Abstract
Marine sponges have been considered as a gold mine during the past 50 years, with respect to the diversity of their secondary metabolites. The biological effects of new metabolites from sponges have been reported in hundreds of scientific papers, and they are reviewed here. Sponges have the potential to provide future drugs against important diseases, such as cancer, a range of viral diseases, malaria, and inflammations. Although the molecular mode of action of most metabolites is still unclear, for a substantial number of compounds the mechanisms by which they interfere with the pathogenesis of a wide range of diseases have been reported. This knowledge is one of the key factors necessary to transform bioactive compounds into medicines. Sponges produce a plethora of chemical compounds with widely varying carbon skeletons, which have been found to interfere with pathogenesis at many different points. The fact that a particular disease can be fought at different points increases the chance of developing selective drugs for specific targets.
Keywords: sponge medicine, natural product, cancer, inflammation, virus
INTRODUCTION
The relationship between sponges and medicines goes back to Alexandrian physicians and was thoroughly describes by the Roman historian Plinius. Physicians used sponges that were saturated with iodine to stimulate coagulation of the blood, or with bioactive plant extracts to anesthetize patients. Sponges were soaked with pure wine and put on the left part of the chest in case of heartaches and soaked in urine to treat bites of poisonous animals. Plinius recommended the use of sponges against sunstrokes, and they were used against all kinds of wounds, bone fractures, dropsy, stomach aches, infectious diseases, and testicular tumors (Hofrichter and Sidri, 2001), or even as implants after breast operations (Arndt, 1938). At least since the 18th century, Russian, Ukrainian, and Polish physicians have used a freshwater sponge they call Badiaga (Figure 1) for the treatment of patients (Nozeman, 1788). The dry powder of this sponge is rubbed on the chest or back of patients with lung diseases or on the sore places in cases of foot and leg aches (such as rheumatism (Schroder, 1942). Oficjalski (1937) discovered that Badiaga is not really one sponge, but mixtures of several freshwater sponges that differ depending on the region. In Poland it consisted of powder of Euspongilla lacustris, Ephydatia fluviatilis, and Meyenia muelleri, while the Russian Badiaga was a mixture of Euspongilla lacustris, Ephydatia fluviatilis, Spongilla fragilis, and Carterius stepanowi. He suggested that the high iodine concentration in all sponge species gives rise to the wholesome effect of Badiaga. At present Stodal, syrup containing roasted Spongia officinalis, is used for homeopathic treatment of dry and asthmatic cough in the Western world (Stodal, 2003).
Figure 1.

Examples of homeopathic drugs based on sponge extracts currently in use (Badiaga and Stodal syrup).
Pharmaceutical interest in sponges was aroused in the early 1950s by the discovery of a nucleosides spongothymidine and spongouridine in the marine sponge Cryptotethia crypta (Bergmann and Feeney, 1950, 1951). These nucleosides were the basis for the synthesis of Ara-C, the first marine-derived anticancer agent, and the antiviral drug Ara-A (Proksch et al., 2002). Ara-C is currently used in the routine treatment of patients with leukemia and lymphoma. One of its fluorinated derivatives has also been approved for use in patients with pancreatic, breast, bladder, and lung cancer (Schwartsmann, 2000). At the same time it was revealed that certain lipid components such as fatty acids, sterols and other unsaponifiable compounds occur in lower invertebrates in a diversity far greater than that encountered among animals of higher organization (Bergmann and Swift, 1951). These early promises have now been substantiated by an overwhelming number of bioactive compounds that have been discovered in marine organisms. More than 15,000 marine products have been described thus for (MarinLit, 1999; Faulkner, 2000, 2001, 2002). Sponges, in particular, are responsible for more than 5300 different products, and every year hundreds of new compounds are being discovered (Faulkner 2000, 2001, 2002).
Most bioactive compounds from sponges can be classified as antiinflammatory, antitumor, immunosuppressive or neurosuppressive, antiviral, antimalarial, antibiotic, or antifouling. The chemical diversity of sponge products is remarkable. In addition to the unusual nucleosides, bioactive terpenes, sterols, cyclic peptides, alkaloids, fatty acids, peroxides, and amino acid derivatives (which are frequently halogenated) have been described from sponges (Figure 2).
Figure 2.
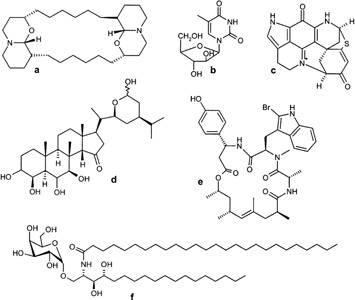
An illustration of the chemical diversity of sponge-derived molecules. a: Xestospongin C (Xestospongia sp. / macrocyclic bis-oxaquinolizidine. b: Spongothymidine (Cryptotethia crypta / unusual nucleoside). c: discorhabdin D (Latrunculia brevis; Prianos sp. / fused pyrrolophenanthroline alkaloid. d: Contignasterol (Petrosia contignata / oxygenated sterol). e: Jaspamide (Hemiastrella minor / macrocyclic lactam/lactone). f: agelasphin (Agelas mauritianus / α-galactosylceramide).
For this review we have surveyed the discoveries of products derived from marine sponges up to now, and attempted to show the variety of potential medical applications of metabolites from sponges and the mechanisms by which they interfere with the pathogenesis of human diseases. This knowledge is a prerequisite for the development of a drug from a bioactive compound. For example, many secondary metabolites inhibit growth of cancer cell lines, but this does not imply that they will be suitable as a medicine against cancer, because they may exhibit important side effects. The following sections summarize compounds by disease type and describe their mode of action, and discuss the reasons why sponges would produce these metabolites.
SPONGE PRODUCTS
Antiinflammatory Compounds
Acute inflammations in the human body can result from microbial infection, physical damage, or chemical agents. The body reacts by changing the blood flow, increasing the permeability of blood vessels, and allowing the escape of cells from the blood into the tissues (Tan et al., 1999). Chronic inflammation of the skin or joints may severely damage the body if it leads to psoriasis or rheumatic arthritis (Pope et at., 1999). Sponges have proved to be an interesting source of antiinflammatory compounds (Table 1).
Table 1.
Examples of Antiinflammatory Products from Sponges
| Compound | Compound class | Species/order | Mode of action | Reference |
|---|---|---|---|---|
| Manoalide | Cyclohexane sesterterpenoid | Luffariella variabilis/Dictyoceratida | Phospholipase A2 inhibitor | Bennet et al., 1987 |
| Dysidotronic acid | Drimane sesquiterpenoid | Dysidea sp./Dendroceratida | Phospholipase A2 inhibitor | Giannini et al., 2000 |
| Ircinin-1 and -2 | Acyclic sesterterpenoid | Ircinia oros/Dictyoceratida | Phospholipase A2 inhibitor | Ciminoe et al., 1972 |
| Petrosaspongiolides M-R | Cheilantane sesterterpenoid | Petrosaspongia nigra/Dictyoceratida | Phospholipase A2 inhibitor | Randazzo et al., 1998a |
| Spongidines A-D | Pyridinium alkaloid | Spongia sp./Dictyoceratida | Phospholipase A2 inhibitor | De Marino et al., 2000 |
| Topsentin | Bis-indole alkaloid | Topsentia genitrix/Halichondrida | Phospholipase A2 inhibitor | Jacobs et al., 1994 |
| Scalaradial | Scalarane sesterterpene | Cacospongia scalaris/Dictyoceratida | Phospholipase A2 inhibitor | De Carvalho and Jacobs, 1991 |
| Cacospongionolide B | Sesterterpene lactone | Fasciospongia cavernosa/Dictyoceratida | Phospholipase A2 inhibitor | Garcia Pastor et al., 1999 |
| Jaspaquinol | Diterpene benzenoid | Jaspis splendens/Astrophorida | Lipoxygenase inhibitor | Carroll et al., 2001 |
| Subersic acid | Diterpene benzenoid | Suberea sp./Verongida | Lipoxygenase inhibitor | Carroll et al., 2001 |
Manoalide, one of the first sesterterpenoids to be isolated from a marine sponge (Luffariella variabilis), was found to be an antibiotic (De Silva and Scheuer, 1980) and an analgesic (Mayer and Jacobs, 1988). In addition, its antiinflammatory properties have been studied extensively (Bennet et al., 1987). The antiinflammatory action is based on the irreversible inhibition of the release of arachidonic acid from membrane phospholipids by preventing the enzyme phospholipase A2 from binding to the membranes (Glaser et al., 1989). A rise in the intracellular arachidonic acid concentration would lead to upregulation of the synthesis of inflammation mediators as prostaglandins and leukotrienes (Figure 3). Phospholipase A2 inhibition has been recorded for many sesterterpenes from sponges of the order Dictyoceratida, but also for bis-indole alkaloids such as topsentin (Jacobs et al., 1994). The mechanism by which they affect the inflammation process is different from commonly used nonsteroidal antiinflammatory drugs. Only a few sponge-derived terpenoids have been found to inhibit lipoxygenase, another enzyme that is involved in the inflammatory response (Carroll et al., 2001).
Figure 3.
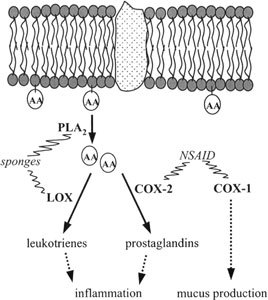
Inflammatory cascade inside the cell. Phospholipase A2 (PLA2) catalyzes the release of membrane-bound arachidonic acid (AA) to free arachidonic acid. Arachidonic acid is converted to leukotrienes and prostaglandins by lipoxygenase (LOX) and cyclooxygenase-2 (COX-2), respectively. Sponge-derived antiinflammatory molecules are mainly inhibitors of PLA2 or LOX, while nonsteroidal antiinflammatory drugs inhibit COX-2, but also the constitutive COX-1.
The antiinflammatory sponge products are selective inhibitors of specific enzymes of a range of diseases, like psoriasis or rheumatic arthritis. The currently used nonsteroidal antiinflammatory drugs often fail to control the disease and present important side effects such as risk of gastrointestinal bleeding and renal complications (De Rosa, 2002). These are caused by unselective inhibition of cyclooxygenases, some of which are also involved in the promotion of the production of the natural mucus that protects the gastrointestinal tract (Bjarnason et al., 1993).
Antitumor Compounds
A number of isolated sponge compounds are inhibitors of protein kinase C (PKC). PKC inhibitors have attracted interest worldwide, as there is evidence that too high levels of PKC enzyme are involved both in the pathogenesis of arthritis and psoriasis (owing to regulation of phospholipase A2 activity), and in tumor development (Bradshaw et al., 1993; Yoshiji et al., 1999). PKC is believed to be the receptor protein of tumor-promoting phorbol esters, and PKC inhibitors prevent binding of carcinosarcoma cells to the endothelium (B. Liu et al., 1991). Glycosylation of the receptors, and especially the presence of fucose residues, plays an important role in the binding of carcinosarcoma cells and leukocytes to the receptors in the endothelium (Springer and Lasky, 1991).
Fucosyltransferase inhibitors, such as the octa- and nonaprenylhydroquinone sulfates that were isolated from a Sarcotragus sp. (Wakimoto et al., 1999), may therefore be promising candidates for controlling inflammatory processes such as arthritis or for combating tumor growth.
In addition to PKC inhibitors and fucosyl transferase inhibitors, numerous anticancer molecules with a different mode of action have been discovered in marine sponges (Table 2). These compounds can be divided in 3 classes:
Table 2.
