Abstract
The common cold is the most frequent, although generally mild, human disease. Human Rhinoviruses are the prevalent causative agents, but other viruses are also implicated. Being so common, viral colds, have significant implications on public health and quality of life, but may also be life-threatening for vulnerable groups of patients. Specific diagnosis and treatment of the common cold still remain unmet needs. Molecular diagnostic techniques allow specific detection of known pathogens as well as the identification of newly emerging viruses. Although a number of medications or natural treatments have been shown to have some effect, either on the number or on the severity of common colds, no single agent is considerably effective. Virus-specific management remains in most cases a challenging potential as many factors have to be taken into account, including the diversity of the viral genomes, the heterogeneity of affected individuals, as well as the complexity of this long standing host-virus relationship.
Keywords: Common cold, Prevention, Cure, Rhinovirus, Upper respiratory tract infections, Antiviral therapy, Vaccines, Influenza, RSV, Molecular diagnosis, Management, Passive immunization, Vaccination
Introduction
The common cold is an acute viral infection of the upper respiratory tract (URTI) that is usually self-limited. In the European Position Paper on Rhinosinusitis and Nasal Polyps 2012 (EPOS 2012), common cold is defined as acute viral rhinosinusitis with symptoms lasting less than 10 days [1]. It is the most frequent human illness, with approximately 25 million of affected individuals annually in the U.S.A. [2]. As a result, it is associated with significant burden in terms of medical visits as well as work and school absenteeism. Because of its increased frequency, the overall impact of this relatively mild clinical entity is considerable [3]. Moreover, the lack of established diagnostic procedures or specific therapy, result in the use of diverse over-the-counter medications, whereas significant number of medical visits (up to 30 %) result in inappropriate and unnecessary antibiotic prescription, contributing to antibiotic overuse and microbial resistance[4]. Finally, the common cold can be a trigger for severe and even fatal disease in individuals with preexisting conditions. For all the above reasons, prevention and effective treatment of this condition are important unmet needs.
Epidemiology
Several studies have demonstrated that adults usually experience 1–3 URTIs per year [5], whereas children have considerably more (up to 11, depending on age) and experience more prolonged symptoms [6]. The rates of common cold per year usually decline with age. Other risk factors include day-care attendance [7], genetic factors [8], psychological stress [9], smoking [10] and heavy physical training [11].
The list of pathogens that may cause common cold symptoms include human rhinoviruses (RVs), respiratory syncytial virus (RSV), influenza viruses, parainfluenza viruses, coronaviruses and adenoviruses. Less frequently, enteroviruses (coxsackieviruses, echoviruses), bocavirus, EBV and human metapneumovirus (hMPV) [12] are implicated. However, among all causal agents, RVs are the most common, with up to 80 % of cases [13].
In the North hemisphere, the incidence of the common cold is low during the summer, and increases from late August/beginning of September until early spring [14]. Factors that contribute to the increased incidence of URTIs during the colder months include time spent indoors, resulting in a higher possibility of intimacy to infected individuals, and increased levels of environmental humidity and low indoor moisture that favor the survival of most viruses. Coronavirus infections are mainly seen in the winter and early spring, whereas enterovirus respiratory infections occur primarily during early autumn and summer. The infuenza season is between November and March, whereas for parainfluenza (PIV), PIV1 & PIV2 are usually isolated during autumn whereas PIV3 causes outbreaks during spring and early summer [15]. RSV and hMPV peak incidence is from December to February [16]. Similarly, adenoviral respiratory infections have a peak incidence in late winter, spring and early summer. Human bocavirus (HBoV) URTIs are more common during the winter most frequently as co-infections [17]. During winter, RVs are not the main cause of colds [12]. In temperate climates, RV infection peaks during early fall and spring, with a second smaller peak during summer months [18], whereas in tropical regions, it mainly presents during the rainy months [19]. Nevertheless, RVs are the most common viruses found in the respiratory tract, independent of season [20, 21].
Pathophysiology
Transmission of viruses causing URTIs may occur via inhalation of viral-particles or by hand contact [22].
The pathophysiology of common cold represents a dynamic interplay between host immunity and the infecting virus (Fig. 1). A considerable amount of information comes from studies of RV infection in volunteers [23]. After deposition in the nasopharynx, RV attaches to specific receptors on epithelial cells; the intracellular adhesion molecule-1 (ICAM-1) receptor for RV major serotypes, the low-density lipoprotein (LDL) receptor for RV minor serotypes and a yet unknown receptor for type-C RVs. It then initiates inflammation via an NF-kB-dependent mechanism [24•]. In contrast to other respiratory viruses such as RSV and influenza, RV does not have major cytopathic effects on upper respiratory tract airway epithelial cells [25]. It is thus recognized that common cold symptoms are not caused by the cytopathic effects of RV on human cells, but rather by the inflammatory response [26•]. However, RVs disrupt the epithelial barrier function[27], facilitating epithelial cells exposure to bacteria and promoting secondary bacterial infections, as well as other external stimuli such as irritants and allergens.
Fig. 1.
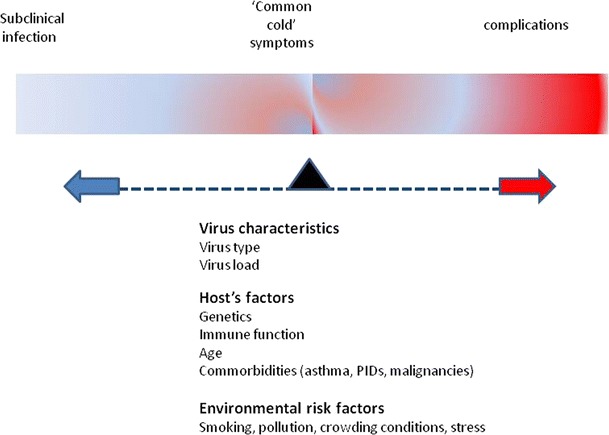
Common cold symptoms and possible complications result from a dynamic interplay between infecting virus characteristics and host’s immune response. In the majority of cases, common cold is a relatively mild and self limited illness. However, affected individuals with defective immunity -either because of an underlying condition (i.e. asthma, immunodeficiency, young or old age) or as a result of environmental conditions (i.e. smoking, stress, pollution)- may experience severe (even fatal) complications.
Clinical Presentation & Complications
Common cold symptoms are indistinguishable among different viral causes, and include sore throat, nasal congestion, rhinorrhea, sneezing, cough, low-grade fever, malaise and headache. Following inoculation, the incubation period of viruses varies significantly; 12 hours for influenza B, 1.5 days for influenza A, 3 days for coronavirus, 4 days for RSV or PAI and 5.5 days for adenovirus, whereas 12-72 h, (usually 24-48 h), for RVs [28, 29]. Symptoms peak at 2–3 days post-inoculation and generally last for 7–11 days, but occasionally longer [30]. RVs can also be detected in 12.5 % to 33 % of asymptomatic children [31, 32], but only in 2 % of adults [33].
Although in immunocompetent hosts the common cold is mild and usually self-limited, it has been linked with several complications.
Acute otitis media (AOM) presents in more than one-third of viral URTIs in children [34], resulting from changes in middle ear pressure and eustachian tube function or by increasing the susceptibility to bacterial co-infection in the middle ear [35].
Similarly, sinuses frequently present with effusion on computed tomography (CT) and magnetic resonance imaging (MRI) [36] during URTIs, possibly through the increased intranasal pressure during nose blowing. Moreover, pre-existing chronic rhinosinusitis may present with exacerbations triggered by URTIs, especially in combination with cigarette smoking [1].
Postviral olfactory disorders, including parosmia, phantosmia, hyposmia, or anosmia, are associated with common cold in a percentage varying from 11 % to 40 % in different studies [37]). These disorders most commonly affect middle and old aged people, mainly women. In one third of cases, spontaneous recovery may occur within 2 years. Yet no specific therapy can be applied.
