Abstract
We compared the outcomes in patients receiving unrelated peripheral blood stem cell transplants (PBSCT) with those receiving bone marrow transplants (BMT) in a matched pair analysis. Seventy-four patients with hematological malignancies with HLA-matched (77%) and mismatched (23%) donors were analyzed in this study. Thirty-four patients (45%) were considered as high risk patients. Sixty-eight patients received standard conditioning regimens with Bu/Cy or TBI/Cy. Six patients received an intensified conditioning regimen with the addition of etoposide, thiotepa or melphalan. GVHD prophylaxis consisted of prednisolone, cyclosporine and methotrexate. Groups were matched for patient, donor, transplant characteristics and HLA compatibility. Peripheral blood stem cell collection led to the collection of a higher number of CD34+ and CD3+ cells in comparison to bone marrow collection. Leukocyte engraftment in the PBSCT group occurred in 14 days (median; range 6–26 days) and in the BMT group in 19 days (range 9–29 days; P < 0.02). The time of platelet engraftment did not differ significantly. The incidence of grades II–lV acute GVHD in the group of HLA-identical patients was 35% in the PBSCT group and 25% in the BMT group (P < 0.33, log-rank). However, there was a significant difference (P < 0.05, log-rank) in incidence and time to onset of acute GVHD II–IV comparing all patients, including the 17 mismatched transplants. Disease-free survival was 51% (19 patients) with a median of 352 days and 59% (21 patients) with a median of 760 days for PBSC and BMT transplants, respectively. In conclusion, our results indicate that allogeneic PBSCT led to significantly faster leukocyte engraftment but is associated with a higher incidence and more rapid onset of severe acute GVHD comparing all patients, including the 17 mismatched transplants. However, the incidence of severe acute GVHD in HLA-identical patients was not different between the PBSCT and BMT groups. Bone Marrow Transplantation (2001) 27, 27–33.
Keywords: GVHD, PBSCT, BMT, HLA compatibility
Main
Since 1989, peripheral blood stem cells (PBSC), after stimulation with granulocyte colony-stimulating factor (G-CSF), have been increasingly used as an alternative to hematopoietic stem cells from bone marrow in related donor transplants.1 In the following years allogeneic PBSCT began to displace BMT, and in 1996 the EBMT published data on more than 1000 transplants.2 The potential advantages of PBSC for allogeneic transplants are several. Firstly, rapid engraftment could be demonstrated in patients with PBSC transplants in comparison to those with BMT.3,4 This might reasonably be expected to reduce infection and bleeding risk. Thus, published reports have indicated that PBSCT is associated with a reduced need for antibiotics, shortened hospital stays and lower 200-day mortality.5 Moreover, reports have also suggested that immunological recovery is more rapid following allogeneic PBSC transplants.6 Typical PBSC products contain three to four times more CD34+ cells and up to 20 times higher T cell levels than do typical marrow products.7 Therefore, the main concern with allogeneic PBSC transplantation is graft-versus-host disease (GVHD). Reports to date, however, have not identified an increased incidence or severity of acute GVHD following PBSCT.8 On the other hand, increased incidence and severity of chronic GVHD have been reported by some groups.9,10 The results of the first randomized trial comparing allogeneic PBSCT and BMT presented at the ASH meeting in 1999 confirmed no difference in incidence of aGVHD and a tendency to higher incidence of chronic GVHD in patients transplanted with mobilized peripheral stem cells.11 It should be noted that this trial showed a higher mortality rate in patients transplanted with bone marrow stem cells and a significantly higher overall survival for PBSCT patients. This important message should be confirmed in other randomized trials. For instance, other authors12 presented the results of a randomized trial comparing allogeneic BMT and PBSCT from related donors. The incidence of GVHD was equivalent in the two groups (48% vs 57%), the incidence of extensive chronic GVHD was significantly higher in the PBSCT group (77%) than in the BMT group (27%) and no difference of overall survival could be demonstrated (at 1 year 67% and 59%, respectively). The higher number of T cells may also induce a more potent GVL effect. PBSCT may be advantageous for allogeneic donors.13,14 Only 3–7% of donors experienced complications15 from stimulation by G-CSF but no general anesthesia was necessary. The procedure is carried out in an outpatient setting. Nevertheless, one concern has been the ethics of using unrelated donors to mobilize PBSC, although the side-effects after administration of G-CSF to normal donors are tolerable after several years of follow-up. Thus, in the last 2 years only a few patients who received PBSCT from unrelated donors have been reported. We started unrelated allogeneic PBSC transplants in 1995 and 37 patients have been transplanted in our department. We report our experience with allogeneic PBSCT in comparison to BMT in a matched pair analysis. In addition, we analyze the incidence of acute GVHD in all patients as well as HLA-identical transplant patients.
