Abstract
The sudden death of three calves, one diarrheic calf, and one aborted fetus from four farms in southern Brazil was investigated. Two Histophilus somni-associated syndromes were identified: systemic histophilosis (n = 4) and abortion (n = 1). The principal pathological findings included vasculitis, meningoencephalitis with thrombosis, necrotizing myocarditis, renal infarctions, hepatic abscesses, and bronchopneumonia. PCR assays were used to amplify specific amplicons of the ovine herpesvirus 2, bovine herpesvirus 1 and −5, Listeria monocytogenes, H. somni, and pestivirus; bovine group A rotavirus (BoRV-A) and bovine coronavirus (BCoV) were investigated in calves with diarrhea. H. somni DNA was amplified in tissues from all calves and the brain of the aborted fetus with pathological alterations consistent with histophilosis. All other PCR assays were negative; BoRV-A and BCoV were not identified. These findings confirm the participation of H. somni in the pathological alterations observed in this study and represent the first description of histophilosis in cattle from Brazil.
Keywords: Bovine disease, Histophilosis, Necrotizing myocarditis, Thrombotic meningoencephalitis, Molecular diagnostics
Introduction
Histophilus somni (formerly Haemophilus somnus) is a gram-negative bacterium that is associated with a disease complex (histophilosis) that can produce clinical syndromes such as thrombotic meningoencephalitis (TME), pleuritis, polysynovitis, suppurative bronchopneumonia, septicemia, myocarditis, otitis media, infertility, abortion, and mastitis in affected animals (Harris and Janzen 1989; Divers 2008; Pérez et al. 2010). Additionally, H. somni is one of the bacterial pathogens associated with the clinical entity known as bovine respiratory disease of feedlot cattle (Divers 2008) and is a normal habitat of the reproductive and respiratory mucus membranes of asymptomatic cattle (Harris and Janzen 1989; Corbeil 2007; Woolums et al. 2009). Furthermore, based on sequence analyses of the 16S rRNA and rpoB genes, it was proposed that the organisms referred to as Haemophilus somni, Haemophilus agni, and Histophilus ovis are in fact H. somni and should represent a novel genus within the Pasteurellaceae family (Angen et al. 2003). However, differences between ovine and bovine strains of H. somni have been identified (Tanaka et al. 2005) .
Although H. somni-related diseases are of economic importance to livestock owners (George 2009), clinical manifestations of disease in cattle is predominant within the USA (Fecteau and George 2004; Woolums et al. 2009) and Canada (Van Donkersgoed et al. 1990; Gagea et al. 2006), with reports of sporadic outbreaks occurring in Europe (Tegtmeier et al. 1999; Janosi et al. 2009b), Argentina (Descarga et al. 2002), Nigeria (Odugbo et al. 2009), Japan (Momotani et al. 1985; Tanaka et al. 2005), and South Africa (Last et al. 2001); there is no description of this pathogen affecting cattle in Brazil. Nevertheless, H. somni has also been isolated from healthy goats in Mexico (Perez-Romero et al. 2011) and Hungary (Janosi et al. 2009a) and has been associated TME in sheep within Europe (Cassidy et al. 1997), reproductive disease in sheep from Canada (Lees et al. 1990), and in one case of endometritis in a sheep from Brazil (Rizzo et al. 2012). Furthermore, H. somni-induced infections are more predominant in young cattle (Harris and Janzen 1989; Van Donkersgoed et al. 1990) and in feedlot relative to dairy and pastured animals (Fecteau and George 2004), occurring within the first 2 weeks or months of the feeding period (Woolums et al. 2009).
Despite the various syndromes associated with H. somni in cattle (Harris and Janzen 1989; Pérez et al. 2010), the frequency of manifestations due to septicemic disease, such as myocarditis and arthritis, and bronchopneumonia might be increasing relative to TME (Harris and Janzen 1989; Van Donkersgoed et al. 1990). Consequently, due to the large number of organs/tissue that can be affected, manifestations of H. somni-induced infections in cattle are directly related to the organ and/or system infected (Harris and Janzen 1989; Woolums et al. 2009). Nevertheless, vasculitis and thrombosis due to endothelial cell damage are the predominant pathological features of H. somni-induced infections (Harris and Janzen 1989; Divers 2008). This manuscript describes the pathological and molecular findings associated with H. somni-induced infections in cattle from southern Brazil.
Materials and methods
Farms affected, clinical manifestations, and necropsy
Four cattle farms (three beef and one dairy) within the state of Paraná, southern Brazil (Table 1), had unexplained episodes of calf fatality and abortion. The calves were of different breeds, with ages varying from 1 day to 2 months, were maintained on three of these farms and died within 3 to 4 days after the initial manifestation of disease. The clinical signs observed were not common within the farms and varied between sudden death (farms A and B), reproductive problems (farm C), and gastrointestinal manifestations (farm D). Animals at farms A and B demonstrated predominantly neurological manifestations. Reproductive complaints included infertility, 20 abortions, and a reduced conception rate (35 %). Most calves at farm D had a clinical diagnosis of coccidiosis and suffered from intermittent diarrhea during the last few months. All cattle were reared on pasture; water and commercial mineral salts were administered ad libitum.
Table 1.
