Abstract
A prospective study was conducted from November 2013 to February 2014 to estimate the spatial clustering; cumulative incidence and risk factors associated with avian influenza (AI) subtype H9 infection on commercial poultry farms of Pakistan. A total of 400 farms were enrolled and followed during the study period. Among these, 109 farms submitted samples suspected for AI to the laboratory, and only 47 farms were confirmed positive by hemagglutinin inhibition (HI) test. Data was collected from these 109 farms about their demography, management, and biosecurity practices. The cumulative incidence of H9N2 was 11.75 % (95 % confidence interval (CI) 8.76–15.23). The highest number of cases (40.42 %) was reported in January. One most likely cluster (p = 0.009, radius = 4.61 km) occurred in the Kasur district. Multivariable logistic regression analyses showed that the presence of wild birds on the farms (odds ratio (OR) = 16.18; 95 % CI 3.94–66.45) was independently associated with H9N2 infection. Cleaning of cages before delivery on farm (OR = 0.16; 95 % CI = 0.06–0.47), presence of a footbath at the entrance of farm (OR = 0.24; 95 % CI 0.08–0.79), and changing of gloves (OR = 0.33; 95 % CI 0.11–0.99) were protective factors against H9N2 infection. Reducing the exposure to risk factors and adapting biosecurity measures may reduce the risk of AI H9N2 infection on commercial poultry farms in Pakistan.
Keywords: Avian influenza, Attack rate, Prospective study, H9N2 serotype, Commercial poultry farms, Zoonosis
Introduction
Influenza is a continuing threat to human and animal health. Every year, thousands of people are infected with seasonal influenza and may be exposed to subtypes of avian (H5, H6, H7, H9, and H10) and swine (H1 and H3) origin (García-Sastre and Schmolke 2014).
Avian influenza viruses (AIVs) of subtype H9N2 have spread widely since their first identification in turkeys in Wisconsin, USA, in 1966 (Homme and Easterday 1970). H9N2 viruses were isolated from pigs in 1998 and were subsequently isolated from humans with an influenza-like illness in both Hong Kong and Mainland China (Peiris et al. 1999). H9N2 are significantly important due to their extensive circulation in domestic poultry in different regions of world from the Far East to the Middle East (Fusaro et al. 2011). Genetic analysis of H9N2 viruses has showed extensive re-assortment of these viruses with many subtypes of AIVs including HPAI H5N1 and H7N3 viruses (Chaudhry et al. 2015; Fusaro et al. 2011).
In Pakistan, commercial poultry production has attained the shape of an industry in recent years with investment of billions of rupees. Since 1995, AIV subtypes H9, H7, and H5 are responsible for five massive epidemics in Pakistan affecting poultry and poultry products across the country (Naeem et al. 2007). Although H9N2 viruses are of low pathogenicity, the frequent heavy losses caused by them have raised serious concerns for the poultry industry in many countries.
Advancement has been made in disease investigations with new tools like geographic information system (GIS), which is used for spatiotemporal analysis of important emerging infections, e.g., severe respiratory syndrome (SARS), AIV H5N1, and influenza A (H1N1) (Tiensin et al. 2009; Martin et al. 2011; Lai et al. 2013). Disease clustering can be detected by using space-time scan statistics (Kulldorff et al. 2005).
Few studies have examined the association of risk factors with AI on poultry farms in Pakistan (Abbas et al. 2012; Chaudhry et al. 2015). To date, very little information is available on spatial clustering of H9 infection in this region. Awareness about risk factors responsible for disease introduction and spatial clustering is critically important in developing risk-based surveillance strategies, policies, and timely recommendation for control. The primary objectives of this study were to calculate attack rate (AR) of H9N2 infection and to identify risk factors associated with this infection among poultry farms of Pakistan. The other objective was to identify any clustering of unusually high number of H9 cases than expected for early detection of any emerging outbreak of this disease in different areas of Pakistan when only the number of cases is available.
Materials and methods
A prospective study was conducted from November 2013 to February 2014. All commercial poultry farms of Pakistan raising domesticated poultry for sale were considered as the target population of study. The final study population was commercial poultry farms submitting samples for laboratory analysis to the collaborating private poultry laboratory for routine screening and suspected infections. Each commercial farm was taken as a sampling unit. All poultry farms, which were included in the study, were considered negative for H9 at the start of study due to the absence of any influenza or influenza-like illness in the flock. None of broiler flock was vaccinated against H9, H5, or H7. Breeder and layers were vaccinated against H9.
