Abstract
Knowledge of coronaviruses in wild carnivores is limited. This report describes coronavirus genetic diversity, species specificity and infection prevalence in three wild African carnivores. Coronavirus RNA was recovered from fresh feces from spotted hyena and silver-backed jackal, but not bat-eared fox. Analysis of sequences of membrane (M) and spike (S) gene fragments revealed strains in the genus Alphacoronavirus, including three distinct strains in hyenas and one distinct strain in a jackal. Coronavirus RNA prevalence was higher in feces from younger (17 %) than older (3 %) hyenas, highlighting the importance of young animals for coronavirus transmission in wild carnivores.
Keywords: Gene Fragment, Wild Carnivore, Spotted Hyena, Fresh Feces, Coronavirus Infection
Knowledge of coronaviruses in wild carnivores is very limited. Novel coronaviruses have been reported in palm civet (Paguma larvata), raccoon dog (Nyctereutes procyonoides) [7], Asian leopard cat (Prionailurus bengalensis) and Chinese ferret badger (Melogale moschata) [3] in China, and in spotted hyena (Crocuta crocuta) in Tanzania [5]. Serological surveys provide evidence of coronavirus exposure in wild carnivores, suggesting that members of many terrestrial carnivore families serve as hosts for coronaviruses (Felidae, e.g. the European wild cat (Felis silvestris) [14], cheetah (Acinonyx jubatus) [19] and lion (Panthera leo) [10]; Hyaenidae, e.g., spotted hyena [5]; Canidae, e.g., wolf (Canis lupus) [22] and raccoon dog [7]; Mustelidae, e.g., Chinese ferret-badger [7]); Viverridae, e.g., civet cat [7]).
Coronaviruses belong to the family Coronaviridae in the order Nidovirales [8]. They are enveloped, single-stranded RNA viruses with the largest genomes of all RNA viruses [13, 21].
Our study reports the prevalence of infection and genetic diversity of coronaviruses belonging to the genus Alphacoronavirus (see http://talk.ictvonline.org/files/ictv_official_taxonomy_updates_since_the_8th_report/m/vertebrate-2008/default.aspx) in African wild carnivores (Figs. 1 and 2). This genus also includes feline coronavirus (FCoV), canine coronavirus (CCoV) and transmissible gastroenteritis virus (TGEV) [8]. We investigated fragments from two genes that produce structural proteins, the relatively conserved membrane (M) gene, which has been used for phylogenetic investigations [2, 8], and the highly variable spike (S) gene. The M protein is required for virus assembly and incorporation of the S protein into the viral envelope [4]. The S protein mediates virus attachment to host-cell receptors, fusion of the virus envelope to the host-cell membrane and cell-to-cell fusion [6, 12]. Because the S protein mediates virus-host cell receptor attachment, it is thought to influence host-species specificity [12], and as it induces production of virus-neutralizing antibodies, it is subject to positive selection [2, 6, 20].
