Abstract
The arthritis-susceptible DA rat is one of the most commonly used rat strains for genetic linkage analysis and is instrumental for the identification of many genetic loci. Even though DA rats were kept as inbred lines at different institutes and suppliers, it became obvious that the various breeding stocks differed genetically. To be able to compare the results from different linkage studies it is very import to verify the genetic background of the substrains used in those studies. We performed a genetic and phenotypic analysis of two DA substrains, DA/ZtmRhd and DA/OlaHsd, and found several genetic differences. One of the allelic differences between the DA/ZtmRhd and the DA/OlaHsd strain was located at rat chromosome 3, a 17-Mb large fragment, including the peak marker of a previously identified quantitative trait locus (QTL) for collagen-induced arthritis, Cia11. In addition, the substrains exhibited a significant difference in the susceptibility to pristane-induced arthritis (PIA) and disease severity of collagen-induced arthritis and PIA. However, by generating and testing a congenic line, we could demonstrate that phenotypic differences were not due to the contaminating fragment on chromosome 3. Nevertheless, we conclude that DA substrains show distinct genetic differences and caution should be taken when comparing arthritis data from different DA substrains.
Electronic supplementary material
The online version of this article (doi:10.1007/s00335-008-9125-x) contains supplementary material, which is available to authorized users.
Keywords: Quantitative Trait Locus, Pristane, Arthritis Severity, Ncf1 Gene, SSLP Marker
Introduction
Rheumatoid arthritis (RA) is a chronic autoimmune disease that affects 0.5–1% of most populations, with a higher propensity in females (Alamanos and Drosos 2005). RA is a multifactorial disease of unknown etiology but with a strong genetic component (Dieude and Cornelis 2005), and recent studies based on various gene-finding strategies have identified the first genes implicated in the susceptibility to RA (Plenge et al. 2007; The Wellcome Trust Consortium 2007; Worthington 2005). However, many susceptibility genes still remain unknown. Among the difficulties involved in gene identification in complex traits are factors such as variable penetrance, variable relative risk associated to the disease allele, epistasis, genetic heterogeneity of the human population, and complex interactions between environmental and genetic factors (Lander and Schork 1994).
Despite the recent advances in human genetics, animal models of RA remain attractive tools, not only to overcome genetic complexity, but also to permit studies under stable environmental conditions (Holmdahl 2003). Experimental animal models can be used for gene identification, but more important they allow the functional characterization of arthritis-regulating loci and genes, which consequently could result in the development of novel drugs or targets for human RA. Rat models are particularly useful because large numbers of progeny can be generated from inbred strains that differ in their susceptibility to various arthritis models, including collagen-induced arthritis (CIA), adjuvant-induced arthritis (AIA), oil-induced arthritis (OIA), and pristane-induced arthritis (PIA) (Holmdahl et al. 2001). To date, linkage analysis studies in rat models of RA have identified more than 20 loci that are either specific for the given animal model or shared with other arthritis models or with RA in humans (Joe 2006).
The DA rat strain is highly susceptible to most of the experimentally induced models of arthritis and was therefore chosen as the arthritis-susceptible parental strain in a large number of conducted genetic linkage studies. Nevertheless, caution should be taken when comparing physiologic, biochemical, or genetic data among studies in which the same strains but from different breeding facilities have been used. In this study we aimed at characterizing the genetic and phenotypic differences between the two most commonly used DA substrains (DA/Ztm and DA/OlaHsd) in arthritis linkage studies. This approach provided additional information and a better understanding of previously identified quantitative trait loci (QTL) in those strains. Our original colony of inbred DA rats (referred to as DA/ZtmRhd) was established from DA/Ztm breeding pairs and we also used DA rats from Harlan Europe (referred to as DA/OlaHsd). We performed a genetic and phenotypic analysis of DA/ZtmRhd and DA/OlaHsd rats and report here several phenotypic and genetic differences between these strains.
Material and methods
Animals
DA/ZtmRhd rats were kindly provided by Prof. H.J. Hedrich in 1996 from the Zentralinstitut fuer Versuchstierkunde (Hannover, Germany) and were bred for more than 20 generations of brother-sister mating in the animal facility in Lund. DA/OlaHsd rats originating from Harlan Europe (Netherlands) were kept and bred as brother-sister matings for more than five generations in the animal facility in Lund in a climate-controlled environment with 12-h light/dark cycles. In the same facility breeding was performed to produce DA.DA/ZtmRhd-Cia11 (≥N3) congenic rats. Shortly, one DA/ZtmRhd male and two DA/OlaHsd rats were used to produce the F1 generation. Six F1 females were further backcrossed with DA/OlaHsd male rats. After three to four backcross generations, Cia11 heterozygous rats were then intercrossed and only strictly littermate-controlled experiments were performed to ensure that all phenotypic differences originated exclusively from the genetic differences in the Cia11 region. Rats were housed in polystyrene cages containing wood shavings and fed standard rodent chow and water ad libitum. The rats were free from common pathogens including Sendai virus, Hantaan virus, corona virus, reovirus, cytomegalo virus, and Mycoplasma pulmonis. All experiments had been approved by the local ethical committee (Malmö/Lund, Sweden).
