Abstract
Vaccination against the SARS-CoV infection is an attractive means to control the spread of viruses in public. In this study, we employed a DNA vaccine technology with the levamisole, our newly discovered chemical adjuvant, to generate Th1 type of response. To avoid the enhancement antibody issue, genes encoding the nucleocapsid, membrane, and envelope protein of SARS-CoV were cloned and their expressions in mammalian cells were determined. After the intramuscular introduction into animals, we observed that the constructs of the E, M, and N genes could induce high levels of specific antibodies, T cell proliferations, IFN-γ, DTH responses, and in vivo cytotoxic T cells activities specifically against SARS-CoV antigens. The highest immune responses were generated by the construct encoding the nucleocapsid protein. The results suggest that the N, M, and E genes could be used as the targets to prevent SARS-CoV infection in the DNA vaccine development.
Keywords: SARS-CoV, Nucleocapsid protein, Membrane protein, Envelop protein, DNA vaccine, In vivo CTL, Th1 response
Severe acute respiratory syndrome (SARS) is a highly contagious disease, caused by a human coronavirus infection (SARS-CoV). The genome of SARS-CoV is a single-stranded positive-sense RNA including a 5′-cap and a 3′-polyadenylation structure, and encodes five major proteins typical of coronaviruses including the replicate polyproteins, the spike protein (S), the envelope protein (E), the membrane glycoprotein (M), and the nucleocapsid protein (N). To develop a safe and yet effective SARS vaccine is a new challenge for laboratories in the world. Spike protein has been indicated as the key protein to interact with the cell receptor in coronaviral family including the SARS-CoV [1]. Immunization with the spike protein or killed viral vaccine induces an enhancement antibody which accelerates subsequent infection of the feline coronavirus [2], [3]. It suggests that the spike protein may not be a good candidate antigen used as vaccine development.
The M and E are two proteins playing an essential role in coronaviral particle assembling, and immune responses against these antigens should also be effective for inhibiting the viral infectivity or helping host cells to induce the production of protective IFN-α [4], [5]. As for the viral vaccine design, much attention has been focused on the induction of T cell responses, especially for the generation of cytotoxic T lymphocytes (CTL). Several studies have demonstrated that protective CTL responses were induced by the surface antigen of HBsAg virus [6], [7]. However, more reports have shown that the nucleocapsid protein can be a good target to induce the protective CTL response in viruses, such as Ebola [8], influenza [9], [10], and measles [11].
DNA vaccination has emerged as a practical technology for the generation of humoral and cellular immunity [12], and potentially used against some viral infections including human immunodeficiency virus [13], hepatitis B [14], and influenza [15].
Taking this into account, we examined the immune responses generated with DNA vaccines encoding the N, M, and E proteins of SARS-CoV into mice. All the three DNA vaccines induced potent Th1 polarized immune responses as well as specific antibodies in these animals. And the highest levels of humoral response and T cell proliferation activity were induced by the N gene construct. We also observed that in vivo CTL activities were successfully induced in mice by these DNA constructs.
Materials and methods
Animals. Female BALB/c mice 4–6 weeks old were purchased from Beijing Vital Laboratory Animal Technology (Beijing, China), randomly divided into groups at 10 per group, and received pathogen-free water and food for maintenance.
Virus isolation. A 35-year-old female patient was hospitalized and confirmed with diagnosis as infected with SARS-CoV. The SARS-CoV isolation was established by infecting Vero cells with the filtered stool sample from this patient under BSL-3 condition. Cytopathic effects were observed after 3-day cultures. cDNA was generated from the RNA isolated from the supernatant of the culture and used for the sequence analysis to confirm its SARS-CoV identity.
Plasmid constructs for immunization. All DNA vaccine constructs, encoding the E protein, the M glycoprotein, and the N protein of SARS-CoV, were obtained as follows: these genes were amplified from the cDNA by PCR amplifications using each set of specific primers, respectively. For the N gene, the upstream primer is 5′-AAGAATTC GCCATGTCTGATAATGGACCC-3′ and the downstream one is 5′-AAGAATTCGTTTATGCCTGAGTTGAATC-3′; for the M gene, the upstream primer is 5′-AAGAATTC GCCATGGCAGACAACGGTACTAT-3′ and the downstream one is 5′-AATCTAGATTACTGTACTAGCAAAGCAATAT-3′; and for the E gene, the upstream primer is 5′-AAGAATTC GCCATGTACTCATTCGTTTCG-3′ and the downstream one is 5′-AATCTAGATTAGACCAGAAGATCAG-3′. To express the three genes in eukaryotic cells, the sequences containing an ATG start codon and a Kozak consensus sequence, GCC, were incorporated into 5′ end of each upstream primer. A restriction digestion site was also incorporated into each primer indicated as underlined. All the amplified products were first cloned into pDM18-T vectors and confirmed by the sequence analysis, respectively. The three products were, respectively, subcloned into appropriate sites of pcD3d, a 3.4 kb eukaryotic expression vector derived from the pcD3d vector (Invitrogen) by a deletion of the regions for the SV40 promoter, neomycin ORF, and SV40 poly(A). The final products were designated as the pcD3d/E, pcD3d/M, and pcD3d/N, respectively. All three DNA vaccines were transformed into the Escherichia coli strain Top10 (Invitrogen), extracted and purified by PEG8000 precipitation [16]. The final products were dissolved by saline solution and stored at 4 °C until immunization.
