Abstract
Many emerging infectious diseases in human populations are associated with zoonotic origins. Attention has often focused on wild animal reservoirs, but most zoonotic pathogens of recent concern to human health either originate in, or are transferred to, human populations from domesticated animals raised for human consumption. Thus, the ecological context of emerging infectious disease comprises two overlapping ecosystems: the natural habitats and populations of wild animals, and the anthropogenically controlled habitats and populations of domesticated species. Intensive food animal production systems and their associated value chains dominate in developed countries and are increasingly important in developing countries. These systems are characterized by large numbers of animals being raised in confinement with high throughput and rapid turnover. Although not typically recognized as such, industrial food animal production generates unique ecosystems—environments that may facilitate the evolution of zoonotic pathogens and their transmission to human populations. It is often assumed that confined food animal production reduces risks of emerging zoonotic diseases. This article provides evidence suggesting that these industrial systems may increase animal and public health risks unless there is recognition of the specific biosecurity and biocontainment challenges of the industrial model. Moreover, the economic drivers and constraints faced by the industry and its participants must be fully understood in order to inform preventative policy. In order to more effectively reduce zoonotic disease risk from industrial food animal production, private incentives for the implementation of biosecurity must align with public health interests.
Keywords: influenza A virus, avian, poultry, zoonoses, agriculture, biosecurity
Introduction
The high-profile emergence of human diseases from animal populations, such as Nipah virus infection in 1999, SARS in 2002, and highly pathogenic avian influenza (HPAI) from 1997 to the present, have heightened public awareness of linkages between animal populations and human health. It is estimated that three out of four emerging pathogens affecting humans over the past 10 years have originated from animals or animal products (Taylor et al., 2001).
Considerable research attention is devoted to understanding the ecological dynamics that foster pathogen spillover from the wild animal host to susceptible species (Daszak et al., 2000; Woolhouse, 2002; Pulliam, 2008). Yet the environments of domesticated food animals—systems that are driven by unique ecological, social, and economic factors—are not usually recognized as ecosystems in and of themselves, with intense interactions between animals, humans, and pathogens. For the purposes of this article, we define the industrial food animal ecosystem as a distinct entity consisting of organisms (including humans, domesticated and wild animals, and microorganisms) interacting with each other and an anthropomorphically designed environment designed to maximize profit rather than biological sustainability. Understanding the complex dynamics that define the food animal ecosystem of the 21st century is central to mitigating risks of emerging zoonoses.
Domesticated animals raised to produce human food play a significant role in the emergence of zoonotic pathogens through two primary mechanisms (as shown schematically in Figure 1): as a bridge between wild animal reservoirs and human populations, and as the locus of pathogen evolution itself. The outbreak of Nipah virus in 1999 exemplifies the former mechanism, in which domesticated animals (pigs) served as a biological vector between the host species (wild fruit bats) and the human population (Field et al., 2007). The development of antimicrobial-resistant bacteria is a central example of the latter mechanism, in which the food animal environment influences the evolution of pathogens. The use of antimicrobial agents in industrial food animal production, particularly at nontherapeutic doses for growth promotion, provides constant pressure on bacterial pathogens to select for resistance (Gaze et al., 2008; Silbergeld et al., 2008).
Figure 1.
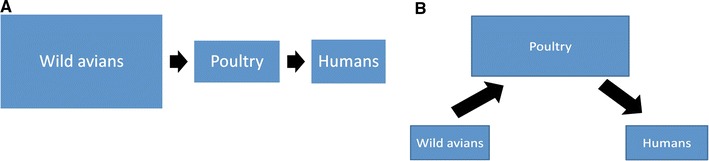
Simplified schematics of avian influenza transmission dynamics among wild avians, poultry, and humans. A, B: Two simplistic models of the avian influenza transmission dynamics among wild birds, domesticated birds, and humans. This model is not comprehensive (i.e., does not include direct wild bird transmission to humans, the possible role of swine or other intermediate hosts, or disease transmission from poultry to wild birds), but is merely a representation of two different functions poultry may play in AI transmission to humans. In (A), poultry serve as a pass-through between the wild avian host and humans. Poultry are infected by wild avians, and the virus replicates within the poultry host without undergoing significant alterations. Infectious poultry, in turn, transmit the virus to human populations, notably poultry workers. Model (B) demonstrates another role poultry play in the avian influenza ecosystem: the locus of pathogen evolution and adaptation to new host species. While poultry remain a vehicle of transmission of infection between wild species and humans, the virus undergoes significant changes and adaptations during replication within the poultry host. These mutations may increase the virus’s affinity for poultry hosts, and, in the presence of human poultry workers, may facilitate viral adaptation to humans.
The recent H5N1 situation illustrates how both of these mechanisms can occur in tandem—how domesticated animals can act simultaneously as a vector and a locus for pathogen evolution. H5N1 demonstrates how a viral challenge emerged from wildlife, adapted to domestic poultry, and after circulating in these populations, acquired limited ability to infect humans. Notably, recent evidence of the North American H7N2 subtype’s increased affinity for human cellular receptors implies that the virus has adapted following contact between infectious domesticated poultry and human hosts (Belser et al., 2008).
HPAI also exemplifies how ecological conditions of animal husbandry and the food production supply chain can influence health risks for human populations worldwide. In the industrial farm, pathogens can move by unregulated and unrecognized pathways, such as on airborne dust, via nuisance insects, in animal wastes utilized in agriculture and aquaculture, in contaminated water, and by wild animals (Graham et al., 2008). While individual countries have taken steps to contain outbreaks at the farm level and to reduce local dissemination of HPAI, the globalized nature of the food animal production industry and supply chain must be recognized for its role in augmenting rapid transmission of pathogens across long distances.
This article reviews some of the major organizational and ecological changes in food animal production that have occurred over the past 50 years, starting in the USA and now influencing food animal production worldwide, in the context of our current understanding of the emergence of novel influenza A viruses. We then review direct and indirect evidence on the limitations of biosecurity and biocontainment in the industrial model that are relevant to the transmission of influenza viruses and other pathogens into, among, and from industrial food animal production systems. Finally, we analyze the incentives associated with the management of animal and public health risks with an eye towards designing policy that recognizes the importance of economic incentives and the industrial food animal sector.
Modern Industrial Food Animal Production
In most societies, increasing affluence, coupled with global population growth, intensifies consumer demand for livestock-derived protein (Popkin and Du, 2003; McMichael et al., 2007). In the USA, growth in wealth and population coincided with increased efficiencies in animal production and government subsidies to agriculture, contributing to changes in the price and availability of livestock-derived food products (Pollan, 2006). The industrialization of the industry has now spread to much of the world (World Bank, 2005), resulting in dramatic changes in the ecological and organizational landscape of food animal production from 1918 to the present (Silbergeld et al., 2008). Although beyond the scope of this article, the growing centralization of the food animal production industry also has ramifications for food security particularly in the developing world, where reduced access to markets and increased regulations limit small-holder animal production and availability of animal protein.
