Abstract
Since the discovery of human bocavirus (hBoV), the virus has been detected worldwide in respiratory tract samples from young children by various polymerase chain reaction (PCR) assays and real-time PCRs (Q-PCR). Until now, no data have been reported on the presence of hBoV in Belgium and the detection of hBoV in a multiplex Q-PCR setting has not been described. The aim of this study was to develop a fast and reliable multiplex Q-PCR for the simultaneous detection of hBoV DNA and adenovirus (AdV) DNA. During the winter of 2004–2005, 445 nasopharyngeal aspirates (NPAs) were analysed from 404 Belgian children up to 5 years old with acute respiratory tract infections (ARTIs). (Co)infections with hBoV, AdV, respiratory syncytial virus (RSV), human metapneumovirus (hMPV) and influenza A virus were investigated. A viral agent was detected in 61% (n = 272/445) of the NPAs. Multiplex Q-PCR found a prevalence of 11% (n = 51/445) hBoV and 13% (n = 58/445) AdV. Coinfections were more frequently found with AdV (62%; n = 36/58) than with hBoV (49%; n = 25/51). Follow-up samples were available from 22 patients with ARTIs. In three patients, hBoV DNA persisted for one month. Multiplex Q-PCR may help in closing the diagnostic gap by addressing a broader range of potential respiratory pathogens.
Keywords: Respiratory Syncytial Virus, Lower Respiratory Tract Infection, Severe Acute Respiratory Syndrome, Human Bocavirus, Respiratory Tract Sample
Introduction
Human bocavirus (hBoV) was discovered by Allander et al. using random polymerase chain reaction (PCR) amplification and large-scale sequencing for virus screening in a pool of human respiratory tract samples [1]. Two genomes of hBoV were distinguished in Stockholm, ST1 and ST2. The deduced amino acid sequences were related to bovine parvovirus and canine minute virus in the family of the Parvoviridae, from which the name bocavirus was derived.
The first prevalence studies detected hBoV in the respiratory tract of 1.5% to 19% of young symptomatic children [2, 3]. Later publications reported the virus in the gastro-intestinal tract and serum, referring to a systemic dissemination [2, 4]. In Australia and Korea, prevalence studies showed that hBoV was the second most frequently encountered virus after respiratory syncytial virus (RSV) among children with acute respiratory tract infections (ARTIs) [5, 6]. Until now, the clinical significance and the causal role of hBoV has not yet been proven, partly because of a high coinfection rate ranging from 12% to 83% [3].
Viruses are the major cause of ARTIs in children in developed countries [7]. Up to half of the infants have more than one episode of lower respiratory tract infection in their first year of life [8]. Thanks to molecular techniques, the panel of potential respiratory pathogens has recently been enlarged with human metapneumovirus (hMPV), the coronaviruses NL63, HKU1 and severe acute respiratory syndrome (SARS), the polyomaviruses Wu and Ki, and hBoV [9, 10]. To narrow the diagnostic gap, a broader range of potential pathogens should be addressed in the routine laboratory. This can be realised by fast and reliable multiplex real-time PCR (Q-PCR).
Studies integrating hBoV DNA in a multiplex Q-PCR were rarely published, probably because most of the respiratory pathogens are RNA viruses for which a reverse-transcription step is necessary in PCR-based platforms. In our laboratory, the concept of multiplex Q-PCR was already in use for the routine analysis of RNA viruses, RSV and hMPV [11] and influenza A and B viruses. In this study, we developed a multiplex Q-PCR to simultaneously detect hBoV DNA and adenovirus (AdV) DNA in nasopharyngeal aspirates (NPAs) of children with ARTIs.
The frequencies of hBoV, AdV, RSV, hMPV and influenza A virus were determined in this paediatric population. Coinfection rates of hBoV and AdV were calculated and subsequent episodes of ARTIs were investigated.