Determination of expression of DNA construct. HeLa cells were seeded at 4 × 105 cells per plate in a 16 cm2 plate containing a complete DMEM, supplemented with 10% fetal bovine serum (Gibco/BRL). The cells were cultured in a humidified incubator at 37 °C and 5% CO2 until 80% confluent. The three constructs were transfected into the HeLa cells using the Lipofectamine according to the manufacturer’s instruction (Invitrogen, CA, USA), respectively. Each total cellular RNA was extracted from the harvested cells using an extraction kit (Sangon, Shanghai, China) 24 h after the transfection. The cDNA was synthesized from 2 μl of the total RNA in a 20 μl reaction system consisting of 4 μl reverse transcriptase buffers (250 mM Tris–HCl, pH 8.3, 375 mM KCl, 40 mM MgCl2, and 50 mM DTT), 50 pmol oligo(dT)18 primer, 0.5 mM dNTP, 10 U AMV reverse transcriptase, and 20 U RNase inhibitor. The reaction was performed at 42 °C for 30 min and 99 °C for 5 min. RNAs derived from HeLa cells that were transfected with a control vector were processed in parallel as a negative control. The cDNAs were amplified by PCR with each pair of SARS-CoV-specific primers described above for 30 cycles (94 °C for 30 s, 55 °C for 30 s, and 72 °C for 50 s) and with an extension for 7 min at 72 °C in the last cycle. The PCR products were loaded on the 1.5% agarose gel and visualized under a 302 nm UV light.
Immunizations. For immunization, the DNA constructs were mixed with 1% levamisole (LMS) as an adjuvant for the final injection according to our previous study [17]. Each mouse was injected with 100 μg/100 μl DNA vaccine intramuscularly twice at a biweekly interval. Mice sera were collected regularly on day 0, 14, 28, 42, and 56, and stored at −20 °C for later use.
Detection of anti-SARS-CoV antibody. Serum anti-SARS-CoV antibody levels were assayed by enzyme-linked immunosorbent assay (ELISA) [18]. Ninety-six wells of microtiter plates were coated with 2 μg/ml of a chemically killed SARS-CoV antigen per well diluted in 100 μl of 0.05 M bicarbonate buffers (pH 9.6) at 4 °C overnight. Following the blocking of each well with 3% BSA-PBST at 37 °C for 1 h, the mouse serum with a serial dilution was added to each well and incubated at 37 °C for 1 h. A secondary goat anti-mouse antibody conjugated with horseradish peroxidase (Sigma, St. Louis, IL) at 1:1000 was added into each well and incubated at 37 °C for 1 h. To develop the ELISA, 10 mg TMB tablet (Sigma, St. Louis) was dissolved in 1 ml of 0.025 M phosphate–citrate buffer and 50 μl of the resulting solution was added to each well. The reaction was stopped by addition of 0.2 M H2SO4 and the resulting optical density (OD) at 450/620 nm was analyzed with a plate reader (Magellan, Tecan Austria GmbH). Titer values were assigned as the highest dilution at which the optical density was 2 SDs higher than the OD produced by the serum of pre-immune mice at equivalent dilution.
T cell proliferation activity. On the 14th day after the second injection, the mice were sacrificed and single-cell suspension was prepared from spleens for each group as described previously [17]. The splenocytes were cultured in triplicate in 96-well microtiter plates at 5 × 104 cells per well in RPMI-10. The cultures were, respectively, stimulated for 24 h with each of the following: 5 μg/ml Con A (positive control), 2 μg/ml of the chemically killed SARS-CoV as the specific antigen, 2 μg/ml BSA (irrelevant antigen), or none (negative control). The cells were incubated at 37 °C and 5% CO2 for 48 h. The colorimetric reaction was performed according to “Cell Titer 96 Aqueous Non-Radioactive Cell Proliferation Assay” protocol (Promega, USA). Briefly, the cells were mixed with a mixture of MTS/PMS (20 μl each well) and further cultured for 4 h. The OD value was quantitatively read by absorbance at 490 nm with the plate reader (Magellan, Tecan Austria GmbH).
The stimulation indexes (SI) were determined by mean OD492 reading of triplicate antigen containing wells divided by mean OD492 reading of triplicate wells without antigen.
Antigen-specific DTH. Five mice from each group were tested for DTH response. Seven days after the second immunization, all mice were challenged with the chemically killed SARS-CoV in the right footpad and also received injections with saline solution at the left footpad as the control. The thickness of the footpad was measured at 24, 48, and 72 h later with an engineer’s micrometer.
