Abstract
Canine parvovirus (CPV) and canine coronavirus (CCoV) are considered the main pathogens responsible for acute gastroenteritis in dogs. From a collection of 250 samples, seven CPV strains and three CCoV strains were identified in symptomatic Irish dogs. Samples were screened for the viruses using polymerase chain reaction (PCR) and typed via DNA sequence analysis. Three CPV strains were characterized as CPV-2a, while four others were characterized as CPV-2b. To date, CPV-2c remains unreported in Ireland. Two CCoV strains were characterized as CCoV-II and one as CCoV-I. In the case of one sample, PH4/09/Ire, a mixed infection with CPV and CCoV was detected.
Keywords: Severe Acute Respiratory Syndrome, Canine Distemper Virus, Severe Acute Respiratory Syndrome, Polymerase Chain Reaction Amplicon, Feline Panleukopenia Virus
Canine parvovirus type 2 (CPV) and canine coronavirus (CCoV) are considered the main pathogens responsible for acute gastroenteritis in dogs [1]. CPV was first identified in the late 1970s as a major cause of haemorrhagic gastroenteritis and myocarditis in puppies [2]. CPV has a small single-stranded DNA genome of 5.2 kilobases (kb). Symptoms of CPV infection vary from mild to haemorrhagic enteritis , fever, vomiting and death in severe cases. Coronaviruses are single-stranded, positive-sense RNA viruses with a large genome of 27-31 kb in length. Enteric canine coronavirus typically causes mild enteritis in dogs, with more severe clinical signs observed in young animals [3].
The single-stranded genome of CPV makes the virus more susceptible to modifications, and error-prone replication can lead to significant DNA sequence variation and greater potential for evolution of these viruses, when measured over defined periods, or during growth of CPV through serial passage in cell culture [4]. It is presumed that CPV originated from feline panleukopenia virus via genetic mutations and evolution [2]. Shortly after its emergence, CPV-2 was entirely replaced by a new type, designated CPV-2a, which possessed the ability to infect both cats and dogs [5]. In the mid-eighties, another type, designated as CPV-2b, began to emerge. CPV-2b is distinguished from CPV-2a by a single amino acid (aa) substitution, asparagine (Asn) to aspartic acid (Asp), at position 426 of the major antigenic site of the VP2 capsid protein [6].
In 2000, a novel CPV type, called CPV-2c, was detected in Italy, and this is now progressively replacing other CPV types [7]. CPV-2c has since been detected throughout parts of Europe, including Spain [8], Germany [9], Portugal [10], the United Kingdom [11], as well as Korea [12], the United States [13], South America [14] and Vietnam [15]. CPV-2c is distinguishable from CPV-2a or 2b by substitution of glutamic acid (Glu) in place of Asn or Asp, respectively, at the 426th aa residue of the VP2 protein. Therefore, due to the positioning of these aa substitutions in an antigenic site, it is possible to differentiate between types by employing monoclonal antibodies [15]. Alternatively, the nucleotide sequence required to substitute Glu at aa position 426 created a novel MboII restriction site (GAAGA), which is unique to CPV-2c, and therefore, CPV-2c can be distinguished from CPV-2a/2b by restriction enzyme analysis [7]. Infection with CPV-2c has been reported to result in clinical symptoms similar to those exhibited by dogs infected with CPV-2a and 2b [13]. Some studies reported milder symptoms in infected animals [16], while other reports found evidence of more severe clinical symptoms and higher rates of mortality, even in vaccinated dogs [7, 8, 17].
CCoV was first discovered as an enteropathogen in 1971 [18]. Enteric CCoV exists in two closely related forms, the original form, which was isolated in the 1970s, is named CCoV-II. A new CCoV strain, which was identified in Italy in 2003, is designated as CCoV-I. CCoV-I and II share a high nucleotide homology in the viral genome but are highly divergent in the spike protein gene [19]. The strains are so named according to genetic similarities observed between CCoV-I and II, and feline coronavirus (FCoV)-I and II, respectively [20]. As coronaviruses contain large RNA genomes, they are highly susceptible to frequent mutation as a result of the high error rate of RNA polymerase, which results in the accumulation of several base substitutions per round of replication [21]. Coronaviruses can accumulate small insertions and deletions in their genomes, which promotes their evolution [22]. They also undergo a high rate of homologous RNA recombination, mediated by a “copy-choice” mechanism based on sequence homology surrounding the recombination sites [23]. As a result of all of these mechanisms, coronaviruses can mutate rapidly, leading to new genotypes (CCoV-I), biotypes (pantropic CCoV) and host variants (canine respiratory coronavirus). Following the emergence of severe acute respiratory syndrome (SARS) in humans, the continual epidemiological surveillance on CoVs has been reinforced, as they are considered to be potential agents of direct and indirect zoonoses [24].
Infection can occur with a single CCoV strain, or both strains may be present simultaneously [25]. CCoV infection generally has a low mortality; however, a recently characterized strain detected in Italy (CB/05) resulted in a fatal disease as a consequence of systemic spread of the virus [24]. Deaths have also been caused by the synergistic effect produced when a mixed infection occurs with other canine enteric viruses. Mixed infections have been reported between CCoV and CPV-2, canine distemper virus and canine adenovirus type I [8, 26, 27]. Mixed infections have also been reported between CPV-2 and calicivirus [28, 29]. Dual infection results in illnesses that are usually more severe than either virus can produce alone [28]. Synergistic mechanisms are common with other gastroenteritis viruses such as rotavirus. Studies have demonstrated this effect with calves co-infected with group A rotavirus and group C rotavirus, or with group A rotavirus and Escherichia coli [30, 31].
