Abstract
The 3C-like protease (3CLpro) of severe acute respiratory syndrome coronavirus (SARS-CoV) plays key roles in viral replication and is an attractive target for anti-SARS drug discovery. In this report, a fluorescence resonance energy transfer (FRET)-based method was developed to assess the proteolytic activity of SARS-CoV 3CLpro. Two internally quenched fluorogenic peptides, 1NC and 2NC, corresponding to the N-terminal and the C-terminal autocleavage sites of SARS-CoV 3CLpro, respectively, were used as substrates. SARS-CoV 3CLpro seemed to work more efficiently on 1NC than on 2NC in trans-cleavage assay. Mutational analysis demonstrated that the His41 residue, the N-terminal 7 amino acids, and the domain III of SARS-CoV 3CLpro were important for the enzymatic activity. Antibodies recognizing domain III could significantly inhibit the enzymatic activity of SARS-CoV 3CLpro. The effects of class-specific protease inhibitors on the trans-cleavage activity revealed that this enzyme worked more like a serine protease rather than the papain protease.
Keywords: SARS coronavirus, 3C-like protease, FRET-based proteolytic assay
Proteolytic processing of viral polyproteins is a key step in the replication cycle of many positive-stranded RNA viruses, and such processing in severe acute respiratory syndrome coronavirus (SARS-CoV) is performed by papain-like protease and 3C-like protease (3CLpro) [1]. The crystal structure of SARS-CoV 3CLpro was recently resolved by two groups [2], [3], which reveal great similarity to the structure of the 3CLpro of porcine transmissible gastroenteritis virus (TGEV) and of human coronavirus (strain 229E), all of which form dimers [2], [4]. Each monomer of the enzyme contains three structural domains: the first two domains form a chymotrypsin fold containing the catalytic site comprised of His41 and Cys145, and the third domain is a globular α-helical structure which is linked to domain II by a long loop [2], [3], [4] The extra helical domain III is unique for coronavirus 3CLpro because the picornavirus 3C proteases only contain the chymotrypsin fold, but have no extra domain [5], [6], [7]. The role of domain III in SARS-CoV 3CLpro is not quite clear but seems to be involved in dimer formation [3], [4], [8]. Due to its functional importance, SARS-CoV 3CLpro becomes an attractive target for structure-based drug designs against SARS [9], [10], [11], [12].
To fulfill the needs of efficient and robust high-throughput screening of the anti-protease drugs, a convenient assay system for measuring the enzymatic activity is needed. In this study, we have established a fluorescence resonance energy transfer (FRET)-based method to assess the proteolytic activity of SARS-3CLpro. Two internally quenched fluorogenic (IQF) 11-mer peptides with N-terminus conjugated with an ortho-aminobenzoic acid (Abz) and C-terminus conjugated with a lysine-2,4-dinitrophenylamide (Lys-DNP) were synthesized and used as substrates. Upon cleavage of the substrate, the fluorophore was separated from the quenching group, generating a fluorescence signal [13], [14]. The enzymatic reactions were monitored in real time. Using the FRET-based method, we have characterized the enzymatic activities of a catalytic site-mutated 3CLpro (H41A), a N-terminus-deleted 3CLpro (Δ1–7), and a domain III-deleted 3CLpro (ΔIII). We also used the system to demonstrate that some protease inhibitors and monoclonal antibodies recognizing domain III had inhibitory effects on the proteolytic activity of SARS-3CLpro. The FRET-based real time assay provides an ideal platform for exploring the inhibitors against SARS-CoV 3CLpro and is a convenient system to study the kinetics of proteolytic reactions.
Materials and methods
Construction of pET28b-3CL pro . The cDNA corresponding to SARS-CoV (strain TW1) nt 9985–10,902 coding for the 3CLpro protein was amplified by PCR from the reverse transcriptional mixture provided by Dr. P.J. Chen (Graduate Institute of Clinical Medicine, National Taiwan University, Taipei, Taiwan). The PCR product was cloned into the pET28b vector (Novagen) and the resulting plasmid, designated as pET28b-3CLpro, was verified by sequencing.
