Abstract
Purpose
Pyruvate kinase muscle isoenzyme 2 (PKM2) is a key enzyme in aerobic glycolysis and is thought to contribute to cancer cell metabolic reprogramming. The aim of this study was to evaluate PKM2 immunohistochemical expression as a potential prognostic biomarker in pancreatic ductal adenocarcinoma (PDAC).
Methods
A tissue microarray was constructed using surgical specimens for 115 patients who underwent resections for PDAC, stained with PKM2 antibody, and scored for expression level. Statistical analyses were performed to investigate the association between PKM2 and patient survival, tumor stage, tumor grade, surgical margin status, lymph node ratio, perineural invasion status, or the use of adjuvant chemotherapy.
Results
Fifty-three percent of tumors had positive PKM2 expression, and 47 % of tumors had negative PKM2 expression. PKM2 expression was associated with overall survival (HR 0.56, p=0.007) and CA 19–9 levels (p=0.035), but was not associated with tumor stage, tumor grade, surgical margin status, lymph node ratio, perineural invasion, or adjuvant chemotherapy use.
Conclusions
PKM2 expression is associated with overall survival in PDAC. Further studies are warranted to validate the value of PKM2 as a prognostic biomarker and to examine the potential utility of PKM2 in predicting treatment response, as well as a potential therapeutic target in PDAC.
Keywords: Pyruvate muscle kinase isoenzyme 2, Prognostic biomarker, Pancreatic adenocarcinoma, Immunohistochemical expression
Introduction
In the 1920s, Otto Warburg observed that cancer cells displayed a unique metabolism compared to normal cells. He noted cancer cells metabolize glucose by glycolysis, rather than utilizing pyruvate to undergo the tricarboxylic acid (TCA) cycle and oxidative phosphorylation, which is the more efficient energy production used by normal cells in aerobic conditions [1]. This has been termed the “Warburg effect” or aerobic glycolysis. In contrast to normal cells, cancer cells upregulate glucose uptake and thereby give rise to intermediate glycolytic metabolites and the end product pyruvate. Glycolysis in cancer cells is uncoupled from the mitochondrial TCA cycle and oxidative phosphorylation. The glycolysis-derived pyruvate is diverted into lactate and kept away from the mitochondrial oxidative metabolism. Though described more than 80 years ago, there has been little progression in knowledge until recently. There is a renewed interest in studying the role of this distinct cancer metabolism in tumor initiation and progression [reviewed in 2]. Additionally, it has been suggested that this unique metabolism of cancer cells may be a potential way to selectively target cancer cells with oncologic therapy [reviewed in 3].
Pyruvate kinase (PK) is an enzyme that catalyzes the conversion of phosphoenolpyruvate to pyruvate in the final step of the glycolysis pathway and is thought to be a key player in regulating the switch to aerobic glycolysis in cancer cell metabolism. There are four isoforms of PK—L, R, M1, and M2 [4]. Isoenzymes L and R are encoded by the gene PKLR, while isoenzymes M1 and M2 are encoded by the same gene PKM2 but represent different splice variants [5, 6]. The M1 isoform (PKM1) is typically found in differentiated skeletal muscle, heart, and brain cells, while the M2 isoform (PKM2) is expressed in most embryonic cells, stem cells, and tumor cells. Among the isoenzymes, PKM2 has received the most attention given its preferential expression in cancer and its role in cancer cell anabolic metabolism.
The biological functions of PKM2 in cancer initiation and progression have not been well defined. Most in vitro studies based on analysis of cultured cells demonstrated that switching PKM2 for PKM1 reverses the Warburg effect and reduces cancer cell proliferation. Knockdown of PKM2 has been shown to repress proliferation and migration of colon cancer cells in vitro [7]. Studies using xenograft tumors have yielded contradictory results regarding the role of PKM2 in tumor growth [8, 9]. Both selective small molecular activators and inhibitors of PKM2 have been studied in cell culture and xenograft tumor models. Inhibition of PKM2 has been shown to suppress xenograft tumor graft growth [10]. Interestingly, PKM2 activators TEPP-46 and DASA-58 specifically activate PKM2 but not PKM1 in vitro, and TEPP-46 also impairs tumor growth in vivo in a non-small cell lung carcinoma xenograft [11]. A recent report that loss of PKM2 accelerates tumor formation in a mouse breast cancer model further indicates the complicated functions of PKM2 under different cellular contexts [12].
Pancreatic ductal adenocarcinoma is currently the fourth leading cause of cancer death in the USA and continues to have a dismal prognosis, with 5-year overall survival rates estimated at 6 % [13]. The only potential curative option is surgical resection, for which less than 20 % of patients are eligible. Even in this subset of patients, the 5-year overall survival remains only 18–24 % [14–18]. Given the poor survival with surgery alone, attempts have been made to improve outcomes with adjuvant therapy. However, the role of adjuvant therapy in the management of localized pancreatic cancer remains controversial as many of the randomized clinical trials were statistically underpowered and used outdated radiation fractionation schema and techniques. Therefore, tumor biomarkers that could be used to predict which subset of patients is likely to benefit from adjuvant therapy would be very useful for clinicians to tailor therapy based on that individual patient’s tumor characteristics.
Evidence from preclinical studies suggests a complicated role of PKM2 activity in cancer development. It is important to understand the context-dependent metabolic needs of cancer cells on PKM2. To this end, this study sought to evaluate the potential role of immunohistochemical PKM2 expression as a prognostic biomarker in pancreatic adenocarcinoma.
Materials and Methods
Patient Selection
This study was approved by the Vanderbilt University Medical Center Institutional Review Board. From 2000 to 2009, 115 patients were identified who had undergone curative resections for pancreatic adenocarcinoma and for whom both clinical data and tumor tissue were available. Only patients with histologically confirmed ductal adenocarcinomas were included. All tumors were restaged by a single pathologist (SCS) according to AJCC 7th edition criteria [19]. Data collected included patient demographics, operative details, treatment details, and survival. Pathologic data obtained included tumor location, total number of nodes involved, total number of nodes resected, tumor size, differentiation, pancreatic extension, and margin status. A positive margin was defined as tumor within 1 mm of the inked resection margin on microscopic examination. Tumor differentiation was recorded according to the guidelines outlined by the College of American Pathologists [20]. The lymph node ratio (LNR) was defined as the number of positive lymph nodes as a fraction of the total number of lymph nodes examined/resected.