The original CPV-2 strain is still employed in many commercial vaccines. However, this strain was entirely replaced by its variant types (CCoV-I and II) shortly after its emergence. There is concern that these current vaccines may fail to protect pups against the CPV-2 variants [32, 33]. A plan to use current strains in vaccine formulations has led to a CPV-2b-based vaccine being licensed in Europe [34]. Currently, all canine vaccines used to protect against CCoVs are based on CCoV-II. However, it has been shown that the level of cross-reactivity between CCoV-I and II is limited [35]. Recombinant CCoVs have been reported that were derived from CCoV-II and transmissible gastroenteritis virus of swine (TGEV), related in the N terminal domain of the S protein. Antigenic differences have also been found between TGEV-like CCoVs and reference CCoV strains [17]. As a result of this viral evolution, the efficacy of prophylaxis programs in place to protect dogs against CCoV challenges may need to be assessed.
To date, there is no epidemiological information on the circulation of coronaviruses and parvoviruses in Ireland. This study was undertaken to investigate the prevalence of these viruses in a wide spectrum of Irish dogs, both with and without clinical symptoms, and to assess the efficacy of vaccination programs currently in place to protect against these viruses.
A total of 250 faecal samples were collected from both symptomatic and asymptomatic dogs in 2008 and 2009 (Table 1a). The samples were collected from dogs of all ages (2 months to 15 years) from a wide spectrum of groups, including the Irish Society for Prevention of Cruelty to Animals (ISPCA), the Guide Dogs Association, racing greyhounds, hunting hounds and domestic dogs presenting in veterinary practices, which represent the major groups of canines in Ireland.
Table 1a.
Samples analysed in 2008 and 2009
| Sample source | Number of samples | Number of CCoV positives | Number of CPV positives |
|---|---|---|---|
| Veterinary Clinics | 131 | 3 | 6 |
| Guide Dogs Association | 89 | - | - |
| Hunting Hounds | 13 | - | - |
| ISPCA | 11 | - | 1 |
| Racing Greyhounds | 6 | - | - |
| Total | 250 | 3 | 7 |
Total nucleic acids were extracted from the samples by a standard phenol-chloroform method with ethanol precipitation. The extracted nucleic acids were resuspended in 100 μl of sterile DEPC-H2O and stored at -80°C prior to use.
Samples were screened for parvovirus by PCR amplification of a 583-bp segment at the carboxy terminus of CPV open reading frame 2, using primers and reaction conditions described by Buonavoglia et al. [7]. The reaction was carried out on an MJ Researcher PTC-200 Thermocycler (GMI Inc, Minnesota, USA). Reaction conditions were as follows: 94°C for 10 min, followed by 35 cycles of 94°C for 30s, 50°C for 1 min and 72°C for 1 min, with a final extension at 72°C for 10 min. Positive and negative controls were included in the reaction.
Samples were subjected to screening for the presence of canine coronavirus (CCoV) by RT-PCR amplification of the polymerase gene, using primers described by Stephenson et al. [36] and an Enhanced Avian Reverse Transcriptase Kit (Sigma-Aldrich). Reaction conditions were as follows: 45°C for 50 min, and 94°C for 2 min, followed by 35 cycles of 94°C for 1 min, 48°C for 1 min and 68°C for 1 min, with a final extension at 68°C for 10 min. All samples that tested positive for the presence of the CoV conserved polymerase gene were subjected to RT-PCR amplification of an N gene segment in order to specifically detect canine coronavirus. A segment of the S gene was amplified for application in sequence and phylogenetic analysis, using primers and reaction conditions as described by Erles and Brownlie [37].
PCR products were run on a 1.5% agarose gel following ethidium bromide staining and visualized using UV light transillumination.
All samples that tested positive for the VP2 gene segment of CPV-2 via PCR amplification were subjected to RFLP analysis of this PCR product. For restriction enzyme digests, 10 μl of each PCR amplicon was digested with five units of the restriction enzyme MboII (Fermentas, Germany) and visualized on a 1.5% agarose gel to determine the cleavage pattern of the nucleic acid.
PCR amplicons were purified using a QIAquick PCR purification kit (Qiagen Ltd, West Sussex, England) and sequenced using a commercial service (MWG-biotech, Ebersberg, Germany). Nucleotide sequences were submitted to the GenBank database, and their accession numbers are displayed in Tables 1b and 1c. Nucleotide and amino acid sequence alignment was performed using the ClustalW application with Bioedit Sequence Alignment Editor [38].
Table 1b.
CPV strain information
| Isolate | Accession no. | Type | Age | Symptoms | Breed | Vaccinated |
|---|---|---|---|---|---|---|
| CG4/09/Ire | GQ387486 | 2b | 13m | HD, V, D | Sheepdog | Yes |
| PH4/09/Ire | GQ387487 | 2b | 3m | HD | Jack Russell | No |
| PH1/09/Ire | GQ387488 | 2b | 3m | HD, V, P | Collie Cross | No |
| PH2/09/Ire | GQ387489 | 2a | 15m | HD, V | Spaniel | Yes |
| BC2/09/Ire | GQ414751 | 2a | 3m | HD | Mixed | Yes |
| BC4/09/Ire | GU014701 | 2a | 2m | HD | Collie Cross | No |
| BC5/09/Ire | GU475122 | 2b | 3m | HD | Mixed | Yes |
Table 1c.
