Summary
Respiratory infections are among the most important diseases of growing pigs. In order to elucidate the multifactorial aetiology of porcine respiratory disease complex (PRDC) in Denmark, lungs from 148 finishing pigs with cranioventral bronchopneumonia (case group) and 60 pigs without lung lesions (control group) were collected from abattoirs. The pathogens involved in PRDC and their interactions were identified and linked to the histopathological diagnosis. The lung samples were cultured for bacteria and tested by multiplex polymerase chain reaction for presence of swine influenza virus (type A), porcine reproductive and respiratory syndrome virus (both European and US type), porcine circovirus type 2 (PCV2), porcine respiratory coronavirus, porcine cytomegalovirus, Mycoplasma hyopneumoniae and Mycoplasma hyorhinis. All cases had cranioventral lobular bronchopneumonia consistent with PRDC. There was a broad range of microscopical lesions and the cases were characterized as acute (n = 10), subacute (n = 24) or chronic (n = 114) bronchopneumonia. Five bacterial species, five viruses and two Mycoplasma spp. were detected in different combinations. PCV2, M. hyopneumoniae, M. hyorhinis and Pasteurella multocida were detected most frequently among the PRDC affected swine and the diversity and number of pathogens were higher in these animals compared with controls. No clear-cut associations were detected between pathogens and histological lesions or histopathological diagnoses. PRDC occurs more frequently than enzootic pneumonia among Danish finishing pigs and has complex and varied histopathology.
Keywords: bronchopneumonia, histopathology, pig, porcine respiratory disease complex (PRDC)
Introduction
Respiratory infections constitute some of the most important diseases of growing pigs and result in substantial economic losses and reduced welfare (Sørensen et al., 2006). Porcine respiratory disease complex (PRDC) is a multifactorial disease of finishing pigs from 14 to 22 weeks of age (Thacker, 2001, Kim et al., 2003). Morbidity ranges from 10% to 40% and mortality from 2% to 20% (Harding and Halbur, 2002, Harms et al., 2002). Lesions are primarily located in the cranioventral parts of the lung, where consolidation, discoloration and failure of the lung tissue to collapse may be observed (Harms et al., 2002). Histopathology may vary according to the pathogens involved, but bronchopneumonia, sometimes in combination with interstitial pneumonia, is often reported (Harms et al., 2002, Kim et al., 2003). In the USA, the most commonly isolated pathogens are porcine reproductive and respiratory syndrome virus (PRRSV), swine influenza virus (SIV), porcine circovirus type 2 (PCV2), Pasteurella multocida and Mycoplasma hyopneumoniae. Other important pathogens associated with PRDC are Streptococcus suis, Actinobacillus pleuropneumoniae and [Haemophilus] parasuis (Thacker, 2001, Harms et al., 2002, Choi et al., 2003). A study from 1999 showed that 25% of Danish finishing pigs had cranioventral bronchopneumonia (CBP) at slaughter (Christensen and Enoe, 1999). Concurrent abattoir surveys in Switzerland and Belgium showed a similar prevalence (Grest et al., 1997, Maes et al., 2001), whereas earlier surveys revealed a higher prevalence, from 37% in Canada (Osborne et al., 1981), 45% in Australia (Davies et al., 1992) to 78% in another Canadian study (Wilson et al., 1986). Abattoir surveys of porcine bronchopneumonia usually focus on the gross pathology and/or the microbiology, whereas the histopathology and microbiology are seldom linked. A survey of the pathogens involved in CBP in Danish pigs has not been performed for decades and since new respiratory viruses (i.e. PCV2, porcine respiratory coronavirus [PRCV], PRRSV and new strains of SIV) have been identified in recent years, a survey of respiratory pathogens in this species has become relevant.
The aims of the present study were to elucidate the complexity of pathogens involved in PRDC and their interactions in finishing pigs, and to associate these findings with the histopathological diagnosis, in order to broaden the understanding of the pathogenesis of porcine pneumonia.
Materials and Methods
Animals
During spring 2006 and winter 2007, lungs, including tracheobronchial lymph nodes, from 148 finishing pigs with CBP (cases) and 60 pigs without gross lung lesions (controls) were collected from two Danish abattoirs in Zealand and Jutland. Cases of CBP were defined as those with lesions affecting a minimum of three cranioventral lung lobes (i.e. the apical, cardiac and intermediate lobes; Sørensen et al., 2006). The gross lesions were of acute and chronic inflammation and included consolidation, swelling, hyperaemia, failure to collapse and/or fibrosis. The samples were cooled and processed (see below) within 5 h of sampling. Tracheobronchial lymph nodes were not obtained from two animals with CBP and five control animals.
Gross Pathology
Lungs and tracheobronchial lymph nodes were evaluated macroscopically and the morphological pattern, type of exudate and estimated duration of the pulmonary lesions were recorded. A preliminary diagnosis was made based on these observations. The bronchopneumonia was of the lobular type as evidenced by sharp (lobular) delineation between lesional and non-lesional tissue. Acute lesions were oedematous, hyperaemic and swollen. Subacute lesions were those in which swelling and hyperaemia were no longer present, but in these cases there may still have been mild oedema and often marked exudation into the bronchi. Chronic lesions were characterized by atelectic, fibrotic and grey-red lung tissue with dilation of exudate-filled bronchi. Lymph nodes were scored according to their size as normal (0), moderate enlargement (1) or marked enlargement (2).
Histopathology
Tissue samples were taken from lung lesions (cases), lung tissue of normal appearance from the dorsal surface of the right diaphragmatic lobe (all controls) and from the right apical lobe (20 controls). The samples were fixed in 10% neutral buffered formalin for 24 h, embedded in paraffin wax, sectioned (3–5 μm) and stained with haematoxylin and eosin (HE). Tissue sections for immunohistochemistry (IHC) were mounted on SuperFrost®Plus slides (Mensel-gläser, Braunschweig, Germany). Selected sections were stained by Masson's trichrome and phosphotungstic acid haematoxylin to detect fibrin and collagen. IHC for cytokeratin (Soerensen et al., 2005) was applied to confirm the presence of epithelial hyperplasia, hyperplasia of type II pneumocytes and atelectasis.