In vivo CTL assay. In vivo cytotoxicity assay has been recently documented [19], [20]. Briefly, to prepare target cells for the detection of in vivo cytotoxic activity, splenocytes were isolated from naïve BALB/c spleen after erythrocytes were removed. The single suspension cells were then washed and equally split into two parts, one of which was pulsed with the chemically killed SARS-CoV (50 μg/ml) at 37 °C for 4 h and labeled with a high concentration of CFSE (2.5 μM, Molecular Probe, Portland, USA) designated as the CFSEhigh cells; while the other, as a control target, was only labeled with a low concentration of CFSE (0.25 μM) designated as the CFSElow cells. The two populations of cells were mixed together and injected intravenously at 2 × 107 cells into each mouse, which was previously immunized with E, M, N or control DNA constructs 7 days earlier. The mouse was sacrificed 4 h later to isolate lymph nodes. Cell suspensions from the lymph nodes were analyzed on a FACSCalibur analyzer (BD Biosciences, Mountain View, CA), and each population was detected via its differential CFSE fluorescent intensities. To calculate specific lysis, the following formula was used: ratio = (percentage CFSElow/percentage CFSEhigh). Percentage-specific lysis = [1 − (ratio unprimed/ratio primed) × 100].
Cytokine assay. Relative amounts of cytokine levels were determined by semi-quantitative RT-PCR with a known amount of competitor, pQRS, as previously described [21]. An extraction kit (Sangon, Shanghai, China) was used to extract total RNA. The cDNA was synthesized by AMV reverse transcriptase and oligo(dT)18 primer. Each PCR was optimized with specific primers for the housekeeping or cytokine genes as described previously [17]. The amounts of specific competitor products of each reaction were assessed from scanned images of ethidium bromide-stained gels using Bio-Rad Image software (Quantity One 4.2.0). The total volume (intensity/area) of the specific product was determined and the statistical analysis was performed.
Statistical analysis. Statistical analysis was performed using Student’s t test, one-sided. Differences were considered statistically significant with p < 0.05.
Results
Expression of DNA constructs in HeLa cells
The identifications of the three constructs were analyzed by restriction digestions and further confirmed by the sequence analysis (data not shown).
In order to confirm that the DNA constructs, pcD3d/E, pcD3d/M, and pcD3d/N, can express the E, M, and N proteins in the eukaryotic systems, each construct was transfected into 80% of confluent HeLa cells. The total RNA was extracted from the cells 24 h after the transfection and used to perform the RT-PCR assays to detect the presence of the E, M, and N mRNAs with each pair of their specific primers. The target bands were observed from the cells transfected with these constructs, whereas no band with a similar size was detected from the pcD3d vector control transfected cells (Fig. 1 ). This result suggests that the three DNA constructs encoding the E, M, and N proteins can be successfully expressed in eukaryotic cells.
Fig. 1.
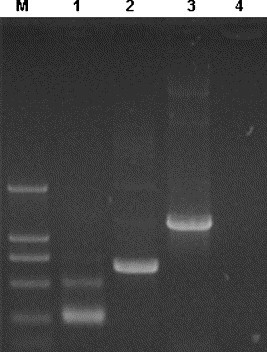
Eukaryotic expression of DNA vaccines in HeLa cells. Total RNA was extracted from HeLa cells 12 h after transfected with pcD3d/E (lane 1), pcD3d/M (lane 2), pcD3d/N (lane 3), and pcD3d (lane 4). The RNAs were used to perform RT-PCR with specific primers, respectively. Positive band of each construct was observed in the transfected sample.
Generation of specific IgG in mice
To examine the humoral responses in mice, the levels of anti-SARS-CoV-specific antibodies of the mice immunized with pcD3d/E, pcD3d/M, and pcD3d/N, respectively, were determined by ELISA. Sera from day 14, 28, 42, and 56 were determined with the titers of specific IgG and shown in Fig. 2 . All three constructs induced significantly higher levels of SARS-CoV-specific antibodies than that of the control group immunized with pcD3d vector. Moreover, among all inoculations, pcD3d/N induced the highest specific antibody titers against SARS-CoV.
Fig. 2.
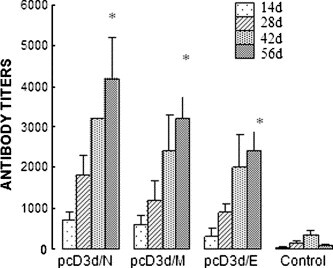
Antibody responses to SARS-CoV mice (five per group) that were bled on day 14, 28, 42, and 56 after initial immunization. The mean titers with standard deviation (SD) of antibodies in five animals against CoV were evaluated by ELISA with 2 μg/ml of chemically killed SARS-CoV as antigen coated on each well in a 96-well plate and by using an absolute ratio of Post/Naïve serum at a cutoff of 2.1. *Indicates values that are statistically significant at a p value <0.05 by Student’s t test compared with the group of control vector.
T cell proliferation after DNA vaccine immunization
To determine the T cell proliferative response, single suspension of splenocytes was prepared from the mice spleen 14 days after the second immunization to perform the T cell proliferation assay. Fig. 3 shows that the highest stimulation indexes are achieved in the T cells isolated from spleens (Fig. 3A) and lymph nodes (Fig. 3B) of mice injected with pcD3d/N. The levels of proliferative responses of the mice immunized with the pcD3d/E and pcD3d/M are significantly higher than those of animals immunized with the pcD3d vector control.
