Abstract
Infectious diseases are a leading cause of mortality worldwide, with viruses and bacteria in particular having enormous impacts on global healthcare. One major challenge in combatting such diseases is a lack of effective drugs or specific treatments. In addition, drug resistance to currently available therapeutics and adverse effects caused by long-term overuse are both serious public health issues. A promising treatment strategy is to employ cell membrane mimics as decoys to trap and to detain the pathogens. In this Perspective, we briefly review the infection mechanisms adopted by different pathogens at the cellular membrane interface and highlight the applications of cell membrane mimicking nanodecoys for systemic protection against infectious diseases. We also discuss the implication of nanodecoy–pathogen complexes in the development of vaccines. We anticipate this Perspective will provide new insights on design and development of advanced materials against emerging infectious diseases.
Keywords: cell membrane, biomimetic nanoparticle, infectious disease, bacteria, virus
Graphical Abstract
Infectious diseases have recently emerged as a serious global public health concern underscored by the rapidly increasing number of drug-resistant strains of existing pathogens and the emergence of new pathogens.1,2 Collectively, these pathogen infections cause millions of deaths and, thus, have a huge impact on global healthcare and socioeconomical development.3 Very recently, the World Health Organization (WHO) declared the outbreak of a novel severe acute respiratory syndromes coronavirus (SARS-nCoV-2) in China a Public Health Emergency of International Concern.4 The new disease, first made public by Wuhan, China at the end of December 2019, has already spread to 48 countries; the number of people in China infected with SARS-nCoV-2 reached 82,000 on 27 February 2020 and more than 2800 of them have died.5,6 The primary challenge in fighting such emerging diseases is the lack of effective drugs or specific treatments.7,8 In addition, adverse side effects and increasing drug resistances owing to long-term overuse are also serious issues.9,10 New and effective prevention and treatment strategies need to be developed urgently.
Pathogens, or disease-causing infection agents, are all over the world in which we live.11,12 Although these microbes can come in different forms, they all have one thing in common: to cause infection, they must invade a host.13,14 Cell membrane is one of the major barriers that pathogens need to conquer.13 For example, a virus first needs to attach itself to the cell membrane and then injects its genetic material into the host cell prior to replication.15 Such structural similarity provides the opportunity to develop new therapeutic platforms for broad-spectrum anti-infection applications. Recent efforts have shown multiple anti-infection applications by utilizing cell-membrane-based nanostructures.16–19 These novel formulations take advantage of the fact that despite their different infection modes, pathogens must interact with host cell membranes, at some point. Furthermore, biomimetic nanoparticles have many inherent properties that can benefit anti-infection applications, including long systemic circulation and multivalent pathogen interaction.20–22
In light of these recent advances, in this Perspective, we review the evolution of cellular-membrane-based nanodecoys with a specific focus on infectious-disease-related studies. We first discuss multiple pathogen–membrane interactions to elucidate the fundamental action mechanism of such biomimetic nanodecoys against pathogen infections. We then provide an overview of the design and preparation of cell-membrane-mimicking formulations as nanodecoys to trap and to detain pathogens. Finally, we discuss the implication of utilizing nanodecoy–pathogen complexes for vaccination. We believe this Perspective can provide new insights on the development of new and effective prevention and treatment strategies for infectious diseases.
PATHOGEN–MEMBRANE INTERACTIONS
According to their morphology and structure, pathogens can generally be divided into four main categories: viruses, bacteria, fungi, and parasites.23 Despite the diversity of pathogens, one similarity they share is how they cause infectious disease: these pathogens usually invade the host by interacting with the host cell membrane first.13 Here, we briefly review the interactions between different pathogens and hosts at the cell-membrane interface.