Examples of Antitumor Products from Sponges
| Compound | Compound class | Species/order | Mode of action | Reference |
|---|---|---|---|---|
| BRS1 | Diamino-dihydroxy polyunsaturated lipid | Calcareous sponge/? | Protein kinase C inhibitora | Willis and De Vries, 1997 |
| Isoaaptamine | Benzonaphthyridine alkaloid | Aaptos aaptos/Hadromerida | Protein kinase C inhibitora | Fedoreev et al., 1989 |
| Debromohymenialdisine | Pyrrole-guanidine alkaloid, prenylhydroquinone derivative | Hymeniacidonaldis/Halichondrida | Protein kinase C inhibitora | Kitagawa et al., 1983 |
| Sarcotragus sp./Dictyoceratida | A1, 3-fucosyltransferase inhibitor | Wakimoto et al., 1999 | ||
| Adociasulfates | Triterpenoid hydroquinones | Haliclona (aka Adocia) sp./Haplosclerida | Kinesin motor protein inhibitors | Blackburn et al., 1999 |
| Discodermolide | Linear tetraene lactone | Discodermia dissolute/Lithistida | Stabilization of microtubules | Ter Haar et al., 1996 |
| Laulimalide | Macrocyclic lactone | Cacospongia mycofljiensis/Dictyoceratida | Stabilization of microtubules | Mooberry et al., 1999 |
| Peloruside A | Macrocyclic lactone | Mycdle hentschett/Poecilosclerida | Stabilization of microtubules | Hood et al., 2002 |
| Hemiasterlin | Unusual tripeptide | Auletta sp./Halichondrida | Stabilization of microtubules | Anderson et al., 1997 |
| Dictyostatin | Macrocyclic lactone | Corallistidae sp./Lithistida | Stabilization of microtubules | Isbrucker et al., 2003 |
| Spongistatin 1 | Bis(spiroacetal) macrolide | Spongia sp./Dictyoceratida | Tubulin polymerisation inhibitor | Bai et al., 1993 |
| Halichondrin B | Polyether macrolide | e.g., Halichondria okadai/Halichondrida | Tubulin polymerisation inhibitor | Hirata and Uemura, 1986; Bai et al., 1991 |
| Arenastatin A | Macrocyclic lactan/lactone | Dysidea arenaria/Dendroceratida | Tubulin polymerisation inhibitor | Koiso et al., 1996 |
| Latrunculin A | Thiazole macrolide | Latrunculia magnified/Poecilosclerida | Actin-depolymerisation | Kashman et al., 1980 Coue et al., 1987 |
| Swinholide A | Macrocyclic lactone | Theonella swinhoei/Lithistida | Actin-depolymerization | Bubb et al., 1995 |
| Mycalolide B | Oxazole macrolide | Mycale sp./Poecilosclerida | Actin-depolymerization | Fusetani et al., 1989 Saito et al., 1994 |
| Jaspamide | Macrocyclic lactam/lactone | Hemiastrella minor/Hadromerida | Actin filament stabilizer | Fabian et al., 1999 |
| Fascaplysin | Bisindole alkaloid | Fascaplysinopsis sp./Dictyoceratida | Cyclin-dependent kinase 4 inhibitor | Soni et al., 2000 |
| Spongiacidin B | Pyrrole alkaloid | Hymeniacidon sp. / Halichondrida | Cyclin-dependent kinase 4 inhibitor | Inaba et al., 1998 |
| Mycalamide A and B | Polyether amide (pederin-like) | Mycale sp./Poecilosclerida | Protein synthesis inhibitor | Burres and Clement, 1989 |
| Girodazole | Imidazole alkaloid | Cymbastela cantharella/Halichondrida | Protein synthesis inhibitor | Ahond et al., 1988; Colson et al., 1992 |
| Aragusterol A | Sterol | Xestospongia sp./Haplosclerida | Protein synthesis inhibitor | Iguchi et al., 1993; Fukuoka et al., 2000 |
| Neoamphimedine | Pyridoacridine alkaloid | Xestospongia cf. carbonaria/Haplosclerida | Topoisomerase II inhibitor | De Guzman et al., 1999 |
| Elenic acid | Alkylphenol | Plakinastrella sp./Homosclerophorida | Topoisomerase II inhibitor | Juagdan et al., 1995 |
| Naamine D | Imidazole alkaloid | Leucetta cf. chagosensis/Calcinea | Nitric oxide synthetase inhibitorb | Dunbar et al., 2000 |
| Agelasphin (KRN7000) | α-Galactosylceramide | Agelas mauritianus /Agelasida | NKT cell activator | Shimosaka, 2002 |
| Agosterol A | Sterol | Spongia sp./Dictyoceratida | Reverses drug resistancy of dancer cells | Aoki et al., 1998 |
| Salicylihalamide A | Salicylate macrolide | Haliclona sp./Haplosclerida | v-ATPase inhibitor | Erickson et al., 1997 |
| Chondropsin A and B | Macrolide lactam | Chondropsis sp./Poecilosclerida | v-ATPase inhibitor | Cantrell et al., 2000; Bowman et al., 2003 |
| 6-Hydroximino-4-en-3-one steroids | Oximated steroid | Cinachyrella sp./Spirophorida | Aromatase inhibitor | Holland et al., 1992 |
| Crambescidins 1-4 | Pentacyclic guanidine derivative | Crambe crambe/Poecilosclerida | Ca2+/channel blocker | Jares-Erijman et al., 1991; Berlinck et al., 1993 |
| Haligramides A and B | Cyclic peptide | Haliclona nigra/Haplosclerida | Unknown | Rashid et al., 2000 |
| Discorhabdin D | Fused pyrrolophenanthroline alkaloid | Latrunculia brevis/Poecilosclerida; Prianos sp./Haplosclerida | Unknown | Perry et al., 1988 |
| Callystatin A | Polyketide | Callyspongia truncata/Haplosclerida | Unknown | Kobayashi et al., 1997 |
| Tedanolide | Macrocyclic lactone | Tedania ignis/Poecilosclerida | Unknown | Schmitz et al., 1984 |
| Glaciasterols A and B | 9, 11-Secosterol | Aplysilla glacialis/Dendroceratida | Unknown | Pika et al., 1992 |
| Axinellins A and B | Cyclic peptide | Axinella carter!/Halichondrida | Unknown | Randazzo et al., 1998b |
| Incrustasterols A and B | Sterol | Dysidea incrustans/Dendroceratida | Unknown | Casapullo et al., 1995 |
aAlso has antiinflammatory activity.
bAlso has immunosuppressive activity.
(1) nonspecific inhibitors of cell growth; (2) specific inhibitors of cancer cells; and (3) inhibitors of cancer cells of a certain type of cancer (as the aforementioned PKC inhibitors).
Many nonspecific cell growth inhibitors have been discovered in sponges. They are valuable for treating cancer under certain conditions, but they also affect the division of healthy cells. Therefore, their applications are limited, depending on their specific characteristics. The cytoskeleton is an interesting target for cancer therapy, as the microtubules and microfilaments are involved in cellular organization during cell division. A number of adociasulfates (triterpenoid hydroquinones) from a Haticlona sp. were the first inhibitors of the kinesin motor protein to be discovered. These toxins are believed to inhibit the protein by binding to the microtubule binding site, “locking up” the protein’s motor function, and thereby blocking cell division (Blackburn et al., 1999). In addition to these triterpenoid hydroquinones, a number of potent microtubule-interfering compounds have been discovered in marine sponges, such as halichondrin B (Bai et al., 1991), spongistatin (Bai et al., 1993), discodermolide (Ter Haar et al., 1996), laulimalide (Moobeny et al., 1999), peloruside A (Hood et al., 2002), and dictyostatin (Isbrucker et al., 2003). Other metabolites, such as latrunculin A from Latrunculia magnifica (Coue et al., 1987) and swinholide A from Theonella swinhoei (Bubb et al., 1998), disrupt the polymerization of actin. Actin which is the key element of the microfilaments, and it can block many cellular processes including cell division. Spongiacidin B (Inaba et al., 1998) and fascaplysin (Soni et al., 2000) are examples of sponge-derived metabolites that inhibit cell division by inhibition of cyclin-dependent kinase 4, which leads to arrest of cells in the G1 phase. Other metabolites, such as mycalamide (Burres and Clement, 1989) and aragusterol (Fukuoka et al., 2000), disturb cell division by inhibition of protein synthesis. Neoamphimedine (De Guzman et al., 1999) and elenic acid (Juagdan et al., 1995) inhibit the development of tumors by blocking topoisomerase II, the nuclear enzyme which makes transient DNA breaks that are required for replication (L.F. Liu and Chen, 1994).
Nitric oxide synthetase inhibitors, such as the imidazole alkaloid Na amine D that was isolated from the calcareous sponge Leucetta cf. chagosensis (Dunbar et al., 2000), are not involved in growth inhibition of cancer cells, but may prevent events in the early phases of tumorigenesis. Nitric oxide could participate in the tumorigenesis by mediating DNA damage and support tumor progression through the induction of angiogenesis (Lala and Orucevic, 1998). However, inhibition of nitric oxide synthetase may also affect other physiologic processes in which nitric oxide is involved, such as intracellular or transcellular messaging, and it is involved in regulation of the immunogenic respons by T lymphocytes. Agelasphin (KRN7000) from Agelas mauritianus (E. Kobayashi et al., 1995) has been found to stimulate the immune system by activation of dendritic and natural killer T (NKT) cells. The NKT cell level is lower in the blood of patients with cancer or autoimmune disease, such as type 1 diabetes (Shimosaka, 2002), and in mice it was shown that tumors could be rejected by stimulation of the immune system by agelasphin (Yamaguchi et al., 1996).
The activity of other compounds is more specific toward tumor cells. Multidrug resistance in human carcinoma cells caused by overexpression of two kinds of membrane glycoproteins is reversed by agosterol A from the marine sponge Spongia sp. It has been suggested that an altered cytosolic pH plays a role in drug resistance. Vascular (H+) ATPase (v-ATPase) is an enzyme involved in many cellular processes that are often upregulated in cancer cells, such as acidic vesicular organelle formation, which is a response to radiation injury or manipulation of the pH to decrease entry of chemotherapeutics into the cells (Martinez-Zaguilan et al., 1999). Salicylihamide A was isolated from a Haliclona sp. as a selective inhibitor of v-ATPase and has been shown to be 60-fold more cytotoxic to certain cancer cells than to their normal noncancerous counterparts (Erickson et al., 1997). The first natural 6-hydroximino-4-en-3-one steroids were isolated from Cinachyrella spp. (Rodriguez et al., 1997) and are examples of molecules that can be deployed against a specific type of cancer. They displayed high affinity to aromatase (Holland et al., 1992), which is the rate-limiting enzyme that catalyzes the conversion of androgens to estrogens (Figure 4). Blockade of this step allows treatment of hormone-sensitive breast cancer that is dependent on estrogen (Lonning et al., 2003). A peculiar fact about the 6-hydroximi no-4-en-3-one steroids is that they were chemically synthesized before they were even discovered in nature.
Figure 4.
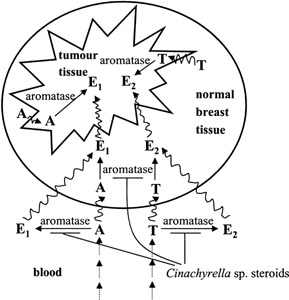
Inhibition of breast cancer by Cinachyrella sp. steroids. Aromatase is the key enzyme in the formation of the estrogens estrone (E1) and estradiol (E2). It catalyzes the final steps, from androstenedione (A) to estron and from testosterone (T) to estradiol, in the estrogen pathway. Estrogen conversion can occur in the blood, in normal breast tissue, as well as in breast tumor tissue (adapted from Geisler, 2003). The 6-hydroximino-4-en-3-one steroids from Cinachyrella sp. are inhibitors of aromatase. The inhibition of aromatase in the tumor tissue is not shown to maintain the clarity of the illustration.
In addition, many more compounds that displayed growth inhibition activity of tumor cell lines have been isolated (Table 2), although their exact effects are still unclear. Discorhabdin D (Perry et al., 1988), chondropsin A and B (Cantrell et al., 2000), haligramides A and B (Rashid et al., 2000), and glaciasterols A and B (Pika et al., 1992) are only a few examples of these molecules.
Immunosuppressive Compounds
In addition to their potential for treatment of cancer, nitric oxide synthetase inhibitors downregulate T-cells are, suppressing the immune system, and they diminish the fierceness of migraine attacks (Griffith and Gross, 1996). Immune system suppression is desired in cases of hypersensitivity to certain antigens (e.g., allergies) or organ transplantations. Patients who receive a donor organ need life-long medication to prevent rejection by the immune system, and for that reason it is extremely important that these medicines are very specific suppressors. Therefore there is a continuous demand for new immunosuppressives. A number of new molecules with immunosuppressive activity, which interfere at different points of the immune response have been discovered in marine sponges (Table 3; Figure 5).
Table 3.
Examples of Immunosuppressive Products from Sponges
| Compound | Compound class | Species/order | Mode of action | Reference |
|---|---|---|---|---|
| Simplexides | Glycolipid | Plakortis simplex/Homosclerophorida | Inhibitor of T-cell proliferation | Costantino et al.,1999 |
| Polyoxygenated sterols | Sterol | Dysidea sp./Dendroceratida | IL-8 inhibitor | Leone et al., 2000 |
| Contignasterol | Oxygenated sterol | Petrosia contignata/Haplosclerida | Histamine release inhibitor | Takei et al., 1994; Bramley et al., 1995 |
| Xestobergsterols A and B | Pentacyclic sterol | Xestosponga berquistia/Haplosclerida | Histamine release inhibitor | Shoji et al., 1992 |
| Taurodispacamide A | Pyrrole-imidazole alkaloid | Agelas oroides/Agelasida | IL-2 inhibitor | Fattorusso and Taglialatela-Scafati, 2000 |
| Pateamine A | Thiazole macrolide | Mycale sp./Poecilosclerida | IL-2 inhibitor | Northcote et al., 1991 |
Figure 5.
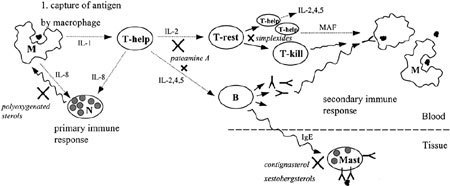
Simplified representation of the immune respons after capture of an antigen by macrophages (M). Both macrophages, but especially T-helper cells (T-help), secrete many interleukins (IL-x) or macrophage activation factor (MAP), to trigger the primary immune response via neutrophils (N), or the secondary immune respons by activating resting T cells (T-rest) and B cells (B). Activated B cells secrete antibodies that bind to macrophages that have phagocytized an antigen, and they are subsequently destroyed by T-killer cells (T-kill). Mast cells (Mast) release histamine as a response to binding of an antigen to IgE molecules present in their cell membranes. The black crosses indicate position where sponge-derived immunosuppressive compounds interfere with the immune response.
Three polyoxygenated sterols from a Dysidea sp. from Northern Australia are selective immunosuppressive compounds that inhibit the binding of interleukin 8 (IL-8), a cytokine that attracts neutrophils into an area of tissue injury, to the IL-8 receptor (Leone et al., 2000). The simplexides from the Caribbean sponge Plakortis simplex are a group of immunosuppressive glycolipids that inhibit proliferation of activated T cells by a noncytotoxic mechanism (Costantino et al., 1999). Pateamine A, from a Mycale sp., inhibits the production of IL-2 (Romo et al., 1998) and thereby the activation of resting T cells and B cells to a lesser extent. Contignasterol from Petrosia contignata (Burgoyne and Andersen, 1992) inhibits allergen-induced histamine release from rat mast cells (Takei et al., 1994) and from guinea-pig lung tissue in vitro (Bramley et al., 1995), and the activation of eosinophils into airways in guinea-pigs and could be used to treat asthma (Langlands et al., 1995).
Cardiovascular Agents
In addition to regulators of the white blood cells, a number of sponge-derived molecules have been found to interfere with other blood-related diseases such as thrombosis, atherosclerosis, or diabetes (Table 4). The process of blood coagulation is triggered by a complex proteolytic cascade that leads to the formation of fibrin. Thrombin is a serine protease that cleaves a peptide fragment from fibrinogen, which then leads to the generation of fibrin, a major component of blood clots (Shuman et al., 1993). Cyclotheonarnide A, isolated from a Theonella sp. (Fusetani et al., 1990), represents an unusual class of serine protease inhibitors and is a potential drug for the treatment of thrombosis (Maryanoff et al., 1993). Eryloside F from Erylus formosus was found to be a potent thrombin receptor antagonist (Stead et al., 2000). Thrombin receptor activation is likely to play a key role not only in arterial thrombosis but also in atherosclerosis (Chackalamannil, 2001). Atherosclerosis starts with damage to the endothelium and subsequent deposition of fats, cholesterol platelets, cellular waste products, calcium, and other substances in the artery wall. These may stimulate endothelial cells to produce a vascular cell adhesion molecule that results in further buildup of cells and shrinkage of the arterial diameter (Zapolska-Downar et al., 2001). Halichlorine from Halichondria okadai is an inhibitor of the expression of vascular cell adhesion molecule 1 (Kuramoto et al., 1996) and may thus impede atherogenesis (Arimoto et al., 1998).