Genotyping
DNA was prepared from toe biopsies by alkaline lysis, amplified with fluorescence-labeled SSLP markers by multiplex PCR according to the standard protocol, and analyzed on MegaBACE 1000 (Amersham Pharmacia Biotech). Primer sequences for SSLP markers used for the congenic fragment DA/ZtmRhd, the whole-genome screen of the DA substrains, and fine typing of certain genomic fragments were retrieved from http://www.ensembl.org/index.html. A list of all used SSLP markers and their allele sizes is given in Supplementary Table 1. Single-nucleotide polymorphism (SNP) typing was performed using specific primers designed with PSQ Assay Design software (Biotage, Uppsala, Sweden). The reactions were amplified by PCR using biotinylated forward and reverse primers followed by separation with streptavidin. Then the SNPs were detected in sequencing reactions using Pyrosequencing® equipment according to protocols supplied by the manufacturer (Biotage, Uppsala, Sweden). Inbred and outbred rat strain DNA was retrieved from collections at the Central Animal Laboratory, Hannover (H.J. Hedrich, DA/Ztm, ACI/Ztm), Scanbur (outbred SD/Sca, outbred W/Sca), Taconic (outbred SD/MolTac), animal facilities in Lund (R. Holmdahl, DA/OlaHsd, DA/ZtmRhd, E3/ZtmRhd, LEW.1F/ZtmRhd), Stockholm (J. Lorentzen, DA/ZtmKiru, BN/Kiru), Malmö (H. Luthmann, F344/Swe), and Manhasset (P. Gulko, DA/OlaHsd, DA/BklArbNsi).
Table 1.
Whole-genome scan of DA/ZtmRhd and DA/OlaHsd rats using SSLP markers and regions with clusters of polymorphic SNPs between DA/HanKini and DA/OlaHsd rats
| RNO | Different SSLP allelesa/all SSLP tested | Additional SSLP marker | Regions with polymorphic SNPsb in Mb |
|---|---|---|---|
| 1 | 4/30 | 227.3–228.9 | |
| 2 | 3/15 | 0/4 | 202.8–206.6 |
| 218.3–219.0 | |||
| 3 | 3/14 | 13/26 | 35.6–52.3 |
| 74.8–77.9 | |||
| 4 | 1/16 | ||
| 5 | 2/13 | 0/3 | |
| 6 | 2/17 | ||
| 7 | 0/13 | 45.9–48.4 | |
| 8 | 1/10 | ||
| 9 | 0/8 | ||
| 10 | 3/13 | 0/3 | |
| 11 | 2/11 | ||
| 12 | 1/10 | ||
| 13 | 4/9 | 0/6 | 32.9–36.3 |
| 14 | 2/10 | ||
| 15 | 0/5 | ||
| 16 | 0/8 | ||
| 17 | 1/8 | ||
| 18 | 2/10 | ||
| 19 | 1/7 | ||
| 20 | 1/10 | ||
| X | 2/11 | ||
| Total | 35/248 |
aAlleles with ≥2 bp length differences
bData retrieved from http://www.ensembl.org/index.html
Induction and evaluation of arthritis
Lathyritic collagen II (CII) was purified from Swarm rat chondrosarcoma, grown in male rats receiving β-aminopropionitrile monofumaratic salt in drinking water during the tumor-growing period as previously described (Miller and Rhodes 1982; Smith et al. 1975). Collagen-induced arthritis (CIA) was induced by a single intradermal injection of 75 μg lathyritic rat collagen II dissolved in 50 μl 0.1 M acetic acid and emulsified in 50 μl incomplete Freund’s adjuvant (IFA). Pristane-induced arthritis (PIA) was induced by a single intradermal injection of 50 μl pristane (2,6,10,14-tetramethylpentadecane, ACROS Organics, Geel, Belgium) at the base of the tail. Arthritis was induced in age- and cage-matched rats at the age of 7–9 weeks, and arthritis development was monitored in all four limbs using a macroscopic scoring system. Briefly, one point was given for each swollen and red toe, one point for each affected midfoot, digit, or knuckle, and five points for a swollen ankle (maximum score per limb was 15 and maximum score per rat was 60). The rats were examined in a blinded fashion every second to third day for one month after induction of the disease (Holmdahl 1997). All experiments in congenic rats were carried out with DA/OlaHsd littermate controls originating from F1 intercross between heterozygous congenic rats to ensure that all phenotypic differences in the congenic animals originated exclusively from the genetic differences in the chromosome 3 region.