Industrial food animal production involves high throughput animal husbandry. Thousands of animals of similar genotypes are raised for one purpose (such as pigs, layer hens, broiler chickens, ducks, turkeys) with rapid population turnover at one site under highly controlled conditions, often in confined housing, with nutrient dense and artificial feeds replacing access to forage crops.
Globally, pig and poultry production are the fastest growing and industrializing livestock subsectors with annual production growth rates of 2.6% and 3.7% over the past decade (Table 1). In the industrialized countries, the vast majority of chickens and turkeys are now produced in houses in which 15,000–70,000 birds are confined throughout their lifespan. Increasingly, pigs and cattle are also raised under similar conditions of confinement and high density.
Table 1.
Changes in Global Human Population, Pig and Poultry Inventories, and Production and International Trade of Pig and Poultry Meat between 1996 and 2005a
| 1996 | 2005 | Annual change (%) | |
|---|---|---|---|
| Human population | 5762 | 6451 | 1.1 |
| Inventory | |||
| Pigs (million) | 859 | 963 | 1.1 |
| Poultry (million) | 14,949 | 18,428 | 2.1 |
| Production | |||
| Pig meat (thousand tons) | 79,375 | 103,226 | 2.6 |
| Poultry meat (thousand tons) | 56,408 | 81,856 | 3.7 |
| International trade | |||
| Pig meat (thousand tons) | 6398 | 9557 | 4.0 |
| Poultry meat (thousand tons) | 5359 | 9234 | 5.3 |
aSource: FAOSTAT (2007)
For poultry and pigs, industrial production is organized in stages with separate primary breeders, multipliers, and producers (often contract growers). A small number of globally operating companies form the apex of the breeding pyramid. The feeds supplied to animals in industrial operations are highly formulated and substantially different from the foraged feeds traditionally available to these same species (Sapkota et al., 2007). This sector is also dominated by a small number of commercial entities.
The consolidation of poultry and pig production was undertaken for reasons of competitive advantage. This has greatly affected the geography of food animal populations. In the USA, poultry production is highly concentrated in the southeastern states, with more than 40% of total production occurring in Georgia, Arkansas, and Alabama (USDA, 2008). Pig production is concentrated in some of these same states. Similar trends have occurred worldwide (Figure 2).
Figure 2.
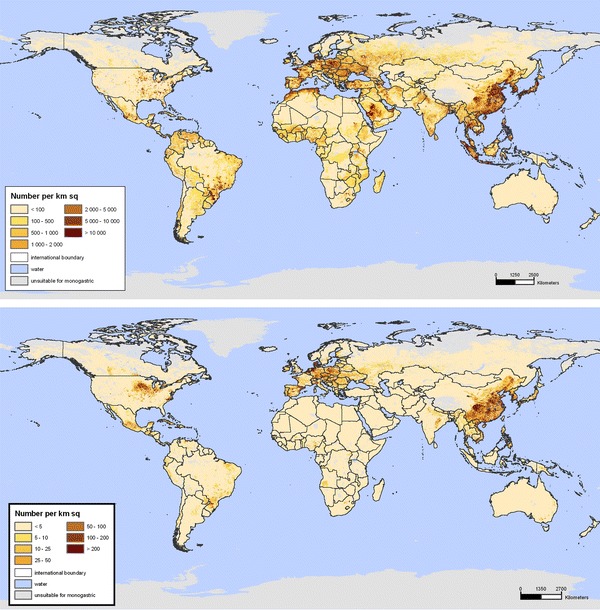
Global poultry (top) and swine (bottom) distributions (FAO, 2007).
The geographical concentration of pig and poultry production has also increased both regional and global trade and movement of pig and poultry meat products, which over the past decade has increased at an average annual rate of 4.0% and 5.3%, respectively (Table 1). In the industrial model, different production stages are often undertaken at different sites, requiring a significant amount of live animal transfers, some of which cross national borders. In 2005, for example, more than 25 million live pigs, i.e., more than 2 million pigs per month, were traded internationally (FAOSTAT, 2007).
After the 2007 HPAI H5N1 outbreak in Sussex, UK, investigations revealed extensive international transfers of hatching eggs, birds, and poultry products between facilities in the UK and Hungary, highlighting the implications of these transfers for long-distance pathogen transmission (Lucas, 2007). The geographic concentration of animal slaughter operations has also increased average distances for transport to slaughter (MLC, 2001; Burrell, 2002). The poultry trade has been implicated in the cross-border spread of H5N1 in Asia and Africa (Kilpatrick et al., 2006).
The geographic intensity and coincident location of industrial pig and poultry production, along with the frequent movement of animals between production stages, provides significant opportunities for interactions between large populations of confined poultry and pigs, which may contribute to the evolution and transmission of pathogens, including zoonotic agents. This may be of particular relevance to the evolution of human-to-human transmissible avian influenza, as suggested by genetic analyses in which both avian and swine influenza viral elements can be detected in farm workers (Gray et al., 2007; Ma et al., 2007), as well as in strains from human outbreaks, including 1918 strain (Vana and Westover, 2008).
Furthermore, animals held in confinement produce large amounts of waste, estimated to exceed 314 million metric tons in the USA each year (Gerba and Smith, 2005). Much of this waste, which contains large quantities of pathogens, is disposed of on land without any requirements for pretreatment, posing an opportunity for human contact (Zheng et al., 2006; Nachman et al., 2005) and transmission to wild animals, both avian and mammalian. As shown in Figure 3, for example, open cesspits of hog waste—called “lagoons”—may attract wild avians to the poultry environment. Wastes are often used for land-based cultivation of finfish in aquaculture (Little and Edwards, 2003), which results in the creation of artificial wetlands and thereby increases direct opportunities for contact with wild avians (Figure 4). This was noted as a risk factor for emergence and outbreaks of HPAI in Vietnam (Pfeiffer et al., 2007).
Figure 3.

Seagulls and egrets at a hog waste lagoon near Lima, Peru (courtesy of C.S. Arriola).
Figure 4.

a: Duck house with exit onto fish pond, Thailand (courtesy of S. Kasemsuwan). b: Chicken house over fish pond, Cambodia (courtesy of M.J. Otte). c: Ducks on fish pond, Vietnam (courtesy of J. Hinrichs).
Industrial Poultry Ecosystems and the Emergence of Novel Influenza A Viruses
Wild aquatic birds are believed to be the primary reservoir of influenza A viruses, and all influenza A viruses in mammals likely have ancestral links to avian lineages (Webby and Webster, 2001; Alexander, 2006). An important feature of influenza A viruses is their capacity to undergo molecular transformation through recombination and reassortment, which facilitates adaptation to new host populations and thereby the potential to cause major disease outbreaks in humans and other species (Vana and Westover, 2008). Influenza A viruses are classified by subtypes on the basis of their hemagglutinin (HA) and neuraminidase (NA) antigens and their pathogenicity to chickens. Strains that cause severe disease and high levels of mortality are classified as highly pathogenic avian influenza while viruses causing milder disease in domesticated poultry are classified as low pathogenic avian influenza (LPAI).