Patients and methods
Patients
Patients with a diagnosis indicating the need for a transplant were included in this study after approved informed consent was obtained from the donor and recipient. Patient and donor characteristics are given in Table 1.
Table 1.
Patient and donor characteristics

Informed consent
Patients and donors were asked to participate in a study evaluating transplantation with PBSC as an alternative to BM. All patients and donors gave informed consent approved by our institutional revue board (IRB) and also approved by the local ethical committee (Ethik Kommission, Landesärztekammer Rheinland-Pfalz).
Bone marrow control group
All 37 patients transplanted in our institution between January 1995 and May 1999 with PBSCT from unrelated donors were included in this study. Each patient who received an unrelated PBSCT was matched with a patient who received BMT from an unrelated donor. From the total of 170 unrelated marrow transplants performed in our department from May 1994 to June 1999, we selected 37 matched patients for this study. The following prognostic factors were matched: diagnosis, disease stage, age, HLA compatibility and GVHD prophylaxis.
Donors
Twenty-seven donors of PBSC were HLA-identical to the patients, while two had a class-I locus mismatch. One of these was a locus-A mismatch and the other a locus B-mismatch. Eight donors had one DR-locus mismatch, respectively. Thirty donors of bone marrow were HLA-identical to the patients, two had a B-locus mismatch and five a DR-locus mismatch. HLA typing was done serologically for class I and using polymerase chain reaction-single strand polymorphism (PCR-SSP) for class II. All PBSC donors received G-CSF (Rhone-Poulenc Rorer, Lyon, France or Amgen, Thousand Oaks, CA, USA). The G-CSF dose ranged from 7 to 12 μg/kg and was administered subcutaneously twice daily. G-CSF was administered for 4 days in five donors, for 5 days in 27 donors and for 6 days in five donors. One leukapheresis was performed in 15 donors and two in 22 donors. The donors experienced skeletal pain in most cases, which was relieved by paracetamol. Calcium was administered prophylactically during the leukapheresis to reduce signs of hypocalcemia. In 80% of donors the leukapheresis was performed in an outpatient setting.
Treatment regimens
The conditioning regimen included busulfan (16 mg/kg) orally administered over 4 days and cyclophosphamide (60 mg/kg/day i.v.) (Bu/Cy) administered for 2 consecutive days for standard risk patients with myeloid diseases (31 patients). ALL and NHL patients (36 patients) received 2.25 Gy TBI twice daily for 3 consecutive days (13.5 Gy) and cyclophosphamide (60 mg/kg/day i.v.) for 2 consecutive days; six patients with refractory leukemias received an intensified conditioning regimen with etoposide (60 mg/kg/day i.v.) in addition to Bu/Cy once (two patients) or thiothepa (10 mg/kg i.v.) once (two patients) or melphalan (140 mg/m2 i.v.) once (one patient). The GVHD prophylaxis regimen used was prednisolone from day 7 to day 14 at 0.5 mg/kg/day i.v., followed by 1 mg/kg/day from day 15 and then tapered; cyclosporine (10 mg/kg/day) from day −3 and methotrexate (15 mg/m2 i.v.) were given on day 1 followed by 10 mg/m2 i.v. on days 3 and 6. Eight patients transplanted from HLA non-identical donors additionally received mycophenolate mofetil (2 × 1 g/day) orally as GVHD prophylaxis.16 Three of them were transplanted with BM and five with PBSC. CD34+ cell selection for T cell depletion using immunomagnetic beads (Isolex 300 SA; Baxter, Deerfield, IL, USA) was performed in one PBSC patient in January 1995. All patients required prophylaxis for infectious diseases. Patient management was performed according to the standard procedures in our center and was the same in the two different groups. It should be noted that post-transplantation G-CSF was not given in the BMT group of patients. The prophylaxis was the same in the two groups. Antibacterial medication contained a combination of trimethoprim with a sulfanilamide and a non-reabsorptive antibiotic. Antifungal prophylaxis included itraconazol and amphotericin B orally, while the antiviral prophylaxis contained acyclovir up to day 14.
