Abstract
Arboviruses are often maintained in complex cycles involving vertebrates such as mammals or birds and blood-feeding mosquitoes. However, the role of wildlife hosts in their emergence or re-emergence in human populations has received little attention. The recent emergence of Zika virus in America, and previous occurrences of chikungunya and dengue, forces us to confront a potential new disease-emergence phenomenon. Using a spatial data mining framework to identify potential biotic interactions, based on the degree of co-occurrence between different species, we identified those mammal species with the highest potential for establishing mammal–vector interactions, considering as principal vector Aedes aegypti. Seven of the top ten identified mammal species with highest potential were bats, with two of them having previously been confirmed as positive hosts for dengue in Mexico. We hope that this will raise interest of Mexican public health authorities and academic institutions to assess the role of wild hosts in the maintenance and spread of arboviruses.
Keywords: Wildlife host, Dengue, Chikungunya, Zika virus, Arbovirus, Aedes aegypti
The current outbreak of Zika virus (ZIKV) is the most recent disease threatening human health worldwide (Lucey and Gostin 2016; WHO 2016). ZIKV is a mosquito-borne virus (arbovirus) belonging to the family Flaviviridae, which was first isolated from a rhesus monkey in the Zika forest of Uganda in 1952 (Dick et al. 1952). Until 2007, sporadic human disease cases had been reported from countries in Africa and Asia (Musso et al. 2014). However, last year the virus was detected in America (Zanluca et al. 2015), where it has been spreading explosively (ECDC 2015; Petersen et al. 2016). A great number of cases with neurological and autoimmune complications have been reported from 20 countries in the Americas, mainly in South America (Ventura et al. 2016; WHO 2016), though a substantial number of human cases have recently been identified in North America (Chen and Hamer 2016; McCarthy 2016).
Arboviruses are often maintained in complex cycles involving vertebrates and blood-feeding vectors (Blum et al. 2016) and are transmitted by the bite of infected female mosquitoes (Lefèvre et al. 2009). Consequently, humans could be at high risk of infections from pathogens circulating within the blood system of other wildlife vertebrate species (Kaddumukasa et al. 2015). Due to their current relevance to public health, the identification of mosquitoes’ wildlife blood-meal sources and their relative importance is important in order to determine the linkages between potential hosts and vectors.
The arboviruses ZIKV, dengue (DENV) and chikungunya (CHIKV) have been isolated from several Aedes mosquito species (Klimpel and Mehlhorn 2014), most notably Aedes aegypti and Aedes albopictus, which are widespread worldwide (Grard et al. 2014; Kraemer et al. 2015). Although other mosquito species have been identified as having links with these viruses, A. aegypti has generally been considered to be the most relevant in Mexico. However, the role of potential wildlife hosts in maintenance of these arboviruses has received relatively little attention, even though there are results that indicate that they are not necessarily restricted to epizootic cycles in the New World. For instance, although DENV is widely believed to be absent from New World wildlife, all four serotypes of DENV have been found in a wide variety of South American mammals in French Guiana (de Thoisy et al. 2009), including bats, rodents and marsupials. Notably, identified hosts were encountered in both DENV endemic areas as well as areas where the disease was nearly absent. Genetic evidence was provided that was consistent with the hypotheses that (1) there exist spillover infections from humans to mammals and (2) there exists an enzootic cycle. In Mexico, six bat species have been identified as positives for DENV (Artibeus jamaicensis, Artibeus lituratus, Carollia brevicauda, Myotis nigricans, Pteronotus parnellii and Natalus stramineus) (Aguilar-Setién et al. 2008; Machain-Williams et al. 2013; Sotomayor-Bonilla et al. 2014).
In contrast, there are no identified hosts of CHIKV or ZIKV in the New World, although both have multiple mammal hosts in Africa and Asia. In particular, for bats, CHIKV has been isolated in three Chinese bats (Rousettus aegyptiacus, Hipposideros caffer, Chaerephon pumilus) and one from Senegal (Scotophilus sp.). For ZIKV, no natural infections of bats have been documented; however, the bat Myotis lucifugus was infected successfully in laboratory conditions (Reagan et al. 1955).