Table 4.
Examples of Sponge Products that Affect Blood-Related Diseases
| Compound | Compound class | Species/order | Mode of Action | Reference |
|---|---|---|---|---|
| Cyclotheonamide A | Cyclic pentapeptide | Theonella sp./Lithistida | Serine protease inhibitor | Maryanoff et al., 1993 |
| Eryloside F | Penasterol disaccharide | Eryltus formosus/Astrophorida | Thrombin receptor antagonist | Stead et al., 2000 |
| Halichlorine | Cyclic aza-polyketide | Halichondria okadai/Halichondrida | VCAM-1 inhibitor | Arimoto et al., 1998 |
| Callyspongynic acid | Polyacetylene | Callyspongia truncata/Haplosclerida | α-glucosidase inhibitora | Nakao et al., 2002 |
aAlso has potential antiviral activity.
Callyspongynic acid, isolated from Callyspongia truncata, is an α-glucosidase inhibitor (Nakao et al., 2002). α-Glucosidase inhibitors interfere with the hydrolysis of glycogen, keeping the glucose concentration in the blood at a lower level, and can be used to treat patients with diabetes (Lebovitz, 1992).
Neurosuppressive Compounds
Keramidine, isolated from an Agelas sp. (Nakamura et al., 1984), is an example of a number of neurosuppressive compounds that have been isolated from marine sponges (Table 5). It is a serotonergic receptor antagonist and blocks serotonin-mediated neural communication. Several different serotonin receptors have been identified. They are related to (1) platelet aggregation, and may therefore be useful against thrombosis (Ruomei et al., 1996); (2) smooth muscle contraction (Garcia-Colunga and Miledi, 1996); (3) vomiting, owing to their presence in the gastrointestinal tract (Lang and Marvig, 1989); (4) and most interestingly, may function as antidepressant drugs in the brain (Nagayama et al., 1980).
Table 5.
Examples of Neurosuppressives and Muscle Relaxants from Sponges
| Compound | Compound class | Species/order | Mode of action | Reference |
|---|---|---|---|---|
| Dysiherbaine | Unusual amino acid | Dysidea herbacea/Dendroceratida | Glutamate receptor antagonist | Sakai et al., 1997 |
| Keramadine | Pyrrole-guanidine alkaloid | Agelas sp. / Agelasida | Serotonergic receptor antagonist | Nakamura et al., 1984 |
| 1-Methylisoguanosine | Nucleoside analogue | Tedania digitata /Poecilosclerida | Unknown (muscle relaxant, antiallergic) | Quinn et al., 1980 |
| Xestospongin C | Macrocyclic bis-oxaquinolizidine | Xestospongia sp./Haplosclerida | IP3-inhibitor | De Smet et al.,1999 |
| Okinonellin B | Furanosesterterpenoid | Spongionella sp./Dendroceratida | Unknown (muscle relaxant) | Kato et al., 1986 |
| Bromotopsentin | Bis-indole alkaloid | Spongosorites sp./Halichondrida | α1-Adrenergic receptor antagonist | Phife et al., 1996 |
| Penaresidin A | Azetidine alkaloid | Penares sp./Astrophorida | Actomyosin ATPase inhibitor | Kobayashi et al., 1991 |
| S1319 | Benzothiazole derivative | Dysidea sp./Dendroceratida | Unknown (antiasthmatic, uterine relaxation) | Suzuki et al., 1999 |
Dysiherbaine from Dysidea herbacea (Sakai et al., 1997) is a potent excitatory amino acid that causes seizures by interfering with the L-glutamate-based neurotransmitter communication and may provide a lead compound in therapeutical agents for neurologic disorders (Sakai et al., 2001).
Muscle Relaxants
Disturbances in neuromuscular communication resulting from stress cause permanent muscle activation (Lundberg, 1995; Edgar et al., 2002). In addition to the above-mentioned centrally acting muscle relaxants, which mediate neuromuscular communication, peripherally acting muscle relaxant may be used for local muscle relaxation. They are applied for relief of strokes, or during intubations and surgery (Frakes, 2001). 1-Methylguanosine from Tedania digitata (Quinn et al., 1980) and xestospongin C, which was isolated from a Xestospongia sp. (Gafni et al., 1997), are examples of muscle relaxants that discovered in sponges (Table 5). Xestospongin C is a potent inhibitor of the inositol 1,4,5-triphosphate (IP3) receptors and the endoplasmic-reticulum Ca2+ pumps (De Smet et al., 1999) and inhibits IP3-induced increase in the oscillatory contraction of muscles (Miyamoto et al., 2000). ß-Adrenoreceptor agonists, such as S1319 isolated from a Dysidea sp. (Suzuki et al., 1999), have utero-relaxant properties, which can be therapeutically used for the preterm delivery of infants (Dennedy et al., 2002), and are widely used as antiasthmatic drugs (Suzuki et al., 1999). However, owing to their low selectivity ß-adrenoreceptor agonists may have severe side effects such as arterial hypertension, corony heart disease, and tachycardia (Borchard, 1998). Therefore, there is continued interest in finding more selective ß-adrenoreceptor agonists such as S1319.
Antiviral Compounds
Sponges are also a rich source of compounds with antiviral properties (Table 6). The high number of HIV-inhibiting compounds discovered does not reflect greater potential of sponges to fight AIDS compared with other viral diseases, but rather the interest of many researchers. The strong focus on screening for anti-HIV activity has led to discovery of numerous compounds, but the mechanism of inhibition is still poorly characterized. Papuamides C and D (Ford et al., 1999), haplosamates A and B (Qureshi and Faulkner, 1999), and avarol (Muller et al., 1987), which has also been patented as antipsoriasis (Muller et al., 1991), are examples of HIV-inhibiting compounds from different sponges. Avarol is one of the few compounds for which the mechanism by which it inhibits progression of HIV infection is more or less known. In vitro and animal data indicate that avarol combines useful properties of an increased humoral immune response, as IgG and IgM production is significantly increased, and interference with the posttranscriptional processes of viral infection (Muller et al., 1987). Avarol inhibits HIV by almost completely blocking the synthesis of the natural UAG suppressor glutamine transfer tRNA. Synthesis of this tRNA is upregulated after viral infection, and it is important for the synthesis of a viral protease, which is necessary for viral proliferation (Muller and Schroder, 1991). Low concentrations of only 0.9 and 0.3 μM avarol resulted in 80% and 50% inhibition of virus release from infected cells, respectively (Schroder et al., 1991), while uninfected cells were highly resistant against avarol (Muller et al., 1985; Kuchino et al., 1988). Furthermore, it was shown that the avarol derivatives, 6′-hydroxy avarol and 3′-hydroxy avarone (Figure 6), were very potent inhibitors of HIV reverse transcriptase. This enzyme has a key role in the early stages of HIV infection and is a specific target for antiviral drugs, as it is responsible for converting the viral genomic RNA into proviral double-stranded DNA, which is subsequently integrated into the host chromosomal DNA (Loya and Hizi, 1990).
Table 6.
Examples of Antiviral Products from Sponges
| Compound | Compound class | Species/order | Activity | Reference |
|---|---|---|---|---|
| Dragmacidin F | Indole alkaloid | Halicortex sp./? | Antiviral | Cutignano et al., 2000 |
| Papuamides C and D | Cyclic peptide | Theonella mirabilis, T. swinhoei/Lithistida | Antiviral (HIV-1) | Ford et al., 1999 |
| Mololipids | Tyramine lipid | ?/Verongida | Antiviral (HIV-1) | Ross et al., 2000 |
| Haplosamates A and B | Sulfamated steroid | Xestospongia sp./Haplosclerida | Antiviral (HIV-1 integrase inhibitor) | Qureshi and Faulkner, 1999 |
| Hamigeran B | Phenolic macrolide | Hamigera tarangaensis/Poecilosclerida | Antiviral (herpes and polio) | Wellington et al., 2000 |
| Weinbersterols A and B | Sulfated sterol | Petrosia weinbergi/Haplosclerida | Antiviral (feline leukemia, mouse influenza, mouse corona) | Sun et al., 1991 |
| Variolin B | Pyridopyrrolopyrimidine alkaloid | Kirkpatrickia varialosa/Poecilosclerida | Antiviral | Perry et al., 1994 |
| Avarol | Hydroquinone, sesquiterpenoid | Dysidea avara/Dendroceratida | UAG suppressor glutamine tRNA inhibitora | Muller et al., 1987; Muller et al., 1991 |
| 2-5A | 2′, 5′ Linked oligonucleotide | Many sponges | Interferon mediator | Kelve et al., 2003 |
| Hennoxazole A | Bisoxazole | Polyfibrospongia sp./Dictyoceratida | Antiviral | Ichiba et al., 1991 |
aAlso has antiinflammatory potential antitumor activity.
Figure 6.
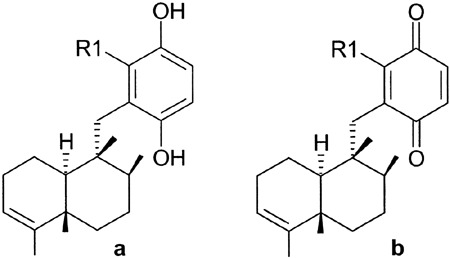
Molecular structures of avarol (a: R1 = H) and 6′-hydroxy avarol (a: R1 = OH) and avarone (b: R1 = H) and 3′-hydroxy avarone (b: R1 = OH).
In addition to their applications to treat diabetes, α-glucosidase inhibitors, such as callyspongymc acid, are potentially broad-based antiviral agents. They disturb protein glycosylation and cause some viral envelope proteins to be misfolded, which leads to arrest of these proteins within the endoplasmic reticulum, where protein folding takes place. It has been demonstrated that alteration of the glycosylation pattern of HIV, hepatitis B virus, and bovine viral diarrhea virus by α-glucosidase inhibitors attenuates viral infectivity (Ratner et al., 1991; Mehta et al., 1998).
A very different class of virus inhibitors that has been found in many different sponges are 2′-5′ oligoadenylates (2–5A), which are involved in the interferon-mediated response against a wide range of viruses in mammals. The antiviral action is based on the activation of a latent endoribonuclease that prevents viral replication by degradation of its mRNA as well as cellular RNA (Kelve et al., 2003). For many other antivirals, the mechanism of inhibition is still unclear, but they are active against range of viruses. Hamigeran B from Hamigera tarangaensis, for example, showed 100 % in vitro inhibition against both the herpes and polio viruses (Wellington et al., 2000), and the weinbersterols A and B from Petrosia weinbergi exhibited in vitro activity against feline leukemia virus, mouse influenza virus, and mouse corona virus (Sun et al., 1991; Koehn et al., 1991).
In general, antiviral molecules from sponges do not give protection against viruses, but they may result in drugs to treat already infected persons. In addition, broad-based antiviral agents such as 2-5A and α-glucosidase inhibitors may be useful in cases of sudden outbreaks of (unfamiliar) viruses like SARS and Ebola.
Antimalarial Compounds
Several sponge-derived antimalarial compounds have been discovered during the last decade (Table 7). New antimalarial drugs are needed to cope with the increasing number of multidrug-resistant Plasmodium strains that cause malaria. Plasmodium falciparum has become resistant against chloroquinone, pyrimethamine, and sulfadoxine (Bwijo et al., 2003). Kalihinol A from a Acanthella sp. (Miyaoka et al., 1998) and a number of terpenoid isocyanates, isothiocyanates, and isonitriles from Cymbastela hooperi (Konig et al., 1996) display selective in vitro antimalarial activity against P. falciparum. Also a number of free carboxylic acids from Diacarnus levii were used as precursors to yield new cyclic norditerpene peroxides after esterification. These epidioxy-substituted norditerpenes and norsesterterpenes displayed selective activity against both chloroquine-sensitive and chloroquine-resistant P. falciparum strains (D’ Ambrosio et al., 1998). The manzamines, the most promising antimalarial compound, have been discovered in a number of sponges (Sakai et al., 1986; Ang et al., 2000; Youssaf et al., 2002). It has been suggested that the antimalarial effect of manzamine A is due to an enhanced immune response (Ang et al., 2001).
Table 7.
Examples of Antimalarial Products from Sponges
| Compound | Compound class | Species/order | Reference |
|---|---|---|---|
| Axisonitrile-3 | Sesquiterpenoid isocyanide | Acanthella klethra/Halichondrida | Angerhofer et al., 1992 |
| Manzamine A | Manzamine alkaloid, diterpene isocyanates, isothiocyanates and isonitriles, norditerpenoid and norsesterterpenoid endoperoxides | e.g., Haliclona sp./Haplosclerida | Ang et al., 2001 |
| Cymbastela hooperi/Halichondrida | Konig et al., 1996 | ||
| Diacarnus levii/Poecilosclerida | D’Ambrosio et al., 1998 | ||
| Kalihinol A | Isonitril-containing kalihinane diterpenoid | Acanthella sp./Halichondrida1 | Miyaoka et al., 1998 |
Antibiotics and Fungicides
With respect to antibiotics and fungicides, similar multiresistance problems have concerned physicians for a long time. Many new molecules with antibiotic properties are discovered every year, but in marine sponges their ubiquity is remarkable (Table 8). An early screening by Burkholder and Ruetzler (1969) revealed that 18 of 31 sponges tested showed antimicrobial effects, of which some were very strong against a range of gram-positive and gram-negative bacteria. The added value of some new sponge-derived antibiotics was shown by the inhibitory effect of arenosclerins A–C from Arenosclera brasiliensis on 12 antibiotic-resistant bacteria isolated from a hospital (Torres et al., 2002). Fungicides that are currently used are less diverse than antimicrobials, and the use of many of them is restricted because of toxic effects to humans, animals, and plants (Nakagawa and Moore, 1995; Rahden-Staron, 2002). It remains to be demonstrated whether antifungals like topsentiasterols D and E from Topsentia sp. (Fusetani et al., 1994), acanthosterol sulfates I and J from an Acanthodendrilla sp. (Tsukamoto et al., 1998) or the macrolide leucascandrolide A from the calcareous sponge Leucascandra caveolata (D’Ambrosio et al., 1996) will have different characteristics than the fungicides that are currently used, but the fact that they are produced by eukaryotic organism (if not produced by a symbiont) may imply that they are less toxic to other nonfungal eukaryotes.