A total of 400 commercial poultry farms of different production categories (breeders, broiler, and layer farms) located in Punjab Province and Islamabad Capital Territory of Pakistan were enrolled in the study. Out of these 400 farms, only 109 farms submitted samples to laboratory for suspected infection with AIV, and a pretested questionnaire was filled from the owner/supervisor of these 109 farms in a face-to-face interview after explaining the objectives of study to the farmers. Prior to interview, written consent of the owner/attendant was obtained. The questionnaire contained 26 questions about risk factors, which were known to influence the disease occurrence and were selected after reviewing literature about AI (Nishiguchi et al. 1999; Ward et al. 2008; McQuiston et al. 2005; Fang et al. 2008; Woo and Park 2008; Abbas et al. 2012; Chaudhry et al. 2015; Nishiguchi et al. 2007) and from the observations of technical staff working on these farms.
The farmers were requested to provide five to ten dead birds from total mortality on farm, which were carefully examined by conducting postmortem examination for specific disease lesions. Typical pathological lesions in respiratory system, i.e., rhinitis, sinusitis, congestion, and inflammation in the trachea (Swayne 2008), were suspected for AIV. Confirmation was done by Anigen Rapid AIV Ag Detection Kit (BIONOTE Inc., Korea). The outcome of interest was H9 status, i.e., infected and non-infected farms. Samples confirmed by rapid test were further tested by virus isolation in embryonated chicken eggs, and subtyping was done by hemagglutinin inhibition (HI) test.
AR of H9N2 was calculated (Thrusfield 2007). All biologically plausible and relevant variables were screened in univariable analysis by using glm function of the epicalc package (version, 2.15.1.0) in R statistical software (available at http://www.R-project.org). A multivariable model was derived by forward stepwise selection procedure (Dohoo et al. 2003). Variables with significant univariable relationship at P < 0.25 were selected for inclusion in the final model. Odds ratios (ORs) and corresponding 95 % confidence intervals (CIs) were calculated (Hosmer and Lemeshow 2000).
Space-time scan statistic
All laboratory-confirmed cases of H9N2 between December 2013 to February 2014 were geocoded to street addresses. SatScan software version 9.1.1 developed by Martin Kulldorff, Havard Medical School (Boston, USA) and the Information Management Services Inc. (Maryland, USA) was used (available at http://www.satscan.org). The prospective space-time permutation scan statistic module was used to detect a local excess of events and to test if this excess could have occurred by chance (Hyder et al. 2011). This method consists of thousands of cylinders that move across space and/or time. Each cylinder has a base, which represents geographical area (in this study, a commercial farm), and height, which is time (in this study, a day). The base of each cylinder comprised a maximum of 50 % of the population, while height was a maximum of 50 % of the study time (60 days). The cylinder with more observed cases than expected, with respect to cases reported outside the cylinder, is called “most likely cluster.” For each location and size of the cylinder, the number of observed and expected cases is counted. Among these, the most “unusual” excess of observed cases is noted. The statistical significance of this cluster is then evaluated taking into account the multiple testing stemming from the many potential cluster locations and sizes evaluated (Kulldorff et al. 2005). ArcGIS version 10 was used for the map display.
Results
During the study, we followed 400 commercial poultry farms of which 109 submitted samples. Among these 109 farms, 47 poultry farms get infected with H9. The AR among total enrolled farms (47/400) was 11.75 % (95 % CI 8.76–15.23), while AR among total examined (47/109) was 43.10 % (95 % CI 34.20–52.50). Among the infected farms, majority (66 %) reported 5–9 % mortality due to H9 (Table 1).
Table 1.
Average mortality from AIV subtype H9 infection
| S. no | Mortality on the farms (%) | No. of farms | Average (%) among total (47) |
|---|---|---|---|
| 1 | 5–9 % | 31 | 66.0 |
| 2 | 10–14 % | 10 | 21.3 |
| 3 | 15 % and above | 6 | 12.8 |
The AR was highest in Lahore district (11/28) followed by Kasur (24/38) and Sheikhupura (6/13) districts (Fig. 1). No sample was positive for Newcastle disease virus, H5, and H7 AIVs.
Fig. 1.