Although the question remains as to whether wildlife and/or domestic hosts can maintain DENV, CHIKV and ZIKV in enzootic cycles and, therefore, play a role in its re-emergence in human populations, or that infections in human populations can spillover into wildlife hosts, these preliminary results highlight the need to conduct studies focused on identifying and understanding the role of wildlife species in the spread and maintenance of these arboviruses. Thus, we urgently need research on these viruses and the ecologic, entomologic and host determinants of viral maintenance and emergence (Blum et al. 2016). A further complication comes from the complex evolution of arboviruses (Weaver and Barrett 2004; Turner et al. 2010) which, as RNA viruses, are highly mutable, thus allowing for potentially rapid adaptive changes to new hosts and changes in host range.
Additionally, the steady increase in contact between human and wildlife by the invasion and destruction of natural forest, as well as climate and environmental changes, is allowing mosquitoes to be exposed to potentially new wildlife species hosts and pathogens, thus being able to potentially create new disease patterns (Kaddumukasa et al. 2015). Therefore, a major short-term goal should be to conduct specific, directed studies of potential wildlife hosts involved in arbovirus maintenance. Although host selection by mosquitoes depends on a great number of factors, a first step should be to determine which wildlife species have a high risk of being in contact with the disease vectors in order to optimize resource allocation for these studies.
The increased availability of spatial data and recent methodological developments in species distribution modelling (Stephens et al. 2009; González-Salazar et al. 2013) allow us to implement spatial analysis that can be used to build predictive models for the presence of emerging diseases. In particular, they allow for the direct incorporation of biotic factors, which are not explicitly accounted for in most niche modelling studies, including inter-specific interactions, as well as abiotic factors. Biotic factors, such as host range, are obviously of crucial importance in the dynamics of zoonoses. The method uses point collection data for potential/known vectors and potential/known hosts and constructs a diagnostic, epsilon (Stephens et al. 2009), that measures the statistical significance of the degree of co-occurrence of a pair of species (e.g. a potential vector and a potential host) relative to the null hypothesis that they are uncorrelated. In the present case, the mammal data set used contains 37,297 unique point collections from geo-referenced localities for 427 terrestrial mammals occurring in Mexico (GBIF; www.gbif.org, and CONABIO; www.conabio.gob.mx). For A. aegypti, there were 302 collection points taken from the Sistema Nacional de Información sobre la Biodiversidad of the Comisión Nacional para Uso y Conservación de la Biodiversidad (www.conabio.gob.mx). Co-occurrences were defined on a uniform grid covering Mexico of 3337 rectangular cells of size 25 km × 25 km. With these data, a predictive model for the potential distribution of A. aegypti in Mexico was created based only on biotic factors. This approach gives us two principal results: (1) a ranked list of mammals that can potentially be blood-meal sources for the mosquito and therefore potential hosts and (2) a map of potential presence of A. aegypti linked to the presence of potential mammal hosts (Stephens et al. 2009).
Of 427 potential vector–host pair interactions, we show in Table 1 the top 25% (108) of most statistically significant positive associations based on our co-occurrence index, epsilon. This ranked mammal list is a first-pass predictive model for the most important potential blood-meal sources for A. aegypti; consequently, they are, at this level of description, the main candidates to be positive for DENV, ZIKV or CHIKV. Of course, such a statistical association does not necessarily prove that there is a direct “causal” interaction between these taxa. Neither does it allow for a more detailed description of the potential host role, i.e., as a competent host, dead-end host, alternative host. However, previous studies using and validating this method led to the prediction and subsequent discovery of 22 previously unknown mammals hosts for Leishmaniasis (Stephens et al. 2016). Additionally, evaluating our mammal list against the known results for DENV in Mexico, five of the six bat species positive to DENV (Aguilar-Setién et al. 2008) are in the top 50 highest ranked species (P < 10−4). It is notable that 7 of the top 10 species are bats, with two being previously identified as positive for the presence of dengue, and, interestingly, four of them (Glossophaga soricina, A. jamaicensis, A. lituratus and Sturnira lilium) are identified hosts of Leishmania (L.) mexicana and Trypanosoma cruzi (Villegas-García and Santillán-Alarcón 2001; Berzunza-Cruz et al. 2015; López-Cancino et al. 2015). Within the highest ranked species are all major groups of mammals, with many of them being synanthropic species (Table 1) (Estrada et al. 1994; Medellín et al. 2000; Ruiz-Piña and Cruz-Reyes 2002; Martínez-Hernández et al. 2014; Hennessy et al. 2015; López-Cancino et al. 2015; Cruz-Salazar et al. 2016). Additionally, the genus Artibeus, Sturnira and Carollia have all been identified as hosts for other pathogens in fragmented landscapes, including coronavirus, pegivirus, hepacivirus, rabies and leptospira (Anthony et al. 2013; Quan et al. 2013; Chávez et al. 2015).