The introduction of LPAI viruses into domestic poultry populations usually requires direct or indirect contact with infectious wild waterfowl or from wild waterfowl to domestic ducks (Alexander, 2006). Incursions of LPAI virus into domestic poultry have been reported over the past decade, mostly in North America and Europe, but also in Mexico, Chile, and Pakistan, as summarized by Capua and Alexander (2004).
The transition from LPAI to HPAI can result from a single point mutation affecting the hemagglutinin surface protein. The probability of such a mutation is amplified in the setting of industrial poultry production due to the rapid viral replication that occurs in an environment of thousands of confined, susceptible animals. In Mexico in 1994, a LPAI H5N2 virus mutated into a HPAI virus and spread to Guatemala in 2000 and to El Salvador in 2001, presumably via trade in poultry (Lee et al., 2004). LPAI H5N2 is now established in domestic chicken populations in Central America. In both the 2003 H7N7 HPAI epidemic in the Netherlands (Stegeman et al., 2004) and the 2004 H7N3 HPAI epidemic in British Columbia, Canada (Power, 2005), LPAI infections in poultry preceded the emergence of HPAI in different poultry houses on the same commercial farms. In Italy, the 1999/2000 H7N1 HPAI epidemic was preceded by 199 reported outbreaks of LPAI H7N1 in the same region.
Pigs may potentially assume a separate important role in the emergence of novel influenza A viruses, as they can be infected by both avian and human viruses (Schulz et al., 1991; Kida et al., 1994; Alexander, 2006). Gilchrist et al. (2007) note the proximity of concentrated poultry and swine operations as a source of disease risk from influenza A viruses, although to date there have only been reports of avian influenza viruses in pigs, not swine influenza in poultry. Classical H1N1 swine influenza viruses are very similar to the virus implicated in the 1918 human influenza pandemic and circulate predominantly in the USA and Asia. H3N2 viruses of human origin have been isolated from pigs in Europe and the Americas shortly after their emergence in humans (Webby and Webster, 2001), and are now endemic in pigs in southern China (Peiris et al., 2001), where they cocirculate with H9N2 viruses with the potential of reassortment with H5N1. Evidence for the concurrent circulation of H1N2, H1N1, and H3N2 influenza A viruses in pigs has been reported from Spain (Maldonado et al., 2006). In the United States, outbreaks of respiratory disease in swine herds have been caused by influenza A viruses which arose from reassortment of human, swine, and avian viral genes (Zhou et al., 1999). Evidence for viral reassortment of avian, human, and swine influenzas within pigs has been published by Shieh et al. (2008), Zhou et al. (1999), and Ma et al. (2007).
Biosecurity in Industrial Animal Systems
Biosecurity is broadly defined as any practice or system that prevents the spread of infectious agents from infected to susceptible animals, or prevents the introduction of infected animals into a herd, region, or country in which the infection has not yet occurred (Radostits, 2001). Farm biosecurity combines “bioexclusion,” i.e., measures for preventing a pathogen from being introduced to a herd/flock, and “biocontainment,” which addresses events after introduction, i.e., the ability for a pathogen to spread among groups of animals at a farm or, more generally, in terms of releases from the farm (Dargatz et al., 2002).
Disease transmission between farms depends on the combination of individual bioexclusion practices and biocontainment measures. The importance of biocontainment is largely determined by the magnitude and direction of resource (feed, water, and air) and waste flows into, within, out of, and between farm populations. In the livestock sector, these flows can be complex because of specialization at different stages of animal production and processing, and intricate formal and informal market chains.
The design and operational requirements of large-scale poultry and swine houses in and of themselves result in compromises of biosecurity. Ventilation of animal houses poses a major, and often unrecognized, gap in biosecurity. The confinement of thousands of animals requires controls to reduce heat and regulate humidity. Poultry and swine houses are ventilated with high-volume fans that result in considerable bidirectional movement of particles, dust, and biomaterials between the animal house and the external environment (Jones et al., 2005). Measurement of aerosol emissions from a broiler operation revealed a million-fold elevated concentration of aerosolized invisible dust near a poultry barn fan as compared to outdoor air in a semi-rural area (Power, 2005). In a study of an LPAI outbreak (H5N2) in Japan in 2006, indirect transmission among groups of chickens was considered to involve dust or bioaerosol movement (Okamatsu et al., 2007). Although little is known about the survival of influenza A viruses on dust particles, high concentrations of infectious avian influenza virus have been detected in air samples from an infected barn (Power, 2005).
Other pathogens have been shown to readily move in and out of poultry and swine houses. Pathogen entry was demonstrated in a recent study of Campylobacter-free broiler flocks, housed in sanitized facilities, using standard biosecurity measures, and fed Campylobacter-free feed and water. Seven out of 10 flocks became colonized with Campylobacter by the time of slaughter, and two flocks were colonized by Campylobacter strains genetically indistinguishable from strains isolated from puddles outside of the facility prior to flock placement (Bull et al., 2006). Campylobacter strains with identical DNA fingerprints to those colonizing broilers have been measured in air up to 30 m downwind of broiler facilities housing colonized flocks (Lee et al., 2002). Although the route of entry was not determined, these studies clearly demonstrate that some pathogens can overcome standard bioexclusion measures.
Insects are another means for pathogen entry to, and exit from, poultry houses. Research carried out during an HPAI outbreak in Kyoto, Japan in 2004, found that flies caught in proximity to broiler facilities where the outbreak took place, carried the same strains of H5N1 influenza virus as found in chickens of an infected poultry farm (Sawabe et al., 2006). A study in Denmark found that as many as 30,000 flies may enter a broiler facility during a single flock rotation in the summer months (Hald et al., 2004), suggesting that the impact of insect vectors may be significant.
Further evidence of the limits of current bioexclusion measures in large-scale industrial poultry operations is provided by HPAI H5N1 outbreaks reported to OIE (http://www.oie.int). For instance, outbreaks of HPAI (H5N1) have been reported in large-scale industrial poultry units with supposedly high biosecurity standards in South Korea (a 300,000 bird unit), in Russia (two 200,000 bird units), and in Nigeria (a 50,000 bird unit) in 2006, and in the UK (a 160,000 turkey unit) in 2007 (http://www.oie.int). Moreover, large(r) industrial flocks appear to be overrepresented in the list of HPAI H5N1 outbreaks reported to OIE as compared to outbreaks in backyard/village flocks, in relation to their respective shares of total national flocks. Around 40% of the HPAI H5N1 outbreaks in domestic poultry reported to OIE between late 2005 and early 2007 occurred in poultry units of 10,000 birds or more (more than 25% occurred in units of more than 10,000 birds), while, even in many OECD countries (e.g., Germany, France, UK, and Belgium), less than 10% of flocks consist of more than 10,000 birds (Table 2).
