Abstract
Aims/introduction
The predictive low glucose management (PLGM) system was introduced in March 2018 in Japan. Although there are some reports demonstrating the benefit of PLGM in preventing hypoglycemia, no data are currently available in Japanese patients with type 1 diabetes mellitus (T1DM). The aim of the present study is to evaluate the effect of PLGM with sensor-augmented pump therapy in the prevention of hypoglycemia in Japanese patients.
Materials and methods
We included 16 patients with T1DM who used the MiniMed®640G system after switching from the MiniMed®620G system. We retrospectively analysed the data of the continuous glucose monitoring system in 1 month after switching to MiniMed®640G.
Results
The area under the curve (AUC) of hypoglycemia of < 70 mg/dL was lowered from 0.42 ± 0.43 mg/dL day to 0.18 ± 0.18 mg/dL day (P = 0.012). Correspondingly, the duration of severe hypoglycemia (< 54 mg/dL) was reduced significantly from 15.3 ± 21.7 min/day to 4.8 ± 6.9 min/day (P = 0.019). The duration of hypoglycemia was reduced, but the reduction was not significant. Regarding the AUC for hyperglycemia > 180 mg/dL and the duration of hyperglycemia did not change. With the PLGM function, 79.3% of the predicted hypoglycemic events were avoided.
Conclusions
The hypoglycemia avoidance rate was comparable to those in previous reports. In addition, we demonstrated that PLGM can markedly suppress severe hypoglycemia without deteriorating glycemic control in Japanese T1DM patients. It is necessary to further investigate the effective use of the PLGM feature such as establishing a lower limit and the timing of resumption.
Keywords: Hypoglycemia, Predictive low glucose management (PLGM), Type 1 diabetes mellitus (T1DM), Sensor-augmented pump therapy (SAP)
Introduction
Currently, insulin pump therapy is one of the important treatment methods for type 1 diabetes mellitus (T1DM). Since sensor-augmented pumps (SAP) equipped with real-time continuous glucose monitoring (CGM) were launched in recent years, the management of glycemic control for individuals with T1DM has greatly advanced. Although these new medical devices such as CGM and SAP have enabled the prevention of severe hypoglycemia [1–6], the preventive effects have still not been satisfactory, especially in cases aiming for strict glycemic control. For these circumstances, the MiniMed®640G system (Medtronic, Northridge, CA, USA) was made commercially available in Japan in March 2018. This system includes a predictive low glucose management (PLGM) algorithm, in which insulin delivery is suspended when the sensor glucose (SG) value is predicted to be 20 mg/dL above the pre-set limit in 30 min. Previous studies have shown preventive effects on hypoglycemia in which 75–83% of cases of predicted hypoglycemia can be avoided without deteriorating glycemic control using the PLGM feature [7, 8], but the clinical utility of the device in Japan has remained unknown.
In this study, we investigated the hypoglycemia-preventive effect of switching from the MiniMed®620G (Medtronic, Northridge, CA, USA) system to the MiniMed®640G system in Japanese patients with T1DM. We analysed glycemic profiles with the use of CGM data before and after changing the equipment.
Materials and methods
This study was a retrospective observational study that was approved by the ethics committee of Okayama University Graduate School of Medicine, Dentistry and Pharmaceutical Sciences (approval no: ken1812-016, approval date: 14 December 2018). We switched from the MiniMed®620G system to the MiniMed®640G system between April 2018 and October 2018 for all 21 patients with T1DM who are on SAP therapy at Okayama University Hospital. We included only 16 of these patients (4 males and 12 females) because the inclusion criteria for this study was patients who used CGM for 5 days/week or more. Baseline characteristics of the participants are shown in Table 1.
Table 1.
