Abstract
Mycobacteriosis caused by non-tuberculous mycobacteria (NTM) is a rising concern in human medicine both in immunocompromised and immunocompetent patients. In cats, mycobacteriosis caused by NTM is considered mostly to be a focal or dermal infection, with disseminated disease mostly caused by Mycobacterium avium. We describe three cases of disseminated mycobacteriosis in cats, caused by Mycobacterium malmoense, Mycobacterium branderi/shimoidei and M. avium, with no identified underlying immunosuppression. In all cases, extracellular mycobacteria were seen in the pulmonary epithelium, intestinal lumen and glomerular tufts, which could affect the shedding of the organism. The present study highlights the importance of mycobacteriosis as a differential even in immunocompetent animals. Considering the close relationship of owners and pets and the potential presence of free mycobacteria in secretions, cats should be considered as a possible environmental reservoir for mycobacteria.
Keywords: cat, mycobacteriosis, non-tuberculous mycobacteria
Introduction
Mycobacteria are acid-fast, aerobic, non-spore forming rod shaped bacteria that range from obligate pathogens to environmental saprophytes. Mycobacteria can be divided into three groups based on the disease they cause: Mycobacterium leprae and Mycobacterium lepromatosis cause leprosy in man. The Mycobacterium tuberculosis complex (MTC) is formed from species that cause tuberculosis in man and animals, such as M. tuberculosis, Mycobacterium bovis and Mycobacterium microti. In human medicine the word ‘tuberculosis’ is reserved for infections caused by the MTC species (Esteban and Muños-Egea, 2016). The rest of the mycobacterial species are grouped as non-tuberculous mycobacteria (NTM), which are mostly opportunistic environmental pathogens (Koh, 2017). Most mycobacteria have a particular host preference, but some can cause infections in other species, including man (Quinn et al., 2011).
Human leprosy is caused by either M. leprae or the more recently discovered M. lepromatosis, with varying skin lesions and peripheral nerve thickening and occasionally with systemic or mucosal involvement (Renault and Ernst, 2010). It was at first presumed that these species infected only man, but both species have also been found in Eurasian red squirrels (Sciurus vulgaris) (Avanzi et al., 2016) and M. leprae is known to infect nine-banded armadillos (Dasypus novemcinctus) (Balamayooran et al., 2015).
Tuberculosis can cause organ-specific infections, systemic infections or classical granulomatous pulmonary infections (Fitzgerald et al., 2010, Pesciaroli et al., 2014). MTC species are closely related to each other with a high sequence similarity (Esteban and Muños-Egea, 2016) and all of them have an established zoonotic potential (Thirunavukkarasu et al., 2017). M. tuberculosis is the most common cause of tuberculosis in man, but M. bovis has been found in 5–10% of cases (Esteban and Muños-Egea, 2016) and M. microti has been described in over 20 human cases (Esteban and Muños-Egea, 2016). In man, most MTC species cause a disease similar to classical tuberculosis (Esteban and Muños-Egea, 2016). In animals, the most common cause of tuberculosis is M. bovis, which has been described to affect, with varying degree, all common domestic mammals (Pesciaroli et al., 2014).
The Mycobacterium avium complex (MAC) is the most common NTM in man (Daley, 2017, Koh, 2017), but a multitude of NTM species are known to cause disease (Koh, 2017, Thirunavukkarasu et al., 2017). NTM infections affect both immunocompromised and immunocompetent patients (Koh, 2017) and the incidence of NTM isolates in human samples has been rising (Shah et al., 2016). Infections in people are classified into four distinct clinical syndromes: chronic pulmonary disease, lymphadenitis, cutaneous disease and disseminated disease (Koh, 2017). In animals MAC species are generally considered to be the most important NTM (Quinn et al., 2011) and these include M. avium, causing avian tuberculosis, and M. avium subspecies paratuberculosis, causing paratuberculosis or Johne's disease. Other NTM species are described to cause mostly sporadic, non-specific infections in several species (Thirunavukkarasu et al., 2017).
There are few data on the prevalence of mycobacterial infections in domestic cats. In the UK, a recent publication studying histopathological samples reported typical lesions with acid-fast bacteria in approximately 0.3% of all cases examined, with the most common mycobacteria isolated being M. bovis and M. microti (Gunn-Moore et al., 2013). Broughan et al. (2013) reported M. bovis-positive cultures in 16% of feline samples over a period of 7 years when examining microbiological samples submitted to the Animal Health and Veterinary Laboratories Agency, UK, for confirmation of suspected mycobacteriosis. Cats with access to wildlife reservoirs or bovine milk infected with mycobacteria are predisposed to mycobacterial infections (Gunn-Moore, 2014, Pesciaroli et al., 2014) and there might be an increased risk associated with immunosuppressive diseases, especially with NTM infections (Gunn-Moore, 2014).
