Abstract
Background
A lymph node harvest (LNH) of < 12 is a predictor for poor prognosis in rectal cancer patients. However, neoadjuvant chemoradiotherapy (NACRT) is known to decrease LNH; hence, a cut-off of 12 is inappropriate in such patients. This paper aims to establish a LNH cut-off predictive for disease-free and overall survival in NACRT patients.
Methods
A retrospective review of patients who underwent elective surgery for rectal cancer from 2006 to 2013 was performed. All patients with R1/2 resections and presence of metastases and those operated on for recurrence were excluded. Patient demographics, clinical features, operative details, LNH, 30-day mortality and disease-free and overall survival were recorded. P values of < 0.05 were considered significant.
Results
A total of 257 patients were studied, with 174 (68%) males and a median age of 66 years. Ninety-four (37%) patients received long-course NACRT, and 122 (48%) patients were stage 2 and below. Median LNH was 17, which was reduced in the NACRT group (14 versus 23, P < 0.01). Average length of stay was 9 ± 8 days, with a major post-operative complication rate of 4%. Using hazard ratio plots for the NACRT subgroup, LNH cut-offs of 16.5 and 8.5 were obtained for disease-free survival (DFS) and overall survival (OS) respectively. Survival analysis showed that a LNH cut-off of 8.5 was a significant predictor of OS (P < 0.001).
Conclusion
LNH is reduced in patients receiving NACRT before rectal cancer surgery. A LNH of 9 and above is associated with improved overall survival. We propose that this can be used as a tool for prognosis.
Keywords: Rectal cancer, Neoadjuvant, Chemotherapy, Radiotherapy, Lymph node
Introduction
The number of harvested lymph nodes (LN) is a well-established prognostic factor in patients with rectal cancer. The American Joint Committee on Cancer (AJCC) recommends that at least 12 LN are needed to confirm node-negative disease for rectal cancer [1, 2].
Neoadjuvant chemoradiotherapy (NACRT) is known to decrease the lymph node harvest (LNH) in the resected specimen [3–5]. While studies have shown that LNH is associated with recurrence and survival in rectal cancer patients who have undergone upfront surgery [6, 7], its prognostic value in patients who have received NACRT is debatable [8–13]. Although some studies show that LNH has no significant correlation with recurrence or survival in NACRT patients [9, 12], some report that a suboptimal LNH of < 12 independently predicts worse overall survival irrespective of neoadjuvant treatment [11, 13]. A low LN count could represent either poor sampling of the draining LN basin and hence inadequate staging or a good response to neoadjuvant treatment and therefore a surrogate marker for improved survival. Currently, the National Comprehensive Cancer Network (NCCN) guidelines indicate that a minimal LNH of 12 is no longer applicable for patients who have undergone neoadjuvant therapy.
Hence, the aim of this project is to determine a new LNH cut-off in the NACRT subgroup for both disease-free and overall survival in our patient population.
Methodology
This was a single-centre retrospective review of all patients who underwent elective surgery for rectal cancer in our institution from January 2006 to December 2013. Inclusion criteria were all patients who underwent elective surgical resection for histologically proven rectal adenocarcinoma. Exclusion criteria are as follows: all patients with R1/2 resections, involvement of the circumferential radial margin, high rectal tumours (defined as tumours for which a high anterior resection and partial mesorectal excision was performed), presence of metastases, emergency surgeries, and those operated on for recurrence. This study was conducted with the approval of our institution’s ethical review board (Domain Specific Review Board reference number: 2018/00540). We chose a period of study before 2014 to allow for sufficient follow-up for survival analysis.
Pre-operative conduct
All patients had a pre-operative diagnosis of rectal cancer based on endoscopically obtained biopsy with histological confirmation of the primary tumour. All patients received a staging computed tomography (CT) scan of the thorax, abdomen and pelvis, as well as a magnetic resonance imaging (MRI) of the rectum prior to surgery. Patient demographic details and tumour characteristics were recorded accordingly. Patients in the NACRT group received long-course neoadjuvant chemoradiotherapy—comprising 45 Gy of radiotherapy given in 25 fractions over 5 weeks, with concomitant oral capecitabine 825 mg/m2 twice daily as a radiosensitiser. All patients were discussed at the multidisciplinary tumour board. Patients with T3-, T4- or N-positive disease on pre-operative MRI were selected to undergo NACRT. In this group of patients, upfront surgery may still proceed if the surgeon determines that the circumferential resection margin was sufficiently wide to reduce the risk of a R0 resection or if the patient declines neoadjuvant treatment.
