Abstract
Ulcerative colitis (UC) is a complex chronic pathological condition of the gut in which microbiota targeted treatment, such as fecal microbiota transplantation (FMT), has shown an encouraging effect. The aim of the present study was to investigate the efficacy and safety of FMT in patients with mild or moderate UC. A single-center, open-label study was designed, including 47 patients with mild or moderate active UC who received three treatments of fresh FMT via colonic transendoscopic enteral tubing within 1 week. The inflammatory bowel disease questionnaire, partial Mayo scores, colonoscopy, erythrocyte sedimentation rate, C-reactive protein level and procalcitoin values were used to assess the efficacy of FMT and alteration in gut microbiota was detected by 16S ribosomal RNA-sequencing. Before FMT, microbiota Faecalibacterium prausnitzii (F. prausnitzii) levels were significantly decreased in patients with UC compared with healthy donors (P<0.01). At 4 weeks post-FMT, F. prausnitzii levels were significantly increased (P<0.05), and the Mayo score was significantly decreased (1.91±1.07 at baseline vs. 4.02±1.47 at week 4; P<0.001) in patients with UC compared with healthy donors. Steroid-free clinical responses were reported in 37 patients (84.1%), and steroid-free clinical remission was achieved in 31 patients (70.5%) at week 4 post-FMT, however, steroid-free remission was not achieved in any patient. No adverse events were reported in 41 (93.2%) patients after FMT or during the 12-week follow-up. Shannon's diversity index and Chao1 estimator were also improved in patients with UC receiving FMT. In conclusion, the results of the present study suggested that FMT resulted in clinical remission in patients with mild to moderate UC, and that the remission may be associated with significant alterations to the intestinal microbiota of patients with UC. Furthermore, F. prausnitzii may serve as a diagnostic and therapeutic biomarker for the use of FMT in UC.
Keywords: fecal microbiota transplantation, ulcerative colitis, gut microbiota, colonoscopy, Faecalibacterium prausnitzii
Introduction
Inflammatory bowel disease (IBD), including ulcerative colitis (UC) and Crohn's disease, is characterized by chronic relapsing inflammation of the gastrointestinal (GI) tract (1). Over the past 20 years, the incidence and prevalence of IBD has risen sharply in developing areas of the world, including in Asia, South America, the Middle East and Africa (2). In China, the total number of IBD cases between 2005 and 2014 was ~350,000 and it has been predicted that the number of patients with IBD will reach 1.5 million by 2025(2). IBD is primarily caused by the aberrant activation of the immune system in response to abnormal alterations to the gut environment (3). Furthermore, the diverse microbiota in the GI tract serves a critical role in the development of IBD (4). In a previous study, significantly increased levels of Fusobacterium spp. and Enterrococcus faecalis were identified in the feces of patients with IBD compared with healthy controls (5). IBD has also been reported to be correlated with decreased levels of Erysipelotrichales, Bacteroidales, Clostridiales and Faecalibacterium prausnitzii, and an increased abundance of Enterobacteriaceae, Pasteurellacaea, Veillonellaceae, Proteobacter and Fusobacteriaceae (6).
The treatment options for UC are based on a variety of parameters, including patient age, severity of disease, relapse frequency and disease course (7). At present, the standard treatment strategies for UC include anti-inflammatory therapeutics, systemic administration of steroids, immunosuppressants and biologics, and even surgery (8). However, a substantial proportion of patients with UC are resistant or intolerant to the standard treatment strategies, therefore, the manipulation of enteric microbiota has become a focus for the treatment of UC (6).
Fecal microbiota transplantation (FMT) is a therapeutic process in which the fecal microbiota of a healthy donor is transplanted into a diseased recipient to reconstruct the gut microbial community and restore microbial homeostasis (9). Based on a previous study that reported an overall cure rate of 90% for refractory or recurrent Clostridium difficile (C. difficile) infection with FMT, researchers are investigating the use of FMT in intestinal diseases, including in IBD (10). Previous studies have demonstrated the therapeutic effects of FMT in IBD (11,12); however, its effects on UC have not been investigated extensively.
The safety and efficacy of FMT in patients with UC has been assessed in three small, randomized controlled clinical trials (RCTs) (13-15). The three studies differed in terms of infusion protocol, weekly treatment and clinical outcomes. The study conducted by Moayyedi et al (13) reported that FMT induced remission in patients with active UC. A total of 70 patients with active UC were treated weekly with FMT or water enema (placebo) for 6 weeks. The remission rate (full Mayo score ≤2; endoscopic Mayo score=0) in the FMT group was significantly higher compared with the placebo group (24% vs. 5%, respectively). A recently published systematic review conducted by Costello et al (16) meta-analyzed 14 cohort studies and 4 RCTs, including 308 FMT-treated patients with UC. In these meta-analyses of RCTs, it was reported that FMT effectively treated UC with a clinical remission rate of 28% (39/140) in patients treated with FMT, compared with 9% (13/137) in patients treated with the placebo. Furthermore, clinical response was achieved in 49% (69/140) of patients treated with FMT compared with 28% (38/137) of patients treated with the placebo. In the 14 cohort studies, 24% (39/168) of patients treated with FMT achieved clinical remission.
Although a number of studies have reported the beneficial effect of FMT for UC, it is still unclear how the GI microbiota impacts UC status. In the present study, the efficacy and safety of FMT was assessed in patients with mild to moderate active UC by assessing clinical responses and identifying associated components in the fecal microbiota.
Materials and methods
Study design and patients.
The present study was conducted to assess the efficacy and safety of FMT in patients who were treated for mild to moderate active UC at Guangzhou First People's Hospital between January 2017 and December 2017 and approved by the Ethics Committee of Guangzhou First People's Hospital (approval no. K-2017-103-02). In total, 47 patients (age, 18-75 years) were enrolled over this period. The baseline demographic and clinical characteristics of the patients are summarized in Table I. Written informed consent was obtained from all participants. UC was diagnosed based on clinical, endoscopic and histological criteria, including clinically and endoscopically active UC, a total Mayo score of 3-10, a Mayo endoscopy subscore ≥1 and a Physician's Global Assessment subscore ≤2(17). Mild activity was defined as a total Mayo score of 3-5 and moderate activity was defined as a score of 6-10. The exclusion criteria for patients with UC were as follows: Severe disease activity (total Mayo clinical score ≥10); indeterminate colitis; co-morbid chronic disease; food allergy; irritable bowel syndrome; history of bowel cancer, pregnancy or other severe diseases, including diabetes and cancer; GI surgery (except for appendicectomy) during the 3 months before enrollment; used antibiotics or probiotics during the 4 weeks before enrollment; and had been followed up for <8 weeks. Furthermore, patients with GI infections, including parasitic and C. difficile infections, were excluded from the study. During the current study, concomitant treatments using 5-aminosalicylic acid, immunomodulators or anti-tumor necrosis factor (TNF) agents were permitted, as long as the dose was stable prior to enrollment. Furthermore, patients were not allowed to take antibiotics, probiotics or corticosteroids during the present study, and patients who were previously on steroid treatment (corticosteroids) were taken off of the treatment 1 month prior to enrollment.
Table I.
Baseline patient characteristics.
| Parameters | Data |
|---|---|
| Total patients | 44.0 |
| Age (years) | 44.4±15.5 |
| Sex | |
| Male | 25.0 (57%) |
| Female | 19.0 (43%) |
| Smoke | |
| Smoker | 9.0 (20%) |
| Non-smoker | 35.0 (80%) |
| Disease extent | |
| Proctitis | 10.0 (23%) |
| Left-sided | 24.0 (54%) |
| Extensive | 10.0 (23%) |
| Disease duration (months) | 55.7±25.3 |
| Concomitant drugs | |
| None | 4.0 (9%) |
| Oral 5-aminosalicylate | 28.0 (64%) |
| Oral immunomodulator (azathioprine, cyclosporine, methotrexate) | 10.0 (23%) |
| Oral steroids | 11.0 (25%) |
| Previous anti-TNF therapy | 4.0 (9%) |
| Total Mayo score | 5.9±2.0 |
| Mayo endoscopic subscore | 1.9±0.7 |
| 1 | 13.0 (30%) |
| 2 | 23.0 (52%) |
| 3 | 8.0 (18%) |
| UCEIS score | 4.4±2.1 |
| Mayo clinical score | 4.0±1.5 |
| IBDQ score | |
| ESR (mm/h) | 25.5±20.6 |
| CRP (mg/l) | 3.0±0.7 |
| PCT (ng/ml) | 0.1±0.0 |
| White blood cell count (x109 cells/l) | 7.3±1.9 |
| Red blood cell count (x1012 cells/l) | 4.4±0.7 |
| Hemoglobin (g/l) | 121.5±24.8 |
| Platelet count (x109 cells/l) | 309.3±112.7 |
| Albumin (g/l) | 37.8±4.4 |
Data are presented as the number of patients (%) or the mean ± standard deviation. Total Mayo scores range from 0-12 and Mayo endoscopic subscores range from 0-3; higher scores indicate severe disease. UCEIS scores range from 0-8; higher scores indicate severe endoscopic disease. IBDQ scores range from 32-224; higher scores indicate an improved quality of life. TNF, tumor necrosis factor; UCEIS, Ulcerative Colitis Endoscopic Index of Severity; IBDQ, Inflammatory Bowel Disease Questionnaire; ESR, erythrocyte sedimentation rate; CRP, C-reactive protein; PCT, procalcitonin.