Tuberculosis in cats usually presents as a localized cutaneous disease or, less commonly, as a disseminated or systemic respiratory or alimentary disease (Gunn-Moore et al., 2011a, Gunn-Moore, 2014, Pesciaroli et al., 2014). NTM infections cause mostly cutaneous or subcutaneous nodules or granulomatous panniculitis with variable lymph node involvement (Gunn-Moore et al., 2011a, Gunn-Moore, 2014). The ‘feline leprosy syndrome’, a leprosy-like ulcerative and nodular skin disease especially affecting the head and limbs (Gunn-Moore, 2014), is also part of the NTM group of infections. Of the NTM species, a few cases of disseminated or systemic disease have been reported with M. avium (Morfitt et al., 1989, Barry et al., 2002, Griffin et al., 2003, Knippel et al., 2004, Baral et al., 2006, De Groot et al., 2010, Rivière et al., 2011), two cases with Mycobacterium xenopi (MacWilliams et al., 1998, Meeks et al., 2008) and single cases with Mycobacterium heckeshornense (Elze et al., 2013), Mycobacterium simiae (Dietrich et al., 2003) and Mycobacterium kansasii (Lee et al., 2017). Despite the reported cases, systemic mycobacteriosis caused by NTM species is still considered relatively rare, especially outside the MAC species (Lloret et al., 2013, Gunn-Moore, 2014, Mauldin and Peters-Kennedy, 2016).
There is close contact between domestic cats and their owners. Given that the incidence of NTM isolates from human patients is rising and that there are still lingering views that systemic NTM mycobacteriosis rarely affects cats, the aim of this study was to describe the pathological and bacteriological findings in three cases of disseminated mycobacteriosis in cats caused by different NTM species.
Materials and Methods
Animals
Three privately owned cats from urban areas of Southern Finland were subjected to necropsy examination at the Department of Veterinary Biosciences, Faculty of Veterinary Medicine, University of Helsinki, Finland, between December 2014 and November 2016. The cats all had a cytological diagnosis of mycobacterial infection. The cats had tested negative for feline immunodeficiency virus (FIV) and feline leukaemia virus (FeLV) within the 8 months preceding their death by humane destruction. Cats A and B had current vaccinations against feline panleucopenia virus, felid herpesvirus and feline calicivirus at the time of death, while cat C had not been vaccinated for several years.
Cytology
Cytological samples, air-dried and stained with May–Grünwald–Giemsa (MGG), were provided from all cases during clinical workup. Samples were provided from initial lesion sites: the liver for cat A, an abdominal mass for cat B and an abdominal mass and a submandibular lymph node for cat C.
Necropsy Examination, Histopathology and Immunohistochemistry
A full necropsy examination was performed within 24 h after death, with tissue samples taken from internal organs, intestinal lymph nodes, the central nervous system (CNS) and bone marrow and from the eye in cats A and B. Tissue samples were fixed in 10% neutral buffered formalin, processed routinely and embedded in paraffin wax. Sections (4 μm) were stained by haematoxylin and eosin (HE) and Ziehl–Neelsen (ZN) stains.
Immunohistochemistry (IHC) was used to detect feline coronavirus antigen. Antigen retrieval was in 0.01 M citrate buffer (pH 6.0) and the primary reagent was mouse anti-feline coronavirus (FIPV3-70, MCA2194, Bio-Rad AbD Serotec Ltd, Oxford, UK) diluted at 1 in 600. The secondary reagent was anti-mouse antibody polymer linked to horseradish peroxidase (BrightVision + Poly-HRP-anti Ms/Rb; Immunologic, Duiven, the Netherlands) with 3,3′-diaminobenzidine tetrahydrochloride as chromogen. The FIPV3-70 anti-coronavirus antibody is routinely used for diagnosis of feline infectious peritonitis (Kipar et al., 1998, Poncelet et al., 2008).
Microbiology
Fresh tissue samples from the liver of cat A, the mass from cat B and the lymph node from cat C were submitted to the Helsinki University Hospital, Division of Clinical Microbiology, Helsinki, Finland, for mycobacterial culture. Firstly, the presence of the M. tuberculosis complex was excluded by use of a specific, direct nucleic acid amplification method (GeneXpert Mycobacterium tuberculosis/rifampicin (MTB/RIF), Cepheid, Sunnyvale, California, USA) (Ioannidis et al., 2011) from the tissue sample. The tissue was cultured on solid and in liquid mycobacterial culture media. One month later, the bacterial deoxyribonucleic acid (DNA) was extracted from positive mycobacterial culture for a mycobacterial DNA line probe assay (GenoType Mycobacterium common mycobacteria (CM), Hain Lifescience, Nehren, Germany) (Russo et al., 2006) and sequencing of a 525 base pair (bp) region of the 16S ribosomal ribonucleic acid (rRNA) gene (Tuuminen et al., 2006) in order to identify the mycobacteria to species level. The sequence obtained was compared with those in the database of the National Center for Biotechnology Information (NCBI, Rockville Pike, Bethesda Maryland, USA) by use of the BLAST (basic logical alignment search tool) database (Cummings et al., 2002).
Results
Animals
The signalment, main clinical complaint and affected organs for each case are presented in Table 1 . All were middle-aged domestic shorthair cats, with at least a partial history of outdoor life. Cat A was a shelter cat, which had been adopted approximately 2 years before diagnosis. Cat B had been a barn cat for most of its life. Cat C lived partly outdoors during the summer, but was kept indoors during other seasons.
Table 1.
