements for all survey measures over baseline (P<0.0001, P<0.0001, P<0.0001, and P=0.0001). Placebo supplementation resulted in significant improvements for depression and perceived stress over baseline (P<0.001 and P<0.05, respectively), but were not significant for generalized anxiety or aggression (P=0.231 and P=0.132, respectively). The improvement in scores for the oxaloacetate/vitamin C group was much larger than results for all the measurements in the placebo group.
Data from parts A and B of this clinical trial for the 48 women were compiled and summarized in Table 3. As shown, the improvements in depression (54.1%, P<0.0001), perceived stress (35.8%, P<0.001), general anxiety (51.4%, P<0.001), and aggression (17.8%, P<0.0001) were significantly improved over baseline with oxaloacetate/vitamin C treatment. Furthermore, although the improvements over baseline with placebo ranged from 6.8% to 30.7% (P<0.001, P<0.001, P<0.001 and P<0.05, respectively), the difference between oxaloacetate/vitamin C and placebo was highly significant (P<0.01, P<0.001, P<0.001, P<0.01, respectively).
Table 3. Overall comparison of changes from baseline within and between oxaloacetate/vitamin C and placebo for parts A and B participants.
| Characteristics | Oxaloacetate | Placebo | P-valueb) | ||||
|---|---|---|---|---|---|---|---|
| Mean improvement (%) | Standard error (%) | P-valuea) | Mean improvement (%) | Standard error (%) | P-valuea) | ||
| Becks depression | 54.1 | ±4.2 | <0.0001 | 30.7 | ±5.9 | <0.001 | <0.01 |
| Cohen perceived stress | 35.8 | ±3.3 | <0.0001 | 18.0 | ±4.3 | <0.001 | <0.001 |
| Generalized anxiety disorder | 51.4 | ±4.4 | <0.0001 | 27.0 | ±7.6 | <0.001 | <0.001 |
| Buss-Perry aggression | 17.8 | ±2.5 | 0.0001 | 6.8 | ±2.8 | <0.05 | <0.01 |
a)P-value for evaluating changes from baseline within treatment group; b)P-value for evaluating changes from baseline between oxaloacetate and placebo group (primary analysis).
Based on average scores from the 4 surveys, as shown in Table 4, women treated with oxaloacetate moved from a classification of “moderate depression” to “mild mood disturbance”; from “above average stress” to “slightly above average stress”; from “moderate anxiety” to “mild anxiety”; and from “above average aggression” to “slightly above average aggression.”
Table 4. Overall averages for oxaloacetate/vitamin C mix treated women in the study using self-assessment surveys.
| Characteristics | Becks depression inventory | Cohen perceived stress | Generalized anxiety disorder | Buss-Perry aggression |
|---|---|---|---|---|
| Raw mean score | 13.0 | 16.5 | 6.8 | 70.9 |
| Standard deviation | 10.6 | 6.6 | 4.9 | 23.0 |
| Classification of group after oxaloacetate treatment | Mild mood disturbance | Slightly above average stress | Mild anxiety | Slightly above average aggression |
As a subcategory analysis, the suicidal ideation question from the Beck's Depression Inventory demonstrated a mean improvement of 47.9% with oxaloacetate/vitamin C supplementation, which was highly significant over baseline (P<0.0001) for women who had an initial baseline measurement of suicidal ideation (n=31). This was examined due to the concern that some anti-depressants may increase the risk for suicidal ideation [31]. During the study, no side effects and/or adverse effects in the study population were reported with the oxaloacetate/vitamin C mix.
Discussion
Results of this study demonstrated that a mix of oxaloacetate/vitamin C produced a highly significant improvement over baseline and placebo in depression, perceived stress, anxiety, and aggression related to PMS. In part A, there was a significant difference between the oxaloacetate/vitamin C and placebo treatment groups for all PMS surveys except aggression, which demonstrated a trend toward statistical significance. Placebo treatment did not significantly improve the anxiety and aggression scores and produced a lower significance level of improvement over baseline than the oxaloacetate/vitamin C treatment in depression and perceived stress.
This was observed despite the observation of a carryover effect when oxaloacetate/vitamin C was administered first, which would skew the part A results in favor of the placebo group. These results indicate that oxaloacetate/vitamin C has lasting effects on mood, which may be related to gene modulation effects rather than pharmacological effects.
Due to the observed carryover effect, the study was repeated in part B, with a modification to the study design, such that the placebo was administered first to eliminate any carryover, rather than randomizing the order of placebo and oxaloacetate/vitamin C in the trial. Part B of the study produced results nearly identical to those of part A, with all oxaloacetate/vitamin C results significantly improved over baseline and placebo.
