Abstract
Bone marrow chimeras represent a key tool employed to understand biological contributions stemming from the hematopoietic versus the stromal compartment. In most institutions, cesium irradiators are used to lethally irradiate recipient animals prior to the injection of donor bone marrow. Cesium irradiators, however, have significant liabilities—including concerns around domestic security. Recently, X-ray irradiators have been implemented as a potential alternative to cesium sources. Only a small number of publications in the literature have attempted to compare these two modalities and, in most cases, the emphasis was on irradiation of human blood productions. We were able to find only a single study that directly compared X-ray and cesium technologies in the generation of murine bone marrow chimeras, a standard laboratory practice. This study focused on chimerism in the blood of recipient animals. In the present study, we begin by comparing cesium and X-ray based sources for irradiation, then transition to using X-ray-based systems for immunology models with an emphasis on immunotherapy of cancer in immunocompetent mouse models—specifically evaluating chimerism in the blood, spleen, and tumor microenvironment. While our data demonstrate that the two platforms are functionally comparable and suggest that X-ray based technology is a suitable alternative to cesium sources. We also highlight a difference in chimerism between the peripheral (blood, spleen) and tumor compartments that is observed using both technologies. While the overall degree of chimerism in the peripheral tissues is very high, the degree of chimerism in the tumor is cell type specific with T and NK cells showing lower chimerism than other cell types.
Keywords: immunotherapy, bone marrow chimera, X-ray irradiation, cesium irradiation, tumor model, syngeneic
INTRODUCTION
Animals that receive a lethal dose of irradiation and are reconstituted with exogenous bone marrow are known as bone marrow chimeras. The generation of bone marrow chimeras allow investigators to isolate the stromal versus the hematopoietic compartment when evaluating the contribution of a particular gene to an observed phenotype [1]. The generation of bone marrow chimeras has traditionally relied on a cesium irradiator (Cesium137) and most protocols described in the literature focus on this form of radiation. However, cesium irradiators are currently being targeted for replacement by the Office of Radiological Security (ORS), a division within the National Nuclear Security Administration in the Department of Energy [2]. The overall goal of the ORS is to promote the use of alternative radiation sources that do not present a potential security risk. In addition, many of the cesium sources that are in use today were instituted decades ago. The efficiency of these cesium irradiators has decreased over time due to significant radio-isotopic decay, therefore requiring costly annual maintenance fees.
Unfortunately, there are limited publications describing how best to integrate non-cesium based technology into a working laboratory. Many publications focus on the irradiation of human blood products, reflecting a common use of this equipment in the hospital setting [3,4]. We were able to find only a single study that directly compared X-ray and cesium technologies in the generation of murine bone marrow chimeras [5], a standard laboratory practice. We have recently purchased and validated an X-ray based irradiator for use in basic scientific research. In the current study, we compared cesium and X-ray-based technologies to generate bone marrow chimeras for future work and demonstrate how these chimeras can be used for the study of the immune system, particularly with regards to cancer immunotherapy. We show that X-ray-sourced irradiators provide a comparable ability to generate chimeras and are well suited for basic scientific research. Here, we provide a practical protocol describing use of an X-ray irradiator for the establishment of bone marrow chimeras. Furthermore, we describe additional considerations for using these animals to study therapeutics that target the immune system in the context of cancer.
MATERIALS AND METHODS
Animals
6–8 week old female C57BL6 and CD45.1 congenic animals were purchased from Jackson Labs. All experimental studies were conducted under protocols approved by the Institutional Animal Care and Use Committee of Amgen. Animals were housed at Association for Assessment and Accreditation of Laboratory Animal Care International-accredited facilities (at Amgen) in ventilated micro-isolator housing on corncob bedding. Animals had access ad libitum to sterile pelleted food and reverse osmosis-purified water and were maintained on a 12:12 hour light:dark cycle with access to environmental enrichment opportunities. Following irradiation, animals were given neomycin sulfate in drinking water (1.1 mg/ml) to prevent infection for five days.
Irradiation
Animals were irradiated either with a Cesium-137 irradiator model 143-68R (JL Shepherd, San Fernando, CA) or a MultiRad 350 (Faxitron, Tucson, AZ). For the cesium irradiator, animals were irradiated based on the intended dosage in gray for a duration calculated based on the isotopic emission. Typically, animals received between 7 and 11 grays of radiation in total. The overall radiation dose was divided into two separate and equal doses, given 4–6 h apart. For the X-ray irradiator, efforts were made to deliver equivalent dose per unit time. That is, the machine was programmed to 3.5–5.5 grays per dose, with an approximate dose rate of 2 grays per minute and the current set to 11.4 mA and 350 kV. The X-ray machine was operated with the 0.75 mm tin, 0.25 mm copper, and 1.5 mm aluminum beam conditioning filter (Thoraeus filter) installed and the internal shelf set to the 4th position.
Bone marrow isolation and reconstitution of irradiated animals
Bone marrow was harvested from the femur of donor animals using standard techniques [6]. Red blood cells were removed from the bone marrow with an ACK lysis (Thermo Fisher, Waltham, MA). Cells were resuspended to a final concentration of 10–25 × 106 cells/ml in phosphate buffered saline (PBS) and 200 μl of cells were injected into each irradiated recipient animal for a final delivered dose of 2–5 × 106 cells/recipient.
Analysis of chimerism: Preparation of samples
For terminal analysis of chimerism, animals were bled by cardiac puncture under isoflurane anesthesia. For interim analysis of chimerism, animals were bled by retro-orbital route to collect 75–100 μl of whole blood. Blood was collected into EDTA tubes (Becton Dickinson, Franklin Lakes, NJ) and used for flow cytometry analysis.