Fig. 1.
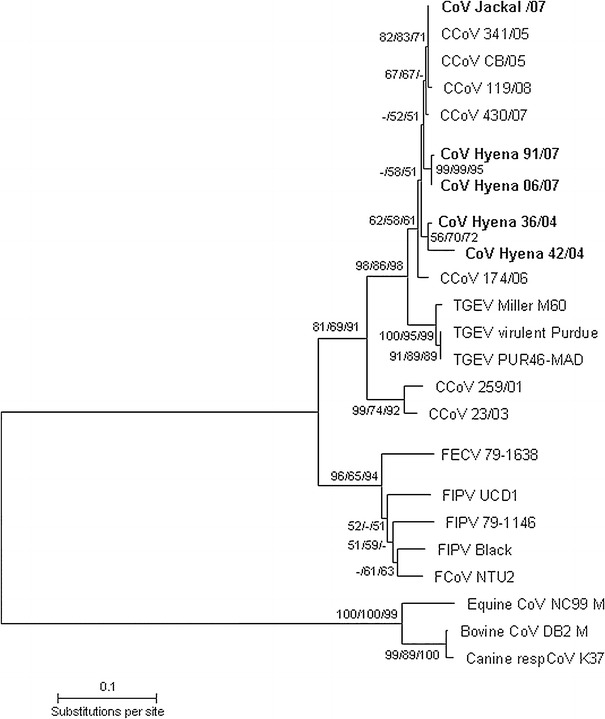
Phylogenetic relationship between coronavirus strains from spotted hyenas (Hyena), a silver-backed jackal (Jackal) and other published alphacoronavirus strains (canine, feline and porcine coronaviruses), plus betacoronavirus strains (bovine, canine and equine) based on a 369-nt fragment of the M gene. The unrooted neighbor joining (NJ) tree was constructed using the Tamura Nei model as implemented in Mega 5.0 [18]. Numbers at nodes represent bootstrap values for NJ, maximum-likelihood (ML) and maximum-parsimony (MP) trees in the order NJ/ML/MP. The scale bar indicates the estimated number of nt substitutions per site. Identical sequences from spotted hyenas in 2006 and 2007 were grouped as CoV Hyenas 06/07. Accession numbers of sequences generated in this study are as follows: CoV Hyena 36/04 (HQ339898), CoV Hyena 42/04 (HQ339900), CoV Hyena 91/07 (HQ339910), CoV Hyena 110/07 (HQ339911) and CoV Hyena Jackal/07 (HQ339912); ‘CoV Hyenas 06/07’: CoV Hyena 64/06 (HQ339901), CoV Hyena 33/07 (HQ339902), CoV Hyena 34/07 (HQ339903), CoV Hyena 35/07 (HQ339904), CoV Hyena 36/07 (HQ339905), CoV Hyena 37/07 (HQ339906), CoV Hyena 44/07 (HQ339907), CoV Hyena 62/07 (HQ339908), CoV Hyena 82/07 (HQ339909). The accession numbers of published coronavirus sequences used in this study are as follows: canine CoVs type I: CCoV 23/03 (AY548235), CCoV 259/01 (AF502583, only M gene sequence available); canine CoVs type II: CCoV 430/07 (EU924790), CCoV 341/05 (EU856361), CCoV CB/05 (DQ112226), CCoV 119/08 (EU924791), CCoV 174/06 (EU856362); feline CoVs type I: FIPV Black (AB086903), FCoV/NTU2/R/2003 (DQ160294), FIPV UCD1 (AB088222); feline CoVs type II: FIPV 79-1146 (DQ010921), FECV 79-1683 (Y13921, only M gene sequence available) and porcine CoVs: TGEV virulent Purdue (DQ811789), TGEV PUR46-MAD (AJ271965) and TGEV Miller M60 (DQ811786); betacoronavirus strains: canine respiratory CoV K37 (GQ918142), equine CoV NC99 (AY316300), bovine CoV DB2 (DQ811784)
Fig. 2.
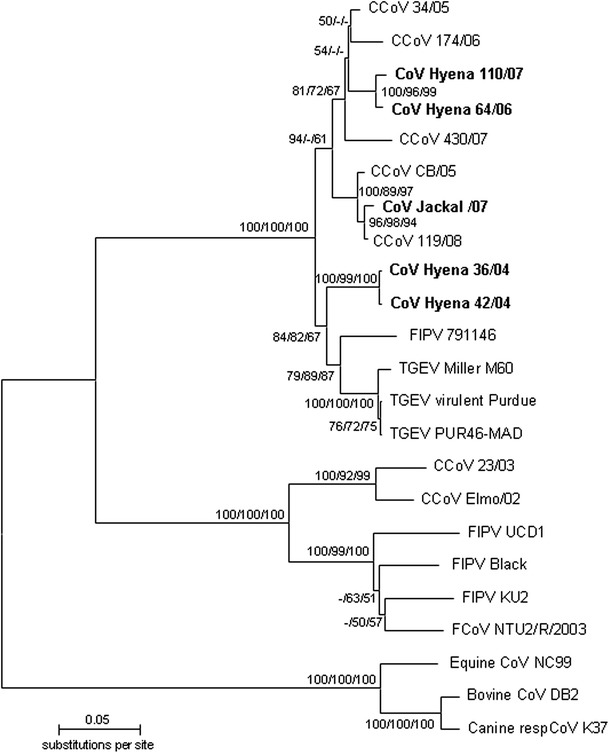
Phylogenetic relationship between coronavirus strains from spotted hyenas (Hyena), a silver-backed jackal (Jackal), other published alphacoronavirus strains, plus betacoronavirus strains (canine, bovine and equine), based on a 624-nt portion of the S gene. Identical sequences obtained from spotted hyena in 2007 were grouped as CoV Hyenas 07. The unrooted neighbor joining (NJ) tree was constructed using the Tamura Nei model as implemented in Mega 5.0 [18]. Numbers at nodes represent bootstrap values for (NJ), maximum-likelihood (ML) and maximum-parsimony (MP) trees in the order NJ/ML/MP. Scale bar indicates the estimated number of nt substitutions per site. Accession numbers of sequences generated in this study are as follows: CoV Hyena 36/04 (HQ339884), CoV Hyena 42/04 (HQ339885), CoV Hyena 64/06 (HQ339886) and CoV Jackal/07 (HQ339897); ‘CoV Hyenas 07’: CoV Hyena 33/07 (HQ339887), CoV Hyena 34/07 (HQ339888), CoV Hyena 35/07 (HQ339889), CoV Hyena 36/07 (HQ339890), CoV Hyena 37/07 (HQ339891), CoV Hyena 44/07 (HQ339892), CoV Hyena 62/07 (HQ339893), CoV Hyena 82/07 (HQ339894), CoV Hyena 91/07 (HQ339895), CoV Hyena 110/07 (HQ339896). The accession numbers of published sequences from alphacoronaviruses used in this study are as follows: canine CoVs type I: CCoV 23/03 (AY548235), CCoV Elmo/02 (AY307020, only S gene sequence available); canine CoVs type II: CCoV 430/07 (EU924790), CCoV 341/05 (EU856361), CCoV CB/05 (DQ112226), CCoV 119/08 (EU924791), CCoV 174/06 (EU856362); feline CoVs type I: FIPV Black (AB086903), FCoV/NTU2/R/2003 (DQ160294), FIPV UCD1 (AB088222), FIPV KU-2 (D32044); feline CoV type II: FIPV 79-1146 (DQ010921), and porcine coronaviruses: TGEV virulent Purdue (DQ811789), TGEV PUR46-MAD (AJ271965) and TGEV Miller M60 (DQ811786); betacoronavirus strains: canine respiratory CoV K37 (GQ918142), equine CoV NC99 (AY316300), bovine CoV DB2 (DQ811784)
Our study was conducted between 2003 and 2008 in an area of approximately 250 km2 in the center (2°27′S, 34°49′E) of the extensive (14,763 km2) Serengeti National Park (SNP) in northern Tanzania, where domestic hosts of coronaviruses were prohibited. Fresh feces were collected immediately after deposition from 165 individually known spotted hyena (hereafter termed hyena) in three closely monitored social groups termed clans [5] and from silver-backed jackals Canis mesomelas (17 feces) (hereafter termed jackals) and bat-eared foxes Otocyon megalotis (9 feces).
Samples were stored and transported frozen at −80 °C, or preserved in RNAlater (Sigma-Aldrich Inc., St. Louis, MO, USA), initially stored at −10 °C, transported frozen, and then stored at −80 °C. Samples were homogenized as a 10 % solution in DEPC-treated water then centrifuged for 5 minutes at 13,000 rpm. Viral RNA was extracted using a MinElute Virus Spin Kit (QIAGEN, Hilden, Germany) following the manufacturer’s instructions, using 200 μl of the supernatant of the homogenate. We used published primer pairs designed to detect alphacoronavirus strains, including CCV1 (5′-TCCAGATATGTAATGTTCGG-3′) and CCV2 (5′- TCTGTTGAGTAATCACCAGCT – 3′) [15, 16] to amplify a 409-nucleotide (nt)-long M gene fragment and primers S5 (5′-TGCATTTGTGTCTCAGACTT-3′) and S6 (5′-CCAAGGCCATTTTACATAAG-3′) [17] to amplify a 694-nt-long S gene fragment (3′ region). Reverse transcription PCR (RT-PCR) was performed in duplicate using a SuperScriptTM III One-Step RT-PCR System (Invitrogen, Karlsruhe, Germany) following the manufacturer’s instructions, in a total reaction volume of 25 μl. The FCoV-positive control strains used were KU2Japan, FCoVNW-1, FCoV DF-2, and FCoV WSU 79-1146. The CCoV-positive control strain was CCoV 1-71.