Common cold complications are not restricted to the upper airways, as common cold viruses, including RV [27], have the potential to infect the lower airways. In addition to laryngotracheobronchitis and bronchiolitis, which usually start with an URTI, RVs have been isolated in 15-25 % of children hospitalized with community acquired pneumonia (CAP) [38]. In all the aforementioned cases, bacterial co-infection is present in a range of 50 %-60 % [39]. Viral pathogens are recognized as causes of CAP in adults in lower rates. However, in the elderly, RV infections are associated with significant morbidity and mortality [39].
RV is critically implicated in asthma exacerbations [40, 41], through innate immune response deficiency in asthmatic individuals [42]. RV infection has also an important role in exacerbations of chronic obstructive pulmonary disease (COPD)[43] and cystic fibrosis [44].
Immunocompromised individuals, including children with primary immunodeficiencies and patients with organ transplantations, malignancies, HIV-infection, diabetes and auto-immune diseases are extremely susceptible to infections, including those caused by common cold viruses[45, 46•].
Diagnosis
Identification of causative pathogens is a crucial step for subsequent, potentially specific, management.
Samples for the identification of upper respiratory pathogens include nasopharyngeal (NP) aspirates, NP washes, NP swabs, NP swabs plus oropharyngeal (OP) swabs [47], and liquid specimens, such as sputum, tracheal aspirates and bronchoalveolar lavages (BAL). The sensitivity and specificity of virus detection for each sampling method can vary significantly [48].
Antigen detection tests and different cell cultures formats have been used. A new chip to detect antibody responses to different RV subtypes is currently being developed in the context of the EU project (www.predicta.eu, News, April 2013).
PCR has proved valuable in the diagnosis of viral infections, and particularly RV infections, for which other methods have been suboptimal [12]. It can be used in single virus detection and quantification assays as well as in multiplex formats allowing the simultaneous detection of up to 15 viruses and/or subtypes. Up to date, several real-time PCR assays such as hydrolysis probes, molecular beacons and dual hybridization probes have been incorporated into diagnostic devices to identify respiratory pathogens. The sensitivity, specificity, high detection rate, cost-effectiveness (especially in multiplexing), subtyping capability and adaptability to emerging new viruses [49] has made real-time RT-PCR the method of choice amongst most respiratory virus detection techniques. Furthermore, available multiplex RT-PCR-based approaches have significantly improved the diagnostic yield of co-infections. Other molecular based methods are also available, more in the research domain [50].
Despite the fact that molecular testing has improved specific diagnosis of respiratory viruses, its limitations cannot be ignored. For example, it can only be applied to a virus once its genome has been sequenced. PCRs do not measure viable virus. The detection of low levels of virus late in the course of infection, in a dual infection, or in asymptomatic people makes it difficult to determine the clinical relevance of the virus presence [51]. The lack of available external reference standards, difficulties in virus quantification, different levels of sensitivity when various types of clinical specimens are used, makes normalization hard to achieve or clinically undetermined [50].
Prevention
Physical Interventions
URT viruses are spread primarily through direct contact, airborne particles, and aerosols. Physical interventions include basic hygiene measures, which nevertheless, can be quite effective in preventing the transmission of the disease. Thorough hand washing [22], which is more effective than ethanol sanitizer [52], and general protection against auto-inoculation by minimizing finger-to-nose spread help to reduce the rates of transmission. Implementing barriers to transmission, such as isolation, and hygiene measures (wearing masks, gloves and gowns) can be effective in containing respiratory virus epidemics or in hospital wards [22].
Vaccination
is currently available only for influenza and is efficient in preventing infection. Recently a new Modified Vaccinia virus Ankara (MVA) vector encoding nucleoprotein and matrix protein 1 (MVA-NP + M1) was evaluated in phase I clinical trial in healthy adults and found to be both safe and immunogenic, leading to high frequencies of responding T cells [53•].
Despite the ongoing efforts there are no licensed vaccines for parainfluenza. HPIV3 cp45 is being evaluated in clinical trials [54].
Adenoviral vaccines delivered orally have been used for decades to prevent respiratory illnesses in USA military training installations[55].
Several approaches have been used to develop vaccines against RSV but currently no vaccines have been approved for use in humans[55]. Several viruses have been used as vectors for RSV vaccine to stimulate immunogenicity [56]. Intranasal administration gains more attention. Recently the immunostimulatory effects of TLR ligands have been investigated with encouraging results [57•, 58•].
As RV is the most common culprit, vaccine development is of great interest. An obstacle to overcome is the more than 100 different serotypes and the high-mutation rate during viral replication. In addition natural humoral immune response in humans is not well investigated and there are vague data on the dominant or more virulent strains. Recent progress in the sequencing of all known RV genomes [59], including the newly characterized RV-C [60•],as well as analysis of RV’s genomic signature [61•] makes the perception of a RV vaccine not as impossible as it used to seem in the past. The description of a mouse model of RV infection and immunization has allowed study of RV antibody responses [62]. Insights in the way that RV misdirects the immune response against non-neutralizing epitopes [63••] and identification of recombinant VP1 and VP4 proteins of the RV capsid, which can induce cross-neutralizing antibodies to diverse strains [64, 65•] have stimulated renewed interest in RV vaccination.
Passive Immunization
is only available for RSV prophylaxis of children with high risk of respiratory complications. Palivizumab reduces RSV hospitalizations by 50 % in premature infants and in children aged <24 months with hemodynamically congenital heart disease [66]. The more recent Motavizumab, was shown to be more effective but resulted in more hypersensitivity adverse events [66], hence has not yet been approved by FDA. MEDI-557, a long-acting anti-RSV MAb to be administrated only once or twice during the RSV season is under development [67]. Recently, RSV F-specific nanobodies that bind to epitopes with high affinity and specificity, were found to protect BALB/c mice from RSV infection and therapeutic intranasal administration resulted in reduced viral replication and reduced pulmonary inflammation [68].
Regarding influenza, two human antibodies, PN-SIA28 [69••] and Fi6v3 [70••], against hemagglutinin, the main target for the influenza-neutralizing antibody response, were found to be protective against several subtypes, suggesting new therapeutic options.
Treatment
Symptomatic Treatment
Since Sir William Osler said that “The only way to treat common cold is with contempt” little have changed. The treatment remains primarily supportive, including over-the-counter products aimed at symptom relief.
NSAIDs and acetaminophen are equally effective against common cold-induced pain and malaise [71, 72]. First-generation antihistamines improve runny nose and sneezing, but their use in children should be avoided [73]. In combination with decongestant they are more effective but have adverse effects such as drowsiness, dry mouth, insomnia and dizziness [74]. Second-generation antihistamines do not seem to have an effect [74]. Nasal and oral decongestants provide short term relief but their use is limited by adverse effects (rhinitis medicamentosa and rebound congestion) [75]. The potential immunomodulatory activities of pseudoephedrine, including interleukin-2 and tumor necrosis factor inhibition in-vitro, may be further explored [76]. Topical ipratropium reduces rhinorrhea and sneezing but has no effect on nasal congestion. Its use is well tolerated [77]. Steam inhalation, unlike the common belief, does not improve symptoms [78] and has been associated with severe scalds, especially in children [79]. Cough due to URTIs is usually transient and self-limited. A paucity of data exists for the efficacy of cough products [80]. Antitussives are not recommended for young children [81]. Current evidence does not support the use of intranasal corticosteroids for the common cold [75, 82]. Because of the viral etiology, antibiotics do not work for common cold and many people are affected by antibiotic side effects that may also contribute to community bacterial resistance to antibiotics[4].
Complementary and Alternative Treatment
Another approach, which has become very popular, is the use of herbal and nutritive remedies for general health maintenance, enhancing the immune system and treating of common cold. A major issue in regard to trials of herbal remedies is that there is no standardization of extracts used; combinations of various ingredients in several proportions further muddle the outcome. Additionally the occasionally strong beliefs of participants about treatment may influence the outcome [83].
Zinc inhibits RV replication in vitro, blocks RV binding to ICAM-1, alters the configuration of viral capsid proteins, and decreases histamine release [24•]. In clinical trials, it reduced the average duration but not the severity of the common cold in healthy people, when taken within 24 hours of onset. There is a significant reduction in the duration of cold at a dose of >/= 75 mg/day but the likely benefit has to be balanced against side effects (bad taste and nausea) [84]. Regarding prophylactic zinc supplementation, currently no firm recommendation can be made because of insufficient data [84].