Fig. 3.
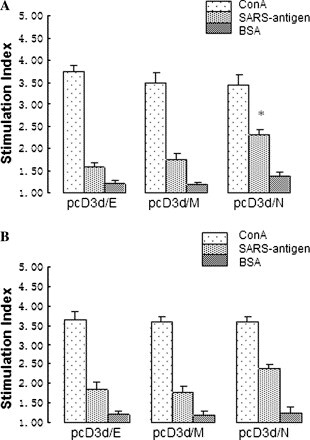
T cell proliferation with MTS colorimetric detection. Single suspension of splenocytes and lymphocytes of the immunized mice was isolated 2 weeks after the second immunization and stimulated in vitro either with the killed SARS-CoV for test groups or an unrelevant protein, BSA for negative control, or with Con A as the positive control. Splenocytes from a group of normal mice were stimulated with chemically killed SARS-CoV in vitro which served as the sham control. The stimulation index was derived from the value of test group divided by medium control group. (A) T cell proliferation responses from the spleen of the animals. (B) T cell proliferation responses from the spleen of the animals. *Indicates values that are statistically significant at a p value <0.05 by Student’s t test compared with all other groups.
SARS-CoV-dependent DTH responses
Since DTH is a reflection of antigen-specific T cell activation and proliferation in vivo from the antigen-sensitized animal reacting on the site challenged antigen, we next evaluated whether the SARS-CoV-dependent DTH could also be induced by the three DNA vaccines. On the 7th day after the second immunization, each group of mice was challenged with the chemically killed SARS-CoV on their right footpads, and saline solution on the left footpads as the negative control. The thickness of footpad was measured at 24, 48, and 72 h after challenging. From Table 1 , we observed that the group of mice injected with the pcD3d/N showed the highest level of DTH, followed by the groups immunized with the pcD3d/M and pcD3d/E constructs.
Table 1.
Determination of DTH responsesa
| Immunization groups | Thickness of footpad (mm)b |
|||||
|---|---|---|---|---|---|---|
| Left foot |
Right foot |
|||||
| 24 h | 48 h | 72 h | 24 h | 48 h | 72 h | |
| pcD3d/E | 1.43 ± 0.15 | 1.6 ± 0.17 | 1.4 ± 0.01 | 2.4 ± 0.15⁎ | 2.67 ± 0.05⁎ | 2.1 ± 0.01⁎ |
| pcD3d/M | 1.47 ± 0.05 | 1.6 ± 0.01 | 1.43 ± 0.11 | 2.53 ± 0.05⁎ | 2.83 ± 0.05⁎ | 2.27 ± 0.05⁎ |
| pcD3d/N | 1.57 ± 0.05 | 1.7 ± 0.01 | 1.53 ± 0.05 | 2.7 ± 0.17⁎ | 3.07 ± 0.15⁎ | 2.2 ± 0.01⁎ |
| pcD3d vector | 1.53 ± 0.05 | 1.55 ± 0.06 | 1.4 ± 0.01 | 1.76 ± 0.11 | 1.9 ± 0.11 | 1.63 ± 0.15 |
Mice used for challenge on day 7 after boost immunizations with indicated immunogens in two weeks. 146S antigen (20 μl, 1 mg/ml) was injected intradermally on each right footpad and and 20 μl saline on each left footpad for the challenges.
Thickness was measured at 24, 48, and 72 h after the injection of antigen. Shown are means ± SDs (n = 3).
p < 0.05 when compared to the vector control group.
In vivo CTL
Specific CTL activity has been demonstrated as an important factor to control viral infections. Previously many studies to access the CTL activity were performed in in vitro system, which may not directly reflect the situation in vivo. To detect the presence of anti-SARS-CoV-specific CTL in vivo, we have adopted the method previously documented [19], [20]. After the target splenocytes differential labeled with CSFE were transferred into immunized or control mouse intravenously, specific antigen-labeled target cells were killed while the antigen unlabeled target would have survived. Fig. 4 shows that the degree of specific lysis is achieved in target cells isolated from the lymph node 4 h after the transfer into the immunized syngeneic host. The specific lysis of target cells at 50.6% (Fig. 4E) was observed in the mice immunized with the pcD3d/N construct, at 17% with those injected with the pcD3d/M construct (Fig. 4D), and at 5.6% with the group injected with the pcD3d/E construct (Fig. 4C). These data suggest that the mice vaccinated with the N construct produce the highest of CTL activities in vivo, which can be further concluded from the previous results that the N gene contains the more immunogenetic regions for T and B cell stimulations.
Fig. 4.