Blood sampling and detection of antibodies
Peripheral blood was collected from rats after CIA induction at termination day by cutting the tip of the tail. The blood was left for 1 h at 4°C and centrifuged at 13,000 rpm for 10 min. Serum samples were frozen at −20°C until use. For detection of anti-collagen II antibodies (a-CII) in Eu3+LISA, plates were coated with pepsin-digested CII and incubated overnight at 4°C. After blocking with 2% BSA, diluted serum samples were added to the plates and incubated. Thereafter, biotin-labeled mouse anti-rat antibodies (anti-IgG1, anti-IgG2a, anti-IgG2b, anti-IgG2c, PharMingen, BD Bioscience, San Jose, CA, USA; polyclonal anti-rat IgG; Zymed, San Francisco, CA, USA) were added and incubated. Then Europium-labeled streptavidin (in Assaybuffer, Wallac Oy, Turku, Finland) was added. For final detection, Enhancement Solution (Wallac) was added and fluorescence emissions were read using Victor/Wallac (Wallac).
Statistics
The Statview software program was used for all statistical analyses. Incidence of arthritis was analyzed by Fisher’s exact test. The nonparametric Mann-Whitney U test (comparison of two groups) or the Kruskal-Wallis test (comparison of three groups) was used in all other statistical analyses. p values less than 0.05 were considered significant.
Results
Genetic comparison between DA/OlaHsd and DA/ZtmRhd rats
First we determined the degree of genetic heterogeneity of the two most commonly used DA substrains for linkage studies of arthritis. We therefore performed a whole-genome scan of DA/OlaHsd and DA/ZtmRhd rats using 248 well-established SSLP markers covering all 20 autosomal chromosomes and the X chromosome and compared the allele sizes in the two substrains. The marker selection was based on previous results from crosses between E3/ZtmRhd and DA/ZtmRhd rats and the E3 strain also was included in the typing as an internal control. As expected, all 248 initially typed markers were found in homozygous form in all three strains and were polymorphic between E3 and DA/ZtmRhd rats. However, we also found markers that differed in allele size between DA/ZtmRhd and DA/OlaHsd rats. As shown in Table 1, 35 of 248 markers were found to be polymorphic between the two DA substrains. This means that calculated on those markers, the two substrains differed in 14.1% of their genome. When analyzing markers from each rat chromosome (RNO) alone, the variability ranges from 0% (RNO7, 9, 15, 16) to 20% (RNO2, 3, 10, 14, 18) and up to 44% (RNO13). Detailed information on all polymorphic and nonpolymorphic markers is listed in Supplementary Table 1.
In our genome scan we identified seven regions (one on RNO2, 3, 5, 10, X and two on RNO13) where two or three consecutive SSLP markers were polymorphic between the DA substrains. We added additional markers in close proximity to those polymorphic regions. For RNO2, RNO5, RNO10, and RNO13 all additional markers were nonpolymorphic, ruling out the possibility of a large contaminating fragment. However, on RNO3 additional markers were found to be polymorphic, suggesting the presence of a large contaminating fragment in this region. It is well known that SSLP regions exhibit a higher mutation rate than the average genome and, thus, the surprisingly high genetic variability could be the result of mutations in the SSLP regions analyzed. However, it is far more likely that it is the result of contamination of one of the DA substrains with another rat strain. To support this theory, we took advantage of the recently performed whole-genome typing of 20,238 SNPs in 167 inbred rat strains, including DA/OlaHsd (Saar et al. 2008). Although the DA/ZtmRhd substrain was not among those strains, a relatively closely related substrain, DA/HanKini, was included. By comparing this strain with DA/OlaHsd we found seven distinct clusters of polymorphic SNPs on five rat chromosomes (Table 1). While six of those regions with sizes between 1 and 4 Mb were considerably small, we found one cluster of SNPs on RNO3 that was more that 10 Mb. We conclude that DA/OlaHsd and DA/ZtmRhd rats differed at a high number of SSLP and at a large contaminating DNA fragment on RNO3.
Fine-mapping of the contaminating fragment on RNO3
To identify the genetic border of the chromosomal fragment on RNO3 that differed between DA/OlaHsd and DA/ZtmRhd rats, we typed 26 additional markers in the region and generated a physical map that indicates the position of all polymorphic and nonpolymorphic markers (Fig. 1). We found 13 markers differing between DA/OlaHsd and DA/ZtmRhd, including the original peak marker D3Wox9 of the Cia11 QTL. Most of them were clustered between position 35.9 and 49.0 Mb. To verify that this difference is indeed the result of contamination and not due to randomly occurring mutations in the SSLP regions, we typed four SNP markers. Because at that point of investigation only a limited amount of strains had available SNP information and the DA strain was not included, we chose the SNPs randomly. Two of the tested SNPs were found to be polymorphic between the substrains. To compare the contaminating fragment to other strains, we included DA substrains (DA/Ztm, DA/ZtmKiru, DA/AlbNSI, DA/Hsd) from different breeders as well as other rat inbred strains such as BN, ACI, F344, and LEW.1F. First, DA/Ztm, DA/ZtmRhd, and DA/ZtmKiru rats share the same haplotype on RNO3, confirming the close relationship of these substrains (Supplementary Table 2). At the same time, DA/OlaHsd rats from Harlan Europe share the same haplotype with DA/Hsd rats from Harlan USA and DA/AlbNSI rats from the USA. Second, BN and F344 are highly similar to the DA/ZtmRhd haplotype, whereas ACI is nearly identical to the DA/OlaHsd strain. Analyses of all published SNPs in that locus revealed a 100% identical haplotype of DA/OlaHsd and all substrains of ACI, whereas the DA/HanKini haplotype appeared to be unique. Even so, no conclusion of the origin of the contamination can be drawn at that point and extensive SNP typing will be required to do so. In summary, combination of SSLP analyses and available SNP data conclusively identified a contaminating fragment on RNO3 stretching from 35.6 to 52.3 Mb.