Spleens were injected with digestion buffer and incubated for 15 min at 37°C. Enzymatic digestion buffer was prepared as RPMI + 2% FBS + 0.2 mg/ml Liberase TL (Roche, Basel, Switzerland) and 20 U/ml DNase I (Roche). After incubation, the spleens were quenched with FBS. Dissociated spleens were passed through a 100 micron mesh filter and centrifuged at 1500 rpm for 5 min at 4°C. The pellet of cells was ACK lysed to remove red blood cells and resuspended in PBS with 2% FBS. Cells were filtered a second time through a 40 micron mesh filter.
Analysis of chimerism: Flow cytometry
Cells were stained with the following cell surface antibodies for a lymphoid panel: CD19 clone 1D3, CD4 clone RM4-5, CD8 clone 53-6.7, CD11b clone M1/70, TCRb clone H57-597, CD105 clone MJ7/18, NKp46 clone 29A1.4, CD45.2 clone 104, CD45.1 clone A20 (all from BD Biosciences, San Jose, CA) and PD1 clone J43 (eBioscience, San Diego, CA). In addition, cells were stained for a myeloid panel with the following antibodies: Ly6G clone IA8, CD24 clone M1/69 (from BD Biosciences); F4/80 clone BM8, TCRb clone H57-597, CD19 clone 6D5, NKp46 clone 29A1.4, Ly6C clone HK11.4, CD105 clone MJ7/18, Cd86 clone GL-1, CD11c clone N418, CD11b clone M1/70 (from Biolegend, San Diego, CA); and PDL1 clone MIH5 and MHC II clone M5/114.15 (eBioscience). All flow cytometry data was analyzed in FlowJo (FlowJo LLC, Ashland, OR).
Implantation and evaluation of syngeneic tumors
Mouse melanoma (cell line B16F10) or mouse colon carcinoma cells (cell line MC38) were injected subcutaneously in the right flank of mice at the indicated cell inoculum. Tumor volume (mm3) was measured using electronic calipers (Fowler High Precision, Newtown, MA) twice a week. All tumor measurements were recorded in Studylog software (Studylog Systems, South San Francisco, CA).
Analysis of tumor immune infiltrates in syngeneic tumors
To evaluate immune infiltrates within subcutaneous tumors, the tumors were excised and dissociated by enzymatic digestion. Tumors were digested using a Gentle MACS Dissociator (Miltenyi Biotec, San Diego, CA) in C-tubes. Enzymatic digestion buffer was prepared as RPMI + 2% FBS + 0.2 mg/ml Liberase TL (Roche, Basel, Switzerland) and 20 U/ml DNase I (Roche). Post-digestion, the tumor slurry was passed through a 100-micron mesh filter, centrifuged at 1500 rpm for 5 min at 4°C, filtered again with a 40 micron filter, and resuspended in FACS buffer for staining. Tumor derived cells were stained as described in the section above (Analysis of chimerism).
Analysis of tumor growth response to treatment in syngeneic tumors
For treatment studies, animals were randomized into treatment groups (10 animals per group) once tumors reached an average of approximately 75–100 mm3. The average tumor volume at the beginning of treatment was uniform across treatment groups. Upon randomization, animals were treated with anti-CTLA4 (clone 9D9 formatted on a mouse IgG2a backbone), anti-PD1 (clone J32 or 29F1A12 formatted on a mouse IgG1 N297G backbone), or an isotype control reagent. Animals were dosed three times, three days apart starting with the day of randomization. Body weight was measured once weekly and tumor volumes were measured twice weekly until the end of study.
Statistical analysis of tumor studies
Comparison of tumor volumes over time were conducted by comparing median survival of each group to an arbitrary tumor volume of greater than 800 mm3. Median survival for each group was compared using a Log-rank (Mantel-Cox) test in Prism (GraphPad, San Diego, CA). For chimerism and reconstitution analyses, all groups evaluated were compared to each other using a one-way ANOVA with multiple comparisons test in Prism.
RESULTS
To begin, we evaluated the reconstitution of hematopoietic cells in animals that had been irradiated using an X-ray based source. The X-ray irradiator used was purchased from Faxitron and irradiation was performed using the Thoraeus filter, a hybrid filter composed of tin, copper, and aluminum. This filter provides beam hardening at the expense of a lower dose rate. However, the overall dose rate was adjusted to provide roughly the same irradiation rate constant as the cesium irradiator used in these experiments. While previous publications had suggested that X-ray irradiation could provide comparable relative biological equivalent (RBE) at lower doses [3], our initial experiments compared a dose range of X-ray irradiation. A total dose range of 7 to 11 grays was performed. Animals were irradiated twice, with half the total dose delivered approximately four hours apart, prior to the introduction of congenic labeled bone marrow (CD45.1+). Animals were then provided antibiotics in drinking water supplied ad libitum for 5 d post irradiation to prevent any infections due to immunosuppression. Six to eight weeks later, animals were euthanized to determine the degree of chimerism in the blood and spleen. In these experiments, the recipient animal was CD45.1 (congenic) and the donor bone marrow was CD45.2 (WT for C57BL6). The overall degree of chimerism in the blood (Fig. 1A-1C) and the spleen (Fig. 1D-1F) was high with X-ray irradiation, with most populations showing >90% reconstitution with donor marrow (CD45.2+). Neutrophils (Ly6G+), myeloid cells (Ly6G-, Ly6C+), B cells (B220+), and dendritic cells (CD11b+) all showed a significant and near complete reconstitution with donor marrow (Table 2). As previously published, a population of CD3+ T cells remains from the original host (CD45.1+) and represents a radio-resistant subset of the T cell population that would be expected to diminish over time [7].