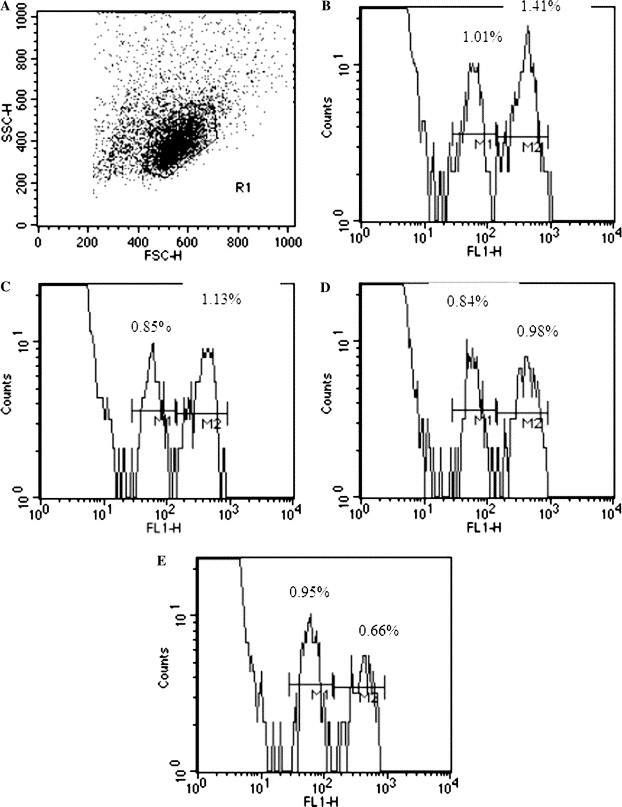
In vivo CTL assay. To analyze SARS-CoV-specific cytotoxicity in vivo, a 1:1 mixture of 107 cells of each target cell population was injected i.v. into naïve BALB/c mice 7 days after the second injection with the DNA vaccine. After 4 h, the mice were killed and the splenocytes were analyzed for the presence of CFSEhigh and CFSElow target cell populations. The percentage of specific lysis was then determined as described in Materials and methods. (A) Representative histogram plot of lymph node cells obtained from the mice injected with SARS-CoV DNA vaccines 4 h after transfer of CFSE-labeled target cells. (B–E) The SARS-CoV-specific cytotoxic responses in the lymph nodes are shown from mice previously injected by the pcD3d vector control, pcD3d/E, pcD3d/M, and pcD3d/N constructs, respectively. Each datum has been repeated twice.
Th1 and Th2 cytokine mRNA levels in mice
Having demonstrated a better immunogenicity generated from the N gene, the profile of cytokines may lead to understand such significance. To measure the relative level of cytokines, all RNA samples were competed with the same amount of a competitor, pQRS, at 150 pg/ml [21], which showed the expression of housekeeping gene, HPRT, and allowed a semi-quantitative comparison to be made. All groups of RNAs used in the RT-PCRs were initially normalized according to the level of the expression level of the HPRT. As depicted in Fig. 5 , it shows that the highest levels of mRNAs for IL-2 and IFN-γ were induced in the group immunized with the pcD3d/N construct, which suggests that the Th1 polarized response, followed by the groups immunized with the pcD3d/M and pcD3d/E constructs. For the Th2 cytokines, IL-4 mRNA expressions were observed after immunizations with N and M DNA vaccines. No IL-10 expression was observed, except in the group injected with M gene construct.
Fig. 5.
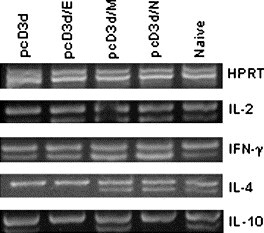
Semi-quantitative RT-PCR assay for cytokine gene expression. Total RNA was isolated from spleen of the immunized groups or normal mice (3 for each group). The levels of the cytokines IL-2, IFN-γ, IL-4, and IL-10 were semi-quantitatively measured by a competitive RT-PCR with the addition of the 150 pg/ml of the pQRS competitor after the normalization of tested cDNA concentration to a constant amount of HPRT as described in Materials and methods.
Discussion
DNA vaccination is receiving considerable attention due to its ability to induce both humoral and cellular immune responses. It induces immune responses to, and in some cases even leads to, the protection against various types of infection, such as influenza, malaria, and SARS [22], [23], [24]. A common feature of the DNA vaccination is that the synthesis of the antigen occurs in intracellular compartments, allowing the processed antigen to enter the MHC class I pathway [25], in turn to generate CD8+ CTL activities. In this report, we demonstrated that both humoral- and cellular-mediated specific responses could be induced by DNA constructs encoding N, M, and E antigens against the SARS-CoV. All three DNA constructs induced SARS-CoV-specific antibodies in mice and the pcD3d/N group showed the highest titer of antibodies (1:4800), followed by the pcD3d/M group (1:3200) and the pcD3d/E group (1:2400) (Fig. 2). In addition, the T cell proliferation and DTH responses were also successfully induced in mice after vaccinations with these constructs (Fig. 3 and Table 1). These results suggested that the DNA vaccine was effective to prime a specific anti-SARS-CoV response to and apparently generate a broad range of both T-helper and B-cell memory during the priming. This was also consistent with previous observation of the protective immunity [26], [27]. Furthermore, the administration with DNAs generated a lower level of IL-10 (Fig. 5), suggesting that a locomotor’s role of DNA initiated Th1 polarized immunity.
