Abstract
The hemagglutination (HA) activity of porcine epidemic diarrhea virus (PEDV) was investigated. Two cell-adapted strains of PEDV (KPEDV-9 and SM98LVec) were subjected to HA test against erythrocytes of various origin. Both strains showed HA activity with rabbit erythrocytes only after treatment with trypsin or neuraminidase. Optimal conditions for inducing HA activity of PEDV were 2 h incubation at 37°C using phosphate-buffered saline containing 0.1% BSA. These results suggest that the HA activity of PEDV is most likely caused by proteolytic action on it, which could be developed as a new diagnostic method to rapidly detect and differentiate PEDV infections from other enteric diseases.
Keywords: Porcine Epidemic Diarrhea Virus, Infectious Bronchitis Virus, Rabbit Erythrocyte, Neuraminidase Treatment, Porcine Epidemic Diarrhea Virus Strain
Porcine epidemic diarrhea virus (PEDV) is a member of the family Coronaviridae and is closely related to human coronavirus 229E, transmissible gastroenteritis virus (TGEV) and feline infectious peritonitis virus (FIPV) [3, 30]. This virus causes porcine epidemic diarrhea, an enteric disease characterized by acute watery diarrhea and dehydration [8]. The clinical and pathological symptoms of PEDV infection are similar to transmissible gastroenteritis, making it difficult to differentiate between the two [21]. Although PEDV has become enzootic in several countries [4], seasonal outbreaks, resulting in death of neonatal piglets as well as weight loss in fattening pigs, continue to cause serious economic losses in the swine industry [5]. Prophylactic vaccines capable of preventing the initial stages of viral infection usually induce neutralizing antibodies against the viral pathogen [20]. Due to the economic importance of PEDV, cell-adapted strains of the virus have been developed as live attenuated vaccines [16]. Other vaccine candidates have focused on the CO-26K equivalent (COE) domain located on the spike protein, which is reported to contain a neutralizing epitope against PEDV [12].
Hemagglutination (HA) is a specific form of agglutination involving red blood cells. Several viruses are capable of inducing hemagglutination through interaction of the viral surface proteins with the cellular receptors found on the surface of erythrocytes. This property has been widely used for quantitation and analysis of several viruses, including orthomyxoviruses, paramyxoviruses, and encephalomyocarditis virus [18]. Some viruses belonging to the family Coronaviridae have also been reported to possess HA activity. These include human coronavirus OC43 (HCoV OC43) [13, 31], bovine coronavirus (BCoV) [22], hemagglutinating encephalomyelitis virus (HEV) [10], TGEV [19], murine hepatitis virus (MHV) [28], and avian infectious bronchitis virus (IBV) [1].
For some of these coronaviruses, the HA activity is attributed to the presence of an additional surface glycoprotein, the hemagglutinin-esterase glycoprotein (HE), which carries the functional sites for both hemagglutination and acetylesterase activities [27]. The HE protein of these coronaviruses has a structural and functional relationship to the hemagglutinin protein of influenza C virus and may have been derived by recombination between the HE mRNA of influenza C virus and the genomic RNA of an ancestral coronavirus [17]. For others, like IBV, TGEV and BCoV, the spike (S) protein has been observed to possess sialic-acid-binding activity [15, 23]. In the case of IBV, it was reported that HA activity can be induced by trypsin or neuraminidase treatment [6].
The present study investigates the HA activity of PEDV. Two cell-adapted strains of PEDV, KPEDV-9 and SM98LVec, were subjected to HA test against erythrocytes of various origin. The following report describes the prerequisite conditions for inducing the HA activity of PEDV.
African green monkey kidney cells (Vero, CCL-81) from the American Type Culture Collection (ATCC) were prepared in minimum essential medium (MEM, Gibco) containing 5% fetal bovine serum (FBS, Gibco) and 100 U penicillin, 100 μg streptomycin and 34 μg ampotericin B per milliliter of media. Cell-adapted strains of the Korean PEDV isolate, KPEDV-9 and SM98LVec, provided by the Korea National Veterinary Quarantine and Research Services of Korea, were propagated using Vero cells following to the method described by Hofmann and Wyler, with some modifications [11]. Briefly, Vero cells were inoculated with stock virus at a multiplicity of infection ≥1 and then cultured in serum-free MEM at 37°C, 5% CO2 for 48–60 h. Virus was harvested by collecting the supernatant, which was then clarified by centrifugation (2,500 rpm for 10 min, 4°C). The viruses were kept at −70°C until use.