Operative details
All surgeries were performed by four specialists in the colorectal subspecialty service in our institution. Surgeries were performed either using the traditional open technique or via the laparoscopic approach, according to the discretion of the primary consultant and in consultation with the patient. If NACRT was given, surgery was performed within 6–10 weeks from completion of chemoradiotherapy. Oncologic en bloc resection of the specimen was performed, along with total mesorectal excision to ensure adequate clearance of the draining lymphovascular basin. All resected specimens were sent for histopathological assessment. Lymph node retrieval and assessment were performed by trained pathologists from our institution. In order to maximise the lymph node yield, all pathologists performed chemical fat clearance and took additional random sections of mesorectal fat for sampling. In cases with less than 12 lymph nodes found during the initial assessment, a repeat examination was performed by a second pathologist.
Outcome measures
All patients were followed up for at least 5 years post-operatively. Length of stay, 30-day mortality, post-operative complications, disease-free survival (DFS) and overall survival (OS) were calculated for all patients. All patients who received neoadjuvant therapy also received adjuvant chemotherapy. Patients were given 3-month follow-up clinic visits for the first 2 years, including physical examination and CEA levels. Patients were then seen 6 months from the third year onwards, until the 5th post-operative year. Surveillance colonoscopies were performed at 1st, 3rd and 5th year post-surgery, then 3–5 years thereafter. For patients with an incomplete scope pre-surgery, completion colonoscopy was performed within 6 months post-surgery to rule out synchronous lesions. Surveillance CT scans (thorax, abdomen and pelvis) were performed at 12-month intervals until the 5th post-operative year.
Disease-free survival was defined as the time from surgery to death or disease progression, and patients without these events were censored at last follow-up. Overall survival was defined as the time from surgery to death from any cause, and patients who were alive at last follow-up were censored.
LNH was categorised into < 12 and ≥ 12 based on AJCC and UICC guidelines of 12 being the optimal LNH cut-off in resected specimens.
Statistical analysis
Chi-square test, Mann-Whitney U test, Kruskal-Wallis test and multiple linear regression analyses were used as appropriate. For the NACRT subgroup, we investigated the relationship between LNH and survival outcomes by iteratively dichotomizing the LNH at each integer value and computing hazard ratios at each cut-off value. The optimal LNH cut-off was determined based on the cut-off which was associated with the most significant split in Kaplan-Meier curves (i.e. smallest univariable log-rank P value). Kaplan-Meier survival curves were then plotted based on these LNH cut-offs for both DFS and OS respectively. P values of < 0.05 were considered significant. All statistical analysis was performed using SPSS version 21.0 and R software version 3.4.2 (The R Foundation for Statistical Computing).
Results
Clinical characteristics and operative details
There were a total of 257 patients included in this study, with 174 (68%) males and a mean age of 66 ± 11 years old. The rectal tumours were at a mean distance of 6 ± 3 cm from the anal verge.
Ninety-four (37%) patients received pre-operative NACRT. With regard to the type of surgery, there were 32 (12%) ultra-low anterior resections, 168 (65%) low anterior resections, 19 (7.4%) Hartmann’s procedure and 38 (15%) abdominoperineal resections. One hundred fifty (58%) patients underwent laparoscopic surgery, while 107 (42%) patients underwent open surgery. One hundred twenty-two (48%) of cancer patients were stage 1 or 2, while the remaining 135 (52%) were stage 3.
When stratified according to the upfront surgery versus NACRT, a significantly higher proportion of patients in the NACRT group had lower rectal tumours (mean distance of 5.5 versus 7.0 cm from anal verge, P < 0.001). Accordingly, a greater proportion of patients in the NACRT group also underwent abdominoperineal resections or ultra-low anterior resections as compared to the upfront surgery group (Table 1).
Table 1.