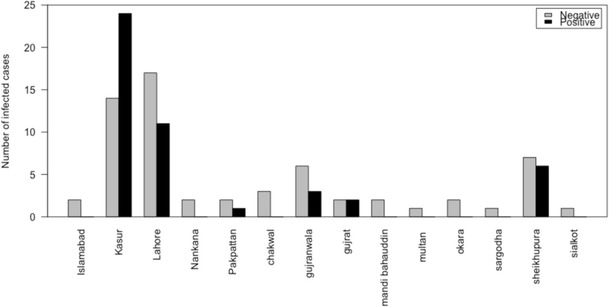
Attack rate of H9 infection in different districts of Pakistan
The study showed a high AR in the month of January (21/34) followed by December (11/29), November (9/23), and lowest incidence in February (6/23) (Fig. 2).
Fig. 2.
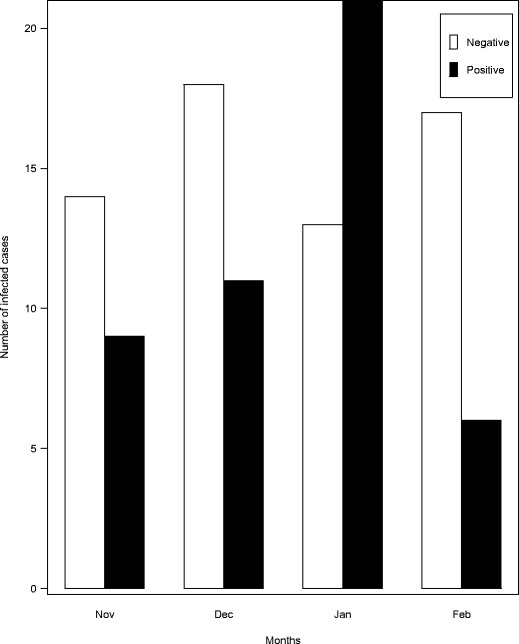
Frequency of positive cases according to reporting month in the study
Risk factors identified during the study
Out of 26, 11 factors were selected for inclusion in final model of multivariable analysis (Table 2). Factors with p > 0.25 were excluded from further analysis.
Table 2.
Univariable analysis of potential factors for AIV subtype H9 infection
| Factors | Level of response | H9 +ve | H9 −ve | OR | 95 % CI | p value |
|---|---|---|---|---|---|---|
| Wild birds on farm | Yes | 44 | 36 | 10.59 | 2.96–37.86 | 0.000281a |
| No | 3 | 26 | ||||
| Dropping removal | Before catching | 5 | 47 | 1.0280 | 1.01–7.73 | 0.0476a |
| After catching | 42 | 15 | ||||
| Farm fully fenced | Yes | 3 | 17 | 0.18 | 0.05–0.66 | 0.0096a |
| No | 44 | 45 | ||||
| Rubber boots channing | Yes | 12 | 33 | 0.3 | 0.13–0.69 | 0.00432b |
| No | 35 | 29 | ||||
| Clean cages before entering farm | Yes | 9 | 31 | 0.24 | 0.1–0.57 | 0.00135b |
| No | 38 | 31 | ||||
| Movement of workers within farms | No | 34 | 55 | 3 | 1.09–8.28 | 0.0334a |
| Yes | 13 | 7 | ||||
| Gloves changing | Yes | 8 | 24 | 0.32 | 0.13–0.81 | 0.162 |
| No | 39 | 38 | ||||
| Vehicles entry into the farm | Yes | 38 | 42 | 2.01 | 0.82–4.95 | 0.12a |
| No | 9 | 20 | ||||
| Foot bath | Yes | 40 | 44 | 0.43 | 0.16–1.13 | 0.014b |
| No | 7 | 18 | ||||
| Share equipment | Yes | 7 | 4 | 2.537 | 0.7–9.24 | 0.1580a |
| No | 40 | 58 | ||||
| Waste disposal | Properly disposed | 9 | 1 | 0.24 | 0.02–2.39 | 0.224a |
| Not properly disposed | 38 | 61 | ||||
| Ventilation system | Fan | 45 | 54 | 3.33 | 0.67–16.5 | 0.1401a |
| Natural | 2 | 8 |
aRisk factors
bProtective factors
In the final multivariable model, four factors were identified as significant (Table 3). Among those four factors, one factor was identified as risk factor (OR > 1), i.e., wild birds on the farm, and three factors were proved to be protective factor (OR < 1), namely cleaning of cages before entering the farm area, having foot bath/dipping area at the entrance of farm, and workers change gloves while entry into bird area.