Erythrocytes were obtained from rabbit, pig, chicken, mouse and dog. Briefly, whole blood were collected in a sterile anticoagulant tube containing K3EDTA (Vacutainer, BD Bioscience), then immediately washed three times with 10 mM phosphate-buffered saline (PBS, pH 7.2). The erythrocytes were placed in 10% Alsever’s solution at 4% (v/v) suspension and kept at 4°C for a maximum of 1 week.
The HA test was performed using V-shaped microtiter plates (SPL Labware). KPEDV-9 and SM98LVec cultured in serum-free MEM were serially diluted twofold in PBS containing 0.1% (w/v) bovine serum albumin (BSA Fraction V, Merck), and then 50 μl of sample was distributed to each well. PBS containing 0.1% BSA was used as a negative control. The erythrocytes were reconstituted to a 1% (v/v) suspension in PBS containing 0.1% BSA, and a 50-μl aliquot of this suspension was added to each well (final concentration of 0.5% v/v) containing the diluted virus. The mixture was incubated at either 4 or 37°C and observed until all of the erythrocytes in the negative control have sedimented to the bottom of the well. The HA titer was reported as the reciprocal value of the maximum dilution that causes complete agglutination.
Preliminary findings showed that none of the erythrocytes agglutinated in the presence of PEDV (data not shown). As mentioned previously, some strains of IBV showed HA activity after treatment of the virus with trypsin [6]. Therefore, the HA test was repeated using trypsin-treated PEDV. Prior to the HA test, KPEDV-9 and SM98LVec were treated with 5–100 μg/ml trypsin (Sigma) at 37°C for 30 min. Afterwards, trypsin was inactivated with 2 μg/ml aprotinin (Sigma). The HA test using erythrocytes from different species was repeated at 37°C as described above. Trypsin treatment of KPEDV-9 and SM98LVec did not induce appreciable HA activity against erythrocytes from pig, chicken, mouse or dog. However, both strains exhibited HA activity to rabbit erythrocytes at 37°C when treated with 5 μg/ml trypsin, which was more clearly defined at higher concentrations (Fig. 1). Based on these findings, subsequent HA tests were conducted with 0.5% rabbit erythrocytes and virus pre-treatment with 10 μg/ml trypsin.
Fig. 1.
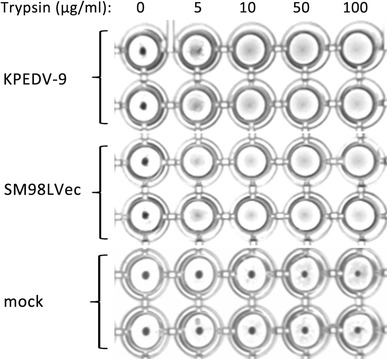
Trypsin treatment of PEDV induces HA activity with rabbit erythrocytes. KPEDV-9 and SM98LVec were treated with various concentrations of trypsin at 37°C for 30 min prior to HA test. Spent culture media from non-infected Vero cells was used as mock virus
The correlation between HA activity and infectious titer of PEDV was also investigated. Vero cells, grown to confluence in a 25-cm2 TC flask (SPL Labware), was inoculated with 20 HA units of KPEDV-9 and then cultured in serum-free MEM at 37°C, 5% CO2. Samples of the culture supernatant were taken at 0, 8, 16, 20, and 24 h post-inoculation (hpi) and titrated by focus formation assay as described previously [7]. At the same time, the same samples were treated with 10 μg/ml trypsin and subjected to the HA test. Figure 2 shows the comparative levels of HA units/focus-forming units of KPEDV-9. Within a 24-h period, the KPEDV-9 titer in the culture supernatant increased from 8 × 102 ffu per ml at 8 hpi to 1 × 105 ffu per ml at 24 hpi. Similarly, the HA titer of KPEDV-9 was measured at 2 HA units at 8 hpi and 32 HA units at 24 hpi. There is a positive correlation between the HA titer and infectious titer by ffa in KPEDV-9. However, the ratio of HA unit/ffu was not consistent among the samples tested, suggesting that the HA titer of PEDV cannot be precisely translated into infectious units. Nonetheless, measuring the HA titer may be sufficient for monitoring the rate of virus growth.