Clinicopathologic characteristics and outcomes stratified by neoadjuvant treatment (values in either median ± SD or no. (%) unless otherwise stated)
| Variable | Category | Total (N = 257) | Upfront surgery (N = 163) | NACRT group (N = 94) | P value |
|---|---|---|---|---|---|
| Age at diagnosis (years) | 66 ± 11 | 67 ± 11 | 64 ± 10 | 0.132 | |
| Gender | Male | 174 (68) | 106 (65) | 68 (72) | 0.227 |
| Female | 83 (32) | 57 (35) | 26 (28) | ||
| Distance from anal verge (centimetres) | 6 ± 3 | 7.0 ± 3.4 | 5.5 ± 2.4 | < 0.001 | |
| Type of operation | Ultra-low anterior resection | 32 (12) | 16 (10) | 16 (13) | 0.001 |
| Low anterior resection | 168 (65) | 117 (72) | 51 (65) | ||
| Hartmann’s procedure | 19 (8) | 15 (9) | 4 (7) | ||
| Abdominoperineal resection | 38 (15) | 15 (9) | 23 (15) | ||
| Lymph node harvest | 17 ± 11 | 23 ± 13 | 14 ± 6.7 | < 0.001 | |
| Lymph node harvest | < 12 | 53 (21) | 17 (10) | 36 (38) | 0.002 |
| ≥ 12 | 204 (79) | 146 (90) | 58 (62) | ||
| Post-operative complications | None | 225 (88) | 140 (86) | 85 (88) | 0.729 |
| Clavien-Dindo 1 and 2 | 27 (11) | 19 (11) | 8 (10) | ||
| Clavien-Dindo 3 and above | 5 (1) | 4 (3) | 1 (2) | ||
| Length of stay (days) | 9 ± 8 | 10 ± 8 | 8 ± 8 | 0.190 | |
| 30-day mortality | Yes | 255 (99) | 2 (1) | 0 (0) | 0.281 |
| No | 2 (1) | 161 (99) | 94 (100) | ||
| Recurrence | Yes | 60 (23) | 40 (25) | 20 (21) | 0.551 |
| No | 197 (77) | 123 (75) | 74 (79) | ||
| Disease-free survival (months) | 66 ± 41 | 62 ± 40 | 73 ± 42 | 0.067 | |
| Mortality | Yes | 90 (35) | 60 (37) | 30 (32) | 0.428 |
| No | 167 (65) | 103 (63) | 64 (68) | ||
| Overall survival (months) | 71 ± 38 | 68 ± 38 | 77 ± 39 | 0.059 | |
NACRT neoadjuvant chemoradiotherapy
Operative outcomes
There were 5 (2%) patients with post-operative complications of Clavien-Dindo classification 3 and above, of which there were 4 anastomotic leaks and 1 intestinal obstruction. The median length of stay was 9 ± 8 days, and 30-day mortality incidence was 0.8% (n = 2). Twenty-three percent (n = 60) of patients had recurrence within 5 years post-surgery, of which 11 (18%) were locoregional, 41 (68%) were systemic and 8 (13%) were with both locoregional and systemic. The overall 5-year disease-free survival was 66 ± 41 months. For the 5-year duration of follow-up analysed, the overall mortality was n = 90 (35%), with an overall survival of 71 ± 38 months.
There was no statistically significant difference between the upfront surgery and NACRT group in terms of operative outcomes (Table 1). There was also no statistically significant difference between the lymph node yield when stratified according to the laparoscopic versus open surgery (17 versus 16, P = 0.56).
Impact of NACRT on LNH
An overall median of 17 ± 11 lymph nodes were retrieved. Patients who received NACRT had a significantly reduced LNH compared to those that had upfront surgery (14 ± 7 versus 23 ± 13, P < 0.001). Similarly, 90% (n = 146) of patients with upfront surgery met the recommended LNH cut-off of ≥ 12, versus only 62% (n = 58) in the NACRT group.
Factors associated with DFS and OS
Table 2 shows the multivariate analysis of clinicopathological factors on disease-free and overall survival. On multivariate analysis, tumour staging was the only significant prognostic variable for both DFS and OS, with lower stage 1 and 2 tumours having a longer DFS and OS versus stage 3 tumours. Patients in the LNH < 12 subgroup had a decreased DFS and OS compared to the LNH ≥ 12 subgroup, although this was not statistically significant (DFS = 61 versus 62 months, OS = 68 versus 73 months). Table 3 shows the subgroup analysis with separation of patients into the NACRT versus upfront surgery, and looks into factors related to tumour staging and lymph node yield alone. When stratified according to the NACRT versus upfront surgery, tumour staging remained significantly associated with a longer disease-free and overall survival. However, during subgroup analysis of patients who received upfront surgery, our data shows that a LNH of 12 and above was significantly associated with a longer disease-free and overall survival, with P values of 0.040 and 0.013 respectively (Table 3). Contrastingly, LNH > 12 was not a significant predictor of long-term outcome for the NACRT subgroup.
Table 2.