Principal manifestations observed in farms with H. somni-induced infections in the state of Paraná, southern Brazil
| Geographical location | Breed | Age range (days) | Principal clinical manifestations | Calves affected | ||
|---|---|---|---|---|---|---|
| Fatality | Total | Necropsied | ||||
| A: Arapongas | Nelore | 2–10 | Lateral recumbency | 8 | 40 | 2 |
| Muscular weakness | ||||||
| Sudden death within 2–3 days | ||||||
| B: Congoinhas | Nelore | 25–30 | Abnormal gait | 4 | 174 | 1 |
| Motor incoordination | ||||||
| Sudden death within 2 days | ||||||
| C: Guaraci | Girolando | 8-month-old fetus | Abortion | 20a | 20 | 1 |
| Infertility | ||||||
| D: Guarapuava | Shorthorn × Hereford | 50–60 | Coccidiosis | 11 | 110 | 1 |
| Unresolved diarrhea | ||||||
| Poor feed conversion ratio | ||||||
| Death within 5–7 days | ||||||
aNumber of abortions informed by the consulting veterinarian
Four calves, that died spontaneously, and one aborted fetus were submitted for routine necropsy that was done soon after death. The first case occurred in late May (animal 1) and the others during late October to early November, 2012. Selected tissue fragments (brain, liver, lungs, kidneys, spleen, intestine, myocardium, and lymph nodes) from each animal necropsied were fixed by immersion in 10 % buffered formalin solution and routinely processed for histopathological evaluation. Duplicate sections of the organs mentioned above were collected freshly during necropsy and maintained at −20 °C until processed for molecular diagnostics. Additionally, fecal samples from calves (n = 10) with history of diarrhea from farm D were evaluated for the presence of rotavirus and coronavirus.
DNA extraction, PCR assays, and sequencing
DNA and/or RNA extracted from freshly collected tissue fragments from the calves and the aborted fetus (Boom et al. 1990), associated with proteinase K (Ambion, Grand Island, NY, USA), and a combination of the phenol/chloroform/isoamyl alcohol and silica/guanidine isothiocyanate method (Alfieri et al. 2006), was used in PCR assays designed to amplify specific amplicons of principal infectious disease agents of cattle. These PCR protocols targeted the ovine herpesvirus type 2 (OvHV-2) tegument protein gene (Baxter et al. 1993), the glycoprotein C gene of bovine herpesvirus-1 (BoHV-1) and −5 (Claus et al. 2005), the listeriolysin (hlyA) gene of Listeria monocytogenes (Wesley et al. 2002), the 16S rRNA gene of H. somni (Angen et al. 1998), and the 5′-UTR region of pestivirus (Vilček et al. 1994). Additionally, the feces from the calf with clinical diagnosis of enteritis were evaluated for the presence of the nucleoprotein gene of bovine coronavirus BCoV (Takiuchi et al. 2006) and G (VP7) and P (VP4) genes of bovine group A rotavirus, BoRV-A (Gouvea et al. 1990; Gentsch et al. 1992).
Positive controls consisted of DNA/RNA from previous cases: OvHV-2 (Headley et al. 2012b), L. monocytogenes (Headley et al. 2012a), cell culture-adapted Los Angeles and AA Par strains of BoHV-1 and −5, respectively (Claus et al. 2005), BVDV (NADL strain)-infected Madin Darby bovine kidney cells, and BCoV (Takiuchi et al. 2006). No positive control was included in the H. somni PCR assays. Nuclease-free water (Invitrogen Corp. Carlsbad, CA, USA) was used as negative controls in all PCR assays. All PCR products were separated by electrophoresis in 2 % agarose gels, stained with ethidium bromide, and examined under ultraviolet light.
The amplified PCR products were then purified (illustra GFX PCR DNA and Gel Band Purification Kit, GE Healthcare, Little Chalfont, Buckinghamshire, UK) and submitted for direct sequencing using the forward and reverse primers. The partial nucleotide sequences were initially compared by the BLAST (http://www.ncbi.nlm.nih.gov/BLAST) program with similar selected sequences deposited in GenBank. Phylogenetic tree and sequence alignments based on the 16S rRNA gene of the Pasteurellaceae family were then created by using MEGA 5.10 (Tamura et al. 2011), constructed by the neighbor-joining method, based on 1,000 bootstrapped data sets; distance values were calculated by using the Kimura 2 parameter model. Escherichia coli was used as the out-group to provide stability to the generated tree.
Results
Pathological findings and syndromes associated with H. somni
The principal pathological lesions and syndromes observed in these animals are resumed in Table 2. Although there were three distant clinical manifestations at the farms investigated, only two major pathological syndromes associated with H. somni were identified: systemic histophilosis in the calves (n = 4) from farms A, B, and D, and abortion from farm C.
Table 2.
Signalment with principal pathological findings and syndromes in cattle infected with H. somni
| Farms | Animals | Gross alterations | Histopathology | Pathological syndrome |
|---|---|---|---|---|
| A | 1: 1-day-old Nelore calf | Bloody fluid within the intestine | Hemorrhagic ruminitis | Systemic histophilosis |
| Cerebrum; shallow sulci with distended gyri | Cerebellum; meningoencephalitis with vasculitis, thrombosis and infarctions | |||
| Congestion of cerebral cortex | Lymphadenitis with vasculitis, congestion, and edema | |||
| Corneal opacity | Necrohemorrhagic enteritis | |||
| Hemorrhage of meningeal vessels | Pulmonary edema and hemorrhage | |||
| Hemorrhagic rhinotracheitis | Renal hemorrhages | |||
| Lymphadenopathy | Vasculitis (myocardial, renal, and pulmonary) | |||
| Pulmonary hemorrhage | ||||
| Ruminal and abomasal hemorrhages | ||||