Previous work has demonstrated that the half-life of orally supplemented oxaloacetate/vitamin C in the blood stream is short, under 2 hours. This conflicts with the observed results that that a cross-over effect occurs when oxaloacetate/vitamin C was administered first for approximately 28 days, then the placebo was ingested. This may be due to changes in gene expression rather than pharmacological effects. Mice administered oxaloacetate exhibited changes in gene expression change similar to those of calorie restriction [19]. Of importance were gene changes associated with better regulation of glucose, such as increases in the expression levels of FOXO3a. Genomic effects occur over weeks or months, and typically take the same length of time to dissipate [32]. Thus, the carryover effect of oxaloacetate supplementation improving depression related to PMS for time periods past its pharmacological presence could be due to beneficial gene expression and better regulation of systemic glucose.
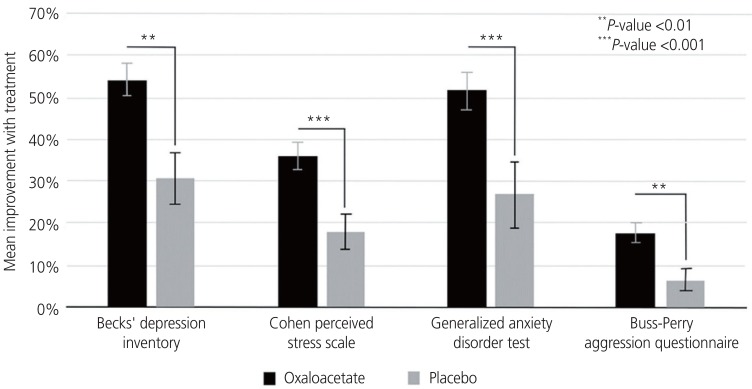
Fig. 2 presents the results of all women in the 2 cohorts normalized to a scale from 0% to 100%, and shows the high statistical improvement compared with placebo in all surveys taken. As shown, the improvement in depression, perceived stress, general anxiety, and aggression were highly significantly improved over both baseline and placebo with oxaloacetate/vitamin C treatment. Depression and anxiety were the most improved, helping women significantly reduce these negative emotions by >50% compared with baseline. Perceived stress and aggression were also significantly reduced compared with placebo.
Fig. 2. An analysis of changes from baseline measurements of 4 emotional symptoms surveys versus treatment with an oxaloacetate/vitamin C mix or placebo in 48 participants with premenstrual syndrome.
Modern science does not have a consensus answer as to why some women experience terrible mood issues before their menstrual cycle. It may be a combination of factors and, while hormone fluctuations are often believed to be causative for emotional PMS, there may be other explanations. Neuroimaging studies of women experiencing extreme mood swings with PMS have demonstrated a large increase in glucose consumption in the cerebellum [9]. In a study involving approximately 70,000 women, scientists at Columbia University (New York, NY, USA) investigated the link between sugary foods and depression, where they found a statistically significant increase in depression in women who consumed foods with added sugars. In contrast, women who consumed fruits and vegetables had a significantly lower incidence of depression [33]. Rather than glucose supplementation, oxaloacetate may provide relief of emotional symptoms in PMS through improved glucose availability on demand. Cellular studies indicate that oxaloacetate is an important metabolite in the liver, allowing the conversion of fatty acids to glucose through gluconeogenesis [12]. We speculate that PMS-related mood symptoms are due to a lack of glucose supply to the brain, particularly the cerebellum, which oxaloacetate may help to alleviate. Further evidence that oxaloacetate supports healthy glucose levels is through clinical trials involving subjects with diabetes. Supplementing with oxaloacetate helps drive glucose into cells, thus lowering fasting glucose levels in diabetics, on average, by 24%. No side effects were reported in the diabetic study [18].
In addition to improved glucose system processing, oxaloacetate has been shown in animal trials to modify neurotransmitter levels in the brain that may also affect mood. In a recent study by Wilkins et al. [25], oxaloacetate was found to increase in vivo levels of several key neurochemicals. In that study, 5-month old male C75B1/6 mice were analyzed using proton magnetic resonance spectroscopy (1H-MRS). Scans were performed before receiving treatment and on the same mice 1 week after receiving treatment. Mice were administered intraperitoneal injections of oxaloacetate at a dose of 2 g/kg/day for 1 week. The authors found that after 1 week of treatment, oxaloacetate induced changes in several key brain biochemical intermediates. Brain GABA levels were elevated by 15% compared with pretreatment levels. Glutamine has been shown to be elevated in the brains of depressed rats [34]. The metabolic by-product of glutamine—glutamate—may play a key role in pathological anxiety via related excitatory dysfunction [35]. Oxaloacetate has been shown to reduce excess free glutamate levels in the brain [36], and these changes in glutamate and GABA may alleviate the emotional symptom component of PMS.