Multivariate analysis of clinicopathologic factors related to disease-free and overall survival
| Variables | Category | N | Disease-free survival | Overall survival | ||
|---|---|---|---|---|---|---|
| Median ± SD (months) | P value | Median ± SD (months) | P value | |||
| Age at diagnosis (years) | < 50 | 18 | 81 ± 46 | 0.180 | 90 ± 38 | 0.370 |
| 50–64 | 101 | 68 ± 42 | 73 ± 39 | |||
| 65–74 | 74 | 63 ± 39 | 69 ± 37 | |||
| > 75 | 64 | 63 ± 39 | 65 ± 38 | |||
| Gender | Male | 174 | 63 ± 42 | 0.160 | 69 ± 39 | 0.470 |
| Female | 83 | 73 ± 38 | 76 ± 36 | |||
| Neoadjuvant therapy | No | 163 | 62 ± 40 | 0.060 | 68 ± 38 | 0.191 |
| Yes | 94 | 73 ± 41 | 77 ± 42 | |||
| Type of operation | Ultra-low anterior resection | 32 | 56 ± 38 | 0.908 | 61 ± 39 | 0.659 |
| Low anterior resection | 168 | 67 ± 43 | 73 ± 40 | |||
| Hartmann’s procedure | 19 | 55 ± 42 | 59 ± 38 | |||
| Abdominoperineal resection | 38 | 77 ± 44 | 59 ± 42 | |||
| Tumour staging | Stage 1 | 61 | 84 ± 39 | < 0.001 | 85 ± 40 | < 0.001 |
| Stage 2 | 61 | 67 ± 35 | 72 ± 38 | |||
| Stage 3 | 135 | 58 ± 38 | 64 ± 37 | |||
| Lymph node harvest | < 12 | 53 | 61 ± 44 | 0.070 | 62 ± 42 | 0.051 |
| ≥ 12 | 204 | 68 ± 42 | 73 ± 36 | |||
Table 3.
Subgroup analysis of neoadjuvant chemoradiotherapy (NACRT) and upfront surgery patients
| Variables | Category | N | Disease-free survival | Overall survival | ||
|---|---|---|---|---|---|---|
| Median ± SD (months) | P value | Median ± SD (months) | P value | |||
| NACRT group (N= 94) | ||||||
| Tumour staging | Stage 1 | 18 | 88 ± 39 | 0.026 | 88 ± 39 | 0.051 |
| Stage 2 | 32 | 73 ± 40 | 76 ± 39 | |||
| Stage 3 | 44 | 67 ± 37 | 74 ± 36 | |||
| Lymph node harvest | < 12 | 36 | 61 ± 38 | 0.801 | 63 ± 41 | 0.436 |
| ≥ 12 | 58 | 80 ± 40 | 86 ± 39 | |||
| Upfront surgery group (N= 163) | ||||||