Vitamin D is important for the innate immune response against URTIs. Three meta-analysis published in 2013 reached conflicting conclusions [85–87]. While low 25(OH) D levels were associated with increased risk of viral RTIs in children and adolescents in a dose-dependent manner [88•], monthly administration of 100 000 IU of vitamin D did not reduce the incidence or severity of URTIs in healthy adults [89]. Baseline levels of vitamin D, age, and dose of vitamine D need to be taken under consideration.
Vitamin C in doses of up to 4 g daily does not demonstrate any therapeutic benefit in symptom duration or severity [90]. A combination of 100 mg Vitamin C with 10 mg of Zinc may alleviate rhinorrhea [91]. Daily supplementation with large doses of vitamin C does not seem to prevent common colds; however modest but consistent effects in reducing the duration and severity of common colds have been demonstrated [92].
As with other herbal treatments, results on Echinacea are inconclusive [83, 93, 94]. In a recent randomized, double-blind, placebo-controlled trial, daily supplementation with Echinacea purpurea over a 4-month period resulted in reduction of the total number of colds. Echinacea inhibited virally confirmed colds and especially prevented enveloped virus infections [95].
Probiotics have a marginal effect on prevention and duration of colds [96–98]. In the elderly, probiotics appear to reduce the common cold incidence in a dose-dependent way [99, 100]. Heterogenecity of studies, strains and dose of probiotics tested does not allow to make a safe conclusion.
Quercetin inhibits viral endocytosis, RV and poliovirus protease activity, and RNA polymerase activity of some RNA viruses. In cell cultures and in C57BL/6 mice, treatment with quercetin after RV infection resulted in reduced viral load and inflammatory response [101•, 102]. However, in a randomized blinded trial, quercetin supplementation over 12 weeks had no influence on URTI rates or severity. A reduction in URTI total sick days and severity was noted only in middle aged and older subjects ingesting 1000 mg quercetin/day [103].
Arabinogalactan is a long polysaccharide which is naturally found in mycobacteria’s cells wall and plants. In a recent placebo-controlled trial, supplementation with 4.5 g of arabinogalactan over a period of 12 weeks, reduced the number of common cold episodes by 23 %, but had no effect on duration or severity of the episodes [104•].
Iota-caraageenan has antiviral effect against several respiratory viruses [105, 106]. Application of a nasal spray containing iota-carraageenan three times per day alleviated local symptoms of common cold, reduced the viral load in the nasal mucosa and reduced pro-inflammatory mediators; however, systemic symptoms remained the same [107]. In a cohort of 135 children, nasal application of iota-caraageenan did not alleviate symptoms but reduced the duration of the disease and the viral load and resulted in lower incidence of secondary infections with other respiratory viruses [108].
Beta-glucans are natural polysaccharides of the cell walls of Saccharomyces cerevisiae, fungi and some bacteria and they seem to have immunostimulatory effects [109]. In two recent trials they were able to reduce the occurrence of symptomatic common cold infections and reduced severity [109, 110].
The overall conclusion from the above is that although many of the over-the-counter treatments seem to have some activity against the common cold, this is not enough to produce robust results; therefore cost-effectiveness is questionable, while more effective treatments are needed.
A rational approach to tackle with common cold is the development of anti-viral agents. However, the diverse etiology makes the development of a uniform anti-viral drug unlikely.
Antiviral Agents
have been intensely investigated for many years. However, only a small number has reached the clinical trial phase, and even less, bedside. The only currently commercially available agents are against influenza and RSV. Amantadine and Rimantadine, the first antivirals against influenza have been replaced by neuraminidase inhibitors (NIs), Zanamivir and Oseltamivir, because of widespread resistance [111]. NIs are used for prophylaxis within 48 h after exposure and for the influenza treatment within 36 h after first symptoms. The treatment benefit is small (shortens symptoms by about 1 day) but may reduce disease severity [112]. Laninamivir octanoate which has in vitro neuraminidase-inhibitory activity against various influenza A and B viruses, including subtypes of N1 to N9 and oseltamivir resistant viruses, is currently being developed [113]. Ribavirin is the only approved therapy for lower respiratory tract disease caused by RSV. Ribavirin inhibits RSV-specific IgE production in nasal secretions, improves pulmonary function and may reduce the duration of severe RSV complications such as mechanical ventilation and hospitalization [114]. Inhibitors of RSV entry in the host cells, targeting the envelop F protein TMC-353121 and MDT-637 are being under development [115•] [116].
Extensive research efforts have led to the discovery of many potent antiviral agents against RV, but most have not found their way to the clinic [117, 118, 119•], mostly due to safety concerns. Capsid-binding inhibitors were among the first developed agents. Pleconaril was able to reduce the viral load and the duration of common cold by 1 day. Concerns regarding safety precluded further development as an oral treatment. The outcomes of a phase II study of an intranasal formulation of pleconaril indicate that there is no difference in the incidence of RV infections and asthma exacerbations between pleconaril and placebo group, however relevant publications are still pending (http://www.clinicaltrials.gov , NCT00394914). Vapendavir (BTA-798) exhibits antiviral activity against known RV-A and RV-B serotypes as well as selected RVs; activity against RV-C has not yet been established. In healthy volunteers vapendavir was well tolerated and reduced the peak viral load after experimental challenge[24•]; currently is being evaluated for the treatment of RV infections in patients with asthma (http://www.clinicaltrials.gov, NCT01175226). Another promising molecule was RV 3C protease inhibitor Ruprintrivir. Topical administration inhibited symptoms in experimental RV infections even when administered 24 h after exposure; however in a natural setting it failed to ameliorate clinical manifestations or viral load [24•].
Interferons regulate immune response to viruses by inhibiting virus replication and enhancing phagocytosis and cytotoxicity. Intranasal IFN-a2 reduces respiratory illness when administered either continuously during a respiratory virus season or intermittently as post-exposure prophylaxis in the family setting, but has little to no effect on the development of infection or symptoms when provided after infection [118]. Combining interferon with conventional compounds provides greater benefits [120]. However, adverse reactions (nasal irritation, mucosal friability, and bleeding) have limited its potential. Recently, a low-dose recombinant human IFNα-2b nasal spray was developed [121] in order to reduce adverse reactions and seem to be efficient for prevention of infections caused by influenza A and B, PIV 1–3 and adenovirus species but not RSV. New insights into the role of IFN-b in combating viral infections, nominate it as promising antiviral agent, especially for asthmatic individuals [122••].
In the context of exploring possible antiviral and/or anti-inflammatory means of therapy in common cold, a variety of pharmacological agents [123–131] that are beneficial in different disease entities (Table 1) have been also tested, however with inconclusive results.
Table 1.