Fig. 1.

Genomic map of the contaminating fragment (black line) on RNO3 that differed between DA/ZtmRhd and DA/OlaHsd rats. The border of an uncertain genotype is shown as a dotted line. Nonpolymorphic markers are in regular text and polymorphic markers are in boldface. Allele sizes from polymorphic SSLP and SNP markers are indicated (left side DA/ZtmRhd, right side DA/OlaHsd)
Table 2.
List of all arthritis experiments of DA substrains
| EXP-ID | Immunogen | DA substrains | Incidence of arthritis | Day of onset (mean ± SD)a | Maximal arthritis score (mean ± SD)a |
|---|---|---|---|---|---|
| EXP-01 | 50 μl pristane | DA/OlaHsd | 25/26 | 12 ± 2 | 39 ± 11 |
| DA/ZtmRhd | 13/27* | 14 ± 2* | 25 ± 11* | ||
| EXP-02 | 50 μl pristane | DA/OlaHsd | 26/29 | 12 ± 2 | 46 ± 9 |
| DA/ZtmRhd | 13/27* | 12 ± 1 | 40 ± 8* | ||
| EXP-03b | 150 μl pristane | DA/ZtmRhd | 9/10 | 13 ± 2 | 27 ± 14 |
| EXP-04 | 75 μg CII/100 μl | DA/OlaHsd | 20/20 | 15 ± 2 | 36 ± 18 |
| DA/ZtmRhd | 20/22 | 18 ± 3* | 21 ± 15* | ||
| EXP-05 | 75 μg CII/100 μl | DA/OlaHsd | 18/19 | 14 ± 1 | 53 ± 10 |
| DA/ZtmRhd | 17/17 | 16 ± 2* | 33 ± 17* |
SD = standard deviation
aOnly affected animals included
bExperiment was terminated at day 16
* Significantly different (p < 0.05) from DA/OlaHsd
DA/OlaHsd rat suffered from more severe pristane-induced arthritis
After we clearly established the genetic difference between the substrains, we wanted to address whether they also display a phenotypic difference. Because the DA strain is the most commonly used rat strain in genetic studies of arthritis, we investigated the susceptibility to and severity of PIA, a well characterized model for RA. In two independent experiments we immunized the DA substrains DA/OlaHsd and DA/ZtmRhd with a single intradermal injection of 50 μl pristane, which previously had been found to be the optimal dose to induce arthritis in nearly all DA/OlaHsd rats. Data from the two experiments showed similar results (Table 2) and thus were pooled, revealing a significant difference in arthritis incidence between the two strains (Fig. 2a). Fifty-two of 55 (95%) DA/OlaHsd rats developed arthritis while only 48% (26/54) of DA/ZtmRhd rats developed arthritis (p < 0.0001). In addition, the arthritis severity was significantly reduced in DA/ZtmRhd rats. When all animals were included in the analysis, p < 0.0001 for all scoring days from day 12 to day 21, when the experiment was terminated (Fig. 2b). Because there was such a drastic difference in incidence, we performed the analysis including only affected rats. DA/OlaHsd rats (n = 52) also had a significantly enhanced arthritis severity compared to DA/ZtmRhd rats (n = 26) (Fig. 2c). The p value was less than 0.01 from day 14 to the end of the experiment. However, the day of disease onset was not different between the rat substrains. The low incidence of DA/ZtmRhd was surprising to us because earlier we found that this substrain displayed a 90–100% susceptibility to PIA. However, previous experiments with the DA/ZtmRhd strain induced PIA with a higher dose of pristane (150 μl). To confirm that the lower incidence seen in DA/ZtmRhd strain in this study was due to the lower amount of pristane injected, we immunized ten DA/ZtmRhd rats with 150 μl pristane. The result confirmed the data from previous experiments using the DA/ZtmRhd strain; nine of ten rats developed arthritis at day 16 after immunization and the day of onset ranged from day 10 to day 16 (Table 2). In conclusion, we demonstrated that DA/OlaHsd rats were significantly more sensitive to the induction of PIA, which is reflected in the enhanced incidence and severity of arthritis after a single “low-dose” injection of pristane.
Fig. 2.