Figure 1.
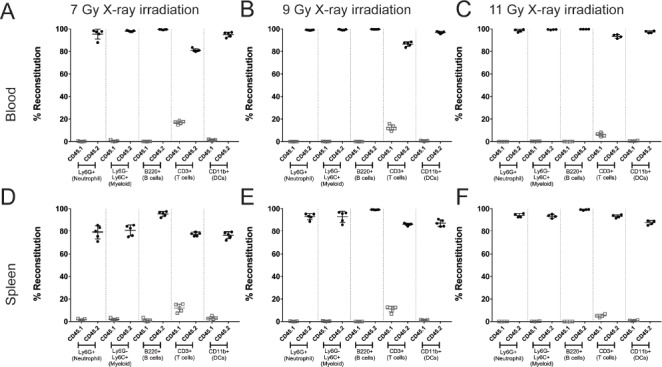
Chimerism achieved with various doses of X-ray irradiation. Animals were irradiated with varying doses of X-ray irradiation and analyzed 8 weeks post reconstitution to evaluate chimerism. The reconstitution of the donor hematopoietic cells (CD45.2+; closed circles) or residual recipient hematopoietic cells (CD45.1+; open squares) is shown as a percent of total cells in the blood (A-C) or in the spleen (D-F) of animals. In general, the range of irradiation used demonstrated comparable reconstitution. This figure is representative of two independent experiments. Data represents mean +/- standard deviation with 5 mice per group.
Table 2.
Statistics for X-ray vs. cesium on blood populations.
| Compartment | Cell type | Control vs. Cs 9 gy | Control vs. X-ray 7 gy | Cs 9 gy vs. X-ray 7 gy |
|---|---|---|---|---|
| Blood | Neutrophil | NS | NS | NS |
| Myeloid | NS | NS | NS | |
| B cell | *** | *** | NS | |
| T cell | ** | ** | NS | |
| Dendritic cell | *** | ** | NS |
All groups were compared to each other using a one way ANOVA with multiple comparisons. P values shown are: NS = non-significant,
*P < 0.05,
**P < 0.01,
***P < 0.001, and
****P < 0.0001.
As the intended use of the bone marrow chimeras in our laboratory was to study the contribution of particular hematopoietic cells to anti-tumor immune responses, we next evaluated the effect of X-ray irradiation and reconstitution on the implantation of syngeneic tumor cell lines. Consistent with previous publications using cesium irradiators and suggesting that homeostatic proliferation can result in anti-tumor responses [8], we saw a delay in the growth of subcutaneously implanted B16F10 tumor cells (Fig. 2A-2E). Further, higher doses of radiation were associated with both health issues and failure to engraft implanted tumor cells. A significant number of animals (8 out of 9) that received 11 grays of irradiation had to be removed from study due to health issues (largely malocclusion and tooth deformation). In addition, only 4 of the animals implanted with tumor cells generated a palpable tumor (Fig. 2E and 2F). Taken together, the overall trend observed was that increasing doses of radiation correlated with an increase in median survival (the time required for the tumor to exceed 2000 mm3). In addition, the median survival at any dose of radiation was significantly longer than age matched animals that had not undergone irradiation and reconstitution (median survival of 18 d for controls as compared to 30 d, 39 d, and undefined for animals that received 7 grays, 9 grays, or 11 grays of radiation; Table 1). This effect was confirmed in a second syngeneic tumor model, MC38 (Fig. S1) and was observed in animals irradiated with both X-ray and cesium sources (data not shown). The delay in tumor growth was likely not due to issues with chimerism (Fig. 1), but rather other changes induced by the irradiation itself.
Figure 2.
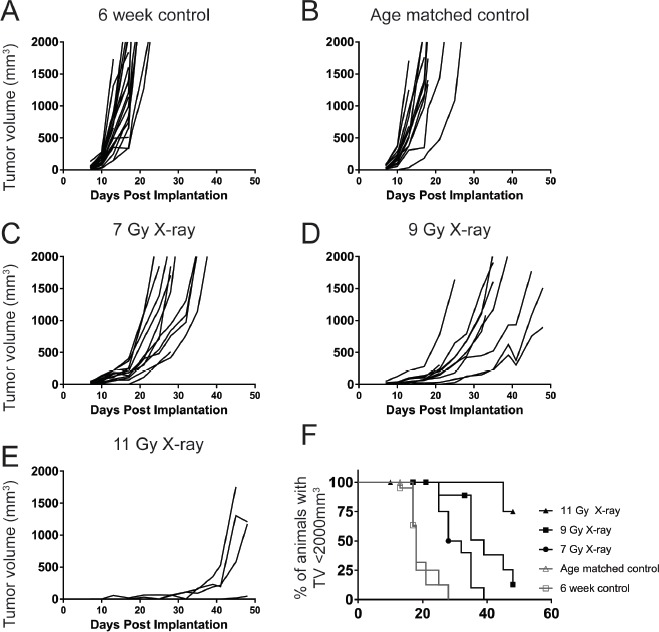
Irradiation and reconstitution results in a delay in B16F10 tumor growth dependent of initial radiation dose. 6 week old (A), age matched (B), or 7-11 Gy irradiated and reconstituted (C-E) C57Bl6 animals were implanted with 2 × 105 B16F10 syngeneic melanoma tumor cells. In animals irradiated with 11 Gy, 5/9 tumors failed to engraft (E). In groups irradiated with 7 Gy and 9 Gy, all tumors (10/10) grew out. The growth rate of the tumors, however, was slower with increasing radiation doses (F). This figure is representative of at least two independent experiments.