Construction of Tissue Microarray
Tissue microarrays were constructed using 1-mm cores of both tumor and background normal/reactive pancreas from 115 curative resection specimens, including pancreaticoduodenectomy/gastrojejunostomy procedures (Whipple procedures) and total or distal pancreatectomies. The microarrays were composed of single or duplicate cores from tumor and background pancreas. The microarrays were cut at 5-μm thickness and stained with hematoxylin and eosin.
Immunohistochemistry Study
Five-micrometer thick sections of formalin-fixed, paraffin-embedded tissue microarrays were de-paraffinized in xylene and rehydrated graded alcohol. Antigen enhancement was done by incubating the sections in citrate buffer pH 6 (Dako, Carpentaria, CA) as recommended. Staining was performed using Peroxidase Histostain-Plus Kit (Zymed) according to the manufacturer’s protocol. PKM2 rabbit polyclonal antibody (Cell Signaling, Beverly, MA) was used at a 1:800 dilution. Diaminobenzidine (DAB) (Life Technologies, Camarillo, CA) was used as substrate for peroxidase. Slides were counterstained with hematoxylin. Positive and negative controls for PKM2 staining were performed and are shown in Fig. 1. PKM2 expression was quantified after scoring both the intensity and the extent of the reaction by a 12-tier scoring method previously cited in studies by Han et al. and Koo et al. [21, 22]. The intensity of PKM2 expression was quantified using the following scores: 0=negative, 1=weakly positive, 2=moderately positive, 3=strongly positive. The extent of PKM2 expression was quantified by evaluating the percentage of the positive staining areas in relation to the whole cancer areas in the core, where a score of 0 was given for 0 % reactivity, 1 point was assigned for 1–10 % reactivity, 2 points were assigned for 11–50 % reactivity, 3 points were given for 51–80 % reactivity, and samples with >80 % reactivity were assigned a total of 4 points. The final immunore-active score was determined by multiplying the intensity score by the extent score, with the minimum score attainable being 0 and a maximum score of 12. The 12-tier scoring was then simplified by combining scores so that 0–2=negative PKM2 expression (final score=0), 3–6=weak PKM2 expression (final score=1), 7–9=intermediate PKM2 expression (final score=2), and 10–12=strong PKM2 expression (final score=3). Representative images for PKM2 expression scoring are shown in Fig. 2.
Fig. 1.
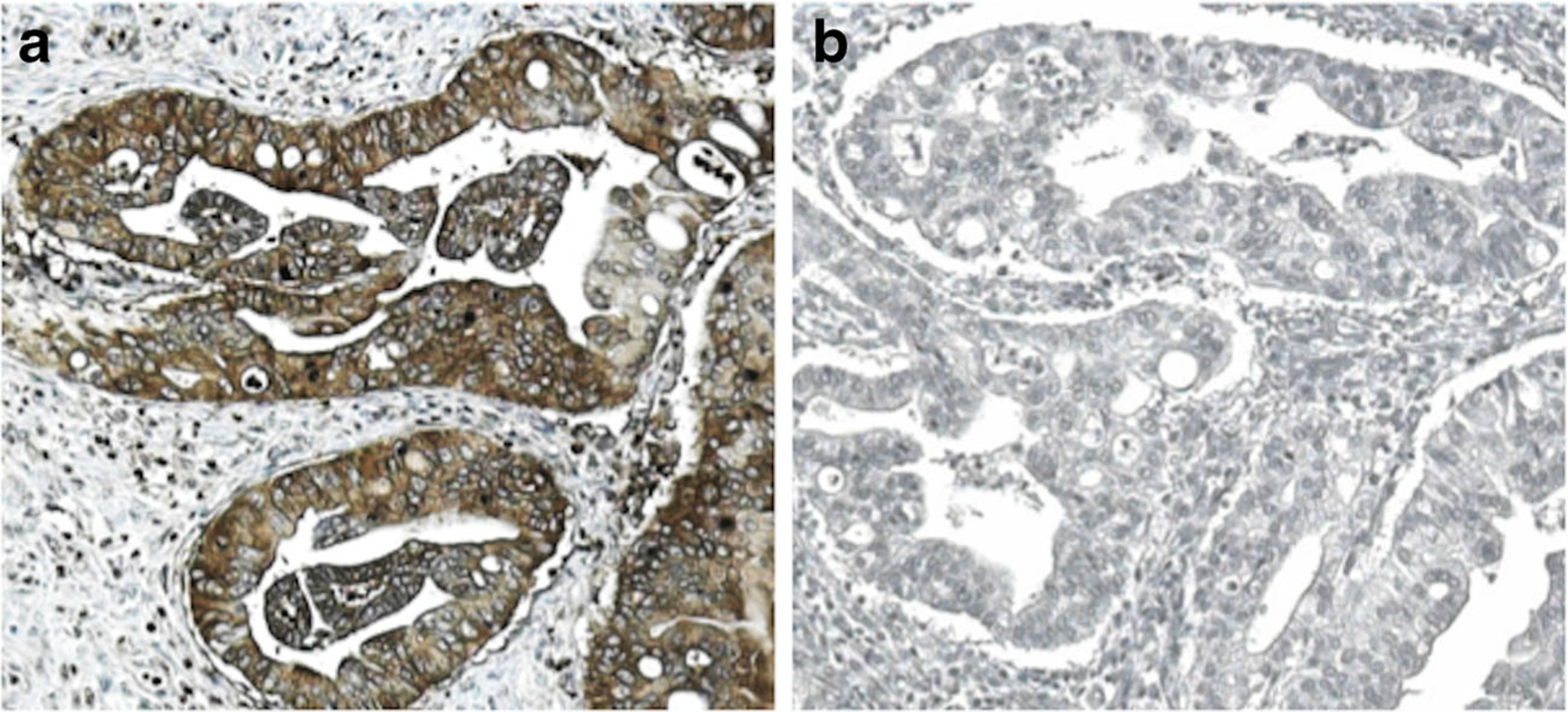
Representative images of positive and negative controls for PKM2 staining. a Positive IHC staining by PKM2 rabbit antibody (1:800). b Negative IHC staining by rabbit IgG at the same titer as PKM2 antibody
Fig. 2.