Materials and methods
Specimen collection
NPAs (n = 445) from 404 children (≤5 years of age) of age with ARTIs who presented at the Onze Lieve Vrouw (OLV) Hospital (844 beds; 54 paediatric beds) between 1st November 2004 and 31st May 2005 were analysed for hBoV, AdV, RSV, hMPV and influenza A virus. All infants included in the study suffered from respiratory distress, dyspnoea and/or acute expiratory wheezing. A paediatrician decided for hospitalisation or ambulatory treatment on the basis of the degree of hypoxaemia and the clinical and respiratory status of each child. NPAs were extracted with the QIAamp Viral RNA Mini Kit from Qiagen and RSV RNA and hMPV RNA were analysed by multiplex Q-PCR on a daily basis [11]. Influenza A virus RNA was investigated by real-time PCR during the influenza season (according to the national surveillance data; http://www.iph.fgov.be/flu). Nucleic acid extracts were stored at −70°C and retrospectively analysed in batches for the presence of hBoV DNA and AdV DNA by the multiplex Q-PCR described in this study.
Real-time PCR and sequencing
The target for hBoV was a highly conserved region of 88 bp in the nonstructural protein 1 (NP-1) gene. The hBoV primers and probe were designed using the Primer Express 2.0 software from Applied Biosystems. Subsequently, the hBoV Q-PCR was combined with an in-house previously validated Q-PCR targeting a 132-bp fragment from the hexon gene of AdV (based on [12]) to obtain a multiplex Q-PCR (Table 1). The multiplex Q-PCR detecting simultaneously hBoV DNA and AdV DNA was performed on the ABI 7000 Sequence Detection System (ABI). The assay was carried out in a total volume of 25 µL containing ABgene buffer (Absolute QPCR ROX Master Mix from ABgene), 300 nM of each forward and reverse primer from both viruses, 200 nM of probes from both viruses and 5 µL of extracted DNA from the sample. The Q-PCR protocol consisted of 2 min of decontamination at 50°C, 15 min of denaturation and activation of the Taq-DNA-polymerase at 95°C, followed by 45 cycles of 15 sec of denaturation at 95°C and 1 min annealing and extension at 60°C.
Table 1.
Nucleotide sequences of the primers and probes used for multiplex Q-PCR and sequencing
| Multiplex Q-PCR primers and probes | Sequence |
| hBoV NP-1 gene (88 bp)a | |
| Forward primer | 5′-AGAGGCTCGGGCTCATATCA-3′ (2478)c |
| Reverse primer | 5′-TCTTCATCACTTGGTCTGAGGTCTT-3′ (2541)c |
| Probe | 5′-VIC-AGGAACACCCAATCARCCACCTATCGTCT-TAMRA-3′ (2500)c |
| AdV hexon gene (132 bp)b | |
| Forward primer | 5′-GCCACGGTGGGGTTTCTAAACTT-3′ (21)e |
| Reverse primer | 5′-GCCCCAGTGGTCTTACATGCACATC-3′ (152)e |
| Probe | 5′-FAM-TGCACCAGACCCGGGCTCAGGTACTCCGA-TAMRA-3′ (89)e |
| Sequencing primers | Sequence |
| hBoV NP-1 gene (313 bp)a | |
| Forward primer | 5′-CTCCAGCAAGTCCTCCAAAC-3′ (2253)c |
| Reverse primer | idem hBoV Q-PCR |
| hBoV VP2 gene (965 bp) a | |
| Forward primer | 5′-CAGCTGGAGGCAATGCTAC-3′ (4059)d |
| Reverse primer | 5′-ATGACGTGGGCTGGATGTAT-3′ (5005)d |
| Forward primer semi-nested | 5′-AATTGCAAACCCATCACTCTCA-3′ (4558)d |
| Reverse primer semi-nested | 5′-CCCAGACTTGATTTGGAAACATC-3′ (4645)d |
hBoV = human bocavirus; AdV = adenovirus
aDesigned as part of the present study
bHeim et al. [12], adapted according to the sequence information available in GenBank
c, d, eThe location of the primers and probes is indicated in parentheses (the reference sequence is DQ000495(c) and DQ000496(d) for hBoV and AB330083(e) for AdV)
An hBoV-positive control was kindly provided by Dr. B. Weissbrich [13]. The positive control consisted of a plasmid construct containing a NP-1 gene fragment. A positive control for AdV consisted of a DNA extract from an AdV-positive sample.