Healthy stool donors were recruited and screened using previously described criteria (18). Healthy donors (3; sex, male; age, 24-29 years; median age, 25 years) were selected from volunteers able to attend Guangzhou First People's Hospital, who were not pregnant and had good dietary and sleep habits (reported sleep of 7-8 h per day; reported taking part in physical exercise >3 times per week; did not eat fast food; did not smoke; did not drink alcohol). Prior to sample donation, the following laboratory tests were performed: Blood (complete blood count), erythrocyte sedimentation rate (ESR), procalcitonin (PCT), C-reactive protein (CRP), biochemical, hepatitis A, hepatitis B, hepatitis C, human immunodeficiency virus (HIV), syphilis and stool tests (stool parasites, ova and culture). Donor exclusion criteria were as follows: Infectious diseases (HIV, hepatitis B or C); high-risk sexual behavior; use of illicit drugs; communicable disease (for example, upper respiratory tract infection); GI co-morbidities (history of or current IBD, irritable bowel syndrome, chronic constipation, chronic diarrhea, intrinsic GI illness/condition, history of or current GI malignancy or polyposis, family history of colorectal cancer or history of major GI surgery); use of antimicrobials, probiotics or systemic antineoplastic agents during the 12 weeks prior to sample collection; other conditions, including systemic autoimmunity, metabolic syndrome, obesity (body mass index ≥30) or moderate to severe undernutrition/malnutrition; history of malignant illness; and ongoing oncologic therapy.
Transendoscopic enteral tubing (TET) tube insertion.
Standard bowel preparation (19) was performed using polyethylene glycol-electrolyte solution and subsequently, a colonoscopy was performed in each patient to examine the whole colon and distal ileum. A TET (FMT Medical Co., Ltd.) tube was inserted via the anus as far as the terminus of the ileum, using an endoscope, and while the head of the TET tube was kept stable, the endoscope was carefully removed. The endoscope was reinserted and the head of the TET was fixed with a clamp that was attached to the intestinal wall (Fig. S1A). An additional two loops of the TET were fixed with clamps to the intestinal wall while removing the endoscope (Fig. S1B and C). The end of the TET was fixed with tape to the sacral skin (Fig. S1D), as previously described (20,21). The appearance of the TET tube is detailed in Fig. S2.
Feces preparation and intervention.
The fresh fecal samples were collected in a clean room next to the FMT operating room. The feces was inspected visually by examining the form and the presence of blood and/or mucous. Within 0.5 h of collection, ~150-200 g donated fresh feces was dissolved in 1000 ml physiological saline and was purified using the GenFMTer automatic purification system (FMT Medical Co., Ltd.), which performs microfiltration and centrifugation steps to obtain a centrifuged microbiota sample (according to the manufacturers protocol). At 1 day post-TET insertion, 150 ml physiological saline containing ~50 cm3 centrifuged microbiota was infused into the entire colon of the patient via the TET tube. Patients were required to remain in the right lateral position for ≥30 min after FMT (to maintain the largest contact area between microbiota and intestines) and were allowed to eat 2 h later. The FMT procedure was repeated every other day for a total of 3 treatments, with each patient only receiving FMT obtained from the same donor (20). The FMT of each donor was batched and fecal samples were stored at -80˚C until further use.
Follow-up.
FMT follow-up visits were scheduled at weeks 1, 4 and 12. At each visit, bowel frequency, bleeding, GI symptoms, adverse events (AEs), changes in medication and quality of life of the patient were assessed using the Inflammatory Bowel Disease Questionnaire (IBDQ) (22). The partial Mayo scores were calculated based on bowel frequency, rectal bleeding and the Physician's Global Assessment score. At weeks 4 and 12, blood and stool samples were collected for blood tests and the determination of ESR, CRP and PCT values. Molecular microbiological analyses were performed using fecal samples obtained from patients 1 day prior to FMT, and 4 and 12 weeks after FMT.
Extraction of genomic DNA.
Total genomic DNA was extracted from the fecal samples using the cetyl trimethylammonium bromide/sodium dodecyl sulfate method and DNA concentration and purity were monitored by gel electrophoresis using 1% agarose gels, as previously described (23). DNA was diluted to 1 ng/µl with sterile water before loading on the gel.
Amplicon generation.
16S ribosomal (r)RNA/18S rRNA/internal transcribed spacer (ITS) genes in different regions (16S V4, 16S V3-V4, 16S V4-V5, 18S V4, 18S V9, ITS1 and ITS2) were amplified using the following primers: 16S V4, 515 forward, 5'-GTGCCAGCMGCCGCGGTAA-3' and 806 reverse, 5'-GGACTACHVGGGTWTCTAAT-3'; 16S V3-V4, 341 forward, 5'-CCTAYGGGRBGCASCAG-3' and 806 reverse, 5'-GGACTACNNGGGTATCTAAT-3'; 16S V4-V5, 515 forward, 5'-GTGCCAGCMGCCGCGGTAA-3' and 907 reverse, 5'-CCGTCAATTCCTTTGAGTTT-3';18S V4 528 forward, 5'-GCGGTAATTCCAGCTCCAA-3' and 706 reverse, 5'-AATCCRAGAATTTCACCTCT-3'; 18S V9 1380 forward, 5'-CCCTGCCHTTTGTACACAC-3' and 1510 reverse, 5'-CCTTCYGCAGGTTCACCTAC-3'; ITS1 1F forward, 5'-CTTGGTCATTTAGAGGAAGTAA-3' and 1F reverse, 5'-GCTGCGTTCTTCATCGATGC-3'; ITS2 ITS3-2024 forward, 5'-GCATCGATGAAGAACGCAGC-3' and ITS4-2409 reverse, 5'-TCCTCCGCTTATTGATATGC-3'. All PCR reactions were carried out in 30 µl reactions with 15 µl of Phusion® High-Fidelity PCR Master Mix (New England Biolabs, Inc.); 0.2 µM of forward and reverse primers, and ~10 ng template DNA. Thermal cycling consisted of initial denaturation at 98˚C for 1 min, followed by 30 cycles of denaturation at 98˚C for 10 sec, annealing at 50˚C for 30 sec and elongation at 72˚C for 30 sec. Final extension was at 72˚C for 5 min. Subsequently, the PCR products were mixed with the same volume of 1X Loading Buffer (Takara Bio, Inc.) and visualized by gel electrophoresis on a 2% agarose gel. The PCR products were excised and purified from the agarose gel using the GeneJETTM Gel Extraction kit (Thermo Fisher Scientific, Inc.) according to the manufacturer's protocol.
Library preparation and sequencing.
Sequencing libraries were generated from the amplified genomic DNA using the Ion Plus Fragment Library kit (48 rxns; Thermo Fisher Scientific, Inc.), according to the manufacturer's protocol. Library quality was assessed on the Qubit® 2.0 Fluorometer (Thermo Fisher Scientific, Inc.). The library was sequenced on the Ion S5™ XL system (Thermo Fisher Scientific, Inc.) and single-end reads (400/600 bp) were generated.
Sequence analyses.