Table 2.
HPAI H5N1 Outbreaks Reported to OIE, by Flock Type of Index Case, 2003–2007a
| Year | Type/strain | Country | Index case/primary outbreak(s) | Comment | |
|---|---|---|---|---|---|
| Commercial | Backyard | ||||
| 2003 | H5N1 | Vietnam | x | Two poultry breeding farms | |
| 2003 | H5N1 | Korea | x | Broiler-breeder farm, spread to 13 more farms (total of 123,888 birds) | |
| 2004 | H5N1 | Cambodia | x | Layer farm (7500 birds) in Phnom Penh area | |
| 2004 | H5N1 | Indonesia | Mostly layer/breeder hens | ||
| 2004 | H5N1 | Japan | x | Layer farm, total of four farms affected, three commercial and one hobby, total of 274,553 cases | |
| 2004 | H5N1 | Lao PDR | x | “Sector 3” around Vientiane, nearly entire “commercial” sector wiped out | |
| 2004 | H5N1 | Malaysia | x | Fighting cocks on Thai border | |
| 2004 | H5N1 | Thailand | x | Layer flock (66,350 birds) “traditional farming practice” | |
| 2005 | H5N1 | Turkey | x | Free-range turkey farm (2500 birds) | |
| 2005 | H5N1 | Ukraine | x | Five village flocks (1800–6000 birds each) | |
| 2006 | H5N1 | Afghanistan | x | Small backyard farm (187 birds) | |
| 2006 | H5N1 | Albania | x | Village (backyard) flocks (60 birds) | |
| 2006 | H5N1 | Burkina Faso | x | Village (backyard) flocks (130 guinea fowl) | |
| 2006 | H5N1 | Cameroon | x | Three village (backyard) duck flocks (58 birds) | |
| 2006 | H5N1 | Denmark | x | Mixed backyard flock (102 birds) | |
| 2006 | H5N1 | Djibouti | x | Backyard flock (22 birds) | |
| 2006 | H5N1 | Egypt | Massive onset both in commercial and backyard farms | ||
| 2006 | H5N1 | France | x | Turkey producer (~10,000 turkeys) | |
| 2006 | H5N1 | India (Maharashtra) | x | Backyard also affected | |
| 2006 | H5N1 | Ivory Coast | x | Village (backyard) flocks (17 birds) | |
| 2006 | H5N1 | Iraq | x | Village (backyard) flocks (3150 birds) | |
| 2006 | H5N1 | Israel | Three commercial flocks (62,000 birds) and one village flock (6500 birds) | ||
| 2006 | H5N1 | Jordan | x | Farm, 20,075 birds | |
| 2006 | H5N1 | Korea | x | Broiler-breeder flock, 13,200 birds (currently another six commercial flocks infected—from 9000 to 295,000 birds) | |
| 2006 | H5N1 | Lao PDR | x | State farm, parent stock, 6000 birds (different clade from 2004) | |
| 2006 | H5N1 | Malaysia | x | Backyard farms, suspected introduction from Indonesia by boat | |
| 2006 | H5N1 | Myanmar | x | Village flock, 780 birds | |
| 2006 | H5N1 | Niger | x | Village flock, 20,000 birds | |
| 2006 | H5N1 | Nigeria | x | One commercial layer farm, 46,000 birds | |
| 2006 | H5N1 | Pakistan | x | Two commercial farms with 10,000 and 16,000 birds | |
| 2006 | H5N1 | Palestinian Auton. Terr. | x | Eight commercial farms, 1900 to 29,000 birds | |
| 2006 | H5N1 | Romania | x | x | One commercial farm (80,000 birds) and four village flocks |
| 2006 | H5N1 | Russia | x | Two commercial farms, >200,000 birds each | |
| 2006 | H5N1 | Serbia and Montenegro | x | One backyard flock with 23 birds | |
| 2006 | H5N1 | Sudan | x | Two commercial farms, 35,000 and 3400 birds | |
| 2007 | H5N1 | Hungary | x | One geese flock (3355 geese) | |
| 2007 | H5N1 | Japan | x | One commercial farm, 12,000 birds | |
| 2007 | H5N1 | Kuwait | Four small farms, 15, 47, 114, and 670 birds | ||
| 2007 | H5N1 | Lao PDR | One layer duck farm (1380 ducks) and one backyard flock (54 birds) | ||
| 2007 | H5N1 | Myanmar | x | A commercial layer flock, 1360 birds | |
| 2007 | H5N1 | Pakistan | x | Two backyard flocks with 40 and 187 birds, respectively | |
| 2007 | H5N1 | Russia | x | Three village flocks with 41, 49, and 57 birds | |
| 2007 | H5N1 | UK | x | Turkey farm (~160,000 turkeys); trade link to commercial farm in Hungary | |
aSource: OIE WAHID Interface; Available: http://www.oie.int/wahid-prod/public.php?page=home [accessed August 9, 2008]
It is likely that some of the overrepresentation of large industrial-type flocks in reported outbreaks is due to ascertainment bias because outbreaks are more likely to be detected and reported in large-scale operations than in backyard systems. Nevertheless, it demonstrates that, whatever the source of the virus (wild avians, backyard poultry, and other commercial units), bioexclusion measures implemented by some large-scale industrial poultry units, including those in industrialized countries, may be insufficient to protect against avian influenza incursion when challenged.
Once poultry are infected, biocontainment poses a substantial challenge, even in countries with advanced animal-health services, and depends on early detection of outbreaks and action before the virus has spread widely in infected premises. Once influenza A viruses have entered industrial production facilities, they can be transferred among operations via contaminated shipping containers and trucks, as observed by Rule et al. (2008) in a study of bacteria released from poultry transport trucks in the USA. Given that a gram of infected feces can contain as many as 10 billion infectious virus particles, a small amount of contaminated fecal material or litter adhering to boots, clothing, or equipment may be sufficient to transmit virus from an infected to a susceptible flock (Power, 2005).
This empirical evidence of suboptimal biosecurity among commercial operations points to a need for greater oversight and/or regulation of biosecurity of industrial poultry production. For instance, Power (2005) reported that a survey of commercial broiler and table egg farm operators in Fraser Valley, Canada indicated that more than three-quarters did not provide disinfection footbaths nor did they require a change of clothes/coveralls by employees on entering their barns. In Maryland, USA, Price et al. (2007) found that poultry workers are provided little or no protective clothing or opportunities for personal hygiene or decontamination on site, and that almost all take their clothes home for washing.