Baseline characteristics of the participants
| Parameter | Mean ± standard deviation | Range |
|---|---|---|
| N (F) | 16 (12) | – |
| Age (years) | 46.1 ± 16.9 | 17–78 |
| BMI (kg/m2) | 22.2 ± 2.6 | 17.7–27.4 |
| Duration of diabetes (years) | 15.8 ± 12.6 | 2–44 |
| Experience with CSII (years) | 5.4 ± 2.7 | 1.3–7.8 |
| Experience with SAP (years) | 2.2 ± 1.0 | 0.8–3.9 |
| HbA1c (%) | 7.0 ± 0.8 | 5.8–8.6 |
| GA (%) | 19.5 ± 4.2 | 13.0–27.5 |
| TDD (μ/day) | 40.9 ± 13.0 | 22.2–64.9 |
| TBD (μ/day) | 12.2 ± 5.2 | 5.1–22.4 |
| %TBD (%) | 30.9 ± 14.4 | 17–46 |
| Carbohydrate input value (g/day) | 177 ± 64 | 91–251 |
BMI body mass index, CSII continuous subcutaneous insulin infusion, SAP sensor-augmented pump, GA glycated albumin, TDD total daily insulin dose, TBD total basal insulin dose, %TBD the percentage of the total basal insulin dose out of the total daily insulin dose
We retrospectively investigated the glycemic profile, insulin administration status and CGM data before and 1 month after changing to the MiniMed®640G system using the CGM analysis software (CareLink® Pro Therapy Management Software, Medtronic). Insulin administration status included total daily insulin dose (TDD), total basal insulin dose (TBD), and the percentage of total basal insulin dose to total daily insulin dose (%TBD); HbA1c and glycated albumin (GA) were used as the glycemic profile. We analysed the mean SG value, the number of insulin suspensions by PLGM (PLGM event), the duration of insulin suspension, severe hypoglycemia (< 54 mg/dL), hypoglycemia (< 70 mg/dL) and hyperglycemia (> 180 mg/dL), the areas under the curve (AUCs) for hypoglycemia and hyperglycemia and the carbohydrate values that the patients input for automatic bolus calculation as the CGM data. In addition, we investigated the SG value at the time of suspension initiation, the lowest SG value during suspension, the SG value at the resumption of insulin infusion and the SG value 1 h after initiating insulin infusion. All patients did not change the basal insulin setting or carbohydrate-to-insulin ratio after the initiation of PLGM, and all patients used the automatic bolus calculation function of the insulin pump. The preset lower limit value when the PLGM feature introduced was 70 mg/dL. Because previous report advocated that in case of suspension, patients should monitor the trend and delay hypoglycemia treatment to ‘‘let the pump do the work’’ [9], we recommended all patients not to resume insulin infusion manually during suspension. However, we also advised them to resume insulin infusion manually at meals, even in suspension mode, because they could not administer bolus insulin infusion while suspended. In addition, we instructed patients not to perform preventive supplementation as much as possible during suspension.
Statistical analysis
All data are presented as mean ± SD. Differences in HbA1c, GA, TDD, TBD, %TBD, and the carbohydrate values measured before and after the change in treatment were analysed with the Wilcoxon signed-rank test. Other statistical data were assessed with a Mann–Whitney U test. All statistical analyses were performed with SPSS software version 20 (IBM SPSS statistics). A P value of < 0.05 was considered statistically significant.
Results
Glycemic control markers and insulin administration status
Both the HbA1c and GA values were unchanged from before to after the PLGM feature was introduced (HbA1c: 7.0 ± 0.8% vs. 7.0 ± 0.9%, P = 0.867, GA: 19.5 ± 4.2% vs. 20.1 ± 3.5%, P = 0.400). TDD, TBD and %TBD all decreased after the PLGM feature was introduced, but this reduction was not statistically significant (Table 2).
Table 2.
Comparison of glycemic control markers and insulin administration status before and after PLGM was introduced
| Baseline | PLGM | P | |
|---|---|---|---|
| HbA1c (%) | 7.0 ± 0.8 | 7.0 ± 0.9 | 0.867 |
| GA (%) | 19.5 ± 4.2 | 20.1 ± 3.5 | 0.400 |
| TDD (μ/day) | 40.9 ± 13.0 | 38.1 ± 11.4 | 0.564 |
| TBD (μ/day) | 12.2 ± 5.2 | 11.0 ± 5.2 | 0.402 |
| %TBD (%) | 30.9 ± 14.4 | 29.3 ± 13.2 | 0.616 |
| Carbohydrate input value (g/day) | 177 ± 64 | 189 ± 73 | 0.696 |
All data are presented as the mean ± standard deviation (SD)
GA glycated albumin, TDD total daily insulin dose, TBD total basal insulin dose, %TBD the percentage of the total basal insulin dose out of the total daily insulin dose
CGM data
There was no significant change in mean SG value, the duration of hyperglycemia or the AUC of hyperglycemia > 180 mg/dL from before to after the PLGM feature was introduced. No significant decrease was observed in the duration (58.2 ± 49.4 min/day vs. 30.1 ± 25.9 min/day, P = 0.067) of hypoglycemia. In contrast, the duration of severe hypoglycemia (15.3 ± 21.7 min/day vs. 4.8 ± 6.9 min/day, P = 0.019) and the AUC of hypoglycemia < 70 mg/dL (0.42 ± 0.43 mg/dL day vs. 0.18 ± 0.18 mg/dL day, P = 0.012) were both significantly reduced (Table 3). There were no episodes of diabetic ketoacidosis (DKA) or serious device-related adverse events during the research period.