The cut-off for a positive result was set at cycle threshold (Ct) ≤ 36 (this corresponds to ≥10e4 copies/mL of the plasmid control) based on 90% concordance between hBoV single-locus Q-PCR and hBoV multiplex Q-PCR, as shown by a difference of a maximum of one Ct in 18/20 samples, ranging from Ct 15.48 (10e10 copies/mL) to Ct 36.45 (10e4 copies/mL). For samples with a Ct > 36 (<10e4 copies/mL), reproducibility was not guaranteed and the results were reported as ‘indeterminate’.
Amplicon sequencing for the confirmation of hBoV identity was performed on two samples with high copy numbers, respectively Ct = 15.48 (10e10 copies/mL) and Ct = 20.00 (10e8 copies/mL). The sequencing primers developed in this study are listed in Table 1. Both a 313-bp fragment of the NP-1 gene and a 965-bp fragment of the viral capsid protein 2 (VP2) gene polymorphous region were sequenced. For cycle sequencing, the CEQ Dye Terminator Cycle Sequencing (DTCS) Quick Start Kit was used (Analis, Suarlée, Belgium). Sequencing reactions were analysed on a CEQ 8000 Capillary Sequencer (Beckman Coulter, Fullerton, CA, USA) according to the manufacturer’s instructions. The resulting sequences were aligned with each other and with sequences in GenBank with the use of the ClustalW program (http://www.ebi.ac.uk/clustalw).
Statistical analysis
Data are expressed as the median and inter-quartile range. Independent samples t-test was used for the comparison of continuous variables. A two-sided test of P < 0.05 was judged to be significant. Statistical analysis was done with MedCalc®v9.4.1.0 (Mariakerke, Belgium).
Results
Optimisation of real-time PCR
hBoV probe and primer concentrations were initially optimised in a single-locus hBoV Q-PCR. The specificity was confirmed by sequencing two highly positive samples of Ct = 15.48 (10e10 copies/mL) and Ct = 20.00 (10e8 copies/mL). BLAST analysis of the sequenced NP-1 and VP2 gene fragments showed a perfect homology with known hBoV sequences in GenBank (accession numbers DQ000495, DQ000496, AM160609, AM160610, AM160611, AM160612, AM160613, AM160614, AM160615). There was no significant homology with other organisms. The exact sequences of the forward primer and probe were recovered in the NP-1 gene fragment. Phylogenetic analysis of the sequenced VP2 fragment classified both samples as ST2.
The hBoV Q-PCR was combined with an AdV Q-PCR to develop a multiplex hBoV/AdV Q-PCR for the simultaneous detection of both DNA viruses. The multiplex Q-PCR was further optimised using a ten-fold dilution series covering a range of seven log10 dilutions of the positive controls for hBoV respectively AdV (Fig. 1). The multiplex Q-PCR was linear from 10e4 to 10e8 copies/mL. The 92% efficiency of the hBoV Q-PCR in a multiplex setting was slightly higher than the 82% efficiency for AdV. The multiplex Q-PCR was specific for both viruses independently, without mutual cross-reactivity. No interference was observed with hMPV, RSV, influenza A and B virus, and Mycoplasma pneumoniae.
Fig. 1.
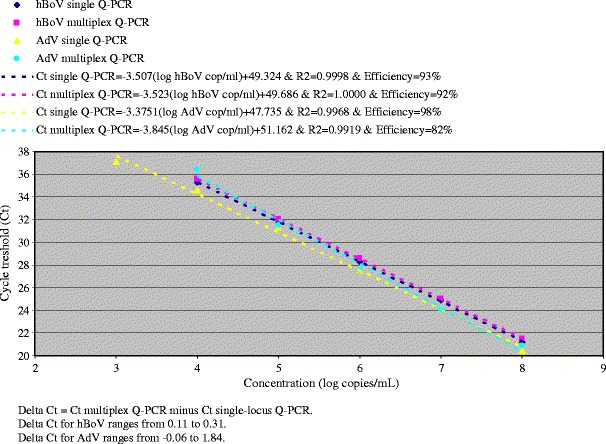
Cycle threshold (Ct) values obtained for the analysis of the human bocavirus (hBoV)- and adenovirus (AdV)-positive dilution series in single-locus and multiplex Q-PCRs
Checkerboard analysis of combined dilutions of hBoV- and AdV-positive controls ranging from 10e2 to 10e10 copies/mL was performed with the multiplex Q-PCR. The sensitivity of the multiplex Q-PCR for hBoV detection was 10e4 copies/mL (Ct = 36). The sensitivity of AdV diminished when high hBoV concentrations (10e6–10e10 copies/mL) were analysed. Interventions in the composition of the assay format did not alter the sensitivity. The AdV sensitivity was improved to 10e4 copies/mL (Ct = 36) by a two-stage algorithm: if the result of the multiplex Q-PCR gave hBoV ≥ 10e5 copies/mL (Ct ≤ 32), the sample needed to be retested at a second stage with the AdV single-locus Q-PCR.