Clinical information and affected organs
| Cat A | Cat B | Cat C | |
|---|---|---|---|
| Age (years) | 8 | 10 | 12 |
| Sex | FN | MN | FN |
| Significant clinical finding | Hepatomegaly | Abdominal mass | Generalized lymphadenomegaly |
| Lymph nodes | |||
| Macroscopical | + | + | + |
| Microscopical | + | + | + |
| Liver | |||
| Macroscopical | + | − | + |
| Microscopical | + | + | + |
| Spleen | |||
| Macroscopical | + | + | + |
| Microscopical | + | + | + |
| Lung | |||
| Macroscopical | + | – | + |
| Microscopical | + | + | + |
| Adrenal gland | |||
| Macroscopical | + | – | – |
| Microscopical | + | + | – |
| Peritoneum | |||
| Macroscopical | − | + | + |
| Microscopical | – | + | + |
| Bone marrow | |||
| Macroscopical | – | – | – |
| Microscopical | + | + | + |
| Kidney | |||
| Macroscopical | – | – | – |
| Microscopical | + | + | + |
FN, neutered female; MN, neutered male.
Cytology
In all cases, fine needle aspiration yielded a highly cellular sample with macrophages as the main cellular population. The macrophages were enlarged and distended, with the nucleus often at the periphery of the cytoplasm. Numerous MGG-negative, slender, rod-shaped bacteria were readily seen within macrophages and freely in the background. Cat A had numerous uni- or multinucleated macrophages within the liver sample and mild hepatic lipidosis. In cat B, the abdominal mass was comprised of severe granulomatous inflammation with scant neutrophils and lymphocytes. In cat C, the cytology of both the abdominal mass and the lymph node were consistent with granulomatous inflammation.
Macroscopical Findings
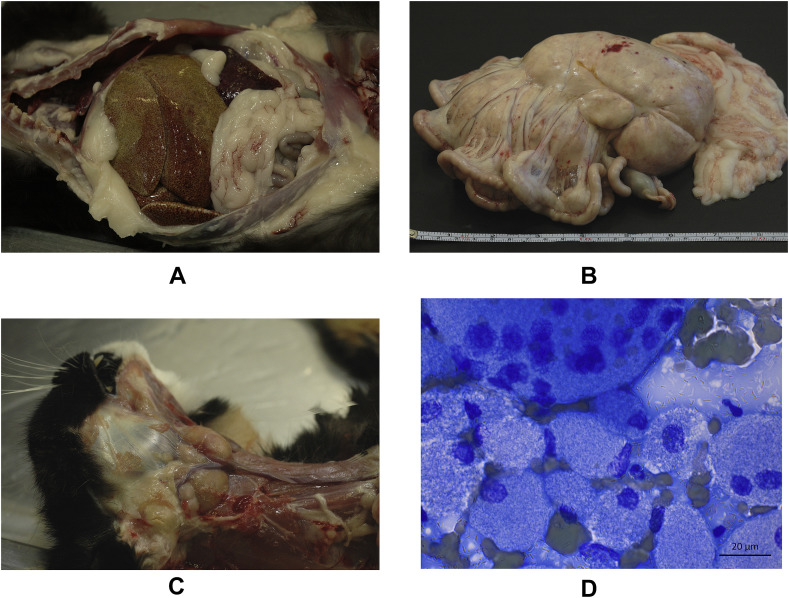
In all cases, innumerable miliary to partly coalescing, 1–5 mm, slightly raised, white or pale grey, oval to round nodules were seen in multiple organs, both on the serosal surfaces and on the cut surfaces. Cat A had severe hepato- and splenomegaly (Fig. 1 A) and a moderate generalized lymphadenomegaly of the peripheral, thoracic and abdominal lymph nodes, with 1–5 mm pale grey nodules on the cut surfaces. Cat B had a large mass replacing the mesenteric lymphoid tissue (Fig. 1B) and moderate lymphadenomegaly of the retropharyngeal and prescapular lymph nodes and severe lymphadenomegaly of the sternal lymph nodes. Cat C had a 7 × 8 × 7 cm, partly necrotic and cystic mass replacing the mesenteric and ileocaecal lymphoid tissue, two approximately 1 cm diameter pale mural nodules in the wall of the small intestine and a severe generalized lymphadenomegaly (Fig. 1C). In all cats, enlarged lymph nodes were firm and pale. In addition, cat B had chronic infarction of the left kidney and cat C had bilaterally small, pale kidneys.
Fig. 1.
(A) Severe diffuse hepatomegaly (cat A). (B) Mesenteric mass (cat B), approximately 16 × 18 × 15 cm, soft and pale, weighing 500 g. (C) Severe lymphadenomegaly of the retropharyngeal lymph nodes (cat C). (D) Cytology of the mesenteric mass (cat B) showing markedly distended macrophages, giant cells and numerous unstained rod-shaped bacteria within the macrophages intra- and extracellularly. MGG.