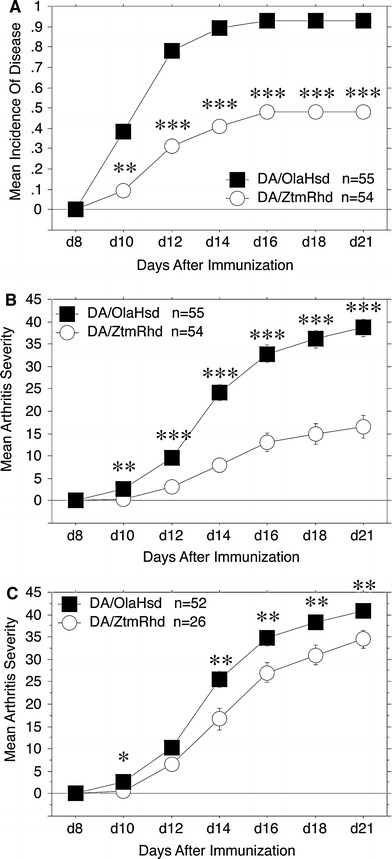
Pristane-induced arthritis in DA/OlaHsd and DA/ZtmRhd rats. (a) Arthritis incidence in DA/OlaHsd (n = 55) and DA/ZtmRhd (n = 54) rats after a 50-μl pristane injection. Data from two independent experiments were pooled and shown as mean of arthritis incidence at the indicated number of days after immunization. All significant p values between DA/OlaHsd and DA/ZtmRhd are indicated (* p < 0.05, ** p < 0.001, *** p < 0.0001). DA/ZtmRhd had a significantly reduced arthritis incidence compared with DA/OlaHsd. (b) Arthritis severity is shown as mean ± SEM of clinical arthritis score. All animals were included. DA/OlaHsd rats developed significantly more severe PIA than DA/ZtmRhd rats. (c) Mean arthritis severity score of only diseased animals (all rats with an arthritis score of 0 between day 8 and day 21 were excluded from the analysis). There was still a significant difference in arthritis severity between DA/OlaHsd (n = 52) and DA/ZtmRhd (n = 26) even after the exclusion of healthy rats
DA/OlaHsd had an earlier onset and increased severity of collagen II-induced arthritis
As a second model to study the difference between the two DA substrains, we chose CIA. We immunized rats with 75 μg lathyritic collagen type II emulsified in IFA. Two independent experiments with similar results (Table 2) were performed and the data were pooled. In contrast to PIA, the arthritis incidence at the end of the experiment (day 28) was not significantly different between the two DA substrains. Thirty-eight of 39 (97%) DA/OlaHsd rats developed CIA and 95% (37/39) of DA/ZtmRhd rats developed it. However, a significantly earlier onset was observed in DA/OlaHsd rats (p < 0.0001), leading to a significantly higher incidence in DA/OlaHsd rats at day 14 to day 17 (Fig. 3a, c). In addition, the DA/ZtmRhd rats showed a reduced arthritis severity with a p < 0.0001 from day 14 to day 24 (Fig. 3b). The levels of anti-CII IgG isotypes and total anti-CII-IgG antibodies in serum at day 24 were examined. Anti-CII-IgG2a, anti-CII-IgG2b, and anti-CII-IgG were found to be significantly reduced in DA/ZtmRhd rats (Fig. 3d). Anti-CII-IgG1 and anti-CII-IgG2c were not significantly different (data not shown).
Fig. 3.
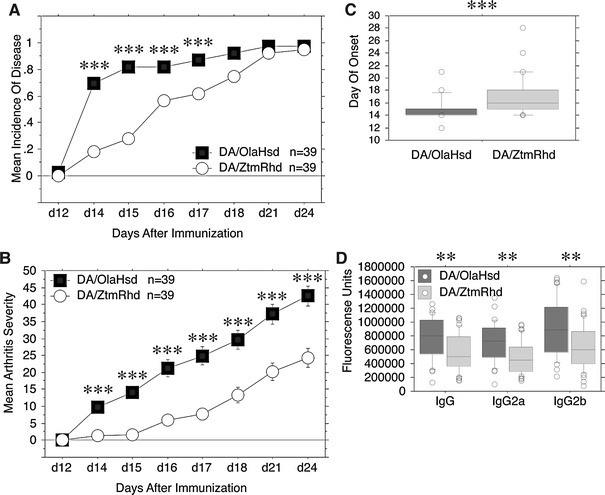
Collagen II-induced arthritis in DA/OlaHsd and DA/ZtmRhd rats. (a) Mean of disease incidence in DA/OlaHsd (n = 39) and DA/ZtmRhd (n = 39) rats after injection of 75 μg collagen type II. Data from two independent experiments were pooled and shown as mean of arthritis incidence. All significant p values between DA/OlaHsd and DA/ZtmRhd are indicated (* p < 0.05, ** p < 0.001, *** p < 0.0001). (b) Arthritis severity is shown as mean ± SEM of clinical arthritis score. All animals were included. DA/OlaHsd rats developed significantly more severe CIA than DA/ZtmRhd rats. (c) Day of onset. The days where the first macroscopic signs of arthritis were observed are shown as boxplots. Each box represents the second and third quartiles (from the 25th to 75th percentiles) of all values. Bold lines inside the boxes highlight the median value (50th percentile). Lines outside the boxes represent the 10th and 90th percentiles. Outlines (°) are indicated. DA/OlaHsd (n = 38) had a significantly earlier onset of arthritis than DA/ZtmRhd (n = 37). (d) Anti-CII antibodies in DA substrains after CIA are shown as relative fluorescence units measured in Eu3+LISA. Data are shown as boxplots. DA/OlaHsd rats had significantly higher levels of total anti-CII-IgG, anti-CII-IgG2a, and anti-CII-IgG2b
Having observed such a dramatic difference between DA/OlaHsd and DA/ZtmRhd rats in the development of CIA and PIA, we wanted to investigate whether this difference was due to the contaminating fragment on RNO3, which covers parts of the Cia11 QTL. We generated a partly congenic rat line by backcrossing DA/ZtmRhd rats to DA/OlaHsd rats for at least three generations to introduce the DA/ZtmRhd fragment on RNO3 into the DA/OlaHsd background. Despite the genetic and phenotypic differences that we observed between the two DA substrains, both strains displayed a high susceptibility to collagen-induced arthritis, which suggested that only a small number of arthritis QTLs differed in these substrains. Thus, we estimated that three backcross generations, a considerably high number of tested animals, and the strict use of littermate controls will be sufficient to investigate the effect of a single locus like the contaminating fragment on RNO3. No difference in arthritis and antibody response between DA.DA/ZtmRhd-Cia11 congenic rats and DA/OlaHsd littermate controls was observed in PIA or CIA (Fig. 4).