Table 3.
Comparison of the CGM data before and after PLGM was introduced
| Baseline | PLGM | P | |
|---|---|---|---|
| SG value (mg/dL) | 155.3 ± 34.0 | 152.9 ± 44.2 | 0.669 |
| Time > 180 mg/dL (min/day) | 421.3 ± 262.0 | 440.4 ± 290.1 | 0.926 |
| AUC > 180 (mg/dL/day) | 17.8 ± 19.2 | 16.1 ± 14.0 | 0.926 |
| Time < 70 mg/dL (min/day) | 58.2 ± 49.4 | 30.1 ± 25.9 | 0.067 |
| Time < 54 mg/dL (min/day) | 15.3 ± 21.7 | 4.8 ± 6.9 | 0.019* |
| AUC < 70 (mg/dL/day) | 0.42 ± 0.43 | 0.18 ± 0.18 | 0.012* |
SG sensor glucose, AUC area under the curve
All data are presented as the mean ± standard deviation (SD), *P < 0.05
PLGM operation status and hypoglycemia avoidance rate
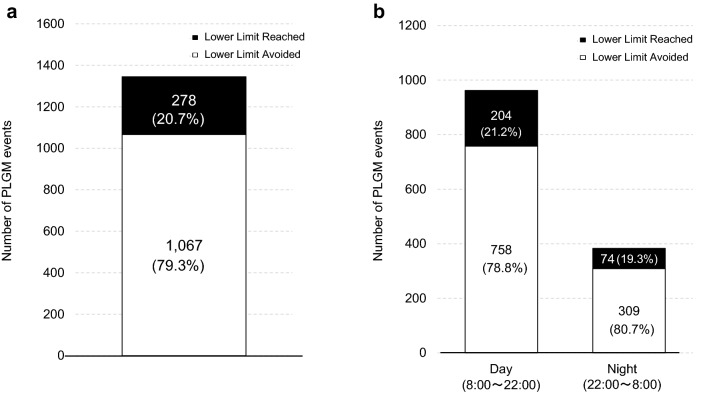
There were 1345 PLGM events during the research period, for a rate of 3.0 ± 1.2 per subject per day. In addition, the overall mean duration of suspension before hypoglycemic events was 156 ± 46 min/day. Moreover, 1067 out of the 1345 events did not reach the preset threshold; that is, 79.3% of the predicted hypoglycemia was avoided (Fig. 1a). This hypoglycemia avoidance rate was similar in the daytime (8:00 until 22:00) and night-time (22:00 until 8:00) (Fig. 1b).
Fig. 1.
The number of PLGM events and the hypoglycemia avoidance rate, a over 24 h, b during the daytime and night-time
The change in SG value during suspension and after resumption of insulin infusion
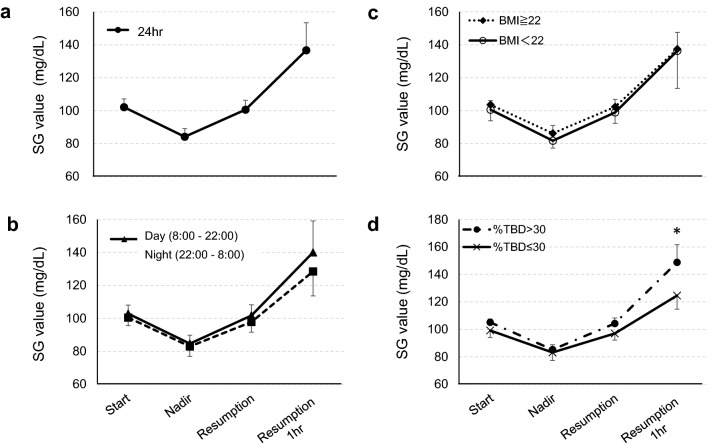
We further investigated the SG value at the time of suspension initiation (start), the lowest SG value during suspension (nadir), the SG value at the resumption (resumption) and the SG value 1 h after resumption (resumption 1 h). The mean SG value 1 h after resumption increased to 136.7 ± 16.7 mg/dL (Fig. 2a), and the mean suspension duration was 55.2 ± 10.6 min. In the comparison of daytime and nighttime SG values, it was determined that the mean SG value at the start, nadir, resumption and resumption 1 h were all higher in the daytime, but the differences were not significant (start: 102.8 ± 5.2 mg/dL vs. 100.5 ± 4.9 mg/dL, P = 0.110, nadir: 84.6 ± 5.1 mg/dL vs. 82.9 ± 6.1 mg/dL, P = 0.323, resumption: 101.7 ± 6.5 mg/dL vs. 97.8 ± 6.2 mg/dL, P = 0.094, resumption 1 h: 140.1 ± 19.1 mg/dL vs. 128.5 ± 14.8 mg/dL, P = 0.110) (Fig. 2b). In addition, the mean suspension time was significantly longer in the night-time than in the daytime (49.1 ± 10.2 min vs. 70.2 ± 16.0 min, P < 0.0005).