Table 1.
Rank list of Potentials Mammal Blood Sources to Aedes aegypti in Mexico.
| Rank | Mammal | Epsilon | Rank | Mammal | Epsilon |
|---|---|---|---|---|---|
| 1 | Glossophaga soricina + | 12.78 | 55 | Caluromys derbianus | 5.78 |
| 2 | Molossus rufus | 11.99 | 56 | Molossus molossus | 5.76 |
| 3 | Artibeus jamaicensis* ,+ | 11.68 | 57 | Oryzomys rostratus | 5.76 |
| 4 | Liomys pictus | 11.06 | 58 | Osgoodomys banderanus | 5.76 |
| 5 | Oryzomys couesi | 11.04 | 59 | Myotis carteri | 5.66 |
| 6 | Carollia subrufa + | 10.49 | 60 | Micronycteris microtis | 5.52 |
| 7 | Sturnira lilium + | 10.28 | 61 | Sylvilagus brasiliensis | 5.47 |
| 8 | Artibeus lituratus* ,+ | 9.91 | 62 | Sylvilagus floridanus | 5.37 |
| 9 | Choeroniscus godmani | 9.42 | 63 | Spermophilus annulatus | 5.36 |
| 10 | Liomys salvini | 9.33 | 64 | Peromyscus leucopus | 5.3 |
| 11 | Oligoryzomys fulvescens | 9.15 | 65 | Conepatus leuconotus | 5.3 |
| 12 | Dermanura phaeotis + | 9.12 | 66 | Chaetodipus pernix | 5.27 |
| 13 | Rhogeessa tumida | 9.06 | 67 | Sciurus yucatanensis | 5.23 |
| 14 | Pteronotus personatus | 9.05 | 68 | Sigmodon mascotensis | 5.13 |
| 15 | Baiomys musculus | 8.97 | 69 | Eira barbara | 5.12 |
| 16 | Glossophaga commissarisi + | 8.8 | 70 | Ateles geoffroyi | 5.11 |
| 17 | Didelphis virginiana + | 8.58 | 71 | Neotoma phenax | 5.07 |
| 18 | Pteronotus parnellii* | 8.58 | 72 | Noctilio leporinus | 5.06 |
| 19 | Orthogeomys hispidus | 8.53 | 73 | Reithrodontomys fulvescens | 4.95 |
| 20 | Sciurus aureogaster + | 8.52 | 74 | Megasorex gigas | 4.92 |
| 21 | Molossus sinaloae | 8.51 | 75 | Heteromys gaumeri + | 4.9 |
| 22 | Desmodus rotundus + | 8.23 | 76 | Eumops bonariensis | 4.85 |
| 23 | Saccopteryx bilineata | 8.22 | 77 | Thyroptera tricolor | 4.85 |
| 24 | Lasiurus intermedius | 8.15 | 78 | Baiomys taylori | 4.84 |
| 25 | Phyllostomus discolour | 8.12 | 79 | Orthogeomys grandis | 4.79 |
| 26 | Philander opossum + | 8.1 | 80 | Nyctinomops laticaudatus | 4.76 |
| 27 | Peromyscus gymnotis | 7.9 | 81 | Peromyscus mexicanus + | 4.69 |
| 28 | Balantiopteryx plicata | 7.81 | 82 | Glossophaga leachii | 4.65 |
| 29 | Eptesicus furinalis | 7.69 | 83 | Cratogeomys gymnurus | 4.56 |
| 30 | Pteronotus davyi | 7.55 | 84 | Cuniculus paca | 4.55 |
| 31 | Dermanura tolteca | 7.48 | 85 | Xenomys nelsoni | 4.54 |
| 32 | Sciurus variegatoides | 7.48 | 86 | Ototylomys phyllotis | 4.52 |
| 33 | Mormoops megalophylla | 7.45 | 87 | Sciurus alleni | 4.43 |
| 34 | Oryzomys melanotis | 7.42 | 88 | Chiroderma villosum | 4.41 |