Table 3.
Multivariable analysis of potential risk factors
| S. no | Factors | Level of response | Positive | Negative | OR | 95 % CI | p value |
|---|---|---|---|---|---|---|---|
| 1 | Wild birds entry into the farm | Yes | 44 | 36 | 16.18 | 3.94–66.45 | <0.001 |
| No | 3 | 26 | |||||
| 2 | Cleaning of cages before delivery | Yes | 9 | 31 | 0.16 | 0.06–0.47 | <0.001 |
| No | 38 | 31 | |||||
| 3 | Foot bath dipping area at the entrance | Yes | 40 | 44 | 0.24 | 0.08–0.79 | 0.018 |
| No | 7 | 18 | |||||
| 4 | Worker change gloves | Yes | 8 | 24 | 0.33 | 0.11–0.99 | 0.048 |
| No | 39 | 38 |
Spatiotemporal cluster analysis
Total examined farms in Punjab Province were 107, while 2 farms were examined from Islamabad Capital Territory (Fig. 3).
Fig. 3.
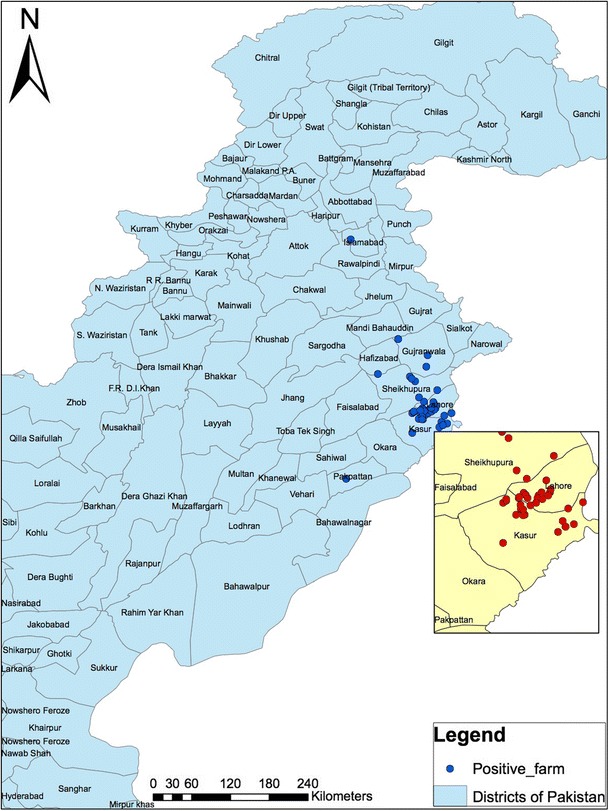
Spatial distribution of positive poultry farms in different districts of Pakistan
From November 1, 2013 to February 28, 2014, one most likely cluster (p = 0.009, radius = 4.61 km) occurred in the Kasur district of Punjab, Pakistan (Fig. 4). This signal had four cases observed over 25 days when 0.52 cases were expected [relative risk (RR) = 7.67], with a null occurrence rate of once every 111 days.
Fig. 4.