Table 8.
Examples of Antibacterial and Antifungal Products from Sponges
| Compound | Compound class | Species/order | Activity | Reference |
|---|---|---|---|---|
| Discodermins B, C, and D | Cyclic peptide | Discodermia kiiensis/Lithistida | Antibacterial | Matsunaga et al., 1985 |
| Topsentiasterol sulfates A–E | Sulfated sterol | Topsentia sp./Halichondrida | Antibacterial/antifungal (D and E) | Fusetani et al., 1994 |
| Arenosclerins A, B, and C | Alkylpiperidine alkaloid | Arenosclera brasiliensis /Haplosclerida | Antibacterial | Torres et al., 2002 |
| Axinellamines B–D | Imidazo-azolo-imidazole alkaloid | Axinella sp./Halichondrida | Antibacterial | Urban et al., 1999 |
| Acanthosterol I and J | Sulfated sterol | Acanthodendrilla sp./Dendroceratida | Antifungal | Tsukamoto et al., 1998 |
| Oceanapiside | Bisaminohydroxylipid glycoside | Oceanapia phillipensis/Haplosclerida | Antifungal | Nicolas et al., 1999 |
| Spongistatin | Polyether macrolide lactone | Hyrtios erecta/Dictyoceratida | Antifungal | Pettit et al., 1998 |
| Leucascandrolide A | Oxazole-containing polyether macrolide | Leucascandra caveolata/Calcarea | Antifungal | D’Ambrosio et al., 1996 |
Antifouling Compounds
A last class of bioactive compounds from marine sponges are antifouling molecules (Table 9). They are not associated with the development of new drugs, but could be environmentally friendly substitutes for chemical antifoulants. Biofouling organisms such as blue mussels, barnacles, and macroalgae cause serious problems to ship’s hulls, cooling systems of power plants, and aquaculture materials (Holmes, 1970; Houghton, 1978). Long-term use of chemical antifoulants has led to increased concentrations of tributyltin and its current replacements in coastal sediments (Konstantinou and Albanis, 2004) and to mortality and change of sex of nontarget organisms (Katranitsas et al., 2003). Natural marine antifouling molecules have recently been reviewed (Fusetani, 2004) and may provide less toxic and more specific antifouling activity. Sponge-derived antifouling molecules have been found to inhibit the settlement of barnacle larvae (Okino et al., 1995; Tsukamoto et al., 1996a, 1996b), inhibit fouling by macroalgae (Hattori et al., 1998; Kubanek et al., 2002), or repell the blue mussel Mytilus edulis galloprovincialis (Sera et al., 1999).
Table 9.
Examples of Antifouling Products from Sponges
| Compound | Compound class | Species/order | Reference |
|---|---|---|---|
| Kalihinene X | Isocyanoterpenoid | Acanthella cavernosa/Halichondrida | Okino et al., 1995 |
| Kalihipyran B | Isocyanoterpenoid | Acanthella cavernosa/Halichondrida | Okino et al., 1996 |
| 10β-Formarnidokalihinol | Isocyanoterpenoid | Acanthella cavernosa/Halichondrida | Hirota et al., 1996 |
| Pseudoceratidine 2 | Dibromopyrrole-containing spermidine derivative | Pseudoceratina purpurea/Verongida | Tsukamoto et al., 1996b |
| Ceratinamide A and B | Bromotyrosine derivative | Pseudoceratina purpurea/Verongida | Tsukamoto et al., 1996a |
| C22 ceramide | Ceramide | Haliclona koremella/Haplosclerida | Hattori et al., 1998 |
| Formoside | Striterpene glycoside, sterol diperoxide | Erylus formosus/Astrophorida | Kubanek et al., 2002 |
| Lendenfeldia chondrodes /Dictyoceratida | Sera et al., 1999 | ||
| Axinyssimides | Sesquiterpene carbonimide dichlorides | Axinyssa sp./Halichondrida | Hirota et al., 1998 |
Ecologic Role of Sponge Metabolites
Such an extensive collection of sponge-derived bioactive compounds raises the question of why sponges produce so many metabolites that can be useful to treat our human diseases. The huge number of different secondary metabolites discovered in marine sponges and the complexity of the compounds and their biosynthetic pathways (and corresponding kilobases of DNA for the programming of their synthesis) can be regarded as an indication of their importance for survival. An obvious example of the benefits of their secondary metabolites for the sponge itself, is the presence of antifouling products. To safeguard their water-pumping capacity, sponges cannot tolerate biofilm formation or settlement of barnacles or bryozoans on their surface (Proksch, 1994), The level of cytotoxicity of some sponge products is high enough to even create a bare zone around the sponge (Thompson, 1985) that is maintained by the emission of a mucus containing the toxins (Sullivan et al., 1981). This allows the conquest of densely populated rocks or corals and competition with faster growing organisms, but it is striking that the sponge can selectively use its poisons without self-destruction.
Secondary metabolites can protect the organism against predation, which is especially important for physically unprotected sessile organisms like sponges (Becerro et al., 1997). Relatively few animals, such as the hawksbill turtle and some highly evolved teleost fishes (Meylan, 1990), are largely dependent on sponges for their diet. Also some nudibranches feed on sponges and even manage to use the sponge’s metabolites for their own chemical defence (Pawlik et al., 1988). However, these spongivores represent only a tiny fraction of the animals inhabiting the seas. Secondary metabolites can also protect their producers against bacteria, fungi, or parasites (Davies, 1992). In sponges the role of the chemical constituents is clouded by the complexity of the sponge-symbiont relationship (Dumdei et al., 1998). Many different bacterial species permanently inhabit sponges and contribute considerably to the total sponge biomass (Wilkinson, 1978). It has been suggested that the growth of “useful” microorganisms may be under control of the sponge host and serve as source of food or supply other metabolic products (Muller et al., 1981). However, it has also been found that associated bacteria might be the actual producers of a number of compounds that have been isolated from sponges. Oscillatoria spongelia, a cyanobacterial symbiont that can constitute up to 40% of Dysidea herbacea, is the producer of antimicrobial polybrominated biphenyl ethers and might keep the sponge free of other bacteria (Unson, et al., 1994).
For many products it is not yet known whether they are produced by the sponge or by a symbiont. It is clear, however, that sponges are responsible for the production of a rich arsenal of “chemical weapons.” Their early appearance in evolution has given them a lot of time for the development of an advanced chemical defense system. It is interesting to note that the synthesis of secondary metabolites is regulated depending on conditions that the sponge experiences. Specimens of Crambe crambe in well-illuminated regions grow faster than their counterparts exposed to darker conditions, but the specimens in the dark are better defended as they accumulate higher concentrations of cytotoxic metabolites (Turon et al., 1998). Another example is the production of halichondrin B by Lissodendoryx sp., which varies seasonally, with depth, and with the condition of the sponge. Halichondrin B yields could be enhanced by an order of magnitude during serial cloning, suggesting a defensive response to damage (Battershill et al., 2002). The ability to stimulate the production of secondary metabolites by sponges is an important consideration when one wants to harvest compounds from sponges for the production of potential new medicines.
CONCLUSION
Marine sponges produce an enormous array of antitumor, antiviral, antiinflammatory, immunosuppressive, antibiotic, and other bioactive molecules that can affect the pathogenesis of many human diseases. The relationship between the chemical structures of the secondary metabolites from sponges and the diseases they affect is usually not obvious. Different components affect the targeted disease by different mechanisms (e.g., microtubule stabilization or interaction with DNA to combat tumors). Moreover, inhibitors of transcription may be effective against both cancer and viral diseases. To make things more complex, there are many relations between, for instance, inflammation, cancer, and viral infections via the immune system, which plays a key role in certain responses of the body to these diseases. Chronic inflammation of the lungs by cigarette smoke often leads to lung cancer (Ohwada et al., 1995) and cervical and liver cancer can follow chronic inflammation caused by papilloma viruses (Smith-McCune et al., 1996) and hepatitis B and C viruses, respectively (Zhu et al., 1997). In addition, limited activity testing (e.g., only on cell growth inhibition and not on antiviral properties) yields an incomplete overview of the actual properties of the metabolites. Finally, for many bioactive molecules from sponges, the exact mode of action and their origin (sponge or symbiont) are still unclear. Most bioactive metabolites from sponges are inhibitors of certain enzymes, which often mediate or produce mediators of intracellular or intercellular messengers involved in the pathogenesis of a disease. As this is usually a cascade of reactions inside the cell or tissue, many enzymes in the cascade are targets for potential therapy. The different enzymes in the cascade can be structurally completely different proteins; therefore, it is not surprising that a wide range of metabolites can be used for the treatment of one disease. This applies in particular to a complex disease, such as cancer, which is affected by so many different factors. Furthermore, antiviral molecules also appear to encamps a wide array of chemical structures, such as peptides, lipids, alkaloids, sterols, oligonucleotides, and a phenolic macrolide. A similar diverse pattern is observed for antibacterial and immunosuppressive metabolites. Most compounds that display antiinflammatory activity are sesterterpenoids. Nevertheless, in these cases the activity of the sponge metabolites is concentrated on certain steps; for instance, most antiinflammatory compounds act against phospholipase A2.
The potency of sponge-derived medicines lies in the fact that each of these thousands of metabolites and their derivatives has its own specific dose-related inhibitory effect, efficacy, and potential (diminished) side effects that determine its suitability for medicinal use. In addition, the skeleton or active core of these molecules may be used as a vehicle to develop derivatives with their own specific efficacy and side effects. Therefore, the most important challenge in transforming bioactive molecules into medicines is now to screen the treasure-house of sponge metabolites and select those that display a specific mode of action with the desired characteristics against a disease. An important question for the future remains how to actually prepare the potential novel drugs on a large scale.