Viruses are small infectious agents that do not grow by cell division, but instead use the host metabolism to complete self-replication. Viruses can infect all types of life forms on earth, from animals and plants to microorganisms. The modes of viral infection and replication differ greatly between species but attachment is the basic stage, in which viral capsid proteins specifically bind to the receptors on the host cellular membrane.8 For example, HIV infects human leukocytes through the specific interaction between its surface protein gp120 and the CD4 receptor and the CCR5 or CXCR4 coreceptors on CD4+ T cells.24
Bacteria are one kind of biological cell that compose a wide range of prokaryotic microorganisms. The relationship between bacteria and humans is complex. Sometimes bacteria offer us a helping hand. For example, probiotics help us digest. In other cases, bacteria are subversive, causing infectious diseases like methicillin-resistant Staphylococcus aureus (MRSA).25 Typically, pathogenic bacteria express and/or release a large domain of molecules that target the host membrane and facilitate many individual host responses.16 The molecular mechanisms of the interactions between bacteria and hosts is different across different species. For example, many Gram-positive pathogens secrete pore-forming toxins that can form pores in the cell membranes and lead to cell lysis, while Gram-negative pathogens secrete endotoxins that can activate macrophages to produce inflammatory cytokines.26
A fungus is any member of eukaryotic organisms, including mushrooms and microorganisms such as molds and yeasts. According to their different structures, sizes, surface properties, and ability to secrete pathogenicity factors, fungi can attack host cells in different ways, including membrane disruption by mechanical forces and membrane remodeling by fungal lipases.27 In addition, Moyes et al. discovered that Candida albicans is able to produce a peptide toxin that can directly damage host membranes and activate host immunity.28
Parasitism is a relationship between different species in evolutionary biology, where one life form, the parasite, lives on or in another life form, the host. Although the parasite may cause the host some harm, the host is adapted structurally to live with the parasite. For example, to complete their life cycle in the blood circulation, the malaria parasite produces merozoite that can attach to and enter into red blood cells (RBCs) where the parasite asexually divides to form many copies of itself. The parasite copy then exits the infected RBC and infects more RBCs.29
TRAPPING AND DETENTION OF PATHOGENS WITH NANODECOYS
Due to the close interactions between pathogens and cell membranes, various cell-membrane-mimicking nanostructures have been employed as decoys to mitigate pathogen infection.16,17,26,30 In the following section, we review previous infectious disease research involving cell membrane mimics, with a special emphasis on the platforms developed for pathogen trap and detention.
Liposomes
Liposomes are artificial vesicles composed of one or more hydrophobic phospholipid bilayers; they have been used extensively for systemic delivery of therapeutic compounds. Because liposomes imitate the capabilities of cell membranes in several ways, they have been widely utilized as model membranes to study the infection mechanisms of many pathogens at the biomembrane interface (Figure 1a).31 Research on the interaction between pathogens and liposomes can be traced back to the 1960s, when Cornatzer et al. found that streptolysin-S is able to regulate the cationic permeability of multilayer liposomes.32 Following the early mechanistic studies, researchers further applied liposomes as pathogen substrates in the emerging infectious disease area. For example, liposome microarrays can be used for ultrasensitive detection of various types of pathogens including viruses, bacteria, and fungi.33 These cell membrane mimics, consisting of synthetical lipids along with functional ligands, promote the formation of pathogen–liposome complexes that effectively improve the detection sensitivity in various electrochemical and immunological assays. Another example that utilizes the liposome–pathogen interaction is direct inhibition of the pathogen infection with liposomes. The pathogens were trapped and detained by cell-membrane-mimicking liposomes, thereby preventing them from attacking their cellular targets.34 In viral and bacterial infection models, treatment with functionalized liposomes was demonstrated to improve subject survival as well as to reduce the overall infection.34,35 Engineered liposomes have also been used as a secondary therapeutic to supplement traditional anti-infective drugs. Systemic administration of nanoscale liposomes along with penicillin effectively protected animals from S. pneumoniae and S. aureus-caused septicemia.36 Moreover, researchers have also exploited the liposome–pathogen interaction for infection-triggered drug release. Motivated by targeting therapeutic drugs to the specific site, pore-forming toxins are able to bore holes on the synthetic liposomal vehicles, resulting in the selective release of vancomycin at the infection sites.37 Surface engineering of liposomes with specific molecules may further promote the applicability of synthetic liposomes against infectious diseases. Magee et al., for instance, successfully modified liposomes with antiviral antibodies for effective protection against coxsackie A-21 virus infection.38 With the rapid developments of biotechnology and nanotechnology, the physical and chemical properties of phospholipid bilayers may be tuned finely to improve their efficacy in various anti-infection applications.
Figure 1.