This work was purposefully not performed on participants with PMDD nor those diagnosed with clinical depression because these conditions are classified as “diseases” by the US FDA and greatly increase the cost of clinical investigations. In future work, it may be beneficial to examine both conditions with oxaloacetate supplementation.
A major concern with anti-depressant compounds is a potential inverse relationship with suicidal ideation [37,38]. The suicidal ideation question in the Beck's Depression Inventory demonstrated a mean improvement of 47.9% with oxaloacetate/vitamin C supplementation, which was highly significant over baseline. The improvement in suicidal ideation with oxaloacetate supplementation should be examined further, as there are few pharmacological methods to affect this issue that may have dire consequences.
In conclusion, results of this study, involving 48 women with emotional symptoms while experiencing PMS, demonstrated that capsules containing a mixture of oxaloacetate/vitamin C significantly improved depression, perceived stress, anxiety, aggression, and suicidal ideation. Further work investigating the effects of oxaloacetate on anxiety and depression is warranted, as patients with formally diagnosed PMDD and clinical depression were excluded from this study. Physical PMS symptoms were not addressed in this study and should be examined in future investigations. Additional attention should be devoted to patients with suicidal ideation because oxaloacetate/vitamin C supplementation may be an effective method of saving lives.
Acknowledgements
Kathy Cash assisted in day-to-day implementation of this study. Funding for this study was provided by Terra Biological LLC. Author Alan Cash is CEO of Terra Biological LLC, a company which holds patent and commercial interests in oxaloacetate.
Footnotes
Conflict of interest: No potential conflict of interest relevant to this article was reported.
Ethics approval: The procedures followed were in accordance with the ethical standards of the institution or regional committee on human experimentation and that approval was obtained from the relevant committee on human subjects and/or animal welfare. The Institutional Review Board (IRB) for this clinical trial was “The Institute for Regenerative and Cellular Medicine” number IRCM-2016-086.
Patient consent: Each participant in the clinical trials describe in this article provided written consent to the inclusion of material pertaining to themselves, and the authors have fully anonymized them.
References
- 1.Zendehdel M, Elyasi F. Biopsychosocial etiology of premenstrual syndrome: a narrative review. J Family Med Prim Care. 2018;7:346–356. doi: 10.4103/jfmpc.jfmpc_336_17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Hoyer J, Burmann I, Kieseler ML, Vollrath F, Hellrung L, Arelin K, et al. Menstrual cycle phase modulates emotional conflict processing in women with and without premenstrual syndrome (PMS)--a pilot study. PLoS One. 2013;8:e59780. doi: 10.1371/journal.pone.0059780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Beck AT, Beamesderfer A. Assessment of depression: the depression inventory. Mod Probl Pharmacopsychiatry. 1974;7:151–169. doi: 10.1159/000395074. [DOI] [PubMed] [Google Scholar]
- 4.Beck AT, Epstein N, Brown G, Steer RA. An inventory for measuring clinical anxiety: psychometric properties. J Consult Clin Psychol. 1988;56:893–897. doi: 10.1037//0022-006x.56.6.893. [DOI] [PubMed] [Google Scholar]
- 5.Löwe B, Decker O, Müller S, Brähler E, Schellberg D, Herzog W, et al. Validation and standardization of the Generalized Anxiety Disorder Screener (GAD-7) in the general population. Med Care. 2008;46:266–274. doi: 10.1097/MLR.0b013e318160d093. [DOI] [PubMed] [Google Scholar]