References
- Ahond A., Bedoya Zurita M., Colin M., Laboute P., Lavelle F., Laurent D., Poupat C., Pusset J., Pusset M., Thoison O., Potier P. La girolline, nouvelle substance antitumorale extraite de l’éponge, Pseudaxinyssa cantharella n. sp. (Axinellidae) C R Acad Sci Paris 307 Series. 1988;II:145–148. [Google Scholar]
- Anderson H.J., Coleman J.E., Andersen R.J., Roberge M. Cytotoxic peptides hemiasterlin, hemiasterlin A and hemiasterlin B induce mitotic arrest and abnormal spindle formation. Cancer Chemother Pharmacol. 1997;39:223–226. doi: 10.1007/s002800050564. [DOI] [PubMed] [Google Scholar]
- Ang K.K.H., Holmes M.J., Higa T., Hamann M.T., Kara U.A.K. In vivo antimalarial activity of the β-carboline alkaloid manzamine A. Antimicrob Agents Chemother. 2000;2000:1645–1649. doi: 10.1128/AAC.44.6.1645-1649.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ang K.K.H., Holmes M.J., Kara U.A.K. Immune-mediated parasite clearance in mice infected with Plasmodium berghei following treatment with manzamine A. Parasitol Res. 2001;87:715–721. doi: 10.1007/s004360000366. [DOI] [PubMed] [Google Scholar]
- Angerhofer C.K., Pezzuto J.M., Konig G.M., Wright A.D., Stichter O. Antimalarial activity of sesquiterpenes from the marine sponge Acanthella klethra. J Nat Prod. 1992;55:1787–1789. doi: 10.1021/np50090a014. [DOI] [PubMed] [Google Scholar]
- Aoki S., Yoshioka Y., Miyamoto Y., Higuchi K., Setiawan A., Murakami N., Chen Z.-S., Sumizawa T., Akiyama S.-I., Kobayashi M. Agosterol A, a novel polyhydroxylated sterol acetate reversing multidrug resistance from a marine sponge Spongia sp. Tetrahedron Lett. 1998;39:6303–6306. doi: 10.1016/S0040-4039(98)01336-7. [DOI] [Google Scholar]
- Arimoto H., Hayakawa I., Kuramoto M., Uemura D. Absolute stereochemistry of halichlorine; a potent inhibitor of VCAM-1 induction. Tetrahedron Lett. 1998;39:861–862. doi: 10.1016/S0040-4039(97)10714-6. [DOI] [Google Scholar]
- Arndt, W. (1938) “Schwamme” In: arndt, W., Pax, F. (eds.), Die Rohstoffe des Tierreichs 1, 2 Hälfte, Gebr, Borntraeger, Berlin, pp 1577-2000
- Bai R.L., Paull K.D., Herald C.L., Malspeis L., Pettit G.R., Hamel E. Halichondrin B and homohalichondrin B, marine natural products binding in the vinca domain of tubulin: discovery of tubulin-based mechanism of action by analysis of differential cytotoxicity data. J Biol Chem. 1991;266:15882–15889. [PubMed] [Google Scholar]
- Bai R., Cichacz Z.A., Herald C.L., Pettit G.R., Hamel E. Spongistatin 1, a highly cytotoxic, sponge-derived, marine natural product that inhibits mitosis, microtubule assembly, and the binding of vinblastine to tubulin. Mol Pharmacol. 1993;44:757–766. [PubMed] [Google Scholar]
- Battershill C.N., Page M.J., Munro M.H.G. A chemical ecology of sponges in culture. Boll Mus 1st Biol Univ Genova. 2002;66–67:23. [Google Scholar]
- Becerro M.A., Turon X., Uriz M.J. Multiple functions for secondary metabolites in encrusting marine invertebrates. J Chem Ecol. 1997;23:1527–1547. doi: 10.1023/B:JOEC.0000006420.04002.2e. [DOI] [Google Scholar]
- Bennet C.F., Mong S., Clark M.A., Kruse L.J., Crooke S.T. Differential effects of manoalide on secreted intracellular phospholipases. Biochem Pharmacol. 1987;36:2079–2086. doi: 10.1016/0006-2952(87)90134-1. [DOI] [PubMed] [Google Scholar]
- Bergmann W., Feeney R.J. The isolation of a new thymine pentoside from sponges. J Am Chem Soc. 1950;72:2809–2810. doi: 10.1021/ja01162a543. [DOI] [Google Scholar]
- Bergmann W., Feeney R.J. Contributions to the study of marine products, 32: the nucleosides of sponges, I. J Org Chem. 1951;16:981–987. doi: 10.1021/jo01146a023. [DOI] [Google Scholar]
- Bergmann W., Swift A.N. Contributions to the study of marine products, 30: Component acids of lipids of sponges, I. J Org Chem. 1951;16:1206–1221. doi: 10.1021/jo50002a005. [DOI] [Google Scholar]
- Berlinck R.G.S., Braekman J.C., Daloze D., Bruno I., Riccio R., Ferri S., Spampinato S., Speroni E. Polycyclic guanidine alkaloids from the marine sponge Crambe crambe and Ca++ channel blocker activity of crambescidin 816. J Nat Prod. 1993;56:1007–1015. doi: 10.1021/np50097a004. [DOI] [PubMed] [Google Scholar]
- Bjarnason I., Hayllar J., Macpherson A.J., Russell A.S. Side effects of nonsteroidal anti-inflammatory drugs on the small and large intestine in humans. Gastroenterology. 1993;104:1832–1847. doi: 10.1016/0016-5085(93)90667-2. [DOI] [PubMed] [Google Scholar]
- Blackburn C.L., Hopmann C., Sakowicz R., Berdelis M.S., Goldstein L.S.B., Faulkner D.J. Adociasulfates 1–6, inhibitors of kinesin motor proteins from the sponge Haliclona (aka Adocia) sp. J Org Chem. 1999;64:5565–5570. doi: 10.1021/jo9824448. [DOI] [PubMed] [Google Scholar]
- Borchard U. Pharmacological properties of β-adrenoreceptor blocking drugs. J Clin Bas Cardiol. 1998;1:5–9. [Google Scholar]
- Bowman E.J., Gustafson K.R., Bowman B.J., Boyd M.R. Identification of a new chondropsin class of antitumor compound that selectively inhibits V-ATPases. J Biol Chem. 2003;278:44147–44152. doi: 10.1074/jbc.M306595200. [DOI] [PubMed] [Google Scholar]
- Bradshaw D., Hill C.H., Nixon J.S., Wilkinson S.E. Therapeutic potential of protein kinase C inhibitors. Agents Actions. 1993;35:135–147. doi: 10.1007/BF02027225. [DOI] [PubMed] [Google Scholar]
- Bramley A.M., Langlands J.M., Jones A.K., Burgoyne D.L., Li Y., Andersen R.J., Salari H. Effects of IZP-94005 (contignasterol) on antigen-induced bronchial responsiveness in ovalbumin-sensitized guinea-pigs. Br J Pharmacol. 1995;115:1433–1438. doi: 10.1111/j.1476-5381.1995.tb16634.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bubb M.R., Spector I., Bershadsky A.D., Korn E.D. Swinholide A is a microfilament disrupting marine toxin that stabilizes actin dimers and severs actin filaments. J Biol Chem. 1995;270:3463–3466. doi: 10.1074/jbc.270.8.3463. [DOI] [PubMed] [Google Scholar]
- Burgoyne D.L., Andersen R.J. Contignasterol, a highly oxygenated steroid with the ‘unnatural’ 14β configuration from the marine sponge Petrosia contignata Thiele, 1899. J Org Chem. 1992;57:525–528. doi: 10.1021/jo00028a024. [DOI] [Google Scholar]
- Burkholder P.R., Ruetzler K. Antimicrobial activity of some marine sponges. Nature. 1969;222:983–984. doi: 10.1038/222983a0. [DOI] [PubMed] [Google Scholar]
- Burres N.S., Clement J.J. Antitumor activity and the mechanism of action of the novel marine natural products mycalamide-A and -B and onnamide. Cancer Res. 1989;49:2935–2940. [PubMed] [Google Scholar]
- Bwijo B., Kaneko A., Takechi M., Zungu I.L., Moriyama Y., Lum J.K., Tsukahara T., Mita T., Takahashi N., Bergqvist Y., Björkman A., Kobayakawa T. High prevalence of quintuple mutant dhpsldhfr genes in Plasmodium falciparum infections seven years after introduction of sulfadoxine and pyrimethamine as first line treatment in Malawi. Acta Tropica. 2003;85:363–373. doi: 10.1016/S0001-706X(02)00264-4. [DOI] [PubMed] [Google Scholar]
- Cantrell C.L., Gustafson K.R., Cecere M.R., Pannell L.K., Boyd M.R. Chondropsins A and B: novel tumor cell growth-inhibitory macrolide lactams from the marine sponge Chondropsis sp. J Am Chem Soc. 2000;122:8825–8829. doi: 10.1021/ja0010711. [DOI] [Google Scholar]
- Carroll J., Johnsson E.N., Ebel R., Hartman M.S., Holman T.R., Crews P. Probing sponge-derived terpenoids for human 15-L-lipoxygenase inhibitors. J Org Chem. 2001;66:6847–6851. doi: 10.1021/jo015784t. [DOI] [PubMed] [Google Scholar]
- Casapullo A., Minale L., Zollo F. New cytotoxic polyoxygenated steroids from the sponge Dysidea incrustans. Tetrahedron Lett. 1995;36:2669–2672. doi: 10.1016/0040-4039(95)00329-B. [DOI] [Google Scholar]
- Chackalamannil S. Thrombin receptor antagonists as nove therapeutic agents. Curr Opin Drug Discov Dev. 2001;4:417–427. [PubMed] [Google Scholar]
- Cimino G., De De Stefano S., Minale L., Fattorusso Ircinin 1 and 2, linear sesterterpenes from the marine sponge Ircinia oros. Tetrahedron. 1972;28:333–341. doi: 10.1016/0040-4020(72)80140-6. [DOI] [Google Scholar]
- Colson G., Rabault B., Lavelle F., Zerial A. Mode of action of the antitumor compound girodazole (RP 49532A, NSC 627434) Biochem Pharmacol. 1992;43:1717–1723. doi: 10.1016/0006-2952(92)90701-J. [DOI] [PubMed] [Google Scholar]
- Costantino V., Fattorusso E., Mangoni A., Di Rosa M., Ianaro A. Glycolipids from sponges, VII: simplexides, novel immunosuppressive glycolipids from the Caribbean sponge Plakortis simplex. Bioorg Med Chem Lett. 1999;9:271–276. doi: 10.1016/S0960-894X(98)00719-7. [DOI] [PubMed] [Google Scholar]
- Coue M., Brenner S.L., Spector I., Korn E.D. Inhibition of actin polymerization by latrunculin A. FEBS Lett. 1987;213:316–318. doi: 10.1016/0014-5793(87)81513-2. [DOI] [PubMed] [Google Scholar]
- Cutignano A., Bifulco G., Bruno I., Casapullo A., Gomez-Paloma L., Riccio R. Dragmacidin F: a new antiviral bromoindole alkaloid from the Mediterranean sponge Halicortex sp. Tetrahedron. 2000;56:3743–3748. doi: 10.1016/S0040-4020(00)00281-7. [DOI] [Google Scholar]
- D’Ambrosio M., Guerriero A., Debitus C., Pietra F. Leucascandrolide A, a new type of macrolide: the first powerfully bioactive metabolite of calcareous sponges (Leucascandra caveolata, a new genus from the coral sea) Helv Chim Acta. 1996;79:51–60. doi: 10.1002/hlca.19960790107. [DOI] [Google Scholar]
- D’Ambrosio M., Guerriero A., Deharo E., Debitus C., Munoz V., Pietra F. New types of potentially antimalarial agents: epidioxy-substituted norditerpene and norsesterpenes from the marine sponge Diacarnuslevii. Helv Chim Acta. 1998;81:1285–1292. doi: 10.1002/hlca.19980810539. [DOI] [Google Scholar]
- Davies, J. (1992) “Introduction” In: Chadwick, D.J. , Whelan, J. (eds.), Secondary Metabolites: Their Function and Evolution, Wiley, Chichester, U.K., pp 1-2
- De De Carvalho M.S., Jacobs R.S. Two-step inactivation of bee venom phospholipase A2 by scalaradial. Biochem Pharmacol. 1991;42:1621–1626. doi: 10.1016/0006-2952(91)90432-5. [DOI] [PubMed] [Google Scholar]
- De De Guzman F.S., Carte B., Troupe N., Faulkner D.J., Harper M.K., Conception G.P., Mangalindan G.C., Matsumoto S.S., Barrows L.R., Ireland C.M. Neoamphimedine: a new pyridoacridine topoisomerase II inhibitor which catenates DNA. J Org Chem. 1999;64:1400–1402. doi: 10.1021/jo982047x. [DOI] [Google Scholar]
- De De Marino S., Iorizzi M., Zollo F., Debitus C., Menou J.-L., Ospina L.F., Alcaraz M.J., Paya M. New pyridinium alkaloids from a marine sponge of the genus Spongia with a human phospholipase A2 inhibitor profile. J Nat Prod. 2000;63:322–326. doi: 10.1021/np990374+. [DOI] [PubMed] [Google Scholar]
- Dennedy M.C., Houlihan D.D., McMillan H., Morrison J.J. β2− and β3−Adrenoreceptor agonists: human myometrial selectivity and effects on umbilical artery tone. Am J Obstet Gynecol. 2002;187:641–647. doi: 10.1067/mob.2002.125277. [DOI] [PubMed] [Google Scholar]
- De Rosa, S. (2002) “Mediterranean marine organisms as source of new potential drugs” In: Rauter, A., Palma, F.B., Justino, J., Araujo, M.E., Santos, S.P. (eds.), Natural Products in the New Millennium: Prospects and Industrial Applications, Kluwer Academic Publishers, The Netherlands, pp 441-461
- De De Silva E.D., Scheuer P.J. Manoalide, an antibiotic sesterterpenoid from the marine sponge Luffariella variabilis. Tetrahedron Lett. 1980;21:1611–1614. doi: 10.1016/S0040-4039(00)77766-5. [DOI] [Google Scholar]
- De De Smet P., Parys J.B., Callewaert G., Weidema A.F., Hill E., De De Smedt H., Erneux C., Sorrentino V., Missiaen L. Xestospongin C is an equally potent inhibitor of the inositol 1,4,5-triphosphate receptor and the endoplasmic-reticulum Ca2+ pumps. Cell Calcium. 1999;26:9–13. doi: 10.1054/ceca.1999.0047. [DOI] [PubMed] [Google Scholar]
- Dumdei, E.J., Blunt, J.W., Munro, M.H.G., Battershill, C.N., Page, M.J. (1998) “The whys and whats of sponge chemistry: why chemists extract sponges and what problems does this cause?” In: Watanabe, Y., Fusetani, N. (eds.), Sponge Sciences; Multidisciplinary Perspectives, Springer-verlag, Tokyo, Japan, pp 353-364
- Dunbar D.C., Rimoldi J.M., Clark A.M., Kelly M., Hamann M.T. Anti-cryptococcal and nitric oxide synthase inhibitory imidazole alkaloids from the calcareous sponge Leucetta cf. chagosensis. Tetrahedron. 2000;56:8795–8798. doi: 10.1016/S0040-4020(00)00821-8. [DOI] [Google Scholar]