| 35 | Artibeus intermedius + | 7.4 | 89 | Spermophilus adocetus | 4.32 |
| 36 | Chaetodipus artus | 7.2 | 90 | Cryptotis mayensis | 4.32 |
| 37 | Nasua narica + | 7.18 | 91 | Cryptotis obscura | 4.32 |
| 38 | Dasypus novemcinctus | 7.11 | 92 | Geomys tropicalis | 4.32 |
| 39 | Sigmodon hispidus + | 7.02 | 93 | Molossus nigricans | 4.32 |
| 40 | Uroderma bilobatum | 6.82 | 94 | Leptonycteris curasoae | 6.75 |
| 41 | Carollia perspicillata + | 6.71 | 95 | Heteromys desmarestianus | 4.23 |
| 42 | Centurio senex | 6.61 | 96 | Leptonycteris nivalis | 4.19 |
| 43 | Sciurus colliaei | 6.59 | 97 | Musonycteris harrisoni | 4.17 |
| 44 | Lontra longicaudis | 6.49 | 98 | Lasiurus blossevil | 4.14 |
| 45 | Didelphis marsupialis + | 6.49 | 99 | Lasiurus ega | 4.10 |
| 46 | Cratogeomys bulleri | 6.35 | 100 | Cratogeomys fumosus | 4.08 |
| 47 | Carollia sowelli* ,+ | 6.27 | 101 | Tapirus bairdii | 4.08 |
| 48 | Myotis elegans | 6.12 | 102 | Vampyrum spectrum | 4.08 |
| 49 | Myotis nigricans* | 6.06 | 103 | Conepatus semistriatus | 4.05 |
| 50 | Sigmodon arizonae | 6.00 | 104 | Marmosa mexicana | 4.05 |
| 51 | Rhynchonycteris naso | 5.95 | 105 | Rhogeessa parvula | 4.05 |
| 52 | Tlacuatzin canescens | 5.87 | 106 | Mustela frenata | 4.01 |
| 53 | Leopardus pardalis | 5.84 | 107 | Procyon lotor + | 4.01 |
| 54 | Pteronotus rubiginosus | 4.32 | 108 | Peromyscus perfulvus | 3.99 |
* Mammal species confirmed positives to dengue virus.
+ Synanthropic mammal species.
To identify those Mexican states with a high probability of significant mosquito–mammal interaction, we modelled the potential distribution of A. aegypti based on mammal point collection data using a score function (see Stephens et al. 2009) (Fig. 1a). This map shows areas where vector and potential mammal hosts are most likely to both be present and, therefore, potentially interact. As stated, an important potential use of this information is to target further investigation by concentrating field studies and/or surveillance in those areas. Note that, our list is not restricted to bat species, including several other groups of mammals, for instance rodents, marsupials, carnivores, etc., indicating that field studies should have the capacity to collect a wide, representative set of potential mammal hosts.
Figure 1.
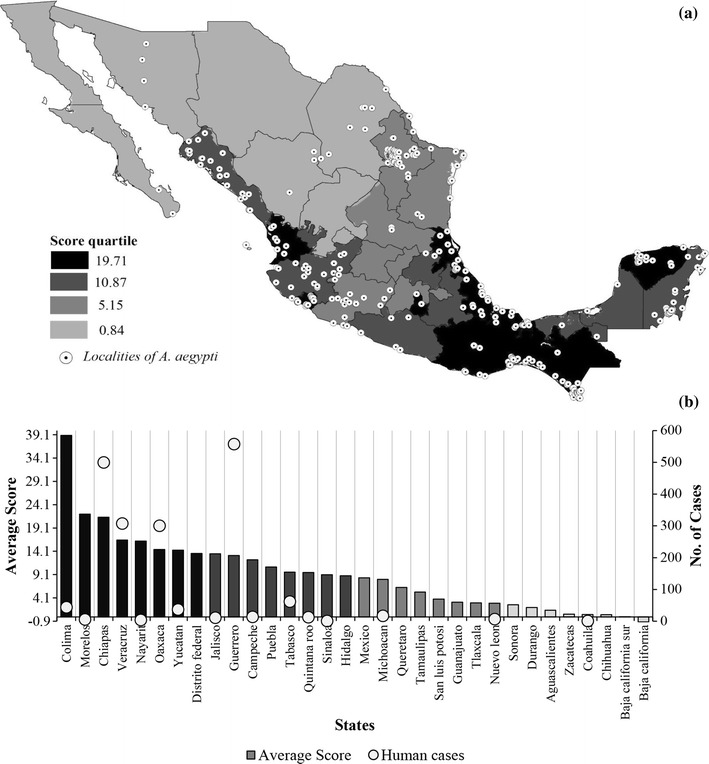
a Potential distribution of Aedes aegypti based on mammal ranges by States in Mexico. b Average score (i.e. probability of A. aegypti presence) by these states and number of human cases confirmed of Zika virus at present.