Fig. 4.
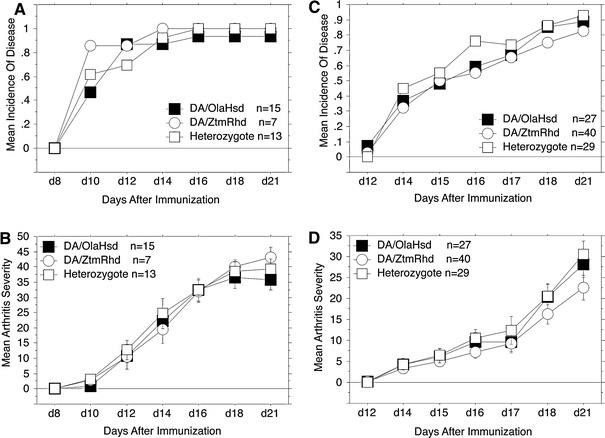
Arthritis development in DA.DA/ZtmRhd-Cia11 congenic rats. (a) Disease incidence after PIA in DA.DA/ZtmRhd-Cia11 homozygous rats (DA/Ztm, n = 7), DA.DA/ZtmRhd-Cia11 heterozygous rats (Heterozygous, n = 13), and DA littermate controls (DA/OlaHsd, n = 15). Data from two independent experiments were pooled and shown as mean of arthritis incidence. (b) Arthritis severity is shown as mean ± SEM of clinical arthritis score. No significant difference between the groups was observed. (c) Arthritis incidence after CIA in DA.DA/ZtmRhd-Cia11 homozygous rats (DA/Ztm, n = 40), DA.DA/ZtmRhd-Cia11 heterozygous rats (Heterozygous, n = 29), and DA littermate controls (DA/OlaHsd, n = 27). Data from four independent experiments were pooled and shown as mean of arthritis incidence. (d) Mean arthritis severity after CIA. There was no significant difference between the groups
Discussion
Several reports of genetically contaminated rat strains and observed phenotypic differences between rats of different colonies have been described earlier. It has been shown that BUF rats from different commercial vendors have polymorphic rat CD45 molecules (Jones et al. 1994) and that Dahl/Rapp rats are polymorphic at the Nos2 gene (Hojna et al. 2005). Furthermore, a polymorphism in the Ncf1 gene in inbred LEW rats from different sources was reported and it was shown that the substrains react differently to the induction of PIA (Olofsson et al. 2004). All these examples clearly show that when it comes to studies in which genetic purity is crucial to the experimental design and interpretation, one should act with caution when comparing animal studies from different institutes.
In arthritis research one of the most commonly used rat strains is the DA rat. The DA rat has a leading role in the search for arthritis-susceptible genes. Worldwide four different institutes have reported linkage studies with collagen II-induced arthritis that were performed using the arthritis-susceptible DA strain in combination with various resistant rat strains such as ACI, BN, E3, and F344 (Dracheva et al. 1999; Griffiths et al. 2000; Gulko et al. 1998; Meng et al. 2004; Olofsson et al. 2003b; Remmers et al. 1996). These studies provide a unique tool for the study of arthritis-regulating loci and for in silico fine-mapping by comparing the QTLs in those linkage studies.
However, caution must be taken so that only different resistant strains are used and not different sources of the susceptible DA rat strain. Here we report the finding of genetic polymorphisms of DA substrains from different origins. The largest contaminating fragment on RNO3 stretches from 35.6 to 52.3 Mb, colocalizing with the previously reported QTL Cia11. Interestingly, the Cia11 QTL was found in two different studies. In the first study by Griffiths et al. (2000), a F2 hybrid of (DA × BN) was used, and it was found that the DA allele from DA/Bkl rats at Cia11 recessively promotes CIA severity. In a different study by Olofsson et al. (2003b), another substrain of DA (DA/ZtmRhd) was used for an F2 backcross with the E3 strain, and in this study the DA allele had a recessive-promoting effect on arthritis severity as well. The Cia11 QTL was found to have a recessive-promoting allele in both DA substrains, but because of their genetic differences on RNO3 between 35.6 and 52.3 Mb we can exclude this region for being responsible for the Cia11 effect. This is confirmed through the comparison of the DA.DA/ZtmRhd-Cia11 congenic rat, which is as susceptible to CIA and PIA as their littermates harboring the DA/OlaHsd allele.