- 6.Andreou E, Alexopoulos EC, Lionis C, Varvogli L, Gnardellis C, Chrousos GP, et al. Perceived Stress Scale: reliability and validity study in Greece. Int J Environ Res Public Health. 2011;8:3287–3298. doi: 10.3390/ijerph8083287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Lee EH. Review of the psychometric evidence of the perceived stress scale. Asian Nurs Res. 2012;6:121–127. doi: 10.1016/j.anr.2012.08.004. [DOI] [PubMed] [Google Scholar]
- 8.Diamond PM, Magaletta PR. The short-form Buss-Perry Aggression Questionnaire (BPAQ-SF): a validation study with federal offenders. Assessment. 2006;13:227–240. doi: 10.1177/1073191106287666. [DOI] [PubMed] [Google Scholar]
- 9.Rapkin AJ, Berman SM, Mandelkern MA, Silverman DH, Morgan M, London ED. Neuroimaging evidence of cerebellar involvement in premenstrual dysphoric disorder. Biol Psychiatry. 2011;69:374–380. doi: 10.1016/j.biopsych.2010.09.029. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Wolf U, Rapoport MJ, Schweizer TA. Evaluating the affective component of the cerebellar cognitive affective syndrome. J Neuropsychiatry Clin Neurosci. 2009;21:245–253. doi: 10.1176/jnp.2009.21.3.245. [DOI] [PubMed] [Google Scholar]
- 11.Kimbrell TA, Ketter TA, George MS, Little JT, Benson BE, Willis MW, et al. Regional cerebral glucose utilization in patients with a range of severities of unipolar depression. Biol Psychiatry. 2002;51:237–252. doi: 10.1016/s0006-3223(01)01216-1. [DOI] [PubMed] [Google Scholar]
- 12.Schmahmann JD, Weilburg JB, Sherman JC. The neuropsychiatry of the cerebellum - insights from the clinic. Cerebellum. 2007;6:254–267. doi: 10.1080/14734220701490995. [DOI] [PubMed] [Google Scholar]
- 13.Schmahmann JD, Caplan D. Cognition, emotion and the cerebellum. Brain. 2006;129:290–292. doi: 10.1093/brain/awh729. [DOI] [PubMed] [Google Scholar]
- 14.Schmahmann JD. The role of the cerebellum in cognition and emotion: personal reflections since 1982 on the dysmetria of thought hypothesis, and its historical evolution from theory to therapy. Neuropsychol Rev. 2010;20:236–260. doi: 10.1007/s11065-010-9142-x. [DOI] [PubMed] [Google Scholar]
- 15.Trout KK, Basel-Brown L, Rickels MR, Schutta MH, Petrova M, Freeman EW, et al. Insulin sensitivity, food intake, and cravings with premenstrual syndrome: a pilot study. J Womens Health (Larchmt) 2008;17:657–665. doi: 10.1089/jwh.2007.0594. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Michener W, Rozin P, Freeman E, Gale L. The role of low progesterone and tension as triggers of perimenstrual chocolate and sweets craving: some negative experimental evidence. Physiol Behav. 1999;67:417–420. doi: 10.1016/s0031-9384(99)00094-3. [DOI] [PubMed] [Google Scholar]
- 17.Møller SE. Serotonin, carbohydrates, and atypical depression. Pharmacol Toxicol. 1992;71(Suppl 1):61–71. doi: 10.1111/j.1600-0773.1992.tb01630.x. [DOI] [PubMed] [Google Scholar]
- 18.Yoshikawa K. Studies on the anti-diabetic effect of sodium oxaloacetate. Tohoku J Exp Med. 1968;96:127–141. doi: 10.1620/tjem.96.127. [DOI] [PubMed] [Google Scholar]
- 19.Cash A. Oxaloacetic acid supplementation as a mimetic of calorie restriction. Open Longev Sci. 2009;3:22–27. [Google Scholar]
- 20.Marco R, Pestaña A, Sebastian J, Sols A. Oxaloacetate metabolic crossroads in liver. Enzyme compartmentation and regulation of gluconeogenesis. Mol Cell Biochem. 1974;3:53–70. doi: 10.1007/BF01660077. [DOI] [PubMed] [Google Scholar]
- 21.Ford TC, Nibbs R, Crewther DP. Glutamate/GABA+ ratio is associated with the psychosocial domain of autistic and schizotypal traits. PLoS One. 2017;12:e0181961. doi: 10.1371/journal.pone.0181961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Stan AD, Schirda CV, Bertocci MA, Bebko GM, Kronhaus DM, Aslam HA, et al. Glutamate and GABA contributions to medial prefrontal cortical activity to emotion: implications for mood disorders. Psychiatry Res. 2014;223:253–260. doi: 10.1016/j.pscychresns.2014.05.016. [DOI] [PubMed] [Google Scholar]