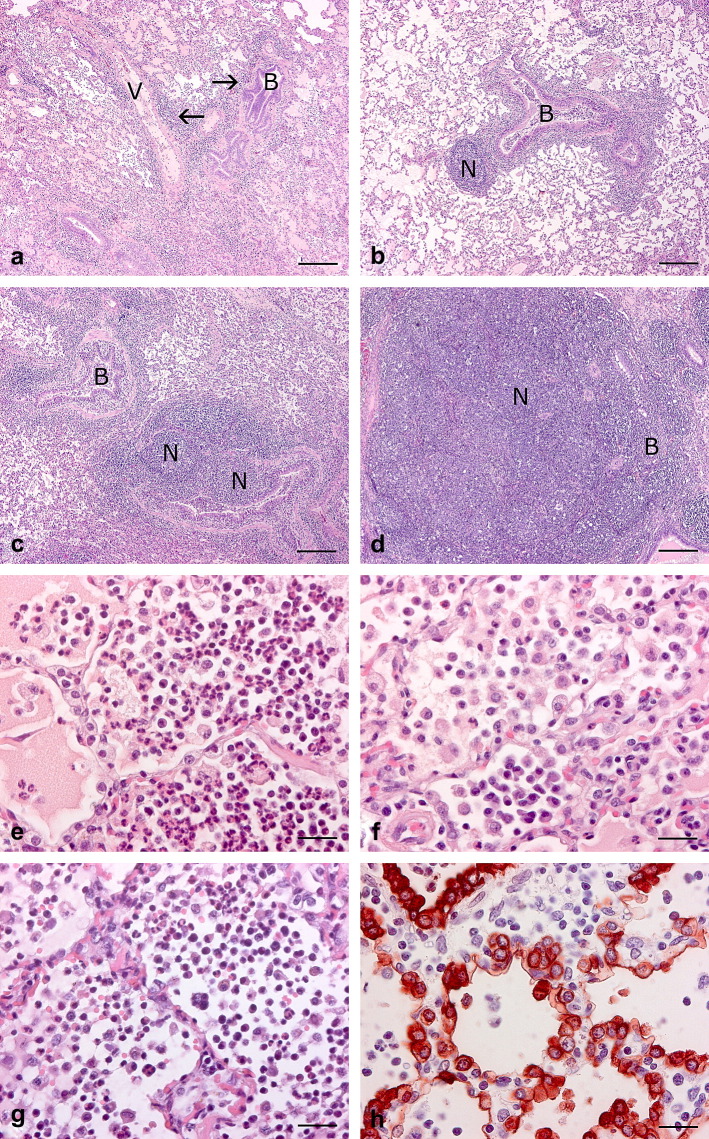
Sections were examined systematically by evaluation of the following structures in each section: bronchi, bronchioles and bronchus-associated lymphoid tissue (BALT); alveolar ducts and alveoli, including alveolar septa; peribronchial, peribronchiolar and interlobular connective tissues; and pleura. BALT hyperplasia was graded as follows (Ross, 1999): (0) absent; (+) mild diffuse infiltration of lymphocytes in the peribronchial, peribronchiolar and perivascular tissues including the lamina propria of the airways; (++) moderate increased diffuse infiltration of lymphocytes and/or presence of a few lymphoid nodules; (+++) marked number of lymphoid nodules; or (++++) extensive number of lymphoid nodules affecting most of the lung section (Fig. 1 a–d). Alveolar exudates were classified according to Bochsler and Slauson (2002) as suppurative (neutrophils dominated), non-suppurative (mononuclear inflammatory cells dominated) or mixed (intermediate grades between suppurative and non-suppurative; Fig. 1e–g). Hyperplasia of type II pneumocytes was present when these cells lined more than 3% of the alveolar surface area (Plopper and Adams, 2006) (Fig. 1h). Crowding of type II pneumocytes in close proximity to the interlobular connective tissue was not considered a significant lesion. Acute lesions were defined as those with neutrophils as the dominant inflammatory cell type, extensive oedema and/or fibrin exudation and absence of chronic signs. Chronic lesions were characterized by fibroplasias, BALT hyperplasia (of grade ++ to ++++), hyperplasia of bronchial or bronchiolar epithelium, presence of bronchiolar polyps and alveolar polyp-like structures with a fibrous core, hypertrophy of the smooth muscle layer around bronchioles and alveolar ducts, and cellular infiltrates primarily consisting of lymphocytes and plasma cells. Lesions that were not strictly acute or chronic (e.g. cellular infiltration dominated by neutrophils, combined with moderate BALT hyperplasia as the only chronic lesion) were regarded as subacute. Thus, the extent of acute versus chronic signs determined the duration of the inflammatory response. The histopathological diagnosis was regarded as the definitive diagnosis.
Fig. 1.
Porcine lung tissue with different histological lesions found in cases of CBP. (a) Mild BALT hyperplasia (+) showing diffuse infiltration of lymphocytes (arrows) into the peribronchial, peribronchiolar and perivascular tissues including the lamina propria of the airways. B, bronchiole; V, blood vessel. HE. Bar, 250 μm. (b) Moderate BALT hyperplasia (++) showing greater diffuse infiltration of lymphocytes and/or the presence of a few lymphoid nodules (N). B, bronchiole. HE. Bar, 250 μm. (c) Marked BALT hyperplasia (+++) showing a considerable number of lymphoid nodules (N). B, bronchiole. HE. Bar, 250 μm. (d) Extensive BALT hyperplasia (++++) showing an extensive number of lymphoid nodules (N) affecting most of the lung section. A compressed bronchiole (B) can be seen. HE. Bar, 250 μm. (e) Suppurative alveolar exudates showing a cellular infiltrate dominated by neutrophils in acute suppurative bronchopneumonia (ASBP). HE. Bar, 25 μm. (f) Non-suppurative exudates showing a cellular infiltrate dominated by mononuclear inflammatory cells in chronic non-suppurative bronchopneumonia (CNBP). HE. Bar, 25 μm. (g) Mixed exudates showing cellular infiltrate with both neutrophils and mononuclear cells in subacute mixed bronchopneumonia (SMBP). HE. Bar, 25 μm. (h) Marked hyperplasia of type II pneumocytes. More than 50% of the alveolar surface is lined by type II pneumocytes. IHC. Bar, 25 μm.
Microbiology
Bacterial Culture
Swabs from lung lesions (cases) and from macroscopically normal lung tissue of the right diaphragmatic lobe (controls) were taken under sterile conditions, plated on blood agar plates (blood agar base CM 55; Oxoid, Basingstoke, UK; with 5% calf blood) and incubated aerobically in a sealed plastic bag at 37°C for 18–24 h. All samples were cross-inoculated with a v-factor producing Acinetobacter calcoaeceticus. Bacterial isolates were identified using standard methods for phenotypic characterization as previously described (Barrow and Feltham, 1993). Growth of P. multocida was evaluated semi-quantitatively according to the number of bacterial colonies on the primary plates: (+) weak growth with 1–25 colonies, (++) dense growth with 25–300 colonies or (+++) marked growth with >300 colonies. Streptococcus spp. were differentiated by pulsed-field gel electrophoresis and strains representing different genotypes were identified by sequencing 16S rRNA (Chadfield et al., 2004). All v-factor-dependent cultures were also subjected to 16S rRNA sequencing to obtain a final identification. Mixed cultures demonstrating less than 25 colonies were regarded as contamination.
Mycoplasma and Virus Detection by Polymerase Chain Reaction
Tissue samples from lung lesions (cases) and macroscopically normal lung tissue (controls) were frozen at −20°C and investigated by multiplex polymerase chain reaction (PCR; Pulmotest®, Landeslabor Schleswig-Holstein, Food, Veterinary and Environmental Diagnostic Institute of Schleswig-Holstein, Neumünster, Germany), according to the method described by Palzer et al. (2007). Pathogens detected by the multiplex PCR were: SIV (influenza A), PRRSV (both European and US type), PCV2, PRCV, porcine cytomegalovirus (PCMV), M. hyopneumoniae and Mycoplasma hyorhinis.
Statistics
Data were analyzed statistically by Chi-square test or by Fisher's exact test when a small sample size necessitated this. The analysis of number of pathogens in relation to lesion duration was carried out as a Poisson regression. The degree of association was estimated by Spearman-rho factor. All analyses were done with SAS version 9.1 (SAS Institute, Inc., Cary, North Carolina) with significance of P < 0.05. Statistical analysis was not carried out on groups that included less than nine pigs and for this reason the results for PRRSV European and US types were pooled. When looking for associations between pathogens or combination of pathogens and lesion duration, type of exudate or histopathological diagnosis, we tested against the remaining cases of CBP, excluding the control animals.