Fig. 2.
The change in the SG value during suspension and after resumption of insulin infusion. a All PLGM data, b classified by event starting time: daytime and night-time. The differences between day and night are not significant. c Classified by the BMI of patients. The differences between high BMI group and low BMI group are not significant. d Classified by %TBD of patients. The SG value at resumption 1 h was significantly higher in the high %TBD group than in the low %TBD group. SG sensor glucose, All data are presented as the mean ± standard deviation (SD). *Statistically significant, P < 0.05
Furthermore, we compared the SG value and the suspension time by 2-quantile groups such as BMI (high BMI group with BMI ≥ 22, n = 8; low BMI group with BMI < 22, n = 8), TDD (high TDD group with TDD ≥ 38.7, n = 8; low TDD group with TDD < 38.7, n = 8), TBD (high TBD group with TDD ≥ 17.8, n = 8; low TDD group with TDD < 17.8, n = 8) and %TBD (high %TBD group with %TBD > 30, n = 8; low %TBD group with %TBD ≤ 30, n = 8). We did not find a difference in SG value by BMI (Fig. 2c), TDD and TBD at any time point. Similarly, there was no difference in the mean suspension time. In contrast, the SG value at resumption 1 h was significantly higher in the high %TBD group than in the low %TBD group (148.8 ± 12.9 mg/dL vs. 124.6 ± 9.9 mg/dL, P = 0.001) (Fig. 2d) without differences of the mean suspension time (57.2 ± 8.4 min vs. 53.2 ± 12.1 min, P = 0.645).
Hyperglycemia after suspension
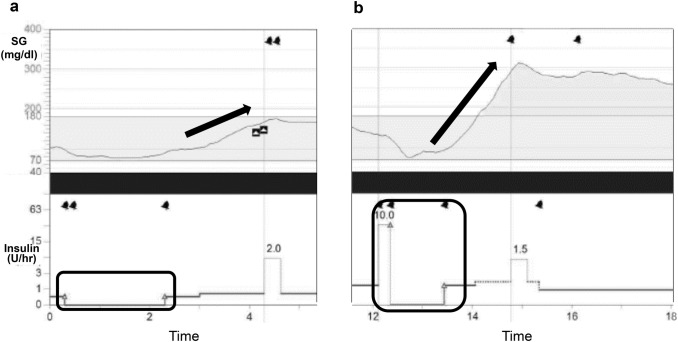
There were some cases in which the duration of hyperglycemia was increased by completely relying on the algorithm. Figure 3a shows a case of hyperglycemia after 2 h suspension while sleeping. Figure 3b shows a case of severe hyperglycemia after suspension of insulin infusion because PLGM started just after meal.
Fig. 3.
The cases of hyperglycemia after suspension. a A case of hyperglycemia after 2 h suspension at bedtime. b A case of severe hyperglycemia after suspension of insulin infusion because PLGM started just after a meal. Arrows show the degree of SG value increase, and squares indicate the time during which PLGM was operating
Discussion
Despite the advances in medicine and medical devices, hypoglycemia remains as a critical issue in the treatment of T1DM. In recent years, the MiniMed®640G system equipped with a PLGM function became available, and an improvement in the hypoglycemia avoidance rate is expected. We analysed hypoglycemia prevention in Japanese T1DM patients using SAP therapy before and after switching from the MiniMed®620G system to the MiniMed®640G system. In this study, there was a declining trend in the duration of hypoglycemia, and the duration of severe hypoglycemia and the AUC of < 70 mg/dL were both significantly reduced. The hypoglycemia avoidance rate was 79.3%, which was almost the same in both the daytime and night-time. Moreover, the duration of hyperglycemia, the AUC of > 180 mg/dL, and the values of HbA1c and GA did not increase. From these results, it was shown that the risk of hypoglycemia is lowered using the PLGM feature without deteriorating glycemic control, at least in the short term.