Expression and purification of the 3CL pro protein. The plasmid transformed Escherichia coli cells were grown at 37 °C until A 600 reached 0.6–0.8 and then induced to express the recombinant protein with 0.5 mM isopropyl-1-thio-β-d-galactopyranoside (IPTG) for 3 h. The cells harvested were disrupted in buffer A (20 mM Tris–HCl, pH 8.0, 500 mM NaCl, and 5 mM imidazole) and cell lysate was applied to a nickel affinity column (Amersham Pharmacia Biotech AB, Uppsala, Sweden). After wash, the 3CLpro protein was eluted with a 12–100% linear gradient of buffer B (20 mM Tris–HCl, pH 8.0, 500 mM NaCl, and 250 mM imidazole) in buffer A. The fractions containing 3CLpro were pooled and dialyzed against the storage buffer (20 mM Tris–HCl, pH 8.0, 50 mM NaCl, and 1 mM EDTA). The sample was concentrated and stored at −80 °C in storage buffer containing 50% glycerol. The proteins were more than 95% pure as assessed by SDS–PAGE.
Deletion and point mutation. Using pET28b-3CLpro as a template, three mutant clones (H41A, Δ1–7, and ΔIII) were generated by one-primer PCR method as described previously [15]. The single primer 5′-CACAGTATACTGTCCAAGAGCTGTCATTTGCACAGCAG-3′ was used for the substitution of His41 with an alanine, primer 5′-CAGCAAATGGGTCGGGATCCCTTCCCGTCAGGCAAAGTTGAA-3′ was used for the deletion of the N-terminal 1–7 amino acids of 3CLpro, and primer 5′-GCAGGTACAGACACAACCATAGCGGCCGCACTCGAGCACCAC-3′ was used for the deletion of the C-terminal 201–306 amino acids of 3CLpro. All the mutant clones were verified by sequencing.
Protease activity assay using the IQF peptide substrates. Two synthetic IQF peptides, 1NC (Abz-Thr-Ser-Ala-Val-Leu-Gln↓Ser-Gly-Phe-Arg-Lys-DNP) and 2NC (Abz-Ser-Gly-Val-Thr-Phe-Gln↓Gly-Lys-Phe-Lys-Lys-DNP) (Genemed Synthesis, South San Francisco, CA), were used in this study (↓ indicates the cleavage site). The reaction mixture (30 μl) contained 5 mM Hepes, pH 7.3, 1 mM DTT, 25 mM NaCl, 0.025% Triton X-100, 100 μM peptide substrate, and 6 μM 3CLpro. Reactions were performed in a 384-well black microtiter plate incubated at 32 °C. After the enzyme was added, the increase of fluorescence was recorded continuously using a Labsystems fluorometer (Fluoroskan Ascent) with a plate reader accessory with excitation and emission wavelengths of 320 and 420 nm, respectively. The kinetic parameters were determined by Lineweaver–Burk plot using 6 μM enzyme and 25–400 μM peptide substrates.
Analysis of peptide cleavage by HPLC. The cleavage assays were carried out in a reaction mixture as described above for 3 h at 32 °C and then stopped by the addition of 1% formic acid. The reaction products were resolved on a C18 analytic column (4.6 mm × 250 mm, Beckman, Fullerton, CA) using a 0–60% linear gradient of 80% acetonitrile in 0.06% trifluoroacetic acid, at 1 ml min−1 flow rate. The elution was monitored at an absorbance wavelength of 220 nm.
Inhibition assays. The inhibitory activities of protease inhibitors or antibodies toward 3CLpro were measured in a reaction mixture lacking DTT in the presence of various concentrations (0–400 μM) of the inhibitors or different amounts (0–5 μl) of antiserum or monoclonal antibody ascites. Two cysteine protease inhibitors, N-ethylmaleimide (NEM) and E-64, and two serine protease inhibitors, Nα-p-Tosyl-l-lysine chloromethyl ketone (TLCK) and N-p-Tosyl-l-phenylalanine chloromethyl ketone (TPCK) (Sigma), were used in this study. The antibodies used were rabbit anti-SARS-CoV 3CLpro antiserum and three mouse anti-3CLpro monoclonal antibodies (our unpublished data).
Results and discussion
Expression and purification of SARS-CoV 3CLpro and its mutants
To establish a FRET-based assay, the wild-type SARS-CoV 3CLpro was first expressed in E. coli and purified to nearly homogeneity (Fig. 1 A). Meanwhile, three mutant proteins, the 3CLpro having the His41 substituted with an Ala (H41A), a deletion of the N-terminal 1–7 amino acid residues (Δ1–7), and a deletion of domain III (from aa 201 to 306) (ΔIII), were expressed as well to assess the roles of the residue and the domains in the proteolytic activity of SARS-CoV 3CLpro. Due to the extra sequences derived from pET28b, the full-length SARS-CoV 3CLpro was expressed as a 39.5 kDa protein containing His6-tag at both the N-terminus and the C-terminus. However, it was realized that the C-terminal sequences of SARS-CoV 3CLpro, VTFQ, while in connection with the 11 amino acid residues of vector pET28b, AAALEHHHHHH, could actually form a consensus cutting site for SARS-CoV 3CLpro, VTFQ↓AAA. The resulting protein would then be 37.4 kDa instead of 39.5 kDa (Fig. 1A). Thus, the results clearly suggest that the SARS-CoV 3CLpro expressed possesses cis-cleavage activity.