To test model accuracy as a function of score, we grouped Mexican states by score quartiles. The 4th quartile corresponds to the 25% of states with the highest score values, the 3rd quartile to the next 25% of states with highest score values, etc. This allows us to establish predictability profiles across the different score quartiles for our biotic model. We randomly selected 30% of mosquito records (92 points) to calculate for each score quartile the percentage of associated A. aegypti records. Forty percentages of points were observed in the top quartile, and this percentage significantly decreased towards the lower quartiles (X 2 = 38.64, P < 0.00001). Taking as a null hypothesis that A. aegypti are distributed randomly in Mexico, the outcomes showed that biotic model predicted accurately the most important potential areas for A. aegypti presence.
Considering the correlation between current human cases of ZIKV in Mexico with the predicted distribution by state of A. aegypti using our biotic model we see that the vast majority of autochthonous cases have occurred in states with high values of scores (i.e. high probability to find significant mosquito–mammal interactions). For instance, Chiapas and Guerrero reported 500 and 558 cases, respectively, corresponding to 56% of the 1884 cases confirmed at present (http://www.epidemiologia.salud.gob.mx/). We believe there is a significant risk that synanthropic mammals, such as bats, rodents or marsupials, could be infected thus favouring virus maintenance. We have restricted our analysis, conservatively, to A. aegypti, as vector as it has been identified positively for all three viruses—DENV, CHIKV and ZIKV. However, our methodology can be applied to consider the relation between potential mammal hosts and any other potential vector or combination thereof.
In distinction to important zoonosis like Leishmaniasis and Chagas, for which the role of wild hosts is better understood (De Almeida Curi et al. 2006; Herrera 2010), for DENV, CHIKV and ZIKV and other arbovirus, our understanding of the role of non-human hosts, either as a spillover from an epizootic cycle or via the potential existence of enzootic cycles, is very poor. However, given the potentially important epidemiological consequences of either of these scenarios, it is important to explore these possibilities. Our results are a first step in identifying ecological characteristics of those regions with the highest risk of a potentially significant role for non-human hosts. We hope that this will raise the interest of Mexican public health authorities and academic institutions in considering the risk arising from potential non-human hosts of ZIKV. Obviously our methodology is also applicable to prediction of potential non-human hosts for ZIKV in other countries, the only requirement being the relevant collection data analogous to that used in the present study. Finally, we emphasise again that the scope of the model of the present paper is to serve as a focus for future studies and show that potentially useful information can be gleaned from the method, which, at this level, is not capable of predicting detailed elements such as potential host competency.
Acknowledgements
Mosquito data were kindly provided by the National Commission for the Knowledge and Use of Biodiversity (Comision Nacional para el Conocimiento y Uso de la Biodiversidad, CONABIO) of Mexico. We deeply thank two anonymous referees for their significant comments and suggestions to improve this manuscript. We are grateful for financial support from PAPIIT-UNAM grant number IG200217. CGS is grateful for financial support from the Centro de Ciencias de la Complejidad and the Laboratorio Nacional de Ciencias de la Complejidad.
References