Fig. 2.
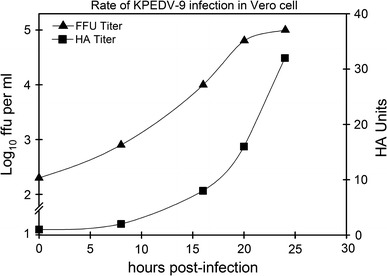
Comparison of HA titer and infectious titer by ffa. Culture supernatant was sampled from PEDV-infected Vero cells at various times and subjected to HA test and focus formation assay. Infectious titer is described in Log10 ffu per ml; HA titer is described in HA units
Based on the full genome sequence of PEDV strain CV777 (GenBank accession no. AF353511.1), this virus lacks the HE protein responsible for the HA activity of other coronaviruses like MHV-DVIM, huCoV OC43 and BRCV G95 [3]. Therefore, our primary assumption is that the HA activity of PEDV is attributed to the S protein. We further investigated the HA activity of the PEDV S protein by subjecting the virus or rabbit erythrocytes to neuraminidase treatment. This is based on several reports that focused on the HA activity of the spike protein of IBV and TGEV. Schultz et al. [24, 25] reported that the HA activity of IBV and TGEV can be induced by treating the virions with the neuraminidase enzyme, which removes the alpha 2,3-linked N-acetylneuraminic acid from the virus surface, while Schwegmann et al. [26] showed that neuraminidase treatment of bovine erythrocytes at less than 3 U/ml did not completely abolish agglutination by TGEV, indicating that a large amount of sialic acid on the surface of the erythrocytes was not necessary for hemagglutination by this virus. Briefly, KPEDV-9 and SM98LVec were prepared by culturing in Vero cells without supplementing with trypsin, following the method described earlier. The supernatants were harvested after 24 h then clarified by centrifugation. The virus suspensions were separated into two groups. One group was treated with 10 μg/ml trypsin as described earlier, while the other group was treated with 50 mU/ml of neuraminidase from Clostridium perfringens (Sigma-Aldrich) at 37°C for 30 min with shaking and then purified by ultracentrifugation. The purified virus was diluted to the original volume using PBS and then subjected to HA test. Rabbit erythrocytes were also treated with 2 U/ml of the neuraminidase enzyme at 37°C for 30 min and washed three times with PBS before being used in a separate HA test with the two strains of PEDV.
Similar to the effect of trypsin, the HA activity of KPEDV-9 and SM98LVec was induced by neuraminidase treatment (Table 1). The newly prepared cultures of KPEDV-9 and SM98LVec showed HA titers of 8 HA units after trypsin treatment and 2 and 4 HA units, respectively, after neuraminidase treatment. The discrepancy in the HA titer of KPEDV-9 obtained with the recent virus preparation compared to the previous preparation may have been caused by variations in the efficiency of virus replication under non-trypsin conditions. Nonetheless, the resulting HA titers were sufficient for this particular study. Differences in the HA activity between trypsin-treated and neuraminidase-treated virus can be attributed to two factors: efficiency of virus recovery during purification after neuraminidase treatment and inherent differences in the mechanism of activation of HA activity by trypsin and neuraminidase, which were not elucidated in this study. Meanwhile, neuraminidase treatment of rabbit erythrocytes did not abolish the HA activity of PEDV (Table 2). While neuraminidase-induced HA activity of PEDV was observed in rabbit erythrocytes, a similar treatment was not established for erythrocytes from other species.
Table 1.
PEDV strains KPEDV-9 and SM98LVec were subjected to trypsin or neuraminidase treatment at 37°C for 30 min prior to HA test
| Virus strain | Treatment | ||
|---|---|---|---|
| Non-treated (HA units) | Trypsina (HA units) | Neuraminidaseb (HA units) | |
| KPEDV-9 | 0 | 8 | 2 |
| SM98LVec | 0 | 8 | 4 |
a10 μg/ml trypsin at 37°C, 30 min
b50 mU/ml neuraminidase at 37°C, 30 min
Table 2.