Sequence analyses were performed using Uparse software (version 7.0.1001; drive5.com/uparse) (24). Sequences with ≥97% similarity were assigned to the same operational taxonomic units (OTUs). Representative sequences from each OTU were screened for further annotation. After the samples were rarefied to the same sequencing depth, and α and β diversities, differential OTU abundance analyses were performed using Uparse and Quantitative Insights Into Microbial Ecology software (version 1.7.0; qiime.org/) (25). Shannon's diversity index was used to display the diversity of the gut microbiota. Chao1 estimator was used to display the richness of the microbiota. Metastats analysis was used to analyze the diversity of bacteria in different groups (26).
Outcomes.
The primary outcomes referred to as steroid-free clinical responses at week 4 after FMT were as follows: A decrease of ≥3 points in the Mayo score, a reduction ≥50% in total Mayo subscores of rectal bleeding plus stool frequency, or both. The secondary outcomes were as follows: Steroid-free clinical remission (total Mayo subscore ≤1 for rectal bleeding plus stool frequency), steroid-free remission (total Mayo score ≤2 with no individual subscore ≥1, and mucosal healing defined by a Mayo endoscopy subscore ≤1), quality of life (assessed with the IBDQ) and safety (assessed by AEs) (27). At week 12 after FMT, the patients were categorized into responder and non-responder groups, a responder was defined by a decrease of ≥3 points in the Mayo score, a reduction ≥50% in total Mayo subscores of rectal bleeding plus stool frequency, or both; a non-responder was defined by a decrease of <3 points in the Mayo score and a reduction <50% in total Mayo subscores of rectal bleeding plus stool frequency.
Statistical analysis.
Data are presented as the percentage of patients or the mean ± standard deviation. The χ2 test, unpaired t-test, Fisher's exact test or Wilcoxon signed rank test were used to compare data containing two groups. One-way ANOVA followed by Tukey's post hoc test was used to compare data containing ≥3 groups. All statistical analyses were performed using SPSS software (version 23.0; IBM Corp.). P<0.05 was considered to indicate a statistically significant difference.
Results
Clinical results.
Between January 2017 and December 2017, a total of 78 patients were recruited, however, only 47 were enrolled in the present study, as some did not meet the inclusion criteria/met one of the exclusion criteria. After FMT, three patients dropped out of the study before week 4, therefore, only 44 patients were included in the analyses at week 4 and 12. The baseline demographic and clinical characteristics of the patients are summarized in Table I, and the FMT outcomes are summarized in Table II. At week 4, steroid-free clinical response and steroid-free clinical remission were observed in 37 (84.1%) and 31 (70.5%) patients, respectively. Steroid-free remission was not achieved in any patient (Table II). Accordingly, the patients were categorized into responder and non-responder groups. These two groups displayed no significant differences in their baseline demographic and clinical characteristics (Table III). The clinical response rate was higher in patients with mild active UC (18/23, 78%) compared with patients with moderate active UC (16/21, 76%), although no statistical significance was observed between these two groups. The clinical response rate in patients with a lower Mayo endoscopic subscore (10/13, 77%) was similar compared with patients with a higher subscore (24/31, 77%; Table III). Furthermore, the UC endoscopic index of severity scores suggested a similar outcome to the Mayo endoscopic subscores (Table III). The association between the clinical response rate and clinical parameters, including age, sex, smoking, disease extent, disease duration, IBDQ scores, concomitant drug use and donors, was also assessed, and no significant associations were identified (P>0.05; Table III). The Mayo clinical score was significantly decreased at week 4 compared with the baseline score in the FMT group (4.02±1.47 at baseline; 1.91±1.07 at week 4; P<0.001; Fig. 1). At week 4, the mucosal activity was reassessed by colonoscopy in 10 responders. All 10 responders displayed a steroid-free endoscopic response (Mayo endoscopy subscore ≤1 with a reduction of ≥1 point from the baseline) and none achieved steroid-free endoscopic remission (Mayo endoscopy subscore=0; Table II). Representative images of the colons of two responders displaying steroid-free endoscopic remission are presented in Fig. 2. No serious complications, including anaphylactic shock or septicemia, occurred in 41/44 patients following FMT or during the 12-week follow-up. Increased diarrhea frequency was reported in three patients within 24 h of FMT, and two patients experienced abdominal pain within 6 h of FMT. These AEs were short-term, and disappeared within 1 day without any medical intervention. Of the patients with UC, 94% (32/34) of responders and 60% (6/10) of non-responders were willing to undergo FMT again (data not shown).
Table II.
Outcomes of fecal microbiota transplantation.
| Outcome | Week 4 | Week 12 |
|---|---|---|
| Steroid-free clinical remission | 31 (70.5%) | 30 (68.2%) |
| Steroid-free clinical response | 37 (84.1%) | 34 (77.3%) |
| Steroid-free remission | 0 | 0 |
Data are presented as the mean±standard deviation (= 44). Steroid-free clinical remission was defined as a total Mayo subscore ≤1 for rectal bleeding plus stool frequency. Steroid-free clinical response was defined as a decrease by ≥3 points, reduction by ≥50% from baseline or both in total Mayo subscores for rectal bleeding plus stool frequency. Steroid-free remission was defined as a total Mayo score ≤2, no individual subscore ≥1 and Mayo endoscopy subscore ≤1.
Table III.
Baseline characteristics of responders and non-responders.
| Parameters | Responders (n=34) | Non-responders (n=10) | P-value |
|---|---|---|---|
| Age (years) | 42.7±14.5 | 49.1±14.4 | 0.25 |
| Sex | 0.30 | ||
| Male | 22.0 (62%) | 4.0 (40%) | |
| Female | 12.0 (38%) | 6.0 (60%) | |
| Smoke | 0.69 | ||
| Smoker | 6.0 (18%) | 3.0 (30%) | |
| Non-smoker | 28.0 (82%) | 7.0 (70%) | |
| Disease extent | 0.93 | ||
| Proctitis | 8.0 (24%) | 2.0 (20%) | |
| Left-sided | 18.0 (52%) | 6.0 (60%) | |
| Extensive | 8.0 (24%) | 2.0 (20%) | |
| Disease duration (months) | 56.4±24.4 | 53.3±29.5 | 0.74 |
| Concomitant drugs | |||
| None | 3.0 (9%) | 1.0 (10%) | |
| Oral 5-aminosalicylate | 21.0 (62%) | 7.0 (70%) | 0.92 |
| Oral immunomodulator | 8.0 (24%) | 2.0 (20%) | 0.84 |
| Oral steroids | 9.0 (26%) | 2.0 (20%) | 1.00 |
| Previous anti-TNF therapy | 3.0 (9%) | 1.0 (10%) | |
| Total Mayo score | 5.9±2.0 | 5.8±2.0 | 0.84 |
| 3-5 | 18.0 (53%) | 5.0 (50%) | |
| 6-10 | 16.0 (47%) | 5.0 (50%) | |
| Mayo endoscopic subscore | 0.98 | ||
| 1 | 10.0 (29%) | 3.0 (30%) | |
| 2 | 18.0 (53%) | 5.0 (50%) | |
| 3 | 6.0 (18%) | 2.0 (20%) | |
| UCEIS score | 4.3±2.1 | 4.5±2.2 | 0.81 |
| Mayo clinical score | 4.1±1.5 | 3.9±1.5 | 0.77 |
| IBDQ score | |||
| ESR (mm/h) | 22.4±18.4 | 35.8±25.1 | 0.07 |
| CRP (mg/l) | 2.7±3.5 | 5.1±2.0 | 0.42 |
| PCT (ng/ml) | 0.1±0.1 | 0.1±0.0 | 0.85 |
| White blood cell count (x109 cells/l) | 7.1±1.9 | 8.0±1.5 | 0.13 |
| Red blood cell count (x1012 cells/l) | 4.4±0.7 | 4.4±0.6 | 0.82 |
| Hemoglobin (g/l) | 123.0±23.7 | 116.5±29.2 | 0.74 |
| Platelet count (x109 cells/l) | 308.5±118.8 | 358.6±57.8 | 0.13 |
| Albumin (g/l) | 38.2±4.3 | 36.6±5.0 | 0.33 |
| Donor identifier no. | 0.30 | ||
| 1 | 17.0 (50%) | 3.0 (30%) | |
| 2 | 1.0 (3%) | 1.0 (10%) | |
| 3 | 16.0 (47%) | 6.0 (60%) |
Data are presented as the number of patients (%) or the mean ± SD. Total Mayo scores range from 0-12 and Mayo endoscopic subscores range from 0-3; higher scores indicate severe disease. UCEIS scores range from 0-8; higher scores indicate severe endoscopic disease. IBDQ scores range from 32-224; higher scores indicate an improved quality of life. TNF, tumor necrosis factor; UCEIS, Ulcerative Colitis Endoscopic Index of Severity; IBDQ, Inflammatory Bowel Disease Questionnaire; ESR, erythrocyte sedimentation rate; CRP, C-reactive protein; PCT, procalcitonin.