Dynamics and Control of HPAI in Industrial Poultry Areas
The 1999–2000 H7N1 epidemic in northern Italy, the 2003 H7N7 epidemic in the Netherlands, and the 2004 H7N3 epidemic in the Fraser Valley (British Columbia, Canada) highlight the difficulties faced by animal health authorities when HPAI infects flocks in densely populated poultry production areas (DPPAs). Table 3 provides a summary of these three epidemics.
Table 3.
Summary of HPAI Outbreaks in Densely Populated Poultry Production Areas
| Farm type | Italya | Netherlands | Canada | |||
|---|---|---|---|---|---|---|
| 1999–2000 | 2003 | 2004 | ||||
| Industrial | Backyard/hobby | Industrial | Backyard/hobby | Industrial | Backyard/hobby | |
| Farms in the affected area | 3271 | NA | 1362 | 17,431 | ~800 | 533 |
| Farms declared infected | 382 | 10 | 233 | 22 | 42 | 11 |
| Infection risk | 12% | NA | 17% | 0.1% | ~5% | 2% |
| Farms depopulated | All in 5500 km2 | 1255 | 17,421 | 410 | 533 | |
| Proportion farms depopulated | 100% | 92% | 100% | ~50% | 100% | |
| Birds culled | ~16 million | ~30 million | 13.6 million | 17,977 | ||
aFigures are for Veneto and Lombardia, the hardest hit provinces; source: I. Capua, personal communication (JO)
In all three epidemics, animal health authorities noted the high geographic density of poultry farms, frequent contact among farms by trucks, and low levels of biosecurity practiced by some operators as having been associated with the considerable spread of virus (Capua et al., 2002; Stegeman et al., 2004; Power, 2005). Retrospective analysis of between-flock transmission in the 2003 outbreak in the Netherlands and two outbreaks in Italy in 2003–2004 estimated reproduction ratios (Rh, the average number of secondary infections caused by one infectious flock) of 6.5 in the Netherlands, and 3.1 and 2.9 for two distinct regions in Italy, during the outbreak prior to the implementation of control measures (Stegeman et al., 2004; Capua and Marangon, 2007). This clearly indicates that standard bioexclusion and biocontainment measures in a number of the predominantly industrial flocks were insufficient to prevent disease spread, and that disease detection or reporting was delayed. For caged layers, for example, Capua and Alexander (2006) cite a flock incubation period of up to 18 days, which, in areas with intense between-farm traffic, provides sufficient time for extensive movement of the virus.
Control of the three epidemics was only achieved through massive depopulation of commercial and backyard/hobby flocks (vaccination was not applied). Retrospective analysis of the Dutch outbreak also revealed that between-flock transmission continued even after the implementation of strict movement controls in the affected areas. The authors concluded that containment of the epidemic was more likely to be the result of the depletion of susceptible flocks by depopulation than the reduction of the transmission rate through biocontainment measures (Stegeman et al., 2004).
The lower probability of infection of backyard/hobby flocks compared to that of industrial flocks in the Dutch and Canadian epidemics is consistent with findings from the HPAI epidemic in Thailand in 2004, and the 2002 outbreak of Newcastle disease in Denmark (Otte et al., 2007). This suggests that limits in biosecurity and inter-farm commercial transactions are an important route for disease transmission between industrial farms.
Food Animal Production Workers at Risk
A number of recent studies demonstrate that influenza A viruses from animals can move across the animal:human interface in the context of food animal production and processing. In a comprehensive study of the 1997 H5N1 outbreak in Hong Kong, only occupational tasks involving contact with live poultry were associated with increased risks of seropositivity, and the probability of carrying H5 antibodies increased with increased numbers of such occupational contacts (Bridges et al., 2002). A study in Italy found anti-H7 antibodies in 3.8% of serum samples from poultry workers during the 2003 LPAI H7N3 outbreak (Puzelli et al., 2005). A study of persons reporting influenza-like illness in the Netherlands, during the 2003 H7N7 outbreak in poultry there, documented highest levels of active H7 infection among poultry cullers (41.2%), followed by veterinarians (26.3%), and farmers and their family members (14.7%) (Koopmans et al., 2004). Among swine farmers, Myers et al. (2006), observed higher titers of H1N1 and H1N2 antibodies and greatly elevated risk of seropositivity to these two influenza A viruses (35.3 and 13.8 odds ratios, respectively), as compared to community referents. These studies suggest that workers in industrial food animal settings may act as a “bridge” for influenza A viruses between animal and human communities at large (Saenz et al., 2006; Graham et al., 2008).
Risk Management Strategies at the Animal:Human Interface
Given the role industrial production plays in sustaining and transmitting influenza A viruses, designing interventions specifically targeted to the industry is critical. For interventions to be effective, particular attention must be paid to the incentives driving behavior within the industrial food animal production network.
Animal and public health risks have a complex relationship with economic incentives. Generally speaking, incentives for risk reduction are associated with a “virtuous cycle” of product quality, reputation, and profit. Behaviors that increase risk usually arise from uncertainty, loss aversion, or illicit profit incentives.
Disease risk can be considered as the outcome of an initial process of infection, followed by within-farm transmission, exposure of other actors in the production process, and reaction. The magnitude of each of these processes can be positively or negatively affected by economic incentives and policy interventions. For example, the risk posed by an infectious disease related to food animal production may be influenced by:
Providing incentives and introducing regulations to promote adoption of practices that reduce the probability of initial outbreaks (e.g., bioexclusion measures);
Introduction of incentives and standards that facilitate early detection, on-farm containment, and eradication;
Establishing incentives and standards to reduce release from farms and other routes of exposure of others (e.g., biocontainment measures); and
Developing strategies to mitigate disease impact, for example, through emergency or preventive vaccination in high-risk areas.
Outbreaks of HPAI and other livestock diseases have economic repercussions reaching far beyond primary producers. These impacts are, to a large extent, a result of public and private responses to the risk (real or perceived) of the disease and its potential effects, rather than to the direct on-farm impact. Thus, any control program needs to take into account this plurality of stakeholder reactions and interests, as well as their potential to contribute to (as well as undermine) control programs.
A critical issue of incentives and disincentives relates to early detection of infection in an animal population. The importance of early detection cannot be overstated, as the magnitude of disease epidemics is exponentially related to the time elapsed between pathogen introduction and implementation of control measures. Timely reaction heavily relies on early detection and disclosure by those in daily contact with food animals, however, current disease control policy tends to discourage this behavior.
Compensation schemes that offer equal compensation for lost animals, irrespective of timing of disclosure, fail to create incentives consistent with the policy objective of early reaction (Graming et al., 2006). Likewise, depopulation of entire premises in the event of selected pathogen entry into a poultry house does not offer an incentive for reporting or for major investments in within-farm biocontainment (let alone between-farm biocontainment). Furthermore, compensation for depopulation usually only covers (partial) costs to producers that are directly affected by the depopulation, rather than costs to farmers who may be the first to detect signals of HPAI infection. Compensation and infection control policies should be designed to coordinate human behavior and public health goals, rather than set them at odds. Greater consideration of the human behavioral consequences of control strategies, and in particular the participation of social scientists in their design, would be beneficial in facilitating early detection and control.