Similar results have been reported in Western countries. Zhong A and colleagues retrospectively examined the effect of PLGM, and reported that both hypoglycemic (SG < 70 mg/dL) and hyperglycemic (SG > 240 mg/dL) exposure time decreased in patients using PLGM [8]. Biester et al. reported that the frequency and duration of hypoglycemia and the AUC < 70 mg/dL decreased significantly in a prospective study of children with T1DM using PLGM for 6 weeks [9]. In addition, Battelino T and colleagues carried out a randomized controlled trial of children with T1DM divided into two groups with or without the use of PLGM for 14 days. They showed that the incidence of hypoglycemic events with SG values < 65 mg/dL was significantly reduced in the PLGM-treated group during both daytime and night-time, and there was no increase in hyperglycemic exposure time at any level (SG value > 140 mg/dL, > 180 mg/dL and > 250 mg/dL) [10]. However, their data demonstrated that PLGM did not prevent severe hypoglycemia below 50 mg/dL, which is different from our results. Battelino T and colleagues considered that the participants in their study were relatively well managed, the study period was very short, and the overall number of hypoglycemic events below 50 mg/dL was too small to provide statistically significant results. More recently, the results of a randomized controlled trial on the presence or absence of 6 months of PLGM use in 154 children and adolescent patients with T1DM were reported. This trial showed that the incidence of hypoglycemic events with SG values < 63 mg/dL decreased significantly in the PLGM group and that the HbA1c value at 6- month was not significantly different between the two groups: 7.6 ± 1.0% in the non-PLGM group and 7.8 ± 0.8% in the PLGM group. This result indicated that even when using PLGM for a long time, glycemic control does not deteriorate [11]. Besides that, Maahs et al. reported that the suspension system reduced nocturnal hypoglycemia (< 60 mg/dL) by 12%, median hypoglycemia area under the curve by 81% and hypoglycemia lasting > 2 h by 74%. They also reported that overnight hyperglycemia (> 180 mg/dL) was almost in same level with or without suspension [12]. Regarding the hypoglycemia avoidance rate, Choudhary et al. reported that 82.7% and 84.0% of predicted hypoglycemia was avoidable in the daytime and the night-time, respectively, using the PLGM feature [7], while Zhong et al. reported that the avoidance rate was 73.9% and 77.4%, respectively [8]. Since our study also showed a rate of hypoglycemia avoidance similar to those in these reports, PLGM could be expected to suppress hypoglycemic events in Japanese patients with T1DM.
Next, we investigated the change in the SG value during suspension and after resumption of insulin infusion. We confirmed that the SG value increased 1 h after resumption, which is the same as the findings of previous reports [7, 9, 13–15]. When comparing daytime and night-time, the suspension time was significantly longer at night, and the SG value at the start, resumption and resumption 1 h tended to be lower at night. Biester et al. reported similar results in which the SG value 1 h after resumption was lower at night (174 mg/dL in the daytime vs. 137 mg/dL in the night-time), and the suspension time during daytime was shorter than during night-time (54 min vs. 68 min) [9]. They discussed that manual resumption was performed at approximately 46% after insulin suspension, and as a result, the suspension time was shorter during the daytime, and the rise in SG value 1 h after resumption was noted in their article. Although, we instructed the participants to avoid manual resumption and preventive carbohydrate intake after suspension was initiated as much as possible after the introduction of the PLGM feature, we obtained a similar result as those previously reported. However, there was no significant difference in SG value after suspension between the daytime and night-time, and it was considered that higher SG values in the daytime were prevented by instructing the patients to follow the insulin pump algorithm. The reason the suspension time during the daytime was longer than during the night-time is that it was necessary to resume insulin infusion manually at the time of bolus administration at a meal. Moreover, since we could not confirm the manual resumption rate, there is a possibility that manual resumption was carried out to the same extent as in the previous report. These results suggested that better glycemic control could be obtained by following the insulin pump algorithm rather than resuming the insulin infusion manually after PLGM was initiated.