Results
Gross Pathology
The gross lesions of all lungs in the case group (n = 148) were consistent with cranioventral, lobular bronchopneumonia (Fig. 2 a). Mucopurulent, purulent or seromucous exudates were found in 7% (11/148) acute, 18% (26/148) subacute and 75% (111/148) chronic cases of CBP, respectively. Focal or multifocal fibrotic pleurisy of the diaphragm lobes, without any relation to the pneumonic lesions, was present in 36% (54/148) of these animals. Except for 13% (8/60) of the control pigs, which had chronic focal pleurisy, none of the controls had gross lesions. No gross lesions were detected in the lymph nodes in animals from either group. Lymph node scores for the cases of CBP were: score 0, 3% (4/146); score 1, 66% (96/146); and score 2, 32% (46/146) and for the control pigs: score 0, 62% (34/55); score 1, 35% (19/55); and score 2, 4% (2/55). A significant association was present between lymph node enlargement and CBP (P < 0.001). When the lymph node scores were compared with the lesion duration (data not shown), no significant association was found (P = 0.16).
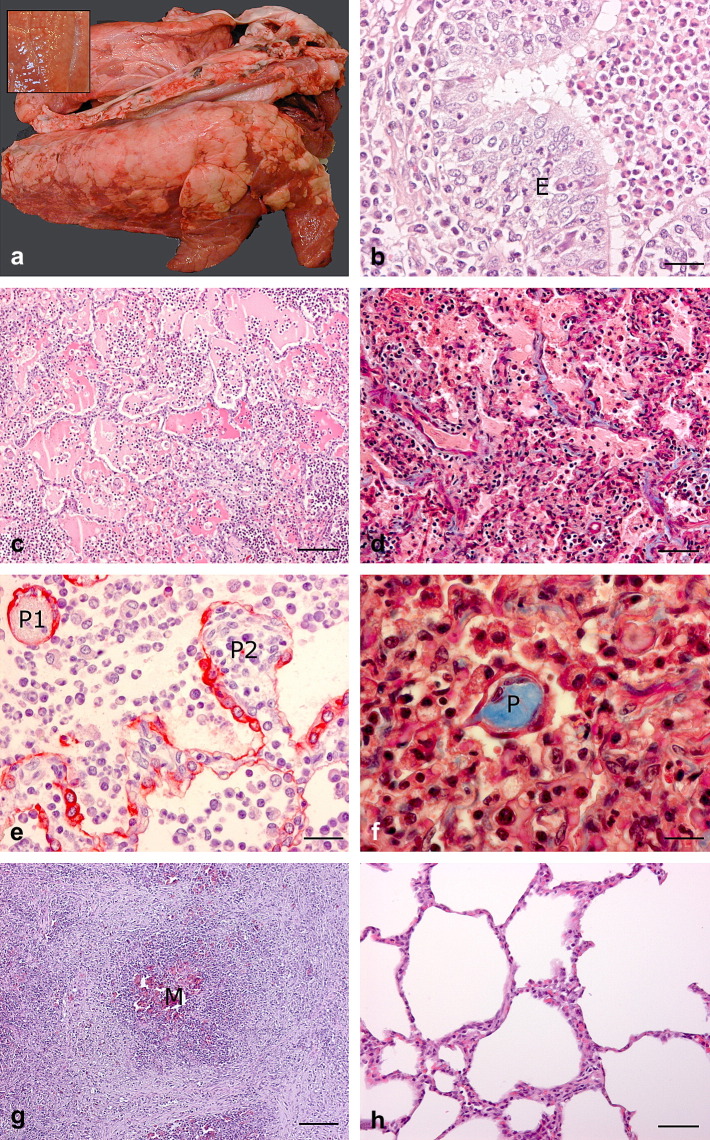
Fig. 2.
Porcine lung tissue with different lesions. (a) Gross appearance of chronic CBP. Inset shows lung tissue with a bronchial pattern of pulmonary consolidation. (b) Suppurative bronchiolitis and concurrent epithelial hyperplasia (E). HE. Bar, 25 μm. (c) Oedematous flooding of alveoli. HE. Bar, 50 μm. (d) Thickening of alveolar septa by collagenous (blue) tissue. Masson's trichrome. Bar, 50 μm. (e) Alveolar polyp-like structures, P1 with a fibrinous core and P2 with cellular infiltrate. Both are covered by type I and type II pneumocytes as shown by immunohistochemical labelling for cytokeratin expression. Bar, 25 μm. (f) Alveolar polyp-like structure (P) with a collagenous core (blue). Masson's trichrome. Bar, 15 μm. (g) Focal necrotic encapsulated tissue with central mineralization (M). HE. Bar, 100 μm. (h) Mild thickening of alveolar septa from a pig in the control group. HE. Bar, 50 μm.
Histopathology
Based on the histopathological examination, the lungs in the case group (n = 148) were divided into acute (7%, 10/148), subacute (16%, 24/148) or chronic (77%, 114/148) cases of bronchopneumonia (BP). The histopathological diagnosis (definitive diagnosis) for all of the acute cases (n = 10) was suppurative BP (ASBP). The subacute cases (n = 24) were subdivided into suppurative BP (SSBP) (67%, 16/24), mixed BP (SMBP) (25%, 6/24) or non-suppurative BP (SNBP) (8%, 2/24), and the chronic cases (n = 114) were subdivided into suppurative BP (CSBP) (46%, 52/114), mixed BP (CMBP) (32%, 37/114) or non-suppurative BP (CNBP) (22%, 25/114). The main histopathological findings are summarized in Table 1 .
Table 1.
Histopathological findings in the lungs from control animals and cases of CBP
| Control |
CBP∗ |
|||||||
|---|---|---|---|---|---|---|---|---|
| (n = 60) |
Acute (n = 10) |
Subacute (n = 24) |
Chronic (n = 114) |
|||||
| n | % | n | % | n | % | n | % | |
| Bronchitis | 1 | 2 | 3 | 30 | 3 | 12 | 14 | 12 |
| Bronchiolitis | 1 | 2 | 6 | 60 | 8 | 33 | 30 | 26 |
| Bronchiolar polyps | 0 | – | 0 | – | 2 | 8 | 21 | 18 |
| BALT hyperplasia† | ||||||||
| 0 | 41 | 68 | 4 | 40 | 2 | 8 | 14 | 12 |
| + | 15 | 25 | 6 | 60 | 4 | 17 | 26 | 23 |
| ++ | 4 | 7 | 0 | – | 16 | 67 | 52 | 46 |
| +++ | 0 | – | 0 | – | 0 | – | 12 | 11 |
| ++++ | 0 | – | 0 | – | 2 | 8 | 10 | 9 |
| Alveolar oedema | 1 | 2 | 8 | 80 | 18 | 75 | 67 | 59 |
| Fibrin exudation into alveoli | 0 | – | 5 | 50 | 7 | 29 | 10 | 9 |
| Type II pneumocyte hyperplasia | 3 | 5 | 3 | 30 | 7 | 29 | 40 | 35 |
| Thickening of alveolar septa | 16 | 27 | 1 | 10 | 8 | 33 | 28 | 25 |
| Alveolar polyp-like structures | 0 | – | 2 | 20 | 9 | 38 | 30 | 26 |
| Smooth-muscle hyperplasia | 2 | 3 | 1 | 10 | 3 | 13 | 31 | 27 |
| Interstitial oedema | 2 | 3 | 7 | 70 | 10 | 42 | 34 | 30 |
| Interstitial fibroplasia | 2 | 3 | 1 | 10 | 5 | 21 | 34 | 30 |
| Necrotic foci | 0 | – | 0 | – | 1 | 4 | 8 | 7 |
Cases of CBP were grouped as acute, subacute or chronic based on histological evaluation.