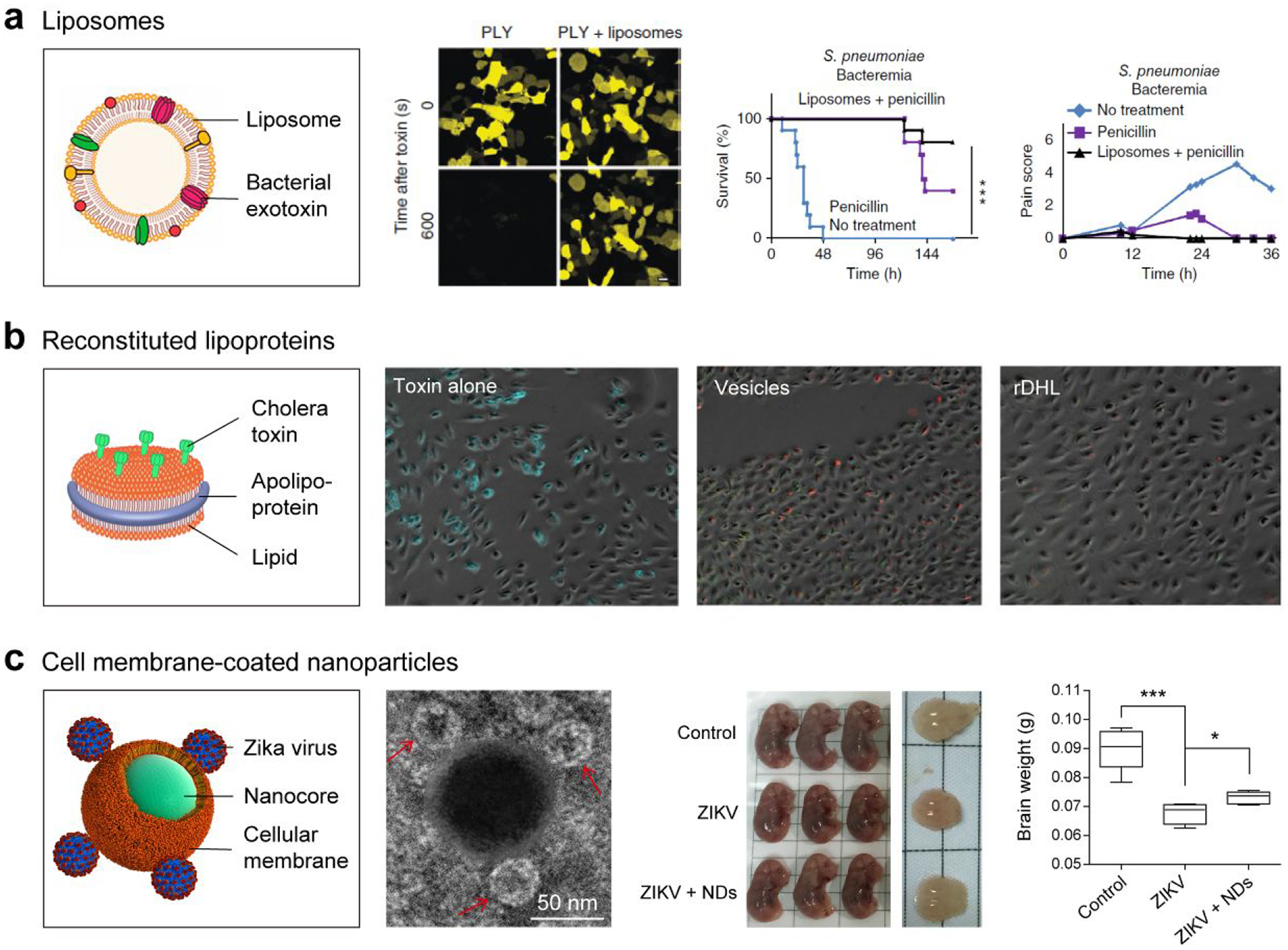
Trapping and detention of pathogens/toxins with nanodecoys. a) Artificial liposomes sequester bacterial exotoxins and rescue mice from septicemia. Reprinted with permission from refs 16 and 34. Copyrights 2015 Elsevier B.V. and 2014 Nature Publishing Group, respectively. b) Modified reconstituted high-density lipoproteins (rHDL) effectively inhibit the cholera toxin attachment to epithelial cells and prevent toxin-associated negative outcomes. Reprinted with permission from ref 41. Copyright 2010 American Society for Biochemistry & Molecular Biology. c) Cellular-membrane-coated nanodecoys (NDs) effectively trap Zika virus (ZIKV) and mitigate the ZIKV-caused fetal microcephaly in vivo. Reprinted from ref 17. Copyright 2019 American Chemical Society.
Reconstituted Lipoproteins
In addition to liposomes, reconstituted lipoproteins have also been extensively employed in the development of anti-infection decoys. Lipoproteins are inherently present in the human body as a disk-like patch of phospholipids bound by apolipoproteins. By using their constructing ingredients, these proteins can be controllably reconstituted.39 This method promises the controllable functionalization of lipoprotein components at the molecular level, opening the door for any lipid-compatible ingredients to be incorporated into the lipoprotein nanostructure. Proton pumps, signaling proteins, and membrane-associating enzymes have all been integrated into the reconstituted lipoproteins.39
Engineered lipoproteins have been demonstrated to interact frequently with pathogens, leading to the effective suppression of pathogen infectivity. This inhibition can be ascribed to the lipoproteins’ microdomains, which are also rich in pathogen/toxin receptors such as sphingomyelin and cholesterol.40 Furthermore, to develop anti-infection nanostructures for clinical use, Bricarello et al. prepared reconstituted high-density lipoprotein (rHDL) to integrate ganglioside GM1 to improve affinity to certain pathogenic molecules (Figure 1b).41 The modified rHDL effectively inhibited the cholera toxin attachment to epithelial cells and prevented toxin-associated negative outcomes. Although promising, further work along the lines of how to alleviate the toxicity of such nanostructures and how to improve the rHDL–host interaction is necessary before successful translation of rHDL as an anti-infection candidate.