- Aguilar-Setién A, Romero-Almaraz ML, Sánchez-Hernández C, Figueroa R, Juárez-Palma LP, García-Flores MM, Vázquez-Salinas C, Salas-Rojas M, Hidalgo-Martínez AC, Pierlé SA, García-Estrada C, Ramos C. Dengue virus in Mexican bats. Epidemiology and Infection. 2008;136:1678–1683. doi: 10.1017/S0950268808000460. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anthony SJ, Ojeda-Flores R, Rico-Chávez O, Navarrete-Macias I, Zambrana-Torrelio CM, Rostal MK, Epstein JH, Tipps T, Liang E, Sanchez-Leon M, Sotomayor-Bonilla J, Aguirre AA, Ávila-Flores RA, Medellín RA, Goldstein T, Suzán G, Daszak P, Lipkin WI. Coronaviruses in bats from Mexico. Journal of General Virology. 2013;94:1028–1038. doi: 10.1099/vir.0.049759-0. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berzunza-Cruz M, Rodríguez-Moreno Á, Gutiérrez-Granados G, González-Salazar C, Stephens CR, Hidalgo-Mihart M, Marina CF, Rebollar-Téllez EA, Bailón-Martínez D, Balcells CD, Ibarra-Cerdeña CN, Sánchez-Cordero V, Becker I. Leishmania (L.) mexicana infected bats in Mexico: novel potential reservoirs. PLOS Neglected Tropical Diseases. 2015;9:1–15. doi: 10.1371/journal.pntd.0003438. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blum JD, Conway PH, Sharfstein JM (2016) Zika virus in the Americas—yet another arbovirus threat. New England Journal of Medicine 493–494. doi:10.1056/NEJMp1002530. [DOI] [PubMed]
- Chávez OR, Flores RO, Bonilla JS, Zambrana-Torrelio C, Rubio EL, Aguirre AA, Suzán G. Viral diversity of bat communities in human-dominated landscapes in Mexico. Veterinaria México OA. 2015;2:1–22. [Google Scholar]
- Chen LH, Hamer DH. Zika virus: rapid spread in the Western hemisphere. Annals of Internal Medicine. 2016 doi: 10.7326/M16-0150. [DOI] [PubMed] [Google Scholar]
- Cruz-Salazar B, Ruiz-Montoya L, Vázquez-Domínguez E, Navarrete-Gutiérrez D, Espinoza-Medinilla EE, Vázquez L-B. Genetic diversity of Didelphis virginiana related to different levels of disturbance in the Highlands and the Central Depression regions of Chiapas, Mexico. Journal of Tropical Ecology. 2016;32:146–157. doi: 10.1017/S0266467416000080. [DOI] [Google Scholar]
- De Almeida Curi NH, Miranda I, Talamoni SA. Serologic evidence of Leishmania infection in free-ranging wild and domestic canids around a Brazilian National Park. Memorias do Instituto Oswaldo Cruz. 2006;101:99–101. doi: 10.1590/S0074-02762006000100019. [DOI] [PubMed] [Google Scholar]
- de Thoisy B, Lacoste V, Germain A, Muñoz-Jordán J, Colón C, Mauffrey J-F, Delaval M, Catzeflis F, Kazanji M, Matheus S, Dussart P, Morvan J, Setién AA, Deparis X, Lavergne A. Dengue infection in neotropical forest mammals. Vector-Borne and Zoonotic Diseases. 2009;9:157–170. doi: 10.1089/vbz.2007.0280. [DOI] [PubMed] [Google Scholar]
- Dick GWA, Kitchen SF, Haddow AJ. Zika virus (I). Isolations and serological specificity. Transactions of the Royal Society of Tropical Medicine and Hygiene. 1952;46:509–520. doi: 10.1016/0035-9203(52)90042-4. [DOI] [PubMed] [Google Scholar]
- European Centre for Disease Prevention and Control (ECDC) (2015) Rapid risk assessment: Zika virus epidemic in the Americas: potential association with microcephaly and Guillain-Barré syndrome. Available from: http://ecdc.europa.eu/en/publications/Publications.
- Estrada A, Coates-Estrada R, Meritt D. Non flying mammals and landscape changes in the tropical rain forest region of Los Tuxtlas, Mexico. Ecography (Cop) 1994;17:229–241. doi: 10.1111/j.1600-0587.1994.tb00098.x. [DOI] [Google Scholar]
- González-Salazar C, Stephens CR, Marquet PA. Comparing the relative contributions of biotic and abiotic factors as mediators of species’ distributions. Ecological Modelling. 2013;248:57–70. doi: 10.1016/j.ecolmodel.2012.10.007. [DOI] [Google Scholar]
- Grard G, Caron M, Mombo IM, Nkoghe D, Mboui Ondo S, Jiolle D, Fontenille D, Paupy C, Leroy EM. Zika virus in Gabon (Central Africa)-2007: a new threat from Aedes albopictus? PLOS Neglected Tropical Diseases. 2014;8:1–6. doi: 10.1371/journal.pntd.0002681. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hennessy C, Tsai CC, Beasley JC, Beatty WS, Zollner PA, Rhodes OE. Elucidation of population connectivity in synanthropic mesopredators: using genes to define relevant spatial scales for management of raccoons and virginia opossums. Journal of Wildlife Management. 2015;79:112–121. doi: 10.1002/jwmg.812. [DOI] [Google Scholar]