Hyperplasia of the BALT was scored as absent (0), mild (+), moderate (++), marked (+++) or extensive (++++).
Microscopical lesions were found in the alveoli of all lungs with CBP. The majority also had lesions affecting bronchioles and/or alveolar ducts, often together with lesions in the interlobular connective tissue. Bronchitis and bronchiolitis were mainly suppurative, with concurrent epithelial hyperplasia in the subacute and chronic cases (Fig. 2b). In cases of bronchiolitis, destruction of the epithelial lining in the bronchioles was observed in 17% (1/6), 50% (4/8) and 37% (11/30) of the acute, subacute and chronic cases of CBP, respectively. The main finding in the alveoli was infiltration of inflammatory cells (neutrophils, lymphocytes and plasma cells), reflecting the duration of the inflammatory response, whereas macrophages were identified at all stages. Oedematous flooding was a common finding in the alveoli in acute, subacute and chronic cases of CBP, with the degree of oedema being more pronounced in acute stages (Fig. 2c). Varying degrees of thickening of the alveolar septa, mainly by collagenous tissue, were seen in 25% (37/148), of cases of CBP (Fig. 2d). Alveolar polyp-like structures were seen in acute, subacute and chronic bronchopneumonia, whereas bronchiolar polyps only occurred in subacute and chronic lesions. Alveolar polyp-like structures with a fibrinous core could be observed in both acute and chronic cases, whereas polyps consisting of collagen were not present in acute cases (Fig. 2e,f). The major findings in the interlobular connective tissues were oedema, diffuse lymphocyte infiltration and/or fibroplasia. Other findings included hypertrophy of the muscles around bronchioles and alveolar ducts and hyperplasia of type II pneumocytes. Occasional findings were vasculitis, thrombosis and areas of necrosis with or without mineralization (Fig. 2g).
Not all combinations of these lesions were seen in the lung sections. The lungs from three animals, diagnosed as acute (n = 1) or subacute (n = 2) according to gross pathology, were definitely diagnosed as chronic on the basis of histopathology. Microscopical lung lesions were seen in 50% (30/60) of the control animals and no difference was observed between the samples from the diaphragmatic and the cranioventral lobes. The findings in these animals mainly included focal or multifocal thickening of alveolar septa (Fig. 2h) and/or mild BALT hyperplasia (Table 1).
Microbiology
A total of 12 potential pathogens (five bacterial species, five viruses and two Mycoplasma spp.) were detected and these are listed in Table 2 . In general, the frequencies of the pathogens were higher in the case group compared with controls, except for PCMV, which was more frequent in the controls (though not significant). PRCV was not detected in any of the pigs. Staphylococcus aureus, [H.] parasuis, A. pleuropneumoniae and SIV-A were only detected in one to five animals. S. aureus, [H.] parasuis, A. pleuropneumoniae, PRRSV European and US type were only found in the cases of CBP. When comparing cases and controls according to pathogens (Table 2), all grades of P. multocida (P < 0.001–P = 0.004), M. hyopneumoniae (P < 0.001) and M. hyorhinis (P < 0.001) were found more frequently in the cases of CBP. Furthermore, the finding of mixed cultures and sterile samples was more frequent in the control group than in the case group (P < 0.001). Significant associations were observed between eight pairs of pathogens among the diseased animals and between two pathogens in the control group (Table 3 ).
Table 2.
Bacterial culture and multiplex PCR for viruses and Mycoplasma
| Pigs with CBP (n = 148) |
Control animals (n = 60) |
P value∗ | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Acute |
Subacute |
Chronic |
|||||||||||||||
| ASBP (n = 10) |
SSBP (n = 16) |
SMBP (n = 6) |
SNBP (n = 2) |
CSBP (n = 52) |
CMBP (n = 37) |
CNBP (n = 25) |
|||||||||||
| n | % | n | % | n | % | n | % | n | % | n | % | n | % | n | % | ||
| Bacterial culture | |||||||||||||||||
| P. multocida† | |||||||||||||||||
| + | 0 | – | 2 | 13 | 1 | 17 | 0 | – | 4 | 8 | 4 | 11 | 6‡ | 24 | 0 | – | 0.004 |
| ++ | 2 | 20 | 5‡ | 31 | 3‡ | 50 | 1 | 50 | 5 | 10 | 2 | 5 | 3 | 12 | 1 | 2 | 0.004 |
| +++ | 7 | 70 | 8 | 50 | 1 | 17 | 1 | 50 | 31 | 60 | 18 | 49 | 10 | 40 | 1 | 2 | <0.001 |
| S. suis | 0§ | – | 4 | 25 | 2 | 33 | 0 | – | 7 | 13 | 8 | 22 | 6 | 24 | 6 | 10 | NS |
| S. aureus | 0 | – | 0 | – | 0 | – | 1 | 50 | 0 | – | 0 | – | 0 | – | 0 | – | NS |
| [H.] parasuis | 0 | – | 0 | – | 0 | – | 0 | – | 2 | 4 | 2 | 5 | 1 | 4 | 0 | – | NS |
| A. pleuropneumoniae | 0 | – | 0 | – | 0 | – | 0 | – | 1 | 2 | 0 | – | 0 | – | 0 | – | NS |
| Mixed culture | 2 | 20 | 0 | – | 0 | – | 0 | – | 0 | – | 4 | 11 | 0 | – | 27 | 45 | <0.001 |
| Sterile | 0 | – | 0 | – | 0 | – | 0 | – | 5 | 10 | 3 | 8 | 4 | 16 | 25 | 42 | <0.001 |
| Multiplex PCR | |||||||||||||||||
| PCV2 | 10 | 100 | 14 | 88 | 6 | 100 | 2 | 100 | 50 | 96 | 37 | 100 | 24 | 96 | 56 | 93 | NS |
| PRRSV | |||||||||||||||||
| EU type | 1 | 10 | 3 | 19 | 0 | – | 0 | – | 2 | 4 | 1 | 3 | 1 | 4 | 0 | – | NS |
| US type | 0 | – | 0 | – | 0 | – | 0 | – | 1 | 2 | 0 | – | 0 | – | 0 | – | NS |
| PCMV | 3 | 30 | 3 | 19 | 1 | 17 | 0 | – | 15 | 29 | 7 | 19 | 6 | 24 | 26 | 43 | NS |
| SIV-A | 0 | – | 0 | – | 0 | – | 0 | – | 0 | – | 2 | 5 | 1 | 4 | 1 | 2 | NS |
| M. hyopneumoniae | 10 | 100 | 15 | 94 | 6 | 100 | 1 | 50 | 51 | 98 | 35 | 95 | 24 | 96 | 21 | 37 | <0.001 |
| M. hyorhinis | 9 | 90 | 13 | 81 | 6 | 100 | 2 | 100 | 41 | 79 | 30 | 81 | 17 | 68 | 22 | 35 | <0.001 |
Numbers and percentage of positive animals are listed for the 148 pigs with CBP in relation to diagnosis.