Cell Membrane Nanostructures
Cell-membrane-derived nanoparticles present an attractive platform for anti-infection applications because pathogens have evolved to utilize abundant molecules on the cell membrane.42 Although synthetic liposomes can be straightforwardly functionalized with specific ligands, it has proven difficult to replicate or to mimic the complicated protein compositions and functions in natural cell membranes. Natural cell-membrane-derived vesicles possess a unique advantage of maintaining most of the natural compositions and functions of source cell membranes, thereby enabling each platform to exhibit properties that would otherwise be hard to accomplish.43,44 Moreover, emerging genetic editing technology can be further employed to amend the antigen profile of natural cellular membranes to fit different purposes.18
To develop biomimetic anti-infection nanodecoys, de Carvalho et al. demonstrated that CD4+ T cell-derived exosomes could efficiently suppress HIV-1 infection in vitro.45 These exosomes exhibited the same completed membrane protein composition as their source CD4+ T cells, ensuring that they attached effectively to HIV-1, thereby preventing the HIV-1 from binding to and entering healthy CD4+ T cells. In addition, exosomes released from certain host cells have also been utilized to suppress bacterial and parasitic invasion of specific host cells as well.46
Because many exotoxins secreted by bacteria and fungi are known for their unique capability to lyse erythrocytes (red blood cells, RBCs), RBC-derived nanovesicles have been widely employed as a model to examine the kinetics of these exotoxins.47 After removal of the intracellular contents, these cell-membrane-derived nanovesicles inherit the lipids, glycans, and proteins from their source cells, enabling the pathogen–nanoparticle interaction to occur in a natural manner.
In developing a biomimetic formulation that traps exotoxins secreted by bacteria, Hu et al. demonstrated that poly(lactic-co-glycolic acid) (PLGA) nanoparticles camouflaged with RBC membranes are able to serve as toxin “nanosponges,” absorbing pore-forming toxins and diverting them away from their intended targets.26 Moreover, Rao et al. showed that mosquito host cell-membrane-wrapped nanodecoys effectively trap Zika virus (ZIKV) and divert it away from its healthy cellular targets (Figure 1c).17 In mouse models, the research team further demonstrated that these nanodecoys successfully mitigate the ZIKV-caused inflammatory responses and fetal microcephaly. Compared to cell-membrane-derived vesicles, the cell-membrane-coated nanostructures are not prone to membrane fusion and therefore do not transfer detained pathogens to host cells.48,49 In addition, due to the core–shell structure, the lipid membrane shell is stabilized by the nanoparticle core, benefiting in vivo applications.20,50
NANODECOY–PATHOGEN COMPLEX FOR VACCINATION
Considering that cell-membrane-mimicking nanodecoys are able to trap pathogens in a natural manner, the obtained nanoparticle–pathogen complexes can be applied to develop vaccines.21 Typically, vaccine preparation relies on heat or chemical treatments to destruct the protein’s tertiary structure, leading to antigen alteration and reduced immunogenicity.51 However, the inherent balance between efficacy and safety presents a great limitation to vaccine development. Recently, more efforts have focused on weakening a pathogen’s infectivity while maintaining its original structure, thus enhancing both the efficacy and safety of vaccines.3 Trapping and detention of pathogens/exotoxins by nanodecoys offer the chance to denude the pathogens’ infectious capabilities without compromising immunogenicity. Demonstrating a novel concept of pathogen/toxin sequestration by cell-membrane-mimicking nanodecoys for vaccination, Hu et al. showed that the spontaneous interactions between RBC membrane-coated nanodecoys and pore-forming toxins presented a facile approach for developing safe and effective vaccines.21 The nanoparticle vehicles are endowed with many properties that are beneficial to antigen processing: the pathogens/toxins were displayed on immune cells in their native manner; the nanoparticles’ small size and long circulation promoted antigen presentation; and the nanoparticle carriers were primarily absorbed by cells through endocytosis, thereby promoting the localization and metabolism of pathogens/exotoxins in endolysosomal sections. These properties together contributed to the improved safety and efficiency by nanoparticle-trapped pathogens/toxins.