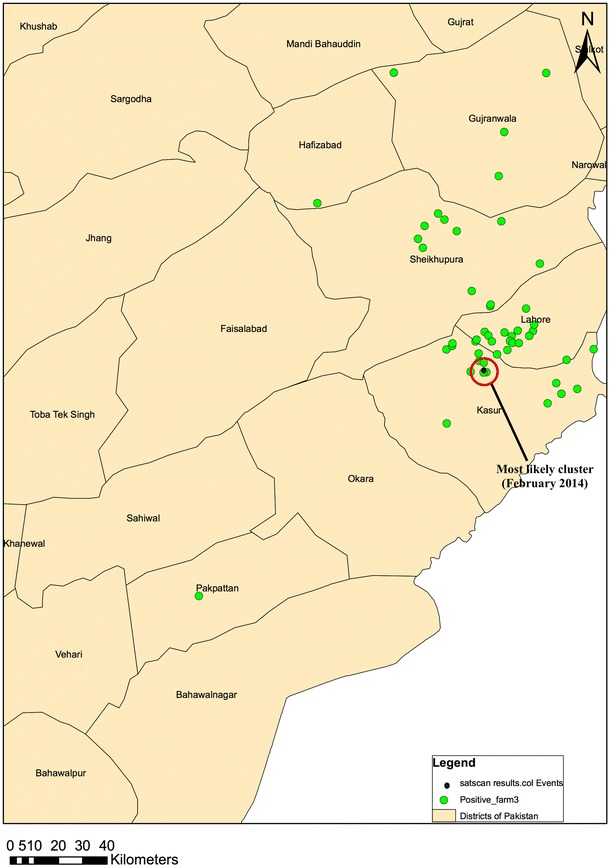
Spatiotemporal cluster of cases of H9N2 in Kasur district of Pakistan, November 2013 to February 2014
Discussion
Attack rate was highest in Lahore district (17/28) followed by Kasur (24/38) and Sheikhupura (6/13) districts. The reason for this high AR could be the high density of commercial poultry farms in these districts. Association of HPAI H5N1 and high and medium density of poultry farms has been studied and was identified as a risk factor in different countries (Henning et al. 2009; Abbas et al. 2012). Poultry farm densities in these regions ranged from 0.05 to 4 farms per square kilometer (Hamilton et al. 2009). As distance between the farms plays a significant role in the transmission of infection, poultry farms in these areas are exposed almost every year during the endemic (Abbas et al. 2012; Chaudhry et al. 2015). Furthermore, in densely populated poultry areas, movement of vehicles and people from farm to farm is considerably high, subsequently facilitating the spread of this virus through fomites. In the current study, the two districts (Lahore and Kasur) are on the main highway road (Grand Trunk road), which have a significant number of commercial poultry farms alongside. The vehicles over loaded with infected birds or mortality move on this road routinely. Previously, many studies have demonstrated that proximity to major roads was associated with avian influenza (Ward et al. 2008; Chaudhry et al. 2015). Movement of veterinarian and para-veterinary staff between different farms to implement control measures or to investigate mortalities can also contribute to the spread of virus among farms. In Pakistan, poultry farmers who apply strict biosecurity measures sometimes relax these rules for visitors, who enter the bird areas (Chaudhry et al. 2015).
Spatiotemporal analysis detected a most likely cluster in Kasur district (p = 0.009, radius = 4.61 km). Poultry farm in this cluster has a significantly higher risk of being infected as compared to poultry farms outside the cluster (RR = 7.67). These results represent the most important hotspot of expected outbreak and are valuable for improving knowledge and understanding of spatial pattern of H9N2 in specific areas. Targeted surveillance of these districts is needed for early detection of any future AI outbreak and their possible re-assortment. Spatial analyses have been used to study different outbreaks of human and avian influenza infections (Tiensin et al. 2009; Leveau et al. 2015).
Risk factor analyses showed that the presence of wild birds on farm could enhance the probability of infection (OR = 16.18; 95 % CI 3.94–66.45). Wild birds could serve as a potential source of propagation of AI virus especially when biosecurity measures are poorly implemented on farm. They can act both as mechanical and biological vectors (shedding the virus in droppings) and are important source of introducing virus to new areas (McQuiston et al. 2005; Henning et al. 2009; Chaudhry et al. 2015).
Cleaning of cages before delivery was strongly associated with decrease in risk of H9 (OR = 0.16, 95 % CI 0.06–0.47). Though statistically non-significant, cleaning of cages has been studied as a protective factor in lowering risk of H9 infection (Chaudhry et al. 2015). Poultry traders are well aware about the importance of cleaning cages and vehicles as an effective biosecurity measure (Kurscheid et al. 2015).
Presence of a footbath/dipping area at the entrance of farm and changing of gloves are also important as part of biosecurity measures on farm and have proved effective in decreasing risk of AI previously (Biswas et al. 2009; Chaudhry et al. 2015).
This study found evidence of clustering, in space and time, and identified some well-known factors mainly responsible for increasing risk of AIV infection. Enhancing good management practices and strict biosecurity can lower the risk of infection among poultry farms. Spatial clustering of disease provides information to health authorities to more effectively target and improve their surveillance and control strategies in affected areas.
Acknowledgments
The authors are highly indebted to respondents of the commercial farms, who participated in study for data collection. We also acknowledge the support of staff of commercial poultry laboratory for providing us data from logbook about case farms, which were included in the study. Without their great cooperation and help, this study would not have been possible.
Abbreviations
- AI
Avian influenza
- AIVs
Avian influenza viruses
- GIS
Geographic information system
- HI
Hemagglutinin inhibition test
- OR
Odds ratios
- CI
Confidence intervals
- RR
Relative risk
- AR
Attack rate
Compliance with ethical standards
Ethical approval
This article does not contain any studies with animals performed by any of the authors.