ASBP, acute suppurative bronchopneumonia (BP); SSBP, subacute suppurative BP; SMBP, subacute mixed BP; SNBP, subacute non-suppurative BP; CSBP, chronic suppurative BP; CNBP, chronic non-suppurative BP; CMBP, chronic mixed BP. PRRSV (EU type, European type; US type, North American type); SIV-A, swine influenza virus type A; NS, not significant; PCV2, porcine circovirus type 2; PCMV, porcine cytomegalovirus.
P values for differences between cases and controls were calculated by either the Chi-square or Fischer's exact test as appropriate.
Growth of P. multocida was graded as weak growth (+), dense growth (++) or marked growth (+++).
The frequency of each pathogen detected in the diagnosis group was tested against the remaining cases of CBP by the Chi-square or Fischer's exact test. Significant associations are marked by ‘‡’.
Table 3.
Significant associations between pairs of pathogens
| Associated pathogens | P value∗ | Spearman-rho factor |
|---|---|---|
| Animals with CBP (n = 148) | ||
| PCV2 and P. multocida (+)† | 0.017 | 0.067 |
| PCV2 and S. suis | 0.021 | 0.088 |
| PCV2 and M. hyopneumoniae | 0.012 | −0.038 |
| PCV2 and PRRSV | 0.013 | 0.048 |
| PCV2 and PCMV | 0.025 | 0.104 |
| P. multocida (++) and M. hyorhinis | 0.050 | 0.157 |
| S. suis and M. hyopneumoniae | 0.022 | 0.097 |
| PRRSV and M. hyopneumoniae | 0.014 | 0.052 |
| Control animals (n = 60) | ||
| PCV2 and S. suis | 0.032 | 0.105 |
P values were tested by the Fischer's exact test and the degree of association was calculated by the Spearman-rho factor-test.
Weak growth of P. multocida on the plate (+).
A total of 63 different combinations of pathogens were present, many of which were only found in a single animal (n = 34; data not shown). Among the cases of CBP and the control animals there were 51 and 23 different combinations of pathogens, respectively. The 10 most frequent combinations are presented in Table 4 . Some combinations were seen in either diseased or healthy animals. The three most frequent combinations, only isolated from cases of CBP, were combinations 1, 6 and 10 (Table 4). Likewise, the two most frequent combinations, only seen among the control animals, were mixed culture and PCV2, and PCV2 as a solitary pathogen.
Table 4.
The 10 most frequent combinations of pathogens among cases of CBP and control animals
| Combination number |
||||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | |
| Mixed culture | × | |||||||||
| P. multocida + | × | |||||||||
| P. multocida ++ | × | |||||||||
| P. multocida +++ | × | × | × | |||||||
| S. suis | × | |||||||||
| PCMV | × | × | × | |||||||
| PCV2 | × | × | × | × | × | × | × | × | × | × |
| M. hyopneumoniae | × | × | × | × | × | × | × | |||
| M. hyorhinis | × | × | × | × | × | × | ||||
| Frequency of combination (n=)∗ | ||||||||||
| CBP | ||||||||||
| Acute | 3 | – | 3† | 1 | – | 1 | – | – | – | – |
| Subacute | 4 | 1 | 3 | 3 | – | – | – | – | – | 1 |
| Chronic | 33 | 9 | 8 | 6 | 1 | 8 | – | – | 3 | 4 |
| Controls | – | 4 | 1 | 1 | 8 | – | 9 | 7 | 2 | – |
Growth of P. multocida defined as weak (+), dense (++) or marked (+++).
Number of animals with a given combination of pathogens.
Significant association.
Associations between Microbiological and Histopathological Findings
When testing associations of selected histological lesions (bronchiolar polyps, BALT hyperplasia, fibrin exudation into alveoli, hyperplasia of type II pneumocytes, thickening of alveolar septa, alveolar polyp-like structures, necrotic foci) within the case group, and subsequently within lesion duration groups, to pathogens, significant results were obtained for the following: in chronic cases with bronchiolar polyps, P. multocida (+++) was isolated more frequently (P = 0.016); BALT hyperplasia (grade ++ to ++++) was associated with PCMV in chronic cases (P < 0.001); fibrin exudation into alveoli was associated with the isolation of P. multocida (++) (P = 0.018); thickening of alveolar septa was never seen when P. multocida (+) was isolated (P = 0.013). Furthermore, in 78% (7/9) of cases with necrotic foci, P. multocida (+++) was present, although with an association of only borderline significance (P = 0.062). No association between histological lesions and pathogens was found in the control group.
Table 2 presents the frequencies of single pathogens, divided according to the diagnosis groups. P. multocida was, as the only pathogen, isolated more frequently from the following groups: P. multocida (++) and SSBP (P = 0.054); P. multocida (++) and SMBP (P = 0.037); and P. multocida (+) and CNBP (P = 0.043). S. suis was not isolated from any case of ASBP (P = 0.021). Significant associations were not seen when comparing single pathogens and type of exudate (suppurative, mixed and non-suppurative) or lesion duration (acute, subacute and chronic).
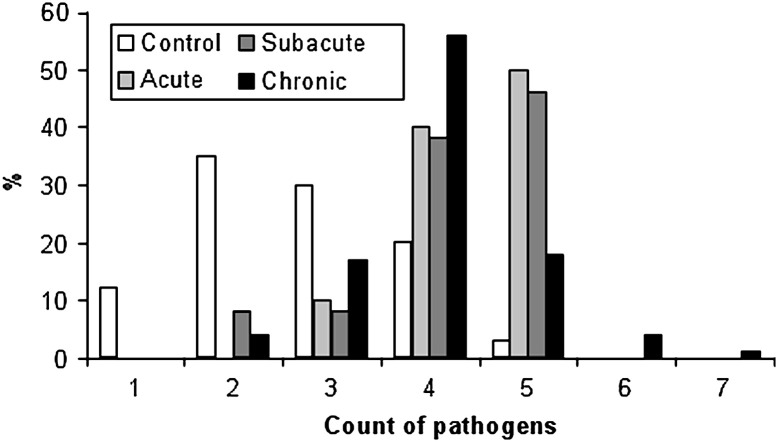
In Fig. 3 the number of pathogens detected in each pig is listed for the lesion duration groups and the control group. At least one pathogen was detected in all lung samples (cases and controls). Detection of a single pathogen (PCV2) occurred in seven animals, all from the control group. The majority of pigs in the control group had a significantly lower number of pathogens, while cases of CBP had increased counts (P < 0.001). Between lesion duration groups there was a tendency to a lower count in the chronic cases compared with acute and subacute cases; however, this difference was not significant.
Fig. 3.
The frequency of pathogen count detected in control animals (n = 60) and in animals with CBP of acute (n = 10), subacute (n = 24) or chronic (n = 114) type.
When comparing the 10 most frequent combinations of pathogens and lesion duration groups (Table 4) the only significant association was combination number 3, which was seen more frequently in the acute cases (30%, 3/10) (P = 0.044) than in the subacute (13%, 3/24) and chronic cases (7%, 8/114). There were no significant associations when comparing these combinations of pathogens with types of exudate or histopathological diagnosis (data not shown).