For vaccine design, much attention has been focused on the induction of T cell responses especially of the antigen-specific CTL. Several studies have found that the nucleocapsid protein could induce CTL responses in the cases of the human Hantaan virus [28], the lethal coronavirus peritonitis virus [29], and SHIV-1 [30]. Recently, Zhu et al. [31] have found that a DNA construct based on the pcD3d vector could successfully induce SARS-CoV N protein-specific immune responses in a manner of inducing high antibody titers and CTL responses. Kim [32] has demonstrated that the vaccination with N DNA vaccine could successfully induce a SARS-CoV antigen-specific CD8+ T cell response and distinctly reduce the titer of recombinant vaccinia virus expressing SARS-CoV N protein after the challenge and that the co-expression of calreticulin (CRT), a 46 kDa Ca2+-binding protein, with N gene could enhance its ability to protect against viral challenges. In the present study, it was consistent with their work that the N protein construct could induce the highest SARS-specific IgG, T cell proliferation, and in vivo CTL response (lysis rate of 50.6%) compared with M protein gene (lysis rate of 17%) and E protein gene (lysis rate of 5.6%) (Fig. 4). Furthermore, the highest Th1 type response based on expressions of the IFN-γ and IL-2 was also induced by N protein. However, the higher level of immune responses generated by the pcD3d/N vaccine may be due to more epitopes in the N nucleocapsid protein since it contains 422 amino acid residues, while the M protein has 220 amino acids; and the E has 76 amino acids. In addition, the differential concentrations of each antigen presented in the killed SARS-CoV preparation could have also contributed to the differences.
Specific CTL activity has been demonstrated as an important factor to confront virus infections. It has been previously documented that monkeys vaccinated with the DNA vaccine encoding the Gag protein, the nuclear protein, can induce specific CTLs in monkeys, and thus confer partial protection against the challenge with the SHIV (simian/human immunodeficiency virus) [30]. We have accessed the CTL in in vivo situation with the three DNA vaccines tested in this study and indicated that high levels of in CTL can be generated against the SARS-CoV-labeled target cells. Whether the in vivo CTL could control the SARS-CoV infection is needed for further investigation.
It is well known that the protective immunity against viral infections is associated with the induction of Th1 immune response. Production of the antigen-specific IL-2 and IFN-γ in the splenocytes was all augmented significantly by vaccinations of these three DNA constructs compared with the vector or the naïve control. The group receiving the immunization of the pcD3d/N induced the highest levels for both IL-2 and IFN-γ shown in Fig. 5, which is consistent with the highest levels of both antigen-specific T cell activity shown in Fig. 3 and DTH response in mice shown in Table 1.
In summary, the administrations with all three SARS-CoV DNA vaccines in our study were able to induce high levels of the antigen-specific IgG antibody, the T cell proliferation, IFN-γ, DTH, and in vivo CTL responses. It suggests that the Th1 type of response is induced by the three structural genes of SARS-CoV in DNA vaccinations, which may lead to a desirable means when a therapeutic approach against SARS-CoV infection is considered.
Acknowledgments
This work was supported in part by the special research fund to B.W. provided by CAU. We thank Dr. Jane Q.L. Yu, Mr. Qinghong Zhu, and Mr. Zhonghuai He for their assistance of the work.
Footnotes
Abbreviations: SARS, severe acute respiratory syndrome; RT-PCR, reverse transcriptase-PCR; LB, Luria–Bertani; BSA, bovine serum albumin; OD, optical density; HPRT, hypoxanthine phosphoribosyl transferase; MTT, 3-(4,5-dimethylthiazol-2-yi)-2,5-diphenyltetrazolium; DTH, delayed-type hypersensitivity; CTL, cytotoxic T lymphocyte.