| 2: 7-day-old Nelore calf | Abomasal ulcerations | Abomasal ulcerations | Systemic histophilosis | |
| Arthritis | Cerebrum and cerebellum; vasculitis | |||
| Blood-filled intestine | Centrilobular hepatocellular necrosis | |||
| Cardiac valvular thrombosis | Hemorrhagic enteritis | |||
| Foamy secretion within trachea | Hepatitis with thrombosis and vasculitis | |||
| Lymphadenopathy | Lymphadenitis with vasculitis, congestion, and edema | |||
| Pale areas at the myocardium | Necrotizing myocarditis with vasculitis and dystrophic mineralization | |||
| Wet and heavy lungs | Pulmonary edema | |||
| B | 3: 20-day-old Nelore calf | Cerebrum; shallow sulci with distended gyri | Hepatic abscesses and thrombosis | Systemic histophilosis |
| Congestion of meningeal vessels | Kidney, vasculitis and thrombosis | |||
| Pale hepatic foci (0.2–0.5 cm diameter) | Meningoencephalitis with vasculitis, thrombosis and infarctions | |||
| Pulmonary hemorrhage | Myocarditis with vasculitis | |||
| Congestion of meningeal vessels | Pleuropneumonia | |||
| C | 4: 8-month-old Girolando fetus | Abomasal hemorrhages | Pulmonary hemorrhage with vasculitis and thrombosis | Abortion |
| Brain; shallow sulci with distended gyri | Meningoencephalitis with vasculitis and thrombosis | |||
| D | 5: 60-day-old Shorthorn calf | Congestion of cerebral cortex | Vasculitis (myocardial and renal) | Systemic histophilosis |
| Hemorrhage of meningeal vessels | Bronchopneumonia | |||
| Cranioventral consolidation of lung | Hemorrhagic enteritis | |||
| Pale areas at the wall of the left myocardial ventricle | Hepatitis with vasculitis and thrombosis and sinusoidal congestion | |||
| Epicardial and endocardial petechiae | Lymphadenitis with vasculitis, congestion, and edema | |||
| Fetid diarrhea with | Meningoencephalitis with vasculitis and thrombosis | |||
| Lymphadenopathy | Multifocal coagulative cortical renal necrosis and vasculitis | |||
| Several wedge-shaped pale areas at the cortex of the kidney (Renal infarctions) | Necrotizing myocarditis with vasculitis | |||
| Thymus hemorrhages |
Systemic histophilosis was characterized by lesions to the central nervous system, liver, myocardium, lungs, joints, hematopoietic, and gastrointestinal tract. The gross alterations observed in most calves were suggestive of widespread vascular damage, with congestion of meningeal vessels and cerebral edema being the predominant (75 %, 3/4) alterations in the animals necropsied. One calf (animal no. 1) demonstrated marked congestion of the cerebral cortex (Fig. 1a); fresh impression smears were made at the sectioned cerebral surface to detect the possible presence of Babesia bovis. Hemorrhage of meningeal vessels (Fig. 1b) was more pronounced in animal no. 5 relative to the other calves. Frequent non-CNS alterations included blood-filled intestinal content (Fig. 1c) and circulatory pulmonary lesions, occurring in most (75 %, 3/4) of these animals. Two calves with bloody intestines (animal nos. 2 and 5) also demonstrated pale, irregularly shaped areas that varied from 2 × 5 mm to 0.5 × 1.2 cm and which contrasted the normal color of the left myocardial ventricle (Fig. 1d). One of these (animal no. 2) also had an enlarged left hock joint that contained a cloudy synovial fluid and a large thrombus attached to the left atrioventricular valve. Other significant gross findings included hemorrhagic rhinotracheitis, abomasal ulcerations (Fig. 1e), and multiple, pale, wedge-shaped areas at the renal cortex (Fig. 1f). Congestion of meningeal vessels was the most significant gross lesions observed in the aborted fetus.
Fig. 1.
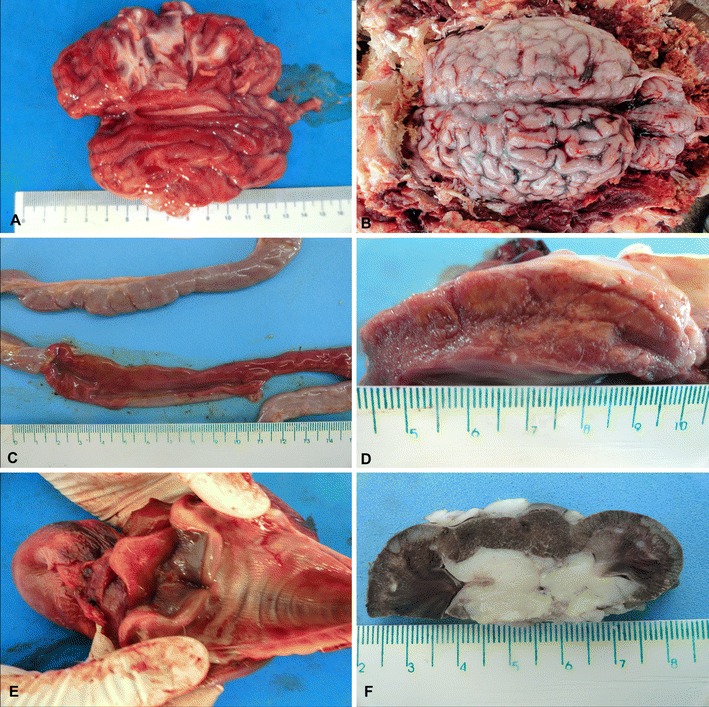
Gross demonstration of H. somni-induced manifestations in cattle. Observe the severely congested gray matter of the cerebral cortex (a), and the hemorrhage of meningeal vessels (b). The small intestine is hemorrhagic (c), and there are several, different-sized, pale areas at the left myocardial ventricle (d), and hemorrhage of the trachea and epiglottis (e). There are several infarctions at the renal cortex (f). Scale in centimeters
All calves demonstrated vasculitis with thrombosis (Fig. 2a). Meningoencephalitis with vasculitis, thrombosis, hemorrhage, and infarctions (Fig. 2b–c) occurred in most calves (75 %, 3/4) evaluated, except the calf with valvular myocardial thrombosis. The irregularly shaped, pale areas observed at the left ventricular myocardium of animal nos. 2 and 5 grossly demonstrated severe, multifocal, necrotizing myocarditis (Fig. 2d) with vasculitis and dystrophic mineralization associated with intralesional bacteria. Necrotizing myocarditis with vasculitis was also observed in animal no. 3. The pale areas at the renal cortex of animal no. 5, revealed severe, multifocal, coagulative tubular necrosis with vasculitis (Fig. 2e); multifocal hemorrhagic nephritis with vasculitis was predominant at the medullary region of the kidney. The pale hepatic foci observed in animal no. 3 were, in fact, hepatic abscesses (Fig. 2f). Three calves (75 %) demonstrated enteritis, which contained either a hemorrhagic or necrohemorrhagic inflammatory exudate. Other significant histopathological findings included centrilobular hepatocellular necrosis, ulcerative abomasitis, and bronchopneumonia. Impression smears made from the sectioned cerebral surface of animal no. 1 did not reveal the presence of hemoparasites, and these were not observed by histopathology. The principal lesions within the aborted fetus were characterized by meningoencephalitis with vasculitis and thrombosis.