| Tumour staging | Stage 1 | 43 | 82 ± 38 | 0.001 | 84 ± 41 | 0.003 |
| Stage 2 | 29 | 61 ± 37 | 68 ± 40 | |||
| Stage 3 | 91 | 54 ± 35 | 60 ± 37 | |||
| Lymph node harvest | < 12 | 17 | 61 ± 38 | 0.040 | 61 ± 39 | 0.013 |
| ≥ 12 | 146 | 63 ± 41 | 68 ± 38 | |||
Optimal LNH cut-offs for the NACRT group
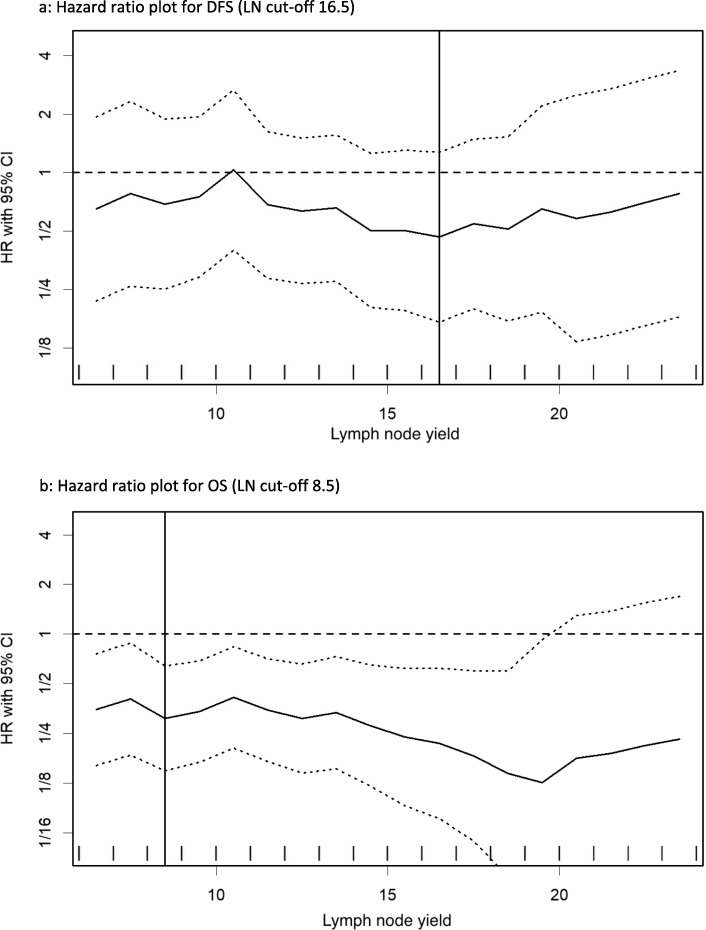
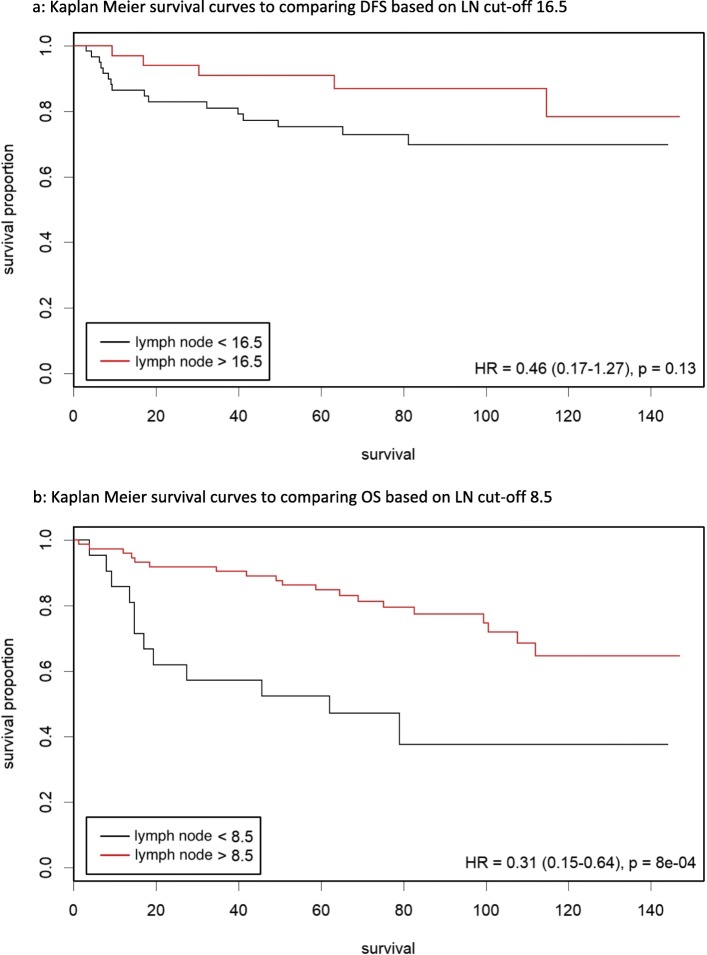
As our data shows that NACRT significantly reduces LNH and that a cut-off of 12 was not applicable for this subgroup, we proceeded on to establish if there was a new optimal LNH cut-off for both DFS and OS in NACRT patients. Hazard ratio plots demonstrate that the optimal LNH cut-off for DFS and OS are 16.5 and 8.5 respectively (Fig. 1). The Kaplan-Meier curve survival analyses show that a LNH cut-off of 8.5 was a significant predictor for OS (HR 0.31, 95% CI 0.15–0.64, P < 0.001); however, a LNH cut-off of 16.5 did not significantly predict DFS (HR 0.46, 95% CI 0.17–1.27, P = 0.13) (Fig. 2).
Fig. 1.
Hazard ratio plots to determine optimal lymph node (LN) cut-offs for disease-free survival (DFS) and overall survival (OS) in the neoadjuvant chemoradiotherapy (NACRT) group. a Hazard ratio plot for DFS (LN cut-off 16.5). b Hazard ratio plot for OS (LN cut-off 8.5)
Fig. 2.
Kaplan-Meier survival curves to compare disease-free survival (DFS) and overall survival (OS) based on lymph node (LN) cut-offs in the neoadjuvant chemoradiotherapy (NACRT) group. a Kaplan-Meier survival curves to comparing DFS based on LN cut-off 16.5. b Kaplan-Meier survival curves to comparing OS based on LN cut-off 8.5
Using this new LNH cut-off of 9, we proceeded on to further subgroup analysis of the NACRT cohort. Table 4 demonstrates that on the multivariable analysis, a LNH cut-off of 9 was a significant predictor for both disease-free and overall survival.
Table 4.