PCR products were analyzed by gel electrophoresis and visualized by GelRed (Biotium Inc, Hayward, California) staining. RT-PCR products of the expected length were purified using a QIAquick PCR Purification Kit (QIAGEN, Hilden, Germany). Sequencing was bidirectional and conducted using an ABI PRISM BigDye Terminator Cycle Sequencing Kit 1.1 (Applied Biosystems [ABI], Darmstadt, Germany) following the manufacturer’s instructions. Sequences were visualized on an ABI3130xl Genetic Analyzer (ABI). Editing and translation of sequences and determination of nt and amino acid (aa) similarities were carried out with BioEdit v.7.0.9.0 [9]. We sequenced 14 M gene fragments, 13 from hyena (10 identical sequences are collectively termed CoV Hyenas 06/07, Fig. 1) and one from a jackal, plus 14 S gene fragments, 13 from hyena (10 identical sequences are collectively termed CoV Hyenas 07, Fig. 2) and one from a jackal. All sequences for M and S gene fragments generated in this study were submitted to GenBank.
We aligned our sequences with other published sequences from alphacoronavirus (FCoV, CCoV, TGEV) and three betacoronavirus sequences, using Mega 5.05 [18]. Gaps in the nucleotide alignments were adjusted manually according to the gaps in the corresponding amino acid sequence alignments. Subsequent construction of trees was carried out using the neighbor-joining (NJ), maximum likelihood (ML) and maximum parsimony (MP) approaches for both the S gene and M gene sequences. NJ and ML were based on the Jukes-Cantor model as determined by and implemented in Mega 5.05 [18]. Statistical support was obtained by bootstrapping (NJ1000/ ML100/ MP1000), and percentages are given at the nodes (NJ/ ML/ MP). The alignment for the M gene was based on 369 positions, and that for the S gene, on 624 positions.
Analysis of the M gene fragment revealed that the two ‘hyena’ strains in 2004 (CoV Hyena 42/04 and CoV Hyena 36/04) were positioned separately from ‘hyena’ strains in 2006 and 2007 (Fig. 1). The M gene fragment from the ‘jackal’ strain in 2007 was distinct from the ‘hyena’ strain found in that year. The ‘jackal’ strain clustered with CCoV-II strains from domestic dogs (Fig. 1).
Comparison of deduced aa sequences from coronavirus M gene fragments revealed a high similarity between strains recovered from hyenas in 2006 and 2007 (99.1 %). These strains had a lower similarity to the two ‘hyena’ strains recovered in 2004 (98.3 % and 97.5 % respectively). The M protein fragment from the jackal in 2007 differed from fragments obtained from hyenas in that year (98.3 and 99.1 %) but was identical to published CCoV strains. Coronavirus M protein fragments from hyenas and the jackal were more similar to published CCoV and TGEV strains (between 95.9 % and 98.3 %) than to FCoV strains (between 79.3 % and 85.2 %).
Analysis of the S gene fragment also revealed that both ‘hyena’ strains in 2004 clustered together and were separated from the distinct cluster of strains from hyena in 2006 and 2007 (Fig. 2). In relation to published coronavirus strains from domestic animals, the S gene fragments from ‘hyena’ strains in 2004 were placed closer to FCoV type II and TGEV strains than CCoV strains, whereas ‘hyena’ strains in 2006 and 2007 were placed closer to CCoV-II strains than FCoV type II and TGEV strains. The strain from a jackal in 2007 clustered with CCoV-II strains (Fig. 2).
Comparison of deduced aa sequences of S gene fragments revealed the highest similarity between the two distinct ‘hyena’ strains recovered in 2004 (99.0 %) and the lowest similarity between the ‘hyena’ strains CoV Hyena 36/04 and CoV hyena 07 (96.6 %). The similarity between the S protein fragments obtained from ‘hyena’ strains and the ‘jackal’ strain was higher in 2004 (97.0 %) and 2006 (97.5 %) than in 2007 (96.6 %).
Our results revealed five genetically distinct coronavirus strains, including two distinct strains in the hyena population within one year (2004) and distinct strains in hyenas between years 2004 and 2007. The ‘jackal’ strain was distinct from all ‘hyena’ strains, suggesting a degree of host-species specificity in strains that infected hyenas and jackals. Species specificity was also suggested by a previous study [5] that revealed a novel ‘hyena’ coronavirus strain with 20 unique substitution sites in a highly variable S gene fragment (5′ region). The diversity and between-year difference in ‘hyena’ strains suggests rapid coronavirus evolution [11] and/or repeated transmission of strains from other host species.