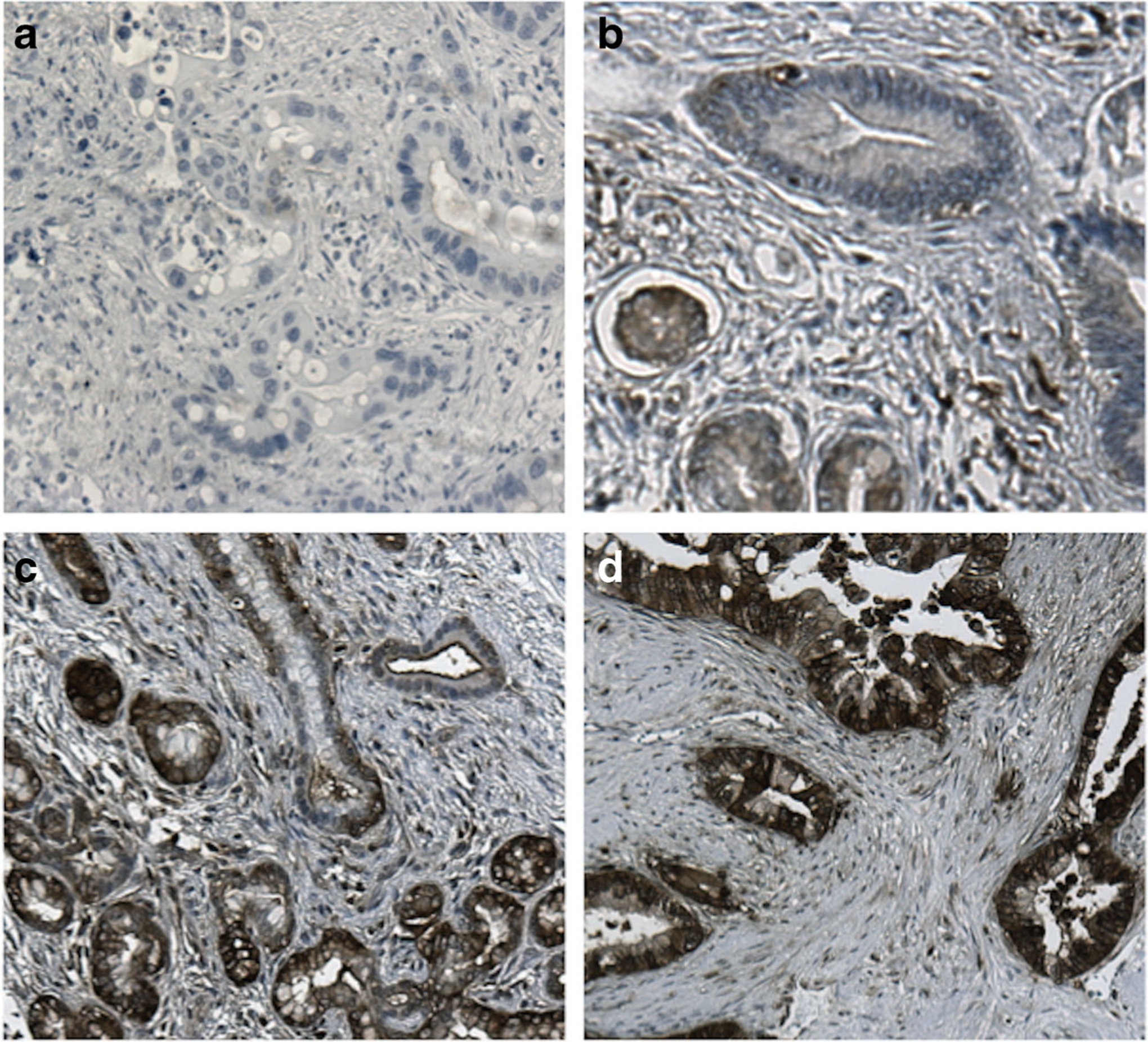
Representative images of PKM2 expression intensity. a Negative PKM2 expression, b weak PKM2 expression, c intermediate PKM2 expression, and d strong PKM2 expression at 100× magnification
Statistical Analysis
PKM2 expression scores, the main biomarker of this study, were unevenly distributed as 0, 1, 2, and 3 with frequency 54, 20, 30, and 11, respectively. To achieve the highest statistical power to detect differences between clinically meaningful groups, we further classified the PKM2 expression as positive (score 1, 2, or 3) or negative (score 0). Patient demographic and clinical variables were then summarized by PKM2 expression (negative versus positive), using the median with the 25th and 75th percentiles (quartiles) for continuous variables. For categorical variables, frequency and percentages were shown. Comparisons between PKM2 negative and positive expression groups were conducted with Wilcoxon rank-sum test (continuous variables) and Pearson’s test (categorical variables). The primary endpoint was overall survival (OS), which was defined as the time from surgery to the date of all-cause death or last follow-up. The Kaplan-Meier method and log-rank test were used to compare the overall survival between PKM2 expression groups. To assess the association between PKM2 expression and the overall survival, a multivariable Cox proportional hazard (Cox PH) model was employed. All variables adjusted in the multivariable model were selected a priori based on our clinical experiences and examinations using directed acyclic graphs (DAGs) [23]. Multiple imputations (MI) with five iterations were used to deal with the missing values [24]. All analyses were performed with R version 3.0.2 [25], and statistical significance was based on two-sided tests at the 5 % level.
Results
Patient Clinical and Pathologic Characteristics
From 2000–2009, 115 patients were identified who had undergone curative resections for pancreatic adenocarcinoma for whom tissue samples were also available for study. Table 1 summarizes the demographic and treatment details as well as clinicopathologic findings. The average patient age was 67 years. The majority of patients (90.4 %) underwent a Whipple resection, and 70.0 % of all patients in the study had microscopically negative surgical margins. The median overall survival for all patients was 13.2 months (IQR 7.6–31.6). Fifty-three percent of tumors had positive PKM2 expression intensity, and 47 % of tumors had negative PKM2 expression. The percentages of patients with weak, intermediate, and strong PKM2 expression are shown in Table 2. In positive-staining tumor cells, PKM2 was predominantly expressed in the cytoplasm (Figs. 1 and 2). In the normal pancreas tissue, weak to moderate positive staining of PKM2 was evident in the epithelial cells of the ducts, especially in small and intercalated ducts. PKM2 is also expressed in the interspersed islet cells (Fig. 3).
Table 1.