Rabbit erythrocytes were subjected to neuraminidase treatment at 37°C for 30 min prior to HA test with trypsin-treated PEDV strains
| Virus strain | Rabbit erythrocytes | |
|---|---|---|
| Non-treated (HA units) | Neuraminidase treateda (HA units) | |
| KPEDV-9 | 8 | 8 |
| SM98LVec | 8 | 8 |
a2 U/ml at 37°C, 30 min
Based from the above findings, PEDV possess HA activity even though it lacks the HE surface protein that confers HA activity in other coronaviruses. The HA activity of PEDV was elicited by trypsin or neuraminidase treatment, suggesting that proteolytic processing or removal of the N-acetylneuraminic acid from the virus surface, most likely on the spike protein, is responsible for this attribute. While the target erythrocytes of PEDV (rabbit origin) differ from those of TGEV (bovine origin), their HA activities share a common trait: neither virus requires a large number of surface-bound sialic acid residues on their target erythrocytes for a hemagglutination reaction.
HA is a useful diagnostic and serologic property shared by many different viruses. However, the conditions governing HA and the properties of hemagglutinins are very different, depending on the virus [29]. In previous studies, it was reported that some coronavirus agglutinate a variety of erythrocytes only under special conditions [2, 6]. Large variations exist among members of the family Coronaviridae in their ability to agglutinate erythrocytes. HEV readily agglutinates chicken erythrocytes at room temperature and forms a relatively permanent bond with the erythrocytes [10]. In the case of MHV, while several strains contain the HE glycoprotein, only the MHV-DVIM strain has been shown to cause hemagglutination [9, 27]. HA activity of IBV was first revealed after trypsin, phospholipase C, and neuraminidase treatment [6, 14]. Noda et al. [19] reported in 1987 that TGEV grown in primary swine kidney cell cultures agglutinated erythrocytes from chicken, guinea pig and cattle but not erythrocytes from mouse and goose at 4°C. It was also found that the HA reaction is effectively inhibited by specific antiserum.
Here, we describe the HA activity of two different strains of PEDV against erythrocytes of different origin. Both strains were able to agglutinate rabbit erythrocytes, but not the other types of erythrocytes. In addition, HA activity was only induced by treatment with either trypsin or neuraminidase, similar to that of TGEV and IBV. On the basis of these results, it can be concluded that the HA activity of PEDV is dependent on the removal of the neuraminic acid on the virion surface through either trypsin or neuraminidase treatment. The ability of PEDV to agglutinate rabbit erythrocytes, but not chicken or pig erythrocytes, can be used as a diagnostic tool to differentiate PED from TGE infection. This is particularly useful because both PEDV and TGEV infections exhibit similar clinical manifestations, and differentiation by other methods such as RT-PCR, ELISA or other commercial test kits is rather expensive. Further studies are needed to understand the mechanism of PEDV HA activity. Nonetheless, these findings provide primary insight for the development of new diagnostic methods for PEDV detection.
Acknowledgments
This study was supported by the Technology Development Program for Agriculture and Forestry, Ministry for Food, Agriculture, Forestry and Fisheries, Republic of Korea and the Korean Research Foundation grant funded by the Korean Government (MOEHRD No. E00157, E00045, E00027).