Evaluation and definition
CD34+, CD3+ and CD56+ cells were determined in BM or PBSC grafts by flow cytometry, as previously described.17 Engraftment was assessed according to published criteria.7 Briefly, myeloid engraftment was defined as the first of 3 consecutive days when the absolute neutrophil count was higher than 0.5 × 109/l. Platelet engraftment was defined as the first of 7 consecutive days when the platelet count exceeded 20 × 109/l without transfusions. Acute GVHD was graded according the standard criteria.18,19 Chronic GVHD was assessed in patients alive after day 100.20 Bacteremia was defined as the first positive blood culture related to a febrile episode during the first 30 days after transplantation. CMV reactivation was determined weekly by PCR during hospitalization. CMV disease was recognized from the clinical manifestation of this infection with PCR positivity in the blood, bronchoalveolary lavage or urine. Mycological methods were used to document fungal infections in patients who appeared to have fungi. Transplant-related mortality (TRM) was defined as death in the absence of relapse. Relapse was diagnosed from BM specimens taken on day 30, day 100 and every hundred days regularly or when clinically indicated.
Statistics
Analysis was performed on 21 September 1999 with a median follow up of 558 days (76–1860 days), corresponding to 18.6 months (2.5–62 months). The Kaplan–Meier method with log-rank test (Mantel–Haenszel) was used to estimate probability of overall and disease-free survival, acute GVHD, myeloid and platelet engraftment. The Mann–Whitney U test was used to compare cell yield, time to engraftment, transfusions, and hospitalization.21,22 Only patients surviving more than 30 days were included in the analysis of acute GVHD. A minimum of 100 days of follow-up was the criterion for chronic GVHD.
Results
Cell yield in grafts
The numbers of nucleated cells, CD34+, CD3+ and CD56+ cells are given in Table 2. A significant difference in the cell count in grafts was demonstrated between the BMT and PBSCT groups. The numbers of counted cells were higher in the PBSCT group.
Table 2.
Cell yield

Myeloid and platelet engraftment, transfusions and hospitalization
Thirty-two (87%) patients in the PBSCT group and 36 (97.5%) patients in the BMT group showed myeloid and 28 (75%) and 26 (70%) platelet engraftment. Late graft failure was seen in one female after PBSCT and one male after BMT. Median time to leukocyte engraftment in the PBSCT group was 14.5 days (8–35 days) and 19 days (10–33 days) in the BMT group. Time to ANC greater than 0.5 × 109/l was significantly faster in the PBSCT than in the BMT group (Figure 1, P < 0.02). Time to reach a platelet count greater than 20 × 109/l, without platelet transfusion, tended to be longer in the patients receiving BMT than in the PBSCT group (median 14 days; range 12–78 days and median 19 days; range 19–100 days; Figure 2, P = 0.66). The PBSCT group required fewer platelet transfusions than the BMT group, but no statistically significant difference in the number of transfusions was detected between the two groups (6 vs 7 and 6 vs 6 transfusion units, respectively). The median time to discharge from the hospital was 48.5 days in the PBSC group and 45.5 days in the BM group without a statistically significant difference.
Figure 1.
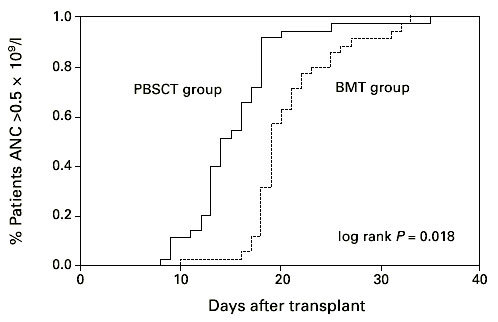
Days to an ANC recovery >0.5 × 109/l after hematopoietic stem cell transplantation with unmanipulated PBSC or BM. PBSCT group vs BMT group P < 0.02.
Figure 2.
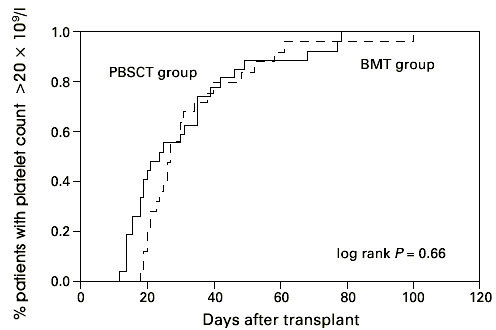
Days to platelet recovery >20 × 109/l after hematopoietic stem cell transplantation with unmanipulated PBSC or BM. PBSCT group vs BMT group P = 0.66.