Discussion
Pneumonia can be classified as embolic, bronchopneumonia and interstitial or bronchointerstitial (Caswell and Williams, 2007) based on the morphological pattern observed in experimental studies and/or infections with single pathogens. The present study of naturally occurring pneumonia recorded a range of pneumonic lesions, but the predominant pattern was bronchopneumonia associated with thickening of the alveolar septa. In some animals chronic lesions were accompanied by acute lesions, which may represent the healing of acute pneumonic lesions or the presence of two different disease incidents. These cases of bronchopneumonia were also characterized microscopically according to the estimated duration of the disease process (i.e. as acute, subacute or chronic). Few previous studies have focused on the histopathological findings in porcine lungs with naturally occurring PRDC (Harms et al., 2002), whereas many studies describe naturally occurring gross lesions or the histopathology of experimental coinfections. The pigs examined by Harms et al. (2002) had clinical signs of respiratory disease and therefore the lesions observed in that study were more severe than in the pigs of the present investigation.
The results of the present study show that the histopathology of PRDC is complex, as nearly all pulmonary reaction patterns can co-exist. Thickened alveolar septa were observed in pigs from both control and case groups, indicating that this finding is not entirely related to bronchopneumonia. This lesion may therefore be regarded as a pulmonary response to environmental factors associated with swine production (e.g. dust and ammonia). Furthermore, thickening of alveolar septa was not associated with the presence of specific pathogens. Type II pneumocyte hyperplasia occurred more frequently in pigs with CBP compared with controls; however, the lesion did not appear to be associated with the duration of disease, since it was present in all diagnosis groups. In addition to the bronchiolar polyps in the pigs with pneumonic lesions, there were also structures that resembled alveolar polyps, (i.e. they protruded from the epithelial surface in the alveoli and were covered by epithelium). Three different forms of alveolar polyp-like structures were identified (fibrinous, cell-infiltrated fibrinous and collagenous) and these probably represented different stages of the same initial lesion. Alveolar polyp-like structures have not been described previously in pigs and their significance is undetermined.
Most authors define PRDC as a multifactorial respiratory disease involving several pathogens (Harms et al., 2002, Kim et al., 2003, Opriessnig et al., 2007, Fachinger et al., 2008), whereas Thacker (2006) specifies PRDC as enzootic pneumonia (i.e. infection with Mycoplasma spp. and opportunistic bacteria) aggravated by respiratory viruses. Except for 14 animals, Mycoplasma spp., bacteria and respiratory viruses were identified in all cases of CBP in the present study. This pathogen profile, combined with the pathological findings, is in accordance with the diagnosis of PRDC, although it is not possible to determine the order of infection. Based on the pathogen combination, ‘classical’ enzootic pneumonia was only present in five animals with CBP, which indicates that PRDC is more prevalent in Danish finishing pigs compared with enzootic pneumonia. This probably corresponds to the findings in other countries, where a mixture of Mycoplasma spp., bacteria and viruses are identified in the majority of porcine pneumonias (Harms et al., 2002, Kim et al., 2003, Palzer et al., 2007).
Twelve different pathogens were obtained from the lungs and the spectrum of pathogens correlated with observations made in other countries (Chiou et al., 2004, Palzer et al., 2008). The following potential pathogens (Thacker, 2001, Liljegren et al., 2003) were only present in the case group: S. aureus, [H.] parasuis, A. pleuropneumonia and PRRSV (European and US type). Therefore, these agents might represent more aggressive pathogens in cases of PRDC, although the difference in frequency between cases and controls was not significant. The diversity and number of pathogens were higher among the diseased animals compared with the control group, supporting the definition of PRDC as a multifactorial disease (Harms et al., 2002), which should be considered when diagnosing and treating cases of PRDC and in prophylactic strategies. The observed tendency towards a lower number of pathogens in the chronic cases of CBP (Fig. 3), compared with the acute and subacute cases, could be explained by regression of the infection and elimination of pathogens.
The pathogens detected most frequently from the PRDC affected swine were PCV2, M. hyopneumoniae, M. hyorhinis and P. multocida. These pathogens were also found, but in different proportions, from pigs with pneumonia in Germany (Palzer et al., 2008), Taiwan (Chiou et al., 2004) and the USA (Choi et al., 2003). The divergence in ratio of pathogens between these studies depends on the identification methods used, differences in health status of the animals, management factors of the farms and seasonal variation, or it may simply reflect the complex nature of pneumonia in swine (Sørensen et al., 2006).
In agreement with other studies (Chiou et al., 2004, Palzer et al., 2008), with the exception of results from the USA (Choi et al., 2003), PCV2 was detected in the majority of the lung samples in the present study. The mere identification of PCV2 by PCR is not regarded as diagnostic for PCV2-associated disease (i.e. porcine circovirus disease), therefore the animals in the present study may have been subclinically infected (Opriessnig et al., 2007). PCV2 has the potential to reduce acquired immunity to other pathogens (Opriessnig et al., 2006) and vaccination against PCV2 reduced co-infections by secondary pathogens and lowered the incidence of pulmonary coinfections (Fachinger et al., 2008). In recent years it has been speculated that PCV2 plays a part in PRDC and this is now widely accepted (Harms et al., 2002, Thacker, 2006). In the present study, PCV2 was part of the majority of significant associations between pairs of pathogens in the cases of CBP (Table 3). Except for M. hyopneumoniae, PCV2 and the associated pathogens had a positive influence on each other (i.e. the presence of one increased the chance of finding the other). Although PCV2 was also detected in samples from most control animals, there was only a single significant association between PCV2 and other pathogens in this group. These findings indicate that PCV2 could be involved in PRDC in Danish finishing pigs, preparing the way for other respiratory pathogens or occurring as a secondary invader.
M. hyopneumoniae was significantly more prevalent in cases of CBP compared with controls. This organism is an important pathogen in porcine pneumonia, able to cause disease on its own and in combination with other pathogens (Sørensen et al., 1997, Thacker, 2004). Co-occurrence was seen between M. hyopneumoniae and PRRSV or S. suis, which is in accordance with other reports on pathogen interaction in respiratory disease (Thacker, 2001, Thacker, 2004). The negative association found between M. hyopneumoniae and PCV2 is of unknown significance. Recent studies indicate that M. hyorhinis is also important in enzootic pneumonia and respiratory disease in general (Falk et al., 1991, Kawashima et al., 1996, Lin et al., 2006). In the present study, the isolation of M. hyorhinis in a significantly higher frequency from the CBP affected pigs indicates that this pathogen does have an effect on the development of PRDC in Danish swine.
Confirming previous results (Gois et al., 1975, Falk et al., 1991), P. multocida was isolated from approximately 80% of the cases of CBP, which was significantly higher than in the control group. Furthermore, P. multocida was found with increased frequency in some subacute (SSBP, SMBP) and chronic (CNBP) lesions compared with other diagnosis groups. P. multocida was also significantly associated with histological lesions. This indicates that P. multocida most likely is a secondary invading pathogen and aggravates the disease in cases of CBP, which is in agreement with the literature (Morrison et al., 1985, Hoie et al., 1991, Ross, 2006). The finding of P. multocida in association with necrotic lesions has also been reported in both naturally and experimentally infected pigs (Berndt et al., 2002, Ono et al., 2003, Pijoan, 2006). In the present study there was a significant co-occurrence between P. multocida and M. hyorhinis in natural cases of CBP, which is in accordance with field studies by Falk et al. (1991).