Potential antiviral activity of drugs used for other conditions
| Description | Method | Virus tested | Results | Outcome | Suggested mechanism | Ref. | |
|---|---|---|---|---|---|---|---|
| Antibiotics | |||||||
| • Levofloxacin | Quinolone antibiotic | In vitro/human tracheal epithelial cells | RV14, RV2, RV15 | LVFX pretreatment reduced the RV14 and RV15 titer, the level of cytokines in the supernatant, the amount of RV14 RNA in the cells, and the cells’ susceptibility to RV14 infection, but did not reduce the titers of RV2. | Inhibits major-group rhinovirus infections by impeding the viral entrance and may also modulate airway inflammation in rhinoviral infections. | Reduces ICAM-1 expression levels, the receptor of RV, the number of acidic endosomes which allow RV RNA to enter the cytoplasm and inhibits the activation of NFκΒ proteins. | [123] |
| • Macrolides | Antibiotics | In vitro/in vivo/ randomized controlled trials | RV, RSV, Influenza | In vitro and in vivo studies show that macrolides reduce viral titers and infection-induced cytokines by respiratory viruses. Clinical trials have failed to prove a clinical benefit in healthy population, where for asthmatics individuals results are more promising. | Reduce inflammation induced by common cold but their clinical benefit is still uncertain. | Modulate the function of inflammatory cells , neutrophil migration, phagocytosis, the production of proinflammatory mediators, and eosinophilic inflammation. | [124, 125] |
| • Niclosamide | Anti-helmintic compound | In vitro/cell lines | RV1A, 2, 14, 16 CVB3, B4, A21, Influenza PR8, HSV | Niclosamide protects HeLa cells from HRV1A, 2, 14 and 16 infections and A549 cells from Influenza, while CVB3 and HSV1 infections were not affected. | Broad range antiviral against viruses which require a low-pH step for infectious entry, but not against pH-independent viruses. | Neutralizes pH and alters the distribution of endosomes, in a proton carrier mode-of-action independent of any protein target. | [126] |
| Leukotriene receptor antagonists (LTRAs) | Treatment of asthma and allergic rhinitis | Randomized controlled trials in healthy or asthmatic children and adults | Natural infection with common old virus [127, 128], Experimental inoculation of RV16 [129] | In experimental rhinovirus infection, LTRAs did not improve asthma control or cold symptoms in astmatic patients, but attenuated eosinophilic inflammation in the airway. In asthmatic children aged <4 years old, improved asthma control during viral exacerbation, in healthy children, though, prophylactic use of LTRAs did not reduce the incidence of common cold. | LTRAs may prevent virus-induced reductions in lung function in asthmatic, and this effect is not mediated by a reduction in viral replication. | Reduce inflammatory cell infiltration, bronchial smooth muscle contraction, mucus secretion and vascular permeability. | [127–129] |
| Anticholinergic | |||||||
| • Tiotropium | Anti-cholinergic agent | In vitro/human airway epithelial cells | RV14 | Tiotropium reduced RV14 titers, cytokine concentrations, and cells’ susceptibility to RV14 infection. | Inhibits RV infection by impeding the viral entrance and reducing inflammation. | Reducing the levels of ICAM-1 and acidic endosomes and inhibits the activation of NFκ B proteins. | [130] |
| Low-pH buffers | Chemical agents | In vitro/cell lines Clinical trial | RV1A, 2, 14, 16, 49, 85 PIV-3 Influenza A, RSV | Inhibits replication of HRV1A, 2, 14, 16, and 49 and Influenza A in cell lines but not RV 85, RSV or PIV-3. Intranasal administration reduced the viral shedding after experimental inoculation of RV16. | Nasal irrigation with low-pH buffers solution may improve clinical symptoms in patients with the common cold. | Low-pH alters capsid proteins leading to the loss of VP4. | [131] |
While most of the anti-viral therapies have significant results in vitro and sometimes in animal models, they have failed to show benefit in humans. Practical issues, as the need of administration at the onset of symptoms of common cold, are among the reasons currently impeding their use. To reduce the risk of resistance and enhance the effectiveness of agents, the combined use of antiviral agents with different mechanisms of action, may be a useful strategy [24•].
Conclusions
Molecular-based diagnosis as well as emerging therapeutic options targeting specific viruses or host immunity related mechanisms are promising regarding the development of effective treatment strategies for the common cold and its complications. In particular, groups such as asthmatics, immunocompromised individuals, young children and the elderly, remain vulnerable.
However, since the common cold is generally a mild and self-limiting condition, a potential therapy has to be safe and effective with practically no side effects. Moreover, respiratory viruses present with great plasticity, and their evading mechanisms may result in drug resistance. To further complicate this issue, research on viral molecular genetics highlights evidence of intimate relationship among some viruses and humans, e.g. influencing the selection pressure that contributes to the maintenance of major histocompatibility complex (MHC) diversity and the host immune response [132]. Possible consequences from the disturbance of this long standing and elegant balance by eliminating viral pathogens should be carefully considered.
Finally, practical and logistical issues such as time and cost related limitations of molecular diagnosis, timing of therapy administration and macroeconomics comparing health care costs, have to be taken into account when balancing the pros and cons of common cold treatments in the general population.
Compliance with Ethics Guidelines
Conflict of Interest
Nikolaos G. Papadopoulos has served as a consultant for AbbVie, Novartis, Menarini, and Meda; has received grant support from Merck, Sharp & Dohme, Nestle, and GlaxoSmithKline; has received payment for giving lectures/serving on speakers bureaus from Novartis, Uriach, GlaxoSmithKline, Allergopharma, and Stallergen; and has received payment for development of educational presentations from Uriach and Meda.
Maria Passioti, Paraskevi Maggina, and Spyridon Megremis declare that they have no conflict of interest.
Human and Animal Rights and Informed Consent
This article does not contain any studies with human or animal subjects performed by any of the authors.
Footnotes
This article is part of the Topical Collection on Rhinosinusitis
References
Papers of particular interest, published recently, have been highlighted as: • Of important •• Of major important
- 1.Fokkens WJ, et al. European position paper on rhinosinusitis and nasal polyps 2012. Rhinol Suppl. 2012;23:3. [PubMed] [Google Scholar]
- 2.Heikkinen T, Jarvinen A. The common cold. Lancet. 2003;361(9351):51–9. doi: 10.1016/S0140-6736(03)12162-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Roelen CA, Koopmans PC, Notenbomer A, Groothoff JW, et al. Job satisfaction and short sickness absence due to the common cold. Work. 2011;39(3):305–13. doi: 10.3233/WOR-2011-1178. [DOI] [PubMed] [Google Scholar]
- 4.Kenealy T, Arroll B. Antibiotics for the common cold and acute purulent rhinitis. Cochrane Database Syst Rev. 2013;6:CD000247. doi: 10.1002/14651858.CD000247.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Leder K, Sinclair MI, Mitakakis TZ, Hellard ME, Forbes A. A community-based study of respiratory episodes in Melbourne, Australia. Aust N Z J Public Health. 2003;27(4):399–404. doi: 10.1111/j.1467-842x.2003.tb00416.x. [DOI] [PubMed] [Google Scholar]