Figure 1.
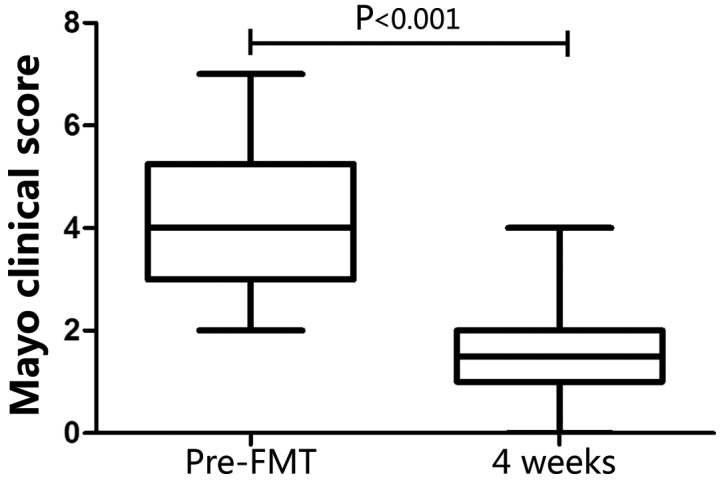
Clinical and endoscopic responses to triple infusions of FMT. The efficacy of FMT at week 4 was evaluated using the Mayo clinical score. The lower and upper margins of the box represent the 25th and 75th percentiles, with the extended arms representing the 10th and 90th percentiles, respectively. The median is presented as a horizontal line within the box. FMT, fecal microbiota transplantation.
Figure 2.
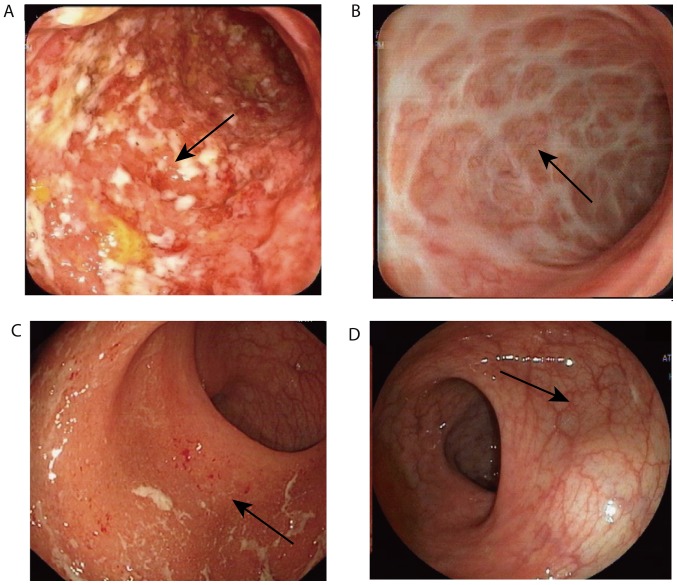
Steroid-free endoscopic responses after FMT in two cases. A 37-year-old woman with a six-year history of left-sided UC and acute colitis (diarrhea six times per day with bleeding and abdominal pain) was treated with 4 g oral 5-aminosalicylate and 40 mg prednisone per day. (A) Baseline endoscopic appearance of a 30 cm rectosigmoid active colitis: Total Mayo score=8 and endoscopic subscore=3. Arrows: Diffuse mucosal erosion with marked hyperemia, edema, hemorrhage and purulent secretions. (B) Endoscopic appearance at the end of week 4 after FMT: Total Mayo score=1, endoscopic subscore=1. Arrows: White scar formation. The patient displayed steroid-free clinical remission until the follow-up at 12 weeks post-FMT. A 61-year-old woman with a two-year history of left-sided UC and acute colitis (diarrhea five times per day with bleeding) was treated with 4 g oral 5-aminosalicylate and 40 mg prednisone per day. (C) Baseline endoscopic appearance of a 25 cm rectosigmoid active colitis: Total Mayo score=7 and endoscopic subscore=2. Arrows: Erosion, mucosal congestion and edema and a small amount of purulent discharge. (D) Endoscopic appearance at 4 weeks post-FMT; total Mayo score=1 and endoscopic subscore=0. Arrow: Mucosa is intact and the blood vessel texture is slightly blurred. After FMT, the patient was completely weaned off corticosteroids and maintained their clinical and endoscopic remission until week 4 post-FMT. FMT, fecal microbiota transplantation; UC, ulcerative colitis.
Microbiome results.
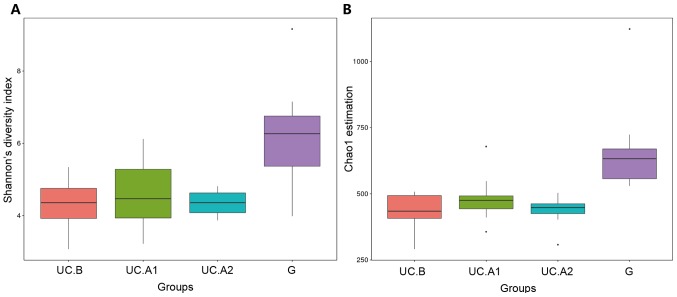
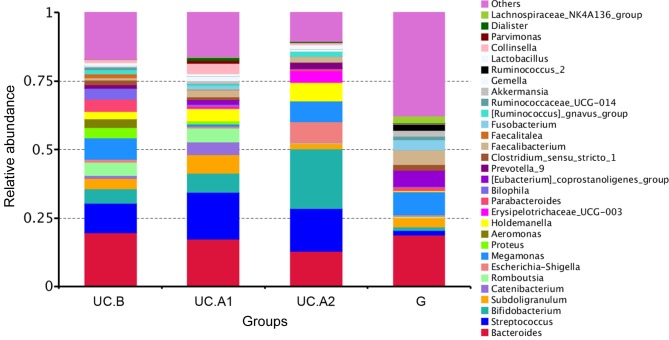
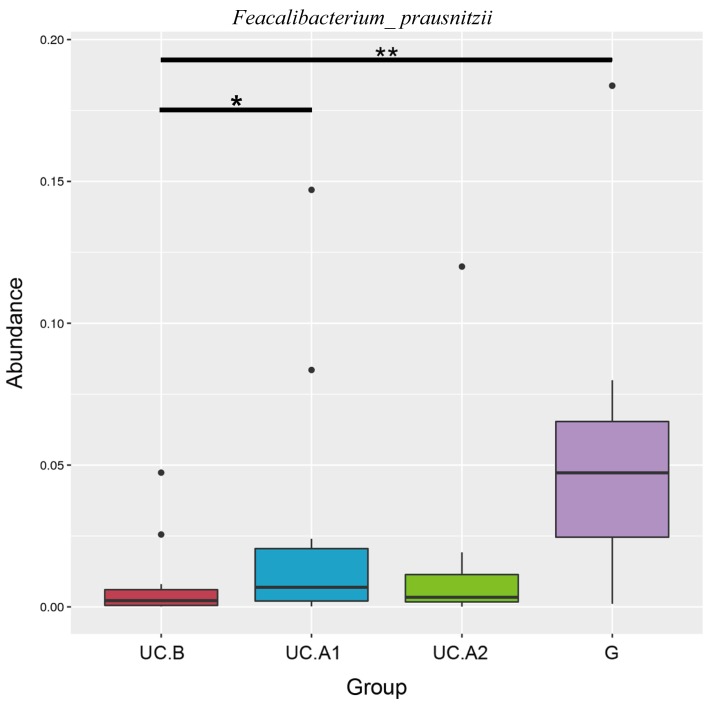
Fecal samples obtained 1 day before FMT, and at weeks 4 and 12 post-FMT were evaluated. Microbiota analyses were performed in 32 fecal samples from 12 patients, who were all responders, and 12 donors. The fecal samples were categorized into four groups: Pre-FMT (UC.B), week 4 (UC.A1), week 12 (UC.A2) and donors (G). The diversity (Shannon's diversity index) and richness of the fecal microbiota (Chao1 estimator) were markedly decreased in patients with UC pre-FMT compared with healthy donors (P<0.05; Fig. 3A and B). After FMT, these two indicators suggested that the diversity and richness of the fecal microbiota were increased at week 4 post-FMT but then slightly decreased at week 12 post-FMT (Fig. 3A and B). Patients with UC displayed a decreased abundance in Ruminococcus_2 and Faecalibacterium, and an increased abundance in Bifidobacterium, Escherichia-Shigella, Faecalitalea, Streptococcus, Aeromonas and Proteobacter (Fig. 4). After FMT, some alterations in the abundance of microbiota in patients with UC were markedly similar to that of healthy donors. F. prausnitzii abundance displayed a significant decrease in patients with UC compared with healthy donors (P<0.01; Fig. 5). At week 4 post-FMT, F. prausnitzii abundance was significantly increased (P<0.05) in patients with UC compared with the baseline abundance, and was decreased compared with healthy donors (P<0.05; Fig. 5).