S. suis is a natural inhabitant of the porcine respiratory tract, but is also associated with suppurative bronchopneumonia; however, whether this organism is a primary or secondary pathogen is still to be determined (Thacker, 2001, Higgins and Gottschalk, 2006). Although not significant, S. suis was isolated from a higher proportion of PRDC affected animals (45%) than controls (13%), suggesting that S. suis is involved in PRDC. S. suis was isolated from significantly fewer acute cases (ASBP) compared with subacute and chronic cases, suggesting that it is a secondary invading pathogen. Furthermore, co-occurrence was detected between S. suis and M. hyopneumoniae or PCV2, which supports the role of S. suis as an opportunistic pathogen.
PCMV mainly causes rhinitis and pneumonia in nursery pigs, but it has been associated with respiratory disease in pigs of other ages (Orr et al., 1988, Yoon and Edington, 2006). In the present study, PCMV was the only pathogen isolated at a higher frequency from the control animals (43%) compared with the PRDC affected animals (24%), but a significant association between PCMV and PCV2 was only found in the cases of PRDC. Among the pathogen combinations, combination number 3 (P. multocida, PCMV, PCV2, M. hyopneumoniae and M. hyorhinis) was seen more frequently in acute cases of CBP than in subacute and chronic cases. This combination only differed from combination numbers 1 and 4 by the presence of PCMV, thus it can be speculated that presence of PCMV causes exacerbation of the disease. To our knowledge PCMV has not been associated with PRDC before, but the role of PCMV in PRDC is controversial and needs to be elucidated.
In conclusion, PRDC occurs more frequently than enzootic pneumonia among Danish finishing pigs. The previously reported pathogen profile and multifactorial aetiology of PRDC are confirmed by the results of the present study. Furthermore, PCV2, S. suis and M. hyorhinis are associated with PRDC, and S. suis and P. multocida are likely to be involved as secondary pathogens. The role of PCMV in PRDC should be further elucidated. The histopathology of PRDC is complex and nearly all pulmonary reaction patterns can be observed in combination.
Acknowledgments
The authors would like to acknowledge B. Andersen, L. Kioerboe, H. H. Moeller and K. Madsen for excellent technical assistance. The authors would also like to thank ELANCO, Eli Lilly Denmark A/S, who funded part of this project.
References
- Barrow G.I., Feltham R.K.A. 3rd Edit. Cambridge University Press; Cambridge: 1993. Cowan and Steel's Manual for the Identification of Medical Bacteria. pp. 50–164. [Google Scholar]
- Berndt A., Heller M., Kosmehl H. Cytokine mRNA expression in experimental porcine pneumonia. Deutsche Tierarztliche Wochenschrift. 2002;109:205–209. [PubMed] [Google Scholar]
- Bochsler P.N., Slauson D.O. Inflammation and repair of tissue. In: Slauson D.O., Cooper B.J., editors. Mechanisms of Disease. A Textbook of Comparative General Pathology. 3rd Edit. Mosby Inc.; St. Louis: 2002. pp. 140–245. [Google Scholar]
- Caswell J.L., Williams K.J. Respiratory system. In: Maxie M.G., editor. Jubb, Kennedy, and Palmer's Pathology of Domestic Animals. 5th Edit. Saunders Elsevier; Philadelphia: 2007. pp. 523–653. [Google Scholar]
- Chadfield M.S., Christensen J.P., Christensen H., Bisgaard M. Characterization of streptococci and enterococci associated with septicaemia in broiler parents with a high prevalence of endocarditis. Avian Pathology. 2004;33:610–617. doi: 10.1080/03079450400013089. [DOI] [PubMed] [Google Scholar]
- Chiou MT, Lin CN, Chuang MS, Chiang HC, Tsai CP (2004) Etiological and epidemiological survey of PRDC associated pathogens in Taiwan. In: Proceedings of the 18th International Pig Veterinary Society Congress, Hamburg, p. 191.
- Choi Y.K., Goyal S.M., Joo H.S. Retrospective analysis of etiologic agents associated with respiratory diseases in pigs. Canadian Veterinary Journal. 2003;44:735–737. [PMC free article] [PubMed] [Google Scholar]
- Christensen G., Enoe C. The prevalence of pneumonia, pleuritis, pericarditis and liver spots in Danish slaughter pigs in 1998, including comparation with 1994. Dansk Veterinaertidsskrift. 1999;82:1006–1015. [Google Scholar]
- Davies PR, Moore MJ, Pointon AM (1992) The association between lesions of pleuritis and enzootic pneumonia in slaughtered pigs. In: Proceedings of the 12th International Pig Veterinary Society Congress, The Hague, p. 306.
- Fachinger V., Bischoff R., Jedidia S.B., Saalmuller A., Elbers K. The effect of vaccination against porcine circovirus type 2 in pigs suffering from porcine respiratory disease complex. Vaccine. 2008;26:1488–1499. doi: 10.1016/j.vaccine.2007.11.053. [DOI] [PubMed] [Google Scholar]
- Falk K., Hoie S., Lium B.M. An abattoir survey of pneumonia and pleuritis in slaughter weight swine from 9 selected herds. II. Enzootic pneumonia of pigs: microbiological findings and their relationship to pathomorphology. Acta Veterinaria Scandinavica. 1991;32:67–77. doi: 10.1186/BF03546998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gois M., Sisak F., Kuksa F., Sovadina M. Incidence and evaluation of the microbial flora in the lungs of pigs with enzootic pneumonia. Zentralblatt fur Veterinarmedizin. Reihe B. 1975;22:205–219. doi: 10.1111/j.1439-0450.1975.tb00581.x. [DOI] [PubMed] [Google Scholar]
- Grest P., Keller H., Sydler T., Pospischil A. The prevalence of lung lesions in pigs at slaughter in Switzerland. Schweizer Archiv fur Tierheilkunde. 1997;139:500–506. [PubMed] [Google Scholar]
- Harding JC, Halbur PG (2002) PMWS or a group of PCV2-associated syndromes: ever-growing concerns. In: Proceedings of the 17th International Pig Veterinary Society Congress. PMWS and PCV2 Diseases, Beyond the Debate, Keynotes on the Merial Symposium and Brief Epidemiological Updates, Ames, pp. 19–31.