Patient results
During the winter season of 2004–2005, a viral agent was detected in as many as 61% (n = 272/445) of NPAs from children (≤5 years of age) with ARTIs. The prevalence of the aetiologic agents was 35% (n = 154/445) for RSV, 13% (n = 58/445) for AdV, 13% (n = 37/290) for influenza A, 11% (n = 51/445) for hBoV and 6% (n = 28/445) for hMPV.
Box-and-whisker plots of the multiplex Q-PCR results (Figs. 2 and 3) showed a median hBoV Ct of 32.02 [27.29–35.06] and a median AdV Ct of 30.26 [23.62–34.81]. These ranges correspond to 1.03 10e5 [10e4–10e6] hBoV copies/mL and 2.73 10e5 [10e4–10e7] AdV copies/mL. The majority of the hBoV-positive samples had a high Ct, indicating a low viral load. The AdV-positive samples, on the other hand, showed an equal spread between Ct 17.91 (10e9 copies/mL) and 36 (10e4 copies/mL). The highest hBoV viral load of 10e10 copies/mL (Ct = 13.04) was found in the NPA of an 11-month-old boy and the highest AdV viral load of 10e9 copies/mL (Ct = 17.91) was found in the NPA of a 7-month-old boy.
Fig. 2.
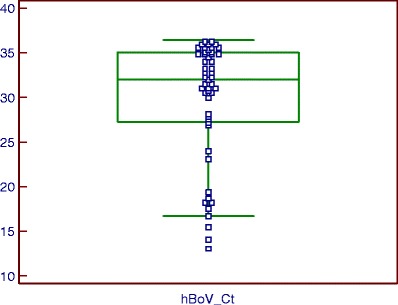
Ct distribution of hBoV detected with the multiplex Q-PCR algorithm (box-and-whisker plot). Fifty-one nasopharyngeal aspirates (NPAs) from children up to 5 years old were positive for hBoV with a median Ct of 32.02 [27.29–35.06], corresponding to 1.03 10e5 [10e4–10e6] hBoV plasmid control copies/mL
Fig. 3.
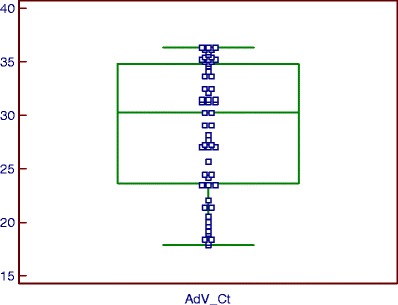
Ct distribution of AdV detected with the multiplex Q-PCR algorithm (box-and-whisker plot). Fifty-eight NPAs from children up to 5 years old were positive for AdV with a median Ct of 30.26 [23.62–34.81], corresponding to 2.73 10e5 [10e4–10e7] AdV copies/mL
Of the 272 diagnostic NPAs, 53 (19%) were coinfected by at least two potential pathogens (Table 2). We found more coinfections with AdV (62%; n = 36/58) than with hBoV (49%; n = 25/51). The viral loads in the coinfected samples were not statistically different from the monoinfections (P = 0.3012 for hBoV and P = 0.2912 for AdV). hBoV coinfections were observed with RSV (n = 10), AdV (n = 7), influenza A (n = 3), hMPV (n = 1) and triple infections of hBoV were seen with AdV/influenza A virus (n = 2) and with AdV/RSV (n = 2). In four of the AdV coinfections, the results of the multiplex Q-PCR transgressed the limit of detection for AdV (Ct ≥ 35 or ≤10e4 copies/mL) and the viral load was obtained by the two-stage algorithm.
Table 2.