CONCLUSIONS AND OUTLOOK
Conventional anti-infection strategies primarily depend on structure-customized platforms such as antibodies and anti-sera. Although effective, these formulations often require accurate identification of the pathogenic species and have proven to be impractical to administer at times. Amidst the increasing awareness of emerging virus epidemics as well as the rising threat of drug-resistant bacteria, broadly applicable platforms have tremendous potential for the treatment of infectious diseases. In-depth biological research has elucidated different mechanisms of pathogen infection, most of which involve attaching to the cellular membrane biointerface and invading the host cells. Recent nanotechnology developments have resulted in a variety of nanoscale cell-membrane-mimicking formulations, including liposomes, reconstituted lipoproteins, and cell-membrane nanostructures. Much effort has been directed toward exploring the interactions between these biomimetic nanodecoys and pathogens/exotoxins, and researchers have demonstrated successful protection against major pathogenic infections through nanoparticle functionalization.17,18,26,30 These nanoformulations can be infused in vivo to alleviate the disease burden in multiple infectious diseases. Moreover, pathogen/toxin trapping and detention by cellular-membrane-based nanostructures has wider implications in the development of safe and effective vaccines.18, 21
Cell-membrane-mimicking nenodecoys represent a promising technology that has enormous translation potential.16 To realize this potential, further exploration and detailed study are required. Scalability is a crucial factor that is required for any clinical translational nanostructures. Previous work on the robust preparation of liposomes, lipoproteins, and cell-membrane nanoparticles indicates the need for further explorations on the large-scale production of these biomimetic nanodecoys.17 Moving forward, the host–pathogen affinity is a key issue that needs to be further considered. For many pathogens with clear infection mechanisms, strategies including bioconjugate chemistry and genetic editing can be employed to enhance the specific protein expression, resulting in enhanced platform efficacy.18,52 A promising property of these bioinspired nanodecoys is that they are highly customizable, especially for cell-membrane-coated nanoparticles. Although most of the previous designs employed a PLGA nanoparticle as the core, it is easy to imagine such cell-membrane-coated platforms can be formed with many other types of nanoparticle cores,43,47,53–55 such as magnetic nanoparticles. With an additional magnet, cell membrane-wrapped magnetic particles can be localized to targeted sites and away from susceptible tissues.54 Finally, although nanoparticle-trapped pathogens have many advantages over traditional vaccine developments, the nanocomplexes containing nondenatured pathogens unavoidably increase safety risks. Further in-depth studies are needed prior to successful clinical translation of these novel nanotechnologies.
Suggested pull quotes.
Despite their different infection modes, pathogens must interact with host cell membranes, at some point.
With the rapid developments of biotechnology and nanotechnology, the physical and chemical properties of phospholipid bilayers may be tuned finely to improve their efficacy in various anti-infection applications.
Cell-membrane-derived nanoparticles present an attractive platform for anti-infection applications because pathogens have evolved to utilize abundant molecules on the cell membrane.
Trapping and detention of pathogens/exotoxins by nanodecoys offer the chance to denude the pathogens’ infectious capabilities without compromising immunogenicity.
ACKNOWLEDGMENTS
We would like to thank Prof. Liangfang Zhang at University of California, San Diego for manuscript discussion and editing. This work was supported by the Intramural Research Program, National Institute of Biomedical Imaging and Bioengineering (NIBIB), National Institutes of Health (NIH).
Footnotes
The authors declare no competing financial interest.