- 6.Gruber C, et al. History of respiratory infections in the first 12 yr among children from a birth cohort. Pediatr Allergy Immunol. 2008;19(6):505–12. doi: 10.1111/j.1399-3038.2007.00688.x. [DOI] [PubMed] [Google Scholar]
- 7.Ball TM, et al. Influence of attendance at day care on the common cold from birth through 13 years of age. Arch Pediatr Adolesc Med. 2002;156(2):121–6. doi: 10.1001/archpedi.156.2.121. [DOI] [PubMed] [Google Scholar]
- 8.Pitkaranta A, et al. Lowered yields of virus-induced interferon production in leukocyte cultures and risk of recurrent respiratory infections in children. J Clin Virol. 1999;14(3):199–205. doi: 10.1016/S1386-6532(99)00056-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Cohen S, et al. Positive emotional style predicts resistance to illness after experimental exposure to rhinovirus or influenza a virus. Psychosom Med. 2006;68(6):809–15. doi: 10.1097/01.psy.0000245867.92364.3c. [DOI] [PubMed] [Google Scholar]
- 10.Herlov-Nielsen H, Permin H. Common cold–risk factors, transmission and treatment. Ugeskr Laeger. 2001;163(41):5643–6. [PubMed] [Google Scholar]
- 11.Nieman DC. Exercise, upper respiratory tract infection, and the immune system. Med Sci Sports Exerc. 1994;26(2):128–39. doi: 10.1249/00005768-199402000-00002. [DOI] [PubMed] [Google Scholar]
- 12.Makela MJ, Puhakka T, Ruuskanen O, Leinonen M, Saikku P, Kimpimäki M, et al. Viruses and bacteria in the etiology of the common cold. J Clin Microbiol. 1998;36(2):539–42. doi: 10.1128/jcm.36.2.539-542.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Monto AS. Epidemiology of viral respiratory infections. Am J Med. 2002;112(Suppl 6A):4S–12S. doi: 10.1016/s0002-9343(01)01058-0. [DOI] [PubMed] [Google Scholar]
- 14.Grief SN. Upper respiratory infections. Prim Care. 2013;40(3):757–70. doi: 10.1016/j.pop.2013.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Jartti T, Jartti L, Ruuskanen O, Söderlund-Venermo M. New respiratory viral infections. Curr Opin Pulm Med. 2012;18(3):271–8. doi: 10.1097/MCP.0b013e328351f8d4. [DOI] [PubMed] [Google Scholar]
- 16.Weigl JA, et al. Ten years' experience with year-round active surveillance of up to 19 respiratory pathogens in children. Eur J Pediatr. 2007;166(9):957–66. doi: 10.1007/s00431-007-0496-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kahn J. Human bocavirus: clinical significance and implications. Curr Opin Pediatr. 2008;20(1):62–6. doi: 10.1097/MOP.0b013e3282f3f518. [DOI] [PubMed] [Google Scholar]
- 18.Gwaltney JM, Jr, et al. Rhinovirus infections in an industrial population. I. The occurrence of illness. N Engl J Med. 1966;275(23):1261–8. doi: 10.1056/NEJM196612082752301. [DOI] [PubMed] [Google Scholar]
- 19.Monto AS, Johnson KM. A community study of respiratory infections in the tropics. II. The spread of six rhinovirus isolates within the community. Am J Epidemiol. 1968;88(1):55–68. doi: 10.1093/oxfordjournals.aje.a120867. [DOI] [PubMed] [Google Scholar]
- 20.Briese T, et al. Global distribution of novel rhinovirus genotype. Emerg Infect Dis. 2008;14(6):944–7. doi: 10.3201/eid1406.080271. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Arakawa M, Okamoto-Nakagawa R, Toda S, Tsukagoshi H, Kobayashi M, Ryo A. Molecular epidemiological study of human rhinovirus species A, B and C from patients with acute respiratory illnesses in Japan. J Med Microbiol. 2012;61(3):410–9. doi: 10.1099/jmm.0.035006-0. [DOI] [PubMed] [Google Scholar]
- 22.Jefferson T, Del Mar C, Dooley L, Ferroni E, Al-Ansary LA, Bawazeer GA, et al. Physical interventions to interrupt or reduce the spread of respiratory viruses. Cochrane Database Syst Rev. 2011;7:CD006207. doi: 10.1002/14651858.CD006207.pub4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Gern JE, et al. Relationship of upper and lower airway cytokines to outcome of experimental rhinovirus infection. Am J Respir Crit Care Med. 2000;162(6):2226–31. doi: 10.1164/ajrccm.162.6.2003019. [DOI] [PubMed] [Google Scholar]
- 24.•.Jacobs SE, et al. Human rhinoviruses. Clin Microbiol Rev. 2013;26(1):135–62. doi: 10.1128/CMR.00077-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Winther B, et al. Light and scanning electron microscopy of nasal biopsy material from patients with naturally acquired common colds. Acta Otolaryngol. 1984;97(3–4):309–18. doi: 10.3109/00016488409130994. [DOI] [PubMed] [Google Scholar]
- 26.•.Triantafilou K, Vakakis E, Richer EA, Evans GL, Villiers JP, Triantafilou M. Human rhinovirus recognition in non-immune cells is mediated by Toll-like receptors and MDA-5, which trigger a synergetic pro-inflammatory immune response. Virulence. 2011;2(1):22–9. doi: 10.4161/viru.2.1.13807. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Papadopoulos NG, et al. Rhinoviruses infect the lower airways. J Infect Dis. 2000;181(6):1875–84. doi: 10.1086/315513. [DOI] [PubMed] [Google Scholar]
- 28.Lessler J, et al. Incubation periods of acute respiratory viral infections: a systematic review. Lancet Infect Dis. 2009;9(5):291–300. doi: 10.1016/S1473-3099(09)70069-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Harris JM, 2nd, Gwaltney JM., Jr Incubation periods of experimental rhinovirus infection and illness. Clin Infect Dis. 1996;23(6):1287–90. doi: 10.1093/clinids/23.6.1287. [DOI] [PubMed] [Google Scholar]
- 30.Gwaltney JM, Jr, Hendley JO, Patrie JT. Symptom severity patterns in experimental common colds and their usefulness in timing onset of illness in natural colds. Clin Infect Dis. 2003;36(6):714–23. doi: 10.1086/367844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Iwane MK, Prill MM, Lu X, Miller EK, Edwards KM, Hall CB. Human rhinovirus species associated with hospitalizations for acute respiratory illness in young US children. J Infect Dis. 2011;204(11):1702–10. doi: 10.1093/infdis/jir634. [DOI] [PubMed] [Google Scholar]
- 32.Singleton RJ, Bulkow LR, Miernyk K, DeByle C, Pruitt L, Hummel KB. Viral respiratory infections in hospitalized and community control children in Alaska. J Med Virol. 2010;82(7):1282–90. doi: 10.1002/jmv.21790. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Graat JM, Schouten EG, Heijnen ML, Kok FJ, Pallast EG, de Greeff SC, et al. A prospective, community-based study on virologic assessment among elderly people with and without symptoms of acute respiratory infection. J Clin Epidemiol. 2003;56(12):1218–23. doi: 10.1016/S0895-4356(03)00171-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Armengol CE, Hendley JO, Winther B. Occurrence of acute otitis media during colds in children younger than four years. Pediatr Infect Dis J. 2011;30(6):518–20. doi: 10.1097/INF.0b013e3182044930. [DOI] [PubMed] [Google Scholar]
- 35.Marom T, Nokso-Koivisto J, Chonmaitree T. Viral-bacterial interactions in acute otitis media. Curr Allergy Asthma Rep. 2000;12(6):551–8. doi: 10.1007/s11882-012-0303-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Esposito S, Marchisio P, Tenconi R, Tagliaferri L, Albertario G, Patria MF, et al. Diagnosis of acute rhinosinusitis. Pediatr Allergy Immunol. 2012;23(22):17–9. doi: 10.1111/j.1399-3038.2012.01319.x. [DOI] [PubMed] [Google Scholar]
- 37.Welge-Lussen A, Wolfensberger M. Olfactory disorders following upper respiratory tract infections. Adv Otorhinolaryngol. 2006;63:125–32. doi: 10.1159/000093758. [DOI] [PubMed] [Google Scholar]
- 38.Papadopoulos NG. Do rhinoviruses cause pneumonia in children? Paediatr Respir Rev. 2004;5(A):S191–S195. doi: 10.1016/s1526-0542(04)90036-x. [DOI] [PubMed] [Google Scholar]
- 39.Ruuskanen O, et al. Viral pneumonia. Lancet. 2011;377(9773):1264–75. doi: 10.1016/S0140-6736(10)61459-6. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Corne JM, et al. Frequency, severity, and duration of rhinovirus infections in asthmatic and non-asthmatic individuals: a longitudinal cohort study. Lancet. 2002;359(9309):831–4. doi: 10.1016/S0140-6736(02)07953-9. [DOI] [PubMed] [Google Scholar]