Fig. 1.

Expression and purification of recombinant SARS-CoV 3CLpro and its mutants. The recombinant proteins purified by Ni-affinity column were analyzed by SDS–PAGE on a 15% polyacrylamide gel and stained with Coomassie brilliant blue (A), or reacted with rabbit anti-SARS-CoV 3CLpro polyclonal antiserum (B), or with a mouse monoclonal antibody, mAb 4, against SARS-CoV 3CLpro (C). Molecular size markers (in kDa) are indicated on the left.
On the contrary, all the other three mutant proteins apparently did not possess proteolytic activities, their sizes were the same as anticipated, i.e., 39.5 kDa (H41A), 38.7 kDa (Δ1–7), and 27.7 kDa (ΔIII). All the 3CLpro and mutant proteins were recognized by a rabbit anti-SARS-CoV 3CLpro antiserum (Fig. 1B). However, all but ΔIII were recognized by a mouse anti-SARS-CoV 3CLpro monoclonal antibody (mAb 4) (Fig. 1C), suggesting that this monoclonal antibody might recognize the epitope residing in domain III.
Enzymatic activity of SARS-CoV 3CLpro and its mutants
Two IQF peptides, 1NC and 2NC, were used for in vitro trans-cleavage assay. The sequences of these correspond to the N-terminal and the C-terminal autocleavage sites of SARS-CoV 3CLpro, respectively. The enzymatic activities of SARS-CoV 3CLpro toward 1NC and 2NC were analyzed by direct monitoring of the increase of fluorescence in real time (Fig. 2 A). In parallel, the cleaved products obtained at the 3-h reaction were analyzed by reverse-phase HPLC (Fig. 2B). Both FRET-based and HPLC-based assays indicated that the wild-type SARS-CoV 3CLpro had better cleavage efficiency on 1NC than on 2NC. According to the HPLC data, more than 90% of 1NC were cleaved by SARS-CoV 3CLpro in 3 h, whereas only about 50% of 2NC were cleaved (Fig. 2B). Kinetic parameters were calculated based on the FRET-based assays. The K m value for 1NC was 145.69 μM, the K cat was 0.604 min−1, and the K cat/K m was 4.143 mM−1 min−1. For 2NC, the K m was 252.47 μM, the K cat was 0.303 min−1, and the K cat/K m was 1.20 mM−1 min−1.
Fig. 2.
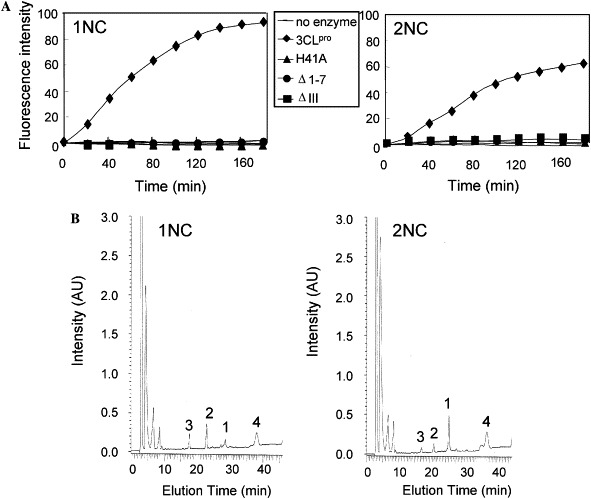
Protease activity assays. (A). FRET-based method. The reaction contained 100 μM IQF substrate (1NC or 2NC) and 6 μM the wild-type SARS-CoV 3CLpro or the mutant proteins, and carried out for 3 h as described under Materials and methods. The fluorescence was monitored in real time by a plate reader. (B) HPLC-based method. The reaction was carried out for 3 h and the cleaved products were analyzed by HPLC as described under Materials and methods. Peak 1 are the original peptide. Peaks 2 and 3 are the cleaved N-terminal and C-terminal fragments, respectively. Peak 4 are impurities in the original substrate solution.