Fig. 2.
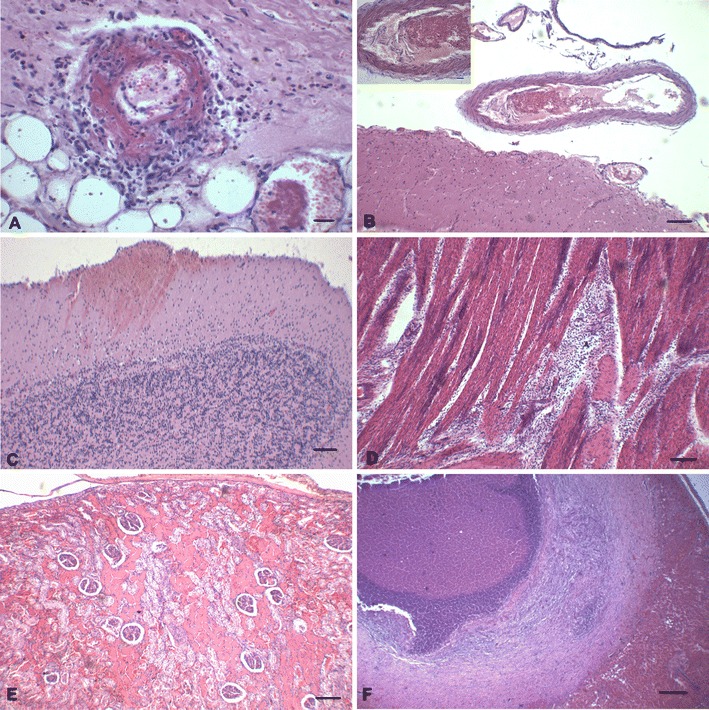
Histopathological findings associated with H. somni-induced infections in cattle. There is vasculitis with thrombosis of the artery at the renal pelvis (a). Observe vasculitis with thrombosis at the meninges of the cerebrum (b), with the enlarged vessel (insert). Observe hemorrhage at the molecular layer of the cerebellum (c) and necrotizing myocarditis (d). There is multifocal to coalescing coagulative necrosis of renal tubules (e) and a hepatic abscess (f). Hematoxylin and eosin stain; Bar: a and insert at b, 20 μm; b–e, 100 μm; f, 300 μm
Molecular characterization of H. somni
The H. somni 16S rRNA PCR assays successfully amplified the desired amplicon in tissue fragments of all animals; these results are summarized in Table 3. However, all other PCR assays revealed negative results. Additionally, H. somni DNA was amplified within multiple tissues of animal no. 3 and from fragments of the left ventricular myocardium of animal 2 that demonstrated necrotizing myocarditis. Partial sequences of the 16S rRNA gene of H. somni were obtained from two animals (nos. 1 and 3). BLAST analyses of these sequences (GenBank accession numbers KC414854 and KC414855) demonstrated 99 % similarity with other sequences deposited in GenBank. Phylogenetic analyses revealed that these sequences clustered with other bovine sequences of H. somni derived from different geographical regions, being more closely related to cases from Canada and Australia (Fig. 3). However, ovine-derived H. somni sequences clustered apart from their bovine counterparts, but all H. somni sequences (bovine and ovine) formed a cluster that was distinct from other members of the Pasteurellaceae family. The nucleotide sequences used for phylogenetic analyses during this study are given in Fig. 3.
Table 3.
Distribution of H. somni DNA in calves from southern Brazil
| Farm | Animal | Organs evaluated | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Brain | Lung | Liver | Kidney | Myocardium | Nasal mucosa | Intestine | Spleen | Lymph node | ||
| A | 1 | − | − | − | − | − | + | − | − | − |
| 2 | − | − | − | − | + | NC | ND | − | − | |
| B | 3 | + | + | + | + | − | NC | ND | − | + |
| C | 4 | + | − | − | − | − | NC | ND | − | − |
| D | 5 | − | − | − | − | − | NC | − | − | + |
+ positive, − negative, ND not determined, NC not collected
Fig. 3.
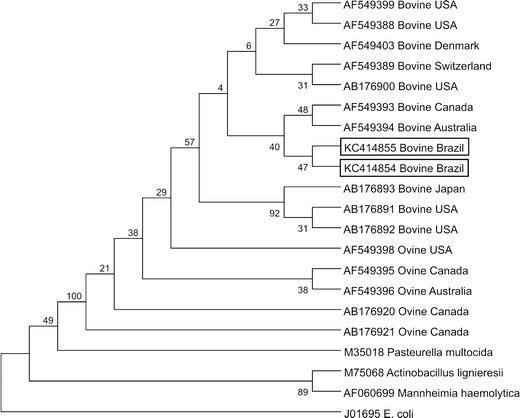
Phylogenetic tree based on the 16S rRNA gene sequences of H. somni and selected Pasteurellaceae members generated by MEGA 5.10. The tree was constructed by the neighbor-joining method, based on 1,000 bootstrapped data sets; distance values were calculated by using the Kimura 2 parameter model. The GenBank accession numbers, animal species, and country of origin of the sequences are given. The sequences derived from this study are highlighted within the boxes
After a diagnosis of H. somni-induced infection was confirmed at farm D, all remaining diarrheic Shorthorn calves were medicated with oxytetracycline. The consulting veterinarian has informed that the episodes of diarrhea have ceased, and these calves have since recuperated and are gaining body weight. Feces from the calf necropsied from this farm were negative for BCoV and BoRV-A.