References
- 1.Li W., Moore M.J., Vasilieva N., Sui J., Wong S.K., Berne M.A., Somasundaran M., Sullivan J.L., Luzuriaga K., Greenough T.C., Choe H., Farzan M. Angiotensin-converting enzyme 2 is a functional receptor for the SARS coronavirus. Nature. 2003;426:450–454. doi: 10.1038/nature02145. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Weiss R.C., Scott F.W. Antibody-mediated enhancement of disease in feline infectious peritonitis: Comparisons with dengue hemorrhagic fever. Comp. Immunol. Microbiol. Infect. Dis. 1981;4:175–189. doi: 10.1016/0147-9571(81)90003-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Vennema H., de Groot R.J., Harbour D.A., Dalderup M., Gruffydd-Jones T., Horzinek M.C., Spaan W.J. Early death after feline infectious peritonitis virus challenge due to recombinant vaccinia virus immunization. J. Virol. 1990;64:1407–1409. doi: 10.1128/jvi.64.3.1407-1409.1990. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Charley B., Laude H. Induction of alpha interferon by transmissible gastroenteritis coronavirus: Role of transmembrane glycoprotein E1. J. Virol. 1988;62:8–11. doi: 10.1128/jvi.62.1.8-11.1988. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Laude H., Gelfi J., Lavenant L., Charley B. Single amino acid changes in the viral glycoprotein M affect induction of alpha interferon by the coronavirus transmissible gastroenteritis virus. J. Virol. 1992;66:743–749. doi: 10.1128/jvi.66.2.743-749.1992. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Loirat D., Lemonnier F.A., Michel M.-L. Multiepitopic HLA-A*0201-restricted immune response against hepatitis B surface antigen after DNA-based immunization. J. Immunol. 2000;165:4748–4755. doi: 10.4049/jimmunol.165.8.4748. [DOI] [PubMed] [Google Scholar]
- 7.Kiszka I., Kmieciak D., Gzyl J., Naito T., Bolesta E., Sieron A., Singh S.P., Srinivasan A., Trinchieri G., Kaneko Y., Kozbor D. Effect of the V3 loop deletion of envelope glycoprotein on cellular responses and protection against challenge with recombinant vaccinia virus expressing gp160 of primary human immunodeficiency virus type 1 isolates. J. Virol. 2002;76:4222–4232. doi: 10.1128/JVI.76.9.4222-4232.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Wilson J.A., Hart M.K. Protection from Ebola virus mediated by cytotoxic T lymphocytes specific for the viral nucleoprotein. J. Virol. 2001;75:2660–2664. doi: 10.1128/JVI.75.6.2660-2664.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Chen Z., Matsuo K., Asanuma H., Takahashi H., Iwasaki T., Suzuki Y., Aizawa C., Kurata T., Tamura S. Enhanced protection against a lethal influenza virus challenge by immunization with both hemagglutinin- and neuraminidase-expressing DNAs. Vaccine. 1999;17:653–659. doi: 10.1016/s0264-410x(98)00247-3. [DOI] [PubMed] [Google Scholar]
- 10.Chen Z., Yoshikawa T., Kadowaki S.-e., Hagiwara Y., Matsuo K., Asanuma H., Aizawa C., Kurata T., Tamura S.-i. Protection and antibody responses in different strains of mouse immunized with plasmid DNAs encoding influenza virus haemagglutinin, neuraminidase and nucleoprotein. J. Gen. Virol. 1999;80:2559–2564. doi: 10.1099/0022-1317-80-10-2559. [DOI] [PubMed] [Google Scholar]
- 11.Schadeck E.B., Partidos C.D., Fooks A.R., Obeid O.E., Wilkinson G.W., Stephenson J.R., Steward M.W. CTL epitopes identified with a defective recombinant adenovirus expressing measles virus nucleoprotein and evaluation of their protective capacity in mice. Virus Res. 1999;65:75–86. doi: 10.1016/s0168-1702(99)00103-3. [DOI] [PubMed] [Google Scholar]
- 12.Gurunathan S., Klinman D.M., Seder R.A., DNA vaccines: Immunology, application, and optimization* Annu. Rev. Immunol. 2000;18:927–974. doi: 10.1146/annurev.immunol.18.1.927. [DOI] [PubMed] [Google Scholar]
- 13.O’Hagan D., Singh M., Ugozzoli M., Wild C., Barnett S., Chen M., Schaefer M., Doe B., Otten G.R., Ulmer J.B. Induction of potent immune responses by cationic microparticles with adsorbed human immunodeficiency virus DNA vaccines. J. Virol. 2001;75:9037–9043. doi: 10.1128/JVI.75.19.9037-9043.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Schirmbeck R., Zheng X., Roggendorf M., Geissler M., Chisari F.V., Reimann J., Lu M. Targeting murine immune responses to selected T cell- or antibody-defined determinants of the hepatitis B surface antigen by plasmid DNA vaccines encoding chimeric antigen. J. Immunol. 2001;166:1405–1413. doi: 10.4049/jimmunol.166.2.1405. [DOI] [PubMed] [Google Scholar]
- 15.Webster R.G., Fynan E.F., Santoro J.C., Robinson H. Protection of ferrets against influenza challenge with a DNA vaccine to the haemagglutinin. Vaccine. 1994;12:1495–1498. doi: 10.1016/0264-410x(94)90071-x. [DOI] [PubMed] [Google Scholar]