Patient clinical and pathologic characteristics
| Number | No. (%) | |
|---|---|---|
| Age, years | 115 | 67 (57–73)a |
| Gender | 115 | |
| Female | 52 (45) | |
| Male | 63 (55) | |
| Race | 112 | |
| African American | 5 (4.5) | |
| Caucasian | 107 (95.5) | |
| Tumor grade | 115 | |
| 1 | 15 (13) | |
| 2 | 65 (57) | |
| 3 | 35 (30) | |
| Tumor stage | 115 | |
| I-IIA | 29 (25) | |
| IIB-IV | 86 (75) | |
| Operation type | 115 | |
| Whipple | 104 (90.4) | |
| Distal pancreatectomy | 9 (7.8) | |
| Total pancreatectomy | 1 (0.9) | |
| En bloc resection | 1 (0.9) | |
| Surgical margin status | 115 | |
| Negative | 81 (70) | |
| Positive | 34 (30) | |
| Adjuvant chemotherapy | 110 | |
| No | 30 (27) | |
| Yes | 80 (73) | |
| Adjuvant radiation therapy | 110 | |
| No | 56 (51) | |
| Yes | 54 (49) | |
| CA 19–9 | 97 | 150 (49–369)a |
| Lymph node ratio | 113 | 0.136 (0.000–0.300)a |
| PKM2 expression | 115 | |
| Negative | 54 (47) | |
| Positive | 61 (53) | |
| Overall survival status | 115 | |
| Alive | 23 (20) | |
| Deceased | 92 (80) | |
| Survival time, months | 115 | 13.2 (7.6–31.6)a |
Median (IQR)
Table 2.
Patient PKM2 expression levels
| PKM2 expression score | Patients, N = 115 No. (%) |
|---|---|
| Negative (score 0–2) | 54 (47) |
| Weak (score 3–6) | 20 (17) |
| Intermediate (score 7–9) | 30 (26) |
| Strong (score 10–12) | 11 (10) |
Fig. 3.
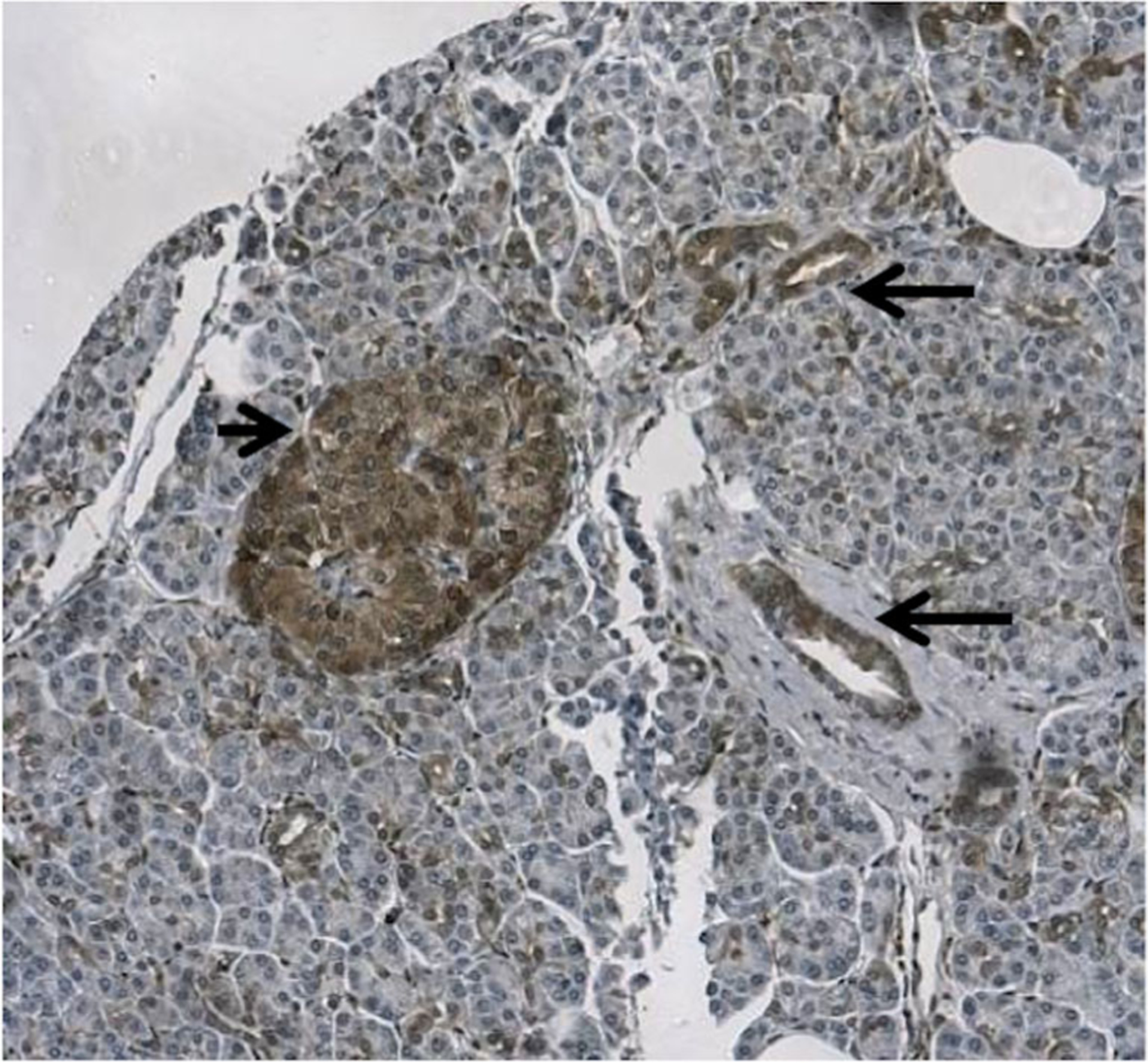
Expression of PKM2 in normal pancreas. Weak to moderate staining of PKM2 is demonstrated in the epithelial cells of the ducts, especially in small and intercalated ducts (long arrows). PKM2 is also expressed in the interspersed islet cells (short arrow)
The Associations Between PKM2 Expression and Overall Survival
PKM2 expression was associated with CA 19–9 levels. Lower CA 19–9 levels were observed among patients who had positive PKM2 expressions (median 85 versus 205, p=0.034; Table 3, Fig. 4). Patients with positive PKM2 expression tended to have lower lymph node ratio than those who had negative PKM2 expression (median 0.091 versus 0.195); however, the difference did not reach statistical significance (p=0.077). PKM2 expression was not observed to be associated with age at surgery, tumor stage, tumor grade, surgical margin status, perineural invasion status, or adjuvant chemotherapy use. In the univariate survival analysis, PKM2 expression was associated with overall survival (HR 0.56, 95 % CI 0.37–0.86, p=0.007). When we further adjusted for age at surgery, surgical margin status, perineural invasion status, peripancreatic extension status, and stratified on adjuvant chemotherapy use status in the multivariable Cox model, the association between PKM2 expression and overall survival remained statistically significant (HR 0.57, 95 % CI 0.36–0.91, p=0.017) (Table 4 and Fig. 5).
Table 3.