Because we have only limited information on polymorphic markers on RNO3, the origin of the putative contamination remains unclear. SNP data revealed identical haplotypes between ACI and DA/OlaHsd and no match of the DA/HanKini haplotype to any of the included strains, suggesting the contamination originated from ACI. However, previously performed SSLP typing of the recombinant inbred strain DXEC (unpublished data) showed a number of alleles not matching either of the parental strains DA/Ztm and E3/Ztm. Some of these alleles were found to be identical in DA/OlaHsd rats. Additional SNP typing will be required to conclusively determine the origin of the contamination.
In conclusion, we found considerable genetic differences and also major differences in arthritis incidence, severity, and onset of PIA and CIA between the DA/OlaHsd and DA/ZtmRhd substrains. Thus, there must be at least one yet unidentified CIA and PIA locus between those substrains. In our whole-genome scan we found a second region with polymorphic markers colocalizing with a previously described arthritis-regulating region. The polymorphic SSLP markers D10Rat26 and D10Rat93 on RNO10 are considerably close to the confirmed Cia5 and Oia3 QTLs (Brenner et al. 2005; Holm et al. 2001) and to the Pia10 QTL (Olofsson et al. 2003a). Although our additional marker screen or the SNP data did not reveal a large contaminating fragment, there is still the possibility of a minor fragment that differs between the DA/OlaHsd and DA/ZtmRhd substrains, inducing the differences in arthritis severity in those rats. Another possibility is that there is a novel arthritis QTL in one of the other contaminated regions or that the difference in arthritis susceptibility is due to a spontaneous mutation in one of the substrains.
In summary, we demonstrated that DA substrains show distinct genetic differences and exhibited a significantly diverse susceptibility to experimentally induced arthritis. Thus, this study shows how important it is to consider genetic variability in rat inbred strains and that one cannot uncritically compare studies performed with animals of different origin.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Acknowledgments
We thank the technicians at Medical Inflammation Research (Lund, Sweden), Carlos Palestro and Isabelle Bohlin, for taking excellent care of the animals. We also thank Dr. Lina Olsson and Dr. Robert Bockermann for critically reading the manuscript and for valuable comments. This work was supported by grants from the Swedish Research Council, the Swedish Association against Rheumatism, and the Swedish Foundation for Strategic Research, and European Union Grants MUGEN (LSHG-CT-2005-005203) and EURATools (European Commission contract No. LSHG-CT-2005-019015).
References
- Alamanos Y, Drosos AA. Epidemiology of adult rheumatoid arthritis. Autoimmun Rev. 2005;4:130–136. doi: 10.1016/j.autrev.2004.09.002. [DOI] [PubMed] [Google Scholar]
- Brenner M, Meng HC, Yarlett NC, Joe B, Griffiths MM, et al. The non-MHC quantitative trait locus Cia5 contains three major arthritis genes that differentially regulate disease severity, pannus formation, and joint damage in collagen- and pristane-induced arthritis. J Immunol. 2005;174:7894–7903. doi: 10.4049/jimmunol.174.12.7894. [DOI] [PubMed] [Google Scholar]
- Dieude P, Cornelis F. Genetic basis of rheumatoid arthritis. Joint Bone Spine. 2005;72:520–526. doi: 10.1016/j.jbspin.2005.09.001. [DOI] [PubMed] [Google Scholar]
- Dracheva SV, Remmers EF, Gulko PS, Kawahito Y, Longman RE, et al. Identification of a new quantitative trait locus on chromosome 7 controlling disease severity of collagen-induced arthritis in rats. Immunogenetics. 1999;49:787–791. doi: 10.1007/s002510050552. [DOI] [PubMed] [Google Scholar]
- Griffiths MM, Wang J, Joe B, Dracheva S, Kawahito Y, et al. Identification of four new quantitative trait loci regulating arthritis severity and one new quantitative trait locus regulating autoantibody production in rats with collagen-induced arthritis. Arthritis Rheum. 2000;43:1278–1289. doi: 10.1002/1529-0131(200006)43:6<1278::AID-ANR10>3.0.CO;2-S. [DOI] [PubMed] [Google Scholar]
- Gulko PS, Kawahito Y, Remmers EF, Reese VR, Wang J, et al. Identification of a new non-major histocompatibility complex genetic locus on chromosome 2 that controls disease severity in collagen-induced arthritis in rats. Arthritis Rheum. 1998;41:2122–2131. doi: 10.1002/1529-0131(199812)41:12<2122::AID-ART7>3.0.CO;2-#. [DOI] [PubMed] [Google Scholar]