Figure 3.
α diversity analysis. (A) The diversity of the fecal microbiota (Shannon's diversity index). (B) The richness of the fecal microbiota (Chao1 estimator). The lower and upper margins of the box represent the 25th and 75th percentiles, with the extended arms representing the 10th and 90th percentiles, respectively. The median is presented as a horizontal line within the box. UC, ulcerative colitis; UC.B, patients before FMT; UC.A1, patients 4 weeks after FMT; UC.A2, patients 12 weeks after FMT; G, healthy donors.
Figure 4.
Relative abundance of fecal microbiota composition at the genus level. UC, ulcerative colitis; UC.B, patients before FMT; UC.A1, patients 4 weeks after FMT; UC.A2, patients 12 weeks after FMT; G, healthy donors.
Figure 5.
F. prausnitzii abundance in different groups. F. prausnitzii, Faecalibacterium prausnitzii; UC.B, patients before FMT; UC.A1, patients 4 weeks after FMT; UC.A2, patients 12 weeks after FMT; G, healthy donors (*: P<0.05; **: P<0.01).
Discussion
The present study demonstrated that three rounds of FMT effectively treated mild to moderate active UC, as evidenced by the steroid-free clinical responses that occurred in 77.3% (34/44) of patients at 12 weeks post-FMT. The Mayo clinical score significantly decreased at week 4 compared with the baseline in FMT treated patients (1.91±1.07 vs. 4.02±1.47; P<0.001). No AEs occurred in 93.2% (41/44) of patients after FMT or during the 12 weeks of follow-up (data not shown). The results suggested an improved effect of FMT in patients with UC compared with previous studies (13,28-30). The improved efficacy displayed in the present study may be explained by the modified FMT procedures used, including stool preparation (feces exposure to air for <30 min), microbiota resource (fresh; all FMTs for one patient were derived from the same donor), delivery route (via TET tube; remained in the right lateral position for 30 min to maintain the largest contact area between microbiota and intestines), dosage (150 ml to avoid inducing intestinal peristalsis) and intensity (three times within one week).
Fresh donor feces have to be diluted and homogenized to an administrable form before FMT can be performed (31). In the majority of previous studies (32-35), donor stool was mixed in normal sterile, non-bacteriostatic saline, which is presumed to guarantee the quality of microbiota. Subsequently, the mixture was homogenized, often manually, and filtered using a gauze, strainer or coffee filter. This purification process may alter the bacterial levels in the fecal suspension. In the present study, the fresh donor feces were microfiltered and centrifuged using an automatic purification system, which ensured the quality of fecal microbiota in the suspension.
It is not clear whether fresh or frozen-thawed microbiota improves the efficiency of FMT; however, frozen-thawed microbiota is a more convenient method (36,37). Hamilton et al (38) reported the successful use of standardized, partially purified and frozen fecal microbiota to treat C. difficile infection. High-throughput 16S rRNA gene sequencing has displayed stable engraftment of gut microbiota following FMT (39). Moayyedi et al (13) reported efficacy of frozen-thawed stool in FMT for active UC. Furthermore, other previous studies have reported that both frozen (15) and fresh (13,14) donor stool were effective for UC. However, Nishida et al (40) doubted the efficacy of using fresh stool for FMT in patients with UC, but the present study further suggested that this application was efficacious in UC. Another debate regarding FMT is whether pooled or single donor stool results in the highest efficacy. In a study including 81 patients who received FMT or placebo enemas for 5 days a week for 8 weeks, Paramsothy et al (15) used pooled donor stool. The aim when using pooled donor stool was to increase the diversity of microorganisms in the stool suspension. Cao et al (12) reported that 27% of the patients in the FMT group displayed steroid-free clinical and endoscopic remission at week 8 compared with 8% in the placebo group (P=0.02). However, in a study conducted by Rossen et al (14), no statistically significant difference in clinical and endoscopic remission between the FMT and control groups of patients with mild to moderate active UC was reported. However, the microbiota of the responders displayed distinct features compared with the non-responders. A key advantage of a pooled stool is that it increases the chances of transmitting key bacteria to the recipient; however, it is not clear whether this hypothesis is translated into real efficacy (41,42).
The frequency and duration between each FMT also impacts the outcomes. Paramsothy et al (15) used a large dose over a long duration (5 days per week for 8 weeks; 40 doses; 1500 g), and Moayyedi et al (13) delivered 8.3 g stool per week for 6 weeks via enema (6 doses; a total of 49.8 g). These two aforementioned studies achieved similar remission rates, therefore, it could be hypothesized that moderate dose and frequent application may improve the efficiency of FMT.
A number of systematic reviews have reported various approaches of FMT for the treatment of IBD, including colonoscopy, retention enema, nasoduodenal tube, pills or a combination (43,44). Paramsothy et al (15) used the retention enema approach to perform FMT in patients with UC, while Gordon et al (11) employed a nasoduodenal tube approach, and Cold et al (28) investigated the use of FMT capsules. According to two retrospective studies (45,46), nasointestinal delivery can be uncomfortable and result in a number of AEs, including severe nausea, vomiting, reduced food intake and sensory discomfort; delivery of the fecal microbiota suspension to the cecum may be difficult with the retention enema method; furthermore, the FMT capsule preparation increases the exposure time of the microbiota to the air, thus affecting bacterial activity. In the present study, a TET fixed to the cecum was used for the delivery of the fecal microbiota suspension to ensure the infusion of fecal microbiota into the whole colon. The high remission rate in the present study may be explained by the standardized and automatic purification process, fresh microbiota, appropriate dosage and intensity, and use of TET.
The gut microbiota is involved in the development of intestinal inflammation and UC. Therefore, microbial manipulation could be an alternative therapeutic approach for UC (47). FMT may serve as an effective treatment strategy for UC, as it is able to correct the altered gut microbial community and restore microbial homeostasis (48). In the present study, both the diversity (Shannon's diversity index) and abundance (Chao1 estimator) of the fecal microbiota were markedly decreased in patients with UC compared with healthy donors. However, both indexes at pre-FMT, and weeks 4 and 12 post-FMT displayed no significant difference. The results of the present study suggested that the majority of patients with UC achieved steroid-free clinical response or remission, but no steroid-free remission, after receiving FMT, indicating that FMT may ameliorate UC, but did not cure the disease. Therefore, it could be suggested that patients with UC may require repeated FMT to treat the disease.