On the contrary, the three mutants had extremely low proteolytic activities on both peptide substrates, as measured by FRET-based assays (Fig. 2A). The results indicate that the N-terminal 7 amino acids and the domain III are as important as the catalytic His41 residue in the proteolytic activity of SARS-CoV 3CLpro.
Inhibitory effects of protease inhibitors on SARS-CoV 3CLpro
SARS-CoV 3CLpro belongs to the cysteine protease family with a chymotrypsin fold, thus it is interesting to know whether it is inhibited by serine protease inhibitors or by cysteine protease inhibitors. The inhibitory effects of two serine protease inhibitors, TLCK and TPCK, and two cysteine protease inhibitors, NEM and E-64, were tested in this FRET-based assay. Data shown in Fig. 3 demonstrate that both serine protease inhibitors reduced the enzymatic activity of SARS-CoV 3CLpro in a dose-dependent manner. However, the results for the two cysteine protease inhibitors were inconsistent: while NEM had significant inhibitory effects on SARS-CoV 3CLpro, E-64 did not inhibit it at all. The data support the idea that SARS-CoV 3CLpro works following a general base mechanism like the serine proteases, but not following a thiolate–imidazolium ion pair catalytic mechanism like the papain protease [16]. However, NEM probably inhibits the proteolytic activity of SARS-CoV 3CLpro via directly blocking the catalytic Cys145 residue, due to its ability to bind stoichiometrically to the sulfhydryl (SH) group of cysteine in the cysteine protease family.
Fig. 3.
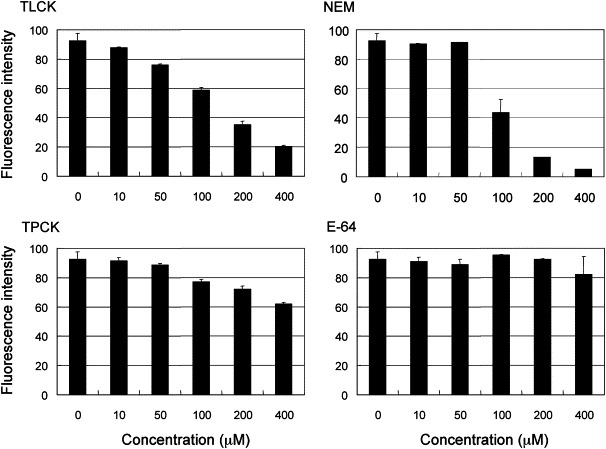
Effects of protease inhibitors on the activity of SARS-CoV 3CLpro. Two serine protease inhibitors, TLCK and TPCK, and two cysteine protease inhibitors, NEM and E-64, ranging from 0 to 400 μM were added to the reaction mixture containing the 1NC substrate. Fluorescence was monitored continuously. The data shown are the (means ± SD) fluorescence intensity at the 3-h reaction.
Inhibition of SARS-CoV 3CLpro activity by monoclonal antibodies that recognized domain III
Previously we have generated three monoclonal antibodies, mAb 3, mAb 4, and mAb 6, that could recognize full-length SARS-CoV 3CLpro but not the protein lacking domain III (Fig. 1C and data not shown). We interpreted that these monoclonal antibodies probably recognized the epitopes residing in domain III. Therefore, we were interested to test whether they could inhibit the proteolytic activity of SARS-CoV 3CLpro in the trans-cleavage assay. A rabbit anti-SARS-CoV 3CLpro polyclonal antiserum was also tested. Data shown in Fig. 4 reveal that all three monoclonal antibodies could inhibit the enzymatic activity, whereas the polyclonal antiserum did not have the inhibitory activity. To our knowledge, this is the first report demonstrating that the enzymatic activity of SARS-CoV 3CLpro could be inhibited by antibodies. The results further confirmed the pivotal roles of domain III in the proteolytic activity of SARS-CoV 3CLpro.
Fig. 4.
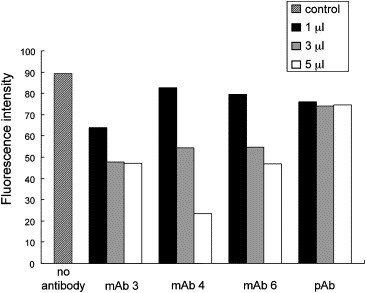
Effects of antibodies on the activity of SARS-CoV 3CLpro. Different amounts (1–5μl) of mouse anti-SARS-CoV 3CLpro monoclonal antibody (mAb 3, mAb 4, or mAb 6) or rabbit anti-SARS-CoV 3CLpro polyclonal antiserum (pAb) were added to the proteolytic reaction mixture containing the 1NC substrate and the fluorescence was monitored continuously. The data shown are the fluorescence intensity at the 3-h reaction.