The associations between PKM2 expression and other prognostic factors
| PKM2 expression | |||
|---|---|---|---|
| Negativea (N=54) |
Positivea (N=61) |
p valueb | |
| Tumor grade | 0.33 | ||
| 1 | 7 (13 %) | 8 (13 %) | |
| 2 | 27 (50 %) | 38 (62 %) | |
| 3 | 20 (37 %) | 15 (25%) | |
| Tumor stage | 0.49 | ||
| I-IIA | 12 (22 %) | 17 (28 %) | |
| IIBIV | 42 (78 %) | 44 (72 %) | |
| Surgical margin status | 0.099 | ||
| Negative | 34 (63 %) | 47 (77 %) | |
| Positive | 20 (37 %) | 14 (23 %) | |
| Perineural invasion | 0.3 | ||
| Negative | 12 (22 %) | 9 (15 %) | |
| Positive | 42 (78 %) | 52 (85 %) | |
| Peripancreatic extension | 0.29 | ||
| Negative | 10 (19%) | 7 (11 %) | |
| Positive | 44 (81 %) | 54 (89 %) | |
| Adjuvant chemotherapy | 0.28 | ||
| No | 17 (32 %) | 13 (23 %) | |
| Yes | 36 (68 %) | 44 (77 %) | |
| CA 19–9 level | 205 (98–551) | 85 (32–336) | 0.034 |
| Lymph node ratio | 0.195 (0.036–0.435) | 0.091 (0.00–0.286) | 0.077 |
| Overall survival, months | 10.6 (6.6–19.0) | 16.6 (9.8–35.8) | 0.008 |
| Age at surgery | 68.8 (60.7–72.9) | 63.6 (56.5–73.0) | 0.22 |
Frequency (percent) for categorical variables and median (IQR) for continuous variables
Pearson test for categorical variables and Wilcoxon rank-sum test for continuous variables
Fig. 4.
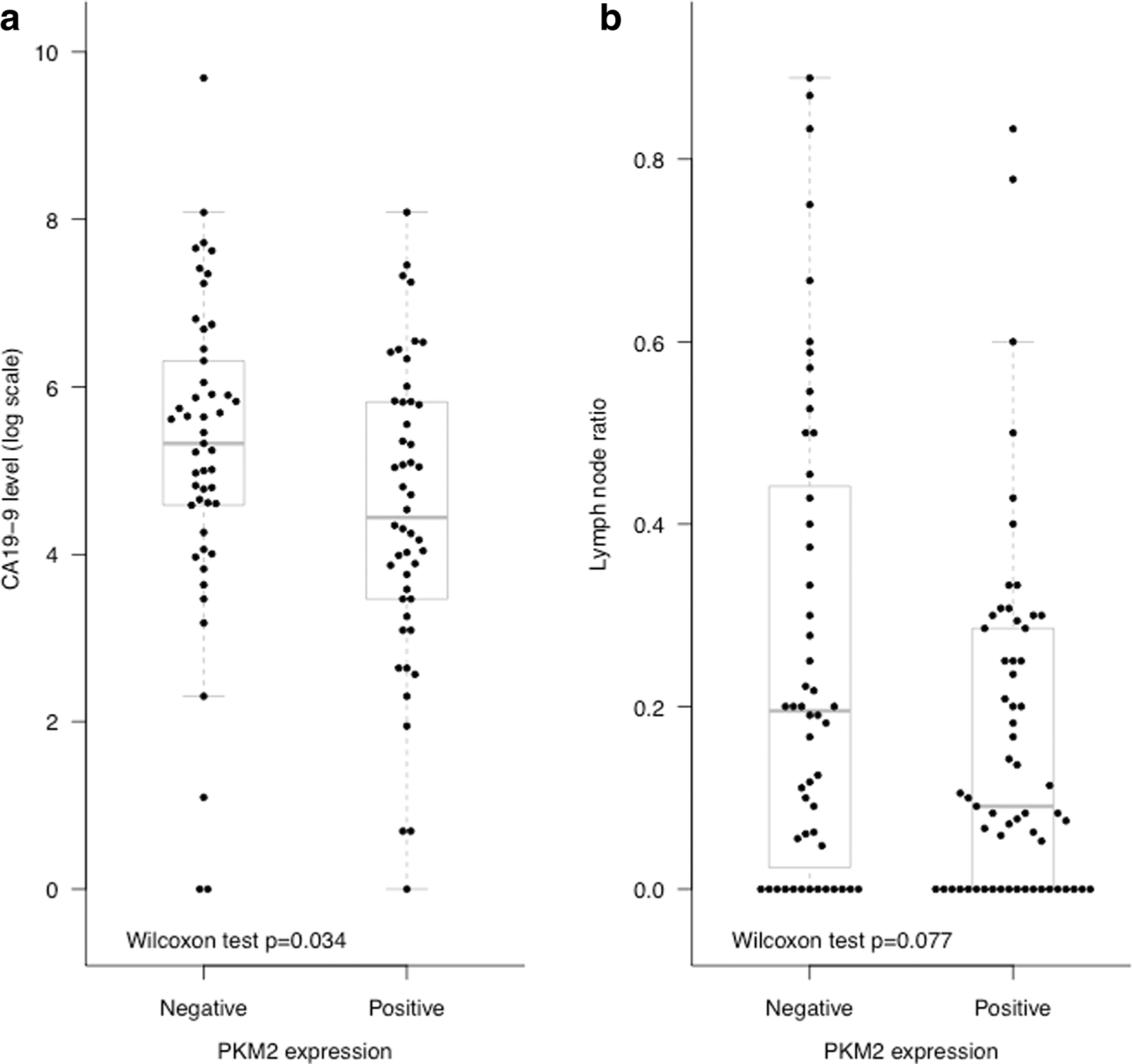
CA19.9 level (a) and lymph node ratio value (b) by PKM2 expression
Table 4.
The associations between PKM2 expression and selected prognostic factors with overall survival form the multivariable Cox model
| Overall survivala | |||
|---|---|---|---|
| Variables | HR | 95 % CI | p value |
| Age at surgery (73 vs 57.2)b | 1.18 | 0.83–1.68 | 0.048 |
| PKM2 expression (positive vs negative) | 0.57 | 0.36–0.91 | 0.017 |
| Surgical margin status (positive vs negative) | 1.54 | 0.95–2.49 | 0.079 |
| Perineural invasion (positive vs negative | 0.81 | 0.46–1.43 | 0.471 |
| Peripancreatic extension (positive vs negative) | 2.36 | 1.21–4.60 | 0.012 |
The multivariable Cox model stratified on adjuvant chemotherapy (yes or no)
Age at surgery was included in the model nonlinearly by using a restricted cubic spline with three knots. Reported here is the HR for upper quartile vs lower quartile, the p value reported is for the whole curve
Fig. 5.