Subgroup multivariate analysis of clinicopathologic factors related to disease-free and overall survival for the neoadjuvant chemoradiotherapy (NACRT) group, with a new LN cut-off of 9
| Variables | Category | N = 94 | Disease-free survival | Overall survival | ||
|---|---|---|---|---|---|---|
| Median ± SD (months) | P value | Median ± SD (months) | P value | |||
| Age at diagnosis (years) | < 50 | 7 | 82 ± 38 | 0.236 | 91 ± 35 | 0.082 |
| 50–64 | 44 | 65 ± 41 | 72 ± 40 | |||
| 65–74 | 26 | 63 ± 40 | 67 ± 36 | |||
| > 75 | 17 | 62 ± 33 | 64 ± 38 | |||
| Gender | Male | 68 | 64 ± 35 | 0.770 | 68 ± 40 | 0.835 |
| Female | 26 | 72 ± 36 | 74 ± 38 | |||
| Type of operation | Ultra-low anterior resection | 51 | 56 ± 35 | 0.838 | 60 ± 41 | 0.907 |
| Low anterior resection | 16 | 67 ± 40 | 72 ± 40 | |||
| Hartmann’s procedure | 4 | 57 ± 41 | 61 ± 37 | |||
| Abdominoperineal resection | 23 | 74 ± 39 | 60 ± 36 | |||
| Tumour staging | Stage 1 | 18 | 82 ± 40 | 0.008 | 86 ± 35 | 0.012 |
| Stage 2 | 32 | 64 ± 37 | 60 ± 37 | |||
| Stage 3 | 44 | 53 ± 37 | 57 ± 38 | |||
| Lymph node harvest | < 9 | 21 | 52 ± 36 | 0.028 | 55 ± 39 | 0.009 |
| ≥ 9 | 73 | 63 ± 45 | 72 ± 40 | |||
Discussion
Our study shows that NACRT results in a significant reduction in LNH as compared to upfront surgery. This finding is well supported by the current literature [3–5], with prior large-scale retrospective studies by Ha et al. [4] and Amajoyi et al. [5] demonstrating mean LNH of 14.5 and 9 respectively in the neoadjuvant group, versus 21.5 and 13 in the upfront surgery group. A meta-analysis by Mechera et al. also shows that NACRT decreases the LNH by approximately two to four lymph nodes [14]. It has been suggested that this is due to a reduction in lymph node size due to apoptosis and involution induced by chemoradiotherapy [4, 15, 16]. Given that NACRT significantly reduces LNH, the utility of LNH as a prognostic factor in such patients should be interpreted with caution.
Traditionally, a cut-off of 12 lymph nodes is used as an indicator of adequate oncological clearance, in order to confirm node-negative rectal cancer as per AJCC guidelines [1, 2]. However, LN yield is known to be affected by pre-operative NACRT [3–5], and it is proposed that the traditional cut-off of 12 should not be applied to this subgroup of patients. The current NCCN guidelines do not give a guideline of a minimum number of harvested nodes in patients who have undergone neoadjuvant therapy. A low LNH in NACRT patients could either represent either poor oncological clearance and hence inadequate staging or a good response to NACRT and therefore a predictor of good outcome. While some studies show that LNH predicts worse overall survival regardless of NACRT [11, 13], majority of the literature are proponents of the notion that a LNH metric may not be clinically relevant in the era of neoadjuvant therapy [9, 12, 17, 18]. Our paper demonstrates that a LNH cut-off of 12 is not significantly associated with poorer DFS and OS in patients who received pre-operative NACRT. However, further survival curve analysis within the NACRT group demonstrates that a cut-off of 8.5 significantly predicted overall survival.
As NACRT significantly reduces LNH in rectal cancer patients undergoing surgical resection, it is important for surgeons to recognise that when using LNH as a prognosticating factor. These two groups of patients—upfront surgery versus NACRT—should be evaluated using a different metric. Establishing a new LNH cut-off in NACRT patients is important as it allows clinicians to establish a more accurate surrogate judgement for the quality of their surgery, as well as better counsel their patients regarding future prognosis. In some patients who have concerns about undergoing advised completion chemotherapy after NACRT, having a lymph node yield of < 9 provides added reason to proceed. Conversely, in elderly patients who have a lymph node yield of less than 12 but more than 9 after NACRT, consideration should also be taken as to whether completion chemotherapy will be beneficial. Given the increasing prevalence of colorectal cancer in older adults [19], studies have shown that the elderly are less likely to be recommended adjuvant therapy in view of significant side effects and limited survival benefit [20, 21]. Lastly, our results are especially pertinent to our local population, as it allows us to apply these results to patients from a similar background. A recent local paper published by Chan et al. demonstrated that the LNH cut-off of 12 had no significant impact on overall survival in patients who received neoadjuvant therapy [22]. Perhaps, a new lower cut-off of 9 can be used in our local population instead.