Consistent with previous studies, the present study suggested that alterations to the enteric microbiota following FMT were primarily manifested by the decrease of pathogenic bacteria (for example, Faecalitalea and Proteobacter) and the increase of probiotic bacteria (for example, Bifidobacterium, Ruminococcus and Faecalibacterium). However, pathogenic bacterium Escherichia-Shigella decreased at 4 weeks post-FMT but then increased at 12 weeks post-FMT compared with pre-FMT, possibly due to a reduction in the inhibitory effect of probiotics on pathogens over time. The present study focused on alterations to the abundance of F. prausnitzii, which is a type of anti-inflammatory and health-promoting probiotic (49). In the intestine, F. prausnitzii produce butyrate, a major energy source for colonocytes to fight against IBD (50). In addition, butyrate can reduce intestinal mucosa inflammation by inhibiting NF-κβ transcription factor activation, upregulating peroxisome proliferator-activated receptor-γ expression and inhibiting interferon-γ expression (51). F. prausnitzii abundance is correlated with various IBD-related signaling pathway mediators, including T helper 17 cells/interleukin (IL)-17, forkhead box 3-T regulatory-transforming growth factor-β/IL-10 and IL-23(52). A decreased abundance of F. prausnitzii is associated with the development and recurrence of UC (49). Similarly, it has been suggested that F. prausnitzii can be used as a biomarker for the diagnosis and treatment of IBD (53). In the present study, the abundance of F. prausnitzii was significantly decreased in patients with UC compared with healthy donors. After FMT, the levels of F. prausnitzii were significantly increased at week 4 compared with the baseline, but remained lower compared with healthy donors. F. prausnitzii abundance is strongly correlated with the diagnostic and therapeutic effectiveness of FMT (54).
The present study had a number of limitations. Firstly, the present study was an open-label study and not a double-blind RCT; therefore, the results may have included potential bias from the researchers and patients, and the placebo effect cannot be ruled out. Secondly, the limitations of TET are as follows: i) Patients had to take purgatives to clean the colon, which may have influenced the gut flora; ii) although the TET tube was maintained in the same position for repeated FMT delivery, the tube usually fell out of place spontaneously at ~1 week, therefore it may be difficult to use the TET method for long-term delivery; and iii) the TET tube was positioned using colonoscopy, therefore it could be suggested that the method is only suitable for diseases that occur near to the anus, and is not ideal for diseases localized in the small intestine. Thirdly, partial Mayo scores were used as important criteria for classifying disease severity, however, these scores are primarily focused on clinical symptoms. Furthermore, the mucosal activity was only determined by colonoscopy in 10 responders and not in all patients. Therefore, endoscopic remission or responses could not be investigated in the present study. Fourthly, the ESR and other related inflammatory indicators in non-responders were slightly increased after FMT. It was hypothesized that FMT could introduce a large number of exogenous gut microbiota into the patient's intestine, which might increase intestinal immunity. However, in the present study individual differences were not excluded and a larger sample size is required to further investigate the efficiency of FMT for UC. Finally, the observation period was only 12 weeks; therefore, future studies are required to assess the long-term outcomes of the TET therapeutic strategy for UC.
The therapeutic role of FMT in UC varies in different reports, as evidenced by the rate of clinical response ranging between 39 and 55% in four RCTs investigating the use of FMT in UC (55). Ramai et al (36) reported that the clinical response in patients at 1 month and 3 months after FMT was 74.3 and 51.4%, respectively. A pilot study in India reported 87.1% clinical response, 58.1% endoscopic remission and 45.2% histological remission at week 48 post-FMT (29). The patients with UC treated with FMT in the present study displayed a steroid-free clinical response rate of 84.1 and 77.3% at week 4 and week 12, respectively. The high response rate may have been due to the following reasons: i) The modified FMT procedures, including stool preparation, microbiota resource, delivery route, dosage, and intensity; ii) certain patients had an irregular medical history of taking antibiotics and hormones due to repeated enteritis, which may have been a reason for repeated episodes of UC; iii) the donors in the present study were young and had good habits and lifestyles, making the composition of the gut microbiota active and compatible. However, the FMT method requires further investigation into the effects of antibiotic use before FMT, fresh or frozen administration, and the location (upper GI or lower GI tract) and frequency of administration. In the present study, the TET procedure, frequency of administration and use of 150-200 g fresh donated feces were based on previous studies investigating the use of FMT treatment for UC (21,31,56,57). In addition, the 16S sequencing results of the present study suggested that the relative abundance of F. prausnitzii significantly increased at 4 weeks post-FMT, which had not been reported in previous FMT studies. F. prausnitzii is an important short-chain fatty acid bacteria in the intestine; therefore, it has been suggested that FMT should focus on the separation and cultivation of functional bacteria (49).
In conclusion, the results of the present study suggested that the delivery of fresh microbiota suspension via TET was an effective and safe method for patients with mild to moderate active UC. Furthermore, F. prausnitzii may serve as a diagnostic and therapeutic biomarker of FMT in patients with UC. Additionally, the results suggested that the donor selection, stool preparation, delivery route, dosage and intensity of FMT should be standardized. Further investigation using larger multi-center studies with a longer follow up period and data analysis on histology, endoscopy and the microbiome are required to confirm the efficacy and safety of FMT for UC remission.
Supplementary Material
Acknowledgements
Not applicable.
Funding
The present study was funded by the Guangzhou General Science and Technology Project of Health and Family Planning (grant nos. 20181A011007 and 20191A011001), the National Natural Science Foundation of China (grant no. NSFC 81871905), the Natural Science Foundation of Guangdong Province (grant. no. 2018A030313676) and the Guangzhou Planned Project of Science and Technology (grant nos. 201707010275 and 201904010132).
Availability of data and materials
The datasets analyzed during the present study are not publicly available due to patient privacy concerns but are available from the corresponding author on reasonable request.
Authors' contributions
HTC contributed to the design of the study, recruited the patients and drafted the manuscript. HLH performed the statistical analysis, interpreted the data and drafted the manuscript. HMX and QLL performed the sample collection and performed DNA extraction. JH collected the clinical information and performed the follow-up examinations. YQL and YLZ prepared the fecal samples into filtrate for administration during the FMT procedure. YQN and YJZ designed and supervised the study, interpreted the data and revised the manuscript.
Ethics approval and consent to participate
The present study was approved by the Ethics Committee of Guangzhou First People's Hospital (approval no. K-2017-103-02). Written informed consent was obtained from all patients.
Patient consent for publication
All patients provided written consent for publication of all figures/pictures including endoscopy and patient images.
Competing interests
The authors declare that they have no competing interests.
References