Infections
We assessed all viral, bacterial and fungal infections during the time of hospitalization up to day 100 (Figure 3). Bacteremia occurred in 15 patients including seven in the BMT group and eight in the PBSCT group. Out of the total number of 74 patients, eight (11%) suffered from Gram-positive coccal infections (three in the BMT group and five in the PBSCT group). In a comparatively high number of patients (three in the PBSC group and four in the BM group) Gram-negative bacteria were identified in blood cultures: four cases of Pseudomonas aeruginosa and three of Stenotrophomonas maltophilia. No one died from septicemia. Only one patient (in the PBSCT group) developed bacterial pneumonia that was followed by a therapy refractory ARDS. CMV-PCR was positive in 24 patients, namely, 13 in the PBSC group and 11 in the BM group. The number of patients with CMV reactivation (PCR positivity) was low due to suitable pre-emptive treatment and prophylaxis strategy: gancyclovir (5 mg/kg i.v.) 5 days a week was started in all high risk patients for CMV infection after engraftment. Only one patient developed a CMV infection and died from interstitial pneumonia (in the BMT group). One patient after BMT and acute GVHD, grade IV, that was successfully treated by combined immunosuppressive therapy died from herpes simplex pneumonia refractory to acyclovir. Five patients (all in the PBSCT group) suffered from hemorrhagic cystitis not earlier than day 40. BK virus was detected in all cases. Invasive fungal infection was diagnosed in five patients, three in the BMT and two in the PBSCT group. All these patients had a pretransplant history of pulmonary aspergillosis. Three patients died from progressive fungal pneumonia, refractory to liposomal (one patient) or conventional (two patients) amphotericin B. Two patients – one from each group – are alive and without any signs of pneumonia after treatment with liposomal amphotericin B. Other types of fungi were not detected. One patient in the PBSCT group, who was anti-toxoplasma IgM positive before the transplant, developed cerebral toxoplasmosis (pathological finding in section) with clinical signs of encephalitis. Excluding the hemorrhagic cystitis, because the reason for its occurrence is still not clearly determined (the role of BK virus in the pathogenesis is under investigation), the overall number of infectious complications in the two groups did not differ (12 patients in each group, Figure 3).
Figure 3.
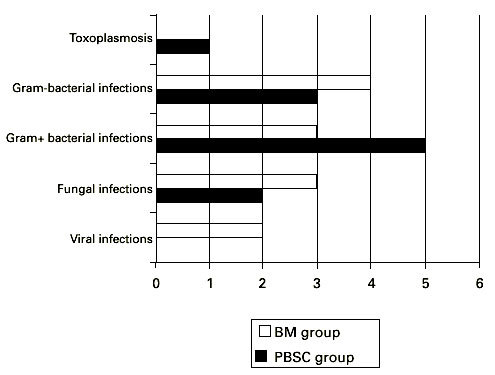
Number of patients with infections complications after hematopoietic stem cell transplantation with unmanipulated PBSC or BM.
Causes of death, relapses and transplant-related mortality (TRM)
Overall, TRM and relapse rate did not differ between the two groups. Eight relapses were diagnosed and all patients died in the BMT group. Seven patients in the PBSCT group were diagnosed with relapsed disease and six died, but one of them is in complete remission and well more than 1 year after chemotherapy in combination with donor lymphocyte infusion. Four patients died from severe infections (two with fungal infections and two with herpes virus infections) in the BMT group and two patients in the PBSCT group (toxoplasmic encephalitis, bacterial pneumonia). GVHD and multiorgan failure as causes of death were seen more often in the PBSCT group (four and two patients and four and one patient, respectively), but without any significant differences.
Acute GVHD and chronic GVHD
We found no significant difference in the incidence and time of onset of acute GVHD in patients transplanted with hematopoietic stem cells from matched unrelated donors. The incidence of acute GVHD grades II–IV in the HLA-identical PBSCT group (27 patients) was 35% and in the BM transplant group (30 patients) with HLA-identical donors it was 25% (Figure 4, P = 0.33; log-rank test). However, we found a significant difference (log-rank test, P < 0.05) in incidence and time to onset of severe acute GVHD comparing all patients, including the 17 mismatched transplants (Figure 5). Twelve patients suffered from acute GVHD higher than grade II in the PBSCT group (four died) and eight (four died) in the BMT group, respectively. Acute GVHD grade I developed in eight patients in the PBSCT group and in nine patients in the BMT group. In addition, we analyzed the cumulative incidence of chronic GVHD in the two groups. The cumulative probability of chronic GVHD was 58% among patients receiving a PBSC graft and 42% for patients receiving a BM graft (NS) (Figure 6). Two patients in the PBSCT group and four patients in the BMT group developed extensive chronic GVHD.