References
- 1.Alexander DJ, Chettle NJ. Procedures for the haemagglutination and the haemagglutination inhibition tests for avian infectious bronchitis virus. Avian Pathol. 1977;6:9–17. doi: 10.1080/03079457708418208. [DOI] [PubMed] [Google Scholar]
- 2.Biswal N, Nazerian K, Cunningham CH. A hemagglutinating fraction of infectious bronchitis virus. Am J Vet Res. 1966;27:1157–1167. [Google Scholar]
- 3.Bridgen A, Kocherhans R, Tobler K, Laude H, Ackermann M. Sequence determination of the nucleocapsid protein gene of the porcine epidemic diarrhea virus confirms that this is a coronavirus related to human coronavirus 229E and porcine transmissible gastroenteritis virus. J Gen Virol. 1993;74:1795–1804. doi: 10.1099/0022-1317-74-9-1795. [DOI] [PubMed] [Google Scholar]
- 4.Carvajal A, Lanza I, Diego R, Rubio P, Carmenes P. Seroprevalence of porcine epidemic diarrhea virus infection among different types of breeding swine farms in Spain. Prev Vet Med. 1995;23:33–40. doi: 10.1016/0167-5877(94)00429-M. [DOI] [Google Scholar]
- 5.Chae C, Kim O, Choi C, Min K, Cho WS, Kim J, Tai JH. Prevalence of porcine epidemic diarrhea virus and transmissible gastroenteritis virus infection in Korean pigs. Vet Rec. 2000;18:606–608. doi: 10.1136/vr.147.21.606. [DOI] [PubMed] [Google Scholar]
- 6.Corbo L, Cunningham CH. Hemagglutination by trypsin-modified infectious bronchitis virus. Am J Vet Res. 1959;20:876–883. [PubMed] [Google Scholar]
- 7.Cruz DJM, Shin HJ. Application of a focus formation assay for detection and titration of porcine epidemic diarrhea virus. J Virol Methods. 2007;145:56–61. doi: 10.1016/j.jviromet.2007.05.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Debouck P, Pensaert MB. Experimental infection of pigs with a new porcine enteric coronavirus CV777. Am J Vet Res. 1980;41:219–223. [PubMed] [Google Scholar]
- 9.Gagneten S, Gout O, Dubois-Dalco M, Rottier P, Rossen J, Hokmes KV. Interaction of mouse hepatitis virus (MHV) spike glycoprotein with receptor glycoprotein MHVR is required for infection with an MHV strain that expresses the hemagglutinin-esterase glycoprotein. J Virol. 1995;69:889–895. doi: 10.1128/jvi.69.2.889-895.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Girard A, Greig AS, Mitchell D. Encephalomyelitis of swine caused by a hemagglutinating virus. III. Serological studies. Res Vet Sci. 1964;5:294–302. [Google Scholar]
- 11.Hofmann M, Wyler R. Propagation of the virus of porcine epidemic diarrhea in cell culture. J Clin Microbiol. 1988;26:2235–2239. doi: 10.1128/jcm.26.11.2235-2239.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Kang TJ, Han SC, Yang SY, Jang YS. Expression of synthetic neutralizing epitope of porcine epidemic diarrhea virus fused with synthetic B subunit of Escherichia coli heat-labile enterotoxin in tobacco plants. Protein Expr Purif. 2006;46:16–22. doi: 10.1016/j.pep.2005.07.026. [DOI] [PubMed] [Google Scholar]
- 13.Kapikian AZ, James HD, Jr, King LM, Vaughn AL, Chanock RM. Hemadsorption by coronavirus strain OC43. Proc Soc Exp Biol Med. 1972;139:179–186. doi: 10.3181/00379727-139-36105. [DOI] [PubMed] [Google Scholar]
- 14.King EJ. Observations on the preparation and stability of infectious bronchitis virus hemagglutination antigen from virus propagated in chicken embryo and chicken kidney cell cultures. Avian Dis. 1984;28:504–518. doi: 10.2307/1590358. [DOI] [PubMed] [Google Scholar]
- 15.Krempl C, Ballesteros ML, Zimmer G, Enjuanes L, Klenk HD, Herrler G. Characterization of the sialic acid binding activity of transmissible gastroenteritis coronavirus by analysis of haemagglutination-deficient mutants. J Gen Virol. 2000;81:489–496. doi: 10.1099/0022-1317-81-2-489. [DOI] [PubMed] [Google Scholar]