Using life histories of individually known hyenas [5], we compared the prevalence of coronaviral RNA in feces from juveniles (<12 months of age) and older hyenas (>12 months of age) using a log likelihood ratio test performed in SYSTAT 12 (Systat Software Inc., Richmond, USA). We detected coronaviral RNA significantly more often in feces from juveniles (16 positive of 94 sampled juveniles, 17 %) than older hyenas (2 of 71 sampled individuals, 3 %; G = 9.7, d.f. = 1, n = 165, p < 0.01), which demonstrated the importance of juvenile wild carnivores in the epidemiology of coronaviruses. Only one of 17 (6 %) feces from adult jackals was positive, and all nine feces from adult bat-eared foxes were negative. The lack of feces from juvenile canids in our analysis probably explains why infection was rarely detected in these canids. Extensive screening of feces from young wild carnivores will most likely reveal a considerable diversity of coronavirus strains in the carnivore community in the SNP and elsewhere.
Virus transmission requires susceptible hosts. The proportion of these hosts in a population changes with the level of herd immunity and can differ between host age categories [1]. The adult hyena population in the SNP has a high (between 68 % and 74 %) seroprevalence of antibodies against coronavirus, whereas juvenile hyenas often have no protective titers or titers well below those in adults [5]. This suggests that an important component of coronavirus epidemiology in hyena populations is spatially separated groups of susceptible juveniles at communal dens inside clan territories [5]. This clumped distribution of juveniles may result in the ‘burn-out’ of some ‘hyena’ strains when virus transmission between susceptible individuals in different territories is prevented by high herd immunity in adults.
Our study demonstrates that RT-PCR screening of fresh feces, particularly from young wild carnivores, can be used to track coronavirus infection. This non-invasive method is appropriate for monitoring coronavirus infection in rare and endangered species and those in protected areas where invasive monitoring methods are prohibited. Wild carnivore species in protected areas may be infected with a diversity of novel coronavirus strains that are distinct from those described in domestic species, and a degree of host-species specificity among coronavirus strains in wild carnivores can be expected.
Acknowledgments
We thank COSTECH, TAWIRI and TANAPA for permission to conduct the study. We thank V. Benetka, N. Burgener, A. Francis, G. Orio, T. Shabani, D. Thierer and K. Wilhelm for assistance, V. Benetka, O. P. Höner and K. Möstl and for fruitful discussions, and two reviewers for helpful comments. The study was conducted within the GRAKO 1121 “Genetic and Immunologic Determinants of Pathogen-Host-Interactions” at the Center for Infection Biology and Immunity (ZIBI) of the Humboldt University Berlin and was financed by the DFG and IZW, Berlin.
References
- 1.Anderson RM, May RM. Infectious diseases of humans: dynamics and control. Oxford: Oxford University Press; 1991. [Google Scholar]
- 2.Decaro N, Buonavoglia C. An update on canine coronaviruses: viral evolution and pathobiology. Vet Microbiol. 2008;132:221–234. doi: 10.1016/j.vetmic.2008.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Dong BQ, Liu W, Fan XH, Vijaykrishna D, Tang XC, Gao F, Li LF, Li GJ, Zhang JX, Peiris JSM, Smith GJD, Chen H, Guan Y. Detection of a novel and highly divergent coronavirus from Asian leopard cats and Chinese ferret badgers in southern China. J Virol. 2007;81:6920–6926. doi: 10.1128/JVI.00299-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.de Haan CAM, Smeet M, Vernooij F, Vennema H, Rottier PJM. Mapping the coronavirus membrane protein domains involved in interaction with the spike protein. J Virol. 1999;73:7441–7452. doi: 10.1128/jvi.73.9.7441-7452.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.East ML, Moestl K, Benetka V, Pitra C, Höner OP, Wachter B, Hofer H. Coronavirus infection of spotted hyenas in the Serengeti ecosystem. Vet Microbiol. 2004;102:1–9. doi: 10.1016/j.vetmic.2004.04.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gallagher TM, Buchmeiert MJ. Coronavirus spike proteins in viral entry and pathogenesis. Virology. 2001;279:371–374. doi: 10.1006/viro.2000.0757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Guan YBJ, Zheng YQH, Liu XL, Zhyang ZX, Cheung CL, Luo SW, Li PH, Zhang LJ, Guan YJ, Butt KM, Wong KL, Chan KW, Lim W, Shortridge KF, Yuen KY, Peiris JSM, Poon LLM. Isolation and characterization of viruses related to the SARS coronavirus for animals in southern China. Science. 2003;302:276–278. doi: 10.1126/science.1087139. [DOI] [PubMed] [Google Scholar]