As per all retrospective reviews, the authors acknowledge that there are limitations to this paper. The variables analysed can only be associated with the outcomes, and a directional causality cannot be proven. The treatment modality was also patient and surgeon dependent; hence, the allocation of patients into the two treatment groups was not entirely random. However, this study has a long follow-up interval of 5 years, with a sizeable patient population. More prospective longer-term studies are needed to establish if a new LNH cut-off of 9 and above should be used as a surrogate marker for better patient outcome instead.
Conclusion
Instead of the traditionally accepted cut-off value of 12 lymph nodes, perhaps a lower number can be considered optimal in NACRT patients. Our study shows that a LN cut-off of 9 and above can be used to predict improved overall survival in NACRT patients. This data will help clinicians better prognosticate their patients’ outcomes and may influence their decisions for post-operative management should lymph node yield be insufficient.
Acknowledgements
Not applicable.
Authors’ contributions
CSY performed the data collection and data analysis and contributed in writing the manuscript. NS performed the data analysis and contributed in writing the manuscript. The authors read and approved the final manuscript.
Funding
Not applicable.
Availability of data and materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Ethics approval and consent to participate
This study has obtained ethical approval from the relevant institution’s institutional review board (reference number: 2018/00540).
Consent for publication
Not applicable.
Competing interests
The authors declare that they have no competing interests.
Footnotes
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
- 1.Compton CC, Fielding LP, Burgart LJ, Conley B, Cooper HS, Hamilton SR, Hammond ME, Henson DE, Hutter RV, Nagle RB, Nielsen ML, Sargent DJ, Taylor CR, Welton M, Willett C. Prognostic factors in colorectal cancer. College of American Pathologists Consensus Statement 1999. Archives of pathology & laboratory medicine. 2000;124(7):979–994. doi: 10.1043/0003-9985(2000)124<0979:Pficc>2.0.Co;2. [DOI] [PubMed] [Google Scholar]
- 2.Edge SB, Compton CC. The American Joint Committee on Cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Annals of surgical oncology. 2010;17(6):1471–1474. doi: 10.1245/s10434-010-0985-4. [DOI] [PubMed] [Google Scholar]
- 3.Miller ED, Robb BW, Cummings OW, Johnstone PA. The effects of preoperative chemoradiotherapy on lymph node sampling in rectal cancer. Diseases of the colon and rectum. 2012;55(9):1002–1007. doi: 10.1097/DCR.0b013e3182536d70. [DOI] [PubMed] [Google Scholar]
- 4.Ha YH, Jeong SY, Lim SB, Choi HS, Hong YS, Chang HJ, Kim DY, Jung KH, Park JG. Influence of preoperative chemoradiotherapy on the number of lymph nodes retrieved in rectal cancer. Annals of surgery. 2010;252(2):336–340. doi: 10.1097/SLA.0b013e3181e61e33. [DOI] [PubMed] [Google Scholar]
- 5.Amajoyi Robert, Lee Yoori, Recio Patrick J., Kondylis Philip D. Neoadjuvant therapy for rectal cancer decreases the number of lymph nodes harvested in operative specimens. The American Journal of Surgery. 2013;205(3):289–292. doi: 10.1016/j.amjsurg.2012.10.020. [DOI] [PubMed] [Google Scholar]
- 6.Gunderson LL, Jessup JM, Sargent DJ, Greene FL, Stewart A. Revised tumor and node categorization for rectal cancer based on surveillance, epidemiology, and end results and rectal pooled analysis outcomes. J Clin Oncol. 2010;28(2):256–263. doi: 10.1200/jco.2009.23.9194. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Kim YW, Kim NK, Min BS, Lee KY, Sohn SK, Cho CH. The influence of the number of retrieved lymph nodes on staging and survival in patients with stage II and III rectal cancer undergoing tumor-specific mesorectal excision. Ann Surg. 2009;249(6):965–972. doi: 10.1097/SLA.0b013e3181a6cc25. [DOI] [PubMed] [Google Scholar]