- Edgar V.A., Cremaschi G.A., Sterin-Borda L., Genaro A.M. Altered expression of autonomic neurotransmitter receptors and proliferative responses in lymphocytes from a chronic mild stress model of depression: effects of fluoxetine. Brain Behav Immun. 2002;16:333–350. doi: 10.1006/brbi.2001.0632. [DOI] [PubMed] [Google Scholar]
- Erickson K.L., Beutler J.A., Cardellina J.H., II, Boyd M.R. Salicylihalamides A and B, novel cytotoxic macrolides from the marine sponge Haliclona sp. J Org Chem. 1997;62:8188–8192. doi: 10.1021/jo971556g. [DOI] [PubMed] [Google Scholar]
- Fabian I., Halperin D., Lefter S., Mittelman L., Altstock R.T., Season O., Tsarfaty I. Alteration of actin organisation by jaspamide inhibits ruffling, but not phagocytosis or oxidative burst, in HL-60 cells and human monocytes. Blood. 1999;93:3994–4005. [PubMed] [Google Scholar]
- Fattorusso E., Taglialatela-Scafati O. Two novel pyrrole-imidazole alkaloids from the Mediterranean sponge Agelas oroides. Tetrahedron Lett. 2000;41:9917–9922. doi: 10.1016/S0040-4039(00)01764-0. [DOI] [Google Scholar]
- Faulkner D.J. Marine natural products. Nat Prod Rep. 2000;17:7–55. doi: 10.1039/a809395d. [DOI] [PubMed] [Google Scholar]
- Faulkner D.J. Marine natural products. Nat Prod Rep. 2001;18:149. doi: 10.1039/b006897g. [DOI] [PubMed] [Google Scholar]
- Faulkner D.J. Marine natural products. Nat Prod Rep. 2002;19:1–48. doi: 10.1039/b009029h. [DOI] [PubMed] [Google Scholar]
- Fedoreev S.A., Prokof’eva N.G., Denisenko V.A., Rebachuk N.M. Cytotoxic activity of aaptamines from suberitid marine sponges. Pharm Chem J. 1989;22:615–618. doi: 10.1007/BF00763625. [DOI] [Google Scholar]
- Ford P.W., Gustafson K.R., McKee T.C., Shigematsu N., Maurizi L.K., Pannell L.K., Williams D.E., De De Silva E.D., Lassota P., Alien T.M., Van Van Soest R., Andersen R.J., Boyd M.R. Papuamides A–D, HIV-inhibitory and cytotoxic depsipeptides from the sponges Theonella mirabilis and Theonella swinhoei collected in Papua New Guinea. J Am Chem Soc. 1999;121:5899–5909. doi: 10.1021/ja990582o. [DOI] [Google Scholar]
- Frakes M.A. Muscle relaxant choices for rapid sequence induction. Air Med J. 2001;20:20–21. doi: 10.1067/mmj.2001.112417. [DOI] [PubMed] [Google Scholar]
- Fukuoka K., Yamagishi T., Ichihara T., Nakaike S., Iguchi K., Yamada Y., Fukumoto H., Yoneda T., Samata K., Ikeya H., Nanaumi K., Hirayama N., Narita N., Saijo N., Nishio K. Mechanism of action of aragusterol A (YTA0040), a potent anti-tumor marine steroid targeting the G1 phase of the cell cycle. Int J Cancer. 2000;88:810–819. doi: 10.1002/1097-0215(20001201)88:5<810::AID-IJC20>3.0.CO;2-P. [DOI] [PubMed] [Google Scholar]
- Fusetani N. Biofouling and antifouling. Nat Prod Rep. 2004;21:94–104. doi: 10.1039/b302231p. [DOI] [PubMed] [Google Scholar]
- Fusetani N., Yasumuro K., Matsunaga S., Hashimoto K. Mycalolides A–C, hybrid macrolides of ulapualides and halichondramide, from a sponge of the genus Mycale. Tetrahedron Lett. 1989;30:2809–2812. doi: 10.1016/S0040-4039(00)99131-7. [DOI] [Google Scholar]
- Fusetani N., Matsunaga S., Matsumoto H., Takebayashi Y. Cyclotheonamides, potent thrombin inhibitors, from a marine sponge Theonella sp. J Am Chem Soc. 1990;112:7053–7054. doi: 10.1021/ja00175a045. [DOI] [Google Scholar]
- Fusetani N., Takahashi M., Matsunaga S. Topsentiasterol sulfates, antimicrobial sterol sulfates possessing novel side chains, from a marine sponge, Topsentia sp. Tetrahedron. 1994;50:7765–7770. doi: 10.1016/S0040-4020(01)85260-1. [DOI] [Google Scholar]
- Gafni J., Munsch J.A., Lam T.H. Xestospongins: potent membrane permeable blockers of the inositol 1,4,5-triphosphate receptor. Neuron. 1997;19:723–733. doi: 10.1016/S0896-6273(00)80384-0. [DOI] [PubMed] [Google Scholar]
- Garcia-Colunga J., Miledi R. Serotonergic modulation of muscle acetylcholine receptors of different subunit composition. Proc Natl Acad Sci U S A. 1996;93:3990–3994. doi: 10.1073/pnas.93.9.3990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Garcia Pastor P., De De Rosa S., De De Giulio A., Payá M., Alcaraz M.J. Modulation of acute and chronic inflammatory processes by cacospongionolide B, a novel inhibitor of human synovial phospholipase A2. Br J Pharmacol. 1999;126:301–311. doi: 10.1038/sj.bjp.0702302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geisler J. Breast cancer tissue estrogens and their manipulation with aromatase inhibitors and inactivators. J Steroid Biochem Mol Biol. 2003;86:245–253. doi: 10.1016/S0960-0760(03)00364-9. [DOI] [PubMed] [Google Scholar]
- Giannini C., Debitus C., Posadas I., Paya M., D’Auria M.V. Dysidotronic acid, a new and selective human phospholipase A2 inhibitor from the sponge Dysidea sp. Tetrahedron Lett. 2000;41:3257–3260. doi: 10.1016/S0040-4039(00)00362-2. [DOI] [Google Scholar]
- Glaser K.B., De De Carvalho M.S., Jacobs R.S., Kernan M.R., Faulkner D.J. Manoalide: structure-activity studies and definition of the pharmacophore for phospholipase A2 inactivation. Mol Phys. 1989;36:782–788. [PubMed] [Google Scholar]
- Griffith, O.W, Gross, S.S. (1996) “Inhibitors of nitric oxide synthases” In: Stamler, J., Feelish, M. (eds.), Methods in Nitric Oxide Research, Wiley & Sons, New York, N.Y., pp 187-208
- Hattori T., Adachi K., Shizuri Y. New ceramide from marine sponge Haliclona koremella and related compounds as antifouling substances against macroalgae. J Nat Prod. 1998;61:823–826. doi: 10.1021/np970527y. [DOI] [PubMed] [Google Scholar]
- Hirata Y., Uemura D. Halichondrins — antitumor polyether macrolides from a marine sponge. Pure Appl Chem. 1986;58:701–710. [Google Scholar]
- Hirota H., Tomono Y., Fusetani N. Terpenoids with antifouling activity against barnacle larvae from the marine sponge Acanthella cavernosa. Tetrahedron. 1996;52:2359–2368. doi: 10.1016/0040-4020(95)01079-3. [DOI] [Google Scholar]
- Hirota H., Okino T., Yoshimura E., Fusetani N. Five new antifouling sesquiterpenes from two marine sponges of the genus Axinysssa and the nudibranch Phyllidia pustulosa. Tetrahedron. 1998;54:1397–13980. doi: 10.1016/S0040-4020(98)00867-9. [DOI] [Google Scholar]
- Hofrichter, R., Sidri, M. (2001) “Ein Mittel fur jeden Zweck: der Badeschwamm” In: Hofrichter, R. (ed.), Das Mittelmeer. Flora, Fauna, Ökologie, Spektrum Verlag, Bd. 1, pp 608-809
- Holland H.L., Kumaresan S., Tan L., Njar V.C.O. Synthesis of 6-hydroximino-3-oxo steroids, a new class of aromatase inhibitor. J Chem Soc Perkin Trans. 1992;1:585–587. doi: 10.1039/p19920000585. [DOI] [Google Scholar]
- Holmes N. Marine fouling in power stations. Mar Pollut Bull. 1970;1:105–106. doi: 10.1016/0025-326X(70)90217-1. [DOI] [Google Scholar]
- Hood K.A., West L.M., Rouwé B., Northocote P.T., Berridge M.V., Wakefield S.J., Miller J.H. Peloruside A, a novel antimitotic agent with paclitaxel-like microtabule-stabilizing activity. Cancer Res. 2002;62:3356–3360. [PubMed] [Google Scholar]
- Houghton D.R. Marine fouling and offshore structures. Ocean Manage. 1978;4:347–352. doi: 10.1016/0302-184X(78)90033-1. [DOI] [Google Scholar]
- Ichiba T., Yoshida W.Y., Scheuer P.J., Higa T. Hennoxazoles, bioactive bisoxazoles from a marine sponge. J Am Chem Soc. 1991;113:3173–3174. doi: 10.1021/ja00008a056. [DOI] [Google Scholar]
- Iguchi K., Fujita M., Nagaoka H., Mitome H., Yamada Y. Aragusterol A: a potent antitumor marine steroid from the Okinawan sponge of the genus, Xestospongia. Tetrahedron Lett. 1993;34:6277–6280. doi: 10.1016/S0040-4039(00)73731-2. [DOI] [Google Scholar]
- Inaba K., Sato H., Tsuda M., Kobayashi J. Spongiacidins A–D, new bromopyrrole alkaloids from Hymeniacidon sponge. J Nat Prod. 1998;61:693–695. doi: 10.1021/np970565h. [DOI] [PubMed] [Google Scholar]
- Isbrucker R.A., Cummins J., Pomponi S.A., Longley R.E., Wright A.E. Tubulin polymerizing activity of dictyostatin 1, a polyketide of marine sponge origin. Biochem Pharmacol. 2003;66:75–82. doi: 10.1016/S0006-2952(03)00192-8. [DOI] [PubMed] [Google Scholar]
- Jacobs, R.S., Koehn, F.E., Gunasekera, S.P. (1994). Topsentin, a unique phosphohpase A2 inhibitor [abstract]. Presented at the Japan–U.S. Seminar on Bioorganic Marine Chemistry
- Jares-Erijman E.A., Sakai R., Rinehart K.L. Crambescidins: new antiviral and cytotoxic compounds from the sponge Crambe crambe. J. Ore Chem. 1991;56:5712–5715. doi: 10.1021/jo00019a049. [DOI] [Google Scholar]
- Juagdan E.G., Kalindindi R.S., Scheuer P.J., Kelly-Borges M. Elenic acid, an inhibitor of topoisomerase II, from a sponge, Plakinastrella sp. Tetrahedron Lett. 1995;36:2905–2908. doi: 10.1016/0040-4039(95)00432-C. [DOI] [Google Scholar]
- Kashman Y., Groweiss A., Shmueli U. Latruncutin, a new 2-thiazolidinone macrolide from the marine sponge Latrunculia magnifica. Tetrahedron Lett. 1980;21:3629–3632. doi: 10.1016/0040-4039(80)80255-3. [DOI] [Google Scholar]
- Kato Y., Fusetani N., Matsunaga S., Hashimoto K. Okinonellins A and B, two novel furanosesterterpenes, which inhibit cell division of fertilized starfish eggs, from the marine sponge Spongionella sp. Experientia. 1986;42:1299–1300. doi: 10.1007/BF01946432. [DOI] [Google Scholar]
- Katranitsas A., Castritsi-Catharios J., Persoone G. The effects of a copper-based antifouling paint on mortality and enzymatic activity of a non-target marine organism. Mar Pollut Bull. 2003;46:1491–1494. doi: 10.1016/S0025-326X(03)00253-4. [DOI] [PubMed] [Google Scholar]
- Kelve M., Kuusksalu A., Lopp A., Reintamm T. Sponge (2’,5’)oligoadenylate synthetase activity in the whole sponge organism and in a primary cell culture. J Biotechnol. 2003;100:177–180. doi: 10.1016/S0168-1656(02)00254-7. [DOI] [PubMed] [Google Scholar]
- Kitagawa I., Kobayashi M., Kitanaka K., Kido M., Kyogoku Marine natural products, XII: on the chemical constituents of the Okinawan marine sponge Hymeniacidon aldis. Chem Pharm Bull. 1983;31:2321–2328. [Google Scholar]
- Kobayashi J., Cheng J.F., Ishibashi M., Walchli M.R., Yamamura S., Ohizumi Y. Penaresidin A and B, two novel azetidine alkaloids with potent actomyosin ATPase activating activity from the Okinawan marine sponge Penares sp. J Chem Soc Perkin Trans. 1991;1:1135–1138. doi: 10.1039/p19910001135. [DOI] [Google Scholar]
- Kobayashi E., Motoki K., Uchida T., Fukushima H., Koezuka Y. KRN7000, a novel immunomodulator, and its antitumor activity. Oncol Res. 1995;7:529–534. [PubMed] [Google Scholar]
- Kobayashi M., Higuchi K., Murakami N., Tajima H., Aoki S. Callystatin A, a potent cytotoxic polyketide from the marine sponge, Callyspongia truncata. Tetrahedron Lett. 1997;38:2859–2862. doi: 10.1016/S0040-4039(97)00482-6. [DOI] [Google Scholar]
- Koehn F.E., Gunasekera M., Cross S.S. New antiviral sterol disulfate ortho esters from the marine sponge Petrosia weinbergi. J Org Chem. 1991;56:1322–1325. doi: 10.1021/jo00003a080. [DOI] [Google Scholar]
- Koiso Y., Morita K., Kobayashi M., Wang W., Ohyabu N., Iwasaki S. Effects of arenastatin A and its synthetic analogs on microtubule assembly. Chemico-Biol Interact. 1996;102:183–191. doi: 10.1016/S0009-2797(96)03743-X. [DOI] [PubMed] [Google Scholar]
- Konig G.M., Wright A.D., Angerhofer C.K. Novel potent antimalarial diterpene isocyanates, isothiocyanates, and isonitriles from the tropical marine sponge Cymbastela hooperi. J Org Chem. 1996;61:3259–3267. doi: 10.1021/jo952015z. [DOI] [Google Scholar]
- Konstantinou I.K., Albanis T.A. Worldwide occurrence and effects of anifouling paint booster biocides in the aquatic environment: a review. Environment Int. 2004;30:235–248. doi: 10.1016/S0160-4120(03)00176-4. [DOI] [PubMed] [Google Scholar]
- Kubanek J., Whalen K.E., Engel S., Kelly S.R., Henkel T.P., Fenical W., Pawfik J.R. Multiple defensive roles for triterpene glycosides from two Carribean sponges. Oecologia. 2002;1:125–136. doi: 10.1007/s00442-001-0853-9. [DOI] [PubMed] [Google Scholar]
- Kuchino Y., Nishimura S., Schroder H.C., Rottmann M., Müller W.E.G. Selective inhibition of formation of suppressor glutamine tRNA in Moloney murine leukemia virus–infected NIH-3T3 cells by avarol. Virology. 1988;165:518–526. doi: 10.1016/0042-6822(88)90596-X. [DOI] [PubMed] [Google Scholar]