Figure 4.
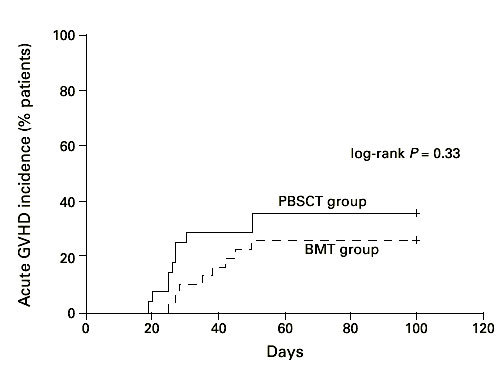
Time to and incidence of grades II–IV acute GVHD after hematopoietic stem cell transplantation with unmanipulated HLA-identical PBSC (n = 27) or BM (n = 30). PBSCT group vs BMT group (HLA-identical) P = 0.33.
Figure 5.
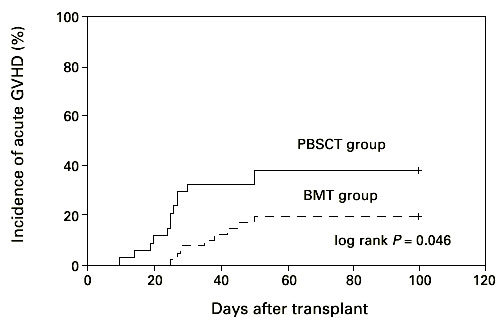
Time to and incidence of grades II–IV acute GVHD after hematopoietic stem cell transplantation with unmanipulated PBSC (n = 37) or BM (n = 37). PBSCT group vs BMT group P < 0.05.
Figure 6.
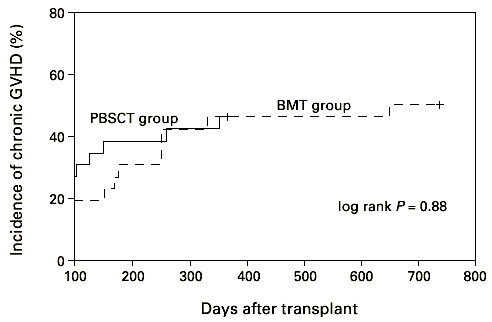
Time to and incidence of chronic GVHD after hematopoietic stem cell transplantation with unmanipulated PBSC or BM. PBSCT group vs BMT group P = 0.88.
Overall survival and disease-free survival
The median overall survival was 1123 days in the BMT group and 511 days in the PBSCT group (Figure 7, P = 0.33; log-rank test). Disease-free survival reached 51% and 41%, respectively, and was not significantly different between the two groups (Figure 8, P = 0.27; log-rank test).
Figure 7.
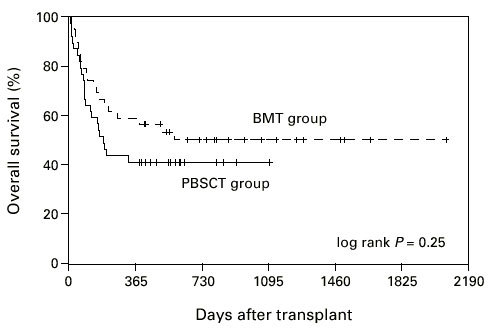
Overall survival of patients grafted with unmanipulated PBSC or BM. PBSCT group vs BMT group P = 0.33.
Figure 8.
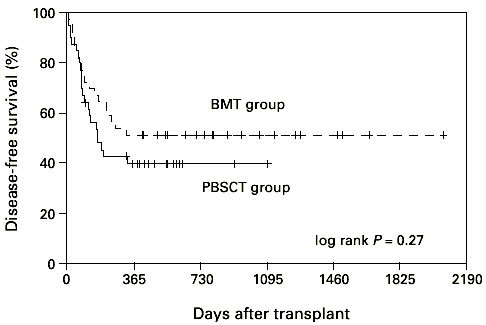
Disease-free survival of patients grafted with unmanipulated PBSC or BM. PBSCT group vs BMT group P = 0.63.