- Herrera L. Una revisión sobre reservorios de Trypanosoma (Schizotrypanum) cruzi (Chagas, 1909), agente etiológico de la Enfermedad de Chagas. Bol Malariol y Salud Ambient. 2010;50:3–15. [Google Scholar]
- Kaddumukasa MA, Kayondo JK, Masiga D, Akol AM, Lutwama JJ, Masembe C. High proportion of mosquito vectors in Zika forest, Uganda, feeding on humans has implications for the spread of new arbovirus pathogens. African Journal of Biotechnology. 2015;14:1418–1426. doi: 10.5897/AJB2015.14474. [DOI] [Google Scholar]
- Klimpel S, Mehlhorn H. Bats (Chiroptera) as Vectors of Diseases and Parasites. Parasitology Research Monographs. Heidelberg: Springer; 2014. [Google Scholar]
- Kraemer MUG, Sinka ME, Duda KA, Mylne AQN, Shearer FM, Barker CM, Moore CG, Carvalho RG, Coelho GE, Van Bortel W, Hendrickx G, Schaffner F, Elyazar IR, Teng HJ, Brady OJ, Messina JP, Pigott DM, Scott TW, Smith DL, William Wint GR, Golding N, Hay SI. The global distribution of the arbovirus vectors Aedes aegypti and Ae. albopictus. Elife. 2015;4:1–18. doi: 10.7554/eLife.08347. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lefèvre T, Gouagna LC, Dabiré KR, Elguero E, Fontenille D, Renaud F, Costantini C, Thomas F. Beyond nature and nurture: phenotypic plasticity in blood-feeding behavior of Anopheles gambiae s.s. when humans are not readily accessible. American Journal of Tropical Medicine and Hygeine. 2009;81:1023–1029. doi: 10.4269/ajtmh.2009.09-0124. [DOI] [PubMed] [Google Scholar]
- López-Cancino SA, Tun-Ku E, De la Cruz-Felix HK, Ibarra-Cerdena CN, Izeta-Alberdi A, Pech-May A, Mazariegos-Hidalgo CJ, Valdez-Tah A, Ramsey JM. Landscape ecology of Trypanosoma cruzi in the southern Yucatan Peninsula. Acta Tropica. 2015;151:58–72. doi: 10.1016/j.actatropica.2015.07.021. [DOI] [PubMed] [Google Scholar]
- Lucey DR, Gostin LO. The emerging Zika pandemic: enhancing preparedness. Journal of the American Medical Association. 2016 doi: 10.1001/jama.2016.0904. [DOI] [PubMed] [Google Scholar]
- Machain-Williams C, López-Uribe M, Talavera-Aguilar L, Vera-Escalante L, Puerto-Manzano F, Ulloa A, Farfán-ale JA, Garcia-Rejon J, Blitvich BJ, Alba M. Serologic evidence of flavivirus infection in bats in the Yucatan Peninsula of Mexico. Wildlife Disease Association. 2013;49:1–8. doi: 10.7589/2011-10-287. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martínez-Hernández F, Rendon-Franco E, Gama-Campillo LM, Villanueva-García C, Romero-Valdovinos M, Maravilla P, Alejandre-Aguilar R, Rivas N, Córdoba-Aguilar A, Muñoz-García CI, Villalobos G. Follow up of natural infection with Trypanosoma cruzi in two mammals species, Nasua narica and Procyon lotor (Carnivora: Procyonidae): evidence of infection control? Parasites and Vectors. 2014;7:405. doi: 10.1186/1756-3305-7-405. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCarthy M. Zika virus was transmitted by sexual contact in Texas, health officials report. BMJ. 2016;352:i720. doi: 10.1136/bmj.i720. [DOI] [PubMed] [Google Scholar]
- Medellín RA, Equihua M, Amin MA. Bat diversity and abundance as indicators of disturbance in neotropical rainforest. Conservation Biology. 2000;14:1666–1675. doi: 10.1046/j.1523-1739.2000.99068.x. [DOI] [PubMed] [Google Scholar]
- Musso D, Nilles EJ, Cao-Lormeau VM. Rapid spread of emerging Zika virus in the Pacific area. Clinical Microbiology and Infection. 2014;20:O595–O596. doi: 10.1111/1469-0691.12707. [DOI] [PubMed] [Google Scholar]