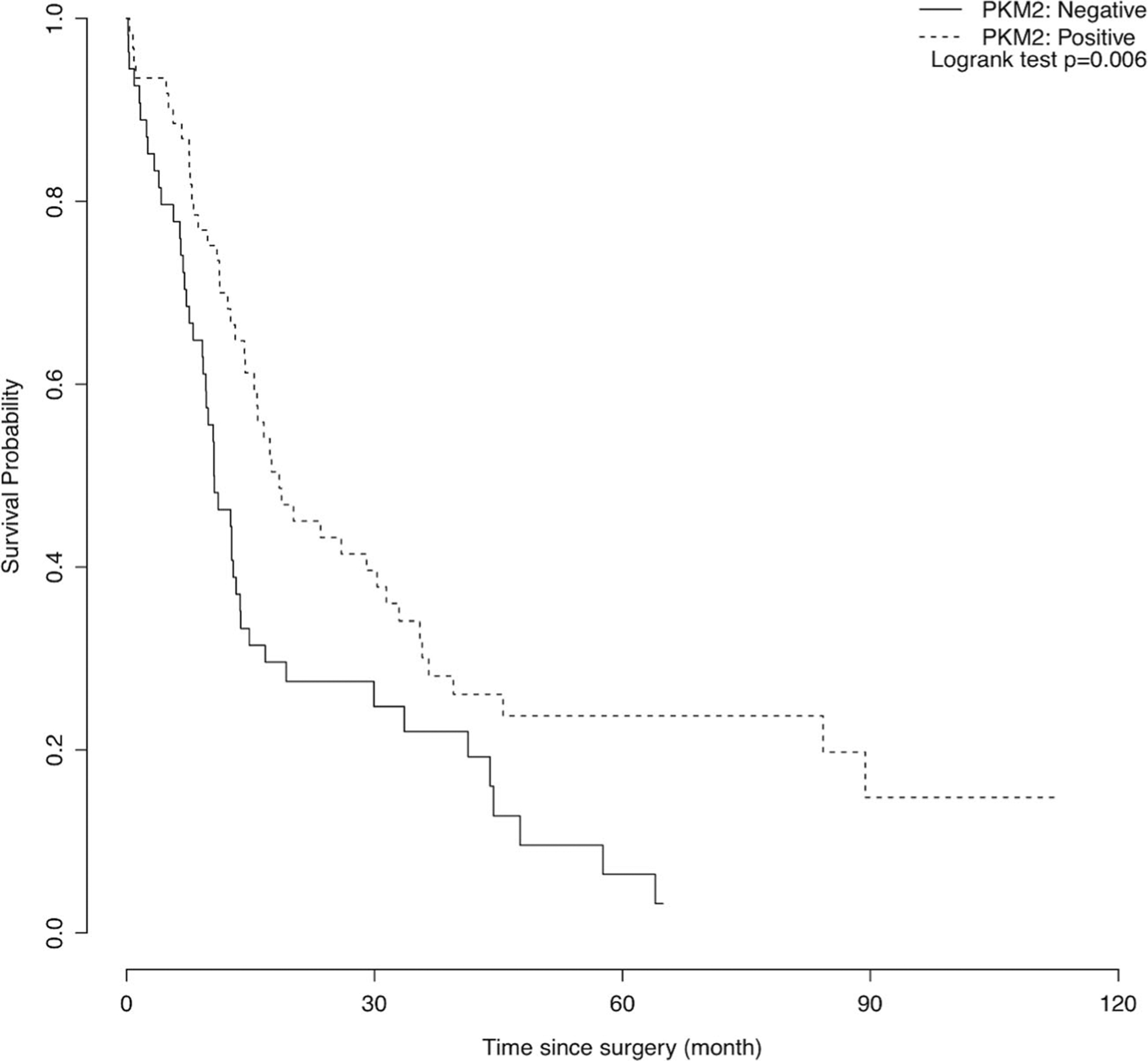
Kaplan-Meier curve for overall survival. PKM2 intensity was associated with overall survival (p=0.006)
Discussion
The aim of this study was to evaluate the potential role of PKM2 immunohistochemical expression as a prognostic biomarker in pancreatic adenocarcinoma. We found that PKM2 expression in pancreatic adenocarcinoma was associated with overall survival (p=0.007) and CA 19–9 levels (p=0.035). PKM2 expression was not found to be associated with tumor stage, tumor grade, surgical margin status, lymph node ratio, perineural invasion status, or the use of adjuvant chemotherapy.
The cellular functions and the pathological roles of PKM2 in cancer metabolism and progression are just beginning to be uncovered. The current understanding of the prognostic value of PKM2 in cancer is limited, and there is wide discrepancy in the reported literature. PKM2 may function differently in cancer development and response to treatment among different tissue and cell types, as supported by a review article by Iqbal et al. detailing conflicting observations of the role of PKM2 among various studies in the literature [26]. PKM2 function may be tissue specific, and its functional regulation within different microenvironments is yet to be determined. Supporting this view, Lim et al. investigated PKM2 expression by mRNA levels and immunohistochemical staining in gastric cancers and found that PKM2 expression was associated with shorter overall survival in signet-ring gastric cancers but was not associated with survival in other gastric cancer histological subtypes [27]. Additionally, studies have shown that normal brain tissue is PKM2 negative while gliomas have strong PKM2 expression that is correlated with glioma tumor grade [28]. In our study, we observed strong PKM2 expression in normal pancreas (Fig. 3). PKM2 expression and function therefore may vary based on cell of origin or molecular environment, which is an interesting future question to explore.
To date, the prognostic value of PKM2 expression in histological specimens of pancreatic cancer has not been published. Our current study is the first to examine the prognostic value of PKM2 immunohistochemical expression in pancreatic cancer and demonstrates that positive PKM2 expression is associated with increased overall survival in PDAC specimens (HR 0.56, 95 % CI 0.37–0.86, p=0.007). Pancreatic cancer inherently features strong inflammatory and extensive fibrotic changes, and it is not totally surprising that our observation in PDAC is different from that in other gastrointestinal malignancies. Zhou et al. investigated PKM2 expression by both mRNA expression and immunohistochemical staining in colorectal cancers and reported that increased PKM2 expression was associated with more advanced stage [10]. High PKM2 immunohistochemical expression in the nucleus has been reported to be associated with shorter survival in esophageal squamous cell carcinoma [29]. In gallbladder cancer, positive PKM2 immunohistochemical expression has been reported to be associated with lymph node metastasis, invasion, and TMN stage as well as shorter average survival by univariate analysis [30].