CCoV strain information
| Isolate | Type | Age | Symptoms | Breed | Vaccinated |
|---|---|---|---|---|---|
| PH4/09/Ire | 1 | 3m | HD | Jack Russell | No |
| AVC2/09/Ire | 2 | 8y | d | Collie | Unknown |
| AVC3/09/Ire | 2 | 4m | d | Mixed | Unknown |
HD: haemorrhagic diarrhoea
d: diarrhoea
V: vomiting
P: pyrexia
D: death
Preliminary analysis was accomplished by comparison with sequences available in the database using the web-based program BLAST (http://www.ncbi.nlm.nih.gov/BLAST). Phylogenetic analysis was conducted using the MEGA 4 program [39]. Two phylogenetic trees, based on the partial CPV VP2 (Figure 1) and CCoV S (Figure 2) gene segments, were constructed by the maximum-parsimony (100 replicates) and maximum -ikelihood (1000 replicates) method, respectively, supplying a statistical support with bootstrapping, using CPV-2 and CCoV reference strains obtained from the GenBank database, as displayed in Tables 2 and 3, respectively.
Fig. 1.
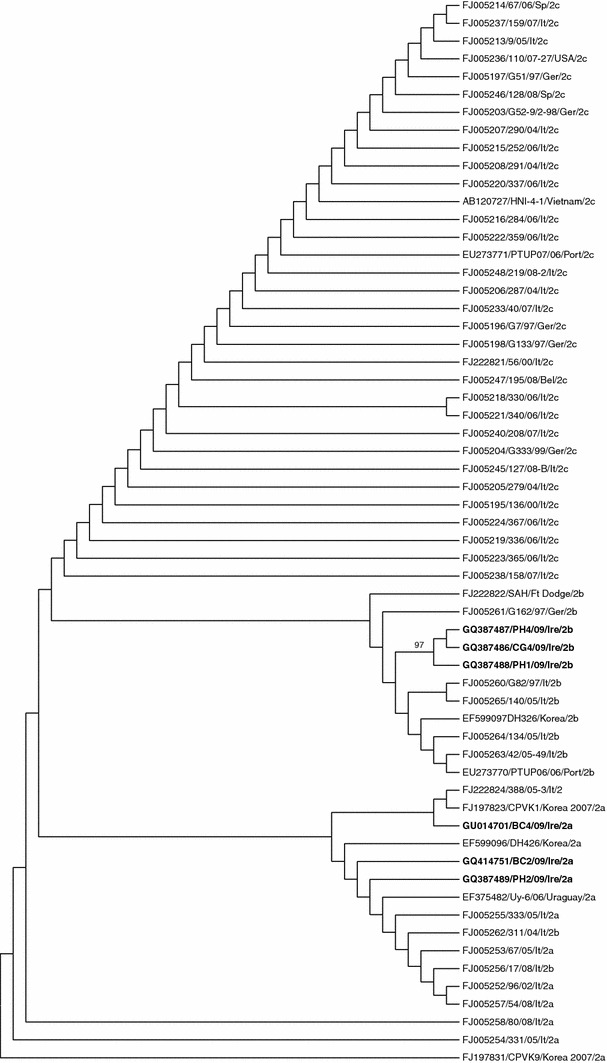
Maximum-parsimony tree based on partial VP2 gene nucleotide sequences of CPVs described in this study and reference strains
Fig. 2.
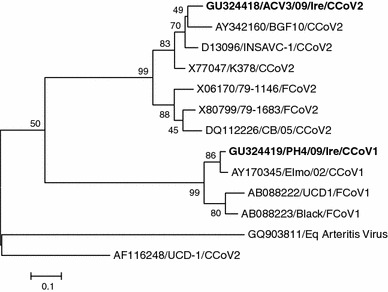
Phylogenetic tree based on partial S gene nucleotide sequences of CCoVs described in this study and reference strains. Evolutionary history was assessed using the maximum-likelihood method based on the Tamura-Nei model [48]. Sequence alignments were performed using MUSCLE [49]. The tree with the highest log likelihood (-3027.2510) is shown. The percentage of replicate trees in which the associated taxa clustered together in the bootstrap test (1,000 replicates) is shown next to the branches. The tree is drawn to scale, with branch lengths measured in the number of substitutions per site. The analysis involved 13 nucleotide sequences. All ambiguous positions were removed for each sequence pair
Table 2.
VP2 reference strains
| CPV type | Name | Origin | Year | GenBank accession no. |
|---|---|---|---|---|
| 2 | 338/05-3 | Italy | 2005 | FJ222824 |
| 2a | 96/02 | Italy | 2002 | FJ005252 |
| 2a | 67/05 | Italy | 2005 | FJ005253 |
| 2a | 331/05 | Italy | 2005 | FJ005254 |
| 2a | 333/05 | Italy | 2005 | FJ005255 |
| 2a | 17/08 | Italy | 2008 | FJ005256 |
| 2a | 54/08-3 | Italy | 2008 | FJ005257 |
| 2a | 80/08 | Italy | 2008 | FJ005258 |
| 2a | DH426 | Korea | 2007 | EF599096 |
| 2b | DH326 | Korea | 2007 | EF599097 |
| 2a | CPVK1 | Korea | 2007 | FJ197823 |
| 2a | CPVK9 | Korea | 2007 | FJ197831 |
| 2b | G82/97 | Germany | 1997 | FJ005260 |
| 2b | G162/97 | Germany | 1997 | FJ005261 |
| 2b | 311/04 | Italy | 2004 | FJ005262 |
| 2b | 42/05-49 | Italy | 2005 | FJ005263 |
| 2b | 134/05 | Italy | 2005 | FJ005264 |
| 2b | 140/05 | Italy | 2005 | FJ005265 |
| 2b | SAH | Fort Dodge | NA | FJ222822 |
| 2c | 56/00 | Italy | 2000 | FJ222821 |
| 2c | 136/00 | Italy | 2000 | FJ005195 |
| 2c | G7/97 | Germany | 1997 | FJ005196 |
| 2c | G51/97 | Germany | 1997 | FJ005197 |
| 2c | G133/97 | Germany | 1997 | FJ005198 |
| 2c | G52-9/2-98 | Germany | 1998 | FJ005203 |
| 2c | G333/99 | Germany | 1999 | FJ005204 |
| 2c | 279/04 | Italy | 2004 | FJ005205 |
| 2c | 287/04 | Italy | 2004 | FJ005206 |
| 2c | 290/04 | Italy | 2004 | FJ005207 |
| 2c | 291/04 | Italy | 2004 | FJ005208 |
| 2c | 9/05 | Italy | 2005 | FJ005213 |
| 2c | 67/06 | Spain | 2006 | FJ005214 |
| 2c | 252/06 | Italy | 2006 | FJ005215 |
| 2c | 284/06 | Italy | 2006 | FJ005216 |
| 2c | 330/06 | Italy | 2006 | FJ005218 |
| 2c | 336/06 | Italy | 2006 | FJ005219 |
| 2c | 337/06 | Italy | 2006 | FJ005220 |
| 2c | 340/06-A | Italy | 2006 | FJ005221 |
| 2c | 365/06 | Italy | 2007 | FJ005223 |
| 2c | 40/07 | Italy | 2007 | FJ005233 |
| 2c | 110/07-27 | USA | 2007 | FJ005236 |
| 2c | 158/07 | Italy | 2007 | FJ005237 |
| 2c | 159/07 | Italy | 2007 | FJ005238 |
| 2c | 208/07 | Italy | 2007 | FJ005240 |
| 2c | 127/08-B | Italy | 2008 | FJ005245 |
| 2c | 128/08 | Spain | 2008 | FJ005246 |
| 2c | 195/08-81 | Belgium | 2008 | FJ005247 |
| 2c | 219/08-2 | Italy | 2008 | FJ005248 |