Discussion
This study is one of the first to prospectively establish that PBSCT from unrelated donors is feasible and leads to a faster leukocyte recovery in comparison to BMT. Thirty-seven patients received PBSCT from unrelated donors from 1996 up to May 1999 and 37 patients received BMT. Ringden et al23 also reported a significantly faster neutrophil engraftment in the PBSCT group. It should be noted that the patients in BMT group were not treated with G-CSF post-transplantation, which may influence the difference in neutrophil recovery between BMT and PBSCT. The use of G-CSF after transplantation is not the practice in our center because of the effect on residual leukemia which could not be excluded.5 However, the incidence of infections was not different in the two study groups, probably as a consequence of the small number of patients with documented infections in both groups. Nevertheless, the overall number of documented infectious complications and death from severe infections was lower in the PBSCT group. Time to platelet engraftment did not differ in the two groups, which contrasted with the experience of other studies in related settings.2,3 Recently, Blaise et al24 reported in a randomized trial that PBSCT resulted in quicker neutrophil and platelet recovery than does BMT from sibling donors. However, a higher occurrence of chronic GVHD was observed. The main finding in this study is the significantly higher incidence and shorter time of onset of severe acute GVHD in patients who received PBSCT. The rate of acute GVHD observed after allogeneic PBSCT from HLA-identical siblings has been reported to be similar to BMT.25 In a recently published multicenter analysis26 we demonstrated a trend to a higher incidence of severe acute GVHD in the PBSC group compared to the BM group (30% vs 20%). The results in our recent analysis of transplants from unrelated HLA-identical donors26 corresponds with the published data,27 especially with the results of prospective, randomized trials in transplants from related donors presented at the meeting of ASH last year.11,12 Including the data from transplants of HLA-non-identical unrelated donors might introduce differences due to matching mistakes. The number of patients with donors who were not HLA identical was 10 in the PBSCT group and seven in the BM group. Another reason for the higher incidence of acute GVHD might be the number of high risk patients. In the PBSCT group it was somewhat higher in comparison to the BMT group (20 vs 14). In addition, the CMV positivity of patients prior to transplantation was known to be a risk factor for acute GVHD. CMV positivity was not matched, so there were significant differences between the two groups (the number of negative patients was 14 in the PBSCT and 23 in the BMT group, respectively). It should also be noted that the CD34+ as well as CD3+ cell differential is significantly higher in PBSC grafts in comparison to BM grafts, and may partly explain the higher incidence of acute GVHD in PBSCT group. The impact of G-CSF on T cell functions as well as on alloreactivity for both GVH and GVL effects is still unclear and needs further investigation.28,29 The incidence of chronic GVHD in the PBSCT and BMT groups was not different (58% and 42%, respectively). With regard to overall survival, relapse and relapse-free survival, there was no difference between outcome in patients receiving PBSCT compared with the BMT group. However, it should be noted that the end survival and DFS in the BMT group showed a trend to be better probably due to the slightly worse TRM in the BMT group. This is in agreement with the experience in the HLA-identical siblings as well as in the unrelated setting.2,3,26,27 The follow-up of our patients was short and the data need to be re-evaluated with time. We conclude that PBSCT from unrelated donors is a safe procedure and results in faster neutrophil engraftment than BMT. The eventual disadvantages for the donors need several years follow-up in order to answer different questions raised with regard to the treatment with G-CSF. However, according to the data published to date,30,31 it appears that G-CSF treatment and PBSC collection have an acceptable short- and long-term safety profile in normal donors. Thus, for donors the benefits are obvious, but for recipients the benefits are less so. The incidence of acute GVHD in HLA-identical PBSC transplants was not higher than in BM transplant patients. Finally, the results of this study provide evidence that PBSCT from unrelated HLA-compatible donors can be safely administered as an alternative to BMT. However, the use of PBSCT from HLA-mismatched donors needs further investigation and follow-up with regard to acute and chronic GVHD. On the other hand, the important antileukemic effect of chronic GVHD should not be neglected. We propose to use PBSC as a source for unrelated donors providing that these donors are HLA identical, identified by using serological for class I and genomic for class II HLA typing. With regard to PBSC from HLA-mismatched donors further investigations and follow-up are needed in order to answer the question on the incidence of acute as well as chronic GVHD. In conclusion, we believe that PBSCT by using HLA-mismatched unrelated donor is feasible and safe in a carefully selected patient population.