In addition, we speculated that the time-course changes in the SG value during and after suspension might be influenced by BMI, TDD, TBD or %TBD, so we compared the SG value and the suspension time classified with these parameters. Though there were no differences in the SG value and mean suspension time when classified with BMI, TDD, and TBD, we found the differences of the SG value at resumption 1 h classified with %TBD. The reason for this is not clear, but in the low %TBD group, relatively large amount of bolus insulin might overlay and mask the lack of basal insulin supplement while basal insulin suspension, resulting in suppression of SG elevation after PLGM.
As shown in Fig. 3, there were some cases of hyperglycemia after suspension. Zisser H examined how much blood glucose would rise after interrupting insulin delivery. He demonstrated the rate of rise in glucose concentration over 3 h was ~ 1 mg/dL for each minute insulin infusion was interrupted [16]. Sherr et al. also reported the sensor glucose level rose by 18 ± 58 mg/dL by the end of the 2 h suspension, and by 55 ± 73 mg/dL 4 h after the suspension [17], and Fig. 3a follows their findings. Figure 3b shows a rebound hyperglycemia after suspension. In this case, PLGM started just after meal and the participant ingested carbohydrates without resumption, resulting in rebound hyperglycemia. Furthermore, this participant may have consumed excess amount of carbohydrate during hypoglycemia. Collectively, we recognized that early insulin resumption was necessary to prevent rebound hyperglycemia after carbohydrate intake for hypoglycemia, and we also need to inquire the patient about amount of carbohydrate during hypoglycemia. Although the initial setting of the lower limit was 70 mg/dL in this study, depending on the case, the timing of PLGM initiation was early or the timing of the resumption of insulin infusion was late, and the subsequent increase in the SG value was marked in some cases. Even though similar severe rebound hyperglycemia after PLGM has been reported, such risk is believed to be very low [14, 15, 17, 18]. Indeed, there were no cases with marked hyperglycemia with ketoacidosis in our study.
This study had several limitations. First, this was a small retrospective observational study in a single facility. Second, the participants were relatively well-managed patients with T1DM (mean HbA1c 7.0%), so the frequency of hypoglycemia and hyperglycemia may be low. Third, the current research period is short, only 1 month before and after PLGM introduction. Because of short research period, we could not assess the sustained effects on metabolic control and HbA1c. Most of the published studies were limited to short-term evaluation for 2–6 weeks [7, 9, 10, 12]. Under longer observation period for more than 2 months, other factors besides PLGM, such as a change in pump settings and lifestyle. Therefore, we set 1 month to assess the real clinical picture of PLGM. In addition, since we conducted thorough education for the patients before the introduction of MiniMed®640G system to use the new system safely and effectively, we obtained such good results. Besides these, we did not consider physical activity in this study. Recently, interesting results were reported that assessed the optimal setting of the PLGM algorithm for preventing exercise-induced hypoglycemia in adolescents with T1DM [19]. They concluded that setting a PLGM threshold to 90 mg/dL during night in adolescents performing frequent physical exercise reduced time of hypoglycemia; however, a threshold of 70 mg/dL seems to be safe during physical exercise.
It is necessary to further investigate the setting of the lower limit according to each case, time zone, and the frequency of physical exercise; the timing of manual resumption; and the approach of preventive carbohydrate intake against hypoglycemia. Finally, although PLGM is a very effective mechanism, there are some points to be addressed, such as the fact that hypoglycemia cannot be avoided completely and that there is a possibility of severe hyperglycemia after suspension of insulin infusion. Therefore, when introducing PLGM, it is necessary to take sufficient time to explain the features and precautions of this system to patients and to ensure that they use it with full understanding.
Conclusion
In this research, we demonstrated the short-term effects in which the PLGM feature can markedly suppress hypoglycemia, particularly severe hypoglycemia, without deteriorating glycemic control in Japanese patients with T1DM. Further examination is necessary to determine the long-term effects and to further improve the hypoglycemia avoidance rate.
Acknowledgements
We thank the participants of this study.
Compliance with ethical standards
Conflict of interest
Author Atsuhito T. received lecture fees from Medtronic Japan, Sanofi and Eli Lilly. Author Jun W. received lecture fees from Astellas, Astra Zeneca, Boeringer Ingelheim Japan, Daiichi Sankyo, MSD, Novartis, Tanabe Mitsubishi and Taisho Toyama, and received research funding from Bayer, Baxter, Chugai, Dainippon Sumitomo, Kyowa Hakko Kirin, MSD, Novartis, Novo Nordisk, Ono, Takeda, Tanabe Mitsubishi and Teijin. Other authors declare that they have no conflict of interest associated with this research.