- 8.Gill A, Brunson A, Lara P, Jr, Khatri V, Semrad TJ. Implications of lymph node retrieval in locoregional rectal cancer treated with chemoradiotherapy: a California Cancer Registry Study. Eur J Surg Oncol. 2015;41(5):647–652. doi: 10.1016/j.ejso.2015.01.037. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Kim HJ, Jo JS, Lee SY, Kim CH, Kim YJ, Kim HR. Low lymph node retrieval after preoperative chemoradiation for rectal cancer is associated with improved prognosis in patients with a good tumor response. Ann Surg Oncol. 2015;22(6):2075–2081. doi: 10.1245/s10434-014-4235-z. [DOI] [PubMed] [Google Scholar]
- 10.Tsai CJ, Crane CH, Skibber JM, Rodriguez-Bigas MA, Chang GJ, Feig BW, Eng C, Krishnan S, Maru DM, Das P. Number of lymph nodes examined and prognosis among pathologically lymph node-negative patients after preoperative chemoradiation therapy for rectal adenocarcinoma. Cancer. 2011;117(16):3713–3722. doi: 10.1002/cncr.25973. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Xu Z, Berho ME, Becerra AZ, Aquina CT, Hensley BJ, Arsalanizadeh R, Noyes K, Monson JRT, Fleming FJ. Lymph node yield is an independent predictor of survival in rectal cancer regardless of receipt of neoadjuvant therapy. J Clin Pathol. 2017;70(7):584–592. doi: 10.1136/jclinpath-2016-203995. [DOI] [PubMed] [Google Scholar]
- 12.Persiani R, Biondi A, Gambacorta MA, Bertucci Zoccali M, Vecchio FM, Tufo A, Coco C, Valentini V, Doglietto GB, D’Ugo D. Prognostic implications of the lymph node count after neoadjuvant treatment for rectal cancer. Br J Surg. 2014;101(2):133–142. doi: 10.1002/bjs.9341. [DOI] [PubMed] [Google Scholar]
- 13.Lykke J, Jess P, Roikjaer O. Increased lymph node yield is associated with improved survival in rectal cancer irrespective of neoadjuvant treatment: results from a national cohort study. Dis Colon Rectum. 2015;58(9):823–830. doi: 10.1097/dcr.0000000000000429. [DOI] [PubMed] [Google Scholar]
- 14.Mechera R, Schuster T, Rosenberg R, Speich B. Lymph node yield after rectal resection in patients treated with neoadjuvant radiation for rectal cancer: a systematic review and meta-analysis. Eur J Cancer. 2017;72:84–94. doi: 10.1016/j.ejca.2016.10.031. [DOI] [PubMed] [Google Scholar]
- 15.Scott N, Thorne C, Jayne D. Lymph node retrieval after neoadjuvant radiotherapy for rectal adenocarcinoma. J Clin Pathol. 2004;57(3):335–336. doi: 10.1136/jcp.2003.013441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Baxter NN, Morris AM, Rothenberger DA, Tepper JE. Impact of preoperative radiation for rectal cancer on subsequent lymph node evaluation: a population-based analysis. Int J Radiat Oncol Biol Phys. 2005;61(2):426–431. doi: 10.1016/j.ijrobp.2004.06.259. [DOI] [PubMed] [Google Scholar]
- 17.Abdel-Misih SR, Wei L, Benson AB, 3rd, Cohen S, Lai L, Skibber J, Wilkinson N, Weiser M, Schrag D, Bekaii-Saab T. Neoadjuvant therapy for rectal cancer affects lymph node yield and status without clear implications on outcome: the case for eliminating a metric and using preoperative staging to guide therapy. J Natl Compr Canc Netw. 2016;14(12):1528–1534. doi: 10.6004/jnccn.2016.0164. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Rullier A, Laurent C, Capdepont M, Vendrely V, Belleannee G, Bioulac-Sage P, Rullier E. Lymph nodes after preoperative chemoradiotherapy for rectal carcinoma: number, status, and impact on survival. Am J Surg Pathol. 2008;32(1):45–50. doi: 10.1097/PAS.0b013e3180dc92ab. [DOI] [PubMed] [Google Scholar]
- 19.Greenlee RT, Hill-Harmon MB, Murray T, Thun M. Cancer statistics, 2001. CA Cancer J Clin. 2001;51(1):15–36. doi: 10.3322/canjclin.51.1.15. [DOI] [PubMed] [Google Scholar]
- 20.Krzyzanowska MK, Regan MM, Powell M, Earle CC, Weeks JC. Impact of patient age and comorbidity on surgeon versus oncologist preferences for adjuvant chemotherapy for stage III colon cancer. J Am Coll Surg. 2009;208(2):202–209. doi: 10.1016/j.jamcollsurg.2008.10.016. [DOI] [PubMed] [Google Scholar]
- 21.Biondi A, Vacante M, Ambrosino I, Cristaldi E, Pietrapertosa G, Basile F. Role of surgery for colorectal cancer in the elderly. World journal of gastrointestinal surgery. 2016;8(9):606–613. doi: 10.4240/wjgs.v8.i9.606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Chan DKH, Tan KK. Lower lymph node yield following neoadjuvant therapy for rectal cancer has no clinical significance. Journal of gastrointestinal oncology. 2019;10(1):42–47. doi: 10.21037/jgo.2018.10.02. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Data Availability Statement
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.