- Kuramoto M., Tong C., Yamada K., Chiba T., Hayashi Y., Uemura D. Halichlorine, an inhibitor of VCAM-1 induction from the marine sponge Halichondria okadai Kadata. Tetrahedron Lett. 1996;37:3867–3870. doi: 10.1016/0040-4039(96)00703-4. [DOI] [Google Scholar]
- Lala P.K., Oracevic A. Role of nitric oxide in tumor progression: lessons from experimental tumors. Cancer Metastasis Rev. 1998;17:91–106. doi: 10.1023/A:1005960822365. [DOI] [PubMed] [Google Scholar]
- Lang I.M., Marvig J. Functional localization of specific receptors mediating gastrointestinal motor correlates of vomiting. Am J Physiol Gastrointest Liver Physiol. 1989;256:G92–G99. doi: 10.1152/ajpgi.1989.256.1.G92. [DOI] [PubMed] [Google Scholar]
- Langlands J.M., Hennan J.K., Bramley A.M., Pendleton N., Burgoyne D.L., Andersen R.J. Effects of IZP-94005 on eosinophil number and eosinophul peroxidase activity in lung lavage fluid from sensitized guenea pigs. Am J Respir Crit Care Med. 1995;151:A700. [Google Scholar]
- Lebovitz H.E. Oral antidiabetic agents: the emergence of α-glucosidase inhibitors. Drugs. 1992;44:21–28. doi: 10.2165/00003495-199200443-00004. [DOI] [PubMed] [Google Scholar]
- de de Leone P. A., Redburn J., Hooper J.N.A., Quinn R.J. Polyoxygenated Dysidea sterols that inhibit the binding of [I125] IL-8 to the human recombinant IL-8 receptor type A. J Nat Prod. 2000;63:694–697. doi: 10.1021/np9904657. [DOI] [PubMed] [Google Scholar]
- Liu B., Timar J., Howlett J., Diglio C.A., Honn K.V. Lipoxygenase metabolites of arachidonic and linoleic acids modulate the adhesion of tumor cells to endothelium via regulation of protein kinase C. Cell Regul. 1991;2:1045–1055. doi: 10.1091/mbc.2.12.1045. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu L.F., Chen A.Y. DNA topoisomerases: essential enzymes and lethal targets. Annu Rev Pharmacol Toxicol. 1994;34:191–218. doi: 10.1146/annurev.pa.34.040194.001203. [DOI] [PubMed] [Google Scholar]
- Lonning P.E., Geisler J., Bhatnager A. Development of aromatase inhibitors and their pharmacologic profile. Am J Clin Oncol. 2003;26:S3–S8. doi: 10.1097/00000421-200308001-00002. [DOI] [PubMed] [Google Scholar]
- Loya S., Hizi A. The inhibition of human immunodeficiency virus type 1 reverse transcriptase by avarol and avarone derivatives. FEBS. 1990;269:131–134. doi: 10.1016/0014-5793(90)81137-D. [DOI] [PubMed] [Google Scholar]
- Lundberg U. Methods and applications of stress research. Technol Health Care. 1995;3:3–9. [PubMed] [Google Scholar]
- MarinLit (1999). A marine literature database maintained by the Marine Chemistry Group, University of Canterbury, Christchurch, New Zealand
- Martinez-Zaguilan R., Raghunand N., Lynch R.M., Bellamy W., Martinez G.M., Rojas B., Smith D., Dalton W.S., Gillies R.J. pH and drug resistance, I: functional expression of plasmalemmal V-type H+-ATPase in drug-resistant human breast carcinoma cell lines. Biochem Pharmacol. 1999;57:1037–1046. doi: 10.1016/S0006-2952(99)00022-2. [DOI] [PubMed] [Google Scholar]
- Maryanoff B.E., Qiu X., Padmanabhan K.P., Tulinsky A., Almond H.R., Andrade-Gordon P., Greco M.N., Kauffman J.A., Nicolaou Liu KC A., Brungs P.H., Fusetani N. Molecular basis for the inhibition of human α-thrombin by the macrocyclic peptide cyclotheonamide A. Proc Natl Acad Sci U S A. 1993;90:8048–8052. doi: 10.1073/pnas.90.17.8048. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Matsunaga S., Fusetani N., Konosu S. Bioactive marine metabolites, VII: structures of discodermins B, C, and D, antimicrobial peptides from the marine sponge Discodermia kiiensis. Tetrahedron Lett. 1985;26:855–856. doi: 10.1016/S0040-4039(00)61947-0. [DOI] [PubMed] [Google Scholar]
- Mayer A.M.S., Jacobs R.S. Manoalide: an antiinflammatory and analgesic marine natural product. Memoirs Calif Acad Sci. 1988;13:133. [Google Scholar]
- Mehta A., Zitzmann N., Rudd P.M., Block T.M., Dwek R.A. α-Glucosidase inhibitors as potential broad based anti-viral agents. FEBS Lett. 1998;430:17–22. doi: 10.1016/S0014-5793(98)00525-0. [DOI] [PubMed] [Google Scholar]
- Meylan, A. (1990) “Nutritional characteristics of the sponges in the diet of the hawksbill turtle” In: Rützler, K. (ed.), New Perspectives in Sponge Biology, Institution Press, Washington, D.C.: Smithsonian, pp 472-477
- Miyamoto S., Izumi M., Hori M., Kobayashi M., Ozaki H., Karaki H. Xestospongin C, a selective and membrane-permeable inhibitor of IP3 receptor, attenuates the positive inotropic effect of α-adrenergic stimulation in guinea-pig papillary muscle. Br J Pharmacol. 2000;130:650–654. doi: 10.1038/sj.bjp.0703358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyaoka H., Shimomura M., Kimura H., Yamada Y., Kim H.-S., Wataya Y. Antimalarial activity of kalahinol A and new relative diterpenoids from the Okinawan sponge, Acanthella sp. Tetrahedron. 1998;54:13467–13474. doi: 10.1016/S0040-4020(98)00818-7. [DOI] [Google Scholar]
- Mooberry S.L., Tien G., Hernandez A.H., Plubrukarn A., Davidson B.S. Laulimalide and isolaulimalide, new paclitaxel-like microtubule-stabilizing agents. Cancer Res. 1999;59:653–660. [PubMed] [Google Scholar]
- Muller W.E.G., Schroder H.C. Cell biological aspects of HIV-1 infection: effects of the anti-HIV-1 agent avarol. Int J Sports Med. 1991;12:S43–S49. doi: 10.1055/s-2007-1024749. [DOI] [PubMed] [Google Scholar]
- Muller W.E.G., Zahn R.K., Kurelec B., Lucu C., Muller I., Uhlenbruck G. Lectin, a possible basis for symbiosis between bacteria and sponges. J Bacterial. 1981;145:548–558. doi: 10.1128/jb.145.1.548-558.1981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Muller W.E.G., Maidhof A., Zahn R.K., Schroder H.C., Gasic M.J., Heidemann D., Bernd A., Kurelec B., Eich E., Seibert G. Potent antileukemic activity of the novel cytostatic agent avarone and its analogues in vitro and in vivo. Cancer Res. 1985;45:4822–4826. [PubMed] [Google Scholar]
- Muller W.E.G., Sobel C., Diehl-Seifert B., Maidhof A., Schroder H.C. Influence of the antileukemic and anti-human immunodeficiency virus agent avarol on selected immune responses in vitro and in vivo. Biochem Pharmacol. 1987;36:1489–1494. doi: 10.1016/0006-2952(87)90115-8. [DOI] [PubMed] [Google Scholar]
- Muller, W.E.G., Schatton, W.F.H., Gudrum, M. (1991). Verwendung von avarol oder dessen derivaten zur bekämpfung von entzündlichen systemischen und dermatologischen erkrankungen. Patent Application DE 1991-4137093
- Nagayama H., Hingtgen J.N., Aprison M.H. Pre- and postsynaptic serotonergic manipulations in an animal model of depression. Pharmacol Biochem Behav. 1980;13:575–579. doi: 10.1016/0091-3057(80)90283-X. [DOI] [PubMed] [Google Scholar]
- Nakagawa Y., Moore G.A. Cytotoxic effects of postharvest fungicides, ortho-phenylphenol, thiabendazole and imazalil, on isolated rat hepatocytes. Life Sci. 1995;57:1433–1440. doi: 10.1016/0024-3205(95)02106-S. [DOI] [PubMed] [Google Scholar]
- Nakamura H., Ohizumi Y., Kaboyashi J. Keramadine, a novel antagonist of serotonergic receptors isolated from the Okinawan sea sponge Agelas sp. Tetrahedron Lett. 1984;25:2475–2478. doi: 10.1016/S0040-4039(01)81208-9. [DOI] [Google Scholar]
- Nakao Y., Uehara T., Matsunaga S., Fusetani N., Van Van Soest R.W.M., Matsunaga S. Callyspongynic acid, a polyacetylenic acid which inhibits α-glucosidase, from the marine sponge Callyspongia truncata. J Nat Prod. 2002;65:922–924. doi: 10.1021/np0106642. [DOI] [PubMed] [Google Scholar]
- Nicolas G.M., Hong T.W., Molinski T.F., Lerch M.L., Cancilla M.T., Lebrilla C.B. Oceanapiside, an antifungal bis-α,ω-amino alcohol glycoside from the marine sponge Oceanapia phillipensis. J Nat Prod. 1999;62:1678–1681. doi: 10.1021/np990190v. [DOI] [PubMed] [Google Scholar]
- Northcote P.T., Blunt J.W., Munro M.H.G. Pateamine: a potent cytotoxin from the New Zealand marine sponge, Mycale sp. Tetrahedron Lett. 1991;32:6411–6414. doi: 10.1016/0040-4039(91)80182-6. [DOI] [Google Scholar]
- Nozeman C. Verhandeling over de inlandsche zoetwater-spongie, eene huisvesting der Maskers van puistenbijteren. (1788). Published by the Bataafs Genoodtschap, Part IX:1–16
- Oficjalski P. Spongia fluviatilis (Badiaga) Pharmazeutische Zentralhalle für Deutschland. 1937;78:173–175. [Google Scholar]
- Ohwada A., Takahashi H., Nagaoka I., Iwabuchi K., Mikami O., Kira S. Effect of cigarette smoke on the mRNA and protein expression of carcinoembryonic antigen (CEA), a possible chemoattractant for neutrophils in human bronchioloalveolar. Thorax. 1995;50:651–657. doi: 10.1136/thx.50.6.651. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okino T., Yoshimura E., Hirota H., Fusetani N. Antifouling kalihinenes from the marine sponge Acanthella cavernosa. Tetrahedron Lett. 1995;36:8637–8640. doi: 10.1016/0040-4039(95)01861-B. [DOI] [Google Scholar]
- Okino T., Yoshimura E., Hirota E., Fusetani N. New antifouling kalihipyrans from the marine sponge Acanthella cavernosa. J Nat Prod. 1996;59:1081–1083. doi: 10.1021/np960496r. [DOI] [Google Scholar]
- Pawlik J.R., Kernan M.R., Molinski T.F., Kay-Harper M., Faulkner D.J. Defensive chemicals of the Spanish dancer nudibranch Hexabranchus sanguineus and its egg ribbons: macrolides derived from a sponge diet. J Exp Mar Biol Ecol. 1988;119:99–109. doi: 10.1016/0022-0981(88)90225-0. [DOI] [Google Scholar]
- Perry N.B., Blunt J.W., Munro M.H.G., Higa T., Sakai R. Discorhabdin D an antitumor alkaloid from the sponges Latrunculia brevis and Prianos sp. J Org Chem. 1988;53:4127–4128. doi: 10.1021/jo00252a052. [DOI] [Google Scholar]
- Perry N.B., Ettouati L., Litaudon M., Blunt J.W., Munro M.H.G. Alkaloids from the antarctic sponge Kirkpatrickia varialosa, part 1: variolin B, a new antitumour and antiviral compound. Tetrahedron. 1994;50:3987–3992. doi: 10.1016/S0040-4020(01)89673-3. [DOI] [Google Scholar]
- Pettit R.K., McAllister S.C., Pettit G.R., Herald C.L., Johnson J.M., Cichacz Z.A. Abroad-spectrum antifungal from the marine sponge Hyrtios erecta. Int J Antimicrob Agents. 1998;9:147–152. doi: 10.1016/S0924-8579(97)00044-7. [DOI] [PubMed] [Google Scholar]
- Phife D.W., Ramos R.A., Feng M., King I., Gunasekera S.P., Wright A., Patel M., Pachter J.A., Coval SJ. Marine sponge bis(indole) alkaloids that displace ligand binding to α1-adrenergic receptors. Bioorg Med Chem Lett. 1996;6:2103–2106. doi: 10.1016/0960-894X(96)00376-9. [DOI] [Google Scholar]
- Pika J., Tischler M., Andersen RJ. Glaciasterols A and B, 9,11-secosteroids from the marine sponge Aplysilla glacialis. Can J Chem. 1992;70:1506–1510. [Google Scholar]
- Pope R.M., Lovis R., Mungre S., Perlman H., Koch A.E., Haines G.K., III C/EBPβ in rheumatoid arthritis: correlation with inflammation, not disease specifity. Clin Immunol. 1999;91:271–282. doi: 10.1006/clim.1999.4723. [DOI] [PubMed] [Google Scholar]
- Proksch P. Defensive roles for secondary metabolites from marine sponges and sponge-feeding nudibranchs. Toxicon. 1994;32:639–655. doi: 10.1016/0041-0101(94)90334-4. [DOI] [PubMed] [Google Scholar]
- Proksch P., Edrada R.A., Ebel R. Drugs from the seas—current status and microbiological implications. Appl Microbiol Biotechnol. 2002;59:125–134. doi: 10.1007/s00253-002-1006-8. [DOI] [PubMed] [Google Scholar]
- Quinn R.J., Gregson R.P., Cook A.F., Bartlett A.F. Isolation and synthesis of 1-methylisoguanisine, a potent pharmacologically active constituent from the marine sponge Tedania digitata. Tetrahedron Lett. 1980;21:567–568. doi: 10.1016/S0040-4039(01)85558-1. [DOI] [Google Scholar]
- Qureshi A., Faulkner D.J. Haplosamates A and B: new steroidal sulfamate esters from two haplosclerid sponges. Tetrahedron. 1999;55:8323–8330. doi: 10.1016/S0040-4020(99)00465-2. [DOI] [Google Scholar]
- Rahden-Staron I. The inhibitory effect of the fungicides captan and captafol on eukaryotic topoisomerases in vitro and lack of recombinagenic activity in the wing spot test of Drosophila melanogaster. Mutat Res. 2002;518:205–213. doi: 10.1016/s1383-5718(02)00107-9. [DOI] [PubMed] [Google Scholar]