- 16.Sambrook J., Fritsch E., Maniatis T. Cold Spring Harbor Laboratory Press; New York: 1989. Molecular Cloning: A Laboratory Manual. [Google Scholar]
- 17.Jin H., Li Y., Ma Z., Zhang F., Xie Q., Gu D., Wang B. Effect of chemical adjuvants on DNA vaccination. Vaccine. 2004;22:2925–2935. doi: 10.1016/j.vaccine.2003.12.026. [DOI] [PubMed] [Google Scholar]
- 18.Biragyn A., Belyakov I.M., Chow Y.-H., Dimitrov D.S., Berzofsky J.A., Kwak L.W. DNA vaccines encoding human immunodeficiency virus-1 glycoprotein 120 fusions with proinflammatory chemoattractants induce systemic and mucosal immune responses. Blood. 2002;100:1153–1159. doi: 10.1182/blood-2002-01-0086. [DOI] [PubMed] [Google Scholar]
- 19.Oehen S., Brduscha-Riem K. Differentiation of naive CTL to effector and memory CTL: Correlation of effector function with phenotype and cell division. J. Immunol. 1998;161:5338–5346. [PubMed] [Google Scholar]
- 20.Coles R.M., Mueller S.N., Heath W.R., Carbone F.R., Brooks A.G. Progression of armed CTL from draining lymph node to spleen shortly after localized infection with herpes simplex virus 1. J. Immunol. 2002;168:834–838. doi: 10.4049/jimmunol.168.2.834. [DOI] [PubMed] [Google Scholar]
- 21.Reiner S.L., Zheng S., Corry D.B., Locksley R.M. Constructing polycompetitor cDNAs for quantitative PCR. J. Immunol. Methods. 1993;165:37–46. doi: 10.1016/0022-1759(93)90104-f. [DOI] [PubMed] [Google Scholar]
- 22.Sauzet J.-P., Perlaza B.-L., Brahimi K., Daubersies P., Druilhe P. DNA immunization by Plasmodium falciparum liver-stage antigen3 induces protection against Plasmodium yoelii sporozoite challenge. Infect. Immun. 2001;69:1202–1206. doi: 10.1128/IAI.69.2.1202-1206.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Sedegah M., Weiss W., Sacci J.B., Jr., Charoenvit Y., Hedstrom R., Gowda K., Majam V.F., Tine J., Kumar S., Hobart P., Hoffman S.L. Improving protective immunity induced by DNA-based immunization: Priming with antigen and GM-CSF-encoding plasmid DNA and boosting with antigen-expressing recombinant poxvirus. J. Immunol. 2000;164:5905–5912. doi: 10.4049/jimmunol.164.11.5905. [DOI] [PubMed] [Google Scholar]
- 24.Ulmer J.B., Donnelly J.J., Parker S.E., Rhodes G.H., Felgner P.L., Dwarki V.J., Gromkowski S.H., Deck R.R., DeWitt C.M., Friedman A. Heterologous protection against influenza by injection of DNA encoding a viral protein. Science. 1993;259:1745–1749. doi: 10.1126/science.8456302. [DOI] [PubMed] [Google Scholar]
- 25.Robinson H.L., Torres C.A. DNA vaccines. Semin. Immunol. 1997;9:271–283. doi: 10.1006/smim.1997.0083. [DOI] [PubMed] [Google Scholar]
- 26.Dunachie S.J., Hill A.V.S. Prime-boost strategies for malaria vaccine development. J. Exp. Biol. 2003;206:3771–3779. doi: 10.1242/jeb.00642. [DOI] [PubMed] [Google Scholar]
- 27.McShane H., Brookes R., Gilbert S.C., Hill A.V.S. Enhanced immunogenicity of CD4+ T-cell responses and protective efficacy of a DNA-modified vaccinia virus ankara prime-boost vaccination regimen for murine tuberculosis. Infect. Immun. 2001;69:681–686. doi: 10.1128/IAI.69.2.681-686.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Van Epps H.L., Schmaljohn C.S., Ennis F.A. Human memory cytotoxic T-lymphocyte (CTL) responses to Hantaan virus infection: Identification of virus-specific and cross-reactive CD8+ CTL epitopes on nucleocapsid protein. J. Virol. 1999;73:5301–5308. doi: 10.1128/jvi.73.7.5301-5308.1999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Glansbeek H.L., Haagmans B.L., te Lintelo E.G., Egberink H.F., Duquesne V., Aubert A., Horzinek M.C., Rottier P.J.M. Adverse effects of feline IL-12 during DNA vaccination against feline infectious peritonitis virus. J. Gen. Virol. 2002;83:1–10. doi: 10.1099/0022-1317-83-1-1. [DOI] [PubMed] [Google Scholar]
- 30.Akahata W., Ido E., Akiyama H., Uesaka H., Enose Y., Horiuchi R., Kuwata T., Goto T., Takahashi H., Hayami M. DNA vaccination of macaques by a full-genome simian/human immunodeficiency virus type 1 plasmid chimera that produces non-infectious virus particles. J. Gen. Virol. 2003;84:2237–2244. doi: 10.1099/vir.0.19082-0. [DOI] [PubMed] [Google Scholar]
- 31.Zhu M.S., Pan Y., Chen H.Q., Shen Y., Wang X.C., Sun Y.J., Tao K.H. Induction of SARS-nucleoprotein-specific immune response by use of DNA vaccine. Immunol. Lett. 2004;92:237–243. doi: 10.1016/j.imlet.2004.01.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Kim T.W., Lee J.H., Hung C.-F., Peng S., Roden R., Wang M.-C., Viscidi R., Tsai Y.-C., He L., Chen P.-J., Boyd D.A.K., Wu T.C. Generation and characterization of DNA vaccines targeting the nucleocapsid protein of severe acute respiratory syndrome coronavirus. J. Virol. 2004;78:4638–4645. doi: 10.1128/JVI.78.9.4638-4645.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]