Table 1.
Tumor growth in irradiated and reconstituted animals is slower than unirradiated controls.
| Radiation dose | Median survival (d) | Statistically significant compared to control |
|---|---|---|
| Control | 18 | NS |
| Control (age matched) | 18 | NA |
| 7 grays | 30 | P = 0.0002 |
| 9 grays | 39 | P < 0.0001 |
| 11 grays | NA | P = 0.0018 |
Median survival statistics were compared using Log Rank (Mantel Cox) survival comparison test and compared to the age matched control group. NA = not applicable. NS = not-significant.
The lowest dose that achieved comparable chimerism was used for all subsequent studies with the X-ray irradiator. Based on the previous set of experiments, we chose 7 grays of X-ray irradiation and compared it to our historically used Cesium137 dose of 9 grays. The overall degree of chimerism in the blood compartment was compared as in the previous experiments and suggested that the two methods were comparable (Fig. 3A and 3B). In addition, the cellularity of the blood was not significantly different between X-ray and cesium irradiated animals, suggesting that the two methods produced comparably reconstituted populations (Fig. 3C).
Figure 3.
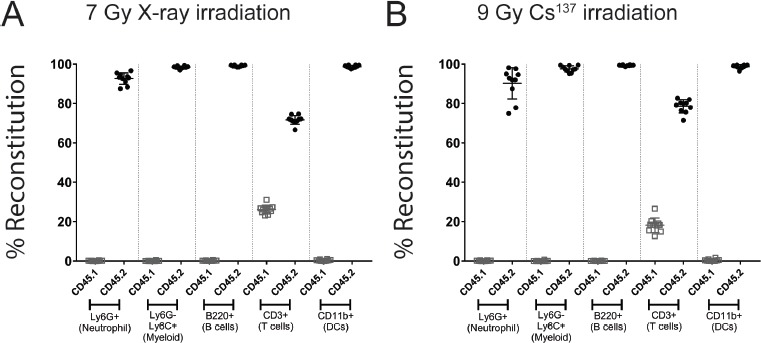
Chimerism at 8 weeks post reconstitution in the blood of X-ray and cesium irradiated animals. Donor marrow reconstitution was evaluated in animals that were irradiated with X-ray (A) or Cesium137 (B). Donor hematopoietic cells (CD45.2+; closed circles) were differentiated from recipient hematopoietic cells (CD45.1+; open squares) by flow cytometry using congenic markers. Hematopoietic cells were identified based on commonly expressed leukocyte antigens. The figure is representative of at least two independent experiments. Data represents mean +/- standard deviation with 10 mice per group.
Having confirmed that the cellularity and reconstitution were comparable for the two methods of irradiation, we next evaluated whether we could increase the tumor growth rate. Due to concerns that the slow growth of the tumors would result in changes to their response to immunotherapy, we increased the initial cell inoculum to determine if that would alter the tumor growth trajectory. Animals were irradiated with (7 grays) of X-ray irradiation, reconstituted with congenic labeled bone marrow, and subcutaneously implanted with a dose titration of B16F10 syngeneic tumor cells ranging from 2 × 105 to 3.2 × 106 cells. Increasing the number of implanted cells from 2 × 105 to 4 × 105 decreased the median survival by 12.5% to 28 d (as compared to 32 d for 2 × 105). Further increases in the number of cells implanted (8 × 105, 1.6 × 106 and 3.2 × 106), however, did not appear to produce additional benefit (Fig. 4). All irradiated animals demonstrated increased median survival compared to the unirradiated control group (19 d for unirradiated control, 32 d for 2 × 105, 28 d for 4 × 105, 28 d for 8 × 105, 27 d for 1.6 × 106 and 28 d for 3.2 × 106). Thus, 4 × 105 cells was the selected inoculation number for irradiated animals, as the median survival was closest to that of the unirradiated control with the lowest number of increased cells implanted.
Figure 4.
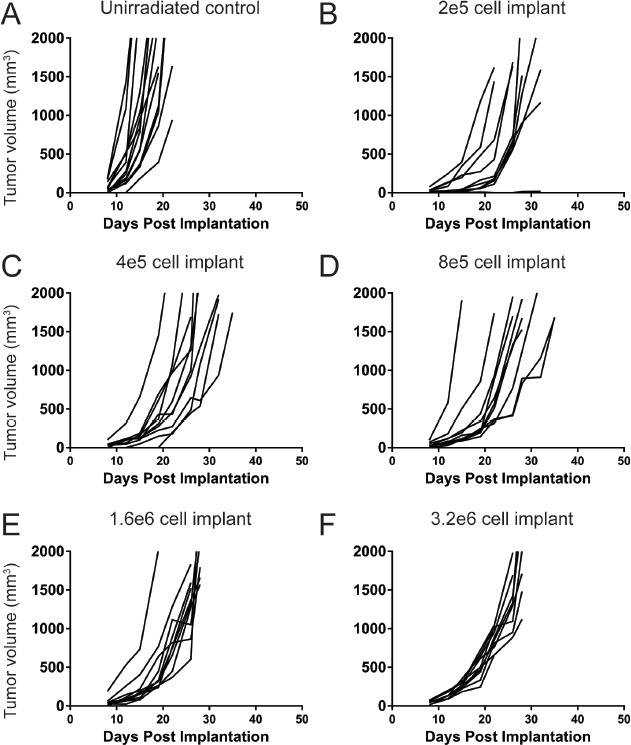
Starting cell implantation has a minimal effect on tumor growth rate post irradiation and reconstitution. Unirradiated, age matched animals were used as a control comparison (A). Animals were irradiated with 7 grays of X-ray irradiation and reconstituted with congenic labeled bone marrow. 8 weeks post reconstitution, animals were implanted with varying doses of B16F10 syngeneic melanoma tumor cells ranging from 2 × 105 to 3.2 × 106 cells per mouse (B-F).