Viral coinfections in the paediatric study population. Of the 272 diagnostic nasopharyngeal aspirates (NPAs), 53 (19%) were coinfected by at least two potential pathogens
| n (%) | |
|---|---|
| Tested NPAs | 445 |
| Mono- and coinfections | 272 (61%) |
| Coinfections | 53 (19%) |
| AdV/RSV | 18 |
| hBoV/RSV | 10 |
| hBoV/AdV | 7 |
| AdV/hMPV | 4 |
| hBoV/influenza A | 3 |
| AdV/influenza A | 3 |
| hMPV/RSV | 2 |
| hBoV/AdV/RSV | 2 |
| hBoV/AdV/influenza A | 2 |
| RSV/influenza A | 1 |
| hBoV/hMPV | 1 |
| hMPV/influenza A | 0 |
Follow-up samples were available from 22 infected patients (two AdV, four hBoV, four hMPV, eleven RSV and one RSV/AdV infected samples). Neither RSV nor hMPV persisted in subsequent samples, while AdV (n = 1/4) and hBoV (n = 3/4) DNA could still be found after at least one month. Two patients showed persistence of a low hBoV viral load of 10e4 copies/mL during one month, with the appearance of RSV at the end of this period. One hBoV patient showed a gradually decreasing viral load from 10e10 copies/mL to 10e6 copies/mL in 2 months. The clearance of hBoV was seen in two patients 3 to 4 months after initial infection with 10e9 copies/mL and 10e5 copies/mL, respectively. Less than six months after the first ARTI, a new episode of ARTI caused by a different aetiologic agent was observed in 15 patients. Cluster analysis of positive hBoV patients was in no case suggestive for a nosocomial transmission.
Discussion
A linear and sensitive AdV/hBoV multiplex Q-PCR was developed and used to detect hBoV and AdV DNA in NPAs. The multiplex Q-PCR was less sensitive than the single-locus Q-PCR for AdV when a high amount of hBoV DNA was present in the sample. Analysis of clinical samples from paediatric patients with ARTIs showed that the majority of the hBoV-positive samples had a low viral load. Therefore, the number of samples that had to be retested for AdV with the single-locus Q-PCR was minimal and the optimised algorithm which includes the multiplex Q-PCR provides a valuable tool in the diagnosis of viral respiratory infections since the work load is considerably reduced.
hBoV has been found in children with upper and lower respiratory tract infection in America, Europe, Africa, Asia and Australia, confirming a global distribution. The prevalence ranged between 1.5 and 19%, depending on the PCR target used and the defined population [2, 3]. Until now, no prevalence of hBoV was known in Belgium. Based on surveys mentioning the predominance of hBoV in infants and young children, a study population aged ≤5 years was selected. This report shows that, in the winter of 2004–2005, hBoV was present in 11% of the NPAs of young Belgian children with ARTIs.
Up to half of the infants have more than one episode of lower respiratory tract infection in their first year of life [8]. In this study population, follow-up samples were available for 22 patients of age 14 days to 4 years who suffered from ARTIs. Many patients (n = 15/22) had a subsequent episode of ARTI caused by a new pathogen 11 days to 6 months following the first episode. Most initial infections were completely cleared at the time when the second infection was detected. hBoV DNA, in contrast, could still be found one month after the first sample was taken, even in patients with a very weak primary infection (10e4 copies/mL) and a coinfecting agent (RSV) in the follow-up sample.
The clinical relevance of hBoV as a respiratory pathogen remains unclear due to the high coinfection rates (12%–83%), persistent viral shedding with low hBoV viral loads and the lack of prospective studies with negative control populations [3, 7, 14–18]. Consistent with the literature, many hBoV coinfections (49%) were found in this study. However, the coinfection rate of AdV, a well-known respiratory pathogen, was even higher (62%). The causal role of hBoV in respiratory tract diseases is still a topic for debate. Koch’s postulates from the 1890s cannot be used to establish the causal relationship of newly identified viruses because of the difficulty in culturing them. Culture-independent criteria for evidence of causation are proposed by Fredericks and Relman [19]. Those criteria are correlated to the copy number of pathogen-associated nucleic acid sequences. Some authors suggested an association between high hBoV viral loads and hBoV being the sole virus detected in the respiratory tract, systemic dissemination of hBoV and the resolution of symptoms after decline of the hBoV DNAemia [3]. In this study, no significant difference in hBoV viral load between monoinfected and coinfected samples (P = 0.3012) was found. However, the finding of the persistence of low hBoV viral loads after several months and the high coinfection rate suggests that a quantitative approach has to be considered when testing for hBoV.