| 2c | HNI-4-1 | Vietnam | 2004 | AB120727 |
Table 3.
CoV reference strains
| Strain | Name | Origin | GenBank accession no. |
|---|---|---|---|
| CCoV-II | UCD-1 | USA | AF116248 |
| CCoV-II | CB/05 | Italy | DQ112226 |
| CCoV-II | K378 | Holland | X77047 |
| CCoV-II | BGF10 | United Kingdom | AY342160 |
| CCoV-II | INSAVC | United Kingdom | D13096 |
| FCoV-II | 79-1683 | United Kingdom | X80799 |
| FCoV-II | 79-1146 | Holland | X06170 |
| CCoV-I | Elmo/02 | Italy | AY170345 |
| FCoV-I | UCD1 | Japan | AB088222 |
| FCoV-I | Black | Japan | AB088223 |
The sequences of the VP2 gene segments analysed, in the CPV-2 viruses under study, were deposited in GenBank under the following accession numbers: CG4/09/Ire, GQ387486; PH4/09/Ire, GQ387487; PH1/09/Ire, GQ387488; PH2/09/Ire, GQ387489; BC2/09/Ire, GQ414751; BC4/09/Ire, GU014701. The sequences of the S gene segments analysed, in the CCoV viruses under study, were deposited in GenBank under the following accession numbers: PH4/09/Ire, GU324419; AVC3/09/Ire, GU324418.
Following PCR amplification of a segment of the VP2 gene, a single band of 583-bp was observed in seven samples (Table 1b) as well as the positive control when visualized on a 1.5% agarose gel. Samples from clinically healthy dogs were used as negative controls, and none of these produced an amplified product. Out of the seven positive parvovirus samples, one sample (PH4/09/Ire) also tested positive for the presence of CCoV.
A total of three out of 250 samples (Table 1c) tested positive for the presence of the CoV polymerase gene. Subsequently, the presence of canine-specific CoV in these three samples was confirmed by the successful amplification of a 280-bp segment of the N gene.
RFLP analysis indicated that none of the CPV strains were variant type 2c, as the PCR amplicon was not digested and remained as a single band when run on an agarose gel. Sequence analysis confirmed these findings.
The N terminus of the S gene was amplified in all isolates using a set of three primers that were designed to differentiate between CCoV-I and CCoV-II [37]. Amplification of the S gene of CCoV-I produced a band of 1108 bp, whereas amplification of the S gene of CCoV-II produced a band of 578 bp. Strain PH4/09/Ire was characterized as CCoV-I, whereas strains AVC2/09/Ire and AVC3/09/Ire were characterized as CCoV-II.
Sequencing of the seven CPV strains revealed that three strains, PH2/09/Ire, BC2/09/Ire and BC4/09/Ire, possessed the aa Asn at position 426 of the VP2 protein. Therefore, these strains were characterized as CPV-2a. Strains CG4/09/Ire, PH4/09/Ire, PH1/09/Ire and BC5/09/Ire possessed the aa Asp at position 426 and were therefore characterized as CPV-2b. In addition, the sequence of the S gene of the three CCoV strains, PH4/09/Ire, AVC2/09/Ire and AVC3/09/Ire, was obtained. However, the sequence of strain AVC2/09/Ire was not exploitable for further analysis. Following alignment of PH4/09/Ire and AVC3/09/Ire with other CCoV-I and II reference strains, the PCR typing results were confirmed.
This twelve-month epidemiological study was conducted in southern Ireland from October 2008 to October 2009. Two hundred fifty samples were collected in total, from both symptomatic and asymptomatic dogs in counties Cork and Kerry. Samples were sourced from several veterinary clinics, the Irish Guide Dogs Association, hunting hounds, racing greyhounds and the Irish Society for Prevention of Cruelty to Animals (ISPCA) (Table 1a). The primers 555f and 555r were used to target a VP2 gene segment of CPV. CoV detection was performed by PCR amplification of the conserved region of the polymerase gene, which allows the detection of all three groups of coronavirus, and subsequently, canine-specific CoV presence was confirmed via PCR amplification of the N gene. In total, out of 250 samples, seven samples tested positive for the presence of CPV, three samples tested positive for the presence of CCoV, and a dual infection was detected in one sample.