Additionally, blood samples collected in EDTA tubes from calves (n = 10) at farm D that were in contact with the necropsied animal, and from cows (n = 11) with history of abortion from farm C, after the diagnosis was made, were negative for H. somni. Furthermore, fecal samples collected from calves at farm D were negative for BCoV and BoRV-A.
Discussion
The results of this study have revealed that the necropsied calves and aborted fetus, from different farms within southern Brazil, demonstrated pathological syndromes that are consistent with those described in cattle infected with H. somni (Harris and Janzen 1989; Divers 2008; George 2009). Additional confirmation of the participation of H. somni in the etiopathogenesis of these manifestations was obtained by the successful amplification of the 16S rRNA gene of this bacterium from tissue fragments of all animals evaluated; similar results have been described (Angen et al. 1998, 2003). These findings represent the first characterization of H. somni-induced infections in cattle from Brazil and extend the geographical distribution of this important pathogen of cattle, since most descriptions of H. somni in livestock have originated from North America (Harris and Janzen 1989; Fecteau and George 2004; Pérez et al. 2010).
All calves herein described demonstrated pathological manifestations consistent with systemic histophilosis, with meningoencephalitis with vasculitis and thrombosis occurring in most animals, except in one calf with severe necrotizing myocardial histophilosis. Although TME is a common neurological manifestation of histophilosis (Fecteau and George 2004; George 2009), studies have suggested that systemic histophilosis is more frequently diagnosed relative to TME (Harris and Janzen 1989; Van Donkersgoed et al. 1990). Furthermore, most of these calves had pulmonary disease, which might be the source of dissemination in these cases (Harris and Janzen 1989; Fecteau and George 2004; Pérez et al. 2010). The hepatic abscess observed in animal no. 3 and multiple renal infarctions of animal no. 5 were previously described in systemic histophilosis (Pérez et al. 2010). The wedge-shaped patterns observed grossly associated with the histopathological findings are characteristic of infarction due to obstruction of the interlobular artery. The finding of thrombi within several organs of these animals is a frequent manifestation of systemic histophilosis (Momotani et al. 1985). Although the pathogenesis of H. somni-induced infections is not well elucidated and is very complex (Corbeil 2007), the disseminated vasculitis observed during this study has been associated with several virulence factors such as lipooligosaccharide, immunoglobulin-binding proteins, major outer membrane proteins, histamine production, and exopolysaccharide (Corbeil 2007; Pérez et al. 2010). However, apoptotic cell death of endothelial cells caused by this bacterium might be the key to explain thrombosis in histophilosis (Corbeil 2007; Woolums et al. 2009; Pérez et al. 2010).
The hemorrhagic enteritis observed in calves in this study could have been associated with endotoxemia induced by H. somni (Woolums et al. 2009), but the participation of this pathogen in gastrointestinal disease has never been demonstrated, and H. somni DNA was not amplified in the intestine of any affected calf. Alternatively, the principal pathogens associated with diarrhea in calves are BCoV, BoRV-A, BVDV, Clostridium perfringens-induced enterotoxemia, enterotoxigenic E. coli, Cryptosporidium spp., and Salmonella spp. (Gunn et al. 2009). However, intralesional protozoa were not observed in the necropsied calf from the farm with intermittent diarrhea or in the other calves with hemorrhagic enteritis, while BCoV, BVDV, and BoRV-A were not identified in these animals. The age of the calves (5–60 days) on the farm with diarrhea is not consistent with that associated of infections by E. coli (Gunn et al. 2009), while the histopathological manifestations characteristic of salmonellosis were not observed. However, the pathological enteric findings of these calves are similar with those associated with C. perfringens type A enterotoxemia (Gunn et al. 2009). Consequently, these diarrheic calves might have been simultaneously infected with H. somni and C. perfringens.
During this study, one calf demonstrated marked congestion of the cerebral cortex and was initially diagnosed as cerebral babesiosis during necropsy (Rodrigues et al. 2005). Although congenital babesiosis has been described in calves that have not received colostrum (Bracarense et al. 2001), rapid staining of the affected cerebral cortex did not reveal hemoparasites, and characteristic histopathological alterations associated with B. bovis were not observed. Additionally, the pathogenesis of bovine babesiosis is incompatible with the age (1 day) of this calf because of the following: (1) clinical manifestations of bovine babesiosis occurs 2 to 3 weeks after tick infestation; (2) the incubation period varies from 5 days to around 3 weeks; and (3) calves in endemic areas, as was this animal, have a natural immunity to babesiosis, which is reinforced by colostral antibodies (Zaugg 2009). Consequently, cerebral babesiosis was excluded as part of the etiopathogenesis associated with this manifestation, but it is proposed that cerebral histophilosis be included as differential diagnosis in cases of cerebral cortical congestion in cattle, particularly in young animals.
Two calves demonstrated pale areas at the myocardium which revealed necrotizing myocarditis by histopathology, and H. somni DNA was amplified from one of these. Similar findings have been described in a feedlot calf from Wyoming, USA, where myocardial disease has been associated with 42 % of mortality (O’Toole et al. 2009), and in Alberta, Canada, where H. somni-induced myocarditis is the principal cause of myocardial disease in feedlot cattle, resulting in mortality of 76 % (70/92) in one study (Haines et al. 2004). Myocardial histophilosis was also one of the main causes of calf mortality in feedlot cattle in another Canadian study (Van Donkersgoed et al. 1990). Nevertheless, bovine myocardial histophilosis is considered rare and sporadic in the USA compared to Canada (O'Toole et al. 2009). Additionally, in this study, myocardial histophilosis was observed at the left ventricular myocardium in both calves; this also occurred in the case from Wyoming (O'Toole et al. 2009) and seems to be a frequent anatomic location for bovine myocardial histophilosis (Harris and Janzen 1989; O'Toole et al. 2009).