- 41.Papadopoulos NG, et al. Viruses and bacteria in acute asthma exacerbations–a GA(2) LEN-DARE systematic review. Allergy. 2011;66(4):458–68. doi: 10.1111/j.1398-9995.2010.02505.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Wark PA, et al. Asthmatic bronchial epithelial cells have a deficient innate immune response to infection with rhinovirus. J Exp Med. 2005;201(6):937–47. doi: 10.1084/jem.20041901. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Kherad O, Kaiser L, Bridevaux PO, Sarasin F, Thomas Y, Janssens JP, et al. Upper-respiratory viral infection, biomarkers, and COPD exacerbations. Chest. 2010;138(4):896–904. doi: 10.1378/chest.09-2225. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.de Almeida MB, Zerbinati RM, Tateno AF, Oliveira CM, Romão RM, Rodrigues JC, et al. Rhinovirus C and respiratory exacerbations in children with cystic fibrosis. Emerg Infect Dis. 2010;16(6):996–9. doi: 10.3201/eid1606.100063. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Crooks BN, Taylor CE, Turner AJ, Osman HK, Abinun M, Flood TJ, et al. Respiratory viral infections in primary immune deficiencies: significance and relevance to clinical outcome in a single BMT unit. Bone Marrow Transplant. 2000;26(10):1097–102. doi: 10.1038/sj.bmt.1702656. [DOI] [PubMed] [Google Scholar]
- 46.•.Kraft CS, Jacob JT, Sears MH, Burd EM, Caliendo AM, Marshall Lyon G. Severity of human rhinovirus infection in immunocompromised adults is similar to that of 2009 H1N1 influenza. J Clin Microbiol. 2012;50(3):1061–3. doi: 10.1128/JCM.06579-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Luinstra K, Petrich A, Castriciano S, Ackerman M, Chong S, Carruthers S, et al. Evaluation and clinical validation of an alcohol-based transport medium for preservation and inactivation of respiratory viruses. J Clin Microbiol. 2011;49(6):2138–42. doi: 10.1128/JCM.00327-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Landry ML, Cohen S, Ferguson D. Impact of sample type on rapid detection of influenza virus A by cytospin-enhanced immunofluorescence and membrane enzyme-linked immunosorbent assay. J Clin Microbiol. 2000;38(1):429–30. doi: 10.1128/jcm.38.1.429-430.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 49.Jartti T, Hedman K, Jartti L, Ruuskanen O, Allander T, Söderlund-Venermo M. Human bocavirus-the first 5 years. Rev Med Virol. 2012;22(1):46–64. doi: 10.1002/rmv.720. [DOI] [PubMed] [Google Scholar]
- 50.Mahony JB, Petrich A, Smieja M. Molecular diagnosis of respiratory virus infections. Crit Rev Clin Lab Sci. 2011;48(5–6):217–49. doi: 10.3109/10408363.2011.640976. [DOI] [PubMed] [Google Scholar]
- 51.Jartti T, Söderlund-Venermo M, Hedman K, Ruuskanen O, Mäkelä MJ. New molecular virus detection methods and their clinical value in lower respiratory tract infections in children. Paediatr Respir Rev. 2013;14(1):38–45. doi: 10.1016/j.prrv.2012.04.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Turner RB, Fuls JL, Rodgers ND, Goldfarb HB, Lockhart LK, Aust LB. A randomized trial of the efficacy of hand disinfection for prevention of rhinovirus infection. Clin Infect Dis. 2012;54(10):1422–6. doi: 10.1093/cid/cis201. [DOI] [PubMed] [Google Scholar]
- 53.•.Berthoud TK, Hamill M, Lillie PJ, Hwenda L, Collins KA, Ewer KJ, et al. Potent CD8+ T-cell immunogenicity in humans of a novel heterosubtypic influenza A vaccine, MVA-NP+M1. Clin Infect Dis. 2011;52(1):1–7. doi: 10.1093/cid/ciq015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Englund JA, Karron RA, Cunningham CK, Larussa P, Melvin A, Yogev R, et al. Safety and infectivity of two doses of live-attenuated recombinant cold-passaged human parainfluenza type 3 virus vaccine rHPIV3cp45 in HPIV3-seronegative young children. Vaccine. 2013;31(48):5706–12. doi: 10.1016/j.vaccine.2013.09.046. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 55.Simancas-Racines D, Guerra CV, Hidalgo R. Vaccines for the common cold. Cochrane Database Syst Rev. 2013;6:CD002190. doi: 10.1002/14651858.CD002190.pub4. [DOI] [PubMed] [Google Scholar]
- 56.Fu YH, He JS, Qiao W, Jiao YY, Hua Y, Zhang Y, et al. Intranasal immunization with a helper-dependent adenoviral vector expressing the codon-optimized fusion glycoprotein of human respiratory syncytial virus elicits protective immunity in BALB/c mice. Virol J. 2013;10:183. doi: 10.1186/1743-422X-10-183. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.•.Kamphuis T, Meijerhof T, Stegmann T, Lederhofer J, Wilschut J, de Haan A. Immunogenicity and protective capacity of a virosomal respiratory syncytial virus vaccine adjuvanted with monophosphoryl lipid A in mice. PLoS One. 2012;7(5):e36812. doi: 10.1371/journal.pone.0036812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.•.Rigter A, Widjaja I, Versantvoort H, Coenjaerts FEJ, van Roosmalen M, Leenhouts K, et al. A protective and safe intranasal RSV vaccine based on a recombinant prefusion-like form of the F protein bound to bacterium-like particles. PLoS One. 2013;8(8):e71072. doi: 10.1371/journal.pone.0071072. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 59.Palmenberg AC, et al. Sequencing and analyses of all known human rhinovirus genomes reveal structure and evolution. Science. 2009;324(5923):55–9. doi: 10.1126/science.1165557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 60.•.Bochkov YA, Palmenberg AC, Lee WM, Rathe JA, Amineva SP, Sun X, et al. Molecular modeling, organ culture and reverse genetics for a newly identified human rhinovirus C. Nat Med. 2011;17(5):627–32. doi: 10.1038/nm.2358. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.•.Megremis S, Demetriou P, Makrinioti H, Manoussaki AE, Papadopoulos NG. The genomic signature of human rhinoviruses A, B and C. PLoS One. 2012;7(9):e44557. doi: 10.1371/journal.pone.0044557. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Bartlett NW, Walton RP, Edwards MR, Aniscenko J, Caramori G, Zhu J, et al. Mouse models of rhinovirus-induced disease and exacerbation of allergic airway inflammation. Nat Med. 2008;14(2):199–204. doi: 10.1038/nm1713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.••.Niespodziana K, Napora K, Cabauatan C, Focke-Tejkl M, Keller W, Niederberger V, et al. Misdirected antibody responses against an N-terminal epitope on human rhinovirus VP1 as explanation for recurrent RV infections. FASEB J. 2012;26(3):1001–8. doi: 10.1096/fj.11-193557. [DOI] [PubMed] [Google Scholar]
- 64.Katpally U, Fu T-M, Freed DC, Casimiro DR, Smith TJ, et al. Antibodies to the buried N terminus of rhinovirus VP4 exhibit cross-serotypic neutralization. J Virol. 2009;83(14):7040–8. doi: 10.1128/JVI.00557-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.•.McLean GR, Walton RP, Shetty S, Peel TJ, Paktiawal N, Kebadze T, et al. Rhinovirus infections and immunisation induce cross-serotype reactive antibodies to VP1. Antiviral Res. 2012;95(3):193–201. doi: 10.1016/j.antiviral.2012.06.006. [DOI] [PubMed] [Google Scholar]
- 66.Andabaka T, Nickerson JW, Rojas-Reyes MX, Rueda JD, Bacic Vrca V, Barsic B. Monoclonal antibody for reducing the risk of respiratory syncytial virus infection in children. Cochrane Database Syst Rev. 2013;4:CD006602. doi: 10.1002/14651858.CD006602.pub4. [DOI] [PubMed] [Google Scholar]
- 67.Groothuis JR, Hoopes JM, Jessie VG. Prevention of serious respiratory syncytial virus-related illness. I: Disease pathogenesis and early attempts at prevention. Adv Ther. 2011;28(2):91–109. doi: 10.1007/s12325-010-0100-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Schepens B, Ibañez LI, De Baets S, Hultberg A, Bogaert P, De Bleser P. Nanobodies(R) specific for respiratory syncytial virus fusion protein protect against infection by inhibition of fusion. J Infect Dis. 2011;204(11):1692–701. doi: 10.1093/infdis/jir622. [DOI] [PubMed] [Google Scholar]