Ethical standards
All procedures followed were in accordance with the ethical standards of the responsible committee on human experimentation (institutional and national) and with the Helsinki Declaration of 1964 and later versions. This study was approved by the ethics committee of Okayama University Graduate School of Medicine, Dentistry and Pharmaceutical Sciences (approval no: ken1812-016, approval date: 14 December 2018). We did not receive the informed consent, but we provided the participants with the opportunity to deny by publishing the Opt Out document.
Footnotes
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Beck RW, Riddlesworth T, Ruedy K, Ahmann A, Bergenstal R, Haller S, Kollman C, Kruger D, McGill JB, Polonsky W, Toschi E, Wolpert H, Price D, DIAMOND study group Effect of continuous glucose monitoring on glycemic control in adults with type 1 diabetes using insulin injections: the DIAMOND randomized clinical trial. JAMA. 2017;317(4):371–378. doi: 10.1001/jama.2016.19975. [DOI] [PubMed] [Google Scholar]
- 2.Heinemann L, Freckmann G, Ehrmann D, Faber-Heinemann G, Guerra S, Waldenmaier D, Hermanns N. Real-time continuous glucose monitoring in adults with type 1 diabetes and impaired hypoglycemia awareness or severe hypoglycemia treated with multiple daily insulin injections (HypoDE): a multicentre, randomised controlled trial. Lancet. 2018;391(10128):1367–1377. doi: 10.1016/S0140-6736(18)30297-6. [DOI] [PubMed] [Google Scholar]
- 3.Matsuoka A, Hirota Y, Urai S, Hamaguchi T, Takeuchi T, Miura H, Suematsu N, So A, Nakamura T, Komada H, Okada Y, Sakaguchi K, Ogawa W. Effect of switching from conventional continuous subcutaneous insulin infusion to sensor augmented pump therapy on glycemic profile in Japanese patients with type 1 diabetes. Diabetol Int. 2018;9(3):201–7. doi: 10.1007/s13340-018-0344-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Battelino T, Conget I, Olsen B, Schütz-Fuhrmann I, Hommel E, Hoogma R, Schierloh U, Sulli N, Bolinder J, SWITCH study group The use and efficacy of continuous glucose monitoring in type 1 diabetes treated with insulin pump therapy: a randomized controlled trial. Diabetologia. 2012;55(12):3155–3162. doi: 10.1007/s00125-012-2708-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bergenstal RM, Tamborlane WV, Ahmann A, Buse JB, Dailey G, Davis SN, Joyce C, Peoples T, Perkins BA, Welsh JB, Willi SM, Wood MA, STAR 3 study group Effectiveness of sensor augmented insulin-pump therapy in type 1 diabetes. N Engl J Med. 2010;363(4):311–320. doi: 10.1056/NEJMoa1002853. [DOI] [PubMed] [Google Scholar]
- 6.Bosi E, Choudhary P, de Valk HW, Lablanche S, Castañeda J, de Portu S, Da Silva J, Ré R, Vorrink-de Groot L, Shin J, Kaufman FR, Cohen O, SMILE study group Efficacy and safety of suspend-before-low insulin pump technology in hypoglycaemia-prone adults with type 1 diabetes (SMILE): an open-label randomised controlled trial. Lancet Diabetes Endocrinol. 2019;7(6):462–472. doi: 10.1016/S2213-8587(19)30150-0. [DOI] [PubMed] [Google Scholar]
- 7.Choudhary P, Olsen BS, Conget I, Welsh JB, Vorrink L, Shin JJ. Hypoglycemia prevention and user acceptance of an insulin pump system with predictive low glucose management. Diabetes Technol Ther. 2016;18(5):288–291. doi: 10.1089/dia.2015.0324. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Zhong A, Choudhary P, McMahon C, Agrawal P, Welsh JB, Cordero TL, Kaufman FR. Effectiveness of automated insulin management features of the MiniMed® 640G sensor-augmented insulin pump. Diabetes Technol Ther. 2016;18(10):657–663. doi: 10.1089/dia.2016.0216. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Biester T, Kordonouri O, Holder M, Remus K, Kieninger-Baum D, Wadien T, Danne T. “Let the algorithm do the work”: reduction of hypoglycemia using sensor-augmented pump therapy with predictive insulin suspension (SmartGuard) in pediatric type 1 diabetes patients. Diabetes Technol Ther. 2017;9(3):173–182. doi: 10.1089/dia.2016.0349. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Battelino T, Nimri R, Dovc K, Phillip M, Bratina N. Prevention of hypoglycemia with predictive low glucose insulin suspension in children with type 1 diabetes: a randomized controlled trial. Diabetes Care. 2017;40(6):764–770. doi: 10.2337/dc16-2584. [DOI] [PubMed] [Google Scholar]