REFERENCES
- (1).Cohen ML Changing Patterns of Infectious Disease. Nature 2000, 406, 762–767. [DOI] [PubMed] [Google Scholar]
- (2).Braden CR; Dowell SF; Jernigan DB; Hughes JM Progress in Global Surveillance and Response Capacity 10 Years after Severe Acute Respiratory Syndrome. Emerg. Infect. Dis 2013, 19, 864–869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (3).Pati R; Shevtsov M; Sonawane A Nanoparticle Vaccines Against Infectious Diseases. Front. Immunol 2018, 9, 2224. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (4).Wu JT; Leung K; Leung GM Nowcasting And Forecasting The Potential Domestic and International Spread of the 2019-Ncov Outbreak Originating in Wuhan, China: A Modelling Study. The Lancet 2020, DOI: 10.1016/S0140-6736(20)30260-9. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (5).Zhu N; Zhang D; Wang W; Li X; Yang B; Song J; Zhao X; Huang B; Shi W; Lu R; A Novel Coronavirus from Patients with Pneumonia in China, 2019. N. Engl. J. Med 2020, 382, 727–733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (6).Li Q; Guan X; Wu P; Wang X; Zhou L; Tong Y; Ren R; Leung KSM; Lau EHY; Wong JY Early Transmission Dynamics in Wuhan, China, of Novel Coronavirus–Infected Pneumonia. N. Engl. J. Med 2020, DOI: 10.1056/NEJMc2001468. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (7).Sullivan NJ; Sanchez A; Rollin PE; Yang ZY; Nabel GJ Development of a Preventive Vaccine for Ebola Virus Infection in Primates. Nature 2000, 408, 605–609. [DOI] [PubMed] [Google Scholar]
- (8).Abbink P; Larocca RA; De La Barrera RA; Bricault CA; Moseley ET; Boyd M; Kirilova M; Li Z; Ng’ang’a D; Nanayakkara O; Nityanandam R Protective Efficacy of Multiple Vaccine Platforms Against Zika Virus Challenge in Rhesus Monkeys. Science 2016, 353, 1129–1132. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (9).Iseman MD Treatment of Multidrug-Resistant Tuberculosis. N. Engl. J. Med 1993, 329, 784–791. [DOI] [PubMed] [Google Scholar]
- (10).Giles J Superbug Genome Excels at Passing on Drug Resistance. Nature 2004, 430, 126. [DOI] [PubMed] [Google Scholar]
- (11).Palella FJ; Delaney KM; Moorman AC; Loveless MO; Fuhrer J; Satten GA; Aschman DJ; Holmberg SD; Investigators HIVOS Declining Morbidity and Mortality Among Patients with Advanced Human Immunodeficiency Virus Infection. N. Engl. J. Med 1998, 338, 853–860. [DOI] [PubMed] [Google Scholar]
- (12).Costerton JW; Stewart PS; Greenberg EP Bacterial Biofilms: A Common Cause of Persistent Infections. Science 1999, 284, 1318–1322. [DOI] [PubMed] [Google Scholar]
- (13).Akira S; Uematsu S; Takeuchi O Pathogen Recognition and Innate Immunity. Cell 2006, 124, 783–801. [DOI] [PubMed] [Google Scholar]
- (14).Bricarello DA; Patel MA; Parikh AN Inhibiting Host–Pathogen Interactions Using Membrane-Based Nanostructures. Trends Biotechnol. 2012, 30, 323–330. [DOI] [PubMed] [Google Scholar]
- (15).Lindenbach BD; Evans MJ; Syder AJ; Wolk B; Tellinghuisen TL; Liu CC; Maruyama T; Hynes RO; Burton DR; McKeating JA; Rice CM Complete Replication of Hepatitis C Virus in Cell Culture. Science 2005, 309, 623–626. [DOI] [PubMed] [Google Scholar]
- (16).Fang RH; Luk BT; Hu C-MJ; Zhang L Engineered Nanoparticles Mimicking Cell Membranes For Toxin Neutralization. Adv. Drug Delivery Rev 2015, 90, 69–80. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (17).Rao L; Wang W; Meng Q-F; Tian M; Cai B; Wang Y; Li A; Zan M; Xiao F; Bu L-L A Biomimetic Nanodecoy Traps Zika Virus To Prevent Viral Infection and Fetal Microcephaly Development. Nano Lett. 2019, 19, 2215–2222. [DOI] [PubMed] [Google Scholar]
- (18).Zhang P; Chen Y; Zeng Y; Shen C; Li R; Guo Z; Li S; Zheng Q; Chu C; Wang Z; Zheng Z; Tian R; Ge S; Zhang X; Xia N-S; Liu G; Chen X VirusMimetic Nanovesicles as a Versatile Antigen-Delivery System. Proc. Natl. Acad. Sci. U. S. A 2015, 112, E6129–E6138. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (19).Rao L; Bu L-L; Ma L; Wang W; Liu H; Wan D; Liu J-F; Li A; Guo S-S; Zhang L; Zhang W-F; Zhao X-Z; Sun Z-J; Liu W Platelet-Facilitated Photothermal Therapy of Head and Neck Squamous Cell Carcinoma. Angew. Chem. Int. Ed 2018, 57, 986–991. [DOI] [PubMed] [Google Scholar]