Having identified an optimal dose of cells to use for tumor studies, we further investigated the degree of reconstitution and compared the blood, spleen, and tumor chimerism after both X-ray and cesium irradiation. Animals were irradiated with either 7 grays of X-ray or 9 grays of Cesium137 irradiation, reconstituted with congenic labeled donor bone marrow, and then implanted subcutaneously with 4 × 105 B16F10 syngeneic melanoma tumor cells. Established tumors on day 13 post implantation (average tumor size 212 mm3 for unirradiated controls, 140 mm3 for irradiated and reconstituted animals) were evaluated by flow cytometry (for gating, see Fig. S2) to determine the immune infiltrate present in the tumor. Absolute number of cells was quantified in the blood, spleen, and tumor of animals that had been irradiated and reconstituted versus unirradiated controls. In irradiated and reconstituted animals, there was an increase in B cells, NK cells, and CD4+ T cells in the blood compared to unirradiated controls. However, there was no difference between Cesium137 and X-ray irradiation sources (Fig. 5).
Figure 5.
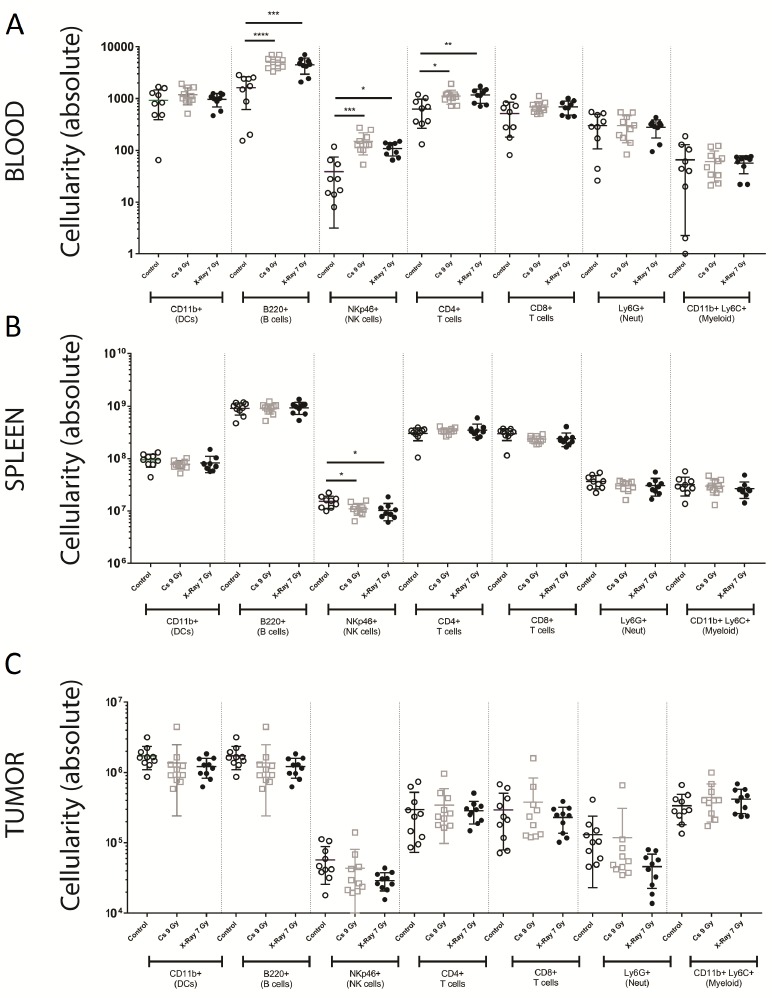
The tumor microenvironment in reconstituted animals is comparable. Age matched unirradiated animals (control; open circles), Cesium137 irradiated and reconstituted (Cs 9 Gy; open squares), and X-ray irradiated and reconstituted (X-ray 7 Gy; closed circles) animals were implanted with 4 × 106 B16F10 syngeneic tumor cells subcutaneously. 13 d post implantation, blood (A), spleen (B), and tumors (C) were analyzed for the degree of donor cell reconstitution. Absolute cell number is shown for CD11b+ dendritic cells, B220+ B cells, NKp46+ NK cells, CD4+ T cells, CD8+ T cells, Ly6G+ neutrophils, and CD11b+Ly6C+Ly6G- myeloid cells. Data represents mean +/- standard deviation with 10 mice per group.
Similarly, we saw only a minor but significant difference in NK cell numbers in the spleen and no differences in overall cellularity in the tumor populations (Fig. 5). To be thorough, we evaluated the degree of chimerism in the various compartments as performed previously (Fig. 1). While the overall degree of chimerism in the blood and spleen was highly comparable and near complete (Fig. S3), the degree of chimerism in the tumor microenvironment showed a significant presence of host hematopoietic cells regardless of the radiation source used (Fig. 6; Table 3).
Figure 6.