- Randazzo A., Debitus C., Minale L., Pastor P.G., Alcaraz M.J., Paya M., Gomez-Paloma L. Petrosaspongiolides M-R: new potent and selective phospholipase A2 inhibitors from the New Caledonian marine sponge Petrosaspongia nigra. J Nat Prod. 1998a;61:571–575. doi: 10.1021/np9704922. [DOI] [PubMed] [Google Scholar]
- Randazzo A., Dal Piaz F., Orru S., Debitus C., Roussakis C., Pucci P., Gomez-Paloma L. Axinellins A and B: new proline-containing antiproliferative cyclopeptides from the Vanuatu sponge Axinella carteri. Eur J Org Chem. 1998b;11:2659–2665. doi: 10.1002/(SICI)1099-0690(199811)1998:11<2659::AID-EJOC2659>3.3.CO;2-8. [DOI] [Google Scholar]
- Rashid M.A., Gustafson K.R., Boswell J.L., Boyd M.R. Haligramides A and B, two new cytotoxic hexapeptides from the marine sponge Haliclona nigra. J Nat Prod. 2000;63:956–959. doi: 10.1021/np000051+. [DOI] [PubMed] [Google Scholar]
- Ratner L., Vander Heyden N., Dedera D. Inhibition of HIV and SIV infectivity by blockade of α-glucosidase activity. Virology. 1991;181:180–192. doi: 10.1016/0042-6822(91)90483-R. [DOI] [PubMed] [Google Scholar]
- Rodriguez J., Nunez L., Peixinho S, Jimenez C. Isolation and synthesis of the first natural 6-hydroximino 4-en-3-one-steroids from the sponges Cinachyrella spp. Tetrahedron Lett. 1997;38:1833–1836. doi: 10.1016/S0040-4039(97)00163-9. [DOI] [Google Scholar]
- Romo D., Rzasa R.M., Shea H.A., Park K., Langenhan J.M., Sun L., Akhiezer A., Liu J.O. Total synthesis and immunosuppressive activity of (-)-pateamine A and related compounds: implementation of a β-lactam-based macrocyclization. J Am Chem Soc. 1998;120:12237–12254. doi: 10.1021/ja981846u. [DOI] [Google Scholar]
- Ross S.A., Weete J.D., Schinazi R.F., Wirtz S.S., Tharnish P., Scheuer P.J., Hamann M.T. Mololipids, a new series of anti-HIV bromotyramine-derived compounds from a sponge of the order Verongida. J Nat Prod. 2000;63:501–503. doi: 10.1021/np980414u. [DOI] [PubMed] [Google Scholar]
- Ruomei Q., Ozaki Y., Satoh K., Kurota K., Asazuma N., Yatomi Y., Kume S. Quantitative measurement of various 5-HT receptor antagonists on platelet activation induced by serotonin. Thromb Res. 1996;81:43–54. doi: 10.1016/0049-3848(95)00212-X. [DOI] [PubMed] [Google Scholar]
- Saito S., Watabe S., Ozaki H., Fusetani N., Karaki H. Mycalolide, a novel actin depolymerizing agent. J Biol Chem. 1994;269:29710–29714. [PubMed] [Google Scholar]
- Sakai R., Higa T., Jefford C W., Bernardinelli G. Manzamin A, a novel antitumor alkaloid from a sponge. J Am Chem Soc. 1986;108:6404–6405. doi: 10.1021/ja00280a055. [DOI] [Google Scholar]
- Sakai R., Kamiya H., Murata M., Shimamoto K. A new neurotoxic amino acid from the Micronesian marine sponge Dysidea herbacea. J Am Chem Soc. 1997;119:4112–116. doi: 10.1021/ja963953z. [DOI] [Google Scholar]
- Sakai R., Swanson G.T., Shimamoto K., Green T., Contractor A., Ghetti A., Tamura-Horikawa Y., Oiwa C., Kamiya H. Pharmacological properties of the potent epileptogenic amino acid dysiherbaine, a novel glutamate receptor agonist isolated from the marine sponge Dysidea herbacea. J Pharmacol Exp Ther. 2001;296:650–658. [PubMed] [Google Scholar]
- Schmitz F.J., Gunasekera S.P., Yalamanchili G., Hossain M.B., Van der Van der Helm D. Tedanolide: a potent cytotoxic macrolide from the Caribbean sponge Tedania ignis. J Am Chem Soc. 1984;106:7251–7252. doi: 10.1021/ja00335a069. [DOI] [Google Scholar]
- Schroder K. Die Verwendung der Susswasserschwamme in der Ukraine. Die Umschau Wissenschaft Technik. 1942;46:507–509. [Google Scholar]
- Schroder H.C., Begin M.E., Klocking R., Matthes E., Sarma A.S., Gasic M.J., Muller W.E.G. Avarol restores the altered prostaglandin and leukotrin metabolism in monocytes infected with human immunodeficiency virus type 1. Virus Res. 1991;21:213–223. doi: 10.1016/0168-1702(91)90034-S. [DOI] [PubMed] [Google Scholar]
- Schwartsmann G. Marine organisms and other novel natural sources of new cancerdrugs. Ann Oncol. 2000;11:235–243. doi: 10.1023/A:1011160906608. [DOI] [PubMed] [Google Scholar]
- Sera Y., Adachi K., Shizuri Y. A new epidioxy sterol as an antifouling substance from a Palauan marine sponge, Lendenfeldia chondrodes. J Nat Prod. 1999;62:152–154. doi: 10.1021/np980263v. [DOI] [PubMed] [Google Scholar]
- Shimosaka A. Role of NKT cells and α-galactosyl ceramide. Int J Hematol. 2002;76:277–279. doi: 10.1007/BF03165262. [DOI] [PubMed] [Google Scholar]
- Shoji N., Umeyama A., Shin K., Takeda K., Arihara S., Kobayashi J., Takei M. Two unique pentacyclic steroids with cis C/D ring junction from Xestospongia bergquistia Fromont, powerful inhibitors of histamine release. J Org Chem. 1992;57:2996–2997. doi: 10.1021/jo00037a009. [DOI] [Google Scholar]
- Shuman R.T., Rothenberger R.B., Campell C.S., Smith G.F., Gifford-Moore D.S., Gesellchen P.D. Highly selective tripeptide thrombm inhibitors. J Med Chem. 1993;36:314–319. doi: 10.1021/jm00055a002. [DOI] [PubMed] [Google Scholar]
- Smith-McCune K.K., Reddy S., Robbins C., Zhu Y.-H. Induction of apoptosis by HPV 16E7: implications for cin and cervical cancer. J Soc Gynecologic Invest. 1996;3:376A. doi: 10.1016/1071-5576(96)83082-6. [DOI] [Google Scholar]
- Soni R., Muller L., Furet P., Schoepfer J., Stephan C., Zumstein-Mercker S., Fretz H., Chaudhuri B. Inhibition of cyclin-dependent kuiase 4 (Cdk4) by frascaplysin, a marine natural product. Biochem Biophys Res Commun. 2000;275:877–884. doi: 10.1006/bbrc.2000.3349. [DOI] [PubMed] [Google Scholar]
- Springer T.A., Lasky L.A. Sticky sugars for selectins. Nature. 1991;349:196–197. doi: 10.1038/349196a0. [DOI] [PubMed] [Google Scholar]
- Stead P., Hiscox S., Robinson P.S., Pike N.B., Sidebottom P.J., Roberts A.D., Taylor N.L., Wright A.E., Pomponi S.A., Langley D. Eryloside F, a novel penasterol disaccharide possessing potent thrombin receptor antagonist activity. Bioorg Med Chem Lett. 2000;10:661–664. doi: 10.1016/S0960-894X(00)00063-9. [DOI] [PubMed] [Google Scholar]
- Stodal (2003). Available at http://www.sblglobal.com/stodal.html
- Sullivan B., Djura P., McIntyre D.E., Faulkner D.J. Antimicrobial constituents of the sponge Siphonodictyon coralliphagum. Tetrahedron. 1981;37:979–982. doi: 10.1016/S0040-4020(01)97672-0. [DOI] [Google Scholar]
- Sun H.H., Cross S.S., Gunasekera M., Koehn F.E. Weinbersteroldisulfates A and B, antiviral steroid sulfates from the sponge Petrosia weinbergi. Tetrahedron. 1991;47:1185–1190. doi: 10.1016/S0040-4020(01)86375-4. [DOI] [Google Scholar]
- Suzuki H., Shindo K., Ueno A., Miura T., Takei M., Sakakibara M., Fukamachi H., Tanaka J., Higa T. S1319: A novel ß2-adrenoceptor agonist from a marine sponge Dysidea sp. Bioorg Med Chem Lett. 1999;9:1361–1364. doi: 10.1016/S0960-894X(99)00205-X. [DOI] [PubMed] [Google Scholar]
- Takei M., Burgoyne D.L., Andersen R.J. Effect of contignasterol on histamine release induced by anti-immunoglobulin E from rat peritoneal mast cells. J Pharm Sci. 1994;83:1234–1235. doi: 10.1002/jps.2600830909. [DOI] [PubMed] [Google Scholar]
- Tan P., Luscinskas F.W., Homer-Vanniasinkam S. Cellular and molecular mechanisms of inflammation and thrombosis. Eur J Endovasc Surg. 1997;17:373–389. doi: 10.1053/ejvs.1998.0759. [DOI] [PubMed] [Google Scholar]
- Ter Haar E., Kowalski R.J., Hamel E., Lin C.M., Longley R.E., Gunasekera S.P., Rosenkranz H.S., Day B.W. Discodermolide, a cytotoxic marine agent that stabilizes microtubules more potently than taxol. Biochemistry. 1996;35:243–250. doi: 10.1021/bi9515127. [DOI] [PubMed] [Google Scholar]
- Thompson J.E. Exudation of biologically-active metabolites in the sponge Aplysina fistularis, I: biological evidence. Mar Biol. 1985;88:23–26. doi: 10.1007/BF00393039. [DOI] [Google Scholar]
- Torres Y.R., Berlinck R.G.S., Nascimento G.G.F., Fortier S.C., Pessoa C., De De Moraes M.O. Antibacterial activity against resistant bacteria and cytotoxicity of four alkaloid toxins isolated from the marine sponge Arenosclera brasiliensis. Toxicon. 2002;40:885–891. doi: 10.1016/S0041-0101(01)00286-0. [DOI] [PubMed] [Google Scholar]
- Tsukamoto S., Kato H., Hirota H., Fusetani N. Ceratinamides A and B: new antifouling dibromotyrosine derivatives from the marine sponge Pseudoceratina purpurea. Tetrahedron. 1996a;52:8181–4186. doi: 10.1016/0040-4020(96)00387-0. [DOI] [PubMed] [Google Scholar]
- Tsukamoto S., Kato H., Hirota H., Fusetani N. Pseudoceratidine: a new antifouling spermidine derivative from the marine sponge Pseudoceratina purpurea. Tetrahedron Lett. 1996b;37:1439–1440. doi: 10.1016/0040-4039(96)00025-1. [DOI] [Google Scholar]
- Tsukamoto S., Matsunaga S., Fusetani N., Van Van Soest R.W.M. Acanthosterol sulfates A–J: ten new antifungal steroidal sulfates from a marine sponge Acanthodendrilla sp. J Nat Prod. 1998;61:1374–1378. doi: 10.1021/np980178n. [DOI] [PubMed] [Google Scholar]
- Turon X., Tarjuelo I., Uriz MJ. Growth dynamics and mortality of the encrusting sponge Crambe crambe (Poecilosclerida) in contrasting habitats: correlation with population structure and investment in defence. Functional Ecol. 1998;12:631–639. doi: 10.1046/j.1365-2435.1998.00225.x. [DOI] [Google Scholar]
- Unson M.D., Holland N.D., Faulkner D J. A brominated secondary metabolite synthesized by the cyanobacterial symbiont of a marine sponge and accumulation of the crystalline metabolite in the sponge tissue. Mar Biol. 1994;119:1–11. doi: 10.1007/BF00350100. [DOI] [Google Scholar]
- Urban S., De De Almeida Leone P., Carroll A.R., Fechner G.A., Smith J., Hooper J.N.A., Quinn R.J. Axinellamines A–D, novel imidazo-azolo-imidazole alkaloids from the Australian marine sponge Axinell asp. J Org Chem. 1999;64:731–735. doi: 10.1021/jo981034g. [DOI] [PubMed] [Google Scholar]
- Wakimoto T., Maruyama A., Matsunaga S., Fusetani N., Shinoda K., Murphy P.T. Octa- and nonaprenylhydroquinone sulfates, inhibitors of α1,3-fucosyltransferase VII, from an Australian marine sponge Sarcotragus sp. Bioorg Med Chem Lett. 1999;9:727–730. doi: 10.1016/S0960-894X(99)00059-1. [DOI] [PubMed] [Google Scholar]
- Wellington K.D., Cambie R.C., Rutledge P.S., Bergquist P.R. Chemistry of sponges, 19: Novel bioactive metabolites from Hamigera tarangaensis. J Nat Prod. 2000;63:79–85. doi: 10.1021/np9903494. [DOI] [PubMed] [Google Scholar]
- Wilkinson C.R. Microbial associations in sponges, III: ultrastructure of the in situ associations in coral reef sponges. Mar Biol. 1978;49:177–185. doi: 10.1007/BF00387117. [DOI] [Google Scholar]
- Willis R.H., De De Vries D J. BRS1, a C30 bis-amino, bis-hydroxy polyunsaturated lipid from an Australian calcareous sponge that inhibits protein kinase C. Toxicon. 1997;35:1125–1129. doi: 10.1016/S0041-0101(96)00218-8. [DOI] [PubMed] [Google Scholar]
- Yamaguchi Y., Motoki K., Ueno H., Maeda K., Kobayashi E., Inoue H., Fukushima H., Koezuka Y. Enhancing effects of (2S,3S,4R)-1-O-(alpha-D-galactopyranosyl)-2-(N-hexacosanoylamino)-1,3,4-octadecanetriol (KRN7000) on antigen-presenting function of antigen-presenting cells and antimetastatic activity of KRN7000-pretreated antigen-presenting cells. Oncol Res. 1996;8:399–407. [PubMed] [Google Scholar]
- Yoshiji H., Kuriyama S., Ways D.K., Yoshii J., Miyamoto Y., Kawata M., Ikenaka Y., Tsujinoue H., Nakatani T., Shibuya M., Fukui H. Protein kinase C lies on the signaling pathway for vascular endothelial growth factor–mediated tumor development and angiogenesis. Cancer Res. 1999;59:4413–4418. [PubMed] [Google Scholar]
- Yousaf M., El Sayed K.A., Rao K.V., Lim C.W., Hu J.-F., Kelly M., Franzblau S.G., Zhang F., Peraud O., Hill R.T., Hamann M.T. 12,34-Oxamanzamines, novel biocatalytic and natural products from rnanzamine producing Indo-Pacific sponges. Tetrahedron. 2002;58:7397–7402. doi: 10.1016/S0040-4020(02)00825-6. [DOI] [Google Scholar]
- Zapolska-Downar D., Zapolska-Downar A., Markiewski M., Ciechanowicz M., Kaczmarczyk M., Naruszewicz M. Selective inhibition by procubol of vascular cell adhesion molecule 1 (VCAM-1) expression in human vascular endothelial cells. Atherosclerosis. 2001;155:123–130. doi: 10.1016/S0021-9150(00)00553-0. [DOI] [PubMed] [Google Scholar]
- Zhu K., Levine R.S., Brann E.A., Baum M.K. The relationship of hepatitis history and pathological diagnosis of primary liver cancer. J Clin Epidemiol. 1997;50:197–301. doi: 10.1016/S0895-4356(96)00364-2. [DOI] [PubMed] [Google Scholar]