The 16S rRNA PCR assay amplified the desired amplicon (Angen et al. 1998, 2003) in all animals with histopathological alterations consistent with those induced by H. somni (Harris and Janzen 1989; Fecteau and George 2004; Divers 2008). Furthermore, confirmation of the participation of H. somni in these lesions was achieved by the direct sequencing of these amplicons, while the absence of a H. somni-positive control excluded possible contamination. A positive H. somni control was not included due to the availability of a reliable control DNA, considering that there is only one case description of this disease within Brazil. The sequences from this study clustered with other bovine sequences from other geographical locations, while those derived from sheep formed a somewhat separate cluster. These findings are in accordance with the theory that ovine and bovine H. somni sequences clustered separately and can be differentiated by analyses of the H. somni rpoB gene (Tanaka et al. 2005). Furthermore, all H. somni sequences formed a cluster that was distinct from other members of the Pasteurellaceae family, confirming the genus differentiation proposed (Angen et al. 2003) .
During this study, the PCR assays of common pathogens associated with sudden death in cattle were negative, while those in calves with diarrhea were negative for BCoV and BoRV-A. Additionally, all diarrheic calves treated with oxytetracycline recovered completely; this is important in associating the participation of H. somni-induced disease in the diarrheic animals since oxytetracycline is the drug of choice for H. somni-induced infections (Harris and Janzen 1989; Fecteau and George 2004; George 2009; Woolums et al. 2009), and standing animals medicated during the progression of histophilosis are known to recover (Harris and Janzen 1989; Woolums et al. 2009). Although other common pathogens were not identified in these animals, the possible participation of a non-evaluated viral or bacterial agent cannot be completely excluded as stressor associated with the development of systemic histophilosis. Since only one aborted fetus from farm C and one calf was affected at farm D, whole blood was collected from other calves of similar age range and from the cow of the aborted fetus and cows with history of abortion from farm C, in an attempt to identify additional bacteremic calves at the first location and the source of infection at the latter. However, all of these samples from farms C and D were negative for H. somni; this suggests that these animals were not bacteremic, but the PCR-negative cows could have been asymptomatic carriers (Pérez et al. 2010) and the source of infection at the farm with a history of abortion.
The finding of pathological manifestations consistent with histophilosis within the brain and myocardium, associated with the amplification of H. somni DNA from the brain of the fetus, confirms the participation of this pathogen as a cause of abortion in this case. H. somni-associated abortion in cattle was not previously described in livestock from Brazil or South America. Infectious agents frequently associated with abortions in cattle from South America include Neospora caninum, Brucela abortus, Leptospira spp., Campylobacter spp., BoHV-1, and BVDV (Alfieri et al. 1998). During this study the participation of BoHV-1 and BVDV were excluded due to the negative PCR results, while the other infectious agents were ruled out due to the absence of associated histopathological findings.
Most of these cases occurred during the 2012 Brazilian spring, which was very atypical with sudden variations in temperature and relative humidity; adverse climatic conditions are common stressors associated with the development of bovine (Harris and Janzen 1989; Divers 2008; George 2009; Pérez et al. 2010) and ovine (Cassidy et al. 1997) histophilosis and could have participated in the development of systemic histophilosis at these farms. The animals of most farms are reared exclusively on pastures; H. somni is more frequently diagnosed in feedlot cattle (Fecteau and George 2004). Nevertheless, since these infections occurred at farms from distinct areas and that there was no movement of cattle between the affected farms, the authors believe that this bacterium was always present within the Brazilian cattle herds but was never diagnosed or the possibility of H. somni-induced infections, investigated. This might be because the diverse clinical manifestations associated with H. somni, as observed in these cases, can be easily confused with other diseases (such as bovine viral diarrhea; infectious bovine rhinotracheitis; rotavirus and coronavirus enteritis; herpesvirus-induced meningoencephalitis; and malignant catarrhal fever) that are more frequently associated with cattle mortality in Brazil. Consequently, it is recommended that H. somni be included in the list of differentials to investigate livestock mortality in Brazil and South America, since this pathogen was previously diagnosed in a sheep with endometritis from Brazil (Rizzo et al. 2012) and sporadic outbreaks have occurred in Argentina (Descarga et al. 2002).
In conclusion, H. somni DNA was identified in tissue samples of calves from different farms with unresolved causes of mortality in southern Brazil. These calves demonstrated pathological lesions consistent with systemic histophilosis, while H. somni DNA was amplified within the brain of an aborted fetus with meningoencephalitis. The results of this study represent the first characterization of this important bovine disease in cattle from Brazil.
Acknowledgments
The authors thank Drs. Celso Koetz Júnior, Gilmar Sartori Júnior, Antônio Francisco Chaves Neto, and Simone de Fátima Ferreira for submitting these cases for necropsy. Drs. SA Headley, AF Alfieri, and AA Alfieri are recipients of the National Council for Scientific and Technological Development (CNPq; Brazil) fellowships. This study was partially funded by the Office of the Pro-Rector for Research and Graduate Studies, Universidade Norte do Paraná, Londrina, Brazil.