- Hojna S, Kadlecova M, Zicha J, Kunes J. Polymorphism in Nos2 gene is absent in Prague colony of Dahl/Rapp salt-sensitive and salt-resistant rats. Physiol Res. 2005;54:201–206. [PubMed] [Google Scholar]
- Holm BC, Xu HW, Jacobsson L, Larsson A, Luthman H, et al. Rats made congenic for Oia3 on chromosome 10 become susceptible to squalene-induced arthritis. Hum Mol Genet. 2001;10:565–572. doi: 10.1093/hmg/10.6.565. [DOI] [PubMed] [Google Scholar]
- Holmdahl R (1997) Genetic analysis of mouse models for rheumatoid arthritis. In: Textbook of human genome methods. CRC Press, Boca Raton, FL
- Holmdahl R. Dissection of the genetic complexity of arthritis using animal models. J Autoimmun. 2003;21:99–103. doi: 10.1016/S0896-8411(03)00096-9. [DOI] [PubMed] [Google Scholar]
- Holmdahl R, Lorentzen JC, Lu S, Olofsson P, Wester L, et al. Arthritis induced in rats with nonimmunogenic adjuvants as models for rheumatoid arthritis. Immunol Rev. 2001;184:184–202. doi: 10.1034/j.1600-065x.2001.1840117.x. [DOI] [PubMed] [Google Scholar]
- Joe B. Quest for arthritis-causative genetic factors in the rat. Physiol Genomics. 2006;27:1–11. doi: 10.1152/physiolgenomics.00034.2005. [DOI] [PubMed] [Google Scholar]
- Jones RE, Weinberg A, Bourdette D. Evidence for genetic contamination of inbred buffalo rats (RT-1b) obtained from a commercial vendor. J Neuroimmunol. 1994;52:215–218. doi: 10.1016/0165-5728(94)90115-5. [DOI] [PubMed] [Google Scholar]
- Lander ES, Schork NJ. Genetic dissection of complex traits. Science. 1994;265:2037–2048. doi: 10.1126/science.8091226. [DOI] [PubMed] [Google Scholar]
- Meng HC, Griffiths MM, Remmers EF, Kawahito Y, Li W, et al. Identification of two novel female-specific non-major histocompatibility complex loci regulating collagen-induced arthritis severity and chronicity, and evidence of epistasis. Arthritis Rheum. 2004;50:2695–2705. doi: 10.1002/art.20366. [DOI] [PubMed] [Google Scholar]
- Miller EJ, Rhodes RK. Preparation and characterization of the different types of collagen. Methods Enzymol 82 Pt. 1982;A:33–64. doi: 10.1016/0076-6879(82)82059-4. [DOI] [PubMed] [Google Scholar]
- Olofsson P, Holmberg J, Pettersson U, Holmdahl R. Identification and isolation of dominant susceptibility loci for pristane-induced arthritis. J Immunol. 2003;171:407–416. doi: 10.4049/jimmunol.171.1.407. [DOI] [PubMed] [Google Scholar]
- Olofsson P, Lu S, Holmberg J, Song T, Wernhoff P, et al. A comparative genetic analysis between collagen-induced arthritis and pristane-induced arthritis. Arthritis Rheum. 2003;48:2332–2342. doi: 10.1002/art.11100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Olofsson P, Johansson A, Wedekind D, Kloting I, Klinga-Levan K, et al. Inconsistent susceptibility to autoimmunity in inbred LEW rats is due to genetic crossbreeding involving segregation of the arthritis-regulating gene Ncf1. Genomics. 2004;83:765–771. doi: 10.1016/j.ygeno.2003.10.005. [DOI] [PubMed] [Google Scholar]
- Plenge RM, Seielstad M, Padyukov L, Lee AT, Remmers EF, et al. TRAF1–C5 as a risk locus for rheumatoid arthritis—a genomewide study. N Engl J Med. 2007;357:1199–1209. doi: 10.1056/NEJMoa073491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Remmers EF, Longman RE, Du Y, O’Hare A, Cannon GW, et al. A genome scan localizes five non-MHC loci controlling collagen-induced arthritis in rats. Nat Genet. 1996;14:82–85. doi: 10.1038/ng0996-82. [DOI] [PubMed] [Google Scholar]
- Saar K, Beck A, Bihoreau MT, Birney E, Brocklebank D, et al. SNP and haplotype mapping for genetic analysis in the rat. Nat Genet. 2008;40:560–566. doi: 10.1038/ng.124. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith BD, Martin GR, Miller EJ, Dorfman A, Swarm R. Nature of the collagen synthesized by a transplanted chondrosarcoma. Arch Biochem Biophys. 1975;166:181–186. doi: 10.1016/0003-9861(75)90378-1. [DOI] [PubMed] [Google Scholar]
- The Wellcome Trust Consortium Genome-wide association study of 14, 000 cases of seven common diseases and 3, 000 shared controls. Nature. 2007;447:661–678. doi: 10.1038/nature05911. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Worthington J. Investigating the genetic basis of susceptibility to rheumatoid arthritis. J Autoimmun. 2005;25(suppl):16–20. doi: 10.1016/j.jaut.2005.09.011. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