There are several limitations in our current study. First, tumor or intratumoral heterogeneity is a possible limitation within our study, though tumor heterogeneity is an inherent issue with tissue microarrays [31]. Despite potential tumor heterogeneity, however, if a marker has a strong association, it should be detectable. Second, all of the reported studies to date have grouped the scoring of PKM2 expression differently from one another [27, 29, 30], and the optimal grouping of scoring of PKM2 expression has yet to be established. In the current study, we grouped the scoring of PKM2 immunohistochemical expression into negative versus positive based on sample distribution and statistical power considerations. However, the heterogeneity of PMK2 expression scores impedes the comparison of the results of different studies. Third, adjuvant chemotherapy status may affect the effect of PKM2 on overall survival. This was not our hypothesis for this particular study, and a larger study size would be required to test any interactions. Therefore, we did not test this interaction term directly. However, in our multivariable Cox model, we did allow the patients who received adjuvant therapy had a different baseline survival function than those who received no adjuvant therapy, by fitting the Cox model stratified by the adjuvant therapy status. This is a limitation of our study due to small sample size and is worth further investigation in future larger studies.
PKM2 levels can be measured in EDTA-plasma. A systematic review of PKM2 levels in pancreaticobiliary and colorectal cancer patients found that PKM2 is elevated in malignancy with the degree of elevation corresponding to disease extent. It has been proposed that plasma PKM2 can be used as an adjunctive diagnostic test in conjunction with CA 19–9 and also as a biomarker for adverse prognosis [32]. A meta-analysis found that the specificity of plasma PKM2 for pancreatic cancer was 60 % with a 95 % sensitivity [33]. Given that this is a retrospective study, we were unable to measure concurrent plasma PKM2 levels, which would have added another perspective on PKM2 utility in pancreatic cancer. Future investigations will evaluate if the plasma levels versus histological expression of PKM2 are correlated and whether they have similar or different pathophysiologic functions.
In addition to the evaluated expression levels, PKM2 forms either a dimer with low activity or a tetramer with high activity, and it is thought that the low-activity form of PKM2 contributes to cancer cell progression by promoting aerobic glycolysis [reviewed in 2, 34]. The high-activity PKM2 tetramer is expressed in normal cells and promotes conversion of pyruvate to acetyl-CoA driving the TCA cycle. Conversely, PKM2 activity is suppressed by tyrosine kinase phosphorylation into the low-activity dimer, which is expressed predominately in cancer cells, and promotes the conversion of pyruvate to lactate in glycolysis [reviewed in 33]. The in vivo factors influencing the ratio of PKM2 tetramer to dimer have been studied by Kumar et al. in metastatic and non-metastatic human pancreatic cancer cell lines. It was demonstrated that the metastatic Colo357 cells contained higher levels of PKM2 than the non-metastatic Panc-1 cells, and additionally Colo357 cells contained 3.5-fold more of the dimeric form of PKM2. Changes in pancreatic tumor microenvironment such as hypoxia, glucose deprivation, and low pH had no effect on overall PKM2 levels but increased PKM2 dimeric form in Colo357 cells and also impacted tumor cell proliferation [35]. The prognostic and predictive values of the specific form of PKM2 and the nuclear versus cytosolic expression of PKM2 need to be tested in future studies.
Our finding that positive PKM2 expression is associated with an improved overall survival could have important implications both as a prognostic marker as well as a predictive marker of therapeutic response. Both selective small molecular activators and inhibitors of PKM2 have been studied in cell culture and xenograft tumor modes. Anastasiou et al. has demonstrated that the small molecule PKM2 activators TEPP-46 and DASA-58 specifically activate PKM2 but not PKM1 in vitro and that TEPP-46 impairs tumor growth in vivo in a non-small cell lung carcinoma xenograft [11]. PKM2 inhibitors have also been studied and shown to suppress colorectal cancer tumor growth in an in vivo model [10]. It is apparent in light of these studies that the role of PKM2 in cancer pathogenesis and progression is likely complex, as both inhibitors and activators of PKM2 have shown to be beneficial. Understanding the mechanisms that underlie improved overall survival in pancreatic cancer will be critical for future studies in exploring the predictive value of PKM2 expression, as well as in the development of novel treatment strategies targeting the PKM2 signaling pathway in pancreatic ductal adenocarcinoma.
In summary, PKM2 is emerging as a key player in the switch to cancer cell metabolism and cancer cell progression. This is the first study to examine the prognostic value of PKM2 immunohistochemical expression in pancreatic adenocarcinoma, and we propose that PKM2 expression may be a useful biomarker in pancreatic adenocarcinoma. Further studies are warranted to better understand the role of PKM2 in pancreatic cancer.
Funding
The project described was supported by Cancer Center Support Grant (CCSG) National Institutes of Health (NIH) P30 CA68485 and Grant UL1 RR024975-01 and is now at the National Center for Advancing Translational Sciences, Grant 2 UL1 TR000445-06. The content is solely the responsibility of the authors and does not necessarily represent the official views of the NIH.
Footnotes
Conflict of Interest The authors declare they have no competing interests.