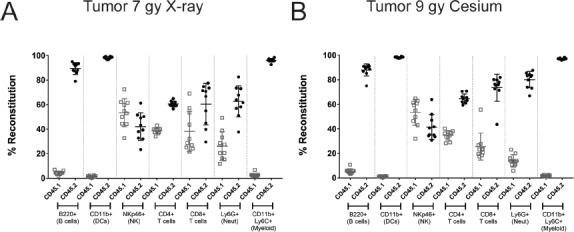
Chimerism at 8 weeks post reconstitution in the tumor microenvironment of X-ray and cesium irradiated animals. Donor cell reconstitution was evaluated in animals that were irradiated with X-ray (A) or Cesium137 (B) and implanted with 4 × 105 B16F10 melanoma tumor cells. Tumors were implanted and allowed to establish before dissociation and analysis on day 13 post implantation (approximately 140–220 mm3. Donor hematopoietic cells (CD45.2+; closed circles) were differentiated from recipient hematopoietic cells (CD45.1+; open squares) by flow cytometry using congenic markers. Hematopoietic cells were identified based on commonly expressed leukocyte antigens. The figure is representative of at least two independent experiments.
Table 3.
Statistics for X-ray vs. cesium on blood, spleen and tumor populations.
| Compartment | Cell type | Control vs. Cs 9 gy | Control vs. X-ray 7 gy | Cs 9 gy vs. X-ray 7 gy |
|---|---|---|---|---|
| Blood | Dendritic cell | NS | NS | NS |
| B cell | **** | *** | NS | |
| NK cell | *** | * | NS | |
| CD4+ T cell | * | ** | NS | |
| CD8+ T cell | NS | NS | NS | |
| Neutrophil | NS | NS | NS | |
| Myeloid | NS | NS | NS | |
| Spleen | Dendritic cell | NS | NS | NS |
| B cell | NS | NS | NS | |
| NK cell | * | * | NS | |
| CD4+ T cell | NS | NS | NS | |
| CD8+ T cell | NS | NS | NS | |
| Neutrophil | NS | NS | NS | |
| Myeloid | NS | NS | NS | |
| Tumor | Dendritic cell | NS | NS | NS |
| B cell | NS | NS | NS | |
| NK cell | NS | NS | NS | |
| CD4+ T cell | NS | NS | NS | |
| CD8+ T cell | NS | NS | NS | |
| Neutrophil | NS | NS | NS | |
| Myeloid | NS | NS | NS |
All groups were compared to each other using a one way ANOVA with multiple comparisons. P values shown are: NS = non-significant,
*P < 0.05,
**P < 0.01,
***P < 0.001, and
****P < 0.0001.
We next evaluated whether the change in tumor growth after irradiation and reconstitution would have a meaningful impact on the response to checkpoint therapy. For these experiments, we chose an anti-mouse CTLA4 blocking antibody. This molecule was generated on a mouse IgG2a isotype. Crosslinking of CTLA4 on T cells (particularly regulatory T cells (Tregs)) and Fc receptor on myeloid cells results in the subsequent depletion of Tregs. In inflamed tumor models, such as CT-26 syngeneic colon carcinoma, CTLA4 mediated-Treg depletion can result in robust tumor growth inhibition [9,10]. In contrast, in a relatively cold tumor model such as B16F10, we do not see efficacy with anti-CTLA4 treatment (Fig. 7A and 7B). However, treatment of B16F10 tumors implanted in irradiated and reconstituted animals resulted in a significant decrease in tumor growth kinetics, although it was insufficient to eradicate the tumors completely (Fig. 7C and 7D). Thus, the effects of irradiation and reconstitution may augment or enhance the effects of conventional immunotherapeutic modalities, even when the radiation dose is administered prior to tumor implantation.
Figure 7.
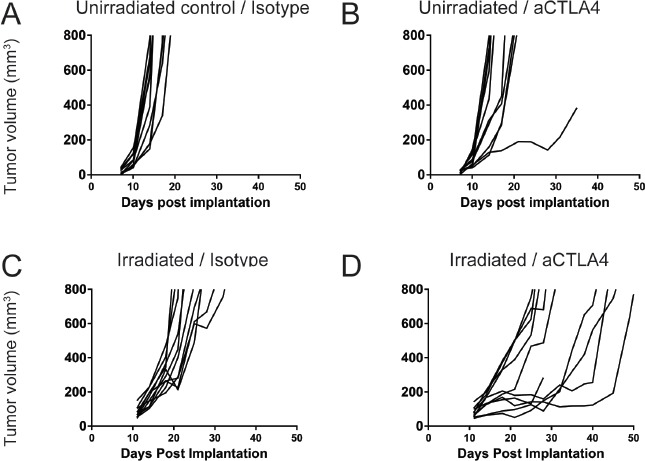
Response to checkpoint therapy in B16F10 is increased in reconstituted animals, consistent with increases in baseline inflammation. Unirradiated, age matched controls (A and B) and animals that were irradiated with 7 gray of X-Ray irradiation and reconstituted with donor bone marrow (C and D) were implanted with 4 × 105 B16F10 syngeneic melanoma cells subcutaneously. On day 11 post-implantation, animals were randomized into treatment groups that received either isotype antibody (A and C) or anti-CTLA-4 antibody (B and D). Tumor volumes were measured twice a week until the tumors exceeded 800 mm3.