Histology and Immunohistochemistry
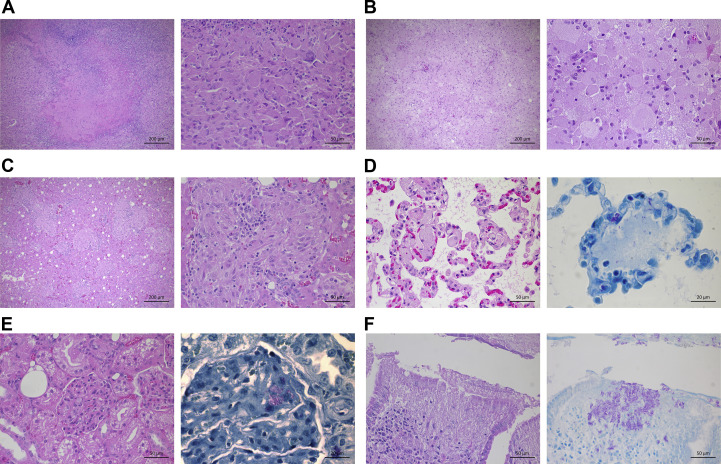
Cat A had a uniform sheet-like infiltration of large polygonal macrophages in almost all affected organs, with these having abundant, pale eosinophilic, slightly granular cytoplasm containing numerous acid-fast bacteria. Admixed with the macrophages were moderate numbers of giant cells and small numbers of neutrophils, lymphocytes and plasma cells. Depending on the affected organ, histiocytic infiltrates were either arranged as multifocal to coalescing nodules or as a diffuse infiltration encompassing large areas of the affected tissue. The nodular infiltrations were mostly associated with vascular or lymphatic structures and no clear granuloma formation was seen in the affected organs. In addition, varying sized areas of coagulative necrosis were present multifocally (Fig. 2 A) and there was a mild histiocytic infiltration bilaterally in the collector vessels of the limbus of the eye and in the choroid plexus.
Fig. 2.
(A) Adrenal (cat A), showing a large area of necrosis with surrounding histiocytic infiltration and scattered giant cells. HE. Inset: closer view of the histiocytic infiltration. HE. (B) Abdominal mass (cat B), showing marked infiltration of enlarged macrophages. HE. Inset: high-power view of the diffuse histiocytic infiltration. HE. (C) Liver (cat C), showing multifocal nodular granulomas. HE. Inset: high-power view of a granuloma. HE. (D) Lung (cat B), showing multifocal large histiocytes within the alveolar walls. HE. Inset: acid-fast rods within the histiocytes and free in the alveolar space. ZN. (E) Kidney (cat B), showing mild basement membrane thickening in the glomeruli and multifocal aggregates of histiocytes within the glomerular tufts. HE. Inset: acid-fast rods within the glomerular tufts. ZN. (F) Small intestine (cat C) with focal epithelial ulceration. HE. Inset: numerous acid-fast rods within the ulcerated villus and free in the intestinal lumen. ZN.
Cat B had a similar sheet-like infiltration of macrophages in all affected organs. The abdominal mass was almost completely comprised of markedly enlarged macrophages and giant cells (Fig. 2B), with small lymphocytic foci admixed with the inflammatory cells, interpreted as possible replacement of normal lymph node tissue by the histiocytic infiltrate. In addition, cat B had a moderate, multifocal, histiocytic infiltration within the small intestine and in the connective tissue of the renal papilla, retro-orbital fat, gastric submucosa and surrounding the thyroid glands. Free mycobacteria were also seen within the lamina propria of the small intestinal villi.
In the affected organs of cat C, the macrophages were smaller and spindle shaped, with a moderate amount of eosinophilic cytoplasm and numerous intracytoplasmic mycobacteria; only a few multinucleated giant cells were seen. The macrophages were arranged in concentric circles, reminiscent of classical granulomas (Fig. 2C), with multifocal areas of coagulative necrosis and moderate numbers of lymphocytes and plasma cells admixed with the inflammation. Individual granulomas were readily distinguished even in heavily infiltrated tissues. In addition, cat C had a severe, multifocal, partly transmural, granulomatous enteritis in the small intestine and microscopical miliary granulomas in the subepicardial area of the left ventricle and within the thyroid glands.
In all cats, macrophages laden with ZN-positive bacteria were present within the alveolar walls and were free in the alveoli and occasionally, free mycobacteria were seen within the alveolar fluid (Fig. 2D). Cat C had multifocal histiocytic infiltration of the tracheal lymphoid tissue with ulceration of the overlying epithelium and free mycobacteria in the tracheal lumen. In cats A and B, single mycobacteria-filled macrophages were present multifocally within the glomerular tufts of the kidney (Fig. 2E) and in cat C, mild focal histiocytic infiltration was noted around a renal cortical vein, with free mycobacteria within glomerular tufts, separated from glomerular filtrate by the basement membrane. The infiltration was mild without granuloma formation and at times, macrophages were difficult to ascertain in HE-stained sections. In cat B the epithelium of the small intestinal villi was eroded multifocally, with mycobacteria-laden macrophages separated from faeces by the basement membrane, and in cat C, multifocal epithelial ulceration was seen with free mycobacteria in the intestinal lumen (Fig. 2D).
In addition, cat A had moderate multifocal chronic lymphoplasmacytic interstitial nephritis with mild fibrosis and mild, multifocal, chronic neutrophilic subendocardial myocarditis affecting the left ventricle. IHC for feline coronavirus antigen was negative in all cases.
Microbiology
All cats had different NTM species isolated from tissue samples. In cat A, Mycobacterium malmoense was isolated from the liver. In cat B, DNA sequence closest to Mycobacterium branderi/shimoidei was amplified from the abdominal mass. In cat C, M. avium was isolated from a lymph node.
Discussion
This case series describes three cases of disseminated NTM infections in cats with no apparent underlying immunocompromise. To the authors' knowledge, M. bronderi/shimodei has not previously been verified as the cause of a generalized infection in cats. M. malmoense has been reported in a soft tissue mass (Hetzel et al., 2012) and as an unspecified infection (Gunn-Moore et al., 2011a).