- 16.Kweon CH, Kwon BJ, Lee JG, Kwon GO, Kang YB. Derivation of attenuated porcine epidemic diarrhea virus (PEDV) as vaccine candidate. Vaccine. 1999;17:2546–2553. doi: 10.1016/S0264-410X(99)00059-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Luytjes W, Bredenbeek PJ, Noten ARN, Horzinek MC, Spaan WJM. Sequence of mouse hepatitis virus A59 mRNA 2: indications for RNA recombination between coronaviruses and influenza C virus. Virology. 1988;166:415–422. doi: 10.1016/0042-6822(88)90512-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Markwell MAK. Viruses as hemagglutinins and lectins. In: Mirelman D, editor. Microbial lectins and agglutinins: properties and biological activity. New York: Wiley; 1986. pp. 21–53. [Google Scholar]
- 19.Noda M, Yamachita H, Koide F, Kadoi K, Omori T, Asagi M, Inaba Y. Hemagglutination with transmissible gastroenteritis virus. Arch Virol. 1987;96:109–115. doi: 10.1007/BF01310994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Pensaert MB. Porcine epidemic diarrhea. In: Straw BE, D’Allaire S, Mengeling WL, Taylor DI, editors. Diseases of swine. 8. Iowa: Iowa State University Press; 1999. pp. 179–185. [Google Scholar]
- 21.Pritchard GC, Panton DJ, Wibberley G, Ibata G. Transmissible gastroenteritis and porcine epidemic diarrhea in Britain. Vet Rec. 1999;144:616–618. doi: 10.1136/vr.144.22.616. [DOI] [PubMed] [Google Scholar]
- 22.Sato K, Inada Y, Kurogi H, Takahashi E, Satoda K, Omori T, Matumoto M. Haemagglutination by calf diarrhea coronavirus. Vet Microbiol. 1977;2:83–87. doi: 10.1016/0378-1135(77)90035-9. [DOI] [Google Scholar]
- 23.Schultze B, Gross HJ, Brossmer R, Herrler G. The S protein of bovine coronavirus is a hemagglutinin recognizing 9-O-acetylated sialic acid as a receptor determinant. J Virol. 1991;65:6232–6237. doi: 10.1128/jvi.65.11.6232-6237.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Schultze B, Cavanagh D, Herller G. Neuraminidase treatment of avian infectious bronchitis coronavirus reveals a hemagglutinating activity that is dependent on sialic acid-containing receptors on erythrocytes. Virology. 1992;189:792–794. doi: 10.1016/0042-6822(92)90608-R. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Schultze B, Krempl C, Ballesteros ML, Shaw L, Schauer R, Enjuanes L, Herrler G. Transmissible gastroenteritis coronavirus, but not the related porcine respiratory coronavirus, has a sialic acid (N-glycolylneuraminic acid) binding activity. J Virol. 1996;70:5634–5637. doi: 10.1128/jvi.70.8.5634-5637.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Schwegmann C, Zimmer G, Yoshino T, Enss M-L, Herrler G. Comparison of the sialic acid binding activity of transmissible gastroenteritis coronavirus and E. coli K99. Virus Res. 2001;75:69–73. doi: 10.1016/S0168-1702(01)00228-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Sugiyama K, Kasai M, Kato S, Kasai H, Hatakeyama K. Hemagglutinin-esterase protein (HE) of murine coronavirus: DVIM. Arch Virol. 1998;143:1523–1534. doi: 10.1007/s007050050395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Talbot PJ. Hemagglutination by murine hepatitis viruses. Intervirology. 1989;30:117–120. doi: 10.1159/000150109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Turner R, Lathey JL, Van Voris LP, Belshe RB. Serological diagnosis of influenza B virus infection: comparison of an enzyme-linked immunosorbent assay and the hemagglutination inhibition test. J Clin Micobiol. 1982;15:824–829. doi: 10.1128/jcm.15.5.824-829.1982. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Yaling Z, Ederveen K, Egberink H, Pensaert MB, Horzinek MC. Porcine epidemic diarrhea virus (CV777) and feline infectious peritonitis virus (FIPV) are antigenically related. Arch Virol. 1988;102:63–71. doi: 10.1007/BF01315563. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Zhang XM, Kousoulas KG, Storz J. The hemagglutinin/esterase gene of human coronavirus strain OC43: phylogenetic relationships to bovine and murine coronavirus and influenza C virus. Virology. 1992;186:318–323. doi: 10.1016/0042-6822(92)90089-8. [DOI] [PMC free article] [PubMed] [Google Scholar]