Dogs infected with CPV and CCoV may shed the virus once clinical symptoms have ceased. In addition, recovered dogs may serve as asymptomatic carriers and shed the virus periodically [1, 40]. This is an important mechanism for the continued circulation and persistence of CPV and CCoV in the environment. However, in this study, both viruses were isolated exclusively from dogs that presented with clinical symptoms. The viruses were isolated from samples sourced in veterinary clinics (n=8) and the ISPCA (n=1), from dogs aged two months to 15 years, several of which had been vaccinated previously. Out of 40 samples sourced from symptomatic dogs, seven were positive for the presence of CPV (17.5%). All dogs presented with haemorrhagic enteritis, while three also presented with vomiting. Additionally, one dog presented with pyrexia, and another case resulted in death (Table 1b). Three CCoV were detected in 40 samples from symptomatic dogs (7.5%), one of which was present in combination with CPV, and this mixed infection produced severe haemorrhagic gastroenteritis (Table 1c).
RFLP analysis was initially used to diagnose CPV-2c infection in vitro, as the nucleotide sequence required to substitute Glu at aa position 426 created a new MboII restriction site (GAAGA), which is unique to this strain [7]. This restriction site was not detected in any of the current isolates. Subsequently, the VP2 gene segment was sequenced in order to confirm the RFLP results, to determine which amino acid each isolate contained at position 426 and thus reveal if isolates were CPV-2a or CPV-2b. Three isolates possessed the aa Asn at position 426 of the VP2 protein and were therefore characterised as CPV-2a. All other isolates possessed the aa Asp at position 426 and thus were characterised as CPV-2b. There does not appear to be any correlation between CPV type and symptom severity. A variety of phylogenetic trees were constructed from this information. However, it can be seen in Figure 1 that the VP2 gene has a low variability, and thus is not particularly informative from a phylogenetic view. Typically, a 99% maximum identity is displayed between the VP2 gene of CPV-2 strains. The phylogenetic tree prepared using the maximum-parsimony method proved to give the best possible resolution (Figure 1). CPV strains are subtyped according to a single amino acid in a strategic location, which involves few changes in the nucleotide sequence. As a result, when comparing CPV nucleotide sequence data, the CPV subgroups 2a, b and c are not segregated into three distinct groups, as there are several other nucleotide differences at other less antigenically significant locations on the VP2 gene, which must also be taken into consideration.
The CCoV strains identified were differentiated using a typing PCR, which was based on the variable nature of the S gene [37]. The spike protein is the major antigenic determinant of CoVs. CCoV-I and II share a high nucleotide homology in the viral genome but are highly divergent in the spike protein gene [19]. The strain PH4/09/Ire was characterized as CCoV-I, with strains AVC2/09/Ire and AVC3/09/Ire being characterized as CCoV-II. CCoV-I was first discovered in Italy in 2003 [19]. Since then, it has been reported in other countries, such as the United Kingdom, Greece, Portugal, Belgium, Romania, Sweden and Slovenia [41]. This is the first report of CCoV-I and II in Ireland. In order to confirm PCR typing results, the sequence of the S gene of the three CCoV samples was determined. Subsequent sequence analysis confirmed that CCoV-II was more prevalent than CCoV-I, which is in accordance with other published findings [42]. Although the low numbers of positive samples in this study lack statistical support, it does provide a snapshot of the epidemiology of CCoV in Ireland. Using BLAST analysis, it was determined that the CCoV-I strain PH1/09/Ire displayed 96% nucleotide sequence homology with CCoV-I strain Elmo from Italy. The CCoV-II strain AVC3/09/Ire displayed 91% nucleotide homology with CCoV-II strain INSAVC and 88% nucleotide homology with CCoV-II strain BGF10, both of which were isolated in the United Kingdom.
Considering that the single-stranded nature of CPV renders it more susceptible to mutation than a double-stranded virus, with a mutation rate similar to that observed in rapidly evolving RNA viruses [9], it is imperative to continuously monitor the genotypes of the virus present in the population to ensure any new types that have evolved are detected. The latest CPV variant type to emerge, CPV-2c, has been shown to produce a more pathogenic clinical outcome in some cases, whereas others have a lower severity and mortality rate [16]. As the aa that determines the type is located in a major antigenic site, a mutation that affects the type may have serious implications for vaccine efficacy and the antibody response generated in pups. A number of commercially licensed vaccines are available in Ireland and are given to pups at six weeks and again four weeks later, followed by an annual vaccination booster. It has been shown that the vaccines based on the original CPV-2 isolate protect against CPV-2 variant type challenges; however, a plan to use current strains in vaccine formulations has led to a CPV-2b- based vaccine being licensed in Europe [34].
A number of commercially licensed vaccines are available in Ireland and are given to pups at a young age to protect against CCoV-related enteritis. Currently, all vaccines are based on CCoV-II. However, it has been shown that the level of cross-reactivity between CCoV-I and II is limited [35]. A lack of cross-reactivity has also been reported between the original CCoV-II strain, designated CCoV-IIa, and a more recently described CCoV-II strain that is closely related to TGEV in the 5’ end of the spike gene, designated CCoV-IIb [43]. As a result of RNA recombination, insertions, deletions and a high susceptibility to frequent mutation, coronaviruses can mutate rapidly, leading to new genotypes (CCoV-I), biotypes (pantropic CCoV) and host variants (canine respiratory coronavirus), all of which may present possible difficulties regarding successful vaccination of dogs. Such dilemmas have been encountered with vaccination against other RNA viruses, such as influenza virus and rotaviruses. The evolution of these viruses is well documented to play an important role in the continuous requirement to assess the efficiency of the vaccines and vaccinal updates. For example, the H3 haemagluttinin protein (the major antigenic determinant) exhibits a rate of accumulation of amino acid substitutions of approximately 5 × 10-3 per year [44]. Similarly, human rotavirus strains, detected over an 18-year period from 1986 to 2004 in Palermo, Italy, displayed a rate of accumulation of amino acid substitutions of approximately 2 × 10-3 per year [45].