DISCUSSION
The generation of bone marrow chimeras has been a useful tool to understand the cellular context of a genetic phenotype. Bone marrow chimeras have been used to identify the contribution of hematopoietic cells to a variety of preclinical disease models, including infectious diseases, transplantation, autoimmunity & asthma, and other tissue disorders [11-19]. At most institutions, irradiation is performed using a cesium137 source, which has increasingly come under scrutiny as a potential domestic security risk owing to the possibility of their use in dirty bombs. In addition, depending on when these machines were originally acquired, they may have undergone significant isotopic decay. At the same time, newer technologies have emerged that are equivalent in function but have fewer challenges with the aforementioned issues. X-ray irradiators, supplied by several vendors, can be implemented at most institutions as a cost-effective replacement. In this study, we have established a protocol to irradiate and reconstitute recipient animals with donor bone marrow. In addition, we have evaluated the effects of irradiation and reconstitution on tumor implantation and growth, as well as the ability of the immune system to mount an anti-tumor response.
Consistent with expectations, the irradiation and subsequent reconstitution resulted in the majority of the CD45+ cells being derived from the donor bone marrow. The only exception was a population of radioresistant T cells that decrease over time but do not entirely disappear. This phenomenon was observed regardless of the radiation dose and has been previously described using cesium irradiators [7]. Reconstitution of animals with congenic, but fully immunocompetent, bone marrow also resulted in a significant delay in tumor growth compared to age matched, unirradiated animals. Previous reports have suggested that this may be mediated by changes to the skin compartment due to radiation exposure [20], homeostatic proliferation of immune cells during reconstitution [8], or inflammation induced by radiation damage (particularly in the gut) [21]. Doses from 7 to 11 grays demonstrated tumor growth delay. To further investigate the nature of this delay, we evaluated the tumor immune infiltrate present in the subcutaneously implanted tumors. While we did see significant increases in certain blood compartment immune cells, we did not observe any major changes in the immune cell populations found in the tumor microenvironment. Increased sensitivity to checkpoint therapy may have resulted from the homeostatic expansion of immune cells required to produce a lympho-replete host. The B16F10 syngeneic melanoma cell line, in our laboratory, is resistant to single therapy checkpoint blockade with either anti-CTLA4 (Fig. 6A and 6B) or anti-PD1 (data not shown). In contrast, B16F10 tumors implanted on reconstituted animals showed a marked response to anti-CTLA4 antibody monotherapy treatment. Although it is beyond the scope of this manuscript to determine what drives this increased sensitivity to checkpoint inhibition, it is important to note this phenomenon, particularly when interpreting data published in bone marrow chimeras.
In summary, we have established and optimized conditions for the generation of bone marrow chimeras using an X-ray-based source. This protocol conforms with current goals set by the Office of Radiological Security while providing comparable data to a Cesium137 source. Additionally, based on our experience with these two modalities, there are additional advantages we have experienced with X-ray irradiation. X-ray irradiators typically measure the delivered dose of radiation directly with a sensor at the site where the animals are located within the X-ray cabinet. As a result, the delivered dose of radiation is extremely accurate. In contrast, cesium irradiators typically rely on a calculation based on the most recent isotopic emission measurement. Thus, the delivered dose may be less accurate depending on when the most recent reading was taken. For this reason, reproducibility between experiments with an X-ray irradiator should be improved as compared to cesium methods.
At present, access to cesium irradiators has been further restricted based on United States Nuclear Regulatory Commission Order EA-05-090 [22,23]. The necessary but increased regulatory burden on corporations and universities that hold permits for these instruments, as well as the financial and legal liability, could potentially restrict access to users who have a legitimate and scientific use for an irradiator. Since X-ray technology does not pose comparable security risks, it will be available to users more readily and may increase the number of experiments performed and reported in the scientific literature. This, in turn, would significantly enrich our understanding of the ways in which various genetic manipulations contribute to changes in the hematopoietic and the stromal compartments. We hope that the data reported here can provide a blueprint for institutions and users to incorporate X-ray irradiator technology into their experimental work flow.
Supplementary Material
Figure S1. Irradiation and reconstitution results in a delay in MC38 tumor growth.
Figure S2. Gating schemes for identification of populations by flow cytometry.
Figure S3. Confirmation of chimerism in blood and spleen of tumor engrafted animals.
Supplementary information of this article can be found online at
Acknowledgments
We would like to thank the cytometry lab at Amgen for help and support with the flow analysis. We thank Angela Coxon and Jackson Egen for critical review of the manuscript.