All animals had at least a partial history of an outdoor life and as hunting and fighting has been described as predisposing factors for mycobacterial infections (Gunn-Moore, 2014), the infections most likely originated from contact with infected prey or soil. Of the systemic NTM infections described, only M. avium and M. simiae have been described in cats with no apparent immunocompromise (Morfitt et al., 1989, Barry et al., 2002, Dietrich et al., 2003, Knippel et al., 2004, Baral et al., 2006, De Groot et al., 2010, Rivière et al., 2011). An idiopathic CD4+ (cluster of differentiation) T-cell lymphopenia was suspected to be the cause of a M. xenopi infection (Meeks et al., 2008) and local surgery may have caused fulmination of another M. xenopi infection (MacWilliams et al., 1998). FIV infection was described with M. heckeshornense infection (Elze et al., 2013). No apparent immunocompromising factors were noted in the history or macroscopical or histological findings in the present cases. Mycobacterial infection should be considered a differential diagnosis for systemic granulomatous inflammation, even in immunocompetent cats, especially in shelter or barnyard animals.
The cytological appearance of samples from all cases correlated well with findings described in the literature (Raskin, 2010, Cowell and Arndt, 2014) and cytological interpretation was straightforward from MGG staining. Cytology is described to be a useful tool in lepromatous lesions, but has lower sensitivity in tuberculoid lesions (Raskin, 2010). However, all of the present cases had abundant mycobacteria within macrophages and organisms free in the background, readily allowing cytological diagnosis.
There were slight differences in the gross findings between the cases. Lymph nodes and spleen were affected in all cases, while only cats A and C had gross changes in the liver and lungs. The presence of necrosis also varied between the cases. M. malmoense is phylogenetically closer to M. avium than to M. branderi/shimodei (Schrenzel, 2012), which could in part account for the different gross findings. However, no morphological differences are reported between different mycobacterial infections (Gunn-Moore et al., 2011a) and more cases are needed to determine whether there are differences between the infections caused by different mycobacterial species. In previous studies (Morfitt et al., 1989, Knippel et al., 2004; Baral et al., 2006; De Groot et al., 2010, Gunn-Moore et al., 2011a), lymph node involvement was common, as it was in the present study. Sampling an enlarged lymph node can be a fast and relatively easy way to confirm a diagnosis. M. avium has been described to cause enlarged abdominal lymph nodes (Barry et al., 2002, Knippel et al., 2004; Rivière et al., 2011), similar to cats B and C in this study and in addition to feline infectious peritonitis and lymphoma, mycobacteriosis should be considered as a differential diagnosis when markedly enlarged abdominal lymph nodes are noted.
There were clear histological differences between the cases. The lesions of cat C resembled classical tuberculous granulomas, while cats A and B had more diffuse infiltration with no apparent granuloma formation. There also appeared to be an affinity for vascular structures in cats A and B, which was absent from the lesions in cat C. Histological patterns similar to those reported in cat C are described with M. avium infection (Morfitt et al., 1989, Knippel et al., 2004, Baral et al., 2006). To the best of our knowledge, there are no detailed descriptions in the literature concerning the histological pattern for M. branderi/shimoidei or M. malmoense infection, but Gunn-Moore et al. (2011b) found no significant differences between the histological changes related to infections by different mycobacteria. However, most of those cases involved infection with MTC species, MAC species or were culture-negative, with only one cat with M. malmoense infection studied. When considering giant cells, the present findings match the results of Gunn-Moore et al. (2011b), with giant cells appearing with M. malmoense infection, but being rare in lesions caused by M. avium. In cat C, mycobacteria-laden macrophages were also often seen within the adipose tissue of different organs, which could indicate similar predilection for adipose tissue as reported for some NTM species (Gunn-Moore, 2014).
All grossly affected organs had an abundance of mycobacteria-laden macrophages, with strong acid-fast staining. Hetzel et al. (2012) described similar results with M. malmoense, but M. avium has been described with either myriad or only few intralesional bacteria (Baral et al., 2006). In addition, all cases had several histologically-affected organs where gross lesions were absent. Based on these results, mycobacterial spreading between organ systems may happen faster than the formation of visible lesions. This should be taken into account when considering surgical excision of lesions.
The presence of free mycobacteria within pulmonary structures, the intestinal lumen and glomerular tufts could lead to shedding of the mycobacteria. NTM species are widespread in the environment (Koh, 2017) and because of the composition of their outer membrane are relatively resistant to chlorine and ozone (Daley, 2017). Even though people and animals are readily exposed to the bacteria in the environment (Griffith et al., 2007), infections caused by NTM are rare and, to our knowledge, there are no reported animal-to-human infections of NTM species outside the MAC. However, considering the close relationship of owners and pets and the survivability of mycobacteria in the household environment, a cat shedding mycobacteria in its faeces and sputum may increase the owner's exposure to mycobacteria. This should be taken into consideration, especially if the animal is infected with MAC species or if the owner has any immunocompromise.