In this study, viruses were isolated from vaccinated and unvaccinated pups. It is possible, in the cases where vaccination had been carried out, that maternal antibody had interfered with the immune response of the pup at time of vaccination. A suspected lack of efficacy of vaccines based on the original CPV-2 strain against the new CPV variants in circulation has been suggested previously after worldwide CPV outbreaks were observed in regularly vaccinated dogs [46, 47]. As a result of this study, it can be concluded that although it appears that the viral evolution of CPV type 2 is not yet a significant problem for the canine population in Ireland, vaccine efficacy may need to be re-evaluated by the animal healthcare industry, as it is a possibility that the new CPV-2c strain will eventually emerge into the population as a result of importing dogs from other parts of the world. This study provides evidence that both CPV and CCoV are causative agents of gastroenteritis in southern Ireland in symptomatic dogs, with no evidence of viral shedding in asymptomatic dogs. It has also been demonstrated that dual infection of CCoV along with CPV is of significant concern regarding animal health and well-being. Continued surveillance would be beneficial to evaluate better vaccine efficacy, to understand the underlying mechanisms of vaccine breakthroughs and to implement successful prophylactic measures.
Acknowledgements
This work was funded by the Irish Research Council for Science, Engineering and Technology (IRCSET). We are grateful to all those who participated in the study to collect research samples: veterinary surgeons Bill Cashman, Pat O’Doherty, Conor Goold and Peter Hill, as well as the staff at the Guide Dogs Association and the CSPCA. We also thank Patrick O’Donoghue and Conn O’Shea for their help organizing the sample collection of hunting dogs. A CPV-positive control was kindly provided by Professor Canio Buonavoglia from the Department of Animal Health and Wellbeing in Bari, Italy.
References
- 1.Greene CE, Decaro N. Canine viral enteritis. In: Greene, C.E. editor, Infectious Diseases of the Dog and Cat (4th ed). W.B. Saunders Philadelphia, USA, in press
- 2.Appel MJ, Scott FW, Carmichael LE. Isolation and immunisation studies of a canine parvo-like virus from dogs with hemorrhagic enteritis. Vet Rec. 1979;105:156–159. doi: 10.1136/vr.105.8.156. [DOI] [PubMed] [Google Scholar]
- 3.Decaro N, Buonavoglia C. An update on canine coronaviruses: viral evolution and pathobiology. Vet Microbiol. 2008;132:221–234. doi: 10.1016/j.vetmic.2008.06.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Tsao J, Chapman MS, Agbandje M. The three-dimensional structure of canine parvovirus and its functional implications. Science. 1991;251:1456–1464. doi: 10.1126/science.2006420. [DOI] [PubMed] [Google Scholar]
- 5.Truyen U, Evermann JF, Vieler E, Parrish CR. Evolution of canine parvovirus involved loss and gain of feline host range. Virology. 1996;215:186–189. doi: 10.1006/viro.1996.0021. [DOI] [PubMed] [Google Scholar]
- 6.Parrish CR, Aquadro CF, Strassheim ML, Evermann JF, Sgro JY, Mohammed HO. Rapid antigenic-type replacement and DNA evolution of canine parvovirus. J Virol. 1991;65:6544–6552. doi: 10.1128/jvi.65.12.6544-6552.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Buonavoglia C, Martella V, Pratelli A, Tempesta M, Cavalli A, Buonavoglia D, Bozzo G, Elia G, Decaro N, Carmichael L. Evidence for evolution of canine parvovirus type 2 in Italy. J Gen Virol. 2001;82:3021–3025. doi: 10.1099/0022-1317-82-12-3021. [DOI] [PubMed] [Google Scholar]
- 8.Decaro N, Martella V, Desario C, Bellacicco AL, Camero M, Manna L, D’aloja D, Buonavoglia C. First detection of canine parvovirus type 2c in pups with haemorrhagic enteritis in Spain. J Vet Med B Infect Dis Vet Public Health. 2006;53:468–472. doi: 10.1111/j.1439-0450.2006.00974.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Shackleton LA, Parrish CR, Truyen U, Holmes EC. High rate of viral evolution associated with the emergence of carnivore parvovirus. Proc Natl Acad Sci USA. 2005;102:379–384. doi: 10.1073/pnas.0406765102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Vieira MJ, Silva E, Oliveira J, Vieira AL, Decaro N, Desario C, Muller A, Carvalheira J, Buonavoglia C, Thompson G. Canine parvovirus 2c infection in central Portugal. J Vet Invest. 2008;20:488–491. doi: 10.1177/104063870802000412. [DOI] [PubMed] [Google Scholar]
- 11.Decaro N, Desario C, Elia G, Martella V, Mari V, Lavazza A, Nardi M, Buonavoglia C. Evidence for immunisation failure in vaccinated adult dogs infected with canine parvovirus type 2c. New Microbiol. 2008;31:125–130. [PubMed] [Google Scholar]
- 12.Kang BK, Song DS, Lee CS, Jung KI, Park SJ, Kim EM, Park BK. Prevalence and genetic characterization of canine parvoviruses in Korea. Virus Genes. 2008;36:127–133. doi: 10.1007/s11262-007-0189-6. [DOI] [PubMed] [Google Scholar]