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
References
- Alfieri AA, Alfieri AF, Medici KC. The effects of Bovine herpesvirus type 1 on reproductive system of cattle. Semina Ciêncas Agrárias. 1998;19:86–93. [Google Scholar]
- Alfieri AA, Parazzi ME, Takiuchi E, Medici KC, Alfieri AF. Frequency of group A rotavirus in diarrhoeic calves in Brazilian cattle herds, 1998–2002. Tropical Animal Health and Production. 2006;38:521–526. doi: 10.1007/s11250-006-4349-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Angen O, Ahrens P, Kuhnert P, Christensen H, Mutters R. Proposal of Histophilus somni gen. nov., sp. nov. for the three species incertae sedis 'Haemophilus somnus', 'Haemophilus agni' and 'Histophilus ovis'. International Journal of Systematic and Evolutionary Microbiology. 2003;53:1449–1456. doi: 10.1099/ijs.0.02637-0. [DOI] [PubMed] [Google Scholar]
- Angen O, Ahrens P, Tegtmeier C. Development of a PCR test for identification of Haemophilus somnus in pure and mixed cultures. Veterinary Microbiology. 1998;63:39–48. doi: 10.1016/S0378-1135(98)00222-3. [DOI] [PubMed] [Google Scholar]
- Baxter SI, Pow I, Bridgen A, Reid HW. PCR detection of the sheep-associated agent of malignant catarrhal fever. Archives of Virology. 1993;132:145–159. doi: 10.1007/BF01309849. [DOI] [PubMed] [Google Scholar]
- Boom R, Sol CJ, Salimans MM, Jansen CL, Wertheim-van Dillen PM, van der Noordaa J. Rapid and simple method for purification of nucleic acids. Journal of Clinical Microbiology. 1990;28:495–503. doi: 10.1128/jcm.28.3.495-503.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bracarense APFL, Vidotto O, Cruz GD. Prenatal Babesia bovis infection. Arquivo Brasileiro de Medicina Veterinária e Zootecnia. 2001;53:1–3. doi: 10.1590/S0102-09352001000400017. [DOI] [Google Scholar]
- Cassidy JP, McDowell SW, Reilly GA, McConnell WJ, Forster F, Lawler D. Thrombotic meningoencephalitis associated with Histophilus ovis infection in lambs in Europe. The Veterinary Record. 1997;140:193–195. doi: 10.1136/vr.140.8.193. [DOI] [PubMed] [Google Scholar]
- Claus MP, Alfieri AF, Folgueras-Flatschart AV, Wosiacki SR, Medici KC, Alfieri AA. Rapid detection and differentiation of bovine herpesvirus 1 and 5 glycoprotein C gene in clinical specimens by multiplex-PCR. Journal of Virological Methods. 2005;128:183–188. doi: 10.1016/j.jviromet.2005.05.001. [DOI] [PubMed] [Google Scholar]
- Corbeil LB. Histophilus somni host-parasite relationships. Animal Health Research Reviews. 2007;8:151–160. doi: 10.1017/S1466252307001417. [DOI] [PubMed] [Google Scholar]
- Descarga CO, Piscitelli HG, Zielinski GC, Cipolla AL. Thromboembolic meningoencephalitis due to Haemophilus somnus in feedlot cattle in Argentina. The Veterinary Record. 2002;150:817. doi: 10.1136/vr.150.26.817. [DOI] [PubMed] [Google Scholar]
- Divers TJ. Respiratory diseases. St. Louis, Missouri: Rebhun's diseases of diary cattle. Saunders/Elsevier; 2008. pp. 79–96. [Google Scholar]
- Fecteau G, George LW. Bacterial meningitis and encephalitis in ruminants. The Veterinary clinics of North America. Food Animal Practice. 2004;20:363–377. doi: 10.1016/j.cvfa.2004.03.002. [DOI] [PubMed] [Google Scholar]
- Gagea MI, Bateman KG, van Dreumel T, McEwen BJ, Carman S, Archambault M, Shanahan RA, Caswell JL. Diseases and pathogens associated with mortality in Ontario beef feedlots. Journal of Veterinary Diagnostic Investigation. 2006;18:18–28. doi: 10.1177/104063870601800104. [DOI] [PubMed] [Google Scholar]
- Gentsch JR, Glass RI, Woods P, Gouvea V, Gorziglia M, Flores J, Das BK, Bhan MK. Identification of group A rotavirus gene 4 types by polymerase chain reaction. Journal of Clinical Microbiology. 1992;30:1365–1373. doi: 10.1128/jcm.30.6.1365-1373.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- George LW. Thrombotic meningioencephalitis (Histophilus somni [Haemophilus somuns] infection; sleeper calves) In: Smith BP, editor. Large Animal Internal Medicine. 4. Missouri: Mosby/Elsevier, St. Louis; 2009. pp. 1048–1050. [Google Scholar]
- Gouvea V, Glass RI, Woods P, Taniguchi K, Clark HF, Forrester B, Fang ZY. Polymerase chain reaction amplification and typing of rotavirus nucleic acid from stool specimens. Journal of Clinical Microbiology. 1990;28:276–282. doi: 10.1128/jcm.28.2.276-282.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gunn AA, Naylor JA, House JK. Diarrhea. In: Smith BP, editor. Large Animal Internal Medicine. 4. Missouri: Mosby/Elsevier, St. Louis; 2009. pp. 340–362. [Google Scholar]
- Rizzo H, Gregory L, Carvalho AF, Pinheiro ES. Histophilus somni isolation on sheep with endometritis first case report on Brazil. Revista Brasileira de Reprodução Animal. 2012;36:136–138. [Google Scholar]
- Haines DM, Moline KM, Sargent RA, Campbell JR, Myers DJ, Doig PA. Immunohistochemical study of Hemophilus somnus, Mycoplasma bovis, Mannheimia hemolytica, and bovine viral diarrhea virus in death losses due to myocarditis in feedlot cattle. The Canadian Veterinary Journal. 2004;45:231–234. [PMC free article] [PubMed] [Google Scholar]
- Harris FW, Janzen ED. The Haemophilus somnus disease complex (Hemophilosis): A review. The Canadian Veterinary Journal. 1989;30:816–822. [PMC free article] [PubMed] [Google Scholar]
- Headley, S.A., Bodnar, L., Fritzen, J.T.T., Rodrigues, W.B., Bronkhorst, D.E., Alfieri, A.F., Okano, W., Alfieri, A.A., 2012a. Clinical, histopathological, and molecular characterization of encephalitic listeriosis in small ruminants from northern Paraná, Brazil. Brazilian Journal of Microbiology, Accepted for publication. [DOI] [PMC free article] [PubMed]
- Headley, S.A., Sousa, I.K.F., Minervino, A.H.H., Barros, I.O., Júnior, R.A.B., Alfieri, A.F., Ortolani, E.L., Alfieri, A.A., 2012b. Molecular confirmation of ovine herpesvirus 2-induced malignant catarrhal fever lesions in cattle from Rio Grande do Norte, Brazil. Pesquisa Veterinária Brasileira 32, 1213–1218.