As it can be more profitable to raise animals in areas where animal feed is abundant, e.g., close to feed mills, areas of high livestock density have emerged in a number of regions worldwide, as discussed earlier. Semi-vertical integration of production processes, where a large company supplies young stock and feed, while farmers provide animal housing and labor, has often not been accompanied by systematic spatial planning of the units in the system. Although spatial concentration is convenient from an organizational point of view, as illustrated in the case of the HPAI outbreaks in DPPAs, it has serious drawbacks for the control of epidemic diseases. In addition to economics, a consideration of disease control should play a role in siting of animal production facilities, and greater participation of local and central governments in these siting decisions may be valuable in this regard.
The benefits of reducing the risks of highly contagious diseases are shared by all market participants. But as achievement and maintenance of disease prevention is heavily dependent on individual behavior, industrial food production stakeholders may become locked into a “Nash equilibrium” in which no one has anything to gain by changing their behavior unless others change theirs as well. The result is a tragedy of the commons, in which actions that benefit the individual have a negative impact on the system as a whole. Public intervention is required to align individual and societal interests, but to do so successfully requires detailed understanding of the individual incentives of market participants and of the full set of consequences of potential interventions.
Conclusions
An ecosystem approach has been useful in understanding the emergence of zoonotic diseases and identifying opportunities for control (Taylor et al., 2001). However, to date, there has been little consideration of industrial food animal production as an ecosystem, in its own right, for understanding emerging infectious disease. The recent concern regarding HPAI provides a valuable opportunity to demonstrate the importance of including this system in a comprehensive examination of biological, as well as economic and social, motivators with implications for both animal and human health.
In applying an ecosystem perspective to avian influenza, attention to systems-level dynamics is critical. Interactions between food animals, pathogens, and humans must be viewed as part of a dynamic whole, and the interface between them better understood. From this perspective, the role of collaborations between industry, government, and farm communities is increasingly important in establishing regulations to guide the development of the food animal industry to reduce the risk of disease emergence.
Other lessons to draw from an ecosystem approach are the importance of sustainability in environmental systems and the importance of adopting a long-term perspective. As food production becomes increasingly international, local public health risks become global ones. Viewing infectious disease risk as a facet of food production technologies, that can compromise their sustainability, may encourage the development of production methods that are less vulnerable to this risk in the long term.
However, food animal production is also inherently an economic activity, driven by financial incentives and profit motives. These economic forces exist in parallel with the biological pressures that moderate pathogen evolution and spread. Improving the sustainability of current food animal production methods relies on a clear understanding of these economic incentives and their relationship to biological drivers of disease emergence. Policies that address the economic realities, relationships, and drivers in industrial food animal production, and reshape economic incentives accordingly, may prove most fruitful in reducing overall disease risk.
Footnotes
This article is a modified version of a Research Report of the Pro-Poor Livestock Initiative of the Food and Agriculture Organization (FAO), originally published in June 2007 at: http://www.fao.org/AG/AGAINFO/programmes/en/pplpi/docarc/rep-hpai_industrialisationrisks.pdf. Full permission to reproduce this work has been granted from the FAO.
References
- Alexander DJ. Avian influenza viruses and human health. Developmental Biology. 2006;124:77–84. [PubMed] [Google Scholar]
- Belser JA, Blixt O, Chen LM, Pappas C, Maines TR, Hoeven N, et al. Contemporary North American influenza H7 viruses possess human receptor specificity: implications for virus transmissibility. Proceedings of the National Academy of Sciences of the United States of America. 2008;105:7558–7563. doi: 10.1073/pnas.0801259105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bridges CB, Lim W, Hu-Primmer J, Sims L, Fukuda K, Mak KH, et al. Risk of influenza A (H5N1) infection among poultry workers, Hong-Kong, 1997–1998. Journal of Infectious Diseases. 2002;185:1005–1010. doi: 10.1086/340044. [DOI] [PubMed] [Google Scholar]
- Bull SA, Allen VM, Domingue G, Jørgensen F, Frost JA, Ure R, et al. Sources of Campylobacter spp. colonizing housed broiler flocks during rearing. Applied and Environmental Microbiology. 2006;72:645–652. doi: 10.1128/AEM.72.1.645-652.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burrell A. Animal disease epidemics: implications for production, policy and trade. Outlook on Agriculture. 2002;31:151–160. [Google Scholar]
- Capua I, Mutinelli F, Pozza MD, Donatelli I, Puzelli S, Cancellotti FM. The 1999–2000 avian influenza (H7N1) epidemic in Italy: veterinary and human health implications. Acta Tropica. 2002;83:7–11. doi: 10.1016/S0001-706X(02)00057-8. [DOI] [PubMed] [Google Scholar]
- Capua I, Alexander DJ. Human health implications of avian influenza viruses and paramyxoviruses. European Journal of Clinical Microbiology and Infectious Diseases. 2004;23:1–6. doi: 10.1007/s10096-003-1059-3. [DOI] [PubMed] [Google Scholar]
- Capua I, Alexander DJ. The challenge of avian influenza to the veterinary community. Avian Pathology. 2006;35:189–205. doi: 10.1080/03079450600717174. [DOI] [PubMed] [Google Scholar]
- Capua I, Marangon S. The use of vaccination to combat multiple introductions of notifiable avian influenza viruses of the H5 and H7 subtypes between 2000 and 2006 in Italy. Vaccine. 2007;25:4987–4995. doi: 10.1016/j.vaccine.2007.01.113. [DOI] [PubMed] [Google Scholar]
- Dargatz DA, Garry FB, Traub-Dargatz JL. An introduction to biosecurity of cattle operations. Veterinary Clinics of North America. Food Animal Practice. 2002;18:1–5. doi: 10.1016/S0749-0720(02)00002-6. [DOI] [PubMed] [Google Scholar]