- 13.Hong C, Decaro N, Desario C, Tanner P, Pardo MC, Sanchez S, Buonavoglia C, Saliki JT. Occurrence of canine parvovirus type 2c in the United States. J Vet Diagn Invest. 2007;19:535–539. doi: 10.1177/104063870701900512. [DOI] [PubMed] [Google Scholar]
- 14.Perez R, Francia L, Romero V, Maya L, Lopez I, Hernandez M. First detection of canine parvovirus type 2c in South America. Vet Microbiol. 2007;124:147–152. doi: 10.1016/j.vetmic.2007.04.028. [DOI] [PubMed] [Google Scholar]
- 15.Nakamura M, Tohya Y, Miyazawa T, Mochizuki M, Phung HT, Nguyen NH, Huynh LM, Nguyen LT, Nguyen PN, Nguyen PV, Nguyen NP, Akashi H. A novel antigenic variant of canine parvovirus from a Vietnamese dog. Arch Virol. 2004;149:2261–2269. doi: 10.1007/s00705-004-0367-y. [DOI] [PubMed] [Google Scholar]
- 16.Decaro N, Desario C, Campolo M, Elia G, Martella V, Ricci D, Lorusso E, Buonavoglia C. Clinical and virological findings in pups naturally infected by canine parvovirus type 2 Glu-426 mutant. J Vet Diagn Invest. 2005;17:133–138. doi: 10.1177/104063870501700206. [DOI] [PubMed] [Google Scholar]
- 17.Decaro N, Viviana M, Campolo M, Lorusso A, Camero M, Elia G, Martella V, Cordioli P, Enjuanes L, Buonavoglia C. Recombinant canine coronaviruses related to transmissible gastroenteritis virus of swine are circulating in dogs. J Virol. 2008;83:1532–1537. doi: 10.1128/JVI.01937-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Binn LN, Lazar EC, Keenan KP, Huxsoll DL, Marchwicki RH, Strano AJ. Recovery and characterization of a coronavirus from military dogs with diarrhea. Proc Annu Meet U S Anim Health Assoc. 1974;78:359–366. [PubMed] [Google Scholar]
- 19.Pratelli A, Martella V, Decaro N, Tinelli A, Camero M, Cirone F, Elia G, Cavalli A, Corrente M, Greco M, Buonavoglia D, Gentile M, Tempesta M, Buonavoglia C. Genetic diversity of a canine coronavirus detected in pups with diarrhea in Italy. J Virol Methods. 2003;110:9–17. doi: 10.1016/S0166-0934(03)00081-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Lorusso A, Decaro N, Schellen P, Rottier PJM, Buonavoglia C, Haijema BJ, Groot RJ. Gain, preservation and loss of a group 1a coronavirus accessory glycoprotein. J Virol. 2008;82:10312–10317. doi: 10.1128/JVI.01031-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Jarvis TC, Kirkegaard K. The polymerase in its labyrinth: mechanisms and implications of RNA recombination. Trends Genet. 1991;7:186–191. doi: 10.1016/0168-9525(91)90434-R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Dolja VV, Carrington JC. Evolution of positive-strand RNA viruses. Semin Virol. 1992;3:315–326. [Google Scholar]
- 23.Makino S, Keck JG, Stohlman SA, Lai MMC. High frequency RNA recombination of murine coronavirus. J Virol. 1986;57:729–739. doi: 10.1128/jvi.57.3.729-737.1986. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Decaro N, Campolo M, Lorusso A, Desario C, Mari V, Colaianni ML, Elia G, Martella V, Buonavoglia C. Experimental infection of dogs with a novel strain of canine coronavirus causing systemic disease and lymphopenia. Vet Microbiol. 2008;128:253–260. doi: 10.1016/j.vetmic.2007.10.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Decaro N, Martella V, Ricci D, Elia G, Desario C, Campolo M, Cavaliere N, Di Trani L, Tempesta M, Buonavoglia C. Genotype-specific fluorogenic RT-PCR assays for the detection and quantitation of canine coronavirus type I and type II RNA in faecal samples of dogs. J Virol Methods. 2005;130:72–78. doi: 10.1016/j.jviromet.2005.06.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Decaro N, Camero M, Greco G, Zizzo N, Elia G, Campolo M, Pratelli A, Buonavoglia C. Canine distemper and related diseases: report of a severe outbreak in a kennel. New Microbiol. 2004;27:177–181. [PubMed] [Google Scholar]
- 27.Decaro N, Campolo M, Elia G, Buonavoglia D, Colaianni ML, Lorusso A, Mari V, Buonavoglia C. Infectious canine hepatitis: an “old” disease reemerging in Italy. Res Vet Sci. 2007;83:269–273. doi: 10.1016/j.rvsc.2006.11.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Pratelli A, Tempesta M, Roperto FP, Sagazio P, Carmichael L, Buonavoglia C. Fatal coronavirus infection in puppies following canine parvovirus 2b infection. J Vet Diagn Invest. 1999;11:550–553. doi: 10.1177/104063879901100615. [DOI] [PubMed] [Google Scholar]
- 29.Martella V, Lorusso E, Decaro N, Elia G, Radogna A, D’Abramo M, Desario C, Cavalli A, Corrente M, Camero M, Germinario CA, Banyai K, Di Martino B, Marsilio F, Carmichael C, Buonavoglia C. Detection and molecular characterization of a canine norovirus. Emerg Infect Dis. 2008;14:1306–8. doi: 10.3201/eid1408.080062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Gouet P, Contrepois M, Dubourguier HC, Riou Y, Scherrer R, Laporte J, Voutherot JF, Cohen J, Hardion RL. The experimental production of diarrhoea in colostrum deprived axenic and gnotoxenic calves with enteropathogenic Escherichia coli, rotavirus, coronavirus and in a combined infection of rotavirus and E. coli. Ann Rech Vet. 1978;9:433–40. [PubMed] [Google Scholar]