- 8.González JM, Gomez-Puertas P, Cavanagh D, Gorbalenya AE, Enjuanes L. A comparative sequence analysis to revise the current taxonomy of the family Coronaviridae. Arch Virol. 2003;148:2207–2235. doi: 10.1007/s00705-003-0162-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Hall TA. BioEdit: a user-friendly biological sequence alignment editor and analysis program for Windows 95/98/NT. Nucleic Acids Symp Ser. 1999;41:95–98. [Google Scholar]
- 10.Hofmann-Lehmann R, Fehr D, Grob M, Elgizoli M, Packer C, Martenson JS, O’Brien SJ, Lutz H. Prevalence of antibodies to feline parvovirus, calicivirus, herpervirus, coronavirus, and immunodeficiency virus and of feline leukemia virus antigen and the interrelationship of these viral infections in free-ranging lions in East Africa. Clin Diagn Lab Immunol. 1996;3:554–562. doi: 10.1128/cdli.3.5.554-562.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Jenkins GM, Rambaut A, Pybus OG, Holmes EC. Rates of molecular evolution in RNA viruses: a quantitative phylogenetic analysis. J Mol Ecol. 2002;54:156–165. doi: 10.1007/s00239-001-0064-3. [DOI] [PubMed] [Google Scholar]
- 12.Kuo L, Godeke GJ, Raamsman MJB, Masters PS, Rottier PJM. Retargeting of cooronavirus by substitution of the spike glycoprotein ectodomain: Crossing the host cell species barrier. J Virol. 2000;74:1393–1406. doi: 10.1128/JVI.74.3.1393-1406.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Lai MMC, Holmes KV. Coronaviridae: the viruses and their replication. In: Knipe DM, Howley PM, editors. Fields virology. 4. Philadelphia: Williams and Wilkins; 2001. pp. 1163–1185. [Google Scholar]
- 14.Leutenegger CM, Hofmann-Lehmann R, Riols C, Liberek M, Worel G, Lups P, Fehr D, Hartmann M, Weilenmann P, Lutz H. Viral infections in free-living populations of the European wildcat. J Wildl Dis. 1999;35:678–686. doi: 10.7589/0090-3558-35.4.678. [DOI] [PubMed] [Google Scholar]
- 15.Pratelli A, Tempesta M, Greco G, Martella V, Buonavoglia C. Development of a nested PCR assay for the detection of canine coronavirus. J Virol Methods. 1999;80:11–55. doi: 10.1016/S0166-0934(99)00017-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Pratelli A, Tinelli A, Decaro N, Camero M, Elia G, Gentile A, Buonavoglia C. PCR assay for the detection and the identification of atypical canine coronavirus in dogs. J Virol Methods. 2002;106:209–213. doi: 10.1016/S0166-0934(02)00165-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Pratelli A, Decaro N, Tinelli A, Martella V, Elia G, Tempesta M, Cirone F, Buonavoglia C. Two genotypes of canine coronavirus simultaneously detected in the fecal samples of dogs with diarrhea. J Clin Microbiol. 2004;42:1797–1799. doi: 10.1128/JCM.42.4.1797-1799.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S. MEGA5: Molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Mol Biol Evol. 2011;28:2731–2739. doi: 10.1093/molbev/msr121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Thalwitzer S, Wachter B, Robert N, Wibbelt G, Müller M, Lonzer J, Meli ML, Bay G, Hofer H, Lutz H. Seroprevalences to viral pathogens in free-ranging and captive cheetahs (Acinonyx jubatus) on Namibian farmland. Clin Vac Immunol. 2010;17:232–238. doi: 10.1128/CVI.00345-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Wege H, Siddell S, Ter Meulen V. The biology and pathogenesis of coronaviruses. Curr Top Microbiol Immun. 1982;99:165–200. doi: 10.1007/978-3-642-68528-6_5. [DOI] [PubMed] [Google Scholar]
- 21.Weiss SR, Navas-Martin S. Coronavirus pathogenesis and the emerging pathogen severe acute respiratory syndrome coronavirus. Microbiol Mol Biol Rev. 2005;69:635–664. doi: 10.1128/MMBR.69.4.635-664.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Zarnke RL, Evermann J, Ver Hoef JM, McNay ME, Boertje RD, Gardner CL, Adams LG, Dale BW, Burch J. Serological survey for canine coronavirus in wolves from Alaska. J Wildl Dis. 2001;37:740–745. doi: 10.7589/0090-3558-37.4.740. [DOI] [PubMed] [Google Scholar]