References
- 1.Kessinger A, Smith DM, Strandjord SE. Allogeneic transplantation of blood-derived, T cell-depleted hematopoietic stem cells after myeloablative treatment in a patient with acute lymphoblastic leukemia. Bone Marrow Transplant. 1989;4:643–646. [PubMed] [Google Scholar]
- 2.Schmitz N, Bacigalupo A, Labopin M. Transplantation of allogeneic peripheral blood progenitor cells – the EBMT experience. Bone Marrow Transplant. 1996;17:40–46. [PubMed] [Google Scholar]
- 3.Körbling M, Przepiorka D, Huh YO. Allogeneic blood stem cell transplantation: peripheralization and yield of donor-derived primitive hematopoietic progenitor cells (CD34+ Thy-1dim) and lymphoid subsets, and possible predictors of engraftment and graft-versus-host disease. Blood. 1995;86:2842–2848. [PubMed] [Google Scholar]
- 4.Russell JA, Brown C, Bowen T. Allogeneic blood cell transplants for haematological malignancy: preliminary comparison of outcomes with bone marrow transplantation. Bone Marrow Transplant. 1996;17:703–708. [PubMed] [Google Scholar]
- 5.Bensinger WI, Clift R, Martin P. Allogeneic peripheral blood progenitor cell transplantation in patients with advanced hematologic malignancies: a retrospective comparison with marrow transplantation. Blood. 1996;88:2794–2800. [PubMed] [Google Scholar]
- 6.Ottinger HD, Beelen DW, Scheulen B. Improved immune reconstitution after allotransplantation of peripheral blood stem cells instead of bone marrow. Blood. 1996;88:2775–2779. [PubMed] [Google Scholar]
- 7.Sierra J, Storer B, Hansen JA. Transplantation of marrow cells from unrelated donors for treatment of high-risk acute leukemia: the effect of leukemia burden, donor HLA-matching, and marrow cell dose. Blood. 1997;89:4226–4235. [PubMed] [Google Scholar]
- 8.Urbano-Ispizua A, Solano C, Brunet S. Allogeneic peripheral blood progenitor cell transplantation: analysis of short-term engraftment and acute GVHD incidence in 33 cases. Bone Marrow Transplant. 1996;18:35–40. [PubMed] [Google Scholar]
- 9.Storek J, Gooley T, Siadak M. Allogeneic peripheral blood stem cell transplantation may be associated with a high risk of chronic graft-versus-host disease. Blood. 1997;90:4705–4709. [PubMed] [Google Scholar]
- 10.Scott MA, Gandhi MK, Jestice HK. A trend towards an increased incidences of chronic graft-versus-host disease following allogeneic peripheral blood progenitor cell transplantation: a case controlled study. Bone Marrow Transplant. 1998;22:273–276. doi: 10.1038/sj.bmt.1701327. [DOI] [PubMed] [Google Scholar]
- 11.Bensinger W, Martin P, Clift R. A prospective, randomized trail of peripheral blood stem cell (PBSC) transplants or marrow (BM) for patients undergoing allogeneic transplantation for hematologic malignancies. Blood. 1999;10:368a. [Google Scholar]
- 12.Durant S. Morton AJ. A randomized trial of filgrastim (G-CSF) stimulated donor marrow (BM) versus peripheral blood (PBSC) for allogeneic transplantation: increased extensive chronic GVHD following PBSC transplantation. Blood. 1999;10:608a. [Google Scholar]
- 13.Anderlini P, Przepiorka D, Körbling M, Champlin R. Blood stem cell procurement: donor safety issues. Bone Marrow Transplant. 1998;21:35–39. [PubMed] [Google Scholar]
- 14.Bensinger WI, Price TH, Dale DC. The effects of daily recombinant human granulocyte colony stimulating factor administration on normal granulocyte donors undergoing leukapheresis. Blood. 1998;81:1883–1888. [PubMed] [Google Scholar]
- 15.Becker PS, Wagle M, Matous S. Spontaneous splenic rupture following administration of granulocyte colony stimulating factor (G-CSF): occurence in an allogeneic donor of peripheral blood stem cells. Blood Marrow Transplant. 1997;3:45–49. [PubMed] [Google Scholar]