- 23.Zlotnik A, Sinelnikov I, Gruenbaum BF, Gruenbaum SE, Dubilet M, Dubilet E, et al. Effect of glutamate and blood glutamate scavengers oxaloacetate and pyruvate on neurological outcome and pathohistology of the hippocampus after traumatic brain injury in rats. Anesthesiology. 2012;116:73–83. doi: 10.1097/ALN.0b013e31823d7731. [DOI] [PubMed] [Google Scholar]
- 24.Zlotnik A, Gurevich B, Tkachov S, Maoz I, Shapira Y, Teichberg VI. Brain neuroprotection by scavenging blood glutamate. Exp Neurol. 2007;203:213–220. doi: 10.1016/j.expneurol.2006.08.021. [DOI] [PubMed] [Google Scholar]
- 25.Wilkins HM, Harris JL, Carl SM, e L, Lu J, Eva Selfridge J, et al. Oxaloacetate activates brain mitochondrial biogenesis, enhances the insulin pathway, reduces inflammation and stimulates neurogenesis. Hum Mol Genet. 2014;23:6528–6541. doi: 10.1093/hmg/ddu371. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Method for extending lifespan and delaying the onset of age-related disease. European Patent Office EP1824470B1. 2005. [Google Scholar]
- 27.von Glischinski M, von Brachel R, Hirschfeld G. How depressed is “depressed”? A systematic review and diagnostic meta-analysis of optimal cut points for the Beck Depression Inventory revised (BDI-II) Qual Life Res. 2019;28:1111–1118. doi: 10.1007/s11136-018-2050-x. [DOI] [PubMed] [Google Scholar]
- 28.Cohen S. Perceived stress in a probability sample of the United States. In: Spacapan S, Oskamp S, editors. The social psychology of health: the Claremont symposium on applied social psychology. Newbury Park, CA: Sage Publications, Inc.; 1988. pp. 31–67. [Google Scholar]
- 29.Spitzer RL, Kroenke K, Williams JB, Löwe B. A brief measure for assessing generalized anxiety disorder: the GAD-7. Arch Intern Med. 2006;166:1092–1097. doi: 10.1001/archinte.166.10.1092. [DOI] [PubMed] [Google Scholar]
- 30.Buss AH, Perry M. The aggression questionnaire. J Pers Soc Psychol. 1992;63:452–459. doi: 10.1037//0022-3514.63.3.452. [DOI] [PubMed] [Google Scholar]
- 31.Ducasse D, Jaussent I, Olié E, Guillaume S, Lopez-Castroman J, Courtet P. Personality traits of suicidality are associated with premenstrual syndrome and premenstrual dysphoric disorder in a suicidal women sample. PLoS One. 2016;11:e0148653. doi: 10.1371/journal.pone.0148653. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Dhahbi JM, Mote PL, Wingo J, Tillman JB, Walford RL, Spindler SR. Calories and aging alter gene expression for gluconeogenic, glycolytic, and nitrogen-metabolizing enzymes. Am J Physiol. 1999;277:E352–60. doi: 10.1152/ajpendo.1999.277.2.E352. [DOI] [PubMed] [Google Scholar]
- 33.Gangwisch JE, Hale L, Garcia L, Malaspina D, Opler MG, Payne ME, et al. High glycemic index diet as a risk factor for depression: analyses from the Women's Health Initiative. Am J Clin Nutr. 2015;102:454–463. doi: 10.3945/ajcn.114.103846. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Cui T, Qiu HM, Huang D, Zhou QX, Fu XY, Li HY, et al. Abnormal levels of seven amino neurotransmitters in depressed rat brain and determination by HPLC-FLD. Biomed Chromatogr. 2017;31:e3937. doi: 10.1002/bmc.3937. [DOI] [PubMed] [Google Scholar]
- 35.Li CT, Lu CF, Lin HC, Huang YZ, Juan CH, Su TP, et al. Cortical inhibitory and excitatory function in drug-naive generalized anxiety disorder. Brain Stimulat. 2017;10:604–608. doi: 10.1016/j.brs.2016.12.007. [DOI] [PubMed] [Google Scholar]
- 36.Zlotnik A, Gruenbaum SE, Artru AA, Rozet I, Dubilet M, Tkachov S, et al. The neuroprotective effects of oxaloacetate in closed head injury in rats is mediated by its blood glutamate scavenging activity: evidence from the use of maleate. J Neurosurg Anesthesiol. 2009;21:235–241. doi: 10.1097/ANA.0b013e3181a2bf0b. [DOI] [PubMed] [Google Scholar]
- 37.Courtet P, Lopez-Castroman J. Antidepressants and suicide risk in depression. World Psychiatry. 2017;16:317–318. doi: 10.1002/wps.20460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Friedman RA, Leon AC. Expanding the black box - depression, antidepressants, and the risk of suicide. N Engl J Med. 2007;356:2343–2346. doi: 10.1056/NEJMp078015. [DOI] [PubMed] [Google Scholar]