- 69.••.Clementi N, De Marco D, Mancini N, Solforosi L, Moreno GJ, Gubareva LV, et al. A human monoclonal antibody with neutralizing activity against highly divergent influenza subtypes. PLoS One. 2011;6(12):e28001. doi: 10.1371/journal.pone.0028001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.••.Corti D, Voss J, Gamblin SJ, Codoni G, Macagno A, Jarrossay D, et al. A neutralizing antibody selected from plasma cells that binds to group 1 and group 2 influenza A hemagglutinins. Science. 2011;333(6044):850–6. doi: 10.1126/science.1205669. [DOI] [PubMed] [Google Scholar]
- 71.Kim SY, Chang YJ, Cho HM, Hwang YW, Moon YS. Non-steroidal anti-inflammatory drugs for the common cold. Cochrane Database Syst Rev. 2013;6:CD006362. doi: 10.1002/14651858.CD006362.pub3. [DOI] [PubMed] [Google Scholar]
- 72.Li S, et al. Acetaminophen (paracetamol) for the common cold in adults. Cochrane Database Syst Rev. 2013;7:CD008800. doi: 10.1002/14651858.CD008800.pub2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Simons FE. H1-antihistamines in children. Clin Allergy Immunol. 2002;17:437–64. [PubMed] [Google Scholar]
- 74.De Sutter AI, van Driel ML, Kumar AA, Lesslar O, Skrt A. Oral antihistamine-decongestant-analgesic combinations for the common cold. Cochrane Database Syst Rev. 2012;2:CD004976. doi: 10.1002/14651858.CD004976.pub3. [DOI] [PubMed] [Google Scholar]
- 75.Meltzer EO, Caballero F, Scadding G. Treatment of congestion in upper respiratory diseases. Int J Gen Med. 2010;3:69–91. doi: 10.2147/ijgm.s8184. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 76.Fiebich BL, Collado JA, Stratz C, Valina C, Hochholzer W, Muñoz E, et al. Pseudoephedrine inhibits T-cell activation by targeting NF-kappaB, NFAT and AP-1 signaling pathways. Immunopharmacol Immunotoxicol. 2012;34(1):98–106. doi: 10.3109/08923973.2011.582118. [DOI] [PubMed] [Google Scholar]
- 77.Albalawi ZH, Othman SS, Alfaleh K. Intranasal ipratropium bromide for the common cold. Cochrane Database Syst Rev. 2013;6:CD008231. doi: 10.1002/14651858.CD008231.pub3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 78.Singh M. Heated, humidified air for the common cold. Cochrane Database Syst Rev. 2013;6:CD001728. doi: 10.1002/14651858.CD001728.pub5. [DOI] [PubMed] [Google Scholar]
- 79.Baartmans M, et al. Steam inhalation therapy: severe scalds as an adverse side effect. Br J Gen Pract. 2012;62(600):e473–7. doi: 10.3399/bjgp12X652337. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 80.Dicpinigaitis PV. Cough: an unmet clinical need. Br J Pharmacol. 2011;163(1):116–24. doi: 10.1111/j.1476-5381.2010.01198.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 81.Sen EF, Verhamme KM, Felisi M, 't Jong GW, Giaquinto C, Picelli G, et al. Effects of safety warnings on prescription rates of cough and cold medicines in children below 2 years of age. Br J Clin Pharmacol. 2011;71(6):943–50. doi: 10.1111/j.1365-2125.2010.03860.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Hayward G, Thompson MJ, Perera R, Del Mar CB, Glasziou PP, Heneghan CJ. Corticosteroids for the common cold. Cochrane Database Syst Rev. 2012;8:CD008116. doi: 10.1002/14651858.CD008116.pub2. [DOI] [PubMed] [Google Scholar]
- 83.Barrett B, et al. Echinacea for treating the common cold: a randomized trial. Ann Intern Med. 2010;153(12):769–77. doi: 10.7326/0003-4819-153-12-201012210-00003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Singh M, Das RR. Zinc for the common cold. Cochrane Database Syst Rev. 2013;6:CD001364. doi: 10.1002/14651858.CD001364.pub4. [DOI] [PubMed] [Google Scholar]
- 85.Mao S, Huang S. Vitamin D supplementation and risk of respiratory tract infections: a meta-analysis of randomized controlled trials. Scand J Infect Dis. 2013;45(9):696–702. doi: 10.3109/00365548.2013.803293. [DOI] [PubMed] [Google Scholar]
- 86.Jolliffe DA, Griffiths CJ, Martineau AR. Vitamin D in the prevention of acute respiratory infection: systematic review of clinical studies. J Steroid Biochem Mol Biol. 2013;136:321–9. doi: 10.1016/j.jsbmb.2012.11.017. [DOI] [PubMed] [Google Scholar]
- 87.Bergman P, Lindh AU, Björkhem-Bergman L, Lindh JD, et al. Vitamin D and respiratory tract infections: a systematic review and meta-analysis of randomized controlled trials. PLoS One. 2013;8(6):e65835. doi: 10.1371/journal.pone.0065835. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.•.Science M, Maguire JL, Russell ML, Smieja M, Walter SD, Loeb M. Low serum 25-hydroxyvitamin D level and risk of upper respiratory tract infection in children and adolescents. Clin Infect Dis. 2013;57(3):392–7. doi: 10.1093/cid/cit289. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Murdoch DR, Slow S, Chambers ST, Jennings LC, Stewart AW, Priest PC, et al. Effect of vitamin D3 supplementation on upper respiratory tract infections in healthy adults: the VIDARIS randomized controlled trial. JAMA. 2012;308(13):1333–9. doi: 10.1001/jama.2012.12505. [DOI] [PubMed] [Google Scholar]
- 90.Arroll B. Common cold. Am Fam Physician. 2011;84(12):1390–1. [PubMed] [Google Scholar]
- 91.Maggini S, Beveridge S, Suter M. A combination of high-dose vitamin C plus zinc for the common cold. J Int Med Res. 2012;40(1):28–42. doi: 10.1177/147323001204000104. [DOI] [PubMed] [Google Scholar]
- 92.Hemila H, Chalker E. Vitamin C for preventing and treating the common cold. Cochrane Database Syst Rev. 2013;1:CD000980. doi: 10.1002/14651858.CD000980.pub4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Linde K, Barrett B, Wölkart K, Bauer R, Melchart D. Echinacea for preventing and treating the common cold. Cochrane Database Syst Rev. 2006;1:CD000530. doi: 10.1002/14651858.CD000530.pub2. [DOI] [PubMed] [Google Scholar]
- 94.Nahas R, Balla A. Complementary and alternative medicine for prevention and treatment of the common cold. Can Fam Physician. 2011;57(1):31–6. [PMC free article] [PubMed] [Google Scholar]
- 95.Jawad M, et al. Safety and efficacy profile of echinacea purpurea to prevent common cold episodes: a randomized, double-blind, Placebo-Controlled Trial. Evid Based Complement Alternat Med. 2012;2012:841315. doi: 10.1155/2012/841315. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Kang EJ, Kim SY, Hwang IH, Ji YJ. The effect of probiotics on prevention of common cold: a meta-analysis of randomized controlled trial studies. Korean J Fam Med. 2013;34(1):2–10. doi: 10.4082/kjfm.2013.34.1.2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 97.Hao Q, Lu Z, Dong BR, Huang CQ, Wu T. Probiotics for preventing acute upper respiratory tract infections. Cochrane Database Syst Rev. 2011;9:CD006895. doi: 10.1002/14651858.CD006895.pub2. [DOI] [PubMed] [Google Scholar]
- 98.Rerksuppaphol S, Rerksuppaphol L. Randomized controlled trial of probiotics to reduce common cold in schoolchildren. Pediatr Int. 2012;54(5):682–7. doi: 10.1111/j.1442-200X.2012.03647.x. [DOI] [PubMed] [Google Scholar]
- 99.Shinkai S, Toba M, Saito T, Sato I, Tsubouchi M, Taira K, et al. Immunoprotective effects of oral intake of heat-killed Lactobacillus pentosus strain b240 in elderly adults: a randomised, double-blind, placebo-controlled trial. Br J Nutr. 2013;109(10):1856–65. doi: 10.1017/S0007114512003753. [DOI] [PubMed] [Google Scholar]
- 100.Marotta F, et al. Is there a potential application of a fermented nutraceutical in acute respiratory illnesses? An in-vivo placebo-controlled, cross-over clinical study in different age groups of healthy subjects. J Biol Regul Homeost Agents. 2012;26(2):285–94. [PubMed] [Google Scholar]