- 11.Abraham MB, Nicholas JA, Smith GJ, Fairchild JM, King BR, Ambler GR, Cameron FJ, Davis EA, Jones TW, PLGM study group Reduction in hypoglycemia with the predictive low-glucose management system: a long-term randomized controlled trial in adolescents with type 1 diabetes. Diabetes Care. 2018;41(2):303–310. doi: 10.2337/dc17-1604. [DOI] [PubMed] [Google Scholar]
- 12.Maahs DM, Calhoun P, Buckingham BA, Chase HP, Hramiak I, Lum J, Cameron F, Bequette BW, Aye T, Paul T, Slover R, Wadwa RP, Wilson DM, Kollman C, Beck RW, In Home Closed Loop Study Group A randomized trial of a home system to reduce nocturnal hypoglycemia in type 1 diabetes. Diabetes Care. 2014;37(7):1885–1891. doi: 10.2337/dc13-2159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Bergenstal RM, Klonoff DC, Garg SK, Bode BW, Meredith M, Slover RH, Ahmann AJ, Welsh JB, Lee SW, Kaufman FR, ASPIRE In-Home Study Group Threshold-based insulin-pump interruption for reduction of hypoglycemia. N Engl J Med. 2013;369(3):224–232. doi: 10.1056/NEJMoa1303576. [DOI] [PubMed] [Google Scholar]
- 14.Agrawal P, Zhong A, Welsh JB, Shah R, Kaufman FR. Retrospective analysis of the real-world use of the threshold suspend feature of sensor-augmented insulin pumps. Diabetes Technol Ther. 2015;17(5):316–319. doi: 10.1089/dia.2014.0257. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Buckingham BA, Cameron F, Calhoun P, Maahs DM, Wilson DM, Chase HP, Bequette BW, Lum J, Sibayan J, Beck RW, Kollman C. Outpatient safety assessment of an in-home predictive low-glucose suspend system with type 1 diabetes subjects at elevated risk of nocturnal hypoglycemia. Diabetes Technol Ther. 2013;15(8):622–627. doi: 10.1089/dia.2013.0040. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Zisser H. Quantifying the impact of a short-interval interruption of insulin-pump infusion sets on glycemic excursions. Diabetes Care. 2008;31(2):238–239. doi: 10.2337/dc07-1757. [DOI] [PubMed] [Google Scholar]
- 17.Sherr JL, Palau Collazo M, Cengiz E, Michaud C, Carria L, Steffen AT, Weyman K, Zgorski M, Tichy E, Tamborlane WV, Weinzimer SA. Safety of nighttime 2-h suspension of basal insulin in pump-treated type 1 diabetes even in the absence of low glucose. Diabetes Care. 2014;37(3):773–779. doi: 10.2337/dc13-1608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Calhoun PM, Buckingham BA, Maahs DM, Hramiak I, Wilson DM, Aye T, Clinton P, Chase P, Messer L, Kollman C, Beck RW, Lum J, In Home Closed Loop Study Group Efficacy of an overnight predictive low-glucose suspend system in relation to hypoglycemia risk factors in youth and adults with type 1 diabetes. J Diabetes Sci Technol. 2016;10(6):1216–1221. doi: 10.1177/1932296816645119. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Cherubini V, Gesuita R, Skrami E, Rabbone I, Bonfanti R, Arnaldi C, D’Annunzio G, Frongia A, Lombardo F, Piccinno E, Schiaffini R, Toni S, Tumini S, Tinti D, Cipriano P, Minuto N, Lenzi L, Ferrito L, Ventrici C, Ortolani F, Cohen O, Scaramuzza A. Optimal predictive low glucose management (PLGM) settings during physical exercise in adolescents with type 1 diabetes. Pediatr Diabetes. 2019;20(1):107–112. doi: 10.1111/pedi.12792. [DOI] [PubMed] [Google Scholar]