- 1.Feuerstein JD, Cheifetz AS. Ulcerative colitis: Epidemiology, diagnosis, and management. Mayo Clin Proc. 2014;89:1553–1563. doi: 10.1016/j.mayocp.2014.07.002. [DOI] [PubMed] [Google Scholar]
- 2.Kaplan GG. The global burden of IBD: From 2015 to 2025. Nat Rev Gastroenterol Hepatol. 2015;12:720–727. doi: 10.1038/nrgastro.2015.150. [DOI] [PubMed] [Google Scholar]
- 3.Loftus EV Jr. Clinical epidemiology of inflammatory bowel disease: Incidence, prevalence, and environmental influences. Gastroenterology. 2004;126:1504–1517. doi: 10.1053/j.gastro.2004.01.063. [DOI] [PubMed] [Google Scholar]
- 4.Takahashi K, Nishida A, Fujimoto T, Fujii M, Shioya M, Imaeda H, Inatomi O, Bamba S, Sugimoto M, Andoh A. Reduced abundance of butyrate-producing bacteria species in the fecal microbial community in Crohn's disease. Digestion. 2016;93:59–65. doi: 10.1053/j.gastro.2004.01.063. [DOI] [PubMed] [Google Scholar]
- 5.Zhou Y, Chen H, He H, Du Y, Hu J, Li Y, Li Y, Zhou Y, Wang H, Chen Y, Nie Y. Increased Enterococcus faecalis infection is associated with clinically active Crohn disease. Medicine (Baltimore) 2016;95(e5019) doi: 10.1097/MD.0000000000005019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kostic AD, Xavier RJ, Gevers D. The microbiome in inflammatory bowel disease: Current status and the future ahead. Gastroenterology. 2014;146:1489–1499. doi: 10.1053/j.gastro.2014.02.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Jackson B, De Cruz P. Algorithms to facilitate shared decision-making for the management of mild-to-moderate ulcerative colitis. Expert Rev Gastroenterol Hepatol. 2018;12:1079–1100. doi: 10.1080/17474124.2018.1530109. [DOI] [PubMed] [Google Scholar]
- 8.Coskun M, Vermeire S, Nielsen OH. Novel targeted therapies for inflammatory bowel disease. Trends Pharmacol Sci. 2017;38:127–142. doi: 10.1016/j.tips.2016.10.014. [DOI] [PubMed] [Google Scholar]
- 9.D'Haens GR, Jobin C. Fecal microbial transplantation for diseases beyond recurrent clostridium difficile infection. Gastroenterology. 2019;157:624–636. doi: 10.1053/j.gastro.2019.04.053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Sbahi H, Di Palma JA. Faecal microbiota transplantation: Applications and limitations in treating gastrointestinal disorders. BMJ Open Gastroenterol 3. 2016;e000087 doi: 10.1136/bmjgast-2016-000087. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Gordon H, Harbord M. A patient with severe Crohn's colitis responds to Faecal Microbiota Transplantation. J Crohns Colitis. 2014;8:256–257. doi: 10.1016/j.crohns.2013.10.007. [DOI] [PubMed] [Google Scholar]
- 12.Cao Y, Zhang B, Wu Y, Wang Q, Wang J, Shen F. The value of fecal microbiota transplantation in the treatment of ulcerative colitis patients: A systematic review and meta-analysis. Gastroenterol Res Pract 2018. 2018;(5480961) doi: 10.1155/2018/5480961. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Moayyedi P, Surette MG, Kim PT, Libertucci J, Wolfe M, Onischi C, Armstrong D, Marshall JK, Kassam Z, Reinisch W, Lee CH. Fecal microbiota transplantation induces remission in patients with active ulcerative colitis in a randomized controlled trial. Gastroenterology. 2015;149(102.e6-109.e6) doi: 10.1053/j.gastro.2015.04.001. [DOI] [PubMed] [Google Scholar]
- 14.Rossen NG, Fuentes S, van der Spek MJ, Tijssen JG, Hartman JH, Duflou A, Löwenberg M, van den Brink GR, Mathus-Vliegen EM, de Vos WM, et al. Findings from a randomized controlled trial of fecal transplantation for patients with ulcerative colitis. Gastroenterology. 2015;149(110.e4-118.e4) doi: 10.1053/j.gastro.2015.03.045. [DOI] [PubMed] [Google Scholar]
- 15.Paramsothy S, Kamm MA, Kaakoush NO, Walsh AJ, van den Bogaerde J, Samuel D, Leong RWL, Connor S, Ng W, Paramsothy R, et al. Multidonor intensive faecal microbiota transplantation for active ulcerative colitis: A randomised placebo-controlled trial. Lancet. 2017;389:1218–1228. doi: 10.1016/S0140-6736(17)30182-4. [DOI] [PubMed] [Google Scholar]
- 16.Costello SP, Soo W, Bryant RV, Jairath V, Hart AL, Andrews JM. Systematic review with meta-analysis: Faecal microbiota transplantation for the induction of remission for active ulcerative colitis. Aliment Pharmacol Ther. 2017;46:213–224. doi: 10.1111/apt.14173. [DOI] [PubMed] [Google Scholar]
- 17.Schroeder KW, Tremaine WJ, Ilstrup DM. Coated oral 5-aminosalicylic acid therapy for mildly to moderately active ulcerative colitis. A randomized study. N Engl J Med. 1987;317:1625–1629. doi: 10.1056/NEJM198712243172603. [DOI] [PubMed] [Google Scholar]
- 18.Paramsothy S, Borody TJ, Lin E, Finlayson S, Walsh AJ, Samuel D, van den Bogaerde J, Leong RW, Connor S, Ng W, et al. Donor recruitment for fecal microbiota transplantation. Inflamm Bowel Dis. 2015;21:1600–1606. doi: 10.1097/MIB.0000000000000405. [DOI] [PubMed] [Google Scholar]
- 19.Hassan C, Bretthauer M, Kaminski MF, Polkowski M, Rembacken B, Saunders B, Benamouzig R, Holme O, Green S, Kuiper T, et al. Bowel preparation for colonoscopy: European Society of Gastrointestinal Endoscopy (ESGE) guideline. Endoscopy. 2013;45:142–150. doi: 10.1055/s-0032-1326186. [DOI] [PubMed] [Google Scholar]
- 20.Cui B, Li P, Xu L, Zhao Y, Wang H, Peng Z, Xu H, Xiang J, He Z, Zhang T, et al. Step-up fecal microbiota transplantation strategy: A pilot study for steroid-dependent ulcerative colitis. J Transl Med. 2015;13(298) doi: 10.1186/s12967-015-0646-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Peng Z, Xiang J, He Z, Zhang T, Xu L, Cui B, Li P, Huang G, Ji G, Nie Y, et al. Colonic transendoscopic enteral tubing: A novel way of transplanting fecal microbiota. Endosc Int Open. 2016;4(E610-E613) doi: 10.1055/s-0042-105205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Guyatt G, Mitchell A, Irvine EJ, Singer J, Williams N, Goodacre R, Tompkins C. A new measure of health status for clinical trials in inflammatory bowel disease. Gastroenterology. 1989;96:804–810. [PubMed] [Google Scholar]
- 23.Cui B, Gai Z, She X, Wang R, Xi Z. Effects of chronic noise on glucose metabolism and gut microbiota-host inflammatory homeostasis in rats. Sci Rep. 2016;6(36693) doi: 10.1038/srep36693. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Edgar RC. UPARSE: Highly accurate OTU sequences from microbial amplicon reads. Nat Methods. 2013;10:996–998. doi: 10.1038/nmeth.2604. [DOI] [PubMed] [Google Scholar]
- 25.Caporaso JG, Kuczynski J, Stombaugh J, Bittinger K, Bushman FD, Costello EK, Fierer N, Peña AG, Goodrich JK, Gordon JI, et al. QIIME allows analysis of high-throughput community sequencing data. Nat Methods. 2010;7:335–336. doi: 10.1038/nmeth.f.303. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.White JR, Nagarajan N, Pop M. Statistical methods for detecting differentially abundant features in clinical metagenomic samples. PLoS Comput Biol. 2009;5(e1000352) doi: 10.1371/journal.pcbi.1000352. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Feagan BG, Patel H, Colombel JF, Rubin DT, James A, Mody R, Lasch K. Effects of vedolizumab on health-related quality of life in patients with ulcerative colitis: Results from the randomised GEMINI 1 trial. Aliment Pharmacol Ther. 2017;45:264–275. doi: 10.1111/apt.13852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Cold F, Browne PD, Günther S, Halkjaer SI, Petersen AM, Al-Gibouri Z, Hansen LH, Christensen AH. Multidonor FMT capsules improve symptoms and decrease fecal calprotectin in ulcerative colitis patients while treated-an open-label pilot study. Scand J Gastroenterol. 2019;54:289–296. doi: 10.1080/00365521.2019.1585939. [DOI] [PubMed] [Google Scholar]
- 29.Sood A, Mahajan R, Singh A, Midha V, Mehta V, Narang V, Singh T, Singh Pannu A. Role of faecal microbiota transplantation for maintenance of remission in patients with ulcerative colitis: A pilot study. J Crohns Colitis. 2019;13:1311–1317. doi: 10.1093/ecco-jcc/jjz060. [DOI] [PubMed] [Google Scholar]
- 30.Tian Y, Zhou Y, Huang S, Li J, Zhao K, Li X, Wen X, Li XA. Fecal microbiota transplantation for ulcerative colitis: A prospective clinical study. BMC Gastroenterol. 2019;19(116) doi: 10.1186/s12876-019-1010-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Zhang F, Zhang T, Zhu H, Borody TJ. Evolution of fecal microbiota transplantation in methodology and ethical issues. Curr Opin Pharmacol. 2019;49:11–16. doi: 10.1016/j.coph.2019.04.004. [DOI] [PubMed] [Google Scholar]