References
- 1.Warburg O, Posener K, Negelein E. Ueber den stoffwechsel der tumoren. Biochem Z. 1924;152:319–44. [Google Scholar]
- 2.Vander Heiden MG, Cantley LC, Thompson CB. Understanding the Warburg effect: the metabolic requirements of cell proliferation. Science. 2009;324:1029–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Sutendra G, Michelakis ED. Pyruvate dehydrogenase kinase as a novel therapeutic target in oncology. Front Oncol. 2013;3:38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Harada K, Saheki S, Wada K, et al. Purification of four pyruvate kinase isoenzymes of rats by affinity elution chromatography. Biochem Biophys Acta. 1978;524:327–39. [DOI] [PubMed] [Google Scholar]
- 5.Noguchi T, Yamada K, Inoue H, et al. The L- and R-type isoenzymes of rate pyruvate kinase are produced from a single gene by use of different promoters. J Biol Chem. 1987;262:14366–71. [PubMed] [Google Scholar]
- 6.Noguchi T, Inoue H, Tanaka T. The M1- and M2-type isoenzymes of rate pyruvate kinase are produced from the same gene by alternative RNA splicing. J Biol Chem. 1986;261:13807–12. [PubMed] [Google Scholar]
- 7.Christofk HR, Vander Heiden MG, Harris MH, et al. The M2 slice isoform of pyruvate kinase is important for cancer metabolism and tumor growth. Nature. 2008;452:230–3. [DOI] [PubMed] [Google Scholar]
- 8.Cortes-Cros M, Hemmerlin C, Ferretti S, et al. M2 isoform of pyruvate kinase is dispensable for tumor maintenance and growth. Proc Natl Acad Sci U S A. 2013;110:489–94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Goldberg MS, Sharp PA. Pyruvate kinase M2-specific siRNA induces apoptosis and tumor regression. J Exp Med. 2012;209:217–24. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Zhou CF, Li XB, Sun H, et al. Pyruvate kinase M2 is upregulated in colorectal cancer and promotes proliferations and migration of colon cancer cells. IUBMB Life. 2012;64:775–82. [DOI] [PubMed] [Google Scholar]
- 11.Anastasiou D, Yu Y, Israelsen WJ, et al. Pyruvate kinase M2 activators promote tetramer formation and suppress tumorigenesis. Nat Chem Biol. 2012;8:839–47. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Israelsen WJ, Dayton TL, Davidson SM, et al. PKM2 isoform-specific deletion reveals a differential requirement for pyruvate kinase in tumor cells. Cell. 2013;155:397–409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Siegel R, Naishadham D, Jermal A. Cancer statistics, 2013. CA Cancer J Clin. 2013;63:11–30. [DOI] [PubMed] [Google Scholar]
- 14.Yeo CJ, Abrams RA, Grochow LB, et al. Pancreaticoduodenectomy for pancreatic adenocarcinoma: postoperative adjuvant chemoradiation improves survival. A prospective, single-institution experience. Ann Surg. 1997;225:621–33. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Sohn TA, Yeo CJ, Cameron JL, et al. Resected adenocarcinoma of the pancreas-616 patients: results, outcomes, and prognostic indicators. J Gastrointest Surg. 2000;4:567–79. [DOI] [PubMed] [Google Scholar]
- 16.Cameron JL, Pitt HA, Yeo CJ, et al. One hundred and forty-five consecutive pancreaticoduodenectomies without mortality. Ann Surg. 1993;217:430–8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Balcom JH, Rattner DW, Warshaw AL, et al. Ten-year experience with 733 pancreatic resections: changing indications, older patients, and decreasing length of hospitalization. Arch Surg. 2001;136:391–8. [DOI] [PubMed] [Google Scholar]
- 18.Birkmeyer JD, Finlayson SR, Tosteson AN, et al. Effect of hospital volume on in-hospital mortality with pancreaticoduodenectomy. Surgery. 1999;125:250–6. [PubMed] [Google Scholar]
- 19.Edge SB, Byrd DR, Compton CC, et al. , editors. AJCC cancer staging manual. 7th ed New York: Springer; 2010. [Google Scholar]
- 20.Washington K, Tang LH, Berlin J, et al. Protocol for the examination of specimens from patients with carcinoma of the endocrine pancreas. College of American Pathologists Cancer Protocols: Pancreas (exocrine) 2009. [Google Scholar]
- 21.Han CP, Lee MY, Tzeng SL, et al. Nuclear receptor interaction protein (NRIP) expression assay using human tissue microarray and immunohistochemistry technology confirming nuclear localization. J Exp Clin Cancer Res. 2008;27:25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Koo CL, Kok LF, Lee MY, et al. Scoring mechanisms of p16INK4a immunohistochemistry based on either independent nucleic stain or mixed cytoplasmic with nucleic expression can significantly signal to distinguish between endocervical and endometrial adenocarcinomas in a tissue microarray study. J Transl Med. 2009;7:25. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Greenland S, Perl J, Robins JM. Causal diagrams for epidemiologic research. Epidemiology. 1999;10:37–48. [PubMed] [Google Scholar]
- 24.White IR, Royston P, Wood AM. Multiple imputation using chained equations: issues and guidance for practice. Stat Med. 2011;30:377–99. [DOI] [PubMed] [Google Scholar]
- 25.R Core Team. R: a language and environment for statistical computing. Vienna: R Foundation for Statistical Computing; 2013. ISBN 3-900051-07-0, URL http://www.R-project.org/. [Google Scholar]
- 26.Iqbal MA, Gupta V, Gopin P, et al. Pyruvate kinase M2 and cancer: an updated assessment. FEBS Lett. 2014. 10.1016/j.febslet.2014.04.011 [DOI] [PubMed] [Google Scholar]
- 27.Lim JY, Yoon SO, Seol SY, et al. Overexpression of the M2 isoform of pyruvate kinase is an adverse prognostic factor for signet ring cell gastric cancer. World J Gastroenterol. 2012;18:4037–43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Mukherjee J, Phillips JJ, Wiencke J, et al. Pyruvate kinase M2 expression, but not pyruvate kinase activity, is up-regulated in a grade-specific manner in human glioma. PLoS One. 2013;8: e57610. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Zhang X, He C, He C, et al. Nuclear PKM2 expression predicts poor prognosis in patients with esophageal squamous cell carcinoma. Pathol Res Pract. 2013;209:510–5. [DOI] [PubMed] [Google Scholar]
- 30.Li J, Yang Z, Zou Q, et al. PKM2 and ACVR 1C are prognostic markers for poor prognosis of gallbladder cancer. Clin Transl Oncol. 2014;16:200–7. [DOI] [PubMed] [Google Scholar]
- 31.Rimm DL, Camp RL, Charette LA. Tissue microarray: a new technology for amplification of tissue resources. Cancer J. 2001;7:24–31. [PubMed] [Google Scholar]
- 32.Hathurusinghe HR, Goonetilleke KS, Siriwardena AK. Current status of tumor M2 pyruvate kinase (tumor M2-PK) as a biomarker of gastrointestinal malignancy. Ann Surg Oncol. 2007;14:2714–20. [DOI] [PubMed] [Google Scholar]
- 33.Kumar Y, Gurusamy K, Pamecha V, et al. Tumor M2-pyruvate kinase as a tumor marker in exocrine pancreatic cancer: a meta-analysis. Pancreas. 2007;35:114–9. [DOI] [PubMed] [Google Scholar]
- 34.Tamada M, Suematsu M, Saya H. Pyruvate kinase M2: multiple faces for conferring benefits on cancer cells. Clin Cancer Res. 2012;18:5554–61. [DOI] [PubMed] [Google Scholar]
- 35.Kumar Y, Mazurek S, Yang S, et al. In vivo factors influencing tumor M2-pyruvate kinase level in human pancreatic cancer cell lines. Tumour Biol. 2010;31:69–77. [DOI] [PubMed] [Google Scholar]