In summary, a FRET-based method for detecting the proteolytic activity of SARS-CoV 3CLpro was evaluated in this study. The kinetic studies showed that the N-terminal autocleavage site of SARS-CoV 3CLpro was a better substrate than the C-terminal autocleavage site in the trans-cleavage assay. Recently, Fan et al. [17] have systematically analyzed the substrate specificity of SARS-CoV 3CLpro and found that the P2 position of the substrate required a large hydrophobic residue for the hydrophobic interaction between the P2 position and the S2 pocket of the enzyme, as indicated by the crystal structure [2], [4]. Although Phe, Val, and Met are tolerant at P2 for SARS-CoV 3CLpro [18], the mutation of Leu-P2 to other hydrophobic residues decreases the reaction activity [17]. Since 1NC has a Leu at the P2 position and 2NC has a Phe, this may explain why 2NC was cleaved less efficiently than 1NC by SARS-CoV 3CLpro.
Our deletion data (Fig. 2) and antibody blocking data (Fig. 4) indicate that both the N-terminal 7 amino acids and the domain III are important for the enzymatic activity of SARS-CoV 3CLpro. Similar conclusion has also been reached for the 3CLpro of TGEV [4]. Previous crystal structure data have attributed this importance to the dimerization function mediated by these two domains [3], [4], [8], [12]. However, a recent study by Chen et al. reported that the N-terminal seven amino acids of SARS-CoV 3CLpro were not required for enzyme dimerization. Instead, they proposed that the N-terminus-deleted mutant had a conformation that reduced the size of the binding pocket, thus creating unfavorable conditions for substrate binding [19]. The discrepancy about the roles of the N-terminal seven amino acid residues is not completely resolved at this moment and it needs to be further investigated by different analytical methods. However, no matter what function the N-terminal domain may play, it is definitely important for the proteolytic activity of SARS-CoV 3CLpro, as demonstrated by our activity assay.
Acknowledgment
This work was supported by Grant NSC 92-2751-B-002-004-Y from National Science Council of the Republic of China.
References
- 1.Thiel V., Ivanov K.A., Putics A., Hertzig T., Schelle B., Bayer S., Weissbrich B., Snijder E.J., Rabenau H., Doerr H.W., Gorbalenya A.E., Ziebuhr J. Mechanisms and enzymes involved in SARS coronavirus genome expression. J. Gen. Virol. 2003;84:2305–2315. doi: 10.1099/vir.0.19424-0. [DOI] [PubMed] [Google Scholar]
- 2.Anand K., Ziebuhr J., Wadhwani P., Mesters J.R., Hilgenfeld R. Coronavirus main proteinase (3CLpro) structure: basis for design of anti-SARS drugs. Science. 2003;300:1763–1767. doi: 10.1126/science.1085658. [DOI] [PubMed] [Google Scholar]
- 3.Yang H., Yang M., Ding Y., Liu Y., Lou Z., Zhou Z., Sun L., Mo L., Ye S., Pang H., Gao G.F., Anand K., Bartlam M., Hilgenfeld R., Rao Z. The crystal structures of severe acute respiratory syndrome virus main protease and its complex with an inhibitor. Proc. Natl. Acad. Sci. USA. 2003;100:13190–13195. doi: 10.1073/pnas.1835675100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Anand K., Palm G.J., Mesters J.R., Siddell S.G., Ziebuhr J., Hilgenfeld R. Structure of coronavirus main proteinase reveals combination of a chymotrypsin fold with an extra alpha-helical domain. EMBO J. 2002;21:3213–3224. doi: 10.1093/emboj/cdf327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Bergmann E.M., Mosimann S.C., Chernaia M.M., Malcolm B.A., James M.N. The refined crystal structure of the 3C gene product from hepatitis A virus: specific proteinase activity and RNA recognition. J. Virol. 1997;71:2436–2448. doi: 10.1128/jvi.71.3.2436-2448.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Matthews D.A., Smith W.W., Ferre R.A., Condon B., Budahazi G., Sisson W., Villafranca J.E., Janson C.A., McElroy H.E., Gribskov C.L. Structure of human rhinovirus 3C protease reveals a trypsin-like polypeptide fold, RNA-binding site, and means for cleaving precursor polyprotein. Cell. 1994;77:761–771. doi: 10.1016/0092-8674(94)90059-0. [DOI] [PubMed] [Google Scholar]