- 101.•.Ganesan S, Faris AN, Wang Q, Nanua S, Hershenson MB, et al. Quercetin inhibits rhinovirus replication in vitro and in vivo. Antiviral Res. 2012;94(3):258–71. doi: 10.1016/j.antiviral.2012.03.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Song JH, Park KS, Kwon DH, Choi HJ. Anti-human rhinovirus 2 activity and mode of action of quercetin-7-glucoside from Lagerstroemia speciosa. J Med Food. 2013;16(4):274–9. doi: 10.1089/jmf.2012.2290. [DOI] [PubMed] [Google Scholar]
- 103.Heinz SA, Henson DA, Austin MD, Jin F, Nieman DC. Quercetin supplementation and upper respiratory tract infection: A randomized community clinical trial. Pharmacol Res. 2010;62(3):237–42. doi: 10.1016/j.phrs.2010.05.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.•.Riede L, Grube B, Gruenwald J. Larch arabinogalactan effects on reducing incidence of upper respiratory infections. Curr Med Res Opin. 2013;29(3):251–8. doi: 10.1185/03007995.2013.765837. [DOI] [PubMed] [Google Scholar]
- 105.Leibbrandt A, Meier C, König-Schuster M, Weinmüllner R, Kalthoff D, Pflugfelder B, et al. Iota-carrageenan is a potent inhibitor of influenza A virus infection. PLoS One. 2010;5(12):e14320. doi: 10.1371/journal.pone.0014320. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Grassauer A, Meier C, Pretsch A, Prieschl-Grassauer E, Unger H. Iota-carrageenan is a potent inhibitor of rhinovirus infection. Virol J. 2008;5:107. doi: 10.1186/1743-422X-5-107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Eccles R, Meier C, Prieschl-Grassauer E. Efficacy and safety of an antiviral iota-carrageenan nasal spray: a randomized, double-blind, placebo-controlled exploratory study in volunteers with early symptoms of the common cold. Respir Res. 2010;11:108. doi: 10.1186/1465-9921-11-108. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Fazekas T, Eickhoff P, Pruckner N, Vollnhofer G, Fischmeister G, Diakos C, et al. Lessons learned from a double-blind randomised placebo-controlled study with a iota-carrageenan nasal spray as medical device in children with acute symptoms of common cold. BMC Complement Altern Med. 2012;12:147. doi: 10.1186/1472-6882-12-147. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Auinger A, Riede L, Bothe G, Busch R, Gruenwald J. Yeast (1,3)-(1,6)-beta-glucan helps to maintain the body's defence against pathogens: a double-blind, randomized, placebo-controlled, multicentric study in healthy subjects. Eur J Nutr. 2013;52(8):1913–8. doi: 10.1007/s00394-013-0492-z. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Graubaum H-J, Busch R, Stier H, Gruenwald J. A doubleblind, randomized, placebo-controlled nutritional study using an insoluble yeast beta-glucan to improve the immune defense system. Food Nut Sci. 2012;3:738–746. [Google Scholar]
- 111.Yates PJ, et al. Virus susceptibility analyses from a phase IV clinical trial of inhaled zanamivir treatment in children infected with influenza. Antimicrob Agents Chemother. 2013;57(4):1677–84. doi: 10.1128/AAC.02145-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Michiels B, et al. The value of neuraminidase inhibitors for the prevention and treatment of seasonal influenza: a systematic review of systematic reviews. PLoS One. 2013;8(4):e60348. doi: 10.1371/journal.pone.0060348. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 113.Wang K, et al. Neuraminidase inhibitors for preventing and treating influenza in children (published trials only) Cochrane Database Syst Rev. 2012;4:CD002744. doi: 10.1002/14651858.CD002744.pub4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Tregoning JS, Schwarze J. Respiratory viral infections in infants: causes, clinical symptoms, virology, and immunology. Clin Microbiol Rev. 2010;23(1):74–98. doi: 10.1128/CMR.00032-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.•.Olszewska W, Ispas G, Schnoeller C, Sawant D, Van de Casteele T, Nauwelaers D, et al. Antiviral and lung protective activity of a novel respiratory syncytial virus fusion inhibitor in a mouse model. Eur Respir J. 2011;38(2):401–8. doi: 10.1183/09031936.00005610. [DOI] [PubMed] [Google Scholar]
- 116.Sun Z, et al. Respiratory syncytial virus entry inhibitors targeting the F protein. Virus. 2013;5(1):211–25. doi: 10.3390/v5010211. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Maugeri C, et al. New anti-viral drugs for the treatment of the common cold. Bioorg Med Chem. 2008;16(6):3091–107. doi: 10.1016/j.bmc.2007.12.030. [DOI] [PubMed] [Google Scholar]
- 118.Patick AK. Rhinovirus chemotherapy. Antiviral Res. 2006;71(2–3):391–6. doi: 10.1016/j.antiviral.2006.03.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 119.•.Rollinger JM, Schmidtke M. The human rhinovirus: human-pathological impact, mechanisms of antirhinoviral agents, and strategies for their discovery. Med Res Rev. 2011;31(1):42–92. doi: 10.1002/med.20176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 120.Gwaltney JM, Jr, et al. Combined antiviral-antimediator treatment for the common cold. J Infect Dis. 2002;186(2):147–54. doi: 10.1086/341455. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 121.Gao L, et al. A randomized controlled trial of low-dose recombinant human interferons alpha-2b nasal spray to prevent acute viral respiratory infections in military recruits. Vaccine. 2010;28(28):4445–51. doi: 10.1016/j.vaccine.2010.03.062. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 122.••.Campbell-Harding G, Sawkins H, Bedke N, Holgate ST, Davies DE, Andrews AL, et al. The innate antiviral response upregulates IL-13 receptor alpha2 in bronchial fibroblasts. J Allergy Clin Immunol. 2013;131(3):849–55. doi: 10.1016/j.jaci.2012.08.030. [DOI] [PubMed] [Google Scholar]
- 123.Yamaya M, et al. Levofloxacin inhibits rhinovirus infection in primary cultures of human tracheal epithelial cells. Antimicrob Agents Chemother. 2012;56(8):4052–61. doi: 10.1128/AAC.00259-12. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 124.Koutsoubari I, et al. Effect of clarithromycin on acute asthma exacerbations in children: an open randomized study. Pediatr Allergy Immunol. 2012;23(4):385–90. doi: 10.1111/j.1399-3038.2012.01280.x. [DOI] [PubMed] [Google Scholar]
- 125.Min JY, Jang YJ. Macrolide therapy in respiratory viral infections. Mediators Inflamm. 2012;2012:649570. doi: 10.1155/2012/649570. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 126.Jurgeit A, et al. Niclosamide is a proton carrier and targets acidic endosomes with broad antiviral effects. PLoS Pathog. 2012;8(10):e1002976. doi: 10.1371/journal.ppat.1002976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Yoshihara S, et al. Inhibition of common cold-induced aggravation of childhood asthma by leukotriene receptor antagonists. Allergol Int. 2012;61(3):405–10. doi: 10.2332/allergolint.11-OA-0400. [DOI] [PubMed] [Google Scholar]
- 128.Kozer E, et al. RCT of montelukast as prophylaxis for upper respiratory tract infections in children. Pediatrics. 2012;129(2):e285–90. doi: 10.1542/peds.2011-0358. [DOI] [PubMed] [Google Scholar]
- 129.Kloepfer KM. Effects of montelukast on patients with asthma after experimental inoculation with human rhinovirus 16. Ann Allergy Asthma Immunol. 2011;106(3):252–7. doi: 10.1016/j.anai.2010.11.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 130.Yamaya M, et al. Inhibitory effects of tiotropium on rhinovirus infection in human airway epithelial cells. Eur Respir J. 2012;40(1):122–32. doi: 10.1183/09031936.00065111. [DOI] [PubMed] [Google Scholar]
- 131.Gern JE, et al. Inhibition of rhinovirus replication in vitro and in vivo by acid-buffered saline. J Infect Dis. 2007;195(8):1137–43. doi: 10.1086/512858. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 132.Van Blerkom LM. Role of viruses in human evolution. Am J Phys Anthropol. 2003;37:14–46. doi: 10.1002/ajpa.10384. [DOI] [PMC free article] [PubMed] [Google Scholar]