- Daszak P, Cunningham AA, Hyatt AD. Emerging infectious diseases of wildlife—threats to biodiversity and human health. Science. 2000;287:443–449. doi: 10.1126/science.287.5452.443. [DOI] [PubMed] [Google Scholar]
- FAO (2007) Gridded Livestock of the World, 2007, Rome: Food and Agriculture Organization of the United Nations, Animal Production and Health Division. Available: http://www.fao.org/docrep/010/a1259e/a1259e00.htm
- FAOSTAT (2007) Statistical Yearbook (FAOSTAT), 2007. Rome: Food and Agriculture Organization of the United Nations. Available: http://www.fao.org/statistics/yearbook/vol_1_1/index_en.asp [accessed August 28, 2008]
- Field HE, Mackenzie JS, Daszak P. Henipaviruses: emerging paramyxoviruses associated with fruit bats. Current Topics in Microbiology and Immunology. 2007;315:133–159. doi: 10.1007/978-3-540-70962-6_7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gaze W, O’Neill C, Wellington E, Hawkey P. Antibiotic resistance in the environment, with particular reference to MRSA. Advances in Applied Microbiology. 2008;63:249–280. doi: 10.1016/S0065-2164(07)00007-X. [DOI] [PubMed] [Google Scholar]
- Gerba CP, Smith JE., Jr Sources of pathogenic microorganisms and their fate during land application of wastes. Journal of Environmental Quality. 2005;34:42–48. [PubMed] [Google Scholar]
- Gilchrist MJ, Greko C, Wallinga DB, Beran GW, Riley DG, Thorne PS. The potential role of concentrated animal feeding operations in infectious disease epidemics and antibiotic resistance. Environmental and Health Perspectives. 2007;115:313–316. doi: 10.1289/ehp.8837. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Graham JP, Leibler JH, Price LB, Otte JM, Pfeiffer DU, Tiensin T, et al. The animal:human interface and infectious disease in industrial food animal production: rethinking biosecurity and biocontainment. Public Health Reports. 2008;123:282–299. doi: 10.1177/003335490812300309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gramig BM, Barnett BJ, Skees JR, Black JR. Incentive compatibility in risk management of contagious livestock disease. In: Koontz SR, Hoag DL, Thilmany DD, Green JW, Grannis JL, editors. The Economics of Livestock Disease Insurance: Concepts, Issues and International Case Studies. Wallingford, UK: CABI Publishing; 2006. pp. 39–52. [Google Scholar]
- Gray GC, McCarthy T, Capuano AW, Setterquist SF, Olsen CW, Alavanja MC. Swine workers and swine influenza virus infections. Emerging Infectious Diseases. 2007;13:1871–1878. doi: 10.3201/eid1302.060680. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hald B, Skovgård H, Bang DD, Pedersen K, Dybdahl J, Jespersen JB, et al. Flies and Campylobacter infection in broiler flocks. Emerging Infectious Diseases. 2004;10:1490–1492. doi: 10.3201/eid1008.040129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jones TA, Donnelly CA, Stamp Dawkins M. Environmental and management factors affecting the welfare of chickens on commercial farms in the United Kingdom and Denmark stocked at five densities. Poultry Science. 2005;84:1155–1165. doi: 10.1093/ps/84.8.1155. [DOI] [PubMed] [Google Scholar]
- Kida H, Ito T, Yasuda J, Shimizu Y, Itakura C, Shortridge KF, et al. Potential for transmission of avian influenza viruses to pigs. Journal of General Virology. 1994;75:2183–2188. doi: 10.1099/0022-1317-75-9-2183. [DOI] [PubMed] [Google Scholar]
- Kilpatrick AM, Chmura AA, Gibbons DW, Fleischer RC, Marra PP, Daszak P. Predicting the global spread of H5N1 avian influenza. Proceedings of the National Academy of Sciences of the United States of America. 2006;103:19368–19373. doi: 10.1073/pnas.0609227103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Koopmans M, Wilbrink B, Conyn M, Natrop G, van der Nat H, Vennema H, et al. Transmission of H7N7 avian influenza A virus to human beings during a large outbreak in commercial poultry farms in the Netherlands. Lancet. 2004;363:587–593. doi: 10.1016/S0140-6736(04)15589-X. [DOI] [PubMed] [Google Scholar]
- Lee CW, Senne DA, Suarez DL. Effect of vaccine use in the evolution of Mexican lineage H5N2 avian influenza virus. Journal of Virology. 2004;78:8372–8981. doi: 10.1128/JVI.78.15.8372-8381.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee MD, Sanchez S, Zimmer M, Idris U, Berrang ME, McDermott PF. Class 1 integron-associated tobramycin-gentamicin resistance in Campylobacter jejuni isolated from the broiler chicken house environment. Antimicrobial Agents and Chemotherapy. 2002;46:3660–3664. doi: 10.1128/AAC.46.11.3660-3664.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Little E, Edwards P. Integrated Livestock-Fish Farming Systems. Rome: Food and Agriculture Organization of the United Nations; 2003. [Google Scholar]
- Lucas C (2007) Bird flu’s link with the crazy trade in poultry. Financial Times, February 26, 2007
- Ma W, Vincent AL, Gramer MR, Brockwell CB, Lager KM, Janke BH, et al. Identification of H2N3 influenza A viruses from swine in the United States. Proceedings of the National Academy of Sciences of the United States of America. 2007;104:20949–20954. doi: 10.1073/pnas.0710286104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Maldonado J, Van Reeth K, Riera P, Sitjà M, Saubi N, Espuña E, et al. Evidence of the concurrent circulation of H1N2, H1N1 and H3N2 influenza A viruses in densely populated pig areas in Spain. Veterinary Journal. 2006;172:377–381. doi: 10.1016/j.tvjl.2005.04.014. [DOI] [PubMed] [Google Scholar]
- McMichael AJ, Powles JW, Butler CD, Uauy R. Food, livestock production, energy, climate change and health. Lancet. 2007;370:1253–1263. doi: 10.1016/S0140-6736(07)61256-2. [DOI] [PubMed] [Google Scholar]
- Meat and Livestock Commission (MLC) (2001) The Abattoir and Meat Processing Industry in Great Britain. Supplement to the 1999 Edition. Available: 2001 Meat and Livestock Commission, PO Box 44, Winterhill House, Snowdon Drive, Milton Keynes MK6 1AX, UK
- Myers KP, Olsen CW, Setterquist SF, Capuano AW, Donham KJ, Thacker EL, et al. Are swine workers in the United States at increased risk of infection with zoonotic influenza virus? Clinical Infectious Diseases. 2006;42:14–20. doi: 10.1086/498977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nachman KE, Graham JP, Price LB, Silbergeld EK. Arsenic: a roadblock to potential animal waste management solutions. Environmental and Health Perspectives. 2005;113:1123–1124. doi: 10.1289/ehp.7834. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okamatsu M, Saito T, Mase M, Tsukamoto K, Yamaguchi S. Characterization of H5N2 influenza A viruses isolated from chickens in Japan. Avian Diseases. 2007;51(1 Suppl):474–475. doi: 10.1637/7573-033106R1.1. [DOI] [PubMed] [Google Scholar]