- 7.Seipelt J., Guarne A., Bergmann E., James M., Sommergruber W., Fita I., Skern T. The structures of picornaviral proteinases. Virus Res. 1999;62:159–168. doi: 10.1016/s0168-1702(99)00043-x. [DOI] [PubMed] [Google Scholar]
- 8.Shi J., Wei Z., Song J. Dissection study on the severe acute respiratory syndrome 3C-like protease reveals the critical role of the extra domain in dimerization of the enzyme: defining the extra domain as a new target for design of highly specific protease inhibitors. J. Biol. Chem. 2004;279:24765–24773. doi: 10.1074/jbc.M311744200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Holmes K.V. SARS coronavirus: a new challenge for prevention and therapy. J. Clin. Invest. 2003;111:1605–1609. doi: 10.1172/JCI18819. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Blanchard J.E., Elowe N.H., Huitema C., Fortin P.D., Cechetto J.D., Eltis L.D., Brown E.D. High-throughput screening identifies inhibitors of the SARS coronavirus main proteinase. Chem. Biol. 2004;11:1445–1453. doi: 10.1016/j.chembiol.2004.08.011. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wu C.Y., Jan J.T., Ma S.H., Kuo C.J., Juan H.F., Cheng Y.S., Hsu H.H., Huang H.C., Wu D., Brik A., Liang F.S., Liu R.S., Fang J.M., Chen S.T., Liang P.H., Wong C.H. Small molecules targeting severe acute respiratory syndrome human coronavirus. Proc. Natl. Acad. Sci. USA. 2004;101:10012–10017. doi: 10.1073/pnas.0403596101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Bacha U., Barrila J., Velazquez-Campoy A., Leavitt S.A., Freire E. Identification of novel inhibitors of the SARS coronavirus main protease 3CLpro. Biochemistry. 2004;43:4906–4912. doi: 10.1021/bi0361766. [DOI] [PubMed] [Google Scholar]
- 13.Jean F., Basak A., DiMaio J., Seidah N.G., Lazure C. An internally quenched fluorogenic substrate of prohormone convertase 1 and furin leads to a potent prohormone convertase inhibitor. Biochem. J. 1995;307(Pt. 3):689–695. doi: 10.1042/bj3070689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Kuang W.F., Lin Y.C., Jean F., Huang Y.W., Tai C.L., Chen D.S., Chen P.J., Hwang L.H. Hepatitis C virus NS3 RNA helicase activity is modulated by the two domains of NS3 and NS4A. Biochem. Biophys. Res. Commun. 2004;317:211–217. doi: 10.1016/j.bbrc.2004.03.032. [DOI] [PubMed] [Google Scholar]
- 15.Makarova O., Kamberov E., Margolis B. Generation of deletion and point mutations with one primer in a single cloning step. Biotechniques. 2000;29:970–972. doi: 10.2144/00295bm08. [DOI] [PubMed] [Google Scholar]
- 16.Huang C., Wei P., Fan K., Liu Y., Lai L. 3C-like proteinase from SARS coronavirus catalyzes substrate hydrolysis by a general base mechanism. Biochemistry. 2004;43:4568–4574. doi: 10.1021/bi036022q. [DOI] [PubMed] [Google Scholar]
- 17.Fan K., Ma L., Han X., Liang H., Wei P., Liu Y., Lai L. The substrate specificity of SARS coronavirus 3C-like proteinase. Biochem. Biophys. Res. Commun. 2005;329:934–940. doi: 10.1016/j.bbrc.2005.02.061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Fan K., Wei P., Feng Q., Chen S., Huang C., Ma L., Lai B., Pei J., Liu Y., Chen J., Lai L. Biosynthesis, purification, and substrate specificity of severe acute respiratory syndrome coronavirus 3C-like proteinase. J. Biol. Chem. 2004;279:1637–1642. doi: 10.1074/jbc.M310875200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chen S., Chen L., Tan J., Chen J., Du L., Sun T., Shen J., Chen K., Jiang H., Shen X. Severe acute respiratory syndrome coronavirus 3C-like proteinase N terminus is indispensable for proteolytic activity but not for enzyme dimerization. Biochemical and thermodynamic investigation in conjunction with molecular dynamics simulations. J. Biol. Chem. 2005;280:164–173. doi: 10.1074/jbc.M408211200. [DOI] [PMC free article] [PubMed] [Google Scholar]