To our knowledge, M. branderi/shimoidei has not been described previously as a naturally occurring infection in animals. Both are relatively uncommon pathogens in human medicine (Bittner and Preheim, 2016) and have been described mostly in the lung of both immunocompromised and immunocompetent patients (Galizzi et al., 2013, Kanaji et al., 2013, Turvey et al., 2017). The sequencing method used in this study could not differentiate between the two species. M. malmoense has been isolated from natural waters in Finland (Bittner and Preheim, 2016) and causes mainly pulmonary infections in people (Bittner and Preheim, 2016, Koh, 2017), with cases reported in immunocompetent patients (Jeon et al., 2015). M. avium causes respiratory disease in both immunocompetent and immunocompromised patients, as well as disseminated disease in immunocompromised patients (Daley, 2017).
Our findings in this case series highlight that systemic mycobacterial infection in cats is not only restricted to immunocompromised animals and that NTM species outside the MAC can be causative agents of feline disease. Mycobacterial infection should be considered as a differential diagnosis in cats with generalized lymphadenomegaly or an abdominal mass, especially in free-roaming or shelter animals. Cytology or biopsy of affected tissue can give a speedy diagnosis in systemic forms of the disease.
Acknowledgments
The authors thank the laboratory technicians from the histology laboratory and post-mortem room for invaluable assistance, J. Linden for help with methodology and T. Grönthal for assistance with images. This research did not receive any specific grant from funding agencies in the public, commercial or not-for-profit sectors.
Footnotes
Supplementary data related to this article can be found at https://doi.org/10.1016/j.jcpa.2018.02.002.
Conflict of Interest Statement
The authors declare no conflict of interest with respect to publication of this manuscript.
Appendix A. Supplementary data
The following is the supplementary data related to this article:
References
- Avanzi C., del-Pozo J., Benjak A., Stevenson K., Simpson V.R. Red squirrels in the British Isles are infected with leprosy bacilli. Science. 2016;354:744–747. doi: 10.1126/science.aah3783. [DOI] [PubMed] [Google Scholar]
- Balamayooran G., Pena M., Sharma R., Truman R.W. The armadillo as an animal model and reservoir host for Mycobacterium leprae. Clinics in Dermatology. 2015;33:108–115. doi: 10.1016/j.clindermatol.2014.07.001. [DOI] [PubMed] [Google Scholar]
- Baral R.M., Metcalfe S.S., Krockenberger M.B., Catt M.J., Barrs V.R. Disseminated Mycobacterium avium infection in young cats: overrepresentation of Abyssinian cats. Journal of Feline Medicine & Surgery. 2006;8:23–44. doi: 10.1016/j.jfms.2005.06.004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Barry M., Taylor J., Woods J.P. Disseminated Mycobacterium avium infection in a cat. Canadian Veterinary Journal. 2002;43:369–371. [PMC free article] [PubMed] [Google Scholar]
- Bittner M.J., Preheim L.C. Other slow-growing nontuberculous mycobacteria. Microbiology Spectrum. 2016;4 doi: 10.1128/microbiolspec.TNMI7-0012-2016. [DOI] [PubMed] [Google Scholar]
- Broughan J.M., Downs S.H., Crawshaw T.R., Upton P.A., Brewer J. Mycobacterium bovis infections in domesticated non-bovine mammalian species. Part 1: Review of epidemiology and laboratory submissions in Great Britain 2004–2010. The Veterinary Journal. 2013;198:339–345. doi: 10.1016/j.tvjl.2013.09.006. [DOI] [PubMed] [Google Scholar]
- Cowell R.L., Arndt T.P. Selected infectious agents. In: Valenciano A.C., Cowell R.L., editors. Cowell and Tyler's Diagnostic Cytology and Hematology of the Dog and Cat. 4th Edit. Elsevier Mosby; St Louis: 2014. pp. 49–50. [Google Scholar]
- Cummings L., Riley L., Black L., Souvorov A., Resenchuk S. Genomic BLAST: custom-defined virtual databases for complete and unfinished genomes. FEMS Microbiology Letters. 2002;216:133–138. doi: 10.1111/j.1574-6968.2002.tb11426.x. [DOI] [PubMed] [Google Scholar]
- Daley C.L. Mycobacterium avium complex disease. Microbiology Spectrum. 2017;5 doi: 10.1128/microbiolspec.TNMI7-0045-2017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Groot P.H.S., van Ingen J., de Zwaan R., Boeree M.J., van Soolingen D. Disseminated Mycobacterium avium subsp. avium infection in a cat, the Netherlands. Veterinary Microbiology. 2010;144:527–529. doi: 10.1016/j.vetmic.2010.05.028. [DOI] [PubMed] [Google Scholar]
- Dietrich U., Arnold P., Guscetti F., Pfyffer G.E., Spiess B. Ocular manifestation of disseminated Mycobacterium simiae infection in a cat. Journal of Small Animal Practice. 2003;44:121–125. doi: 10.1111/j.1748-5827.2003.tb00132.x. [DOI] [PubMed] [Google Scholar]