- (20).Hu C-MJ; Zhang L; Aryal S; Cheung C; Fang RH; Zhang L Erythrocyte Membrane-Camouflaged Polymeric Nanoparticles as a Biomimetic Delivery Platform. Proc. Natl. Acad. Sci. U. S. A 2011, 108, 10980–10985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (21).Hu C-MJ; Fang RH; Luk BT; Zhang L Nanoparticle-Detained Toxins for Safe and Effective Vaccination. Nat. Nanotechnol 2013, 8, 933–938. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (22).Rao L; Bu L-L; Xu J-H; Cai B; Yu G-T; Yu X; He Z; Huang Q; Li A; Guo S-S Red Blood Cell Membrane as a Biomimetic Nanocoating for Prolonged Circulation Time and Reduced Accelerated Blood Clearance. Small 2015, 11, 6225–6236. [DOI] [PubMed] [Google Scholar]
- (23).Khezerlou A; Alizadeh-Sani M; Azizi-Lalabadi M; Ehsani A Nanoparticles and their Antimicrobial Properties Against Pathogens Including Bacteria, Fungi, Parasites and Viruses. Microb. Pathogenesis 2018, 123, 505–526. [DOI] [PubMed] [Google Scholar]
- (24).Trkola A; Dragic T; Arthos J; Binley JM; Olson WC; Allaway GP; ChengMayer C; Robinson J; Maddon PJ; Moore JP CD4-Dependent, Antibody-Sensitive Interactions between HIV-1 and its Co-Receptor CCR-5. Nature 1996, 384, 184–187. [DOI] [PubMed] [Google Scholar]
- (25).Hooper LV; Midtvedt T; Gordon JI How Host–Microbial Interactions Shape the Nutrient Environment of the Mammalian Intestine. Annu. Rev. Nutr 2002, 22, 283–307. [DOI] [PubMed] [Google Scholar]
- (26).Hu C-MJ; Fang RH; Copp J; Luk BT; Zhang L A Biomimetic Nanosponge that Absorbs Pore-Forming Toxins. Nat. Nanotechnol 2013, 8, 336–340. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (27).Westman J; Hube B; Fairn GD Integrity under Stress: Host Membrane Remodelling and Damage by Fungal Pathogens. Cell. Microbiol 2019, 21, e13016. [DOI] [PubMed] [Google Scholar]
- (28).Moyes DL; Wilson D; Richardson JP; Mogavero S; Tang SX; Wernecke J; Höfs S; Gratacap RL; Robbins J; Runglall M Candidalysin is a Fungal Peptide Toxin Critical for Mucosal Infection. Nature 2016, 532, 64–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (29).Cowman AF; Crabb BS Invasion of Red Blood Cells by Malaria Parasites. Cell 2006, 124, 755–766. [DOI] [PubMed] [Google Scholar]
- (30).Liu X; Yuan L; Zhang L; Mu Y; Li X; Liu C; Lv P; Zhang Y; Cheng T; Yuan Q; Xia N; Chen X; Liu G Bioinspired Artificial Nanodecoys for Hepatitis B Virus. Angew. Chem. Int. Ed 2018, 57, 12499–12503. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (31).Goyal AK; Rawat A; Mahor S; Gupta PN; Khatri K; Vyas SP Nanodecoy System: A Novel Approach To Design Hepatitis B Vaccine for Immunopotentiation. Int. J. Pharm 2006, 309, 227–233. [DOI] [PubMed] [Google Scholar]
- (32).Bangham AD; Standish MM; Weissmann G The Action of Steroids and Streptolysin S on the Permeability of Phospholipid Structures to Cations. J. Mol. Biol 1965, 13, 253–259. [DOI] [PubMed] [Google Scholar]
- (33).Tallury P; Malhotra A; Byrne LM; Santra S Nanobioimaging and Sensing of Infectious Diseases. Adv. Drug Deliv. Rev 2010, 62, 424–437. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (34).Henry BD; Neill DR; Becker KA; Gore S; Bricio-Moreno L; Ziobro R; Edwards MJ; Mühlemann K; Steinmann J; Kleuser B Engineered Liposomes Sequester Bacterial Exotoxins and Protect from Severe Invasive Infections in Mice. Nat. Biotechnol 2014, 33, 81–88. [DOI] [PubMed] [Google Scholar]
- (35).Nussbaum O; Lapidot M; Loyter A Reconstitution of Functional Influenza Virus Envelopes and Fusion with Membranes and Liposomes Lacking Virus Receptors. J. Virol 1987, 61, 2245–2252. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (36).Zolnik BS; González-Fernández A; Sadrieh N; Dobrovolskaia MA Nanoparticles and the Immune System. Endocrinology 2010, 151, 458–465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (37).Thamphiwatana S; Gao W; Pornpattananangkul D; Zhang Q; Fu V; Li J; Li J; Obonyo M; Zhang L Phospholipase A2-Responsive Antibiotic Delivery via Nanoparticle-Stabilized Liposomes for the Treatment of Bacterial Infection. J. Mater. Chem. B 2014, 2, 8201–8207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (38).Magee WE; Miller OV Liposomes Containing Antiviral Antibody Can Protect Cells from Virus Infection. Nature 1972, 235, 339–341. [DOI] [PubMed] [Google Scholar]