- 32.Eiseman B, Silen W, Bascom GS, Kauvar AJ. Fecal enema as an adjunct in the treatment of pseudomembranous enterocolitis. Surgery. 1958;44:854–859. [PubMed] [Google Scholar]
- 33.Schwan A, Sjölin S, Trottestam U, Aronsson B. Relapsing clostridium difficile enterocolitis cured by rectal infusion of homologous faeces. Lancet. 1983;2(845) doi: 10.1016/s0140-6736(83)90753-5. [DOI] [PubMed] [Google Scholar]
- 34.Cammarota G, Ianiro G, Tilg H, Rajilić-Stojanović M, Kump P, Satokari R, Sokol H, Arkkila P, Pintus C, Hart A, et al. European consensus conference on faecal microbiota transplantation in clinical practice. Gut. 2017;66:569–580. doi: 10.1136/gutjnl-2016-313017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Ott SJ, Waetzig GH, Rehman A, Moltzau-Anderson J, Bharti R, Grasis JA, Cassidy L, Tholey A, Fickenscher H, Seegert D, et al. Efficacy of sterile fecal filtrate transfer for treating patients with clostridium difficile infection. Gastroenterology. 2017;152(799.e7-811.e7) doi: 10.1053/j.gastro.2016.11.010. [DOI] [PubMed] [Google Scholar]
- 36.Ramai D, Zakhia K, Ofosu A, Ofori E, Reddy M. Fecal microbiota transplantation: Donor relation, fresh or frozen, delivery methods, cost-effectiveness. Ann Gastroenterol. 2019;32:30–38. doi: 10.20524/aog.2018.0328. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Hui W, Li T, Liu W, Zhou C, Gao F. Fecal microbiota transplantation for treatment of recurrent C. difficile infection: An updated randomized controlled trial meta-analysis. PLoS One. 2019;14(e0210016) doi: 10.1371/journal.pone.0210016. [DOI] [PMC free article] [PubMed] [Google Scholar] [Retracted]
- 38.Hamilton MJ, Weingarden AR, Sadowsky MJ, Khoruts A. Standardized frozen preparation for transplantation of fecal microbiota for recurrent Clostridium difficile infection. Am J Gastroenterol. 2012;107:761–767. doi: 10.1038/ajg.2011.482. [DOI] [PubMed] [Google Scholar]
- 39.Hamilton MJ, Weingarden AR, Unno T, Khoruts A, Sadowsky MJ. High-throughput DNA sequence analysis reveals stable engraftment of gut microbiota following transplantation of previously frozen fecal bacteria. Gut Microbes. 2013;4:125–135. doi: 10.4161/gmic.23571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Nishida A, Imaeda H, Ohno M, Inatomi O, Bamba S, Sugimoto M, Andoh A. Efficacy and safety of single fecal microbiota transplantation for Japanese patients with mild to moderately active ulcerative colitis. J Gastroenterol. 2017;52:476–482. doi: 10.1007/s00535-016-1271-4. [DOI] [PubMed] [Google Scholar]
- 41.Allegretti JR, Fischer M, Sagi SV, Bohm ME, Fadda HM, Ranmal SR, Budree S, Basit AW, Glettig DL, de la Serna EL, et al. Fecal microbiota transplantation capsules with targeted colonic versus gastric delivery in recurrent clostridium difficile infection: A comparative cohort analysis of high and lose dose. Dig Dis Sci. 2019;64:1672–1678. doi: 10.1007/s10620-018-5396-6. [DOI] [PubMed] [Google Scholar]
- 42.El-Salhy M, Hausken T, Hatlebakk JG. Increasing the dose and/or repeating faecal microbiota transplantation (FMT) increases the response in patients with irritable bowel syndrome (IBS) Nutrients. 2019;11 doi: 10.3390/nu11061415. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kelly CR, Kahn S, Kashyap P, Laine L, Rubin D, Atreja A, Moore T, Wu G. Update on fecal microbiota transplantation 2015: Indications, methodologies, mechanisms, and outlook. Gastroenterology. 2015;149:223–237. doi: 10.1053/j.gastro.2015.05.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Bafeta A, Yavchitz A, Riveros C, Batista R, Ravaud P. Methods and reporting studies assessing fecal microbiota transplantation: A systematic review. Ann Intern Med. 2017;167:34–39. doi: 10.7326/M16-2810. [DOI] [PubMed] [Google Scholar]
- 45.Li N, Tian H, Ma C, Ding C, Ge X, Gu L, Zhang X, Yang B, Hua Y, Zhu Y, Zhou Y. Efficacy analysis of fecal microbiota transplantation in the treatment of 406 cases with gastrointestinal disorders. Zhonghua Wei Chang Wai Ke Za Zhi. 2017;20:40–46. (In Chinese) [PubMed] [Google Scholar]
- 46.Li N, Tian HL, Chen QY, Yang B, Ma CL, Lin ZL, Zhang XY, Zhao D, Huang ZX, Jiang J, Qin HL. Efficacy analysis of fecal microbiota transplantation in the treatment of 2010 patients with intestinal disorders. Zhonghua Wei Chang Wai Ke Za Zhi. 2019;22:861–868. doi: 10.3760/cma.j.issn.1671-0274.2019.09.011. (In Chinese; Abstract available in Chinese from the publisher) [DOI] [PubMed] [Google Scholar]
- 47.Shen ZH, Zhu CX, Quan YS, Yang ZY, Wu S, Luo WW, Tan B, Wang XY. Relationship between intestinal microbiota and ulcerative colitis: Mechanisms and clinical application of probiotics and fecal microbiota transplantation. World J Gastroenterol. 2018;24:5–14. doi: 10.3748/wjg.v24.i1.5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Paramsothy S, Paramsothy R, Rubin DT, Kamm MA, Kaakoush NO, Mitchell HM, Castaño-Rodríguez N. Faecal microbiota transplantation for inflammatory bowel disease: A systematic review and meta-analysis. J Crohns Colitis. 2017;11:1180–1199. doi: 10.1093/ecco-jcc/jjx063. [DOI] [PubMed] [Google Scholar]
- 49.Lopez-Siles M, Duncan SH, Garcia-Gil LJ, Martinez-Medina M. Faecalibacterium prausnitzii: From microbiology to diagnostics and prognostics. ISME J. 2017;11:841–852. doi: 10.1038/ismej.2016.176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 50.Duncan SH, Hold GL, Harmsen HJ, Stewart CS, Flint HJ. Growth requirements and fermentation products of Fusobacterium prausnitzii, and a proposal to reclassify it as Faecalibacterium prausnitzii gen. nov., comb. nov. Int J Syst Evol Microbiol. 2002;52:2141–2146. doi: 10.1099/00207713-52-6-2141. [DOI] [PubMed] [Google Scholar]
- 51.Falony G, Joossens M, Vieira-Silva S, Wang J, Darzi Y, Faust K, Kurilshikov A, Bonder MJ, Valles-Colomer M, Vandeputte D, et al. Population-level analysis of gut microbiome variation. Science. 2016;352:560–564. doi: 10.1126/science.aad3503. [DOI] [PubMed] [Google Scholar]
- 52.Qin J, Li R, Raes J, Arumugam M, Burgdorf KS, Manichanh C, Nielsen T, Pons N, Levenez F, Yamada T, et al. A human gut microbial gene catalogue established by metagenomic sequencing. Nature. 2010;464:59–65. doi: 10.1038/nature08821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 53.Zhou Y, Xu ZZ, He Y, Yang Y, Liu L, Lin Q, Nie Y, Li M, Zhi F, Liu S, et al. Gut microbiota offers universal biomarkers across ethnicity in inflammatory bowel disease diagnosis and infliximab response prediction. mSystems. 2018;3 doi: 10.1128/mSystems.00188-17. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Hourigan SK, Ahn M, Gibson KM, Pérez-Losada M, Felix G, Weidner M, Leibowitz I, Niederhuber JE, Sears CL, Crandall KA, Oliva-Hemker M. Fecal transplant in children with clostridioides difficile gives sustained reduction in antimicrobial resistance and potential pathogen burden. Open Forum Infect Dis. 2019;6(ofz379) doi: 10.1093/ofid/ofz379. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Costello SP, Hughes PA, Waters O, Bryant RV, Vincent AD, Blatchford P, Katsikeros R, Makanyanga J, Campaniello MA, Mavrangelos C, et al. Effect of fecal microbiota transplantation on 8-week remission in patients with ulcerative colitis: A randomized clinical trial. JAMA. 2019;321:156–164. doi: 10.1001/jama.2018.20046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Wang JW, Wang YK, Zhang F, Su YC, Wang JY, Wu DC, Hsu WH. Initial experience of fecal microbiota transplantation in gastrointestinal disease: A case series. Kaohsiung J Med Sci. 2019;35:566–571. doi: 10.1002/kjm2.12094. [DOI] [PubMed] [Google Scholar]
- 57.Long C, Yu Y, Cui B, Jagessar SAR, Zhang J, Ji G, Huang G, Zhang F. A novel quick transendoscopic enteral tubing in mid-gut: Technique and training with video. BMC Gastroenterol. 2018;18(37) doi: 10.1186/s12876-018-0766-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Data Availability Statement
The datasets analyzed during the present study are not publicly available due to patient privacy concerns but are available from the corresponding author on reasonable request.