- Elze J., Grammel L., Richter E., Aupperle H. First description of Mycobacterium heckeshornense infection in a feline immunodeficiency virus-positive cat. Journal of Feline Medicine & Surgery. 2013;15:1141–1144. doi: 10.1177/1098612X13490711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Esteban J., Muños-Egea M.C. Mycobacterium bovis and other uncommon members of the Mycobacterium tuberculosis complex. Microbiology Spectrum. 2016;4 doi: 10.1128/microbiolspec.TNMI7-0021-2016. [DOI] [PubMed] [Google Scholar]
- Fitzgerald D.W., Sterling T.R., Haas D.W. Mycobacterium tuberculosis. In: Mandell G.L., Bennet J.E., Dolin R., editors. 7th Edit. Vol. 2. Churchill Livingstone Elsevier; Philadelphia: 2010. pp. 3129–3163. (Mandell, Douglas, and Bennett's Principles and Practice of Infectious Diseases). [Google Scholar]
- Galizzi N., Tortoli E., Gori A., Morini F., Lapadula G. A case of mild pulmonary disease due to Mycobacterium shimoidei with a favourable outcome. Journal of Clinical Microbiology. 2013;51:3467–3468. doi: 10.1128/JCM.01028-13. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griffin A., Newton A.L., Aronson L.R., Brown D.C., Hess R.S. Disseminated Mycobacterium avium complex infection following renal transplantation in a cat. Journal of the American Veterinary Medical Association. 2003;222:1097–1101. doi: 10.2460/javma.2003.222.1097. [DOI] [PubMed] [Google Scholar]
- Griffith D.E., Aksamit T., Brown-Elliott B.A., Catanzaro A., Daley C. An official ATS/IDSA statement: diagnosis, treatment, and prevention of nontuberculous mycobacterial diseases. American Journal of Respiratory and Critical Care Medicine. 2007;174:367–416. doi: 10.1164/rccm.200604-571ST. [DOI] [PubMed] [Google Scholar]
- Gunn-Moore D.A. Feline mycobacterial infections. Veterinary Journal. 2014;201:230–238. doi: 10.1016/j.tvjl.2014.02.014. [DOI] [PubMed] [Google Scholar]
- Gunn-Moore D.A., Gaunt C., Shaw D.J. Incidence of mycobacterial infections in cats in Great Britain: estimate from feline tissue samples submitted to diagnostic laboratories. Transboundary and Emerging Diseases. 2013;60:338–344. doi: 10.1111/j.1865-1682.2012.01352.x. [DOI] [PubMed] [Google Scholar]
- Gunn-Moore D.A., McFarland S.E., Brewer J.I., Crawshaw T.R., Clifton-Hadley R.S. Mycobacterial disease in cats in Great Britain: I. Culture results, geographical distribution and clinical presentation of 339 cases. Journal of Feline Medicine & Surgery. 2011;13:934–944. doi: 10.1016/j.jfms.2011.07.012. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gunn-Moore D.A., McFarland S.E., Schock A., Brewer J.I., Crawshaw T.R. Mycobacterial disease in a population of 339 cats in Great Britain: II. Histopathology of 225 cases, and treatment and outcome of 185 cases. Journal of Feline Medicine & Surgery. 2011;13:945–952. doi: 10.1016/j.jfms.2011.09.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hetzel U., Dixon C.E., Just A.E., Birtles R.J., Kipar A. Mycobacterium malmoense as an extrapulmonary pathogen of cats. Veterinary Record. 2012;171:274. doi: 10.1136/vr.100926. [DOI] [PubMed] [Google Scholar]
- Ioannidis P., Papaventsis D., Karabela S., Nikolaou S., Panagi A. Cepheid GeneXpert MTB/RIF assay for Mycobacterium tuberculosis detection and rifampin resistance identification in patients with substantial clinical indications of tuberculosis and smear-negative microscopy results. Journal of Clinical Microbiology. 2011;49:3068–3070. doi: 10.1128/JCM.00718-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jeon M.K., Yoon J.A., Kim J., Yi S., Sung H. Lung disease caused by Mycobacterium malmoense in an immunocompetent patient. Tuberculosis and Respiratory Diseases. 2015;78:293–296. doi: 10.4046/trd.2015.78.3.293. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanaji N., Kushida Y., Bandoh S., Ishii T., Haba R. Membranous glomerulonephritis associated with Mycobacterium shimoidei pulmonary infection. American Journal of Case Reports. 2013;14:543–547. doi: 10.12659/AJCR.889684. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kipar A., Bellmann S., Kremendahl J., Köhler K., Reinacher M. Cellular composition, coronavirus antigen expression and production of specific antibodies in lesions in feline infectious peritonitis. Veterinary Immunology and Immunopathology. 1998;65:243–257. doi: 10.1016/S0165-2427(98)00158-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knippel A., Hetzel U., Baumgärtner W. Disseminated Mycobacterium avium-intracellulare infection in a Persian cat. Journal of Veterinary Medicine. B. 2004;51:464–466. doi: 10.1111/j.1439-0450.2004.00803.x. [DOI] [PubMed] [Google Scholar]