- 16.Basara N, Blau IW, Kiehl MG. Mycophenolate mofetil for the prophylaxis of acute GVHD in HLA-mismatched bone marrow transplant patients. Clin Transplant. 2000;14:121–126. doi: 10.1034/j.1399-0012.2000.140204.x. [DOI] [PubMed] [Google Scholar]
- 17.Hägglund H, Ringden O, Remberger M. Faster neutrophil and platelet engraftment, but no differences in acute GVHD or survival using peripheral blood stem cells from related and unrelated donors, compared to bone marrow. Bone Marrow Transplant. 1998;22:131–136. doi: 10.1038/sj.bmt.1701298. [DOI] [PubMed] [Google Scholar]
- 18.Thomas ED, Storb R, Clift RA. Bone marrow transplantation. Parts I, II. New Engl J Med. 1975;293:832–840, 895–902. doi: 10.1056/NEJM197512112932409. [DOI] [PubMed] [Google Scholar]
- 19.Przepiorka D, Weisdorf D, Martin P. Meeting report: concensus conference on acute GVHD grading. Bone Marrow Transplant. 1995;15:825–828. [PubMed] [Google Scholar]
- 20.Shulman HM, Sullivan KM, Weiden PI. Chronic graft-versus-host syndrome in man. A long term clinicopathologic study of 20 Seattle patients. Am J Med. 1980;69:204–217. doi: 10.1016/0002-9343(80)90380-0. [DOI] [PubMed] [Google Scholar]
- 21.Peto R, Pike MC, Armitage P. Design and analysis of randomized clinical trails requiring prolonged observation of each patient. II. Analysis and examples. Br J Cancer. 1977;35:1–39. doi: 10.1038/bjc.1977.1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Armitage P, Berry G. Statistical Methods in Medical Research. 1987. [Google Scholar]
- 23.Ringden O, Lönnqvist B, Hägglind H. Transplantation with peripheral blood stem cells from unrelated donors without serious graft-versus-host disease. Bone Marrow Transplant. 1995;16:856–857. [PubMed] [Google Scholar]
- 24.Blaise D, Kuentz M, Fortanier C. Randomized trial of bone marrow versus lenograstim-primed blood cell allogeneic transplantation in patients with early-stage leukemia: a report from the Societe Française de Greffe de Moelle. J Clin Oncol. 2000;18:537–546. doi: 10.1200/JCO.2000.18.3.537. [DOI] [PubMed] [Google Scholar]
- 25.Schmitz N, Bacigalupo A, Hasenclever D. Allogeneic bone marrow transplantation vs filgrastim-mobilized peripheral blood progenitor cell transplantation in patients with early leukaemia: first results of a randomized multicenter trial of the European group for Blood and Marrow Transplantation. Bone Marrow Transplant. 1998;21:995–1003. doi: 10.1038/sj.bmt.1701234. [DOI] [PubMed] [Google Scholar]
- 26.Ringden O, Remberger M, Runde V. Peripheral blood stem cell transplantation from unrelated donors: a comparison with marrow transplantation. Blood. 1999;94:455–464. [PubMed] [Google Scholar]
- 27.Stockschläder M, Löliger C, Krüger W. Transplantation of allogeneic rhG-CSF mobilized peripheral CD34+ cells from an HLA-identical unrelated donor. Bone Marrow Transplant. 1995;16:719–722. [PubMed] [Google Scholar]
- 28.Pan L, Delmonte J Jr, Jalonen C. Pretreatment of donor mice with granulocyte colony stimulating factor polarizes donor T-lymphocytes toward type-2 cytokine production and reduces severity of experimental graft-versus-host-disease. Blood. 1995;86:4422–4429. [PubMed] [Google Scholar]
- 29.Tayebi H, Tiberghien P, Kuentz M. Comparison of HLA-identical allogeneic bone marrow (BM) versus peripheral blood stem cells (PBSC) transplantation: phenotypical and functional study of donor T cells in the graft and after hematopoietic reconstitution. Blood. 1998;92:140 (Abstr. 562). [Google Scholar]
- 30.Anderlini P, Körbling M, Dale D. Allogeneic blood stem cell transplantation: considerations for donors. Blood. 1997;90:903–908. [PubMed] [Google Scholar]
- 31.Storek J, Dawson M, Maloney DG. Normal T,B, and NK cell counts in healthy donors at 1 year after blood stem cell harvesting (letter) Blood. 2000;95:2993–2994. [PubMed] [Google Scholar]