Clinically, no distinction can be made between symptoms caused by RSV, hMPV or hBoV [20]. Studies frequently found hBoV in NPAs of hospitalised children with respiratory distress and abnormal chest radiography [7, 21–24]. Lower respiratory tract infections, bronchitis, bronchiolitis, pneumonia, croup and asthma exacerbations were the prominent diseases. The most important symptoms were rhinorrhoea, fever, cough and wheezing.
The diagnostic gap for respiratory infections is narrowing thanks to the extension of the list of potential viral pathogens. The broad range of potential aetiologic agents needs expanded surveillance [25]. Many newly identified viruses are not detectable with rapid antigen detection assays nor culture, e.g. coronavirus, polyomavirus and hBoV [6]. Multiplex Q-PCR may help in closing the diagnostic gap efficiently by addressing a broader range of potential pathogens. It is relatively fast for surveillance and outbreak investigation and is more sensitive than antigen detection. Since most current molecular diagnostics have a limited scope, a multiplex Q-PCR for the simultaneous detection of hBoV and AdV DNA was developed. In previous years when only RSV and influenza antigen testing were performed in this hospital, a pathogen was identified in less than 30% of ARTI cases [11]. In this study, the Q-PCR detection of RSV/hMPV RNA, influenza A virus RNA and hBoV/AdV DNA resulted in a diagnosis in 61% of the paediatric ARTIs. Using virus culture alone, Choi et al. found a potential aetiologic agent in 36% of children with ARTIs compared to 61% of the cases using PCRs targeting 11 viruses [6]. When a larger diagnostic panel of 16 respiratory viruses was analysed by Allander et al., a potential aetiologic agent was detected in up to 95% of the cases [7].
In conclusion, the multiplex Q-PCR developed in this study is a reliable method for the fast detection of hBoV and AdV in paediatric NPAs. In 2004, hBoV was found in 11% of the NPAs from young Belgian children with ARTIs. In almost half of the NPAs, hBoV coincided with various respiratory pathogens. However, the presence of hBoV in NPAs should be interpreted cautiously, taking into account the viral load and the clinical context.
References
- 1.Allander T, Tammi MT, Eriksson M, et al. Cloning of a human parvovirus by molecular screening of respiratory tract samples. Proc Natl Acad Sci U S A. 2005;102:12891–12896. doi: 10.1073/pnas.0504666102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Allander T. Human bocavirus. J Clin Virol. 2008;41:29–33. doi: 10.1016/j.jcv.2007.10.026. [DOI] [PubMed] [Google Scholar]
- 3.Schildgen O, Müller A, Allander T, et al. Human bocavirus: passenger or pathogen in acute respiratory tract infections? Clin Microbiol Rev. 2008;21:291–304. doi: 10.1128/CMR.00030-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Kahn J. Human bocavirus: clinical significance and implications. Curr Opin Pediatr. 2008;20:62–66. doi: 10.1097/MOP.0b013e3282f3f518. [DOI] [PubMed] [Google Scholar]
- 5.Sloots TP, McErlean P, Speicher DJ, et al. Evidence of human coronavirus HKU1 and human bocavirus in Australian children. J Clin Virol. 2006;35:99–102. doi: 10.1016/j.jcv.2005.09.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Choi EH, Lee HJ, Kim SJ, et al. The association of newly identified respiratory viruses with lower respiratory tract infections in Korean children, 2000–2005. Clin Infect Dis. 2006;43:585–592. doi: 10.1086/506350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Allander T, Jartti T, Gupta S, et al. Human bocavirus and acute wheezing in children. Clin Infect Dis. 2007;44:904–910. doi: 10.1086/512196. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Arnold JC, Singh KK, Spector SA, et al. Undiagnosed respiratory viruses in children. Pediatrics. 2008;121:e631–e637. doi: 10.1542/peds.2006-3073. [DOI] [PubMed] [Google Scholar]