- Harms P.A., Halbur P.G., Sorden S.D. Three cases of porcine respiratory disease complex associated with porcine circovirus type 2 infection. Journal of Swine Health and Production. 2002;10:27–30. [Google Scholar]
- Higgins R., Gottschalk M. Streptococcal diseases. In: Straw B.E., Zimmerman J.J., D'Allaire S., Taylor D.J., editors. Diseases of Swine. 9th Edit. Blackwell Publishing; Oxford: 2006. pp. 769–783. [Google Scholar]
- Hoie S., Falk K., Lium B.M. An abattoir survey of pneumonia and pleuritis in slaughter weight swine from 9 selected herds. IV. Bacteriological findings in chronic pneumonic lesions. Acta Veterinaria Scandinavica. 1991;32:395–402. doi: 10.1186/BF03546970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawashima K., Yamada S., Kobayashi H., Narita M. Detection of porcine reproductive and respiratory syndrome virus and Mycoplasma hyorhinis antigens in pulmonary lesions of pigs suffering from respiratory distress. Journal of Comparative Pathology. 1996;114:315–323. doi: 10.1016/S0021-9975(96)80053-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kim J., Chung H.K., Chae C. Association of porcine circovirus 2 with porcine respiratory disease complex. Veterinary Journal. 2003;166:251–256. doi: 10.1016/s1090-0233(02)00257-5. [DOI] [PubMed] [Google Scholar]
- Liljegren C.H., Aalbaek B., Nielsen O.L., Jensen H.E. Some new aspects of the pathology, pathogenesis, and aetiology of disseminated lung lesions in slaughter pigs. Acta Pathologica, Microbiologica et Immunologica Scandinavica. 2003;111:531–538. doi: 10.1034/j.1600-0463.2003.1110501.x. [DOI] [PubMed] [Google Scholar]
- Lin J.H., Chen S.P., Yeh K.S., Weng C.N. Mycoplasma hyorhinis in Taiwan: diagnosis and isolation of swine pneumonia pathogen. Veterinary Microbiology. 2006;115:111–116. doi: 10.1016/j.vetmic.2006.02.004. [DOI] [PubMed] [Google Scholar]
- Maes D.G., Deluyker H., Verdonck M., Castryck F., Miry C. Non-infectious factors associated with macroscopic and microscopic lung lesions in slaughter pigs from farrow-to-finish herds. Veterinary Record. 2001;148:41–46. doi: 10.1136/vr.148.2.41. [DOI] [PubMed] [Google Scholar]
- Morrison R.B., Pijoan C., Hilley H.D., Rapp V. Microorganisms associated with pneumonia in slaughter weight swine. Canadian Journal of Comparative Medicine. 1985;49:129–137. [PMC free article] [PubMed] [Google Scholar]
- Ono M., Okada M., Namimatsu T., Fujii S., Mukai T. Septicaemia and arthritis in pigs experimentally infected with Pasteurella multocida capsular serotype A. Journal of Comparative Pathology. 2003;129:251–258. doi: 10.1016/s0021-9975(03)00040-9. [DOI] [PubMed] [Google Scholar]
- Opriessnig T., McKeown N.E., Harmon K.L., Meng X.J., Halbur P.G. Porcine circovirus type 2 infection decreases the efficacy of a modified live porcine reproductive and respiratory syndrome virus vaccine. Clinical and Vaccine Immunology. 2006;13:923–929. doi: 10.1128/CVI.00074-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Opriessnig T., Meng X.J., Halbur P.G. Porcine circovirus type 2 associated disease: update on current terminology, clinical manifestations, pathogenesis, diagnosis, and intervention strategies. Journal of Veterinary Diagnostic Investigation. 2007;19:591–615. doi: 10.1177/104063870701900601. [DOI] [PubMed] [Google Scholar]
- Orr J.P., Althouse E., Dulac G.C., Durham P.J. Epizootic infection of a minimal disease swine herd with a herpesvirus. Canadian Veterinary Journal. 1988;29:45–50. [PMC free article] [PubMed] [Google Scholar]
- Osborne A.D., Saunders J.R., Sebunya T. An abattoir survey of the incidence of pneumonia in Saskatchewan swine and an investigation of the microbiology of affected lungs. Canadian Veterinary Journal. 1981;22:82–85. [PMC free article] [PubMed] [Google Scholar]
- Palzer A., Ritzmann M., Majzoub M., Wolf G., Hermanns W. Frequency of occurrence of pneumonia associated agents and their correlation with clinical and pathological-anatomical findings in pigs. Berliner und Munchener Tierarztliche Wochenschrift. 2007;120:483–489. [PubMed] [Google Scholar]
- Palzer A., Ritzmann M., Wolf G., Heinritzi K. Associations between pathogens in healthy pigs and pigs with pneumonia. Veterinary Record. 2008;162:267–271. doi: 10.1136/vr.162.9.267. [DOI] [PubMed] [Google Scholar]
- Pijoan C. Pneumonic pasteurellosis. In: Straw B.E., Zimmerman J.J., D'Allaire S., Taylor D.J., editors. Diseases in Swine. 9th Edit. Iowa State University Press; Ames: 2006. pp. 719–726. [Google Scholar]
- Plopper C.G., Adams D.R. Respiratory system. In: Eurell J.A., Frappier B.L., editors. Dellmann's Textbook of Veterinary Histology. 6th Edit. Blackwell Publishing; Iowa: 2006. pp. 153–169. [Google Scholar]
- Ross R.F. Mycoplasmal diseases. In: Straw B.E., D'Allaire S., Mengeling W.L., Taylor D.J., editors. Diseases of Swine. 8th Edit. Blackwell Science; Oxford: 1999. pp. 495–509. [Google Scholar]
- Ross R.F. Pasteurella multocida and its role in porcine pneumonia. Animal Health Research Reviews. 2006;7:13–29. doi: 10.1017/S1466252307001211. [DOI] [PubMed] [Google Scholar]
- Sørensen V., Ahrens P., Barfod K., Feenstra A.A., Feld N.C. Mycoplasma hyopneumoniae infection in pigs: duration of the disease and evaluation of four diagnostic assays. Veterinary Microbiology. 1997;54:23–34. doi: 10.1016/s0378-1135(96)01266-7. [DOI] [PubMed] [Google Scholar]
- Soerensen C.M., Holmskov U., Aalbaek B., Boye M., Heegaard P.M. Pulmonary infections in swine induce altered porcine surfactant protein D expression and localization to dendritic cells in bronchial-associated lymphoid tissue. Immunology. 2005;115:526–535. doi: 10.1111/j.1365-2567.2005.02189.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sørensen V., Jorsal S.E., Mousing J. Diseases of the respiratory system. In: Straw B.E., Zimmerman J.J., D'Allaire S., Taylor D.J., editors. Diseases of Swine. 9th Edit. Blackwell Publishing; Oxford: 2006. pp. 149–177. [Google Scholar]
- Thacker E.L. Immunology of the porcine respiratory disease complex. Veterinary Clinics of North America. Food Animal Practice. 2001;17:551–565. doi: 10.1016/S0749-0720(15)30006-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thacker E.L. Diagnosis of Mycoplasma hyopneumoniae. Animal Health Research Reviews. 2004;5:317–320. doi: 10.1079/ahr200491. [DOI] [PubMed] [Google Scholar]
- Thacker E.L. Mycoplasmal diseases. In: Straw B.E., Zimmerman J.J., D'Allaire S., Taylor D.J., editors. Diseases of Swine. 9th Edit. Blackwell Publishing; Oxford: 2006. pp. 701–717. [Google Scholar]
- Wilson M.R., Takov R., Friendship R.M., Martin S.W., McMillan I. Prevalence of respiratory diseases and their association with growth rate and space in randomly selected swine herds. Canadian Journal of Veterinary Research. 1986;50:209–216. [PMC free article] [PubMed] [Google Scholar]
- Yoon K.J., Edington N. Porcine cytomegalovirus. In: Straw B.E., Zimmerman J.J., D'Allaire S., Taylor D.J., editors. Diseases of Swine. 9th Edit. Blackwell Publishing; Oxford: 2006. pp. 323–329. [Google Scholar]