- Petersen E, Wilson ME, Touch S, McCloskey B, Mwaba P, Bates M, Dar O, Mattes F, Kidd M, Ippolito G, Azhar EI, Zumla A (2016) Unexpected and rapid spread of Zika virus in the Americas—implications for public health preparedness for mass gatherings at the 2016 Brazil Olympic Games. International Journal of Infectious Diseases 1–5. doi:10.1016/j.ijid.2016.02.001. [DOI] [PubMed]
- Quan PL, Firth C, Conte JM, Williams SH, Zambrana-Torrelio CM, Anthony SJ, Ellison JA, Gilbert AT, Kuzmin IV, Niezgoda M, Osinubi MO, Recuenco S, Markotter W, Breiman RF, Kalemba L, Malekani J, Lindblade KA, Rostal MK, Ojeda-Flores R, Suzán G, Davis LB, Blau DM, Ogunkoya AB, Alvarez Castillo DA, Moran D, Ngam S, Akaibe D, Agwanda B, Briese T, Epstein JH, Daszak P, Rupprecht CE, Holmes EC, Lipkin WI. Bats are a major natural reservoir for hepaciviruses and pegiviruses. Proceedings of the National Academy of Sciences. 2013;110:8194–8199. doi: 10.1073/pnas.1303037110. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reagan RL, Rumbaugh H, Nelson H, Brueckner AL. Effect of Zika virus and Bwamba virus in the cave bat (Myotis lucifugus) Transactions of the American Microscopical Society. 1955;74:77–79. doi: 10.2307/3223847. [DOI] [Google Scholar]
- Ruiz-Piña HA, Cruz-Reyes A. The opossum Didelphis virginiana as a synanthropic reservoir of Trypanosoma cruzi in Dzidzilché, Yucatán, México. Memorias do Instituto Oswaldo Cruz. 2002;97:613–620. doi: 10.1590/S0074-02762002000500003. [DOI] [PubMed] [Google Scholar]
- Sotomayor-Bonilla J, Chaves A, Rico-Chávez O, Rostal MK, Ojeda-Flores R, Salas-Rojas M, Aguilar-Setien A, Ibáñez-Bernal S, Barbachano-Guerrero A, Gutiérrez-Espeleta G, Aguilar-Faisal JL, Aguirre AA, Daszak P, Suzán G. Short report: dengue virus in bats from southeastern Mexico. American Journal of Tropical Medicine and Hygeine. 2014;91:129–131. doi: 10.4269/ajtmh.13-0524. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stephens CR, González-Salazar C, Sánchez-Cordero V, Becker I, Rebollar-Tellez E, Rodríguez-Moreno A, Berzunza-Cruz M, Domingo Balcells C, Gutiérrez-Granados G, Hidalgo-Mihart M, Ibarra-Cerdeña CN, Ibarra López P, Iñiguez Dávalos LI, Ramírez Martínez MM. Can you judge a disease host by the company it keeps? Predicting disease hosts and their relative importance: a case study for Leishmaniasis. PLOS Neglected Tropical Diseases. 2016;10(10):e0005004. doi: 10.1371/journal.pntd.0005004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Stephens CR, Heau JG, González C, Ibarra-Cerdeña CN, Sánchez-Cordero V, González-Salazar C. Using biotic interaction networks for prediction in biodiversity and emerging diseases. PLoS ONE. 2009;4:e5725. doi: 10.1371/journal.pone.0005725. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turner PE, Morales NM, Alto BW, Remold SK. Role of evolved host breadth in the initial emergence of an RNA virus. Evolution (N Y) 2010;64:3273–3286. doi: 10.1111/j.1558-5646.2010.01051.x. [DOI] [PubMed] [Google Scholar]
- Ventura CV, Maia M, Bravo-Filho V, Góis AL, Belfort R. Zika virus in Brazil and macular atrophy in a child with microcephaly. Lancet. 2016;387:228. doi: 10.1016/S0140-6736(16)00006-4. [DOI] [PubMed] [Google Scholar]
- Villegas-García JC, Santillán-Alarcón S. Sylvatic focus of American trypanosomiasis in the State of Morelos, Mexico. Revista de Biologia Tropical. 2001;49:685–688. [PubMed] [Google Scholar]
- Weaver SC, Barrett ADT. Transmission cycles, host range, evolution and emergence of arboviral disease. Nature Reviews Microbiology. 2004;2:789–801. doi: 10.1038/nrmicro1006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- World Health Organization (WHO) (2016) Report Zika Situation Neurological Syndrome and Congenital Anomalies. Available from: http://www.who.int/emergencies/zika-virus/situation-report/5-february-2016/en/.
- Zanluca C, de Melo VCA, Mosimann ALP, dos Santos GIV, dos Santos CND, Luz K. First report of autochthonous transmission of Zika virus in Brazil. Memorias do Instituto Oswaldo Cruz. 2015;110:569–572. doi: 10.1590/0074-02760150192. [DOI] [PMC free article] [PubMed] [Google Scholar]