References
- 1.Kamen J, Hsu W, Boswell B, Hill C. (2019) Successful migration from radioactive irradiators to X-ray irradiators in one of the largest medical centers in the US. Health Phys 117: 558-570. doi: 10.1097/HP.0000000000001095. PMID: [DOI] [PubMed] [Google Scholar]
- 2.Janatpour K, Denning L, Nelson K, Betlach B, Mackenzie M, et al. (2005) Comparison of X-ray vs. gamma irradiation of CPDA-1 red cells. Vox Sang 89: 215-219. doi: 10.1111/j.1423-0410.2005.00699.x. PMID: [DOI] [PubMed] [Google Scholar]
- 3.Gibson BW, Boles NC, Souroullas GP, Herron AJ, Fraley JK, et al. (2015) Comparison of cesium-137 and X-ray irradiators by using bone marrow transplant reconstitution in C57BL/6J Mice. Comp Med 65: 165-172. PMID: [PMC free article] [PubMed] [Google Scholar]
- 4.Spangrude GJ. (2001) Assessment of lymphocyte development in radiation bone marrow chimeras. Curr Protoc Immunol Chapter 4: doi: 10.1002/0471142735.im0406s10. PMID: [DOI] [PubMed] [Google Scholar]
- 5.Tollefson J. (2016) Biologists struggle with push to eliminate radioactive caesium in labs. Nature 533: 156-157. doi: 10.1038/533156a. PMID: [DOI] [PubMed] [Google Scholar]
- 6.Amend SR, Valkenburg KC, Pienta KJ. (2016) Murine hind limb long bone dissection and bone marrow isolation. J Vis Exp: doi: 10.3791/53936. PMID: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Bosco N, Swee LK, Bénard A, Ceredig R, Rolink A. (2010) Auto-reconstitution of the T-cell compartment by radioresistant hematopoietic cells following lethal irradiation and bone marrow transplantation. Exp Hematol 38: 222-232. doi: 10.1016/j.exphem.2009.12.006. PMID: [DOI] [PubMed] [Google Scholar]
- 8.Dummer W, Niethammer AG, Baccala R, Lawson BR, Wagner N, et al. (2002) T cell homeostatic proliferation elicits effective antitumor autoimmunity. J Clin Invest 110: 185-192. doi: 10.1172/JCI15175. PMID: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Selby MJ, Engelhardt JJ, Quigley M, Henning KA, Chen T, et al. (2013) Anti-CTLA-4 antibodies of IgG2a isotype enhance antitumor activity through reduction of intratumoral regulatory T cells. Cancer Immunol Res 1: 32-42. doi: 10.1158/2326-6066.CIR-13-0013. PMID: [DOI] [PubMed] [Google Scholar]
- 10.Selby MJ, Engelhardt JJ, Johnston RJ, Lu LS, Han M, et al. (2016) Correction: Preclinical development of ipilimumab and nivolumab combination immunotherapy: Mouse tumor models, in vitro functional studies, and cynomolgus macaque toxicology. PLoS One 11: e0167251. doi: 10.1371/journal.pone.0167251. PMID: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Ballinger MN, Newstead MW, Zeng X, Bhan U, Mo XM, et al. (2015) IRAK-M promotes alternative macrophage activation and fibroproliferation in bleomycin-induced lung injury. J Immunol 194: 1894-1904. doi: 10.4049/jimmunol.1402377. PMID: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Hollingsworth JW, Theriot BS, Li Z, Lawson BL, Sunday M, et al. (2009) Both hematopoietic-derived and non-hematopoietic-derived {beta}-arrestin-2 regulates murine allergic airway disease. Am J Respir Cell Mol Biol 43: 269-275. doi: 10.1165/rcmb.2009-0198OC. PMID: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Huang W, Rainbow DB, Wu Y, Adams D, Shivakumar P, et al. (2017) A novel Pkhd1 mutation interacts with the nonobese diabetic genetic background to cause autoimmune cholangitis. J Immunol 200: 147-162. doi: 10.4049/jimmunol.1701087. PMID: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Hwang S, Cobb DA, Bhadra R, Youngblood B, Khan IA. (2016) Blimp-1-mediated CD4 T cell exhaustion causes CD8 T cell dysfunction during chronic toxoplasmosis. J Exp Med 213: 1799-1818. doi: 10.1084/jem.20151995. PMID: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Komada T, Chung H, Lau A, Platnich JM, Beck PL, et al. (2018) Macrophage uptake of necrotic cell DNA activates the AIM2 inflammasome to regulate a proinflammatory phenotype in CKD. J Am Soc Nephrol 29: 1165-1181. doi: 10.1681/ASN.2017080863. PMID: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Kronenberg G, Uhlemann R, Richter N, Klempin F, Wegner S, et al. (2017) Distinguishing features of microglia- and monocyte-derived macrophages after stroke. Acta Neuropathol 135: 551-568. doi: 10.1007/s00401-017-1795-6. PMID: [DOI] [PubMed] [Google Scholar]
- 17.Machein MR, Plate KH. (2014) Bone marrow chimera experiments to determine the contribution of hematopoietic stem cells to cerebral angiogenesis. Methods Mol Biol 1135: 275-288. doi: 10.1007/978-1-4939-0320-7_23. PMID: [DOI] [PubMed] [Google Scholar]
- 18.Majorov KB, Eruslanov EB, Rubakova EI, Kondratieva TK, Apt AS. (2005) Analysis of cellular phenotypes that mediate genetic resistance to tuberculosis using a radiation bone marrow chimera approach. Infect Immun 73: 6174-6178. doi: 10.1128/IAI.73.9.6174-6178.2005. PMID: [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Micklem HS, Clarke CM, Evans EP, Ford CE. (1968) Fate of chromosome-marked mouse bone marrow cells tranfused into normal syngeneic recipients. Transplantation 6: 299-302. PMID: [PubMed] [Google Scholar]
- 20.Milas L, Ito H, Hunter N, Jones S, Peters LJ. (1986) Retardation of tumor growth in mice caused by radiation-induced injury of tumor bed stroma: dependency on tumor type. Cancer Res 46: 723-727. PMID: [PubMed] [Google Scholar]
- 21.McBride WH, Chiang C, Olson JL, Wang C, Hong J, et al. (2004) A sense of danger from radiation. Radiat Res 162: 1-19. doi: 10.1667/rr3196. PMID: [DOI] [PubMed] [Google Scholar]
- 22.United States Nuclear Regulatory Commission. Order imposing increased controls (Effective immediately). Available from: https://www.nrc.gov/docs/ML0531/ML053130218.pdf
- 23.United States Nuclear Regulatory Commission. Increased controls for licensees that possess sources containing radioactive material quantities of concern. Available from: https://www.nrc.gov/docs/ML0531/ML053130364.pdf
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Figure S1. Irradiation and reconstitution results in a delay in MC38 tumor growth.
Figure S2. Gating schemes for identification of populations by flow cytometry.
Figure S3. Confirmation of chimerism in blood and spleen of tumor engrafted animals.
Supplementary information of this article can be found online at