- Otte J, Pfeiffer DU, Tiensen T, Price LB, Silbergeld EK (2007) HPAI risk, biosecurity and smallholder adversity. In: Proceedings of the WPSA Asian Pacific Federation Working Group on Small-Scale Family Poultry Farming Symposium, Bangkok, Thailand, pp 1–8
- Peiris JSM, Guan Y, Markwell D, Ghose P, Webster RG, Shortridge KF. Co-circulation of avian H9N2 and contemporary ‘human’ H3N2 influenza viruses in pigs in Southeastern China: potential for genetic reassortment? Journal of Virology. 2001;75:9679–9686. doi: 10.1128/JVI.75.20.9679-9686.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pfeiffer DU, Minh PQ, Martin V, Epprecht M, Otte MJ. An analysis of the spatial and temporal patterns of highly pathogenic avian influenza occurrence in Vietnam using national surveillance data. Veterinary Journal. 2007;174:302–309. doi: 10.1016/j.tvjl.2007.05.010. [DOI] [PubMed] [Google Scholar]
- Pollan M. The Omnivore’s Dilemma. New York: The Penguin Press; 2006. [Google Scholar]
- Popkin BM, Du S. Dynamics of the nutrition transition towards the animal foods sector in China and its implications: a worried perspective. Journal of Nutrition. 2003;133(11):3898S–3906S. doi: 10.1093/jn/133.11.3898S. [DOI] [PubMed] [Google Scholar]
- Power C (2005) The Source and Means of Spread of the Avian Influenza Virus in the Lower Fraser Valley of British Columbia during an Outbreak in the Winter of 2004—An Interim Report, February 2005. Available: http://www.inspection.gc.ca
- Price LB, Roess A, Graham JP, Baqar S, Vailes R, Sheikh KA, et al. Neurologic symptoms and neuropathologic antibodies in poultry workers exposed to Campylobacter jejuni. Journal of Occupational and Environmental Medicine. 2007;49:748–755. doi: 10.1097/JOM.0b013e3180d09ec5. [DOI] [PubMed] [Google Scholar]
- Pulliam JRC. Viral host jumps: moving towards a predictive framework. EcoHealth. 2008;5:80–91. doi: 10.1007/s10393-007-0149-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Puzelli S, Di Trani L, Fabiani C, Campitelli L, De Marco MA, Capua I, et al. Serological analysis of serum samples from humans exposed to avian H7 influenza viruses in Italy between 1999 and 2003. Journal of Infectious Diseases. 2005;192:1318–1322. doi: 10.1086/444390. [DOI] [PubMed] [Google Scholar]
- Radostits OM (2001) Control of infectious diseases of food-producing animals. In: Herd Health: Food Animal Production Medicine, 3rd ed., Philadelphia: Saunders
- Rule AM, Evans SL, Silbergeld EK. Food animal transport: a potential source of community exposures to health hazards from industrial farming (CAFOs) Journal of Infection and Public Health. 2008;1:33–39. doi: 10.1016/j.jiph.2008.08.001. [DOI] [PubMed] [Google Scholar]
- Saenz RA, Hethcote HW, Gray GC. Confined animal feeding operations as amplifiers for influenza. Vector Borne and Zoonotic Diseases. 2006;6:338–346. doi: 10.1089/vbz.2006.6.338. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sapkota AR, Lefferts LY, McKenzie S, Walker P. What do we feed to food-production animals? A review of animal feed ingredients and their potential impacts on human health. Environmental and Health Perspectives. 2007;115:663–670. doi: 10.1289/ehp.9760. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sawabe K, Hoshino K, Isawa H, Sasaki T, Hayashi T, Tsuda Y, et al. Detection and isolation of highly pathogenic H5N1 avian influenza A viruses from blow flies collected in the vicinity of an infected poultry farm in Kyoto, Japan, 2004. American Journal of Tropical Medicine and Hygiene. 2006;75:327–332. [PubMed] [Google Scholar]
- Schulz U, et al. Evolution of pig influenza viruses. Virology. 1991;183:61–73. doi: 10.1016/0042-6822(91)90118-U. [DOI] [PubMed] [Google Scholar]
- Shieh HK, Chang PC, Chen TH, Li KP, Chan CH. Surveillance of avian, swine influenza in the swine population in Taiwan, 2004. Journal of Microbiology, Immunology and Infection. 2008;41:231–242. [PubMed] [Google Scholar]
- Silbergeld EK, Graham J, Price LB. Industrial food animal production, antimicrobial resistance, and human health. Annual Review of Public Health. 2008;29:151–169. doi: 10.1146/annurev.publhealth.29.020907.090904. [DOI] [PubMed] [Google Scholar]
- Stegeman A, Bouma A, Elbers AR, de Jong MC, Nodelijk G, de Klerk F, et al. Avian influenza A virus (H7N7) epidemic in the Netherlands in 2003: course of the epidemic and effectiveness of control measures. Journal of Infectious Diseases. 2004;190:2088–2094. doi: 10.1086/425583. [DOI] [PubMed] [Google Scholar]
- Taylor LH, Latham SM, Woolhouse ME. Risk factors for human disease emergence. Philosophical Transactions of the Royal Society of London. Series B: Biological Sciences. 2001;356:983–989. doi: 10.1098/rstb.2001.0975. [DOI] [PMC free article] [PubMed] [Google Scholar]
- United States Department of Agriculture (USDA) (2008) National Agricultural Statistics Service Quick Stats, 2008. Available: http://www.nass.usda.gov/Data_and_Statistics/Quick_Stats/ [accessed June 18, 2008]
- Vana G, Westover KM. Origin of the 1918 Spanish influenza virus: a comparative genomic analysis. Molecular Phylogenetics and Evolution. 2008;47:1100–1110. doi: 10.1016/j.ympev.2008.02.003. [DOI] [PubMed] [Google Scholar]
- Webby RJ, Webster RG. Emergence of influenza A viruses. Philosophical Transactions of the Royal Society of London. Series B: Biological Sciences. 2001;356:1817–1828. doi: 10.1098/rstb.2001.0997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woolhouse MEJ. Population biology of emerging and re-emerging pathogens. Trends in Microbiology. 2002;10:S3–S7. doi: 10.1016/S0966-842X(02)02428-9. [DOI] [PubMed] [Google Scholar]
- World Bank . Managing the Livestock Revolution: Policy and Technology to Address the Negative Impacts of a Fast-Growing Sector, June 2005; Report No. 32725-GLB. Washington, DC: The World Bank; 2005. [Google Scholar]
- Zheng Y, Ge S, Zhang J, Guo Q, Ng MH, Wang F, et al. Swine as a principal reservoir of hepatitis E virus that infects humans in eastern China. Journal of Infectious Diseases. 2006;193:1643–1649. doi: 10.1086/504293. [DOI] [PubMed] [Google Scholar]
- Zhou NN, Senne DA, Landgraf JS, Swenson SL, Erickson G, Rossow K, et al. Genetic reassortment of avian, swine, and human influenza A viruses in American pigs. Journal of Virology. 1999;73:8851–8856. doi: 10.1128/jvi.73.10.8851-8856.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]