Informed consent
The manuscript does not contain clinical studies or patient data. The owners of the commercial farms were briefed about the objective of study, and informed consent was obtained from all individual participants included in the study to collect data.
Conflict of interest
The authors declare that they have no conflict of interest.
References
- Abbas T, Wilking H, Horeth-Bontgen D, Conraths FJ. Contact structure and potential risk factors for avian influenza transmission among open-sided chicken farms in Kamalia, an important poultry rearing area of Pakistan. Berliner und Münchener tierärztliche Wochenschrift. 2012;125:110–116. [PubMed] [Google Scholar]
- Biswas, P. K., Christensen, J. P., Ahmed, S., Barua, H., Das, A., Rahman, M., Giasuddin, M., Hannan, A., Habib, A., Debnath, N., 2009. Risk factors for infection with highly pathogenic influenza A virus (H5N1) in commercial chickens in Bangladesh. Veterinary Record, 164 [DOI] [PubMed]
- Chaudhry M, Rashid HB, Thrusfield M, Welburn S, Bronsvoort MB. A case-control study to identify risk factors associated with avian influenza subtype H9N2 on commercial poultry farms in Pakistan. PLoS ONE. 2015;10(3):e0119019. doi: 10.1371/journal.pone.0119019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dohoo, I., Martin, W., Stryhn, H., 2003. Veterinary epidemiologic research (AVC Inc, Charlottetown, Prince Edward Island, Canada)
- Fang, L., de Vlas, S.J., Liang, S., Looman, C.W.N., Gong, P., Xu, B., Yan, L., Yang, H., Richardus, J. H., Cao, W., 2008. Environmental factors contributing to the spread of H5N1 avian influenza in Mainland China. PLoS One, 3, http://dx.doi.org/10.1371/journal.pone.0002268 [DOI] [PMC free article] [PubMed]
- Fusaro A, Monne I, Salviato A, Valastro V, Schivo A, Amarin NM, Gonzalez C, Ismail MM, Al-Ankari AR, Al-Blowi MH, Khan OA, Maken Ali AS, Hedayati A, Garcia Garcia J, Ziay GM, Shoushtari A, Al Qahtani KN, Capua I, Holmes EC, Cattoli G. Phylogeography and evolutionary history of reassortant H9N2 viruses with potential human health implications. J Virol. 2011;85(16):8413–8421. doi: 10.1128/JVI.00219-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- García-Sastre, A., Schmolke, M., 2014. Avian influenza A H10N8?a virus on the verge? Lancet 2014; 383: 676–677. [DOI] [PubMed]
- Hamilton SA, East IJ, Toribio JA, Garner MG. Are the Australian poultry industries vulnerable to large outbreaks of highly pathogenic avian influenza? Australian Veterinary Journal. 2009;87:165–174. doi: 10.1111/j.1751-0813.2009.00423.x. [DOI] [PubMed] [Google Scholar]
- Henning KA, Henning J, Morton J, Long NT, Ha NT, Meers J. Farm-and flock-level risk factors associated with highly pathogenic avian influenza outbreaks on small-holder duck and chicken farms in the Mekong Delta of Viet Nam. Preventive Veterinary Medicine. 2009;91:179–188. doi: 10.1016/j.prevetmed.2009.05.027. [DOI] [PubMed] [Google Scholar]
- Homme PJ, Easterday BC. Avian influenza virus infections: I. Characteristics of influenza A-turkey-Wisconsin-1966 virus. Avian Diseases. 1970;14:66–74. doi: 10.2307/1588557. [DOI] [PubMed] [Google Scholar]
- Hosmer DW, Lemeshow S. Applied logistic regression. New York: John Wiley & Sons, Inc; 2000. [Google Scholar]