- Janosi K, Hajtos I, Makrai L, Gyuranecz M, Varga J, Fodor L. First isolation of Histophilus somni from goats. Veterinary Microbiology. 2009;133:383–386. doi: 10.1016/j.vetmic.2008.07.010. [DOI] [PubMed] [Google Scholar]
- Janosi K, Stipkovits L, Glavits R, Molnar T, Makrai L, Gyuranecz M, Varga J, Fodor L. Aerosol infection of calves with Histophilus somni. Acta Vet Hung. 2009;57:347–356. doi: 10.1556/AVet.57.2009.3.1. [DOI] [PubMed] [Google Scholar]
- Last R.D., Macfarlane M.D., Jarvis C.J. Isolation of Haemophilus somnus from dairy cattle in kwaZulu-Natal. An emerging cause of 'dirty cow syndrome' and infertility? Journal of the South African Veterinary Association. 2001;72:95. doi: 10.4102/jsava.v72i2.625. [DOI] [PubMed] [Google Scholar]
- Lees VW, Meek AH, Rosendal S. Epidemiology of Haemophilus somnus in young rams. Canadian Journal of Veterinary Research. 1990;54:331–336. [PMC free article] [PubMed] [Google Scholar]
- Momotani E, Yabuki Y, Miho H, Ishikawa Y, Yoshino T. Histopathological evaluation of disseminated intravascular coagulation in Haemophilus somnus infection in cattle. Journal of Comparative Ppathology. 1985;95:15–23. doi: 10.1016/0021-9975(85)90073-8. [DOI] [PubMed] [Google Scholar]
- O'Toole D, Allen T, Hunter R, Corbeil LB. Diagnostic exercise: Myocarditis due to Histophilus somni in feedlot and backgrounded cattle. Veterinary Pathology. 2009;46:1015–1017. doi: 10.1354/vp.08-VP-0332-O-DEX. [DOI] [PubMed] [Google Scholar]
- Odugbo MO, Ogunjumo SO, Chukwukere SC, Kumbish PR, Musa A, Ekundayo SO, Okewole PA, Nwankpa ND, Itodo AE, Haruna G. The first report of Histophilus somni pneumonia in Nigerian dairy cattle. Veterinary Journal. 2009;181:340–342. doi: 10.1016/j.tvjl.2008.03.007. [DOI] [PubMed] [Google Scholar]
- Perez-Romero N, Aguilar-Romero F, Arellano-Reynoso B, Diaz-Aparicio E, Hernandez-Castro R. Isolation of Histophilus somni from the nasal exudates of a clinically healthy adult goat. Tropical Animal Health and Production. 2011;43:901–903. doi: 10.1007/s11250-011-9792-6. [DOI] [PubMed] [Google Scholar]
- Pérez DS, Pérez FA, Bretschneider G. Histophilus somni: pathogenecity in cattle an update. Anales de Veterinaria de Murcia. 2010;26:5–21. [Google Scholar]
- Rodrigues A, Rech RR, Barros RR, Fighera RA, Barros CSL. Cerebral babesiosis in cattle: 20 cases. Ciência Rural. 2005;35:121–125. doi: 10.1590/S0103-84782005000100019. [DOI] [Google Scholar]
- Takiuchi E, Stipp DT, Alfieri AF, Alfieri AA. Improved detection of bovine coronavirus N gene in faeces of calves infected naturally by a semi-nested PCR assay and an internal control. Journal of Virological Methods. 2006;131:148–154. doi: 10.1016/j.jviromet.2005.08.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S. MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony methods. Molecular Biology and Evolution. 2011;28:2731–2739. doi: 10.1093/molbev/msr121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanaka A, Hoshinoo K, Hoshino T, Tagawa Y. Differentiation between bovine and ovine strains of Histophilus somni based on the sequences of 16S rDNA and rpoB gene. The Journal of Veterinary Medical Science. 2005;67:255–262. doi: 10.1292/jvms.67.255. [DOI] [PubMed] [Google Scholar]
- Tegtmeier C, Uttenthal A, Friis NF, Jensen NE, Jensen HE. Pathological and microbiological studies on pneumonic lungs from Danish calves. Journal of Veterinary Medicine. Series B. 1999;46:693–700. doi: 10.1046/j.1439-0450.1999.00301.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Donkersgoed J, Janzen ED, Harland RJ. Epidemiological features of calf mortality due to hemophilosis in a large feedlot. The Canadian Veterinary Journal. 1990;31:821–825. [PMC free article] [PubMed] [Google Scholar]
- Vilček Š, Herring AJ, Herring JA, Nettleton PF, Lowings JP, Paton DJ. Pestiviruses isolated from pigs, cattle and sheep can be allocated into at least three genogroups using polymerase chain reaction and restriction endonuclease analysis. Archives of Virology. 1994;136:309–323. doi: 10.1007/BF01321060. [DOI] [PubMed] [Google Scholar]
- Wesley IV, Larson DJ, Harmon KM, Luchansky JB, Schwartz AR. A case report of sporadic ovine listerial menigoencephalitis in Iowa with an overview of livestock and human cases. Journal of Veterinary Diagnostic Investigation. 2002;14:314–321. doi: 10.1177/104063870201400407. [DOI] [PubMed] [Google Scholar]
- Woolums AR, Ames TR, Baker JC. The broncopneumonias (respiratory disease complex of cattle, sheep, and goats) In: Smith BP, editor. Large animal internal medicine. 4. Missouri: Mosby/Elsevier, St. Louis; 2009. pp. 602–643. [Google Scholar]
- Zaugg JL. Babesiosis. In: Smith BP, editor. Large Animal Internal Medicine. 4. Missouri: Mosby/Elsevier, St. Louis; 2009. pp. 1157–1160. [Google Scholar]