- 31.Chang KO, Nielsen PR, Ward LA, Saif LJ. Dual infection of gnotobiotic calves with bovine strains of group A and porcine-like group C rotaviruses influences pathogenesis of the group C rotavirus. J Virol. 1999;73:9284–9283. doi: 10.1128/jvi.73.11.9284-9293.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Truyen U. Evolution of canine parvoviruses - a need for new vaccines? Vet Microbiol. 2006;117:9–13. doi: 10.1016/j.vetmic.2006.04.003. [DOI] [PubMed] [Google Scholar]
- 33.Decaro N, Desario C, Elia G, Campolo M, Lorusso A, Mari V, Martella V, Buonavoglia C. Occurrence of severe gastroenteritis in pups after canine parvovirus vaccine administration: A clinical and laboratory diagnostic dilemma. Vaccine. 2007;25:1161–1166. doi: 10.1016/j.vaccine.2006.10.020. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Martella V, Cavalli A, Decaro N, Elia G, Desario C, Campolo M, Bozzo G, Tarsitano E, Buonavoglia C. Immunogenicity of an intranasally administered modified live canine parvovirus type 2b vaccine in pups with maternally derived antibodies. Clin. Diagn Lab Immunol. 2005;12:1243–1245. doi: 10.1128/CDLI.12.10.1243-1245.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Pratelli A, Elia G, Decaro N, Tola S, Tinelli A, Martella V, Rocca S, Tempesta M, Buonavoglia C. Cloning and expression of two fragments of the S gene of canine coronavirus type I. J Virol Methods. 2004;110:9–17. doi: 10.1016/j.jviromet.2003.12.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Stephensen CB, Casebolt DB, Gangopadhyay NN. Phylogenetic analysis of a highly conserved region of the polymerase gene from 11 coronaviruses and development of a consensus polymerase chain reaction assay. Virus Res. 1999;60:181–189. doi: 10.1016/S0168-1702(99)00017-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Erles K, Brownlie J. Sequence analysis of divergent canine coronavirus strains present in a UK dog population. Virus Res. 2009;141:21–25. doi: 10.1016/j.virusres.2008.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Hall TA (1999) Bioedit: a user friendly biological sequence alignment and analysis program for Windows 95/98/NT. Nucl Acids Symp 41: 95-98
- 39.Tamura K, Dudley J, Nei M, Kumur S. MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0. Mol Biol Evol. 2007;24:1596–1599. doi: 10.1093/molbev/msm092. [DOI] [PubMed] [Google Scholar]
- 40.Patel JR, Heldens JGM. Review of companion animal viral diseases and immunoprophylaxis. Vaccine. 2009;27:491–504. doi: 10.1016/j.vaccine.2008.11.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Decaro N, Viviana M, Elia G, Addie D, Camero M, Lucente MS, Martella V, Buonavoglia C (2010) Recombinant Canine Coronaviruses in Dogs, Europe. Emerg Infectious Dis 16: 41-47 [DOI] [PMC free article] [PubMed]
- 42.Decaro N, Desario C, Billi M, Mari V, Elia G, Cavalli A, Martella V, Buonavoglia, C (2009) Western European epidemiological survey for parvovirus and coronavirus infections in dogs. Vet J. doi:10.1016/j.tvjl.2009.10.027 [DOI] [PMC free article] [PubMed]
- 43.Decaro N, Mari V, Campolo M, Lorusso A, Camero M, Elia G, Martella V, Cordioli P, Enjuanes L, Buonavoglia C. Recombinant canine coronaviruses related to transmissible gastroenteritis virus of Swine are circulating in dogs. J Virol. 2009;83:1532–7. doi: 10.1128/JVI.01937-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Hay AJ, Gregory V, Douglas AR, Lin YP. The evolution of human influenza viruses. Philos Trans R Soc Lond B Biol Sci. 2001;356:1861–70. doi: 10.1098/rstb.2001.0999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Arista S, Giammanco GM, Grazia S, Ramirez S, Lo Biundo C, Colomba C, Cascio A, Martella V. Heterogeneity and temporal dynamics of evolution of G1 human rotaviruses in a settled population. J Virol. 2006;80:10724–33. doi: 10.1128/JVI.00340-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 46.Calderon MG, Mattion N, Bucafusco D, Fogel F, Remorini P, La Torre J. Molecular characterization of canine parvovirus strains in Argentina: detection of the pathogenic variant CPV2c in vaccinated dogs. J Virol Methods. 2009;159:141–145. doi: 10.1016/j.jviromet.2009.03.013. [DOI] [PubMed] [Google Scholar]
- 47.Decaro N, Cirone F, Desario C, Elia G, Lorusso E, Colaianni ML, Martella V, Buonavoglia C. Severe parvovirus in a 12-year-old dog that had been repeatedly vaccinated. Vet Rec. 2009;164:593–5. doi: 10.1136/vr.164.19.593. [DOI] [PubMed] [Google Scholar]
- 48.Tamura K, Nei M. Estimation of the number of nucleotide substitutions in the control region of mitochondrial DNA in humans and chimpanzees. Mol. Biol. Evol. 1993;10:512–526. doi: 10.1093/oxfordjournals.molbev.a040023. [DOI] [PubMed] [Google Scholar]
- 49.Edgar RC. MUSCLE: a multiple sequence alignment method with reduced time and space complexity. BMC Bioinformatics. 2004;5:113. doi: 10.1186/1471-2105-5-113. [DOI] [PMC free article] [PubMed] [Google Scholar]