- Hyder K, Vidal-Diez A, Lawes J, Sayers AR, Milnes A, Hoinville L, Cook AJC. Use of spatiotemporal analysis of laboratory submission data to identify potential outbreaks of new or emerging diseases in cattle in Great Britain. BMC Veterinary Research. 2011;7:14. doi: 10.1186/1746-6148-7-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kulldorff M, Heffernan R, Hartman J, Assunçāo R, Mostashari F. A space-time permutation scan statistics for disease outbreak detection. PloS Medicine. 2005;2(3):e59. doi: 10.1371/journal.pmed.0020059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kurscheid J, Millar J, Abdurrahman M, Ambarawati IGAA, Suadnya W, Yusuf RP, Fenwick S, Toribio JA. Knowledge and perceptions of highly pathogenic avian influenza (HPAI) among poultry traders in live bird markets in Bali and Lombok, Indonesia. PLoS ONE. 2015;10(10):e0139917. doi: 10.1371/journal.pone.0139917. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lai PC, Kwong KH, Wong HT. Spatio-temporal and stochastic modeling of the severe acute respiratory syndrome (SARS) Geospatial Health. 2013;8(1):183–92. doi: 10.4081/gh.2013.65. [DOI] [PubMed] [Google Scholar]
- Leveau C, Uez O, Vacchino MN. Spatiotemporal trends of cases of pandemic influenza A (H1N1)pdm09 in Argentina, 2009-2012. Revista do Instituto de Medicina Tropical de São Paulo. 2015;57(2):133–138. doi: 10.1590/S0036-46652015000200006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin V, Pfeiffer DU, Zhou X, Xiao X, Prosser DJ, Guo F, Gilbert M. Spatial distribution and risk factors of highly pathogenic avian influenza (HPAI) H5N1 in China. PLoS Pathogen. 2011;7(3):e1001308. doi: 10.1371/journal.ppat.1001308. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McQuiston JH, Garber LP, Porter-Spalding BA, Hahn JW, Pierson FW, Wainwright SH, Senne DA, Brignole TJ, Akey BL, Holt TJ. Evaluation of risk factors for the spread of low pathogenicity H7N2 avian influenza virus among commercial poultry farms. Journal of American Veterinary Medicine Association. 2005;226:767–772. doi: 10.2460/javma.2005.226.767. [DOI] [PubMed] [Google Scholar]
- Naeem K, Siddique N, Ayaz M, Jalalee MA. Avian influenza in Pakistan: outbreaks of low- and high-pathogenicity avian influenza in Pakistan during 2003-2006. Avian diseases. 2007;51:189–193. doi: 10.1637/7617-042506R.1. [DOI] [PubMed] [Google Scholar]
- Nishiguchi A, Kobayashi S, Yamamoto T, Ouchi Y, Sugizaki T, Tsutsui T. Risk factors for the introduction of avian influenza virus into commercial layer chicken farms during the outbreaks caused by a low-pathogenic H5N2 virus in Japan in 2005. Zoonoses Public Health. 2007;54:337–343. doi: 10.1111/j.1863-2378.2007.01074.x. [DOI] [PubMed] [Google Scholar]
- Peiris M, Yuen KY, Leung CW, Chan KH, Ip PL, Lai RW, Orr WK, Shortridge KF. Human infection with influenza H9N2. Lancet. 1999;354(9182):916–917. doi: 10.1016/S0140-6736(99)03311-5. [DOI] [PubMed] [Google Scholar]
- Swayne, D. E. 2008. Avian influenza. In: Foreign animal diseases, (Boca Raton, FL: United States Animal Health Association), 137-146.
- Thrusfield, M., 2007. Veterinary Epidemiology (Oxford, Blackwell Science. UK)
- Tiensin, T., Ahmed, S.S., Rojanasthien, S., Songserm, T., Ratanakorn, P., Chaichoun, K., Kalpravidh, W., Wongkasemjit, S., Patchimasiri, T., Chanachai, K., Thanapongtham, W., Chotinan, S., Stegeman, A., Nielen, M., 2009. Ecologic risk factor investigation of clusters of avian influenza A (H5N1) virus infection in Thailand. Journal of Infectious Diseaes, 199, 1735–1743 [DOI] [PubMed]
- Ward MP, Maftei D, Apostu C, Suru A. Environmental and anthropogenic risk factors for highly pathogenic avian influenza subtype H5N1 outbreaks in Romania, 2005–2006. Veterinary Research Communication. 2008;32:627–634. doi: 10.1007/s11259-008-9064-8. [DOI] [PubMed] [Google Scholar]
- Woo JT, Park BK. Seroprevalence of low pathogenic avian influenza (H9N2) and associated risk factors in the Gyeonggi-do of Korea during 2005–2006. Journal of Veterinary Science. 2008;9:161–168. doi: 10.4142/jvs.2008.9.2.161. [DOI] [PMC free article] [PubMed] [Google Scholar]