- 9.Sloots TP, Whiley DM, Lambert SB, et al. Emerging respiratory agents: new viruses for old diseases? J Clin Virol. 2008;42:233–243. doi: 10.1016/j.jcv.2008.03.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Bialasiewicz S, Whiley DM, Lambert SB, et al. Development and evaluation of real-time PCR assays for the detection of the newly identified KI and WU polyomaviruses. J Clin Virol. 2007;40:9–14. doi: 10.1016/j.jcv.2007.07.015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Bonroy C, Vankeerberghen A, Boel A, et al. Use of a multiplex real-time PCR to study the incidence of human metapneumovirus and human respiratory syncytial virus infections during two winter seasons in a Belgian paediatric hospital. Clin Microbiol Infect. 2007;13:504–509. doi: 10.1111/j.1469-0691.2007.01682.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Heim A, Ebnet C, Harste G, et al. Rapid and quantitative detection of human adenovirus DNA by real-time PCR. J Med Virol. 2003;70:228–239. doi: 10.1002/jmv.10382. [DOI] [PubMed] [Google Scholar]
- 13.Weissbrich B, Neske F, Schubert J, et al. Frequent detection of bocavirus DNA in German children with respiratory tract infections. BMC Infect Dis. 2006;6:109. doi: 10.1186/1471-2334-6-109. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Schenk T, Huck B, Forster J, et al. Human bocavirus DNA detected by quantitative real-time PCR in two children hospitalized for lower respiratory tract infection. Eur J Clin Microbiol Infect Dis. 2007;26:147–149. doi: 10.1007/s10096-006-0244-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Lu X, Chittaganpitch M, Olsen SJ, et al. Real-time PCR assays for detection of bocavirus in human specimens. J Clin Microbiol. 2006;44:3231–3235. doi: 10.1128/JCM.00889-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Vicente D, Cilla G, Montes M, et al. Human bocavirus, a respiratory and enteric virus. Emerg Infect Dis. 2007;13:636–637. doi: 10.3201/eid1304.061501. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Neske F, Blessing K, Tollmann F, et al. Real-time PCR for diagnosis of human bocavirus infections and phylogenetic analysis. J Clin Microbiol. 2007;45:2116–2122. doi: 10.1128/JCM.00027-07. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Ricour C, Goubau P. Human bocavirus, a newly discovered parvovirus of the respiratory tract. Acta Clin Belg. 2008;63:329–334. doi: 10.1179/acb.2008.064. [DOI] [PubMed] [Google Scholar]
- 19.Fredericks DN, Relman DA. Sequence-based identification of microbial pathogens: a reconsideration of Koch’s postulates. Clin Microbiol Rev. 1996;9:18–33. doi: 10.1128/cmr.9.1.18. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Völz S, Schildgen O, Klinkenberg D, et al. Prospective study of Human Bocavirus (HBoV) infection in a pediatric university hospital in Germany 2005/2006. J Clin Virol. 2007;40:229–235. doi: 10.1016/j.jcv.2007.07.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Arnold JC, Singh KK, Spector SA, Sawyer MH. Human bocavirus: prevalence and clinical spectrum at a children’s hospital. Clin Infect Dis. 2006;43:283–288. doi: 10.1086/505399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Kesebir D, Vazquez M, Weibel C, et al. Human bocavirus infection in young children in the United States: molecular epidemiological profile and clinical characteristics of a newly emerging respiratory virus. J Infect Dis. 2006;194:1276–1282. doi: 10.1086/508213. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Ma X, Endo R, Ishiguro N, et al. Detection of human bocavirus in Japanese children with lower respiratory tract infections. J Clin Microbiol. 2006;44:1132–1134. doi: 10.1128/JCM.44.3.1132-1134.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Fry AM, Lu X, Chittaganpitch M, et al. Human bocavirus: a novel parvovirus epidemiologically associated with pneumonia requiring hospitalization in Thailand. J Infect Dis. 2007;195:1038–1045. doi: 10.1086/512163. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Fox JD. Respiratory virus surveillance and outbreak investigation. J Clin Virol. 2007;40(Suppl 1):S24–S30. doi: 10.1016/S1386-6532(07)70006-9. [DOI] [PMC free article] [PubMed] [Google Scholar]