- (39).Bricarello DA; Smilowitz JT; Zivkovic AM; German JB; Parikh AN Reconstituted Lipoprotein: A Versatile Class of Biologically-Inspired Nanostructures. ACS Nano 2011, 5, 42–57. [DOI] [PubMed] [Google Scholar]
- (40).Badin J; Barillec A Streptolysin O Inhibition by Serum ΓG-Globulin and Β-Lipoprotein After Blocking of Nonesterified Cholesterol by Digitonin. J. Lab. Clin. Med 1970, 75, 975–982. [PubMed] [Google Scholar]
- (41).Bricarello DA; Mills EJ; Petrlova J; Voss JC; Parikh AN Ganglioside Embedded in Reconstituted Lipoprotein Binds Cholera Toxin with Elevated Affinity. J. Lipid Res 2010, 51, 2731–2738. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (42).Chen Z; Wang Z; Gu Z Bioinspired and Biomimetic Nanomedicines. Acc. Chem. Res 2019, 52, 1255–1264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (43).Parodi A; Quattrocchi N; van de Ven AL; Chiappini C; Evangelopoulos M; Martinez JO; Brown BS; Khaled SZ; Yazdi IK; Enzo MV Synthetic Nanoparticles Functionalized with Biomimetic Leukocyte Membranes Possess Cell-Like Functions. Nat. Nanotechnol 2013, 8, 61–68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (44).Hu C-MJ; Fang RH; Wang K-C; Luk BT; Thamphiwatana S; Dehaini D; Nguyen P; Angsantikul P; Wen CH; Kroll AV Nanoparticle Biointerfacing by Platelet Membrane Cloaking. Nature 2015, 526, 118–121. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (45).de Carvalho JV; de Castro RO; da Silva EZM; Silveira PP; da Silva-Januário ME; Arruda E; Jamur MC; Oliver C; Aguiar RS; daSilva LLP Nef Neutralizes the Ability of Exosomes from CD4+ T Cells to Act as Decoys during HIV-1 Infection. PLoS One 2014, 9, e113691. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (46).Inal Jameel M.; Ansa-Addo Ephraim A.; Lange S Interplay of Host–Pathogen Microvesicles and their Role in Infectious Disease. Biochem. Soc. Trans 2013, 41, 258–262. [DOI] [PubMed] [Google Scholar]
- (47).Li L-L; Xu J-H; Qi G-B; Zhao X-Z; Yu F; Wang H Core–Shell Supramolecular Gelatin Nanoparticles for Adaptive and “On-Demand” Antibiotic Delivery. ACS Nano 2014, 8, 4975–4983. [DOI] [PubMed] [Google Scholar]
- (48).Wei X; Zhang G; Ran D; Krishnan N; Fang RH; Gao W; Spector SA; Zhang L T-Cell-Mimicking Nanoparticles Can Neutralize HIV Infectivity. Adv. Mater 2018, 30, 1802233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (49).Zhang Y; Chen Y; Lo C; Zhuang J; Angsantikul P; Zhang Q; Wei X; Zhou Z; Obonyo M; Fang RH; Gao W; Zhang L Inhibition of Pathogen Adhesion by Bacterial Outer Membrane-Coated Nanoparticles. Angew. Chem. Int. Ed 2019, 58, 11404–11408. [DOI] [PubMed] [Google Scholar]
- (50).Rao L; Cai B; Bu L-L; Liao Q-Q; Guo S-S; Zhao X-Z; Dong W-F; Liu W Microfluidic Electroporation-Facilitated Synthesis of Erythrocyte Membrane-Coated Magnetic Nanoparticles for Enhanced Imaging-Guided Cancer Therapy. ACS Nano 2017, 11, 3496–3505. [DOI] [PubMed] [Google Scholar]
- (51).Cryz SJ; Fürer E; Germanier R Effect of Chemical and Heat Inactivation On the Antigenicity And Immunogenicity Of Vibrio Cholerae. Infec. Immun 1982, 38, 21–26. [DOI] [PMC free article] [PubMed] [Google Scholar]
- (52).Rao L; Meng Q-F; Huang Q; Wang Z; Yu G-T; Li A; Ma W; Zhang N; Guo S-S; Zhao X-Z; Liu K; Yuan Y; Liu W Platelet–Leukocyte Hybrid Membrane-Coated Immunomagnetic Beads for Highly Efficient and Highly Specific Isolation of Circulating Tumor Cells. Adv. Funct. Mater 2018, 28, 1803531. [Google Scholar]
- (53).Rao L; Bu L-L; Cai B; Xu J-H; Li A; Zhang W-F; Sun Z-J; Guo S-S; Liu W; Wang T-H; Zhao X-Z Cancer Cell Membrane-Coated Upconversion Nanoprobes for Highly Specific Tumor Imaging. Adv. Mater 2016, 28, 3460–3466. [DOI] [PubMed] [Google Scholar]
- (54).Rao L; Bu L-L; Meng Q-F; Cai B; Deng W-W; Li A; Li K; Guo S-S; Zhang W-F; Liu W; Sun Z-J; Zhao X-Z Antitumor Platelet-Mimicking Magnetic Nanoparticles. Adv. Funct. Mater 2017, 27, 1604774. [Google Scholar]
- (55).Fang RH; Kroll AV; Gao W; Zhang L Cell Membrane Coating Nanotechnology. Adv. Mater 2018, 30, 1706759. [DOI] [PMC free article] [PubMed] [Google Scholar]