- Koh W.J. Nontuberculous mycobacteria – overview. Microbiology Spectrum. 2017;5 doi: 10.1128/microbiolspec.TNMI7-0024-2016. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee S.H., Go D.M., Woo S.H., Park H.-T., Kim E. Systemic Mycobacterium kansasii infection in a domestic shorthair cat. Journal of Comparative Pathology. 2017;157:215–219. doi: 10.1016/j.jcpa.2017.07.009. [DOI] [PubMed] [Google Scholar]
- Lloret A., Hartmann K., Pennisi M.G., Gruffydd-Jones T., Addie D. Mycobacterioses in cats: ABCD guidelines on prevention and management. Journal of Feline Medicine & Surgery. 2013;15:591–597. doi: 10.1177/1098612X13489221. [DOI] [PMC free article] [PubMed] [Google Scholar]
- MacWilliams P.S., Whitley N., Moore F. Lymphadenitis and peritonitis caused by Mycobacterium xenopi in a cat. Veterinary Clinical Pathology. 1998;27:50–53. doi: 10.1111/j.1939-165x.1998.tb01015.x. [DOI] [PubMed] [Google Scholar]
- Mauldin E.A., Peters-Kennedy J. Integumentary system. In: Maxie M.G., editor. 6th Edit. Vol. 1. Elsevier; St. Louis: 2016. pp. 639–640. (Jubb, Kennedy and Palmer's Pathology of Domestic Animals). [Google Scholar]
- Meeks C., Levy J.K., Crawford P.C., Farina L.L., Origgi F. Chronic disseminated Mycobacterium xenopi infection in a cat with idiopathic CD4+ T lymphocytopenia. Journal of Veterinary Internal Medicine. 2008;22:1043–1047. doi: 10.1111/j.1939-1676.2008.0108.x. [DOI] [PubMed] [Google Scholar]
- Morfitt D.C., Matthews J.A., Thoen C.O., Kluge J.P. Disseminated Mycobacterium avium serotype 1 infection in a seven-month-old cat. Journal of Veterinary Diagnostic Investigation. 1989;1:354–356. doi: 10.1177/104063878900100415. [DOI] [PubMed] [Google Scholar]
- Pesciaroli M., Alvarez J., Boniotti M.B., Cagiola M., Di Marco V. Tuberculosis in domestic animal species. Research in Veterinary Science. 2014;97:S78–S85. doi: 10.1016/j.rvsc.2014.05.015. [DOI] [PubMed] [Google Scholar]
- Poncelet L., Coppens A., Peeters D., Bianchi E., Grant C.K. Detection of antigenic heterogeneity in feline coronavirus nucleocapsid in feline pyogranulomatous meningoencephalitis. Veterinary Pathology. 2008;45:140–153. doi: 10.1354/vp.45-2-140. [DOI] [PubMed] [Google Scholar]
- Quinn P.J., Markey B.K., Leonard F.C., FitzPatrick E.S., Fanning S. Mycobacterium species. In: Quinn P.J., Markey B.K., Leonard F.C., FitzPatrick E.S., Fanning S., editors. Veterinary Microbiology and Microbial Disease. 2nd Edit. Blackwell Science Ltd.; Singapore: 2011. pp. 250–261. [Google Scholar]
- Raskin R.E. Skin and subcutaneous tissue. In: Raskin R.E., Meyer D.J., editors. Canine and Feline Cytology: A Color Atlas and Interpretation Guide. 2nd Edit. Saunders Elsevier; St. Louis: 2010. pp. 34–37. [Google Scholar]
- Renault C.A., Ernst J.D. Mycobacterium leprae. In: Mandell G.L., Bennet J.E., Dolin R., editors. 7th Edit. Vol. 2. Churchill Livingstone Elsevier; Philadelphia: 2010. (Mandell, Douglas, and Bennett's Principles and Practice of Infectious Diseases). 3165–3163. [Google Scholar]
- Rivière D., Pingret J.-L., Etievant M., Jechoux A., Lanore D. Disseminated Mycobacterium avium subspecies infection in a cat. Journal of Feline Medicine & Surgery. 2011;13:125–128. doi: 10.1016/j.jfms.2010.09.002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Russo C., Tortoli E., Menichella D. Evaluation of the new genotype Mycobacterium assay for identification of mycobacterial species. Journal of Clinical Microbiology. 2006;44:334–339. doi: 10.1128/JCM.44.2.334-339.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schrenzel M.D. Molecular epidemiology of mycobacteriosis in wildlife and pet animals. Veterinary Clinics of North America: Exotic Animal Practice. 2012;15:1–23. doi: 10.1016/j.cvex.2011.11.001. [DOI] [PubMed] [Google Scholar]
- Shah N.M., Davidson J.A., Anderson L.F., Lalor M.K., Kim J. Pulmonary Mycobacterium avium-intracellulare is the main driver of the rise in non-tuberculous mycobacteria incidence in England, Wales and Northern Ireland, 2007–2012. BMC Infectious Diseases. 2016;16 doi: 10.1186/s12879-016-1521-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thirunavukkarasu S., Plain K.M., de Silva K., Marais B.J., Whittington R.J. Applying the One Health concept to mycobacterial research – overcoming parochialism. Zoonoses and Public Health. 2017;64:401–422. doi: 10.1111/zph.12334. [DOI] [PubMed] [Google Scholar]
- Tuuminen T., Heinäsmäki T., Kerttula T. First report of bacteremia by Asaia bogorensis, in a patient with a history of intravenous drug abuse. Journal of Clinical Microbiology. 2006;44:3048–3050. doi: 10.1128/JCM.00521-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Turvey S.L., Tyrrell G.J., Hernandez C., Kabbani D., Duocette K. Mycobacterium branderi infection: case report and literature review of an unusual and difficult-to-treat non-tuberculous mycobacterium. International Journal of Infectious Diseases. 2017;58:65–67. doi: 10.1016/j.ijid.2017.03.002. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.